Abstract
Background
The outpatient parenteral antimicrobial therapy (OPAT) programme was introduced to reduce costs and enhance the quality of life for patients requiring prolonged treatment with parenteral antimicrobials. However, given the escalating inflation, the extent of current cost savings achieved through OPAT programmes remains unclear. This systematic review and meta-analysis employ a cost-minimization analysis to quantify the cost savings from OPAT compared to inpatient treatment.
Methods
The Cochrane Library, MEDLINE, Embase, PubMed and Web of Science databases were searched for studies comparing the costs of parenteral antimicrobial treatment without restriction on study design and year. Two reviewers conducted eligibility screening and cross-validated the extracted data. The cost data were adjusted and inflated to 2023 US dollars. A random effect model calculated mean differences (MD) with 95% confidence intervals (CI). The review protocol was registered on PROSPERO (CRD42024584201).
Results
Twenty studies involving 2790 patients were included in the systematic review, and six studies (three randomized controlled trials and three cohorts) were subject to metanalysis. Collectively, these included 560 patients who received treatment in outpatient settings, and 491 treated as inpatients. The cost of parenteral antimicrobial per episode of care was lower in the outpatient settings MD −$5436.73 (95% CI: −$9589.24 to −$1284.22, I² = 96%; P = 0.01) than in inpatient settings.
Conclusions
OPAT significantly saves costs compared to inpatient treatment. We recommend comprehensive analysis of treatment costs from all perspectives, including various cost types.
Introduction
Infections are a major public health challenge worldwide.1 Traditionally, serious infections have been treated with parenteral antimicrobials administered in a hospital environment. However, there has been a broad rise in infections leading to increased hospitalization rates, straining healthcare facilities, and elevating the risk of nosocomial infections.2 Avoiding hospitalization or facilitating early discharge requires the implementation of an effective stewardship programme.3
Outpatient parenteral antimicrobial therapy (OPAT) is a care delivery modality that avoids hospitalization.4,5 Since its implementation in 1974,6 the programme has been offered in various ambulatory settings, including infusion centres, hospital-based clinics, skilled nursing facilities and patient’s homes.7 It is indicated for patients in clinically stable conditions who have the necessary support.8 The disease conditions treated in the programme include skin and soft tissue infections, bone and joint infections, bacteraemia, wound infections, urinary tract infections, intra-abdominal infections and central nervous system infections.9
Treating these conditions in outpatient settings is both safe and effective compared to traditional inpatient treatments.10 OPAT is associated with high patient satisfaction, as it allows patients to receive treatment in the comfort of their homes, engage with family during treatment and maintain their daily routines.11 Additionally, OPAT reduces costs by freeing up hospital beds, thereby improving patient flow and increasing the capacity for new admissions.12 It also minimizes the income and productivity loss associated with prolonged hospitalization by facilitating an earlier return to work.13–17 Even though the savings from the OPAT programme vary depending on the type of patients, the healthcare system, payers and society,18 there is potential for further cost reductions through the advancements of OPAT models of care.19 However, there is a lack of a clear description regarding the additional cost savings of the OPAT programme. Consequently, the extent of cost savings from the OPAT programme compared to inpatient therapy remains uncertain.3
Psaltikidis et al. conducted a review in 2017 to evaluate the cost savings of the OPAT programme. However, the studies included in their review were heterogeneous, and the currency was neither adjusted for inflation nor standardized to a common currency, which precluded a robust meta-analysis.20 Thus, there is a lack of reviews that have pooled cost data comparing the savings associated with OPAT to those from inpatient treatment. Such economic evaluations are crucial for delivering optimal care while ensuring financial viability within the healthcare system. Therefore, this systematic review and meta-analysis assessed the cost savings of OPAT compared to inpatient treatment by inflating and adjusting to a common currency.
Methods
Search strategy
A systematic search was conducted across the Cochrane Library, MEDLINE, Embase, PubMed and Web of Science databases. A manual search of the reference lists of included studies and review articles was also conducted to identify further relevant literature. The complete search strategy is detailed in Table S1 (available as Supplementary data at JAC-AMR Online). No restrictions were imposed on language, publication year, or format. This review adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines,21 and the protocol was registered on PROSPERO (CRD42024584201).
Study selection
Studies were included if they compared the cost, cost–effectiveness, cost–benefit, or cost–utility of inpatient parenteral antimicrobial therapy with OPAT. Studies on OPAT included both early discharge facilitation and admission avoidance. The study population included all patients receiving parenteral antimicrobials in outpatient or inpatient settings. Patients receiving parenteral antimicrobials in the outpatient setting began their treatment either while admitted to the hospital or at the emergency department, and subsequently completed their treatment in an ambulatory setting, such as fixed clinics, emergency departments, or their own homes. Inpatient patients were those who completed their treatment while remaining in hospital beds.
Studies that relied on assumption-based imputation and those that did not distinguish between oral and parenteral antimicrobial treatment were excluded. Additionally, non-peer-reviewed studies, abstracts and reviews were excluded.
Screening of studies
Two independent reviewers (S.A.M. and G.M.A.) screened the abstracts and full-text studies based on predefined criteria. The screening process was facilitated by Covidence (Veritas Health Innovation, Melbourne, Australia). Any conflicts between the reviewers were resolved through discussion, involving a third reviewer, F.S., when necessary. The Kappa statistic was used to assess and report the agreement between the two reviewers.
Data extraction
Data were extracted using a standardized form, which included study characteristics (year, country, design, authors, currency type, type of economic evaluation, disease condition and antimicrobials), participant characteristics (number, age, gender) and study outcomes (cost, days of hospitalization avoided and health outcomes). The studies defined health outcomes differently, and these were extracted as reported. One reviewer (S.A.M.) performed the data extraction, while the senior reviewer (F.S.) verified the extracted data.
Risk of bias assessment
One reviewer (S.A.M.) assessed the risk of bias of non-randomized studies using Newcastle–Ottawa Scale,22 while the Cochrane risk of bias assessment tool was used for randomized studies.23
Data analysis
A cost-minimization analysis was conducted due to the similar clinical outcomes between OPAT and inpatient treatment.10 The results of the review were summarized as follows: (i) the percentage of cost savings per patient for OPAT was calculated by subtracting the average OPAT cost from the average hospitalization cost and dividing by the average hospitalization cost; (ii) the overall average cost savings percentage was determined by dividing the total OPAT cost savings from all studies by the number of studies; and (iii) the average number of hospitalization days avoided was calculated by dividing the total number of hospitalization days averted by the total number of patients or treatment episodes. Detailed calculations and the extracted cost data are presented in Table S2.
The cost data reported across different years and currencies were standardized to a target currency (USD) and year (2023) through a two-stage process.24 In the first stage, the original cost data are adjusted from its initial price year to the target price year using the International Monetary Fund (IMF) Gross Domestic Product (GDP) Deflator Index to account for inflation. For studies that did not specify a price year, the last year of patient recruitment was used as the reference price year. In the second stage, the price–year-adjusted cost data are converted from the original currency to the target currency using the Purchasing Power Parities (PPP) for GDP from the IMF World Economic Outlook Database. The PPP adjusts for differences in price levels between countries.24 This cost standardization was performed using a web-based tool, CCEMG–EPPI Centre Cost Converter (v.1.7).
Studies that reported the mean and standard deviation (SD) of cost data were included in the meta-analysis. The SD was calculated for studies that provided the standard error of the mean. The data were extracted as presented and incorporated into the narrative for studies reporting median and range. The mean cost difference for parenteral antimicrobial treatment was calculated using the Meta package in R software (version 4.3.0) (Posit Software, Vienna, Austria). A random effects model (Mantel–Haenszel method) was employed for the analyses.25 The I-squared statistic was used to assess heterogeneity among included studies, with a value greater than 50% indicating substantial heterogeneity.26 Subgroup analyses were conducted to evaluate variations in OPAT care models: S-OPAT (self or carer-administered OPAT at patients’ homes), H-OPAT (physician or nurse-administered OPAT at patients’ homes) and C-OPAT (outpatient clinic or infusion centre administration). A sensitivity analysis was performed to test the robustness of the primary analysis by restricting it to studies that evaluated similar types of costs (direct and indirect), cost perspectives (health care provider, payer and society), population (adult and paediatric), ways of hospitalization avoidance (admission avoidance and facilitation of discharge) and those published after 2000. Publication bias could not be assessed because a funnel plot requires at least 10 studies.26
Results
Search results
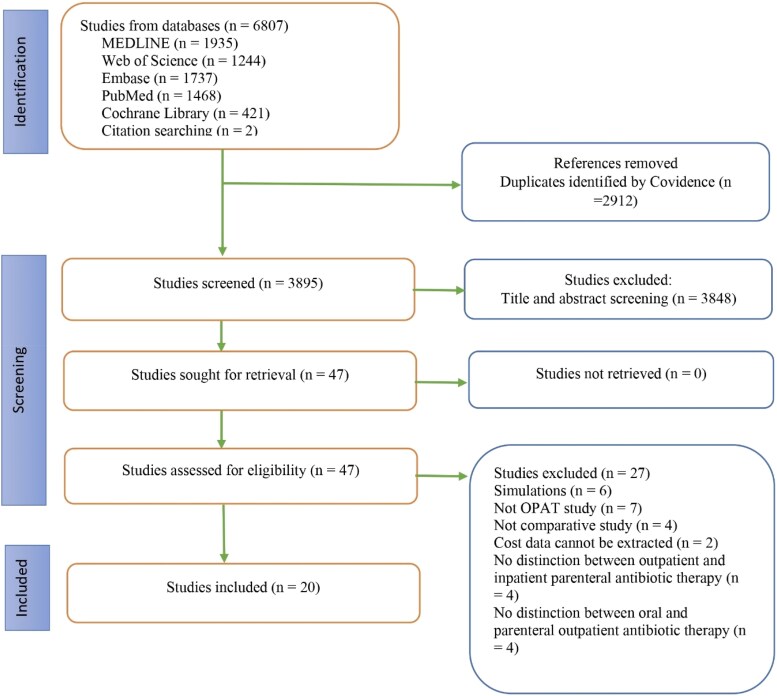
The screening process for the studies is presented in Figure 1. A total of 6807 studies were identified from all databases. After removing 2912 duplicate studies, 3895 were screened for title and abstract. The full length of 47 studies was assessed, and 20 were included in the final analysis.27–46 The level of agreement between the two reviewers was 70.25% for title and abstract screening and 95.98% for full-length screening. The list of excluded studies and the reasons for their exclusion is presented in Table S3.
Figure 1.
Study eligibility screening.
Characteristics of included studies
The characteristics of the included studies are presented in Table 1. These studies were conducted between 197828 and 2022,44 with two-thirds taking place in Australia32–35,42,43 and the USA.28–31,39–41 Among the studies, five were randomized controlled trials (RCTs),27,31,34,42,43 eight were prospective cohorts28–30,33,37,39,40,46 and seven were retrospective cohorts,32,35,36,38,41,44,45 involving a total of 2790 participants. The average percentage of male participants was 51.87% in the OPAT group and 47.49% in the inpatient group. The mean age of participants was 31.88 years in the OPAT group and 32.08 years in the inpatient group.
Table 1.
Characteristics of included articles
| Author and year | Country | Study design | Qualitya | Number of participants | Male % | Mean age | |||
|---|---|---|---|---|---|---|---|---|---|
| OPAT | IPAT | OPAT | IPAT | OPAT | IPAT | ||||
| Hendricks 201131 | USA | RCT | Some concern | 35 | 57 | 54 | 29 | 47.1 | 46.1 |
| Hensey 201732 | Australia | Retrospective cohort | 8 | 41 | 130 | 61 | 30.8 | NR | NR |
| Martel 199437 | Canada | Prospective cohort | 8 | 33 | 17 | NR | NR | NR | NR |
| Antoniskis 197828 | USA | Prospective cohort | 9 | 13 | 7 | 61.5 | 57.1 | 37.2 | 52.7 |
| Kameshwar 201635 | Australia | Retrospective cohort | 8 | 124 | 204 | 64.5 | 54.9 | 59b | 58.5b |
| Donati 198729 | USA | Prospective cohort | 9 | 26 | 38 | 46.1 | 47.4 | 23.3 | 23.3 |
| Warner 199841 | USA | Retrospective cohort | 8 | 120 | 122 | 67.6 | 55.14 | 11.2 | 11.5 |
| Yong 200845 | Singapore | Retrospective cohort | 9 | 69 | 93 | 52.8 | 50.5 | 53 | 56 |
| Ramasubramanian 201838 | India | Retrospective cohort | 8 | 90 | 10 | 43 | 56.2 | ||
| Wolter 200443 | Australia | RCT | High | 44 | 38 | 45.5 | 34.2 | 43b | 49b |
| Ahmed 200727 | Egypt | RCT | High | 61 | 58 | 59 | 60.3 | 7.2 | 6.3 |
| Talcott 199440 | USA | Prospective cohort | 7 | 30 | 27 | 43.3 | NR | 38b | NR |
| Fishman 200030 | USA | Prospective cohort | 8 | 52 | 98 | NR | NR | 10.2 | |
| Lacroix 201436 | France | Retrospective cohort | 7 | 18 | 21 | 11 | 14 | 59.5 | 67.5 |
| Wolter 199742 | Australia | RCT | High | 13 | 18 | 39 | 28 | 22b | 22b |
| Ibrahim 201733 | Australia | Prospective cohort | 7 | 47 | 68 | 55.3 | 58.9 | 6.3 | 6.3 |
| Yadev 202244 | Canada | Retrospective cohort | 7 | 341 | 239 | 59.8 | 51.9 | 61.7 | 65 |
| Ibrahim 201934 | Australia | RCT | High | 89 | 91 | 60 | 47 | 7 | 7.2 |
| Stovroff 199439 | USA | Prospective cohort | 7 | 8 | 8 | 87.8 | 100 | 9.1 | 9 |
| Pena 201346 | Chile | Prospective cohort | 8 | 111 | 81 | 22.5 | 40.7 | 1.17 | 0.83 |
OPAT, outpatient parenteral antimicrobial therapy; IPAT, inpatient parenteral antimicrobial therapy; NR, not reported; RCT, randomized clinical trial; USA, United States of America.
aNewcastle–Ottawa Quality Assessment Scale for non-randomized studies and Cochrane risk of bias assessment tool for randomized studies.
bMedian.
Risk of bias
The summary of risk assessment for the included studies is presented in Table 1. One RCT was assessed as having some concern,31 while four RCTs were evaluated as having high concern.27,34,42,43 All other studies were assessed as high quality. Detailed quality assessments for the randomized and non-randomized studies is presented in Tables S4 and S5, respectively.
Clinical characteristics
Table 2 presents the clinical characteristics and outcomes of the included studies. The primary disease conditions studied were cystic fibrosis,29,33,42 febrile neutropenia,27,31,40 infective endocarditis,28,36 cellulitis34,35,44 and appendicitis.30,39,41 Some studies did not report the specific interventions used to treat these conditions31,32,35,37,43–45 or the types of OPAT models employed.30,36,38,41,45
Table 2.
Clinical characteristics and outcomes of patients receiving parenteral antimicrobial therapy
| Author and year | Disease condition | OPAT model | Intervention | Health outcomes | |
|---|---|---|---|---|---|
| OPAT | IPAT | ||||
| Hendricks 201131 | Febrile neutropenia | H-OPAT | Aminoglycoside and vancomycin or ceftazidime alone; for allergic patients’ imipenem alone or an aztreonam-containing regimen | NR | Complication OPAT 9%, IPAT 8%; readmission OPAT 3 (9%); mortality 0%; patient-reported quality of life was similar between the two groups. |
| Hensey 201732 | Pyelonephritis, meningitis | H-OPAT | NR | NR | Unplanned readmission within 30 days pyelonephritis IPAT 8 (7%), OPAT 1 (8%); meningitis IPAT 2 (13%), OPAT 2 (7%) |
| Martel 199437 | Osteomyelitis, septic arthritis septic bursitis, cellulitis, cystic fibrosis, complicated urinary tract infection, severe external otitis, chronic sinusitis, cutaneous blastomycosis, endocarditis, lung abscess, liver abscess | S-OPAT | Penicillin G, cloxacillin, cephalothin, cefazolin, cefoxitin, cefotaxime, cefoperazone, ceftriaxone, ceftazidime, gentamicin, netilmicin, tobramycin, amikacin, clindamycin, erythromycin, gancyclovir, amphotericin B | NR | Treatment failure OPAT 2 (6.1%), IPAT 1 (5.9%); readmission OPAT 2 (6.1%); OPAT patients had a significantly higher internal locus of control, IPAT group had a significantly higher external locus of control. |
| Antoniskis 197828 | Infective endocarditis, osteomyelitis | S-OPAT | Methicillin, cephalothin, nafcillin, penicillin G, cefazolin, vancomycin, gentamicin, ampicillin | Methicillin, cephalothin, nafcillin, penicillin G, cefazolin, vancomycin, gentamicin, ampicillin | There are similar antibiotic-related complications; there is no infection of the intravenous cannula in either group. readmission OPAT 3 (23.1%) |
| Kameshwar 201635 | Cellulitis | H-OPAT | NR | NR | NR |
| Donati 198729 | Cystic fibrosis | H-OPAT, S-OPAT | Tobramycin, semisynthetic penicillin, cephalosporin | Tobramycin, semisynthetic penicillin, cephalosporin | No complications relating to the catheter; 65% of OPAT patients and 68% of IPAT patients required retreatment for pulmonary exacerbations; 85% of OPAT patients returned to their daily routines. |
| Warner 199841 | Acute appendicitis | NR | Cefotetan, gentamicin, ampicillin sodium/sulbactam sodium | Cefotetan, gentamicin, ampicillin sodium/sulbactam sodium | Negative appendectomy IPAT 12.3%, OPAT 9.2%; perforation IPAT 26.2%, OPAT 18.3% |
| Yong 200845 | Urinary tract infections, infective endocarditis, bone and joint infection, diabetic foot, septicaemia, orbital procedure, mouth, larynx or pharynx disorder | NR | NR | NR | Complication OPAT 18 (25%); cure OPAT 59 (81.9%), IPAT 75 (80.6%); readmission OPAT 13 (18.1%), IPAT 18 (19.4%); mortality OPAT 1 (1.4%), IPAT 1 (1.1%) |
| Ramasubramanian 201838 | Acute pyelonephritis | NR | Ertapenem | Ertapenem | NR |
| Wolter 200443 | Urinary tract infection, cellulitis, pneumonia, osteomyelitis, mycobacterial infection, cystic fibrosis, bronchiectasis | Home infusion | NR | NR | Adverse event OPAT 4 (11.4%), IPAT 4 (11.2%); readmission within 30 days OPAT 7 (15.9%), IPAT 4 (10.5%); no differences in improvements in quality of life between the two groups after treatment. |
| Ahmed 200727 | Febrile neutropenia | C-OPAT | Ceftriaxone plus amikacin | Imipenem | Adverse event OPAT 8 (13.1%), IPAT 4 (6.9%); favourable outcome OPAT 58 (95%), IPAT 56 (97%); mortality OPAT 2 (3.3%), IPAT 2 (3.4%) |
| Talcott 199440 | Febrile neutropenia | S-OPAT | Mezlocillin, gentamicin, ceftazidime, vancomycin | Mezlocillin, gentamicin, ceftazidime, vancomycin | Adverse events OPAT 4 (13.3%); readmission OPAT 9 (30%); quality of life improved during home therapy |
| Fishman 200030 | Perforated appendicitis | NR | Piperacillin-tazobactam, allergic patients gentamicin and clindamycin | Ampicillin, gentamicin, clindamycin | No mortality; readmission OPAT 4 (7.8%), IPAT 5 (5.1%); strong satisfaction with the home antibiotic regimen. |
| Lacroix 201436 | Infective endocarditis | NR | Oxacillin, vancomycin, ceftriaxone, amoxicillin, gentamycin, daptomycin, rifampicin | Oxacillin, vancomycin, ceftriaxone, amoxicillin, gentamycin, daptomycin, rifampicin | Adverse event OPAT 3 (16.7%); readmission OPAT 6 (33.3%), IPAT 7 (33.3%), death OPAT 1 (5.5%), IPAT 1 (4.8%) |
| Wolter 199742 | Cystic fibrosis | S-OPAT | Ceftazidime and tobramycin, imipenem used on allergy or failure | Ceftazidime and tobramycin, imipenem used on allergy or failure | No deaths, no short-term readmissions, no adverse events; no statistical difference in overall improvement in lung function; OPAT patients reported less disruption to their family life, personal life and sleeping pattern. |
| Ibrahim 201733 | Moderate/severe cellulitis | H-OPAT | Ceftriaxone | Flucloxacillin | Complication OPAT 3 (6%), IPAT 6 (10%); treatment failure OPAT 2 (4%), IPAT 8 (14%); readmission OPAT 0%, IPAT 1 (2%) |
| Yadev 202244 | Cellulitis | H-OPAT, C-OPAT | NR | NR | Adverse event OPAT 39 (11.4%); treatment failure OPAT 8 (2.3%) |
| Ibrahim 201934 | Cellulitis | H-OPAT, telemedicine | Ceftriaxone | Flucloxacillin | Readmission OPAT 2.25%; treatment failure OPAT 1 (1.1%), IPAT 7 (7.7%) |
| Stovroff 199439 | Ruptured appendicitis | S-OPAT | Ampicillin, gentamicin, clindamycin | Ampicillin, gentamicin, clindamycin | Complication 0%; readmission 0%; treatment failure 0% |
| Pena 201346 | Urinary tract infection | C-OPAT | Amikacin, ceftriaxone, cefotaxime | Amikacin, ceftriaxone, cefotaxime | OPAT 100% adherence, 0% readmission; efficacy OPAT 105 (100%), IPAT 72 (98.6%); adverse event OPAT 18 (16.2%), IPAT 58 (71.6%); mortality 0% |
OPAT, outpatient parenteral antimicrobial therapy; IPAT, inpatient parenteral antimicrobial therapy; NR, not reported; S-OPAT, self or carer-administered OPAT at patients’ home; H-OPAT, physician or nurse-administered OPAT at patient home; C-OPAT, outpatient clinic or infusion centre administration.
Economic evaluation
The economic evaluations of the included studies are presented in Table 3. The majority of the studies used a cost–consequence analysis.27–33,36–43,45,46 The cost perspective was specified in two-thirds of the studies,28,29,31,33–36,39,40,42–45 and half reported both the direct and indirect costs associated with parenteral antibiotic treatment.31,33,34,36,39,40,42–44,46 The average cost of treatment per episode of care was $10 453.67 in outpatient settings and $16 491.7 in inpatient settings. Notably, the average percentage of cost savings associated with OPAT was 36.11%.
Table 3.
Economic evaluation of patients receiving parenteral antimicrobial therapy
| Author and year | Economic analysis | Perspective | Type of cost | Mean cost (USD) | % of OPAT cost saving | Number of days of hospitalization avoided (mean) | |
|---|---|---|---|---|---|---|---|
| OPAT | IPAT | ||||||
| Hendricks 201131 | CCA | Healthcare Payer’s | Direct and indirect | 11 076.12 | 14 492.76 | 23.57 | NR |
| Hensey 201732 | CCA | NR | NR | 1371.12 | 1647.36 | 16.77 | Polynephritis 1.5, meningitis 9a |
| Martel 199437 | CCA | NR | NR | 7723.67 | 11 020.07 | 29.91 | NR |
| Antoniskis 197828 | CCA | Hospital | NR | 26 510.86 | 41 792.7 | 36.57 | 17.31 |
| Kameshwar 201635 | CEA | Hospital | Direct and overhead | 5205.88 | 4605.78 | −13.03 | 5.6 |
| Donati 198729 | CCA | Hospital | Direct | 23 673.32 | 44 432.82 | 46.72 | 17.7 |
| Warner 199841 | CCA | NR | NR | Non-perforated appendicitis 6561.01, perforated appendicitis 14 108.52 | Non-perforated appendicitis 7385.20, perforated appendicitis 20 153.75 | NR | |
| Yong 200845 | CCA | Hospital | Direct | 18 623.38a | 18 136.45a | −2.68 | 24.3 |
| Ramasubramanian 201838 | CCA | NR | NR | 9373.89 | 19 516.23 | 51.97 | NR |
| Wolter 200443 | CCA | Hospital | Direct and indirect | 8461.92a | 15 922.93a | 46.86 | 6a |
| Ahmed 200727 | CCA | NR | NR | 1878.03 | 2771.96 | 32.25 | 2.9 |
| Talcott 199440 | CCA | NR | Direct and indirect | 804.11 | 796.99 | −0.89 | 5.3 |
| Fishman 200030 | CCA | Hospital | NR | 25 522.56 | 37 226.67 | 31.44 | NR |
| Lacroix 201436 | CCA | Hospital | Direct and indirect | 46 413.96 | 59 972.16 | 22.61 | 16.6 |
| Wolter 199742 | CCA | Hospital | Direct and indirect | 4747.45 | 9628.95 | 50.70 | 9a |
| Ibrahim 201733 | CCA | Hospital | Direct and indirect | 1037.97 | 2309.17 | 55.05 | 3.7 |
| Yadev 202244 | CBA | Healthcare Payer’s | Direct and indirect | 2247.78 | 11 115.3 | 79.78 | NR |
| Ibrahim 201934 | CEA | Societal | Direct and indirect | 1672.27 | 3212.63 | 47.95 | NR |
| Stovroff 199439 | CCA | Hospital | Direct and indirect | 7663.52 | 12 754.49 | 39.92 | 5.1 |
| Pena 201346 | CCA | NR | Direct and indirect | 789.89 | 3062.94 | 74.21 | 3.2 |
OPAT, outpatient parenteral antimicrobial therapy; IPAT, inpatient parenteral antimicrobial therapy; NR, not reported; CBA, cost–benefit analysis; CCA: cost–consequence analysis; CEA, cost–effectiveness analysis; USD, US dollar.
aMedian.
Cost of parenteral antibiotic treatment
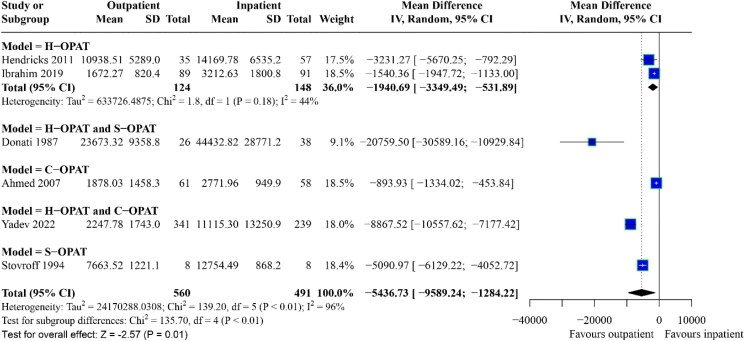
Six studies, involving three RCTs and three cohort studies, were included in the meta-analysis. The mean cost of OPAT per episode of care was $8012.24 (range: $1672.27–$23 673.32), and inpatient parenteral antimicrobial treatment was $14 742.83 (range: $2771.96–$44 432.82). The cost of parenteral antimicrobial treatment per episode of care was lower in the outpatient settings, with mean differences of −$5436.73 (95% CI: −$9589.24 to −$1284.22, I² = 96%; P = 0.01) compared to inpatient settings (Figure 2).
Figure 2.
Cost of treatment: Forest plot comparison of outpatient and inpatient antimicrobial therapy. S-OPAT, self or carer-administered OPAT at patients’ home; H-OPAT, physician or nurse-administered OPAT at patient home; C-OPAT, outpatient clinic or infusion centre administration; IV, inverse variance; SD, standard deviation.
A sensitivity analysis was conducted on studies evaluating parenteral antimicrobial treatment’s direct and indirect costs, cost perspectives (payer), population (adult and paediatric), ways of hospitalization avoidance (admission avoidance and facilitation of discharge) and those published after 2000 (Figures S1–S9). The results confirmed those of the primary analysis (Figures S1–S3). The cost savings from the healthcare provider perspective favour OPAT, though the result is not statistically significant (Figure S4). Moreover, the cost savings of OPAT for the paediatric population, discharge facilitation and admission avoidance favour outpatient treatment, though the results are not statistically significant (Figures S7–S9).
Discussion
To our knowledge, this is the first meta-analysis to assess the cost savings from OPAT compared to inpatient parenteral antimicrobial treatment. We conducted a quantitative analysis by inflating and adjusting the costs of parenteral antimicrobial treatment to 2023 US dollars. Parenteral antimicrobial treatment through OPAT programmes had a significantly lower cost compared to inpatient (hospital-bed based) treatment, with an estimated average cost saving of −$5436.73 per episode of care.
This finding of cost saving aligns with a previous review,20 although in that review, a robust estimate of the magnitude of cost saving was not possible due to the quality and methodological heterogeneity of the included studies, limiting direct comparisons. A gross estimate of the average cost saving associated with OPAT from the 35 heterogeneous studies they included, calculated from a ratio of average cost for OPAT to average cost for inpatient treatment, was 57.19% (−13.03% to 95.47%); in this review, the average percentage of cost savings calculated in the same way was 36.11%.
Most studies in the review examined indirect and direct costs, while only a few focused exclusively on direct costs. The cost savings from the OPAT programme are primarily attributed to the freeing up of hospital beds.47 However, a simple comparison of treatment costs between outpatient and inpatient settings does not provide a comprehensive assessment. A thorough evaluation should include all direct, indirect and intangible costs associated with treatment. Ultimately, the costs of parenteral antimicrobial treatment should be weighed against the benefits it offers to patients, healthcare providers, payers and the society.48
The cost–benefit of OPAT varies depending on the perspectives of patients, healthcare providers, payers and society. Despite the differing perspectives that lead to varied analyses and economic outcomes, from all viewpoints, the savings are primarily attributed to reductions in inpatient costs. However, in some healthcare systems, outpatient costs are typically borne directly by patients including co-payments for drugs and services, transportation to and from the healthcare facility, accommodation and home care services.49 These expenses may partially offset the savings from outpatient treatment.47 Nonetheless, the cost–benefit can be substantial when indirect benefits, such as increased productivity, savings on patient or carer transportation and accommodation costs, are taken into account.48
The substantial cost–benefit of the OPAT programme would mean that it can serve as an invaluable cost-saving tool for healthcare systems in the wake of increasing pressure to deliver high-quality patient-centred care while optimizing resources at a reduced cost, without compromising clinical outcomes.50 According to the World Health Organization, governments allocate between half and two-thirds of their health expenditures to hospital care. Alarmingly, nearly $300 billion is wasted annually due to hospital-related inefficiencies.51 Therefore, wider implementation of OPAT is likely to contribute to efficient resource/funding relocation, enhancing the overall capacity of the healthcare system. The OPAT programme improves patient flow and increases hospital bed capacity by allowing patients to receive treatment closer to home, thereby enabling hospitals to focus on acute medical conditions.52
Treatment in OPAT is both safe and effective compared to inpatient treatment.10 OPAT enables the treatment of patients infected with antimicrobial-resistant organisms. The risk of acquiring nosocomial infections with such organisms is low, thereby avoiding unplanned huge treatment costs associated with such infections.53 Beyond financial gains, it offers clinical benefits by improving patient’s quality of life through home-based treatment, which minimizes stress and disruptions to daily routines. It also promotes emotional well-being and allows for flexible scheduling, making the treatment experience more manageable and supportive.14
Despite the multiple benefits OPAT offers, it also has limitations. Some patients encountered difficulties in administering medications due to a lack of confidence in their skills,17,54 which could lead to complications related to administration devices.55 Others experienced challenges with showering, walking and sleeping during continuous intravenous drug administration,56 impacting their social activities54 and school attendance.52 A few families perceived OPAT as increasing their workload.57 Additionally, patients with limited transportation or those living far from healthcare facilities may need to travel for monitoring and medication administration.58
The strengths of this review include adherence to PRISMA guidelines and a comprehensive search across multiple databases, ensuring a robust methodology. Moreover, the review included studies that compared the actual costs associated with OPAT to those of inpatient treatment, with costs inflated and adjusted to a common currency and year for accurate comparison. However, there are notable limitations among the included studies. First, most studies were cohort studies, with only six being RCTs, and these RCTs were assessed as having a high risk of bias. Additionally, there was heterogeneity among the studies, which arose from their diverse healthcare system contexts. Second, over half of the studies reported limited cost data, which precluded their inclusion in the meta-analysis. Furthermore, publication bias could not be assessed due to the small number of included studies. Third, the included studies demonstrated heterogeneity in the disease conditions evaluated, with many focusing on uncommon indications for OPAT. Fourth, the included studies reported different ways of hospitalization avoidance (early discharge and admission avoidance) and patient populations (adult and paediatric). Fifth, the types of costs used to calculate the costs of parenteral antimicrobial treatment varied widely. However, a sensitivity analysis did not alter the primary results. Sixth, cost data in most of the included studies were evaluated as secondary outcomes. Furthermore, most studies did not specify the cost perspective, and those that did were limited to the hospital perspective. Consequently, pharmacoeconomic data comparing the costs of parenteral antimicrobial treatment in outpatient versus inpatient settings are scarce. Therefore, we recommend conducting more robust pharmacoeconomic studies that compare outpatient and inpatient parenteral antimicrobial treatment costs, incorporating all cost types from the perspective of patients, society, the healthcare system and payers. Moreover, studies comparing the cost of OPAT to complex outpatient oral antimicrobial therapy are warranted.
Conclusions
Parenteral antimicrobial treatment through OPAT programmes significantly saves costs compared to inpatient settings from the payer perspective. For a comprehensive comparison of treatment costs in both settings, further pharmacoeconomic studies that include all types of treatment costs are essential. We recommend conducting studies that evaluate the costs of parenteral antimicrobial treatment from the perspectives of patients, healthcare providers, payers and the society.
Supplementary Material
Acknowledgements
F.B.S. acknowledges support from the Australian National Health and Medical Research Council (NHMRC) Investigator Grant (APP1197866). J.A.R. would like to acknowledge funding from the Australian National Health and Medical Research Council for a Centre of Research Excellence (APP2007007) and an Investigator Grant (APP2009736) as well as an Advancing Queensland Clinical Fellowship. M.G. is a member of BSAC OPAT Initiative.
Contributor Information
Solomon Ahmed Mohammed, UQ Centre for Clinical Research, The University of Queensland, Brisbane, QLD, Australia; Department of Pharmacy, Wollo University, Dessie, Ethiopia.
Jason A Roberts, UQ Centre for Clinical Research, The University of Queensland, Brisbane, QLD, Australia; Department of Pharmacy and Intensive Care Medicine, Royal Brisbane and Women’s Hospital, Brisbane, QLD, Australia; Herston Infectious Disease Institute (HeIDI), Metro North Health, Brisbane, QLD, Australia; Division of Anaesthesiology Critical Care Emerging and Pain Medicine, Nimes University Hospital, University of Montpellier, Nîmes, France.
Manuel Mirón-Rubio, Hospital Universitario de Torrejón, Unidad de Hospitalización a Domicilio, Madrid, Spain.
Luis Eduardo López Cortés, Unidad Clínica de Enfermedades Infecciosas y Microbiología, Instituto de Biomedicina de Sevilla (IBiS)/CSIC, Seville, Spain; Hospital Universitario Virgen Macarena, and Departamento de Medicina, Universidad de Sevilla, Seville, Spain; CIBERINFEC, Instituto de Salud Carlos III, Madrid, Spain.
Getnet Mengistu Assefa, UQ Centre for Clinical Research, The University of Queensland, Brisbane, QLD, Australia; Department of Pharmacy, Wollo University, Dessie, Ethiopia.
James Pollard, Cabrini @ Home, Cabrini Health, Melbourne, Australia.
Kate McCarthy, Royal Brisbane Clinical School, Faculty of Medicine, The University of Queensland, Brisbane, QLD, Australia; Department of Infectious Diseases, Royal Brisbane and Women’s Hospital, Brisbane, QLD, Australia.
Mark Gilchrist, Department of Pharmacy/Infection, Imperial College Healthcare NHS Trust, London, UK; Department of Infectious Diseases, Imperial College, London, UK.
Menino Cotta, UQ Centre for Clinical Research, The University of Queensland, Brisbane, QLD, Australia.
Fekade B Sime, UQ Centre for Clinical Research, The University of Queensland, Brisbane, QLD, Australia.
Funding
This study was carried out as part of our routine work.
Transparency declarations
None to declare.
Supplementary data
Figures S1–S9 and Tables S1–S5 are available as Supplementary data at JAC-AMR Online.
References
- 1. McCracken K, Phillips DR. Global Health: An Introduction to Current and Future Trends. Routledge, 2017. [Google Scholar]
- 2. Suetens C, Latour K, Kärki T et al. Prevalence of healthcare-associated infections, estimated incidence and composite antimicrobial resistance index in acute care hospitals and long-term care facilities: results from two European point prevalence surveys, 2016 to 2017. Euro Surveill 2018; 23: 1800516. 10.2807/1560-7917.ES.2018.23.46.1800516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Chapman AL. Outpatient parenteral antimicrobial therapy in a changing NHS: challenges and opportunities. Clin Med 2013; 13: 35–6. 10.7861/clinmedicine.13-1-35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Chapman AL, Patel S, Horner C et al. Updated good practice recommendations for outpatient parenteral antimicrobial therapy (OPAT) in adults and children in the UK. JAC Antimicrob Resist 2019; 1: dlz026. 10.1093/jacamr/dlz026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. López-Cortés LE, Luque R, Cisneros JM et al. Next step, outpatient antimicrobial therapy programs as a tool of stewardship programs. Clin Infect Dis 2019; 68: 2155. 10.1093/cid/ciy1052 [DOI] [PubMed] [Google Scholar]
- 6. Rucker RW, Harrison GM. Outpatient intravenous medications in the management of cystic fibrosis. Pediatrics 1974; 54: 358–60. 10.1542/peds.54.3.358 [DOI] [PubMed] [Google Scholar]
- 7. Mirón-Rubio M, González-Ramallo V, Estrada-Cuxart O et al. Intravenous antimicrobial therapy in the hospital-at-home setting: data from the Spanish Outpatient Parenteral Antimicrobial Therapy Registry. Future Microbiol 2016; 11: 375–90. 10.2217/fmb.15.141 [DOI] [PubMed] [Google Scholar]
- 8. Tice AD, Rehm SJ, Dalovisio JR et al. Practice guidelines for outpatient parenteral antimicrobial therapy. Clin Infect Dis 2004; 38: 1651–71. 10.1086/420939 [DOI] [PubMed] [Google Scholar]
- 9. Williams DN, Baker CA, Kind AC et al. The history and evolution of outpatient parenteral antibiotic therapy (OPAT). Int J Antimicrob Agents 2015; 46: 307–12. 10.1016/j.ijantimicag.2015.07.001 [DOI] [PubMed] [Google Scholar]
- 10. Mohammed SA, Roberts JA, Cotta MO et al. Safety and efficacy of outpatient parenteral antimicrobial therapy: a systematic review and meta-analysis of randomized clinical trials. Int J Antimicrob Agents 2024; 64: 107263. 10.1016/j.ijantimicag.2024.107263 [DOI] [PubMed] [Google Scholar]
- 11. Saillen L, Arensdorff L, Moulin E et al. Patient satisfaction in an outpatient parenteral antimicrobial therapy (OPAT) unit practising predominantly self-administration of antibiotics with elastomeric pumps. Eur J Clin Microbiol Infect Dis 2017; 36: 1387–92. 10.1007/s10096-017-2944-5 [DOI] [PubMed] [Google Scholar]
- 12. Kwok CS, Whittaker JJ, Malbon C et al. Outpatient parenteral antimicrobial therapy (OPAT) service is associated with inpatient-bed cost savings. Br J Cardiol 2021; 28: 38. 10.5837/bjc.2021.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Durojaiye OC, Kritsotakis EI. Evaluation of health-related quality of life in patients receiving outpatient parenteral antimicrobial therapy (OPAT) in a UK setting. Expert Rev Anti Infect Ther 2024; 22: 987–95. 10.1080/14787210.2024.2334059 [DOI] [PubMed] [Google Scholar]
- 14. Wee LE, Sundarajoo M, Quah W-F et al. Health-related quality of life and its association with outcomes of outpatient parenteral antibiotic therapy. Eur J Clin Microbiol Infect Dis 2020; 39: 765–72. 10.1007/s10096-019-03787-6 [DOI] [PubMed] [Google Scholar]
- 15. Erba A, Beuret M, Daly M-L et al. OPAT in Switzerland: single-center experience of a model to treat complicated infections. Infection 2020; 48: 231–40. 10.1007/s15010-019-01381-8 [DOI] [PubMed] [Google Scholar]
- 16. Poretz DM, Eron LJ, Goldenberg RI et al. Intravenous antibiotic therapy in an outpatient setting. JAMA 1982; 248: 336–9. 10.1001/jama.1982.03330030042023 [DOI] [PubMed] [Google Scholar]
- 17. Tonna A, Anthony G, Tonna I et al. Home self-administration of intravenous antibiotics as part of an outpatient parenteral antibiotic therapy service: a qualitative study of the perspectives of patients who do not self-administer. BMJ Open 2019; 9: e027475. 10.1136/bmjopen-2018-027475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Krauth C, Busse R, Smaczny C et al. Cost analysis of hospital and home iv therapy in adults with cystic fibrosis. Results of a prospective controlled study: ergebnisse einer prospektiven kontrollierten studie. Med Klin 1999; 94: 541–8. 10.1007/BF03044951 [DOI] [PubMed] [Google Scholar]
- 19. Dimitrova M, Gilchrist M, Seaton R. Outpatient parenteral antimicrobial therapy (OPAT) versus inpatient care in the UK: a health economic assessment for six key diagnoses. BMJ Open 2021; 11: e049733. 10.1136/bmjopen-2021-049733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Psaltikidis EM, Silva E, Bustorff-Silva JM et al. Economic evaluation of outpatient parenteral antimicrobial therapy: a systematic review. Expert Rev Pharmacoecon Outcomes Res 2017; 17: 355–75. 10.1080/14737167.2017.1360767 [DOI] [PubMed] [Google Scholar]
- 21. Page MJ, McKenzie JE, Bossuyt PM et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg 2021; 88: 105906. 10.1016/j.ijsu.2021.105906 [DOI] [PubMed] [Google Scholar]
- 22. Wells GA, Shea B, O’Connell D et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm.
- 23. Higgins JPT, Savovic J, Page MJ et al. Assessing risk of bias in a randomized trial. In: Higgins JPT, Thomas J, Chandler J et al., eds. Cochrane Handbook for Systematic Reviews of Interventions Version 6.4: Cochrane Collaboration. Wiley, 2023. [Google Scholar]
- 24. Shemilt I, James T, Marcello M. A web-based tool for adjusting costs to a specific target currency and price year. Evid Policy 2010; 6: 51–9. 10.1332/174426410X482999 [DOI] [Google Scholar]
- 25. Kuritz SJ, Landis JR, Koch GG. A general overview of Mantel–Haenszel methods: applications and recent developments. Annu Rev Public Health 1988; 9: 123–60. 10.1146/annurev.pu.09.050188.001011 [DOI] [PubMed] [Google Scholar]
- 26. Higgins JPT, Thomas J, Chandler J et al. Cochrane handbook for systematic reviews of interventions version 6.5. In: Cochrane Collaboration. Wiley, 2024. [Google Scholar]
- 27. Ahmed N, El-Mahallawy HA, Ahmed IA et al. Early hospital discharge versus continued hospitalization in febrile pediatric cancer patients with prolonged neutropenia: a randomized, prospective study. Pediatr Blood Cancer 2007; 49: 786–92. 10.1002/pbc.21179 [DOI] [PubMed] [Google Scholar]
- 28. Antoniskis A, Anderson BC, Van Volkinburg EJ et al. Feasibility of outpatient self-administration of parenteral antibiotics. West J Med 1978; 128: 203–6. [PMC free article] [PubMed] [Google Scholar]
- 29. Donati MA, Guenette G, Auerbach H. Prospective controlled study of home and hospital therapy of cystic fibrosis pulmonary disease. J Pediatr 1987; 111: 28–33. 10.1016/s0022-3476(87)80337-2 [DOI] [PubMed] [Google Scholar]
- 30. Fishman SJ, Pelosi L, Klavon SL et al. Perforated appendicitis: prospective outcome analysis for 150 children. J Pediatr Surg 2000; 35: 923–6. 10.1053/jpsu.2000.6924 [DOI] [PubMed] [Google Scholar]
- 31. Hendricks AM, Loggers ET, Talcott JA. Costs of home versus inpatient treatment for fever and neutropenia: analysis of a multicenter randomized trial. J Clin Oncol 2011; 29: 3984–9. 10.1200/JCO.2011.35.1247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Hensey CC, Sett A, Connell TG et al. A comparison of hospital versus outpatient parenteral antibiotic therapy at home for pyelonephritis and meningitis. Pediatr Infect Dis J 2017; 36: 827–32. 10.1097/INF.0000000000001605 [DOI] [PubMed] [Google Scholar]
- 33. Ibrahim LF, Hopper SM, Connell TG et al. Evaluating an admission avoidance pathway for children in the emergency department: outpatient intravenous antibiotics for moderate/severe cellulitis. J Emerg Med 2017; 34: 780–5. 10.1136/emermed-2017-206829 [DOI] [PubMed] [Google Scholar]
- 34. Ibrahim LF, Huang L, Hopper SM et al. Intravenous ceftriaxone at home versus intravenous flucloxacillin in hospital for children with cellulitis: a cost–effectiveness analysis. Lancet Infect Dis 2019; 19: 1101–8. 10.1016/S1473-3099(19)30288-9 [DOI] [PubMed] [Google Scholar]
- 35. Kameshwar K, Karahalios A, Janus E et al. False economies in home-based parenteral antibiotic treatment: a health-economic case study of management of lower-limb cellulitis in Australia. J Antimicrob Chemother 2016; 71: 830–5. 10.1093/jac/dkv413 [DOI] [PubMed] [Google Scholar]
- 36. Lacroix A, Revest M, Patrat-Delon S et al. Outpatient parenteral antimicrobial therapy for infective endocarditis: a cost-effective strategy. Med Mal Infect 2014; 44: 327–30. 10.1016/j.medmal.2014.05.001 [DOI] [PubMed] [Google Scholar]
- 37. Martel AY. Home intravenous self-injection of antibiotic therapy. Can J Infect Dis Med Microbio 1994; 5: 51C–5C. 10.1155/1994/673183 [DOI] [Google Scholar]
- 38. Ramasubramanian V, Murlidharan P, Nambi S et al. Efficacy and cost comparison of ertapenem as outpatient parenteral antimicrobial therapy in acute pyelonephritis due to extended-spectrum beta-lactamase-producing Enterobacteriaceae. Indian J Nephrol 2018; 28: 351–7. 10.4103/ijn.IJN_207_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Stovroff MC, Totten M, Glick PL. PIC lines save money and hasten discharge in the care of children with ruptured appendicitis. J Pediatr Surg 1994; 29: 245–7. 10.1016/0022-3468(94)90327-1 [DOI] [PubMed] [Google Scholar]
- 40. Talcott JA, Whalen A, Clark J et al. Home antibiotic therapy for low-risk cancer patients with fever and neutropenia: a pilot study of 30 patients based on a validated prediction rule. J Clin Oncol 1994; 12: 107–14. 10.1200/JCO.1994.12.1.107 [DOI] [PubMed] [Google Scholar]
- 41. Warner BW, Kulick RM, Stoops MM et al. An evidenced-based clinical pathway for acute appendicitis decreases hospital duration and cost. J Pediatr Surg 1998; 33: 1371–5. 10.1016/s0022-3468(98)90010-0 [DOI] [PubMed] [Google Scholar]
- 42. Wolter J, Bowler S, Nolan P et al. Home intravenous therapy in cystic fibrosis: a prospective randomized trial examining clinical, quality of life and cost aspects. Eur Respir J 1997; 10: 896–900. 10.1183/09031936.97.10040896 [DOI] [PubMed] [Google Scholar]
- 43. Wolter J, Cagney R, McCormack J. A randomised trial of home vs hospital intravenous antibiotic therapy in adults with infectious diseases. J Infect 2004; 48: 263–8. 10.1016/S0163-4453(03)00135-X [DOI] [PubMed] [Google Scholar]
- 44. Yadav K, Kumar S, Chhabra S et al. Outpatient parenteral antibiotic therapy (OPAT) and inpatient treatment strategies for emergency department patients with cellulitis: a cost analysis. J Emerg Med 2022; 24: 520–8. 10.1007/s43678-022-00320-1 [DOI] [PubMed] [Google Scholar]
- 45. Yong C, Fisher DA, Sklar GE et al. A cost analysis of outpatient parenteral antibiotic therapy (OPAT): an Asian perspective. Int J Antimicrob Agents 2009; 33: 46–51. 10.1016/j.ijantimicag.2008.07.016 [DOI] [PubMed] [Google Scholar]
- 46. Pena A, Zambrano A, Alvarado M et al. Evaluation of the effectiveness, safety and costs of outpatient intravenous antimicrobial treatment (OPAT) vs hospitalized in urinary infection in pediatrics. Rev Chilena Infectol 2013; 30: 426–34. 10.4067/S0716-10182013000400012 [DOI] [PubMed] [Google Scholar]
- 47. Eisenberg JM, Kitz DS. Savings from outpatient antibiotic therapy for osteomyelitis: economic analysis of a therapeutic strategy. JAMA 1986; 255: 1584–8. 10.1001/jama.1986.03370120062024 [DOI] [PubMed] [Google Scholar]
- 48. Milkovich G. Costs and benefits. Hosp Pract 1993; 28: 39–43. 10.1080/21548331.1993.11442934 [DOI] [PubMed] [Google Scholar]
- 49. Nolet BR. Update and overview of outpatient parenteral antimicrobial therapy regulations and reimbursement. Clin Infect Dis 2010; 51: S216–9. 10.1086/653522 [DOI] [PubMed] [Google Scholar]
- 50. Chen X, Geng S, Zhu Y et al. Impact of infection on healthcare costs and clinical outcomes in elderly hospitalized patients with multimorbidity. Heliyon 2024; 10: e31560. 10.1016/j.heliyon.2024.e31560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. WHO . WHO Global Health Expenditure Atlas. 2014. https://cdn.who.int › health-financing › atlas2014.
- 52. Carter B, Fisher-Smith D, Porter D et al. Being ‘at-home’ on outpatient parenteral antimicrobial therapy (OPAT): a qualitative study of parents’ experiences of paediatric OPAT. Arch Dis Child 2020; 105: 276–81. 10.1136/archdischild-2019-317629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Barr DA, Semple L, Seaton RA. Outpatient parenteral antimicrobial therapy (OPAT) in a teaching hospital-based practice: a retrospective cohort study describing experience and evolution over 10 years. Int J Antimicrob Agents 2012; 39: 407–13. 10.1016/j.ijantimicag.2012.01.016 [DOI] [PubMed] [Google Scholar]
- 54. Hamad Y, Dodda S, Frank A et al. Perspectives of patients on outpatient parenteral antimicrobial therapy: experiences and adherence. Open Forum Infect Dis 2020; 7: 6. 10.1093/ofid/ofaa205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Agnihotri G, Gross AE, Seok M et al. Decreased hospital readmissions after programmatic strengthening of an outpatient parenteral antimicrobial therapy (OPAT) program. Antimicrob Steward Healthc Epidemiol 2023; 3: e33. 10.1017/ash.2022.330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Berrevoets MAH, Oerlemans AJM, Tromp M et al. Quality of outpatient parenteral antimicrobial therapy (OPAT) care from the patient’s perspective: a qualitative study. BMJ Open 2018; 8: e024564. 10.1136/bmjopen-2018-024564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Cabrera López IM, Agúndez Reigosa B, Adrados García S et al. Home–hospital care for children with acute illnesses: a 2-year follow-up study. Paediatr Child Health 2022; 58: 969–77. 10.1111/jpc.15870 [DOI] [PubMed] [Google Scholar]
- 58. Mehta M, Benning M, Johnson JE et al. Facilitating OPAT in rural areas. Ther Adv Infect Dis 2023; 10: 20499361231210353. 10.1177/20499361231210353 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.