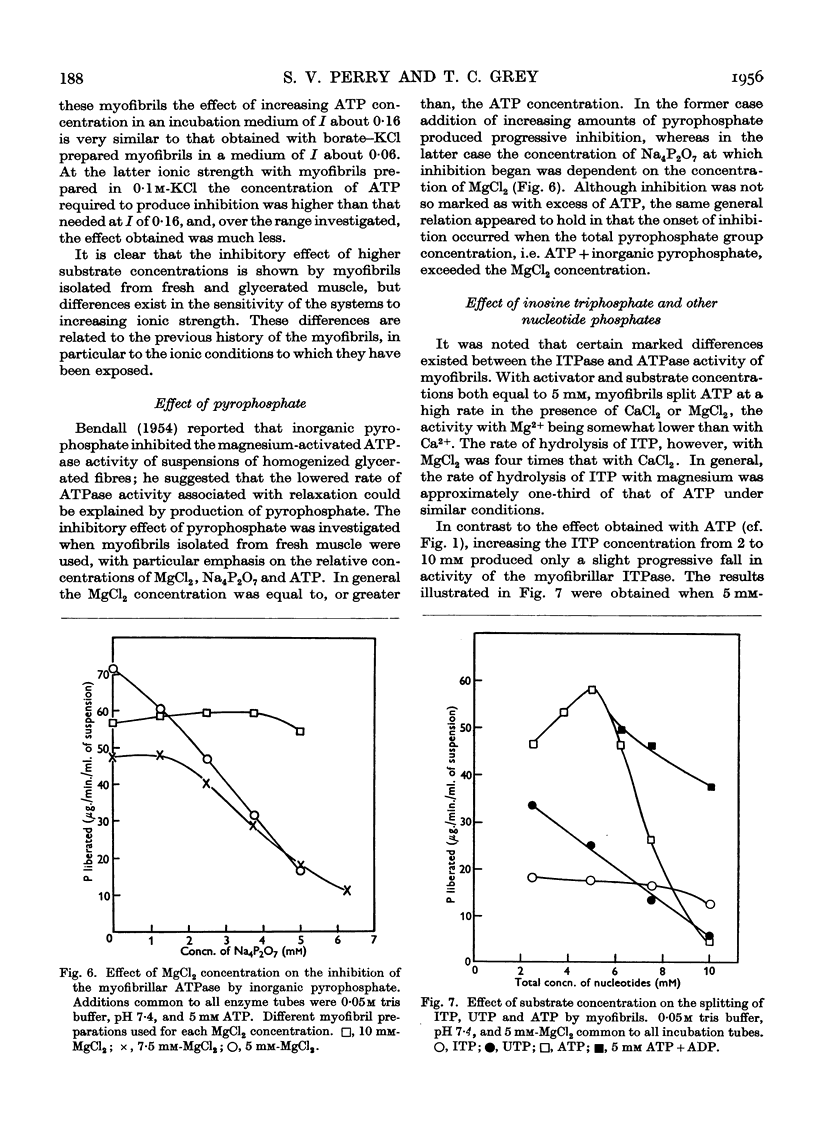
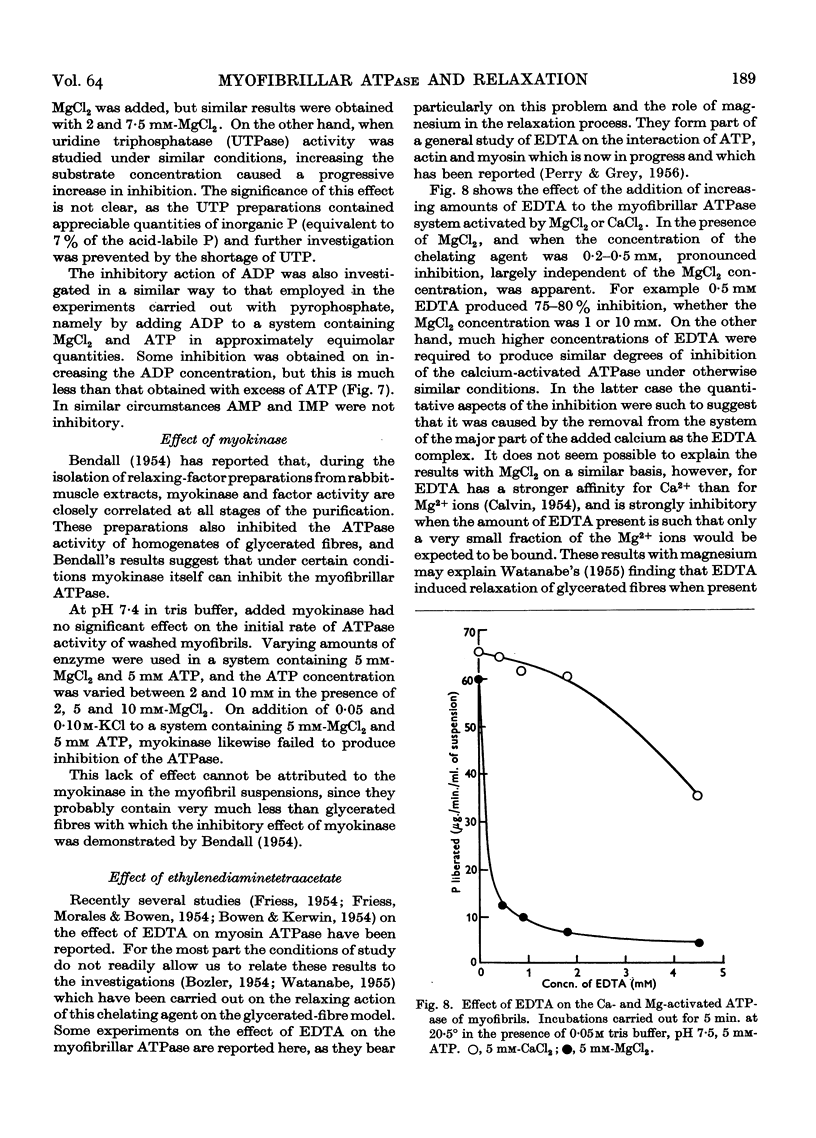
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BENDALL J. R. Effect of pyrophosphate on the shortening of muscle-fibre models in presence of adenosine triphosphate. Nature. 1953 Sep 26;172(4378):586–587. doi: 10.1038/172586a0. [DOI] [PubMed] [Google Scholar]
- BENDALL J. R. Further observations on a factor (The `Marsh' factor) effecting relaxation of ATP-shortened muscle-fibre models and the effect of Ca and Mg ions upon it. J Physiol. 1953 Aug;121(2):232–254. doi: 10.1113/jphysiol.1953.sp004944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BENDALL J. R. The relaxing effect of myokinase on muscle fibres; its identity with the Marsh factor. Proc R Soc Lond B Biol Sci. 1954 May 27;142(908):409–426. doi: 10.1098/rspb.1954.0033. [DOI] [PubMed] [Google Scholar]
- BLOCH-FRANKENTHAL L. The role of magnesium in the hydrolysis of sodium pyrophosphate by inorganic pyrophosphatase. Biochem J. 1954 May;57(1):87–92. doi: 10.1042/bj0570087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BOWEN W. J., KERWIN T. D. A study of the effects of ethylenediamine-tetra-acetic acid on myosin adenosinetriphosphatase. J Biol Chem. 1954 Nov;211(1):237–247. [PubMed] [Google Scholar]
- BOZLER E. Mechanism of relaxation in extracted muscle fibers. Am J Physiol. 1951 Oct;167(1):276–283. doi: 10.1152/ajplegacy.1951.167.1.276. [DOI] [PubMed] [Google Scholar]
- BOZLER E., PRINCE J. T. The control of energy release in extracted muscle fibers. J Gen Physiol. 1953 Sep;37(1):53–61. doi: 10.1085/jgp.37.1.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bailey K., Webb E. C. Purification and properties of yeast pyrophosphatase. Biochem J. 1944;38(5):394–398. doi: 10.1042/bj0380394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHAPPELL J. B., PERRY S. V. The stimulation of the adenosinetriphosphatase activities of myofibrils and L-myosin by 2:4-dinitrophenol. Biochim Biophys Acta. 1955 Feb;16(2):285–287. doi: 10.1016/0006-3002(55)90217-1. [DOI] [PubMed] [Google Scholar]
- FRIESS E. G., MORALES M. F., BOWEN W. J. Some further observations on the interaction of EDTA with the myosin-ATP system. Arch Biochem Biophys. 1954 Nov;53(1):311–313. doi: 10.1016/0003-9861(54)90256-8. [DOI] [PubMed] [Google Scholar]
- FRIESS E. T. The effect of a chelating agent on myosin ATPase. Arch Biochem Biophys. 1954 Jul;51(1):17–23. doi: 10.1016/0003-9861(54)90448-8. [DOI] [PubMed] [Google Scholar]
- GILMOUR D., CALABY J. H. Myokinase and pyrophosphatase of insect muscle. Enzymologia. 1953 Apr 15;16(1):34–40. [PubMed] [Google Scholar]
- HASSELBACH W., WEBER H. H. Der Einfluss des MB-Faktors auf die Kontraktion des Fasermodells. Biochim Biophys Acta. 1953 May;11(1):160–161. doi: 10.1016/0006-3002(53)90021-3. [DOI] [PubMed] [Google Scholar]
- HEPPEL L. A., HILMOE R. J. Purification of yeast inorganic pyrophosphatase. J Biol Chem. 1951 Sep;192(1):87–94. [PubMed] [Google Scholar]
- HERS H. G. Rôle du magnésium et du potassium dans la réaction fructokinasique. Biochim Biophys Acta. 1952 Apr;8(4):424–430. doi: 10.1016/0006-3002(52)90068-1. [DOI] [PubMed] [Google Scholar]
- KREBS H. A., HEMS R. Some reactions of adenosine and inosine phosphates in animal tissues. Biochim Biophys Acta. 1953 Sep-Oct;12(1-2):172–180. doi: 10.1016/0006-3002(53)90136-x. [DOI] [PubMed] [Google Scholar]
- KUMAGAI H., EBASHI S., TAKEDA F. Essential relaxing factor in muscle other than myokinase and creatine phosphokinase. Nature. 1955 Jul 23;176(4473):166–166. doi: 10.1038/176166a0. [DOI] [PubMed] [Google Scholar]
- MARSH B. B. The effects of adenosine triphosphate on the fibre volume of a muscle homogenate. Biochim Biophys Acta. 1952 Sep;9(3):247–260. doi: 10.1016/0006-3002(52)90159-5. [DOI] [PubMed] [Google Scholar]
- McELROY W. D., COULOMBRE J., HAYS R. Properties of firefly pyrophosphatase. Arch Biochem Biophys. 1951 Jun;32(1):207–215. doi: 10.1016/0003-9861(51)90255-x. [DOI] [PubMed] [Google Scholar]
- PERRY S. V. Relation between chemical and contractile function and structure of the skeletal muscle cell. Physiol Rev. 1956 Jan;36(1):1–76. doi: 10.1152/physrev.1956.36.1.1. [DOI] [PubMed] [Google Scholar]
- PERRY S. V. The adenosinetriphosphatase activity of myofibrils isolated from skeletal muscle. Biochem J. 1951 Mar;48(3):257–265. doi: 10.1042/bj0480257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PERRY S. V. The bound nucleotide of the isolated myofibril. Biochem J. 1952 Jul;51(4):495–499. doi: 10.1042/bj0510495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ROBBINS E. A., STULBERG M. P., BOYER P. D. The magnesium activation of pyrophosphatase. Arch Biochem Biophys. 1955 Jan;54(1):215–222. doi: 10.1016/0003-9861(55)90024-2. [DOI] [PubMed] [Google Scholar]


