Abstract
Background
Chikungunya, and Zika emerged in the 2010s in the Americas, causing simultaneous epidemics with dengue. However, little is known of these Aedes-borne diseases (ABDs) joint patterns and contributors at the population-level.
Methods
We applied a novel Poisson-multinomial spatial model to the registered cases of dengue (n = 291,820), chikungunya (n = 75,913), and Zika (n = 72,031) by municipality in Colombia, 2014–2016. This model estimates the relative risk of total ABDs cases and associated factors, and, simultaneously, the odds of presence and contributors of each disease using dengue as a baseline category. This approach allows us to identify combined characteristics of ABDs, since they are transmitted by the same mosquitoes, while also identifying differences between them.
Results
We found an increased ABDs risk in valleys and south of the Andes, the Caribbean coast, and borders, with temperature as the main contributor (Relative Risk 2.32, 95% Credible Interval, CrI, 2.05–2.64). Generally, dengue presence was the most probable among the ABDs, although that of Zika was greater on Caribbean islands. Chikungunya and Zika were more likely present than dengue in municipalities with less vegetation (Odds Ratio, OR, 0.75, 95%CrI 0.65–0.86, and 0.85, 95%CrI 0.74–0.99, respectively). Chikungunya tended to be present in more socially vulnerable areas than dengue (OR 1.20, 95%CrI 0.99–1.44) and Zika (OR 1.19, 95%CrI 0.95–1.48).
Conclusions
Important differences between the ABDs were identified and can help guide local and context-specific interventions, such as those aimed at preventing cases importation in border and tourism locations and reducing chikungunya burden in socially vulnerable regions.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12879-025-10782-0.
Introduction
Dengue affects tropical and subtropical regions, with an estimated 30-fold increase in incidence over the past 50 years and 6.5 million cases in 2023 [1, 2]. Caused by four distinct viral serotypes and transmitted by Aedes mosquitoes, dengue control is difficult, and epidemics occur cyclically [3]. The last decades, dengue-endemic countries faced the emergence of other Aedes-borne diseases, notably chikungunya and Zika, leading to co-circulation and simultaneous epidemics, i.e. syndemics, a greater challenge to public health [3–5]. This was the situation in Colombia between 2014 and 2016, one of the countries most affected by dengue in the Americas since the 1970s [6, 7]. Chikungunya emerged in Colombia in 2014, followed by Zika in 2015, all while a dengue epidemic was also ongoing [7]. Together, the three diseases were responsible for over 485 thousand registered cases in the country during this syndemic period [8].
Dengue, chikungunya, and Zika distribution at the population-level is associated with factors affecting the presence, density, and vector competence of Aedes mosquitoes, such as climate, vegetation, and sanitation [9–11]. Warmer temperatures caused by climate change are also thought to drive dengue expansion to previously unaffected regions [12–15]. Urban, densely populated, and socially vulnerable locations are considered at increased risk for Aedes-borne diseases, reflecting the mosquito's adaptation to living close to humans [11, 16–18].
Because these arboviruses share the same vector and co-circulate, it is expected that their spatial distribution is correlated, advocating for a joint analysis. In Colombia, a study applied multivariate scan statistics to detect simultaneous clusters of dengue, chikungunya, and Zika [7]. Although risk-areas for their joint presence were identified, clusters were also found for only one or two diseases at a time. We consider that certain population- and environmental-level factors (e.g., sanitation, humidity, etc.) impact each arbovirus distribution differently.
Aiming to simultaneously study and compare the spatial patterns and contributors of dengue, chikungunya, and Zika in Colombia during their first period of co-occurrence, we applied a previously proposed joint multivariate spatial model [19]. This model allows us to describe spatial trends of the total cases associated with Aedes-borne diseases and to compare the odds ratio of a municipality having Zika or chikungunya (which emerged during the study period) with dengue, which has been endemic in the region for decades. Since all inference is made under a single framework, a joint model has the advantage of capturing interdependencies among the diseases and improving the estimations as it relies on more data than individual models. Ultimately, our results aim to support the development of public health interventions that may impact all three Aedes-borne diseases simultaneously, as well as interventions that address the specificities of each disease.
Methods
This ecological study used national surveillance data of notified dengue, chikungunya, and Zika cases in Colombia grouped by municipality, from 2014 to 2016. The period of study corresponds to the period when the three diseases co-circulated causing simultaneous epidemics in Colombia (Supplementary Figure S1). Hereon, we use "Aedes-borne diseases" to encompass dengue, chikungunya, and Zika together.
We applied a Poisson-multinomial spatial model to estimate i) the relative risk of the total cases of Aedes-borne diseases, ii) the probability of presence of each disease, given the total, and ii) the association of covariates with the relative risk of Aedes-borne diseases and with the odds of presence of one disease in comparison to each other.
Study site
Colombia is located in northern South America and its territory is divided into 1,121 municipalities, grouped into 33 departments (Supplementary Figure S2). Colombia borders five countries, has two coastlines, an archipelago in the Caribbean Sea, three mountain ranges forming the Colombian Andes, a savanna region east of the Andes, and an Amazon rainforest in the south.
Data
Anonymized individual records of dengue (2014–2016), chikungunya (2014–2016), and Zika (2015–2016) cases were obtained from the Colombian National Public Health Surveillance System (SIVIGILA). Data correspond to individuals diagnosed with dengue, chikungunya, or Zika at a healthcare facility, using case definitions from Colombian National Health Institute protocols [20–22]. Cases matching a suspected or confirmed case definition (Supplementary Text S1) and with symptom onset between January 3 2014 to October 1 2016 (epidemiological weeks, EWs, 01/2014 to 39/2016) were included. Cases that were classified as discarded in the database were excluded from the analysis. Then, cases were aggregated by municipality of residence.
Environmental data for the same period, including the Normalised Difference Vegetation Index (NDVI, which quantifies vegetation greenness, with values near + 1 representing dense vegetation cover), mean temperature, relative humidity, and total precipitation by week and municipality, were obtained from data previously processed [23, 24]. We calculated the average values of these variables for each municipality (Supplementary Figure S3). Population estimates for 2014–2016 were obtained from Colombia's National Administrative Department of Statistics (DANE) and used to calculate the population density per km2(Supplementary Figure S4A). Socioeconomic data by municipality, including the percentage of people using rainwater for cooking, the percentage of homes with garbage collection, and the percentage of people with unsatisfied basic needs (UBN) were sourced from the national census of 2018 (DANE) (Supplementary Figure S4B-D). The UBN index captures socioeconomic vulnerabilities based on housing conditions, overcrowding, and access to basic sanitation, among others [25]. Two healthcare access indices for 2016–2017 were obtained from the Colombian National Health Observatory (ONS). The "real" index is related to the quality of the healthcare service (e.g., percentage of births attended by qualified personnel), while the "potential" is related to the offer of the service (e.g., number of healthcare beds and general practitioners). These indexes range from 0 to 100, with 0 representing the worst healthcare access and 100 the best (Supplementary Figure S4E-F) [26].
Statistical methods
We applied a Poisson-multinomial spatial model under the Bayesian framework, originally proposed by Schmidt et al. [19] Let , and denote the number of dengue, chikungunya, and Zika cases, respectively, in municipality i = 1, 2, …, n = 1121, during the study period. And let be the total number of cases of the three Aedes-borne diseases. We assume that.
| 1 |
being the number of expected cases considering a homogenous incidence rate across the municipalities, calculated as and the relative risk associated with the total number of cases of Aedes-borne diseases, modelled as
| 2 |
where is a vector of covariates, including an intercept, is a vector of coefficients, and is a latent effect for municipality i.
Now let be a three-dimensional vector with the number of registered cases of each disease in municipality i. When total > 0:
| 3 |
where is the vector of probabilities of occurrence of dengue, chikungunya, or Zika in municipality i, with . We assume that
| 4 |
to model the conditional probability of chikungunya presence (k = 2) given that it is chikungunya or dengue (k = 1), and of Zika presence (k = 3) given that it is Zika or dengue. Dengue was considered the baseline category due to endemicity before the introduction of chikungunya and Zika. The covariates' coefficients vary with k and represents the odds ratio of chikungunya or Zika presence compared to dengue associated with one standard deviation increase in covariate j. The odds ratio associated with covariates and chikungunya presence compared to Zika is obtained from , and vice-versa from .
Note that equations [2] and [4] include latent effects , k = 1, 2, 3. These components are present to accommodate overdispersion with respect to the Poisson () and the multinomial distributions (). Besides accommodating overdispersion, they also account for whatever is left in the relative risk of the total and the log odds of the multinomial after accounting for the covariates. As inference follows a Bayesian approach, a prior distribution needs to be assigned to Considering that the three diseases are transmitted by the same vectors, we assume that , i.e., conditional on a local effect , follows conditional multivariate normal distributions with covariance , where 13 is a 3-dimensional column-vector with all elements equal to 1. This induces a correlation among the latent effects, as within municipality i, the 's share, a priori, a common spatial effect , that captures unobserved effects that are common to the three diseases. We assume that follows a proper conditional autoregressive prior distribution (CAR) [27]. The covariance matrix captures potential covariance among the diseases within each municipality, accommodating any structure that is left after accounting for the common effect .
Models were fitted using the Stan platform [28] in R (version 4.3.3), with the package rstan (version 2.32.6). Covariates were standardised. Convergence was checked by the R-hat statistic and by visually inspecting the chains. Figures were created using the packages ggplot2 (version 3.5.0) and colorspace (version 2.1–0) in R.
Results
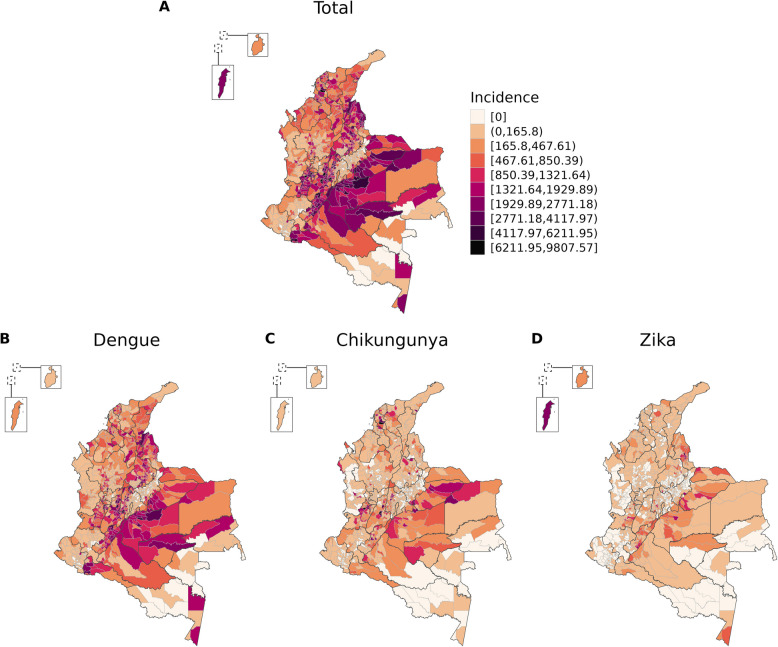
Between EWs 01/2014 and 39/2016, 291,820 dengue, 75,913 chikungunya and 72,031 Zika cases were registered in Colombia, totalling 439,769 cases of Aedes-borne diseases. The cumulative incidence at the national level was 629.8 cases per 100,000 inhabitants for dengue, 163.8 for chikungunya, 155.4 for Zika, and 949.1 for the three diseases. The incidence distribution by municipality is shown in Fig. 1.
Fig. 1.
Aedes-borne diseases (dengue, chikungunya, and Zika) total incidence (per 100,000 inhabitants) and for each disease by municipality, Colombia, EW 01/2014 to 39/2016
Total relative risk and probability of presence of each disease
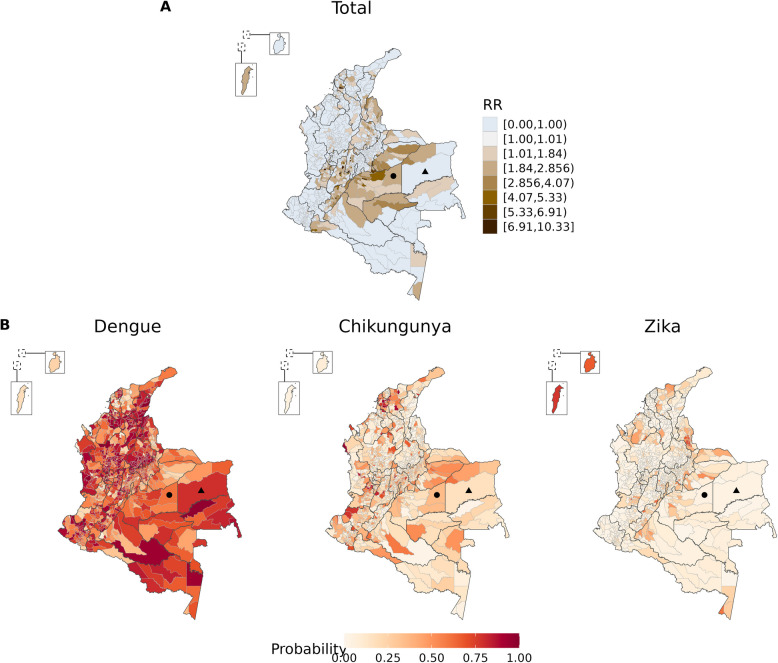
Figure 2A shows the relative risk for Aedes-borne diseases, i.e., for the total cases of the three diseases (dengue, chikungunya, and Zika) combined. Of the 1,121 municipalities, 357 (31.8%) had posterior mean relative risks > 1. Higher risks were concentrated south of the Andean region (Meta, Casanare and Guaviare departments), in valleys in the Andean region (Tolima, Huila, Cundinamarca and inland Valle del Cauca), near the border with Venezuela (Santander and Norte de Santander) and Ecuador (Putumayo), in the Caribbean coast (Bolívar and San Andrés island), and in the southernmost municipality of the country, Leticia, that borders Brazil and Peru.
Fig. 2.
Posterior mean (A) of the relative risk (RR) for the total cases of Aedes-borne diseases (dengue, chikungunya, and Zika) and (B) of probabilities of presence of each disease, given the total, by municipality, Colombia, epidemiological week 01/2014 to 39/2016. The solid circle and the solid triangle are the locations of two municipalities (Puerto Gaitán and Cumaribo, respectively) to serve as examples of the results interpretation in the main text
Figure 2B shows the conditional probability of presence of each disease. Generally, the probability of dengue presence was the highest among the arboviruses (Supplementary Figure S5). The probability of presence of one disease should be interpreted comparatively to the others and conditioned to the relative risk for the total Aedes-borne diseases. For example, from Fig. 2A, Puerto Gaitán (solid circle) had an overall relative risk for Aedes-borne diseases > 1 (above the expected for its population) and, comparatively from Fig. 2B, the probability of dengue presence was higher than that of chikungunya and Zika. In contrast, Cumaribo (solid triangle) had an overall relative risk < 1 (below the expected given its population). And, under low risk of Aedes-borne diseases, dengue was more likely present in Cumaribo than chikungunya and Zika.
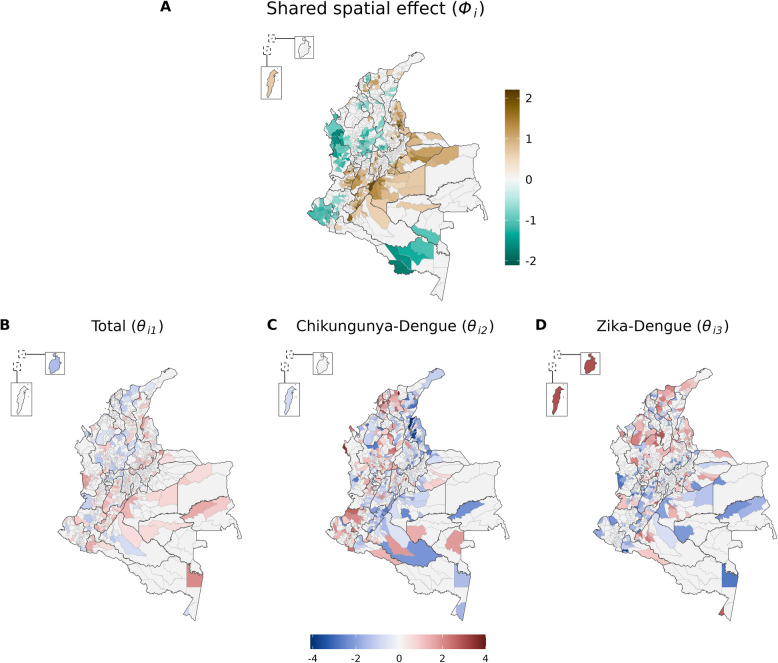
Latent effects
Figure 3A depicts the posterior mean of the shared spatial effect ( and Fig. 3B the disease-specific latent effects (). The distribution of the spatial effect was similar to that of the posterior relative risk (Fig. 2A). High values of and/or represent an increased probability of presence of chikungunya and/or Zika, respectively, compared to dengue, not explained by the covariates and the shared spatial effect.
Fig. 3.
Posterior mean of the shared spatial effect for Aedes-borne diseases (dengue, chikungunya, and Zika) (A) and of the latent effects associated with the total cases (equation [2]) (B) and with the presence of chikungunya (C) or Zika (D) compared to dengue (equation [4]) by municipality in Colombia, epidemiological week 01/2014 to 39/2016. Municipalities for which the 95% credible interval of the effect included 0 are shown in light grey
Association with covariates
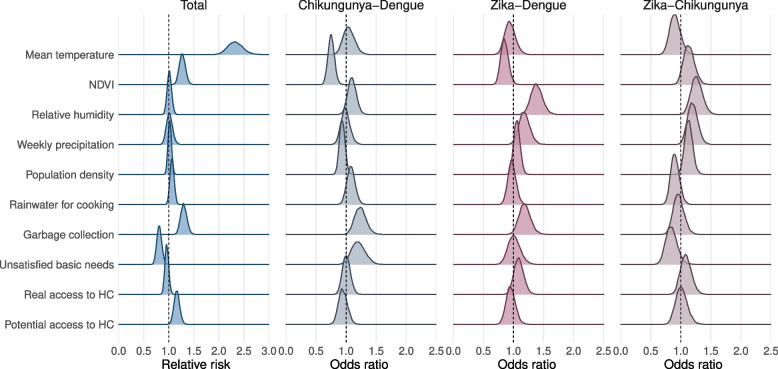
The posterior distribution of the estimated associations with covariates is presented in Fig. 4. To facilitate the interpretation of these results, we summarise them in the text with point estimates and the respective 95% posterior credible interval (CrI). Temperature showed the strongest association with the risk of Aedes-borne diseases (mean 2.32, 95%CrI 2.05–2.64). Warmer locations tended to have decreased odds of Zika presence compared to dengue (0.94, 95%CrI 0.78–1.13) and chikungunya (0.90, 95%CrI 0.74–1.09).
Fig. 4.
Posterior distribution of the relative risks associated with each of the covariates and the total number of cases of Aedes-borne diseases (dengue, chikungunya, and Zika), and of the odds ratios associated with each of the covariates and the odds of a municipality having chikungunya or Zika in comparison to dengue, and Zika in comparison to Chikungunya. NDVI = Normalized Difference Vegetation Index, HC = Healthcare
The NDVI was positively associated with the total Aedes-borne diseases risk (1.27, 95%CrI 1.16–1.40), and inversely associated with the odds of chikungunya (0.75, 95%CrI 0.65–0.86) and Zika (0.85, 95%CrI 0.74–0.99) presence compared to dengue. Locations with higher NDVI had slightly increased odds of Zika compared to chikungunya (1.13, 95%CrI 0.97–1.33).
Humidity, precipitation, and population density were not associated with the Aedes-borne diseases risk. However, municipalities with higher humidity and/or weekly precipitation had increased odds of Zika presence compared to dengue (1.38, 95%CrI 1.21–1.58; and 1.18, 95%CrI 1.01–1.37, respectively) and chikungunya (1.26, 95%CrI 1.09–1.46; and 1.20, 95%CrI 1.04–1.37). More humid municipalities tended to have higher odds of chikungunya presence compared to dengue (1.10, 95%CrI 0.97–1.24). Densely populated municipalities had increased odds of having Zika compared to chikungunya (1.13, 95%CrI 1.01–1.27). These areas also tended to have increased odds of Zika presence (1.06, 95%CrI 0.96–1.17), and lower odds of chikungunya presence (0.93, 95%CrI 0.85–1.03) compared to dengue.
Municipalities with a higher percentage of people using rainwater for cooking tended to have a positive association with the total risk (1.07, 95%CrI 0.99–1.17), and higher odds of chikungunya presence compared to dengue (1.08, 95%CrI 0.95–1.22) and Zika (1.11, 95%CrI 0.95–1.29).
Municipalities with a higher percentage of homes with garbage collection presented a higher risk of total Aedes-borne diseases (1.30, 95%CrI 1.18–1.43), and higher odds of presence of chikungunya (1.23, 95%CrI 1.06–1.44) and Zika (1.19, 95%CrI 1.01–1.39) compared to dengue.
The UBN was inversely associated with the risk of Aedes-borne diseases (0.81, 95%CrI 0.72–0.91), and borderline associated with increased odds of chikungunya presence compared to dengue (1.20, 95%CrI 0.99–1.44) and Zika (1.19, 95%CrI 0.95–1.48).
Real access to healthcare was not associated with the risk of Aedes-borne diseases. Increased Zika presence probability was estimated in locations with better real access to healthcare compared to dengue (1.09, 95%CrI 0.94–1.26) and chikungunya (1.08, 95%CrI 0.92–1.28). Finally, better potential healthcare access was positively associated with the total Aedes-borne diseases cases (1.16, 95%CrI 1.06–1.28), with no differences in the odds of presence between the diseases.
Discussion
We used a novel syndemic approach to estimate the relative risk of Aedes-borne diseases and the conditional probability of presence of each disease. The overall spatial distribution of Aedes-borne diseases varied across Colombia, with temperature identified as the main contributor to arbovirus presence at the municipal level. To our knowledge, this is the first study to use a joint spatial model for dengue, chikungunya, and Zika risk simultaneously for an entire country. Furthermore, our approach goes beyond a simple joint model as we model the total cases together with the distribution of case counts for each disease, conditioned on the total. This allows us to discuss how different covariates are associated with the relative risk of the total cases, as well as the odds of having chikungunya or Zika relative to dengue, which has been present for decades.
Increased risk of Aedes-borne diseases was estimated in regions historically burdened by dengue [6] (in valleys and south of the Andes), tourist locations (in the Caribbean coast and islands), and near international borders. These regions also showed strong common spatial effects in agreement with the consideration of shared measured and unmeasured characteristics associated with the presence and distribution beyond the shared vector. Some of these regions were previously identified as high-risk areas for dengue using a Climate Risk Factor analysis [29], as high-risk clusters for dengue, chikungunya, and Zika using Kulldorff's scan statistics [7, 30, 31], and with elevated burden for Aedes-borne diseases [32].
The higher probability of dengue presence compared to chikungunya and Zika in most municipalities is likely due to dengue’s endemicity in Colombia, with the four serotypes in co-circulation [33]. Conversely, chikungunya and Zika were emerging in the country. Importantly, Zika causes milder symptoms and is likely the most difficult Aedes-borne disease to detect by passive surveillance. Despite this, Zika was the one most likely present in the archipelago of San Andrés, Providencia y Santa Catalina, where thousands of tourists go annually, and the disease was potentially introduced in Colombia [7].
The temperature was identified as the main contributor to Aedes-borne diseases, which is supported by several other studies [11, 14, 34–37]. Warmer temperatures accelerate both the extrinsic incubation period of arboviruses and the Aedes life cycle, boosting transmission and increasing mosquitoes population [34]. Many Colombian municipalities located in mountains have been protected against arboviruses due to altitude, but rising temperatures from climate change may put them at risk.
We identified a positive association between NDVI and Aedes-borne diseases cases, likely driven by dengue, which was widespread in Colombia. Accordingly, more urbanised (i.e. lower NDVI) municipalities had a higher probability of chikungunya and/or Zika presence compared to dengue. These indicate the ruralisation of dengue in Colombia, similar to trends in other Latin American countries [13, 38–40], and the concentration of chikungunya and Zika first epidemics in more urbanised municipalities. Notably, we found that Zika had increased odds of presence than dengue and chikungunya in municipalities with lower temperatures. This contradicts prior research suggesting Zika virus has a higher thermal minimal for vector competence and extrinsic incubation rate than dengue virus [41]. However, the same research predicted that mosquitoes infected with Zika virus live longer than unexposed mosquitoes, especially at lower temperatures (24 °C versus 28 °C). Comparisons with dengue and chikungunya viruses were not available, therefore, more research is necessary to investigate if this represents an advantage for Zika transmission at cooler temperatures. On the other hand, our findings align with a previous study on the spatial variation of Zika epidemics in Latin America that found highest force of infection in lower temperatures (20–26 °C) than expected in laboratory experiments (29 °C) [42, 43].
Despite dengue's ruralisation, Aedes-borne diseases cases were still concentrated in large urban centres, where better socioeconomic indicators were often observed. This may explain the increased risk of Aedes-borne diseases in municipalities with a lower UBN and/or a higher percentage of homes with garbage collection. A similar positive association with garbage collection was found in Brazil [44]. The authors discussed this indicator does not necessarily mean proper solid waste disposal, and disposal in open pits could increase mosquito breeding sites.
Interestingly, we found that, among the Aedes-borne diseases, chikungunya had a higher probability of presence in more socially vulnerable locations. Similar findings were described in a nationwide study in Brazil [44]. Although this could result from disease spread from different entry sites, chikungunya may require more favourable transmission conditions, such as increased vector density and crowding, than dengue and Zika. This is supported by evidence that Ae. aegyptitransmits chikungunya at lower rates than other arboviruses [45, 46].
Municipalities with better potential access to healthcare had more Aedes-borne diseases cases, likely reflecting better reporting and detection rates in these areas. A tendency of a higher probability of Zika presence compared to dengue and chikungunya was observed in locations with better real access to healthcare, which includes good prenatal and gynaecological services. Zika infection during pregnancy had been recently described to cause congenital malformations [47], which probably increased awareness, and, in turn, the sensitivity of Zika detection among pregnant women of childbearing age. Also, nearly 90% of pregnant women attended at least four prenatal visits in Colombia in 2016 [48], improving their access to the health system and infection detection compared to other groups.
Differences in the spatial distribution of disease presence probabilities could not be fully explained by the covariates. This advocates for local-specific interventions and preparing the healthcare system considering the epidemiological scenario and other local contexts. Strategies in tourist and border locations should aim at preventing the introduction of viruses, with active surveillance sensitive to detect asymptomatic and mild cases. Nevertheless, on the border with Ecuador and Venezuela, there is an important context of social vulnerability that such strategies must also alleviate [49]. Collaboration with neighbouring countries in data sharing and strategy alignment is crucial, especially in twin cities like Letícia (Colombia) and Tabatinga (Brazil).
This study has limitations. Surveillance data are prone to underreporting, particularly in locations with limited access to healthcare [50]. However, the Colombian surveillance system is robust, with mandatory reporting and national coverage. Also, given the quality of the Aedes-borne diseases surveillance system, its data is used for public health decision-making [33]. To account for the potential differential reporting given access to healthcare, we included two indicators of healthcare access in our models. Surveillance data can be affected by misclassification of cases, and the complexity of syndemicity further complicates accurate classification [50]. In our study, 29.5% of cases were confirmed by laboratory diagnostics and 35.0% by clinical diagnosis and epidemiological criteria. The remainder received a suspected diagnosis, without confirmation. Nonetheless, the surveillance system has a higher sensitivity in the identification of cases, for which the analysis of the total number of Aedes-borne diseases relative risk is desirable and granted.
In conclusion, we present, in an unprecedented way, a nationwide robust and in-depth study of the syndemics of dengue, chikungunya, and Zika in Colombia using a multivariate model that allows comparative, yet simultaneous, analysis of disease distribution. This Poisson-multinomial spatial model can be applied in other Aedes-endemic countries, at other scales, and for other diseases causing simultaneous outbreaks. As for the generalization of the results, they are specific to Colombia, but similar locations with comparable conditions may yield similar findings. While dengue continues to be a major public health concern in Colombia, chikungunya and Zika did not cause further epidemics after the study period (Supplementary Figure S1). However, a small number of cases continue to be reported, indicating that these diseases are still circulating. If re-emergence occurs in Colombia, our results may be further useful, as it is likely that the distribution and contributing factors of these diseases will be similar to those observed during emergence.
Supplementary Information
Acknowledgements
The authors would like to thank the Instituto Nacional de Salud of Colombia for making the diseases’ surveillance data publicly available, the Observatorio Nacional de Salud for sharing the healthcare access indexes data, and Gina Alexandra Vargas Sandoval, Carlos Andres Castañeda-Orjuela and Karol Patricia Cotes Cantillo, for their help in the interpretation of the indexes.
Authors’ contributions
L.P.F., K.Z., A.M.S., M.C., G.I.J.R., C.G.B., B.N.R. conceptualized research and interpreted data. L.P.F. and A.M.S. designed research and analyzed data. L.P.F. and M.C. wrote the paper. A.M.S., J.E.S.F.,B.A.M., C.G.B., B.N.R., G.I.J.R., and K.Z. reviewed and approved the paper.
Funding
This work was supported by a grant from the Canadian Institutes of Health Research [grant number 428107]. Mabel Carabali holds a Fonds de recherche du Québec - Santé (FRQS) Chercheur Boursier Junior 1 [329874]. Alexandra M. Schmidt acknowledges financial support from the Natural Sciences and Engineering Research Council (NSERC) of Canada (Discovery Grants) [RGPIN-2024-04312]. Gloria I. Jaramillo-Ramirez acknowledges financial support from Universidad Cooperativa de Colombia (INV3065). Kate Zinszer holds a FRQS Chercheur Boursier Junior 2 [310728]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Data availability
The data used in this study are secondary data and are publicly available. Cases data are available at the Colombian National Public Health Surveillance System (Sistema Nacional de Vigilancia en Salud Pública - SIVIGILA) website (http://portalsivigila.ins.gov.co/). Environmental data was organised and made available by Siraj et al. (2019) (10.5061/dryad.83nj1). Population and socioeconomic data are available at the National Administrative Department of Statistics of Colombia (Departamento Administrativo Nacional de Estadística - DANE) website (https://www.dane.gov.co/). A version of the healthcare access indexes can be found at the Colombian National Health Observatory (Observatorio Nacional de Salud - ONS) website (https://www.ins.gov.co/Direcciones/ONS/). Codes for the model are available at https://github.com/laispfreitas/joint_DZC_model.
Declarations
Ethics approval and consent to participate
This study was approved by the Science and Health Research Ethics Committee (Comité d’éthique de la recherche en sciences et en santé - CERSES) of the University of Montreal, approval number CERSES-19-018-D.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.San Martín JL, Brathwaite Dick O, Del Diego J, Montoya RH, Dayan GH, Zambrano B. The History of Dengue Outbreaks in the Americas. The American Journal of Tropical Medicine and Hygiene. 2012;87:584–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.WHO. Dengue and severe dengue. 2024; published online April 23. https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue (accessed June 6, 2024).
- 3.Santos LLM, De Aquino EC, Fernandes SM, Ternes YMF, Feres VCDR. Dengue, chikungunya, and Zika virus infections in Latin America and the Caribbean: a systematic review. Revista Panamericana de Salud Pública. 2023;47:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Farias PCS, Pastor AF, Gonçales JP, et al. Epidemiological profile of arboviruses in two different scenarios: dengue circulation vs. dengue, chikungunya and Zika co-circulation. BMC Infect Dis. 2023;23:177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.De Souza WM, Ribeiro GS, De Lima STS, et al. Chikungunya: a decade of burden in the Americas. The Lancet Regional Health - Americas. 2024;30:100673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Padilla JC, Rojas DP, Sáenz Gómez R. Dengue en Colombia: epidemiología de la reemergencia a la hiperendemia, 1st edn. Bogotá, D.C., Colombia, 2012.
- 7.Freitas LP, Carabali M, Yuan M, et al. Spatio-temporal clusters and patterns of spread of dengue, chikungunya, and Zika in Colombia. PLoS Negl Trop Dis. 2022;16:e0010334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Instituto Nacional de Salud de Colombia. Reporte: Datos históricos 2007-2023. Portal Sivigila. https://portalsivigila.ins.gov.co/Paginas/datos.aspx?cod=154 (accessed March 4, 2025).
- 9.Lowe R, Gasparrini A, Van Meerbeeck CJ, et al. Nonlinear and delayed impacts of climate on dengue risk in Barbados: A modelling study. PLOS Medicine. 2018;15:e1002613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gibb R, Colón-González FJ, Lan PT, et al. Interactions between climate change, urban infrastructure and mobility are driving dengue emergence in Vietnam. Nat Commun. 2023;14:8179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Freitas LP, Schmidt AM, Cossich W, Cruz OG, Carvalho MS. Spatio-temporal modelling of the first Chikungunya epidemic in an intra-urban setting: The role of socioeconomic status, environment and temperature. PLoS Negl Trop Dis. 2021;15:e0009537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.López MS, Gómez AA, Müller GV, Walker E, Robert MA, Estallo EL. Relationship between Climate Variables and Dengue Incidence in Argentina. Environ Health Perspect. 2023;131:057008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Codeço CT, Oliveira SS, Ferreira DAC, et al. Fast expansion of dengue in Brazil. The Lancet Regional Health - Americas. 2022;12:100274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Picinini Freitas LP, Douwes-Schultz D, Schmidt AM, et al. Zika emergence, persistence, and transmission rate in Colombia: a nationwide application of a space-time Markov switching model. Sci Rep. 2024;14:10003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barcellos C, Matos V, Lana RM, Lowe R. Climate change, thermal anomalies, and the recent progression of dengue in Brazil. Sci Rep. 2024;14:5948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dalvi APR, Gibson G, Ramos AN, et al. Sociodemographic and environmental factors associated with dengue, Zika, and chikungunya among adolescents from two Brazilian capitals. PLoS Negl Trop Dis. 2023;17: e0011197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Carabali M, Harper S, Lima‐Neto AS, et al. Spatiotemporal Distribution and Socioeconomic Disparities of Dengue, Chikungunya and Zika in Two Latin American cities from 2007 to 2017. Tropical Medicine & International Health 2020; published online Nov 20. 10.1111/tmi.13530. [DOI] [PubMed]
- 18.Romeo-Aznar V, Picinini Freitas L, Gonçalves Cruz O, King AA, Pascual M. Fine-scale heterogeneity in population density predicts wave dynamics in dengue epidemics. Nat Commun. 2022;13:996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schmidt AM, Freitas LP, Cruz OG, Carvalho MS. A Poisson-multinomial spatial model for simultaneous outbreaks with application to arboviral diseases. Stat Methods Med Res. 2022;31:1590–602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zambrano P, Romero SEG, Cruz JAC, Instituto Nacional de Salud, Colombia. Protocolo de vigilancia en salud pública – Dengue. 2017. https://www.ins.gov.co/BibliotecaDigital/PRO-Dengue.pdf.
- 21.Botero DS, Farías AP, Instituto Nacional de Salud, Colombia. Protocolo de vigilancia en salud pública – Chikungunya. 2017. https://www.ins.gov.co/BibliotecaDigital/PRO-Chikungu%C3%B1a.pdf.
- 22.Pérez NT, Farías AP, Instituto Nacional de Salud, Colombia. Protocolo de vigilancia en salud pública – Enfermedad por Virus Zika. 2017. https://www.ins.gov.co/BibliotecaDigital/PRO-Zika.pdf.
- 23.Siraj AS, Rodriguez-Barraquer I, Barker CM, et al. Spatiotemporal incidence of Zika and associated environmental drivers for the 2015–2016 epidemic in Colombia. Scientific Data. 2018;5:180073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Siraj AS, Rodriguez-Barraquer I, Barker CM, et al. Data from: Spatiotemporal incidence of Zika and associated environmental drivers for the 2015-2016 epidemic in Colombia. 2019;1785323094 bytes. https://datadryad.org/dataset/doi:10.5061/dryad.83nj1. Accessed 3 April 2024. [DOI] [PMC free article] [PubMed]
- 25.Feres JC, Mancero X. El método de las necesidades básicas insatisfechas (NBI) y sus aplicaciones en América Latina. Santiago de Chile: Naciones Unidas, CEPAL, Div. de Estadística y Proyecciones Económicas, 2001.
- 26.Instituto Nacional de Salud de Colombia, Observatorio Nacional de Salud. Acceso a servicios de salud en Colombia. Informe técnico 11. Bogotá, D.C., Colombia, 2019 https://www.ins.gov.co/Direcciones/ONS/Informes/11.%20Acceso%20a%20servicios%20de%20salud%20en%20Colombia.pdf (accessed April 3, 2024).
- 27.Banerjee S, Carlin BP, Gelfand AE. Hierarchical modeling and analysis for spatial data, 2. ed. Boca Raton, Fla.: CRC Press, 2015.
- 28.Stan Development Team. Stan Reference Manual, Version 2.23. https://mc-stan.org/docs/2_23/reference-manual/notation-for-samples-chains-and-draws.html (accessed April 29, 2020).
- 29.Lee J-S, Carabali M, Lim JK, et al. Early warning signal for dengue outbreaks and identification of high risk areas for dengue fever in Colombia using climate and non-climate datasets. BMC Infect Dis. 2017;17:480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Desjardins MR, Whiteman A, Casas I, Delmelle E. Space-time clusters and co-occurrence of chikungunya and dengue fever in Colombia from 2015 to 2016. Acta Tropica. 2018;185:77–85. [DOI] [PubMed] [Google Scholar]
- 31.Triana-Vidal LE, Morales-García MA, Arango-Cárdenas MJ, Badiel-Ocampo M, Cuartas DE. Análisis de la distribución espacial y temporal de los virus del Dengue (2006–2017), Zika (2015–2017) y Chikungunya (2014–2017) en Colombia. Infect. 2019;23:352. [Google Scholar]
- 32.Mora-Salamanca AF, Porras-Ramírez A, De la Hoz Restrepo FP. Estimating the burden of arboviral diseases in Colombia between 2013 and 2016. IJID. 2020;97:81–9. [DOI] [PubMed] [Google Scholar]
- 33.Instituto Nacional de Salud de Colombia. Boletín Epidemiológico Semanal. Semana Epidemiológica 12, 2024. 2024. https://www.ins.gov.co/buscador-eventos/BoletinEpidemiologico/2024_Bolet%C3%ADn_epidemiologico_semana_12.pdf (accessed June 12, 2024).
- 34.Delrieu M, Martinet J-P, O’Connor O, et al. Temperature and transmission of chikungunya, dengue, and Zika viruses: A systematic review of experimental studies on Aedes aegypti and Aedes albopictus. Curr Res Parasitol Vector-Borne Dis. 2023;4:100139. [DOI] [PMC free article] [PubMed]
- 35.Morgan J, Strode C, Salcedo-Sora JE. Climatic and socio-economic factors supporting the co-circulation of dengue, Zika and chikungunya in three different ecosystems in Colombia. PLoS Negl Trop Dis. 2021;15:e0009259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dong B, Khan L, Smith M, et al. Spatio-temporal dynamics of three diseases caused by Aedes-borne arboviruses in Mexico. Commun Med. 2022;2:134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Muñoz E, Poveda G, Arbeláez MP, Vélez ID. Spatiotemporal dynamics of dengue in Colombia in relation to the combined effects of local climate and ENSO. Acta Tropica. 2021;224:106136. [DOI] [PubMed] [Google Scholar]
- 38.Katzelnick LC, Quentin E, Colston S, et al. Increasing transmission of dengue virus across ecologically diverse regions of Ecuador and associated risk factors. PLoS Negl Trop Dis. 2024;18:e0011408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lee SA, Economou T, De Castro Catão R, Barcellos C, Lowe R. The impact of climate suitability, urbanisation, and connectivity on the expansion of dengue in 21st century Brazil. PLoS Negl Trop Dis. 2021;15:e0009773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lana RM, Gomes MFDC, Lima TFMD, Honório NA, Codeço CT. The introduction of dengue follows transportation infrastructure changes in the state of Acre, Brazil: A network-based analysis. PLoS Negl Trop Dis. 2017;11:e0006070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tesla B, Demakovsky LR, Mordecai EA, et al. Temperature drives Zika virus transmission: evidence from empirical and mathematical models. Proc R Soc B. 2018;285:20180795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Harris M, Caldwell JM, Mordecai EA. Climate drives spatial variation in Zika epidemics in Latin America. Proc R Soc B. 2019;286:20191578. [DOI] [PMC free article] [PubMed]
- 43.Mordecai EA, Cohen JM, Evans MV, et al. Detecting the impact of temperature on transmission of Zika, dengue, and chikungunya using mechanistic models. PLOS Neglected Tropical Diseases. 2017;11:e0005568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gardini Sanches Palasio R, Marques Moralejo Bermudi P, Luiz De Lima Macedo F, Reis Santana LM, Chiaravalloti-Neto F. Zika, chikungunya and co-occurrence in Brazil: space-time clusters and associated environmental–socioeconomic factors. Sci Rep 2023; 13: 18026. [DOI] [PMC free article] [PubMed]
- 45.Göertz GP, Vogels CBF, Geertsema C, Koenraadt CJM, Pijlman GP. Mosquito co-infection with Zika and chikungunya virus allows simultaneous transmission without affecting vector competence of Aedes aegypti. PLoS Negl Trop Dis. 2017;11:e0005654. [DOI] [PMC free article] [PubMed]
- 46.Crespo M, Guedes D, Paiva M, et al. Exposure to Zika and chikungunya viruses impacts aspects of the vectorial capacity of Aedes aegypti and Culex quinquefasciatus. PLoS ONE. 2024;19:e0281851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.De Araújo TVB, Rodrigues LC, De Alencar Ximenes RA, et al. Association between Zika virus infection and microcephaly in Brazil, January to May, 2016: preliminary report of a case-control study. The Lancet Infectious Diseases. 2016;16:1356–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.UNICEF. UNICEF Data Warehouse. https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=COL.MNCH_ANC4.&startPeriod=1970&endPeriod=2024 (accessed June 12, 2024).
- 49.Grillet ME, Hernández-Villena JV, Llewellyn MS, et al. Venezuela’s humanitarian crisis, resurgence of vector-borne diseases, and implications for spillover in the region. The Lancet Infectious Diseases. 2019;19:e149-61. [DOI] [PubMed] [Google Scholar]
- 50.Carabali M, Jaramillo-Ramirez GI, Rivera VA, Mina Possu N-J, Restrepo BN, Zinszer K. Assessing the reporting of Dengue, Chikungunya and Zika to the National Surveillance System in Colombia from 2014–2017: A Capture-recapture analysis accounting for misclassification of arboviral diagnostics. PLoS Negl Trop Dis. 2021;15:e0009014. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data used in this study are secondary data and are publicly available. Cases data are available at the Colombian National Public Health Surveillance System (Sistema Nacional de Vigilancia en Salud Pública - SIVIGILA) website (http://portalsivigila.ins.gov.co/). Environmental data was organised and made available by Siraj et al. (2019) (10.5061/dryad.83nj1). Population and socioeconomic data are available at the National Administrative Department of Statistics of Colombia (Departamento Administrativo Nacional de Estadística - DANE) website (https://www.dane.gov.co/). A version of the healthcare access indexes can be found at the Colombian National Health Observatory (Observatorio Nacional de Salud - ONS) website (https://www.ins.gov.co/Direcciones/ONS/). Codes for the model are available at https://github.com/laispfreitas/joint_DZC_model.