Abstract
The complexity and expansive nature of thalamic research has led to numerous interventions for varied disease states. At the same time, this complexity along with siloed areas of study can hinder a comprehensive understanding. The goal of this paper is to give the reader a broader and more detailed perspective on the thalamus. In order to accomplish this goal, the paper begins with a summary of the function, electrophysiology, and anatomy of the normal thalamus. With this foundation, thalamic involvement in neurological diseases is discussed with a focus on epilepsy. Therapeutic interventions in the thalamus for epilepsy as well as movement disorders, psychiatric conditions and disorders of consciousness are described. Lastly limitations in the field and future models of data sharing and cooperation are explored.
Keywords: Thalamus, Neuromodulation, Tremor, Coma, Tourette's syndrome, Drug-resistant epilepsy
Introduction
The thalamus, first described by philosopher and physician Claudius Galenus in the 2nd century A.D, derives from the Greek word meaning “inner chamber” or “anteroom” [1]. This waiting room was originally thought to represent a hollow chamber connecting the optic tracts with the lateral ventricle. Over one thousand years later the thalamus became known by its other architectural meaning of “bridal chamber” where information must first pass through a solid mass of ovoid subcortical gray matter with functions still yet to be discovered [2].
As of this writing, there are 67,000 PubMed entries for the word thalamus, and despite this explosion of investigation, the mysteries of the structure and function still abound. This article provides a comprehensive understanding of the structure and function of the thalamus, its role in disease states, as well as the history and promise of thalamic interventions. The target audience includes clinicians, particularly neurologists and psychiatrists as well as basic scientists in need of a wider perspective.
Development of the thalamus
In human development, at 4 weeks, the central nervous system divides into 3 vesicles, the prosencephalon, the mesencephalon and rhombencephalon. By week 5, the prosencephalon has further divided into the telencephalon, which becomes the cerebral hemispheres (including the amygdala and basal ganglia) and the diencephalon. Further differentiation of the diencephalon creates the thalamus and the hypothalamus [3]. Thalamic neurons are born in the ventricular zone surrounding the third ventricle and are thought to migrate laterally along radially oriented glial fibers, with the earliest generated cells taking the most lateral position. The separate nuclei begin to be discernible during gestational weeks 8–16. Interestingly, the development and differentiation of the thalamus precede that of the telencephalon [4].
Thalamic structure and function
The thalamus is a large cluster of gray matter at the center of the brain. Its very anatomical position - between the brainstem and the cerebral cortex – hints at its function as a relay station and a hub for communication. In addition, the thalamus is a key component involved in controlling our level of consciousness and is involved in sleep initiation, stabilization and termination [[5], [6], [7], [8], [9]]. Vastly different experiences like arousal, vision, hearing, touch, movement, and emotional regulation all involve the thalamus but center in different nuclei. What unites the thalamus is not its functions, which are multi-faceted and disparate, but its method.
Thalamic cell types
The thalamus is comprised of two fundamental cell types [10,11]. The first cell type, projection neurons, are excitatory and glutamatergic, extending their axons outside the thalamus to target the cortex, striatum, and amygdala. As they exit the thalamus, these neurons send collaterals to specific subsections of the reticular nucleus [10]. Interestingly, there are no recurrent intra-thalamic excitatory inputs. This is in stark contrast to cortical neurons that receive abundant excitatory input from local and long distance intracortical connections [11]. The lack of local excitatory connections could restrict the thalamus’ function to organizing the perception of a signal rather than modifying the signal itself. The second fundamental cell type, inhibitory GABAergic interneurons, do not leave their nucleus of origin. They are extensively interconnected with the projection neurons and with each other [11].
Burst and tonic mode
Thalamic projection neurons have response modes known as burst and tonic [11,12]. Burst mode is intrinsically linked to the state of the T-type calcium channels in the soma and dendrites. Burst mode was originally described in drowsiness and in sleep [9]. A thalamic cell enters burst mode when there is a large degree of hyperpolarization, and in this state the T-type calcium channel is closed. The T-type calcium channel becomes active with slight depolarization, opening the voltage gated channel, causing an influx of Ca2+ which in turn activates a burst of conventional action potentials. Once the channel is open, intrinsic mechanisms inactivate the channel, preventing further calcium conductance. This inactivation is slow, lasting 100 ms or longer, so at most the neuron can fire a few times per second. In the inactive state, the channel requires a hyperpolarized state to return to the de-inactivated state (which can be considered the closed state). A neuron in burst mode does not reflect the activity of its inputs with any fidelity.
Neurons that are in a more depolarized state are in a tonic mode, meaning that further depolarization causes a train of action potentials whose frequency is proportional to the input magnitude. Tonic firing is sustained by voltage gated sodium and potassium channels whereas the T-type calcium channel is less involved.
One can conceptualize these two modes as a system for gating the specificity of information leaving the thalamus. Focusing attention on facial sensation may put the thalamic neurons of the ventral posteromedial nuclei into tonic mode, a state in which the afferents are transmitted with fidelity. In sleep, thalamic neurons are mostly in burst mode, effectively blocking transmission from the specific inputs. However, cells in burst mode do transmit information. In fact, a cell in burst mode is more likely to fire with only a slight depolarization, making this mode a more sensitive detector of change [12].
Drivers and modulators
Thalamic nuclei can be roughly divided into those subserving a specific function (e.g., vision and the lateral geniculate nucleus) and association nuclei which are involved in broader diffuse networks (e.g., dorsomedial, intralaminar, pulvinar). For the specific nuclei, the afferents can be conceptualized as drivers and modulators. The drivers are easy to grasp sending the content of information to the thalamus, via glutaminergic synapses. Examples of drivers are the retinal cells going to the lateral geniculate or neurons containing sensory information going from the gracile and cuneate nuclei to the ventral posterolateral nucleus (VPL) [11,13]. The second category of afferents are modulatory, consisting of axons originating from cortical layer VI neurons (most often the same area of cortex that receives information from that thalamic nucleus), subcortical structures including the basal ganglia, intrathalamic neurons (which are all inhibitory), the cerebellum and projections from the brainstem reticular formation. The neurotransmitters of the modulatory inputs are more diverse and can be glutamate, gamma-aminobutyric acid (GABA), serotonin, dopamine, acetylcholine and norepinephrine [14]. The modulators far outnumber the drivers and determine whether and in what form the content from the specific inputs reaches the cortex. For many thalamic nuclei like the pulvinar or dorsomedial nucleus the function of the nucleus itself is not fully understood, and hence the afferents cannot be simply subdivided into drivers and modulators. These nuclei are often termed association nuclei and are involved in visual salience, perception, multi-modal sensory processing, behavioral flexibility, and level of consciousness [3,[15], [16], [17], [18], [19]].
Thalamic nuclei
Although the thalamus can be divided into around 30 different nuclei, we will be discussing and illustrating only a handful (Fig. 1, Fig. 2). The thalamic subdivisions are not easily visible on MRI and historically there has been variation in naming convention and anatomical location outlined in different atlases [[20], [21], [22], [23], [24]]. For the purposes of this paper, the Morel atlas is followed most closely.
Fig. 1.
The anatomy of the thalamus. Right and left thalami shown. The left thalamus is shown with the reticular nucleus removed. The anterior division (blue), medial division (purple), lateral division dorsal tier (greens), lateral division ventral tier (reds/oranges), and intralaminar nuclei (yellow/blue) are shown. The reticular nucleus (gray) covers the lateral surface of the right thalamus. This diagram adheres most closely to the Morel atlas with the exception that the ventral intermediate nucleus is considered part of the ventral lateral nucleus in the Morel atlas. Diagram courtesy of artist Vera Liu.
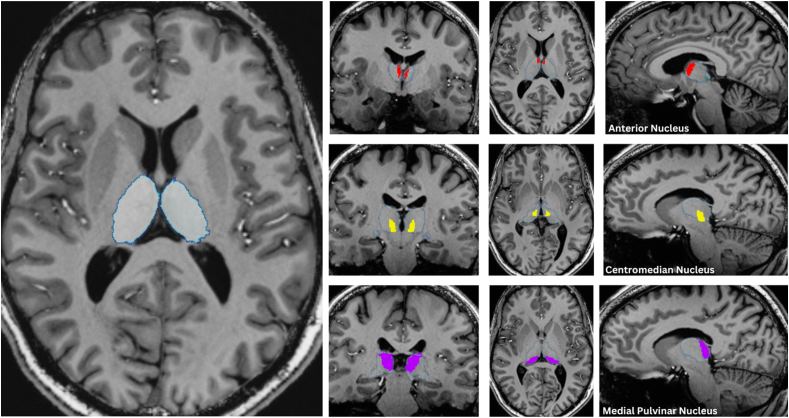
Fig. 2.
Thalamic segmentations using high field imaging and automatic segmentations. Example of single-slice segmentation of the thalamus and three subnuclei of interest, the anterior, centromedian, and medial pulvinar overlaid on a T1-MPRAGE of a single subject (TR = 6000 ms, TE = 3.62 ms, FOV = 240 mm × 320 mm, resolution = 0.7 mm isotropic) acquired using a 7 tesla Siemens MRI scanner. FreeSurfer 7.2 (Fischl et al., 2001; Fischl et al., 2004) with motion correction, intensity normalization, skull stripping and neck removal, automatic segmentation, and parcellation processing steps was used with the T1 image to segment the thalamus and subsequent subnuclei.
The anterior nucleus
The anterior nucleus is comprised of three different subnuclei-anterodorsal, anteroventral and anteromedial all with slightly different connections [25]. It is part of the limbic circuit, historically known as the circuit of Papez, which begins in the hippocampus, moves posteriorly through the crura of the fornix which then fuse into the body of the fornix. The fornix then arches anteriorly and descends with primary projections onto the mammillary bodies. The mammillary bodies project to the anterior nucleus via the mammillothalamic tract. The primary efferent of the anterior nucleus connects to the anterior cingulate cortex, which projects to the entorhinal cortex and then in turn back to the hippocampus [26] (Fig. 3). The anterior thalamic nucleus is involved in arousal as well as sleep. Recently, the anterior thalamic nucleus has been implicated in organizing sleep rhythms, specifically sleep spindles, and coordinating these sleep oscillations in memory consolidation [27].
Fig. 3.
Connectivity of thalamic nuclei in a single subject using diffuse-weighted MRI. Example of 3D rendering of tracts emanating from the anterior, centromedian, and medial pulvinar nucleus in an individual subject. Each subnuclei, segmented with the methods used in Fig. 1 is displayed along with the tracts which were generated from a high-angular-resolved diffusion-weighted imaging MRI (dMRI) sequence (b = 1500 s/mm2, TR = 7200 ms, TE = 67.6 ms, FOV = 210 × 210 mm, resolution = 1.05 mm isotropic, 64 directions, and 5 b0 acquisitions) acquired at 7T. The dMRI series in both anterior-to-posterior and posterior-to-anterior directions were collated into a single volume which was denoised and corrected for eddy current distortions, motion, and B1 inhomogeneity. MRtrix was used to generate tractography seeding from each individual subnuclei to whole brain using SIFT2 and tcksample with 1000 seeds per voxel.
The motor and sensory nuclei
The lateral division of the thalamus is further divided into a dorsal and ventral tier. Within the ventral tier, the motor and sensory nuclei are anatomically arranged from anterior to posterior to communicate with related areas of the telencephalon. The more anterior divisions, the ventral anterior and ventral lateral nuclei are most connected to the motor system, receiving afferent information from the cerebellum. The ventral intermediate nucleus sits behind the ventral lateral nucleus and is thought to receive projections for deep (kinesthetic) sensation and to mediate tremor, thus containing function that is both sensory and motor [28]. Of note, this nucleus is considered a subfield of ventral lateral nucleus in the Morel atlas [[21], [22], [23]]. The ventral posteromedial (face) and lateral (body) nuclei are more posterior and receive input from the sensory tracks from the brainstem and project onto the sensory cortex.
The lateral geniculate and medial geniculate nuclei
The most posterior and inferior nuclei of the ventral group are the lateral geniculate nuclei and the medial geniculate nuclei, which receive direct inputs from the visual and auditory systems, respectively.
The pulvinar
The dorsal tier of the lateral division contains the largest of the thalamic nuclei, the pulvinar, which is a large association nucleus with extensive connections to the posterior parietal, occipital, insular and cingulate cortices (Fig. 3). Further, it is connected to the anterior temporal lobe. It is thought to be involved in multimodal sensory processing and visual attention and salience [18,19]. Of all the nuclei of the thalamus, the pulvinar has increased in size the most in evolution [29].
The reticular nucleus
The thalamic reticular nucleus covers the lateral surface of the thalamus like a blanket, receiving input from projection neurons as they enter or leave the thalamus. Interestingly, thalamic reticular neurons only project onto the thalamus itself, via synapses that are strictly inhibitory. The thalamic reticular nucleus is the only one without excitatory projections outside of the thalamus. In wakefulness, this nucleus is involved in selective attention. In sleep, a loop between the reticular nucleus of the thalamus and the other nuclei is thought to underlie sleep spindles [8,30].
The dorsomedial nucleus
The largest nucleus on the medial surface of the thalamus is dorsomedial nucleus with extensive projections to the prefrontal cortex, the cingulate and insular cortices as well as the brainstem [29,[31], [32], [33]]. It receives input from the limbic system, including the amygdala and the cingulate cortex. It is involved in affect, memory, and level of consciousness. Bilateral lesions here can be a cause of coma [34].
Intralaminar nuclei, with a focus on the centromedian (CM) and central lateral (CL)
Some of the thalamic nuclei are separated by the internal medullary lamina. This lamina further encircles other nuclei, which are aptly termed intralaminar nuclei and includes the centromedian nucleus (CM). The CM projects primarily to the striatum with some direct peri-rolandic projections to the neocortex (Fig. 3). Other regions of the brain are influenced via multisynaptic connections [35]. This CM is part of the reticular activating system, which plays a crucial role in regulating wakefulness, arousal and consciousness. The afferents of the CM are from the brainstem, the cerebellum, the basal ganglia, other thalamic nuclei and the motor cortex. The central lateral nucleus is contiguous with the CM and (not surprisingly) farther from the midline, with a similar efferent and afferent profile [36]. This nucleus is also highly connected to the reticular activating system and is being tested as a target for disorders of consciousness [37].
Lesions of the thalamus
As neurologists, we learn the function of an area from its dysfunction (lesion). Korsakoff syndrome, which is linked to vitamin B1 deficiency due to alcohol use disorder, leads to both anterograde and retrograde amnesia. It is characterized by widespread brain atrophy, with the most significant degeneration occurring in the anterior thalamic nucleus [38,39]. The rare autosomal dominant prion disease fatal familial insomnia is thought to involve preferential destruction of the dorsomedial and anterior nucleus of the thalamus, disrupting the natural sleep cycle causing insomnia, specifically with lack of slow wave sleep, as one of the earliest manifestations [40]. Thalamic vascular lesion can cause a wide range of symptoms, dependent on their exact location, including memory disturbances, decreased level of consciousness or coma, aphasia, hemiataxia, pain syndrome, and sensory loss [41]. Interestingly, acquired lesions to the thalamus do not cause epilepsy. One notable exception is that neonatal thalamic lesions are linked to developmental and/or epileptic encephalopathy with spike-wave activation in sleep (DEE-SWAS), a condition in which epileptic spikes are triggered during non-REM sleep. Clinically, this condition has varying phenotypes and is associated with behavioral disturbances, language regression, acquired aphasia and epilepsy. Children with this condition often have a thalamic lesion, of varying etiologies [[42], [43], [44], [45]]. Furthermore, children with DEE-SWAS and normal MRIs have changes on magnetic resonance spectroscopy and with MRI based quantitative volume analyses to suggest neuronal cell loss as well as smaller thalamic volumes [[46], [47], [48]].
Epilepsy and the thalamus
Neuromodulation of the thalamus in drug-resistant epilepsy
Neuromodulation of the thalamus differs from most other surgical interventions as the thalamus is not considered the seizure onset zone. Rather, the thalamus is involved in seizure propagation and particularly involved in the loss of consciousness with seizures [[49], [50], [51], [52]]. Further, the widespread cortical connections of the thalamic nuclei offer a way to reach multiple brain regions at once in our effort to interrupt the seizure network. In temporal lobe epilepsy, the degree of thalamocortical synchrony correlates with the presence of early loss of consciousness and a high degree of synchrony worsens the prognosis after focal epilepsy surgery [49].
Given the widespread thalamocortical connectivity, it is surprising that thalamic stimulation is tolerated as well as it is. Stimulation side effects when they do occur are location specific. For example, stimulation in the CM can cause facial paresthesia due to the proximity of the ventral posteromedial nucleus. It is worth noting that results from the previous century, often in animal models, were mixed and some thalamic stimulation (particularly high intensity and low frequency) was found to be proconvulsant with high frequency stimulation having a desynchronizing and anticonvulsant effect [53,54].
Thalamic targeting is particularly appealing when the seizure network is large and not amenable to resection. The anterior thalamic, CM, pulvinar, dorsomedial and central lateral nuclei are areas of active investigation and use [50,55]. While there are hundreds of articles on thalamic stimulation and epilepsy, there are only a few published randomized double-blind controlled trials. In the last 20 years there are 2 for deep brain stimulation (DBS) in the anterior nucleus [56,57] in people with drug-resistant focal epilepsy and 1 for DBS in the CM for Lennox Gastaut syndrome (LGS) [58]. Randomized double-blind trials for responsive stimulation in the thalamus for IGE and for patients with LGS are underway [59,60]. There are no head-to-head comparisons testing what thalamic nucleus works best for what type of patient, or whether to use responsive, continuous or duty cycle stimulation. Further, stimulation parameters like pulse width, frequency, and voltage are untested by formal means.
Open loop (duty cycle and continuous) vs closed loop (responsive) stimulation
An open loop for any system means that there is no real time feedback guiding function. In a closed loop, data is being collected and used in real time to drive the system action. Open loop stimulation in epilepsy has a history dating back to the 1980s at least, with a closed loop system being a more recent actor. As of this writing, the randomized, double-blind, controlled trials with thalamic stimulation used an open loop stimulation, delivered on a duty cycle, typically with alternative 1-min active and 5-min inactive phases.
Responsive neurostimulation (RNS) differs from duty cycle stimulation in that the device stimulates only when there is a detected change in electrocortical (ECoG) activity, based on physician specified detection parameters. Detectors are set to deliver hundreds to thousands of therapies per day and are not necessarily limited to the ECoG signature that resembles seizure onset. The two approaches have not been studied head-to-head. However, both approaches are well tolerated by patients and with few side effects. Advantages and disadvantages of these systems have little to do with closed loop versus open loop delivery system and more to do with the specific model versions. Advantages of DBS include at home adjustment capabilities and a rechargeable Implantable Pulse Generator (IPG). Advantages of RNS are detection and response to seizure activity in real-time with targeted stimulation and ECoG data storage to the cloud which allows epileptologists to have a deeper understanding of their patient's epilepsy which goes far beyond the flawed seizure diary [61]. DBS disadvantages include running the risk of mood exacerbation (in the anterior nucleus specifically) whereas RNS requires sophisticated programming and frequent adjustments.
The pivotal RNS study leading to pre-market approval included patients with one or two seizure onset zones, with contacts placed on or in the cortex of the seizure onset with strip or depth electrodes [62]. In the blinded period the sham group showed significantly less reduction in the frequency of seizures and in the open label period the patients originally assigned to the sham group saw a significant reduction in seizures. Further the device efficacy improved with time with a 75 % medium reduction at 9 years [62,63].
The initial concept for the RNS came from the observation that brief pulses of stimulation stopped after-discharges and seizures during brain mapping [64]. However, further study has suggested that the beneficial effects are not from “stopping seizures” but from stimulation-induced modulation of the network [65,66].
Importantly, none of the original stimulation targets were in the thalamus. Since the pivotal RNS paper, many centers have begun to use responsive thalamic stimulation. The current RNS system allows for the placement of two leads with four contacts each. One option with the RNS system is to place one lead in the thalamus and one in the cortex. If seizures are seen in the thalamus at or close to the moment of onset, bilateral thalamic RNS without a cortical lead may be used. Alternatively, as is being tested in an ongoing LGS trial, two RNS systems can be placed bilaterally with detection in the cortex and stimulation in the thalamus [61].
Single and multicenter retrospective series of thalamic RNS [[67], [68], [69], [70], [71], [72], [73], [74], [75], [76]] have reported benefit with the largest multisite retrospective review of 25 patients demonstrating a 54 % at 6 months to a 75 % at 2 years median seizure reduction [71]. Of note, these papers contain heterogeneous patient populations with focal, multifocal, idiopathic, and symptomatic generalized epilepsy. Implantations included different thalamic nuclei (anterior nucleus, CM, and pulvinar), with varying detection and stimulation pathways. In addition, some patients had bilateral thalamic implants, some had a single thalamic lead and a cortical lead and rarely two RNS systems implanted with each system having a cortical and ipsilateral thalamic lead [74]. Numbers were too small to compare the efficacy of any of these variables. The retrospective data published so far does suggest that RNS thalamic stimulation is safe and shows promise [[67], [68], [69], [70], [71], [72], [73], [74], [75]]. Smaller papers which focused on a single thalamic nucleus will be discussed in the section of that nucleus.
Thalamic nucleus selection for the treatment of drug-resistant epilepsy
One must begin to think about the relationship of the thalamic nucleus to the seizure onset zone in terms of time, connectivity, and functional effect. This inquiry is being pursued via several modalities including stereo-EEG (SEEG) with thalamic sampling, structural connectivity studies and the rather obvious methods of examining efficacy outcomes in patients with thalamic stimulation.
One current working hypothesis is efficacy will be optimized by choosing a thalamic nucleus with the greatest connectivity to the seizure onset zone (Table 1). The best evidence for this is in a sub-analysis of the Sante trial of 11 patients with anterior thalamic DBS. Patients with seizures within the network comprised of the hippocampal formation-mammillary bodies-anterior nucleus-cingulate gyrus (historically referred to as the circuit of Papez) had a significantly higher median seizure reduction rate (80 % vs 53 %) compared to those with seizures outside this network at 1 year [77,78].
Table 1.
Working concepts of nucleus selection for thalamic neuromodulation.
| Anterior nucleus | CM | Pulvinar | |
|---|---|---|---|
| Epilepsy type | Focal/Multifocal epilepsy Epilepsy involving the limbic network
|
Generalized epilepsy LGS Frontal lobe epilepsy |
Temporal lobe epilepsy Posterior quadrant epilepsy |
In clinical practice, multiple centers include thalamic leads while designing an individual SEEG implant, knowing that resection or ablation may not be possible. The trajectory of a thalamic lead can be tailored to address the hypothesis for a particular patient. For example, in a patient with seizures of bilateral independent onsets with unresponsiveness ± falls, bilateral leads had their internal contacts in the CM with mid contacts in the posterior insular and external contacts in the parietal operculum (Fig. 4). A review of thalamic SEEG exploration demonstrated that the medial pulvinar, CM, anterior nucleus, and dorsomedial nucleus have been sampled and that this practice is safe with no reported long-term complications [79]. Further, one study suggests that thalamus-driven hypersynchronization may be a mechanism by which seizures are terminated [80].
Fig. 4.
Thalamic recordings from SEEG to RNS. A woman with DRE and bilateral independent seizure onsets on scalp EEG with semiology of unresponsiveness and occasional falling. SEEG had broad bilateral coverage including CM leads whose trajectory included the posterior insular and the parietal operculum [A]. [B] Seizures had broad bilateral independent onsets with CM involvement within 100 msec (right onset shown, only subset of SEEG shown). Patient underwent bilateral CM RNS [C] and seizures were well detected (left sided onset shown). Patient has not had a fall since RNS but continues to have very brief focal impaired awareness seizures.
In one study of focal epilepsy with at least one contact in the thalamus during SEEG, the thalamus was involved within 15 s of onset in most seizures (86 %) and involved in the epileptogenic zone in 20 % of patients. Further, the patients with a higher epileptogenicity index in the thalamus had a poorer response to surgery. Thalamic involvement was independent of epilepsy etiology or lobe of origin [81].
Wu et al. looked at the propagation patterns within the thalamus of 11 patients with presumed temporal lobe epilepsy [82]. Interestingly, onsets during SEEG were seen in the mesial temporal lobe, lateral temporal lobe, insula, and orbital frontal cortex, highlighting the importance of intracranial EEG for precise localization. There was SEEG sampling of the dorsomedial, medial pulvinar and anterior nucleus (the CM was unfortunately not sampled in this study). A stereotyped seizure in a given patient would have a predictable spread pattern in the thalamus. Further, more than half of the subjects showed spread to the medial pulvinar prior to the anterior nucleus. Most interestingly, the seizure onset anatomical location did not predict if the seizure was going to be seen in the pulvinar or anterior nucleus first. This suggests that a patient-specific approach may have the best outcome, and our more textbook knowledge of networks may not be applicable at the individual level. While clinicians are gaining data that feels useful from thalamic exploration during SEEG, there is controversy over this practice. Bernabei et al. suggest that thalamic SEEG be done with IRB approval and that data should be aggregated and shared across centers to further maximize the knowledge gained [83].
Another non-invasive individualized approach is using advanced imaging such as fMRI, high field MRI and diffusion MRI to understand thalamic involvement and to define networks [32,[84], [85], [86]]. The ability to noninvasively differentiate and visualize volumes and connectivity of the thalamic subnuclei may result in greater precision and accuracy for future epilepsy treatment paradigms.
If bilateral thalamic RNS is being considered, the temporal relationship may be of particular importance as stimulation within 1 s of onset is thought to be most effective. In addition, the electrographic signature of the thalamic SEEG should stand out from the background enough that a sensitive and specific detection algorithm can be designed around it. If the thalamic onset is highly subtle or delayed an open loop approach may be best.
In generalized epilepsy, the relationship of the thalamus and the cortex is considered more tightly coupled, with Penfield and Jasper postulating that generalized seizures arise from a deeper source that they termed centroencephalic [87]. As early as the 1950s, the generalized spike and wave discharges of absence seizures were recorded from the thalamus in humans, with several recordings seen in the “electrothalamogram” but not in the cortex [88]. Currently the relationship is seen as bidirectional with generalized onset seizures being generated from a thalamocortical loop network. Regarding nuclei specificity, in a simultaneous EEG-fMRI study, Tyvaert et al. demonstrated that CM is involved prior to the anterior nucleus in the generation of generalized spike and wave discharges [89].
Anterior nucleus
In the first randomized controlled double-blinded investigation of DBS in DRE, the SANTE trial, 110 patients with drug resistant focal epilepsy underwent lead implantation into the anterior nucleus programmed with duty cycle stimulation [57]. For 3 months patients were randomized to receive stimulation or sham stimulation. In this blinded period, the sham participants had a 14.5 % seizure reduction, and the stimulated group had 40.4 % seizure reduction. After 3 months all participants received stimulation. By 2 years, there was a 56 % median reduction of seizure frequency with 54 % of patients having a seizure reduction of at least 50 %. Characteristic of many neuromodulatory studies in epilepsy, the results in long term follow up continued to improve with a 70 % median seizure frequency reduction and improved quality of life [78]. A second smaller randomized trial again tested DBS in the anterior nucleus of 18 patients. This trial, however, failed to show a significant difference in seizure frequency in the stimulated group versus the not stimulated group after 6 months and the results did not improve over time, though only 12 months of data were published [56]. An observation study of DBS in the anterior nucleus with nearly 200 patients showed a more modest efficacy but with a 33 % seizure reduction at 2 years [90].
A common side effect reported in anterior nucleus stimulation is depression which was reported in 37 % of subjects at 7 years in the SANTE trial, though two thirds had depression as a premorbid condition. The rate of SUDEP in the SANTE trial was lower than expected for drug resistant epilepsy patients and similar to the rates of patients who are surgically treated with resection or laser ablation. No adverse events worsened over long term follow up [78,91].
Further analysis of the anterior nucleus target has shown that patients with better outcomes are being stimulated in the most anterior aspect of the anterior nucleus, closer to the mammillothalamic tract [92]. The lead placement in the SANTE trial was subsequently analyzed, with bilateral lead placement scored on a scale of 0 (poor) to 6 (perfect) using axial, coronal, and sagittal images of the thalamus. When the leads failed to be in the ideal spot, they were mostly either too medial or too posterior. Patients with low scores were significantly less likely to be responders [93].
Centromedian nucleus (CM)
The CM has widespread connections with the basal ganglia, brainstem, cerebellum, anterior cingulum, and frontal cortex. Velasco's promising open label trials of CM stimulation in 1987 showed an overwhelming reduction in tonic-clonic seizure activity prompting further study into this nucleus as a potential neurostimulation target [94]. Fisher [95] and Valentin [96] then looked at the CM as a target for DBS in generalized and focal epilepsy, with results suggesting greater efficacy in generalized epilepsy. Multiple further studies (uncontrolled) demonstrated safety as well as efficacy with a mean seizure reduction of about 70% [[97], [98], [99], [100]].
The first randomized double-blind controlled trial looking at stimulation in the CM using DBS in patients with LGS did not show a statistically significant difference in the primarily end point of seizure reduction as documented in caregiver diaries at 3 months between the sham group and the stimulation group, though the trend was improvement in the stimulation group [58]. A strength of this paper was the use of ambulatory EEG data to obtain more objective information. For electrographic seizures, 59 % of the stimulation group and none of the controls had a >50 % reduction at the end of the blinded phase. Differences in responder rates may be secondary to electrode placement, as a follow up study showed that DBS at the anterior and inferior CM border, near the ventral lateral nucleus had the best response [101]. Further, better response was associated with increased connectivity from the CM to the premotor and prefrontal cortex, the putamen, and the pontine brainstem on EEG-fMRI [101,102]. However, this is not a settled matter as a meta-analysis of several studies of CM DBS for generalized epilepsy found that a dorsal lead placement correlated with greater seizure control [100]. Retrospective case series suggest that CM is a promising target for therapy in genetic generalized epilepsies using the responsive neurostimulator (RNS) [71,72,75,103]. A small paper of two pediatric patients with LGS and autism demonstrated 50–99 % seizure reduction with RNS in the CM [69]. More recently radiofrequency ablation of the CM has been tried for drug resistant multifocal or generalized epilepsy with reductions of generalized (but not focal aware) seizures [104]. Currently, there are clinical trials underway looking at bilateral CM RNS for treating LGS and genetic generalized epilepsy [59,60].
Pulvinar
One clue as to saliency of the pulvinar in the epilepsy network is the MRI findings during status epilepticus [84,105]. Specifically, there may be unilateral or bilateral pulvinar diffuse hyperintensity with apparent diffusion coefficient (ADC) dropout, that resolves shortly after the status epilepticus ends. In unilateral cases the finding is ipsilateral to the seizure onset zone. Pulvinar findings are often seen in conjunction with cortical MRI findings, and most often in connection with MRI changes in the temporal lobe, though multiple other areas including the opercular, insular and occipital lobe have been reported. This suggests that during the excessive activity of status epilepticus, the reciprocal connections between the pulvinar and the cortex undergo relative hypoxia and/or excitotoxicity which results in the MRI findings [105]. Interestingly, this is not seen in other thalamic nuclei. The anterior and mesial temporal lobe are highly connected to the medial pulvinar nucleus via a pulvinar temporal white matter bundle [106].
In a study of temporal lobe DRE, ictal activity in the pulvinar was seen in 80 % of seizures during SEEG. Rhythmic spikes were seen most frequently in mesial onsets and low frequency fast activity was more likely in neocortical onsets. The seizures without pulvinar activity were restricted to the seizure onset zone [107]. A brain mapping study in 2019 attempted to abort seizures caused by hippocampal stimulation during brain mapping with a pulse in the ipsilateral medial pulvinar. There was no difference in the overall duration of seizures that received pulvinar stimulation and those that did not. However, seizures that received the pulvinar stimulation were less likely to cause a loss of consciousness, did not generalize, and had a shorter tonic phase [108].
In one case series, 3 patients with posterior quadrant epilepsy were implanted with the RNS system [74]. One patient had 2 RNS systems implanted with bilateral medial pulvinar depths, and bilateral occipital leads. The other 2 patients had a unilateral pulvinar depth with an ipsilateral occipital lead. Seizure frequency reduction of 60–95 % was reported. Published work and clinical practice suggest that pulvinar implantation is feasible and may be efficacious, but more work is needed to characterize safety/efficacy profiles.
Vagal nerve stimulation (VNS) and the thalamus
To date, VNS is the most commonly used neurostimulator device in patients with epilepsy. The antiseizure effect of VNS differs from direct intracranial electrostimulation in the thalamus. The literature supports thalamocortical connections as having a key a role in the efficacy of VNS although its exact mechanism of action is unknown [109,110]. Changes in regional cerebral blood flow and neuronal activation within the thalamus follow efficacious VNS placement [111]. The presumed mechanism of action includes vagus nerve afferents projecting to the nucleus tractus solitarius (NTS). The NTS sends information to the thalamic reticular nucleus which indirectly modulates the cortex via thalamocortical relay. Additionally, the intralaminar nuclei which project diffusely over the cerebral cortex receive ascending inputs from the vagus nerve. Thalamocortical synchronization is also likely involved in the antidepressant effects of VNS [112]. The effects of VNS for seizures and depression improve over time suggesting, like in RNS and DBS, a long-term neuromodulator change which is yet poorly understood [113].
Epilepsy as a disorder of consciousness
Being gripped with fear, seeing flickering lights, or being suddenly unable to understand language are some common manifestations of focal seizures. These experiences are not pleasant, but they are not the source of the major morbidity and mortality of epilepsy. When a seizure causes a depression in the level of consciousness, an individual may lose control of a car, fall into the subway tracks, or spill boiling water on themselves. Apart from any actual injury, the fear of this unpredictable loss of control is a source of depression and anxiety, which can deeply affect quality of life [114,115]. When consciousness does not resume after a seizure is over, one's respiratory state may be compromised, this again is an extremely dangerous state, and one of the causes of sudden unexpected death in epilepsy (SUDEP) [52,116].
Consciousness can be divided into content and level. When the level of consciousness is affected, the thalamus has a starring role, in seizures of both focal and generalized onset. fMRI, SPECT, and EEG studies have shown an activation in parts of the thalamus at the moment of decreased awareness [110,117,118].
The brain's natural loss of awareness, sleep, is epitomized by the electrical synchronization and sleep spindles on an EEG. Spindle formation is driven by rhythmic inhibitory action (GABA containing neurons) of the reticular nucleus on thalamocortical projection neurons, and excitatory cortical feedback that leads to spindle oscillations. During spike and wave discharges, prolonged inhibition of the reticular nucleus may result in depressed levels of consciousness [8]. In a fMRI study of people with absence epilepsy, during generalized spike and wave, there is a symmetric and bilateral activation of the thalamus [119,120].
The loss of awareness in focal seizures is postulated to occur simultaneous to the seizure's thalamic involvement. In an SEEG study of a single patient, gamma activity was seen in the dorsomedial nucleus during wakefulness. During focal seizures, this was predictably interrupted at the moment of loss of consciousness and returned when consciousness was restored [121].
Blumenfeld has long advocated for a systematic thought process to understand and target the treatment for loss of consciousness in seizures [52,122]. He posits that in limbic network seizures there is spread to the subcortical arousal system, including the thalamus, which in turn depresses the level of arousal of the cortex. This may be the reason that as the seizure spreads, focal slowing is often seen in brain regions other than the seizure onset zone. There is an ongoing study to test if intralaminar thalamic stimulation at the central lateral nucleus can restore consciousness more quickly in focal seizures of temporal lobe origin [51,123].
Treatment of the thalamus in other neurological conditions
Thalamic stimulation for disorders of consciousness
We can imagine that the same thalamocortical oscillations that underlie sleep and wake fail us in comatose states. Consciousness does not reside in a specific location but depends upon a network involving the thalamus as well as the upper brainstem reticular activating system, basal forebrain, and frontoparietal association cortices.
Thalamic stimulation in comatose, traumatically brain injured (TBI) patients, is as old as thalamic stimulation for epilepsy [[124], [125], [126]]. In the 1940's Moruzzi and Magoun demonstrated a causal link of thalamic or midbrain stimulation and forebrain arousal [127]. This early stimulation involved the CM and midbrain reticular formation. Steriade and Glenn later identified a monosynaptic pathway from the midbrain reticular formation to the rostral-intralaminar region of the thalamus controlling consciousness [30]. Additionally, the dorsomedial, anterior and ventral anterior nuclei have shown reduced fiber density in disorders of consciousness [128].
There was initially a positive outcome in a single case of DBS placed in the anterior intralaminar nuclei and the adjacent area resulting in increased responsiveness in a minimally conscious patient [129]. Other studies have looked at central thalamic nuclei and DBS with mixed results [[129], [130], [131], [132], [133]]. A more recent study examined the physiology in the thalamus in people with disorders of consciousness who recovered following CM-parafascicular complex DBS. In patients with better outcomes there is more activity including higher firing rates and higher multiunit activity with successful intervention [134]. Outside of level of consciousness, stimulation of the central lateral nucleus was shown to improve executive control with chronic TBI in a small study [135].
Movement disorders
The thalamus has been a consistent and effective target in movement disorder surgery since the 1950s [136]. Before the advent of DBS, a thalamotomy in which a small area is lesioned via various heating techniques, was widely used for treatment of tremor and dystonia. While targets differed (ventral lateral thalamus for dystonia and ventral posterior intermediate for tremor), limitations of this procedure included the risk of waning benefit or permanent neurologic deficit, especially when bilateral procedures were performed [137,138]. The complication profile guided efforts to perform reversible modulation, aided by the advent of DBS in the late 1980s. In their seminal work published in 1987, Benabid and Pollak described stimulation of the thalamic ventral intermediate nucleus creating a reduction in tremor severity [139]. Subsequently the term “informational lesion” was coined by Grill and colleagues to describe how high frequency DBS stimulation is essentially equivalent to a destructive lesion by masking the pathological transmission [140].
The patients likely to be selected for ventral intermediate nucleus neuromodulation include those with essential tremor (ET) and some with tremor-dominant Parkinson's disease. Since a large minority (up to 50 %) of ET patients do not have a sufficient response to medication, neuromodulation has been used as an alternative treatment and was approved by the FDA in 1997. While subthalamic targets are also used, within the borders of the thalamus, the accepted area for stimulation is the ventral intermediate nucleus with expected tremor reductions of 60%–90 % [141]. Stimulation may be most beneficial when it intersects with the dentato-rubro-thalamic tract [142] or the lateral cerebellothalamic tracts [143] . Recent years have seen a resurgence in interest in thalamotomy procedures guided by the advent of magnetic resonance guided focused ultrasound (MRgFUS, also known as high intensity focused ultrasound – HIFU). This procedure was tested in a randomized trial and showed benefit in terms of tremor and quality of life in the active group compared to the sham group [144] but limitations (albeit lesser compared to older methods of lesioning) remain in terms of loss of efficacy and permanent side effects. In the case of focal hand dystonia, thalamotomy of the ventral lateral nuclei has had demonstrated efficacy [145]. Thus, despite evolution over time, over 30 years of experience has cemented thalamic interventions as a cornerstone of movement disorders surgery, but further research to refine and perfect the targets and method continues.
Psychiatric disorders
In the psychiatric realm, thalamic stimulation has been used in about half of the reported DBS cases for Tourette's syndrome, a tic disorder characterized by repetitive movements and involuntary vocalizations. Tourette's syndrome is thought to be caused by a hyperdopaminergic state, and treatments are often dopamine receptor antagonists. The preferred targets are the dorsomedial or CM/parafascicular area. Recent data shows median improvement on the Yale Global Tic Severity Scale of greater than 50 %, supporting the utilization of such stimulation in cases where quality of life is severely affected and if self-harm is a possibility with the uncontrollable movements [146,147]. While the mechanism of action is not fully understood, there is the suggestion that stimulation normalizes the cortico-striate-thalamus circuits. PET scan studies suggest a decrease in dopamine during the stimulation-on times, which may be the mechanism of circuit stabilization [148,149]. Connectomic studies suggest that the anti-tic effects are dependent on precisely targeted stimulation of CM connections to motor cortex while stimulation of nearby dorsomedial thalamic connections to limbic regions can trigger depressed mood [150]. DBS is of burgeoning interest in many psychiatric disorders but in other conditions (major depressive disorder, OCD, eating disorders) the main targets of interest are not thalamic.
Conclusion
In the last 50 years, tens of thousands of articles on the thalamus have been written. Neuroscientists like Jones, Sherman and Guillery have devoted much of their careers to understanding its structure and function. Neurologists have turned to the thalamus hoping that intervention in this central location can steer the brain toward neurological health: Anterior nucleus stimulation is a key therapeutic target in both focal and multifocal epilepsy, with evidence demonstrating its ability to reduce seizure frequency [57,67,71,73,77,78]. The ventral intermediate nucleus is primarily targeted for tremor control [[141], [142], [143]]. While the CM has garnered significant interest in epilepsy [58,69,76,95,96,[98], [99], [100],103,104,151,152] and is also being utilized to manage severe tics in Tourette's syndrome [[147], [148], [149], [150]]. The central lateral nucleus, which is strongly connected to the reticular activating system, is currently being investigated for its potential to improve cognition following traumatic brain injury [135] and to prevent loss of consciousness in epilepsy [122,123]. This massive body of work, while impressive, is fractured. Each publication while increasing specificity about a piece, does not necessarily further our understanding of the whole.
The results in neuromodulation for epilepsy are promising with increased seizure reduction over time, reduced risk of SUDEP, and improved quality of life [62,63,78,113]. And yet, seizure freedom with neuromodulation, including in the thalamus, remains elusive. Every epileptologist reading this paper likely has patients for whom no intervention has yet worked. And the literature may reflect a bias toward positive outcomes as retrospective reviews are often only embarked upon when the results are favorable. This gap creates our ongoing need for further prospective controlled investigation. Prospective randomized multicenter studies are ongoing for the use of RNS in LGS and IGE [59,60].
However, thalamic stimulation parameters in use are not standardized and any ongoing trials will not answer all the questions. In the field of epilepsy, efforts have been made to share EEG data across three central databases (EPILEPSIAE, IEEG.org, Kaggle), two of which are publicly accessible. However, most institutions still store data only locally. Further, the neurophysiology data alone is insufficient; clinical phenotyping, imaging, thalamic stimulation location and settings, as well as outcome data are also essential. Therefore, to maximize the value of the shared data and facilitate the potential for novel insights, data sharing solutions need to be multi-modal with newer standards for data curation, sharing and reanalysis. To address these needs, data sharing protocols such as Brain Imaging Data Structure (BIDS) have been developed that provide standardized organizational criteria for multimodal datasets (imaging, electrophysiological, clinical, behavioral, etc.) common in neuroscience [153]. These standards, originally developed to facilitate sharing of MRI data, have been extended for other relevant types of data (MEG, EEG, etc.) and now include specific extensions for intracranial datasets arguably critical to pool across sites given their rare but highly detailed and valuable depiction of human brain data such as the thalamic data here discussed [154,155].
There are other significant barriers to data sharing, such as physician time constraints, financial costs, and the need to de-identify data. Ideally, data streams into BIDS or a similar standard should be automated (e.g. by data mining from the electronic medical record) to minimize costs, but in practice dedicated efforts are necessary to carry out full standardization. This extra overhead for standardization is starting to be identified as a necessary part of clinical and research datastreams, and therefore is starting to be required or supported by funding agencies. For example, recent changes in NIH Data Management and Sharing (DMS) policies, which requires NIH-funded researchers to submit a plan for managing and sharing scientific data, have already started incentivizing data sharing. Other funding agencies are providing earmarked financial support for data sharing and dissemination, which will likely increase adoption of these practices. Another hurdle is that researchers are often incentivized to publish first rather than develop and curate data for sharing. However, researchers like Wagenaar et al. have suggested rewarding data sharing, such as by replacing the traditional H-index, commonly used for academic promotions, with a “sharing index” (S-index) [156]. Finally, the development of resources devoted to neuroscience data sharing such as OpenNeuro, which facilitate access to accessible online repositories as well as guidance on sharing principles such as data versioning, data standardization and de-identification, should facilitate cross-institutional data integration efforts [157].
In today's “information age,” cross-specialty collaboration may be crucial to understanding this complex structure. Beyond epilepsy, all subspecialties involving the thalamus, whether neurologic or psychiatric, should engage in collaborative data collection, encompassing clinical phenotyping, imaging, intervention, and outcome data. As we continue to investigate the thalamus, we hope, like the structure itself, we can integrate our efforts and forge forward with a clearer message to help our patients.
Author Contributions
Lara V Marcuse: Conceptualization, writing-original draft, writing-review and editing, visualization.
Mackenzie Langan: Writing-review and editing, visualization.
Patrick R.Hof: Writing-review and editing.
Fedor Panov: Writing-review and editing.
Ignacio Saez: Writing-review and editing.
Joohi Jimenez-Shahed: Writing-review and editing.
Martijn Figee: Writing-review and editing.
Helen Mayberg: Writing-review and editing.
Ji Yeoun Yoo: Writing-review and editing.
Saadi Ghatan: Writing-review and editing.
Priti Balchandani: Writing-review and editing.
Madeline C Fields: Conceptualization, writing-original draft, writing-review and editing, visualization.
Declaration of Competing Interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Lara V Marcuse reports was provided by Icahn School of Medicine at Mount Sinai. Lara V Marcuse reports a relationship with Icahn School of Medicine at Mount Sinai that includes:. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Cassel J.-C., Pereira de Vasconcelos A. Routes of the thalamus through the history of neuroanatomy. Neurosci Biobehav Rev. 2021;125:442–465. doi: 10.1016/j.neubiorev.2021.03.001. [DOI] [PubMed] [Google Scholar]
- 2.García-Cabezas M.Á., Pérez-Santos I., Cavada C. The epic of the thalamus in anatomical language. Front Neuroanat. 2021;15 doi: 10.3389/fnana.2021.744095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vanderah T., Gould D. 7th Edition. Elsevier; 2016. Nolte's the Human Brain: An Introduction to Its Functional Anatomy. [Google Scholar]
- 4.Mojsilović J., Zečević N. Early development of the human thalamus: golgi and Nissl study. Early Hum Dev. 1991;27:119–144. doi: 10.1016/0378-3782(91)90033-Y. [DOI] [PubMed] [Google Scholar]
- 5.Andersson S.A., Manson J.R. Rhythmic activity in the thalamus of the unanaesthetized decorticate cat. Electroencephalogr Clin Neurophysiol. 1971;31:21–34. doi: 10.1016/0013-4694(71)90286-0. [DOI] [PubMed] [Google Scholar]
- 6.Andersen P., Andersson S.A., Lømo T. Some factors involved in the thalamic control of spontaneous barbiturate spindles. J Physiol. 1967;192:257–281. doi: 10.1113/jphysiol.1967.sp008299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Beenhakker M.P., Huguenard J.R. Neurons that fire together also conspire together: is normal sleep circuitry hijacked to generate epilepsy? Neuron. 2009;62:612–632. doi: 10.1016/j.neuron.2009.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Steriade M. Sleep, epilepsy and thalamic reticular inhibitory neurons. Trends Neurosci. 2005;28:317–324. doi: 10.1016/j.tins.2005.03.007. [DOI] [PubMed] [Google Scholar]
- 9.Steriade M., McCormick D.A., Sejnowski T.J. Thalamocortical oscillations in the sleeping and aroused brain. Science. 1993;262:679–685. doi: 10.1126/science.8235588. [DOI] [PubMed] [Google Scholar]
- 10.Bickford M. In: The cerebral cortex and thalamus. Bickford M., editor. Oxford University Press; New York: 2023. Thalamus and prethalamus cell types and their interconnections; pp. 13–21. [DOI] [Google Scholar]
- 11.Halassa M.M., Sherman S.M. Thalamocortical circuit motifs: a general framework. Neuron. 2019;103:762–770. doi: 10.1016/j.neuron.2019.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sherman S.M. Tonic and burst firing: dual modes of thalamocortical relay. Trends Neurosci. 2001;24:122–126. doi: 10.1016/S0166-2236(00)01714-8. [DOI] [PubMed] [Google Scholar]
- 13.Sherman S.M., Guillery R.W. On the actions that one nerve cell can have on another: distinguishing “drivers” from “modulators. Proc Natl Acad Sci USA. 1998;95:7121–7126. doi: 10.1073/pnas.95.12.7121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee C.C., Sherman S.M. Synaptic properties of thalamic and intracortical inputs to layer 4 of the first- and higher-order cortical areas in the auditory and somatosensory systems. J Neurophysiol. 2008;100:317–326. doi: 10.1152/jn.90391.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sherman S., Guillery R. Academic Press; 2001. Exploring the Thalamus. [Google Scholar]
- 16.Rikhye R.V., Wimmer R.D., Halassa M.M. Toward an integrative theory of thalamic function. Annu Rev Neurosci. 2018;41:163–183. doi: 10.1146/annurev-neuro-080317-062144. [DOI] [PubMed] [Google Scholar]
- 17.Rikhye R.V., Gilra A., Halassa M.M. Thalamic regulation of switching between cortical representations enables cognitive flexibility. Nat Neurosci. 2018;21:1753–1763. doi: 10.1038/s41593-018-0269-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Michael G.A., Gálvez-García G. Salience-based progression of visual attention. Behav Brain Res. 2011;224:87–99. doi: 10.1016/j.bbr.2011.05.024. [DOI] [PubMed] [Google Scholar]
- 19.Robinson D.L., Petersen S.E. The pulvinar and visual salience. Trends Neurosci. 1992;15:127–132. doi: 10.1016/0166-2236(92)90354-B. [DOI] [PubMed] [Google Scholar]
- 20.Mai J.K., Majtanik M. Toward a common terminology for the thalamus. Front Neuroanat. 2019;12 doi: 10.3389/fnana.2018.00114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gallay M.N., Jeanmonod D., Liu J., Morel A. Human pallidothalamic and cerebellothalamic tracts: anatomical basis for functional stereotactic neurosurgery. Brain Struct Funct. 2008;212:443–463. doi: 10.1007/s00429-007-0170-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Krauth A., Blanc R., Poveda A., Jeanmonod D., Morel A., Székely G. A mean three-dimensional atlas of the human thalamus: generation from multiple histological data. Neuroimage. 2010;49:2053–2062. doi: 10.1016/j.neuroimage.2009.10.042. [DOI] [PubMed] [Google Scholar]
- 23.Morel A. CRC Press; 2007. Stereotactic Atlas of the Human Thalamus and Basal Ganglia. [DOI] [Google Scholar]
- 24.Hassler R. Anatomy of the Normal Human Thalamus. Elsevier; Amsterdam: 1971. Attempt at standardization of nomenclature. [Google Scholar]
- 25.Child N.D., Benarroch E.E. Anterior nucleus of the thalamus. Neurology. 2013;81:1869–1876. doi: 10.1212/01.wnl.0000436078.95856.56. [DOI] [PubMed] [Google Scholar]
- 26.Papez J.W. A proposed mechanism of emotion. Arch Neurol Psychiatr. 1937;38:725. doi: 10.1001/archneurpsyc.1937.02260220069003. [DOI] [Google Scholar]
- 27.Szalárdy O., Simor P., Ujma P.P., Jordán Z., Halász L., Erőss L., et al. Temporal association between sleep spindles and ripples in the human anterior and mediodorsal thalamus. Eur J Neurosci. 2024;59:641–661. doi: 10.1111/ejn.16240. [DOI] [PubMed] [Google Scholar]
- 28.Ohye C., Shibazaki T., Hirai T., Wada H., Hirato M., Kawashima Y. Further physiological observations on the ventralis intermedius neurons in the human thalamus. J Neurophysiol. 1989;61:488–500. doi: 10.1152/jn.1989.61.3.488. [DOI] [PubMed] [Google Scholar]
- 29.Jones E.G. The thalamus of primates. 1998. p. 1–298. [DOI]
- 30.Steriade M., Glenn L.L. Neocortical and caudate projections of intralaminar thalamic neurons and their synaptic excitation from midbrain reticular core. J Neurophysiol. 1982;48:352–371. doi: 10.1152/jn.1982.48.2.352. [DOI] [PubMed] [Google Scholar]
- 31.Giguere M., Goldman-Rakic P.S. Mediodorsal nucleus: areal, laminar, and tangential distribution of afferents and efferents in the frontal lobe of rhesus monkeys. J Comp Neurol. 1988;277:195–213. doi: 10.1002/cne.902770204. [DOI] [PubMed] [Google Scholar]
- 32.Grodd W., Kumar V.J., Schüz A., Lindig T., Scheffler K. The anterior and medial thalamic nuclei and the human limbic system: tracing the structural connectivity using diffusion-weighted imaging. Sci Rep. 2020;10 doi: 10.1038/s41598-020-67770-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Anastasiades P.G., Collins D.P., Carter A.G. Mediodorsal and ventromedial thalamus engage distinct L1 circuits in the prefrontal cortex. Neuron. 2021;109:314–330.e4. doi: 10.1016/j.neuron.2020.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Plum F., Posner J. The diagnosis of stupor and coma. Contemp Neurol Ser. 1972;10:1–286. [PubMed] [Google Scholar]
- 35.Sadikot A.F., Rymar V.V. The primate centromedian-parafascicular complex: anatomical organization with a note on neuromodulation. Brain Res Bull. 2009;78:122–130. doi: 10.1016/j.brainresbull.2008.09.016. [DOI] [PubMed] [Google Scholar]
- 36.Cover K.K., Mathur B.N. Rostral intralaminar thalamus engagement in cognition and behavior. Front Behav Neurosci. 2021;15 doi: 10.3389/fnbeh.2021.652764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sherman S.M. Thalamus plays a central role in ongoing cortical functioning. Nat Neurosci. 2016;19:533–541. doi: 10.1038/nn.4269. [DOI] [PubMed] [Google Scholar]
- 38.Segobin S., Laniepce A., Ritz L., Lannuzel C., Boudehent C., Cabé N., et al. Dissociating thalamic alterations in alcohol use disorder defines specificity of Korsakoff's syndrome. Brain. 2019;142:1458–1470. doi: 10.1093/brain/awz056. [DOI] [PubMed] [Google Scholar]
- 39.Tuladhar A.M., de Leeuw F.-E. Thalamus: a key player in alcohol use disorder and Korsakoff's syndrome. Brain. 2019;142:1170–1172. doi: 10.1093/brain/awz096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Montagna P. Fatal familial insomnia: a model disease in sleep physiopathology. Sleep Med Rev. 2005;9:339–353. doi: 10.1016/j.smrv.2005.02.001. [DOI] [PubMed] [Google Scholar]
- 41.Schmahmann J.D. Vascular syndromes of the thalamus. Stroke. 2003;34:2264–2278. doi: 10.1161/01.STR.0000087786.38997.9E. [DOI] [PubMed] [Google Scholar]
- 42.Gibbs S.A., Nobili L., Halász P. Interictal epileptiform discharges in sleep and the role of the thalamus in encephalopathy related to status epilepticus during slow sleep. Epileptic Disord. 2019;21 doi: 10.1684/epd.2019.1058. [DOI] [PubMed] [Google Scholar]
- 43.Sánchez Fernández I., Takeoka M., Tas E., Peters J.M., Prabhu S.P., Stannard K.M., et al. Early thalamic lesions in patients with sleep-potentiated epileptiform activity. Neurology. 2012;78:1721–1727. doi: 10.1212/WNL.0b013e3182582ff8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Losito E., Battaglia D., Chieffo D., Raponi M., Ranalli D., Contaldo I., et al. Sleep-potentiated epileptiform activity in early thalamic injuries: study in a large series (60 cases) Epilepsy Res. 2015;109:90–99. doi: 10.1016/j.eplepsyres.2014.10.015. [DOI] [PubMed] [Google Scholar]
- 45.Carvalho D., Mendonça C., Carvalho J., Martins A., Leal A. High incidence of early thalamic lesions in the Continuous Spike-Wave related with slow Sleep (CSWS) Epilepsy Behav. 2023;138 doi: 10.1016/j.yebeh.2022.109031. [DOI] [PubMed] [Google Scholar]
- 46.Sánchez Fernández I., Peters J.M., Akhondi-Asl A., Klehm J., Warfield S.K., Loddenkemper T. Reduced thalamic volume in patients with electrical status epilepticus in sleep. Epilepsy Res. 2017;130:74–80. doi: 10.1016/j.eplepsyres.2017.01.010. [DOI] [PubMed] [Google Scholar]
- 47.Öztürk Z., Karalok Z.S., Güneş A. Reduced thalamic volume is strongly associated with electrical status epilepticus in sleep. Acta Neurol Belg. 2021;121:211–217. doi: 10.1007/s13760-019-01202-7. [DOI] [PubMed] [Google Scholar]
- 48.Kilic H., Yilmaz K., Asgarova P., Kizilkilic O., Hatay G.H., Ozturk-Isik E., et al. Electrical status epilepticus in sleep: the role of thalamus in etiopathogenesis. Seizure. 2021;93:44–50. doi: 10.1016/j.seizure.2021.10.010. [DOI] [PubMed] [Google Scholar]
- 49.Guye M., Régis J., Tamura M., Wendling F., McGonigal A., Chauvel P., et al. The role of corticothalamic coupling in human temporal lobe epilepsy. Brain. 2006;129:1917–1928. doi: 10.1093/brain/awl151. [DOI] [PubMed] [Google Scholar]
- 50.Piper R.J., Richardson R.M., Worrell G., Carmichael D.W., Baldeweg T., Litt B., et al. Towards network-guided neuromodulation for epilepsy. Brain. 2022;145:3347–3362. doi: 10.1093/brain/awac234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Blumenfeld H. Arousal and consciousness in focal seizures. Epilepsy Curr. 2021;21:353–359. doi: 10.1177/15357597211029507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Blumenfeld H. Impaired consciousness in epilepsy. Lancet Neurol. 2012;11:814–826. doi: 10.1016/S1474-4422(12)70188-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mirski M.A., Fisher R.S. Electrical stimulation of the mammillary nuclei increases seizure threshold to pentylenetetrazol in rats. Epilepsia. 1994;35:1309–1316. doi: 10.1111/j.1528-1157.1994.tb01803.x. [DOI] [PubMed] [Google Scholar]
- 54.Jasper H., Naquet R., King E.E. Thalamocortical recruiting responses in sensory receiving areas in the cat. Electroencephalogr Clin Neurophysiol. 1955;7:99–114. doi: 10.1016/0013-4694(55)90063-8. [DOI] [PubMed] [Google Scholar]
- 55.Manjunatha R.T., Vakilna Y.S., Chaitanya G., Alamoudi O., Ilyas A., Pati S. Advancing the frontiers of thalamic neuromodulation: a review of emerging targets and paradigms. Epilepsy Res. 2023;196 doi: 10.1016/j.eplepsyres.2023.107219. [DOI] [PubMed] [Google Scholar]
- 56.Herrman H., Egge A., Konglund A.E., Ramm-Pettersen J., Dietrichs E., Taubøll E. Anterior thalamic deep brain stimulation in refractory epilepsy: a randomized, double-blinded study. Acta Neurol Scand. 2018 doi: 10.1111/ane.13047. ane. [DOI] [PubMed] [Google Scholar]
- 57.Fisher R., Salanova V., Witt T., Worth R., Henry T., Gross R., et al. Electrical stimulation of the anterior nucleus of thalamus for treatment of refractory epilepsy. Epilepsia. 2010;51:899–908. doi: 10.1111/j.1528-1167.2010.02536.x. [DOI] [PubMed] [Google Scholar]
- 58.Dalic L.J., Warren A.E.L., Bulluss K.J., Thevathasan W., Roten A., Churilov L., et al. DBS of thalamic centromedian nucleus for Lennox–Gastaut syndrome (ESTEL trial) Ann Neurol. 2022;91:253–267. doi: 10.1002/ana.26280. [DOI] [PubMed] [Google Scholar]
- 59.NeuroPace . clinicaltrials.gov; 2023. RNS® system responsive thalamic stimulation for primary generalized seizures (NAUTILUS) study (clinical trial registration No. NCT05147571) [Google Scholar]
- 60.NeuroPace . clinicaltrials.gov; 2023. RNS system Feasibility study of thalamocortical brain-responsive neurostimulation for the treatment of Lennox-Gastaut syndrome (clinical trial registration No. NCT05339126) [Google Scholar]
- 61.Fan J.M., Baud M.O., Rao V.R. Characterization of multidien rhythms in patients with epilepsy (S36.007) Neurology. 2019;92 doi: 10.1212/WNL.92.15_supplement.S36.007. [DOI] [Google Scholar]
- 62.Bergey G.K., Morrell M.J., Mizrahi E.M., Goldman A., King-Stephens D., Nair D., et al. Long-term treatment with responsive brain stimulation in adults with refractory partial seizures. Neurology. 2015;84:810–817. doi: 10.1212/WNL.0000000000001280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nair D.R., Laxer K.D., Weber P.B., Murro A.M., Park Y.D., Barkley G.L., et al. Nine-year prospective efficacy and safety of brain-responsive neurostimulation for focal epilepsy. Neurology. 2020;95 doi: 10.1212/WNL.0000000000010154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lesser R.P., Kim S.H., Beyderman L., Miglioretti D.L., Webber W.R.S., Bare M., et al. Brief bursts of pulse stimulation terminate afterdischarges caused by cortical stimulation. Neurology. 1999;53:2073. doi: 10.1212/WNL.53.9.2073. 2073. [DOI] [PubMed] [Google Scholar]
- 65.Kokkinos V., Sisterson N.D., Wozny T.A., Richardson R.M. Association of closed-loop brain stimulation neurophysiological features with seizure control among patients with focal epilepsy. JAMA Neurol. 2019;76:800. doi: 10.1001/jamaneurol.2019.0658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rao V.R., Rolston J.D. Unearthing the mechanisms of responsive neurostimulation for epilepsy. Commun Med. 2023;3:166. doi: 10.1038/s43856-023-00401-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Herlopian A., Cash S.S., Eskandar E.M., Jennings T., Cole A.J. Responsive neurostimulation targeting anterior thalamic nucleus in generalized epilepsy. Ann Clin Transl Neurol. 2019;6:2104–2109. doi: 10.1002/acn3.50858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Panov F., Ganaha S., Haskell J., Fields M., La Vega-Talbott M., Wolf S., et al. Safety of responsive neurostimulation in pediatric patients with medically refractory epilepsy. J Neurosurg Pediatr. 2020;26:525–532. doi: 10.3171/2020.5.PEDS20118. [DOI] [PubMed] [Google Scholar]
- 69.Kwon C.-S., Schupper A.J., Fields M.C., Marcuse L.V., La Vega-Talbott M., Panov F., et al. Centromedian thalamic responsive neurostimulation for Lennox-Gastaut epilepsy and autism. Ann Clin Transl Neurol. 2020;7:2035–2040. doi: 10.1002/acn3.51173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Roa J.A., Abramova M., Fields M., Vega-Talbott M La, Yoo J., Marcuse L., et al. Responsive neurostimulation of the thalamus for the treatment of refractory epilepsy. Front Hum Neurosci. 2022;16 doi: 10.3389/fnhum.2022.926337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Fields M.C., Eka O., Schreckinger C., Dugan P., Asaad W.F., Blum A.S., et al. A multicenter retrospective study of patients treated in the thalamus with responsive neurostimulation. Front Neurol. 2023;14 doi: 10.3389/fneur.2023.1202631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kokkinos V., Urban A., Sisterson N.D., Li N., Corson D., Richardson R.M. Responsive neurostimulation of the thalamus improves seizure control in idiopathic generalized epilepsy: a case report. Neurosurgery. 2020;87:E578–E583. doi: 10.1093/neuros/nyaa001. [DOI] [PubMed] [Google Scholar]
- 73.Elder C., Friedman D., Devinsky O., Doyle W., Dugan P. Responsive neurostimulation targeting the anterior nucleus of the thalamus in 3 patients with treatment-resistant multifocal epilepsy. Epilepsia Open. 2019;4:187–192. doi: 10.1002/epi4.12300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Burdette D., Mirro E.A., Lawrence M., Patra S.E. Brain-responsive corticothalamic stimulation in the pulvinar nucleus for the treatment of regional neocortical epilepsy: a case series. Epilepsia Open. 2021;6:611–617. doi: 10.1002/epi4.12524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Welch W.P., Hect J.L., Abel T.J. Case report: responsive neurostimulation of the centromedian thalamic nucleus for the detection and treatment of seizures in pediatric primary generalized epilepsy. Front Neurol. 2021;12 doi: 10.3389/fneur.2021.656585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Nanda P., Sisterson N., Walton A., Chu C.J., Cash S.S., Moura L.M.V.R., et al. Centromedian region thalamic responsive neurostimulation mitigates idiopathic generalized and multifocal epilepsy with focal to bilateral tonic–clonic seizures. Epilepsia. 2024;65:2626–2640. doi: 10.1111/epi.18070. [DOI] [PubMed] [Google Scholar]
- 77.Osorio I., Giftakis J., Stypulkowski P., Tonder L. Anatomical connectivity and efficacy of electro-therapy for seizure control: a SANTE's single-center regression analyses. Epilepsy Behav. 2021;115 doi: 10.1016/j.yebeh.2020.107709. [DOI] [PubMed] [Google Scholar]
- 78.Salanova V., Sperling M.R., Gross R.E., Irwin C.P., Vollhaber J.A., Giftakis J.E., et al. The SANTÉ study at 10 years of follow-up: effectiveness, safety, and sudden unexpected death in epilepsy. Epilepsia. 2021;62:1306–1317. doi: 10.1111/epi.16895. [DOI] [PubMed] [Google Scholar]
- 79.Gadot R., Korst G., Shofty B., Gavvala J.R., Sheth S.A. Thalamic stereoelectroencephalography in epilepsy surgery: a scoping literature review. J Neurosurg. 2022;137:1210–1225. doi: 10.3171/2022.1.JNS212613. [DOI] [PubMed] [Google Scholar]
- 80.Evangelista E., Bénar C., Bonini F., Carron R., Colombet B., Régis J., et al. Does the thalamo-cortical synchrony play a role in seizure termination? Front Neurol. 2015;6 doi: 10.3389/fneur.2015.00192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pizzo F., Roehri N., Giusiano B., Lagarde S., Carron R., Scavarda D., et al. The ictal signature of thalamus and basal ganglia in focal epilepsy. Neurology. 2021;96 doi: 10.1212/WNL.0000000000011003. [DOI] [PubMed] [Google Scholar]
- 82.Wu T.Q., Kaboodvand N., McGinn R.J., Veit M., Davey Z., Datta A., et al. Multisite thalamic recordings to characterize seizure propagation in the human brain. Brain. 2023;146:2792–2802. doi: 10.1093/brain/awad121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bernabei J.M., Litt B., Cajigas I. Thalamic stereo-EEG in epilepsy surgery: where do we stand? Brain. 2023;146:2663–2665. doi: 10.1093/brain/awad178. [DOI] [PubMed] [Google Scholar]
- 84.Di Bonaventura C., Bonini F., Fattouch J., Mari F., Petrucci S., Carnì M., et al. Diffusion-weighted magnetic resonance imaging in patients with partial status epilepticus. Epilepsia. 2009;50(Suppl 1):45–52. doi: 10.1111/j.1528-1167.2008.01970.x. [DOI] [PubMed] [Google Scholar]
- 85.Langan M.T., Verma G., Delman B.N., Marcuse L.V., Fields M.C., Feldman R., et al. Segmentation and quantification of venous structures and perivascular spaces in the thalamus in epilepsy using 7 Tesla MRI. Brain Multiphys. 2024;6 doi: 10.1016/j.brain.2023.100089. [DOI] [Google Scholar]
- 86.Haast R.A.M., Testud B., Makhalova J., Dary H., Cabane A., Le Troter A., et al. Multi-scale structural alterations of the thalamus and basal ganglia in focal epilepsy using 7T MRI. Hum Brain Mapp. 2023;44:4754–4771. doi: 10.1002/hbm.26414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Penfield W., Jasper H. first ed. 1954. Epilepsy and the Functional Anatomy of the Human Brain. [Google Scholar]
- 88.Spiegel E.A., Wycis H.T., Reyes V. Diencephalic mechanisms in petit mal epilepsy. Electroencephalogr Clin Neurophysiol. 1951;3:473–475. doi: 10.1016/0013-4694(51)90035-1. [DOI] [PubMed] [Google Scholar]
- 89.Tyvaert L., Chassagnon S., Sadikot A., LeVan P., Dubeau F., Gotman J. Thalamic nuclei activity in idiopathic generalized epilepsy. Neurology. 2009;73:2018–2022. doi: 10.1212/WNL.0b013e3181c55d02. [DOI] [PubMed] [Google Scholar]
- 90.Peltola J., Colon A.J., Pimentel J., Coenen V.A., Gil-Nagel A., Gonçalves Ferreira A., et al. Deep brain stimulation of the anterior nucleus of the thalamus in drug-resistant epilepsy in the MORE multicenter patient registry. Neurology. 2023;100 doi: 10.1212/WNL.0000000000206887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Sperling M.R., Barshow S., Nei M., Asadi-Pooya A.A. A reappraisal of mortality after epilepsy surgery. Neurology. 2016;86:1938–1944. doi: 10.1212/WNL.0000000000002700. [DOI] [PubMed] [Google Scholar]
- 92.Lehtimäki K., Möttönen T., Järventausta K., Katisko J., Tähtinen T., Haapasalo J., et al. Outcome based definition of the anterior thalamic deep brain stimulation target in refractory epilepsy. Brain Stimul. 2016;9:268–275. doi: 10.1016/j.brs.2015.09.014. [DOI] [PubMed] [Google Scholar]
- 93.Gross R.E., Fisher R.S., Sperling M.R., Giftakis J.E., Stypulkowski P.H. Analysis of deep brain stimulation lead targeting in the stimulation of anterior nucleus of the thalamus for epilepsy clinical trial. Neurosurgery. 2021;89:406–412. doi: 10.1093/neuros/nyab186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Velasco F., Velasco M., Ogarrio C., Fanghanel G. Electrical stimulation of the centromedian thalamic nucleus in the treatment of convulsive seizures: a preliminary report. Epilepsia. 1987;28:421–430. doi: 10.1111/j.1528-1157.1987.tb03668.x. [DOI] [PubMed] [Google Scholar]
- 95.Fisher R.S., Uematsu S., Krauss G.L., Cysyk B.J., McPherson R., Lesser R.P., et al. Placebo-controlled pilot study of centromedian thalamic stimulation in treatment of intractable seizures. Epilepsia. 1992;33:841–851. doi: 10.1111/j.1528-1157.1992.tb02192.x. [DOI] [PubMed] [Google Scholar]
- 96.Valentín A., García Navarrete E., Chelvarajah R., Torres C., Navas M., Vico L., et al. Deep brain stimulation of the centromedian thalamic nucleus for the treatment of generalized and frontal epilepsies. Epilepsia. 2013;54:1823–1833. doi: 10.1111/epi.12352. [DOI] [PubMed] [Google Scholar]
- 97.Velasco F., Velasco M., Jiménez F., Velasco A.L., Brito F., Rise M., et al. Predictors in the treatment of difficult-to-control seizures by electrical stimulation of the centromedian thalamic nucleus. Neurosurgery. 2000;47:295–305. doi: 10.1097/00006123-200008000-00007. [DOI] [PubMed] [Google Scholar]
- 98.Son B., Shon Y.M., Choi J., Kim J., Ha S., Kim S.-H., et al. Clinical outcome of patients with deep brain stimulation of the centromedian thalamic nucleus for refractory epilepsy and location of the active contacts. Stereotact Funct Neurosurg. 2016;94:187–197. doi: 10.1159/000446611. [DOI] [PubMed] [Google Scholar]
- 99.Cukiert A., Cukiert C.M., Burattini J.A., Mariani P.P. Seizure outcome during bilateral, continuous, thalamic centromedian nuclei deep brain stimulation in patients with generalized epilepsy: a prospective, open-label study. Seizure. 2020;81:304–309. doi: 10.1016/j.seizure.2020.08.028. [DOI] [PubMed] [Google Scholar]
- 100.Ilyas A., Snyder K.M., Pati S., Tandon N. Optimally targeting the centromedian nucleus of the thalamus for generalized epilepsy: a meta-analysis. Epilepsy Res. 2022;184 doi: 10.1016/j.eplepsyres.2022.106954. [DOI] [PubMed] [Google Scholar]
- 101.Warren A.E.L., Dalic L.J., Bulluss K.J., BappSci A.R., Thevathasan W., Archer J.S. The optimal target and connectivity for Deep brain stimulation in Lennox–Gastaut syndrome. Ann Neurol. 2022;92:61–74. doi: 10.1002/ana.26368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Warren A.E.L., Harvey A.S., Vogrin S.J., Bailey C., Davidson A., Jackson G.D., et al. The epileptic network of Lennox-Gastaut syndrome. Neurology. 2019;93 doi: 10.1212/WNL.0000000000007775. [DOI] [PubMed] [Google Scholar]
- 103.Zillgitt A.J., Haykal M.A., Chehab A., Staudt M.D. Centromedian thalamic neuromodulation for the treatment of idiopathic generalized epilepsy. Front Hum Neurosci. 2022;16 doi: 10.3389/fnhum.2022.907716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Aguado-Carrillo G., Velasco A.L., Saucedo-Alvarado P.E., Cuellar-Herrera M., Trejo-Martínez D., Navarro-Olvera J.L., et al. Radiofrequency ablation of the centromedian thalamic nucleus in the treatment of drug-resistant epilepsy. Epilepsy Behav. 2021;114 doi: 10.1016/j.yebeh.2020.107560. [DOI] [PubMed] [Google Scholar]
- 105.Ohe Y., Hayashi T., Deguchi I., Fukuoka T., Horiuchi Y., Maruyama H., et al. MRI abnormality of the pulvinar in patients with status epilepticus. J Neuroradiol. 2014;41:220–226. doi: 10.1016/j.neurad.2013.09.003. [DOI] [PubMed] [Google Scholar]
- 106.Kalamatianos T., Mavrovounis G., Skouras P., Pandis D., Fountas K., Stranjalis G. Medial pulvinar stimulation in temporal lobe epilepsy: a literature review and a hypothesis based on neuroanatomical findings. Cureus. 2023 doi: 10.7759/cureus.35772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Rosenberg D.S., Mauguière F., Demarquay G., Ryvlin P., Isnard J., Fischer C., et al. Involvement of medial pulvinar thalamic nucleus in human temporal lobe seizures. Epilepsia. 2006;47:98–107. doi: 10.1111/j.1528-1167.2006.00375.x. [DOI] [PubMed] [Google Scholar]
- 108.Filipescu C., Lagarde S., Lambert I., Pizzo F., Trébuchon A., McGonigal A., et al. The effect of medial pulvinar stimulation on temporal lobe seizures. Epilepsia. 2019;60 doi: 10.1111/epi.14677. [DOI] [PubMed] [Google Scholar]
- 109.Liu W.-C., Mosier K., Kalnin A.J., Marks D. BOLD fMRI activation induced by vagus nerve stimulation in seizure patients. J Neurol Neurosurg Psychiatry. 2003;74:811–813. doi: 10.1136/jnnp.74.6.811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Carney P.W., Masterton R.A.J., Harvey A.S., Scheffer I.E., Berkovic S.F., Jackson G.D. The core network in absence epilepsy. Differences in cortical and thalamic BOLD response. Neurology. 2010;75:904–911. doi: 10.1212/WNL.0b013e3181f11c06. [DOI] [PubMed] [Google Scholar]
- 111.Henry T.R., Votaw J.R., Pennell P.B., Epstein C.M., Bakay R.A., Faber T.L., et al. Acute blood flow changes and efficacy of vagus nerve stimulation in partial epilepsy. Neurology. 1999;52:1166–1173. doi: 10.1212/wnl.52.6.1166. [DOI] [PubMed] [Google Scholar]
- 112.Kamel L.Y., Xiong W., Gott B.M., Kumar A., Conway C.R. Vagus nerve stimulation: an update on a novel treatment for treatment-resistant depression. J Neurol Sci. 2022;434 doi: 10.1016/j.jns.2022.120171. [DOI] [PubMed] [Google Scholar]
- 113.Morris G.L., Mueller W.M. Long-term treatment with vagus nerve stimulation in patients with refractory epilepsy. Neurology. 1999;53:1731. doi: 10.1212/WNL.53.8.1731. 1731. [DOI] [PubMed] [Google Scholar]
- 114.Vickrey B.G., Berg A.T., Sperling M.R., Shinnar S., Langfitt J.T., Bazil C.W., et al. Relationships between seizure severity and health-related quality of life in refractory localization-related epilepsy. Epilepsia. 2000;41:760–764. doi: 10.1111/j.1528-1157.2000.tb00239.x. [DOI] [PubMed] [Google Scholar]
- 115.Sperling M.R. The consequences of uncontrolled epilepsy. CNS Spectr. 2004;9(98–101):106–109. doi: 10.1017/s1092852900008464. [DOI] [PubMed] [Google Scholar]
- 116.Friedman D. Sudden unexpected death in epilepsy. Curr Opin Neurol. 2022;35:181–188. doi: 10.1097/WCO.0000000000001034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Berman R., Negishi M., Vestal M., Spann M., Chung M.H., Bai X., et al. Simultaneous EEG, fMRI, and behavior in typical childhood absence seizures. Epilepsia. 2010;51:2011–2022. doi: 10.1111/j.1528-1167.2010.02652.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Moeller F., LeVan P., Muhle H., Stephani U., Dubeau F., Siniatchkin M., et al. Absence seizures: individual patterns revealed by EEG-fMRI. Epilepsia. 2010;51:2000–2010. doi: 10.1111/j.1528-1167.2010.02698.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Gotman J., Grova C., Bagshaw A., Kobayashi E., Aghakhani Y., Dubeau F. Generalized epileptic discharges show thalamocortical activation and suspension of the default state of the brain. Proc Natl Acad Sci U S A. 2005;102:15236–15240. doi: 10.1073/pnas.0504935102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kostopoulos G.K. Involvement of the thalamocortical system in epileptic loss of consciousness. Epilepsia. 2001;42:13–19. doi: 10.1046/j.1528-1157.2001.042suppl.3013.x. [DOI] [PubMed] [Google Scholar]
- 121.Leeman-Markowski B.A., Smart O.L., Faught R.E., Gross R.E., Meador K.J. Cessation of gamma activity in the dorsomedial nucleus associated with loss of consciousness during focal seizures. Epilepsy Behav. 2015;51:215–220. doi: 10.1016/j.yebeh.2015.07.027. [DOI] [PubMed] [Google Scholar]
- 122.Gummadavelli A., Kundishora A.J., Willie J.T., Andrews J.P., Gerrard J.L., Spencer D.D., et al. Neurostimulation to improve level of consciousness in patients with epilepsy. Neurosurg Focus. 2015;38:E10. doi: 10.3171/2015.3.FOCUS1535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Blumenfeld H. clinicaltrials.gov; 2022. Thalamic Stimulation to Prevent Impaired Consciousness in Epilepsy (Clinical Trial Registration No. NCT04897776) [Google Scholar]
- 124.McLardy T., Ervin F., Mark V., Scoville W., Sweet W. Attempted inset-electrodes-arousal from traumatic coma: neuropathological findings. Trans Am Neurol Assoc. 1968;93:25–30. [PubMed] [Google Scholar]
- 125.Hassler R., Ore G.D., Dieckmann G., Bricolo A., Dolce G. Behavioural and EEG arousal induced by stimulation of unspecific projection systems in a patient with post-traumatic apallic syndrome. Electroencephalogr Clin Neurophysiol. 1969;27:306–310. doi: 10.1016/0013-4694(69)90060-1. [DOI] [PubMed] [Google Scholar]
- 126.Sturm V., Kühner A., Schmitt H.P., Assmus H., Stock G. Chronic electrical stimulation of the thalamic unspecific activating system in a patient with coma due to midbrain and upper brain stem infarction. Acta Neurochir. 1979;47:235–244. doi: 10.1007/BF01406406. [DOI] [PubMed] [Google Scholar]
- 127.Moruzzi G., Magoun H.W. Brain stem reticular formation and activation of the EEG. Electroencephalogr Clin Neurophysiol. 1949;1:455–473. [PubMed] [Google Scholar]
- 128.Yu Y., Zheng W., Tan X., Li X., Zhang X., Gao J., et al. Microstructural profiles of thalamus and thalamocortical connectivity in patients with disorder of consciousness. J Neurosci Res. 2021;99:3261–3273. doi: 10.1002/jnr.24921. [DOI] [PubMed] [Google Scholar]
- 129.Schiff N.D., Giacino J.T., Kalmar K., Victor J.D., Baker K., Gerber M., et al. Behavioural improvements with thalamic stimulation after severe traumatic brain injury. Nature. 2007;448:600–603. doi: 10.1038/nature06041. [DOI] [PubMed] [Google Scholar]
- 130.Cohadon F., Richer E. Deep cerebral stimulation in patients with post-traumatic vegetative state. 25 cases. Neurochirurgie. 1993;39:281–292. [PubMed] [Google Scholar]
- 131.Yamamoto T., Katayama Y. Deep brain stimulation therapy for the vegetative state. Neuropsychol Rehabil. 2005;15:406–413. doi: 10.1080/09602010443000353. [DOI] [PubMed] [Google Scholar]
- 132.Magrassi L., Maggioni G., Pistarini C., Di Perri C., Bastianello S., Zippo A.G., et al. Results of a prospective study (CATS) on the effects of thalamic stimulation in minimally conscious and vegetative state patients. J Neurosurg. 2016;125:972–981. doi: 10.3171/2015.7.JNS15700. [DOI] [PubMed] [Google Scholar]
- 133.Chudy D., Deletis V., Almahariq F., Marčinković P., Škrlin J., Paradžik V. Deep brain stimulation for the early treatment of the minimally conscious state and vegetative state: experience in 14 patients. J Neurosurg. 2018;128:1189–1198. doi: 10.3171/2016.10.JNS161071. [DOI] [PubMed] [Google Scholar]
- 134.He J., Zhang H., Dang Y., Zhuang Y., Ge Q., Yang Y., et al. Electrophysiological characteristics of CM-pf in diagnosis and outcome of patients with disorders of consciousness. Brain Stimul. 2023;16:1522–1532. doi: 10.1016/j.brs.2023.09.021. [DOI] [PubMed] [Google Scholar]
- 135.Schiff N.D., Giacino J.T., Butson C.R., Choi E.Y., Baker J.L., O'Sullivan K.P., et al. Thalamic deep brain stimulation in traumatic brain injury: a phase 1, randomized feasibility study. Nat Med. 2023;29:3162–3174. doi: 10.1038/s41591-023-02638-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Burchiel K. Thalamotomy for movement disorders. Neurosurg Clin. 1995;6:55–71. [PubMed] [Google Scholar]
- 137.Surgical Treatment of Parkinson’s Disease and Other Movement Disorders. Current Clinical Neurology. n.d.
- 138.Witjas-Slucki T. Surgical treatments for tremors. Rev Neurol (Paris) 2018;174:615–620. doi: 10.1016/j.neurol.2018.07.004. [DOI] [PubMed] [Google Scholar]
- 139.Benabid A.L., Pollak P., Louveau A., Henry S., de Rougemont J. Combined (thalamotomy and stimulation) stereotactic surgery of the VIM thalamic nucleus for bilateral Parkinson disease. Appl Neurophysiol. 1987;50:344–346. doi: 10.1159/000100803. [DOI] [PubMed] [Google Scholar]
- 140.Grill W.M., Snyder A.N., Miocinovic S. Deep brain stimulation creates an informational lesion of the stimulated nucleus. Neuroreport. 2004;15:1137–1140. doi: 10.1097/00001756-200405190-00011. [DOI] [PubMed] [Google Scholar]
- 141.Zesiewicz T.A., Elble R., Louis E.D., Hauser R.A., Sullivan K.L., Dewey R.B., et al. Practice parameter: therapies for essential tremor [RETIRED] Neurology. 2005;64:2008–2020. doi: 10.1212/01.WNL.0000163769.28552.CD. [DOI] [PubMed] [Google Scholar]
- 142.Morrison M.A., Lee A.T., Martin A.J., Dietiker C., Brown E.G., Wang D.D. DBS targeting for essential tremor using intersectional dentato-rubro-thalamic tractography and direct proton density visualization of the VIM: technical note on 2 cases. J Neurosurg. 2021;135:806–814. doi: 10.3171/2020.8.JNS201378. [DOI] [PubMed] [Google Scholar]
- 143.Brinda A., Slopsema J.P., Butler R.D., Ikramuddin S., Beall T., Guo W., et al. Lateral cerebellothalamic tract activation underlies DBS therapy for essential tremor. Brain Stimul. 2023;16:445–455. doi: 10.1016/j.brs.2023.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Elias W.J. A trial of focused ultrasound thalamotomy for essential tremor. N Engl J Med. 2016;375:2201–2203. doi: 10.1056/NEJMc1612210. [DOI] [PubMed] [Google Scholar]
- 145.Shimizu T., Maruo T., Miura S., Kishima H., Ushio Y., Goto S. Stereotactic lesioning of the thalamic vo nucleus for the treatment of writer's cramp (focal hand dystonia) Front Neurol. 2018;9 doi: 10.3389/fneur.2018.01008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Lee D.J., Lozano C.S., Dallapiazza R.F., Lozano A.M. Current and future directions of deep brain stimulation for neurological and psychiatric disorders. J Neurosurg. 2019;131:333–342. doi: 10.3171/2019.4.JNS181761. [DOI] [PubMed] [Google Scholar]
- 147.Baldermann J.C., Schüller T., Huys D., Becker I., Timmermann L., Jessen F., et al. Deep brain stimulation for tourette-syndrome: a systematic review and meta-analysis. Brain Stimul. 2016;9:296–304. doi: 10.1016/j.brs.2015.11.005. [DOI] [PubMed] [Google Scholar]
- 148.Kuhn J., Janouschek H., Raptis M., Rex S., Lenartz D., Neuner I., et al. In vivo evidence of deep brain stimulation-induced dopaminergic modulation in Tourette's syndrome. Biol Psychiatry. 2012;71:e11–e13. doi: 10.1016/j.biopsych.2011.09.035. [DOI] [PubMed] [Google Scholar]
- 149.Vernaleken I., Kuhn J., Lenartz D., Raptis M., Huff W., Janouschek H., et al. Bithalamical deep brain stimulation in tourette syndrome is associated with reduction in dopaminergic transmission. Biol Psychiatry. 2009;66:e15–e17. doi: 10.1016/j.biopsych.2009.06.025. [DOI] [PubMed] [Google Scholar]
- 150.Morishita T., Sakai Y., Iida H., Yoshimura S., Ishii A., Fujioka S., et al. Neuroanatomical considerations for optimizing thalamic deep brain stimulation in Tourette syndrome. J Neurosurg. 2022;136:231–241. doi: 10.3171/2021.2.JNS204026. [DOI] [PubMed] [Google Scholar]
- 151.Velasco F, Velasco AL, Velasco M, Jiménez F, Carrillo-Ruiz JD, Castro G. Deep brain stimulation for treatment of the epilepsies: the centromedian thalamic target. Operative Neuromodulation, Vienna: Springer Vienna; n.d., p. 337–342. 10.1007/978-3-211-33081-4_38. [DOI] [PubMed]
- 152.Park S., Permezel F., Agashe S., Osman G., Simpson H.D., Miller K.J., et al. Centromedian thalamic deep brain stimulation for idiopathic generalized epilepsy: connectivity and target optimization. Epilepsia. 2024;65 doi: 10.1111/epi.18122. [DOI] [PubMed] [Google Scholar]
- 153.Gorgolewski K.J., Auer T., Calhoun V.D., Craddock R.C., Das S., Duff E.P., et al. The brain imaging data structure, a format for organizing and describing outputs of neuroimaging experiments. Sci Data. 2016;3 doi: 10.1038/sdata.2016.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Holdgraf C., Appelhoff S., Bickel S., Bouchard K., D'Ambrosio S., David O., et al. iEEG-BIDS, extending the Brain Imaging Data Structure specification to human intracranial electrophysiology. Sci Data. 2019;6:102. doi: 10.1038/s41597-019-0105-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Pernet C.R., Appelhoff S., Gorgolewski K.J., Flandin G., Phillips C., Delorme A., et al. EEG-BIDS, an extension to the brain imaging data structure for electroencephalography. Sci Data. 2019;6:103. doi: 10.1038/s41597-019-0104-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Wagenaar J.B., Worrell G.A., Ives Z., Matthias D., Litt B., Schulze-Bonhage A. Collaborating and sharing data in epilepsy research. J Clin Neurophysiol. 2015;32:235–239. doi: 10.1097/WNP.0000000000000159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Markiewicz C.J., Gorgolewski K.J., Feingold F., Blair R., Halchenko Y.O., Miller E., et al. The OpenNeuro resource for sharing of neuroscience data. Elife. 2021;10 doi: 10.7554/eLife.71774. [DOI] [PMC free article] [PubMed] [Google Scholar]