Abstract
This case-control study of 124 early-pregnant women found that daily supplementation with 400 IU of vitamin D3 significantly increased serum 1,25-(OH)D3 levels and was associated with a lower incidence of threatened miscarriage. The study suggests that vitamin D3 supplementation could reduce the risk of early miscarriage and improve pregnancy outcomes by modulating immune responses and hormonal stability.
Keywords: vitamin D3 supplementation; early pregnancy loss; serum 1,25-(OH)D3; miscarriage prevention; immune modulation
Introduction
Early pregnancy miscarriage refers to the natural death of an embryo within 12 weeks of gestation, representing a significant factor affecting women’s reproductive health. Globally, an estimated 15% to 20% of confirmed pregnancies end in miscarriage, a Fig. likely underestimated due to many early miscarriages occurring before women are aware of their pregnancy.(1–3) Miscarriage not only inflicts physical harm but may also have profound effects on women’s psychological well-being, including but not limited to depression, anxiety, and emotional instability.(4–6) Furthermore, recurrent miscarriages may indicate more severe health issues, such as hormonal imbalances, immune system problems, or anatomical abnormalities.(7,8) These issues underscore early pregnancy miscarriage as a key concern in reproductive medicine and public health research.(9) Research into preventive and management strategies for early pregnancy miscarriage holds crucial clinical significance, aiming to reduce its occurrence, enhance women’s quality of life, and alleviate the burden on public health systems.
Vitamin D is a pivotal nutrient for maintaining human health, especially during pregnancy.(10) It serves as a crucial factor in calcium absorption, bone health maintenance, and plays a significant role in regulating the body’s immune response and cell proliferation.(11–13) Studies suggest that vitamin D deficiency is associated with various health issues, including osteoporosis, cardiovascular diseases, certain cancers, and autoimmune diseases.(14–16) Adequate vitamin D levels during pregnancy are particularly critical for the health of both pregnant women and fetuses.(17–19) Vitamin D not only promotes normal fetal bone development but is also linked to regulating the mother’s immune system, potentially playing a beneficial role in preventing pregnancy complications such as gestational diabetes and placental abruption.(20–22) Furthermore, vitamin D’s anti-inflammatory properties may play a key role in maintaining the stability of the internal environment during pregnancy, potentially lowering the risk of early pregnancy miscarriage.(23–25)
The association between vitamin D levels and the risk of early miscarriage has been extensively investigated in multiple studies.(26,27) Some epidemiological surveys have shown a significant increase in the risk of early miscarriage among pregnant women with lower than standard levels of serum vitamin D.(28–30) For instance, several studies have indicated a higher rate of early miscarriage in women with inadequate vitamin D during early pregnancy.(26,27,31) While these studies offer valuable insights, they are constrained by limitations such as small sample sizes, restricted study designs, and insufficient control over potential confounding factors.(32–34) Furthermore, the specific mechanisms through which vitamin D levels may indirectly affect the risk of miscarriage by influencing immune function, hormone levels, or other biomarkers are not entirely clear.(35–37) Therefore, more systematic and comprehensive research is imperative, utilizing precise measurement techniques, larger sample sizes, and stringent study designs to accurately delineate the relationship between vitamin D levels and the risk of early miscarriage and to explore the underlying biological mechanisms.(38–40)
This study aims to investigate the impact of vitamin D3 supplementation on the serum concentration of 1,25-(OH)D3 and the risk of early miscarriage in early pregnant women. By establishing an experimental group and a control group receiving 400 IU of vitamin D3 supplementation and sunlight advice, respectively, this study monitored the levels of 1,25-(OH)D3 and pregnancy hormones in serum using chemiluminescence immunoassay and liquid chromatography-tandem mass spectrometry techniques, and detected serum cytokines through enzyme-linked immunosorbent assay (ELISA) methods. Additionally, an extensive literature search and meta-analysis were conducted to comprehensively assess the relationship between vitamin D3 levels and the risk of early miscarriage. The results of this study will help verify whether vitamin D3 supplementation can effectively increase serum vitamin D3 levels, thus reducing the risk of early miscarriage. Through scientifically rigorous research methods and systematic data analysis, this study aims to provide evidence-based support for the clinical management of early pregnant women, suggesting monitoring and adjustment of vitamin D levels during pregnancy to enhance stability and safety during this period. This approach not only optimizes maternal health management but also offers scientific grounds for public health policy-making, promoting improvements and advancements in maternal healthcare services.
Materials and Methods
Study participants
This study employed a case-control design, selecting early pregnant patients with fertility requirements who sought treatment at our hospital from January 2022 to September 2023 and completed follow-up as the research subjects. Exclusion criteria were as follows: (1) initial visit occurring more than 50 days after conception; (2) presenting with symptoms of miscarriage at the time of visit, such as abdominal pain, vaginal bleeding, etc.; (3) severe vitamin D deficiency [1,25-(OH)D3 <20 ng/ml, with such patients being advised to supplement vitamin D3]; (4) patients with a history of other internal or surgical diseases, such as hypertension, diabetes, heart disease, etc.; (5) known comorbidities involving reproductive tract abnormalities, infections, chromosomal factors, or autoimmune diseases; (6) history of medication, exposure to radiation, smoking, alcohol abuse, and other unhealthy lifestyle factors; (7) diagnosed with ectopic pregnancy. Any individual meeting any of the aforementioned exclusion criteria was not included in the study. The research adhered to medical ethics standards and obtained approval from the hospital’s ethics committee, and all research subjects provided informed consent. The study subjects were randomly divided into an experimental group of 65 cases and a control group of 59 cases. There were no statistical differences in age, height, weight, season of the initial visit, or duration of amenorrhea between the two groups (Table 1). Refer to Fig. 1 for the specific workflow.
Table 1.
Comparison of baseline data between two groups
| Control group | Experimental group | Statistical value | p | |
|---|---|---|---|---|
| Age | 25 (24, 28) | 25 (23, 28) | Z = −0.324 | 0.746 |
| Season | χ2 = 0.244 | 0.97 | ||
| Spring | 17 (28.8%) | 17 (26.2%) | ||
| Summer | 14 (23.7%) | 17 (26.2%) | ||
| Autumn | 11 (18.6%) | 11 (16.9%) | ||
| Winter | 17 (28.8%) | 20 (30.8%) | ||
| High | 160 (157, 163) | 160 (159, 163) | Z = −0.995 | 0.32 |
| Weight | 54 (51, 56) | 55 (52, 57) | Z = −1.246 | 0.213 |
| First visit (Number of days of menstrual cessation) |
34 (32, 35) | 34 (32, 35) | Z = −0.521 | 0.602 |
Fig. 1.
Inclusion and exclusion process flowchart.
Intervention methods
The experimental group received oral Diqiao vitamin D3 capsules at a dosage of 400 IU/day, along with recommendations to increase sunlight exposure and consume vitamin D3-rich foods. The control group, on the other hand, received only suggestions to increase sunlight exposure and consume foods high in vitamin D3. Both groups were followed up for 12 weeks. In the event of threatened miscarriage, conventional miscarriage preventive treatment was administered. In cases of inevitable miscarriage, follow-up was discontinued. At the 12th week of pregnancy, the incidences of threatened and inevitable miscarriages were recorded for both groups.
Chemiluminescence assay for detection
In this study, chemiluminescence assay was employed to measure human chorionic gonadotropin (HCG) and p values in the control and experimental groups at the initial visit and on days 7, 14, and 21 post-intervention. Blood samples were collected from patients in both groups and centrifuged to obtain serum, which was then cleared of cellular components to yield a clear supernatant suitable for further analysis. The serum samples were incubated with specific antibodies against HCG or progesterone (P), which were labeled with chemiluminescent markers like luminol or acridinium esters to enable luminescence under specific reaction conditions. An appropriate oxidizing agent or substrate, such as hydrogen peroxide, was added to trigger the luminescent reaction, involving an oxidation-reduction process of the labeled markers transitioning from ground state to excited state and releasing photons upon returning to ground state. The emitted photons were captured by sensitive photomultiplier tubes and converted into electrical signals. The intensity of these signals was directly proportional to the levels of HCG and P in the serum. A standard curve was constructed using known concentrations of standard samples, and the concentration of HCG and P in the samples was calculated by comparing the luminescence intensity of the samples with that of the standards.
Liquid chromatography tandem mass spectrometry technique
The serum concentration of 1,25-dihydroxy vitamin D [1,25-(OH)D3] was measured using liquid chromatography tandem mass spectrometry in the control and experimental groups at the initial visit, and on the 7th, 14th, and 21st days post-intervention. Blood samples were collected from both patient groups, and serum was obtained through centrifugation. Sample pretreatment procedures such as protein precipitation, liquid-liquid extraction, or solid-phase extraction were carried out on the serum samples. The pretreated samples were injected into the liquid chromatography system where compounds in the samples were separated by the chromatographic column based on their polarity, size, and charge, distributing between the stationary and mobile phases to achieve separation of different components. Commonly used chromatographic columns in liquid chromatography include C18 columns, while the mobile phase typically consists of an aqueous phase and an organic phase, separated through gradient elution or isocratic elution. The separated compounds were then analyzed using a mass spectrometer. Mass spectrometry analysis employed electrospray ionization (ESI) as the ion source to ionize compounds before introduction into the mass spectrometer. Multiple reaction monitoring (MRM) was utilized in the mass spectrometer to detect specific ion pairs, including precursor and quantifier ions, for quantitative analysis of 1,25-(OH)D. The serum concentration of 1,25-(OH)D was calculated based on the chromatographic peak areas obtained from mass spectrometry analysis, in combination with internal standard correction and a standard curve. The standard curve was established by analyzing a series of known concentrations of standard solutions to quantify the content of 1,25-(OH)D in unknown samples.
ELISA
The concentrations of tumor necrosis factor-α (TNF-α), interleukin (IL)-2, IL-4, and IL-10 in the serum of the control and experimental groups were measured using the ELISA method at the initial visit, on the 7th day post-intervention, 14th day, and 21st day. Blood samples were collected from patients in both groups, and serum was obtained by centrifugation. ELISA kits from Bioss Antibodies (such as Human TNF-α ELISA Kit, Human IL-2 ELISA Kit, Human IL-4 ELISA Kit, and Human IL-10 ELISA Kit) were utilized for serum analysis. Each kit typically included a pre-coated microplate with specific antibodies, standard samples, detection antibodies, enzyme-labeled secondary antibodies, substrate solution, and stop solution. Following the kit instructions, standard samples were diluted to various concentrations and added to the microplate to establish a standard curve. Serum samples were appropriately diluted (if necessary) and added to the microplate. The microplate was then incubated at 37°C for 1–2 h in a constant temperature chamber to allow for antibody binding to cytokines in the samples. Unbound antibodies and other non-specific binding substances were removed by multiple washes with a washing solution. Enzyme-labeled secondary antibodies specific to the target cytokines were added, followed by another incubation. Substrate solution was added, and the enzyme-labeled antibodies catalyzed a color change in the substrate. The reaction time was adjusted according to the kit instructions. Stop solution, typically an acidic solution, was added to halt the enzyme catalysis. The optical density (OD value) of each well was measured using an ELISA reader at a specific wavelength (usually 450 nm), and the concentrations of cytokines in the samples were calculated by comparing them to the standard curve. The concentrations of TNF-α, IL-2, IL-4, and IL-10 in the serum were determined based on the standard curve and the sample’s OD values, followed by relevant biological or clinical analysis.
Meta-analysis literature search strategy
In order to comprehensively assess the impact of vitamin D3 on the risk of early miscarriage in a meta-analysis, a detailed literature search strategy was devised for this study. Initially, a combination of keywords such as “Vitamin D3”, “miscarriage”, “early pregnancy loss”, and “immune modulation” were utilized, and the search criteria was optimized using Boolean operators (AND, OR, NOT). The search was conducted across major medical and life science databases including PubMed, Web of Science, and China National Knowledge Infrastructure (CNKI), encompassing all relevant literature from the establishment of databases up to April 2024. The preliminary screening of all literature was based on the relevance of titles and abstracts, followed by full-text reading and quality assessment of the documents that meet the criteria. This strategy aims to capture and analyze all relevant studies, providing scientifically sound evidence to support the role of vitamin D3 in preventing early miscarriages.
Inclusion and exclusion criteria
For the systematic review and meta-analysis conducted in this study, our focus lies on evaluating the association between vitamin D3 levels and early miscarriage. The aim is to include observational and intervention studies that compare the levels of vitamin D3 between patients experiencing early miscarriage and healthy pregnant control groups.
Initially, we encompassed studies specifically reporting on the levels of vitamin D3 in the serum of pregnant women in both miscarriage and control groups. This includes randomized controlled trials (RCTs), prospective cohort studies, and retrospective cohort studies. All studies included must have a clear definition of miscarriage (such as pregnancies naturally ending before 20 weeks) and a precise method for measuring vitamin D3 levels. We particularly emphasize studies that have appropriately adjusted for sample sizes and baseline characteristics (such as age, ethnicity, pre-pregnancy weight, etc.).
Concerning exclusion criteria, we excluded the following types of studies: those involving underage females, studies that do not provide a comparison of vitamin D3 levels between miscarriage and control groups, studies with significant design flaws (such as noticeable selection bias, information bias, etc.), studies with extremely small sample sizes, and studies not using reasonable statistical methods for data analysis. Additionally, review articles, conference abstracts, case reports, expert opinions, and other non-original research also was excluded.
Moreover, studies that have been previously analyzed in other reviews or meta-analyses underwent specific scrutiny to avoid analysis biases resulting from duplicating data use. These inclusion and exclusion criteria aim to ensure the accuracy and scientific validity of this meta-analysis, ensuring that the collected data through a precise literature screening process can effectively support the assessment of the relationship between vitamin D3 levels and early miscarriage. Such criteria assists in identifying high-quality, reliable studies from a vast pool of relevant literature, thereby providing a solid scientific foundation for further analysis.
Document coding and quality assessment
Each selected document was meticulously coded, including basic information such as authorship, publication year, and sample size. To meet the specific requirements of this study, we also coded key data related to Vitamin D3 levels and early miscarriage, such as participants’ age, race, pre-pregnancy weight, gestational age, and specific measurement values of Vitamin D3 in the serum. All data was systematically organized using Excel to ensure accurate data entry and facilitate subsequent analysis.
In assessing the quality of the literature, we adhere to the standards of the Cochrane Risk of Bias Tool. This involves evaluating random sequence generation, allocation concealment, blinding, completeness of outcome data, selective reporting, and other potential biases. This evaluation process assists us in identifying and eliminating factors that may affect the reliability of the study results.
Literature data extraction
During the data extraction phase, we focus on the methods of measuring vitamin D3 levels reported in each study and their correlation with early miscarriage. Specifically, we extract specific values of serum vitamin D3, time points of measurement, detailed information on miscarriage occurrences, as well as other relevant physiological and clinical data. Additionally, we document sample sizes of each study, experimental and control group settings, and key statistical data such as means, SD, p values, and confidence intervals (CIs).
These data extraction tasks are crucial for subsequent statistical analysis, as they not only help us accurately compare and analyze the relationship between vitamin D3 levels and early miscarriage, but also enable us to evaluate the scientific validity and statistical significance of the study results. Through this systematic and thorough approach, our aim is to ensure that the results of the meta-analysis are highly accurate and reliable, providing a solid scientific basis for exploring the potential role of vitamin D3 in preventing early miscarriages.
Statistical methods
Data management and statistical analysis were conducted using IBM SPSS Statistics 26. Qualitative data were described using n (%) percentages. For quantitative data following a normal distribution, means and SDs were reported, while categorical data were analyzed using the chi-square test. Group comparisons were performed using t tests and analysis of variance for normally distributed quantitative data, while non-normally distributed quantitative data were described using median and interquartile range (P25, P75) and compared using the Mann-Whitney U test. When comparing variables with repeated measurements exceeding two time points, the within-subject correlation was considered, and generalized estimating equations and mixed effects models were employed for modeling, with residual plots used for model diagnostics. The significance level was set at α = 0.05 unless otherwise specified. In this study, individual measurements at each time point were set as level 1 and individuals as level 2. The null model estimations revealed statistically significant individual covariance parameters for serum P, 1,25-(OH)D3, TNF-α, IL-4, IL-10, and IL-2 (p<0.001), with corresponding intraclass correlation coefficients (ICCs) of 63.61%, 62.00%, 81.12%, 60.19%, 33.84%, and 59.09%, respectively. This indicates that a significant proportion of variability in these markers comes from repeated measurements within the same individual at different time points, demonstrating a two-level structure in the data. However, individual covariance parameter estimates for the HCG marker did not reach statistical significance and were consequently analyzed using generalized estimating equations.
The meta-analysis in this study was conducted using RevMan 5.4 and R software, with r values and their 95% CIs selected as measures of effect size. To assess heterogeneity among included studies, Cochran’s Q test and I2 statistics were utilized. Sensitivity analysis was performed to evaluate the stability of study results, and funnel plots were used to assess the potential for publication bias. The significance level for all statistical tests was set at p<0.05.
Results
Dynamic changes in serum 1,25-(OH)D3 concentration under treatment intervention and analysis of mixed effects model
Vitamin D plays a crucial role in calcium metabolism and bone health. The variation in serum 1,25-(OH)D3 concentration, as the primary bioactive form of vitamin D, holds significant importance for human health.(41) Analyzing the impact of different treatment regimens on serum 1,25-(OH)D3 levels in clinical studies can provide a scientific basis for the treatment of related diseases. However, there is currently insufficient research on the dynamic changes in serum 1,25-(OH)D3 concentrations between the experimental and control groups during treatment. This study aims to investigate the trends in serum 1,25-(OH)D3 concentrations between the experimental and control groups in consecutive observations after the initial visit and further analyze their differences using a mixed effects model.
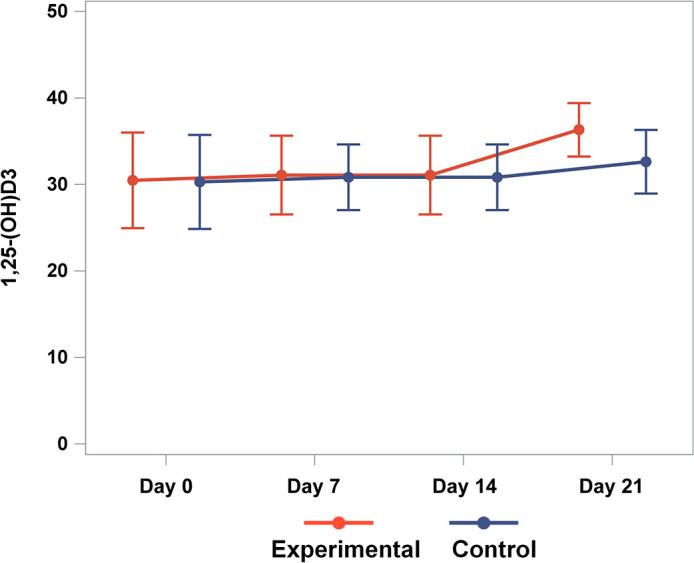
Both the experimental and control groups exhibited a stable trend in serum 1,25-(OH)D3 concentrations post initial visit, remaining steady until the 7th day. However, by the 14th day, both groups showed an increasing trend, with the experimental group demonstrating a greater increase compared to the control group (Fig. 2). Analysis using the mixed effects model revealed a significant increase in serum 1,25-(OH)D3 levels at the 7th, 14th, and 21st days compared to the initial visit, with a statistically significant difference between the experimental and control groups (Table 2 and Fig. 3).
Fig. 2.
Dynamic changes in serum 1,25-(OH)D3 concentration in patients from the experimental and control groups. Represents the time points on the x-axis, and the serum 1,25-(OH)D3 concentration (ng/ml) on the y-axis. Statistical significance: There was no statistically significant difference between the two groups at the initial visit and on the 7th day (p>0.05). However, there was a statistically significant difference between the two groups on the 14th and 21st days (p<0.05).
Table 2.
Mixed-effects model estimates for serum 1,25-(OH)D3 concentration with respect to time and group
| Variable | Group | Estimate | SE | t | p |
|---|---|---|---|---|---|
| Intercept | 28.31 | 0.559 | 50.632 | <0.001 | |
| Time | First visit | 0 | |||
| Day 7 | 0.577 | 0.232 | 2.49 | 0.013 | |
| Day 14 | 2.619 | 0.28 | 9.367 | <0.001 | |
| Day 21 | 4.18 | 0.345 | 12.108 | <0.001 | |
| Group | Control group | 0 | |||
| Experimental group | 3.976 | 0.558 | 7.123 | <0.001 |
Variable, The variables being examined in the analysis. Group, the different categories or groups within each variable. Estimate, the estimated effect size or coefficient for each group or variable. t, the t value, which is the ratio of the estimate to its SE. p, indicating the statistical significance of the estimate.
Fig. 3.
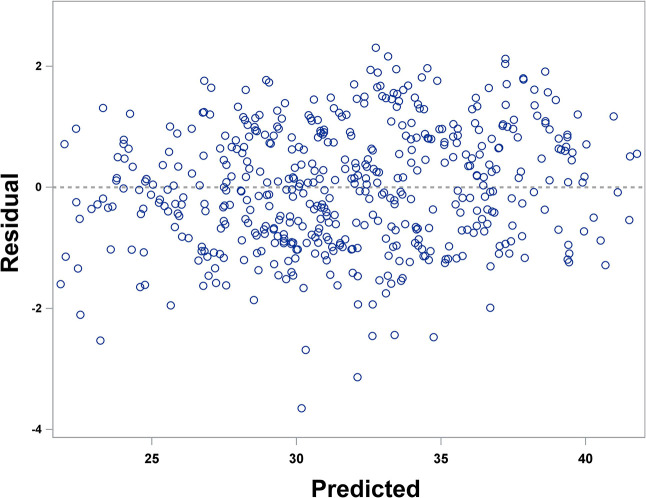
Residual scatter plot for the model diagnosis of serum 1,25-(OH)D3 concentration.
In conclusion, the results of this study indicate that the experimental group exhibited a significantly higher rate of increase in serum 1,25-(OH)D3 concentration during the treatment process than the control group, which may be attributed to differences in treatment regimens or the mechanism of action of the drugs. Therefore, monitoring and adjusting the dynamic changes in serum 1,25-(OH)D3 levels during the treatment of related diseases could potentially help optimize treatment outcomes and improve the quality of life for patients.
Analysis of dynamic changes and mixed effects model of serum HCG and P concentrations
HCG plays a crucial role in the early diagnosis and monitoring of pregnancy. This study investigates the changing trends of serum HCG concentrations in the experimental and control groups during the follow-up period and further analyzes them using generalized estimating equations and mixed effects models.
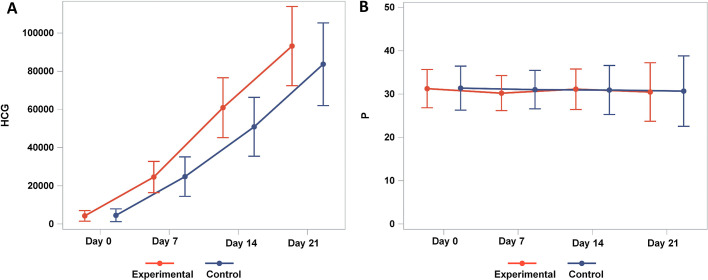
Both the experimental and control groups exhibited an increasing trend in serum HCG concentrations during the follow-up period, with the same magnitude of increase on the 7th day. However, on the 14th and 21st days, the experimental group showed a higher rate of increase than the control group, with statistically significant differences (Fig. 4A). Additionally, the serum p values in both study and control groups remained stable at the initial visit, 7th, 14th, and 21st days, showing no significant trend changes or statistical differences (Fig. 4B). The mixed effects model revealed that the serum HCG values on the 7th, 14th, and 21st days were statistically significant compared to the initial diagnosis time, with significant differences between the two groups (experimental and control) (Table 3). However, the mixed effects model analysis indicated that serum p values showed no statistical significance with time or group (Table 4).
Fig. 4.
Dynamic changes in serum HCG and P concentrations in patients from the experimental and control groups. (A) The horizontal axis represents time points, and the vertical axis represents serum HCG concentration (ng/ml). The sample sizes for the experimental and control groups were both 50. The statistical analysis revealed the following results: there were no statistically significant differences between the two groups at the initial visit and on the 7th day (p>0.05). However, on the 14th and 21st days, the experimental group showed a significant increase compared to the control group, with statistically significant differences observed (p<0.05). (B) The horizontal axis represents time points, while the vertical axis represents serum P concentration (ng/ml). The sample sizes for the experimental and control groups were both 50. The statistical analysis showed no significant differences between the two groups at the initial visit, 7th day, 14th day, and 21st day (p>0.05).
Table 3.
Mixed-effects model estimates for variable effects on serum HCG concentration with respect to time and group
| Variable | Group | Estimate | SE | t | p |
|---|---|---|---|---|---|
| Intercept | 1,931.8 | 1,155.3 | 1.672 | 0.095 | |
| Time | First visit | 0 | |||
| Day 7 | 20,301 | 626.9 | 32.383 | <0.001 | |
| Day 14 | 51,758 | 1,267.7 | 40.828 | <0.001 | |
| Day 21 | 84,257 | 1,766.4 | 47.699 | <0.001 | |
| Group | Control group | 0 | |||
| Experimental group | 4,738.9 | 2,044 | 2.318 | 0.02 |
Table 4.
Mixed-effects model estimates for variable effects on serum P concentration with respect to time and group
| Variable | Group | Estimate | SE | t | p |
|---|---|---|---|---|---|
| Intercept | 31.479 | 0.578 | 54.456 | <0.001 | |
| Time | First visit | 0 | |||
| Day 7 | −0.705 | 0.366 | −1.928 | 0.055 | |
| Day 14 | −0.277 | 0.437 | −0.635 | 0.526 | |
| Day 21 | −0.728 | 0.535 | −1.362 | 0.176 | |
| Group | Control group | 0 | |||
| Experimental group | −0.308 | 0.773 | −0.398 | 0.691 |
These results suggest that the rate of increase in serum HCG concentrations in the experimental group during treatment is higher than that in the control group, potentially reflecting the effectiveness of different treatment regimens. Therefore, monitoring the dynamic changes in serum HCG concentrations, especially on the 14th and 21st days, may be beneficial for evaluating treatment outcomes.
A Comparison of miscarriage rate and serum cytokine concentrations between experimental and control groups
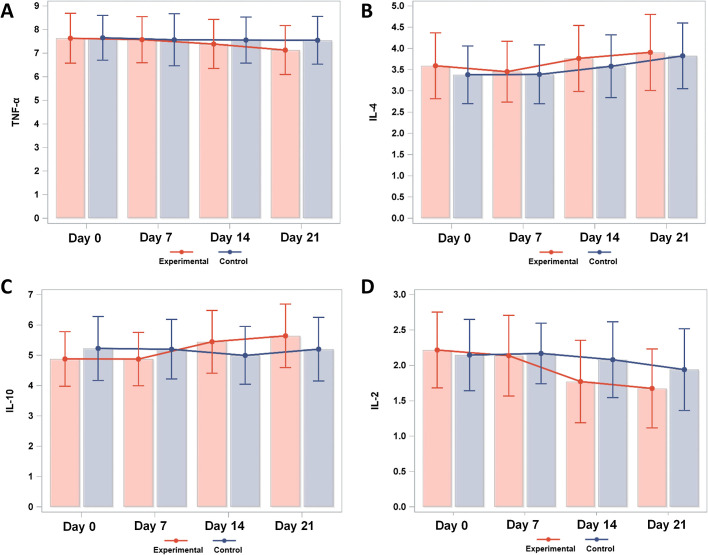
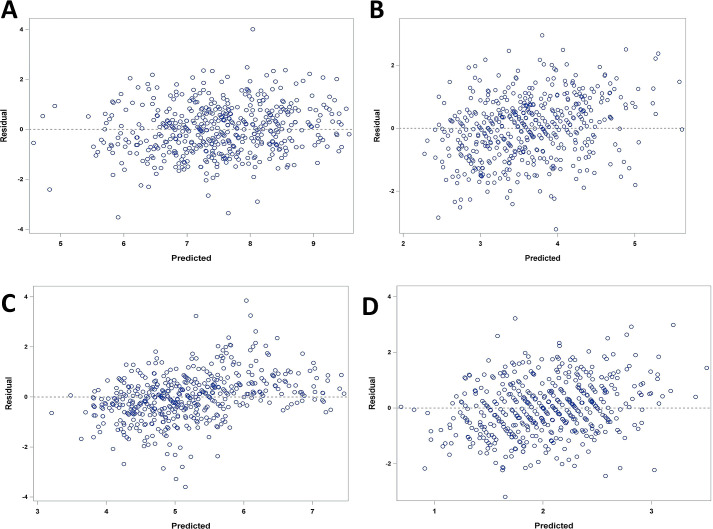
Miscarriage and threatened miscarriage are common complications during pregnancy, with serum cytokine concentrations playing a crucial role in their pathogenesis. There was no statistically significant difference in the rate of miscarriage between the experimental and control groups. However, the number of cases of threatened miscarriage was significantly reduced in the experimental group (Table 5). On the 7th day post initial assessment and intervention, the serum concentrations of Th1 cytokines (TNF-α, IL-2) and Th2 cytokines (IL-4, IL-10) showed no statistical differences between the experimental and control groups (Fig. 5). Yet, by the 14th day post-intervention, the experimental group showed a significant increase in IL-10 compared to the control group, whereas IL-2 significantly decreased (Fig. 5C and D); there were no statistical differences in TNF-α and IL-4 between the two groups (Fig. 5A and B). By the 21st day post-intervention, the experimental group exhibited a significant elevation in IL-10 compared to the control group, while TNF-α and IL-2 decreased significantly, with no statistical difference in IL-4 between the groups. The mixed-effects model analysis revealed that serum TNF-α concentration showed statistical differences at days 14 and 21 compared to the initial assessment (Table 6), with significant differences observed between the experimental and control groups. Serum IL-4 concentration exhibited statistical differences at days 14 and 21 compared to the initial assessment, but differences between the experimental and control groups were not statistically significant (Table 6). Serum IL-10 concentration showed statistical differences at day 21 compared to the initial assessment, while differences between the two groups were not statistically significant (Table 6). Serum IL-2 concentration had statistical differences at days 14 and 21 compared to the initial assessment, yet differences between the experimental and control groups were not statistically significant (Table 6). Model diagnostics indicated that the residuals showed no clear pattern as the predicted values varied, suggesting approximate normal distribution and good model fit, as detailed in Fig. 6.
Table 5.
Comparison of incidence rates a between control and experimental groups for pregnancy outcomes
| Control group | Experimental group | Statistical value | p | |
|---|---|---|---|---|
| Threatened abortion | 10 (16.9%) | 3 (4.6%) | χ2 = 5.013 | 0.025 |
| Inevitable miscarriage | 5 (8.5%) | 3 (4.6%) | χ2 = 0.763 | 0.382 |
Fig. 5.
Dynamic changes in serum cytokine concentrations in patients in the experimental and control groups. (A) Axis X represents the time points, and Axis Y represents the serum TNF-α concentration (ng/ml). The sample sizes of the experimental and control groups were both 50. Statistical analysis results are as follows: at the initial visit and on the 7th and 14th days, there were no statistically significant differences between the two groups (p>0.05). However, on the 21st day, the experimental group showed a decrease compared to the control group, indicating a statistically significant difference (p<0.05). (B) Axis X represents the time points, and Axis Y represents the serum IL-4 concentration (ng/ml). The sample sizes of the experimental and control groups were both 50. Statistical analysis results showed no statistically significant differences between the two groups at the initial visit and on the 7th, 14th, and 21st days (p>0.05). (C) Axis X represents the time points, and Axis Y represents the serum IL-10 concentration (ng/ml). The sample sizes of the experimental and control groups were both 50. Statistical analysis results indicated no statistically significant differences between the two groups at the initial visit and on the 7th day (p>0.05). However, on the 14th and 21st days, the experimental group showed an increase compared to the control group, with statistically significant differences (p<0.05). (D) Axis X represents the time points, and Axis Y represents the serum IL-2 concentration (ng/ml). The sample sizes of the experimental and control groups were both 50. Statistical analysis results demonstrated no statistically significant differences between the two groups at the initial visit and on the 7th day (p>0.05). However, on the 14th and 21st days, the experimental group showed a decrease compared to the control group, indicating statistically significant differences (p<0.05).
Table 6.
Mixed-effects model results of Th1/Th2 cytokines
| Variable | Group | Estimate | SE | t | p |
|---|---|---|---|---|---|
| TNF-α | |||||
| Intercept | 7.725 | 0.13 | 59.606 | <0.001 | |
| Time | First visit | 0 | |||
| Day 7 | −0.073 | 0.052 | −1.401 | 0.162 | |
| Day 14 | −0.173 | 0.055 | −3.157 | 0.002 | |
| Day 21 | −0.311 | 0.06 | −5.21 | <0.001 | |
| Group | Control group | 0 | |||
| Experimental group | −0.155 | 0.077 | −2.023 | 0.044 | |
| IL-4 | |||||
| Intercept | 3.417 | 0.091 | 37.519 | <0.001 | |
| Time | First visit | 0 | |||
| Day 7 | −0.069 | 0.052 | −1.331 | 0.184 | |
| Day 14 | 0.187 | 0.059 | 3.186 | 0.002 | |
| Day 21 | 0.377 | 0.068 | 5.52 | <0.001 | |
| Group | Control group | 0 | |||
| Experimental group | 0.143 | 0.118 | 1.213 | 0.227 | |
| IL-10 | |||||
| Intercept | 5.027 | 0.12 | 41.922 | <0.001 | |
| Time | First visit | 0 | |||
| Day 7 | −0.016 | 0.078 | −0.208 | 0.835 | |
| Day 14 | 0.186 | 0.109 | 1.716 | 0.088 | |
| Day 21 | 0.387 | 0.146 | 2.647 | 0.009 | |
| Group | Control group | 0 | |||
| Experimental group | 0.037 | 0.131 | 0.281 | 0.779 | |
| IL-2 | |||||
| Intercept | 2.248 | 0.067 | 33.66 | <0.001 | |
| Time | First visit | 0 | |||
| Day 7 | −0.031 | 0.036 | −0.863 | 0.389 | |
| Day 14 | −0.265 | 0.041 | −6.463 | <0.001 | |
| Day 21 | −0.383 | 0.048 | −8.023 | <0.001 | |
| Group | Control group | 0 | |||
| Experimental group | −0.121 | 0.086 | −1.416 | 0.159 |
Fig. 6.
Model diagnosis—residual scatter plots. (A) Scatter plot of residual serum TNF-α concentrations. (B) Scatter plot of residual serum IL-4 concentrations. (C) Scatter plot of residual serum IL-10 concentrations. (D) Scatter plot of residual serum IL-2 concentrations.
The results of this study indicate that after intervention, the concentration of serum IL-10 in the experimental group significantly increased, while the concentration of IL-2 significantly decreased, suggesting that the treatment may have affected the immune regulatory pathway. Although there was no significant difference in overall miscarriage rates, the number of threatened miscarriages significantly decreased, which may be related to immune regulation. Changes in serum cytokine concentrations reflect part of the treatment response and miscarriage risk, providing important insights for clinical interventions.
Literature screening and summary with quality assessment of included studies
This study conducted a comprehensive search and selected appropriate papers for meta-analysis from 417 initially identified articles (Fig. 7A). These articles were sourced from various databases including PubMed, Web of Science, CNKI, and Wan Fang Data. After removing duplicates, a detailed screening was performed on the remaining 140 articles. Through reviewing titles and abstracts, 75 articles not aligning with the research objectives or being non-randomized studies were excluded. The remaining 65 articles underwent full-text scrutiny, resulting in the exclusion of 54 articles due to missing key data, incomplete access to full text, or statistical methods not meeting the study requirements. Ultimately, 11 qualifying articles were included in the quantitative analysis. For the included studies, key information such as the first author, publication year, sample size, and patient demographics were summarized, forming a basic characteristics table (Table 7).
Fig. 7.
Literature screening process and quality assessment of included studies. (A) Flowchart of literature inclusion. (B) Summary of bias risk assessment in included studies.
Table 7.
Information about the literature that met the inclusion criteria
| First author | Publication time | Sample size | Age of subject |
Geographical area | Sample source |
|---|---|---|---|---|---|
| Li Ling (Ling LI, Lixiong LUO, Xuebin WU. Discussion on the correlation between serum vitamin D levels in women of childbearing age and early spontaneous abortion [J]. Northern Pharmacy, 2023, 20(12): 65–67.) | 2023 | 224 | — | Fujian Province | Women with early spontaneous abortion |
| Feng Huexhao (DOI: 10.27043/d.cnki.ggzyc.2023.000120) | 2023 | 120 | — | Guangdong Province | RSA |
| Zhao Xueling (DOI: 10.3969/j.issn.1008-1070.2022.12.021) | 2022 | 73 | 21–38 years old |
Henan Province | RSA |
| Xie Ting (DOI: 10.14188/j.1671-8852.2022.0082) | 2022 | 142 | — | Henan Province | RSA |
| Huang Yuehong (Huang Yuehong, Fan Guangqin, Zhang Yanlin. Relationship between vitamin D levels during pregnancy and pregnancy outcomes in patients with recurrent miscarriage [J]. Chinese Modern Doctor, 2022, 60(06): 56–58+69.) | 2022 | 100 | — | Guangdong Province | RSA |
| Wang Huiming (DOI: 10.3969/j.issn.1673-4130.2020.18.022) | 2020 | 138 | 25–35 years old |
Hubei Province | RSA |
| Xu Fang (DOI: CNKI:SUN:ZWJZ.0.2020-05-030) | 2020 | 98 | — | Zhejiang Province | Early threatened abortion |
| Hui Lingyun (DOI: 10.7620/zgfybj.j.issn.1001-4411.2019.15.48) | 2019 | 81 | — | Shaanxi Province | Women with early spontaneous abortion |
| Zhao Xuannan (Zhao Xuenan. Study on the correlation between serum vitamin D and immunological indicators in patients with missed miscarriage [D]. Yan’an University, 2019.) | 2019 | 60 | — | Shaanxi Province | MA |
| Qi Yajie (DOI: 10.27231/d.cnki.gnmyc.2019.000202) | 2020 | 138 | 20–35 years old |
Inner Mongolia Autonomous Region | RSA |
| Hui Lingyun (DOI: 10.3969/j.issn.1673-5293.2017.12.024) | 2017 | 70 | 23–40 years old |
Shaanxi Province | RSA |
RSA, recurrent spontaneous abortion; MA, missed abortion.
For the quality assessment of the literature, a rigorous evaluation was conducted based on the bias risk assessment tool from the Cochrane Collaboration (Fig. 7B). This assessment covered aspects such as random sequence generation, allocation concealment, blinding procedures, and completeness of outcome data, each classified into low, high, or uncertain risk levels. Studies with unclear randomization or allocation concealment measures were deemed high risk, while those providing detailed reports on randomization, allocation concealment, and blinding measures were considered low risk. Through this assessment process, we ensured that the included studies met the high standards required for scientific evidence.
Low serum 1,25-(OH)D3 concentrations associated with increased risk of early miscarriage
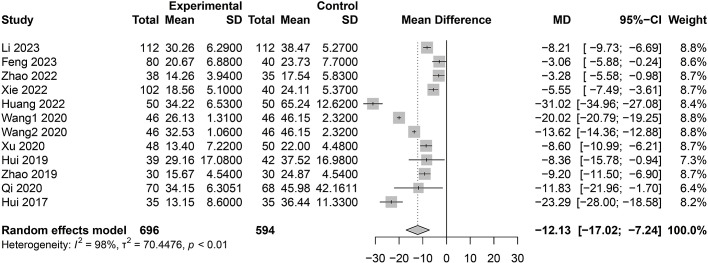
This Meta-analysis focuses on the relationship between serum 1,25-(OH)D3 levels and the risk of early miscarriage. The summarized Forest plot reveals the difference in serum 1,25-(OH)D3 concentrations between the experimental group (patients with early miscarriage) and the control group (pregnant women who did not experience miscarriage) (Fig. 8). Despite a very high level of detected heterogeneity (I2 = 98%), which may indicate methodological differences among studies or other potential variables, the pooled results show that the 1,25-(OH)D3 concentrations in the experimental group are significantly lower than in the control group. Specifically, the pooled mean difference (MD) is −12.13 with a 95% CI of [−17.02, −7.24], indicating a significant statistical association between lower 1,25-(OH)D3 levels and early miscarriage.
Fig. 8.
Meta-analysis forest plot of the association between serum 1,25-(OH)D3 concentration and early miscarriage.
This finding underscores the potential significance of vitamin D metabolic status during pregnancy in relation to early miscarriage. However, given the high level of heterogeneity among studies, these results should be interpreted cautiously. Future research should standardize the measurement of serum 1,25-(OH)D3 levels and control for known confounding factors such as age, lifestyle, and pre-pregnancy weight to ensure the accuracy and consistency of the results.
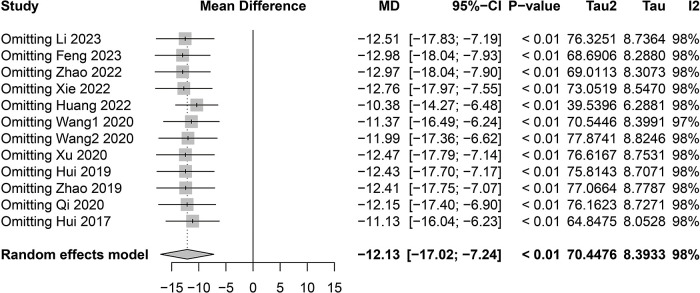
Sensitivity analysis of the relationship between serum 1,25-(OH)D3 levels and risk of early miscarriage reveals robust conclusions. This study conducted a sensitivity analysis to evaluate the impact of removing each individual study on the meta-analysis results regarding the association between serum 1,25-(OH)D3 levels and early miscarriage (Table 8 and Fig. 9). Sensitivity analysis can uncover the influence of specific studies on the overall results, thereby assessing the robustness of the conclusions. As depicted in Fig. 8, the exclusion of each study individually did not lead to significant changes in the pooled MD and its 95% CI, indicating that no single study had a decisive impact on the overall conclusion, thus confirming the stability and reliability of the results.
Table 8.
Sensitivity analysis
| Deleted document | Pooled effect size of remaining literature (Pooled MD) |
95% confidence intervals (CI) for the remaining literature |
|---|---|---|
| Li 2023 | −12.5083 | [−17.8271; −7.1896] |
| Feng 2023 | −12.9805 | [−18.0355; −7.9256] |
| Zhao 2022 | −12.9690 | [−18.0374; −7.9005] |
| Xie 2022 | −12.7586 | [−17.9668; −7.5505] |
| Huang 2022 | −10.3755 | [−14.2722; −6.4789] |
| Wang1 2020 | −11.3661 | [−16.4917; −6.2404] |
| Wang2 2020 | −11.9905 | [−17.3617; −6.6193] |
| Xu 2020 | −12.4669 | [−17.7924; −7.1414] |
| Hui 2019 | −12.4343 | [−17.6979; −7.1707] |
| Zhao 2019 | −12.4108 | [−17.7515; −7.0702] |
| Qi 2020 | −12.1537 | [−17.4037; −6.9038] |
| Hui 2017 | −11.1344 | [−16.0416; −6.2273] |
Fig. 9.
Sensitivity analysis forest plot of the relationship between serum 1,25-(OH)D3 levels and the risk of early miscarriage.
Across all studies, the estimated values of the pooled MD remained between −11.13 and −12.97, which were close to the overall estimated value when no studies were excluded [−12.13, 95% CI: (−17.02; −7.24)]. Furthermore, even under conditions of extremely high heterogeneity (I2 = 98%), the magnitude of the effect between studies did not significantly change, highlighting the resilience of the study conclusions against potential biases. These results emphasize that our findings in the research on the correlation between vitamin D3 levels and the risk of early miscarriage are stable and reliable.
The forest plot visually represents the changes in the meta-analysis pooled results after removing each study, providing a quantitative assessment of the stability of the results and enhancing the credibility of our conclusions. The effect estimates of each study and their 95% CIs are depicted in the plot as squares and horizontal lines, while the diamond represents the overall effect size after combining all studies, all indicating a significant increase in the risk of early miscarriage associated with low levels of serum 1,25-(OH)D3.
The association between vitamin D3 levels and early miscarriage: evidence of no publication bias
This study conducted a test for publication bias to assess the relationship between vitamin D3 levels and early miscarriage. The results were visually displayed through a funnel plot (Fig. 10). The funnel plot revealed the distribution relationship between the effect size (mean difference) of each study and its SE. Ideally, a funnel plot of a meta-analysis without publication bias would show a symmetrical inverted funnel shape, with most studies falling within the central area of the plot. In the funnel plot of this study, although the data points were somewhat scattered, there was no significant asymmetry observed, indicating the absence of significant publication bias in the existing evidence.
Fig. 10.
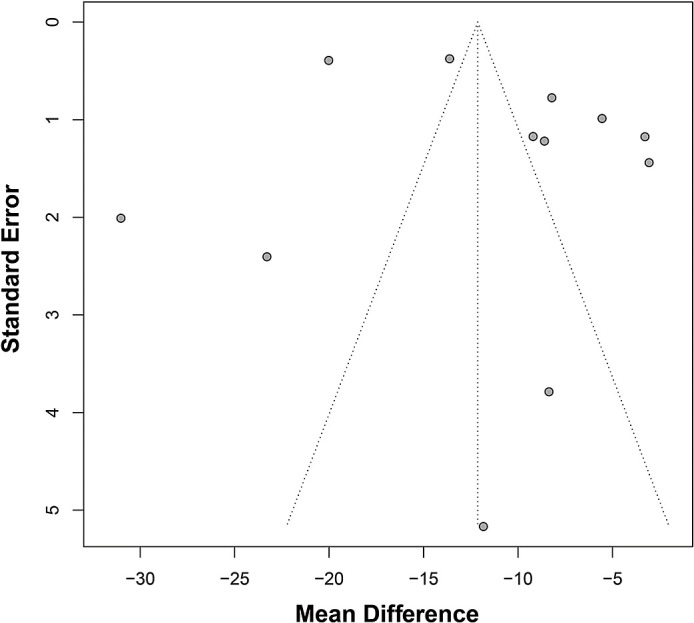
Results of publication bias test in meta-analysis.
Despite some degree of scatter in the distribution of data points in the funnel plot, the majority clustered around the center of the funnel, supporting the robustness and credibility of the meta-analysis results. The deviation of individual studies did not significantly impact the overall trend, suggesting that while there is heterogeneity among studies, the overall conclusion regarding the association between vitamin D3 levels and increased risk of early miscarriage remains solid. This finding holds important implications for clinical and public health practice by emphasizing the significance of monitoring and managing vitamin D levels during pregnancy.
Discussion
In recent years, the impact of vitamin D3 deficiency on pregnancy has become a hot research topic.(42–44) Multiple studies have shown an association between vitamin D3 deficiency and early spontaneous miscarriage, with lower levels of vitamin D3 in the serum of miscarriage patients compared to normal pregnancy patients during the same period. This difference was statistically significant in recurrent miscarriage patients.(45) Some research even suggests that the level of 25-(OH)D3 in the serum can be used as a predictive tool for fetal miscarriage, with a predictive rate of up to 80%.(46–48) Our preliminary research indicated that vitamin D3 deficiency is one of the contributing factors to miscarriage and may impact early pregnancy stability by altering the microenvironment of the decidua tissue.
This study supplemented 400 IU of vitamin D3 as the daily requirement. The results showed a significant increase in serum vitamin D3 levels in the group supplemented with 400 IU of vitamin D3 at the 14th and 21st day follow-ups. Furthermore, the supplemented group exhibited higher serum HCG concentrations on the 14th and 21st days of follow-up compared to the non-supplemented group, although there was no significant effect on progesterone. The probability of miscarriage after vitamin D3 supplementation was similar to that of non-supplemented individuals. However, the likelihood of threatened miscarriage was significantly reduced in the supplemented group, suggesting that vitamin D3 supplementation may increase HCG levels and reduce the occurrence of threatened miscarriage, thereby promoting pregnancy stability.
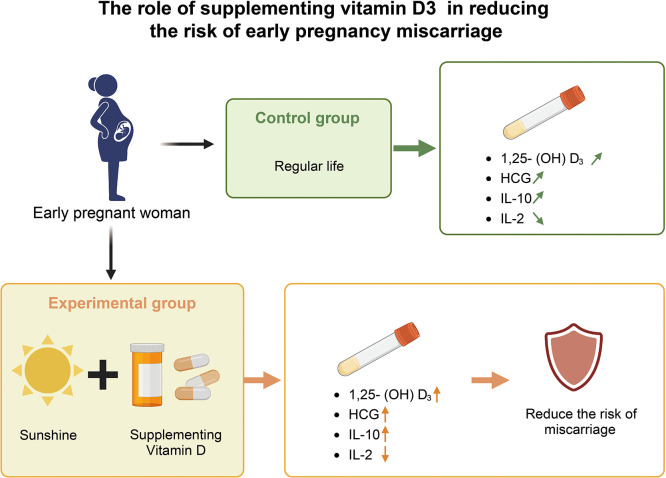
Current evidence suggests that vitamin D3 can prevent immune-related early pregnancy miscarriages by modulating the function of helper T cells. The imbalance between Th1/Th2 types is considered an important factor in natural miscarriage, especially recurrent miscarriage.(49) Previous studies have shown that in patients with natural miscarriage and low vitamin D3 levels, IL-10 levels were significantly lower than in the sufficient vitamin D3 group, while TNF-α and IL-2 levels were significantly higher in the chorionic and decidua tissues of the control group.(50) Researchers have also found that treating women with unexplained recurrent miscarriage with vitamin D3 inhibits IFN-γ and significantly reduces the ratio of IFN-γ/IL-10 after culturing endometrial cells.(51) These studies collectively suggest a preventive effect of vitamin D3 on immune-mediated pregnancy failure.(47,52) This study found that increasing serum vitamin D3 levels through supplementation can reduce serum IL-2 and TNF-α concentrations while increasing serum IL-10 concentrations, with significant statistical differences observed on the 14th and 21st days. A mixed-effects model indicated statistical differences between the experimental and control groups in serum TNF-α levels, highlighting the more significant impact of vitamin D3 on Th1 cell factors, particularly TNF-α. Thus, our study affirms once again that vitamin D3 enhances pregnancy stability by regulating the Th1/Th2 balance, recommending routine supplementation of 400 IU of vitamin D3 for early pregnancy patients, especially those with vitamin D3 deficiency (Fig. 11).
Fig. 11.
The role of vitamin D3 supplementation in reducing the risk of early pregnancy miscarriage.
In this meta-analysis, we conducted a comprehensive search, selection, and quality assessment of a large number of studies to explore the potential association between serum levels of 1,25-(OH)D3 and early miscarriage. Through a rigorous literature screening process, we narrowed down from an initial pool of 417 articles to 11 studies that met strict inclusion criteria for further in-depth analysis. The screening method we employed ensured the quality and relevance of the included literature, crucial for reducing bias and providing trustworthy results. We performed a thorough bias risk assessment using the Cochrane bias risk tool for each study. Although the literature screening process and bias risk assessment indicated the meticulousness of our methodology, caution is advised in interpreting the results, especially in the context of high heterogeneity. Sensitivity analysis further confirmed the robustness of our findings, indicating that no single study result disproportionately influenced the overall meta-analysis. Assessment of publication bias revealed a symmetrical distribution in the funnel plot, suggesting that publication bias may not be a significant concern. Nevertheless, given the detected high heterogeneity, future studies should precisely evaluate the relationship between vitamin D3 levels and the risk of early miscarriage and standardize measurement criteria while controlling for known confounding factors. Overall, the current evidence supports the notion that low serum levels of 1,25-(OH)D3 may be a potential risk factor for early miscarriage compared to the control group. This finding underscores the importance of monitoring and intervening in vitamin D levels during pregnancy.
While these findings are encouraging, this study has limitations. The sample size and geographical restrictions may impact the generalizability of the results. Moreover, although randomized controlled designs can enhance research quality, potential confounding factors may remain uncontrollable.
Future research should aim to increase sample sizes and extend across regions to validate the universality of vitamin D3 supplementation for early pregnancy healthcare. More stringent randomized controlled study designs and control measures for potential confounders enhances the reliability of research results. Furthermore, the impact of vitamin D3 on other pregnancy health indicators, such as gestational diabetes and hypertension, also warrants further investigation.
In conclusion, this study emphasizes the potential importance of vitamin D3 in pregnancy management, laying a foundation for future research directions and clinical applications. Further investigations into the mechanisms may reveal the immunomodulatory role of vitamin D3 in the pregnancy process, offering more precise guidance for nutritional interventions in early pregnancy.
Author Contributions
QZ and LW conceived and designed the study; HX, LW, and LQ performed the experiments; QZ and LQ analyzed the data; QZ and HX wrote the manuscript.
Acknowledgments
This study was supported by Hangzhou Science and Technology Program Guidance Project (No. 20211231Y187).
Ethical Statement
This study was approved by the Clinical Ethics Committee of Tonglu First People’s Hospital (No. IRB-[2021–44]).
Conflict of Interest
No potential conflicts of interest were disclosed.
References
- 1.Choi SE, Choudhary A, Ahern JM, Palmer N, Barrow JR. Association between maternal periodontal disease and adverse pregnancy outcomes: an analysis of claims data. Fam Pract 2021; 38: 718–723. [DOI] [PubMed] [Google Scholar]
- 2.Manda-Taylor L, Kufankomwe M, Chatha G, et al. Perceptions and experiences of intravenous iron treatment for anaemia in pregnancy in Malawi: a formative qualitative study. Gates Open Res 2024; 6: 66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ayen SS, Kasahun AW, Zewdie A. Depression during pregnancy and associated factors among women in Ethiopia: a systematic review and meta-analysis. BMC Pregnancy Childbirth 2024; 24: 220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Quenby S, Gallos ID, Dhillon-Smith RK, et al. Miscarriage matters: the epidemiological, physical, psychological, and economic costs of early pregnancy loss. Lancet 2021; 397: 1658–1667. [DOI] [PubMed] [Google Scholar]
- 5.McNestry C, Killeen SL, Crowley RK, McAuliffe FM. Pregnancy complications and later life women’s health. Acta Obstet Gynecol Scand 2023; 102: 523–531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.D'Angelo A, Petrella C, Greco A, et al. Acute alcohol intoxication: a clinical overview. Clin Ter 2022; 173: 280–291. [DOI] [PubMed] [Google Scholar]
- 7.Vallet-Buisan M, Mecca R, Jones C, Coward K, Yeste M. Contribution of semen to early embryo development: fertilization and beyond. Hum Reprod Update 2023; 29: 395–433. [DOI] [PubMed] [Google Scholar]
- 8.Wichert-Schmitt B, D'Souza R, Silversides CK. Reproductive issues in patients with the fontan operation. Can J Cardiol 2022; 38: 921–929. [DOI] [PubMed] [Google Scholar]
- 9.Bohren MA, Lorencatto F, Coomarasamy A, et al. Formative research to design an implementation strategy for a postpartum hemorrhage initial response treatment bundle (E-MOTIVE): study protocol. Reprod Health 2021; 18: 149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Palma O, Jallah JK, Mahakalkar MG, Mendhe DM. The effects of vegan diet on fetus and maternal health: a review. Cureus 2023; 15: e47971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Goltzman D. Approach to hypercalcemia. In: Feingold KR, Anawalt B, Blackman MR, et al., eds. Endotext. South Dartmouth (MA): MDText.com, Inc., April 17, 2023.. [Google Scholar]
- 12.Sell J, Ramirez S, Partin M. Parathyroid disorders. Am Fam Physician 2022; 105: 289–298. [PubMed] [Google Scholar]
- 13.Jodar E, Campusano C, de Jongh RT, Holick MF. Calcifediol: a review of its pharmacological characteristics and clinical use in correcting vitamin D deficiency. Eur J Nutr 2023; 62: 1579–1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pludowski P, Takacs I, Boyanov M, et al. Clinical practice in the prevention, diagnosis and treatment of vitamin D deficiency: a Central and Eastern European Expert Consensus Statement. Nutrients 2022; 14: 1483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sarris J, Ravindran A, Yatham LN, et al. Clinician guidelines for the treatment of psychiatric disorders with nutraceuticals and phytoceuticals: The World Federation of Societies of Biological Psychiatry (WFSBP) and Canadian Network for Mood and Anxiety Treatments (CANMAT) Taskforce. World J Biol Psychiatry 2022; 23: 424–455. [DOI] [PubMed] [Google Scholar]
- 16.Piuri G, Zocchi M, Della Porta M, et al. Magnesium in obesity, metabolic syndrome, and type 2 diabetes. Nutrients 2021; 13: 320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Agrawal I, Mehendale AM, Malhotra R. Risk factors of postpartum depression. Cureus 2022; 14: e30898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Khan AA, Guyatt G, Ali DS, et al. Management of hypoparathyroidism. J Bone Miner Res 2022; 37: 2663–2677. [DOI] [PubMed] [Google Scholar]
- 19.Song J, Zhou B, Kan J, et al. Gut microbiota: linking nutrition and perinatal depression. Front Cell Infect Microbiol 2022:. 12: 932309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Arnold A, Dennison E, Kovacs CS, et al. Hormonal regulation of biomineralization. Nat Rev Endocrinol 2021; 17: 261–275. [DOI] [PubMed] [Google Scholar]
- 21.Stenhouse C, Suva LJ, Gaddy D, Wu G, Bazer FW. Phosphate, calcium, and vitamin D: key regulators of fetal and placental development in mammals. Adv Exp Med Biol 2022; 1354: 77–107. [DOI] [PubMed] [Google Scholar]
- 22.Kovacs CS. Calcium and phosphate metabolism and related disorders during pregnancy and lactation. In: Feingold KR, Anawalt B, Blackman MR, et al., eds. Endotext. South Dartmouth (MA): MDText.com, Inc., August 1, 2021.. [Google Scholar]
- 23.Minich DM, Henning M, Darley C, Fahoum M, Schuler CB, Frame J. Is melatonin the “next vitamin D”?: a review of emerging science, clinical uses, safety, and dietary supplements. Nutrients 2022; 14: 3934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yalçın Bahat P, Ayhan I, Üreyen Özdemir E, İnceboz Ü, Oral E. Dietary supplements for treatment of endometriosis: a review. Acta Biomed 2022; 93: e2022159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ao T, Kikuta J, Ishii M. The effects of vitamin D on immune system and inflammatory diseases. Biomolecules 2021; 11: 1624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kurmangali Z, Abdykalykova B, Kurmangali A, Zhantagulov D, Terzic M. The influence of vitamin D on pregnancy and outcomes: current knowledge and future perspectives. Gynecol Obstet Invest 2024; 89: 261–266. [DOI] [PubMed] [Google Scholar]
- 27.Yang M, Shen X, Lu D, et al. Effects of vitamin D supplementation on ovulation and pregnancy in women with polycystic ovary syndrome: a systematic review and meta-analysis. Front Endocrinol (Lausanne) 2023; 14: 1148556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Venkatesh U, Sharma A, Ananthan VA, Subbiah P, Durga R;. CSIR Summer Research training team. Micronutrient’s deficiency in India: a systematic review and meta-analysis. J Nutr Sci 2021; 10: e110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tkachuk AS, Vasukova EA, Anopova AD, et al. Vitamin D status and gestational diabetes in Russian pregnant women in the period between 2012 and 2021: a nested case-control study. Nutrients 2022; 14: 2157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hutchings N, Babalyan V, Heijboer AC, et al. Vitamin D status in Armenian women: a stratified cross-sectional cluster analysis. Eur J Clin Nutr 2022; 76: 220–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.D'Ippolito S, Capozzi A, Scambia G, Sorge R, Lello S, Simone ND. Glucose/insulin metabolism and vitamin D in women with recurrent pregnancy loss. Am J Reprod Immunol 2022; 87: e13505. [DOI] [PubMed] [Google Scholar]
- 32.Hemamy M, Pahlavani N, Amanollahi A, et al. The effect of vitamin D and magnesium supplementation on the mental health status of attention-deficit hyperactive children: a randomized controlled trial. BMC Pediatr 2021; 21: 230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chan V, Lo K. Efficacy of dietary supplements on improving sleep quality: a systematic review and meta-analysis. Postgrad Med J 2022; 98: 285–293. [DOI] [PubMed] [Google Scholar]
- 34.Yeung WG, Palmer SC, Strippoli GFM, et al. Vitamin D therapy in adults with CKD: a systematic review and meta-analysis. Am J Kidney Dis 2023; 82: 543–558. [DOI] [PubMed] [Google Scholar]
- 35.Bikle DD. Vitamin D: production, metabolism and mechanisms of action. In: Feingold KR, Anawalt B, Blackman MR, et al., eds. Endotext. South Dartmouth (MA): MDText.com, Inc., December 31, 2021. [Google Scholar]
- 36.Dahiya V, Vasudeva N, Sharma S, Kumar A. Role of dietary supplements in thyroid diseases. Endocr Metab Immune Disord Drug Targets 2022; 22: 985–996. [DOI] [PubMed] [Google Scholar]
- 37.Prud'homme GJ, Kurt M, Wang Q. Pathobiology of the klotho antiaging protein and therapeutic considerations. Front Aging 2022; 3: 931331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mäkitie O, Zillikens MC. Early-onset osteoporosis. Calcif Tissue Int 2022; 110: 546–561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tamblyn JA, Pilarski NSP, Markland AD, et al. Vitamin D and miscarriage: a systematic review and meta-analysis. Fertil Steril 2022; 118: 111–122. [DOI] [PubMed] [Google Scholar]
- 40.Banks N, Sun F, Krawetz SA, et al. Male vitamin D status and male factor infertility. Fertil Steril 2021; 116: 973–979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang Y, Li S, Zhuo F, et al. Additive effects of VDBP and 1,25(OH)2D3 on the viability and apoptosis of rheumatoid arthritis synovial fibroblasts. Front Endocrinol 2021; 11: 583229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bhowmik B, Siddiquee T, Mdala I, et al. Vitamin D3 and B12 supplementation in pregnancy. Diabetes Res Clin Pract 2021; 174: 108728. [DOI] [PubMed] [Google Scholar]
- 43.Bouillon R, Antonio L, Olarte OR. Calcifediol (25OH Vitamin D3) deficiency: a risk factor from early to old age. Nutrients 2022; 14: 1168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Uday S, Högler W. Nutritional rickets & osteomalacia: a practical approach to management. Indian J Med Res 2020; 152: 356–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hou W, Yan XT, Bai CM, Zhang XW, Hui LY, Yu XW. Decreased serum vitamin D levels in early spontaneous pregnancy loss. Eur J Clin Nutr 2016; 70: 1004–1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ota K, Dambaeva S, Han AR, Beaman K, Gilman-Sachs A, Kwak-Kim J. Vitamin D deficiency may be a risk factor for recurrent pregnancy losses by increasing cellular immunity and autoimmunity. Hum Reprod 2014; 29: 208–219. [DOI] [PubMed] [Google Scholar]
- 47.Tafti FD, Zare F, Miresmaeili SM, Fesahat F. Evaluating vitamin D and foxp3 mRNA levels in women with recurrent spontaneous abortion. JBRA Assist Reprod 2022; 26: 232–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mousavi Salehi A, Ghafourian M, Amari A, Zargar M. Evaluation of CD3+ T cell percentage, function and its relationship with serum vitamin D levels in women with recurrent spontaneous abortion and recurrent implantation failure. Iran J Immunol 2022; 19: 369–377. [DOI] [PubMed] [Google Scholar]
- 49.Valencia-Ortega J, Saucedo R, Peña-Cano MI, Hernández-Valencia M, Cruz-Durán JG. Immune tolerance at the maternal-placental interface in healthy pregnancy and pre-eclampsia. J Obstet Gynaecol Res 2020; 46: 1067–1076. [DOI] [PubMed] [Google Scholar]
- 50.Falk MJ, Decherney A, Kahn JP. Mitochondrial replacement techniques—implications for the clinical community. N Engl J Med 2016; 374: 1103–1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ota K, Dambaeva S, Kim MW, et al. 1,25-Dihydroxy-vitamin D3 regulates NK-cell cytotoxicity, cytokine secretion, and degranulation in women with recurrent pregnancy losses. Eur J Immunol 2015; 45: 3188–3199. [DOI] [PubMed] [Google Scholar]
- 52.Chen Y, Wu Q, Wei J, Hu J, Zheng S. Effects of aspirin, vitamin D3, and progesterone on pregnancy outcomes in an autoimmune recurrent spontaneous abortion model. Braz J Med Biol Res 2021; 54: e9570. [DOI] [PMC free article] [PubMed] [Google Scholar]