Abstract
Mangled upper-extremity injuries can have devastating consequences for the patient. The goal of treatment was to recreate a functional hand with a minimum of three sensitive and opposable fingers to achieve an acceptable quality of life. Early management of injuries is critical to treatment success. Initial assessment begins in the emergency department where the injury is quantified and the amputated segments are evaluated, particularly the possibility of replanting them or using them as spare parts as tissue bank. In the operating room, careful debridement is essential for any reconstruction. Despite advances in reconstructive techniques, the management of these injuries is a challenge for the surgeon. The main treatment guidelines and a step-by-step approach to the mangled hand are presented to achieve acceptable results.
Key words: Mangled hand, Reconstructive microsurgery, Replantation, Revascularization
The term “mangled” has a French derivation and defines a complex injury involving several tissues (literally “cut to pieces”). It is often caused by crushing or pulling trauma that does not result in a net injury but in severe multitissue damage that often results in a loss of complete limb function. Different degrees of substance loss are possible, affecting skin, subcutis, muscle-tendon structures, bone, vessels, and nerves.1
These injuries are physically and psychologically devastating, considering that the loss of the thumb alone represents 50% of the function loss of an entire hand and 25% of the body.2
The goal of treatment was to save or restore the prehension function of the hand in order to enable the patient to perform a hand defined as “acceptable” by Piñal,3 which means a hand composed of at least three fingers of near-normal length, with good proximal interphalangeal joint motion, with sufficient sensitivity and an opposable thumb.
However, in extremely complex cases, where the reconstruction of three digital segments is not possible, it is desirable to give the patient a minimal pincer function with two opposable and pain-free fingers. One of the two fingers must be stable, and the second one must be mobile to oppose the first and have an adequate wide commissure to be able to grasp objects: such a hand provides an acceptable grip with variable strength.4 Sensitivity restoration is extremely important and was another main goal of the surgery.
Initial Patient Assessment
Because mutilation injuries of the upper limb and hand are often caused by severe traumas, it is not uncommon for the patient to also have important injuries of other potentially life-threatening body districts. The initial assessment of the patient should also include a comprehensive evaluation using the advanced trauma life support protocol with an examination of the airway, breathing, and circulation.
Gathering the medical history is essential to understand the dynamics of the accident, focusing especially on the timing of the trauma. This is particularly important, especially in the case of devascularizing injuries and if large segments are involved to reduce the risks of muscle necrosis and resorption syndromes. Muscle tissue will be irreversibly damaged after 6 hours of warm ischemia and after 12 hours of cold ischemia. Other tissues have a survival of 12 hours in case of warm ischemia and 24 hours in case of cold ischemia.5,6
The accident setting provides information on the type of injury and the extent of soft tissue involvement: sharp injuries tend to involve a limited area of tissue injury and are easier to manage, whereas high-speed or crush injuries have a very extensive soft tissue involvement that often necessitates more extensive debridement and more difficult reconstructions.
Other aspects to be assessed are the patient’s general health and the conditions of transport and storage of any amputated segments.7 Each amputated segment should be wrapped in moist gauze, placed in a closed plastic bag, and submerged in a container with water and ice to prevent tissue freezing and direct exposure to water if it is to be replanted.8
The lesion assessment involves both clinical and radiographic evaluation. The initial inspection takes place in the emergency department, thus allowing adequate preparation of the surgical instrumentation to be used in the operating theater. This stage allows a cursory assessment mainly of the viability of the segments, as pain contamination or deformity may prevent a more thorough evaluation.
The assessment of finger vascularity is based on the color and capillary refill of the fingertips: it is often easier to assess the refill, at the level of the perionychium from the dorsal surface of the distal phalanx, which is thin, than from the thicker and more frequently soiled pulpal region. However, even a completely devascularized finger may have a capillary refill that appears intact under the nail bed because of the presence of stagnant blood. If in doubt, it is possible to prick the fingertip with a needle (once). Repeated pricking is not recommended because it also causes microtraumatism and consequent clotting at the capillary level in an already compromised tissue. Red bleeding will confirm viability, whereas difficult, slow, dark-colored bleeding, a problem with arterial inflow. When it is possible to perform it, the Allen test allows us to highlight a possible continuity of the palmar arches, which can be confirmed by a Doppler examination.
Evaluation of the musculotendinous system and neurologic functions, including both motor and sensory functions, must be performed with the patient awake. In comatose patients, abnormalities in the finger cascade may suggest a muscle or tendon injury.8
In the case of major bleeding, especially venous bleeding, the best treatment option is to directly compress the bleeding area of the hand (eg, with an elastic bandage). When direct compression is not sufficient, it may be necessary to temporarily inflate the tourniquet until bleeding control is achieved. However, two aspects need to be considered: the first is that the tissues distal to the injury may have already undergone a period of ischemia, and further complete ischemia could permanently compromise them. The second is that in the case of a venous lesion, an incorrectly positioned tourniquet (because of pain caused by pressure) determines a blockage of the venous return but not of the arterial inflow: this consequently leads to an increased blood flow return at the lesion site with increased blood loss (blocked venous return pathways and only one outlet from the injured vein).
Intravenous antibiotic therapy should be administered as soon as possible, theoretically at the scene of the accident, during first aid procedures, and later, in hospital, according to protocols, together with tetanus prophylaxis.9
Surgical Treatment
The first step in the surgical treatment of the patient is debridement, which must be performed immediately to minimize the risk of infection and must be performed by an experienced surgeon.10 It is recommended to perform this procedure with the aid of optical magnification to isolate the noble tissues (vessels and nerves) whose anatomy is often altered. Careless debridement could result in the loss of key anatomical elements. Similarly, failure to remove nonviable tissue causes potential infection, as well as an increase in the zone of necrosis by propagation (coagulative necrosis of neighboring tissues). Observing tissue perfusion while working without limb ischemia may help detect tissue viability, although in high-energy trauma, this is not always easy to ascertain and clearly detectable. Irrigation with large volume saline solution to remove loose debris and all devitalized tissues should be carried out after the first debridement phase to avoid too early irrigation, causing contamination of joints and intermuscular planes.1
As a general rule, cortical bone fragments with no soft tissue adhesion must be considered devitalized and removed, even if such debridement causes a consequent larger bone loss that requires a more complex reconstruction plan; an exception is made for some joint portions that can be reused as “bank tissue.”
Following debridement, bone fixation must be performed: as there is often a bone loss in this type of trauma. The management of bone damage must be based on the size of the residual defect after debridement, the degree of the area contamination, the quality of local perfusion, and the state of the soft tissue bed. Rarely, if the wound bed is clean, well perfused with a low risk of infection, and if soft tissue coverage is possible, the bone defect can be reconstructed immediately, but the use of internal means of synthesis is discouraged, preferring minimal synthesis with Kirschner wires or external fixation.11
When the condition of the soft tissues is compromised and the risk of infection is high, an antibiotic cement spacer may be useful in the case of a bone gap as a temporary strategy to wait for the soft tissues to heal and perform the bone reconstruction at a later time. Therefore, a secondary reconstruction will take place through the induced membrane or a bone flap or a prosthesis or a free vascularized joint graft (Fig. 1).
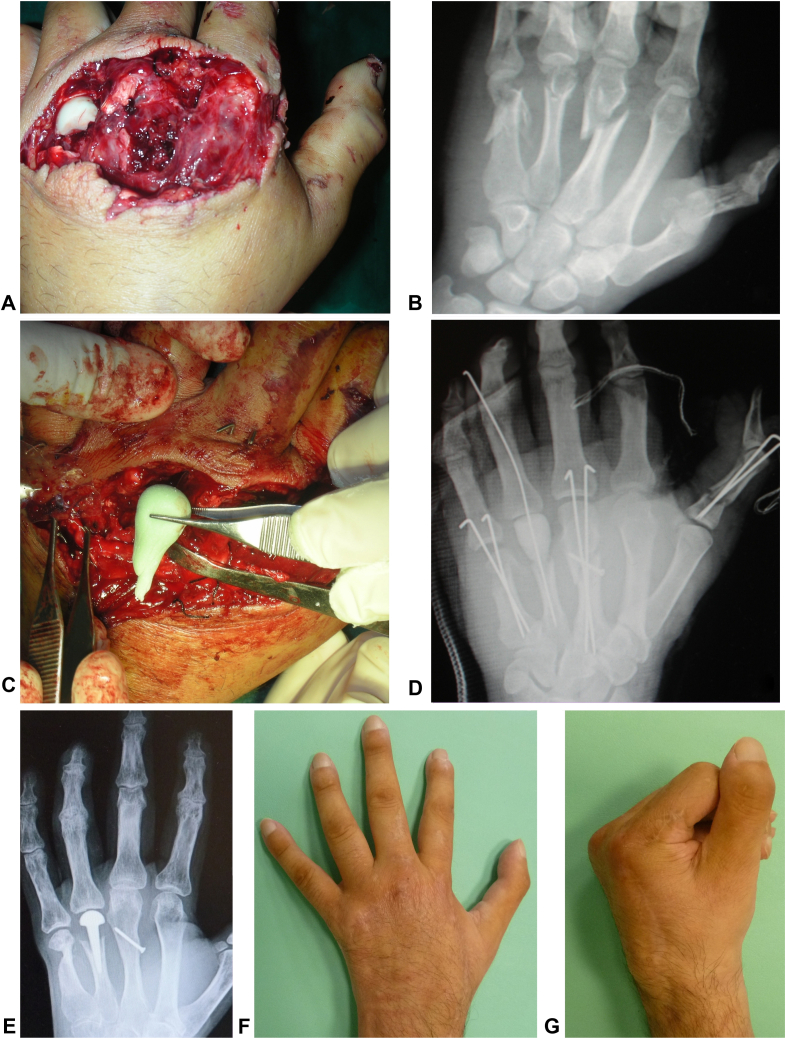
Figure 1.
A Clinical presentation of crush injury on the metacarpophalangeal joint of a left hand, B radiologic aspect of the lesion, C bone cement spacer used to keep the space in the fourth MP joint for a possible secondary reconstruction and was impossible the synthesis of the metacarpal head, D x-ray at the end of surgery with synthesis of fractures, E secondary reconstruction with an endoprosthesis of the head of the fourth metacarpal on the place of the cement “spacer,” and F, G clinical result with a good flexion of all MP joints. MP, metacarpophalangeal.
In the case of joint substance loss, immediate fusion can be performed, especially for the proximal interphalangeal joint of the fingers or the metacarpophalangeal of the thumb. For the metacarpophalangeals of middle fingers, it is preferred to place a cement spacer, which will then be replaced by a prosthesis or a free joint. Literature reports studies indicating that an emergency prosthesis is possible and does not expose to an increased risk of infection (possible silicone prosthesis); this is of course indicated if the coverage is valid, and an accurate debridement is possible.12,13
Before proceeding with skeletal fixation, a decision must be taken on whether to restore the anatomical length or shorten the bone as before performing the fixation. In the presence of nerve or/and arterial lesion, skeletal shortening may allow primary repair of the lesion, whereas length maintenance may require nerve or/and vessel grafting with an increased risk of complications because of vascular thrombosis or/and loss of axonal regeneration through nerve grafting. In cases of severe comminution of the fracture, shortening may also allow primary fixation of the bone, thus avoiding the need for bone graft and primary tendon repair.
If possible, the synthesis must be stable, and in the case of severe trauma with bone exposure, a minimal synthesis with Kirschner wires or external fixation is preferable, especially in those cases that present a risk of contamination. Obviously, if possible, in the case of replantation, an internal synthesis, with intramedullary screws, which allows an easier sliding of the soft tissue, is to be preferred.
The order in which the structures are repaired depends on the surgeon’s preference and the type of injury. Generally, the most common order is bone, extensor tendon, flexor tendon, artery, nerve, vein (usually at least two veins for each repaired artery, as in the reimplantation technique), and skin. However, it should be borne in mind that in the presence of a vascular lesion requiring reconstruction, skeletal and tendon repair should be performed before the repair of the vessel itself to avoid tension in the vascular compartment.
The rationale for this scheme lies in the fact that the coarse movements of bone and tendon repair must precede the fine movements of the microsurgical part where the anastomosis must be handled with care, and “incongruous” maneuvers, which may tear the suture threads, must be avoided. This approach is different in the case of macrosegments in which the presence of muscle masses reduces ischemia time and therefore reperfusion, after rapid bone synthesis stabilization is the most important element to prevent muscle necrosis. Once the macrosegment has been reperfused, tendon sutures can be approached calmly without problems of critical time.
Tendon sutures are performed with 4–6 strands for the flexor tendons to allow early sliding and mobilization (Cruciate/Adelaide or M Tang)—for the extensors, the suture varies depending on the area—they do not require such early mobilization and can also be sutured with “U” stitches. Tendon transfers and/or tendon grafts in urgency are to be considered in case of skeletal segment motor deficit (it can, therefore, be considered to transfer the extensor proper of the index finger or of a superficial flexor of the third or fourth to restore thumb opposition), especially in those cases where the “donor” segment cannot be saved. As a general principle, the surgeon has to try to do “all-in-one procedure,” in an emergency during the first operation, since returning scar tissue in a complex injury is extremely complex and often gives poor results.
Nerve sutures are usually direct sutures, if there are losses of substance of small sensory nerves, it could be taken into consideration to use “bank” tissues from other parts that cannot be used, sensory nerves from the arm or forearm can be used as grafts or biological or synthetic tubulization; we prefer to use biological tubulization by grafting autologous veins filled with muscle as described by Brunelli et al.14
Tubulization can also be used if a nerve graft taken from the leg (sural nerve) is not to be used directly in complex mixed nerve lesions. In these cases, it is sometimes complex to decide where the nerve is to be considered healthy (nerve trimming), and the risk is to graft in an area where a neuroma or intraneural scar will later develop; in doubtful cases, we prefer to use conduits in emergency, whereas other authors use allograft in order to restore the anatomy.15 If the result is not satisfactory, the surgeon can always decide to go back on what has been performed, trim correctly the two stumps, and use nerve grafts.16
The vessel repair is obviously performed by means of the operating microscope; the microsurgical technique involves suturing at least two veins per arterial anastomosis in the case of digital segments. In the case of transmetacarpal lesions, the picture depends on the type of involvement of the palmar arch, whether it is possible to restore or whether it is necessary to suture the common digital branches at the intermetacarpal level individually. In any case, at least two superficial veins must always be present in the case of amputative lesions. In the case of subamputations, tendon and muscle continuity, as well as subcutaneous continuity often guarantee sufficient venous return.
"Tissue Bank" Concept
An important aspect to consider in the management of finger substance loss is the “tissue bank” concept. In the hand, when planning a reconstruction, surgeons should always consider using the structures of a finger that cannot be replanted or used in the original part (bone, tendon, vessels, and nerves) to make up for the loss of substance of another adjacent segment or finger.
In addition, priority for reimplantation should be given to amputated fingers that are less damaged: therefore, an index finger in good condition could be reimplanted or transplanted in place of a thumb in order to achieve a final result that is as functional as possible.17
Sometimes, it is necessary to place silicone bars for possible tendon transfers/grafts to be provided at a later date. Silicone allows a tunnel to be created where tendons can then be passed through; this option is only indicated if there is good coverage of the bars themselves and if there is the possibility of good joint movement in the future.18
This concept in the mangled hand is fundamental: many times, after the initial injury balance, free tissue (bone, tendon, skin, and nerve) or tissue to be revascularized (digital segments and skin) is used. Reconstructive priority should be given to the thumb and at least one or two long digital segments, perhaps replantation should start from the little finger or ring finger to ensure a wide first commissure (Fig. 2).3
Figure 2.
A Clinical preoperative presentation, B radiologic preoperative view, C clinical immediate postoperative static result (thumb reconstruction using ring finger used as sparing part; reduction and fixation of middle finger with longitudinal K-wires; avoiding reconstruction of the little finger), D clinical 6 months postoperative static result (radial forearm flap for volar surface of middle finger), E radiologic 6 months postoperative view, and F functional 6 months postoperative two fingers pinch.
Soft Tissue Coverage
Once all injured structures have been repaired and reconstructed, the last priority in the management of an amputated or mangled hand is to provide a stable and definitive skin cover over the hand that provides protection, a gliding surface for the underlying tendons, brings the limb to healing, and then achieves restoration of maximum function and cosmeticity of the hand.19
Options for coverage of residual loss of substance range from simple skin grafting (or dermal substitutes) to coverage by mobilization of local or free flaps if necessary (especially in cases of urgent need for noble tissue coverage and unavailability of local flaps).20
Muscle flaps allow existing cavities to be filled, and skin flaps are preferred when the possibility of secondary surgery is higher.21
Another option, especially in the case of heavily contaminated wounds, which require a second debridement within 24–48 hours, is the use of a temporary negative pressure cover that provides temporary wound control.22
Conflicts of Interest
No benefits in any form have been received or will be received related directly to this article.
References
- 1.Sabapathy S.R., Del Piñal F., Boyer M.I., Lee D.C., Sebastin S.J., Venkatramani H. Management of a mutilated hand: the current trends. J Hand Surg Eur Vol. 2022;47(1):98–104. doi: 10.1177/17531934211047760. [DOI] [PubMed] [Google Scholar]
- 2.Neumeister M.W., Brown R.E. Mutilating hand injuries: principles and management. Hand Clin. 2003;19(1):1–15, v. doi: 10.1016/s0749-0712(02)00141-5. [DOI] [PubMed] [Google Scholar]
- 3.del Piñal F. Severe mutilating injuries to the hand: guidelines for organizing the chaos. J Plast Reconstr Aesthet Surg. 2007;60(7):816–827. doi: 10.1016/j.bjps.2007.02.019. [DOI] [PubMed] [Google Scholar]
- 4.Baltzer H.L., Moran S.L. The biomechanical impact of digital loss and fusion following trauma: setting the patient up for success. Hand Clin. 2016;32(4):443–463. doi: 10.1016/j.hcl.2016.07.003. [DOI] [PubMed] [Google Scholar]
- 5.Rich N.M., Baugh J.H., Hughes C.W. Acute arterial injuries in Vietnam: 1,000 cases. J Trauma. 1970;10:359–369. doi: 10.1097/00005373-197005000-00001. [DOI] [PubMed] [Google Scholar]
- 6.Wilhelmi B.J., Lee W.P., Pagenstert G.I., May J.W. Replantation in the mutilated hand. Hand Clin. 2003;19:89–120. doi: 10.1016/s0749-0712(02)00137-3. [DOI] [PubMed] [Google Scholar]
- 7.Brown H.C., Williams H.B., Woolhouse F.M. Principles of salvage in mutilating hand injuries. J Trauma. 1968;8:319–332. doi: 10.1097/00005373-196805000-00002. [DOI] [PubMed] [Google Scholar]
- 8.Bernstein M.L., Chung K.C. Early management of the mangled upper extremity. Injury. 2007;38(Suppl 5):S3–S7. doi: 10.1016/j.injury.2007.10.038. [DOI] [PubMed] [Google Scholar]
- 9.Warrender W.J., Lucasti C.J., Chapman T.R., Ilyas A.M. Antibiotic management and operative debridement in open fractures of the hand and upper extremity: a systematic review. Hand Clin. 2018;34(1):9–16. doi: 10.1016/j.hcl.2017.09.001. [DOI] [PubMed] [Google Scholar]
- 10.Ketonis C., Dwyer J., Ilyas A.M. Timing of debridement and infection rates in open fractures of the hand: a systematic review. Hand (N Y) 2017;12(2):119–126. doi: 10.1177/1558944716643294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gupta A., Shatford R.A., Wolff T.W., Tsai T.M., Scheker L.R., Levin L.S. Treatment of the severely injured upper extremity. J Bone Joint Surg Am. 2000;81(A):1628–1651. [PubMed] [Google Scholar]
- 12.Houdek M.T., Wagner E.R., Rizzo M., Moran S.L. Metacarpophalangeal joint arthroplasty in the setting of trauma. J Hand Surg Am. 2015;40(12):2416–2420. doi: 10.1016/j.jhsa.2015.09.012. [DOI] [PubMed] [Google Scholar]
- 13.Xie F., Lan X., Lin J. Arthroplasty for treating traumatic metacarpophalangeal joint defects: a retrospective study over three years. J Pain Res. 2021;14:1457–1464. doi: 10.2147/JPR.S299135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Brunelli G.A., Battiston B., Vigasio A., Brunelli G., Marocolo D. Bridging nerve defects with combined skeletal muscle and vein conduits. Microsurgery. 1993;14(4):247–251. doi: 10.1002/micr.1920140407. [DOI] [PubMed] [Google Scholar]
- 15.Cho M.S., Rinker B.D., Weber R.V., et al. Functional outcome following nerve repair in the upper extremity using processed nerve allograft. J Hand Surg Am. 2012;37(11):2340–2349. doi: 10.1016/j.jhsa.2012.08.028. [DOI] [PubMed] [Google Scholar]
- 16.Tos P., Battiston B., Ciclamini D., Geuna S., Artiaco S. Primary repair of crush nerve injuries by means of biological tubularization with muscle-vein-combined grafts. Microsurgery. 2012;32(5):358–363. doi: 10.1002/micr.21957. [DOI] [PubMed] [Google Scholar]
- 17.Riah Y., Balaguer T., Médard de Chardon V., Chignon-Sicdard B., Dannan E., Lebreton E. The use of the "spare-parts tissue bank" concept in emergency hand surgery. Chir Main. 2010;29(1):23–31. doi: 10.1016/j.main.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 18.Chuinard R.G., Dabezies E.J., Mathews R.E. Two-stage superficialis tendon reconstruction in severely damaged fingers. J Hand Surg Am. 1980;5(2):135–143. doi: 10.1016/s0363-5023(80)80144-4. [DOI] [PubMed] [Google Scholar]
- 19.Ng Z.Y., Salgado C.J., Moran S.L., Chim H. Soft tissue coverage of the mangled upper extremity. Semin Plast Surg. 2015;29(1):48–54. doi: 10.1055/s-0035-1544170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Del Piñal F. An update on the management of severe crush injury to the forearm and hand. Clin Plast Surg. 2020;47(4):461–489. doi: 10.1016/j.cps.2020.06.003. [DOI] [PubMed] [Google Scholar]
- 21.Del Piñal F., Pisani D., García-Bernal F.J., Regalado J., Del Pino F.J., Ayala H. Massive hand crush: the role of a free muscle flap to obliterate the dead space and to clear deep infection. J Hand Surg Br. 2006;31(6):588–592. doi: 10.1016/j.jhsb.2006.07.009. [DOI] [PubMed] [Google Scholar]
- 22.Cherubino M., Valdatta L., Tos P., et al. Role of negative pressure therapy as damage control in soft tissue reconstruction for open tibial fractures. J Reconstr Microsurg. 2017;33(S 01):S08–S13. doi: 10.1055/s-0037-1606542. [DOI] [PubMed] [Google Scholar]