Abstract
Bile acid diarrhea (BAD) is a chronic and socially debilitating disease characterized by abdominal pain, diarrhea, urgency, and fecal incontinence. Recently, in a 6-week randomized controlled trial, we showed that the glucagon-like peptide 1 receptor agonist (GLP-1RA) liraglutide is superior to bile acid sequestration (considered standard-of-care) using colesevelam in reducing BAD symptoms. The emergence of new, more potent, and longer-acting GLP-1RAs has spurred an interest in these treatments in BAD management. Here, we review the literature on different GLP-1RAs in BAD treatment and outline their potential mode of actions, highlight knowledge gaps, and outline the need for further clinical evidence generation.
KEYWORDS: bile acid diarrhea, bile acid malabsorption, glucagon-like peptide 1 receptor agonists, liraglutide, semaglutide
INTRODUCTION
Bile acid diarrhea (BAD) is a chronic disease with diarrhea, fecal urgency, and incontinence as well as abdominal pain as primary symptoms. The symptoms are caused by an exaggerated spillover of bile acids from the small intestine to the large intestine due to increased bile acid synthesis in the liver and/or decreased reabsorption of bile acids in the small intestine. BAD is estimated to affect 1%–2% of the general population (1,2); however, it is heavily underdiagnosed (3). Despite little evidence from randomized controlled trials (RCTs) (4) and highly variable therapeutic effects, bile acid sequestrants (which bind bile acids in the gut lumen preventing them from irritating the colonic mucosa and facilitating their excretion in feces) have been used for decades for off-label treatment of BAD; and notwithstanding the limited evidence (5), this remains the first-line pharmacotherapy in the management of BAD (6).
Efforts to develop alternative treatment strategies have been undertaken. One approach is targeting one of the key elements of BAD pathophysiology: the lack of negative feedback inhibition on the bile acid synthesis from the small intestine. On absorption in the small intestine, bile acids activate the nuclear farnesoid X receptor in enterocytes, which stimulates production and release of fibroblast growth factor 19 (FGF19). This hormone travels together with absorbed bile acids through the circulation to the liver, where they inhibit de novo bile acid synthesis. In BAD caused by malabsorption, this negative feedback mechanism is impaired (7,8). Treatment strategies to restore or enhance the negative feedback inhibition on hepatic bile acid synthesis include the farnesoid X receptor agonists obeticholic acid (9), tropifexor (10), and aldafermin (11). Obeticholic acid has shown promising results in improving BAD symptoms (9) but due to safety concerns (12) and lack of efficacy (13) in patients with primary biliary cholangitis, its use has been significantly restricted. Tropifexor and alderfermin are well tolerated (10,11,14), but RCTs showing efficacy on clinically relevant parameters (stool consistency, bowel movement frequency, and urgency) are still lacking.
In a case report, we documented the curious notion that 2 individuals treated with the glucagon-like peptide 1 (GLP-1) receptor agonist (GLP-1RA) liraglutide for obesity and type 2 diabetes, respectively, experienced remarkable improvement of concomitant BAD symptoms (15). This observation led to further exploration of the potential use of liraglutide for BAD treatment, described in detail below. Subsequently, other publications reported beneficial effects of liraglutide in individuals with BAD (16,17). Together, these findings have spurred the interest in delineating the potential role of GLP-1 in BAD pathophysiology, generated interest among patients with BAD and their healthcare providers, and resulted in considerable off-label use of liraglutide for the treatment of BAD.
GLP-1 is a gut-derived peptide hormone released from enteroendocrine epithelial L cells upon meal ingestion. It is best known for its insulinotropic and glucagonostatic effects, controlling blood glucose levels, and its appetite and food intake-reducing effects, regulating body weight (18). Importantly in the context of BAD, GLP-1 receptor activation also reduces upper gastrointestinal motility decelerating small intestinal transit time (18). The short circulating half-life (t1/2) of native GLP-1 (∼1 minute) makes it unfit as a therapeutic agent, and longer-acting GLP-1RAs with half-lives ranging from ∼2.5 hours to more than a week have been developed for the treatment of both type 2 diabetes and obesity (Table 1) (18). The once-daily subcutaneously administered GLP-1RA liraglutide (Victoza) was approved for the treatment of type 2 diabetes (in doses up to 1.8 mg) by the European Medicines Agency in 2009 and by the US Food and Drug Administration in 2010. In 2015 and 2014, respectively, the 2 agencies approved liraglutide in higher doses (up to 3.0 mg) for the treatment of obesity (Saxenda). Liraglutide in a once-daily dose of 1.8 mg was shown to reduce the risk of major adverse cardiovascular events in patients with type 2 diabetes and high cardiovascular disease risk in the Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes (LEADER) trial published in 2016 (19). Currently, more potent and longer-acting GLP-1RAs suitable for once-weekly subcutaneous administration (exenatide, semaglutide, and dulaglutide) or once-daily oral administration (semaglutide) are available. Furthermore, the combination of GLP-1RAs with other receptor agonists, such as the dual GLP-1–glucagon-dependent insulinotropic peptide receptor agonist tirzepatide, is emerging. Consequently, the question arises as to whether these agents are superior to liraglutide in BAD treatment.
Table 1.
Overview of the available GLP-1RAs listed according to their reported half-lives (t1/2) and the current clinical evidence of their effect in treatment of BAD
| Compound and brand name (therapeutic indication) | Route and timing of administration | Half-life (t1/2) and time for maximum plasma concentration (tmax) | Recommended maintenance doses | Clinical evidence in BAD treatment (reviewed in the main text) |
|
Exenatide Byetta (type 2 diabetes) |
Subcutaneous, twice daily |
t1/2: 2.4 hr tmax: 2.1 hr |
5 or 10 µg/twice daily | None |
|
Lixisenatide Lyxumia/Adlyxin (type 2 diabetes) |
Subcutaneous, once daily |
t1/2: 2–4 hr tmax: 1–3.5 hr |
20 µg/d | None |
|
Liraglutide Saxenda (obesity) Victoza (type 2 diabetes) |
Subcutaneous, once daily |
t1/2: ∼13 hr tmax: ∼8 hr |
3.0 mg/d (Saxenda) 1.8 mg/d (Victoza) |
A case report described complete remission of symptoms in 2 cases of cholecystectomy-induced and idiopathic BAD, respectively, treated with liraglutide for other indications (15) A 6-week head-to-head comparison of liraglutide (1.8 mg/d) with colesevelam (1,875 mg/twice daily) showed a ≥25% reduction in the number of daily bowel movements in 77% of patients treated with liraglutide and 50% in patients treated with colesevelam (20) A registry-based sequence symmetry analysis investigated the sequence of prescriptions for BAS and all GLP-1RAs (liraglutide/semaglutide) (17). This analysis found a sequence ratio <1 indicating that the prescription of BAS was more likely to be followed by a prescription for GLP-1RAs than the other way around. The ratios were lower when looking only at liraglutide but not semaglutide |
|
Exenatide Bydureon |
Subcutaneous, once weekly |
t1/2: ∼5 d tmax: 1 hr |
2.0 mg/wk | None |
|
Dulaglutide Trulicity (type 2 diabetes) |
Subcutaneous, once weekly |
t1/2: ∼5 d tmax: 48 hr |
0.75–4.5 mg/wk | None |
|
Semaglutide Wegovy (obesity) Ozempic (type 2 diabetes) Rybelsus (type 2 diabetes) |
Subcutaneous, once weekly (Wegovy and Ozempic) Oral, once daily (Rybelsus) |
t1/2: ∼5 d tmax: 25–40 hr (Wegovy and Ozempic) t1/2: ∼7 d tmax: 1 hr (Rybelsus) |
1.7 or 2.4 mg/wk (Wegovy) 0.5–2.0 mg/wk (Ozempic) 7 or 14 mg/d (Rybelsus) |
A case report (a patient with primary BAD) showed remission of BAD after liraglutide (uptitration from 0.6 mg/d to 1.8 mg/d), but only a temporary response to semaglutide (0.5 mg/wk subcutaneous injection once weekly) with improvement of BAD symptoms only during the first 2 days after injection followed by lack of effect until the next injection (16) |
BAD, bile acid diarrhea; BAS, bile acid sequestrant; GLP-1RA, glucagon-like peptide 1 receptor agonist; t1/2, half-life.
Here, we review the scarce literature on the use of different GLP-1RAs in BAD treatment and discuss the potential of different GLP-1RAs in the context of their pharmacokinetic profiles, which we speculate may be important determinants of their mode of actions and, thus, pharmacodynamics in BAD. Finally, we highlight knowledge gaps in the present understanding of GLP-1RA treatment of BAD and outline the needs for further clinical evidence generation.
CLINICAL EVIDENCE OF THE USE OF GLP-1RAs IN BAD TREATMENT
Two patients experiencing complete remission of BAD symptoms after initiating liraglutide treatment for type 2 diabetes and obesity, respectively (15), inspired us to compare the effect of liraglutide and the current standard-of-care treatment of BAD, the bile acid sequestrant colesevelam, in patients with idiopathic or postcholecystectomy BAD in a 6-week double-dummy, double-blind RCT (21). Half of the patients (50%) treated with colesevelam (N = 26) experienced ≥25% reduction in the number of bowel movements per day, whereas a significantly greater proportion (77%) of patients treated with liraglutide (N = 26) met the primary endpoint (20). Following the publication of the initial case reports and our RCT, we have experienced a massive interest in using liraglutide for the management of BAD from primary care physicians, gastroenterologists, and patients. Interestingly, using the Danish nationwide health registries, we observe a 341% increase in the number of liraglutide (Victoza) prescription among individuals with BAD concomitant with a 44% decline in the general population in the years following the publication of the abovementioned studies (2019–2022) (Figure 1) (description of the methodology is available in the Supplementary Material, http://links.lww.com/CTG/B266).
Figure 1.
Prescriptions of the GLP-1RA liraglutide (Victoza) in a population with BAD (individuals had at least 1 prescription of a bile acid sequestrant within 1 year after a diagnostic SeHCAT test) (left panel) and in the general population without BAD (right panel). Data are derived from the Danish health registries, and methods are described in the supplementary materials (http://links.lww.com/CTG/B266). BAD, bile acid diarrhea; GLP-1RA, glucagon-like peptide 1 receptor agonist; SeHCAT, 75selenium-homotaurocholic acid.
A study using a registry-based sequence symmetry analysis addressed the effect of GLP-1RAs compared with bile acid sequestrants by analyzing the sequence of redeemed prescriptions for relevant medicine in the Danish population (17). Thus, the study identified individuals who had redeemed a prescription for both a bile acid sequestrant and a GLP-1RA within a 2-year window. Among these individuals, the authors analyzed the sequence of prescriptions, either first GLP-1RA, then bile acid sequestrant, or vice versa (17). The rationale underlying the analysis implies that an individual experiencing efficacy and tolerability of a treatment is unlikely to change to another treatment. Hence, if more individuals change from bile acid sequestrant treatment to GLP-1RA treatment than vice versa, the GLP-1RA is deemed the superior treatment. The study showed that significantly more individuals had a prescription for bile acid sequestrant followed by a prescription for GLP-1RA than vice versa (17), proposedly indicating that GLP-1RAs are superior for BAD treatment compared with bile acid sequestrants in a real world setting. When looking at individual GLP-1RAs (liraglutide and semaglutide), similar results were observed for liraglutide, but not for semaglutide (17). A sensitivity analysis excluding statin users showed similar results (17). However, this method does not take into consideration that some individuals might experience only partial symptom relief. As such, an individual might experience a slight reduction in the number of daily bowel movements, which is better than before treatment, but not nearly optimal. Furthermore, the fact that bile acid sequestrants are the standard treatment for BAD might introduce prescription bias toward bile acid sequestrants as the first medication prescribed. In addition, since the first evidence of a beneficial effect of GLP-1RAs in BAD treatment was not available until 2019, we notice that the prescriptions of GLP-1RAs up until this year were made for other indications than BAD in contrast to bile acid sequestrants. Thus, we find that analyzing the order of prescriptions does not adequately and fairly address the effectiveness of the treatments.
With the emergence of longer-acting and more potent GLP-1RAs such as semaglutide, one might assume that these compounds translate into increased efficacy in BAD compared with liraglutide. No head-to-head RCT data exist, but we recently published a case report describing differences in efficacy of liraglutide compared with semaglutide (once-weekly subcutaneous administration) in an individual with primary BAD (16). The patient experienced full remission of BAD symptoms when treated with liraglutide, but only temporary effect of semaglutide, which only improved BAD symptoms during the 2 postinjection days but had no effect during the following 5 days until the next dose (16). Without RCT data on semaglutide for the management of BAD, it is difficult to evaluate its safety and efficacy compared with other BAD therapeutics, but the abovementioned case underscores that newer, longer-acting, and more potent GLP-1RAs not necessarily provide better treatment results in BAD as compared with older, shorter-acting, and less potent GLP-1RAs such as liraglutide. This apparent discrepancy may be rooted in different pharmacokinetics translating into different pharmacodynamics of importance to BAD. Thus, the mode of action of a given GLP-1RA may be highly dependent on its individual pharmacokinetic properties.
PROPOSED MECHANISMS OF ACTION OF GLP-1RAs IN BAD TREATMENT
As outlined above, only 1 RCT (20) and a few case reports (15,16,22) describing the effects of GLP-1RA treatment in BAD exist. With newer, longer-acting, and more potent GLP-1RAs increasingly taking over the scene in the treatment of type 2 diabetes and, as of recently, also obesity, it is obvious to speculate whether these compounds would be preferable in the treatment of BAD. While the less frequent subcutaneous dosing (once-weekly vs liraglutide's once-daily subcutaneous injection) or the daily oral intake of tablets may seem appealing, there is a lack of RCT data regarding the effects of these compounds on BAD symptoms.
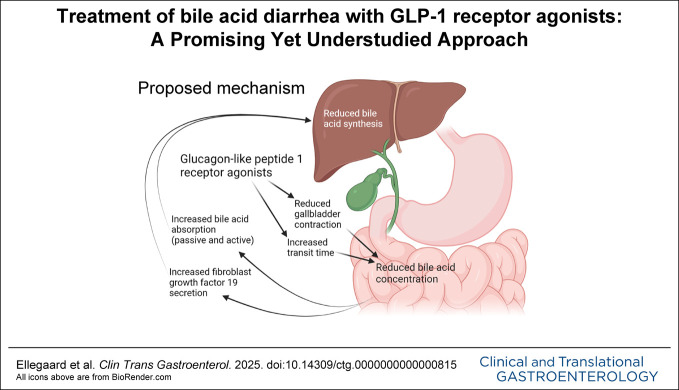
Bile acids are absorbed from the intestinal lumen mainly through active transport by apical sodium-dependent bile acid transporter into enterocytes primarily in the ileum (23). However, unconjugated bile acids are also absorbed through passive diffusion (24). We have previously proposed that the mode of action of liraglutide may involve inhibition of upper gastrointestinal motility resulting in a greater transit time of the luminal content, thus allowing more time for passive and transporter-mediated active absorption of bile acids in the small intestine (20,25) (Figure 2). An increased intestinal absorption of bile acids stimulates the secretion of FGF19 from intestinal enterocytes (26), and increasing circulating levels of both bile acids and FGF19 inhibit hepatic bile acid synthesis, as mentioned above. Furthermore, liraglutide may reduce the amount of bile excreted by the gallbladder or prolonge the time during which bile is excreted (27–29), thus reducing the bile acid concentration in the intestinal lumen. Altogether liraglutide seems to result in improvement of BAD symptoms through mechanisms addressing several aspects of BAD pathophysiology (20). Indeed, we recently found signs that liraglutide increases reabsorption of bile acids (30) and decreases bile acid synthesis (20).
Figure 2.
Illustration of the proposed mode of action of glucagon-like peptide 1 receptor agonists on bile acid diarrhea pathophysiology. See main text for details. Illustration created with BioRender.com.
Gastrointestinal motility is differently affected by different GLP-1RAs due to differences in their pharmacokinetic profiles (16). Short-acting GLP-1RAs (exenatide [t1/2 of 2.4 hours] and lixisenatide [t1/2 of 2–4 hours] administered before meals twice-daily and once-daily, respectively) exhibit intermittent GLP-1 receptor activation translating into potent deceleration of upper gastrointestinal motility, an effect which is sustained during prolonged treatment. However, owing to their short half-lives, the effect plays out only during the meal that they are taken with. By contrast, the long-acting GLP-1RAs (semaglutide [t1/2 of ∼5 days] administered subcutaneously once-weekly or as a once-daily oral formulation and dulaglutide [t1/2 of ∼7 days] administered subcutaneously once-weekly) causing continuous exposure and activation of the GLP-1 receptor resulting in short-lasting effects on upper gastrointestinal motility (31) superseded by subsequent development of tachyphylaxis (16). This may explain the alternating pattern in effect of semaglutide on BAD symptoms described in our recent case report (16). Liraglutide has a t1/2 of ∼13 hours and is administered once daily independently of meals. Thus, liraglutide can be considered an intermediate-acting GLP-1RA providing continuous exposure of the GLP-1 receptor throughout the day, but with clear daily peak and trough plasma concentrations apparently preventing tachyphylaxis on small intestinal motility (32) (Figure 3). Along these lines, daily oral intake of semaglutide presumably results in an overall similar exposure as the subcutaneously administered semaglutide, but with daily pharmacokinetic peaks and troughs. Thus, we speculate that daily oral semaglutide may not induce the same degree of tachyphylaxis as the weekly subcutaneous semaglutide. Overall, theoretically, differences in the pharmacokinetic profiles of different GLP-1RAs may have important implications for their efficacy in BAD and perhaps help explain why liraglutide could be superior in reducing symptoms of BAD as compared with the more potent and longer-acting semaglutide. Nevertheless, relevant RCT data from head-to-head comparisons are needed to test this hypothesis.
Figure 3.
Schematic illustration of the theoretical plasma concentrations of once-daily subcutaneously administered liraglutide (green) and once-weekly subcutaneously administered semaglutide (blue) (arbitrary relationship between the 2 curves). Below the graph, colored boxes indicate the theoretical BAD symptom relief of treatment with the 2 GLP-1RAs based on a case report (16). Dark color indicates strongest symptom relief. Illustration modified from Kårhus et al (16). BAD, bile acid diarrhea; GLP-1RA, glucagon-like peptide 1 receptor agonist.
WHAT IS NEEDED TO PROVIDE SUBSTANTIAL CLINICAL EVIDENCE FOR GLP-1RA THERAPY AS A VIABLE TREATMENT OF BAD?
Currently, the clinical evidence on any GLP-1RA in BAD management is scarce. Thus, as with all types of medicine, off-label use of GLP-1RAs for BAD treatment needs careful consideration of the risks and benefits. Common side effects of GLP-1RA treatment in other patient populations include transient nausea, vomiting, constipation, diarrhea, and abdominal discomfort (33)—largely overlapping with the most common side effects of bile acid sequestrants (5). Rarely but importantly, gallbladder and biliary diseases have been reported in clinical trials with GLP-1RAs (34); however, the extent of these issues in a BAD population is unknown.
The findings with liraglutide treatment reviewed here need verification in larger populations in dedicated placebo-controlled RCTs. Importantly, a consensus on the most clinically relevant primary endpoint needs to be reached to compare the results of different studies. In this regard, we recommend using a composite endpoint of both the number of daily bowel movements and the number of stools of type 6 or 7 on the Bristol stool form scale.
To address the mechanism of action of different GLP-1RAs on BAD pathophysiology, dedicated studies to investigate the proposed mechanisms need to be undertaken. Knowledge on the exact mechanism will provide us with an opportunity for specific targeting of the relevant mechanisms involved.
CONCLUSION
The evidence supporting the use of GLP-1RAs in the treatment of BAD is limited. The strongest evidence comes from a 6-week RCT demonstrating superior efficacy of liraglutide in reducing BAD symptoms as compared with colesevelam currently considered standard-of-care treatment. Sporadic case reports and stories on the internet about the effects of other GLP-1-based drugs are emerging. Ultimately, large, comparative RCTs including all relevant GLP-1RAs and other relevant BAD treatments in parallel treatment designs are warranted.
CONFLICTS OF INTEREST
Guarantor of the article: Anne-Marie Ellegaard, MSc, PhD and Filip K. Knop, MD, PhD.
Specific author contributions: A.M.E. and F.K.K. conceptualized the work; M.W.-J. retrieved and verified the data and performed the data analysis; all authors contributed to data interpretation; A.M.E. wrote the first draft; all authors revised and edited the manuscript; all authors approved the final version of the manuscript.
Financial support: None to report.
Potential competing interests: A.M.E.: None. M.L.K.: None. M.W.-J.: None. A.B.L. have received research funding and speaker honoraria from Novo Nordisk. F.K.K. has been on the advisory panel of, a consultant for, in the speaker's bureau of, owns shares in, and/or has received research support from 89bio, AstraZeneca, Boehringer Ingelheim, Cytoki Pharma, Eli Lilly, Gubra, Novo Nordisk, Merck Sharp & Dohme, Sanofi, Structure Therapeutics, Zealand Pharma, and Zucara; and is co-founder of and a minority shareholder in Antag Therapeutics.
Supplementary Material
Footnotes
SUPPLEMENTARY MATERIAL accompanies this paper at http://links.lww.com/CTG/B266
Currently employed at Novo Nordisk A/S; the present work is independent from Novo Nordisk A/S.
Contributor Information
Martin L. Kårhus, Email: martin.lund.kaarhus@regionh.dk.
Matilde Winther-Jensen, Email: matilde.winther-jensen.01@regionh.dk.
Asger B. Lund, Email: asger.lund.01@regionh.dk.
Filip K. Knop, Email: filip.krag.knop@regionh.dk.
REFERENCES
- 1.Walters JR. Defining primary bile acid diarrhea: Making the diagnosis and recognizing the disorder. Expert Rev Gastroenterol Hepatol 2010;4(5):561–7. [DOI] [PubMed] [Google Scholar]
- 2.Wedlake L, A'Hern R, Russell D, et al. Systematic review: The prevalence of idiopathic bile acid malabsorption as diagnosed by SeHCAT scanning in patients with diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther 2009;30(7):707–17. [DOI] [PubMed] [Google Scholar]
- 3.Kårhus ML, Ellegaard AM, Winther-Jensen M, et al. The epidemiology of bile acid diarrhea in Denmark. Clin Epidemiol 2023;15:1173–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Merza N, Saab O, Nawras Y, et al. Evaluation of alternative treatment strategies for bile acid malabsorption in inflammatory bowel disease patients: A network meta-analysis. J Clin Med Res 2024;16(2-3):33–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borup C, Vinter-Jensen L, Jørgensen SPG, et al. Efficacy and safety of colesevelam for the treatment of bile acid diarrhoea: A double-blind, randomised, placebo-controlled, phase 4 clinical trial. Lancet Gastroenterol Hepatol 2023;8(4):321–331. [DOI] [PubMed] [Google Scholar]
- 6.Camilleri M, BouSaba J. New developments in bile acid diarrhea. Gastroenterol Hepatol 2023;19(9):520–37. [PMC free article] [PubMed] [Google Scholar]
- 7.Johnston IM, Nolan JD, Pattni SS, et al. Characterizing factors associated with differences in FGF19 blood levels and synthesis in patients with primary bile acid diarrhea. Am J Gastroenterol 2016;111(3):423–32. [DOI] [PubMed] [Google Scholar]
- 8.Walters JRF, Tasleem AM, Omer OS, et al. A new mechanism for bile acid diarrhea: Defective feedback inhibition of bile acid biosynthesis. Clin Gastroenterol Hepatol 2009;7(11):1189–94. [DOI] [PubMed] [Google Scholar]
- 9.Walters JRF, Johnston IM, Nolan JD, et al. The response of patients with bile acid diarrhoea to the farnesoid X receptor agonist obeticholic acid. Aliment Pharmacol Ther 2015;41(1):54–64. [DOI] [PubMed] [Google Scholar]
- 10.Camilleri M, Nord SL, Burton D, et al. Randomised clinical trial: Significant biochemical and colonic transit effects of the farnesoid X receptor agonist tropifexor in patients with primary bile acid diarrhoea. Aliment Pharmacol Ther 2020;52(5):808–20. [DOI] [PubMed] [Google Scholar]
- 11.BouSaba J, Torres M, Dilmaghani S, et al. Effects of FGF19 analogue aldafermin in patients with bile acid diarrhea: A randomized, placebo-control trial. Gastroenterology 2023;165(2):499–501.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Food and Drug Administration. Ocaliva (Obeticholic Acid) by Intercept Pharmaceuticals: Drug Safety Communication - Serious Liver Injury Being Observed in Patients without Cirrhosis. FDA; (https://www.fda.gov/safety/medical-product-safety-information/ocaliva-obeticholic-acid-intercept-pharmaceuticals-drug-safety-communication-serious-liver-injury) (2024). Accessed December 18, 2024. [Google Scholar]
- 13.EMA. EMA Recommends Revoking Conditional Marketing Authorisation for Ocaliva. European Medicines Agency; (https://www.ema.europa.eu/en/news/ema-recommends-revoking-conditional-marketing-authorisation-ocaliva) (2024). Accessed November 14, 2024. [Google Scholar]
- 14.Badman MK, Chen J, Desai S, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of the novel non-bile acid FXR agonist tropifexor (LJN452) in healthy volunteers. Clin Pharmacol Drug Dev 2020;9(3):395–410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kårhus ML, Brønden A, Røder ME, et al. Remission of bile acid malabsorption symptoms following treatment with the glucagon-like peptide 1 receptor agonist liraglutide. Gastroenterology 2019;157(2):569–71. [DOI] [PubMed] [Google Scholar]
- 16.Kårhus ML, Knudsen E, Knop FK. Different effects of once-weekly and once-daily administered GLP-1RA semaglutide and liraglutide on bile acid diarrhea. JCEM Case Rep 2023;1(1):luac004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rahbek MT, Lund LC, Hallas J. A case for screening real-world data for collateral drug benefits: Glucagon-like peptide 1 receptor agonists and bile acid diarrhea. Pharmacoepidemiol Drug Saf 2024;33(1):e5673. [DOI] [PubMed] [Google Scholar]
- 18.Andersen A, Lund A, Knop FK, et al. Glucagon-like peptide 1 in health and disease. Nat Rev Endocrinol 2018;14(7):390–403. [DOI] [PubMed] [Google Scholar]
- 19.Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016;375(4):311–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kårhus ML, Brønden A, Forman JL, et al. Safety and efficacy of liraglutide versus colesevelam for the treatment of bile acid diarrhoea: A randomised, double-blind, active-comparator, non-inferiority clinical trial. Lancet Gastroenterol Hepatol 2022;7(10):922–31. [DOI] [PubMed] [Google Scholar]
- 21.Kårhus ML, Brønden A, Lyng Forman J, et al. Protocol for a randomised, double-blinded, placebo-controlled, double-dummy 6-week clinical trial comparing the treatment effects of the glucagon-like peptide 1 receptor agonist liraglutide versus the bile acid sequestrant colesevelam on bile acid malabsorption. BMJ Open 2021;11(2):e044711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Conley TE, White KL, Bond A, et al. Emerging uses of glucagon-like peptide 1 (GLP-1) receptor agonists following ileal resection: Literature review and case examples. Frontline Gastroenterol 2023;14(6):521–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wong MH, Oelkers P, Dawson PA. Identification of a mutation in the ileal sodium-dependent bile acid transporter gene that abolishes transport activity (*). J Biol Chem 1995;270(45):27228–34. [DOI] [PubMed] [Google Scholar]
- 24.Mekhjian HS, Phillips SF, Hofmann AF. Colonic absorption of unconjugated bile acids: Perfusion studies in man. Dig Dis Sci 1979;24(7):545–50. [DOI] [PubMed] [Google Scholar]
- 25.van Can J, Sloth B, Jensen CB, et al. Effects of the once-daily GLP-1 analog liraglutide on gastric emptying, glycemic parameters, appetite and energy metabolism in obese, non-diabetic adults. Int J Obes (Lond) 2014;38(6):784–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Martinot E, Sèdes L, Baptissart M, et al. Bile acids and their receptors. Mol Aspects Med 2017;56:2–9. [DOI] [PubMed] [Google Scholar]
- 27.Rehfeld JF, Knop FK, Asmar A, et al. Cholecystokinin secretion is suppressed by glucagon-like peptide-1: Clue to the mechanism of the adverse gallbladder events of GLP-1-derived drugs. Scand J Gastroenterol 2018;53(12):1429–32. [DOI] [PubMed] [Google Scholar]
- 28.Nexøe-Larsen CC, Sørensen PH, Hausner H, et al. Effects of liraglutide on gallbladder emptying: A randomized, placebo-controlled trial in adults with overweight or obesity. Diabetes Obes Metab 2018;20(11):2557–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gether IM, Nexøe-Larsen C, Knop FK. New avenues in the regulation of gallbladder motility-implications for the use of glucagon-like peptide-derived drugs. J Clin Endocrinol Metab 2019;104(7):2463–72. [DOI] [PubMed] [Google Scholar]
- 30.Ellegaard AM, Kårhus ML, Krych L, et al. Liraglutide and colesevelam change serum and fecal bile acid levels in a randomized trial with patients with bile acid diarrhea. Clin Transl Gastroenterol 2024;15(11):e00772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hjerpsted JB, Flint A, Brooks A, et al. Semaglutide improves postprandial glucose and lipid metabolism, and delays first-hour gastric emptying in subjects with obesity. Diabetes Obes Metab 2018;20(3):610–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nakatani Y, Maeda M, Matsumura M, et al. Effect of GLP-1 receptor agonist on gastrointestinal tract motility and residue rates as evaluated by capsule endoscopy. Diabetes Metab 2017;43(5):430–7. [DOI] [PubMed] [Google Scholar]
- 33.Drucker DJ. Efficacy and safety of GLP-1 medicines for type 2 diabetes and obesity. Diabetes Care 2024;47(11):1873–88. [DOI] [PubMed] [Google Scholar]
- 34.He L, Wang J, Ping F, et al. Association of glucagon-like peptide-1 receptor agonist use with risk of gallbladder and biliary diseases: A systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med 2022;182(5):513–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.