Abstract
Numerous studies on the effects of exercise on antioxidant enzymes have generally concluded that regular exercise positively impacts antioxidant enzyme activity. However, some studies suggest that regular exercise may have no effect on antioxidant enzymes or could even negatively impact them. This suggests that other potential factors may influence antioxidant enzyme activity. Therefore, this study synthesizes existing literature on the effects of exercise interventions on antioxidant enzymes and employs subgroup analysis to identify factors that may influence exercise outcomes, offering insights for individuals aiming to enhance antioxidant capacity through exercise. A systematic review and meta-analysis were performed on exercise intervention studies measuring changes in blood antioxidant enzymes. This study was registered in PROSPERO (identifier: CRD 42023477230). (1) Exercise did not significantly increase superoxide dismutase (SOD) activity in women. (2) In individuals over 45 years of age, exercise did not significantly improve SOD activity or total antioxidant capacity (T-AOC) levels. (3) Regardless of exercise type, trends in SOD and catalase (CAT) activity were similar; however, only resistance exercise increased glutathione peroxidase (GPX) activity and reduced thiobarbituric Acid Reactive Substances (TBARS) levels. (4) High-intensity exercise significantly reduced CAT levels but did not significantly increase GPX levels. (5) Exercise interventions lasting more than 16 weeks showed no significant impact on the activity of SOD, CAT, or GPX. 6. Regular exercise at least three times per week significantly increased SOD and GPX activity and had a notable impact on T-AOC and TBARS levels. This study found that exercise significantly enhanced the activity of most antioxidant enzymes and overall antioxidant capacity. Moderate-to-low intensity exercise, performed at least three times per week for more than 16 weeks, demonstrated the greatest efficacy in enhancing antioxidant enzyme activity. Notably, we also found that women may need to exert more effort than men to achieve increases in antioxidant enzyme activity.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-025-97101-4.
Keywords: Exercise, Antioxidant enzymes, Superoxide dismutase, Catalase, Glutathione peroxidase
Subject terms: Biomarkers, Cardiology, Health care
Introduction
Regular long-term exercise benefits human health, with many positive effects stemming from exercise-induced changes in redox levels. During exercise of a certain intensity, free radicals are generated in areas such as the mitochondria, nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, xanthine oxidase, and phospholipase A21. At normal concentrations, hydrogen peroxide molecules can diffuse across cell membranes and act as signaling molecules. However, excessive free radicals can oxidize biomolecules, disrupting their structure and function2. Thus, free radicals play a dual role, and antioxidant enzymes are key in regulating their concentration in the body3,4. Under normal conditions, free radicals and antioxidant enzymes maintain a balanced state; however, exercise increases the concentration of free radicals5. At the same time, exercise can enhance the activity and/or quantity of antioxidant enzymes, establishing a new balance that allows the body to tolerate greater oxidative stress6.
Different antioxidant enzymes serve various functions. superoxide dismutase (SOD), a primary antioxidant enzyme, comprises copper-zinc superoxide dismutase (Cu/ZnSOD), manganese superoxide dismutase (MnSOD), and extracellular superoxide dismutase (ECSOD), all of which play crucial roles in removing superoxide anions7. catalase (CAT) and glutathione peroxidase (GPX) reduce hydrogen peroxide to water and oxygen; GPX can also reduce organic hydroperoxides but requires glutathione (GSH) as an electron donor, while CAT does not require an electron donor to reduce hydrogen peroxide8. Glutathione reductase (GR) transfers electrons from NADPH to oxidized GSH, thereby reducing it and, together with GPX, forms the GSH system to maintain redox balance9. Thioredoxin reductase (TrxR), a selenocysteine-containing enzyme, is part of the thioredoxin system that maintains redox homeostasis within cells by reducing protein disulfides (S-S) to dithiols (HS-SH)10.
A considerable body of research indicates that the effects of exercise on various antioxidant enzymes are inconsistent. Some studies have shown that running training increases the activity of CAT, GPX, and mitochondrial superoxide dismutase (SOD2) but does not enhance the activity of cytosolic superoxide dismutase (SOD1). In exercise interventions involving older individuals, no changes were observed in the activity of CAT or GPX11,12. Other studies have found that exercise can increase the activity of SOD1 without affecting GPX activity13. Additionally, some studies report that exercise may increase the protein content of SOD1, CAT, and GPX14. Although exercise generally appears to enhance antioxidant enzyme capacity, discrepancies exist among the findings of different studies.
Regular exercise is widely recognized to improve antioxidant capacity; however, variations in exercise type, the specific antioxidant enzymes measured, and the methods used to measure them can all affect experimental outcomes. This variability necessitates a comprehensive analysis of existing results. Furthermore, a single antioxidant enzyme cannot fully represent the body’s overall antioxidant capacity. Therefore, this study additionally selected total antioxidant capacity (T-AOC) and thiobarbituric acid reactive substances (TBARS) levels from the included literature to support the assessment. T-AOC reflects overall antioxidant capacity, encompassing both enzymatic and non-enzymatic systems (such as vitamins C and E)15,16, while TBARS provides a general measure of lipid peroxidation levels17,18. The objectives of this systematic review are as follows: (1) to summarize patterns of change in multiple antioxidant enzymes in response to exercise, and (2) to explore, through subgroup analysis, the factors contributing to differences in antioxidant enzyme changes.
Methods
Protocol and registration
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines19. It has also been pre-registered in the PROSPERO database under the identifier CRD42023477230.
Search strategy
A comprehensive search was conducted from 2004 to October 2024 across the following databases: PubMed, Web of Science, Embase, and the Cochrane Library. Only literature published in English was included. Authors H.Y. and Y.X. independently reviewed and assessed the relevant articles based on the inclusion criteria. Any disagreements regarding the inclusion or exclusion of articles were resolved through discussion with a third author (Y.G.). The search terms used are detailed in Supplementary File 1.
Inclusion criteria and exclusion criteria
Studies comparing the effects of exercise on different antioxidant enzymes in the human body were included. The inclusion criteria were as follows: (1) The study must be a randomized controlled trial (RCT). (2) The study must include a control group. (3) The research subjects must be healthy individuals. (4) The research results must contain data on at least one antioxidant enzyme. (5) The baseline values of the research subjects must not differ significantly.
Data extraction
Data were extracted from the selected literature using a pre-designed Excel 2019 spreadsheet. The extracted information included study titles, participant characteristics, age, intervention methods, intervention duration, and quality assessments. Two authors (Y.H. and Y.X.) independently performed the data extraction, with any disagreements resolved through discussion with a third author (Y.G.). For studies where mean values were presented only in graphical form, Web Plot Digitizer (https://automeris.io/; 2024.10) was utilized to extract the data. If the data were not presented as “Mean ± SD,” a standardized template for evidence-based medicine data extraction was employed to convert them to “Mean ± SD” format.
Risk of bias assessment
The included randomized controlled trials (RCTs) were assessed for quality using the Cochrane risk of bias tool20. This assessment covered seven domains: random sequence generation/allocation concealment (selection bias), blinding of participants/personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other biases. Each domain was rated on three levels: low risk, high risk, and unclear. The quality of the trials was evaluated by two authors (Y.X. and Y.H.), with discrepancies resolved through discussion with a third author (Y.G.) to reach a consensus.
Statistical analysis
Data on antioxidant enzymes were analyzed using Review Manager 5.3, which included subgroup analysis, sensitivity analysis, and assessment of publication bias through funnel plots. The results were summarized using standardized mean differences (SMD) and 95% confidence intervals (95% CI). The I² statistic was categorized as follows: no significant heterogeneity (I² = 0–30%), moderate heterogeneity (I² = 31–49%), significant heterogeneity (I² = 50–74%), and considerable heterogeneity (I² = 75–100%)21. In the presence of significant heterogeneity, sensitivity analysis and subgroup analysis were performed to identify the sources of heterogeneity.
Results
Search results and study selection
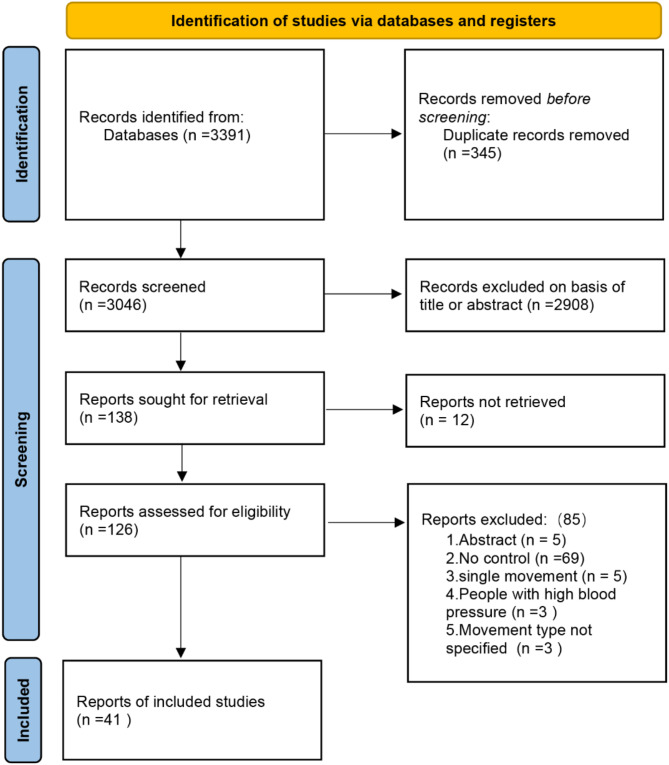
A total of 3391 articles were retrieved from four databases: PubMed (149), Web of Science (431), Embase (2681), and the Cochrane Library (130). After removing 345 duplicate articles, the titles and abstracts of the remaining articles were reviewed, and 2908 articles were excluded, including reviews, animal studies, and non-randomized controlled trials (RCTs). The remaining 138 articles were assessed in full text. During the download process, 12 articles were not found. After thoroughly reviewing the 126 articles, 5 were excluded because they only included abstract information without full text. Additionally, 69 articles lacked a control group and only compared data before and after exercise within the same group, while 5 articles focused on single acute exercise studies, which did not meet the inclusion criteria. Three articles involved participants with diseases, and 3 articles did not specify the type of exercise or provide detailed descriptions of the exercise. Following the full-text review, 85 articles were excluded. Ultimately, 41 articles were included in the analysis (Fig. 1).
Fig. 1.
Flow diagram of study selection
Study characteristics
The meta-analysis included a total of 41 studies (Supplementary Table S1). These studies were conducted between 2002 and 2024 and involved participants aged 15–88 years, with exercise interventions lasting from 14 days to 16 weeks. Based on existing literature on exercise classification, we divided exercise into three categories: resistance exercise aimed at muscle hypertrophy, aerobic exercise aimed at energy expenditure, and combined (mixed) exercise, which incorporates both types. In the studies included in this research, aerobic exercises primarily consisted of activities such as running, soccer, yoga, Pilates, taekwondo, swimming, and cycling. In total, 1,283 participants were included across all studies: 4 studies involved participants under 18 years, 21 studies involved participants aged 18–45 years, 7 studies involved participants aged 45–65 years, and 10 studies involved participants over 65 years. Fifteen studies had intervention durations of less than 8 weeks, 17 studies had durations of 8 to 16 weeks, and 9 studies had durations exceeding 16 weeks. Among the studies, 34 included aerobic exercise, 11 included resistance training, and 4 included combined aerobic and resistance training. Exercise intensity was categorized based on 75% of maximum heart rate or 65% of maximum oxygen uptake22. Thirty-one studies involved low-intensity interventions, and 16 studies involving high-intensity interventions.
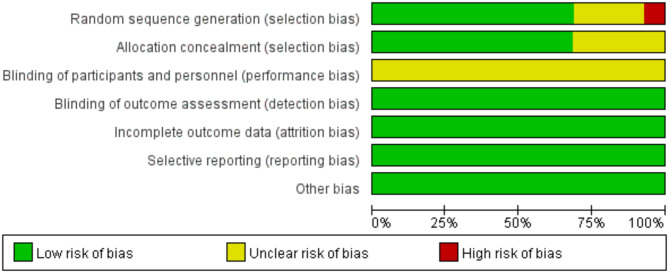
Risk of bias in included studies
Among the 41 included studies, 3 studies were identified as having a high risk of bias in random sequence generation, while 10 studies presented an unclear risk in this area. Additionally, 13 studies exhibited an unclear risk of bias in allocation concealment. Due to the inherent challenges of blinding in exercise interventions, all studies were assessed as having an unclear risk of bias in participants and personnel (Figs. 2 and 3).
Fig. 2.
Risk of bias graph.
Fig. 3.

Risk of bias summary
Effects of the interventions
We first conducted a summary analysis. If significant heterogeneity was identified, subgroup analyses were performed based on age, sex, and type of exercise.
The effect of exercise on SOD
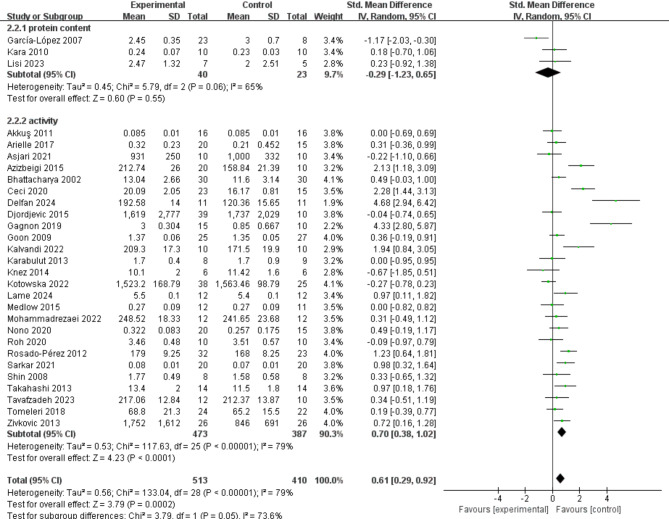
26 studies provided data on the effects of exercise on SOD activity (Fig. 4). The results of the meta-analysis indicated that exercise significantly increases SOD activity (SMD = 0.70; 95% CI: 0.38, 1.02; P < 0.0001). However, the meta-analysis results exhibited considerable heterogeneity (I² = 79%). Funnel plot analysis revealed potential biases in the studies by Delfan (2024) and Gagnon (2019). After excluding these two studies individually, the overall trend remained unchanged, but heterogeneity decreased (I² = 68%). Additionally, three studies provided data on the impact of exercise on SOD protein content, showing no significant effect on increasing SOD protein content (SMD = − 0.29; 95% CI: − 1.23, 0.65; P = 0.55).
Fig. 4.
Effects of exercise on SOD activity
The effect of exercise on CAT
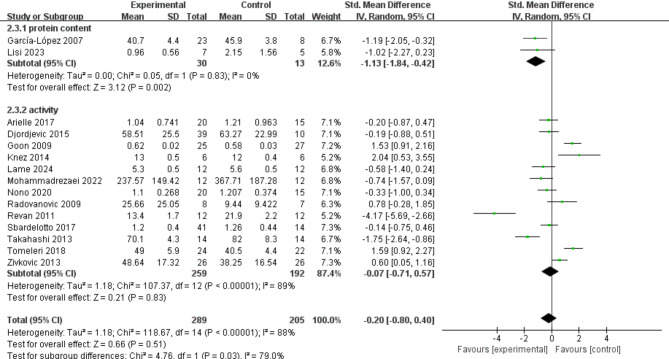
13 studies provided data on the effects of exercise on CAT activity (Fig. 5), and the meta-analysis results indicated that exercise had no significant effect on increasing CAT activity (SMD = − 0.07; 95% CI: − 0.71, 0.57; P = 0.83). The results exhibited high heterogeneity (I² = 89%), although the funnel plot demonstrated good symmetry. Additionally, two studies provided data on the impact of exercise on CAT protein content, showing that exercise significantly reduced CAT protein content (SMD = − 0.07; 95% CI: − 0.71, 0.57; P = 0.83), with low heterogeneity (I² = 0%).
Fig. 5.
Effects of exercise on CAT activity
The effect of exercise on GPX
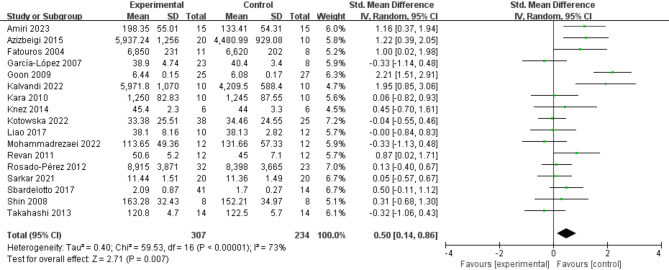
Initially, data from 17 studies were included (Fig. 6), and the meta-analysis results indicated that exercise had a significant effect on increasing GPX activity (SMD = 0.50; 95% CI: 0.14, 0.86; P = 0.007). However, the meta-analysis results exhibited considerable heterogeneity (I² = 73%), although the funnel plot demonstrated good symmetry.
Fig. 6.
Effects of exercise on GPX activity
The effect of exercise on T-AOG
11 studies provided data on the effects of exercise on T-AOC (Fig. 7), and the meta-analysis results indicated that exercise significantly increases T-AOC (SMD = 1.36; 95% CI: 0.54, 2.27; P = 0.003). However, the meta-analysis exhibited considerable heterogeneity (I² = 91%). Funnel plot analysis revealed significant bias in the study by Sinha (2007). Sensitivity analysis using the leave-one-out method did not identify any studies that affected the stability of the results. However, after excluding the Sinha study, heterogeneity significantly decreased (I² = 66%).
Fig. 7.
Effects of exercise on T-AOG
The effect of exercise on TBARS
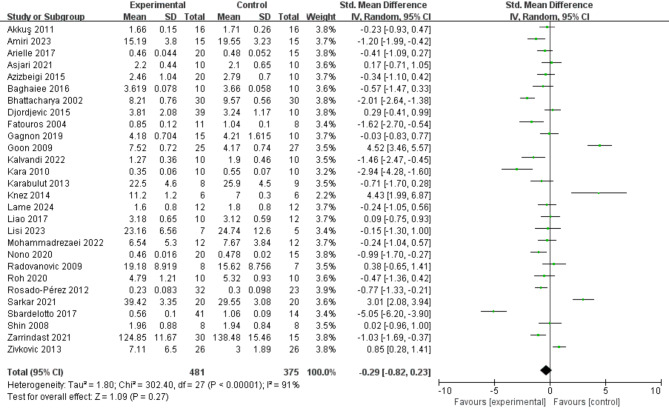
28 studies provided data on the effects of exercise on TBARS (Fig. 8), and the results indicated that exercise had no significant effect on reducing TBARS (SMD = − 0.29; 95% CI: − 0.82, 0.23; P = 0.27). However, the meta-analysis exhibited considerable heterogeneity (I² = 91%). Funnel plot analysis revealed significant bias in the study by Knez (2014). After excluding the Knez study, there was minimal change in both heterogeneity and the overall results.
Fig. 8.
Effects of exercise on TBARS
Subgroup analysis results
Subgroup analyses of SOD, CAT, GPX, T-AOC, and TBARS were conducted based on gender, age, exercise intensity, duration, frequency, and type of exercise.
Subgroup analysis of the effect on SOD activity
In the subgroup analysis by gender, it was found that exercise did not significantly affect the increase in SOD activity in women (SMD = − 0.19; 95% CI: − 0.07, 0.46; P = 0.15), and the results showed no heterogeneity (I² = 0%) (Supplementary Fig. 1). In the subgroup analysis by age, exercise did not significantly improve SOD activity in individuals over 45 years, and the results exhibited considerable heterogeneity. In the subgroup analysis by exercise intensity, both low and high intensities significantly increased SOD activity. In the subgroup analysis by exercise duration, interventions lasting more than 16 weeks did not significantly affect the increase in SOD activity (SMD = 0.28; 95% CI: − 0.33, 0.89; P = 0.37), with considerable heterogeneity in the results (I² = 78%) (Supplementary Fig. 2). In the subgroup analysis by type of exercise, both resistance and aerobic exercises significantly increased SOD activity, while mixed exercise did not show a significant effect on SOD activity (SMD = 1.53; 95% CI: − 0.16, 3.23; P = 0.08), but exhibited considerable heterogeneity (I² = 91%) (Supplementary Fig. 3). In the subgroup analysis by exercise frequency, exercising less than three times per week did not significantly affect the increase in SOD activity (SMD = 0.08; 95% CI: − 1.02, 1.17; P = 0.89), with considerable heterogeneity (I² = 85%) (Supplementary Fig. 4).
Subgroup analysis of the effect on CAT activity
No significant differences were found in the effects of exercise on CAT activity across subgroups based on gender, age, exercise duration, type of exercise, or exercise frequency. However, in the subgroup analysis by exercise intensity, high-intensity exercise significantly reduced CAT activity (SMD = − 0.38; 95% CI: − 0.73, − 0.02; P = 0.04), with moderate heterogeneity in the results (I² = 31%) (Supplementary Fig. 5).
Subgroup analysis of the effect on GPX activity
Regarding the effects of exercise on GPX activity, most studies involved mixed-gender, resulting in insufficient data for subgroup analysis by gender. In the subgroup analysis by age, exercise did not have significant effects on GPX activity across the subgroups. In the subgroup analysis by exercise intensity, high-intensity exercise did not significantly improve GPX activity (SMD = 0.24; 95% CI: -0.42, 0.90; P = 0.48), with significant heterogeneity in the results (I² = 74%) (Supplementary Fig. 6). In the subgroup analysis by exercise duration, only exercise lasting 8 weeks or less significantly improved GPX activity (SMD = 0.54; 95% CI: 0.09, 1.00; P = 0.02), with significant heterogeneity in the results (I² = 59%) (Supplementary Fig. 7). In the subgroup analysis by type of exercise, only resistance exercise significantly increased GPX activity (SMD = 0.80; 95% CI: 0.22, 1.39; P = 0.007), with significant heterogeneity in the results (I² = 60%) (Supplementary Fig. 8). In the subgroup analysis by exercise frequency, exercising less than three times per week did not significantly affect GPX activity (SMD = 0.53; 95% CI: -1.18, 2.24; P = 0.54), although there was considerable heterogeneity (I² = 94%) (Supplementary Fig. 9).
Subgroup analysis of the effect on T-AOC
Regarding the effects of exercise on T-AOC, most studies were mixed-gender, resulting in insufficient data for subgroup analysis by gender. In the subgroup analysis by age, exercise significantly improved T-AOC in the 18–45 age group (SMD = 3.02; 95% CI: 1.05, 4.99; P = 0.003), although considerable heterogeneity was observed in the results (I² = 95%) (Supplementary Fig. 10). In subgroup analyses by exercise intensity, duration, and type, no significant differences were found among the subgroups. In the subgroup analysis by exercise frequency, exercising less than three times per week did not significantly affect T-AOC (SMD = 0.43; 95% CI: − 0.22, 1.09; P = 0.20), with no heterogeneity in the results (I² = 0%) (Supplementary Fig. 11).
Subgroup analysis of the effect on TBARS
In the subgroup analyses by gender, age, and exercise intensity, no significant differences in TBARS were found among the subgroups. However, in the subgroup analysis by exercise duration, exercise lasting 8 to 16 weeks significantly reduced TBARS (SMD = − 0.69; 95% CI: − 1.35, − 0.03; P = 0.04), although considerable heterogeneity was observed in the results (I² = 81%) (Supplementary Fig. 12). In the subgroup analysis by type of exercise, only resistance exercise significantly impacted the reduction of TBARS (SMD = − 1.07; 95% CI: − 1.94, − 0.20; P = 0.02), with significant heterogeneity (I² = 78%) (Supplementary Fig. 13). In the subgroup analysis by exercise frequency, exercising three or more times per week significantly reduced TBARS (SMD = − 0.51; 95% CI: − 0.98, − 0.03; P = 0.04), but there was considerable heterogeneity (I² = 88%) (Supplementary Fig. 14).
4. Discussion
It is generally accepted that exercise can increase antioxidant enzyme levels; however, the trends of changes induced by exercise vary among different antioxidant enzymes. The results of this meta-analysis indicate that long-term physical training does not significantly affect the activity of all antioxidant enzymes, showing a notable impact only on the activities of SOD and GPX, while having no significant effect on CAT activity. Yan’s study suggests that SOD serves as the first line of defense against oxidative stress; if superoxide anions generated by regular exercise training are effectively intercepted by SOD, then exercise may stimulate an increase in SOD without reaching the threshold needed to activate other antioxidant enzymes23. Numerous studies have demonstrated that exercise training increases SOD activity24–26, supporting the findings of this study.
The excess superoxide anions produced during exercise are converted to hydrogen peroxide (H₂O₂) by SOD, and both CAT and GPX function to eliminate excess H₂O₂ in the body. While this study found that exercise does not significantly enhance CAT activity, it does significantly increase GPX activity. Hydrogen peroxide is a unique toxin, stable at environmental temperatures and neutral pH, yet capable of generating highly reactive hydroxyl radicals through the Fenton reaction, which can rapidly kill any type of cell27. To counteract the dangers posed by H₂O₂, all cells possess catalase, a powerful scavenger that allows them to survive in H₂O₂ concentrations thousands of times higher than lethal levels. Due to the transmembrane movement of H₂O₂, it is unlikely to reach toxic concentrations in the body28. The H₂O₂ produced by regular exercise only consumes a small portion of CAT, resulting in a minimal overall effect on total peroxide levels, a conclusion corroborated by several studies29,30. CAT activity was not significantly affected by exercise, which may be because CAT is not the first line of defense among antioxidant enzymes. Therefore, when SOD is able to directly eliminate excess free radicals, CAT may not need to be activated. Another possible explanation is that CAT levels may increase only in individuals who engage in prolonged exercise. In this study, both Goon and Zivkovic underwent approximately six months of aerobic exercise intervention, while Garcia–Lopez’s study, though exceeding 16 weeks, did not reach six months. Excluding Garcia–Lopez’s study from the subgroup analysis on exercise duration yielded a positive result. It has been reported that long-distance runners exhibit higher levels of CAT, which can also be attributed to the duration of exercise. Well-trained individuals or those accustomed to regular exercise may have higher overall antioxidant levels compared to the general population. This could be because long-term training results in a larger reserve of antioxidant enzymes, enabling them to better cope with potential oxidative stress. Although some research published in the 1980s and 1990s indicated that exercise training increases CAT levels in muscle, other studies have not confirmed that exercise induces an increase in CAT activity. Thus, whether exercise increases CAT levels remains a matter of debate8. After synthesizing multiple studies, this research indicates that exercise does not significantly enhance CAT activity.
GPX extends its function beyond merely eliminating hydrogen peroxide; it can also reduce organic hydroperoxides. Most GPXs can effectively reduce small organic hydroperoxides but are unable to address complex hydroperoxides, such as lipid hydroperoxides or cholesterol31. However, GPX4 is an exception; it is the only enzyme in the GPX family capable of directly reducing and eliminating lipid hydroperoxides, even when embedded in biological membranes32,33. The superoxide anions generated during exercise can react with unsaturated fatty acids in the cell membrane, potentially triggering a chain reaction that leads to significant consumption of intracellular GPX434. This may explain the increase in GPX activity following exercise. Several meta-analyses have also indicated that exercise enhances GPX levels in the body13,35,36. Regarding glutathione reductase, thioredoxin peroxidase, and peroxiredoxin, there was insufficient literature available for meta-analysis. T-AOC reflects overall antioxidant capacity, encompassing both the antioxidant enzyme system and non-enzymatic systems (such as vitamins C and E).
Total antioxidant capacity is influenced by both diet and exercise. In the studies included in this research, the intervention group consisted of individuals who only performed exercise, while the control group was a blank control group. Apart from exercise, there were no dietary or other interventions affecting the groups. This approach helps minimize the impact of diet on total antioxidant capacity, makes exercise the main factor affecting the activity of antioxidant enzymes. Measuring the activity of multiple antioxidant enzymes alone does not fully capture the body’s overall antioxidant capacity. For instance, some individuals may have high SOD activity but low CAT activity, and these values cannot simply be added or subtracted to quantify the overall capacity. Therefore, we introduced total antioxidant capacity as a comprehensive measure to assess changes in various antioxidant enzymes. The results of this study indicate that exercise significantly increases T-AOC, which indirectly supports the notion that exercise enhances the activities of SOD and GPX, subsequently leading to an increase in T-AOC. The enhancement of antioxidant enzyme activity improves the body’s ability to adapt to oxidative environments and, to some extent, alleviates oxidative damage. To indirectly assess the effect of exercise on antioxidant capacity, we initially planned to include various markers of oxidative damage in our analysis. In fact, we collected all available markers of oxidative damage from the literature, such as 8-OHdG, protein carbonyl, and TBARS. However, except for TBARS, the data on the other markers were too sparse to conduct a meaningful analysis. As a result, we were only able to include TBARS as an indirect marker of oxidative damage. Regarding the indirect measurement of oxidative stress products using TBARS, the results of this study indicate that exercise has no significant effect on TBARS levels. This may be because the antioxidant enzymes induced by exercise effectively regulate free radicals within physiological ranges, preventing excessive production of TBARS. However, considerable heterogeneity was observed between studies, suggesting that TBARS levels are significantly influenced by certain factors. These results are inconsistent with findings from many other studies. To identify the reasons for these discrepancies, we conducted subgroup analyses and found that exercise frequency and type are the primary factors influencing TBARS levels.
Due to the considerable heterogeneity in the meta-analysis results regarding the effects of exercise on antioxidant enzymes, subgroup analyses were conducted for each enzyme. Interestingly, we found that exercise had minimal impact on SOD activity in women, with no heterogeneity between groups. In contrast, exercise significantly affected SOD activity in men, exhibiting considerable heterogeneity among groups, indicating a potential gender difference in SOD response. In the studies included in this research, 10 studies had male participants, 8 studies had mixed-gender participants, and 8 studies had female participants. Among the 8 studies reporting changes in SOD in women, the total number of participants was 240, with ages ranging from 20 to 71. These studies included both overweight and obese women, as well as women with normal weight. The exercise interventions included resistance training and various types of aerobic exercises. Therefore, we can largely exclude other confounding factors that might affect the results. Wang’s study noted that total SOD activity in men was significantly higher than in women37. Farhat’s study pointed out that the oxidative capacity of the gastrocnemius muscle in female Wistar rats is higher, with greater mitochondrial respiration in states 3 and 4, enhanced enzyme activity in certain electron transport chain complexes, and higher protein content of cytochrome c oxidase. This results in a lower production of free radicals in female rats during exercise compared to males. The effect of exercise on promoting SOD activity is partly attributed to the toxic effects of free radicals. When females experience lower toxic effects, the increase in SOD transcription may be delayed, which could explain why exercise does not significantly affect SOD activity in women. Other studies have shown that estrogen, particularly 17β-estradiol (E2), activates mitogen-activated protein kinase (MAPK), which in turn activates the nuclear factor NF-κB, inducing an increase in the expression of genes encoding antioxidant enzymes. Moderate and regular exercise may reduce estrogen secretion, especially exercises aimed at reducing body fat38. Therefore, in this study, while exercise promotes SOD transcription, the reduction in body fat that lowers estrogen levels may diminish SOD transcription. The combined effect of these factors may explain why exercise does not significantly affect SOD activity in women. This supports the findings of the subgroup analysis, suggesting that specific intervention strategies may be necessary to enhance SOD activity in women, rather than relying solely on regular exercise.
For individuals over 45 years, exercise did not significantly improve SOD activity or T-AOC levels. Silveira’s findings indicated that low-intensity exercise does not increase antioxidant enzyme levels in older individuals39. Ji’s study also pointed out that while aging does not eliminate the training adaptability of antioxidant enzymes, it may weaken this adaptability40. These studies support the results of this research, suggesting that the exercise typically undertaken by older adults, which is often low-intensity aerobic exercise, may not reach the threshold necessary to induce changes in antioxidant enzyme activity.
Regarding the impact of exercise type on antioxidant enzymes, the results of this study indicate that the trends in effects on SOD and CAT activity are similar regardless of the chosen exercise type. However, only resistance exercise was found to significantly increase GPX activity and decrease TBARS levels. Existing evidence suggests that resistance exercise in rats increases the activity of SOD and GPX in skeletal muscle29,30. Studies involving human subjects have shown that resistance exercise enhances the activity of SOD1, SOD2, and GPX41–44. These findings support the results of this research, likely due to the substantial stimulus provided by resistance exercise to local muscles, leading to localized oxidative stress and an increase antioxidant enzyme level. However, this does not imply that resistance exercise is more effective than aerobic exercise in enhancing overall antioxidant enzyme activity, as a single antioxidant enzyme does not represent total antioxidant capacity.
Regarding the effects of exercise intensity on antioxidant enzymes, the results of this study indicate that high-intensity exercise significantly reduces CAT activity but has no significant effect on GPX activity. This suggests that exercise intensity is an important factor influencing antioxidant enzyme activity. One study involving 25 sedentary adults performing cycling exercises at 50%, 60%, and 70% of peak VO2 found a positive correlation between SOD, CAT, and GPX activity and exercise intensity within the range of 70% VO2. It is recommended that the initial exercise intensity for sedentary adults should not exceed 70% VO245. This supports the findings of this study, indicating that moderate to low-intensity exercise is more beneficial for enhancing antioxidant capacity.
When exercise interventions exceed 16 weeks, there is no significant impact on the activity of SOD, CAT, and GPX. This may be related to exercise adaptation; during the initial phase of regular exercise, the free radicals produced may exceed the body’s antioxidant capacity. On one hand, this activates the Nrf2 pathway, increasing the transcription of antioxidant enzymes46,47. On the other hand, it may enhance the efficiency of these enzymes, leading to a significant increase in antioxidant enzyme activity. However, when the duration of exercise is sufficiently long, the adaptive changes in the body’s antioxidant system result in a new equilibrium in the redox system. At this stage, the body has fully adapted to the exercise, leading to no excessive production of free radicals and a reduced need for high levels of antioxidant enzymes.
This study yields an important finding: for enhancing antioxidant capacity in the body, exercising at least three times a week significantly affects the activity of SOD and GPX, as well as T-AOC and TBARS levels. In contrast, if the frequency of exercise falls below three times per week, there are no significant effects on the changes in SOD, GPX, T-AOC, and TBARS. This indicates that among the various subgroup analyses conducted in this study, exercise frequency is a crucial influencing factor. One study that divided participants into high-frequency and low-frequency exercise groups found that lipid peroxidation significantly decreased in the high-frequency group, while CAT activity remained unchanged48. Several studies on mice have similarly confirmed that exercising three times a week has the greatest impact on antioxidant capacity49–51, supporting the findings of this research. When exercise frequency is below three times per week, it is insufficient to stimulate an increase in antioxidant capacity. This suggests that the effect of a single exercise session on antioxidant enzyme levels is short-lived; if the rest period between sessions is too long, the effects will gradually diminish, preventing any cumulative benefit. Fatouros’s study indicated that T-AOC peaks 48 h after exercise and returns to baseline levels after 72 h52, which aligns with the findings of this research.
Most current studies measure antioxidant capacity using two methods: the first involves measuring the protein content of antioxidant enzymes, while the second assesses the activity of these enzymes. Different measurement methods may yield varying results. In the studies included in this analysis, some literature measured the protein content of SOD and CAT. Consequently, this study computed results using both methods, ultimately demonstrating that exercise increased the activity of SOD without raising its protein content. Exercise did not significantly affect CAT activity but resulted in a reduction in CAT protein content. This suggests that the catalytic efficiency of SOD and CAT may have increased. According to the principle of conservation, the body does not require excessive amounts of SOD and CAT, which explains why their protein content did not increase53. Although an increase in antioxidant enzyme activity indicates the positive effects of exercise, a lack of change in antioxidant enzyme activity does not imply that exercise has had no beneficial impact. It may be that exercise enhances the catalytic efficiency of antioxidant enzymes, allowing the body to maintain similar antioxidant capacity while producing fewer enzymes, which could be related to adaptive changes resulting from long-term exercise.
Future research should focus on studying the collaborative interactions between antioxidant enzymes, rather than merely describing the activity of individual enzymes. When measuring antioxidant enzyme outcome indicators, it is recommended to combine both protein content and enzyme activity. This approach can help determine whether the observed increase in antioxidant enzyme activity is due to enhanced activity of individual enzymes or an overall increase in their content.
Limitations
Some studies use the protein content of antioxidant enzymes as an endpoint to reflect changes in antioxidant capacity, which can be misleading. Many experiments select a single antioxidant enzyme to represent overall antioxidant capacity; however, different outcome indicators used to measure antioxidant capacity can yield varying results. Aside from SOD, CAT, and GPX, there is a notable lack of reference data for other antioxidant enzymes, each of which plays an indispensable role in the antioxidant defense system. This presents a significant challenge for analyzing the overall antioxidant enzyme system.
Conclusions
This study found that exercise significantly enhances the activity of most antioxidant enzymes and total antioxidant capacity. Moderate to low-intensity exercise, performed at least three times per week for over 16 weeks, was most effective in improving antioxidant enzyme activity. Interestingly, we also discovered that women may require greater effort than men to achieve enhancements in antioxidant enzyme activity.
Electronic supplementary material
Below is the link to the electronic supplementary material.
The following supporting information can be downloaded at: http://www.mdpi.com/xxx/s1, Supplementary File 1: Searching Strategy; Supplementary Fig. 1. Effect of gender on SOD activity; Supplementary Fig. 2. Effect of exercise time on SOD activity; Supplementary Fig. 3. Effect of exercise types on SOD activity; Supplementary Fig. 4. Effect of exercise frequency on SOD activity; Supplementary Fig. 5. Effect of exercise intensity on CAT activity; Supplementary Fig. 6. Effects of exercise intensity on GPX activity; Supplementary Fig. 7. Effect of exercise duration on GPX activity; Supplementary Fig. 8. Effects of exercise type on GPX activity; Supplementary Fig. 9. Effect of exercise frequency on GPX activity; Supplementary Fig. 10. Effect of age on T-AOC; Supplementary Fig. 11. Effect of exercise frequency on T-AOC; Supplementary Fig. 12. Effect of exercise duration on TBARS; Supplementary Fig. 13. Effect of exercise type on TBARS; Supplementary Fig. 14. Effect of exercise frequency on TBARS; Supplementary Table 1: Characteristic of included trials. References54–93 are cited in the Supplementary Materials
Acknowledgements
Not applicable.
Author contributions
Author Contributions: Conceptualization, Y.H.; methodology, Y.X. and Y.H.; software, Y.X. and Y.G.; validation, Y.X. and Y.H.; formal analysis, Y.X., Y.G., Z.L., L.Z. and Y.H.; investigation, Y.X. and Y.H.; resources, Y.X. and Y.H.; data curation, Y.X. and Y.H.; writing—original draft preparation, Y.X. and Y.G.; writing—review and editing, Y.X. and Y.H.; visualization, Y.H.; supervision, Y.X. and Y.H.; project administration, Y.X., Y.G., Z.L., L.Z. and Y.H.; funding acquisition, Y.X. and Y.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data availability
Data are available upon request, contact: Yongchao Xie, 15637175236@163.com.
Declarations
Competing interests
The authors declare no competing interests.
Institutional review board
Not applicable.
Informed consent
Not applicable.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Powers, S. K., Radak, Z., Ji, L. L. & Jackson, M. Reactive oxygen species promote endurance exercise-Induced adaptations in skeletal muscles. J. Sport Health Sci.13, 780. 10.1016/j.jshs.2024.05.001 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yin, H., Xu, L. & Porter, N. A. Free radical lipid peroxidation: mechanisms and analysis. Chem. Rev.111, 5944–5972. 10.1021/cr200084z (2011). [DOI] [PubMed] [Google Scholar]
- 3.Kuhn, M. A. Oxygen free radicals and antioxidants. Am. J. Nurs.103, 58–62. 10.1097/00000446-200304000-00022 (2003). [DOI] [PubMed] [Google Scholar]
- 4.Sharma, G. N., Gupta, G., Sharma, P. A comprehensive review of free radicals, antioxidants, and their relationship with human ailments. Crit. Rev. Eukaryot. Gene Expr. 28, 139–154. 10.1615/CritRevEukaryotGeneExpr.2018022258 (2018). [DOI] [PubMed] [Google Scholar]
- 5.Morales-Alamo, D. & Calbet, J. L. Free radicals and sprint exercise in humans. Free Radic Res.48, 30–42. 10.3109/10715762.2013.825043 (2014). [DOI] [PubMed] [Google Scholar]
- 6.Cooper, C. E., Vollaard, N. B., Choueiri, T., Wilson, M. T. Exercise free radicals and oxidative stress. Biochem. Soc. Trans.30, 280–285 (2002). [DOI] [PubMed] [Google Scholar]
- 7.Miao, L. & St. Clair, D. K. Regulation of superoxide dismutase genes: implications in diseases. Free Radic Biol. Med.47, 344–356. 10.1016/j.freeradbiomed.2009.05.018 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Powers, S. K. et al. Exercise-induced oxidative stress: friend or foe?? J. Sport Health Sci.9, 415–425. 10.1016/j.jshs.2020.04.001 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xie, Y., Kang, R., Klionsky, D. J. & Tang, D. GPX4 in cell death, autophagy, and disease. Autophagy19, 2621–2638. 10.1080/15548627.2023.2218764 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chupakhin, E. & Krasavin, M. Thioredoxin reductase inhibitors: updated patent review (2017-Present). Expert Opin. Ther. Pat.31, 745–758. 10.1080/13543776.2021.1899160 (2021). [DOI] [PubMed] [Google Scholar]
- 11.Lambertucci, R. H., Levada-Pires, A. C., Rossoni, L. V., Curi, R. & Pithon-Curi, T. C. Effects of aerobic exercise training on antioxidant enzyme activities and mRNA levels in soleus muscle from young and aged rats. Mech. Ageing Dev.128, 267–275. 10.1016/j.mad.2006.12.006 (2007). [DOI] [PubMed] [Google Scholar]
- 12.Oh-ishi, S. et al. Effects of aging and/or training on antioxidant enzyme system in diaphragm of mice. Respir. Physiol.105, 195–202. 10.1016/0034-5687(96)00057-6 (1996). [DOI] [PubMed] [Google Scholar]
- 13.Kwon, I. et al. Elevation of hepatic autophagy and antioxidative capacity by endurance exercise is associated with suppression of apoptosis in mice. Ann. Hepatol.19, 69–78. 10.1016/j.aohep.2019.08.010 (2020). [DOI] [PubMed] [Google Scholar]
- 14.Jang, Y., Kwon, I., Song, W., Cosio-Lima, L. M. & Lee, Y. Endurance exercise mediates neuroprotection against MPTP-mediated Parkinson’s disease via enhanced neurogenesis, antioxidant capacity, and autophagy. Neuroscience379, 292–301. 10.1016/j.neuroscience.2018.03.015 (2018). [DOI] [PubMed] [Google Scholar]
- 15.Çekiç, S. D., Çetinkaya, A., Avan, A. N. & Apak, R. Correlation of total antioxidant capacity with reactive oxygen species (ROS) consumption measured by oxidative conversion. J. Agric. Food Chem.61, 5260–5270. 10.1021/jf3051297 (2013). [DOI] [PubMed] [Google Scholar]
- 16.Sies, H. Total antioxidant capacity: appraisal of a concept. J. Nutr.137, 1493–1495. 10.1093/jn/137.6.1493 (2007). [DOI] [PubMed] [Google Scholar]
- 17.Chakraborty, S., Singh, O. P., Dasgupta, A., Mandal, N. & Nath Das, H. Correlation between lipid peroxidation-induced TBARS level and disease severity in obsessive-compulsive disorder. Prog Neuropsychopharmacol. Biol. Psychiatry. 33, 363–366. 10.1016/j.pnpbp.2009.01.001 (2009). [DOI] [PubMed] [Google Scholar]
- 18.Ialongo, C. Preanalytic of total antioxidant capacity assays performed in serum, plasma, urine and saliva. Clin. Biochem.50, 356–363. 10.1016/j.clinbiochem.2016.11.037 (2017). [DOI] [PubMed] [Google Scholar]
- 19.Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. PRISMA group preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med.6, e1000097. 10.1371/journal.pmed.1000097 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Higgins, J. P. T. et al. The Cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ343, d5928. 10.1136/bmj.d5928 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Higgins, J. P. T., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ327, 557–560. 10.1136/bmj.327.7414.557 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xiong, X. et al. Which type of exercise during radiation therapy is optimal to improve fatigue and quality of life in men with prostate cancer?? A bayesian network analysis. Eur. Urol. Open. Sci.43, 74–86. 10.1016/j.euros.2022.07.008 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yan, Z. & Spaulding, H. R. Extracellular superoxide dismutase, a molecular transducer of health benefits of exercise. Redox Biol.3210.1016/j.redox.2020.101508 (2020). [DOI] [PMC free article] [PubMed]
- 24.Alves, J. O. et al. Strenuous acute exercise induces slow and fast twitch-dependent NADPH oxidase expression in rat skeletal muscle. Antioxidants9, 57. 10.3390/antiox9010057 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hyatt, H. W. et al. Comparative changes in antioxidant enzymes and oxidative stress in cardiac, fast twitch and slow twitch skeletal muscles following endurance exercise training. Int. J. Physiol. Pathophysiol. Pharmacol.8, 160 (2016). [PMC free article] [PubMed] [Google Scholar]
- 26.Lawler, J. M., Kwak, H. B., Song, W. & Parker, J. L. Exercise training reverses downregulation of HSP70 and antioxidant enzymes in Porcine skeletal muscle after chronic coronary artery occlusion. Am. J. Physiol. Regul. Integr. Comp. Physiol.291, R1756–1763. 10.1152/ajpregu.00271.2006 (2006). [DOI] [PubMed] [Google Scholar]
- 27.Toyokuni, S., Ito, F., Yamashita, K., Okazaki, Y. & Akatsuka, S. Iron and thiol redox signaling in cancer: an exquisite balance to escape ferroptosis. Free Radic Biol. Med.108, 610–626. 10.1016/j.freeradbiomed.2017.04.024 (2017). [DOI] [PubMed] [Google Scholar]
- 28.Mahaseth, T. & Kuzminov, A. Potentiation of hydrogen peroxide toxicity. Mutat. Res.773, 274–281. 10.1016/j.mrrev.2016.08.006 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gomes, M. J. et al. Effects of aerobic and resistance exercise on cardiac remodelling and skeletal muscle oxidative stress of infarcted rats. J. Cell. Mol. Med.24, 5352–5362. 10.1111/jcmm.15191 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Scheffer, D. L. et al. Impact of different resistance training protocols on muscular oxidative stress parameters. Appl. Physiol. Nutr. Metab.37, 1239–1246. 10.1139/h2012-115 (2012). [DOI] [PubMed] [Google Scholar]
- 31.Pei, J., Pan, X., Wei, G. & Hua, Y. Research progress of glutathione peroxidase family (GPX) in redoxidation. Front. Pharmacol.1410.3389/fphar.2023.1147414 (2023). [DOI] [PMC free article] [PubMed]
- 32.Jia, M. et al. Redox homeostasis maintained by GPX4 facilitates STING activation. Nat. Immunol.21, 727–735. 10.1038/s41590-020-0699-0 (2020). [DOI] [PubMed] [Google Scholar]
- 33.Forcina, G. C. & Dixon, S. J. GPX4 at the crossroads of lipid homeostasis and ferroptosis. Proteomics19, e1800311. 10.1002/pmic.201800311 (2019). [DOI] [PubMed] [Google Scholar]
- 34.Fujii, J., Homma, T. & Osaki, T. Superoxide radicals in the execution of cell death. Antioxidants1110.3390/antiox11030501 (2022). [DOI] [PMC free article] [PubMed]
- 35.Shin, Y. A., Lee, J. H., Song, W. & Jun, T. W. Exercise training improves the antioxidant enzyme activity with no changes of telomere length. Mech. Ageing Dev.129, 254–260. 10.1016/j.mad.2008.01.001 (2008). [DOI] [PubMed] [Google Scholar]
- 36.Bogdanis, G. C. et al. Short-term high-intensity interval exercise training attenuates oxidative stress responses and improves antioxidant status in healthy humans. Food Chem. Toxicol.61, 171–177. 10.1016/j.fct.2013.05.046 (2013). [DOI] [PubMed] [Google Scholar]
- 37.Wang, D. M., Chen, D. C., Wang, L. & Zhang, X. Y. Sex differences in the association between symptoms and superoxide dismutase in patients with never-treated first-episode schizophrenia. World J. Biol. Psychiatry. 22, 325–334. 10.1080/15622975.2020.1805510 (2021). [DOI] [PubMed] [Google Scholar]
- 38.Farhat, F., Amérand, A., Simon, B., Guegueniat, N. & Moisan, C. Gender-dependent differences of mitochondrial function and oxidative stress in rat skeletal muscle at rest and after exercise training. Redox Rep.22, 508–514. 10.1080/13510002.2017.1296637 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Silveira, E. M. S. et al. Age-related changes and effects of regular low-Intensity exercise on gait, balance, and oxidative biomarkers in the spinal cord of Wistar rats. Braz J. Med. Biol. Res.52 (e8429). 10.1590/1414-431X20198429 (2019). [DOI] [PMC free article] [PubMed]
- 40.Ji, L. L. Exercise-induced modulation of antioxidant defense. Ann. N Y Acad. Sci.959, 82–92. 10.1111/j.1749-6632.2002.tb02085.x (2002). [DOI] [PubMed] [Google Scholar]
- 41.Parise, G., Phillips, S. M., Kaczor, J. J. & Tarnopolsky, M. A. Antioxidant enzyme activity is up-regulated after unilateral resistance exercise training in older adults. Free Radic Biol. Med.39, 289–295. 10.1016/j.freeradbiomed.2005.03.024 (2005). [DOI] [PubMed] [Google Scholar]
- 42.Brinkmann, C. et al. Training alters the skeletal muscle antioxidative capacity in non-insulin-dependent type 2 diabetic men. Scand. J. Med. Sci. Sports. 22, 462–470. 10.1111/j.1600-0838.2010.01273.x (2012). [DOI] [PubMed] [Google Scholar]
- 43.García-López, D. et al. Effects of strength and endurance training on antioxidant enzyme gene expression and activity in middle-aged men. Scand. J. Med. Sci. Sports. 17, 595–604. 10.1111/j.1600-0838.2006.00620.x (2007). [DOI] [PubMed] [Google Scholar]
- 44.Mesquita, P. H. C. et al. Effects of resistance training on the redox status of skeletal muscle in older adults10 (Antioxidants, 2021). 10.3390/antiox10030350 [DOI] [PMC free article] [PubMed]
- 45.Awang Daud, D. M., Ahmedy, F., Baharuddin, D. M. P. & Zakaria, Z. A. Oxidative stress and antioxidant enzymes activity after cycling at different intensity and duration. Appl. Sci.1210.3390/app12189161 (2022).
- 46.Kerins, M. J. & Ooi, A. The roles of NRF2 in modulating cellular iron homeostasis. Antioxid. Redox Signal.29, 1756–1773. 10.1089/ars.2017.7176 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Tonelli, C., Chio, I. I. C. & Tuveson, D. A. Transcriptional regulation by Nrf2. Antioxid. Redox. Signal.2910.1089/ars.2017.7342 (2018). [DOI] [PMC free article] [PubMed]
- 48.Borges, J. P. et al. The impact of exercise frequency upon microvascular endothelium function and oxidative stress among patients with coronary artery disease. Clin. Physiol. Funct. Imaging. 38, 840–846. 10.1111/cpf.12492 (2018). [DOI] [PubMed] [Google Scholar]
- 49.Souza-Rabbo, M. P. et al. Influence of exercise training frequency on cardiac and hepatic oxidative stress in rats. Exp. Clin. Cardiol.8, 201–205 (2003). [PMC free article] [PubMed] [Google Scholar]
- 50.Almeida, A. A. et al. Nephroprotective effect of exercise training in Cisplatin-Induced renal damage in mice: influence of training protocol. Braz J. Med. Biol. Res.55 (e12116). 10.1590/1414-431X2022e12116 (2022). [DOI] [PMC free article] [PubMed]
- 51.Kim, J. S., Lee, Y. H., Choi, D. Y. & Yi, H. K. Expression of heat shock proteins (HSPs) in aged skeletal muscles depends on the frequency and duration of exercise training. J. Sports Sci. Med.14, 347–353 (2015). [PMC free article] [PubMed] [Google Scholar]
- 52.Fatouros, I. G. et al. Time-Course of changes in oxidative stress and antioxidant status responses following a soccer game. J. Strength. Cond. Res.2410.1519/JSC.0b013e3181b60444 (2010). [DOI] [PubMed]
- 53.Gore, M. et al. Endurance training alters antioxidant enzyme gene expression in rat skeletal muscle. Can. J. Physiol. Pharmacol.10.1139/y98-125 (2011). [DOI] [PubMed] [Google Scholar]
- 54.Akkuş, H. Effects of acute exercise and aerobic exercise training on oxidative stress in young men and women. Afr. J. Pharm. Pharmacol.5, 1925–1931. 10.5897/AJPP11.540 (2011). [Google Scholar]
- 55.Amiri, E. & Sheikholeslami-Vatani, D. The role of resistance training and creatine supplementation on oxidative stress, antioxidant defense, muscle strength, and quality of life in older adults. Front. Public. Health. 11 (1062832). 10.3389/fpubh.2023.1062832 (2023). [DOI] [PMC free article] [PubMed]
- 56.Andersen, T. R., Schmidt, J. F., Pedersen, M. T., Krustrup, P. & Bangsbo, J. The effects of 52 weeks of soccer or resistance training on body composition and muscle function in + 65-year-old healthy males—a randomized controlled trial. PloS One. 1110.1371/journal.pone.0148236 (2016). [DOI] [PMC free article] [PubMed]
- 57.Arielle, P., Goedecke, J., Maarman, G. & Mendham, A. Systemic oxidative stress in obese black South African women and effect of exercise training. J. Endocrinol. Metabolism Diabetes South. Afr.22, 33–34 (2017). [Google Scholar]
- 58.Asjari, M., Abedi, B. & Fatolahi, H. Effects of aerobic training and licorice extract consumption on inflammation and antioxidant States in overweight women. Obes. Med.2110.1016/j.obmed.2020.100271 (2021).
- 59.Azizbeigi, K., Azarbayjani, M. A., Atashak, S. & Stannard, S. R. Effect of moderate and high resistance training intensity on indices of inflammatory and oxidative stress. Res. Sports Med.23, 73–87. 10.1080/15438627.2014.975807 (2015). [DOI] [PubMed] [Google Scholar]
- 60.Baghaiee, B., Botelho Teixeira, A. M. & Tartibian, B. Moderate aerobic exercise increases SOD-2 gene expression and decreases leptin and malondialdehyde in middle-aged men. Sci. Sports. 31, e55–e63. 10.1016/j.scispo.2015.12.003 (2016). [Google Scholar]
- 61.Beltran Valls, M. R. et al. Explosive type of moderate-resistance training induces functional, cardiovascular, and molecular adaptations in the elderly. Age36, 759–772. 10.1007/s11357-013-9584-1 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Bhattacharya, S., Pandey, U. S. & Verma, N. S. Improvement in oxidative status with yogic breathing in young healthy males. Indian J. Physiol. Pharmacol.46, 349–354 (2002). [PubMed] [Google Scholar]
- 63.Ceci, R. et al. Endurance Training Improves Plasma Superoxide Dismutase Activity in Healthy Elderly185 (MECHANISMS OF AGEING AND DEVELOPMENT, 2020). 10.1016/j.mad.2019.111190 [DOI] [PubMed]
- 64.Delfan, M. et al. Unveiling the effects of interval resistance training and chlorella vulgaris supplementation on Meteorin-like protein and oxidative stress in obese men. Curr. Dev. Nutr.810.1016/j.cdnut.2024.104428 (2024). [DOI] [PMC free article] [PubMed]
- 65.Djordjevic, D. et al. The effects of two fitness programs with different metabolic demands on oxidative stress in the blood of young females. Serb. J. Exp. Clin. Res.16, 101–107. 10.1515/SJECR-2015-0013 (2015). [Google Scholar]
- 66.Fatouros, I. G. et al. Oxidative stress responses in older men during endurance training and detraining. Med. Sci. Sports. Exerc.36, 2065–2072. 10.1249/01.MSS.0000147632.17450.FF (2004). [DOI] [PubMed] [Google Scholar]
- 67.Gagnon, D. D. et al. Multi-Day prolonged Low- to Moderate-Intensity endurance exercise mimics training improvements in metabolic and oxidative profiles without concurrent chromosomal changes in healthy adults. Front. Physiol.1010.3389/fphys.2019.01123 (2019). [DOI] [PMC free article] [PubMed]
- 68.Goon, J. A. et al. Effect of Tai Chi exercise on DNA damage, antioxidant enzymes, and oxidative stress in middle-age adults. J. Phys. Act. Health. 6, 43–54. 10.1123/jpah.6.1.43 (2009). [DOI] [PubMed] [Google Scholar]
- 69.Kalvandi, F., Azarbayjani, M. A., Azizbeigi, R. & Azizbeigi, K. Elastic resistance training is more effective than vitamin D3 supplementation in reducing oxidative stress and strengthen antioxidant enzymes in healthy men. Eur. J. Clin. Nutr.76, 610–615. 10.1038/s41430-021-01000-6 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kara, E. et al. Effect of zinc supplementation on antioxidant activity in young wrestlers. Biol. Trace Elem. Res.134, 55–63, 10.1007/s12011-009-8457-z (2010). [DOI] [PubMed]
- 71.Karabulut, A. B., Emin Kafkas, M., Kafkas, A. S., Önal, Y. & Kiran, T. R. The effect of regular exercise and massage on oxidant and antioxidant parameters. Indian J. Physiol. Pharmacol.57, 378–383 (2013). [PubMed] [Google Scholar]
- 72.Knez, W. L., Jenkins, D. G. & Coombes, J. S. The effect of an increased training volume on oxidative stress. Int. J. Sports Med.35, 8–13. 10.1055/s-0033-1333746 (2014). [DOI] [PubMed] [Google Scholar]
- 73.Kotowska, J., Jówko, E., Cieśliński, I., Gromisz, W. & Sadowski, J. IL-6 and HSPA1A gene polymorphisms May influence the levels of the inflammatory and oxidative stress parameters and their response to a chronic swimming training. Int. J. Environ. Res. Public Health. 1910.3390/ijerph19138127 (2022). [DOI] [PMC free article] [PubMed]
- 74.Lame-Jouybari, A. H. & Abbasalizad-Farhangi, M. Effects of eight-week regular high-intensity interval training and hemp (Cannabis sativa L.) seed on total testosterone level among sedentary young males: double-blind, randomized, controlled clinical trial. Endocrine84, 273–286. 10.1007/s12020-023-03629-8 (2024). [DOI] [PubMed] [Google Scholar]
- 75.Liao, L. Y., Chung, W. S. & Chen, K. M. Free radicals and antioxidant enzymes in older adults after regular senior elastic band exercising: an experimental randomized controlled pilot study. J. Adv. Nurs.73, 108–111. 10.1111/jan.13094 (2017). [DOI] [PubMed] [Google Scholar]
- 76.Lisi, V. et al. Steady-state redox status in circulating extracellular vesicles: a proof-of-principle study on the role of fitness level and short-term aerobic training in healthy young males. Free Radic. Biol. Med.204, 266–275. 10.1016/j.freeradbiomed.2023.05.007 (2023). [DOI] [PubMed] [Google Scholar]
- 77.Medlow, P. et al. Exercise training protects the LDL I subfraction from oxidation susceptibility in an aged human population. Atherosclerosis239, 516–522. 10.1016/j.atherosclerosis.2015.02.012 (2015). [DOI] [PubMed] [Google Scholar]
- 78.Mohammadrezaei, A., Kavakeb, A., Abbasalizad-Farhangi, M. & Mesgari-Abbasi, M. Effects of hemp seed alone and combined with aerobic exercise on metabolic parameters, oxidative stress, and neurotrophic factors in young sedentary men. J. Food Biochem.46 (e14417). 10.1111/jfbc.14417 (2022). [DOI] [PubMed]
- 79.Nono Nankam, P. A. et al. Changes in systemic and subcutaneous adipose tissue inflammation and oxidative stress in response to exercise training in obese black African women. J. Physiol.598, 503–515. 10.1113/JP278669 (2020). [DOI] [PubMed] [Google Scholar]
- 80.Radovanovic, D. et al. Oxidative stress biomarker response to concurrent strength and endurance training. Gen. Physiol. Biophys.28, 205–211 (2009). [PubMed] [Google Scholar]
- 81.Revan, S. & Erol, A. E. Effects of endurance training on exhaustive exercise-induced oxidative stress markers. Afr. J. Pharm. Pharmacol.5, 437–441. 10.5897/AJPP11.159 (2011). [Google Scholar]
- 82.Roh, H. T., Cho, S. Y. & So, W. Y. Effects of regular Taekwondo intervention on oxidative stress biomarkers and myokines in overweight and obese adolescents. Int. J. Environ. Res. Public Health. 1710.3390/ijerph17072505 (2020). [DOI] [PMC free article] [PubMed]
- 83.Rosado-Pérez, J., Santiago-Osorio, E., Ortiz, R. & Mendoza-Núñez, V. M. Tai Chi diminishes oxidative stress in Mexican older adults. J. Nutr. Health Aging. 1–5. 10.1007/s12603-012-0029-9 (2012). [DOI] [PubMed]
- 84.Sarkar, S. et al. Effect of high intensity interval training on antioxidant status, inflammatory response and muscle damage indices in endurance team male players. Apunts Sports Med.5610.1016/j.apunsm.2021.100352 (2021).
- 85.Sbardelotto, M. L. et al. The effects of physical training are varied and occur in an exercise type-dependent manner in elderly men. Aging Dis.. 8, 887–898. 10.14336/AD.2017.0209 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Sinha, S., Singh, S. N., Monga, Y. P. & Ray, U. S. Improvement of glutathione and total antioxidant status with yoga. J. Altern. Complement. Med.13, 1085–1090. 10.1089/acm.2007.0567 (2007). [DOI] [PubMed] [Google Scholar]
- 87.Takahashi, M. et al. Low-Volume exercise training attenuates oxidative stress and neutrophils activation in older adults. Eur. J. Appl. Physiol.113, 1117–1126. 10.1007/s00421-012-2531-5 (2013). [DOI] [PubMed] [Google Scholar]
- 88.Tavafzadeh, S. S. et al. Effects of aerobic dance exercise and honey supplementation followed by their subsequent cessation on bone metabolism markers and antioxidant status in young collegiate females. Malays. J. Med. Sci.30, 151–166. 10.21315/mjms2023.30.3.14 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Tomeleri, C. M. et al. Correlations between resistance training-induced changes on phase angle and biochemical markers in older women. Scand. J. Med. Sci. Sports. 28, 2173–2182. 10.1111/sms.13232 (2018). [DOI] [PubMed] [Google Scholar]
- 90.Wahyuni, N., Griadhi, I. P. A. & Saraswati, P. A. S. The effects of yoga exercise on manganese superoxide dismutase (Mnsod) levels and anthropometric parameters in abdominal obesity populations. Int. J. Pharma Med. Biol. Sci.10, 35–39. 10.18178/ijpmbs.10.1.35-39 (2020). [Google Scholar]
- 91.Zarrindast, S., Ramezanpour, M. R. & Moghaddam, M. G. Effects of eight weeks of moderate intensity aerobic training and training in water on DNA damage, lipid peroxidation and total antioxidant capacity in sixty years sedentary women. Sci. Sports. 36, e81–e85. 10.1016/j.scispo.2020.04.005 (2021). [Google Scholar]
- 92.Zivkovic, V. et al. Alteration in basal redox state of young male soccer players after a six-month training programme. Acta Physiol. Hung.100, 64–76. 10.1556/APhysiol.100.2013.1.6 (2013). [DOI] [PubMed] [Google Scholar]
- 93.Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ372, n71. 10.1136/bmj.n71 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The following supporting information can be downloaded at: http://www.mdpi.com/xxx/s1, Supplementary File 1: Searching Strategy; Supplementary Fig. 1. Effect of gender on SOD activity; Supplementary Fig. 2. Effect of exercise time on SOD activity; Supplementary Fig. 3. Effect of exercise types on SOD activity; Supplementary Fig. 4. Effect of exercise frequency on SOD activity; Supplementary Fig. 5. Effect of exercise intensity on CAT activity; Supplementary Fig. 6. Effects of exercise intensity on GPX activity; Supplementary Fig. 7. Effect of exercise duration on GPX activity; Supplementary Fig. 8. Effects of exercise type on GPX activity; Supplementary Fig. 9. Effect of exercise frequency on GPX activity; Supplementary Fig. 10. Effect of age on T-AOC; Supplementary Fig. 11. Effect of exercise frequency on T-AOC; Supplementary Fig. 12. Effect of exercise duration on TBARS; Supplementary Fig. 13. Effect of exercise type on TBARS; Supplementary Fig. 14. Effect of exercise frequency on TBARS; Supplementary Table 1: Characteristic of included trials. References54–93 are cited in the Supplementary Materials
Data Availability Statement
Data are available upon request, contact: Yongchao Xie, 15637175236@163.com.