Abstract
Introduction
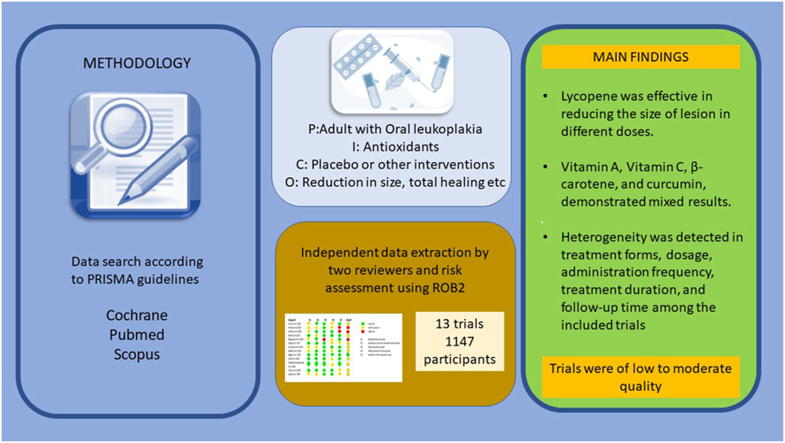
Oral leukoplakia (OL) is a potentially malignant disorder characterized by non-scrabble white lesions that may turn into invasive carcinoma if left untreated. Few antioxidant treatments have shown promising results in the regression of lesions and prevention of their progression to carcinoma. We aim to summarize the current evidence on the effectiveness of antioxidants in the management of OL.
Materials and methods
The present systematic review followed PRISMA guidelines and involved a thorough search of three scientific databases: PubMed, Scopus, and Cochrane. We used specific algorithms related to the keywords “antioxidant” and “Oral leukoplakia” to identify randomized controlled trials (RCTs) that have explored the utility of antioxidants in the management of OL. The risk of bias assessment was conducted using the Cochrane risk of bias tool (ROB 2.0), ensuring the reliability of our findings.
Results
Out of 2490 articles retrieved, only thirteen RCTs involving 1147 participants fulfilled the inclusion criteria. Lycopene was found to be the most consistently effective antioxidant, showing significant effectiveness in reducing lesion size, both in oral and topical forms. The trials incorporating other antioxidants, including vitamin A, vitamin C, β-carotene, and curcumin, demonstrated mixed results. Severe heterogeneity was detected in treatment forms, dosage, administration frequency, treatment duration, and follow-up time among all included trials.
Conclusion
Lycopene may play a significant role in the non-surgical management of OL. Future research with large-scale, well-designed, randomized controlled trials with standardized dosing, delivery methods, and outcome measures is mandatory to provide more robust evidence on the available antioxidant therapy for OL.
Keywords: Antioxidant treatment, OPMD, Oral, Leukoplakia, Premalignant oral lesion
Graphical abstract
1. Introduction
Oral leukoplakia (OL) is a widely recognized oral potentially malignant disorder (OPMD) that affects people of all age groups, genders, and ethnic backgrounds on a global scale.1 Epidemiological studies have shown a higher incidence in males, especially older adults, and individuals with a history of tobacco use, alcohol consumption, and betel quid chewing.2,3 Smoking has been identified as the primary risk factor, with smokers being six times more likely to develop OL.1 Alcohol is acknowledged as a distinct risk factor for OL.4 The consumption of betel quid is a major cause in Southeast Asia, and it is accountable for the higher occurrence of OL in this area.5,6
OL is pathologically characterized by epithelial hyperplasia, keratinization, and dysplastic changes, which mean continuity of morphological alterations that might lead to invasive carcinoma if left untreated.7 Genetic and epigenetic changes, such as mutations in tumor suppressor genes, activation of oncogenes, and disruption of cell cycle control mechanisms, have been identified through molecular studies.8
Presently, numerous therapeutic alternatives exist for OL, encompassing surgical intervention, laser ablation, photodynamic therapy, cryotherapy, and topical therapy, and none are considered entirely curative.9,10 The overall recurrence rate after complete surgical excision of OL lesions was 25 % with an average follow-up time of 76.8 months in a meta-analysis (n = 204) of 5 observational studies on OL surgical therapy.10 The recurrence rate after photodynamic therapy for OL was 25 %, according to a meta-analysis (n = 182) of 5 trials.10 In a meta-analysis (n = 4292) of 27 observational studies on laser management for OL, 24 % of OL lesions recurred after complete remission.11 The total recurrence rate after cryotherapy for OL was 16 %. Even though precision medicine that uses “omics” sciences (genomics, transcriptomics, epigenomics, proteomics, and metabolomics) to integrate data and create highly predictive models of the biological system has been explored for several diseases, for OL, it is still in its infancy. A recent non-randomized controlled trial last year reported the use of immune checkpoint therapy with Nivolumab (programmed cell death 1 protein inhibitor), which showed lesion reduction by size and severity of dysplasia after treatment.12
There has been a continuous effort to develop novel treatment interventions that specifically target the fundamental molecular pathways responsible for the growth of OL and mitigate the likelihood of malignant transformation. In recent years, there has been a growing interest in investigating the use of antioxidants as adjunctive medicines for the treatment and prevention of OL. Oxidative stress is characterized by an uneven generation of reactive oxygen species (ROS) and the protective mechanisms of antioxidants.13 It plays a significant role in the development and advancement of OL.14 The oral cavity, which is regularly exposed to external cancer-causing agents and harmful microorganisms, experiences oxidative harm to its cellular components, including DNA, proteins, and lipids.14,15 The DNA damage caused by reactive oxygen species (ROS) can lead to permanent damage.14,15 Antioxidants, natural or synthetic substances, exert cytoprotective effects by scavenging free radicals, counteracting ROS, and regulating redox signaling pathways important in cellular growth, survival, and inflammation.16,17 Multiple epidemiological studies have demonstrated evidence of a negative correlation between the consumption of antioxidants in one's diet and the chance of developing oral cancer. Individuals who are most vulnerable, namely smokers, are also the ones who have the lowest intake of fruits and vegetables.18,19 Consequently, a hypothesis emerged suggesting that incorporating antioxidants could potentially contribute to both primary prevention and secondary prevention for persons at risk.18 This also implied that diets rich in antioxidants and antioxidant therapy may protect against the development and progression of oral OL. In the early 90s, few studies started to explore the utility of antioxidants in OL.20 Ever since several studies have explored the utility of different established antioxidants in the management of leukoplakia. Even though significant advancement has happened in the field of management of leukoplakia, evidence regarding therapeutic potential of antioxidants in the management of this potentially malignant disorder is still inconclusive. This systematic review aims to assess the existing information on the effectiveness of antioxidant supplementation in the management of OL. This knowledge can aid in creating precise therapy strategies that enhance efficacy and ensure patient safety.
2. Material and methods
A systematic review was conducted to evaluate the efficacy the available antioxidants in the management of OL. The study followed the guidelines outlined in the Cochrane Handbook for Systematic Reviews of Interventions, and the findings are reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).21 The protocol was documented and registered.
2.1. Search strategy
We searched three prominent academic databases, including PubMed, Scopus, and the Cochrane Database, to identify literature related to the use of antioxidants for oral leukoplakia. Boolean operators (AND, OR) also were used to make the search effective. A detailed search strategy is provided in Supplementary Tables 1–3.
We aimed to address the following research questions: Are antioxidants effective in the management of OL? Which of the available antioxidants is the most efficacious in the management of OL? Accordingly, the PICO was formulated as follows:
P- Patients with OL
I- Any antioxidants
C- Placebo or any other treatment
O- Complete cure/partial cure/recurrence
Records were identified by utilizing customized search terms for each search engine and downloaded into a citation manager (Zotero, version 6.0.15). Duplicates were then automatically removed. Two separate reviewers (DG and KF) conducted the screening process, which consisted of two distinct steps. The screening process involved initially reviewing titles and abstracts, followed by a thorough examination of full texts. Any record that satisfied the eligibility requirements was subsequently included in the study. Any difference of opinion between the reviewers was resolved by discussion with the third reviewer (SV). The third reviewer was responsible for scanning the grey literature database and conducting the citation search.
2.2. Inclusion and exclusion criteria
2.2.1. The inclusion criteria
Randomized control trials using antioxidants for the management of oral leukoplakia published between January 2000 and June 2024.
2.2.2. The exclusion criteria
Cross-sectional studies.
Single arm studies.
Animal experiments.
Invitro studies.
Literature reviews and systematic reviews.
Letter to Editor.
Conference abstracts.
2.3. Data extraction and synthesis
Two reviewers (DG and KF) separately extracted the data into a data collection form. The obtained data was inputted into the data gathering form in Excel. The form was designed per the Cochrane Handbook for the systematic review of intervention guidelines. The data obtained from randomized controlled trials (RCTs) were categorized into the following sections: demographic characteristics, intervention characteristics, outcome measures, primary results, and follow-up data.
2.4. Risk of bias
Two reviewers independently assessed the risk of bias using the Cochrane risk of bias tool (ROB 2.0).22
3. Results
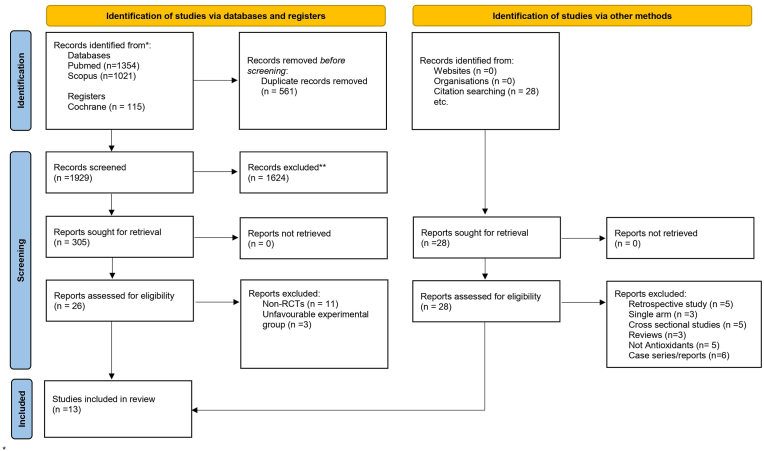
Initial searches of the three databases yielded a total of 2490 articles (Pubmed = 1354, Scopus = 1021, and Cochrane n = 115). After eliminating 561 duplicate entries, a total of 1929 articles were obtained. After a thorough review of the titles and abstracts, 1624 articles were deemed irrelevant and eliminated. A full-text reading was conducted to analyze 26 articles selected from 305 retrieved articles, and finally, 12 RCTs met the inclusion criteria. An additional 28 articles were identified through citation searching and thoroughly reviewed, and one RCT was selected for inclusion. Ultimately, thirteen randomized controlled trials that aligned with our specific outcome criteria were incorporated in our review. The detailed selection of studies is shown in Fig. 1.
Fig. 1.
Prisma flow chart of the review process.
3.1. General characteristics of the studies
The characteristics of the studies are presented in Table 1. Thirteen included trials accounted for a total of 1147 participants. Out of the thirteen trials, one study each was from Italy,23 USA,23 and Japan,24; the rest was conducted in India. Eleven trials utilized a two-arm parallel design, and three trials utilized a three-arm parallel design. The duration of the trials varied. Three investigations employed an open follow-up approach,24, 25, 26 One study had a duration of one year,24 whereas the remaining studies had a duration of less than one year. Most of the studies compared two interventions or interventions with a placebo. Three trials evaluated the effect of the same interventions with varied dosages in different arms, either alone or in combination with others.24,26,27 The primary outcome of all the trials was to evaluate the response toward treatment except for one study, which assessed the recurrence and appearance of new lesions or carcinoma after treatment.23 Nevertheless, five studies additionally followed up on the patients and evaluated recurrence as well as progression of the lesion to higher grades of dysplasia or invasive cancer.24, 25, 26,28,29
Table-1.
Characteristics of the included trials in detail.
| No. | Author, year | Age(years) | Sex(M/F) | Total number of participants) | Treatment and pharmaceutical form | Frequency of administration (a day) | Duration of treatment | Follow-up(weeks) | Outcome measurement | Results | Long term follow up and recurrence | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Ansari et al., 2023 | No data available | No data available | 65(72) | A: 33(36) | A: Moringa oleifera leaves extract mucoadhesive gel (2 %) | 3 | 3 | Monthly during treatment period | Reduction in size | Both treatments reduced the size of lesion. Moringa demonstrated more significant reduction compared to Retinol (p < 0.05) | Long term follow up missing. No data on recurrence |
| B:32(36) | B: Retino-A 0.1 % cream | 3 | ||||||||||
| 2 | Ahmad et al., 2023 | No data available | No data available | 283(300) | A:94(100) | A: curcumin capsules 500 mg | 1 | 3 | Every 30 days during treatment period; monthly during follow up period for 3 months | Number of patients with complete cure/partial cure | Participants cured with oral curcumin - 51 %. Participants cured with oral lycopene-63 % Participants cured with combination curcumin and lycopene- 72 % |
Long term follow up missing. No data on recurrence |
| B:95(100) | B: oral lycopene tablet 4 mg | 1 | ||||||||||
| C:94(100) | C: 4 mg of lycopene tablet + 500 mg curcumin capsule | 1 | ||||||||||
| 3 | Fathima et al., 2022 | 45.15 ± 11.37 | 17 M 3 F | 20(23) | A: 10 | A: 1 % (w/v) Bleomycin solution + oral antioxidant | 1 for bleomycin solution, 2 for antioxidant capsule | 3 | 3 months | Reduction in lesion size and histopathologcal dysplastic features | Significant reduction in lesion size with belomycin antioxidant treatment. (p < 0.05) Reduction in gradess of dysplasia after treatment with curcumin oral gel, but no difference in size |
Long term follow up missing. No data on recurrence |
| B:10 | B: curcumin oral gel + oral antioxidant | 6 for curcumin oral gel, 2 for antioxidant capsules | ||||||||||
| 4 | Mane et al., 2021 | A: 43 ± 4.9 | No data available | 30 | A: 15 | A: topical 5 % Imiquimod + benzocaine jelly | 3(per week) | 1.5 months | Every week for 6 weeks; monthly during follow up period for 6 months | The clinical response measured by reduction in size and Van der Waals (OLEP) staging of oral leukoplakia | Significant reduction in lesion after treatment in Imiquimod,but not with Aquasol (p < 0.05) | No recurrence after 6 momths |
| B: 42 ± 5.2 | B: 15 | B: tablet Aquasol (Vitamin A) (50,000 IU) | 2 | |||||||||
| 5 | Nagarajan et al., 2020 | 43.1 | 10 M | 10 | A: 5 | A: capsule omeprazole of 20 mg | 2 | 1 month | Every week for 4 weeks | Reduction in size and roughness of the lesion | Significant reduction is size and roughness with Omeprazole and Vitamin A, no difference between the groups (p < 0.05) | Long term follow up missing. No data on recurrence |
| B: 5 | B: capsule vitamin A 200000 I.U. | 1(per week) | ||||||||||
| 6 | Singh et al., 2017 | No data available | A: 28M 2F | 60 | A: 30 | A: Calendula officinalis gel containing antioxidants 2 mg by weight, per gram of gel | 3 | 1 month | Every month during follow up period for 3 months | Reduction in size of the lesion | Significant reduction is size with Calendula and Lycopene gel, no difference between the groups (p < 0.05) | Long term follow up missing. No data on recurrence |
| B: 22M 8F | B: 30 | B: lycopene gel containing lycopene antioxidant 2 mg by weight, per gram of gel. | 3 | |||||||||
| 7 | Kuriakose et al., 2016 | No data available | A: 79M 32F | 223 | A: 105(111) | A:600 mg capsules of curcumin, reconstituted with turmeric oil and dispensed in capsules (3.6 g/day) | 2 | 6 months | Monthly for 12 months | Number of patients with complete response/partial response based on clinical and histological criteria | Significantly better response with curcumin clinically and histologically (p < 0.05) | Treatment longer than 6 months did not show additional benefit. |
| B: 82M 30F | B: 108(112) | B: placebo capsules containing cellulose | 2 | |||||||||
| 8 | Mallery et al., 2015 | 44 to 77 (mean 62.2 ± 1.8) |
A: 15M 7F | A:bioadhesive gels that contained either 10 % w/w freeze dried black raspberries (BRB) | 12 weeks | Every 10–14 days during treatment period | Reduction in size of the lesion, histology grade and Loss of heterozygosity events | Significant reduction in lesional sizes, histologic grades with BRB (p < 0.05) | 2 lesions progressed to higher dysplasia in 4–31 months follow up. | |||
| 32 to 78 (mean 57.7 ± 2.9) | B: 9M 9M | B: Placebo | 4 | |||||||||
| 9 | Nagao et al., 2015 | A: 64.8 ± 10.0 | A: 13M 10 F | 33(46) | A: 16(23) | A: 10 mg day−1 of beta-carotene and 500 mg day−1 of vitamin C | 1 | 1 year | Monthly during follow up period for 6 months | Number of patients with complete cure/partial cure | Beta-carotene and vitamin C were neither effective for clinical remission, nor for protection against the development of cancer | 2 patients each from both groups developed oral cancer within 60 months follow up |
| B: 65.9 ± 8.7 | B: 12M 11F | B:17(23) | B: 50 mg day−1 of vitamin C | 1 | ||||||||
| 10 | Patel et al., 2014 | 47 | A: 19M 2F | 41 | A: 21 | A: 6 mg of lycopene + vitamin E (400 I.U.) + selenium (200 mcg) | 2 | 3 months | Every 15 days during treatment period; every month during follow up period for 3 months | Reduction in size and number of patients with complete cure/partial cure | Significant reduction in size after combination treatment. Significant number of patients with complete and partial response (p < 0.05) | Long term follow up missing. No data on recurrence |
| B: 19M 1F | B: 20 | B: placebo capsules | 1 | |||||||||
| 11 | Papadimitrakopoulou et al., 2009 | A: 56.9 ± 14.0 | A: 45M 36F | 154(162) | A: 77(81) | A:13-cis retinoic acid 13cRA (0.5 mg/kg/d orally for 1 year followed by 0.25 mg/kg/d orally for 2 years) | 1 | 3 years | 3 months and 2 years of follow-up whenever possible | Number of patients with complete cure/partial cure | Response to 13cRA was significantly better than BC + RP and RP alone. Response to BC + RP was significantly better than RP alone (p < 0.05) | 4 patients from 13cRA group, 5 pateints from BC + RP group and 2 from RP alone group developed oral cancer within 60 months follow up |
| B:55.0 ± 12.2 | B: 24M 21F | B: 42(45) | B: beta-carotene (BC) (50 mg/d orally) + Retinyl palmitate RP (25,000 U/d orally) | 1 | ||||||||
| C: 55.4 ± 14.4 | C: 16M 20F | C: 35(36) | C: Retinyl palmitate (RP) (25,000 U/d orally) | 1 | ||||||||
| 12 | Chiesa et al., 2005 | No data available | A: 60M 24F | 170 | A: 84 | A: 4-HPR orally at a dose of 200 mg day−1 (2 capsules of 100 mg) | 2 | 1 year | Every 2 months for 1st year; every 3 months for 2 nd year; every 4months for 3rd year; every 6 months thereafter | Number of patients with recurrences | Fenretinide protected against relapses and new lesions up to 19 months after randomization. | Study was underpowered and provided no indication to whether 4-HPR protects against oral cancers in this high-risk population |
| B: 61M 25F | B: 86 | B: Placebo was not given | ||||||||||
| 13 | Singh et al., 2004 | No data available | A: 15M 5F | 58 | A: 20 | A: 8 mg lycopene daily | 2 | 3 mon-ths | Every 7–10 days during the treatment period and then every 15 days during the follow up period for 2 months | Number of patients with complete cure/partial cure | Patients receiving 8 mg as well as 4 mg of lycopene in showed highly significant difference in response as compared to placebo (p < 0.05) |
Long term follow up missing. No data on recurrence |
| B: 14M 6F | B:20 | B: 4 mg lycopene daily | 2 | |||||||||
| C: 15M 3F | C:18 | C: Placebo capsules were given. | 2 | |||||||||
3.2. Characteristics of the interventions
Lycopene was used in four trials, either alone27,30, 31, 32 or in combination31,32 among which two trials utilized capsule form,27,31 whereas two used gel formulation30,32 for topical application. Three trials explored the efficacy of curcumin.29,31,33 Vitamin A was used as capsules28,34 or in other forms like retinol cream,35 retinyl palmitate, 13-cis retinoic acid (13cRA),26 and synthetic retinoid fenretinide.23 β carotene, vitamin C, vitamin E, and selenium were used in trials in combination with other antioxidants rather than sole intervention. Antioxidant-rich compounds, including raspberries,25 Moringa leaves,35 and Calendula officinalis,30 were also used in individual trials. Three trials tested the efficacy of the same interventions in different forms and doses against each other24,26,27
3.3. Characteristics of the outcome measures
Seven out of 13 investigations utilized lesion measurement of the lesion as the clinical metric for evaluating change.25,28,30,32, 33, 34, 35 Additionally, three research studies incorporated photography to measure the lesion's size accurately.25,30,34 Five of the investigations evaluated the number of patients showing complete or partial response.24,26,27,31,32For two trials, complete response was defined as the complete disappearance of the lesion.29,32 However, another two trials defined complete response when there was no evidence of a lesion for at least four weeks.26,27 Histological features were evaluated as a part of treatment response in five studies.24, 25, 26, 27, 28,33 Three investigations utilized distinct categories for clinical assessment. Mane et al. used the bi-dimensional assessment of lesions and color photography, along with the van der Waals (OLEP) staging system, to quantify the clinical and histological aspects of oral leukoplakia.28 Mallery et al. used a responsiveness score that considered the magnitude of changes in lesional size, histologic grade, and loss of heterozygosity.25 In the study by Nagarajan et al., the assessment of the treatment response included size and surface texture, salivary pH, and fractal analysis.34
3.4. Beneficial effects of specific antioxidant lycopene
Antioxidant lycopene has been shown to show consistent results in four trials. Regardless of the form, whether taken orally or applied as a topical agent, lycopene has demonstrated effectiveness in remission OL. Two studies reported 4 mg capsules on a daily dosage as effective.27,31 Gel preparation containing 2 mg of lycopene was adequate for topical application when used daily.30,32 Further, lycopene was found to be more effective than curcumin in a direct comparison trial.31
3.5. Mixed findings on vitamin A and curcumin
As for both of these antioxidants, the results were inconsistent as some studies showed their effectiveness demonstrating significant benefit, in contrast to other studies that did not show a difference between the groups that used and the control groups. Two studies where curcumin capsules were taken orally were shown to be effective in OL remission.29,31 In contrast, topical application of curcumin did not show any reduction in the size of the lesion.33 Curcumin, in combination with lycopene, was shown to be more effective than curcumin.31 Mane et al. did not find any significant reduction with vitamin A capsules (50000 IU), whereas Nagarajan et al. demonstrated effectiveness with 200000 IU, indicating dosage might be a significant factor.34 13-cis retinoic acid (13cRA) was more effective than retinyl palmitate in terms of clinical remission. However, there was no impact on oral cancer-free survival. Fenretinide, a synthetic retinoid, exhibited excellent tolerability and showed efficacy in reducing relapses and the formation of new leukoplakia both during and after treatment.23 The trial was stopped prematurely due to a significantly low number of participants and lacked sufficient statistical power to demonstrate any preventive benefit against oral cancer.
3.6. Effect of combination treatments
There were four trials24,26,31,32 conducted using more than one antioxidant at the same time by combining two or more in the same arm. Lycopene and curcumin combination was shown to be more effective than individual treatments.31 The combination of β carotene and vitamin C was neither effective for clinical remission nor protection against progression into invasive cancer.24 A formulation comprising 6 mg of lycopene + vitamin E (400 I.U.) + selenium (200 mcg) was effective in clinical and histological remission.32 A combination of β carotene and retinyl palmitate showed a better response than treatment with retinyl palmitate alone.26
3.7. Safety and tolerability of the interventions
Only three studies reported on the safety and tolerability of the interventions used in the trials.25,26,28 In the three-arm trial using 13cRA, it was reported that cheilitis, conjunctivitis, and skin reaction were significantly more common in the patients on 13cRA compared to others on β carotene and retinyl palmitate combination or retinyl palmitate alone.26 Other than those, arthralgia, myalgia, headache, and gastrointestinal symptoms were also reported by several patients using 13cRA. Six patients discontinued the 13cRa treatment because of the toxicities. The other two studies reported that the interventions did not cause any reactions in patients and was tolerable.
3.8. Risk of bias
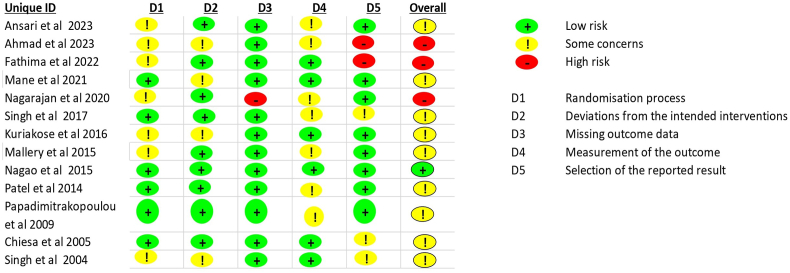
Out of the thirteen trials, one study exhibited an overall low risk of bias according to the standards employed in the critical evaluation of studies.24 Nine studies were determined to have an unclear risk of bias.23,25, 26, 27, 28, 29, 30,32,35 The remaining studies were deemed to have a high risk of bias.31,33,34 The results are shown in Fig. 2.
Fig. 2.
Risk of Bias of the included trials.
4. Discussion
This systematic review assessed the effectiveness of various antioxidants in the management of oral leukoplakia (OL). Lycopene emerged as the most consistently effective antioxidant, showing significant effectiveness in reducing lesion size both in oral and topical forms. Four trials demonstrated its success, where two trials27,31 studied lycopene in capsule form, and another two,30,32 studied its topical form. Both 8 mg and 4 mg lycopene capsules elicited a histological response devoid of any adverse effects, albeit with a limited follow-up period of only 2 months. Additionally, lycopene was more effective when combined with curcumin in one study. However, the findings for other antioxidants, such as vitamin A and curcumin, were mixed. Vitamin A showed variable results based on variable dosages ranging from no significant effect at 50,000 IU in one study28 to a substantial impact at 200,000 IU in another study.34 Similarly, curcumin in oral form showed benefits in two trials,29,31 but its topical application failed to show a reduction in lesion size. In some cases, combination treatments, such as lycopene with vitamin E and selenium, also demonstrated clinical remission. Despite these positive findings, the review identified inconsistencies, particularly in studies involving β-carotene and vitamin C, which did not show clinical remission or prevention of malignant transformation. Notably, three trials highlighted recurrence and progression to dysplasia or invasive cancer even after antioxidant treatment.24,26
The findings of this review align with previous studies that suggest lycopene's role as a potent antioxidant in the management of another OPMD, oral submucous fibrosis (OSF)36,37 Earlier studies have also highlighted lycopene's ability to modulate cell growth and play a role in immune and inflammatory diseases, including mucocutaneous diseases, neurological diseases, cardiovascular diseases, and cancer.38 Conversely, although the results are mixed for vitamin A, its role was never assessed in any other OPMD. The inconsistency in the effectiveness of curcumin, β-carotene, and vitamin C align with previous findings on oral submucous fibrosis (OSMF).36 This could be attributed to study design, dosage, or antioxidant formulation variations. Our review adds value by providing evidence on combination therapies, particularly the effectiveness of lycopene and curcumin when used together. While combination therapy has been underexplored in previous studies, the current review highlights its potential advantages in improving treatment outcomes for OL. Concordantly, the combination therapy has also proven beneficial in OSF.36 The reports on low serum and salivary total antioxidant capacity (TAC) in lichen planus39 and a significant reduction in lipid peroxidation in OL,40 as well as the alteration in levels of superoxide dismutase in OL41 are some of the evidence favoring altered level of antioxidants in OPMD. Therefore, we may say that the antioxidants may have the potential to manage OPMD because of their ability to counteract oxidative stress by scavenging ROS. This is especially relevant in OL, where oxidative damage to DNA, proteins, and lipids contributes to carcinogenesis.14 Lycopene, in particular, has strong antioxidant properties and can regulate redox signaling pathways involved in cellular proliferation and survival, which may explain its effectiveness in lesion remission.42 Lycopene mitigates the effects of ROS via radical addition, adduct formation or electron transfer to the radical.43 Lycopene has been shown to improve the levels of enzymatic antioxidants (catalase, superoxide dismutase, and peroxidase) and nonenzymatic antioxidants, including vitamins C and E, by augmenting the cellular antioxidant defense system.43 Curcumin, another antioxidant explored in this review, is known for its anti-inflammatory and antioxidant properties. Curcumin inhibits lipid peroxidation by oxidizing linoleate, a polyunsaturated fatty acid that forms a radical.44 In vitro and in vivo testing on rat peritoneal macrophages showed that it scavenges superoxide anions, hydrogen peroxide, and nitrite radical. Inducible nitric oxide synthase (iNOS) in macrophages creates significant levels of NO for the ‘oxidative burst'.45 Curcumin decreases macrophage iNOS activity, lowering ROS production in response to oxidative stress.45
However, its inconsistent results owing to different formulations highlights the need for further investigation into its bioavailability and mechanism of action in OL. Similarly, the variation in vitamin A outcomes could be due to its role in cellular differentiation and epithelial repair. Vitamin A can interact with peroxyl radicals, functioning as a chain-breaking antioxidant prior to the radicals' engagement with lipids and the formation of hydroperoxides, thereby averting cellular harm.46 Furthermore, it may enhance antioxidant activity indirectly by upregulating superoxide dismutase (SOD) and catalase.46 Still, the dosage appears to play a critical role in determining its effectiveness.
It is significant to highlight that, despite surgery being the preferred option for many clinicians in the management of leukoplakia, there is a lack of randomized controlled trials comparing the outcomes of surgical excision with no treatment or placebo.47,48 The sole data accessible is derived from observational studies that compare cancer incidence rates in individuals who received surgical therapy for oral leukoplakias in comparison to those who did not.47,48 These studies exhibit diagnostic and inclusion criteria variations, follow-up intervals, participant demographics, and surgical procedures utilized.49,50 The studies also exhibit considerable variability in outcomes and are even contradictory in their conclusions.
Another critical factor to consider is the lack of standardized protocols for evaluating outcomes in this field. Although much research has evaluated the resolution of clinical and histological aspects, it is essential to acknowledge that these analyses may not substantially influence the malignant transformation of OL. Further, despite its clinical presentation, OL comprises a dysplastic component, and a substantial fraction harbors carcinomatous foci.51 During histological investigation, the severity of epithelial dysplasia is often evaluated. Nevertheless, this evaluation is prone to considerable intra- and inter-observer subjectivity, which is a significant concern among pathologists.52
We observed significant heterogeneity in the study designs, intervention types, and outcome measures across the included trials. Ten of the thirteen studies used a two-arm parallel design, while the remaining three used a three-arm design, which impacted comparability. Dosages and formulations of antioxidants also varied, with some trials using oral capsules and others using topical gels or creams. The duration of follow-up was another source of variation, ranging from short-term studies (less than one year) to one study with a one-year follow-up. Short follow-up periods limit the ability to assess long-term outcomes such as recurrence and malignant transformation. The lack of standardization in outcome measures, with some studies focusing solely on lesion size and others evaluating histological changes, further contributed to heterogeneity. This variation makes it challenging to draw definitive conclusions. Further, the small sample sizes in many trials reduce the statistical power of the findings, particularly for vitamin A and curcumin. The reporting of safety and tolerability was limited, with only three studies providing details on adverse events, which hampers the comprehensive evaluation of antioxidant treatments. There was substantial heterogeneity in forms, dosage, administration frequency, treatment duration, and follow-up time among all included studies.
Despite the limitations, the findings of this review provide critical clinical insights. In both oral and topical forms, Lycopene appears to be a promising adjunctive treatment for OL and could be recommended in clinical settings. Its consistent efficacy across multiple studies suggests it may play a significant role in the non-surgical management of OL. The mixed results for vitamin A and curcumin indicate the need for further research to determine optimal dosing and formulations. Clinicians should exercise caution when recommending these treatments, particularly in light of the inconsistent evidence. Combination treatments, incorporating lycopene with curcumin or vitamin E and Selenium, show potential for enhanced efficacy and may offer a promising therapeutic approach. However, clinicians should remain mindful of the limited data on safety and long-term outcomes, particularly for high-dose vitamin A.
Future research should focus on large-scale, well-designed, randomized controlled trials with standardized dosing, delivery methods, and outcome measures to provide more robust evidence. Studies should incorporate long-term follow-up to assess recurrence and progression to dysplasia or invasive cancer. Further investigation is needed to explore the safety and tolerability of high-dose antioxidants, particularly vitamin A, and combination therapies. Bioavailability and formulation differences, especially for curcumin, should be a key focus of future research. As the next-generation sequencing technique becomes more accessible, it may help elucidate this antioxidant treatment's impacts at the molecular level. Linking antioxidant-tailored treatment with patient data, integrating omics-based features, and using artificial intelligence and machine learning may enable the clinical application of individualized OL treatment regimens by rapidly and cost-effectively analyzing patients' genetic and molecular profiles.
5. Conclusion
Based on the evaluation of treatment response, follow-up duration, and the intensity of side effects in the trials analyzed in this review, it can be inferred that lycopene shows potential as an effective antioxidant. Nevertheless, it is crucial to acknowledge that the effectiveness of the other antioxidants has only been assessed in a restricted number of studies. Therefore, further research, including bigger sample sizes and longer follow-up periods, is necessary before making relevant conclusions.
Parent consent
Not applicable.
Data availability statement
Not applicable.
Ethical clearance
Not applicable (Systematic review).
Funding statement
The APC is funded by Ajman University, United Arab Emirates.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowlegements
Nil.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jobcr.2025.03.001.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Warnakulasuriya S. Oral potentially malignant disorders: a comprehensive review on clinical aspects and management. Oral Oncol. 2020;102 doi: 10.1016/j.oraloncology.2019.104550. [DOI] [PubMed] [Google Scholar]
- 2.Zhang C., Li B., Zeng X., Hu X., Hua H. The global prevalence of oral leukoplakia: a systematic review and meta-analysis from 1996 to 2022. BMC Oral Health. 2023;23(1):645. doi: 10.1186/s12903-023-03342-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Speight P.M., Khurram S.A., Kujan O. Oral potentially malignant disorders: risk of progression to malignancy. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology. 2018;125(6):612–627. doi: 10.1016/j.oooo.2017.12.011. [DOI] [PubMed] [Google Scholar]
- 4.Maserejian N.N., Joshipura K.J., Rosner B.A., Giovannucci E., Zavras A.I. Prospective study of alcohol consumption and risk of oral premalignant lesions in men. Cancer Epidemiol Biomarkers Prev. 2006;15(4):774–781. doi: 10.1158/1055-9965.EPI-05-0842. [DOI] [PubMed] [Google Scholar]
- 5.Ranganathan K., Kavitha L. Clinical aspects of oral cancer and potentially malignant disorders in South and Southeast Asia. Oral Dis. 2024 doi: 10.1111/odi.15008. Published online May 30. [DOI] [PubMed] [Google Scholar]
- 6.Lee C.H., Ko A.M.S., Warnakulasuriya S., et al. Intercountry prevalences and practices of betel-quid use in south, southeast and eastern Asia regions and associated oral preneoplastic disorders: an international collaborative study by Asian betel-quid consortium of south and east Asia. Int J Cancer. 2011;129(7):1741–1751. doi: 10.1002/ijc.25809. [DOI] [PubMed] [Google Scholar]
- 7.Kumari P., Debta P., Dixit A. Oral potentially malignant disorders: etiology, pathogenesis, and transformation into oral cancer. Front Pharmacol. 2022;13 doi: 10.3389/fphar.2022.825266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Farah C.S. Molecular, genomic and mutational landscape of oral leukoplakia. Oral Dis. 2021;27(4):803–812. doi: 10.1111/odi.13608. [DOI] [PubMed] [Google Scholar]
- 9.Villa A., Woo S.B. Leukoplakia—a diagnostic and management algorithm. J Oral Maxillofac Surg. 2017;75(4):723–734. doi: 10.1016/j.joms.2016.10.012. [DOI] [PubMed] [Google Scholar]
- 10.Chen Q., Dan H., Pan W., et al. Management of oral leukoplakia: a position paper of the society of oral medicine, Chinese stomatological association. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology. 2021;132(1):32–43. doi: 10.1016/j.oooo.2021.03.009. [DOI] [PubMed] [Google Scholar]
- 11.Dong Y., Chen Y., Tao Y., et al. Malignant transformation of oral leukoplakia treated with carbon dioxide laser: a meta-analysis. Lasers Med Sci. 2019;34(1):209–221. doi: 10.1007/s10103-018-2674-7. [DOI] [PubMed] [Google Scholar]
- 12.Hanna G.J., Villa A., Nandi S.P., et al. Nivolumab for patients with high-risk oral leukoplakia: a nonrandomized controlled trial. JAMA Oncol. 2024;10(1):32–41. doi: 10.1001/jamaoncol.2023.4853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Andrés C.M.C., Pérez de la Lastra J.M., Juan C.A., Plou F.J., Pérez-Lebeña E. Antioxidant metabolism pathways in vitamins, polyphenols, and selenium: parallels and divergences. IJMS. 2024;25(5):2600. doi: 10.3390/ijms25052600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barros CC. da S., Freitas R., de A., Miguel MC. da C., Dantas da Silveira É.J. DNA damage through oxidative stress is an important event in oral leukoplakia. Arch Oral Biol. 2022;135 doi: 10.1016/j.archoralbio.2022.105359. [DOI] [PubMed] [Google Scholar]
- 15.Kumar J., Teoh S.L., Das S., Mahakknaukrauh P. Oxidative stress in oral diseases: understanding its relation with other systemic diseases. Front Physiol. 2017;8:693. doi: 10.3389/fphys.2017.00693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alkadi H. A review on free radicals and antioxidants. IDDT. 2020;20(1):16–26. doi: 10.2174/1871526518666180628124323. [DOI] [PubMed] [Google Scholar]
- 17.Meliante P.G., Petrella C., Fiore M., Minni A., Barbato C. Antioxidant use after diagnosis of head and neck squamous cell carcinoma (HNSCC): a systematic review of application during radiotherapy and in second primary cancer prevention. Antioxidants. 2023;12(9):1753. doi: 10.3390/antiox12091753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McClure J.B., Divine G., Alexander G., et al. A comparison of smokers' and nonsmokers' fruit and vegetable intake and relevant psychosocial factors. Behav Med. 2009;35(1):14–22. doi: 10.3200/BMED.35.1.14-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McPhillips J.B., Eaton C.B., Gans K.M., et al. Dietary differences in smokers and nonsmokers from two southeastern New England communities. J Am Diet Assoc. 1994;94(3):287–292. doi: 10.1016/0002-8223(94)90370-0. [DOI] [PubMed] [Google Scholar]
- 20.Kaugars G.E., Silverman S., Lovas J.G.L., et al. A clinical trial of antioxidant supplements in the treatment of oral leukoplakia. Oral Surg Oral Med Oral Pathol. 1994;78(4):462–468. doi: 10.1016/0030-4220(94)90039-6. [DOI] [PubMed] [Google Scholar]
- 21.Page M.J., McKenzie J.E., Bossuyt P.M., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg. 2021;88 doi: 10.1016/j.ijsu.2021.105906. [DOI] [PubMed] [Google Scholar]
- 22.Sterne J.A.C., Savović J., Page M.J., et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366 doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 23.Chiesa F., Tradati N., Grigolato R., et al. Randomized trial of fenretinide (4‐HPR) to prevent recurrences, new localizations and carcinomas in patients operated on for oral leukoplakia: long‐term results. Intl J Cancer. 2005;115(4):625–629. doi: 10.1002/ijc.20923. [DOI] [PubMed] [Google Scholar]
- 24.Nagao T., Warnakulasuriya S., Nakamura T., et al. Treatment of oral leukoplakia with a low-dose of beta-carotene and vitamin C supplements: a randomized controlled trial. Int J Cancer. 2015;136(7):1708–1717. doi: 10.1002/ijc.29156. [DOI] [PubMed] [Google Scholar]
- 25.Mallery S.R., Tong M., Shumway B.S., et al. Topical application of a mucoadhesive freeze-dried black raspberry gel induces clinical and histologic regression and reduces loss of heterozygosity events in premalignant oral intraepithelial lesions: results from a multicentered, placebo-controlled clinical trial. Clin Cancer Res. 2014;20(7):1910–1924. doi: 10.1158/1078-0432.CCR-13-3159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Papadimitrakopoulou V.A., Lee J.J., William W.N., et al. Randomized trial of 13-cis retinoic acid compared with retinyl palmitate with or without beta-carotene in oral premalignancy. J Clin Oncol. 2009;27(4):599–604. doi: 10.1200/JCO.2008.17.1850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Singh M., Krishanappa R., Bagewadi A., Keluskar V. Efficacy of oral lycopene in the treatment of oral leukoplakia. Oral Oncol. 2004;40(6):591–596. doi: 10.1016/j.oraloncology.2003.12.011. [DOI] [PubMed] [Google Scholar]
- 28.Mane S., Patilsoman B., Bhate P., Das D., Malusare P., Tomar N. To evaluate the efficacy and tolerability of topical 5% imiquimod in cases of oral leukoplakia: a pilot study. J Indian Acad Oral Med Radiol. 2021;33(1):27. doi: 10.4103/jiaomr.jiaomr_147_20. [DOI] [Google Scholar]
- 29.Kuriakose M.A., Ramdas K., Dey B., et al. A randomized double-blind placebo-controlled phase IIB trial of curcumin in oral leukoplakia. Cancer Prev Res. 2016;9(8):683–691. doi: 10.1158/1940-6207.CAPR-15-0390. [DOI] [PubMed] [Google Scholar]
- 30.Singh M., Bagewadi A. Comparison of effectiveness of Calendula officinalis extract gel with lycopene gel for treatment of tobacco-induced homogeneous leukoplakia: a randomized clinical trial. Int J Pharm Investig. 2017;7(2):88–93. doi: 10.4103/jphi.JPHI_19_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ahmad T., Khan I., Sultana A., Rizvi M.M.A. A comparative study to evaluate the therapeutic effects of nutraceuticals in oral leukoplakia:- A randomized clinical trail. Natl J Maxillofac Surg. 2023;14(3):413–419. doi: 10.4103/njms.njms_25_23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Patel J., Umarji H., Dhokar A., Sapkal R., Patel S., Panda A. Randomized controlled trial to evaluate the efficacy of oral lycopene in combination with vitamin E and selenium in the treatment of oral leukoplakia. J Indian Acad Oral Med Radiol. 2014;26(4):369. doi: 10.4103/0972-1363.155630. [DOI] [Google Scholar]
- 33.Fathima A.S., Manoharan G.V.M.G. Comparison of efficacy of topical application of bleomycin with adjuvant antioxidants vs. Topical curcumin oral gel with adjuvant antioxidants in the treatment of oral leukoplakia. J Indian Acad Oral Med Radiol. 2022;34(4):409. doi: 10.4103/jiaomr.jiaomr_201_22. [DOI] [Google Scholar]
- 34.Nagarajan N., Jayachandran S. Therapeutic effect of proton pump inhibitor (Omeprazole) on homogenous leukoplakia with clinico-fractal analysis: a pilot study. J Indian Acad Oral Med Radiol. 2020;32(4):323. doi: 10.4103/jiaomr.jiaomr_160_20. [DOI] [Google Scholar]
- 35.Ansari S., Charantimath S., Fernandes A., Malik J.B., Panta P. Comparison of effectiveness of Moringa oleifera leaves extract gel (2%) with retino A (0.1%) cream for treatment of oral leukoplakia: double blinded randomized control trial. Gulf J Oncolog. 2023;1(43):12–18. [PubMed] [Google Scholar]
- 36.Pérez-Leal M., Lanciano F., Flacco N., Estornut C., Carceller M.C. Antioxidant treatments in patients with oral submucous fibrosis: a systematic review. J Oral Pathol Med. 2024;53(1):31–41. doi: 10.1111/jop.13503. [DOI] [PubMed] [Google Scholar]
- 37.Gopinath D., Hui L.M., Veettil S.K., Balakrishnan Nair A., Maharajan M.K. Comparative efficacy of interventions for the management of oral submucous fibrosis: a systematic review and network meta-analysis. J Personalized Med. 2022;12(8) doi: 10.3390/jpm12081272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Imran M., Ghorat F., Ul-Haq I., et al. Lycopene as a natural antioxidant used to prevent human health disorders. Antioxidants. 2020;9(8):706. doi: 10.3390/antiox9080706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hatami M., Rezaei M., Sadeghi M., Tadakamadla J., Pekiner F.N., Mozaffari H.R. A systematic review and meta-analysis on serum and salivary levels of total antioxidant capacity and C-reactive protein in oral lichen planus patients. Arch Oral Biol. 2022;140 doi: 10.1016/j.archoralbio.2022.105445. [DOI] [PubMed] [Google Scholar]
- 40.Srivastava K.C., Austin R.D., Shrivastava D., Pranavadhyani G. Oxidant-antioxidant status in tissue samples of oral leukoplakia. Dent Res J. 2014;11(2):180–186. [PMC free article] [PubMed] [Google Scholar]
- 41.Kuthoor J.M., Sunil E.A., Raghavan S.E., Purushothaman B.P. A comparative investigation of the activity of superoxide dismutase in patients with leukoplakia and healthy controls. Indian J Dent Res. 2022;33(3):287–291. doi: 10.4103/ijdr.ijdr_1009_21. [DOI] [PubMed] [Google Scholar]
- 42.Trejo-Solís C., Pedraza-Chaverrí J., Torres-Ramos M., et al. Multiple molecular and cellular mechanisms of action of lycopene in cancer inhibition. Evid Based Complement Alternat Med. 2013;2013 doi: 10.1155/2013/705121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shafe M.O., Gumede N.M., Nyakudya T.T., Lycopene Chivandi E. A potent antioxidant with multiple health benefits. Suzuki T., editor. Journal of Nutrition and Metabolism. 2024;2024(1) doi: 10.1155/2024/6252426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wilken R., Veena M.S., Wang M.B., Srivatsan E.S. Curcumin: a review of anti-cancer properties and therapeutic activity in head and neck squamous cell carcinoma. Mol Cancer. 2011;10(1):12. doi: 10.1186/1476-4598-10-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Joe B., Vijaykumar M., Lokesh B.R. Biological properties of curcumin-cellular and molecular mechanisms of action. Crit Rev Food Sci Nutr. 2004;44(2):97–111. doi: 10.1080/10408690490424702. [DOI] [PubMed] [Google Scholar]
- 46.Didier A.J., Stiene J., Fang L., Watkins D., Dworkin L.D., Creeden J.F. Antioxidant and anti-tumor effects of dietary vitamins A, C, and E. Antioxidants. 2023;12(3):632. doi: 10.3390/antiox12030632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lombardi N., Arduino P.G., Lampiano M., et al. Surgical treatment compared with “wait and see” in patients affected by oral leukoplakia to prevent oral cancer: preliminary data from a multicenter randomized controlled trial. Oral Dis. 2024 doi: 10.1111/odi.15058. Published online June 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Arduino P.G., Lodi G., Cabras M., et al. A randomized controlled trial on efficacy of surgical excision of nondysplastic leukoplakia to prevent oral cancer. Cancer Prev Res. 2021;14(2):275–284. doi: 10.1158/1940-6207.CAPR-20-0234. [DOI] [PubMed] [Google Scholar]
- 49.Monteiro L., Barbieri C., Warnakulasuriya S., et al. Type of surgical treatment and recurrence of oral leukoplakia: a retrospective clinical study. Med Oral. 2017 doi: 10.4317/medoral.21645. 0-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.de Pauli Paglioni M., Faustino I.S.P., Pedroso C.M., et al. Oral leukoplakia treated with laser and scalpel: interim trial results of recurrence and malignancy. Oral Dis. 2024;30(7):4161–4164. doi: 10.1111/odi.14961. [DOI] [PubMed] [Google Scholar]
- 51.Gopinath D., Thannikunnath B.V., Neermunda S.F. Prevalence of carcinomatous foci in oral leukoplakia: a clinicopathologic study of 546 Indian samples. J Clin Diagn Res. 2016;10(8):ZC78–83. doi: 10.7860/JCDR/2016/16815.8305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gopinath D., On C.Y., Veettil S.K., Tilakaratne W.M. Is binary grading better than WHO system for grading epithelial dysplasia? A systematic review and meta-analysis. Oral Dis. 2025;31(1):59–68. doi: 10.1111/odi.15160. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Not applicable.