Abstract
Introduction
Acidosis often occurs during clinical complications in newborns and can lead to changes in the mechanisms of arterial tone regulation. However, it is unknown how acidosis affects the activity of Ca2+-activated chloride channels (CaCC) in arteries during early ontogenesis. We hypothesized that their activity may increase during acidosis.
Methods
We studied isometric contractions of saphenous arteries isolated from adult and 10-13-day-old rats. Intracellular pH was measured using a fluorescent indicator BCECF-AM simultaneously with recording the contractile activity of the arterial preparation in isometric mode.
Results
Metabolic acidosis with pH = 6.8 caused a significant decrease in the arterial contractile responses of adult and 10-13-day-old rats. The functional contribution of CaCC was absent in the adult rat arteries both at pH = 7.4 and pH = 6.8. However, in 10-13-day-old rat pups, the functional contribution of CaCC was higher at pH = 6.8 compared to pH = 7.4.
Conclusion
Acidosis augments the functional role of CaCC in arteries during early postnatal ontogenesis, but not in adulthood.
Keywords: Acidosis, Chloride channels, Early postnatal ontogenesis, Intracellular pH, TMEM16A
Highlights
-
•
Acidosis reduces contraction and pHi in saphenous arteries of adult and young rats.
-
•
Acidosis doesn't change TMEM16A channels' role in arterial contraction in adults.
-
•
Acidosis increases procontractile role of arterial TMEM16A channels in young rats.
1. Introduction
The activity of many proteins, including ion channels, is pH dependent. This in turn can have a significant impact on the regulation of vascular tone (Aalkjaer and Peng, 1997; Crimi et al., 2012). Note that even a slight decrease in arterial blood pH below 7.38 (with a normal pH level of 7.4) is described as a state of acidosis (Berend et al., 2014). Acidosis in most vascular regions causes arterial relaxation by influencing various mechanisms at both the smooth muscle and endothelial cell levels (Gaynullina et al., 2022; Boedtkjer and Aalkjaer, 2012).
Previously, we and other authors demonstrated that a decrease in arterial tone under the influence of acidosis is characteristic not only of an adult organism, but also of early postnatal period (Gaynullina et al., 2022; Nakanishi et al., 1997a; Shvetsova et al., 2024). Of note, under normal pH in early ontogenesis many mechanisms regulating vascular tone are different from those in adulthood. In particular, we have previously shown that in the early postnatal period the activity of Ca2+-activated chloride channels (CaCC), which demonstrate the procontractile influence on vascular smooth muscle tone, is higher compared to adults (Kostyunina et al., 2019, 2020). This is consistent with the larger suppressive effect of Cl− substitution in the solution on the arterial contraction in 1-2-week-old compared to 2-3-month-old rats (Kostyunina et al., 2020). Although there is no clear data on the molecular structure of CaCC channels, the TMEM16A protein is considered important for their chloride conductance (Dam et al., 2014a; Thomas-Gatewood et al., 2011; Heinze et al., 2014). Under normal pH conditions, during agonist-induced contraction of arterial smooth muscle the intracellular Ca2+ concentration increases, leading to the activation of CaCC to promote Cl− efflux, thereby causing additional depolarization and additional entry of Ca2+ into the smooth muscle cell (Dam et al., 2014a, 2014b). Thus, under normal pH conditions CaCC play procontractile role. Importantly, our data demonstrated increased levels of TMEM16A mRNA and protein in the arteries of rats in early postnatal ontogenesis compared to adults (Kostyunina et al., 2020). However, it remains unclear how a shift in acid-base balance towards low pH values (i.e. acidosis) will affect the activity of CaCC in the arterial smooth muscle of systemic circulation in early ontogenesis. Interestingly, it was previously demonstrated in the coronary arteries of adult rats, which have high levels of TMEM16A compared with other vascular regions, that extracellular acidosis leads to an increase in the procontractile influence of CaCC (Guo et al., 2021). Note that such an increase in the activity of CaCC in the coronary arteries leads to the development of a contractile response during acidosis (Guo et al., 2021), which is generally not typical for most vascular regions of the systemic circulation (Gaynullina et al., 2022).
Based on the above, we hypothesized that in the systemic arteries of animals during early postnatal ontogenesis, when high levels of expression and activity of CaCC are observed, their functional contribution to the regulation of vascular tone in response to acidosis may also increase. To test this hypothesis, we studied the effect of the TMEM16A blocker MONNA (Page et al., 2019) on the reactivity of the arteries of adult rats and 10-13-day-old rat pups at normal pH = 7.4 and under conditions of acidosis at pH = 6.8.
2. Materials and methods
2.1. Animals
The experiments were evaluated and approved by the Biomedical Ethics Committee of M.V. Lomonosov Moscow State University (Protocol № 97-g-2), and conformed to the European Convention on the protection of animals used for scientific purposes (EU Directive, 2010/63/EU).
In the present study we used Wistar rats received from the vivarium of the Federal State Budgetary Institution Scientific Research Institute of General Pathology and Pathophysiology. All animals had unlimited access to food and water. To get rats during the period of early postnatal ontogenesis, rats of both sexes were bred and offspring was obtained from them. The day after birth, litter size was limited to eight pups per litter.
Experiments were carried out on male rats aged 3–4 months (“adults”), as well as on male rat pups aged 10–13 days. Altogether, 13 adult animals and 14 10-13-day-old rat pups were used in the experiments. The animals were decapitated (anesthesia with CO2 was used for adult rats) and the saphenous artery was isolated in a preparation solution (in mM): NaCl – 145; KCl – 4.5; CaCl2 – 0.1; MgSO4 – 1.0; NaH2PO4 – 1.2; EDTA – 0.025; HEPES – 5.0; pH = 7.4.
2.2. Isometric myography
The study of vascular contractility in isometric mode was carried out using a multichannel myograph (model 620M, DMT). Immediately after isolation, 2 mm long rings were cut out from the artery and mounted on steel wires in a myograph filled with a preparation solution. Mechanical removal of the endothelium was performed using a rat whisker. The signal was recorded at 10 Hz in the PowerGraph 3.3 program (DISoft, Russia) with ADC E14-140 (L-CARD, Russia). The solution in myograph channels was heated to 37C, after which the preparations were normalized to the stretching level at which the vessel demonstrates optimal contractile activity (Mulvany and Halpern, 1977; Shvetsova et al., 2019). During this procedure the internal vascular diameter corresponding to a pressure of 100 mm Hg was determined (d100). Then the solution in the myograph was changed to a working solution (in mM): NaCl – 120; NaHCO3 – 26; KCl – 4.5; CaCl2 – 1.6; MgSO4 – 1.0; NaH2PO4 – 1.2; D-glucose – 5.5; EDTA – 0.025; HEPES – 5, pH = 7.4. The solution was bubbled with carbogen (5 % CO2 + 95 % O2) throughout the experiment.
All arterial preparations were activated by adding (1) norepinephrine (10 μM, 5 min); (2) α1-adrenoceptor agonist methoxamine (10 μM, 5 min) followed by acetylcholine (10 μM, 2 min) to check the removal of endothelium; (3) methoxamine (10 μM, 5 min). 20 min after the end of the activation procedure, the main experimental protocol was carried out (Fig. 1), consisting of two concentration-response relationships to methoxamine (10 nM–100 μM, duration of action of each concentration 2 min). After the end of the first concentration-response relationship to methoxamine and washing, metabolic acidosis was modeled in the experiments. For this purpose, the solution in the myograph chamber was replaced either with a working solution for myograph with pH = 7.4, or with a working solution for myograph with pH = 6.8 (solution composition (in mM): NaCl – 140.5; NaHCO3 – 5.5; KCl – 4.5; CaCl2 – 1.6; MgSO4 – 1.0; NaH2PO4 – 1.2; D-glucose – 5.5; EDTA – 0.025; HEPES – 5.0). After 10 min, the TMEM16A blocker MONNA (3 μM) was added to part of the myograph channels, and the same volume of DMSO (the solvent of MONNA) was added to the remaining part. 20 min later, a second concentration-response relationship to methoxamine was carried out, identical to the first one. The second concentration-response relationships to methoxamine are shown in figures.
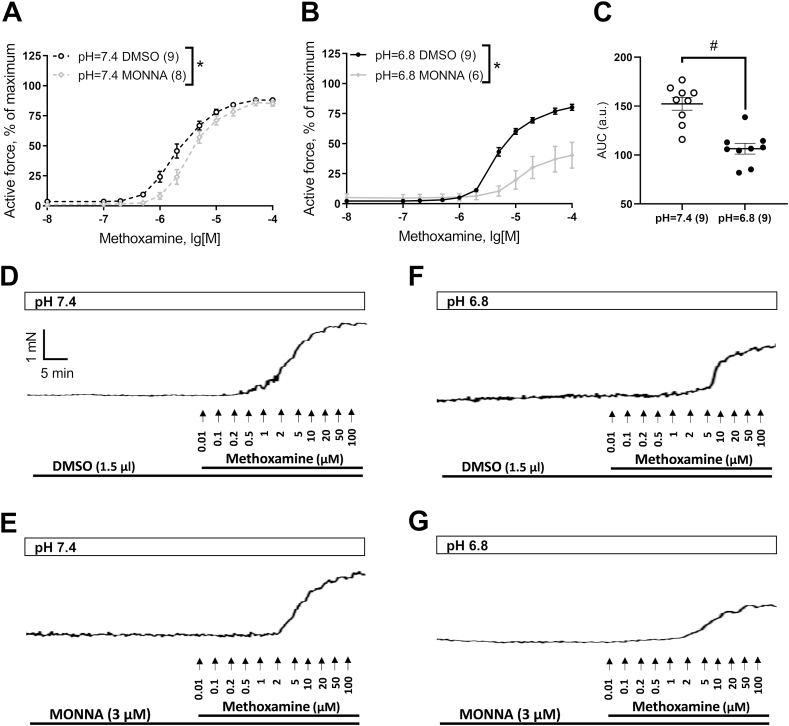
Fig. 1.
MONNA does not change the contractile responses of adult rat arteries either at pH = 7.4 or at pH = 6.8. (A, B) Concentration-response relationships to methoxamine of adult rat arteries in the presence of Solvent (DMSO) or TMEM16A blocker MONNA (3 μM) at pH = 7.4 (A) or at pH = 6.8 (B). (C) The areas under the concentration-response curves to methoxamine in the presence of a solvent at pH = 7.4 or at pH = 6.8 for adult rat arteries. (D–G) Original trace recordings of isometric force measurements in saphenous arteries of adult rats in the presence of solvent (DMSO, D, F) or TMEM16A blocker MONNA (3 μM, E, G) at pH = 7.4 (D, E) or at pH = 6.8 (F, G). The numbers in brackets indicate the number of animals in the group. #p < 0.05 (unpaired Student's t-test).
Processing of the experimental results consisted of determining the “passive” force corresponding to complete arterial dilation, after which it was deducted from the force values obtained in the presence of agonist. Then the obtained values were divided to the maximum force developed during the first concentration-response relationship to methoxamine and presented in %. To determine the effect of acidic pH on the contractile activity of arteries, the areas under the individual concentration-response curves to methoxamine at pH = 7.4 and pH = 6.8 were calculated (in the GraphPad Prism 9.5.1 program (La Jolla, CA, USA)), after which these values were compared. To compare the effect of the blocker, the areas under the curves to agonist were evaluated (in GraphPad Prism 9.5.1), after which the corresponding values for the blocker were divided to the average area in the presence of DMSO and presented in %. In addition, to estimate the sensitivity of arterial preparations to methoxamine, individual concentration–response relationships were fitted to a sigmoidal dose–response (variable slope) equation using GraphPad Prism 9.5.1 to obtain pD2 (the negative logarithm of EC50).
2.3. Measurement of intracellular pH
Intracellular pH (pHi) was measured using a fluorescent indicator BCECF-AM (Invitrogen) (Rochon et al., 2007) simultaneously with recording the contractile activity of the arterial preparation in isometric mode. 2 mm long ring arterial preparations were placed in a myograph (model 360CW, DMT), standard procedures for normalization and activation were performed (see “Isometric myography” section), after which the preparations were loaded with 5 μM BCECF-AM at 37C for 30 min. The indicator was excited alternately with light with wavelengths of 435 and 500 nm using a CoolLED pE800-fura illuminator. The emission signal from the arterial segment was recorded at a frequency of 0.1 Hz using a 535 nm filter in a Nikon Ti2-U microscope and a Photometrics BSI Express camera, this signal at various waves of excitation light was recorded on a computer and processed using NIS-Elements software. For each excitation light wavelength, the baseline fluorescence emission was subtracted (prior to BCECF-AM loading), and then the ratio was calculated for the signals at excitation light wavelengths of 500 and 435 nm (F500/F435).
After loading BCECF-AM, the preparation was washed with a solution with pH = 7.4, which was then replaced with a solution with pH = 6.8 (see section “Isometric myography”) for 10 min.
In a number of separate experiments, calibration was carried out (both for the arteries of adults and for the arteries of 10-13-day-old rats). To do this, after washing from BCECF-AM, the ionophore Nigericine (10 μM) and calibration solutions with pH = 7.50, pH = 7.00, pH = 6.50 were added to the preparations. The composition of the calibration solution was (in mM): NaCl – 56; KCl – 94.5; CaCl2 – 1.6; MgSO4 – 1.0; NaH2PO4 – 1.2; D-glucose – 5.5; EDTA – 0.025; HEPES – 20.0. For each type of artery, the dependence of the fluorescence signal on the pH of the calibration solution was plotted, after which the obtained values were approximated by a linear equation, which was used to convert measurement data obtained by incubating arterial preparations in solutions with extracellular pH = 7.4 and pH = 6.8.
2.4. Statistical data analysis
Statistical data analysis was performed in the GraphPad Prism 9.5.1 software. The type of data distribution was evaluated using the Shapiro-Wilk test. Two-way ANOVA for repeated measures (or Student's t-test were used to compare samples. Statistical significance was reached at p < 0.05. Data are shown as mean ± SEM, n represents the number of rats.
3. Results
3.1. Effect of acidosis on contractile responses to methoxamine and intracellular pH
Internal vascular diameter corresponding to a pressure of 100 mm Hg (d100) was 553 ± 12 μm for arteries of adult animals and 234 ± 3 μm for arteries of 10-13-day-old animals. The maximum force developed by arterial preparations was 32.7 ± 1.9 mN for adult animals and 4.9 ± 0.4 mN for 10-13-day-old animals. The arteries of adult rats and 10-13-day-old rat pups contracted in response to α1-adrenoceptor agonist methoxamine at both normal pH = 7.4 (Fig. 1A (black curves), Fig. 1, Fig. 2A (black curves), Fig. 2D) and at acidic pH = 6.8 (Fig. 1B (black curves), Fig. 1, Fig. 2B (black curves), Fig. 2F). At the same time, the arterial contractile responses in both groups of rats at pH = 6.8 were significantly less than at pH = 7.4. This is confirmed by comparing the areas under the concentration-response curves to methoxamine in the presence of a solvent for adult (Fig. 1C) as well as for 10-13-day-old (Fig. 2C) rats. In addition, acidosis led to a considerable shift of pD2 values for adult animals (5.89 ± 0.03 at pH = 7.4 (n = 7) and 5.55 ± 0.06 at pH = 6.8 (n = 7, p < 0.05)) as well as for 10-13-day-old animals (5.68 ± 0.07 at pH = 7.4 (n = 9) and 5.31 ± 0.03 at pH = 6.8 (n = 9, p < 0.05)).
Fig. 2.
MONNA suppresses contractile responses of arteries of 10-13-day-old rats at pH = 6.8 stronger than at pH = 7.4. (A, B) Concentration-response relationships to methoxamine of 10-13-day-old rat arteries in the presence of Solvent (DMSO) or TMEM16A blocker MONNA (3 μM) at pH = 7.4 (A) or at pH = 6.8 (B). (C) The areas under the concentration-response curves to methoxamine in the presence of a solvent at pH = 7.4 or at pH = 6.8 for 10-13-day-old rat arteries. (D–G) Original trace recordings of isometric force measurements in saphenous arteries of 13-day-old rat pup in the presence of solvent (DMSO, D, F) or TMEM16A blocker MONNA (3 μM, E, G) at pH = 7.4 (D, E) or at pH = 6.8 (F, G). The numbers in brackets indicate the number of animals in the group. ∗p < 0.05 (two-way ANOVA for repeated measures), #p < 0.05 (unpaired Student's t-test).
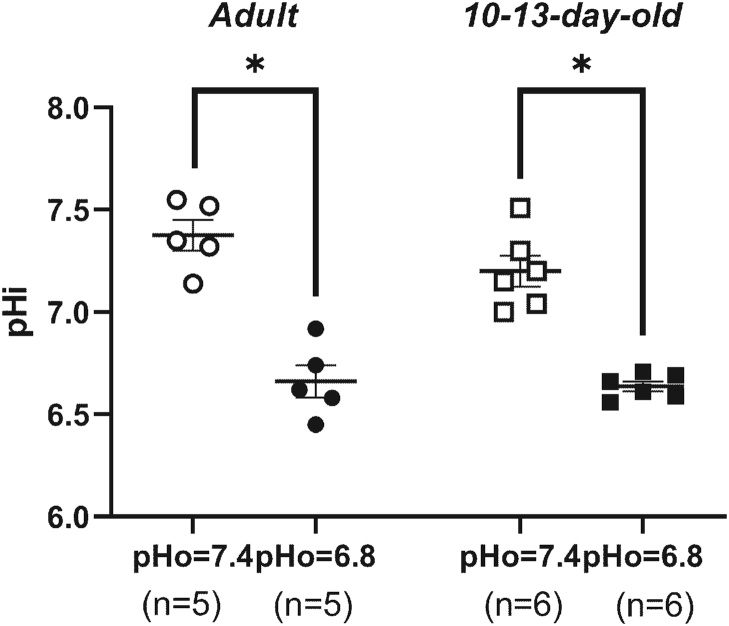
Measurement of intracellular pH showed that extracellular acidosis leads to a significant lowering of pHi. At a solution with pH = 7.4 the pHi values did not differ between adult and 10-13-day-old rats (Fig. 3). Changing the solution to a solution with pH = 6.8 led to considerable intracellular acidification in both age groups to values that did not differ between adult and 10-13-day-old animals as well (Fig. 3).
Fig. 3.
Extracellular acidosis leads to significant intracellular acidification in arteries of adult and 10-13-day-old rats. Measurements of intracellular pH in arteries of adult and 10-13-day-old rats at normal pH (pHо = 7.4) and under acidic conditions (pHо = 6.8). The numbers in brackets indicate the number of animals in the group. ∗p < 0.05 (paired Student's t-test).
3.2. Effect of acidosis on the contribution of TMEM16A channels to contractile responses of arteries of adult rats
At the next stage, the effect of the TMEM16A channel blocker MONNA on contractile responses to methoxamine in adult rats at normal pH and under conditions of acidosis was assessed. Incubation of arteries of adult rats with MONNA (3 μM) did not lead to changes in contractile responses to methoxamine either at pH = 7.4 (Fig. 1A,D, E) or at pH = 6.8 (Fig. 1B,F, G). The pD2 values of arterial contractile responses to methoxamine of adult animals were not affected by MONNA both at pH = 7.4 (5.89 ± 0.03 in Solvent group (n = 7) and 5.85 ± 0.07 in MONNA group (n = 6, p > 0.05)) and at pH = 6.8 (5.55 ± 0.06 in Solvent group (n = 7) and 5.48 ± 0.07 in MONNA group (n = 6, p > 0.05)). Therefore, the contribution of TMEM16A channels to the regulation of contractile responses in adult rat arteries is not altered by acidosis.
3.3. Effect of acidosis on the contribution of TMEM16A channels to contractile responses of arteries of 10–13-day-old rats
Next, we studied the effect of MONNA on the arterial contraction in the early postnatal period at normal pH and under conditions of acidosis. Incubation of arteries of 10-13-day-old rat pups with MONNA (3 μM) led to a decrease in contractile responses to methoxamine both at pH = 7.4 (Fig. 2A,D, E) and at pH = 6.8 (Fig. 2B,F, G). The pD2 values of arterial contractile responses to methoxamine of 10-13-day-old rats were not affected by MONNA at pH = 7.4 (5.68 ± 0.07 in Solvent group (n = 9) and 5.54 ± 0.04 in MONNA group (n = 8, p > 0.05)); similar values for pH = 6.8 were not calculated due to the impossibility of correct approximation of some of the individual curves.
To compare the effects of MONNA at pH = 7.4 and at pH = 6.8, the values of the areas under the curves were calculated and the values of area in the presence of MONNA were expressed as a percentage of the average area in the presence of the solvent. This parameter was 82 ± 5 % at pH = 7.4 (n = 8) and 49 ± 12 % at pH = 6.8 (n = 6, p < 0.05, unpaired Student's t-test). Therefore, during extracellular acidosis the procontractile contribution of TMEM16A channels increases significantly in the arteries of 10-13-day-old rat pups.
4. Discussion
Our data demonstrate that extracellular metabolic acidosis leads to intracellular acidification in both the arteries of adult and 10-13-day-old rats, as well as to a decrease in the arterial contractile responses. In addition, we showed that under conditions of acidosis, the functional contribution of CaCC in the arteries of 10-13-day-old, but not adult rats increases.
Our data on lowering of pHi in response to acidification of extracellular solution in arteries are consistent with literature data. Previously, similar data for adult sexually matured animals were obtained for mesenteric and renal arteries (Niu et al., 2014), aorta (Nakanishi et al., 1997a; Rohra et al., 2003a, 2003b), coronary arteries (Baxter et al., 2006). However, for the arteries of animals in the early postnatal period, data on the effect of extracellular metabolic acidosis on pHi have not been previously published. At the same time, it has been demonstrated that respiratory acidosis (aeration of the solution with a 20 % CO2 gas mixture) causes pHi lowering in the aorta and mesenteric arteries of newborn rabbits (Nakanishi et al., 1997a, 1997b). Note that, despite the fact that the average pHi values in the arteries of 10-13-day-old rats tended to be lower than in adult animals, we did not find statistically significant differences. At the same time, earlier comparisons in pHi between the arteries of adult animals and animals during the period of early ontogenesis were carried out only in a few studies, where no differences in pHi levels were found between different age groups (Nakanishi et al., 1997a, 1997b; Kostyunina et al., 2020).
Acidification led to a significant decrease in arterial contractile responses to methoxamine. The decrease in the arterial contractile responses of adult rats under conditions of acidosis is in good agreement with data previously obtained for other arteries of the systemic circulation (Yartsev et al., 2002; Mohanty et al., 2016; Aoyama et al., 1999; Akanji et al., 2019; Hessellund et al., 2006; Rohra et al., 2005), as well as with our own data for the saphenous artery (Shvetsova et al., 2024). Note that a decrease in contractile reactions under conditions of acidosis in adult animals at the level of smooth muscle cells can be achieved by various mechanisms (Boedtkjer and Aalkjaer, 2012), which, according to our data, are not associated with CaCC.
There are very few studies examining the influence of extracellular acidosis on the vascular tone in early ontogenesis. As in this study, we previously showed a decrease in contractile reactions of the saphenous artery with denuded endothelium under conditions of metabolic acidosis in rat pups aged 12–15 days (Shvetsova et al., 2024). These data are also supported by data on the inhibitory effect of respiratory acidosis (created by aeration of the solution with a gas mixture with 20 % CO2) on the contractile activity of the aorta of newborn rabbits (Nakanishi et al., 1997a, 1997b).
However, the question of the contribution of CaCC to the regulation of arterial smooth muscle contraction under conditions of acidosis in the early postnatal period has not been studied before. We showed for the first time that acidosis, despite the suppression of contractile responses, leads to an increase in the procontractile influence of TMEM16A in the regulation of contraction in early ontogenesis. An increase in CaCC activity due to acidosis has previously been demonstrated in the coronary arteries of adult animals, where TMEM16A was shown to have high expression level compared to other vascular regions (Guo et al., 2021). Interestingly, in the saphenous artery of rat pups during early postnatal ontogenesis, the level of TMEM16A expression is also higher compared to adults (Kostyunina et al., 2020). Moreover, the higher content of TMEM16A in the arteries of animals in the early stages of postnatal ontogenesis is consistent with greater TMEM16A functional contribution to vascular contraction during this period compared to adults, as observed in the present, as well as in our previous studies (Kostyunina et al., 2020). Importantly, relatively high functional contribution of TMEM16A to arterial contractile responses in early period of development is observed at normal pH, when the contraction to agonists is associated with only moderate increase in intracellular Ca2+ concentration (Puzdrova et al., 2014; Akopov et al., 1997, 1998a; Sandoval et al., 2007). Moreover, the high functional contribution of TMEM16A in arteries of animals in the early postnatal ontogenesis persisted under acidic conditions which are known to suppress the intracellular Ca2+ concentration in adult vasculature (Aoyama et al., 1999; Boedtkjer, 2018; Austin and Wray, 1995).
Thus, our data show that during acidosis, the procontractile activity of TMEM16A in arterial smooth muscle cells in early postnatal ontogenesis increases, despite a general decrease in contraction. Apparently, this is due to changes in the functional contribution of other mechanisms regulating vascular tone under the influence of acidosis in early ontogenesis. For example, we previously demonstrated a decrease in the anticontractile effect of TASK-1 channels in the regulation of saphenous artery tone in early ontogenesis in response to acidosis (Shvetsova et al., 2024). We hypothesize that, in contrast to the increased contribution of procontractile mechanisms during acidosis, associated with an increase in the activity of CaCC and a decrease in the activity of TASK-1 channels, a weakening of other procontractile mechanisms may occur in the smooth muscle cells of systemic arteries in early ontogenesis. An example of such a mechanism is Rho-kinase, which plays a pronounced role in the regulation of contraction in the early postnatal period (Mochalov et al., 2018; Akopov et al., 1998b), while its activity significantly decreases under conditions of acidosis (Boedtkjer et al., 2011). Studies of the arterial tone regulation in early ontogenesis under conditions of acidosis are important from the clinical point of view, since they allow us to understand the mechanisms of vascular complications due to pH lowering, which quite often occurs in infants, for example, during neonatal asphyxia (Remzső et al., 2020; Rainaldi and Perlman, 2016).
Several study limitations should be acknowledged. First, only male rats were used in the study, therefore the sex-dependent analysis of TMEM16A contribution to arterial contraction was not performed. Second, the present study does not address other vascular regions of systemic circulation. Third, the contribution of TMEM16A was tested using single blocker. Finally, the functional role of TMEM16A in endothelial cells was not evaluated. All mentioned nuances should be examined in future studies.
5. Conclusion
In conclusion, our novel findings demonstrate a pronounced procontractile role of Ca2+-activated chloride channels in peripheral artery of young rats under conditions of acidosis. It is known, that acidosis leads to a decrease in arterial contractility, which at systemic level may cause a decrease of systemic blood pressure (Celotto et al., 2016). Of note, the level of systemic blood pressure during early postnatal ontogenesis is significantly lower than in adult (Mochalov et al., 2018; Sofronova et al., 2016). We suggest that an increase of procontractile influence of Ca2+-activated chloride channels in peripheral arteries of early postnatal animals under conditions of acidosis may serve as a protective mechanism that prevents lowering already fairly low blood pressure and thereby helps maintain sufficient blood supply to immature organs and tissues. The data presented in this study complement the complex mosaic of changes in the mechanisms of arterial tone regulation in systemic vasculature under conditions of acidosis and indicate the need for further research into this problem, including the development of adequate approaches to correcting the state of acidosis in infants.
CRediT authorship contribution statement
Anastasia A. Shvetsova: Conceptualization, Investigation, Writing – original draft, Data curation. Margarita A. Khlystova: Investigation. Valentina S. Shateeva: Investigation. Sofia D. Simonenko: Investigation. Anna A. Borzykh: Investigation. Denis V. Abramochkin: Writing – original draft, Data curation, all authors approved the final version of the manuscript. Dina K. Gaynullina: Conceptualization, Investigation, Data curation, Writing – original draft.
Funding
The study was supported by Russian Science Foundation (Project № 21-75-10036).
Declaration of competing interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Dina K Gaynullina reports financial support was provided by Russian Science Foundation. Reports a relationship with that includes:. Has patent pending to. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
The part of equipment used in the study was provided by MSU within the framework of federal project “The development of infrastructure for science and education” (Agreement №161).
Data availability
Data will be made available on request.
References
- Aalkjaer C., Peng H.L. pH and smooth muscle. Acta Physiol. Scand. 1997;161:557–566. doi: 10.1046/j.1365-201X.1997.00263.x. [DOI] [PubMed] [Google Scholar]
- Akanji O., Weinzierl N., Schubert R., Schilling L. Acid sensing ion channels in rat cerebral arteries: probing the expression pattern and vasomotor activity. Life Sci. 2019;227:193–200. doi: 10.1016/j.lfs.2019.04.054. [DOI] [PubMed] [Google Scholar]
- Akopov S.E., Zhang L., Pearce W.J. Physiological variations in ovine cerebrovascular calcium sensitivity. Am. J. Physiol. Hear. Circ. Physiol. 1997;41:H2271–H2281. doi: 10.1152/ajpheart.1997.272.5.H2271. [DOI] [PubMed] [Google Scholar]
- Akopov S.E., Zhang L., Pearce W.J. Developmental changes in the calcium sensitivity of rabbit cranial arteries. Biol. Neonate. 1998;74:60–71. doi: 10.1159/000014011. [DOI] [PubMed] [Google Scholar]
- Akopov S.E., Zhang L., Pearce W.J. Regulation of Ca2+ sensitization by PKC and rho proteins in ovine cerebral arteries: effects of artery size and age. Am. J. Physiol. - Hear. Circ. Physiol. 1998;275 doi: 10.1152/ajpheart.1998.275.3.h930. [DOI] [PubMed] [Google Scholar]
- Aoyama Y., Ueda K., Setogawa A., Kawai Y. Effects of pH on contraction and Ca2+ mobilization in vascular smooth muscles of the rabbit basilar artery. Jpn. J. Physiol. 1999;49:55–62. doi: 10.2170/jjphysiol.49.55. [DOI] [PubMed] [Google Scholar]
- Austin C., Wray S. The effects of extracellular pH and calcium change on force and intracellular calcium in rat vascular smooth muscle. J. Physiol. 1995;488:281–291. doi: 10.1113/jphysiol.1995.sp020966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baxter K.A., Laher I., Church J., Hsiang Y.N. Acidosis augments myogenic constriction in rat coronary arteries. Ann. Vasc. Surg. 2006;20:630–637. doi: 10.1007/S10016-006-9109-9. [DOI] [PubMed] [Google Scholar]
- Berend K., de Vries A.P.J., Gans R.O.B. Physiological approach to assessment of acid–base disturbances. N. Engl. J. Med. 2014;371:1434–1445. doi: 10.1056/NEJMRA1003327/SUPPL_FILE/NEJMRA1003327_DISCLOSURES.PDF. [DOI] [PubMed] [Google Scholar]
- Boedtkjer E. Acid–base regulation and sensing: accelerators and brakes in metabolic regulation of cerebrovascular tone. J. Cereb. Blood Flow Metab. 2018;38:588–602. doi: 10.1177/0271678X17733868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boedtkjer E., Aalkjaer C. Intracellular pH in the resistance vasculature: regulation and functional implications. J. Vasc. Res. 2012;49:479–496. doi: 10.1159/000341235. [DOI] [PubMed] [Google Scholar]
- Boedtkjer E., Praetorius J., Matchkov V.V., Stankevicius E., Mogensen S., Füchtbauer A.C., Simonsen U., Füchtbauer E.M., Aalkjaer C. Disruption of NA +,HCO 3- cotransporter NBCn1 (slc4a7) Inhibits no-mediated vasorelaxation, smooth muscle ca 2+ Sensitivity, and hypertension development in mice. Circulation. 2011;124:1819–1829. doi: 10.1161/CIRCULATIONAHA.110.015974. [DOI] [PubMed] [Google Scholar]
- Celotto A.C., Ferreira L.G., Capellini V.K., Albuquerque A.A.S., Rodrigues A.J., Evora P.R.B. Acute but not chronic metabolic acidosis potentiates the acetylcholine-induced reduction in blood pressure: an endothelium-dependent effect. Brazilian J. Med. Biol. Res. 2016;49:1–8. doi: 10.1590/1414-431X20155007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crimi E., Silvio F., Infante T., Scolletta S., Crudele V., Napoli C. Effects of intracellular acidosis on endothelial function : an overview. J. Crit. Care. 2012;27:108–118. doi: 10.1016/j.jcrc.2011.06.001. [DOI] [PubMed] [Google Scholar]
- Dam V.S., Boedtkjer D.M.B., Aalkjaer C., Dam V.S., Boedtkjer D.M.B., Aalkjaer C., Matchkov V. The bestrophin- and TMEM16A-associated Ca(2+)- activated Cl(–) channels in vascular smooth muscles. Channels. 2014;8:361–369. doi: 10.4161/chan.29531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dam V.S., Boedtkjer D.M.B., Nyvad J., Aalkjaer C., Matchkov V. TMEM16A knockdown abrogates two different Ca2+-activated Cl - currents and contractility of smooth muscle in rat mesenteric small arteries. Pflugers Arch. Eur. J. Physiol. 2014;466:1391–1409. doi: 10.1007/s00424-013-1382-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaynullina D.K., Tarasova O.S., Shvetsova A.A., Borzykh A.A., Schubert R. The effects of acidosis on eNOS in the systemic vasculature: a focus on early postnatal ontogenesis. Int. J. Mol. Sci. 2022;23:5987. doi: 10.3390/ijms23115987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo P., Liu Y., Xu X., Ma G., Hou X., Fan Y., Zhang M. Coronary hypercontractility to acidosis owes to the greater activity of TMEM16A/ANO1 in the arterial smooth muscle cells. Biomed. Pharmacother. 2021;139 doi: 10.1016/J.BIOPHA.2021.111615. [DOI] [PubMed] [Google Scholar]
- Heinze C., Seniuk A., Sokolov M.V., Huebner A.K., Klementowicz A.E., Szijártó I.A., Schleifenbaum J., Vitzthum H., Gollasch M., Ehmke H., Schroeder B.C., Hübner C.A. Disruption of vascular Ca2+-activated chloride currents lowers blood pressure. J. Clin. Investig. 2014;124:675–686. doi: 10.1172/JCI70025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hessellund A., Aalkjaer C., Bek T. Effect of acidosis on isolated porcine retinal vessels. Curr. Eye Res. 2006;31:427–434. doi: 10.1080/02713680600681236. [DOI] [PubMed] [Google Scholar]
- Kostyunina D.S., Gaynullina D.K., Matchkov V.V., Tarasova O.S. Pro-contractile role of chloride in arterial smooth muscle: postnatal decline potentially governed by sympathetic nerves. Exp. Physiol. 2019;104:1018–1022. doi: 10.1113/EP087426. [DOI] [PubMed] [Google Scholar]
- Kostyunina D.S., Zhang L., Shvetsova A.A., Selivanova E.K., Tarasova O.S., Matchkov V.V., Gaynullina D.K. Trophic sympathetic influence weakens pro-contractile role of Cl− channels in rat arteries during postnatal maturation. Sci. Rep. 2020;10:1–13. doi: 10.1038/s41598-020-77092-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mochalov S.V., Tarasova N.V., Kudryashova T.V., Gaynullina D.K., Kalenchuk V.U., Borovik A.S., Vorotnikov A.V., Tarasova O.S., Schubert R. Higher Ca2+-sensitivity of arterial contraction in 1-week-old rats is due to a greater Rho-kinase activity. Acta Physiol. 2018;223:1–15. doi: 10.1111/apha.13044. [DOI] [PubMed] [Google Scholar]
- Mohanty I., Suklabaidya S., Parija S.C. Acidosis reduces the function and expression of α1D-adrenoceptor in superior mesenteric artery of capra hircus. Int. J. Pharmacol. 2016;48:399–406. doi: 10.4103/0253-7613.186199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulvany M.J., Halpern W. Contractile properties of small arterial resistance vessels in spontaneously hypertensive and normotensive rats. Circ. Res. 1977;41:19–26. doi: 10.1161/01.RES.41.1.19. [DOI] [PubMed] [Google Scholar]
- Nakanishi T., Gu H., Momma K. Effect of acidosis on contraction, intracellular pH, and calcium in the newborn and adult rabbit aorta. Heart Vessels. 1997;12:207–215. doi: 10.1007/BF02766785. [DOI] [PubMed] [Google Scholar]
- Nakanishi T., Gu H., Momma K. Developmental changes in the effect of acidosis on contraction, intracellular pH, and calcium in the rabbit mesenteric small artery. Pediatr. Res. 1997;42:750–757. doi: 10.1203/00006450-199712000-00006. [DOI] [PubMed] [Google Scholar]
- Niu L., Liu Y., Hou X., Cui L., Li J., Zhang X., Zhang M. Extracellular acidosis contracts coronary but neither renal nor mesenteric artery via modulation of H + ,K + -ATPase, voltage-gated K + channels and L-type Ca 2+ channels. Exp. Physiol. 2014;99:995–1006. doi: 10.1113/expphysiol.2014.078634. [DOI] [PubMed] [Google Scholar]
- Page H.R.A., Jepps T.A., Dalsgaard T., Baldwin S.N., Povstyan O., Olesen S.P., Greenwood I.A. TMEM16A is implicated in the regulation of coronary flow and is altered in hypertension. Br. J. Pharmacol. 2019;176:1635–1648. doi: 10.1111/bph.14598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puzdrova V.A., Kudryashova T.V., Gaynullina D.K., Mochalov S.V., Aalkjaer C., Nilsson H., Vorotnikov A.V., Schubert R., Tarasova O.S. Trophic action of sympathetic nerves reduces arterial smooth muscle Ca2+ sensitivity during early post-natal development in rats. Acta Physiol. 2014;212:128–141. doi: 10.1111/apha.12331. [DOI] [PubMed] [Google Scholar]
- Rainaldi M.A., Perlman J.M. Pathophysiology of birth asphyxia. Clin. Perinatol. 2016;43:409–422. doi: 10.1016/j.clp.2016.04.002. [DOI] [PubMed] [Google Scholar]
- Remzső G., Németh J., Varga V., Kovács V., Tóth-Szűki V., Kaila K., Voipio J., Domoki F. Brain interstitial pH changes in the subacute phase of hypoxic-ischemic encephalopathy in newborn pigs. PLoS One. 2020;15 doi: 10.1371/journal.pone.0233851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rochon P., Jourdain M., Mangalaboyi J., Fourrier F., Soulie-Begu S., Buys B., Dehlin G., Lesage J.C., Chambrin M.C., Mordon S. Evaluation of BCECF fluorescence ratio imaging to properly measure gastric intramucosal pH variations in vivo. J. Biomed. Opt. 2007;12 doi: 10.1117/1.2821698. [DOI] [PubMed] [Google Scholar]
- Rohra D.K., Saito S., Ohizumi Y. Strain-specific effects of acidic pH on contractile state of aortas from Wistar and Wistar Kyoto rats. Eur. J. Pharmacol. 2003;476:123–130. doi: 10.1016/S0014-2999(03)02129-0. [DOI] [PubMed] [Google Scholar]
- Rohra D.K., Saito S., Ohizumi Y. Extracellular acidosis results in higher intracellular acidosis and greater contraction in spontaneously hypertensive rat aorta. Eur. J. Pharmacol. 2003;465:141–144. doi: 10.1016/S0014-2999(03)01490-0. [DOI] [PubMed] [Google Scholar]
- Rohra D.K., Sharif H.M., Zubairi H.S., Sarfraz K., Ghayur M.N., Gilani A.H. Acidosis-induced relaxation of human internal mammary artery is due to activation of ATP-sensitive potassium channels. Eur. J. Pharmacol. 2005;514:175–181. doi: 10.1016/j.ejphar.2005.02.041. [DOI] [PubMed] [Google Scholar]
- Sandoval R.J., Injeti E.R., Williams J.M., Georthoffer W.T., Pearce W.J. Myogenic contractility is more dependent on myofilament calcium sensitization in term fetal than adult ovine cerebral arteries. Am. J. Physiol. Hear. Circ. Physiol. 2007;293:H548–H556. doi: 10.1152/ajpheart.00134.2007. [DOI] [PubMed] [Google Scholar]
- Shvetsova A.A., Gaynullina D.K., Tarasova O.S., Schubert R. Negative feedback regulation of vasocontraction by potassium channels in 10- to 15-day-old rats: dominating role of Kv7 channels. Acta Physiol. 2019;225:1–18. doi: 10.1111/apha.13176. [DOI] [PubMed] [Google Scholar]
- Shvetsova А.A., Borzykh A.A., Gaynullina D.K. vol. 60. 2024. pp. 247–255. (Effect of Extracellular Acidosis on Functional Contribution of K ATP and TASK-1 Potassium Channels to Vascular Tone Regulation in Early Postnatal Ontogenesis). [DOI] [Google Scholar]
- Sofronova S.I., Borzykh A.A., Gaynullina D.K., Kuzmin I.V., Shvetsova A.A., Lukoshkova E.V., Tarasova O.S. Endothelial nitric oxide weakens arterial contractile responses and reduces blood pressure during early postnatal development in rats. Nitric Oxide. 2016;55–56:1–9. doi: 10.1016/j.niox.2016.02.005. [DOI] [PubMed] [Google Scholar]
- Thomas-Gatewood C., Neeb Z.P., Bulley S., Adebiyi A., Bannister J.P., Leo M.D., Jaggar J.H., Jh J. TMEM16A channels generate Ca2+-activated Cl− currents in cerebral artery smooth muscle cells, Am J Physiol Hear. Circ Physiol. 2011;301:H1819–H1827. doi: 10.1152/ajpheart.00404.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yartsev V.N., Karachentseva O.V., Dvoretsky D.P. Effect of pH changes on reactivity of rat mesenteric artery segments at different magnitude of stretch. Acta Physiol. Scand. 2002;174:1–7. doi: 10.1046/j.1365-201x.2002.00923.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made available on request.