Abstract
Background:
Understanding interventions preceding death in children with immunocompromised conditions is important to ensure a peaceful and dignified perideath experience. The aim of this study was to describe the number of interventions performed in the pediatric intensive care unit (PICU) within the 48 hours before death in this population.
Methods:
This was a single-center, retrospective cohort study of all children with an underlying oncologic, hematologic, or immunologic diagnosis admitted to the PICU for at least 72 hours between 2014 and 2021. Medical records were reviewed for interventions within 48 hours preceding death and for palliative care involvement. Interventions were defined as new or escalations in respiratory support, cardiopulmonary resuscitation (CPR), vascular access, drains, and radiographic studies. Associations were evaluated using simple logistic regression.
Results:
A total of 55 patients were included in this study. The predominant PICU admission diagnoses were respiratory (51%), followed by shock (25%), and neurologic diagnoses (9%). These predominant diagnoses were similar perideath. At PICU admission, only 1 patient had a do-not-resuscitate (DNR) order. Forty-six percent had a DNR order 48 hours preceding death, and 91% had DNR orders in place at time of death. During the 48-hour period preceding death, 80% of children received at least one intervention. Radiographic studies were the most common, used in 78% of children, followed by respiratory (20%), vascular (16%), CPR (13%), and drain placement (7%). Palliative care was involved in 38% of cases and was associated with a decrease in the number of radiologic interventions (p = 0.028) and CPR (p = 0.026).
Conclusions:
Children in the PICU with underlying immunocompromised conditions frequently receive interventions within the 48-hour period preceding death. Palliative care involvement was associated with fewer radiographic studies and fewer occurrences of CPR. The impact of interventions on the dying experience warrants further investigation.
Keywords: critical care, immunocompromised host, neoplasms, palliative care, pediatrics, terminal care
Introduction
Mortality rates among children admitted to the pediatric critical care unit with underlying oncologic, hematologic, and immunologic conditions, while improving, remain high. With curative measures being the main objective of pediatric intensivists, the ability to transition to comfort care for the dying child can be difficult for parents and medical providers alike.1–3 This shift should not only focus on the child's quality of remaining life but also on creating a good death experience. While the concept of a good death has been well investigated and represented in the adult literature,4 gaps remain in understanding the end-of-life needs specific to children—particularly those admitted to pediatric critical care units.5
Despite the lack of literature regarding the management of a good death in children, several studies have identified the main components that parents have outlined as being important: symptom management and pain control, communication, privacy with fewer interruptions and interventions, and the avoidance of a protracted death.6–8 The extent to which these factors impact end-of-life care in pediatric critical care medicine has not been well described and warrants further investigation. Certain pediatric populations, such as those with underlying oncologic and hematologic conditions, receive frequent interventions at the end of life when admitted to critical care units.9,10 The pediatric intensivist is almost always initially focused on life-sustaining measures. However, as critical illness progresses, they can be tasked with choosing interventions that focus on enhancing a child's quality of life and foregoing interventions that may cause unnecessary suffering. An understanding of the types of interventions preceding death in children with underlying oncologic and hematologic conditions admitted to the critical care unit is important in alleviating pain and suffering. Further recognizing the types and volume of interventions in the perideath period is the start of understanding the impact on a dignified death and grieving experience family members.4,6,11 This study aims to describe the number and types of interventions performed in the pediatric intensive care unit (PICU) within the 48-hour period preceding death of the immunocompromised child and to investigate the relationship between palliative care involvement on the number and type of these interventions. Understanding the landscape of interventions in the perideath experience is the first step toward identifying if these interventions hinder a peaceful death in children.
Methods
A retrospective cohort study was conducted at a large pediatric academic medical institution. Institutional board approval was obtained before the start of the study. Inclusion criteria included all patients with an active underlying oncologic, hematologic, or immunodeficiency diagnosis who died in the pediatric intensive care unit between January 1, 2014, and December 31, 2021. Specific definition of immunodeficiency included primary or acquired immunodeficiency or receiving immunosuppressive or immunomodulating therapy. This data range was selected based on the approximate implementation and activation of the institution's palliative care program. Subjects were excluded if (1) the duration of admission before death was less than 72 hours or (2) the admission was predesignated as being for provision of end-of-life care. The criteria for admission duration were selected for a few reasons. First, the 72-hour minimum was chosen to provide both the time and opportunity for the adequate development of a provider–patient relationship. We excluded those admitted specifically for end-of-life care because the mindset of clinicians for the type of care we provide is different. In our institution, these patients are often admitted to the PICU due to higher nursing or respiratory therapy needs to provide comfort that may be challenging with staffing on the wards. With this study, we were specifically interested in examining the interventions in patients where providers need to make the challenging shift in mindset from lifesaving to quality and comfort care.
Data were obtained from our local Virtual Pediatric Systems (VPS) Clinical Data Abstractor and from review of the electronic medical record. VPS (www.myvps.org) is a national database for pediatric critical care that houses data aimed at improving outcomes. Individual centers across the country contribute data. For this study, we only used our institution's data supplemented with additional data from our electronic medical record. In addition to demographics, patient data collected on admission included the underlying oncologic/hematologic/immunologic diagnosis, the primary reason for critical care admission and code status. The primary critical care diagnosis and code status were again collected 48 hours preceding death and at time of death. The primary critical care diagnosis was collected from VPS in their standardized and validated manner and was confirmed on review of each patient's medical record.
Both the number and type of interventions were collected within the 48-hour period preceding death. A 48-hour period was used to allow adequate time for realization of disease trajectory and to provide the opportunity to redirect management to comfort-directed rather than curative. Interventions were defined as those that were (1) new or escalated within the 48-hour period preceding death, (2) provided for curative rather than comfort/palliative measures, and (3) clearly documented as a procedure or traceable in the institution's electronic medical record system. Every new intervention or escalation of therapy in each patient was counted. Interventions that were documented to be used solely for palliation or patient comfort were excluded. Details of the specific interventions that were included in the study are detailed and categorized in Table 1. Briefly, we defined interventions as new or escalated respiratory support, any invasive or painful procedures, radiographic studies, or organ support therapies such as dialysis. These interventions were defined a priori. Some of the defined interventions were not experienced by any of the included subjects. Interventional data were collected and further combined into the following subcategories for analysis: imaging, respiratory, lines/access, neurologic, cardiovascular, gastrointestinal, and hematologic. Cardiopulmonary resuscitation (CPR) was designated as its own subcategory for data analysis. Limitations of care including “do not resuscitate” (DNR) orders were noted at PICU admission, 48 hours preceding death, and at time of death. At our institution, during the study period, limitations of care and DNR orders were most commonly obtained by the pediatric intensivist and less frequently by the palliative care team.
Table 1.
Interventions Included in Data Search and Collection
| Category | Intervention |
|---|---|
| Respiratory | High-flow nasal cannula Bilevel positive airway pressure Continuous positive airway pressure Invasive mechanical ventilation High-frequency oscillatory ventilation Prone positioning Bronchoscopy Thoracentesis Chest tube placement Tracheostomy Venovenous extracorporeal membrane oxygenation |
| Cardiovascular | Cardiopulmonary resuscitationa Electrocardiogram Venoarterial extracorporeal membrane oxygenation Pericardiocentesis Catheterization Pacing device placement |
| Neurologic | Electroencephalogram Lumbar puncture Ventriculostomy Intracranial pressure monitor placement External ventricular drain placement |
| Gastrointestinal and renal | Paracentesis (aspiration, drain placement) Foley catheter placement Nephrostomy tube placement Hemodialysis Peritoneal dialysis Continuous renal replacement therapy |
| Hematologic | Bone marrow biopsy Exchange transfusion Pheresis |
| Vascular | Central venous line Peripherally inserted central line Vascaths Arterial lines Intraosseous line |
| Imaging | X-ray imaging Computed tomography Magnetic resonance imaging Ultrasounds Echocardiograms |
Delegated as separate category for data analysis.
Active or established palliative care involvement was defined a priori by the presence of at least three clinical documents within the week preceding death. The criteria to determine active palliative care involvement were selected to allow time for adequate development of a provider–patient relationship and gain a more thorough understanding of the patient's and family's values, beliefs, and concerns, to effectively help represent the family in end-of-life decision-making.
Data are presented as medians with interquartile ranges (IQR) for continuous variables and counts with proportions for categorical variables. The association of palliative care involvement with interventions performed was assessed using the chi-square or Fisher's exact test where appropriate, or otherwise, with a simple logistic regression model. Spearman rank correlation was used to test correlation between continuous variables. The analysis was performed using Stata software (StataCorp LLC, College Station, TX).
Results
A total of 55 patients were identified according to the inclusion criteria. Demographics are found in Table 2. The median age was 7.7 (IQR: 1.8, 15.7) years at time of death. The number of males and females was nearly equivocal, with the majority of the patients being Caucasian and English-speaking. The median length of PICU stay was 17.6 days (IQR: 7.9, 27.9). Leukemia was the most common underlying diagnosis found in 35% (n = 19) of patients, followed by solid tumors and immunodeficiencies. Over a third of the cohort (36%; n = 20) had a history of receiving a hematopoietic cell transplant, and 3 patients (5%) had received solid organ, multivisceral transplants. The predominant indication for PICU admission was respiratory failure (51%; n = 28) followed by shock (25%; n = 14).
Table 2.
Patient Demographics
| Demographic | Median (IQR) or n (%) |
|---|---|
| Mean age | 7.7 (IQR: 1.8, 15.7) years |
| Gender | |
| Female | 31 (56%) |
| Male | 24 (44%) |
| Race | |
| Caucasian | 40 (73%) |
| African American | 6 (11%) |
| Hispanic/Latino | 5 (9%) |
| Other | 4 (7%) |
| Primary spoken language | |
| English | 48 (87%) |
| Spanish | 4 (7%) |
| Other | 3 (5%) |
| Underlying diagnosis | |
| Leukemia | 19 (35%) |
| Solid tumor | 12 (22%) |
| Immunodeficiency | 9 (16%) |
| Brain tumor | 5 (9%) |
| Hematologic | 4 (7%) |
| Lymphoma | 3 (5%) |
| Congenital | 3 (5%) |
| Transplant | |
| Stem cell | 20 (36%) |
| Multivisceral | 3 (5%) |
| Palliative care involvement | |
| No | 34 (62%) |
| Yes | 21 (38%) |
IQR, interquartile ranges.
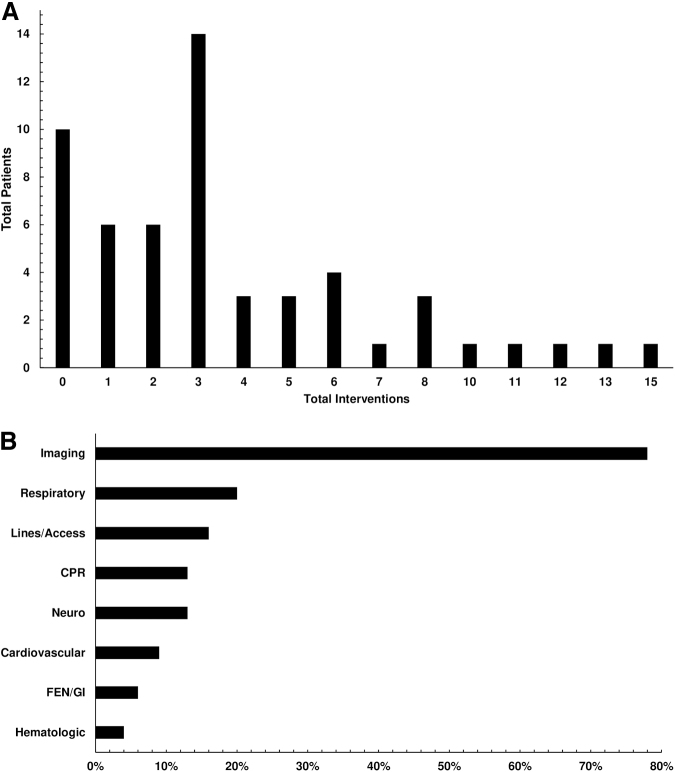
During the 48-hour period preceding death, 81% (n = 45) of children received at least one intervention, with a median of three interventions (IQR: 1, 5) per patient. Ten patients (18.2%) received no interventions, and 1 patient (2.0%) received 15 total interventions (Fig. 1A). Imaging studies were the most commonly received intervention, with 78% of children (n = 43) receiving at least one imaging study. Respiratory interventions were the second most common (20%), followed by placement of vascular access (16%). CPR was performed at least once in 13% (n = 7) of the study population (Fig. 1B). Of these patients, n = 3/7 were transitioned to a DNR after successful CPR with return of spontaneous circulation but before death. Length of PICU stay was not associated with having interventions in the 48 hours preceding death (OR = 0.98, 95% CI: 0.95–1.02, p = 0.368), nor was it correlated with the number of interventions done in the 48-hour period (Spearman's rho = 0.19 p = 0.162).
FIG. 1.
(A) Total number of interventions per patient. (B) Percentage of patients who had an intervention within 48 hours preceding death.
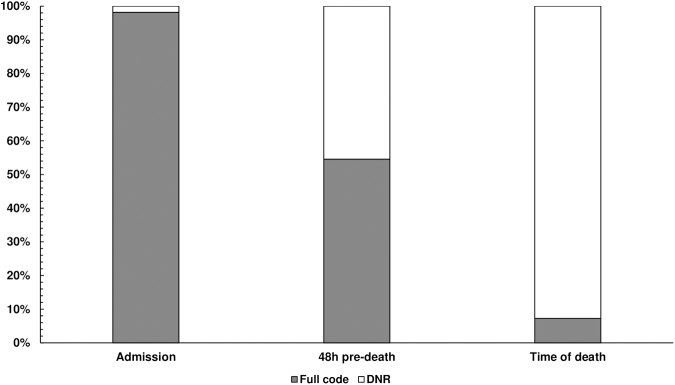
On PICU admission, only one patient had a limitation of care order. This increased as patients neared the time of death, with almost half of the patients (n = 25) having a “do not resuscitate” (DNR) order at 48 hours preceding death, and the majority of patients (n = 51) having DNR orders at time of death (Fig. 2). Those with code limitations at 48 hours preceding death had a lower median number of interventions: 2 (IQR: 1, 3) compared to those with no care limitations with a median of 4.5 interventions (IQR: 3, 8), p = 0.0004. PICU length of stay was not associated with having a DNR at 48 hours preceding death (OR = 1.00, 95% CI: 0.99–1.01, p = 0.916).
FIG. 2.
Code status at admission, 48 hours preceding death, and at time of death.
Palliative care was actively involved in 38% (n = 21) of patients (Table 3). Established palliative care involvement was associated with less radiographic imaging (p = 0.028; OR = 0.22 CI 95% 0.06–0.85). None of the patients with palliative care involvement received CPR (p = 0.026). Every child with palliative care involvement had a DNR in place at time of death, compared with 71% (n = 24) of those without established palliative care. Children with palliative care involvement received a median of two interventions (IQR: 0, 5) compared to those with no palliative care involvement who receive a median of three interventions (IQR: 2, 5); however, this did not reach statistical significance (p = 0.160). There was no association between PICU length of stay and active palliative care involvement (OR = 1.00, 95% CI: 0.99–1.01, p = 0.891).
Table 3.
Differences in Select Interventions Among Immunocompromised Children Admitted to the Pediatric Critical Care Unit With Receipt of Palliative Care Services Versus Those Without
| Intervention | (+) Palliative n = 21 | (−) Palliative n = 34 | Statistic |
|---|---|---|---|
| Radiographic imaging | 7 (31%) | 24 (72%) | OR = 0.22 (95% CI: 0.06–0.85), p = 0.028 |
| Do not resuscitate orders at time of death | 21 (100%) | 24 (71%) | p = 0.006a |
| Cardiopulmonary resuscitation | 0 (0%) | 7 (21%) | p = 0.026a |
As no patient with palliative care involvement received cardiopulmonary resuscitation, a logistic model could not be run for this variable. Models were unadjusted.
Discussion
Patients with underlying immunocompromised conditions frequently receive interventions within the 48 hours preceding death. In our cohort, palliative care involvement decreased the number of radiographic interventions. There was also a notable effect on the rates of DNR orders and CPR at the time of death in those with established palliative care. Furthermore, instituting code limitations would seem to impact the number of interventions performed, as those with a DNR status at 48 hours preceding death had significantly fewer interventions than those without a DNR status.
There is pediatric data to suggest that interventions preceding a patient's death can be seen as disruptive or detrimental to the optimal death experience—oftentimes inflicting further discomfort, consuming valuable quality time for patients and their families, and depriving the patient of an otherwise dignified death.6,12 While our study did not examine the impact of interventions to the overall death experience, our cohort did demonstrate that multiple interventions are being performed in the period preceding death. Future studies should investigate not only if the number of interventions being done impact parental perception of the death experience but also if specific interventions are viewed more negatively that others. There may be opportunities to minimize potentially excessive and burdensome interventions. As suggested by previous literature,6,7,13–15 clear communication and judicious use of interventions may contribute to an improved death experience by (1) providing both privacy and time for the patient and family to prepare, and (2) avoiding prolonged suffering that may otherwise contribute to a protracted death. The characteristics of our cohort help to drive the importance of this. The median length of stay in our cohort was 18 days with over 75% of the cohort having a length of stay longer than a week. These children are likely moving from acute critical illness into a more chronically critically ill state. It can be extreme difficulty for clinical care teams and families to switch a care plan from aggressive interventions with the intent of survival toward a plan of care that emphasizes comfort. Timing of this transition is incredibly challenging. Our study highlights the urgent need to better understand how the multiple interventions being done perideath impact family perceptions of the overall death experience. A better understand may lead to improved shared decision-making as a patient nears death and improve experience for both patients and families.
Invasive interventions performed in the PICU extend beyond those that are painful or physically invasive, such as line placements and intubations. Interventions such as chest X-rays and other forms of imaging, although not painful, are disruptive of the time spent with patients and their families. These interventions require staff and equipment to enter the room, repositioning the patient which can cause discomfort and may even require travel out of the PICU. Similarly, interventions such as dialysis and electroencephalograms often limit the interactions and expressions of intimacy that parents are able to provide to their child. In our cohort, palliative care involvement was associated with a reduced number of radiographic interventions during the 48-hour period preceding death. The reason for this association is unclear. Established palliative care providers provide a source of trust and continuity for patients and their families. They may serve as trusted voices in facilitating difficult conversations and preparing families for medical decisions regarding end-of-life care. Furthermore, when palliative care is already established, the family may have already had the opportunity to consider goals and limits of care in the setting of critical illness. These factors may be the reason we found an association between fewer radiographs, higher rates of DNR orders, and less CPR in children with established palliative care.
These findings align with previous studies demonstrating that children who receive palliative care services have been found to undergo fewer procedures, have a decreased length of hospitalization, and better pain/symptom control.9,10,16–18 Such findings hinge on palliative care services being properly utilized in a timely manner. It is known that palliative care is underutilized and lack of access is a problem globally.19 Although our study did not directly examine the relationship between the number of end-of-life interventions and total duration of palliative care involvement, our data, in conjunction with existing data, suggest that (1) earlier palliative care involvement may allow for more time to develop a meaningful relationship between providers and families, (2) a longer palliative care relationship may allow for more time for discussion and preparation, and (3) with appropriate time to prepare, families can focus on quality of time and experiences during the end-of-life period; therefore, being more likely to forego unnecessary medical interventions.
In our cohort, none of the patients with palliative care involvement received CPR. While it is difficult to determine the exact reason for this retrospectively, it is likely that ongoing and early conversations regarding goals of care play a role. It is also possible that the children who died without palliative care involvement had been admitted to the PICU for a potentially reversible, but unsuccessfully treated, condition. However, at the time of death, all patients with palliative care involvement had a DNR in place, compared to only 71% of patients without palliative care involvement. Conversations regarding code status remain infrequent in pediatrics, with only 30–40% of children having a code status in place at the time of death.20 The absence of a code status necessitates hurried conversations during clinical emergencies. This often occurs with an unfamiliar provider, which may increase the likelihood of unwanted interventions.20 Similar to our study, previous studies have demonstrated that conversations regarding resuscitation status are more likely to be initiated both earlier and more effectively with the establishment of a relationship with palliative care services.11 Early conversations with established providers allow for a shift in focus from curative to comfort measures, avoiding aggressive treatments and interventions.11,20,21 Such evidence should prompt pediatric intensivists to consider consultation with palliative care providers earlier in a patient's PICU admission course. While the majority of the children in our cohort had DNR orders at the time of death, less than half had care limitations in place 48 hours before death, suggesting that many care-limitation conversations occurred very close to the time of death.
Our cohort of immunocompromised patients represents a population with unique needs. Many experience prolonged hospital stays and require considerable health care resources. These are highly complex patients. Palliative care was only actively involved in 38% of cases. This suggests a missed opportunity for more intentional palliative care involvement. There is increasing evidence supporting the integration of pediatric palliative care services for patients with serious illness, such as those in our study.22 This is even more important in light of our data supporting the impact palliative care had on interventions and DNR status at time of death. However, the optimal way to implement palliative care services for patients in the PICU is more difficult to define. While there is no clear consensus regarding the timing of introductory palliative care services for these patients in the PICU, the known benefits to those with established palliative care relationships would suggest earlier introduction for maximum benefit. Given the high acuity and rapidly changing trajectories of these patients in the PICU, effective and cohesive communication is key to facilitating advanced-care planning conversations. A previous study demonstrated that children with complex medical needs who received palliative care services were more likely to receive multidisciplinary care conferences and early exposure to advanced care planning than those without palliative care involvement.23
Limitations of this study include its retrospective nature. Only data that had already been collected and documented in the medical records at the time of review were available. While we excluded limitations specifically documented for comfort only, the retrospective aspect can be challenging to determine the intent and indications of certain interventions due to lack of documentation. It is even more challenging to retrospectively determine the impetus or lack thereof for the initiation of goals of care conversations. Another limitation is the definition of active palliative care. Because the parameters set to define an established palliative care relationship required three charted documents within the week preceding death, it is possible that the pool of patients with an established palliative care relationship was, in actuality, larger than that represented in this study. There is not a formal definition for active palliative care involvement. Our definition may be too restrictive thereby underestimating the effect of palliative care on the number of interventions perideath. Palliative care may have been consulted, but their involvement may have either lacked documentation or the involvement may have been brief. Additionally, it should be acknowledged that there is the possibility for changes in interventions among the patients who have had only one encounter with palliative care. Finally, over the eight-year study period, there was likely unmeasurable practice variability that may have affected the primary outcome of the interventions performed in the period preceding patient death. The single-center nature of the study likely limits some practice variability; however, it would be important to validate these findings in a larger multicenter study.
Conclusion
In this study of 55 children with immunocompromised conditions who died in a pediatric intensive care unit, the integration of palliative care services was associated with a decrease in interventions such as CPR and radiographic imaging studies. Despite palliative care integration, each of our patients received several interventions in the period preceding death. Future investigative considerations should be aimed at directly exploring the impact of the number of interventions performed on the end-of-life experience. Furthermore, almost half of our cohort did not have a comfort-directed resuscitation order in place 48 hours preceding death, prompting discussion regarding the need for goals of care conversations to be initiated earlier in the disease process and/or hospitalization course.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
C.M.R. is supported by The National Heart, Lung, and Blood Institute (K23HL150244).
References
- 1. Rubenfeld GD, Curtis JR. End-of-life care in the intensive care unit: A research agenda. Crit Care Med 2001;10:2001–2006. [DOI] [PubMed] [Google Scholar]
- 2. Suttle ML, Jenkins TL, Tamburro RF. End-of-life and bereavement care in pediatric intensive care units. Pediatr Clin North Am 2017;64(5):1167–1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Forsner M, Mattson J. Caring for dying children in the pediatric intensive care unit, nurses and parents' perspective. J Complement Altern Med 2019;1. [Google Scholar]
- 4. Campos-Calderon C, Montoya-Juarez R, Hueso-Montoro C, et al. Interventions and decision-making at the end of life: The effect of establishing the terminal illness situation. BMC Palliat Care 2016;7(1):91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Conte T, Mitton C, Trenaman LM, et al. Effect of pediatric palliative care programs on health care resource utilization and costs among children with life-threatening conditions: A systematic review of comparative studies. CMAJ Open 2015:E68–E75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Broden EG, Deatrick J, Ulrich C, et al. Defining a “good death” in the pediatric intensive care unit. Am J Crit Care, 2020;29(2):111–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Garros D. [A “good” death in pediatric ICU: Is it possible?]. J Pediatr (Rio J) 2003;79 Suppl 2:S243–S254. [DOI] [PubMed] [Google Scholar]
- 8. Meyer EC, Ritholz MD, Burns JP, et al. Improving the quality of end-of-life care in the pediatric intensive care unit: Parents' priorities and recommendations. Pediatrics 2006;117(3):649–657. [DOI] [PubMed] [Google Scholar]
- 9. Keele L, Keenan HT, Sheetz J, et al. Differences in characteristics of dying children who receive and do not receive palliative care. Pediatrics 2013;132(1):72–78. [DOI] [PubMed] [Google Scholar]
- 10. Osenga K, Postier A, Dreyfus J, et al. A comparison of circumstances at the end of life in a hospital setting for children with palliative care involvement versus those without. J Pain Symptom Manage 2016;52(5):673–680. [DOI] [PubMed] [Google Scholar]
- 11. Wolfe J, Hammel JF, Edwards KE, et al. Easing of suffering in children with cancer at the end of life: Is care changing? J Clin Oncol 2008;26(10):1717–1723. [DOI] [PubMed] [Google Scholar]
- 12. Balluffi A, Kassam-Adams N, Kazak A, et al. Traumatic stress in parents of children admitted to the pediatric intensive care unit. Pediatr Crit Care Med 2004;5(6):547–553. [DOI] [PubMed] [Google Scholar]
- 13. Truog RD, Meyer EC, Burns JP. Toward interventions to improve end-of-life care in the pediatric intensive care unit. Crit Care Med 2006;34(11 Suppl):S373–S379. [DOI] [PubMed] [Google Scholar]
- 14. Starr LT, Ulrich CM, Corey KL, et al. Associations among end-of-life discussions, health-care utilization, and costs in persons with advanced cancer: A systematic review. Am J Hosp Palliat Care 2019;36(10):913–926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Dussel V, Kreicbergs U, Hilden JM, et al. Looking beyond where children die: Determinants and effects of planning a child's location of death. J Pain Symptom Manage 2009;37(1):33–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sedig LK, Spruit JL, Southwell J, et al. Palliative care is not associated with decreased intensity of care: Results of a chart review from a large children's hospital. Pediatr Blood Cancer 2022;69(3):e29391. [DOI] [PubMed] [Google Scholar]
- 17. Friedrichsdorf SJ, Postier A, Dreyfus J, et al. Improved quality of life at end of life related to home-based palliative care in children with cancer. J Palliat Med 2015;18(2):143–150. [DOI] [PubMed] [Google Scholar]
- 18. Arland LC, Hendricks-Ferguson VL, Pearson J, et al. Development of an in-home standardized end-of-life treatment program for pediatric patients dying of brain tumors. J Spec Pediatr Nurs 2013;18(2):144–157. [DOI] [PubMed] [Google Scholar]
- 19. Radbruch L, De Lima L, Knaul F, et al. Redefining palliative care—A new consensus based definition. J Pain Symptom Manage 2020. 40:754–764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Battisti KA, Cohen DM, Bourgeois T, et al. A paucity of code status documentation despite increasing complex chronic disease in pediatrics. J Palliat Med 2020;23(11):1452–1459. [DOI] [PubMed] [Google Scholar]
- 21. Harmoney K, Mobley EM, Gilbertson-White S, et al. Differences in advance care planning and circumstances of death for pediatric patients who do and do not receive palliative care consults: A single-center retrospective review of all pediatric deaths from 2012 to 2016. J Palliat Med 2019;22(12):1506–1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Widger K, Sutradhar R, Rapoport A, et al. Predictors of specialized pediatric palliative care involvement and impact on patterns of end-of-life care in children with cancer. J Clin Oncol 2018;36(8):801–807. [DOI] [PubMed] [Google Scholar]
- 23. Spraker-Perlman HL, Tam RP, Bardsley T, et al. The impact of pediatric palliative care involvement in the care of critically ill patients without complex chronic conditions. J Palliat Med 2019;22(5):553–556. [DOI] [PubMed] [Google Scholar]