Abstract
Background and Aims
In the FLOW trial, semaglutide reduced the risks of kidney and cardiovascular (CV) outcomes and death in participants with type 2 diabetes and chronic kidney disease (CKD). These prespecified analyses assessed the effects of semaglutide on CV outcomes and death by CKD severity.
Methods
Participants were randomized to subcutaneous semaglutide 1 mg or placebo weekly. The main outcome was a composite of CV death, non-fatal myocardial infarction (MI), or non-fatal stroke (CV death/MI/stroke) as well as death due to any cause by baseline CKD severity. CKD was categorized by estimated glomerular filtration rate < or ≥60 mL/min/1.73 m2, urine albumin-to-creatinine ratio < or ≥300 mg/g, or Kidney Disease Improving Global Outcomes (KDIGO) risk classification.
Results
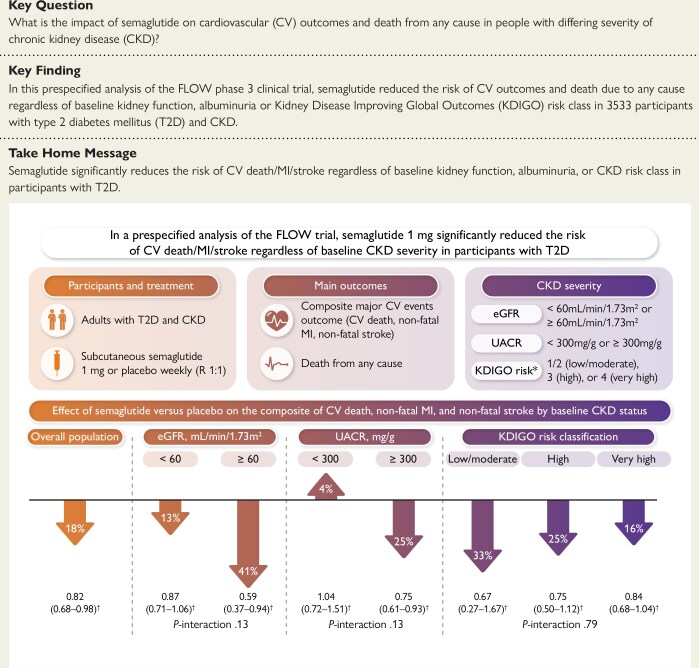
Three thousand, five hundred and thirty-three participants were randomized with a median follow-up of 3.4 years. Low/moderate KDIGO risk was present in 242 (6.8%), while 878 (24.9%) had high and 2412 (68.3%) had very high KDIGO risk. Semaglutide reduced CV death/MI/stroke by 18% [hazard ratio (HR) 0.82 (95% confidence interval 0.68–0.98); P = .03], with consistency across estimated glomerular filtration rate categories, urine albumin-to-creatinine ratio levels, and KDIGO risk classification (all P-interaction > .13). Death due to any cause was reduced by 20% [HR 0.80 (0.67–0.95); P = .01], with consistency across estimated glomerular filtration rate categories and KDIGO risk class (P-interaction .21 and .23, respectively). The P-interaction treatment effect for death due to any cause by urine albumin-to-creatinine ratio was .01 [<300 mg/g HR 1.17 (0.83–1.65); ≥300 mg/g HR 0.70 (0.57–0.85)].
Conclusions
Semaglutide significantly reduced the risk of CV death/MI/stroke regardless of baseline CKD severity in participants with type 2 diabetes.
Keywords: Semaglutide, Cardiovascular outcomes, Type 2 diabetes, Chronic kidney disease
Structured Graphical Abstract
Structured Graphical Abstract.
In a prespecified analysis of the FLOW trial, semaglutide 1.0 mg significantly reduced the risk of CV death/MI/stroke regardless of baseline CKD severity in participants with T2D. *KDIGO-defined risk classification: low, moderate, high, and very high risk based on albuminuria and eGFR. †Data are the HR and 95% CI for the composite major CV event outcome. CI, confidence interval; CKD, chronic kidney disease; CV, cardiovascular; eGFR, estimated glomerular filtration rate; HR, hazard ratio; KDIGO, Kidney Disease Improving Global Outcomes; MI, myocardial infarction; R, randomization; T2D, type 2 diabetes; UACR, urine albumin-to-creatinine ratio.
See the editorial comment for this article ‘Pump, pipes, filter, sugar, weight, and more: the pluripotent prowess of semaglutide’, by S. Verma et al., https://doi.org/10.1093/eurheartj/ehae744.
Introduction
People with type 2 diabetes (T2D) and chronic kidney disease (CKD) are at high risk for cardiovascular (CV) events, including CV death, myocardial infarction (MI), and stroke, as well as death due to any cause.1 The high risk for CV events and mortality is strongly correlated with the severity of CKD, defined by estimated glomerular filtration rate (eGFR), urine albumin-to-creatinine ratio (UACR), or Kidney Disease Improving Global Outcomes (KDIGO) risk classification using both eGFR and UACR.2–5
Renin–angiotensin system inhibitors, sodium–glucose co-transporter 2 inhibitors (SGLT2i), and finerenone provide both kidney protection and reduction of risk of CV events in patients with T2D and high CV risk or CKD.6–12 These therapies are an essential part of a foundation of optimal care as recommended by clinical practice guidelines for this population of people with T2D.13,14 Despite these evidence-based therapies, there is a clear unmet need for additional effective therapies in this patient population as a high residual risk is still present. Treatment with glucagon-like peptide-1 receptor agonists (GLP-1RAs) has reduced CV risk in several CV outcomes trials.15–20 A dedicated kidney outcomes trial, the FLOW (Evaluate Renal Function with Semaglutide Once Weekly) trial, showed that semaglutide reduced the risk of major kidney events by 24%, major CV events by 18%, and death due to any cause by 20%, when compared with placebo, in participants with T2D and CKD.21
In the prespecified analyses of the FLOW trial reported here, the composite of CV death, non-fatal MI or non-fatal stroke (hereafter CV death/MI/stroke), and death due to any cause, as well as safety outcomes, were examined by baseline CKD status to better understand if the treatment effects in people with differing severity of CKD are consistent and support similar therapeutic strategies.
Methods
Study design
The design and primary results of the FLOW trial have been published previously.21,22 The sponsor performed the analyses. The kidney, CV, and death events had been validated by an independent statistical group as part of the validation of the primary analyses previously published.21
Adults with T2D (glycated haemoglobin ≤10%) and CKD defined as eGFR 50–75 mL/min/1.73 m2 (calculated with serum creatinine using the Chronic Kidney Disease Epidemiology Collaboration 2009 formula)23 and UACR >300 to <5000 mg/g or eGFR 25–<50 mL/min/1.73 m2 and UACR >100 to <5000 mg/g while receiving a stable maximal labelled or tolerated dose of renin–angiotensin system inhibitors (angiotensin-converting enzyme inhibitor or angiotensin receptor blocker) were eligible to be enrolled. Full inclusion and exclusion criteria have been published previously.21
Participants were randomly assigned 1:1 to receive subcutaneous semaglutide 1.0 mg or matching placebo once weekly and followed for CV, kidney, and safety outcomes. The trial was stopped early for efficacy based on a recommendation from the independent Data Monitoring Committee.
Study outcomes
The main outcome for this prespecified analysis was a composite of CV death/MI/stroke, which was a secondary confirmatory endpoint included in the testing hierarchy. Other outcomes included death due to any cause and safety events. All major CV events and cause of death were adjudicated by an independent committee according to prespecified endpoint definitions and have been published.21
The efficacy outcomes for these analyses included those in the prespecified hierarchical testing sequence detailed previously.21 All deaths, major adverse CV events, major kidney disease outcomes, and major adverse limb ischaemia events were reviewed by adjudication committees blinded to therapy. The definitions used for the clinical events have been published.21,22 Safety was assessed as the number and nature of serious adverse events (SAEs) or those adverse events (AEs) leading to discontinuation of study drug.
Baseline kidney function
In this analysis, participants were grouped by baseline CKD status according to three methods: (i) eGFR (Chronic Kidney Disease Epidemiology Collaboration 2009) defined as <60 or ≥60 mL/min/1.73 m2; (ii) UACR defined as <300 or ≥300 mg/g; and (iii) KDIGO-defined risk classification: low, moderate, high, and very high risk based on albuminuria and eGFR.2 Enrolment in the study could be based on historical local laboratory data, but baseline data are from a central laboratory analysis only. UACR was calculated as the geometric mean of two first morning urine voids. No imputations were performed for missing data.
Statistical analysis
Efficacy analyses were based on the intention-to-treat principle using in-trial data and included all unique randomized participants. Time-to-event outcomes were analysed using a stratified Cox proportional hazards model with randomized treatment (semaglutide or placebo) as a fixed factor and stratified by SGLT2i use at baseline. P-values were obtained from a score test in the Cox proportional hazards model. For the primary outcome, the hazard ratio (HR), 95% confidence interval (CI), and P-value were adjusted for the group sequential design using the likelihood ratio ordering. Interaction P-values were calculated to evaluate the heterogeneity of treatment effect by CKD status. No adjustment was made for multiple comparisons. A two-sided P-value of 0.05 was considered significant. The absolute risk difference was estimated using a generalized linear regression model with identity link function on the pseudo-observations from the Aalen–Johansen estimate at Week 156. Treatment and SGLT2i use (yes/no) at baseline were included as fixed factors in the regression model. The estimate of the number needed to treat (NNT) is calculated as 1/(cumulative incidence estimate for placebo minus cumulative incidence estimate for semaglutide 1.0 mg). Absolute risk reductions were reported with 95% CI, and corresponding NNTs were reported if the reduction was in favour of semaglutide as indicated by the 95% CI (not including zero). For the comparison of baseline characteristics, P-values were determined using a Kruskal–Wallis test for continuous variables and a χ2 test for categorical variables. Adverse events were summarized by the proportion of participants with an event using data according to randomly assigned treatment and KDIGO risk class.
All statistical analyses were performed with SAS software, version 9.4 TS1M5 (SAS Institute, Cary, NC, USA).
Results
In total, 3533 participants were randomly assigned to semaglutide or placebo with a mean follow-up of 3.4 years. Vital status was known in 98.6% of participants, and adherence to randomized treatment was 89%. Baseline CKD status by baseline eGFR, UACR, and KDIGO risk class was known for 3532 participants (99.97%). At baseline, 2813 participants (79.6%) had eGFR <60 mL/min/1.73 m2 and 719 participants (20.4%) had eGFR ≥60 mL/min/1.73 m2. Additionally, 1113 participants (31.5%) had a baseline UACR <300 mg/g and 2419 participants (68.5%) had a baseline UACR ≥300 mg/g. In terms of KDIGO risk class, 242 participants (6.8%) were low/moderate risk, 878 participants (24.9%) were high risk, and 2412 participants (68.3%) were very high risk.
Table 1 shows the baseline characteristics of the participants enrolled by KDIGO risk class and randomly assigned treatment. Participants with higher KDIGO risk classes tended to be slightly older, were more likely to be male, and had a slightly lower level of high-density lipoprotein cholesterol.
Table 1.
Baseline characteristics by KDIGO risk class
| Parameter | KDIGO low/moderate risk | KDIGO high risk | KDIGO very high risk | |||
|---|---|---|---|---|---|---|
| Semaglutide 1 mg (n = 123) | Placebo (n = 119) | Semaglutide 1 mg (n = 432) | Placebo (n = 446) | Semaglutide 1 mg (n = 1211) | Placebo (n = 1201) | |
| Age, years | 67.0 (62.0–72.0) | 66.0 (60.0–72.0) | 67.0 (60.0–72.0) | 67.0 (62.0–73.0) | 68.0 (62.0–73.0) | 68.0 (61.0–73.0) |
| Male sex | 78 (63.4) | 75 (63.0) | 321 (74.3) | 312 (70.0) | 848 (70.0) | 829 (69.0) |
| Region | ||||||
| North America | 23 (18.7) | 34 (28.6) | 89 (20.6) | 113 (25.3) | 310 (25.6) | 295 (24.6) |
| South America | 5 (4.1) | 8 (6.7) | 36 (8.3) | 38 (8.5) | 80 (6.6) | 85 (7.1) |
| Europe | 28 (22.8) | 23 (19.3) | 134 (31.0) | 134 (30.0) | 310 (25.6) | 334 (27.8) |
| Africa | 8 (6.5) | 3 (2.5) | 17 (3.9) | 8 (1.8) | 26 (2.1) | 39 (3.2) |
| Asia | 26 (21.1) | 28 (23.5) | 106 (24.5) | 89 (20.0) | 346 (28.6) | 317 (26.4) |
| Other | 33 (26.8) | 23 (19.3) | 50 (11.6) | 64 (14.3) | 139 (11.5) | 131 (10.9) |
| Race | ||||||
| Asian | 28 (22.8) | 29 (24.4) | 104 (24.1) | 84 (18.8) | 307 (25.4) | 294 (24.5) |
| Black or African American | 3 (2.4) | 6 (5.0) | 14 (3.2) | 18 (4.0) | 60 (5.0) | 58 (4.8) |
| White | 87 (70.7) | 79 (66.4) | 290 (67.1) | 312 (70.0) | 778 (64.2) | 777 (64.7) |
| Other | 3 (2.4) | 4 (3.4) | 17 (3.9) | 19 (4.3) | 36 (3.0) | 46 (3.8) |
| Not reported | 2 (1.6) | 1 (0.8) | 7 (1.6) | 13 (2.9) | 30 (2.5) | 26 (2.2) |
| Ethnicity | ||||||
| Hispanic/Latino | 14 (11.4) | 21 (17.6) | 80 (18.5) | 80 (17.9) | 179 (14.8) | 182 (15.2 |
| Not Hispanic/Latino | 103 (83.7) | 97 (81.5) | 337 (78.0) | 346 (77.6) | 980 (80.9) | 968 (80.6) |
| Not reported | 6 (4.9) | 1 (0.8) | 15 (3.5) | 20 (4.5) | 52 (4.3) | 51 (4.2) |
| History of CV events | ||||||
| No prior MI or stroke | 91 (74.0) | 90 (75.6) | 329 (76.9) | 340 (76.7) | 924 (77.1) | 913 (77.1) |
| Prior MI or stroke | 32 (26.0) | 29 (24.4) | 99 (23.1) | 103 (23.3) | 274 (22.9) | 271 (22.9) |
| Tobacco use | ||||||
| Current smoker | 13 (10.6) | 13 (10.9) | 61 (14.1) | 61 (13.7) | 149 (12.3) | 132 (11.0) |
| Never smoked | 63 (51.2) | 67 (56.3) | 198 (45.8) | 196 (43.9) | 621 (51.3) | 601 (50.0) |
| Previous smoker | 47 (38.2) | 39 (32.8) | 173 (40.0) | 189 (42.4) | 441 (36.4) | 468 (39.0) |
| BMI, kg/m2 | 31.8 (27.5–35.4) | 30.1 (27.0–35.0) | 31.4 (28.0–35.0) | 31.8 (27.6–36.2) | 31.2 (27.4–35.4) | 31.3 (27.3–35.6) |
| Body weight, kg | 89.0 (77.6–102.0) | 85.8 (73.9–97.1) | 89.8 (76.0–102.3) | 89.1 (75.3–104.0) | 87.0 (75.0–101.0) | 87.2 (74.3–103.0) |
| HbA1c, % | 7.6 (6.9–8.4) | 7.6 (6.9–8.5) | 7.8 (7.0–8.7) | 7.6 (6.9–8.6) | 7.6 (6.9–8.5) | 7.6 (6.8–8.6) |
| eGFR, mL/min/1.73 m2 | 66.0 (61.0–73.0) | 66.0 (61.0–76.0) | 62.0 (52.0–72.0) | 61.0 (50.0–68.0) | 38.0 (32.0–46.0) | 40.0 (33.0–46.0) |
| UACR, mg/g | 137.8 (22.9–240.1) | 137.6 (37.2–215.6) | 442.9 (171.4–1021.7) | 380.2 (171.4–955.1) | 751.9 (337.2–1600.0) | 723.5 (353.1–1530.2) |
| hsCRP, mg/L | 2.6 (1.2–6.9) | 2.5 (1.3–6.8) | 2.3 (1.2–5.6) | 2.4 (1.1–5.3) | 2.5 (1.1–5.6) | 2.6 (1.2–6.3) |
| Total cholesterol, mmol/L | 4.5 (3.8–5.3) | 4.3 (3.7–5.1) | 4.1 (3.5–5.0) | 4.2 (3.6–5.0) | 4.2 (3.6–5.1) | 4.2 (3.5–5.0) |
| LDL-cholesterol, mmol/L | 2.3 (1.7–2.9) | 2.2 (1.6–2.7) | 2.1 (1.5–2.7) | 2.1 (1.6–2.8) | 2.2 (1.6–2.8) | 2.1 (1.6–2.8) |
| HDL-cholesterol, mmol/L | 1.1 (1.0–1.3) | 1.1 (0.9–1.3) | 1.1 (0.9–1.3) | 1.1 (0.9–1.3) | 1.0 (0.9–1.2) | 1.0 (0.9–1.3) |
| Triglycerides, mmol/L | 2.0 (1.5–2.7) | 1.9 (1.2–2.9) | 1.9 (1.4–2.9) | 1.9 (1.4–2.8) | 1.9 (1.4–2.9) | 1.9 (1.4–2.9) |
| SGLT2is | 25 (20.3) | 26 (21.8) | 102 (23.6) | 83 (18.6) | 150 (12.4) | 164 (13.7) |
| CV medications | ||||||
| Beta blockers | 62 (50.4) | 53 (44.5) | 212 (49.1) | 228 (51.1) | 638 (52.7) | 650 (54.1) |
| ACE inhibitors | 55 (44.7) | 50 (42.0) | 171 (39.6) | 159 (35.7) | 398 (32.9) | 406 (33.8) |
| ARBs | 66 (53.7) | 67 (56.3) | 251 (58.1) | 274 (61.4) | 749 (61.8) | 719 (59.9) |
| Calcium channel blockers | 58 (47.2) | 71 (59.7) | 229 (53.0) | 243 (54.5) | 733 (60.5) | 697 (58.0) |
| ARNIs | 1 (0.8) | 4 (3.4) | 1 (0.2) | 4 (0.9) | 9 (0.7) | 5 (0.4) |
| Aldosterone antagonists | 9 (7.3) | 3 (2.5) | 35 (8.1) | 38 (8.5) | 91 (7.5) | 80 (6.7) |
| Lipid-lowering drugs | ||||||
| Statins | 94 (76.4) | 85 (71.4) | 323 (74.8) | 331 (74.2) | 933 (77.0) | 923 (76.9) |
| Ezetimibe | 4 (3.3) | 5 (4.2) | 32 (7.4) | 33 (7.4) | 83 (6.9) | 94 (7.8) |
| PCSK9 inhibitors | 0 | 2 (1.7) | 0 | 1 (0.2) | 2 (0.2) | 4 (0.3) |
| Diuretics | ||||||
| Thiazides | 26 (21.1) | 17 (14.3) | 79 (18.3) | 85 (19.1) | 193 (15.9) | 213 (17.7) |
| Loop diuretics | 15 (12.2) | 15 (12.6) | 73 (16.9) | 84 (18.8) | 340 (28.1) | 363 (30.2) |
| Thiazide-like diuretics | 15 (12.2) | 17 (14.3) | 56 (13.0) | 54 (12.1) | 104 (8.6) | 117 (9.7) |
| Acetylsalicylic acid | 57 (46.3) | 51 (42.9) | 194 (44.9) | 216 (48.4) | 500 (41.3) | 509 (42.4) |
| P2Y12 inhibitors | 12 (9.8) | 11 (9.2) | 48 (11.1) | 42 (9.4) | 136 (11.2) | 144 (12.0) |
| Other | 3 (2.4) | 3 (2.5) | 11 (2.5) | 12 (2.7) | 22 (1.8) | 23 (1.9) |
| DOAC | 7 (5.7) | 12 (10.1) | 28 (6.5) | 30 (6.7) | 76 (6.3) | 75 (6.2) |
| Vitamin K antagonists | 2 (1.6) | 2 (1.7) | 22 (5.1) | 16 (3.6) | 30 (2.5) | 36 (3.0) |
| Atrial fibrillation | 5 (4.1) | 12 (10.1) | 42 (9.7) | 33 (7.4) | 87 (7.2) | 88 (7.3) |
| Heart failure | ||||||
| HFpEF | 10 (8.1) | 15 (12.6) | 39 (9.0) | 37 (8.3) | 118 (9.7) | 106 (8.8) |
| HFrEF | 4 (3.3) | 4 (3.4) | 19 (4.4) | 11 (2.5) | 39 (3.2) | 46 (3.8) |
| No heart failure | 97 (78.9) | 93 (78.2) | 352 (81.5) | 373 (83.6) | 974 (80.4) | 964 (80.3) |
| Unknown | 12 (9.8) | 7 (5.9) | 22 (5.1) | 25 (5.6) | 79 (6.5) | 85 (7.1) |
| LVEF | ||||||
| <40% | 1 (0.8) | 3 (2.5) | 12 (2.8) | 8 (1.8) | 26 (2.1) | 33 (2.7) |
| 40 to <50% | 4 (3.3) | 4 (3.4) | 13 (3.0) | 9 (2.0) | 29 (2.4) | 45 (3.7) |
| ≥50% | 33 (26.8) | 32 (26.9) | 139 (32.2) | 138 (30.9) | 360 (29.7) | 363 (30.2) |
Values are n (%) or median (interquartile range). One subject in the semaglutide group had an unknown KDIGO group at baseline.
ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; ARNI, angiotensin receptor-neprilysin inhibitor; BMI, body mass index; CV, cardiovascular; DOAC, direct oral anticoagulant; eGFR, estimated glomerular filtration rate; HbA1c, glycated haemoglobin; HDL, high-density lipoprotein; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; hsCRP, high-sensitivity C-reactive protein; KDIGO, Kidney Disease Improving Global Outcomes; LDL, low-density lipoprotein; LVEF, left ventricular ejection fraction; MI, myocardial infarction; SGLT2i, sodium–glucose co-transporter 2 inhibitor; UACR, urine albumin-to-creatinine ratio.
The time from randomization to first CV death/MI/stroke event by KDIGO risk class is presented in Figure 1A. Figure 1B shows the time to first CV/MI/stroke event by KDIGO risk class and randomized treatment assignment. Overall, those with a higher KDIGO risk class had higher rates of CV death/MI/stroke, with these events occurring in 19 participants (7.9%) with low/moderate risk, 98 (11.2%) with high risk, and 349 (14.5%) with very high risk over a median (range) follow-up of 3.4 years (0–4.5). Figure 2 shows the effect of semaglutide compared with placebo on the composite of CV death/MI/stroke by baseline CKD status defined by eGFR, UACR, and KDIGO risk class. In the overall population, semaglutide reduced rates of the composite of CV death/MI/stroke compared with placebo [HR 0.82 (95% CI 0.68–0.98)]. Consistent treatment effects were observed across all CKD categories defined by eGFR, UACR, or KDIGO risk class (P-interaction >.05). Supplementary data online, Figure S1, shows CV death/MI/stroke composite and the individual components of the composite by more refined eGFR subgroups defined by eGFR <30, ≥30 to <45, ≥45 to <60, and ≥60 mL/min/1.73 m2. The number of participants was small in these subgroups, and the CIs were broad, but the P-interaction values for treatment by eGFR subgroup were non-significant.
Figure 1.
Time from randomization to first cardiovascular death/myocardial infarction/stroke event by (A) KDIGO risk class and (B) KDIGO risk class and randomized treatment. The cumulative incidence rate is calculated using Aalen–Johansen method with non-cardiovascular death as a competing risk. KDIGO, Kidney Disease Improving Global Outcomes
Figure 2.
Effect of semaglutide 1.0 mg on the composite of CV death, non-fatal MI, and non-fatal stroke by baseline chronic kidney disease status. CI, confidence interval; CV, cardiovascular; eGFR, estimated glomerular filtration rate; KDIGO, Kidney Disease Improving Global Outcomes; MI, myocardial infarction; UACR, urine albumin-to-creatinine ratio
Figure 3 shows the proportions of participants with events, incidence rates, and treatment effects for the individual components of the composite outcome of time to first CV death/MI/stroke. The number of MI and stroke events was lower than the number of CV deaths. Trends towards fewer MI events and more stroke events were observed for semaglutide compared with placebo. Overall, consistent treatment effects with semaglutide were observed for CV death/MI/stroke, with all P-interaction values >.05 except for the non-fatal MI and CKD defined by eGFR (P-interaction .04), for which the benefit observed appeared to be less in participants with eGFR <60 vs. ≥60 mL/min/1.73 m2 [HR 0.94 (95% CI 0.63–1.39) vs. HR 0.28 (95% CI 0.09–0.87), respectively].
Figure 3.
Effect of semaglutide 1.0 mg on (A) CV death, (B) non-fatal MI, and (C) non-fatal stroke by baseline chronic kidney disease. CI, confidence interval; CV, cardiovascular; eGFR, estimated glomerular filtration rate; KDIGO, Kidney Disease Improving Global Outcomes; MI, myocardial infarction; UACR, urine albumin-to-creatinine ratio
The effect of semaglutide on all-cause mortality by baseline eGFR, UACR, and KDIGO risk class is presented in Figure 4. The incidence rates for death were higher in groups with more severe CKD, whether defined by eGFR, UACR, or KDIGO risk class. All-cause mortality was reduced by semaglutide overall [HR 0.80 (95% CI 0.67–0.95)], with consistent results across CKD categories defined by eGFR (P-interaction .21) and KDIGO risk class (P-interaction .23). For UACR category, the randomized treatment effect on all-cause mortality favoured semaglutide in participants with higher UACR [HR 0.70 (95% CI 0.57–0.85) for UACR ≥300 mg/g] compared with a trend towards higher mortality with semaglutide in participants with lower UACR [HR 1.17 (95% CI 0.83–1.65) for UACR <300 mg/g] (P-interaction .01).
Figure 4.
Effect of semaglutide 1.0 mg on death due to any cause by baseline chronic kidney disease. CI, confidence interval; eGFR, estimated glomerular filtration rate; KDIGO, Kidney Disease Improving Global Outcomes; UACR, urine albumin-to-creatinine ratio
Figure 5 shows the treatment effects of semaglutide by KDIGO risk class represented as a heat map. Similar reductions with semaglutide across KDIGO risk class were observed for the composite of CV death/MI/stroke, with HRs ranging from 0.67 to 0.84 and P-value for the interaction not significant (.79).
Figure 5.
KDIGO heat map with composite cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke event rates (semaglutide 1.0 mg vs. placebo). CI, confidence interval; G, grade; GFR, glomerular filtration rate; HR, hazard ratio; KDIGO, Kidney Disease Improving Global Outcomes
Table 2 shows the key safety outcomes by KDIGO risk class for semaglutide and placebo. Participants with higher KDIGO risk class had a higher number of SAEs and fatal events. Although differences were marginal, the proportions of participants with SAEs were numerically lower for the semaglutide than the placebo group within each KDIGO risk category.
Table 2.
Key safety outcomes by KDIGO risk class
| KDIGO low/moderate risk | KDIGO high risk | KDIGO very high risk | ||||
|---|---|---|---|---|---|---|
| Semaglutide 1.0 mg (n = 123) | Placebo (n = 119) | Semaglutide 1.0 mg (n = 432) | Placebo (n = 446) | Semaglutide 1.0 mg (n = 1211) | Placebo (n = 1201) | |
| SAEs | 47 (38.2) | 52 (43.7) | 185 (42.8) | 210 (47.1) | 573 (47.3) | 631 (52.5) |
| AEs leading to permanent discontinuation | 14 (11.4) | 12 (10.1) | 51 (11.8) | 45 (10.1) | 167 (13.8) | 152 (12.7) |
| Fatal events | 9 (7.3) | 7 (5.9) | 43 (10.0) | 51 (11.4) | 121 (10.0) | 155 (12.9) |
| Gallbladder disease | 2 (1.6) | 6 (5.0) | 9 (2.1) | 9 (2.0) | 25 (2.1) | 32 (2.7) |
| Pancreatitis | 1 (0.8) | 2 (1.7) | 4 (0.9) | 0 | 7 (0.6) | 7 (0.6) |
| Retinal disorders | 18 (14.6) | 19 (16.0) | 98 (22.7) | 86 (19.3) | 279 (23.0) | 283 (23.6) |
| Pancreatic cancer | 0 | 2 (1.7) | 2 (0.5) | 0 | 3 (0.2) | 4 (0.3) |
| Abuse or misuse of trial product | 0 | 0 | 1 (0.2) | 1 (0.2) | 0 | 2 (0.2) |
| Acute kidney injury or failure | 4 (3.3) | 4 (3.4) | 21 (4.9) | 26 (5.8) | 128 (10.6) | 145 (12.1) |
| GI disorders | 23 (18.7) | 8 (6.7) | 81 (18.8) | 41 (9.2) | 200 (16.5) | 123 (10.2) |
| COVID-19 | 26 (21.1) | 22 (18.5) | 82 (19.0) | 100 (22.4) | 220 (18.2) | 244 (20.3) |
| Allergic reactions | 3 (2.4) | 1 (0.8) | 7 (1.6) | 8 (1.8) | 23 (1.9) | 29 (2.4) |
| CV disorders | 13 (10.6) | 10 (8.4) | 59 (13.7) | 69 (15.5) | 186 (15.4) | 227 (18.9) |
| Malignant tumours | 14 (11.4) | 6 (5.0) | 25 (5.8) | 21 (4.7) | 64 (5.3) | 62 (5.2) |
| Medication errors | 1 (0.8) | 0 | 6 (1.4) | 3 (0.7) | 11 (0.9) | 8 (0.7) |
| Drug-related hepatic disorders | 2 (1.6) | 1 (0.8) | 11 (2.5) | 4 (0.9) | 16 (1.3) | 18 (1.5) |
Values are n (%).
AE, adverse event; CV, cardiovascular; GI, gastrointestinal; KDIGO, Kidney Disease Improving Global Outcomes; SAE, serious adverse event.
The absolute risk reduction (semaglutide 1.0 mg − placebo) at Week 156 for CV death, MI, or stroke was −0.02 (95% CI −0.04 to −0.002; P = .035), resulting in an NNT of 45 (95% CI 23–623) to prevent one CV death, non-fatal MI, or non-fatal stroke. The absolute risk reduction at Week 156 for death due to any cause was −0.03 (95% CI −0.05 to −0.004; P = .019), resulting in an NNT of 39 (95% CI 21–238) to prevent one death.
Discussion
In the FLOW trial of participants with T2D and CKD, subcutaneous semaglutide 1.0 mg once weekly slowed the decline in kidney function, improved kidney and CV outcomes, and reduced death from any cause compared with placebo.21 The present analyses support a clear and generally consistent benefit on CV outcomes and all-cause mortality across groups with varying severity of CKD at baseline defined by eGFR, UACR, and KDIGO risk class (Structured Graphical Abstract).
Participants with lower eGFR, higher UACR, or higher KDIGO risk class also had higher incidence of CV outcomes and all-cause mortality, a finding that is consistent with prior observations.4,24 The proportion of participants randomized to receive placebo who either died due to CV causes or had a non-fatal MI or non-fatal stroke over a median follow-up of only 3.4 years ranged from 9.2% in the KDIGO low/moderate risk class to 15.5% in the KDIGO very high risk class. The proportion of participants randomized to placebo who died due to any cause was also high and ranged from 7.6% in the KDIGO low/moderate risk class to 17.4% in the KDIGO very high risk class. Notably, compared with other dedicated kidney trials like CREDENCE,9 the FLOW trial population was at much higher risk, with an approximate 50% higher incidence of CV death/MI/stroke and death from any cause, although with slightly longer follow-up (3.4 vs. 2.6 years).
The FLOW trial is the first dedicated kidney outcomes trial in participants with T2D and CKD to evaluate a GLP-1RA. These analyses support the use of semaglutide to reduce CV outcomes and death due to any cause in T2D across a broad range of CKD severity. The results were observed in a population that was receiving a maximum labelled or tolerated dose of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, intensive lipid-lowering therapy (in >80% of participants), and antiplatelet or anticoagulant therapy. In addition, 15.6% of participants were receiving SGLT2i treatment; however, none were receiving finerenone at baseline because FLOW started enrolling in 2019 and finerenone trials were not completed until later. A prior systematic review has shown reductions in CV outcomes with GLP-1RAs in participants with eGFR ≥60 mL/min/1.73 m2 but not in those with eGFR <60 mL/min/1.73 m2.25 Similar to our findings, no heterogeneity in treatment effect with SGLT2i by CKD status defined by eGFR or albuminuria on CV death/MI/stroke was observed in a systematic review of completed SGLT2i trials and similarly with finerenone on CV death/MI/stroke/hospitalized HF.26,27 FLOW now clearly defines benefit in those with lower eGFR and supports use of semaglutide as part of disease management strategies for those with T2D and CKD. As treatment paradigms evolve, further study will be needed.
The analyses of the individual components of the CV composite outcomes showed that there were fewer non-fatal MIs and non-fatal strokes than CV deaths. This is consistent with prior dedicated kidney outcomes trials in T2D and CKD.11,12 A trend towards fewer MIs (52 vs. 64) but more strokes (63 vs. 51) was observed with semaglutide compared with placebo. Prior trials of semaglutide across different indications have not shown any potential increase in strokes,28–30 and systematic reviews of CV outcomes across GLP-1RA trials similarly have not shown a potential increase in strokes.15–20,31 In analyses of treatment by CKD severity with interactions for the composite CV outcome, the components of the composite outcome and all-cause mortality showed general consistency. Two of 15 interactions tested showed a significant P-interaction <.05. The P-interaction for the analysis of death due to any cause by UACR showed the randomized treatment effect on all-cause mortality favoured semaglutide in participants with higher UACR compared with a trend towards higher mortality with semaglutide in participants with lower UACR (P-interaction .01). In addition, the P-interaction for the analysis of non-fatal MI by eGFR showed an apparent larger treatment effect with semaglutide in participants with higher eGFR compared with those with lower eGFR (P-interaction .04). We are not aware of any biologically plausible mechanisms to explain these interactions, and given the number of interactions tested and the fact that we do not correct for multiple interactions, we believe this is likely due to chance.
These analyses have important limitations. The FLOW trial was not designed to have an adequate number of events or statistical power to definitively evaluate treatment effects in subgroups defined by CKD severity using eGFR, and the CIs are wide but no statistically significant interaction was observed. We did not analyse CKD defined by eGFR or UACR as continuous variables because of the small number of events in the extremes of kidney function that limited adequately powered analyses. The MI outcome in subgroups defined by eGFR and death due to any cause defined by UACR had P-interaction values <.05, suggesting greater efficacy in opposite directions by CKD severity measures. There is no clear biological reason for these findings, and given the large number of comparisons performed and opposite directions of effect, they are probably due to chance. Finally, these results are only applicable to patient populations reflective of the FLOW participants as defined by the inclusion CKD parameters applied.
In participants with T2D and CKD, semaglutide reduced the risk of CV death/MI/stroke and across subgroups defined by different levels of eGFR, albuminuria, or KDIGO risk class at baseline. Semaglutide reduced the risk of death from any cause consistently in subgroups defined by eGFR and KDIGO risk. Semaglutide should be considered as part of the therapeutic strategy to reduce CV risk in people living with T2D and CKD.
Supplementary Material
Acknowledgements
We thank all the patients who participated in this trial, the site investigators, and the staff. Editorial assistance in the form of redrawing figures and formatting (but no medical writing support) was provided by Steph Carter, PhD, of Titan, OPEN Health Communications, and funded by Novo Nordisk A/S, in accordance with Good Publication Practice (GPP) guidelines (ismpp.org/gpp-2022). Professor George Bakris contributed to this article as per the ICMJE authorship criteria; however, he sadly passed away during the publication process. We thank him for his invaluable contribution to the FLOW trial and to this publication. This article is dedicated to his memory.
Contributor Information
Kenneth W Mahaffey, Stanford Center for Clinical Research, Department of Medicine, Stanford School of Medicine, 300 Pasteur Drive, Grant S-102, Stanford, Palo Alto, CA 94305, USA.
Katherine R Tuttle, Division of Nephrology, University of Washington School of Medicine, Seattle, WA, USA; Providence Medical Research Center, Providence Inland Northwest Health, Spokane, WA, USA.
Mustafa Arici, Department of Nephrology, Hacettepe University Faculty of Medicine, Ankara, Turkey.
Florian M M Baeres, Novo Nordisk A/S, Søborg, Denmark.
David M Charytan, Department of Medicine, New York University Grossman School of Medicine, New York, NY, USA.
David Z I Cherney, Division of Nephrology, University of Toronto, Toronto, ON, Canada.
Gil Chernin, Kaplan Medical Center, Hebrew University of Jerusalem, Rehovot, Israel.
Ricardo Correa-Rotter, National Institute of Medical Sciences and Nutrition, Salvador Zubirán, Mexico City, Mexico.
Janusz Gumprecht, Department of Clinical and Molecular Medicine, Medical University of Silesia, Katowice, Poland.
Thomas Idorn, Novo Nordisk A/S, Søborg, Denmark.
Giuseppe Pugliese, Department of Clinical and Molecular Medicine, La Sapienza University, Rome, Italy.
Ida Kirstine Bull Rasmussen, Novo Nordisk A/S, Søborg, Denmark.
Søren Rasmussen, Novo Nordisk A/S, Søborg, Denmark.
Peter Rossing, Steno Diabetes Center Copenhagen, Herlev, Denmark; Department of Clinical Medicine, University of Copenhagen, Copenhagen, Denmark.
Ekaterina Sokareva, Novo Nordisk A/S, Søborg, Denmark.
Johannes F E Mann, KfH Kidney Centre, Munich, Germany; Department of Nephrology, Hypertension and Rheumatology, University Hospital, Friedrich-Alexander University, Erlangen, Germany.
Vlado Perkovic, Faculty of Medicine and Health, University of New South Wales, Sydney, NSW, Australia.
Richard Pratley, AdventHealth Translational Research Institute, Orlando, FL, USA.
Supplementary data
Supplementary data are available at European Heart Journal online.
Declarations
Disclosure of Interest
K.W.M. has received grants from AHA, Apple, Inc., Bayer, California Institute for Regenerative Medicine (CIRM), CSL Behring, Eidos, Ferring, Gilead, Google (Verify), Idorsia, Johnson & Johnson, Luitpold, Novartis, PAC-12, Precordior, and Sanifit; has received consulting fees from Applied Therapeutics, Bayer, Bristol Myers Squibb, BridgeBio, CSL Behring, Elsevier, Fosun Pharma, Human, Johnson & Johnson, Moderna, Myokardia, Novartis, Novo Nordisk, Otsuka, PhaseBio, Portola, Quidel, and Theravance; and has equity in Human, Medeloop, Precordior, and Regencor. K.R.T. is supported by NIH research grants R01MD014712, U2CDK114886, UL1TR002319, U54DK083912, U01DK100846, OT2HL161847, UM1AI109568, and OT2OD032581, and CDC project numbers 75D301-21-P-12254 and 75D301-23-C-18264. She has also received investigator-initiated grant support from Travere, Bayer, and the Doris Duke Foundation outside of the submitted work. She reports consultancy fees from Boehringer Ingelheim, Eli Lilly, and Novo Nordisk, and speaker fees from Novo Nordisk. M.A. has received honoraria from Amgen, Astellas, AstraZeneca, Bayer, Boehringer Ingelheim, Menarini, Novo Nordisk, Recordati, and Sanofi. E.S., F.M.M.B., T.I., I.K.B.R., and S.R. are employees and shareholders of Novo Nordisk. G.B. has received honoraria from Bayer, KBP Biosciences, Alnylam, AstraZeneca, Novo Nordisk, and inRegen. D.M.C. reports consulting fees from Eli Lilly/Boehringer Ingelheim, AstraZeneca (Data and Safety Monitoring Board), Allena Pharmaceuticals (Data and Safety Monitoring Board), Gilead, Novo Nordisk, GSK, Medtronic, Merck, CSL Behring, Zogenix, Renalytix, and LG Chem (Data and Safety Monitoring Board); research funding from Medtronic, Gilead, Novo Nordisk, Amgen, and Boehringer Ingelheim/Eli Lilly; patents or royalties from UpToDate.com for authorship/editorials on reviews; and expert witness fees related to proton pump inhibitors. D.Z.I.C. has received honoraria from Boehringer Ingelheim/Eli Lilly, Merck, AstraZeneca, Sanofi, Mitsubishi Tanabe, AbbVie, Janssen, Amgen, Bayer, Prometic, Bristol Myers Squibb, Maze, Gilead, CSL Behring, Otsuka, Novartis, Youngene, Lexicon, Inversago, GSK, and Novo Nordisk, and operational funding for clinical trials from Boehringer Ingelheim/Eli Lilly, Merck, Janssen, Sanofi, AstraZeneca, CSL Behring, and Novo Nordisk. G.C. has received honoraria from Novo Nordisk, Bayer, AstraZeneca, Boehringer Ingelheim, and Sanofi; has participated in a data safety monitoring board for Novo Nordisk, Bayer, AstraZeneca, and Boehringer Ingelheim; and is the President of the Israeli Society of Nephrology and Hypertension. R.C.-R. has received grants from Boehringer Ingelheim, Novo Nordisk, Roche, and Baxter; consulting fees from AstraZeneca, Bayer, Boehringer Ingelheim, MedXL, Chinook, Novo Nordisk, and Alexion; honoraria from AstraZeneca, Boehringer Ingelheim, Amgen, Novo Nordisk, and Bayer; and support for attending meetings from Novo Nordisk. J.G. has received consulting fees from Novo Nordisk, Lilly, Sanofi, Abbott, AstraZeneca, and Boehringer Ingelheim; honoraria from Novo Nordisk, Lilly, Sanofi, Abbott, AstraZeneca, Boehringer Ingelheim, Roche, Bayer, Adamed, and Krka; and support for meetings and/or travel from Novo Nordisk, Lilly, Sanofi, AstraZeneca, and Boehringer Ingelheim. G.P. has received honoraria from Boehringer Ingelheim, Eli Lilly, Mundipharma, and Novo Nordisk; support for attending meetings from Guidotti; has participated in a data safety monitoring board for Abbott, Bayer, Eli Lilly, and Novo Nordisk; and has voluntary membership of the steering committee for World Kidney Day. P.R. has received grants from Bayer, AstraZeneca, and Novo Nordisk; honoraria to the Steno Diabetes Centre Copenhagen from AstraZeneca, Astellas, Boehringer Ingelheim, Gilead, Novo Nordisk, Merck, Mundipharma, Sanofi, and Bayer; and consulting fees from AstraZeneca, Astellas, Boehringer Ingelheim, Gilead, Novo Nordisk, Merck, Mundipharma, Sanofi, and Bayer. J.F.E.M. reports grants from Novo Nordisk, the European Union, and McMaster University, Hamilton, Canada; consulting fees from Novo Nordisk, AstraZeneca, Bayer, and Boehringer Ingelheim; honoraria from Novo Nordisk, AstraZeneca, Bayer, and Novartis; and has participated on a data safety monitoring board or advisory board for AstraZeneca, Bayer, Sanofi, and Boehringer Ingelheim, and has a leadership role in the KDIGO group. V.P. has received consulting fees from AstraZeneca, Bayer, Boehringer Ingelheim, Chinook, GSK, Janssen, Novo Nordisk, Novartis, Otsuka, Gilead, Travere Therapeutics, Mitsubishi Tanabe, Medimmune, and UpToDate, and honoraria from Janssen. R.P. reports speaker fees from Eli Lilly and Novo Nordisk; consulting fees from Bayer AG, Bayer HealthCare Pharmaceuticals, Endogenex, Gasherbrum Bio, Genprex, Getz Pharma, Intas Pharmaceuticals, Eli Lilly, Novo Nordisk, Pfizer, and Sun Pharmaceutical Industries; and grants from Biomea Fusion, Carmot Therapeutics, Dompé, Endogenex, Fractyl, Eli Lilly, Novo Nordisk, and Sanofi.
Data Availability
Data will be shared with bona fide researchers who submit a research proposal approved by the independent review board. Individual participant data will be shared in data sets in a deidentified and anonymized format. Information about data access request proposals can be found at novonordisk-trials.com.
Funding
This study was funded by Novo Nordisk.
Ethical Approval
Approvals from regulatory authorities and institutional review boards were obtained, and each participant provided written informed consent prior to any study-related procedures.
Pre-registered Clinical Trial Number
References
- 1. GBD Chronic Kidney Disease Collaboration . Global, regional, and national burden of chronic kidney disease, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet 2020;395:709–33. 10.1016/S0140-6736(20)30045-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group . KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int 2024;105:S117–314. 10.1016/j.kint.2023.10.018 [DOI] [PubMed] [Google Scholar]
- 3. Gerstein HC, Mann JF, Yi Q, Zinman B, Dinneen SF, Hoogwerf B, et al. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA 2001;286:421–6. 10.1001/jama.286.4.421 [DOI] [PubMed] [Google Scholar]
- 4. Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 2004;351:1296–305. 10.1056/NEJMoa041031 [DOI] [PubMed] [Google Scholar]
- 5. Chronic Kidney Disease Prognosis Consortium; Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, et al. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet 2010;375:2073–81. 10.1016/S0140-6736(10)60674-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001;345:861–9. 10.1056/NEJMoa011161 [DOI] [PubMed] [Google Scholar]
- 7. Lewis EJ, Hunsicker LG, Clarke WR, Berl T, Pohl MA, Lewis JB, et al. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001;345:851–60. 10.1056/NEJMoa011303 [DOI] [PubMed] [Google Scholar]
- 8. Heerspink HJL, Stefánsson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020;383:1436–46. 10.1056/NEJMoa2024816 [DOI] [PubMed] [Google Scholar]
- 9. Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med 2019;380:2295–306. 10.1056/NEJMoa1811744 [DOI] [PubMed] [Google Scholar]
- 10. The EMPA-KIDNEY Collaborative Group; Herrington WG, Staplin N, Wanner C, Green JB, Hauske SJ, et al. Empagliflozin in patients with chronic kidney disease. N Engl J Med 2023;388:117–27. 10.1056/NEJMoa2204233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, et al. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med 2020;383:2219–29. 10.1056/NEJMoa2025845 [DOI] [PubMed] [Google Scholar]
- 12. Pitt B, Filippatos G, Agarwal R, Anker SD, Bakris GL, Rossing P, et al. Cardiovascular events with finerenone in kidney disease and type 2 diabetes. N Engl J Med 2021;385:2252–63. 10.1056/NEJMoa2110956 [DOI] [PubMed] [Google Scholar]
- 13. American Diabetes Association Professional Practice Committee . 9. Pharmacologic approaches to glycemic treatment: standards of care in diabetes-2024. Diabetes Care 2024;47:S158–78. 10.2337/dc24-S009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Naaman SC, Bakris GL. Diabetic nephropathy: update on pillars of therapy slowing progression. Diabetes Care 2023;46:1574–86. 10.2337/dci23-0030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Sattar N, Lee MMY, Kristensen SL, Branch KRH, Del Prato S, Khurmi NS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol 2021;9:653–62. 10.1016/S2213-8587(21)00203-5 [DOI] [PubMed] [Google Scholar]
- 16. de Oliveira Almeida G, Nienkötter TF, Balieiro CCA, Pasqualotto E, Cintra JB, Carvalho HCP, et al. Cardiovascular benefits of GLP-1 receptor agonists in patients living with obesity or overweight: a meta-analysis of randomized controlled trials. Am J Cardiovasc Drugs 2024;24:509–21. 10.1007/s40256-024-00647-3 [DOI] [PubMed] [Google Scholar]
- 17. Adamou A, Barkas F, Milionis H, Ntaios G. Glucagon-like peptide-1 receptor agonists and stroke: a systematic review and meta-analysis of cardiovascular outcome trials. Int J Stroke 2024;19(8):876–87. 10.1177/17474930241253988 [DOI] [PubMed] [Google Scholar]
- 18. Gu X, Jiang S, Yang Y, Li W. Effects of finerenone and glucagon-like peptide 1 receptor agonists on cardiovascular and renal outcomes in type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetol Metab Syndr 2024;16:14. 10.1186/s13098-023-01251-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Rahman A, Alqaisi S, Saith SE, Alzakhari R, Levy R. The impact of glucagon-like peptide-1 receptor agonist on the cardiovascular outcomes in patients with type 2 diabetes mellitus: a meta-analysis and systematic review. Cardiol Res 2023;14:250–60. 10.14740/cr1523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Nreu B, Dicembrini I, Tinti F, Sesti G, Mannucci E, Monami M. Major cardiovascular events, heart failure, and atrial fibrillation in patients treated with glucagon-like peptide-1 receptor agonists: an updated meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis 2020;30:1106–14. 10.1016/j.numecd.2020.03.013 [DOI] [PubMed] [Google Scholar]
- 21. Perkovic V, Tuttle KR, Rossing P, Mahaffey KW, Mann JFE, Bakris G, et al. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med 2024;391:109–21. 10.1056/NEJMoa2403347 [DOI] [PubMed] [Google Scholar]
- 22. Rossing P, Baeres FMM, Bakris G, Bosch-Traberg H, Gislum M, Gough SCL, et al. The rationale, design and baseline data of FLOW, a kidney outcomes trial with once-weekly semaglutide in people with type 2 diabetes and chronic kidney disease. Nephrol Dial Transplant 2023;38:2041–51. 10.1093/ndt/gfad009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009;150:604–12. 10.7326/0003-4819-150-9-200905050-00006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Mann JF, Gerstein HC, Pogue J, Bosch J, Yusuf S. Renal insufficiency as a predictor of cardiovascular outcomes and the impact of ramipril: the HOPE randomized trial. Ann Intern Med 2001;134:629–36. 10.7326/0003-4819-134-8-200104170-00007 [DOI] [PubMed] [Google Scholar]
- 25. Arshad A, Sarween N, Sharif A. Systematic review of cardiovascular outcome trials using new antidiabetic agents in CKD stratified by estimated GFR. Kidney Int Rep 2021;6:2415–24. 10.1016/j.ekir.2021.06.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Patel SM, Kang YM, Im K, Neuen BL, Anker SD, Bhatt DL, et al. Sodium-glucose cotransporter-2 inhibitors and major adverse cardiovascular outcomes: a SMART-C collaborative meta-analysis. Circulation 2024;149:1789–801. 10.1161/CIRCULATIONAHA.124.069568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Sarafidis P, Agarwal R, Pitt B, Wanner C, Filippatos G, Boletis J, et al. Outcomes with finerenone in participants with stage 4 CKD and type 2 diabetes: a FIDELITY subgroup analysis. Clin J Am Soc Nephrol 2023;18:602–12. 10.2215/CJN.0000000000000149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016;375:1834–44. 10.1056/NEJMoa1607141 [DOI] [PubMed] [Google Scholar]
- 29. Husain M, Birkenfeld AL, Donsmark M, Dungan K, Eliaschewitz FG, Franco DR, et al. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2019;381:841–51. 10.1056/NEJMoa1901118 [DOI] [PubMed] [Google Scholar]
- 30. Lincoff AM, Brown-Frandsen K, Colhoun HM, Deanfield J, Emerson SS, Esbjerg S, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med 2023;389:2221–32. 10.1056/NEJMoa2307563 [DOI] [PubMed] [Google Scholar]
- 31. Kelly M, Lewis J, Rao H, Carter J, Portillo I, Beuttler R. Effects of GLP-1 receptor agonists on cardiovascular outcomes in patients with type 2 diabetes and chronic kidney disease: a systematic review and meta-analysis. Pharmacotherapy 2022;42:921–8. 10.1002/phar.2737 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be shared with bona fide researchers who submit a research proposal approved by the independent review board. Individual participant data will be shared in data sets in a deidentified and anonymized format. Information about data access request proposals can be found at novonordisk-trials.com.