Abstract
This inquiry-based lab is designed around genetic diseases with a focus on protein structure and function. To allow students to work on their own investigatory projects, 10 projects on 10 different proteins were developed. Students are grouped in sections of 20 and work in pairs on each of the projects. To begin their investigation, students are given a cDNA sequence that translates into a human protein with a single mutation. Each case results in a genetic disease that has been studied and recorded in the Online Mendelian Inheritance in Man (OMIM) database. Students use bioinformatics tools to investigate their proteins and form a hypothesis for the effect of the mutation on protein function. They are also asked to predict the impact of the mutation on human physiology and present their findings in the form of an oral report. Over five laboratory sessions, students use tools on the National Center for Biotechnology Information (NCBI) Web site (BLAST, LocusLink, OMIM, GenBank, and PubMed) as well as ExPasy, Protein Data Bank, ClustalW, the Kyoto Encyclopedia of Genes and Genomes (KEGG) database, and the structure-viewing program DeepView. Assessment results showed that students gained an understanding of the Web-based databases and tools and enjoyed the investigatory nature of the lab.
Keywords: undergraduate, bioinformatics, protein structure, genetic disease
INTRODUCTION
As computers are used more and more as tools in research laboratories, it becomes increasingly important to introduce undergraduates to bioinformatics tools and databases early in their study of biology. As stated in the National Research Council's (NRC) Bio2010 report:
Computer use is a fact of life for all modern life scientists. Exposure during the early years of their undergraduate careers will help life science students use current computer methods and learn how to exploit emerging computer technologies as they arise… Becoming fully conversant with databases such as the National Center for Biotechnology Information (NCBI) is important for all biology majors (NRC, 2002, 47).
Incorporating bioinformatics into undergraduate biology curriculum has been a focus of several innovative projects that have recently been described in the literature (Campbell, 2003; Centeno et al., 2003; Cooper, 2001; Feig and Jabri, 2002; Honts, 2003; Jungck and Donovan, 2000). In order to reach all biology majors, the curriculum project described here introduces bioinformatics tools and databases in a broad manner in a laboratory that accompanies a large introductory biology course. Students are then offered the option to use these tools more extensively in several smaller, upper-level courses.
This curriculum was designed to incorporate elements of inquiry, while still being compatible with a large course size and limited lab time. The lab described here requires five weekly sessions of 2 h each. Our goal was to provide a laboratory experience in which students approach problems, seek information, synthesize findings, and share results as do active scientists (NRC, 1999, 2000). In particular, we wanted to incorporate evidence-based hypothesis testing, use of primary literature sources, and communication of research results in oral and written reports.
To help students develop a sense of ownership toward their work, individualized projects were developed. The projects provided an opportunity for students to work semi-independently on their own Web-based research. The projects emphasize the link between gene sequence, protein sequence, and gene function in genetic disease. Each project was developed around a single protein with an amino acid substitution that has been linked to a genetic disease. Students begin by reading literature related to their project, then learn more about their protein using NCBI's database, LocusLink (Pruitt and Maglott, 2001), and ExPASy's database, SwissProt (Boeckmann et al., 2003). Students are then guided through a BLAST search to identify homologous proteins, a ClustalW alignment to align the homologous proteins and identify the mutation, and secondary structure prediction of their protein sequence using PSIPRED. Students perform a structure analysis of their protein and model the mutation using the program DeepView, available through the ExPASy Web site. As a result of this analysis, students are asked to predict the effect of the mutation on protein structure and function. Finally, students use the OMIM and KEGG databases to relate their protein's function to human physiology and disease states. A summary of the tools and databases used in the course is provided in Table 1. On the last day of lab, students report their results and defend their predictions in small peer groups. Students within a lab section of 20 are encouraged to serve as support for each other, facilitating group interactions within the framework of a large lecture course.
Table 1.
Bioinformatics tools used in the curriculum
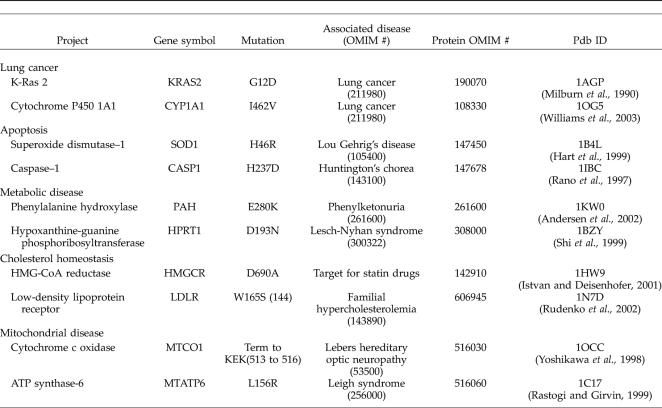
In designing the individual projects (summarized in Table 2), each protein of focus had a single amino acid substitution that had been linked to a human disease, documented in the OMIM database, and had a published crystal structure available of either the exact protein or a close homolog. Ten projects were developed that met these criteria, and we plan to add more in the future. The materials developed for this course are publicly available online (Bednarski et al., 2004), and we hope they will be useful to others in designing bioinformatics labs.
Table 2.
Projects
Context for the Course
The introductory biology core at Washington University (WU) in St. Louis is a three-semester curriculum, Principles of Biology I, II, and III. In the first semester, biological macromolecules (in particular DNA, RNA, and proteins) are introduced in the context of cell biology and microbial genetics. In the second semester, students explore eukaryotic genetics, chromosomes, population genetics, and natural selection. In the third semester, Principles of Biology III, students study protein structure/function, metabolic processes, and human physiology. Principles of Biology I and II are both taught with a weekly 3-h wet lab where students perform experiments in molecular biology and genetics.
Students are required to have taken at least one semester of General Chemistry before enrolling in Principles of Biology I. By the time students are taking Principles of Biology III, they have taken Organic Chemistry I, and they are usually taking Organic Chemistry II concurrently. This allows biochemical principles to be introduced at a fairly advanced level in Principles of Biology III, and the textbook for the biochemistry topics in the course is Berg, Tymoczko, and Stryer's Biochemistry (2002).
The bioinformatics lab described here accompanies the third-semester course, Principles of Biology III. Care has been taken to align the laboratory projects with the proteins and metabolic processes that are discussed in the lecture portion of the course. Lectures for Principles of Biology III include topics relevant to bioinformatics such as protein evolution, multiple sequence alignments, and BLAST algorithms.
LABORATORY DESIGN
Organization
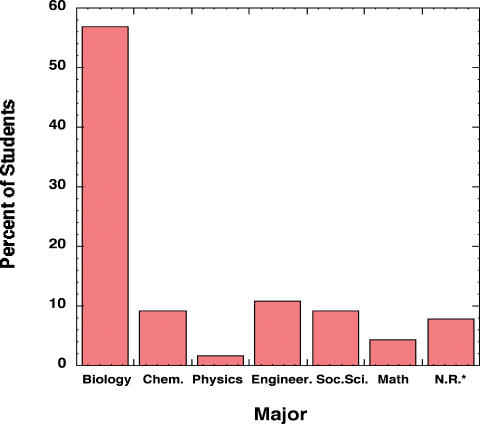
This bioinformatics lab was taught for the first time in Spring 2004 with an enrollment of 246 students. The majority of the students in the course were biology majors, but there were also a significant number of students from a wide variety of other majors, including physics and biomedical engineering (see Figure 1). The students were divided into laboratory sections of 15–20 students, and each section met weekly in a computer lab for a total of five 2-h sessions. The computer lab was equipped with 20 iMac computers (OS 10.x), a projector connected to an instructor's computer, a laser printer, and Ethernet connections for each computer. The students' desks were arranged around the wall on three sides of the classroom, so that the instructor could easily see the students' computer screens while standing in the center of the room (see Figure 2). This arrangement allowed the instructor to quickly assess the progress of the students and their focus on the individual tasks. A laboratory instructor guided each section with help from a graduate teaching assistant. In order to enhance the teaching experience for the graduate students, they had the opportunity to take the lead instructor role in 1–2 sections per week with the instructor serving as an assistant. Students were also encouraged to direct questions to students at nearby computers, since many students had the same type of questions on navigating a particular Web page. This approach helped minimize frustrations over simple Web-page navigation problems.
Figure 1. Distribution of student majors in the Bioinformatics lab Spring 2004. *No response.
Figure 2. Students working in the Bioinformatics computer lab.
The 10 projects that were developed are summarized in Table 2. Since there were approximately 20 students per section, generally two students worked on the same project in each section. These students were encouraged to sit near each other and help each other as they worked. Students often developed different hypotheses for the same project, and this collaboration gave them the opportunity to discuss and defend their ideas in preparation for their final report.
Projects
The 10 projects were grouped with two projects per disease category (as shown in Table 2). These disease categories were used in forming small groups on the last day of lab for students to present their oral reports to each other. These groupings worked well, because projects in the same disease category required similar background reading, and these readings provided some common ground for the presentations. As previously described, each project was formulated using a gene that was known to have a single missense mutation, resulting in a changed amino acid in the protein sequence linked to a disease state in the literature (OMIM database). This model of genetic disease is not generally applicable (since most genetic problems are multifactorial), but it lends itself well to focusing on the role of protein structure in gene function and in providing a format for using the Web-based tools and databases. Students were introduced to more complex models of genetic disease, such as that applicable to atherosclerosis, during the lectures in population genetics given in Principles of Biology II. A review of the structural basis of inherited disease was recently published and contains several additional examples for projects that could be developed for this laboratory (Steward et al., 2003).
Web Site and Materials
A Web site has been designed to provide a source of materials for the laboratory as well as to provide links to the Web addresses that the students commonly use in the lab. All the lab materials are available to download from the Web site including a general laboratory manual, which includes the syllabus, a glossary, and tutorials on the programs and databases common to all of the projects. The glossary is designed to include the content and terminology needed in the lab, regardless of the specific project.
The Web site also contains a separate page for each of the 10 projects where the students can download their project manual, starting DNA sequences, and any articles they are assigned to read. The project manuals contain a short introduction, reading assignments, and guide sheets specific to each project. The reading assignments include short sections from the textbook (Berg et al., 2002), a review article about the disease their protein is related to, and an excerpt from the article that described the crystal structure they will be analyzing. The guide sheets contain instructions and questions specific to each project and the database or program in use. These guide sheets are designed to be very detailed, so that students can easily use the databases and tools with minimum frustration. Although this approach does not encourage a lot of trial and error, it allows students to work at their own pace and to approach the work without anxiety.
In summary, the Web site gives students access to all the lab materials from their home computers as well as other campus computers. While lab time is usually sufficient, students can finish their work outside of class if needed.
IMPLEMENTATION
Lecture
The lecture component of the course presents the key topics needed for understanding the explorations in the laboratory. For example, the topics of protein evolution, multiple sequence alignments, and the BLOSUM-62 substitution matrix (Henikoff and Henikoff, 1992) are presented in lecture before the students work with BLAST (Altschul et al., 1997) and ClustalW in the laboratory. The students have lectures on protein structure, including an introduction to the methods of solving protein structures, before working with the crystal structure data in the laboratory. The lecture also includes discussion of protein function, basic metabolism, enzyme regulation, and cellular energetics that are important in helping the students understand the effects of loss of function of the protein they study in the laboratory, and how that could play a role in a disease. The students are also asked to memorize the amino acid side chains (chemical structure and three- and one-letter codes) during the first week of class. This greatly facilitates working with the multiple sequence alignments and the crystal structure data in the laboratory. Requiring students to be able to use this information immediately in the lab helps reinforce the need to be able to recall the chemical nature of the amino acid side chains based on the one- and three-letter codes.
In the laboratory component of the course, each session begins with a brief review of the topics from lecture that are important for the focus of that day's activities and a demonstration lasting approximately 30 min to show the basics about the Web sites and programs the students are using that day. During the rest of the laboratory session, the students work at their own speed using the guide sheets, with the instructor, the teaching assistant, and other students available for assistance.
Laboratory
In the laboratory, students use a variety of bioinformatics tools and databases. This curriculum emphasizes availability and familiarity of resources, which is consistent with our first goal. To begin each project, students download a cDNA sequence from the lab Web site for a human gene containing a single nonsynonymous mutation. Through their guide sheets, students are instructed to first translate their sequence using a tool on the Sequence Manipulation Suite. Next, students perform a BLAST search (Altschul et al., 1997) with the wild-type human protein sequence to obtain a group of diverse, yet homologous, sequences. Students are instructed to select five sequences, each from a different organism, from the BLAST results. They are asked to include sequences with a range of E-values, so that the selected sequences will be similar, but not almost identical, to their search sequence. To accomplish this, students select sequences with E-values ranging from 10–25 to 10–75. Students then create a document including the homologous sequences, the wild-type sequence, and their mutant sequence in FASTA format, which they load into ClustalW (Thompson et al., 1994) to obtain a multiple sequence alignment. Students use this alignment to identify the mutation and to observe regions of high and low conservation. Next the students use the secondary structure prediction program PSIPRED (McGuffin et al., 2000) to identify and map secondary structure predictions onto their multiple sequence alignment. The aim of this activity is to encourage students to think about the three-dimensional protein structure and provide an opportunity for students to check the prediction method. Students compare the results of the secondary structure predictions with the crystal structure data and generally find some disagreement, although the predictions are very close. This activity helps emphasize the difference between predictions using bioinformatics programs and experimental data obtained from a database.
Before students begin working with the crystal structure data, they download the pdb file for either the human protein or a homologous protein from the Protein Data Bank (Berman et al., 2000). Students then view the crystal structure using DeepView (Guex and Peitsch, 1997). In viewing the structure, students are asked to develop a hypothesis for the role of the wild-type amino acid residue and the effect the mutated residue might have on protein structure and/or function. To accomplish this, students study changes in the noncovalent interactions of the amino acid side chain when the residue is mutated. The main focus of this portion of the lab is to examine the ball-and-stick view of the side chain where the missense mutation occurs and predict noncovalent interactions of the side chain based on its chemical nature and the distance of the neighboring atoms. Examination of noncovalent interactions gives the students a chance to investigate a portion of the crystal structure in depth. The students then use DeepView to “mutate” the selected side chain to the missense mutation they are studying, examine the possible effects of the change on the local noncovalent interactions, and predict how such interactions might be maintained or changed. This activity helps reinforce the concept of noncovalent interactions in a protein structure as well as the different chemical properties of the amino acid side chains. In combination with the structure analysis using DeepView, the students are asked to draw a noncovalent interaction with the residue using Fisher projections to represent the amino acid side chains. This type of activity has been shown to help students interpret three-dimensional structures (Richardson and Richardson, 2002). Additional examples of curriculum developed to aid students in analyzing crystal structures have recently been described in the literature (Centeno et al., 2003; Feig and Jabri, 2002; Honey and Cox, 2003; Richardson and Richardson, 2002).
After studying the structure, students analyze the metabolic pathway(s) containing their protein using the KEGG database. At this point, students can examine their hypothesis regarding the change in protein function and the effect on downstream events in the pathway. Finally, students develop the link between the gene they are studying and human disease using the OMIM database (NCBI, 2000). With the “Allelic Variants” portion of the OMIM entries, the students can read about the specific mutation they are examining and compare their hypothesis with the clinical (and sometimes biochemical) data available. Finally, students organize their results into a report that contains a one-page written summary, their multiple sequence alignment, and figures created using DeepView. By the completion of each project, students have traveled from genotype to phenotype, beginning with a DNA sequence and ending with clinical data.
During the last lab session, the students meet in small groups and present their projects to each other in the form of an oral report. For these presentations, the students who are working on the same project present together to the other pair who are working on the other project in the same disease category. For example, in the Lung Cancer group, the two students working on the K-Ras project present to the two students working on the Cytochrome P450 project (see Table 2). To help ensure that students understand the presentations and the relationship between the two projects, they work together to provide answers to a group quiz about the two projects. This quiz involves four essay questions (see Table 3) that help generate a discussion about the presentations and the hypotheses that the students develop for their projects.
Table 3.
Quiz questions
An Emphasis on Providing an Inquiry-Based Experience
The parts of the curriculum that emphasize inquiry are in analyzing the protein structure, developing a hypothesis about the structure, and writing and discussing the report summaries. In analyzing the protein structure, students are asked to assess the impact of the missense mutation on the protein structure, then relate this impact to effects on protein activity. With this activity, we are asking students to perform the first step in laboratory mutagenesis studies, predicting the experimental outcome of the mutation before the experiment has been performed. For most of the student projects, the mutagenesis experiment has not yet been performed, so the students are not able to look at experimental results of the mutations. In the cases where the mutagenesis results are available, such as with the phenylalanine hydroxylase project, the students read about the results and often discover that the results are not as straightforward as their predictions. For example, in the case of phenylalanine hydroxylase, the students discover that many mutations lead to folding and stability problems. The hypotheses that the students develop concerning the consequences of the mutation are graded on the basis of how well the students explain their reasoning and how well they incorporate the chemical concepts. Students often develop different hypotheses than their partners who are working on the same project, yet both can receive full credit.
After students read about the consequences of the mutation they are studying under the “Allelic Variants” section of the OMIM database and the KEGG database, they are asked to develop a conclusion for their final report that ties together all that they have learned about the mutation they are studying. As part of this conclusion, they are asked to hypothesize how the change in amino acid could lead to a symptom of a disease. Since students often develop different hypotheses to explain the same data, these differences lead to lively discussions and debates during their oral presentations. This activity helps to emphasize the importance of sharing ideas in the process of scientific research.
ASSESSMENT TOOLS
The assessment tools are designed to:
determine how well the students understand bioinformatics terms and the purpose of each database and tool used in the lab;
assess how students feel about the inquiry-based approach. Did they enjoy working on the projects, and did they feel they had gained important skills?
To address the first aspect of the assessment, a multiple-choice test was given before and after the lab. The questions were based on information that was available in the glossary designed for the lab, although students were expected to have learned these topics through the process of their Web-based research projects. The second aspect of the assessment was addressed by a questionnaire designed to obtain students' opinions by ranking their level of agreement to a series of statements. This questionnaire also solicited comments from students on any subject related to the lab. As both of these tools were available online and class time was provided to complete the forms, both tools had a high response rate, and many comments were obtained.
RESULTS AND DISCUSSION
Pre/Posttest
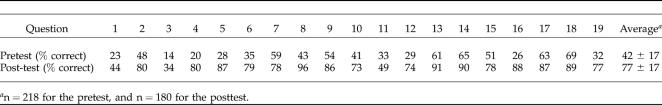
The pre/posttest consists of 19 multiple-choice questions concerning Web-based bioinformatics terms, tools, and databases. Although most students had not previously used the bioinformatics tools in the way required for this lab, several students had used these tools in an undergraduate research experience, and all of the students were introduced to the BLAST program (Altschul et al., 1997) in a previous biology laboratory that was a prerequisite for this course (Principles of Biology I, see Introduction). Students were asked to complete the test online from the lab Web site (Bednarski et al., 2004) on the first day of lab and again on the last day of lab. The average score on the pretest was 42%; this increased to an average score of 77% on the posttest, indicating that the students gained in knowledge of the tools used during this course. There were no significant differences on the scores of the pre/posttest when the scores were separated by lab section. A summary of these results is described in Table 4, and the questions used in the pre/posttest are provided in Appendix A. Although improvement was generally very high, three of the questions were significantly lower than average in the improvement between pre- and posttest, indicating a need to emphasize those points in the future. Overall, we found some of the questions more informative than others, and we are in the process of making revisions to this test.
Table 4.
Pre/posttest results
Postquestionnaire and Comments
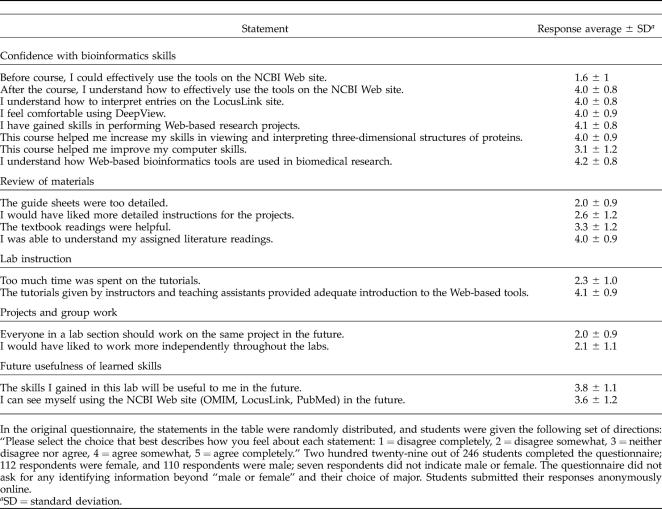
Statements on the postquestionnaire were designed to obtain students' perceptions of their own learning and their views on the curriculum materials, instruction, project assignment, and overall usefulness of the lab. The students were asked to respond to the statements using the Likert scale (Anderson et al., 1983; Likert, 1932). This scale and the results of the survey are summarized in Table 5.
Table 5.
Laboratory assessment questionnaire responses
The postquestionnaire is available online from the laboratory Web site (Bednarski et al., 2004), and the students completed the questionnaire during class on the day of the final presentations after completing the posttest. The questionnaires were anonymous and included an option to provide additional comments about the laboratory. Two hundred twenty-nine students out of 246 enrolled students completed the questionnaire, and 137 students chose to write additional comments. In analyzing the comments, we found that many students chose to make similar statements, and that the comments could be summarized by grouping them into categories. This grouping provides a quantitative view of the comments (see Table 6) (Wolcott, 2001).
Table 6.
Student comment data
Assessment of Goals
The major goals we had for the lab were to introduce all biology students to bioinformatics tools and databases, so that they had a basic knowledge of the types of databases and tools commonly used in biomedical research. Additionally, we wanted to create an inquiry-based lab experience for a large course by providing independent projects, requiring evidence-based predictions, encouraging a collaborative atmosphere, and requiring students to communicate their results.
In regards to our first goal, the pre/posttest results show that students gained a general understanding of bioinformatics terms, as well as the tools and databases used in the lab (Table 4 and Appendix A). On the questionnaire, students also agreed that they had learned how to access and use these tools. They responded with an average of 4.0 ± 0.9 (indicating agreement on a scale of 1–5) to six different questions on the postquestionnaire assessing their comfort in using the tools and databases. In addition, 31 students chose to comment that they were now confident that they could use the bioinformatics tools on their own (Table 6). The students were slightly less confident that they would use these tools in the future; they responded with an average of 3.6 ± 1.2 that they would use the NCBI Web site (including OMIM, LocusLink, and PubMed) in the future and an average of 3.8 ± 1.1 that they would use any of the skills learned in this laboratory in the future. However, 19 students commented directly that they could see themselves using these tools in the future (Table 6). As students encounter these tools in upper-level courses and undergraduate research experiences, we anticipate that student expectations will shift.
Our second goal was most directly addressed by the comments submitted by students. In Table 6, the first three categories of comments, totaling 44 comments, focused on gaining a better understanding of what scientists do and enjoyment of the investigative nature of the projects. Since the inquiry-based approach encourages students to mimic scientists, these comments suggest that this curriculum was successful in providing that experience for many students in the course. By providing independent projects, we hoped students would develop a sense of ownership toward their research; responses to the postquestionnaire and submitted comments suggest that the majority of students enjoyed working on their own projects. In the postquestionnaire, students disagreed with statements that everyone should work on the same project (2.0 ± 0.9), indicating that most students liked the current structure of the laboratory with respect to having their own project to work on. In order to encourage students to read journal articles, we included reading assignments from primary literature sources (the crystal structure paper and several abstracts) and a review article on the disease under study. The assessment results showed that the students slightly preferred the journal articles to the textbook readings. On the questionnaire, students agreed with statements about the helpfulness of the textbook reading assignments with an average of 3.3 ± 1.2 while reporting an understanding of the journal articles with an average of 4.0 ± 0.9. These results suggest that students enjoyed the challenge of reading journal articles. Instructors also noted students commented that they particularly appreciated the introduction to the PubMed database for finding journal articles.
Although we gave students individual projects, we encouraged a collaborative atmosphere in the lab, and students often worked closely with another student nearby. The students also worked in small groups in order to present their research results and work on a joint quiz. In response to these activities, students disagreed with the statement that they would rather work more independently (2.1 ± 1.1). In Table 6, seven comments fit into the category of “Enjoyed partner and group work.” Some comments further stated that the discussions with the group were important to understanding the projects. We interpreted these results to mean that most students enjoyed the collaborative experiences in the laboratory, which would not be possible if students worked completely independently on their projects.
Additional Observations
We observed that there were two areas where students commonly had difficulties in working through their projects. First, students often had a hard time understanding the importance of including sequences of proteins from distantly related species in their sequence alignment, and then interpreting their alignment once they had obtained it. Because of this, we plan to add an activity where students will need to predict which residues are important to protein function based on two different alignments. The first alignment will contain protein sequences from six closely related species, and the second will contain protein sequences from six distantly related species.
The second common difficulty was with predicting hydrogen bonds between amino acid side chains while studying the protein structure. Since many crystal structures are not solved to a resolution that can determine the location of hydrogen atoms, students were required to predict which acidic and basic residues would be protonated in order to predict hydrogen bonding. To help them prepare, we will be including a new written activity with amino acid representations created in DeepView to give students some experience with drawing hydrogen bonds between two residues before they work with the protein structures.
By observing the final presentations and talking with the students, it was clear that the preparation for the report and oral presentation was important for helping students assimilate the information that they had been collecting from the various bioinformatics databases and programs. Students became more motivated to understand the big picture in the Web-based research they were conducting when they needed to explain it to others. In the future, it may be beneficial to encourage students to begin preparing earlier in the course for their presentations to help them make important connections as they work through their projects. The main feature of understanding the big picture is understanding how genotype relates to phenotype. In fact, several students commented on the postquestionnaire that they enjoyed making the connections between DNA sequence, protein sequence, and phenotype as they worked through their projects (Table 6).
The group quizzes given after the oral presentations generated significant discussion within the groups and were important in helping the students understand each other's work. These discussions were often lively, while students fought to defend their own ideas. The presentations were kept informal to encourage discussion, but they gave students an opportunity to use scientific language that was new to them. Figure 3 shows several groups while they were presenting their final reports to each other.
Figure 3. Students presenting their oral reports.
Although the curriculum is written so that most students could complete the projects working on their own time, our observations and student comments suggest that the collaboration with partners, aid from instructors, and the group work were important in helping students to avoid simple but frustrating problems, to understand the exploratory nature of their projects, and to develop their hypotheses.
CONCLUSIONS
Overall, we are satisfied that both goals for the lab were met with this curriculum. The goal of providing an inquiry-based experience for students is difficult to assess. We used the students' comments and our observations to determine that students began to think and act more like scientists while working on the lab projects (Hassard, 2005). Many students commented that the lab gave them a better idea about the ways in which scientists approach problems and share information. The independent projects helped students focus on a single research problem and develop an interest toward that problem. Students used evidence to form hypotheses related to their projects, and, through our informal discussions with the students, we noticed changes in the way they discussed their projects, using more and more scientific language. The oral and written reports gave students a chance to practice their skills in communicating scientific information, and the collaborative nature and group work in the lab gave students opportunities to both defend their ideas and learn from their peers.
In regards to our first goal, the pre/posttest and questionnaire results showed that students learned about accessing and using a variety of Web-based tools and databases. This curriculum allows students to develop a foundation of bioinformatics skills that they will be able to build on in upper-level courses. There are several small, upper-level biology courses at WU that provide additional opportunities for students to work on bioinformatics-based projects.
It is too soon to determine the impact of this lab on the upper-level courses, since this lab was taught for the first time in Spring 2004 to sophomores, and students have 2 yr more to complete upper-level biology courses. It will be interesting to monitor both enrollment and student performance in these courses. Faculty of these upper-level courses have indicated that they found the topics covered in this curriculum to be relevant to the bioinformatics projects in their courses, and that they expect students to perform better on these projects. It appears likely that this will enable us to add additional projects to the upper-level curriculum in the future.
ACCESSING MATERIALS
The curriculum described in this article is available online at http://www.nslc.wustl.edu/courses/Bio3055/bio3055.html
Acknowledgments
This project was supported in part by a grant to Washington University in St. Louis, in support of Sarah C.R. Elgin, from the Howard Hughes Medical Institute through the HHMI Professors program (Grant 52003904). We would like to thank Kathy Westin-Hafer, Frances Thuet, David Heyse, Marcia Mannen, and Rob Compton at Washington University in St. Louis for their teaching and computer support during this course.
Appendix A
PRE/POSTTEST
Directions: Click in the circle next to the option that BEST answers the question.
-
1. Which of the following search programs/databases is NOT found at the NCBI Web site?
LocusLink
OMIM
dbSNP
PSIPRED
PubMed
-
2. Which of the following programs DO NOT use pdb files to create a picture of a three-dimensional molecule?
RasMol
Chime
Swiss-PdbViewer/DeepView
Protein Explorer
BLAST
-
3. All the coordinate files (.pdb) of three-dimensional protein structures that have been published are stored in the same online database. This database is:
ExPASy
RCSB
PubMed
PSIPRED
OMIM
-
4. The online database for genetic disease, located through the NCBI Web site, is called what?
ExPASy
RCSB
PubMed
PSIPRED
OMIM
-
5. To make a multiple sequence alignment, the sequences are saved in FASTA format in a text document, then copied and pasted into what program?
BLAST
ClustalW
PSIPRED
Swiss-PdbViewer
Sequence Manipulation Suite
-
6. Which of the following best describes the scoring pattern by homology search programs such as BLAST?
identical residue = 10 pts, gap = 0 pts
identical residue = 10 pts, conservative substitution = 5 pts, gap = 0 pts
identical residue = 10 pts, conservative substitution = 1–9 pts, gap = −3 pts
identical residue = 10 pts, conservative substitution = 1–9 pts, gap = 0 pts
identical residue = 10 pts, conservative substitution = 10 pts, gap = −3 pts
-
7. In the CPK coloring mode used in Swiss-PdbViewer, what atom type is blue?
hydrogen
carbon
nitrogen
oxygen
phosphorus
-
8. The correct FASTA format for a multiple sequence alignment is best explained by which of the following statements:
Use one-letter amino acid code, all capital letters, spaces separating each letter, and a return separating each line. Headings are put in quotation marks.
Use one-letter amino acid code, no spaces between letters. Returns are used to separate different sequences and headings. Heading lines are marked with a “>.”
Use one-letter amino acid code, all capital letters, no spaces between letters, with returns separating each line of the sequence. Headings are put in all capital letters, followed by a return.
Use three-letter amino acid code; capitalize the first letter of each amino acid, with a space between each amino acid.
Use one-letter amino acid code, put spaces every 10 amino acids, and put the numbering system above each line of amino acids.
-
9. Which font is used for alignments because each letter is the same width?
Times
Geneva
Helvetica
Courier
Arial
-
10. Which of the following sets of information is NOT usually found in a SwissProt entry?
the three-dimensional coordinates for the crystal structure
tissue and subcellular localization
amino acids in the active site
alternate names for the protein
the E.C. number for an enzyme
-
11. Which of the following databases would you select for a BLAST search if you would like to obtain the most homologous protein sequence to your search sequence that has its three-dimensional structure solved?
nr
swissprot
pdb
month
pat
-
12. Which of these sites combines the following information for a particular gene: RefSeq files, KEGG links, PubMed articles, OMIM entries, and dbSNP entries?
GenBank
ExPASy
LocusLink
RCSB
PSIPRED
-
13. Why is GenBank called a redundant database?
because it contains two versions of every entry
because it is saved in multiple locations for safety
because it contains similar sequences for different organisms
because it contains a historical record of sequence discoveries which sometimes includes multiple versions of the same sequence
because it is reread many times to check for mistakes
-
14, RefSeq files are useful to researchers for what reason?
RefSeq files have been edited and contain the most up-to-date sequence information for a particular site.
RefSeq files are edited to contain just the most biologically interesting sequence information.
RefSeq files are generated solely by a computer so contain no mistakes.
RefSeq files contain sequence information generated only from the Human Genome Project.
RefSeq files are the only sequence files that have been obtained experimentally.
-
15. Approximately how close together should two atoms be in a crystal structure to assume hydrogen bonding?
0.25 angstroms
0.5 angstroms
3 angstroms
6 angstroms
20 angstroms
-
16. Which of the following BEST describes the KEGG database?
a database of all macromolecular structure files
a database of metabolic pathways
a database of genetic disease genes
a database of phylogenetic trees
a database of enzyme substrates
-
17. Which of the following terms describes SNP's that result in an amino acid change in the protein?
synonymous change in the noncoding region
synonymous change in the coding region
nonsynonymous change in the noncoding region
nonsynonymous change in the coding region
mutation in the promoter region
-
18. A conservative mutation means:
an amino acid change in a conserved region of the protein
an amino acid change in a nonconserved region of the protein
an amino acid change from acidic to basic
an amino acid change from hydrophobic to hydrophilic
an amino acid change to an amino acid with similar size and chemical properties
-
19. If you were interested in reading a referenced summary about the research done on sickle cell anemia, which Web site would be best to go to?
KEGG
LocusLink
Sequence Manipulation Site
OMIM
SwissProt
REFERENCES
- Altschul S.F, Madden T.L, Schäffer A.A, Zhang J, Zhang Z, Miller W, Lipman D.J. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. (1997).;25:3389–3402. doi: 10.1093/nar/25.17.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen O.A, Flatmark T, Hough E. Crystal structure of the ternary complex of the catalytic domain of human phenylalanine hydroxylase with tetrahydrobiopterin and 3-(2-thienyl)-L-alanine, and its implications for the mechanism of catalysis and substrate activation. J. Mol. Biol. (2002).;320:1095–1108. doi: 10.1016/s0022-2836(02)00560-0. [DOI] [PubMed] [Google Scholar]
- Anderson A, Basilevsky A, Hum D. Measurement: theory and techniques. In: Rossi P, Wright J, Anderson A, editors. Handbook of Survey Research. New York: Academic Press; (1983).. pp. 231–287. In: [Google Scholar]
- Bednarski A, Heyse D, Pakrasi H. The course webpage for Biology 3055 at Washington University in St. Louis. (2004).. http://www.nslc.wustl.edu/courses/Bio3050/bio3055.html (accessed 2 May 2005).
- Berg J, Tymoczko J, Stryer L. Biochemistry. New York: WH Freeman and Company; (2002).. [Google Scholar]
- Berman H.M, Westbrook J, Feng Z, Gilliland G, Bhat T.N, Weissig H, Shindyalov I.N, Bourne P.E. The protein data bank. Nucleic Acids Res. (2000).;28:235–242. doi: 10.1093/nar/28.1.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boeckmann B, Bairoch A, Apweiler R, Blatter M.-C, Estreicher A, Gasteiger E, Martin M.J, Michoud K, O'Donovan C, Phan I, Pilbout S, Schneider M. The SWISS-PROT protein knowledgebase and its supplement TrEMBL in 2003. Nucleic Acids Res. (2003).;31:365–370. doi: 10.1093/nar/gkg095. http://us.expasy.org/sprot/ (accessed 2 May 2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell M.A. Public access for teaching genomics, proteomics, and bioinformatics. Cell Biol. Educ. (2003).;2:98–111. doi: 10.1187/cbe.03-02-0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centeno N.B, Villa-Freixa J, Oliva B. Teaching structural bioinformatics at the undergraduate level. Biochem. Mol. Biol. Educ. (2003).;31:386–391. [Google Scholar]
- Cooper S. Integrating bioinformatics into undergraduate courses. Biochem. Mol. Biol. Educ. (2001).;29:167–168. [Google Scholar]
- Feig A.L, Jabri E. Incorporation of bioinformatics exercises into the undergraduate biochemistry curriculum. Biochem. Mol. Biol. Educ. (2002).;30:224–231. [Google Scholar]
- Guex N, Peitsch M.C. SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling. Electrophoresis. (1997).;18:2714–2723. doi: 10.1002/elps.1150181505. http://www.expasy.org/spdbv (accessed 2 May 2005). [DOI] [PubMed] [Google Scholar]
- Hart P.J, Balbirnie M.M, Ogihara N.L, Nersissian A.M, Weiss M.S, Valentine J.S, Eisenberg D. A structure-based mechanism for copper-zinc superoxide dismutase. Biochemistry. (1999).;38:2167–2178. doi: 10.1021/bi982284u. [DOI] [PubMed] [Google Scholar]
- Hassard J. Assessing Active Science Learning: The Art of Teaching Science. New York: Oxford University Press; (2005).. [Google Scholar]
- Henikoff S, Henikoff J. Amino acid substitution matrices from protein blocks. Proc. Natl. Acad. Sci. U. S. A. (1992).;89:10915–10919. doi: 10.1073/pnas.89.22.10915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honey D.W, Cox J.R. Lesson plan for protein exploration in a large biochemistry class. Biochem. Mol. Biol. Educ. (2003).;31:356–362. [Google Scholar]
- Honts J.E. Evolving strategies for the incorporation of bioinformatics within the undergraduate cell biology curriculum. Cell Biol. Educ. (2003).;2:233–247. doi: 10.1187/cbe.03-06-0026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Istvan E.S, Deisenhofer J. Structural mechanism for statin inhibition of HMG-CoA reductase. Science. (2001).;292:1160–1164. doi: 10.1126/science.1059344. [DOI] [PubMed] [Google Scholar]
- Jones D. Protein secondary structure prediction based on position-specific scoring matrices. J. Mol. Biol. (1999).;292:195–202. doi: 10.1006/jmbi.1999.3091. [DOI] [PubMed] [Google Scholar]
- Jungck J.R, Donovan S. Evolution as a basis for bioinformatics education. Mol. Biol. Cell. (2000).;11:26a. [Google Scholar]
- Kanehisa M. A database for post-genome analysis. Trends Genet. (1997).;13:375–376. doi: 10.1016/s0168-9525(97)01223-7. [DOI] [PubMed] [Google Scholar]
- Kanehisa M, Goto S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. (2000).;28:27–30. doi: 10.1093/nar/28.1.27. http://www.genome.ad.jp/kegg/ (accessed 2 May 2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Likert R. Technique for the measurement of attitudes. Arch. Psych. (1932).;21:140. [Google Scholar]
- McGuffin L, Bryson K, Jones D. The PSIPRED protein structure prediction server. Bioinformatics. (2000).;16:404–405. doi: 10.1093/bioinformatics/16.4.404. [DOI] [PubMed] [Google Scholar]
- Milburn M.V, Tong L, deVos A.M, Brunger A, Yamaizumi Z, Nishimura S, Kim S.H. Molecular switch for signal transduction: structural differences between active and inactive forms of protooncogenic ras proteins. Science. (1990).;247:939–945. doi: 10.1126/science.2406906. [DOI] [PubMed] [Google Scholar]
- National Center for Biotechnology Information. OMIM—Online Mendelian Inheritance in Man. (2000).. http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=OMIM (accessed 2 May 2005).
- National Library of Medicine. PubMed. (2004).. http://www.ncbi.nlm.nih.gov/entrez/query.fcgi (accessed 2 May 2005). [DOI] [PubMed]
- National Research Council. How People Learn: Brain, Mind, Experience, and School. Washington, DC: National Academy Press; (1999).. [Google Scholar]
- National Research Council. Inquiry and the National Science Education Standards: A Guide for Teaching and Learning. Washington, DC: National Academy Press; (2000).. [Google Scholar]
- National Research Council. Bio2010: Undergraduate Education to Prepare Biomedical Research Scientists. Washington, DC: National Academy Press; (2002).. [Google Scholar]
- Pruitt K, Maglott D. RefSeq and LocusLink: NCBI gene-centered resources. Nucleic Acids Res. (2001).;29:137–140. doi: 10.1093/nar/29.1.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rano T.A, Timkey T, Peterson E.P, Rotonda J, Nicholson D.W, Becker J.W, Chapman K.T, Thornberry N.A. A combinatorial approach for determining protease specificities: application to interleukin-1beta converting enzyme (ICE) Chem. Biol. (1997).;4:149–155. doi: 10.1016/s1074-5521(97)90258-1. [DOI] [PubMed] [Google Scholar]
- Rastogi V.K, Girvin M.E. Structural changes linked to proton translocation by subunit c of the ATP synthase. Nature. (1999).;402:263–268. doi: 10.1038/46224. [DOI] [PubMed] [Google Scholar]
- Richardson D.C, Richardson J.S. Teaching molecular 3-D literacy. Biochem. Mol. Biol. Educ. (2002).;30:21–26. [Google Scholar]
- Rudenko G, Henry L, Henderson K, Ichtchenko K, Brown M.S, Goldstein J.L, Deisenhofer J. Structure of the LDL receptor extracellular domain at endosomal pH. Science. (2002).;298:2353–2358. doi: 10.1126/science.1078124. [DOI] [PubMed] [Google Scholar]
- Shi W, Li C.M, Tyler P.C, Furneaux R.H, Grubmeyer C, Schramm V.L, Almo S.C. The 2.0 A structure of human hypoxanthine-guanine phosphoribosyltransferase in complex with a transition-state analog inhibitor. Nat. Struct. Biol. (1999).;6:588–593. doi: 10.1038/9376. [DOI] [PubMed] [Google Scholar]
- Steward R.E, MacArthur M.W, Laskowski R.A, Thornton J.M. Molecular basis of inherited diseases: a structural perspective. Trends Genet. (2003).;19:505–513. doi: 10.1016/S0168-9525(03)00195-1. [DOI] [PubMed] [Google Scholar]
- Stothard P. The sequence manipulation suite: JavaScript programs for analyzing and formatting protein and DNA sequences. BioTechniques. (2000).;28:1102–1104. doi: 10.2144/00286ir01. http://bioinformatics.org/sms/ (accessed 2 May 2005). [DOI] [PubMed] [Google Scholar]
- Thompson J.D, Higgins D.G, Gibson T.J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. (1994).;22:4673–4680. doi: 10.1093/nar/22.22.4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams P.A, Cosme J, Ward A, Angove H.C, Matak Vinkovic D, Jhoti H. Crystal structure of human cytochrome P450 2C9 with bound warfarin. Nature. (2003).;424:464–468. doi: 10.1038/nature01862. [DOI] [PubMed] [Google Scholar]
- Wolcott H.F. Writing up Qualitative Research. Thousand Oaks, CA: Sage Publications; (2001).. [Google Scholar]
- Yoshikawa S, Shinzawa-Itoh K, Nakashima R, Yaono R, Yamashita E, Inoue N, Yao M, Fei M.J, Libeu C.P, Mizushima T, Yamaguchi H, Tomizaki T, Tsukihara T. Redox-coupled crystal structural changes in bovine heart cytochrome c oxidase. Science. (1998).;280:1723–1729. doi: 10.1126/science.280.5370.1723. [DOI] [PubMed] [Google Scholar]