Abstract
Background
Oral and maxillofacial space infections (OMSIs) are a serious emergency disease in oral and maxillofacial departments; untreated or undertreated OMSI can lead to serious complications and can be life-threatening. This study aimed to comprehensively analyse the epidemiological characteristics of OMSI, identify the associated etiological and risk factors, and develop a machine learning-based predictive model for factors influencing hospitalisation.
Methods
Medical records of 217 patients hospitalised with OMSI were retrospectively analysed. Demographic data, clinical characteristics, treatment histories, microbiological profiles, and drug sensitivity test results were reviewed. A risk prediction model for hospitalisation length was established using machine learning.
Results
Odontogenic infections (69.41%) were the most common etiological factors for OMSI, with periapical periodontitis being the most prevalent. Streptococcus spp. was the most frequently cultured aerobic bacteria, whereas Peptostreptococcus anaerobius was the predominant anaerobe. Drug sensitivity tests indicated high resistance rates to clindamycin and erythromycin among aerobic bacteria. The risk prediction model exhibited an area under the curve of 0.726 and was validated by an internal area under the curve of 0.712. Factors such as hypertension, diabetes, pre-admission interventions, and age were significantly associated with prolonged hospitalisation.
Conclusion
Periapical periodontitis remains a primary cause of OMSI; however, the rising incidence of infections due to cosmetic injections and implant surgeries warrants attention. Penicillin, clindamycin, and erythromycin are not recommended as empirical first-choice drugs. The predictive model effectively identified risk factors for extended hospitalisation. Hypertension, diabetes, pre-admission interventions, and age are risk factors for lengthened hospitalisation. Efforts should be made to promote oral hygiene education and healthcare system reforms in regions with similar demographic and socioeconomic conditions.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12903-025-05971-x.
Keywords: Maxillofacial space infection, Hospitalisation time, Risk factors, Machine learning, Microbiology, Epidemiological
Introduction
Oral and maxillofacial space infections (OMSIs) are a common emergency condition in oral and maxillofacial departments, characterised by rapid development and easy spread to adjacent spaces [1]. Although the incidence and complications of OMSI have decreased with antibiotic use, untreated or undertreated OMSI can lead to serious complications, including airway obstruction, Ludwig’s pharyngitis, and intracranial infections [2–5]. Patients with OMSI often require surgery, whereas those with severe symptoms may require tracheotomy, intensive care, or other dispositions. Consequently, OMSI poses significant challenges and burdens on maxillofacial surgeons, medical resources, and socioeconomic factors.
OMSI is primarily caused by odontogenic factors, with the causative organisms of odontogenic infections and even the resident flora of the oral cavity often serving as the source of microorganisms for these infections. Pathogenic bacteria can cause infections through minor injuries caused by patient routine activities, including chewing, routine oral maintenance, oral therapy, and so on. OMSI often requires surgical incision and drainage of infected spaces, complemented by antibiotic therapy, depending on the degree of infection, disease progression, and complications. The use of antibiotics also needs to be adjusted post-surgery based on bacterial culture results [6–9]. Despite clearly defined treatment protocols, the microbial distribution in different geographical regions, lifestyle habits, and empirical medication use by physicians have different effects on the treatment outcome [10]. Thus, analysing the relationships between demographic characteristics, microbial species, drug resistance of pathogens, aetiology of the infection, and the disease’s course and severity is of great significance for diagnosing and treating OMSI.
Long-term hospitalisation for patients with OMSI can have physical, psychological, social, and financial impacts on both clinical work and the patients themselves. Therefore, investigating the factors that influence hospital stay duration will help to reduce hospitalisation time. The application of machine learning (ML) in disease risk prediction is a major current trend [11]. By analysing large amounts of clinical and biological data, ML algorithms can help doctors to more accurately assess the risk of a patient developing a certain disease. Additionally, ML algorithms can uncover patterns and features that traditional medical methods cannot detect, providing doctors with new diagnostic tools and treatment options.
In this study, the epidemiological and microbiological characteristics of OMSIs and the association between the disease and relevant etiological factors were explored. Additionally, a risk prediction model for factors influencing the length of hospitalisation using ML methods was developed to provide a reference for the clinical diagnosis and treatment of OMSIs.
Methods
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki. This study was approved by the Ethics Committee of the Third Affiliated Hospital of Air Force Military Medical University on May 27, 2024. (approval no. KY-OT-20240039, reference no. KQ-YJ-2024-105). The requirement for informed consent was waived by the Institutional Review Board because of the retrospective nature of the study. To ensure patient privacy and data security, all patient data were anonymized and only the results of the evaluation is used for publication.
The medical records of 217 patients with OMSI hospitalised in the Department of Oral and Maxillofacial Survey, School of Stomatology of the Fourth Military Medical University between January 2021 and June 2023 were retrospectively analysed. Inclusion criteria were: (1) a complete record of medical data; (2) clinical manifestations of typical inflammation, including local redness, swelling, elevated skin temperature, spontaneous pain, and local dysfunction; and (3) fulfilment of diagnostic criteria related to OMSIs based on clinical symptoms and signs, medical history, laboratory tests (e.g. WBC > 10 × 109/L, C-reactive protein > 10 mg/L or procalcitonin > 0.5 ng/mL), and other examinations(e.g. Ultrasound, CT, or MRI confirming abscess formation or fascial space involvement, pus culture identifying aerobic/anaerobic pathogens) [1]. Exclusion criteria were: (1) head and neck tumours; (2) significantly improved clinical symptoms (e.g., reduced swelling, normalized body temperature, restored function); (3) inflammatory indicators returned to the normal range.
All patients received surgical and/or antibiotic treatment upon admission, followed by empirical antibiotic therapy. Pus samples were collected and cultured during hospitalisation. Antibiotic sensitivity test was performed using the Kirby-Bauer disk diffusion method for aerobes and the Epsilometer test (E-test) gradient method for anaerobes, in accordance with the guidelines of the Clinical and Laboratory Standards Institute (CLSI).
Wilcoxon rank-sum test was used to identify risk factors influencing the length of hospital stay. It was chosen because the data for several continuous variables were not normally distributed, as confirmed by the Shapiro-Wilk test. The Wilcoxon rank-sum test is robust to outliers and does not assume a normal distribution, making it suitable for comparing two independent groups. A visual predictive model was established using a nomogram. The goodness-of-fit was verified via receiver operating characteristic and calibration curves. Decision curve analysis evaluated the clinical utility of the predictive model. Random forest ML was used by randomly sampling data from 70% to 30% of the training and validation sets, respectively. Cross-validation and receiver operating characteristic curve analyses were conducted on the validation set to verify prediction performance.
Statistical analyses were performed using R version 4.4.40 (R Core Team, R Foundation for Statistical Computing, Vienna, Austria). Categorical data are expressed as the number of cases or percentages (%), with the chi-square test used to assess their correlation with hospital stays exceeding 10 days. Measurement data was presented as mean ± standard deviation, with the Wilcoxon rank-sum test used to assess their correlation with hospital stays. Statistical significance was set at P < 0.05.
Results
General clinical characteristics
The study included 217 patients with OMSI, including 131 men and 86 women. The age range was 4–86 years, with a mean age of 48.8 ± 19.8 years and median age of 52 years, respectively. The mean ages for men and women were 49.21 and 48.23, respectively (Fig. S1).
Over 90% of patients had received antibiotic therapy either independently or at another healthcare facility prior to admission. The mean temperature on admission was 36.77 ± 0.64 °C (range: 35.5–40.8 °C). White blood cell (WBC) counts were measured for 214 patients, with a mean WBC count of 10.41 ± 4.97 × 109/L (range: 1.58 × 109/L–28.22 × 109/L) (Table 1).
Table 1.
Clinical and laboratory characteristics of patients with OMSI at admission
| Condition at admission | Categories | n |
|---|---|---|
| WBCa count (*109/L) | ≤ 10 | 107 |
| 10–20 | 94 | |
| >20 | 16 | |
| Body temperature | ≤ 37℃ | 190 |
| 37.1–38℃ | 17 | |
| >38℃ | 10 | |
| Preadmission intervention | Untreated | 24 |
| Self-medication | 33 | |
| Treated by other medical institution | 125 | |
| Self-medication and treated by other medical institution | 35 |
aWBC, white blood cell
Admission examination and treatment
Clinical symptoms
Primary complaints and symptoms were maxillofacial swelling, trismus, pain at the swelling site, and toothaches. The complaints of most patients encompass more than 3 symptoms (Table 2).
Table 2.
Symptoms of characteristics
| Symptoms | n (%) |
|---|---|
| Swelling | 216 (99.54%) |
| Pain | 176 (81.11%) |
| Trismus | 187 (86.18%) |
| Dyspnoea | 6 (2.76%) |
| Fever | 4 (1.84%) |
| Dysphagia | 16 (7.37%) |
| Toothache | 43 (19.82%) |
| Other symptoms | 1 (0.46%) |
| Number of symptoms | |
| ≤ 2 | 39 (17.97%) |
| 3 | 145 (66.82%) |
| 4 | 29 (13.36%) |
| > 4 | 4 (1.84%) |
Pathogenesis
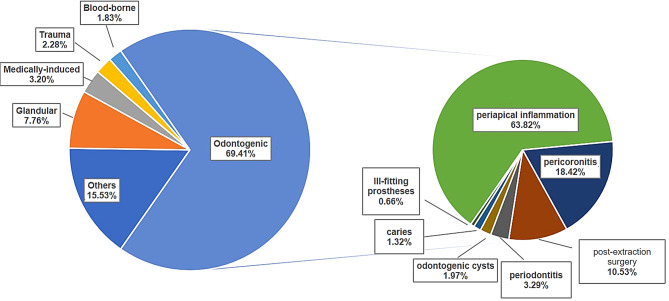
Odontogenic infections accounted for 69.41% of cases, with infections due to periapical inflammation accounting for 63.82%. Other common causes included glandular infections, medically induced infections, trauma, etc. (Fig. 1). In addition to postoperative infections, medical infections include those caused by plastic injections and dental implants.
Fig. 1.
Pathogenesis characteristics of oral and maxillofacial space infection
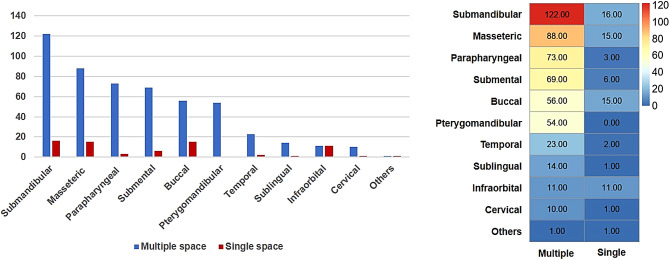
Multiple- and single-space infections were observed in 151 and 66 cases, respectively. The submandibular space was the most commonly involved space. In multi-space infection cases, the most frequently involved spaces were the submandibular, masseteric, and parapharyngeal spaces. In single-space infection cases, the most common sites were the submandibular, buccal, and masseteric spaces (Fig. 2).
Fig. 2.
Distribution of spaces in multiple- and single-space infections
Treatment during hospitalisation
Only 1 patient did not receive antibiotic treatment, 24 used a single type of antibiotic, 161 received 2 antibiotics, 29 used 3 antibiotic drugs, and 3 received 4 types of antimicrobial drugs (Fig. S2). A total of 215 patients underwent surgical treatments, including incision and drainage, puncture extraction, and tooth extraction. Most surgeries were performed under general anaesthesia, whereas puncture pumping and tooth extraction were performed under local anaesthesia. Among the patients under general anaesthesia, 39 underwent postoperative tracheal intubation, and none underwent postoperative tracheotomy (Table S1).
Microbiological findings
Distribution of pathogens
Aerobic bacteria(s) were observed in 98 patient specimens, anaerobic bacteria(s) in 14 patient specimens, and mixed infections in 34 patient specimens. A total of 201 bacterial strains were isolated: 145 aerobic, 50 anaerobic, and 6 fungal. The time for culture results ranged from 18 to 127 h, with a mean and median time of 64.25 ± 17.55 h and 62 h, respectively.
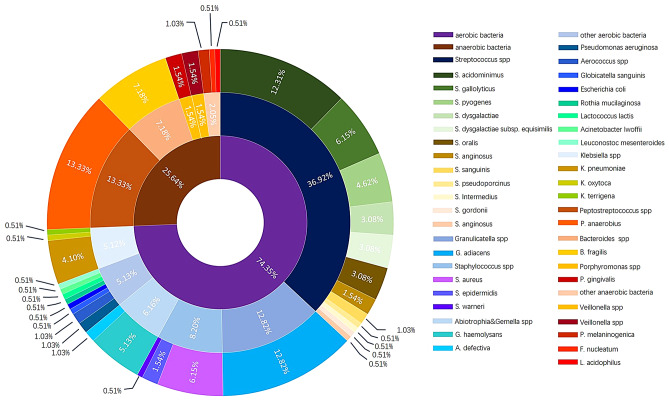
Among the aerobic bacteria, Streptococcus spp. was the most common, followed by Granulicatella spp., Staphylococcus spp., and Klebsiella spp. Peptostreptococcus anaerobius was the most common anaerobic bacterium, followed by Bacteroides fragilis and Porphyromonas gingivalis (Fig. 3).
Fig. 3.
Distribution of microorganisms in positive bacterial culture results
Drug resistance of pathogens
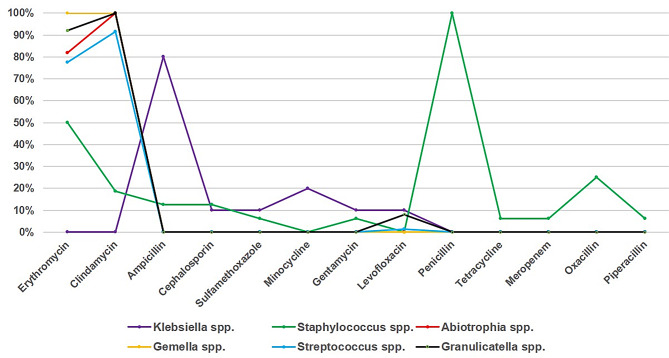
Drug sensitivity tests were conducted on the aerobic bacteria. Figure 4 shows that Streptococcus spp. had the highest resistance rate to clindamycin, a significantly lower resistance rate to levofloxacin, and 100% sensitivity to cephalosporins. Granulicatella spp. exhibited the highest resistance to clindamycin and 100% sensitivity to cephalosporins. Staphylococcus spp. was resistant to multiple antibiotics, including penicillin, erythromycin, benzathine, clindamycin, and cephalosporins. Klebsiella spp. had the highest resistance rate to ampicillin but low resistance to minocycline, cefazolin, gentamicin, and levofloxacin. Gemella spp. showed 100% resistance to both erythromycin and clindamycin, whereas Abiotrophia spp. showed 100% resistance to clindamycin and 81.82% resistance to erythromycin.
Fig. 4.
Antibiotic resistance characteristics of main aerobia
Establishment and validation of the predictive model
Screening risk factors for prolonged hospital stays
Patients were divided into 2 groups based on their hospital stay: those with a stay of ≥ 10 days and those with a stay of < 10 days. Factors influencing the length of hospital stay were examined. Results indicated significant differences in hypertension, diabetes, pre-admission interventions, and age between the 2 groups (Tables 3 and 4).
Table 3.
Results of univariate analysis
| Variable | Categories | Hospitalisation length | P-value | Chi-square | |
|---|---|---|---|---|---|
| < 10 days n (%) |
≥ 10 days n (%) |
||||
| Sex | Female | 43(40.2%) | 43(39.1%) | ||
| Male | 64(59.8%) | 67(60.9%) | 0.979 | <0.001 | |
| Hypertension | No | 97(90.7%) | 79(71.8%) | ||
| Yes | 10(9.3%) | 31(28.2%) | <0.001 | 11.359 | |
| Diabetes | No | 97(90.7%) | 80(72.7%) | ||
| Yes | 10(9.3%) | 30(27.3%) | 0.001 | 10.432 | |
| Dyspnoea/dysphagia | No | 99(92.5%) | 96(87.3%) | ||
| Yes | 8(7.5%) | 14(12.7%) | 0.291 | 1.116 | |
| Pre-admission intervention | No | 17(15.9%) | 7(6.4%) | ||
| Yes | 90(84.1%) | 103(93.6%) | 0.043 | 4.080 | |
| Tracheal intubation | No | 97(90.7%) | 99(90.0%) | ||
| Yes | 10(9.3%) | 11(10.0%) | 1 | <0.001 | |
| Aerobic | Negative | 46(43.0%) | 39(35.5%) | ||
| Positive | 61(57.0%) | 71(64.5%) | 0.318 | 0.996 | |
| Anaerobic | Negative | 82(76.6%) | 87(79.1%) | ||
| Positive | 25(23.3%) | 23(20.9%) | 0.786 | 0.074 | |
Table 4.
Results of w-k test
| Variable | Hospitalisation length | P-value | w-k | |
|---|---|---|---|---|
| < 10 days M (P25, P75) |
≥ 10 days M (P25, P75) |
|||
| Age | 45 (28, 63) | 56 (38.25, 65) | 0.033 | 4897.5 |
| Admission temperature | 36.7 (36.5, 36.9) | 36.6 (36.5, 36.9) | 0.055 | 6766 |
| WBC count | 9.24 (6.5, 12.53) | 10.21 (6.81, 13.16) | 0.501 | 5573 |
| Number of spaces involved | 2 (1, 3) | 3 (1, 4) | 0.584 | 5637.5 |
WBC, white blood cell
Constructing a risk model for prolonged hospital stays in OMSI patients
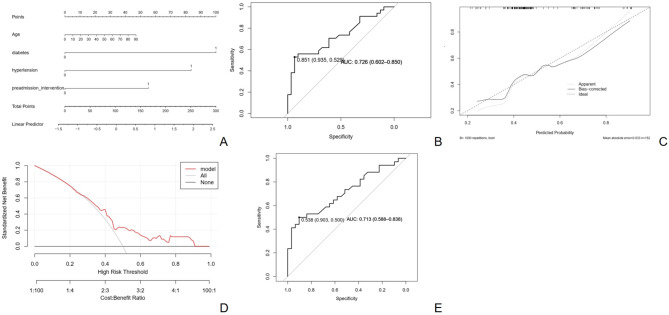
A visual nomogram was constructed (Fig. 5A). The nomogram exhibited an area under the curve (AUC) of 0.726 (Fig. 5B). The calibration plot displays a high degree of overlap between the predicted and actual curves, with an average error value of 0.033 (Fig. 5C). The model’s clinical utility was further evaluated using decision curve analysis (Fig. 5D), which suggested that the risk-prediction model was successfully constructed and preliminarily validated.
Fig. 5.
Construction of a risk model predicting the length of hospital stay. (A) Nomogram for predicting the risk of extended hospital stay. (B) Receiver operating characteristic curve derived from the nomogram, with 95% confidence intervals. (C) Calibration plot derived from the nomogram. (D) Decision curve for the prediction model based on the nomogram. (E) Clinical impact curve derived from the decision curve
Multiple validation and evaluation of the risk model
The predictive model was validated using random forest ML. A total of 217 samples were randomly selected, with 152 samples serving as the training set and 65 as the internal validation set. The predictive model exhibited an area under the curve (AUC) of 0.726 (95% CI: 0.602–0. 850) for the training set and 0.712 (95% CI: 0.588–0.838) for the validation set. Additional validation metrics, including precision, recall, and F1-score, are presented in Table S2. The model demonstrated a precision of 0.644, recall of 0.935, and F1-score of 0.763. At a threshold of 0.538, the sensitivity and accuracy were 0.903 and 0.500, respectively, indicating a predictive capability (Fig. 5E).
Discussion
Among the 217 patients with oral and maxillofacial space infections, 60% were male patients, consistent with a previous study [9]. This may be due to the differences in socioeconomic and psychological factors in different regions, which lead to male patients being more likely to be affected by causative factors, engaging less in comprehensive personal care, and having less timely access to treatment for odontogenic diseases.
Odontogenic infections were by far the most common cause of OMSI in our cohort and counts for more than 2/3 (69%) of all cases. In some previous studies, tonsillar infection was the most common source [12]. These differences may be related to factors such as race, age, and lifestyle habits of the study participants [13]. Among odontogenic factors, apical periodontitis was the most common one in our cohort, possibly explained by socioeconomic factors that prevalence of endodontic diseases in economically developed regions is lower than less developed regions [14, 15]. Notably, medical factors are increasing due to the rising number of patients undergoing cosmetic injections or dental implant surgeries. Although these treatments provide many benefits, doctors need to effectively control infections associated with these treatments to minimise complications.
In this study, the submandibular space was most commonly involved, in concordance with the primary odontogenic cause and patient-reported symptoms. The high proportion of cases with multiple-space infection likely reflects delayed admission which allowed spreading of the infection to additional spaces [16].
OMSI is primarily a mixed infection of aerobic and anaerobic bacteria [17]. The microbiological profile revealed Streptococcus spp. as the predominant aerobic pathogen and Peptostreptococcus anaerobius as the most common anaerobe, consistent with previous studies [3, 4, 18]. In this study, aerobic bacteria exhibited high resistance rates to clindamycin (72.4%) and erythromycin (68.9%), which contrasts with findings from other regions. A study in South Australia reported lower resistance rates to clindamycin (34.5%) among Streptococcus spp [19]. Similarly, in European and American studies, clindamycin had higher sensitivity [4, 20]. However, in several countries in South America and Asia where antibiotic monitoring is insufficient, the prevalence of drug-resistant bacteria is notably higher [21, 22]. These differences may be attributed to variations in antibiotic prescribing practices, patient self-medication habits, and regional bacterial ecology [19]. Therefore, the regional variations underscore the importance of tailoring empirical treatment protocols to local resistance patterns. Globally, the rise in antibiotic resistance among pathogens causing OMSIs is a growing concern. Studies showed that the resistance to penicillin, clindamycin and erythromycin was increasing in varies regions [21, 23, 24]. Moreover, in China, the resistance of pathogenic bacteria including OMSI is also increasing significantly [25, 26]. These trends align with author’s findings and underscore the need for continuous surveillance of antibiotic resistance patterns. Based on the findings, penicillin, clindamycin, and erythromycin were not recommend as first-line empirical treatments for OMSIs in northwestern China. Instead, cephalosporins and nitroimidazoles may be more effective, given their lower resistance rates. And the recommendation also supported by other Chinese scholars [27]. Furthermore, the choice of empirical antibiotics should be guided by local resistance patterns and updated regularly based on surveillance data. Rapid diagnostic tools, such as molecular biology techniques for detecting resistance genes, could further enhance the precision of antibiotic selection [28].
A longer hospital stay adds financial and psychological pressure on the patient and burdens social healthcare resources. Therefore, understanding the factors that influence the length of hospital stay in patients with OMSI is important. Hypertension, diabetes, pre-admission interventions, and age were the factors affect patients with OMSI hospitalisation length. Therefore, in the clinical management of patients with OMSI, doctors should give sufficient attention to these factors to avoid deterioration and serious complications. However, apart from the aforementioned factors, confounding variables such as socioeconomic status, nutritional status and access to medical care also exert influence on the research results. pre-admission interventions may indirectly reflect disparities in socioeconomic status and access to healthcare, thereby accounting for potential confounding factors. Future studies should aim to collect more comprehensive patient data to achieve a deeper understanding of the factors influencing hospitalisation duration.
In recent years, computer-aided techniques, particularly ML methods, has been increasingly employed for the diagnosis and prognostic evaluation of diseases [29]. The establishment of disease risk prediction models based on ML can help medical institutions and individuals achieve precision medicine and personalised health management as well as providing a decision-making basis for public health. In this study, only a few influencing factors with significance were obtained through statistical methods, and logistic regression and random forest ML can be used to predict the impact of the disease. The random forest algorithm was chosen for its ability to handle non-linear relationships and missing data, reduce the risk of overfitting, and ease of interpreting feature importance, which re often present in retrospective studies. While other methods, such as logistic regression or support vector machines, were considered, random forest was deemed the most suitable given the complexity of the clinical data and the study’s sample size [30].
The predictive model developed in this study demonstrated moderate performance, with AUC values of 0.726 and 0.712 for the training and validation sets, respectively. While these results are promising, they highlight the need for further refinement to improve predictive accuracy. Potential limitations include the relatively small sample size, limited number of variables and the retrospective nature of the study, which may have introduced biases in data collection and feature selection. Future studies should explore the inclusion of clinical examinations (e.g., oral diseases and immune status of patients), demographic information (e.g., ethnicity and religion), socio-economic conditions (e.g., family condition, living habits and income), and laboratory variables (e.g., C-reactive protein and neutrophil-to-lymphocyte ratio) to analyse in more detail the factors influencing the length of hospital stay and to improve the model’s predictive power.
Conclusion
This study provides a comprehensive analysis of OMSIs, focusing on their epidemiological and microbiological characteristics, treatment outcomes, and factors influencing hospital stay duration. The machine learning-based predictive model demonstrated moderate accuracy in identifying patients at risk of prolonged hospitalisation, with hypertension, diabetes, pre-admission interventions, and age as key predictors. These findings underscore the importance of integrating comorbidity management and antimicrobial stewardship into clinical protocols. While this study provides region-specific insights, its retrospective design and single-center data limit generalizability. Future research should focus on external validation of the predictive model in diverse populations, prospective studies integrating real-time machine learning applications, and the inclusion of additional variables such as socioeconomic status and nutritional status. Investigating the molecular mechanisms of antibiotic resistance and fostering multi-centers collaborations will further advance our understanding and management of OMSI.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
Not applicable.
Author contributions
Conceptualization: J Wang. Data curation: J Wang, S Qu. Formal analysis: Q Ma, S Qu. Funding acquisition: H Zhang, Y Liu. Investigation: X Chang. Methodology: J Wang, S Qu. Software: S Qu. Validation: Q Ma. Visualization: M Ding, L Tian. Writing - original draft: J Wang. Writing - review & editing: J Wang, S Qu, Q Ma.
Funding
The study was supported by the National Natural Science Foundation of China (Grant No. 82201089 recipient: H Zhang, and 82101050 recipient: Y Liu).
Data availability
All data that support the findings of this study are available from the corresponding authors upon reasonable request.
Declarations
Ethics approval and consent to participate
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki. This study was approved by the Ethics Committee of the Third Affiliated Hospital of Air Force Military Medical University (approval no. KY-OT-20240039). The requirement for informed consent was waived by the Institutional Review Board because of the retrospective nature of the study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Shuang Qu, Email: 18821739525@163.com.
Qin Ma, Email: qinma@fmmu.edu.cn.
References
- 1.Li YP, Shi B, Zhang JR, Liu YP, Shen GF, Guo CB, Yang C, Li ZB, Zhang ZG, Wang HM, et al. [Expert consensus on the treatment of oral and maxillofacial space infections]. Zhonghua Kou Qiang Yi Xue Za Zhi. 2021;56(2):136–44. [DOI] [PubMed] [Google Scholar]
- 2.Pham Dang N, Delbet-Dupas C, Mulliez A, Devoize L, Dallel R, Barthélémy I. Five predictors affecting the prognosis of patients with severe odontogenic infections. Int J Environ Res Public Health 2020, 17(23). [DOI] [PMC free article] [PubMed]
- 3.Qian Y, Ge Q, Zuo W, Cheng X, Xing D, Yang J, Costa Viana MG, Atsawasuwan P. Maxillofacial space infection experience and risk factors: a retrospective study of 222 cases. Ir J Med Sci. 2021;190(3):1045–53. [DOI] [PubMed] [Google Scholar]
- 4.Rasteniene R, Vitosyte M, Jankauskaite D, Aleksejuniene J. Predictors of longer hospitalization of maxillofacial infections-a 17-year retrospective study. Oral Dis. 2022;28(7):1979–86. [DOI] [PubMed] [Google Scholar]
- 5.Sheikh Z, Yu B, Heywood E, Quraishi N, Quraishi S. The assessment and management of deep neck space infections in adults: A systematic review and qualitative evidence synthesis. Clin Otolaryngol. 2023;48(4):540–62. [DOI] [PubMed] [Google Scholar]
- 6.Blankson PK, Parkins G, Boamah MO, Abdulai AE, Ahmed AM, Bondorin S, Nuamah I. Severe odontogenic infections: a 5-year review of a major referral hospital in Ghana. Pan Afr Med J. 2019;32:71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Katoumas K, Anterriotis D, Fyrgiola M, Lianou V, Triantafylou D, Dimopoulos I. Epidemiological analysis of management of severe odontogenic infections before referral to the emergency department. J Craniomaxillofac Surg. 2019;47(8):1292–9. [DOI] [PubMed] [Google Scholar]
- 8.Jevon P, Abdelrahman A, Pigadas N. Management of odontogenic infections and sepsis: an update. Br Dent J. 2020;229(6):363–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zawiślak E, Nowak R. Odontogenic Head and Neck Region Infections Requiring Hospitalization: An 18-Month Retrospective Analysis. Biomed Res Int. 2021;2021:7086763. [DOI] [PMC free article] [PubMed]
- 10.Tent PA, Juncar RI, Onisor F, Bran S, Harangus A, Juncar M. The pathogenic microbial flora and its antibiotic susceptibility pattern in odontogenic infections. Drug Metab Rev. 2019;51(3):340–55. [DOI] [PubMed] [Google Scholar]
- 11.Rajpurkar P, Chen E, Banerjee O, Topol EJ. AI in health and medicine. Nat Med. 2022;28(1):31–8. [DOI] [PubMed] [Google Scholar]
- 12.Vilén ST, Ahde H, Puolakka T, Mäkitie A, Uittamo J, Snäll J. Differences in characteristics and infection severity between odontogenic and other bacterial oro-naso-pharyngeal infections. Head Face Med. 2023;19(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kamiński B, Błochowiak K, Kołomański K, Sikora M, Karwan S, Chlubek D. Oral and maxillofacial Infections-A bacterial and clinical Cross-Section. J Clin Med 2022, 11(10). [DOI] [PMC free article] [PubMed]
- 14.Mutluay M, Mutluay AT. Caries prevalence, oral health practices/behaviours and dental anxiety levels amongst dental hygiene students: A cross-sectional study. Int J Dent Hyg. 2022;20(2):262–72. [DOI] [PubMed] [Google Scholar]
- 15.Elkhodary HM, Abdelnabi MH, Swelem AA, Sabbagh HJ, El Meligy O, Talaat IM, Abdellatif EB, Khader Y, Al-Batayneh OB, Al-Khanati NM, et al. Individual, Familial and country-level factors associated with oral hygiene practices in children: an international survey. BMC Oral Health. 2023;23(1):50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bigus S, Russmüller G, Starzengruber P, Reitter H, Sacher CL. Antibiotic resistance of the bacterial spectrum of deep space head and neck infections in oral and maxillofacial surgery - a retrospective study. Clin Oral Investig. 2023;27(8):4687–93. [DOI] [PubMed] [Google Scholar]
- 17.Böttger S, Zechel-Gran S, Schmermund D, Streckbein P, Wilbrand JF, Knitschke M, Pons-Kühnemann J, Hain T, Weigel M, Imirzalioglu C et al. Clinical relevance of the Microbiome in odontogenic abscesses. Biology (Basel) 2021, 10(9). [DOI] [PMC free article] [PubMed]
- 18.Rasteniene R, Simenaite G, Brukiene V. Maxillofacial infections in Lithuanian hospitalised children and adolescents: a 17-years retrospective study. Eur Arch Paediatr Dent. 2023;24(5):603–11. [DOI] [PubMed] [Google Scholar]
- 19.Liau I, Han J, Bayetto K, May B, Goss A, Sambrook P, Cheng A. Antibiotic resistance in severe odontogenic infections of the South Australian population: a 9-year retrospective audit. Aust Dent J. 2018;63(2):187–92. [DOI] [PubMed] [Google Scholar]
- 20.Heim N, Faron A, Wiedemeyer V, Reich R, Martini M. Microbiology and antibiotic sensitivity of head and neck space infections of odontogenic origin. Differences in inpatient and outpatient management. J Craniomaxillofac Surg. 2017;45(10):1731–5. [DOI] [PubMed] [Google Scholar]
- 21.Umeshappa H, Shetty A, Kavatagi K, Vivek GK, Vaibhav N, Mohammed I. Microbiological profile of aerobic and anaerobic bacteria and its clinical significance in antibiotic sensitivity of odontogenic space infection: A prospective study of 5 years. Natl J Maxillofac Surg. 2021;12(3):372–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gómez-Arámbula H, Hidalgo-Hurtado A, Rodríguez-Flores R, González-Amaro AM, Garrocho-Rangel A, Pozos-Guillén A. Moxifloxacin versus clindamycin/ceftriaxone in the management of odontogenic maxillofacial infectious processes: A preliminary, intrahospital, controlled clinical trial. J Clin Exp Dent. 2015;7(5):e634–639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Farmahan S, Tuopar D, Ameerally PJ. A study to investigate changes in the microbiology and antibiotic sensitivity of head and neck space infections. Surgeon. 2015;13(6):316–20. [DOI] [PubMed] [Google Scholar]
- 24.Judith MJ, Aswath N, Padmavathy K. Microbiota of dental abscess and their susceptibility to empirical antibiotic therapy. Contemp Clin Dent. 2022;13(4):369–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li Y, Yang W, Li Y, Hua K, Zhao Y, Wang T, Liu L, Liu Y, Wang Y, Liu W, et al. The increasing burden of group B Streptococcus from 2013 to 2023: a retrospective cohort study in Beijing, China. Microbiol Spectr. 2025;13(1):e0226624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gong X, Jin Y, Han X, Jiang X, Miao B, Meng S, Zhang J, Zhou H, Zheng H, Feng J, et al. Genomic characterization and resistance features of Streptococcus agalactiae isolated from non-pregnant adults in Shandong, China. J Glob Antimicrob Resist. 2024;38:146–53. [DOI] [PubMed] [Google Scholar]
- 27.Wang P, Huang Y, Long J. A Five-Year retrospective study of 746 cases with maxillofacial space infection in Western China. Infect Drug Resist. 2022;15:5099–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shi H, Li H, Zheng L, Qian W, Wang Z, Xie L, Yang Z, Zheng L, Chen C, Yang X, et al. Metagenomic next-generation sequencing for the diagnosis of oral and maxillofacial space infections. J Dent Sci. 2023;18(3):1199–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kourou K, Exarchos KP, Papaloukas C, Sakaloglou P, Exarchos T, Fotiadis DI. Applied machine learning in cancer research: A systematic review for patient diagnosis, classification and prognosis. Comput Struct Biotechnol J. 2021;19:5546–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pfob A, Lu SC, Sidey-Gibbons C. Machine learning in medicine: a practical introduction to techniques for data pre-processing, hyperparameter tuning, and model comparison. BMC Med Res Methodol. 2022;22(1):282. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data that support the findings of this study are available from the corresponding authors upon reasonable request.