Abstract
This narrative review examines the utility of gait digital biomarkers in Parkinson’s disease (PD) research and clinical trials across four contexts: disease susceptibility/risk, disease progression, response to exercise, and fall prediction. The review of the literature to date suggests that upper body characteristics of gait (e.g., arm swing, trunk motion) may indicate susceptibility/risk of PD, while pace aspects (e.g., gait speed, stride length) are informative for tracking disease progression, exercise response, and fall likelihood. Dynamic stability aspects (e.g., trunk regularity, double-support time) worsen with disease progression but can improve with exercise. Gait variability emerges as a sensitive biomarker across all 4 contexts but with low specificity. The lack of standardized gait testing protocols and the lack of a minimum set of quantified digital gait biomarkers limit data harmonization across studies. Future studies, using a commonly agreed upon protocol, could be used to demonstrate the utility of specific gait biomarkers for clinical practice.
Subject terms: Biomarkers, Parkinson's disease
Introduction
Parkinson’s disease (PD) is one of the first diseases for which digital biomarkers of mobility have been explored1,2. Traditionally, biomarkers have been defined as characteristics (physiologic, pathologic, or anatomic) that can be measured as an indicator of a biological process2–4. More recently, digital biomarkers have been defined as markers that can be measured by validated technologies5, such as wearable sensors coupled with computational tools, either in the clinic or in daily life. Specific biomarker contexts of use have been established by the U.S. Food and Drug Administration and the National Institutes of Health as part of their joint Biomarkers, Endpoints, and other Tools (BEST) resource6,7. Preferred biomarkers for PD are linked to fundamental features of PD neuropathology3,4, may be sensitive to preclinical disease, are correlated to disease progression2, and are responsive to treatment3,8.
Mobility limitations, specifically those related to gait impairments, are among the earliest and most debilitating signs of PD9,10. Gait disturbances in people with PD often impair functional independence and are a major cause of morbidity and mortality. Gait characteristics affected early in PD include9: loss of arm swing (asymmetrically), slow turns, and shuffling, as well as increased temporal variability11,12, see Fig. 1. In addition, as PD progresses, reduction in step length, problems with gait rhythmicity, and freezing of gait can occur9,13. Emerging evidence from studies of gait changes in prodromal PD9 (REM Sleep behavior disorders14,15 or genetic mutations16) demonstrate that gait changes may occur well before a diagnosis of PD. Gait disturbances in people with PD frequently become difficult to treat and are not typically improved through pharmacological or surgical treatment9.
Fig. 1. Stereotypical gait pattern in PD and independent domains of gait with their associated digital gait metrics.
RoM Range of Motion, CoV Coefficient of Variation, SD standard deviation.
Gait is a complex, yet stereotyped, automatic movement pattern that consists of several different physiological characteristics. As it is comprised of multiple characteristics sensitive to survival, age and pathology, several conceptual gait models have been established in the literature9,13. These gait models are helpful in reducing redundancy and aid in the interpretation of different gait domains. The most frequently used gait domain model lists five domains of gait: pace/turning, rhythm, asymmetry, variability, and stability/upper body (Fig. 1)17,18. Gait domains have been shown to be relatively independent and likely controlled by independent neural circuitry (e.g.; pace, rhythm, variability, asymmetry, upper body, and dynamic stability)17,19.
Clinicians agree that gait impairments are highly relevant because of the importance of mobility to quality of life20. Gait impairments are also important because they are among the earliest diagnostic signs of PD, reflect disease severity and progression, are variably responsive to treatment, and have potential to predict falls9,21. However, gait impairments are most often assessed with subjective clinical scales that are limited in the gait domains assessed, such as the postural instability/gait disturbance (PIGD) portion of the revised Movement Disorders Society (MDS) Unified Parkinson’s disease Rating Scale (MDS-UPDRS) Motor Score (Part III)22,23 which has only one item evaluating gait on a scale of 0 to 4 and is not able to measure subtle changes in gait nor predicts falls24. Adding a prescribed test of gait utilizing digital technology offers a wide range of potentially reliable and responsive outcomes that may provide a continuous range of measurement, and be sensitive to small changes in disease state. In addition, there are a wide variety of tools that can be used as gait biomarkers.
Gait Biomarkers: Tools for quantitative analysis
Various tools, including differing types of task parameters (e.g., dual-tasking, courses with or without turns or obstacles) and equipment (e.g., ranging from simple stopwatches and tape measures to optical, pressure-sensitive mats, and wearable Inertial Measurement Units, IMUs), are used to quantify gait. The metrics derived from these tools reflect diverse gait domains and are emerging as potential biomarkers for PD. Here, we review the gait metrics shown to be useful in the literature, given the absence of clear guidelines regarding which specific gait characteristics should be assessed for the aforementioned four different contexts of use, or whether the same gait metrics can be used across all four contexts.
Stopwatches are used for clinic-friendly walk tests, such as the 10-meter, 2- and 6-minute walk tests, that specify distance and/or duration criteria. These tests, which are valid, reliable, and strongly correlated with each other25, assess gait speed or distance, providing a measure of functional status and overall health20. However, gait speed, alone, is not the most specific or sensitive biomarker of PD26,27 and may not best reflect the underlying pathophysiology of PD. Furthermore, the evaluation of gait speed and/or distance walked does not capture important aspects of upper and lower limb spatiotemporal metrics that have potential to provide more PD-sensitive and PD-specific biomarkers of gait28. Capturing continuous kinematic measures during walking requires more advanced instrumentation and technology.
Commercial technologies have emerged as feasible methods to objectively characterize gait29–34. Studies over the past twenty years on quantitative gait characteristics in PD have used various technologies, including footswitches, optical cameras, pressure mats, and IMUs. Optical camera systems, which have been the gold standard to measure gait, allow for upper and lower body measures of gait while walking with the limitation of the calibrated volume, usually contained within 5 to 7 m of walking space35–37. Although this technique is very accurate, it is also time-consuming and requires trained personnel35–37. More portable solutions include footswitches and pressure mats, but are limited to measuring only the lower body during gait and/or a limited walking space (for the pressure mat). Even better, wearable IMUs can quickly and accurately quantify numerous gait measures from both the upper and lower body30,38,39, even as people go about their daily lives. Passive monitoring of gait in daily life holds great promise to derive sensitive information about gait variability across the day or over multiple days which could translate into important gait biomarkers30,38,39.
Although reduced arm swing amplitude and increased arm swing asymmetry have been recognized as early clinical signs of PD40 only a small percentage of gait studies have quantified upper body characteristics during gait19. Together with reduced arm swing range of motion, measures of trunk range of motion during walking in the transverse, coronal or sagittal plane41 as well as measures of trunk motion regularity and smoothness42, have been found to be informative in discriminating between people with early PD and healthy controls. Of the papers that report arm measurements in PD during gait, 70% were published after 2015. Asymmetries of arm swing and/or step during gait can be calculated in various ways but often are expressed as a percent difference between right and left parameters43,44. Trunk-related measures can be calculated from accelerometers placed on the waist at the lumbar level or gyroscope on the sternum42. Studies have included a variety of digital gait measures, so the summary below is based on those reported.
The use of footswitches and IMUs provides reliable measures of gait across long bouts of walking and to quantify stride-to-stride variability of different gait outcomes, which may be a sensitive measure in PD45,46. Usually, a one-minute walk, or a minimum of 20 strides, is the shortest duration or number of strides recommended to capture gait variability45,46. Gait variability is usually quantified with the coefficient of variation or the standard deviation of the number of strides walked45,46. The most common cause of gait variability (temporal or spatial) in PD is poor dynamic postural control of the trunk while walking, which is associated with compensatory postural stepping responses to recover equilibrium that disrupts gait rhythm47. Increased gait variability (stride-time and step-length) in people with PD and elderly people without movement disorders has been associated with a greater risk of falls48–51, and has also been identified as a precursor to freezing of gait (FOG).
Activities of daily living frequently require walking and performing simultaneous cognitive or motor tasks, such as talking with someone or carrying groceries. Another way to assess gait impairments in people with PD is therefore using concurrent dual-task conditions, walking while performing subtractions for example, or other concurrent cognitive tasks52 to exacerbate impairments, such as freezing of gait53,54. Over the years, dual-task walking research has expanded rapidly52,53 and has also been used in research settings to bring out subtle changes in gait, for example, in people with prodromal PD55 or to evaluate susceptibility/risk of future falls. Despite the wide use of dual-task paradigm in gait, it is hard to draw consistent conclusions because of the breadth of methodological variations in using it, such as instructions given on prioritization, type of task chosen, and speed of task52. Standardization has not been reached, although general guidelines in which dual-task should be preferred for PD have recently been reported52.
Gait Biomarkers: Domains for identifying disease risk, progression, response, and fall risk
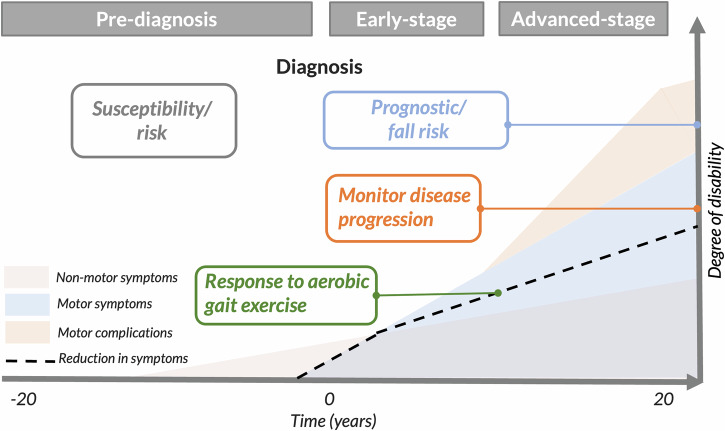
Different gait domains may be useful for understanding different contexts of impairment in PD (e.g., contexts of use). For example, more gait domains are impaired in later stages of PD, compared to earlier stages, as they may be related to the degeneration of different circuit types (e.g., dopaminergic, cholinergic, etc.)56. In particular, levodopa has been shown to increase the pace of gait (e.g., speed, stride length, etc.), but not improve postural stability (e.g., double support time, trunk stability) while walking57. Our focus in this narrative review is to summarize current evidence of specific gait biomarkers for use in clinical trials in the following 4 contexts of use6,7 as indicated in Fig. 2: (1) indicating the potential for symptomatic and pre-symptomatic disease (susceptibility/risk), (2) monitoring disease progression (progression), (3) assessing response to exercise (response), and (4) identifying the likelihood of a fall (prognostic).
Fig. 2.
Context of use for gait biomarkers for Parkinson’s disease summarized in this review.
1) Gait Biomarkers to Indicate Susceptibility/Risk for PD
The clinical diagnosis of PD relies on the manifestation of the classic motor signs of bradykinesia, tremor, and rigidity58. By the time of clinical diagnosis, 50-70% of the nigral dopaminergic neurons are lost59. Therefore, there is still the need to identify reliable, easily accessible and sensitive biomarkers to detect early pathological changes. Might gait biomarkers be used to indicate risk for PD?
One way to address this question is to look at gait studies in people who carry genetic mutations linked to PD, but do not yet have manifest PD (e.g.; normal clinical scales), see Table 1A. Only seven such studies have been published thus far: five in non-manifesting carriers of LRRK2, one in heterozygous Parkin carriers, and one in PINK1 carriers. These studies report a number of significant gait differences between non-manifesting carriers and healthy controls. Among the five studies focused on LRRK2, three studies reported greater stride time variability in non-manifesting LRRK2 carriers relative to healthy controls during regular (comfortably paced)60, fast-paced55,60, and dual-task walking16,55,60. In contrast, the fourth study noted lower stride-time variability in non-manifesting LRRK2 carriers than control subjects during fast walking61. This same study noted that carriers had longer than normal step times on the non-dominant side during fast walking and that longer step times correlated with lower dopamine transporter levels61. In addition, the fifth study of gait during daily life in non-manifesting LRRK2 carriers noted greater variability in arm swing amplitude across different epochs of walking over 7 days, but didn’t measure lower limb variables as only wrist-mounted accelerometers were used62. The single study of gait in non-manifesting Parkin carriers reported the ability to discriminate between this heterozygous carrier group and healthy controls using multivariate analysis with machine learning algorithms and found that dual-task gait best discriminated Parkin carriers from controls63. Finally, a study in heterozygous PINK1 mutation carriers found reduced bilateral arm swing in non-manifest carriers compared to healthy controls64. To date, there are no gait studies in non-manifesting GBA carriers, but there are four studies of people with manifest PD who were GBA carriers. These studies report more asymmetric arm swing65, slower gait speed in single-task66–68 and dual-task conditions68, and increased gait variability67 in those with manifest PD with GBA mutations compared to those with PD without GBA mutations. Across all the studies of genetic mutations related to early PD that we found, the most consistent finding was increased gait variability, with the most pronounced differences generally seen during challenging gait tasks (e.g., dual-task conditions).
Table 1.
List of references found for the 4 different context of use: (A) Gait biomarkers to Indicate Susceptibility/Risk portion. (B) Gait biomarkers to monitor disease progression. (C) Gait Biomarkers for Response to Aerobic Exercise, and (D) Gait Biomarkers for Prognosis (Falls)
| A. Gait biomarkers to Indicate Susceptibility/Risk | ||||
|---|---|---|---|---|
| Publication | Brief study description | Participants | PD Severity/Medication Status | Gait digital biomarkers results and discussion |
|
1 Gait alterations in healthy carriers of the LRRK2 G2019S mutation. Mirelman A, Gurevich T, Giladi N, Bar-Shira A, Orr-Urtreger A, Hausdorff JM.Ann Neurol. 2011 Jan;69(1):193-7. doi: 10.1002/ana.22165.PMID: 21280089 |
Test for association between LRRK2-G2019S mutation and gait Accelerometer on lower back for 3 walking tasks, 1-minute long, over a 20 m corridor: (1) preferred, usual-walking speed; (2) dual-task condition, walking and subtracting serial 7 s; and (3) fast-speed walking. Gait digital biomarkers: Stride time, gait speed, stride length. Stride time variability |
52 first-degree relatives of people with PD who carry LRRK2 mutation - 27 were non-carriers - 25 were carriers |
N/A – participants did not have a diagnosis of PD |
• No differences in gait speed between carriers and non-carriers • Carriers had higher stride time variability in dual task and fast conditions than non-carriers • Amplitude of accelerometer dominant frequency peak was lower in carriers compared to non-carriers in dual task and fast conditions |
|
2 Fall risk and gait in Parkinson’s disease: the role of the LRRK2 G2019S mutation. Mirelman A, Heman T, Yasinovsky K, Thaler A, Gurevich T, Marder K, Bressman S, Bar-Shira A, Orr-Urtreger A, Giladi N, Hausdorff JM; LRRK2 Ashkenazi Jewish Consortium.Mov Disord. 2013 Oct;28(12):1683-90. |
Compare gait in people with PD who are carriers or non-carriers of LRRK2-G2019S mutation Accelerometer on lower back for 3 walking tasks, 1-minute long each, over a 20 m corridor: (1) preferred, usual-walking speed; (2) dual-task condition, walking and subtracting serial 3 s; and (3) fast-speed walking. Gait digital biomarkers: Stride time, gait speed, stride length. Stride time variability |
100 total people with PD - 50 were LRKK2 carriers - 50 were non-carriers |
H&Y II–III Mean UPDRS motor: PD carriers 16.6; PD non-carriers 17.4 Mean levodopa daily dose: carriers 191.8 mg; non-carriers 300.1 mg |
Regardless of walking condition, stride time variability was larger among the carriers compared to non-carriers |
|
3 Arm swing as a potential new prodromal marker of Parkinson’s disease. Mirelman A, Bernad-Elazari H, Thaler A, Giladi-Yacobi E, Gurevich T, Gana-Weisz M, Saunders-Pullman R, Raymond D, Doan N, Bressman SB, Marder KS, Alcalay RN, Rao AK, Berg D, Brockmann K, Aasly J, Waro BJ, Tolosa E, Vilas D, Pont-Sunyer C, Orr-Urtreger A, Hausdorff JM, Giladi N.Mov Disord. 2016 Oct;31(10):1527-1534 |
Evaluate relationship between LRRK2-G2019S mutation, arm swing, and axial rotation in nonmanifesting carriers and noncarriers as well as people with PD Three IMUs (arms and lumbar) for 2, 1-minute long, walking tasks over a 15 m corridor: (1) preferred, usual-walking speed; (2) dual-task condition, walking and subtracting serial 3 s; and. Gait digital biomarkers: Arm swing range of motion, Arm swing variability, arm swing asymmetry, gait variability, stride time variability, axial rotation smoothness |
380 total participants 186 non-manifesting controls, of which 122 were carriers 194 PD patients, of which 67 were carriers |
H&Y I–III UPDRS III: non-carriers 19.9; carriers 15.9 Specific medication info not collected; participants tested on meds |
• Nonmanifesting mutation carriers had greater arm swing asymmetry, greater arm swing variability and lower axial rotation smoothness in dual task compared to nonmanifesting noncarriers; arm swing asymmetry was associated with higher gait variability in nonmanifesting carriers • PD carriers had higher arm swing asymmetry, higher arm swing variability in dual task compared to PD noncarriers • PD carriers had greater stride time variability in dual task compared to PD noncarriers |
|
4 Sensor-based gait analysis in the premotor stage of LRRK2 G2019S-associated Parkinson’s disease. Sánchez-Rodríguez A, Tirnauca C, Salas-Gómez D, Fernández-Gorgojo M, Martínez-Rodríguez I, Sierra M, González-Aramburu I, Stan D, Gutierrez-González A, Meissner JM, Andrés-Pacheco J, Rivera-Sánchez M, Sánchez-Peláez MV, Sánchez-Juan P, Infante J.Parkinsonism Relat Disord. 2022 Apr 7;98:21-26. |
Determine whether there are early gait alterations in carriers of the LRRK2-G2019S mutation that can be detected using inertial sensors Sixteen inertial on head, trunk, arms, legs for 3 walking task over a 15 m corridor: 1) preferred, usual-walking speed, 1-minute; 2) dual-task condition, walking and subtracting serial 3 s, 1-minute; and 3) fast walking (one pass over corridor, no turns). Gait digital biomarkers: Gait speed, stride length, step time, step/stride time variability, step time asymmetry, arm swing amplitude, variability, and asymmetry |
21 idiopathic PD 20 LRRK2 PD 27 nonmanifesting LRRK2 carriers 36 controls |
Idiopathic PD mean MDS-UPDRS-III 29.5, mean LEDD 471 mg LRRK2 PD mean MDS-UPDRS-III 30.5, mean LEDD 705 mg H&Y not specified |
• People with PD and healthy controls differed in speed, stride length and arm swing amplitude, variability and asymmetry in all three tasks (p < 0.01) • In nonmanifesting carriers differences were seen only during fast walking, with greater step time on the non-dominant side (p < 0.05), lower step/stride time variability (p < 0.01) and lower step time asymmetry (p < 0.01). • DaT uptake showed a significant correlation with step time during fast walking on the non-dominant side (r = - 0.52; p < 0.01). • Neural network technique differentiated nonmanifesting carriers and healthy controls based on gait patterns with an accuracy rate of 82.5%. |
|
5 Actigraphy Detects Greater Intra-Individual Variability During Gait in Non-Manifesting LRRK2 Mutation Carriers. van den Heuvel L, Lim AS, Visanji NP, Huang J, Ghate T, Mestre TA, AlDakheel A, Connolly BS, Gasca-Salas C, Kern DS, Jain J, Slow EJ, Pondal M, Faust-Socher A, Rogaeva E, Tomlinson G, Lang AE, Marras C.J Parkinsons Dis. 2018;8(1):131-139 |
Test whether nonmanifesting LRRK2 mutation carriers have more arm swing asymmetry or more movement variability during walking compared to matched controls Bilateral wrist-worn accelerometers for seven days with daily walking task Gait digital biomarkers: Daily activity: fragmentation of activity, fragmentation of sleep, Walking: arm swing asymmetry and variability during walking. |
11 nonmanifesting carriers 14 PD patients, of whom 4 were carriers 29 controls |
H&Y I–IV Mean LEDD 500 mg |
• Compared to healthy controls, nonmanifesting carriers and PD carriers had higher variability in activity during walking • Those with PD (carriers and non-carriers) had lower activity, more arm swing asymmetry and less increase in arm swing with gait speed increase compared to healthy controls |
|
6 Continuous Real-World Gait Monitoring in Idiopathic REM Sleep Behavior Disorder. Del Din S, Yarnall AJ, Barber TR, Lo C, Crabbe M, Rolinski M, Baig F, Hu MT, Rochester L. J Parkinsons Dis. 2020;10(1):283-299. |
Investigate whether wearable gait monitors can detect early gait changes in individuals with RBD Accelerometer on lower back for 7 days of daily activities (24/7). Gait digital biomarkers: Micro gait characteristics (gait pace, rhythm, and step time variability), macro gait characteristics (volume, pattern, and variability of ambulatory bout length) |
63 individuals with RBD 34 healthy controls |
N/A – participants did not have PD diagnosis |
• Micro gait characteristics (gait velocity, rhythm, and step time variability) were seen as the best discriminative domains between RBD and controls. Swing time during gait strongest measure discriminating RBD and healthy controls (AUC = 0.65) • Among the macro gait characteristics (volume, pattern, and variability of ambulatory bout length) only bout length was different between the two groups (longer walking bouts in healthy controls) |
|
7 Subtle gait and balance impairments occur in idiopathic rapid eye movement sleep behavior disorder. Ehgoetz Martens KA, Matar E, Hall JM, Phillips J, Szeto JYY, Gouelle A, Grunstein RR, Halliday GM, Lewis SJG. Mov Disord. 2019 Sep;34(9):1374-1380. |
Determine any gait and balance impairments in iRBD and compare differences to healthy age-matched controls 5 walking conditions on a 6.1 m pressure walkway: 1) normal pace, 2) fast pace, 3) walking while counting backwards from 100 by 1 s, 4) walking while naming as many animals as possible, and 5) walking while subtracting from 100 by 7 s). Gait digital biomarkers: Gait speed, step length, step length variability, step length asymmetry, step width, step width variability |
24 iRBD individuals 14 healthy controls |
N/A – participants did not have PD diagnosis |
• No differences in gait between groups under normal speed gait • Participants’ step length variability worsened as dual-task difficulty increased • iRBD patients demonstrated greater step length asymmetry than healthy controls during fast-paced walking; increased step width variability in response to a more difficult dual task, while healthy controls widened their step width |
|
8 Classifying Idiopathic Rapid Eye Movement Sleep Behavior Disorder, Controls, and Mild Parkinson’s Disease Using Gait Parameters. Cochen De Cock V, Dotov D, Lacombe S, Picot MC, Galtier F, Driss V, Giovanni C, Geny C, Abril B, Damm L, Janaqi S. Mov Disord. 2022 Apr;37(4):842-846. |
Create a multiclass predictive model to distinguish iRBD, healthy controls, and PD Six IMUs on wrists, feet, lumbar and sternum for 3 walking tasks, 13 laps long, over a 20 m corridor: (1) preferred, usual-walking speed; (2) dual-task condition, walking and counting plus 1; (3) dual-task condition, walking and counting minus 3 s. Gait digital biomarkers: 59 gait parameters were extracted, including stride length and velocity; cadence; gait cycle time; double support; swing and stance; ranges of motion; angular velocities of legs, arms, and trunk; and asymmetries of the stride, swing, stance, leg, and arm swing range of motion. |
21 iRBD individuals 21 healthy controls 21 PD patients |
H&Y 1.52 ± 0.75 Disease duration of 4.24 ± 2.77 years Levodopa equivalent daily dose 258.7 ± 214.7 mg |
• A classification model (LASSO) aggregated gait features to predict true health status of participants • Results showed very high accuracy of classification of iRBD from healthy controls and PD patients (95% and 98%, respectively) • Range of motion and asymmetry of lower limbs during walking among the gait biomarkers that contributed most on the classification model • Dual-task did not improve accuracy |
|
9 REM Sleep Without Atonia and Gait Impairment in People with Mild-to-Moderate Parkinson’s Disease. Amundsen-Huffmaster SL, Petrucci MN, Linn-Evans ME, Chung JW, Howell MJ, Videnovic A, Tuite PJ, Cooper SE, MacKinnon CD. J Parkinsons Dis. 2021;11(2):767-778. |
Compare gait disturbances between participants with REM sleep without atonia (RSWA) and those without RSWA among participants with PD One walking conditions on a 10 m pressure walkway to reach 35 steps at comfortable speed Gait digital biomarkers: Gait speed, step length, step width, step time average and variability, double support time |
41 participants with idiopathic PD 21 age- and sex-matched controls |
PD-RSWA (-): H&Y = 2 (2–3); LEDD = 200 (100-317) mg PD-RSWA (+): H&Y = 2 (2–3); LEDD = 373 (300-605) mg |
• PD-RSWA(+) group showed: (1) reduced gait speed, step length and double support compared to healthy controls; and 2) decreased speed, decreased cadence, increased stride velocity variability, increased step and stance times compared to PD-RSWA (-). • The level of muscle activity during REM sleep correlated with measures of gait such that increased tonic or phasic EMG was associated with worsening of gait metrics |
|
10 Subtle gait changes in patients with REM sleep behavior disorder. McDade EM, Boot BP, Christianson TJ, Pankratz VS, Boeve BF, Ferman TJ, Bieniek K, Hollman JH, Roberts RO, Mielke MM, Knopman DS, Petersen RC. Mov Disord. 2013 Nov;28(13):1847-53. |
Investigate differences in gait among subjects with probable RBD compared to controls One walking conditions on a 5 m pressure walkway, up and back at comfortable speed Gait digital biomarkers: Cadence, swing time, swing time variability, stance time, stance time variability, gait velocity, double support time, double support time variability, stride length, and stride length variability, stride time and stride time variability |
• 42 probable RBD subjects • 492 controls |
• N/A – participants did not have PD diagnosis |
• Nearly 80% of variance in gait measures were explained by three factors: measures of rhythm, pace, and stride variability Probable RBD was associated with increased stride-to-stride variability, decreased rhythm and velocity of gait. |
|
11 Detection of Motor Dysfunction With Wearable Sensors in Patients With Idiopathic Rapid Eye Movement Disorder. Ma L, Liu SY, Cen SS, Li Y, Zhang H, Han C, Gu ZQ, Mao W, Ma JH, Zhou YT, Xu EH, Chan P. Front Bioeng Biotechnol. 2021 Apr 15;9:627481. |
Detect subclinical gait changes by using wearable motor sensors in iRBD patients Six IMUs on wrists, ankles, lumbar and sternum for 3 walking tasks, 1-minute each, over a 10 m corridor: (1) preferred, usual-walking speed; (2) dual-task condition, subtracting by 7 s; (3) fastest pace. Gait digital biomarkers: Stride length, stride velocity, stride time, trunk range of motion, stride length asymmetry, stride length variability, stride time variability |
31 iRBD patients 20 healthy controls |
N/A – participants did not have PD diagnosis |
• Compared to healthy controls, iRBD patients showed significantly decreased range of motion and peak angular velocity of the trunk, and longer step time before a turn. • No differences in stride length, stride velocity, stride time, stride length asymmetry, and stride length and stride time variability in the 3 walking conditions |
|
12 Task matters - challenging the motor system allows distinguishing unaffected Parkin mutation carriers from mutation-free controls. Prasuhn J, Borsche M, Hicks AA, Gögele M, Egger C, Kritzinger C, Pichler I, Castelo-Rueda MP, Langlott L, Kasten M, Mascalzoni D, Klein C, Pramstaller PP, Brüggemann N. Parkinsonism Relat Disord. 2021 May;86:101–104. |
Investigate differences in the motor system between Parkin mutation carriers and non-carriers through gradient boosted trees Six IMUs on wrists, ankles, lumbar and sternum for walking tasks, 2-minute each, over a 40 m corridor: (1) preferred, usual-walking speed; (2) fastest pace; (3) dual tasking mental; (4) dual tasking balancing a glass of water Gait digital biomarkers: 132 features encompassing lower limbs, upper limbs gait measures averages, asymmetries and variability. |
27 healthy heterozygous Parkin mutation carriers 24 healthy non-mutation carriers |
N/A – participants did not have PD diagnosis |
• Measures aggregated together for each gait conditions Only the most challenging gait had good AUC (0.78) |
|
13 Gait analysis with wearables predicts conversion to Parkinson’s disease. Del Din S, Elshehabi M, Galna B, Hobert MA, Warmerdam E, Suenkel U, Brockmann K, Metzger F, Hansen C, Berg D, Rochester L, Maetzler W.Ann Neurol. 2019 Sep;86(3):357–367 |
Establish gait impairments and trajectories in the prodromal phase of PD, identifying which gait characteristics are potentially early diagnostic markers of PD. Longitudinal assessment (4 times, 2-year intervals) identified those who converted to PD. Participants walked at different speeds under single and dual tasking, with a wearable device on the lower back; 14 validated clinically relevant gait characteristics were quantified. Gait digital biomarkers: Step time variability, gait asymmetry (all characteristics), gait pace |
696 healthy controls (mean age = 63 ± 7 years) | N/A – participants did not have PD diagnosis | • Sixteen participants were diagnosed with PD on average 4.5 years after first visit (converters; PDC). Higher step time variability and asymmetry of all gait characteristics were associated with a shorter time to PD diagnosis. Gait (lower pace) deviates from that of non-PDC approximately 4 years prior to diagnosis |
| B. Gait biomarkers to monitor disease progression | ||||
|---|---|---|---|---|
| Publication | Brief study description | Participants | PD Severity/Medication Status | Gait digital biomarkers results and discussion |
|
1 Progressive Gait Deficits in Parkinson’s Disease: A Wearable-Based Biannual 5-Year Prospective Study. Hobert MA, Nussbaum S, Heger T, Berg D, Maetzler W, Heinzel S. Front Aging Neurosci. 2019 Feb 13;11:22. (Part of MODEP study, Modeling epidemiological data to study PD progression. Assessment every 6 months for 5 years). |
Longitudinal changes in gait parameters from a lower-back sensor in early- and mid-stage PD compared to older adults. Normal speed and fast speed gait for 2 minutes. (Corridor of 20 m) Accelerometer on lower back for 2 walking tasks, 2-minute long each, over a 20 m corridor: 1) preferred, usual-walking speed; 2) fast-speed walking. Gait digital biomarkers: step number, step time, step velocity, and measures of gait variability, i.e., step time variability (calculated as coefficient of variation), gait asymmetry and phase coordination index (PCI) |
Controls, N = 24 age and gender matched to PD Early PD, N = 22. <4 years from diagnosis Mid-stage PD, N = 18. >4 years from diagnosis |
Visits for PD differed in ON/OFF medication state (E-PD: 18%; M-PD: 25% of visits in ON state) which was accounted for in statistical analyses. |
• normal-pace walking was more sensitive to gait-related changes than fast-pace walking • Changes were significantly larger in E-PD (2.1%/year) than in HC (0.7%/year), and, importantly, showed linear progression over the 5-year observation period. • Significant differences in longitudinal changes were observed between E-PD and HC in gait variability, however, this finding was not (only) due to a (non-significant) increase in PD (8.5%/year) but partly driven by HC decreasing in gait variability over time. • M-PD did not show any significant gait changes compared to HC during the relatively long observation period. |
|
2 Potential Markers of Progression in Idiopathic Parkinson’s Disease Derived From Assessment of Circular Gait With a Single Body-Fixed-Sensor: A 5 Year Longitudinal Study. Micó-Amigo ME, Kingma I, Heinzel S, Rispens SM, Heger T, Nussbaum S, van Lummel RC, Berg D, Maetzler W, van Dieën JH. Front Hum Neurosci. 2019 Feb 19;13:59. (Part of MODEP study, Modeling epidemiological data to study PD progression. Assessment every 6 months for 5 years). |
Longitudinal changes in walking in circle from a lower-back sensor in early- and mid-stage PD compared to older adults. Accelerometer on the lower back for three rounds around a circle of 1.2 m diameter at their preferred speed. This protocol was performed twice at each visit, once in clock-wise and once in counter-clock-wise direction. Gait digital biomarkers: - Number of steps, - Total duration, - Step time asymmetry, - Median stride time, - Stride time variability, - Movement intensity (SD of ap, ml and vt acceleration during the trial) - Step regularity - Stride regularity - Harmonic ratio - Peak power |
Controls, N = 25 age and gender matched to PD Early PD, N = 22. <4 years from diagnosis Mid-stage PD, N = 27. >4 years from diagnosis |
Visits differed in medication state within and between-subjects (16.4% of the assessments were in ON medication state). The condition ON medication was defined as a time period of 30 minutes to 3 hours after the intake of the usual dose of dopaminergic medication |
• Five gait features indicated progressive worsening in early stages of PD: number of steps, total duration and harmonic ratios calculated from vertical (VT), medio-lateral (ML), and anterior-posterior (AP) accelerations. For middle stages of PD, three gait features were identified as potential progression markers: stride time variability, and stride regularity from VT and AP acceleration. |
|
3 Dual vs. Single Tasking During Circular Walking: What Better Reflects Progression in Parkinson’s Disease? Micó-Amigo ME, Kingma I, Heinzel S, Nussbaum S, Heger T, van Lummel RC, Berg D, Maetzler W, van Dieën JH. Front Neurol. 2019 May 14;10:372. |
Same as #2. Addition of DT in circular walking: - Walking in circles while marking crosses Walking in circles while subtracting |
Same as #2. | Same as #2. | • The results support the validity and robustness of some of the gait features already identified in our previous work (2) as progression markers of the disease in single-task circular walking. However, fewer gait features from dual-task than from single-task assessments were identified as markers of progression in PD. |
|
4. Progression of gait dysfunction in incident Parkinson’s disease: impact of medication and phenotype. Galna B, Lord S, Burn DJ, Rochester L. Mov Disord. 2015 Mar;30(3):359-67. ICICLE-PD study (Incidence of Cognitive Impairment in Cohorts with Longitudinal Evaluation—PD). |
To document the extent and progression of gait impairment over 18 months in an incident cohort of PD participants, compare rate of change with respect to motor phenotype. 2-minute walk at the participants’ preferred pace around a 25-m circuit using a 7-m-long instrumented walkway. In addition, cones used to guide participants around the circuit. Gait digital biomarkers: Mean, variability and asymmetry of: Step length Step time Step velocity Stance time Swing time Step width |
Tested at baseline: 121 newly diagnosed participants with PD 184 controls 108 PD participants reassessed at 18 months |
Optimally medicated cohort of incident PD. Tested ON medication Assessed within 4 months of diagnosis |
• Few changes in gait impairment over 18 months in PD, with significant impairment in pace (step length) and rhythm (swing time) • When change scores were explored with respect to motor phenotype, no significant change was found for the PIGD group. In contrast, TD participants showed significant evolution of spatial and variability characteristics, with a shorter and more variable step length. |
|
5 Gait Progression Over 6 Years in Parkinson’s Disease: Effects of Age, Medication, and Pathology. Wilson J, Alcock L, Yarnall AJ, Lord S, Lawson RA, Morris R, Taylor JP, Burn DJ, Rochester L, Galna B. Front Aging Neurosci. 2020 Oct 15;12:577435. ICICLE-PD study (Incidence of Cognitive Impairment in Cohorts with Longitudinal Evaluation—PD). Extension of 36-months. |
(i) identify gait characteristics that significantly changed over 6 years in newly diagnosed PD and healthy age-matched controls; (ii) evaluate gait changes in the PD cohort which related to aging and disease progression by comparing rates of gait change between PD and control groups; (iii) explore the relationship between gait changes and changes in dopaminergic medication dose in early PD. Gait was assessed at 18-month intervals for up to 6 years using an instrumented walkway to measure sixteen spatiotemporal gait characteristics. Same protocol and measures of #4 |
109 newly diagnosed participants with PD 130 controls At least two gait assessments. |
One-hundred and nine newly diagnosed PD participants and 130 controls completed at least two gait assessments. On medication, same as study #4 |
• Gait changes associated primarily with aging rather than PD progression, and also those characteristics due to aging which are accelerated by the presence of PD. • Four characteristics significantly changed in PD only (increased variability of swing time, step time and step width, and reduced swing time asymmetry) and describe changes specifically due to disease progression. These findings extend previous work in this cohort over shorter periods (4) by revealing additional changes in asymmetry and variability that are not evident over 18 or 36 months. • The only gait characteristics related to change in medication is step width variability. Increased step width variability is related to increasing levodopa medication. • Two gait characteristics, step length, and step length variability, significantly worsened within PD and control groups over 6 years and specifically, those changes occurred more rapidly in PD. |
|
6 Progression of postural control and gait deficits in Parkinson’s disease and freezing of gait: A longitudinal study. Vervoort G, Bengevoord A, Strouwen C, Bekkers EM, Heremans E, Vandenberghe W, Nieuwboer A. Parkinsonism Relat Disord. 2016 Jul;28:73-9. |
To identify if postural control deficits and gait dysfunction progress differently in freezers compared to non-freezers Gait assessed with a motion analysis system. Subjects were instructed to perform a continuous overground gait task at a self-preferred pace until at least 40 steps were recorded. Gait digital biomarkers: Gait speed, swing time and step length variability measures of swing time and step length |
76 PD 24 healthy controls Subjects underwent a postural control and gait assessment at baseline and after 12 months follow-up |
All tests were performed in the practically defined ‘off’ state. final sample size of 67 PD patients (11 FOG and 56 NFOG) and 20 controls for the longitudinal analysis. |
• At baseline, there were no differences in spatiotemporal gait variables between PD and controls. • No decline in gait measures over 12-months • (mini-BEST did decline) |
|
7 Identification of motor progression in Parkinson’s disease using wearable sensors and machine learning. Sotirakis C, Su Z, Brzezicki MA, Conway N, Tarassenko L, FitzGerald JJ, Antoniades CA. NPJ Parkinsons Dis. 2023 Oct 7;9(1):142. Oxford Quantification in Parkinsonism (OxQUIP) study |
To establish whether a combination of wearable sensor data and machine learning (ML) algorithms can estimate the clinical rating scale and whether it is possible to monitor the motor symptom progression longitudinally Six IMUs on wrists, feet, lumbar and sternum for one walking task of 2-minute, over a 15 m corridor Gait digital biomarkers: 122 features encompassing lower limbs, upper limbs gait measures averages, asymmetries and variability. |
91 participants wih PD enrolled. All participants were asked to visit the lab once every three months over a period of 18 months, completing a total of 7 visits. |
All tests were performed in the on medication state Final sample size for the longitudinal analysis 74 |
• Automatic feature selection model to predict clinical MDS-UPDRS and the model was able to identify the worsening of PD motor symptoms over time from those data, unlike the actual MDS-UPDRS-III scores. • Symptom progression as early as 15 months after baseline with the ML approach • Individual gait biomarkers that contribute most to an accurate estimate of the MDS-UPDRS-III-score: the angle of the foot at foot strike and toe off, and the stride length • Out of the 29 gait biomarkers that showed statistically significant progression across visits, 19 reflect walking variability |
| C. Gait Biomarkers for Response to Aerobic Exercise | ||||
|---|---|---|---|---|
| Publication | Brief study description | Participants | PD Severity/Medication Status | Gait digital biomarkers results and discussion |
|
1 Effects of 24 wk of treadmill training on gait performance in Parkinson’s disease. Nadeau A, Pourcher E, Corbeil P. Med Sci Sports Exerc. 2014 Apr;46(4):645-55. |
A 24-wk, double-blind, randomized, controlled trial Participants were randomized to speed treadmill training (TT), mixed TT, and control groups. The intervention consisted of 72 one-hour exercise sessions for 24 weeks One walking conditions on a 4.3 m pressure walkway, 8 passes at comfortable speed. 6-minute walking task for distance Gait digital biomarkers: walking speed, stride length, cadence, step width. Variability of stride length and double support time |
34 participants with PD |
H&Y: 1.5-2 On medication |
• Both TT groups improved in terms of speed, cadence, and stride length during self-selected walking conditions at the study end point. • The Control group showed no changes. |
|
2 Exploring gait adaptations to perturbed and conventional treadmill training in Parkinson’s disease: Time-course, sustainability, and transfer. Steib S, Klamroth S, Gaßner H, Pasluosta C, Eskofier B, Winkler J, Klucken J, Pfeifer K. Hum Mov Sci. 2019 Apr;64:123-132. |
Randomized and stratified controlled trial. Participants were randomized to perturbation treadmill training (PTT) or conventional treadmill training (CTT), two times a week for 8 weeks. Overground walking: Two IMUs on shoes for one walking task over a 10 m corridor, 4 passes at preferred, usual-walking speed Gait digital biomarkers: Average, variability and asymmetry of: Stride length, stride time, stance time, swing time |
38 participants with PD |
H&Y: 1-3.5 On medication |
• Significant overground improvements in stance- and swing time were only seen in the PTT group. • A significant between-group interaction effect suggested reduced swing time variability in the PTT group, but an increased variability in the CTT group. |
|
3 Effect of virtual reality training on walking distance and physical fitness in individuals with Parkinson’s disease. de Melo GEL, Kleiner AFR, Lopes JBP, Dumont AJL, Lazzari RD, Galli M, Oliveira CS. NeuroRehabilitation. 2018;42(4):473-480. |
Randomized, controlled clinical trial. Participants were randomly allocated to three intervention groups (control, treadmill and VR) using a block randomization method. 20-minute training sessions three times a week for four weeks (total: 12 sessions) One IMU on lower back for an instrumented 6 min-walking task (6MWT), over a 30 m corridor, as fast as possible. Gait digital biomarkers: Distance traveled during the 6MWT, symmetry index of left/right gait cycles, gait speed. |
42 participants with PD |
H&Y: 1-3 On medication |
• The VR and treadmill groups traveled a longer distance on the 6MWT, had a faster gait speed in comparison to the control group. • No differences in gait in the treadmill and VR group |
|
4 Effects of Treadmill Training with Load on Gait in Parkinson Disease: A Randomized Controlled Clinical Trial. Trigueiro LC, Gama GL, Simão CR, Sousa AV, Godeiro Júnior Cde O, Lindquist AR. Am J Phys Med Rehabil. 2015 Oct;94(10 Suppl 1):830-7. |
Randomized, controlled single-blind trial. Participants were randomly allocated to divided into three groups: treadmill alone (control group [CG]), treadmill with 5% of load (experimental group 5% [EG5%]), and treadmill with 10% of load (experimental group 10% [EG10%]). Gait assessed with a motion analysis system. Subjects were instructed to walk at a self-preferred pace over a space of 8 m for 10 repetitions Gait digital biomarkers: Gait speed, stride length, most affected step length, double support time, stance time, swing time. Hip, knee, and ankle ROM during walking. |
27 participants with PD |
H&Y: 2-3 On medication |
• Irrespective of the load, participants showed increased speed, stride length, and step length and a decrease in the double-support and stance phases after exercise. • Increase in knee ROM during walking after exercise, irrespective of the load. |
| D. Gait Biomarkers for Prognosis (Falls) | ||||
|---|---|---|---|---|
| Publication | Brief study description | Participants | PD Severity/Medication Status | Gait digital biomarkers results and discussion |
|
1 Predicting Fall Counts Using Wearable Sensors: A Novel Digital Biomarker for Parkinson’s Disease Greene, B.R.; Premoli, I.; McManus, K.; McGrath, D.; Caulfield, B. Predicting Fall Counts Using Wearable Sensors: A Novel Digital Biomarker for Parkinson’s Disease. Sensors 2022,22,54. |
To assess how closely associated the output of fall-risk from an instrumented TUG is to actual self-reported fall counts in 3 distinct datasets, report results from various statistical models Two IMU on shanks for the time-up and go test, 3-m corridor Gait digital biomarkers: 71 different parameters of gait, turning, and transfer |
Controls, N = 1015 community-dwelling adults >= 60 PD1, N = 15 PD patients followed longitudinally only PD2, N = 27 PD patients participating in a 12-wk exercise intervention |
PD1: 40-80 yo, levodopa-responsive x 4 yrs, MMSE > 22, able to walk independently at least 3 m, excluded stage 5 and assist device PD2: stage 1-3 PD |
• Used 2 existing trained kinematic models (FRE and Mobility models, that have been previously shown to be related to falls in a number of populations) and 2 statistical approaches • A predictive model for falls counts in PD showed a mean R2 value of 0.43, mean error of 0.42 and a mean correlation of 30% when the results were averaged across two independent sets of PD data • Single biomarkers of gait related to falls not reported |
|
2 Association Between Baseline Gait Parameters and Future Fall Risk in Patients With De Novo Parkinson’s Disease: Forward Versus Backward Gait. Kwon KY, You J, Kim RO, Lee EJ, Lee J, Kim I, Kim J, Koh SB. J Clin Neurol. 2024 Mar;20(2):201-207. |
Determine the falls-related gait dynamics of drug-naïve patients with de novo PD Two walking conditions on a 4.6 m pressure walkway, 10 passes at comfortable speed. Forward walking (FG) and backward walking (BG) Gait digital biomarkers: gait speed, cadence, stride time and length, swing-phase percentage, double-support time, and step width. Included asymmetry and variability of those parameters |
76 individuals with de novo PD assessed at baseline and 1 year follow-up 16 classified as fallers |
Drug naïve, early individuals with PD, disease duration less than 5 years. |
• Fallers had slower gait and shorter stride for FG and BG parameters than did non-fallers, while stride-time variability was greater in fallers but only for BG • Multivariable logistic regression analysis revealed that slow gait was an independent risk factor in BG |
|
3 Fall Risk Prediction in Parkinson’s Disease Using Real-World Inertial Sensor Gait Data. Ullrich M, Roth N, Kuderle A, Richer R, Gladow T, Gasner H, Marxreiter F, Klucken J, Eskofier BM, Kluge F. IEEE J Biomed Health Inform. 2023 Jan;27(1):319-328. |
To compare different data aggregation approaches and machine learning models for the prospective prediction of fall risk using gait parameters derived either from continuous real-world recordings or from unsupervised gait tests (3 times a day, 4 rep x10meters gait) Gait digital biomarkers: 148 gait features for the real-world recordings 116 gait features for the unsupervised gait |
35 participants with PD 3 months of prospective fall recording |
H&Y: 1-3 |
• The highest balanced accuracy of 74.0% (sensitivity: 60.0%, specificity: 88.0%) was achieved with a Random Forest Classifier applied to the real-world gait data when aggregating all walking bouts and days of each participant. Accuracy of unsupervised gait test was 68%. • The 4 most recurrent gait features in real-world to predict falls were: 1) Max variability of stride length, 2) variability of swing time, 3) Average swing time, 4) variability of max foot lift. |
|
4 Gait and turning characteristics from daily life increase ability to predict future falls in people with Parkinson’s disease. Shah VV, Jagodinsky A, McNames J, Carlson-Kuhta P, Nutt JG, El-Gohary M, Sowalsky K, Harker G, Mancini M, Horak FB. Front Neurol. 2023 Feb 28;14:1096401. |
To investigate if digital measures from different components of gait and turning collected from a week of daily activities increased discriminative ability to predict future falls compared to fall history alone Three IMUs on feet and lumbar over 7 days of passive recording during daily life. Gait digital biomarkers: 52 measures grouped into four domains (Lower Body, Lower Trunk, Turning, and Variability) |
34 participants with PD 6 months of retrospective falls + 12 month of prospective fall recording |
H&Y: 2-4 |
• Gait, turning, and variability domains are important to predict future fallers. • Digital measures from different components of gait showed more discriminative ability to predict future fallers from non-fallers compared to falls history, alone. • The top ten models incorporating digital gait and turning measures in this study were able to separate fallers from non-fallers with an AUC over 0.90 compared to fall history alone, which yielded an AUC of 0.77. Gait variability was the most consistent domain selected, with toe out angle variability being the most common variability measure selected, followed by stride time variability. |
|
5 Predicting Falls in Parkinson Disease: What Is the Value of Instrumented Testing in OFF Medication State? Hoskovcová M, Dušek P, Sieger T, Brožová H, Zárubová K, Bezdíček O, Šprdlík O, Jech R, Štochl J, Roth J, Růžička E. PLoS One. 2015 Oct 7;10(10):e0139849. |
To investigate clinical and instrumented tests of balance and gait in both OFF and ON medication states and to verify their utility in the prediction of future falls in PD patients. Five IMUs on shanks, wrists and sternum for the time-up and go test, 7-m corridor, self-selected speed, 3 repetitions. Gait digital biomarkers: Gait speed, cadence, and stride time variability |
45 participants with idiopathic PD were examined in defined OFF and ON medication 6 months of prospective fall diaries |
H&Y:2-3 |
• Stride time variability in the OFF medication state differed between both PD subgroups while other gait and balance measures failed to show any significant differences. • In a multiple logistic regression model of prospective falling based on clinical parameters, a history of falls and depressive symptoms had the highest discriminative value in identifying future fallers, while the combination of stride time variability and cadence in the OFF medication state was the best instrumented predictor of future falls. |
Gait Exercise was operationally defined as activities that involve gait and are designed to meet moderate or vigorous aerobic exercise intensity levels. Assuming they include interventions that meet the operational definition of gait exercise, studies employing technology (i.e., virtual reality, robotic assistance, etc) were included. Of note, to meet the ‘aerobic exercise intensity’ criteria, a study must report quantifying intensity based on measuring participants’ heart rate (HR) or rating of perceived exertion (RPE) and, according to one or both of those metrics, the authors must report prescribing or achieving moderate or vigorous levels (as defined by ACSM guidelines) during gait exercise.
Reported in the table below are the papers including technology more than a stopwatch to measure gait (4 out of 19 described for this context of use).
Another way to determine if digital gait measures can detect early, pathological changes in PD is to examine people with idiopathic, rapid eye movement sleep (REM) behavior disorder (iRBD). IRBD is characterized by elevated muscle tone during REM sleep (termed REM sleep without atonia, or RSWA) along with vigorous, sometimes violent, movements during REM sleep. The presence of iRBD is a strong predictor for developing synucleinopathies, including PD, Lewy body dementia, and multiple system atrophy69–71. More than 80% of iRBD patients convert to PD or another α-synucleinopathy with a median overall prodromal phase of 8 years, and the estimated prevalence of RBD in PD is 42%70,71. We identified 8 studies that attempted to detect prodromal gait abnormalities with quantitative measures in patients with iRBD by comparing them to healthy controls (with some also including individuals with idiopathic PD).
We identified seven studies that reported significant gait differences between people with iRBD and age-matched healthy controls and PD participants. One study in iRBD used a machine learning approach with 59 gait parameters collected during single-task gait using an inertial sensor system and found that arm and trunk range of motion, asymmetry of limb movement, and abnormal gait rhythm (cadence) best discriminated participants with iRBD from healthy controls and people with PD with 100% sensitivity and 91 to and 95% specificity15. Other studies show that gait velocity is lower in participants with iRBD compared to controls72,73. Individuals with PD and RSWA (with or without dream enactment symptoms), were found to have slower gait velocity and shorter step lengths, compared to individuals with PD without RSWA and healthy controls14. People with iRBD have also been shown to have significantly decreased trunk range of motion and peak trunk angular velocity, and longer step time before turning than healthy controls in single-task, dual-task, and fast-paced walking74. Greater step length asymmetry during fast-paced walking75 and increased step-width variability and step-time during dual-task walking76 has also been reported in iRBD compared to healthy controls.
There are conflicting reports on gait variability in people with iRBD, with one study reporting reduced gait velocity variability72 and another reporting increased stride time, double-support time and swing time variability compared to controls73. However, these two reports have fundamental protocol differences. For example, the first study72 collected data during daily life and the second one73 used a research laboratory, this difference in gait protocols is known to impact gait variability46,77. Of course, studies of gait in people with iRBD are limited by not knowing who will eventually express a synucleinopathy and what type they might manifest, as well as how long this expression might take (years to decades)78, so long-term longitudinal studies are needed.
In summary, the most promising gait biomarkers for susceptibility/risk of developing PD are aspects of gait asymmetry and variability, for both upper and lower limbs, reduced trunk range of motion especially while challenging gait at either faster than comfortable speed or by adding a concurrent, dual-task (see Table 2). The gait biomarkers linked to the trunk motion and arm swing during gait may be directly linked to early PD neurophysiology. In fact, recent work79,80 suggests that early dopaminergic denervation in Parkinson’s disease follows a somatotopically-related pattern, starting with the upper-limb representation in the putamen and progressing over a 2-year period to the less-affected hemisphere. These changes could be associated with the clinical presentation and evolution of motor features reported by clinicians, and specifically, be reflected in reduced and asymmetric arm swing range of motion while walking. Lastly, the finding that these subtle changes in gait in prodromal or early disease are more obvious when challenging gait with a concurrent, cognitive dual-task for example, may reflect intact compensatory mechanisms at this stage. In fact, gait control may become less automatic due to damage of the basal ganglia, cortical areas, such as the prefrontal cortex, and require additional attention to compensate as the disease progresses9,16,53. However, when attention is devoted to a cognitive task, this compensatory mechanism breaks down, revealing underlying deficits of automatic axial control of gait.
Table 2.
Summary of the emerging gait biomarkers for the different context of use considered
| Context of use | Gait domain | Proposed gait biomarker | Protocol |
| Susceptibility/risk |
• Stability/Upper body • Asymmetry • Variability |
• Arm swing ROM • Trunk ROM • Asymmetry of step time • Arm swing ROM variability • Stride time variability |
Challenging gait protocol (dual-task gait or fast gait) |
| Monitoring disease progression |
• Pace/Turning • Stability/Upper body • Variability |
• Stride velocity • Stride length • Turn velocity • Angle of the foot at heel strike/toe off • Trunk regularity/stability • Stride time variability ** Mixed results depending on Off/On assessment |
Single-task gait, natural pace |
| Assessing response to aerobic gait exercise |
• Pace/Turning • Stability/Upper body • Variability |
• Gait velocity • Stride length • Cadence • Double-support time • Stride time variability **No upper body measures |
Inconclusive ** No assessment in dual-task gait, limited to treadmill exercise |
| Prognostic/fall risk |
• Pace/Turning • Variability |
• Gait velocity • Cadence • Step time variability |
Single-task gait, natural pace or Backward |
Gait biomarkers to monitor disease progression
A critical limitation in the development of disease-modifying interventions for PD is the lack of reliable, objective measures of progression, particularly early in the disease course81. The etiology of idiopathic PD is unidentified and there is no known cure. There is, however, increasing evidence for neuroprotective and neuromodulatory therapies that may delay disease progression82. Unfortunately, clinical trials that aim to prevent or slow the progress of mobility disability in PD are limited by the low sensitivity of gold-standard outcome measures that depend upon expert qualitative assessment9,83,84.
Objective measures of gait may provide markers of disease progression that can be extremely useful for clinical trials. Table 1B lists potential markers of disease progression summarized from seven studies. A recent, two-year study in 266 individuals with early PD showed that people with PD had greater declines in walking speed (clinically measured with a stop-watch in a 10-meter walk test) compared to declines in fine motor function of the upper extremities or quality of life85. Of the seven studies that have examined longitudinal progression of gait biomarkers in PD, all but one of them included people with PD already on dopaminergic medication for PD. The one, small longitudinal study of people with newly diagnosed PD who had not yet started dopaminergic medication found that the duration of turning 180 deg while walking back and forth over 7 meters showed reliable worsening over 18 months, compared to stable turning duration in healthy controls86.
The MODEP study (MODeling Epidemiological data to study Parkinson’s disease progression)87, found that natural-pace gait characteristics were more sensitive to changes over 5 years than fast-paced gait among people with early and mid-stage PD compared to healthy older adults. Those with early PD significantly reduced their natural paced gait speed compared to healthy controls over time, rate of 2.1 percent per year in early PD compared to less than 1 percent in healthy controls. Interestingly, healthy controls decreased their gait variability over time, whereas those with early PD significantly increased their gait variability over 5 years87. Two other reports from the MODEP study PD88,89 showed that harmonic ratios, calculated from vertical (VT), medio-lateral (ML), and anterior-posterior (AP) accelerations of the trunk, worsened over 5 years in people with early PD71,72. Stride time variability, and stride regularity from trunk VT and AP accelerations were identified as features that worsened over time in mid-stage PD88. No additional longitudinal effect was noticed during dual-task walking compared to single-task walking89.
Another paper, part of the ICICLE-PD study (Incidence of Cognitive Impairment in Cohorts with Longitudinal Evaluation-PD), examined gait over 18 months in 121 people with PD who were newly diagnosed, and already taking PD medications84. Significant reduction in step length and swing time were found over 18 months, compared to 184 healthy controls84. The ICICLE study also tested the same cohort of people with PD over 6 years to examine the relationship between gait changes and medication changes over time90. Gait deterioration associated with aging were accelerated by the presence of PD and 4 gait biomarkers significantly changed with disease progression only in people with PD: increased variability of lower-limb swing time, step time, step width, and reduced swing time asymmetry. These findings extend previous work in this cohort over shorter periods by revealing additional changes in gait asymmetry and variability that were not evident over 18 or 36 months84. Of interest, the increase in medication use over time was related to increase in step width variability, pointing to differences at least partially based on medication status90.
Another report in 91 individuals with early PD, the Oxford Quantification in Parkinsonism (OxQUIP) study, found changes in gait within 15 months from baseline, although clinical rating was not able to capture signs of disease progression91. This study used a machine learning approach to identify the combination of gait measures that best predicted the MDS-UPDRS Part III score and showed such combinations of measures to be more sensitive than the MDS-UPDRS III in capturing changes over time. Among the individual measures sensitive to change over time were: the angle of the foot (at heel strike and toe-off) and stride length. A smaller value in the angle of the foot at heel strike or toe-off indicated more shuffling of steps, that may increase with disease severity91.
It is not known why gait characteristics progress more rapidly in some people with PD than others. PD has been classified into one of three clinical phenotypes based on motor symptoms: Postural Instability and Gait Disability (PIGD), tremor dominant (TD) and Intermediate92–94 but it is unclear if gait progresses faster in one group than another. The distinction was operationalized by creating a tremor subscore and PIGD subscore based on items of the Unified Parkinson’s Disease Rating Scale (UPDRS) or the MDS-UPDRS. Overall, the PIGD phenotype has been associated with slower gait speed and reduced stride length compared to the TD phenotype95–99. In addition, the PIGD phenotype also showed higher variability in gait characteristics84, in particular variability of stride time95 and stride length99, compared to the TD phenotype. However, longitudinal studies of digital gait characteristics in PIGD and tremor phenotypes are lacking in the literature, with only one longitudinal study previously cited tracking gait quantitatively84.
In summary, the most promising gait biomarkers to monitor disease progression, despite the paucity of longitudinal studies, appear to be declines in gait pace (stride velocity, stride length and angle of the foot at heel strike), gait variability (stride time), and trunk stability, detected when walking long distances at a comfortable speed, see Table 2. Interestingly, changes in gait are best detected when participants with PD walk at a comfortable speed during single-task gait, as the addition of a concurrent, cognitive dual-task or fast walking does not help in identifying changes due to disease progression, possibly because of variability to response to the secondary task or because instructions to move as fast as possible invoke compensatory mechanisms to ovecome the decline in the automated gait pattern. As the disease progresses, we think that the degeneration of the putamen may also progress and spread to greater bilateral involvement, resulting in clinically-apparent slowness of gait and increased overall variability of gait accompanied by a progressive decline in gait automaticity; however, large-sample longitudinal studies have not yet included measures of arm swing nor turning characteristics of gait, nor molecular imaging. In addition, discrepancies in findings may be associated with performing gait under different medication conditions. This may be important especially early in the disease, since, medications can mask progression of gait in early PD100.
Gait biomarkers for response to gait specific aerobic exercise
Aerobic exercise has emerged as an intervention to address gait impairments for people with various neurologic conditions, including PD. Over the past 10 years, research studies investigating PD have employed gait specific aerobic exercise in a myriad of ways, including walking overground101,102, walking on a traditional treadmill103–107, walking on treadmills that apply concurrent perturbations to the participant108–111, robot-assisted walking112, Nordic walking113,114, walking in a virtual-reality environment115, walking on a curved treadmill116,117, and walking on a treadmill while carrying an additional load118. While gait velocity and distance, captured with a stop-watch and tape measure, are the two gait biomarkers most commonly assessed, other spatial and temporal characteristics have only been occasionally measured. Table 1C summarizes the evidence on gait metric outcomes for aerobic exercise.
Objective measures of gait are more sensitive to change with exercise than clinical measures in people with PD which has a direct impact on the sample size of exercise studies119. Walking velocity and walking distance consistently benefit from aerobic gait exercise in people with PD. In 11 studies that measured changes in gait velocity after aerobic gait exercise, 10 demonstrated a benefit from the intervention101–103,108,109,112,114,115,117,118, while only one did not104. Similarly, all 11 studies that measured the impact of aerobic gait exercise on walking distance demonstrated a benefit from the intervention102–104,106–109,112,113,115,120.
The literature is promising, but less robust for other spatial and temporal gait characteristics, summary in Table 2. Only 4 studies measured the effects of aerobic gait exercise on step length or stride length, but all showed an improvement103,111,117,118. Both studies that measured step time cadence103,117, as well as the single studies that each measured stride-time variability111 or double-support time103, demonstrated improvement from the aerobic gait exercise intervention. Similarly, both studies assessing freezing of gait showed that freezing improved with aerobic exercise, as well112,117. However, the single study looking at step symmetry did not demonstrate a significant benefit from the gait aerobic exercise111. In addition, no gait-specific aerobic exercise studies have examined spatial and temporal aspects of turning during walking.
To investigate the responsiveness of gait biomarkers to exercise, here we only considered studies using exercise with modes that involve gait and are designed to meet moderate or vigorous aerobic exercise intensity levels. Assuming they include interventions that meet the operational definition of gait exercise, studies employing technology (e.g., virtual reality, robotic assistance, etc.) were included. The overwhelming majority of studies considered a limited set of gait biomarkers, such as gait speed or distance, only assessed with a stopwatch, with only four studies using technology to measure gait spatio-temporal parameters and gait stride-time variability. Collectively, aerobic gait exercise improves aspects of gait pace (e.g., speed and distance), spatial and temporal gait parameters, gait variability, and gait stability.
These positive changes in gait were reported for exercise durations ranging from 4 weeks to 6 months and included participants with a range of disease severities, highlighting the importance of exercise in improving mobility across disease progression. Exercise can modify brain function121 and reduce the symptoms of PD122, thus improving gait function as well. The mechanism by which exercise modifies brain function is not well understood but increased cortical vascularity123, increased brain-derived neurotrophic factor, BDNF, and reduced loss of dopamine121,124,125 are among possibilities.
Gait Biomarkers for Prognosis (Falls)
Gait disturbances lead to falls in PD, which can have a profound impact on independence and quality of life126. Given the importance of falls for quality of life, it is an urgent priority to be able to predict those who are at a higher risk of falling early in the course of the disease. Ultimately, this early predictability could enhance our understanding of the mechanisms underlying falls, while also providing opportunity to initiate evidence-based treatments to minimize falls.
Recent studies have suggested that risk of falls might be predicted with the use of digital biomarkers, measured either in clinic or daily-living environments, see Table 1D127–131. For example, Greene et al. 131 used inertial sensors during a long timed-up and Go (TUG) course to predict fall frequency with moderate accuracy, pointing to multiple measures rather than a single measure. When considering walking speed as a marker, two studies suggested that slow backward, but not forward, walking speed had the potential to be a biomarker for future falls, particularly in de-novo PD130,132. Contributing to the debate about self-paced forward walking, Nemanich & colleagues demonstrated that the difference between self-paced walking and fast-paced walking may be more predictive of future falls than self-paced gait alone133. One small study suggested that increased step time variability and increased walking cadence in the Off-medication state can be important predictors of future falls127. Lastly, two recent studies investigated whether gait and turning biomarkers in daily life could predict falls in people with moderate PD128,129. One study showed that specific gait and turning measures (toe-out angle of the foot, pitch angle of the foot during mid-swing, and peak turn velocity) observed during daily life walking achieved an AUC of 0.94 in predicting future falls while the AUC of past falls as marker of future falls was 0.77128. The other study129 compared data aggregation approaches and machine learning models for the prospective prediction of fall risk using gait parameters derived either from continuous real-world recordings or from unsupervised gait tests. The highest balanced accuracy of 0.74 was achieved with a Random Forest Classifier applied to the real-world gait data when aggregating all walking bouts (short and long) and days of each participant.
Collectively and as shown in Table 2, reductions in gait pace (stride length and overall gait velocity), particularly with backward walking134,135, in addition to increased gait variability, appear to be the strongest predictors of falling, although careful consideration of medication state is important, among other factors126. While previous fall history is a strong predictor of future falls136,137, specific gait parameters that may be modifiable, may also be important biomarkers to consider in interventional trials.
Prediction of falls in PD is complex in nature and abnormal gait is only one of many intrinsic and extrinsic fall-risk factors. Larger and longer studies of the role of gait biomarkers in predicting fall risk for people with PD are needed and other factors, such as previous falls, medication, age, standing balance, cognition, and activity involvement need to be taken into consideration. Lastly, backward walking134,135 or mobility in daily life128 may offer more sensitive measures for fall risk, but larger studies are needed to confirm these preliminary findings.
Highlights and conclusion
There is a critical, unmet need to provide practical, quantitative, easily obtained biomarkers of PD as outcomes for clinical trials. This narrative review focused on gait digital biomarkers as optimal candidates because mobility is affected early in PD138 and gait quality dramatically influences quality of life138.
Table 2 summarizes the evidence for emerging gait digital biomarkers for the 4 specific contexts of use (disease susceptibility/risk, progression, exercise response, and fall risk/prognosis). Upper body aspects of gait, such as arm swing range of motion and asymmetry, trunk range of motion, and gait asymmetry seem to be unique biomarkers indicating the potential for developing a disease in individuals who do not currently have a clinically-apparent disease (susceptibility/risk). Pace aspects of gait, such as speed of gait and spatio-temporal characteristics such as stride length, may be most helpful in tracking disease progression, assessing response to exercise and indicating the likelihood of falls. Dynamic stability aspects of gait, such as trunk regularity (how smooth and consistent trunk movement is during gait)139,140 and double-support time, also worsen with disease progression, but can improve with exercise. Gait variability, specifically stride-time variability, seems to be the only gait biomarker sensitive across the 4 contexts of use, reflecting sensitivity but potentially poor specificity. However, our summary of the best gait measures for each context of use is limited to studies that do not include a wide array of measures as upper body and foot measures are often left out. The gait protocol used to collect data is very important. In fact, while single-task gait at a natural, comfortable speed is best for biomarkers for prognosis and disease progression, a more challenging protocol with a concurrent cognitive dual-task or asking to walk at fast speed seem to best indicate disease risk.
Some contextual considerations need to be listed. The relative benefits of collecting digital biomarkers of gait while individuals are in dopaminergic medication on- versus off-state must be considered. The benefits of collecting digital biomarkers of gait while patients are in the on-medication state are both logistical and physiological. Logistically, it is easier to collect data in the on state because subjects are often more comfortable as they have less rigidity, bradykinesia, and tremor. This comfort level is especially important for long-term or home-based monitoring. Also, when collecting gait measures in the on-medication state, the treating team or clinical trial investigators do not have to wait for medication effects to “turn on” (e.g., waiting for the person with PD and the examiner to feel that levodopa has kicked in) as they would need to do for an on/off medication comparison evaluation. Physiologically, collecting digital biomarkers of gait while people with PD are taking their usual medications is more representative of real-life and may capture aspects of the gait exam that are: (1) not improved by dopaminergic medications (e.g., dynamic postural stability)141 or (2) actually worsened by dopaminergic medications (e.g., postural sway and dyskinesia)142. However, there are several benefits of collecting data in the Off dopaminergic medication state, including the ability to assess the natural physiology of PD early in the disease course, the ease of comparing treatment groups without the need to stratify or match for dopamine equivalent doses, and a greater ability to compare groups of people with PD to groups with atypical parkinsonian disorders (e.g., progressive supranuclear palsy, corticobasal syndrome, multiple system atrophy and vascular parkinsonism), if the goal is early disease identification.
Similar to dopaminergic medications, one must consider the milieu of cholinergic and anticholinergic medications (such as donepezil and oxybutynin) as well as the interaction of deep brain stimulation (DBS) for impact on the results of digital biomarkers of gait. Evidence suggests that anticholinergic medications may worsen gait143,144. Since DBS is usually recommended mid to late in the disease course, testing gait when DBS is turned off may be more poorly tolerated and thus not feasible. Fortunately, there are a number of portable technologies to measure gait simultaneous with DBS stimulation without significant interference. Overall, testing digital gait markers off medication (and off DBS, when applicable) may be more helpful early in the disease course and for diagnostic discrimination, while testing on medication and on stimulation on may be more appropriate for advanced disease monitoring.
Although this narrative review highlights the positive results of digital biomarkers of gait, it also shows the paucity of longitudinal trials including digital gait measures, and the lack of comparison of gait digital biomarkers with the MDS-UPDRS, which is the current clinical gold standard used in clinical trials and only used quantitative comparisons in one study. If digital gait biomarkers are more responsive to change than the MDS-UPDRS, it could result in smaller, more efficient clinical trials for PD. Gait biomarkers may also be helpful in Phase II and Phase III clinical trials as an endpoint aimed at slowing the progression or reducing the severity of PD. Lastly, work is needed to standardize gait assessments before more specific gait biomarkers can be recommended, as important factors vary greatly across studies, including distance/time walked, type of concurrent cognitive task, and inclusion of turns, which all potentially affect gait biomarkers.
The advent of automatic, real-time analysis of gait based on body-worn, inertial sensors has made it feasible to quantify gait characteristics in people with PD in the clinic, as well as in clinical trials. Neurologists can use gait analysis to aid in early diagnosis, monitor response to medications and to track disease progression. Physical therapists would benefit from digital gait measures to measure response to exercise intervention as well as to predict risk of falls. Patients may also want to monitor their gait for themselves or for their physical therapists by wearing sensors during daily life. Digital health technologies to quantify gait are already available for clinicians and patients, but evidence on the usefulness of specific gait biomarkers for clinical practice needs to be accumulated. It should also be mentioned that other promising techniques to quickly characterize gait in the clinic are rapidly developing. In fact, advanced computer vision techniques, often combined with machine learning, are used to analyze gait from video recordings145–147. In the future, after careful validation, quantitative analysis of gait could be even more easily available in clinical practice148.
Acknowledgements
The authors would like to thank the community of people affected by Parkinson’s disease, and specifically those who are exercising within the SPARX3 trial. Also, authors would like to thank Graham Harker, BS, MPH, for his help in editing the manuscript.
Author contributions
Design: MM, FH, CC, DMC Execution: MM, MA, QA, SA-H, LD, GME, TE, MEH, KEJ, LL, JAO, KK, GO, AKR Review and critique: MM, QA, SAH, KB, RC, CC, MLD, LD, GME, TE, GJG, MEH, JH, FBH, KEJ, JOK, AKR, AS, DMC. Writing of the first draft: MM, MA, QA, SA-H, LD, GME, TE, MEH, KEJ, LL, JAO, KK, GO, AKR.
Competing interests
Fay Horak is an employee of APDM Wearable Technologies—a Clario company that may have a commercial interest in the results of this research and technology. This potential conflict has been reviewed and managed by OHSU. All other authors declare no Competing Financial or Non-Financial Interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Deng, K. et al. Heterogeneous digital biomarker integration out-performs patient self-reports in predicting Parkinson’s disease. Commun. Biol.5, 58, 10.1038/s42003-022-03002-x (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Stephenson, D., Badawy, R., Mathur, S., Tome, M. & Rochester, L. Digital progression biomarkers as novel endpoints in clinical trials: a multistakeholder perspective. J. Parkinsons Dis.11, S103–S109, 10.3233/JPD-202428 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gerlach, M. et al. Biomarker candidates of neurodegeneration in Parkinson’s disease for the evaluation of disease-modifying therapeutics. J. Neural Transm.119, 39–52, 10.1007/s00702-011-0682-x (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Horak, F. B. & Mancini, M. Objective biomarkers of balance and gait for Parkinson’s disease using body-worn sensors. Mov. Disord.28, 1544–1551, 10.1002/mds.25684 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Coravos, A., Khozin, S. & Mandl, K. D. Developing and adopting safe and effective digital biomarkers to improve patient outcomes. npj Digit. Med.2, 14, 10.1038/s41746-019-0090-4 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Califf, R. M. Biomarker definitions and their applications. Exp. Biol. Med.243, 213–221, 10.1177/1535370217750088 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cagney, D. N. et al. The FDA NIH biomarkers, EndpointS, and other Tools (BEST) resource in neuro-oncology. Neuro Oncol.20, 1162–1172, 10.1093/neuonc/nox242 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lang, A. E. Clinical trials of disease-modifying therapies for neurodegenerative diseases: the challenges and the future. Nat. Med.16, 1223–1226, 10.1038/nm.2220 (2010). [DOI] [PubMed] [Google Scholar]
- 9.Mirelman, A. et al. Gait impairments in Parkinson’s disease. Lancet Neurol.18, 697–708, 10.1016/s1474-4422(19)30044-4 (2019). [DOI] [PubMed] [Google Scholar]
- 10.Tan, D., Danoudis, M., McGinley, J. & Morris, M. E. Relationships between motor aspects of gait impairments and activity limitations in people with Parkinson’s disease: a systematic review. Parkinson. Relat. Disord.18, 117–124, 10.1016/j.parkreldis.2011.07.014 (2012). [DOI] [PubMed] [Google Scholar]
- 11.Berardelli, A., Rothwell, J. C., Thompson, P. D. & Hallett, M. Pathophysiology of bradykinesia in Parkinson’s disease. Brain124, 2131–2146, 10.1093/brain/124.11.2131 (2001). [DOI] [PubMed] [Google Scholar]
- 12.Teshuva, I. et al. Using wearables to assess bradykinesia and rigidity in patients with Parkinson’s disease: a focused, narrative review of the literature. J. Neural Transm.126, 699–710, 10.1007/s00702-019-02017-9 (2019). [DOI] [PubMed] [Google Scholar]
- 13.Fasano, A. & Bloem, B. R. Gait disorders. Continuum19, 1344–1382, 10.1212/01.Con.0000436159.33447.69 (2013). [DOI] [PubMed]
- 14.Amundsen-Huffmaster, S. L. et al. REM sleep without atonia and gait impairment in people with mild-to-moderate Parkinson’s disease. J. Parkinson Dis.11, 767–778, 10.3233/jpd-202098 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cochen De Cock, V. et al. Classifying idiopathic rapid eye movement sleep behavior disorder, controls, and mild parkinson’s disease using gait parameters. Mov. Disord.37, 842–846, 10.1002/mds.28894 (2022). [DOI] [PubMed] [Google Scholar]
- 16.Mirelman, A. et al. Arm swing as a potential new prodromal marker of Parkinson’s disease. Mov. Disord.31, 1527–1534, 10.1002/mds.26720 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lord, S. et al. Independent domains of gait in older adults and associated motor and nonmotor attributes: validation of a factor analysis approach. J. Gerontol. A Biol. Sci. Med. Sci., 10.1093/gerona/gls255 (2012). [DOI] [PubMed]
- 18.Morris, R. et al. A model of free-living gait: A factor analysis in Parkinson’s disease. Gait Posture52, 68–71, 10.1016/j.gaitpost.2016.11.024 (2017). [DOI] [PubMed] [Google Scholar]
- 19.Horak, F. B., Mancini, M., Carlson-Kuhta, P., Nutt, J. G. & Salarian, A. Balance and gait represent independent domains of mobility in Parkinson disease. Phys. Ther.96, 1364–1371, 10.2522/ptj.20150580 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Middleton, A., Fritz, S. L. & Lusardi, M. Walking speed: the functional vital sign. J. Aging Phys. Act.23, 314–322, 10.1123/japa.2013-0236 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Speelman, A. D., van Nimwegen, M., Borm, G. F., Bloem, B. R. & Munneke, M. Monitoring of walking in Parkinson’s disease: validation of an ambulatory activity monitor. Parkinson Relat. Disord.17, 402–404, 10.1016/j.parkreldis.2011.02.006 (2011). [DOI] [PubMed] [Google Scholar]
- 22.Goetz, C. G. et al. Teaching program for the movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale: (MDS-UPDRS). Mov. Disord.25, 1190–1194, 10.1002/mds.23096 (2010). [DOI] [PubMed] [Google Scholar]
- 23.Goetz, C. G. et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov. Disord.23, 2129–2170, 10.1002/mds.22340 (2008). [DOI] [PubMed] [Google Scholar]
- 24.Bloem, B. R. et al. Measurement instruments to assess posture, gait, and balance in Parkinson’s disease: Critique and recommendations. Mov. Disord.31, 1342–1355, 10.1002/mds.26572 (2016). [DOI] [PubMed] [Google Scholar]
- 25.Chan, W. L. S. & Pin, T. W. Reliability, validity and minimal detectable change of 2-minute walk test, 6-minute walk test and 10-meter walk test in frail older adults with dementia. Exp. Gerontol.115, 9–18, 10.1016/j.exger.2018.11.001 (2019). [DOI] [PubMed] [Google Scholar]
- 26.Shah, V. V. et al. Effect of Bout length on gait measures in people with and without Parkinson’s disease during daily life. Sensors20, 10.3390/s20205769 (2020). [DOI] [PMC free article] [PubMed]
- 27.Shah, V. V. et al. Digital biomarkers of mobility in Parkinson’s disease during daily living. J. Parkinsons Dis.10.3233/jpd-201914 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zanardi, A. P. J. et al. Gait parameters of Parkinson’s disease compared with healthy controls: a systematic review and meta-analysis. Sci. Rep.11, 752, 10.1038/s41598-020-80768-2 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dorsey, E. R., Venuto, C., Venkataraman, V., Harris, D. A. & Kieburtz, K. Novel methods and technologies for 21st-century clinical trials: a review. JAMA Neurol.72, 582–588,, 10.1001/jamaneurol.2014.4524 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dorsey, E. R., Papapetropoulos, S., Xiong, M. & Kieburtz, K. The first frontier: digital biomarkers for neurodegenerative disorders. Digit. Biomark.1, 6–13, 10.1159/000477383 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Erb, M. K. et al. mHealth and wearable technology should replace motor diaries to track motor fluctuations in Parkinson’s disease. NPJ Digit. Med.3, 6, 10.1038/s41746-019-0214-x (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sieberts, S. K. et al. Crowdsourcing digital health measures to predict Parkinson’s disease severity: the Parkinson’s Disease Digital Biomarker DREAM Challenge. NPJ Digit. Med.4, 53, 10.1038/s41746-021-00414-7 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Guo, C. C. et al. Digital devices for assessing motor functions in mobility-impaired and healthy populations: systematic literature review. J. Med. Internet Res.24, e37683, 10.2196/37683 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Viceconti, M. et al. On the use of wearable sensors as mobility biomarkers in the marketing authorization of new drugs: A regulatory perspective. Front. Med9, 996903, 10.3389/fmed.2022.996903 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Benedetti, M. G. et al. SIAMOC position paper on gait analysis in clinical practice: General requirements, methods and appropriateness. Results of an Italian consensus conference. Gait Posture58, 252–260, 10.1016/j.gaitpost.2017.08.003 (2017). [DOI] [PubMed] [Google Scholar]
- 36.Cappozzo, A. Gait analysis methodology. Hum. Mov. Sci.3, 27–50, 10.1016/0167-9457(84)90004-6 (1984). [Google Scholar]
- 37.Summan, R. et al. Spatial calibration of large volume photogrammetry based metrology systems. Measurement68, 189–200, 10.1016/j.measurement.2015.02.054 (2015). [Google Scholar]
- 38.Salchow-Hömmen, C. et al. Review-emerging portable technologies for gait analysis in neurological disorders. Front. Hum. Neurosci.16, 768575, 10.3389/fnhum.2022.768575 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Del Din, S., Kirk, C., Yarnall, A. J., Rochester, L. & Hausdorff, J. M. Body-Worn sensors for remote monitoring of Parkinson’s disease motor symptoms: vision, state of the art, and challenges ahead. J. Parkinsons Dis.11, S35–s47, 10.3233/jpd-202471 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Buchthal, F. & Fernandez-Ballesteros, M. L. Electromyographic study of the muscles of the upper arm and shoulder during walking in patients with Parkinson’s disease. Brain88, 875–896, 10.1093/brain/88.5.875 (1965). [DOI] [PubMed] [Google Scholar]
- 41.Zampieri, C. et al. The instrumented timed up and go test: potential outcome measure for disease modifying therapies in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry81, 171–176, 10.1136/jnnp.2009.173740 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Buckley, C., Galna, B., Rochester, L. & Mazzà, C. Upper body accelerations as a biomarker of gait impairment in the early stages of Parkinson’s disease. Gait Posture71, 289–295, 10.1016/j.gaitpost.2018.06.166 (2019). [DOI] [PubMed] [Google Scholar]
- 43.Sadeghi, H., Allard, P., Prince, F. & Labelle, H. Symmetry and limb dominance in able-bodied gait: a review. Gait Posture12, 34–45, 10.1016/s0966-6362(00)00070-9 (2000). [DOI] [PubMed] [Google Scholar]
- 44.Yogev, G., Plotnik, M., Peretz, C., Giladi, N. & Hausdorff, J. M. Gait asymmetry in patients with Parkinson’s disease and elderly fallers: when does the bilateral coordination of gait require attention? Exp. Brain Res.177, 336–346, 10.1007/s00221-006-0676-3 (2007). [DOI] [PubMed] [Google Scholar]
- 45.Galna, B., Lord, S. & Rochester, L. Is gait variability reliable in older adults and Parkinson’s disease? Towards an optimal testing protocol. Gait Posture37, 580–585, 10.1016/j.gaitpost.2012.09.025 (2013). [DOI] [PubMed] [Google Scholar]
- 46.Rennie, L. et al. The reliability of gait variability measures for individuals with Parkinson’s disease and healthy older adults - The effect of gait speed. Gait Posture62, 505–509, 10.1016/j.gaitpost.2018.04.011 (2018). [DOI] [PubMed] [Google Scholar]
- 47.Kuo, A. D. & Donelan, J. M. Dynamic principles of gait and their clinical implications. Phys. Ther.90, 157–174, 10.2522/ptj.20090125 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ma, L. et al. Gait variability is sensitive to detect Parkinson’s disease patients at high fall risk. Int. J. Neurosci.132, 888–893, 10.1080/00207454.2020.1849189 (2022). [DOI] [PubMed] [Google Scholar]
- 49.Morrison, S. et al. The relation between falls risk and movement variability in Parkinson’s disease. Exp. Brain Res.239, 2077–2087, 10.1007/s00221-021-06113-9 (2021). [DOI] [PubMed] [Google Scholar]
- 50.Hausdorff, J. M. Stride variability: beyond length and frequency. Gait Posture20, 304, 10.1016/j.gaitpost.2003.08.002 (2004). [DOI] [PubMed] [Google Scholar]
- 51.Hausdorff, J. M. Gait variability: methods, modeling and meaning. J. Neuroeng. Rehabil.2, 19, 10.1186/1743-0003-2-19 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Raffegeau, T. E. et al. A meta-analysis: Parkinson’s disease and dual-task walking. Parkinson Relat. Disord.62, 28–35, 10.1016/j.parkreldis.2018.12.012 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kelly, V. E., Eusterbrock, A. J. & Shumway-Cook, A. A review of dual-task walking deficits in people with Parkinson’s disease: motor and cognitive contributions, mechanisms, and clinical implications. Parkinsons Dis.2012, 918719, 10.1155/2012/918719 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Vitorio, R. et al. Dual-task costs of quantitative gait parameters while walking and turning in people with Parkinson’s disease: beyond gait speed. J. Parkinsons Dis.11, 653–664, 10.3233/jpd-202289 (2021). [DOI] [PubMed] [Google Scholar]
- 55.Mirelman, A. et al. Gait alterations in healthy carriers of the LRRK2 G2019S mutation. Ann. Neurol.69, 193–197, 10.1002/ana.22165 (2011). [DOI] [PubMed] [Google Scholar]
- 56.Peterson, D. S. & Horak, F. B. Neural Control of Walking in People with Parkinsonism. Physiol. (Bethesda)31, 95–107, 10.1152/physiol.00034.2015 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Horak, F. B., Mancini, M., Carlson-Kuhta, P., Nutt, J. G. & Salarian, A. Balance and gait represent independent domains of mobility in Parkinson’s Disease. Phys. Ther.96, 1364–1371, 10.2522/ptj.20150580 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Postuma, R. B. et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord.30, 1591–1601, 10.1002/mds.26424 (2015). [DOI] [PubMed] [Google Scholar]
- 59.FEARNLEY, J. M. & LEES, A. J. Ageing and Parkinson’s disease: substantia Nigra regional selectivity. Brain114, 2283–2301, 10.1093/brain/114.5.2283 (1991). [DOI] [PubMed] [Google Scholar]
- 60.Mirelman, A. et al. Fall risk and gait in Parkinson’s disease: the role of the LRRK2 G2019S mutation. Mov. Disord.28, 1683–1690, 10.1002/mds.25587 (2013). [DOI] [PubMed] [Google Scholar]
- 61.Sánchez-Rodríguez, A. et al. Sensor-based gait analysis in the premotor stage of LRRK2 G2019S-associated Parkinson’s disease. Parkinson. Relat. Disord.98, 21–26, 10.1016/j.parkreldis.2022.03.020 (2022). [DOI] [PubMed] [Google Scholar]
- 62.van den Heuvel, L. et al. Actigraphy detects greater intra-individual variability during gait in non-manifesting LRRK2 mutation carriers. J. Parkinsons Dis.8, 131–139, 10.3233/jpd-171151 (2018). [DOI] [PubMed] [Google Scholar]
- 63.Prasuhn, J. et al. Task matters - challenging the motor system allows distinguishing unaffected Parkin mutation carriers from mutation-free controls. Parkinson. Relat. Disord.86, 101–104, 10.1016/j.parkreldis.2021.03.028 (2021). [DOI] [PubMed] [Google Scholar]
- 64.Nürnberger, L. et al. Ultrasound-based motion analysis demonstrates bilateral arm hypokinesia during gait in heterozygous PINK1 mutation carriers. Mov. Disord.30, 386–392, 10.1002/mds.26127 (2015). [DOI] [PubMed] [Google Scholar]
- 65.Gera, A. et al. Gait asymmetry in glucocerebrosidase mutation carriers with Parkinson’s disease. PLoS One15, e0226494, 10.1371/journal.pone.0226494 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Martini, D. N. et al. Sensorimotor inhibition and mobility in genetic subgroups of Parkinson’s disease. Front. Neurol.11, 893, 10.3389/fneur.2020.00893 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Morris, R. et al. Cognition as a mediator for gait and balance impairments in GBA-related Parkinson’s disease. NPJ Parkinsons Dis.8, 78, 10.1038/s41531-022-00344-5 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Srulijes, K. et al. Dual-task performance in GBA Parkinson’s disease. Parkinsons Dis.2017, 8582740, 10.1155/2017/8582740 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Iranzo, A. et al. Rapid-eye-movement sleep behaviour disorder as an early marker for a neurodegenerative disorder: a descriptive study. Lancet Neurol.5, 572–577, 10.1016/s1474-4422(06)70476-8 (2006). [DOI] [PubMed] [Google Scholar]
- 70.Postuma, R. B. et al. Quantifying the risk of neurodegenerative disease in idiopathic REM sleep behavior disorder. Neurology72, 1296–1300, 10.1212/01.wnl.0000340980.19702.6e (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Postuma, R. B. et al. Risk and predictors of dementia and parkinsonism in idiopathic REM sleep behaviour disorder: a multicentre study. Brain142, 744–759, 10.1093/brain/awz030 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Del Din, S. et al. Continuous real-world gait monitoring in Idiopathic REM sleep behavior disorder. J. Parkinsons Dis.10, 283–299, 10.3233/jpd-191773 (2020). [DOI] [PubMed] [Google Scholar]
- 73.McDade, E. M. et al. Subtle gait changes in patients with REM sleep behavior disorder. Mov. Disord.28, 1847–1853, 10.1002/mds.25653 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ma, L. et al. Detection of motor dysfunction with wearable sensors in patients with idiopathic rapid eye movement disorder. Front. Bioeng. Biotechnol.9, 627481, 10.3389/fbioe.2021.627481 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ehgoetz Martens, K. A. et al. Subtle gait and balance impairments occur in idiopathic rapid eye movement sleep behavior disorder. Mov. Disord.34, 1374–1380, 10.1002/mds.27780 (2019). [DOI] [PubMed] [Google Scholar]
- 76.Ehgoetz Martens, K. A. et al. The neural signature of impaired dual-tasking in idiopathic rapid eye movement sleep behavior disorder patients. Mov. Disord.35, 1596–1606, 10.1002/mds.28114 (2020). [DOI] [PubMed] [Google Scholar]
- 77.Tamburini, P. et al. Moving from laboratory to real life conditions: Influence on the assessment of variability and stability of gait. Gait Posture59, 248–252, 10.1016/j.gaitpost.2017.10.024 (2018). [DOI] [PubMed] [Google Scholar]
- 78.Zhang, H. et al. Risk factors for phenoconversion in rapid eye movement sleep behavior disorder. Ann. Neurol.91, 404–416, 10.1002/ana.26298 (2022). [DOI] [PubMed] [Google Scholar]
- 79.Monje, M. H. G. et al. Motor onset Topography and progression in Parkinson’s disease: the upper limb is first. Mov. Disord.36, 905–915, 10.1002/mds.28462 (2021). [DOI] [PubMed] [Google Scholar]
- 80.Pineda-Pardo, J. A., Sánchez-Ferro, Á., Monje, M. H. G., Pavese, N. & Obeso, J. A. Onset pattern of nigrostriatal denervation in early Parkinson’s disease. Brain145, 1018–1028, 10.1093/brain/awab378 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Buckley, C. et al. The role of movement analysis in diagnosing and monitoring neurodegenerative conditions: insights from gait and postural control. Brain Sci.9, 34, 10.3390/brainsci9020034 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Sarkar, S., Raymick, J. & Imam, S. Neuroprotective and therapeutic strategies against Parkinson’s disease: recent perspectives. Int. J. Mol. Sci.17, 904, 10.3390/ijms17060904 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Evans, J. R. et al. The natural history of treated Parkinson’s disease in an incident, community based cohort. J. Neurol. Neurosurg. Psychiatry82, 1112–1118, 10.1136/jnnp.2011.240366 (2011). [DOI] [PubMed] [Google Scholar]
- 84.Galna, B., Lord, S., Burn, D. J. & Rochester, L. Progression of gait dysfunction in incident Parkinson’s disease: impact of medication and phenotype. Mov. Disord.30, 359–367, 10.1002/mds.26110 (2015). [DOI] [PubMed] [Google Scholar]
- 85.Ellis, T. D. et al. Identifying clinical measures that most accurately reflect the progression of disability in Parkinson disease. Parkinson. Relat. Disord.25, 65–71, 10.1016/j.parkreldis.2016.02.006 (2016). [DOI] [PubMed] [Google Scholar]
- 86.Salarian, A. et al. Analyzing 180 degrees turns using an inertial system reveals early signs of progression of Parkinson’s disease. Conf. Proc. IEEE Eng. Med. Biol. Soc.2009, 224–227, 10.1109/IEMBS.2009.5333970 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hobert, M. A. et al. Progressive gait deficits in Parkinson’s disease: a wearable-based biannual 5-year prospective study. Front. Aging Neurosci.11, 22, 10.3389/fnagi.2019.00022 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Micó-Amigo, M. E. et al. Potential markers of progression in idiopathic Parkinson’s disease derived from assessment of circular gait with a single body-fixed-sensor: a 5 year longitudinal Study. Front. Hum. Neurosci.13, 59, 10.3389/fnhum.2019.00059 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Micó-Amigo, M. E. et al. Dual vs. single tasking during circular walking: what better reflects progression in Parkinson’s disease? Front. Neurol.10, 372, 10.3389/fneur.2019.00372 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wilson, J. et al. Gait progression over 6 years in Parkinson’s disease: effects of age, medication, and pathology. Front. Aging Neurosci.12, 577435, 10.3389/fnagi.2020.577435 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Sotirakis, C. et al. Identification of motor progression in Parkinson’s disease using wearable sensors and machine learning. NPJ Parkinsons Dis.9, 142, 10.1038/s41531-023-00581-2 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Álvarez, I., Latorre, J., Aguilar, M., Pastor, P. & Llorens, R. Validity and sensitivity of instrumented postural and gait assessment using low-cost devices in Parkinson’s disease. J. Neuroeng. Rehabil.17, 149, 10.1186/s12984-020-00770-7 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lee, S. H. et al. Parkinson’s disease subtyping using clinical features and biomarkers: literature review and preliminary study of subtype clustering. Diagnostics12, 112, 10.3390/diagnostics12010112 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.von Coelln, R. & Shulman, L. M. Clinical subtypes and genetic heterogeneity: of lumping and splitting in Parkinson disease. Curr. Opin. Neurol.29, 727–734, 10.1097/wco.0000000000000384 (2016). [DOI] [PubMed] [Google Scholar]
- 95.Herman, T., Weiss, A., Brozgol, M., Giladi, N. & Hausdorff, J. M. Identifying axial and cognitive correlates in patients with Parkinson’s disease motor subtype using the instrumented Timed Up and Go. Exp. Brain Res.232, 713–721, 10.1007/s00221-013-3778-8 (2014). [DOI] [PubMed] [Google Scholar]
- 96.Koh, S. B., Park, K. W., Lee, D. H., Kim, S. J. & Yoon, J. S. Gait analysis in patients with Parkinson’s disease: relationship to clinical features and freezing. J. Mov. Disord.1, 59–64 (2008). [Google Scholar]
- 97.Orcioli-Silva, D. et al. Objective measures of unobstructed walking and obstacle avoidance in Parkinson’s disease subtypes. Gait Posture62, 405–408, 10.1016/j.gaitpost.2018.03.046 (2018). [DOI] [PubMed] [Google Scholar]
- 98.Vervoort, G. et al. Distal motor deficit contributions to postural instability and gait disorder in Parkinson’s disease. Behav. Brain Res.287, 1–7, 10.1016/j.bbr.2015.03.026 (2015). [DOI] [PubMed] [Google Scholar]
- 99.Wu, Z. et al. Can quantitative gait analysis be used to guide treatment of patients with different subtypes of Parkinson’s disease? Neuropsychiatr. Dis. Treat.16, 2335–2341, 10.2147/ndt.S266585 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Brzezicki, M. A., Conway, N., Sotirakis, C., FitzGerald, J. J. & Antoniades, C. A. AntiParkinsonian medication masks motor signal progression in de novo patients. Heliyon9, e16415, 10.1016/j.heliyon.2023.e16415 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Uc, E. Y. et al. Phase I/II randomized trial of aerobic exercise in Parkinson disease in a community setting. Neurology83, 413–425, 10.1212/wnl.0000000000000644 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Mak, M. K. Y. & Wong-Yu, I. S. K. Six-month community-based brisk walking and balance exercise alleviates motor symptoms and promotes functions in people with parkinson’s disease: a randomized controlled trial. J. Parkinsons Dis.11, 1431–1441, 10.3233/jpd-202503 (2021). [DOI] [PubMed] [Google Scholar]
- 103.Nadeau, A., Pourcher, E. & Corbeil, P. Effects of 24 wk of treadmill training on gait performance in Parkinson’s disease. Med. Sci. Sports Exerc.46, 645–655, 10.1249/mss.0000000000000144 (2014). [DOI] [PubMed] [Google Scholar]
- 104.Di Martino, S. et al. Aerobic rehabilitation program for improving muscle function in Parkinson’s disease. Restor. Neurol. Neurosci.36, 13–20, 10.3233/rnn-170738 (2018). [DOI] [PubMed] [Google Scholar]
- 105.Schenkman, M. et al. Effect of high-intensity treadmill exercise on motor symptoms in patients with de novo Parkinson disease: a phase 2 randomized clinical trial. JAMA Neurol.75, 219–226, 10.1001/jamaneurol.2017.3517 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Arfa-Fatollahkhani, P. et al. Effects of treadmill training on the balance, functional capacity and quality of life in Parkinson’s disease: A randomized clinical trial. J. Complement. Integr. Med.17, 10.1515/jcim-2018-0245 (2019). [DOI] [PubMed]
- 107.Soke, F., Guclu-Gunduz, A., Kocer, B., Fidan, I. & Keskinoglu, P. Task-oriented circuit training combined with aerobic training improves motor performance and balance in people with Parkinson’s Disease. Acta Neurol. Belg.121, 535–543, 10.1007/s13760-019-01247-8 (2021). [DOI] [PubMed] [Google Scholar]
- 108.Shulman, L. M. et al. Randomized clinical trial of 3 types of physical exercise for patients with Parkinson disease. JAMA Neurol.70, 183–190,, 10.1001/jamaneurol.2013.646 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Steib, S. et al. Perturbation during treadmill training improves dynamic balance and gait in Parkinson’s disease: a single-blind randomized controlled pilot trial. Neurorehabil. Neural Repair31, 758–768, 10.1177/1545968317721976 (2017). [DOI] [PubMed] [Google Scholar]
- 110.Gaßner, H. et al. Perturbation treadmill training improves clinical characteristics of gait and balance in Parkinson’s disease. J. Parkinsons Dis.9, 413–426, 10.3233/jpd-181534 (2019). [DOI] [PubMed] [Google Scholar]
- 111.Steib, S. et al. Exploring gait adaptations to perturbed and conventional treadmill training in Parkinson’s disease: Time-course, sustainability, and transfer. Hum. Mov. Sci.64, 123–132, 10.1016/j.humov.2019.01.007 (2019). [DOI] [PubMed] [Google Scholar]
- 112.Capecci, M. et al. Clinical effects of robot-assisted gait training and treadmill training for Parkinson’s disease. A randomized controlled trial. Ann. Phys. Rehabil. Med.62, 303–312, 10.1016/j.rehab.2019.06.016 (2019). [DOI] [PubMed] [Google Scholar]
- 113.Cugusi, L. et al. Effects of a Nordic Walking program on motor and non-motor symptoms, functional performance and body composition in patients with Parkinson’s disease. NeuroRehabilitation37, 245–254, 10.3233/nre-151257 (2015). [DOI] [PubMed] [Google Scholar]
- 114.Monteiro, E. P. et al. Effects of Nordic walking training on functional parameters in Parkinson’s disease: a randomized controlled clinical trial. Scand. J. Med. Sci. Sports27, 351–358, 10.1111/sms.12652 (2017). [DOI] [PubMed] [Google Scholar]
- 115.de Melo, G. E. L. et al. Effect of virtual reality training on walking distance and physical fitness in individuals with Parkinson’s disease. NeuroRehabilitation42, 473–480, 10.3233/nre-172355 (2018). [DOI] [PubMed] [Google Scholar]
- 116.Cheng, F. Y. et al. Positive effects of specific exercise and novel turning-based treadmill training on turning performance in individuals with Parkinson’s disease: a randomized controlled trial. Sci. Rep.6, 33242, 10.1038/srep33242 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Cheng, F. Y., Yang, Y. R., Wu, Y. R., Cheng, S. J. & Wang, R. Y. Effects of curved-walking training on curved-walking performance and freezing of gait in individuals with Parkinson’s disease: A randomized controlled trial. Parkinson. Relat. Disord.43, 20–26, 10.1016/j.parkreldis.2017.06.021 (2017). [DOI] [PubMed] [Google Scholar]
- 118.Trigueiro, L. C. et al. Effects of treadmill training with load on gait in Parkinson disease: a randomized controlled clinical trial. Am. J. Phys. Med. Rehabil.94, 830–837, 10.1097/phm.0000000000000249 (2015). [DOI] [PubMed] [Google Scholar]
- 119.Hasegawa, N. et al. Responsiveness of objective versus clinical balance domain outcomes for exercise intervention in Parkinson’s disease. Front. Neurol.11, 940, 10.3389/fneur.2020.00940 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Rose, M. H., Løkkegaard, A., Sonne-Holm, S. & Jensen, B. R. Improved clinical status, quality of life, and walking capacity in Parkinson’s disease after body weight-supported high-intensity locomotor training. Arch. Phys. Med. Rehabil.94, 687–692, 10.1016/j.apmr.2012.11.025 (2013). [DOI] [PubMed] [Google Scholar]
- 121.Petzinger, G. M. et al. Enhancing neuroplasticity in the basal ganglia: the role of exercise in Parkinson’s disease. Mov. Disord.25, S141–S145, 10.1002/mds.22782 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Robichaud, J. A. & Corcos, D. M. Motor deficits, exercise, and Parkinson’s disease. Quest57, 79–101, 10.1080/00336297.2005.10491844 (2005). [Google Scholar]
- 123.Rhyu, I. J. et al. Effects of aerobic exercise training on cognitive function and cortical vascularity in monkeys. Neuroscience167, 1239–1248, 10.1016/j.neuroscience.2010.03.003 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Fisher, B. E. et al. The effect of exercise training in improving motor performance and corticomotor excitability in people with early Parkinson’s disease. Arch. Phys. Med. Rehabil.89, 1221–1229, 10.1016/j.apmr.2008.01.013 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Vučković, M. G. et al. Exercise elevates dopamine D2 receptor in a mouse model of Parkinson’s disease: in vivo imaging with [18F]fallypride. Mov. Disord.25, 2777–2784, 10.1002/mds.23407 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Fasano, A., Canning, C. G., Hausdorff, J. M., Lord, S. & Rochester, L. Falls in Parkinson’s disease: A complex and evolving picture. Mov. Disord.32, 1524–1536, 10.1002/mds.27195 (2017). [DOI] [PubMed] [Google Scholar]
- 127.Hoskovcová, M. et al. Predicting falls in Parkinson disease: what is the value of instrumented testing in OFF medication state? PLoS One10, e0139849, 10.1371/journal.pone.0139849 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Shah, V. V. et al. Gait and turning characteristics from daily life increase ability to predict future falls in people with Parkinson’s disease. Front. Neurol.14, 1096401, 10.3389/fneur.2023.1096401 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Ullrich, M. et al. Fall risk Prediction in Parkinson’s disease using real-world inertial sensor gait data. IEEE J. Biomed. Health Inf.27, 319–328, 10.1109/jbhi.2022.3215921 (2023). [DOI] [PubMed] [Google Scholar]
- 130.Kwon, K. Y. et al. Association between baseline gait parameters and future fall risk in patients with De Novo Parkinson’s disease: forward versus backward gait. J. Clin. Neurol., 10.3988/jcn.2022.0299 (2024). [DOI] [PMC free article] [PubMed]
- 131.Greene, B. R., Premoli, I., McManus, K., McGrath, D. & Caulfield, B. Predicting fall counts using wearable sensors: a novel digital biomarker for Parkinson’s Disease. Sensors22, 54, 10.3390/s22010054 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Duncan, R. P. & Earhart, G. M. Should one measure balance or gait to best predict falls among people with Parkinson disease? Parkinson’s. Dis.2012, 923493, 10.1155/2012/923493 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Nemanich, S. T. et al. Predictors of gait speeds and the relationship of gait speeds to falls in men and women with Parkinson disease. Parkinsons Dis.2013, 141720, 10.1155/2013/141720 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Hackney, M. E. & Earhart, G. M. Backward walking in Parkinson’s disease. Mov. Disord.24, 218–223, 10.1002/mds.22330 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Hackney, M. E. & Earhart, G. M. The effects of a secondary task on forward and backward walking in Parkinson’s disease. Neurorehabil. Neural Repair24, 97–106, 10.1177/1545968309341061 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Canning, C. G., Paul, S. S. & Nieuwboer, A. Prevention of falls in Parkinson’s disease: a review of fall risk factors and the role of physical interventions. Neurodegener. Dis. Manag.4, 203–221, 10.2217/nmt.14.22 (2014). [DOI] [PubMed] [Google Scholar]
- 137.Pickering, R. M. et al. A meta-analysis of six prospective studies of falling in Parkinson’s disease. Mov. Disord.22, 1892–1900, 10.1002/mds.21598 (2007). [DOI] [PubMed] [Google Scholar]
- 138.Mammen, J. R. et al. Relative Meaningfulness and Impacts of Symptoms in People with Early-Stage Parkinson’s Disease. J. Parkinsons Dis.13, 619–632, 10.3233/jpd-225068 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Moe-Nilssen, R., Aaslund, M. K., Hodt-Billington, C. & Helbostad, J. L. Gait variability measures may represent different constructs. Gait Posture32, 98–101, 10.1016/j.gaitpost.2010.03.019 (2010). [DOI] [PubMed] [Google Scholar]
- 140.Yack, H. J. & Berger, R. C. Dynamic stability in the elderly: identifying a possible measure. J. Gerontol.48, M225–230,, 10.1093/geronj/48.5.m225 (1993). [DOI] [PubMed] [Google Scholar]
- 141.Jansen, J. A. F. et al. Exploring the levodopa-paradox of freezing of gait in dopaminergic medication-naïve Parkinson’s disease populations. NPJ Parkinsons Dis.9, 130, 10.1038/s41531-023-00575-0 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Curtze, C., Nutt, J. G., Carlson-Kuhta, P., Mancini, M. & Horak, F. B. Levodopa Is a Double-Edged Sword for Balance and Gait in People With Parkinson’s Disease. Mov. Disord.30, 1361–1370, 10.1002/mds.26269 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Duvoisin, R. C. Cholinergic-anticholinergic antagonism in parkinsonism. Arch. Neurol.17, 124–136, 10.1001/archneur.1967.00470260014002 (1967). [DOI] [PubMed] [Google Scholar]
- 144.Bohnen, N. I. et al. Regional cerebral cholinergic nerve terminal integrity and cardinal motor features in Parkinson’s disease. Brain Commun.3, fcab109, 10.1093/braincomms/fcab109 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Hu, K. et al. Vision-Based Freezing of Gait Detection With Anatomic Directed Graph Representation. IEEE J. Biomed. Health Inf.24, 1215–1225, 10.1109/jbhi.2019.2923209 (2020). [DOI] [PubMed] [Google Scholar]
- 146.Sabo, A., Iaboni, A., Taati, B., Fasano, A. & Gorodetsky, C. Evaluating the ability of a predictive vision-based machine learning model to measure changes in gait in response to medication and DBS within individuals with Parkinson’s disease. Biomed. Eng. Online22, 120, 10.1186/s12938-023-01175-y (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Stenum, J., Hsu, M. M., Pantelyat, A. Y. & Roemmich, R. T. Clinical gait analysis using video-based pose estimation: Multiple perspectives, clinical populations, and measuring change. PLOS Digit Health3, e0000467, 10.1371/journal.pdig.0000467 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Alberts, J. L. et al. The Parkinson’s disease waiting room of the future: measurements, not magazines. Front. Neurol.14, 1212113, 10.3389/fneur.2023.1212113 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]