Abstract
Myxoid pleomorphic liposarcoma is a rare and aggressive subtype of soft tissue sarcomas (STS). It primarily arises from adipose tissue and exhibits a high rate of recurrence and metastatic potential. We report the case of a 35-year-old male gym trainer with a 5-month history of a painless, progressively enlarging mass on the right posterior aspect of chest, diagnosed with myxoid pleomorphic liposarcoma following imaging and histopathological evaluation of the excised specimen. Surgical excision with clear margins and adjuvant radiotherapy resulted in a favorable outcome with no recurrence at 7 months. This case emphasizes the importance of early diagnosis and multidisciplinary approach in managing a rare soft tissue sarcoma to prevent complications from a delayed intervention.
Keywords: Myxoid pleomorphic liposarcoma, Soft tissue sarcoma, Oncology, Case report
Introduction
Sarcomas are a rare group of tumors that represent only about <1% of all malignancies in adults [[1], [2], [3]]. Sarcomas are a diverse group of malignant tumors that arise predominantly from connective tissues, including bone and soft tissue [[4], [5], [6]]. Clinically, sarcomas present as large masses due to their silent nature until they reach a significant size or begin encroaching onto the vital organs or nerves [5,7]. They are classified into >100 types based on their tissue of origin and cellular architecture [2]. On the basis of tissue of origin, sarcomas are broadly classified into soft tissue sarcomas and bone sarcomas [8]. Among these, soft tissue sarcomas (STS) constitute ∼80% while bone sarcomas constitute only about ∼20% of sarcomas [9,10]. Soft tissue sarcomas (STS) are rare solid tumors characterized by their heterogeneity and can arise from fat, muscle, nerves, blood vessels, and other connective tissues [7]. They are further classified into ∼70 subtypes based on their histology [2] with leiomyosarcoma, myxofibrosarcoma, unclassified sarcoma, peripheral nerve sheath tumor, fibrosarcoma, and liposarcoma being the most common histological types [[11], [12], [13]]. Among rare ones is the myxoid pleomorphic liposarcoma, which is an aggressive type of adipocytic soft tissue sarcoma [2]. Soft tissue sarcomas have been linked to a variety of factors that play role in their development; including trauma, genetic predisposition, medications (e.g., immunosuppressive drugs, chloramphenicol, alkylating agents, androgen-anabolic steroids), and certain viruses (e.g., HIV, HHV-8), external radiation exposure, smokeless tobacco, occupational exposures such as phenoxy herbicides and increasing age [8,[14], [15], [16]]. Though soft tissue sarcomas (STS) are rare, high-grade STS have a higher rate of metastasis and local recurrence with lungs being the most common site for metastatic spread [17]. Treatment of soft tissue sarcomas depend upon the tumor grade, location, size, histologic type and status of metastasis, and typically includes surgery and radiotherapy, with certain cases i.e. deep seated, locally advanced or metastasized lesions requiring chemotherapy [7,17,18].
Due to the extremely rare nature of myxoid pleomorphic liposarcomas, only a few cases have been reported in the literature. Here, we present a case report of a 35-year-old male with a soft tissue sarcoma, specifically myxoid pleomorphic liposarcoma of the right posterior aspect of chest.
Case presentation
Following is the case report of a 35 y/o male with no co-morbidities, who presented to the out-patient department with the complaint of a slowly growing swelling on his right upper back for 5 months. He complained of mild discomfort when laying down with no associated pain, fever or weight-loss. He also complained of multiple smaller swellings on multiple body areas including right arm, right thigh and left anterior chest, that were not increasing in size. There was no associated chest pain, shortness of breath or cough. The swelling was not associated with any sort of rash, itching or skin discoloration. His systemic review was nonsignificant. The patient had a history of steroid intake for the purpose of bodybuilding for almost 2.5 years 3 months back, for which no record was available. Other than that, there was no family history positive for any congenital diseases or cancer-related morbidity/ mortality. He worked as a gym trainer and didn't report any sort of physical trauma or radiation exposure.
On examination, he was alert, oriented and his observations were within normal range. There was no cervical, supraclavicular, axillary or inguinal lymphadenopathy. No facial deformity or temporal wasting was observed. The patient had 3 small swellings, each measuring 1 × 1 cm on right arm, right thigh and left anterior chest wall. They had smooth surface with soft consistency and no attachment to deep tissue or overlying skin. Slip sign was positive for each of the swelling. A larger 15 × 12 cm globular swelling was noted on the posterior aspect of the right side of chest along the lower and lateral border of right scapula. There was no discoloration, erythema, ulceration, scar marks, draining sinus, visible veins or pulsations in the overlying skin. The swelling itself was nontender with temperature comparable to the normal surrounding skin. The swelling was firm in consistency, had a smooth surface with well-defined margins. It was nonfluctuant, noncompressible, and didn't show any transillumination. It was mobile in all directions and the overlying skin was pinchable. Borders of the scapula were separately palpable. The swelling lateralized with over-head abduction of right arm and it became fixed when the patient was asked to press on the pelvic girdle with his hands (extension and adduction at shoulder joint) (Fig. 1). It had a dull percussion note with no audible bruit. Bilateral chest movements were equal with normal vesicular breathing on auscultation. No added sounds were noted. Rest of the examination revealed no abnormal findings. Based on the history and examination, differential diagnosis of soft tissue sarcoma (STS), lipoma and neurofibromatosis were considered.
Fig. 1.
Preoperative picture showing a swelling location on the right side of back of chest, with lateralization on over-head abduction of the right arm.
Diagnostic assessment
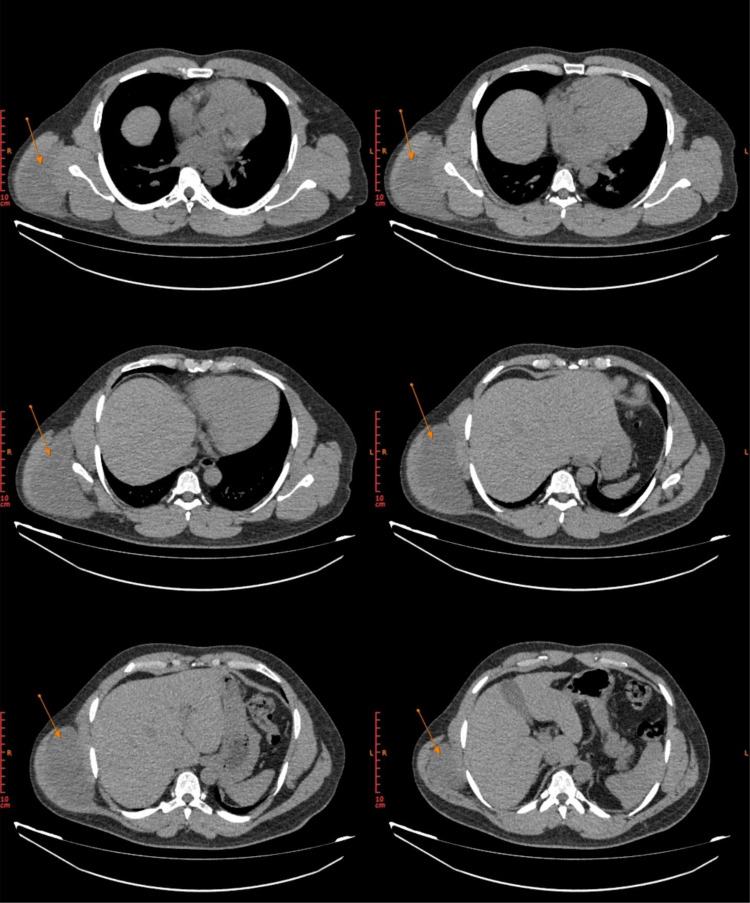
Following the diagnostic protocol, investigations were requested. Patient's baseline investigation (FBC, LFTs, RFTs, S/E) were all within normal range. Chest X-ray showed no pulmonary pathology. USG of the swelling was arranged, which revealed a large echogenic subcutaneous swelling suggestive of a lipoma. Following ultrasound, an FNAC was advised. Micropscopy revealed suspicious looking atypical cells with moderate pleomorphism, irregular nuclear membrane and nuclear hyperchromasia with chondromyxoid background. All the features being suggestive of a spindle cell neoplasm. Based on these investigations, for the accurate localization of the tumor and visualization of any gross metastatic lesions, CT-CAP was requested. CT revealed a well-defined lesion measuring 15 × 10 × 5 cm in the intermuscular plane of right shoulder region below and lateral to the lateral blade of scapula deep to the latissimus dorsi, with lateral displacement of the muscle. However, no local infiltration of the muscle was noted and underlying bone was unremarkable. Additionally, there was no regional lymphadenopathy. All the viscera and organs appeared normal and there was no evidence of metastatic lesions on CT-CAP (Fig. 2).
Fig. 2.
CT films show a right sided mass lesion (arrowhead) in the intermuscular plane deep to latissimus dorsi muscle located lateral to, and extending inferiorly from the scapula. Underlying bone is unremarkable.
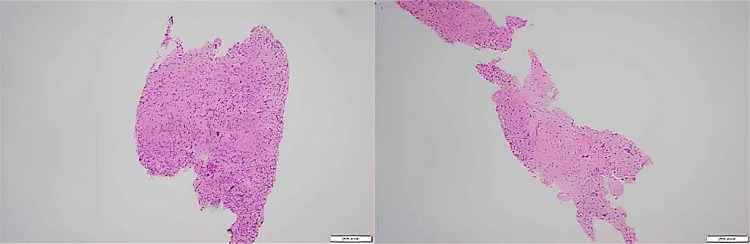
Following these investigations a core biopsy (Tru-Cut biopsy) was requested, which identified spindle cells with ovoid nuclei and tapering eosinophilic neoplasm (Fig. 3). The lesion was found to be microscopically infiltrating into the adjacent muscle. The results of the immune-staining are tabulated below:
| S-100 | -ve |
| SOX-10 | -ve |
| CD-34 | -ve |
Fig. 3.
histological images of the Tru-Cut biopsy showing spindle cells with ovoid nuclei with tapering eosinophilic cytoplasm.
Accordingly, an MRI with contrast was arranged, which revealed a well-defined lobulated heterogenous looking altered signal intensity lesion, T2WI/STIR hyperintense, isointense to muscles on T1 with few patchy internal hyperintense signals, showing heterogenous post contrast enhancement on T1W1. The lesions appeared to be originating from intermuscular plane and involving latissimus dorsi muscle. Underlying bone was found intact with no lymph node involvement (Figs. 4 and 5).
Fig. 4.
MRI Rt. Shoulder shows a well-defined lobulated heterogenous looking altered signal intensity lesion on T2WI/STIR; first row (axial), second row: left (coronal), right (sagittal).
Fig. 5.
MRI Rt. Shoulder shows mass lesion signals isointense to muscles on T1W1/FSE (first row, axial; second row, coronal), with postcontrast enhancement (third row: left, axial; right, coronal). Further, signal void of vessels is unremarkable.
Staging was calculated as stage 3b (T3N0M0).
Treatment and outcome
Based on the investigations, a complete excision of the lesion was planned. The procedure was carried out in general anesthesia. An elliptical skin incision was given and the tumor was dissected out along with 1 cm of muscle margins including periosteum of right inferior angle of scapula (Fig. 6).
Fig. 6.
Intraoperative picture showing excised lesion.
The specimen was labelled, fixed and sent for analysis. Biopsy report revealed the lesion to be myxoid pleomorphic liposarcoma with a histologic grade 3 and negative specimen margins. Immunohistochemical stains on excisional biopsy were:
| Desmin | -ve |
| SMA | -ve |
| S-100 | -ve |
| H3k27me3 | Intact nuclear expression |
After recovering from surgery, the patient was referred to the oncology department for adjuvant radiotherapy. About 25 sessions of radiotherapy were carried out over the course of 3 months. Follow-up CT scan was disease free. After 7 months of excision, there have been no signs of local recurrence. Due to high recurrence rate of STSs, the patient is being followed on OPD bases at regular intervals.
Discussion
Soft tissue sarcomas are a rare group of malignant tumors having a mesenchymal origin and most commonly involves extremities but can also involve trunk, retroperitoneum, head and neck regions [19,20]. Clinically they present as painless mass gradually growing in size [21], those arising from extremities or body wall often present as a palpable swelling or pain on movement [5]. Due to their mostly clinically silent nature, and nonspecific and overlapping imaging features with relatively benign diseases makes the diagnosis difficult and often delays it [5,22]. Cases have been reported in the literature where a delay in presentation and diagnosis have led to significant morbidities and mortality [23,24]. Soft tissue sarcomas are characterized by a relatively aggressive nature with complex genomic profile STS having a high metastatic potential [25]. They exhibit hematogenous invasion with lung as the most common site of secondary metastasis though in rare cases metastasis to oral cavity and brain is also observed [26]. Soft tissue sarcomas have a high local recurrence rate of 5%-80% depending upon the tumor size and locality, type of surgery performed and whether the adjuvant radio-therapy was employed preoperatively or postoperatively [22,[27], [28], [29]].
Myxoid pleomorphic liposarcoma has been recognized as separate entity and categorized as more aggressive type adipocytic sarcomas [2].
Mean age at the diagnosis of myxoid pleomorphic liposarcoma (PML) is 33 years, though it can occur in any age group [30,31], with a female predominance [2]. Our presentation details the case of a 35 years old male.
The diagnosis of soft tissue sarcomas (and myxoid pleomorphic liposarcoma) relies on the histopathology and immunohistochemical staining of the biopsied specimen [17]. Core needle biopsy is often the preferred technique for obtaining the tissue specimen in resource limited countries [17]. Core needle is often preferred over incisional biopsy, because core biopsy is a low cost procedure with low risk of complications [32,33]. But these techniques are limited by the amount and accuracy of sample taken which can pose challenge to providing a valid finding because the tissue sample taken might not be the true representative of the lesion, further the limited quantity of the sample taken can cause the further analyzing of the tumor on histopathology quiet challenging [34]. Complete excision of the lesion is necessary not only for the treatment but also for a more accurate diagnosis of these lesions [35]. Histologically, myxoid pleomorphic liposarcoma shows a mix of both myxoid liposarcoma and pleomorphic liposarcoma types with complex chromosomal aberrations but do not feature any specific immunotypes [36]. In our case the final diagnosis of myxoid pleomorphic liposarcoma was made only after the histopathology of the excised tumor. Previous histopathology on core needle biopsy specimen yielded a diagnosis of spindle cell sarcoma. This difference might be related to the volume of the specimen, depth from where the specimen was taken or the inter-observer variability [34,37].
MRI and CT scans are useful imaging techniques utilized to assess the extent, local invasion, and distant metastasis helping in staging the tumor and guiding the surgical procedure [38]. There is a significant overlap of MRI findings with other tumors, specifically myxomas and myxoid liposarcomas as both can present with homogenous or mildly heterogenous signal characteristics on MRI making them difficult to distinguish on imaging alone [39,40]. Myxoid liposarcomas show well-defined margins which can also be seen in MPL. Further myxoid pleomorphic sarcomas show markedly heterogenous internal structures and moderate to marked enhancement after contrast administration, which can overlap with other high-grade soft tissue tumors particularly liposarcomas [41]. Additionally atypical lipomatous lesions can also mimic pleomorphic lipomas as both can present with prominent myxoid matrix and well defined, encapsulated masses [42]. Hence, differentiating myxoid pleomorphic sarcomas on MRI findings alone can be quite challenging, therefore histological findings particularly excision histology plays a major role in making diagnosis.
Studies suggest complete excision of the tumor lesion as soon as the diagnosis of sarcoma is made [43]. Surgical management is crucial for achieving local control and potential cure, with the goal being complete en bloc excision and confirmation of tumor-free margins [44]. Microscopically negative margins have been associated with a low recurrence rate [45]. Literature suggests ∼1-2 cm-wide resection margin is associated with a reduced local recurrence rate with or without radiotherapy [46,47] while a resection margin of <1 mm is found to have no effect on decreasing local recurrence of the tumor lesion after surgery [48]. Radiotherapy is mostly reserved for large, deep seated high-grade tumors or after marginal resection where further resection is not feasible [49]. Postoperative radiotherapy has been used traditionally for the management soft tissue sarcomas (STS) recurrence, adjuvant radiotherapy, particularly for STS on extremities, significantly increases local control [49]. Preoperative radiotherapy though poses a risk of wound complications may actually help to reduce the risk of later recurrences compared to postoperative radiotherapy [49]. The role of adjuvant chemotherapy in STS has rather conflicting results from studies due to the heterogeneity of the disease; however, meta-analyses of the literature supports its use in large, high-grade, and deep-seated STS [50].
Myxoid pleomorphic liposarcoma is an aggressive type of adipocytic soft tissue sarcomas with early metastatic spread and high recurrence rate with significant morbidity and mortality [2]. They are quite rare and this rarity leads to misdiagnosis or a delay in the diagnosis in a facility with limited resources [51]. Patients with soft tissue sarcomas also usually present late when the tumor has spread locally, or with distant metastasis that leads to significant morbidity or mortality [23,24]. In our case, the patient first presented to a secondary care where he was misdiagnosed with a lipoma on USG, later in tertiary care FNAC yielded a diagnosis of spindle cell carcinoma, for which the excision was performed finally revealing a diagnosis of myxoid pleomorphic liposarcoma. This highlights the difficulties in diagnosing and subsequent treatment opted for such lesions. Had this patient presented later, considering the painless nature of the lesion and the diagnosis of a benign lipoma, the tumor might have had invaded locally or metastasized. Keeping this in regard, it is advised to follow a proper diagnostic protocol and keeping STS in the list of differentials due to its clinically silent nature. People in general should be advised to report any suspicious swelling for clinical analysis and workup.
Conclusion
STS is a heterogenous group of rare tumors. Early detection and surgical intervention play a key role in the improving the prognosis and reducing the associated morbidities. Radiotherapy and chemotherapy can be utilized for high-grade, deep seated or metastatic lesions. Radiotherapy also helps in reducing local recurrences and achieving a better local control. People should be encouraged to report suspicious swellings promptly to health care facilities.
Patient consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Footnotes
Competing Interests: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Siegel R.L., Miller K.D., Fuchs H.E., Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71(1) doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 2.Sbaraglia M., Bellan E., Dei Tos A.P. The 2020 WHO classification of soft tissue tumours: news and perspectives. Pathologica. 2021;113(2):70. doi: 10.32074/1591-951X-213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Grünewald T.G., Alonso M., Avnet S., Banito A., Burdach S., Cidre-Aranaz F., et al. Sarcoma treatment in the era of molecular medicine. EMBO Mol Med. 2020;12(11) doi: 10.15252/emmm.201911131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Potter J.W., Jones K.B., Barrott J.J. Sarcoma–the standard-bearer in cancer discovery. Critic Rev Oncol/Hematol. 2018;126:1–5. doi: 10.1016/j.critrevonc.2018.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Levy A.D., Manning M.A., WB Al-Refaie, Miettinen M.M. Soft-tissue sarcomas of the abdomen and pelvis: radiologic-pathologic features, part 1—Common sarcomas. Radiographics. 2017;37(2) doi: 10.1148/rg.2017160157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Levy A.D., Manning M.A., Miettinen M.M. Soft-tissue sarcomas of the abdomen and pelvis: radiologic-pathologic features, part 2—uncommon sarcomas. Radiographics. 2017;37(3) doi: 10.1148/rg.2017160157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Von Mehren M, RL Randall, Benjamin R.S., Boles S., Bui M.M., Ganjoo K.N., et al. Soft tissue sarcoma, version 2.2018: clinical practice guidelines in oncology. J Nat Comprehens Cancer Network. 2018;16(5):536–563. doi: 10.6004/jnccn.2018.0025. [DOI] [PubMed] [Google Scholar]
- 8.Hui J.Y.C. Epidemiology and etiology of Sarcomas. Surg Clin North Am. 2016;96(5):901–914. doi: 10.1016/j.suc.2016.05.005. [DOI] [PubMed] [Google Scholar]
- 9.Bleloch J.S., Ballim R.D., Kimani S., Parkes J., Panieri E., Willmer T., et al. Managing sarcoma: where have we come from and where are we going? Therapeut Adv Med Oncol. 2017;9(10):637–659. doi: 10.1177/1758834017728927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stiller C.A., Trama A., Serraino D., Rossi S., Navarro C., Chirlaque M.D., et al. Descriptive epidemiology of sarcomas in Europe: report from the RARECARE project. Eur J Cancer. 2013;49(3) doi: 10.1016/j.ejca.2012.09.011. [DOI] [PubMed] [Google Scholar]
- 11.Salas S., Stoeckle E., Collin F., Bui B., Terrier P., Guillou L., et al. Superficial soft tissue sarcomas (S-STS): a study of 367 patients from the French Sarcoma Group (FSG) database. Eur J Cancer. 2009;45(12) doi: 10.1016/j.ejca.2009.03.006. [DOI] [PubMed] [Google Scholar]
- 12.Fletcher C.D.M., Gustafson P., Rydholm A., Willén H., Åkerman M. Clinicopathologic re-evaluation of 100 malignant fibrous histiocytomas: prognostic relevance of subclassification. J Clin Oncol. 2001;19(12) doi: 10.1200/JCO.2001.19.12.3045. [DOI] [PubMed] [Google Scholar]
- 13.Nair S.G., Amadio C.H., Scarlato M., Forman D.A. Frequency of histopathologic types of soft tissue sarcoma (STS) reported from a large community-based hospital over a 20 year period. J Clin Oncol. 2006;24(18_suppl) [Google Scholar]
- 14.Froehner M., Wirth P.M. Etiologic factors in soft tissue sarcomas. Onkologie. 2001;24(2):139–142. doi: 10.1159/000050301. [DOI] [PubMed] [Google Scholar]
- 15.Penel N., Grosjean J., Robin Y.M., Vanseymortier L., Clisant S., Adenis A. Frequency of certain established risk factors in soft tissue sarcomas in adults: a prospective descriptive study of 658 cases. Sarcoma. 2008;2008(1) doi: 10.1155/2008/459386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zahm S.H., Blair A., Holmes F.F., Boysen C.D., Robel R.J. A case-control study of soft-tissue sarcoma. Am J Epidemiol. 1989;130(4):665–674. doi: 10.1093/oxfordjournals.aje.a115388. [DOI] [PubMed] [Google Scholar]
- 17.Cormier J.N., Pollock R.E. Soft tissue sarcomas. CA: a cancer journal for clinicians. 2004;54(2):94–109. doi: 10.3322/canjclin.54.2.94. [DOI] [PubMed] [Google Scholar]
- 18.Ferrari A., Dirksen U., Bielack S. Sarcomas of soft tissue and bone. Prog Tumor Res. 2016;43:128–141. doi: 10.1159/000447083. [DOI] [PubMed] [Google Scholar]
- 19.von Mehren M., Kane J.M., Agulnik M., Bui M.M., Carr-Ascher J., Choy E., et al. Soft tissue Sarcoma, version 2.2022. JNCCN J Natl Compr Cancer Netw. 2022;20(7):815–833. doi: 10.6004/jnccn.2022.0035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Brennan M.F., Casper E.S., Harrison L.B., Shiu M.H., Gaynor J., Hajdu S.I. The role of multimodality therapy in soft-tissue sarcoma. Ann Surg. 1991;214(3):328. doi: 10.1097/00000658-199109000-00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vliet M., Kliffen M., Krestin G.P., Dijke C.F. Soft tissue sarcomas at a glance: clinical, histological, and MR imaging features of malignant extremity soft tissue tumors. Eur Radiol. 2009;19:1499–1511. doi: 10.1007/s00330-008-1292-3. [DOI] [PubMed] [Google Scholar]
- 22.Demetri G.D., Antonia S., Benjamin R.S., Bui M.M., Casper E.S., Conrad E.U., et al. Soft tissue sarcoma. J Natl Compr Cancer Netw. 2010;8(6):630–674. doi: 10.6004/jnccn.2010.0049. [DOI] [PubMed] [Google Scholar]
- 23.Lodhia J., Goodluck G., Tendai J., Urassa E., Nkya G., Mremi A. Case series of high-grade soft tissue sarcoma of the lower limb with delayed diagnosis: experience at a tertiary hospital in northern Tanzania. Int J Surg Case Rep. 2022;97 doi: 10.1016/j.ijscr.2022.107475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Babamiri B., Yavari S., Nikpey S., Faraji N., Goli R., Rahimi K. Undifferentiated soft-tissue sarcoma (STS) in a 34-year-old woman: a case report. Int J Surg Case Rep. 2023;105 doi: 10.1016/j.ijscr.2023.108104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guillou L., Aurias A. Soft tissue sarcomas with complex genomic profiles. Virchows Archiv. 2010;456:201–217. doi: 10.1007/s00428-009-0853-4. [DOI] [PubMed] [Google Scholar]
- 26.Dehal A., Quach L., Garrett E., Jreije K., Hussain F. Soft tissue sarcoma with tongue metastasis: a case report and literature review. J Oral Maxillofac Surg. 2015;73(9):1877–e1. doi: 10.1016/j.joms.2015.04.034. [DOI] [PubMed] [Google Scholar]
- 27.Potter B.K., Adams S.C., Pitcher J.D., Temple H.T. Local recurrence of disease after unplanned excisions of high-grade soft tissue sarcomas. Clin Orthop Relat Res. 2008;466(12):3093–3100. doi: 10.1007/s11999-008-0529-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McMillan R.R., Sima C.S., Moraco N.H., Rusch V.W., Huang J. Recurrence patterns after resection of soft tissue sarcomas of the chest wall. Annal Thorac Surg. 2013;96(4):1223–1228. doi: 10.1016/j.athoracsur.2013.05.015. [DOI] [PubMed] [Google Scholar]
- 29.Schaefer I.M., Hornick J.L., Barysauskas C.M., Raut C.P., Patel S.A., Royce T.J., et al. Histologic appearance after preoperative radiation therapy for soft tissue sarcoma: assessment of the European Organization for Research and Treatment of Cancer–soft tissue and bone sarcoma group response score. Int J Radiat Oncol Biol Physics. 2017;98(2):375–383. doi: 10.1016/j.ijrobp.2017.02.087. [DOI] [PubMed] [Google Scholar]
- 30.Dermawan J.K., Hwang S., Wexler L., Tap W.D., Singer S., Vanderbilt C.M., et al. Myxoid pleomorphic liposarcoma is distinguished from other liposarcomas by widespread loss of heterozygosity and significantly worse overall survival: a genomic and clinicopathologic study. Mod Pathol. 2022;35(11):1644–1655. doi: 10.1038/s41379-022-01107-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Creytens D., Folpe A.L., Koelsche C., Mentzel T., Ferdinande L., van Gorp J.M., et al. Myxoid pleomorphic liposarcoma—a clinicopathologic, immunohistochemical, molecular genetic and epigenetic study of 12 cases, suggesting a possible relationship with conventional pleomorphic liposarcoma. Mod Pathol. 2021;34(11):2043–2049. doi: 10.1038/s41379-021-00862-2. [DOI] [PubMed] [Google Scholar]
- 32.Birgin E., Yang C., Hetjens S., Reissfelder C., Hohenberger P., Rahbari N.N. Core needle biopsy versus incisional biopsy for differentiation of soft-tissue sarcomas: a systematic review and meta-analysis. Cancer. 2020;126(9):1917–1928. doi: 10.1002/cncr.32735. [DOI] [PubMed] [Google Scholar]
- 33.Tuttle R., Kane J.M. Biopsy techniques for soft tissue and bowel sarcomas. J Surg Oncol. 2015;111(5):504–512. doi: 10.1002/jso.23870. [DOI] [PubMed] [Google Scholar]
- 34.Arbiser Z.K., Folpe A.L., Weiss S.W. Consultative (expert) second opinions in soft tissue pathology: analysis of problem-prone diagnostic situations. Am J Clin Pathol. 2001;116(4):473–476. doi: 10.1309/425H-NW4W-XC9A-005H. [DOI] [PubMed] [Google Scholar]
- 35.Gami S., Tiwari S.B., Gautam K., Sharma S., Shrivastav S., Sapkota R. A rare case of myxoid pleomorphic liposarcoma in an infant: a report. Int J Surg Case Rep. 2021;87 doi: 10.1016/j.ijscr.2021.106365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kallen M.E., Hornick J.L. The 2020 WHO classification: what’s new in soft tissue tumor pathology? Am J Surg Pathol. 2021;45(1):e1–23. doi: 10.1097/PAS.0000000000001552. [DOI] [PubMed] [Google Scholar]
- 37.Kang H.J., Kwon S.Y., Kim A., Kim W.G., Kim E.K., Kim A.R., et al. A multicenter study of interobserver variability in pathologic diagnosis of papillary breast lesions on core needle biopsy with WHO classification. J Pathol Transl Med. 2021;55(6):380–387. doi: 10.4132/jptm.2021.07.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.de Juan Ferré A., Álvarez Álvarez R., Casado Herráez A., Cruz Jurado J., Estival González A., Martín-Broto J., et al. SEOM clinical guideline of management of soft-tissue sarcoma (2020) Clin Transl Oncol. 2021;23(5):922–930. doi: 10.1007/s12094-020-02534-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ichikawa J., Kawasaki T., Imada H., Kanno S., Taniguchi N., Ashizawa T., et al. Case report: a typical spindle cell/pleomorphic lipomatous tumor masquerading as a myxoid liposarcoma or intramuscular myxoma. Front Oncol. 2022;12 doi: 10.3389/fonc.2022.1033114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Burt A.M., Huang B.K. Imaging review of lipomatous musculoskeletal lesions. SICOT-J. 2017;3 doi: 10.1051/sicotj/2017015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Arkun R., Memis A., Akalin T., Ustun E.E., Sabah D., Kandiloglu G. Liposarcoma of soft tissue: MRI findings with pathologic correlation. Skeletal Radiol. 1997;26:167–172. doi: 10.1007/s002560050214. [DOI] [PubMed] [Google Scholar]
- 42.Hattori H. Atypical lipomatous tumor of the lip with pleomorphic lipoma-like myxoid area, clinically simulating mucocele. J Oral Pathol Med. 2002;31(9):561–564. doi: 10.1034/j.1600-0714.2002.00151.x. [DOI] [PubMed] [Google Scholar]
- 43.Ramu E.M., Houdek M.T., Isaac C.E., Dickie C.I., Ferguson P.C., Wunder J.S. Management of soft-tissue sarcomas; treatment strategies, staging, and outcomes. SICOT-J. 2017;3 doi: 10.1051/sicotj/2017010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Steele K.H., Raurell A., Ashford R.U. Surgical management of soft tissue sarcoma. Orthop Trauma. 2017;31(3):180–187. [Google Scholar]
- 45.Harati K., Goertz O., Pieper A., Daigeler A., Joneidi-Jafari H., Niggemann H., et al. Soft tissue sarcomas of the extremities: surgical margins can Be close as long as the resected tumor has no ink on it. Oncologist. 2017;22(11):1400–1410. doi: 10.1634/theoncologist.2016-0498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kunisada T., Nakata E., Hasei J., Ozaki T. Resection of high-grade large soft tissue sarcoma with adequate wide margin can lead to good local control without adjuvant radiotherapy. Ann Oncol. 2019;30 [Google Scholar]
- 47.Tarkkanen M., Sampo M., Huuhtanen R., Tukiainen E., Bohling T., Blomqvist C. Impact of the smallest surgical margin on local recurrence in soft tissue sarcoma treated with surgery with or without radiotherapy: first ten year results of a prospective treatment protocol at Helsinki University Central Hospital. J Clin Oncol. 2007;25(18_suppl) [Google Scholar]
- 48.Bilgeri A., Klein A., Lindner L.H., Nachbichler S., Knösel T., Birkenmaier C., et al. The effect of resection margin on local recurrence and survival in high grade soft tissue sarcoma of the extremities: how far is far enough? Cancers (Basel) 2020;12(9):2560. doi: 10.3390/cancers12092560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Larrier N.A., Czito B.G., Kirsch D.G. Radiation therapy for soft tissue sarcoma: indications and controversies for neoadjuvant therapy, adjuvant therapy, intraoperative Radiation therapy, and brachytherapy. Surg Oncol Clin North Am. 2016;25(4):841–860. doi: 10.1016/j.soc.2016.05.012. [DOI] [PubMed] [Google Scholar]
- 50.Loong H.H., Wong K.H., Tse T. Controversies and consensus of neoadjuvant chemotherapy in soft-tissue sarcomas. ESMO Open. 2018;3 doi: 10.1136/esmoopen-2017-000293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kostopoulou O., Delaney B.C., Munro C.W. Diagnostic difficulty and error in primary care: a systematic review. Family Pract. 2008;25(6):400–413. doi: 10.1093/fampra/cmn071. [DOI] [PubMed] [Google Scholar]