Abstract
The mouse scaramanga (ska) mutation impairs mammary gland development such that both abrogation and stimulation of gland formation occurs. We used positional cloning to narrow the interval containing scaramanga (ska) to a 75.6-kb interval containing the distal part of the Neuregulin3 (Nrg3) gene. Within this region the only sequence difference between ska and wild-type mice is in a microsatellite repeat within intron 7. This alteration correlates with variations in Nrg3 expression profiles both at the whole embryo level and locally in the presumptive mammary region in ska mice. Localized expression of Nrg3 and its receptor, Erbb4, in the presumptive mammary region around the future bud site prior to morphological appearance of buds and the expression of bud epithelial markers further support an inductive role. Finally, Neuregulin3 (Nrg3)-soaked beads can induce expression of the early bud marker Lef1 in mouse embryo explant cultures, and epithelial bud formation can be observed histologically, suggesting that initiation of mammary bud development occurs. Taken together, these results indicate that a Neuregulin signaling pathway is involved in specification of mammary gland morphogenesis and support the long-held view that mesenchymal signal(s) are responsible for mammary gland inductive/initiating events.
Keywords: Mammary gland development, Neuregulin3, specification
The selectable and heritable nature of mammary gland number in sheep was demonstrated by Alexander Graham Bell over 100 years ago (Bell 1898). Subsequent analysis in mice supported the suggestion that a genetic component was involved (Little and McDonald 1965). Specification of the mammary epithelial phenotype is thought to occur during embryogenesis when signals from the mesenchyme pattern the overlying ectoderm such that the epithelial cells aggregate locally to form the mammary bud (Propper and Gomot 1967; Hogan 1999). Genetic studies have implicated Fgf10, Lef1, and Tbx3 in the control of developmental processes such as competency and determination during early mammary gland morphogenesis (van Genderen et al. 1994; Mailleux et al. 2002; Davenport et al. 2003; Eblaghie et al. 2004). However, none of these genes have yet been directly implicated in mammary gland specification per se and the precise genetic interactions and hierarchies involved in the initial stages of mammary gland morphogenesis remain to be elucidated (Veltmaat et al. 2003).
Five pairs of mammary glands form at precise and invariant positions along the anterior-posterior, dorsal-ventral axis in most strains of mice. However, variable positions and numbers of mammary glands are observed in the A/J strain of mice, whereas the C57BL/6 (B6) strain is wild type for mammary gland patterns. The locus in A/J mice responsible for aberrant mammary gland phenotypes is called scaramanga (ska) (Howard and Gusterson 2000a,b). In ska homozygotes, the number 3 mammary bud is hypoplastic from embryonic day 11.75 (E11.75), leading to a reduced sized or absent bud, indicating a defective inductive event (Howard and Gusterson 2000a,b). In addition, supernumerary glands are formed at a high frequency in ska mice, demonstrating ectopic induction. The mammary phenotypes observed in the A/J strain are all consistent with defective specification events occurring prior to the morphological appearance of the mammary bud. Here we provide evidence that the gene affected in ska mice encodes the Erbb4 ligand, Neuregulin3 (Nrg3) (Zhang et al. 1997; Hijazi et al. 1998; Gizatullin et al. 2000). Taken together, these results indicate that Nrg3 is a specification signal for mammary buds.
Results
Aberrant mammary gland morphogenesis in ska mutant embryos
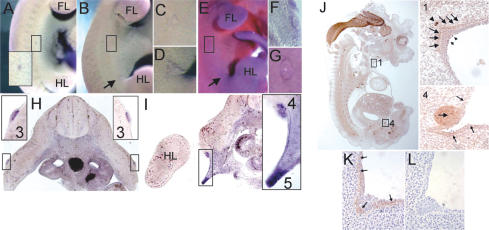
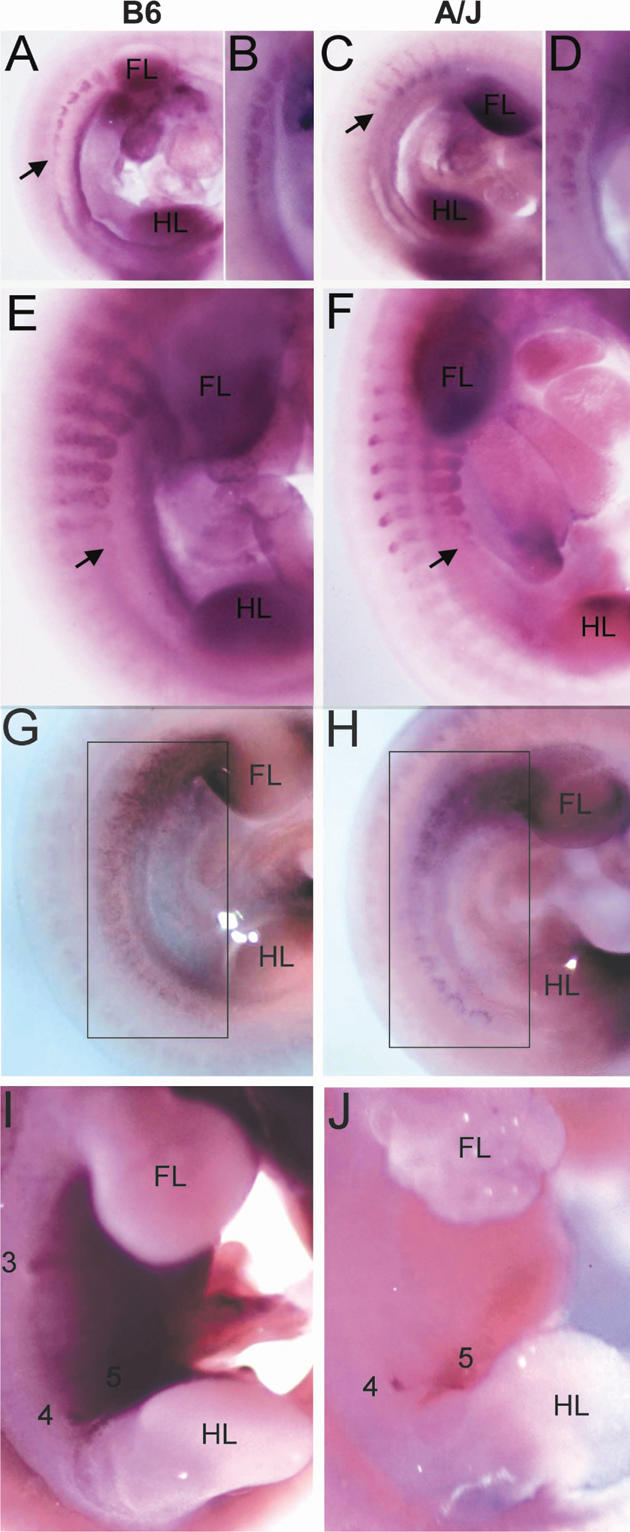
The ska mutant phenotype is easily detected from E11.75 (48-somite stage) onward using differences in the expression of the early mammary epithelial bud marker, Lef1 (Fig. 1). Lef1 is expressed in the buds once they have become morphologically distinct and appears to mark the determined mammary bud epithelia from somite stage 48 onward (Mailleux et al. 2002). However, Lef1 expression is absent in A/J mice in the region where mammary bud 3 would normally form (Fig. 1B). Lef1 expression is also frequently seen slightly dorsal to the endogenous bud 4 at the site of ectopic mammary buds in A/J mice (Fig. 1E). These ectopic sites of Lef1 expression are often much smaller than the normal mammary buds and appear connected to the endogenous bud 4 by a line of Lef1-expressing cells at somite stage 54 (Fig. 1E) and appear in the same location as ectopic mammary glands that form in ska mutant mice (Howard and Gusterson 2000a,b).
Figure 1.
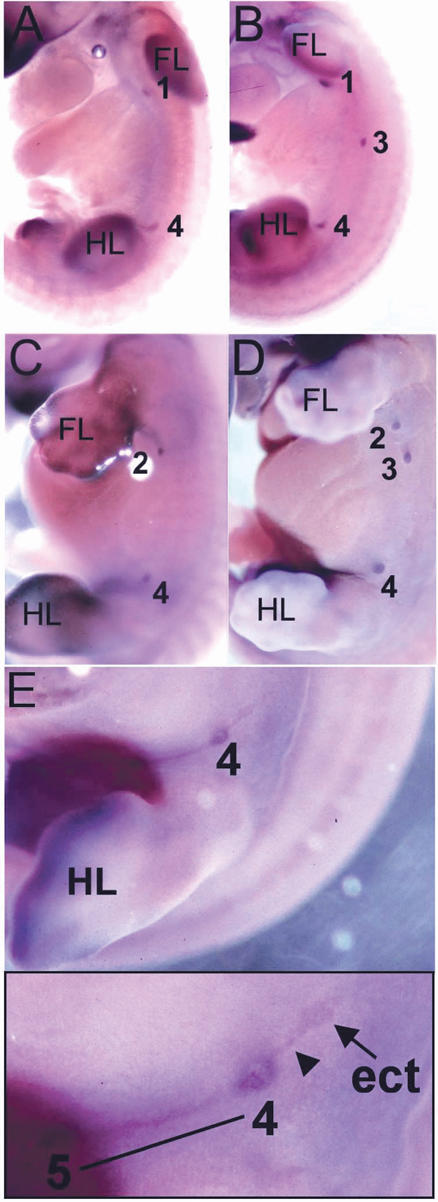
Mammary bud 3 is absent and ectopic mammary buds form in A/J mice. Lef1 expression marks the position of mammary buds. Numbers denote the positions of the five mammary gland anlage. (FL) Forelimb bud; (HL) hindlimb bud. (A,B) Mammary buds are first observed in 48-somite embryos when bud 2 has not yet formed. Mammary bud 3 has not formed in an A/J embryo (A) shown by comparison with B6 (B). (C-E) Mammary buds in E12.5 embryos. (C) A 53-somite A/J embryo with absent bud 3. (D) A 55-somite B6 embryo. (E) A 54-somite A/J embryo with a presumptive supernumerary mammary bud adjacent to mammary bud 4. This is shown in magnified view in the inset. The ectopic Lef1-expressing cells are denoted by an arrow and a line of Lef1-expressing cells connecting bud 4 to the ectopic bud is marked by an arrowhead. At this stage, there is a line of Lef1-expressing cells connecting bud 4 to bud 5 (denoted by a black underline).
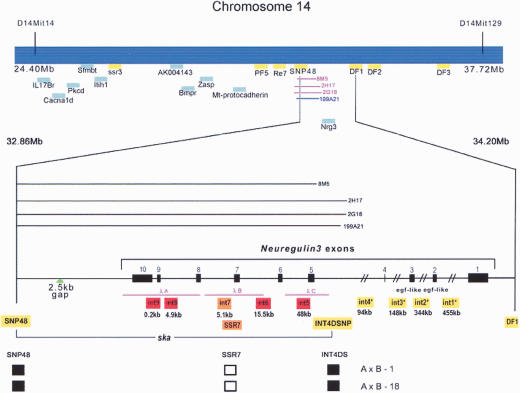
Physical mapping of the ska locus
We previously mapped ska to the proximal region of mouse chromosome (Chr) 14 between microsatellite markers D14Mit14 and D14Mit129 using the AXB/BXA recombinant inbred (RI) mouse strains (Marshall et al. 1992; Howard and Gusterson 2000b). DNA sequence from mouse genome databases (Celera Genomics and Ensembl) indicated that these markers delimited a 13.32-Mb portion of Chr 14 (Waterston et al. 2002). Analysis of this genomic region resulted in the identification of novel markers polymorphic between the two parental strains, A/J and B6. These were mapped onto the RI panel, narrowing the candidate interval to 75.6 kb (Fig. 2; Supplementary Table 1; Supplementary Fig. 1). This critical region contains the distal part of the Neuregulin3 (Nrg3) gene, which encodes a secreted growth factor of the Neuregulin family. Nrg3 is a large gene spanning over 1 Mb of Chr 14, but SNPs or polymorphic SSRs were found at 100 kb and 8 kb upstream from the transcription initiation site of Nrg3, within introns 1, 2, 3, and 4, and 17.3 kb downstream of the 3′UTR and were used to exclude these regions. Intron 4 and exons 5-10 of Nrg3 and 17.3 kb beyond the 3′UTR of the gene remained within the candidate interval (Fig. 2). No DNA sequence differences in any exons or in intron/exon junctions were observed between the A/J and B6 strains. Furthermore, complete sequence analysis of the entire 75,614-bp region revealed that there is only a single sequence difference in a microsatellite within Nrg3 intron 7 between A/J and B6 (Supplementary Fig. 1). This SSR was found to completely cosegregate with the ska phenotype. Therefore, the distal half of Nrg3 is coincident with the critical minimal genomic region containing ska. This same microsatellite variant is also present in several other closely related “Castle” inbred strains (Balb/c, C3H/HeJ, DBA/1, and DBA/2) that also display the ska mutant phenotype, providing further support for ska as an allele of Nrg3 (Supplementary Fig. 1; Witmer et al. 2003).
Figure 2.
Genetic and physical maps of the ska genomic region. Map of a 13.32-Mb genomic interval between markers D14Mit14 and D14Mit129 on mouse Chr 14. Genetic markers from transcription units (in blue) and polymorphic sequences in intragenic regions (in yellow) used to delimit the position of the ska mutation are highlighted. Primer sequences for these markers are tabulated in Supplementary Table 1. The Neuregulin3 intron-exon structure is indicated in the lower exploded view. Asterisks indicate Nrg3 introns, which contain polymorphic genetic marker(s). A/J bacteriophage λ clones (λA, B, and C) are indicated. The location of three A/J BAC clones (Chori27: 2G18, 2H17, and 8M5) and one B6 BAC clone (RP24-199A21) used to sequence the 75.6-kb candidate interval are indicated. A 2.5-kb gap in the Ensembl Celera genomic assemblies was sequenced and this location is indicated. The genomic contig spanning the ska critical region for both A/J and B6 strains will be deposited in GenBank. Haplotype data of informative recombinant inbred mice are shown. Each column represents the type of chromosome identified in these mice. A black square represents the B6 allele and an open square denotes the A/J allele.
Nrg3 expression in the presumptive mammary region
We considered Nrg3 a strong candidate for ska since the most well-known member of the Neuregulin family, Neuregulin1/Heregulin, has been implicated in a variety of cell fate and patterning processes, including lobular-alveolar development of the post-natal mammary gland (Yang et al. 1995; Jones et al. 1996; Burden and Yarden 1997; Troyer and Lee 2001; Li et al. 2002; Falls 2003; Stern 2003). Therefore, we examined the pattern of expression of Nrg3 during embryogenesis to assess whether it was consistent with a role in early morphogenesis of the mammary gland. Nrg3 is expressed at many sites in the mid-gestation embryo, including widespread neural expression as previously reported (Zhang et al. 1997), but also in the atria, branchial arches, olfactory placode, otic vesicles, limb buds, and tail bud (data not shown). We also examined Nrg3 expression using immunohistochemical analysis.
Prior to the development of the mammary gland, Nrg3 expression is observed in the lateral plate mesoderm underlying the future site of mammary bud 3, which is the first of the five buds to form (Fig. 3A-G). Localized Nrg3 expression in the presumptive mammary region is observed from somite stage 38 (E10.75) when Nrg3 is expressed as a comet in the lateral plate mesoderm spanning the level of somites 17-18 (Fig. 3A,B). From somite stage 40, a comet of Nrg3-expressing cells is also observed in the lateral plate mesoderm at the future site of mammary bud 4, followed by the appearance of Nrg3-expressing cells at the other sites of mammary bud formation (Fig. 3B-D). At the 45-somite stage, Nrg3 is expressed in the lateral plate mesoderm around the anlage outlining a distinct bud shape (Fig. 3E-G). Later, from somite stage 48 onward, when bud 3 is morphologically distinct, expression is first detected in the mammary bud epithelia (Fig. 3H). The other four buds form slightly later (and asynchronously in the order 4, 1, 2, and 5), and localized mesenchymal expression is also detected in the lateral plate mesoderm prior to the appearance of each bud, followed by epithelial expression once the bud has formed (Fig. 3I). By somite stage 54, when the bud proper has formed, the epithelial cells of bud 3 express Nrg3 (data not shown). Since the buds form asynchronously, buds 3 and 4, which form first, express Nrg3 in the epithelia, while bud 1, which forms slightly later, expresses Nrg3 in the mesenchyme (Fig. 3J). By E13, all mammary buds express Nrg3 in the epithelia (Fig. 3K). In A/J mice (and also in another ska mutant strain, Balb/c), Nrg3 is not expressed in a localized fashion around the site of presumptive bud 3 at comparable embryo stages (data not shown). Expression prior to actual morphological formation of the mammary buds is consistent with a role for Nrg3 in early initiating events.
Figure 3.
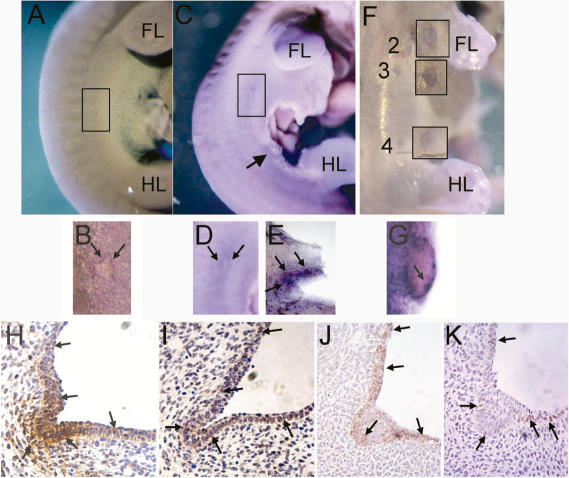
Expression of Neuregulin3 and Neuregulin3 in the presumptive mammary region of E10.75, E11.0, E11.75, and E12.5 (B6) embryos. Whole-mount in situ hybridization of embryos with digoxigenin-labeled Nrg3 probes and immunohistochemical detection of Nrg3. Numbers denote the positions of the mammary gland anlage. (FL) Forelimb bud; (HL) hindlimb bud. The presumptive site of mammary bud 3 (at somite 18) is boxed and of bud 4 (at somite 24) is indicated by an arrow in embryos where mammary buds are not yet morphologically distinct. When the buds are morphologically distinct, they are indicated by numbers. (A-L) Nrg3 transcript and Nrg3 protein distribution in the presumptive mammary region. (A) E10.75: A 38-somite-stage embryo with a comet of Nrg3 expression underlying the future site of number 3 mammary gland. The boxed area at the level of somite 18 is magnified in the inset. (B) E11.0: A 41-somite-stage embryo with a dermal comet of Nrg3 expression surrounding the future site of the number 3 mammary gland. (C) Higher magnification of B at the level of somite 18. (D) Higher magnification of B at the level of somite 24. (E) E11.5: A 45-somite-stage embryo, with Nrg3 expression in the dermal mesenchyme underlying where buds 3 and 4 will subsequently form. (F) A vibratome cross-section (40 μm) across the level of somite 18, where a number 3 mammary bud will form in the embryo shown in E, showing Nrg3 expression in the mesenchyme underlying the nascent mammary anlage. (G) Higher magnification of E at the level of somite 18. (H) E11.75: A vibratome cross-section (60 μm) across the level of somite 18 where mammary bud 3 has just formed in a 47-somite-stage embryo showing Nrg3 expression in the epithelia of the nascent mammary anlage. Bud 3 is boxed and shown in magnification in insets. (I) E11.75: A vibratome cross-section (60 μm) across the level of somite 24, where the number 4 mammary bud has just formed in the same 47-somite-stage embryo in H, showing Nrg3 expression in the nascent mammary anlage 4 and 5. The region where buds 4 and 5 are forming is boxed and shown in magnification in the inset. Nrg3 is expressed in the epithelia of bud 4 and in the mesenchyme underlying the future site of mammary bud 5. (J) E12.5: A sagittal-section of a 54-somite-stage embryo showing Nrg3 expression. Widespread neural expression is observed as well as expression in mammary buds 1 and 4, which are boxed. Inset 1 shows higher magnification of mammary gland anlage 1. Arrows denote Nrg3 expression in the mesenchyme adjacent to the mammary epithelia. A few mammary epithelial cells express Nrg3 (shorter arrows). An arrowhead denotes nerve expressing Nrg3. Inset 4 shows higher magnification of mammary gland bud 4. An arrow denotes the epithelial cells of the mammary bud that express Nrg3. The epithelia adjacent to the mammary bud also express Nrg3 (smaller arrows). (K) E13.0: A section from a 60-somite-stage embryo showing Nrg3 expression in the mammary bud 4 and adjacent epithelia. (L) E13.0: A section stained with hematoxylin from a 60-somite-stage embryo showing mammary bud 4 and adjacent epithelia.
Nrg3 displays a complex transcriptional profile and expression is locally decreased in the presumptive mammary region
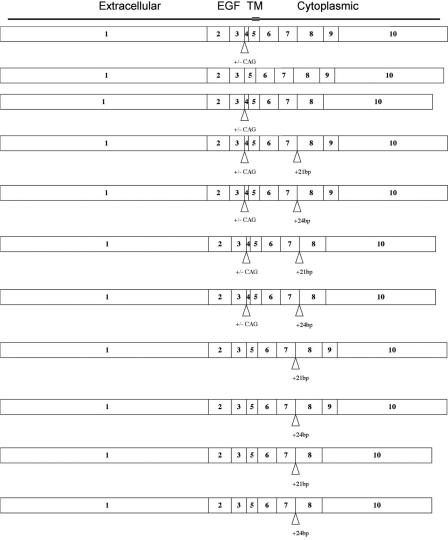
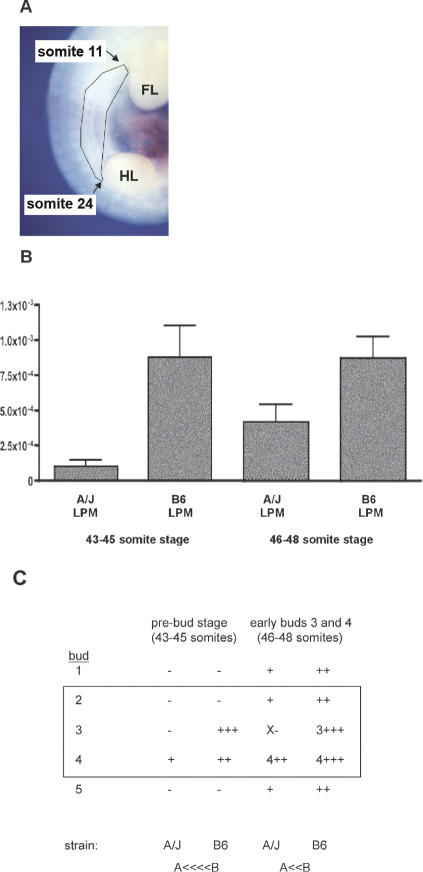
Transcript analysis was determined by direct sequencing of RT-PCR products. This revealed that multiple isoforms of Nrg3 are expressed in mid-gestation mouse embryos, providing further intricacy to this highly complex signaling network. We found that multiple Nrg3 isoforms are locally expressed in the lateral plate mesoderm of the presumptive mammary region prior to the appearance of the mammary bud (Fig. 4; Supplementary Fig. 2). We observed altered levels of expression of Nrg3 in the region where buds 2, 3, and 4 will subsequently form. This was determined by quantitative RT-PCR (qRT-PCR) of microdissected lateral plate mesoderm and its overlying ectoderm from the presumptive mammary region (Fig. 5A) in stage-matched samples from A/J and B6 mice at stages prior to mammary bud formation. To validate the microdissection, we performed semiquantitative RT-PCR using Tbx3 as a marker for lateral plate mesoderm (Supplementary Fig. 3). We observed (on average) a 5.0-fold reduction in Nrg3 mRNA levels in the region where buds 2, 3, and 4 will subsequently form in A/J mice in 43-45-somite-stage embryos (Fig. 5B). In addition, a 1.7-fold reduction in Nrg3 levels in the region where buds 2, 3, and 4 form in A/J mice in 46-48-somite-stage embryos (Fig. 5B) was noted.
Figure 4.
Nrg3 isoforms expressed in presumptive mammary region of B6 and A/J mice. To determine the isoform(s) relevant for mammary gland morphogenesis, we isolated RNA from microdissected lateral plate mesoderm and overlying ectoderm at stages prior to mammary bud formation. RT-PCR analysis revealed the subset of Nrg3 isoforms that are expressed in the presumptive mammary region during embryogenesis. Numbers denote exons. The Egf domain and extracellular, transmembrane, and cytoplasmic domains are indicated. A variant in exon 4 in which the first 3 bp are skipped is indicated by +/- CAG. Inclusion of intronic sequences between exons 7 and 8 is indicated by +21 and +24.
Figure 5.
Nrg3 isoforms expressed in presumptive mammary region. (A) Tissues were microdissected from the presumptive mammary region from E11 embryos between 43 and 45 somites and 46 and 48 somites so that the region spanning where buds 2-4 will form (between somites 11 and 24 of the presumptive mammary region) were isolated. (FL) Forelimb bud; (HL) hindlimb bud. (B) Differential expression of Nrg3 was observed in the lateral plate mesoderm and overlying ectoderm isolated from the A/J strain compared with that isolated from B6. Quantitative real-time RT-PCR was used to determine the level of Nrg3 expressed in the presumptive mammary region. The table shows the relative number of transcripts encoding Nrg3 isoforms containing the Egf domain after normalization to Gapdh. (C) Summary of Nrg3 transcription in the mammary region as determined by qRT-PCR and WMISH. (+) Nrg3 transcript levels at future bud sites; (-) no detectable Nrg3 transcripts at site (as determined by WMISH); (×) hypoplastic bud. Numbers denote buds that are morphologically distinct; these transcripts are post-specification. The region of tissue used for analysis in A is boxed.
Erbb2 and Erbb4 are expressed in the presumptive mammary region
We examined the expression pattern of the gene for the cognate receptor for Nrg3, Erbb4, in the presumptive mammary region (Fig. 6). Like Nrg3, Erbb4 expression also precedes the morphological appearance of the mammary buds and mirrors the expression pattern of Nrg3 (Fig. 6A-E). Erbb4 is expressed in 40-46-somite-stage embryos in the lateral plate mesoderm at the sites of future mammary buds (Fig. 6E). From the 52-somite stage (E12.5), Erbb4 is expressed in the outer layer of the mammary bud epithelial (Fig. 6F,G). Erbb2, the preferred heterodimerization partner for all Erbb receptors, is expressed in the nascent mammary anlage in both the epithelia and adjacent mesenchyme (Fig. 6H). From the first morphological appearance of the mammary bud, Erbb4 is expressed in the bud epithelia (Fig. 6I). By E13, all of the mammary buds express Erbb4, as well as Erbb2 (Fig. 6J,K). Therefore, the Nrg3 ligand and the receptors Erbb2 and Erbb4 are expressed at very early stages of mammary gland organogenesis, consistent with functional early roles.
Figure 6.
Erbb4 expression in the presumptive mammary region. (A-F) Expression of Erbb4 in the presumptive mammary region of E11.0, E11.5, and E12.5 (B6) embryos. Whole mounts of embryos were subjected to in situ hybridization with digoxigenin-labeled Erbb4 probes. Numbers denote the positions of the mammary gland anlage. (FL) Forelimb bud; (HL) hindlimb bud. The site of presumptive mammary bud 3 (at somite 18) is boxed and of bud 4 (at somite 24) is indicated by an arrow in embryos where mammary buds are not yet morphologically distinct. Mammary bud 3 is boxed and indicated by “3” in embryos where this bud is morphologically distinct. (A-F) Erbb4 transcript distribution in the presumptive mammary region. (A) E10.75: A 40-somite-stage embryo with a comet of Erbb4 expression underlying the future site of number 3 mammary gland. (B) Higher magnification of A at the level of somite 18. (C) E11.5: A 46-somite-stage embryo, showing Erbb4 expression in the dermal mesenchyme underlying the area where buds 3 and 4 will subsequently form. (D) Higher magnification of C at the level of somite 18. E. A vibratome sagittal section (60 μm) of mammary bud 4 in the embryo shown in C showing Erbb4 expression in the mesenchyme underlying the future site of mammary bud 4. (F) E12.5: A 53-somite-stage embryo showing Erbb4 expression in mammary buds 2, 3, and 4. Magnified insets are adjacent to the mammary buds. (G) Vibratome cross-section (60 μm) of mammary bud 3 in the embryo shown in E showing Erbb4 expression in the outer epithelial layer of the mammary bud. (H-K) Expression of Erbb2 and Erbb4 in the mammary region at E12.5-E13. (H) E12.5: A section from 54-somite-stage embryo showing Erbb2 expression in the mammary anlage 1 epithelia and underlying mesenchymal cells and the adjacent epithelia. (I) E12.5: A section from a 54-somite-stage embryo showing Erbb4 expression in the mammary anlage 1 and adjacent epithelia. (J) E13.0: A section from 60-somite-stage embryo showing Erbb2 expression in the mammary bud 4 and adjacent epithelia. (K) E13.0: A section from 60-somite-stage embryo showing Erbb4 expression in the mammary bud 4 and adjacent epithelia.
Expression patterns of major regulators of mammary gland organogenesis in the presumptive mammary region
The precise genetic pathway for mammary gland organogenesis has not yet been elucidated, although several genes (Fgf10, Lef1, Tbx3) have been implicated in mammary gland morphogenesis (van Genderen et al. 1994; Mailleux et al. 2002; Davenport et al. 2003). We determined the expression patterns of these genes in both wild-type and ska mutant embryos. Fgf10 expression precedes that of Nrg3 in the presumptive mammary region, with the first appearance of Fgf10 at the 30-somite stage in the presumptive mammary region (Fig. 7A,B; Supplementary Fig. 4A). From 30 to 45 somites, Fgf10 expression extends as a line from the forelimb bud to the level of somite 18 (where bud 3 will subsequently form) (Fig. 7E; Supplementary Fig. 4C) in the dermamyotome (Mailleux et al. 2002). Tbx3 expression is also observed as a line spanning the entire presumptive mammary region prior to mammary gland morphogenesis from 32- to 45-somite stages in the lateral plate mesoderm (Fig. 7G; Supplementary Fig. 4B). Fgf10 and Tbx3 expression patterns appear similar in ska mutants and wild-type embryos at stages prior to mammary bud formation (Fig. 7C,F,H). Lef1 and Wnt10b expression is altered in ska mutants, with decreased or absent expression at bud 3 and ectopic expression associated with supernumerary buds (Figs. 1, 7I,J; Supplementary 4D).
Figure 7.
Expression patterns of major regulators of mammary gland organogenesis in A/J and B6 strains of mice as determined by whole-mount in situ hybridization. Arrows indicate end of mammary line expression at the edge of somite 18. Boxed areas indicate mammary line expression between limb buds. (FL) Forelimb bud; (HL) hindlimb bud. Fgf10 expression in the presumptive mammary region of a B6 33-somite-stage embryo (A), the mammary line from B6 33-somite-stage embryo shown in A (B), an A/J 32-somite-stage embryo (C), the mammary line from A/J 32-somite-stage embryo shown in C (D), a B6 46-somite-stage embryo (E), and an A/J 46-somite-stage embryo (F). Tbx3 expression in the presumptive mammary region of a B6 42-somite-stage embryo (G) and an A/J 40-somite-stage embryo (H). Wnt10b expression in the presumptive mammary region of a B6 50-somite-stage embryo (I) and an A/J 52-somite-stage embryo (J).
Ectopic mammary buds form when Nrg3-soaked beads are placed adjacent to the dense mesenchyme along the mouse `mammary line'
A functional developmental assay was used to investigate whether Nrg3 could induce the expression of mammary epithelial bud markers. We purified recombinant Nrg3 from an insect cell expression system and demonstrated that it was able to stimulate phosphotyrosine formation in MCF7 cells (data not shown). Embryonic explant culture experiments of E10 and E11 B6 embryos were carried out using agarose beads soaked in either recombinant Nrg3 or EGF or Heregulin-1B; these were placed along the ventral surface of E10 and E11 B6 embryos. Remarkably, Nrg3, but not the related factors EGF and Heregulin-1B, induced ectopic expression of the epithelial mammary bud marker Lef1 as well as a morphological bud when placed below the surface of the ventral ectoderm along the presumptive “mammary line” (Fig. 8; Supplementary Fig. 5). Ectopic mammary buds only form when Nrg3-soaked beads are placed adjacent to the dense mesenchyme along the mouse “mammary line”, an entity that has been identified in mouse by molecular marker (Fgf10, Tbx3, and Wnt10b) expression (Fig. 7A-F; Supplementary Fig. 5A-F; Mailleux et al. 2002; Eblaghie et al. 2004; Veltmaat et al. 2004). We observed condensed mesenchyme that the nascent mammary buds abut, similar to dense mesenchyme that has been previously described by Balinsky (1949-1950) underlying the milk line as it forms in the rabbit, in E11.0 B6 embryos in histological sections (Fig. 9). We observe both endogenous and ectopic mammary buds forming adjacent to this line of condensed mesenchyme after 24 h of culture (Figs. 8, 9, 10; Supplementary Fig. 5). We have not observed any difference in responses to Nrg3-soaked beads implanted at various positions along the anterior-posterior axis of this line (Fig. 8; Supplementary Fig. 6). Nrg3-soaked beads implanted in locations outside of the lateral plate such as the head did not result in ectopic mammary bud marker expression (data not shown). We have also implanted Nrg3- and BSA-soaked beads into the BAT-gal reporter mouse strain (which contain a lacZ gene under the control of B-catenin/T cell factor [TCF] responsive elements) (Maretto et al. 2003) and shown by X-gal staining on whole-mounted embryos that TCF activity is increased by implantation with Nrg3-soaked beads when compared with BSA-soaked beads (Supplementary Fig. 6). Consistent with a previous study of canonical Wnt signaling in the mammary region prior to bud formation, we observed X-gal expression in both the surface epithelium and underlying mesenchyme of BAT-gal mice (Supplementary Fig. 6; Chu et al. 2004). Furthermore, both epithelial and mesenchymal expression of X-gal is increased along the mammary line adjacent to implanted Nrg3-soaked beads (Supplementary Fig. 6).
Figure 8.
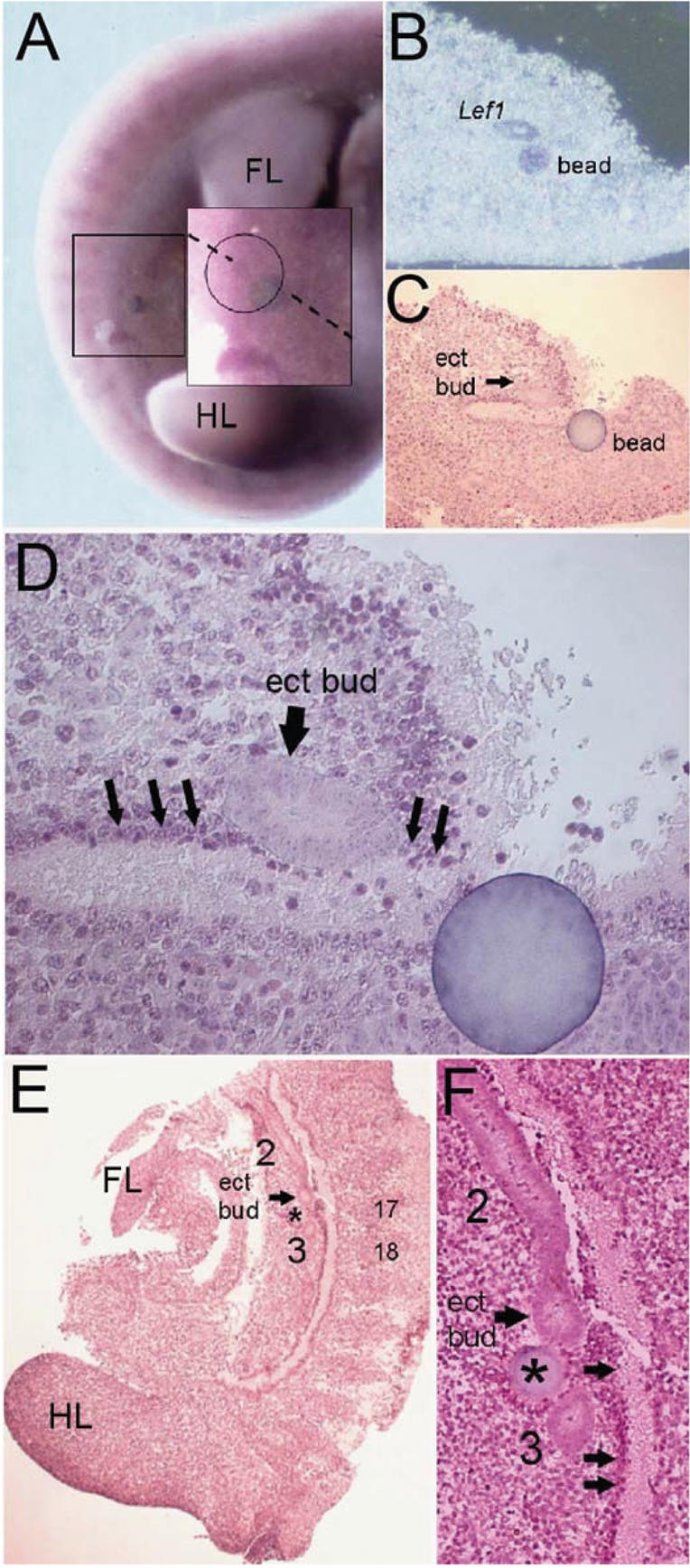
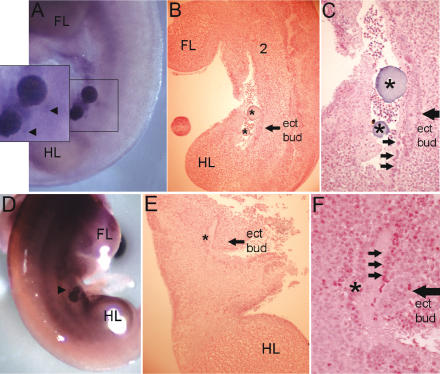
Nrg3 can induce ectopic mammary gland bud formation and Lef1 expression in cultured mouse embryos. Numbers denote the positions of the mammary gland anlage that have formed during the culture period. (FL) Forelimb bud; (HL) hindlimb bud. (A) Whole-mount in situ hybridization with a Lef1 anti-sense probe of a B6 embryo implanted (at the 41-somite stage) with an Nrg3-soaked bead and cultured for 24 h. The bead was implanted along the “mammary line” between the sites where buds 3 and 4 will subsequently form. The boxed area highlights the ectopic bud that has formed adjacent to the implanted bead and is indicated by the circled area in the magnified inset. (B) Paraffin section through an Nrg3-soaked bead shown in A shows that Lef1 expression is confined to the ectopic bud. (C) Hematoxylin-stained section through an Nrg3-soaked bead shown in A shows the location of the ectopic bud. (D) Higher magnification of C. Black arrows indicate the dense mesenchyme adjacent to the ectopic bud. (E) Sagittal hematoxylin-stained section through a B6 embryo implanted (at the 42-somite stage) with an Nrg3-soaked bead and cultured for 24 h. The bead was implanted along the “mammary line” between the sites where buds 2 and 3 will subsequently form. The asterisk indicates the location of the bead. An arrow indicates the location of the ectopic bud. (F) Higher magnification of E. Arrows indicate the dense mesenchyme observed in wild-type (B6) mice along the putative mammary line.
Figure 9.
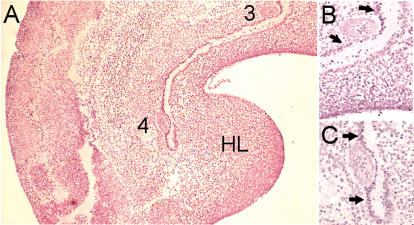
Dense “mammary line” mesenchyme is observed in wild-type (B6) mice at sites where mammary buds form. (HL) Hindlimb bud. (A) Sagittal H&E-stained section of a 46-somite B6 embryo. Numbers denote mammary buds 3 and 4. (B) Magnification of A around bud 3. Arrows indicate the dense mesenchyme at the sites where mammary buds form. (C) Magnification of A around bud 4. Arrows indicate the dense mesenchyme at the sites where mammary buds form.
Figure 10.
Nrg3 can induce ectopic mammary gland bud formation and Lef1 expression in cultured A/J mouse embryos. Numbers denote the positions of the mammary gland anlage, which have formed during the culture period. (FL) Forelimb bud; (HL) hindlimb bud. (A) Whole-mount in situ hybridization with a Lef1 antisense probe of an A/J embryo implanted (at the 40-somite stage) with two Nrg3-soaked beads and cultured for 24 h. The beads were implanted along the “mammary line” between the sites where buds 3 and 4 will subsequently form. The boxed area highlights the ectopic bud that has formed adjacent to the implanted beads and is indicated by arrowheads in the magnified inset. (B) Sagittal hematoxylin-stained section through the A/J embryo shown in A. The asterisks indicate the locations of the beads. An arrow indicates the location of the ectopic bud. This bud spans the anterior-posterior axis that the two beads span and is either larger than a normal bud or the result of the fusion of two nascent buds. (C) Higher magnification of B. Arrows indicate the dense mesenchyme along the putative mammary line, but in ska (A/J) mice this has a more disorganized appearance. (D) Whole-mount in situ hybridization with a Lef1 antisense probe of a 41-somite-stage A/J embryo implanted at the level of somite 20 with an Nrg3-soaked bead and cultured for 24 h. (E) A sagittal hematoxylin-stained section through the A/J embryo shown in D. The asterisk indicates the location of the bead. An arrow indicates the location of the ectopic bud. (F) Higher magnification of E. Arrows indicate dense “mammary line” mesenchyme.
These results indicate that Nrg3 is sufficient, in the context of lateral plate mesoderm, for mammary gland specification. Nrg3-soaked beads implanted into A/J embryos can also induce ectopic mammary bud marker expression and a histological bud both adjacent to the site of bud 3, and at other locations along the presumptive “mammary line” (Fig. 10). This suggests that the ectoderm in ska mutant mice is competent to respond to the signal provided by localized Nrg3 expression and the ska phenotype (absent or hypoplastic gland) is due to a reduction in Nrg3 expression in lateral plate mesoderm.
Discussion
Genetic analysis indicates Nrg3 maps to the ska locus
Genetic analysis delimited an interval within which only a microsatellite sequence varies between wild-type and ska mice. Although we have not yet determined the mechanism by which the sequence change within intron 7 of Nrg3 alters gene expression, similar sequence alterations have been suggested to cause aberrant expression of other genes (Chiba-Falek et al. 1998; Gebhardt et al. 1999; Suzuki et al. 2003; Buerger et al. 2004). We have correlated the presence of this microsatellite sequence variant with the observation of the ska mutant phenotype in several other inbred strains of mice, further supporting this genotype/phenotype association. Taken together, our genetic, gene expression, and functional data strongly suggest that ska is an allele of Nrg3 that results in altered expression of the gene.
Regionally reduced expression of Nrg3 occurs in the presumptive mammary region of ska mice
There is decreased expression of Nrg3 in the presumptive mammary region where buds 2, 3, and 4 will subsequently form in ska mice, as determined by quantitative RT-PCR of microdissected lateral plate mesoderm and overlying ectoderm from the presumptive mammary region (Fig. 5B). We propose that a decreased expression of spatially specific Nrg3 isoforms is the cause of mammary gland abnormalities in ska mice. The decreased expression of Nrg3 is more pronounced at developmental stages (43-45 somites) slightly prior to when the mammary buds 3 and 4 become morphologically distinct (Fig. 5B,C). Mammary bud 3, which forms first, is the most severely affected bud. At slightly later developmental stages (46-48 somites) when the first pairs of buds (3 and 4) are morphologically distinct, Nrg3 levels are also decreased but to a lesser extent such that bud formation proceeds relatively normally in the later-forming buds (Fig. 5B,C). The reduced levels of localized Nrg3 expression in the lateral plate mesoderm and the resulting failure to form mammary bud 3 in ska mice suggests that Nrg3 is a specification signal for bud formation. We suggest that the failure of normal specification of the future mammary cells is due to insufficient levels of Nrg3 in the presumptive bud 3 region. Local expression of Nrg3 around the other future bud sites is presumably not sufficiently altered to prevent overt changes to their specification in ska mutants. However, we have observed subtle phenotypic alterations (variable size and position) associated with the other mammary buds in ska mutant mice, suggesting involvement of Nrg3 signaling in their specification. The mis-specification of cells in the region near bud 4 causing misplaced and ectopic gland formation may be a consequence of the failure of bud 3 formation and/or altered Nrg3 expression surrounding the future bud 4 site. It is possible that the specification of the mammary line is itself defective in ska mutant mice. The dense mammary mesenchyme appears less organized in ska strains when compared with B6 mice (Figs. 8, 9, 10). However, expression of Fgf10 and Tbx3, mesodermal mammary line markers, appears normal in ska mutants (Fig. 7A-H). It is also possible that Nrg3 is acting to transmit signals from Fgf10 and/or Tbx3 to the precursor mammary epithelial cells (which appear to express Wnt10b) (Chu et al. 2004; Veltmaat et al. 2004) and that aberrant Nrg3 signals result in abnormalities in placode formation off the mammary line.
Genetic pathways that regulate early mammary gland morphogenesis
The EGF-receptor superfamily member Erbb4, which is the receptor for Nrg3, is also expressed in the presumptive mammary region (Fig. 6; Zhang et al. 1997). Therefore, the expression patterns of both the ligand Nrg3 and its cognate receptor Erbb4 are consistent with a functional role in mammary gland organogenesis. These two genes are the earliest yet described that are locally expressed in the mesenchyme underlying the future site of mammary buds. Mouse models have established that Erbb4 signaling via Erbb4 homodimers is essential for normal terminal differentiation in the post-natal mammary gland (Long et al. 2003; Tidcombe et al. 2003). As mice lacking Erbb4 do form mammary buds, it is likely that Nrg3 can signal in the absence of Erbb4 homodimers through other Erbbs to specify the embryonic mammary buds. It is of note that Erbb2 is also expressed during early stages of mammary gland morphogenesis (Fig. 6H). This complex and interrelated network may normally elicit mammary specification through the Nrg3/Erbb4 axis, but the inherent redundancy of its signaling capacities is likely to provide alternate means of signaling (Riese et al. 1995; Falls 2003). It is not yet clear if the variety of Nrg3 isoforms contribute to signaling diversity by eliciting distinct biological responses but this is an intriguing possibility.
Knockout studies show that both Fgf10 and Tbx3 are required for bud formation (Mailleux et al. 2002; Davenport et al. 2003). The genetic hierarchy of Fgf10 and Tbx3 remains to be elucidated, but the severity of the phenotype when either is absent indicates that they are both upstream of Nrg3, which is expressed later and in a spatially restricted manner in the presumptive mammary region (Fig. 3; Supplementary Fig. 4C). Both Fgf10 and Tbx3 expression is normal in ska mutants (in stages prior to mammary bud formation as assessed by WMISH) (Fig. 7A-H). The exact role of Fgf10 and Tbx3 in mammary gland morphogenesis remains to be clarified (van Genderen et al. 1994; Davenport et al. 2003). In contrast to Fgf10, Tbx3 is also expressed in the bud proper once the mammary buds have initially formed (Davenport et al. 2003). Lef1-deficient mice can form mammary buds and as both Lef1 and Wnt10b expression is altered in ska mutants, (Figs. 1, 7I,J; Supplementary Fig. 4D), it is likely that Nrg3 is upstream of both of these genes. BAT-gal reporter gene expression is increased in the epithelia and mesenchyme adjacent to implanted Nrg3-soaked beads in cultured explanted embryos, indicating that Wnt signaling can be induced by Nrg3 (Supplementary Fig. 6). Although canonical Wnt signaling is required for mammary gland inductive events, it is not yet known which Wnts are involved and whether the epithelial or mesenchymal Wnts are required for mammary gland initiation (Andl et al. 2002; Chu et al. 2004).
The five pairs of mammary glands have inherent distinctive genetic differences in mouse. Mammary bud 4 is genetically distinct, as both Fgf10 and Fgfr2b null mice fail to form all buds (buds 1, 2, 3, and 5) except for bud 4 (Mailleux et al. 2002). Buds 3 and 5 (which form first and last, respectively) are reported to be absent in Gli3-/- mice (Mailleux et al. 2002). Buds 4 and 5 are initially connected and it would appear that Lef1 and Wnt10b-expressing cells migrate as a line from bud 4 to bud 5 (Figs. 1C-E, 7I,J). It remains to be determined whether the five pairs of mammary buds develop independently of one another or if any type of developmental cascade exists. Our observations suggest that lack of bud 3 formation does not preclude formation of subsequent glands. However, as we often observe unusual placement of the other (later-forming) buds, it is possible that these aberrations are a direct consequence of the abnormal specification of mammary bud 3.
Ectopic mammary buds form adjacent to Nrg3-soaked beads when placed along the `mammary line' mesenchyme
Mammary epithelial buds often form adjacent to Nrg3-soaked beads (Figs. 8, 10; Supplementary Fig. 6). Ectopic mammary buds only form when Nrg3-soaked beads are placed adjacent to the dense mesenchyme along the mouse “mammary line”. Previously both light and scanning electron microscopy have failed to detect a morphological mammary line in the mouse, although a histological ridge has recently been described (Veltmaat et al. 2004). In E11.0 B6 embryos, we have observed condensed mesenchyme that the nascent mammary buds abut, similar to the dense mesenchyme that has been previously observed histologically by Balinsky (1949-1950) underlying the milk line as it forms in the rabbit (Fig. 9). Both endogenous and ectopic mammary buds form adjacent to this line of condensed mesenchyme after 24 h of culture (Figs. 8, 9, 10; Supplementary Fig. 6). In addition, we have demonstrated an increase in TCF activity in explanted embryos implanted along the presumptive mammary line with Nrg3-soaked beads (Supplementary Fig. 6). This suggests that Nrg3 increases Wnt signaling in both mesenchymal and epithelial cells in the presumptive mammary region. Although it has been demonstrated that mammary induction requires Wnt signaling, it is not yet clear which particular Wnt(s) are required or how localized canonical Wnt signaling is achieved (Chu et al. 2004). It is possible that Nrg3 is acting to induce localized Wnt signaling along the mammary line or to prime cells in the presumptive mammary region to respond to Wnt signals. Mammary buds are thought to form from the localized migration of epithelial cells (Propper 1978); it is not yet clear where these epithelial cells come from or how this rapid response is achieved. Fgf, Tbx, Wnt, and Nrg3/Erbb4 signals are expressed in the presumptive mammary region at the time when inductive processes should occur. Erbb4 signaling can regulate neural progenitor migration and placement in the adult forebrain (Anton et al. 2004) and it is possible that Nrg3 signaling may act by influencing the migration of the precursors to mammary epithelial cells.
It has been suggested that the initiating signal for mammary gland formation is mesenchymal (Propper and Gomot 1967; Cunha et al. 1995; Cunha and Hom 1996) and the properties of Nrg3 suggest it as a strong candidate for mediating this signal. Both hypoplastic and ectopic glands should be the result of aberrations during the initial mesenchymal-epithelial signal exchanges. Variable mammary gland phenotypes, with both absent and ectopic glands, may reflect the sensitivity of mammary morphogenesis to local levels of growth factors, as have been described in the human ulnar mammary syndrome (UMS) (Bamshad et al. 1997). The presumptive mammary region in the mouse appears extremely sensitive to variation in local levels of Nrg3, in a manner similar to that observed with respect to the effects of TBX3 haploinsufficiency within UMS families.
The expression pattern of Nrg3 as the gland develops (both embryonically and post-natally) also suggests that this factor might provide a means of signaling that is maintained beyond very early gland development. Nrg3 has the ability to direct precursor epithelial cells to become mammary epithelial cells presumably by defining or specifying a mammary stem cell population. Nrg3 can regulate cell fate decisions, which may have implications for the growth of mammary stem cells in vitro or in tissue engineering. In addition, Bell's approach (Bell 1898) to increase mammary gland number by classical genetic selection methods may now be possible using transgenic Nrg3 expression. Supernumerary nipples in humans are common and it has been estimated that these may be present in as many as one in 18 people (Schmidt 1998). Misregulation of Neuregulin signaling may be responsible, in part, for this phenomenon. Finally, components of the Neuregulin signaling network, such as ERBB2 (the preferred heterodimerization partner for ERBB4) (Graus-Porta et al. 1997) and EGFR, have important clinical relevance in breast cancer (Salomon et al. 1995; Bieche et al. 2003; Lynch et al. 2004; Paez et al. 2004). NRG3 is expressed in some breast cancers (Dunn et al. 2004) and its role in tumorigenesis needs to be explored further.
Materials and methods
Genetic and physical mapping
ska has been previously described and characterized (Howard and Gusterson 2000a,b). Genomic DNAs from the AXB/BXA recombinant inbred mice were purchased from the Jackson Laboratories. PCR genotyping was done by standard protocols (Sambrook et al. 1989). B6 genomic sequence was obtained from the Mouse Genome Sequencing Consortium, Mouse Assembly 3 February 2002, freeze (Waterston et al. 2002) and by directly sequencing BAC clone RP24-199A21, which spans the ska critical region. To obtain DNA sequence from the A/J strain, a bacteriophage λ library from A/J genomic DNA was prepared and screened by hybridization with probes corresponding to nucleotides 1242-1850 and 1923-2538 of GenBank accession no. NM_008734 (Nrg3). Three A/J λ clones (A, B, C on Fig. 2) were directly sequenced. An A/J BAC library CHORI-27 gridded on filters (BACPAC Resource Center) was screened with an Nrg3 probe corresponding to exon 10, and three BAC clones (2G18, 2H17, and 8M5) spanning the ska critical region were sequenced. Additional A/J DNA sequences were obtained from the Celera mouse genome database.
cDNA sequencing
Reverse transcription with oligo(dT) was performed with Thermoscript (Invitrogen) on total RNA isolated from whole embryos or from microdissected tissue. Gene-specific primers were used to amplify cDNA using PCR. PCR products were directly sequenced or subcloned into pTOPO4 (Invitrogen) and sequenced.
Quantitative real-time RT-PCR
Tissues from the presumptive mammary region of E11-E11.5 embryos were microdissected spanning the level of somites 11-24. Five micrograms of total RNA was reverse-transcribed using Thermoscript (Invitrogen). One microliter of the cDNA obtained was subjected to quantitative real-time amplification on the ABI 7700 Sequence Detector using the TaqMan universal PCR Master Mix (ABI). Primer sequences used were Nrg3F, TTGTGATTGAGACCCTGACAGG; Nrg3R, GGCTACCAAGGAGTCCGTTG.
Amplification cycles consisted of an initial 2 min at 50°C, and then 10 min at 95°C, followed by 40 cycles of 15 sec at 95°C and 1 min at 60°C. Analysis was performed using ABI Prism Sequence Detector System (version 1.7) software. Reactions lacking reverse transcriptase were performed to control for genomic DNA amplification. Relative standard curves for both genes were obtained by performing twofold dilutions of a cDNA pool. The relative concentrations were normalized to the Rodent glyceraldehyde 3-phosphate dehydrogenase (Gapdh) (ABI) levels. Fold changes were obtained of A/J relative to B6 samples. Each experiment was repeated using two independent sources of RNA, and within each experimental repeat, amplifications were performed in triplicate. Relative expression of the gene was calculated. The average fold-change ratio and the standard error of the mean were determined.
Tbx3 levels were also determined by semiquantitative RT-PCR as a dissection control to assess microdissection of lateral plate mesoderm and overlying ectoderm. cDNAs used in qRT-PCR experiments were analyzed for Tbx3 mRNA. A 0.5-μL portion of each cDNA was used as a template in PCR containing 0.3 μM of each primer, 2.25 mM MgCl2, 0.5 mM of each dNTP, and 2.5 U of Expand long template enzyme mix (Roche). The amplification conditions were 92°C for 2 min, followed by 25 cycles of 10 sec at 92°C, 30 sec at 65°C, 3 min at 68°C, and a final step of 7 min at 68°C. The following primers were used to amplify part of the Tbx3 coding region: Tbx3F, AAGCGATCACGCAACGTGGCA; Tbx3R, TGCCATTGCCAGTGTCTCGAA. As a control, Gapdh was amplified in parallel.
Whole-mount in situ hybridization
Mouse embryos were from timed B6 or A/J matings. In situ hybridization protocols were as described (Wilkinson and Nieto 1993). Digoxigenin-labeled sense and anti-sense RNA was synthesized following standard protocols (Roche). Probes were generated from plasmids containing mouse Lef (nucleotide positions 1777-2385 of GenBank accession no. NM_010703), Fgf10 (nucleotide positions 266-926 of GenBank accession no. NM_008002), Tbx3 (nucleotide positions 3390-4230 of GenBank accession no. NM_011535), and Wnt10b (nucleotide positions 263-2030 of GenBank accession no. NM_011718). Five Nrg3 probes (corresponding to nucleotides 1-680, 1242-1850, 1923-2538, 827-1321 for exons 2-3, and 1435-1922 of GenBank accession no. NM_008734) were generated. All Nrg3 anti-sense probes gave similar results and sense probes gave no specific signal (data not shown). An Erbb4 anti-sense probe corresponding to nucleotides 558-1527 of GenBank accession no. XM_136682 was generated. A sense probe gave no specific signal (data not shown).
Immunohistochemistry
Sections from Carnoy's-fixed time-mated B6 mouse embryos were incubated with primary antibodies overnight at room temperature. Rabbit polyclonals Erbb2 (C-18, Santa Cruz), Erbb4 (C-18, Santa Cruz), Nrg3 (Orbigen), and Nrg3 (Abgent) and goat polyclonal Nrg3 (R&D Systems) were used. Antibody dilutions were Erbb2, 1:300; Erbb4, 1:400; Nrg3 (Abgent), 1:50; Nrg3 (Orbigen), 1:350; and Nrg3 (R&D Systems), 1:50. Peroxidase-labeled polymer (Envision rabbit, DAKO) was used for detection of primary antibodies raised in rabbits. Biotin-labeled donkey anti-goat IgG antibody (Molecular Probes) was used for detection of the goat antibody using Vector ABC kit (Vector Labs). 3,3′ diaminobenzidine was used as chromagen and sections were counterstained with hematoxylin.
Embryo explant cultures
Recombinant Nrg3 was prepared as described by Hobbs (Hobbs et al. 2002). The coding region of Nrg3 spanning the two EGF repeats (Ser284-Gln360) was amplified and subcloned into pMT-BiP-V5HisB (Invitrogen). S2 Schneider cells were cotransfected with this construct and with pCoHygro. Transfected S2 cells were selected with Hygromycin and were pooled to select stable cell lines. Cell lines were expanded and grown in serum-free insect cell media supplemented with 1 mM CuSO4 to induce recombinant Nrg3 expression. rNrg3 was purified and concentrated by ultrafiltration, dialysis, and chromatography using Ni2 beads.
Affigel blue agarose beads (Bio-Rad) were washed in PBS and incubated with either BSA (1 mg/mL), Nrg3 (1 mg/mL), EGF (1 mg/mL), or Heregulin-1B (37 μg/mL) solution for 2 h at room temperature and then overnight at 4°C in siliconized microfuge tubes. Mouse EGF was obtained from Roche and HRG-1B was obtained from Upstate. Explant cultures were prepared from A/J or C57BL/6, or BAT-gal embryos (Maretto et al. 2003) harvested between E10.5 and E11.5. Beads soaked in protein (EGF, NRG3, HRG-1B, or BSA) were implanted in the flank of the embryo along the line where the mammary buds should form. Organotypic culture (in F12/DMEM 1:1 media supplemented with streptomycin, penicillin, glutamine, and 0.4% BSA [ICN, True Cohn]) of the embryos proceeded in Falcon 353037 organ culture dishes for 24 h in a 5% CO2 chamber. The embryos were fixed in 4% paraformaldehyde and processed for whole-mount in situ hybridization and probed with Lef1 anti-sense and sense probes. No specific signal was observed surrounding BSA-, EGF-, or Heregulin-1B-soaked beads when hybridized with either Lef1 anti-sense or sense probes. BAT-gal and (B6 × BAT-gal) explanted embryos were fixed and stained with X-gal (5-bromo-4-chloro-3-indolyl-D-galactoside) (Hogan et al. 1994). Sections from BAT-gal embryos were counterstained with nuclear fast red.
Acknowledgments
We thank Trevor Dale for BAT-gal transgenic mice; Dawn Steele, Olivia Sopp, and Hakan Goker for assistance with histology; Sally Swift for establishing cell lines expressing rNrg3; staff of the ICR BSU for animal care; and anonymous referees for their helpful comments. B.H. thanks Barry Gusterson for his support and encouragement at the early stages of this work. This work was supported by Breakthrough Breast Cancer whose supporters we would especially like to thank.
Supplemental material is available at http://www.genesdev.org.
Article and publication are at http://www.genesdev.org/cgi/doi/10.1101/gad.338505.
References
- Andl T., Reddy, S.T., Gaddapara, T., and Millar, S.E. 2002. WNT signals are required for the initiation of hair follicle development. Dev. Cell 2: 643-653. [DOI] [PubMed] [Google Scholar]
- Anton E.S., Ghashghaei, H.T., Weber, J.L., McCann, C., Fischer, T.M., Cheung, I.D., Gassmann, M., Messing, A., Klein, R., Schwab, M.H., et al. 2004. Receptor tyrosine kinase ErbB4 modulates neuroblast migration and placement in the adult forebrain. Nat. Neurosci. 7: 1319-1328. [DOI] [PubMed] [Google Scholar]
- Balinsky B. 1949-1950. On the developmental processes in mammary glands and other epidermal structures. Transactions of the Royal Society Edinburgh 62: 1-31. [Google Scholar]
- Bamshad M., Lin, R.C., Law, D.J., Watkins, W.C., Krakowiak, P.A., Moore, M.E., Franceschini, P., Lala, R., Holmes, L.B., Gebuhr, T.C., et al. 1997. Mutations in human TBX3 alter limb, apocrine and genital development in ulnar-mammary syndrome. Nat. Genet. 16: 311-315. [DOI] [PubMed] [Google Scholar]
- Bell A.G. 1898. On the development by selection of supernumerary mammae in sheep. Science 9: 6337-6339. [DOI] [PubMed] [Google Scholar]
- Bieche I., Onody, P., Tozlu, S., Driouch, K., Vidaud, M., and Lidereau, R. 2003. Prognostic value of ERBB family mRNA expression in breast carcinomas. Int. J. Cancer 106: 758-765. [DOI] [PubMed] [Google Scholar]
- Buerger H., Packeisen, J., Boecker, A., Tidow, N., Kersting, C., Bielawski, K., Isola, J., Yatabe, Y., Nakachi, K., Boecker, W., et al. 2004. Allelic length of a CA dinucleotide repeat in the egfr gene correlates with the frequency of amplifications of this sequence—First results of an inter-ethnic breast cancer study. J. Pathol. 203: 545-550. [DOI] [PubMed] [Google Scholar]
- Burden S. and Yarden, Y. 1997. Neuregulins and their receptors: A versatile signaling module in organogenesis and oncogenesis. Neuron 18: 847-855. [DOI] [PubMed] [Google Scholar]
- Chiba-Falek O., Kerem, E., Shoshani, T., Aviram, M., Augarten, A., Bentur, L., Tal, A., Tullis, E., Rahat, A., and Kerem, B. 1998. The molecular basis of disease variability among cystic fibrosis patients carrying the 3849 + 10 kb C → T mutation. Genomics 53: 276-283. [DOI] [PubMed] [Google Scholar]
- Chu E.Y., Hens, J., Andl, T., Kairo, A., Yamaguchi, T.P., Brisken, C., Glick, A., Wysolmerski, J.J., and Millar, S.E. 2004. Canonical WNT signaling promotes mammary placode development and is essential for initiation of mammary gland morphogenesis. Development 131: 4819-4829. [DOI] [PubMed] [Google Scholar]
- Cunha G.R. and Hom, Y.K. 1996. Role of mesenchymal-epithelial interactions in mammary gland development. J. Mammary Gland Biol. Neoplasia 1: 21-35. [DOI] [PubMed] [Google Scholar]
- Cunha G.R., Young, P., Christov, K., Guzman, R., Nandi, S., Talamantes, F., and Thordarson, G. 1995. Mammary phenotypic expression induced in epidermal cells by embryonic mammary mesenchyme. Acta Anat. (Basel) 152: 195-204. [DOI] [PubMed] [Google Scholar]
- Davenport T.G., Jerome-Majewska, L.A., and Papaioannou, V.E. 2003. Mammary gland, limb and yolk sac defects in mice lacking Tbx3, the gene mutated in human ulnar mammary syndrome. Development 130: 2263-2273. [DOI] [PubMed] [Google Scholar]
- Dunn M., Sinha, P., Campbell, R., Blackburn, E., Levinson, N., Rampaul, R., Bates, T., Humphreys, S., and Gullick, W.J. 2004. Co-expression of neuregulins 1, 2, 3 and 4 in human breast cancer. J. Pathol. 203: 672-680. [DOI] [PubMed] [Google Scholar]
- Eblaghie M.C., Song, S.J., Kim, J.Y., Akita, K., Tickle, C., and Jung, H.S. 2004. Interactions between FGF and Wnt signals and Tbx3 gene expression in mammary gland initiation in mouse embryos. J. Anat. 205: 1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falls D.L. 2003. Neuregulins: Functions, forms, and signaling strategies. Exp. Cell Res. 284: 14-30. [DOI] [PubMed] [Google Scholar]
- Gebhardt F., Zanker, K.S., and Brandt, B. 1999. Modulation of epidermal growth factor receptor gene transcription by a polymorphic dinucleotide repeat in intron 1. J. Biol. Chem. 274: 13176-13180. [DOI] [PubMed] [Google Scholar]
- Gizatullin R.Z., Muravenko, O.V., Al-Amin, A.N., Wang, F., Protopopov, A.I., Kashuba, V.I., Zelenin, A.V., and Zabarovsky, E.R. 2000. Human NRG3 gene Map position 10q22-q23. Chromosome Res. 8: 560. [DOI] [PubMed] [Google Scholar]
- Graus-Porta D., Beerli, R.R., Daly, J.M., and Hynes, N.E. 1997. ErbB-2, the preferred heterodimerization partner of all ErbB receptors, is a mediator of lateral signaling. EMBO J. 16: 1647-1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hijazi M.M., Young, P.E., Dougherty, M.K., Bressette, D.S., Cao, T.T., Pierce, J.H., Wong, L.M., Alimandi, M., and King, C.R. 1998. NRG-3 in human breast cancers: Activation of multiple erbB family proteins. Int. J. Oncol. 13: 1061-1067. [DOI] [PubMed] [Google Scholar]
- Hobbs S.S., Coffing, S.L., Le, A.T., Cameron, E.M., Williams, E.E., Andrew, M., Blommel, E.N., Hammer, R.P., Chang, H., and Riese II, D.J. 2002. Neuregulin isoforms exhibit distinct patterns of ErbB family receptor activation. Oncogene 21: 8442-8452. [DOI] [PubMed] [Google Scholar]
- Hogan B.L. 1999. Morphogenesis. Cell 96: 225-233. [DOI] [PubMed] [Google Scholar]
- Hogan B., Beddington, R., Constantani, F., and Lacy, E. 1994. Manipulating the mouse embryo: A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- Howard B.A. and Gusterson, B.A. 2000a. The characterization of a mouse mutant that displays abnormal mammary gland development. Mamm. Genome 11: 234-237. [DOI] [PubMed] [Google Scholar]
- ____. 2000b. Mammary gland patterning in the AXB/BXA recombinant inbred strains of mouse. Mech. Dev. 91: 305-309. [DOI] [PubMed] [Google Scholar]
- Jones F.E., Jerry, D.J., Guarino, B.C., Andrews, G.C., and Stern, D.F. 1996. Heregulin induces in vivo proliferation and differentiation of mammary epithelium into secretory lobuloalveoli. Cell Growth Differ. 7: 1031-1038. [PubMed] [Google Scholar]
- Li L., Cleary, S., Mandarano, M.A., Long, W., Birchmeier, C., and Jones, F.E. 2002. The breast proto-oncogene, HRGα regulates epithelial proliferation and lobuloalveolar development in the mouse mammary gland. Oncogene 21: 4900-4907. [DOI] [PubMed] [Google Scholar]
- Little C.C. and McDonald, H. 1965. Abnormalities of the mammae in the house mouse. J. Hered. 36: 285-288. [Google Scholar]
- Long W., Wagner, K.U., Lloyd, K.C., Binart, N., Shillingford, J.M., Hennighausen, L., and Jones, F.E. 2003. Impaired differentiation and lactational failure of Erbb4-deficient mammary glands identify ERBB4 as an obligate mediator of STAT5. Development 130: 5257-5268. [DOI] [PubMed] [Google Scholar]
- Lynch T.J., Bell, D.W., Sordella, R., Gurubhagavatula, S., Okimoto, R.A., Brannigan, B.W., Harris, P.L., Haserlat, S.M., Supko, J.G., Haluska, F.G., et al. 2004. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350: 2129-2139. [DOI] [PubMed] [Google Scholar]
- Mailleux A.A., Spencer-Dene, B., Dillon, C., Ndiaye, D., Savona-Baron, C., Itoh, N., Kato, S., Dickson, C., Thiery, J.P., and Bellusci, S. 2002. Role of FGF10/FGFR2b signaling during mammary gland development in the mouse embryo. Development 129: 53-60. [DOI] [PubMed] [Google Scholar]
- Maretto S., Cordenonsi, M., Dupont, S., Braghetta, P., Broccoli, V., Hassan, A.B., Volpin, D., Bressan, G.M., and Piccolo, S. 2003. Mapping Wnt/β-catenin signaling during mouse development and in colorectal tumors. Proc. Natl. Acad. Sci. 100: 3299-3304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marshall J.D., Mu, J.-L., Cheah, Y.-C., Nesbitt, M.N., Frankel, W.N., and Paigen, B. 1992. The AXB and BXA set of recombinant inbred mouse strains. Mamm. Genome 3: 669-680. [DOI] [PubMed] [Google Scholar]
- Paez J.G., Janne, P.A., Lee, J.C., Tracy, S., Greulich, H., Gabriel, S., Herman, P., Kaye, F.J., Lindeman, N., Boggon, T.J., et al. 2004. EGFR mutations in lung cancer: Correlation with clinical response to gefitinib therapy. Science 304: 1497-1500. [DOI] [PubMed] [Google Scholar]
- Propper A.Y. 1978. Wandering epithelial cells in the rabbit embryo milk line. A preliminary scanning electron microscope study. Dev. Biol. 67: 225-231. [DOI] [PubMed] [Google Scholar]
- Propper A. and Gomot, L. 1967. Tissue interactions during organogenesis of the mammary gland in the rabbit embryo. C. R. Acad. Sci. Hebd. Seances Acad. Sci. D 264: 2573-2575. [PubMed] [Google Scholar]
- Riese II D.J., van Raaij, T.M., Plowman, G.D., Andrews, G.C., and Stern, D.F. 1995. The cellular response to neuregulins is governed by complex interactions of the erbB receptor family. Mol. Cell. Biol. 15: 5770-5776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salomon D.S., Brandt, R., Ciardiello, F., and Normanno, N. 1995. Epidermal growth factor-related peptides and their receptors in human malignancies. Crit. Rev. Oncol. Hematol. 19: 183-232. [DOI] [PubMed] [Google Scholar]
- Sambrook J., Fritsch, E.F., and Maniatis, T. 1989. Molecular cloning: A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- Schmidt H. 1998. Supernumerary nipples: Prevalence, size, sex and side predilection—A prospective clinical study. Eur. J. Pediatr. 157: 821-823. [DOI] [PubMed] [Google Scholar]
- Stern D.F. 2003. ErbBs in mammary development. Exp. Cell Res. 284: 89-98. [DOI] [PubMed] [Google Scholar]
- Suzuki N., Hirata, M., and Kondo, S. 2003. Traveling stripes on the skin of a mutant mouse. Proc. Natl. Acad. Sci. 100: 9680-9685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tidcombe H., Jackson-Fisher, A., Mathers, K., Stern, D.F., Gassmann, M., and Golding, J.P. 2003. Neural and mammary gland defects in ErbB4 knockout mice genetically rescued from embryonic lethality. Proc. Natl. Acad. Sci. 100: 8281-8286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Troyer K.L. and Lee, D.C. 2001. Regulation of mouse mammary gland development and tumorigenesis by the ERBB signaling network. J. Mammary Gland Biol. Neoplasia 6: 7-21. [DOI] [PubMed] [Google Scholar]
- van Genderen C., Okamura, R.M., Farinas, I., Quo, T.G., Parslow, T.G., Bruhn, L., and Grosschedl, R. 1994. Development of several organs that require inductive epithelial-mesenchymal interactions is impaired in LEF-1-deficient mice. Genes & Dev. 8: 2691-2703. [DOI] [PubMed] [Google Scholar]
- Veltmaat J.M., Mailleux, A.A., Thiery, J.P., and Bellusci, S. 2003. Mouse embryonic mammogenesis as a model for the molecular regulation of pattern formation. Differentiation 71: 1-17. [DOI] [PubMed] [Google Scholar]
- Veltmaat J.M., Van Veelen, W., Thiery, J.P., and Bellusci, S. 2004. Identification of the mammary line in mouse by Wnt10b expression. Dev. Dyn. 229: 349-356. [DOI] [PubMed] [Google Scholar]
- Waterston R.H., Lindblad-Toh, K., Birney, E., Rogers, J., Abril, J.F., Agarwal, P., Agarwala, R., Ainscough, R., Alexandersson, M., An, P., et al. 2002. Initial sequencing and comparative analysis of the mouse genome. Nature 420: 520-562. [DOI] [PubMed] [Google Scholar]
- Wilkinson D.G. and Nieto, M.A. 1993. Detection of messenger RNA by in situ hybridization to tissue sections and whole mounts. Methods Enzymol. 225: 361-373. [DOI] [PubMed] [Google Scholar]
- Witmer P.D., Doheny, K.F., Adams, M.K., Boehm, C.D., Dizon, J.S., Goldstein, J.L., Templeton, T.M., Wheaton, A.M., Dong, P.N., Pugh, E.W., et al. 2003. The development of a highly informative mouse Simple Sequence Length Polymorphism (SSLP) marker set and construction of a mouse family tree using parsimony analysis. Genome Res. 13: 485-491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y., Spitzer, E., Meyer, D., Sachs, M., Niemann, C., Hartmann, G., Weidner, K., Birchmeier, C., and Birchmeier, W. 1995. Sequential requirement of hepatocyte growth factor and neuregulin in the morphogenesis and differentiation of the mammary gland. J. Cell Biol. 131: 215-226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D., Sliwkowski, M.X., Mark, M., Frantz, G., Akita, R., Sun, Y., Hillan, K., Crowley, C., Brush, J., and Godowski, P.J. 1997. Neuregulin-3 (NRG3): A novel neural tissue-enriched protein that binds and activates ErbB4. Proc. Natl. Acad. Sci. 94: 9562-9567. [DOI] [PMC free article] [PubMed] [Google Scholar]