Abstract
Purpose
We studied the regulatory association of Porphyromonas gingivalis (PG) and cataracts.
Methods
PCR and FISH assays were used for detecting PG 16s ribosomal RNA genome, Immunofluorescence was for expression of RpgA in anterior capsular epithelium and fibrosis markers in anterior subcapsular cataract (ASC) model. Flow cytometry was for reactive oxygen species and apoptosis. RNA deep sequencing is for differential gene expression analysis.
Results
PG's 16s ribosomal RNA gene is positively in 43.3% (101/233 cases) of aqueous humor (AH) samples of patients with cataracts, which differs from 4.7% (6/127) of PG-positive AH in patients with glaucoma. Diabetic and high myopia cataracts increase PG-positive AH compared with age-related cataracts. No PG is observed in AH of congenital cataracts. PG is positive in 82% to 94% of the cataractous anterior capsule tissues from high myopia and age-related, congenital, and diabetic cataracts. The PG-positive cells in the cataractous anterior capsular epithelium are CD68+/CD14+ macrophages, but not anterior epithelial cells. In rat ASC models, PG injected via the tail vein or PG-carried bone marrow monocytes can migrate into the equatorial lens epithelium in form of PG-positive macrophages, which promote ASC progression with upregulation of collagen, fibronectin and α smooth muscle actin (α-SMA) expression, and increase 8-OHdG levels and α-SMA expression in the surrounding lens epithelial cells. Kyoto Encyclopedia of Genes and Genomes and Gene Ontology analysis of the RNA sequencing dataset of ASC tissues shows that signaling pathways related to epithelial–mesenchymal transition, oxidative stress, and cell death are up-regulated in PG + ASC compared with that in ASC alone. Co-culture of supernatants of Raw264.7/PG+ cells with rat primary lens epithelial cells increases the 8-OHdG levels, mitochondrial fission, apoptosis, and expression of α-SMA.
Conclusions
Chronic infection with PG can access the lens epithelium via macrophages during stress conditions, which promotes cataract development by possibly elevating oxidative stress, apoptosis, and epithelial–mesenchymal transition in lens tissues. PG infection is a novel a risk factor for cataract development.
Keywords: porphyromonas gingivalis, lens epithelium, macrophage, cataract, aqueous humor
Cataracts are a leading cause of blindness in the world. Depending on age, cataracts are classified as either congenital cataracts or age-related cataracts (ARCs). The prevalence of cataracts increases with age, from 3.9% at 55 to 64 years to 92.6% at age 80 years and older.1,2 Congenital cataracts impact approximately 1 to 15 out of 10,000 newborn children.2 For congenital cataracts, genetic variations, viral infection, or nutrient deficiency are the common risk factors, whereas for ARC, aging is the predominant virulent factor. In addition, UVB light, smoking, alcohol use, glaucoma, high myopia, and systemic diseases such as diabetes, hyperglycemia, and chronic inflammation are common risk factors in triggering ARC development.3 Prevalent studies suggest that the chronic infection of pathogens, such as rubella, hepatitis B virus, or hepatitis C virus, may be associated with both ARC and congenital cataracts.2,4 The chronic infection of Porphyromonas gingivalis (PG), an oral periodontitis-associated gram-negative anerobic bacteria, is associated with the diverse age-associated diseases.5–7 However, the regulatory association of PG infection with cataracts remains unclear.
Chronic infection with PG is associated with the diverse age-associated diseases, such as atherosclerosis, neurodegenerative diseases (e.g., Alzheimer's and Huntingdon's disease), diabetes, cancers, cardiac diseases, kidney diseases, and hypertension.8 PG can invade and survive in the gingival epithelial cells, fibroblast cells, monocytes, and macrophages, through which the bacteria are transmitted to distant tissues.9–11 PG invades host cells through its fimbriae's interaction with surface receptors such as intercellular cell adhesion molecule-1 and integrin α5.12 Intracellular PG can survive in host cells for a long period of time by activating the expression of prosurvival signals, such as, IAP, BCL-2, and increasing the host cell's secretion of nucleoside diphosphate kinase to suppress adenosine triphosphate–dependent apoptosis.13,14 PG infection causes chronic diseases through pathogenic virulent factors, such as gingipains (RgpA, RgpB, and Kgp), fimbriae, lactin-like adhesins, capsular polysaccharide, lipopolysaccharide, hemagglutinins, and hemolysins.15 About 96% of neural biopsy samples from Alzheimer's disease patients are positive for RgpA, and 91% of Alzheimer's disease samples are positive for Kgp.16 Prophylaxis of PG infection or inhibition of gingipains by inhibitors can decrease the incidence of age-related diseases, such as rheumatoid arthritis, atherosclerosis, and Alzheimer's diseases.17,18
The association of PG infection with age-related eye degenerative diseases (e.g., cataract, glaucoma, and AMD) remains unclear. Prevalent studies indicate that serum anti-PG antibody is correlated positively with a high risk of early diabetic retinopathy.19 The expression of the PG 16s-ribosomal RNA (rRNA) gene was detected in the retina of mice with an AMD + Parkinson's disease model, accompanied by the increased angiogenesis and oxidative stress, and high expression of the proinflammatory factors TNF-α, TGF-β1, and IL-1β.20 AMD is associated prevalently with tooth alveolar bone loss, a dominant syndrome of PG-induced periodontitis.21,22 In vitro studies suggest that PG can infect and invade RPE cells, inducing the expression of cytokines such as IL-1, IL-6, and cell apoptosis.23 Recently, prevalent studies suggest that periodontitis patients have a high incidence of cataracts.24 The immune-privileged lens is found to recruit macrophages under stress conditions.25 Monocytes or macrophages are dominant transmitters of PG. Altogether, we propose that there may be a correlation between cataracts and PG infection.
In this study, we analyzed PG's 16s rRNA gene in the AH samples of 233 patients with cataracts and 127 patients with glaucoma, as well as in anterior capsular tissues from 118 cataracts patients, including 45 cases of ARC, 32 cases of diabetic cataract (DC), 17 cases of high myopia cataract (HMC), and 24 cases of congenital cataract. The results showed that PG infection is closely associated with cataracts. More interestingly, we found that PG-positive cells in the anterior capsular epithelium are CD14/CD68-positive macrophage cells, but not lens epithelial cells. Using a rat traumatic ASC model, we found that PG infection increased ASC progression and PG-positive macrophages were observed in the anterior epithelium near the equator and region close to ASC. In contrast, PG could invade and survive intracellularly in Raw264.7 cells in vitro. The supernatants of PG-infected Raw264.7 cells could induce the mitochondria fragmentation, oxidative stress, apoptosis and epithelial–mesenchymal transition (EMT) of rat primary lens epithelial cells. These results suggest that macrophage-hosted PG is a risk factor for cataract development.
Materials and Methods
Cell Culture
The mouse macrophage Raw264.7 cells were bought from ATCC. The cells were maintained and passaged in 10% fetal bovine serum/Dulbecco's Modified Eagle Medium supplemented with antibiotics in a 37°C incubator with 5% CO2.
Primary Rat Lens Epithelial Cell Culture In Vitro and Rat Anterior Subcapsular Cataracts (ASCs) Model
All procedures of animal manipulation followed the principles of the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research, and the protocols were approved by the Ethics Committee of Henan University. For in vitro culture of primary rat lens epithelial cells, Wistar male rats aged 4 to 6 weeks were euthanized using CO2 followed by cervical dislocation. Lenses were isolated under the microscope. The lens capsules were separated from fiber tissues and incubated in 0.12% trypsin at 37°C for 10 minutes. After this, the residual epithelial cells were collected and cultured in M199 medium containing 10% fetal bovine serum for 7 days. For the ASC model, six male and female Wistar rats aged 6 to 8 weeks were recruited to make ASCs. Briefly, the rats were anesthetized using isoflurane. The eyes were dilated with a 5 mg/mL topicamide compound. The injury at the center of the anterior lens capsule was made using a 26G hypodermic needle. The injury was monitored by measuring the stab depth to one-third of the length of the needle. The rats were recovered and treated with antibiotic eye drops for 6 days. The injury cataracts were observed under a slit-lamp microscope. After that, the cataractous lenses were removed, and capsular bags were separated from fiber tissues for whole mount staining or subjected to immunoblotting and quantitative PCR (qPCR) analysis.
Collection of Aqueous Humor (AH) and Anterior Capsular Samples
AH samples and anterior capsular tissues were collected from patients who underwent cataract surgery or glaucoma surgery in Kaifeng Eye Disease Hospital. The AH (100 µL/patient) was collected with a needle before surgery. Anterior capsular tissue was obtained during the procedure. A consent document regarding the research purpose and potential risks and benefits of the research was provided to each patient before sample collection. All procedures regarding to sample collection, transportation, and storage followed the guidance of International Ethical Guidance from the Council for International Organizations of Medical Sciences and World Medical Association's Declaration of Helsinki. The procedures for the manipulation of HA or anterior capsular tissues have been proved by the Ethics Committee of Kaifeng Eye Disease Hospital and Henan University.
PG Culture and Infection to Raw264.7 In Vitro
PG (ATCC33277) was maintained on blood agar plate (Being Land Bridae Technology Co., LTD, Beijing, China) or 41.7 mg/mL GAM Broth, Modified (Nissui, Japan) in a hypoxic incubator with 10% CO2, 10% H2, 80% N2. For cell infection, PG was grown to an optical density at 600 nm of 0.6, followed by three washes in PBS. After this, bacteria resuspended in PBS were added to the cultured Raw264.7 cells at a multiplicity of infection of 1:200 under anoxic conditions for 2 hours, followed by incubation with 100 µg/mL metronidazole for 1 hour to kill the extracellular live bacteria. Cells were washed with medium three times to remove the dead bacteria and cultured in complete medium in a 37°C incubator with 5% CO2 for up to 72 hours. Intracellular PG was detected by immunofluorescence staining.
Rat Tail Vein Injection of PG or PG-Positive Bone Marrow Monocytes (BMMs)
For tail vein injection of PG, PG bacteria at an optical density at 600 nm of 0.6 were collected, washed three times in PBS, and resuspended in PBS at a concentration of 5 × 106/mL. Twelve male and female Wistar rats aged 6 to 8 weeks were grouped into two groups of six rats each. They were injected via the tail vein with 1 × 106 PG or PBS. After 48 hours post injection, ASC surgery was performed on the rat lenses.
For tail vein injection of PG-positive BMMs, two rats aged 6 to 8 weeks were euthanized using CO2 followed by cervical dislocation. The BMMs were isolated from the bone marrow. After washing with PBS, the monocytes were infected with PG at a multiplicity of infection of 1:200 for 2 hours in vitro. The PG-invaded monocytes were validated using immunofluorescence staining. After that, the PG-positive monocytes were injected via the tail vein into six Wistar rats; another six rats that were injected with monocytes only were used as controls. One day after injection, the rats underwent ASC surgery. The development of ASC was analyzed by slit-lamp microscope, hematoxylin and eosin staining, and immunofluorescence staining.
Nested PCR, Quantitative Real-time PCR, and TaqMan qPCR
Nested PCR was used to test the genomic DNA of the PG's 16s rRNA in the AH and the anterior epithelium. Briefly, anterior capsular epithelial tissues were lysed in 500 µL lysis buffer (100 mM Tris [pH 8.0], 500 mM EDTA [pH 8.0], 20 mM NaCl, 10% SDS) containing protease K (2.5 mg/mL final concentration). Tissue DNA was extracted using DNA extraction kits following the manufacturer's protocol (Solarbio, Beijing, China). The DNA samples from tissues or AH were subjected to PCR amplification with the following primer pairs: 5′GGTGCGTAGGTTGTTCGGTA′ (forward) and 5′ TATTACCCTAGTGCGCCCCT3′ (reverse). The PCR products were analyzed on agarose gel and confirmed by DNA sequencing.
Quantitative real-time PCR was used to test messenger RNA expression levels. Briefly, total RNA was extracted with RNAiso reagent following the manufacturer's protocol (Takara Bio, Beijing, China). One microgram of total RNA was used to synthesize cDNA. Equal amounts of cDNA were mixed with Faststart Universal SYBR Green Master Mix (Roche, San Francisco, CA, USA). qRT-PCR was performed using an ABI 7500 system (Applied Biosystems, Foster City, CA, USA). The primers used in this study are listed in Table 1. The data are shown as mean ± SD (n = 4). The unpaired two-tailed t test was used for statistical analysis. A P value of less than 0.05 was considered statistically significant. The primers used for qRT-PCR are listed in Table 1.
Table 1.
Primer Sequences Used in This Study
| Gene | Species | Direction | Sequence |
|---|---|---|---|
| Nadk2 | Rat | FW | GAAACAACTGTGGGGAAGCC |
| RV | AGCCTTTCAATGCAAGCAGC | ||
| Boc | Rat | FW | TGAAGCATCGTAAGCAGGTCA |
| RV | GTTGTAGGCTGCCATCTCCA | ||
| Bcl6 | Rat | FW | AAAGCCCGCAGCTCAAATTC |
| RV | TGTTGTGTTTGCCCAATGCC | ||
| Hmox1 | Rat | FW | TGCACATCCGTGCAGAGAAT |
| RV | AGGAGGCCATCACCAGCTTA | ||
| Lgals3 | Rat | FW | GAGGAGCACTAACCAGGAAAATGG |
| RV | CATCCTCGAGGGTTTGGGTT | ||
| CD24 | Rat | FW | TGCTCCTACCCACGCAGATTTA |
| RV | GTTGGATTTGGGGCAGCAGA | ||
| GAPDH | Rat | FW | AAGGTCGGTGTGAACGGATTT |
| RV | GTTGAACTTGCCGTGGGTAG |
TaqMan qPCR was used to determine the genomic copy of PG in anterior capsular tissues. Briefly, a standard curve was set up using a serial dilution DNA of PG bacteria. The DNA from cataractous tissues was subjected to TaqMan qPCR following the protocol. Multiplex PCR reactions were performed in a total reaction volume of 20 µL, including 2 µL template DNA, 0.5 µL of each primer (16s-TaqManF1: 5′ACCCTTTAAACCCAATAAATC′ and 16s-TAqManR1: 5′ACGAGTATTGCATTGAATG3′), 1 µL of probe (16s-Probe: FAM-CGCTCGCATCCTCCGTATTAC-TAMRA), 10 µL AceQ Unicersal U+Probe Master Mix V2, and 6 µL of ddH2O. Each pair of primers and probes was optimized to a final concentration of 0.2 pM. A real-time PCR reaction was conducted as follows: cycles of 50°C for 2 minutes, 95°C for 10 minutes, followed by 40 cycles of 95°C for 15 seconds, and 65°C for 1 minute.
RNA Sequencing
For analysis of gene expression profile, next-generation RNA deep sequencing and data analysis were performed by Novo Zhiyuan (Beijing, China). The samples of rat lens that were treated with ASC, PG injection alone, and PG injection + ASC were recruited to RNA sequencing analysis. Wild-type (WT) rats were used as controls. One microgram of total RNA (one anterior capsule tissue per sample per rat) was used for RNA sample preparation. The libraries were prepared using the NEBNext Ultra Directional RNA Library Prep Kit for Illumina (San Diego, CA, USA), and quality were evaluated on an Agilent Bioanalyzer 2100 system (Agilent, Santa Clara, CA, USA). The clustering of indexed coded samples was analyzed on the cBot Cluster Generation System using the TruSeq PE Cluster Kit (V3-CBOTt-HS, Illumina). After being fascicled, the library preparation was sequenced on the Illumina Novaseq platform and a 150-bp double-ended read was generated. High-quality clean data were generated using Trimmomatic (v0.30) and the rat reference genome (GRCz11, Ensembl, Cambridge, UK) was compared using Hisat2 (v2.0.5) software. The number of reads for each gene was counted using featurecots (v1.5.0-p3). DESeq2R software package (1.20.0) was used to analyze the differential expression of the WT group, lens ASC group, PG infection group, and PG plus ASC group. P value correction was performed by Benjamini and Hochberg's method. The P value after correction was 0.05, and the multiple change was set as 2 as the threshold for significant differential expression. Gene Ontology enrichment and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment of differentially expressed genes in the WT, lens injury, PG infection, PG infection, and lens injury groups were analyzed using ClusterProfiler R package.
Luminex Cytokine Immunoassays
Serum-free supernatants of Raw264.7 or PG-infected Raw264.7 cells cultured for 24 h and 48 h were collected and subjected to a multicytokine assay (LabEx, Shanghai, China). Cytokine levels were evaluated with a Luminex 200 system (Luminex, Austin, TX, USA) using a panel of 31 mouse cytokines (LX-MultiD-31) in accordance with the recommendations of the manufacturer.
Immunoblot and Immunofluorescent Staining
For immunoblotting, equal amounts of proteins were separated in SDS-PAGE gel and transferred onto polyvinylidene difluoride membranes. After 1 hour of blocking in 5% skim milk/PBS/0.1% Tween 20, the membranes were incubated with primary antibodies at 4°C overnight. The membranes were washed with PBS/0.1% Tween-20 four times, and then incubated with secondary antibodies conjugated with horseradish peroxidase for 1 hour. The protein bands were developed in ECL buffer followed by photographing using chemiluminescence (CLINXChemiScope, Shanghai, China).
For immunofluorescence staining, the cells or capsular bags in whole mount were fixed in 4% polyformaldehyde/PBS for 20 minutes followed by permeabilization in 0.1% Triton x-100 for 8 minutes. The cells were then incubated with 2% BSA/PBS block buffer for 1 hour followed by incubation with antibodies against RgpA or PG bacteria overnight at 4°C. The cells were washed in PBST buffer three times and then incubated in buffer containing a secondary antibody conjugated with fluorescein for 2 hours. After this, the cells were stained with fluorescein-conjugated phalloidin to label the F-actin. The cell nuclei were stained with DAPI. The fluorescent signals were photographed with a confocal microscope (Arial, Japan).
FISH
The capsular bag was fixed in 4% polyformaldehyde/PBS for 20 minutes and then permeabilized in 0.1% triton X-100/PBS for 8 minutes. After incubating the pouch with 2% BSA/PBS block buffer for 1 hour, the capsular bag was hybridized with 5 nM FISH probe P.g-488 (PG 16S rRNA 5′-CAATACTCGTATCGCCCGTTATTC-3′ labeled with Alexafluor-488 (green fluorescent) at the 5′-end) that was diluted in hybridization buffer (0.9M NaCl, 20 mM Tris/HCl, pH 7.3, and 0.01 % SDS, 20% formamide) for 4 hours in the dark at 46°C. The capsular bags were washed with PBST. The nuclei were stained with DAPI at 1:1000 dilution for 30 minutes at room temperature. The signals were photographed under confocal microscopy (Arial, Japan).
Flow Cytometer and Spectrophotometry
For the reactive oxygen species (ROS) assay, cells were incubated with 10 µM 2, 7-dichlorodihydrofluoren diacetate at 37°C for 30 minutes, after which the cell suspension was washed three times with PBS and centrifuged at 800 rpm for 5 minutes. Finally, intracellular ROS levels were measured using flow cytometry (BD FACS Calibur, BD Biosciences, Franklin Lakes, NJ, USA). For measuring ROS in ASC tissues, the lenses were isolated from rats that were left untreated or tail vein injection of PG, lens injury or PG injection + lens injury. The lens tissues were homogenized in homogenate buffer and incubated with ROS probe (dichlorodihydrofluoren diacetate, BestBio, Tainan, Taiwan) at 37°C for 30 minutes. ROS levels were detected with a microplate spectrophotometer at excitation light of 488 nm and emission light of 530 nm. At the same time, the protein concentration of tissue lysate was detected. The ROS level as normalized by the protein concentration was counted for ROS relative levels.
For the apoptosis assay, cells were stained with annexin V-APC/7-AAD double staining solution and apoptosis rate was detected by flow cytometry (BD FACS Calibur). APC+/7-AAD (early apoptosis) and APC+/7-AAD+ (late apoptosis) cells are considered apoptotic.
Statistical Analysis
Data are presented as mean ± SD using GraphPad Prism 8 software. Differences among the three group were analyzed by multiple comparisons using one-way ANOVA. Differences among the two groups were analyzed by an independent two-tailed t test. The χ2 test was used to analyze the frequency of PG in cataract and patients with glaucoma. A P value of less than 0.05 was considered statistically significant.
Results
Identification of PG 16sRNA Genomic DNA in the AH and the Anterior Capsular Tissues of Patients With Cataracts
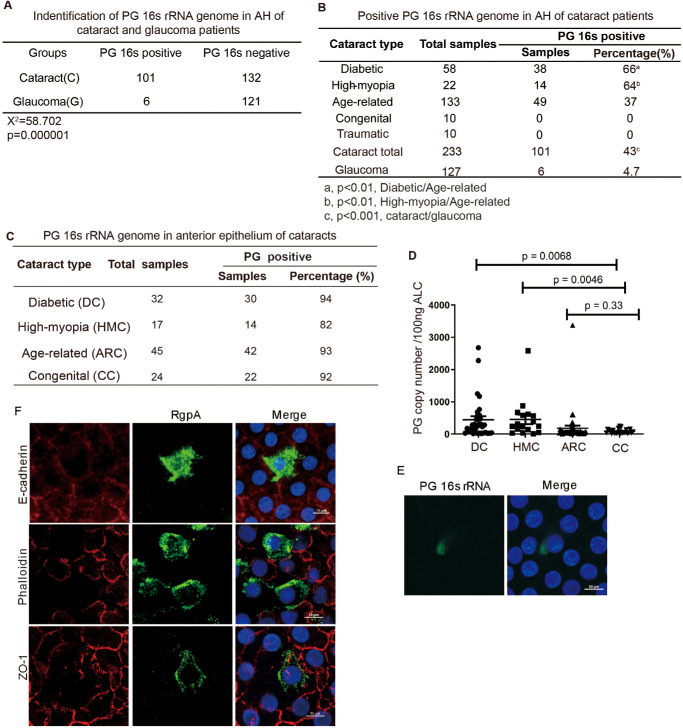
PCR amplification of 16s rRNA genomic DNA has been widely used to test PG infection in the divers tissues including neuron, fibroblast, monocyte, and endothelial cells.26 To determine the association of PG with cataracts, we recruited 233 cataractous AH samples (including 133 ARCs, 22 HMCs, 58 DCs, 10 congenital cataracts, and 10 traumatic cataracts) and 127 AH samples of eyes with glaucoma that were used as a control group (Supplementary Figs. S1A and S1B). Patients with glaucoma with cataracts or AMD were excluded from the study. The recruited 133 patients with ARC were between 60 and 75 years old, which matched with the ages of patients with glaucoma. The PCR results showed that 43.3% (101/233 cases) of overall cataractous AH samples were positive for 16s rRNA genomic DNA, which differs from 4.7% (6/127) of the glaucoma AHs that were PG positive (Fig. 1A). The PCR products were verified by DNA sequencing (Supplementary Figs. S1C, S1D, and S1E). Statistical analysis of the correlation showed that PG infection is closely correlated to cataract incidence (χ2 = 58.702; P = 0.00001). Among cataractous AH samples, PG was positive in 66% (38/58) of the AH samples from DC and 64% (14/22) of the HMC samples, which is significantly higher than in 37% (49/133) of ARC patients (P < 0.02) (Fig. 1B). No PG-positive AH was discernable in the 10 cases of congenital cataracts or traumatic cataracts (Fig. 1B). These results showed that PG infection was closely associated with the incidence of ARCs. High myopia and diabetes significantly elevate PG positive in cataract AH.
Figure 1.
PG infection is closely correlated to cataract incidence. (A) PCR identification of the PG 16s rRNA genomic DNA in the AH of cataracts and glaucoma. The χ2 test was used for statistical analysis. (B) PCR test of the PG 16srRNA genome in AH of different types of cataracts including diabetic, high myopia, age-related, congenital, and traumatic cataracts. χ2 test was used for statistical analysis. (A) Comparison between DC and ARC. (B) Comparison between ARC and HMC. (c) Comparison between cataracts (total) and glaucoma. (C) Percentage of the PGs 16s rRNA genome positive in PCR in the anterior capsular tissues of DCs, HMCs, ARC, and congenital cataracts. (D) Copy number of the PG genome in the anterior capsule of patients with cataracts was detected by TaqMan qPCR. The data shown represent mean ± SD, one-way ANOVA was used for statistical analysis, n = 118. P < 0.01 was considered statistical significance. (E) FISH assay to detect the PGs 16s rRNA (green) in the anterior capsule epithelium of patients with cataracts; cells nuclear was stained by DAPI. Scale bar, 10 µm. (F) Immunofluorescence staining in whole mount for the expression RgpA and E-cadherin; RgpA and ZO-1; RgpA and phalloidin in the anterior capsular epithelium of cataracts. Nuclear was stained with DAPI. Scale bar, 10 µm.
To determine whether PG subsists in cataractous tissues, we analyzed 118 individual anterior capsules including 32 DCs, 17 HMCs, 45 ARCs, and 24 congenital cataracts. The PCR results showed that PG/16s rRNA genomic DNA was positive in the capsular anterior tissues of 94% of DC (30/32), 82% of HMC (14/17), 93% of ARC (42/45), and 92% of congenital cataract (22/24) (Fig. 1C). The PCR products were validated by DNA sequencing (Supplementary Figs. S1F and S1G). The genomic copy number of PG bacteria in the anterior capsules was further determined with TaqMan qPCR. The results showed that PG genomic DNA copies in the anterior capsular tissues of DC and HMC were significantly more than that of ARC and congenital cataract (P = 0.0068; P = 0.0046). There was no significant difference of PG numbers between ARC and congenital cataract (Fig. 1D). These results confirmed again that PG exists in cataractous tissues, and diabetic milieus and high myopia increase the PG infection in lens tissues. The existence of PG in anterior capsular tissues was further confirmed by FISH assay in the anterior capsular epithelium of 10 patients with ARC (Fig. 1E). PG is a periodontitis pathogen. In addition to PG, Actinobacillus actinomycetemcomitans and Treponema denticola are predominant periodontitis-associated pathogens.27 PCR results showed that these two bacteria were not detected (Supplementary Fig. S1H), which excluded the association of A. Actinomycetemcomitans and T. denticola with cataracts. Altogether, the results indicated that periodontopathic bacteria PG, but not the other two bacteria, are closely associated with cataracts.
PG expresses gingipain enzymes (RgpA, RgpB, and Kgp) intracellularly to induce protein denature or innate immune response in host cells. RgpA expression in host cells is a sign of PG infection intracellularly. We then performed whole mount immunofluorescence with antibodies against RgpA to the anterior capsular tissues of 20 patients with ARC. The results showed that there were few RgpA-positive cells observed in the cataractous lens anterior epithelium of all 20 samples (Figs. 1F, Supplementary S1J). Interestingly, those RgpA-positive cells were not stained by the antibodies against E-cadherin and ZO-1 or stained weakly by phalloidin (Fig. 1F and Supplementary Video S1), the markers of lens epithelial cells. These results suggested that PG expressed RgpA in host cells, and PG-positive host cells were not lens anterior epithelial cells. PG-positive cells in the anterior epithelium of ARC were further confirmed by using a home-made rabbit polyclonal antibodies against PG (Supplementary Figs. S1I, S1J). Altogether, these results indicated that there were RgpA-positive cells in the cataractous anterior lens epithelium, and these RgpA-positive cells were other types of cells, rather than lens epithelial cells.
PG Colocalizes With the Macrophages in Cataractous Anterior Capsular Epithelium
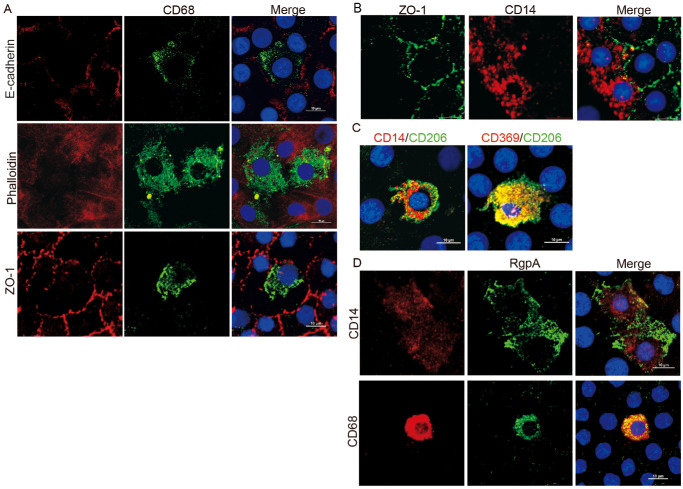
Macrophages and dendritic cells are the common means for PG transmission. The lens is originally considered as an immune-privileged organ.28 However, recent reports have indicated that macrophages can migrate along the ciliary zonule and enter into in the lens epithelium during lens damage and development.29 Macrophages were visible in the lens epithelium of the mice with N-cadherin deficiency.30 Therefore, we proposed that macrophages, being PG hosts, might be recruited to the lens epithelium in stress conditions such as cataracts. To prove this, we stained the macrophages in the anterior capsular epithelium from 13 patients with ARC in whole mount by using antibodies against CD68 for macrophages or phalloidin or antibodies of ZO-1 and E-cadherin for lens epithelial cells. The results showed that there were CD68+ cells that are not colocalized with antibodies against ZO-1 and E-cadherin in the cataractous lens epithelium (Fig. 2A and Supplementary Video S2). The macrophages in the cataractous anterior epithelium were further validated by double staining CD14 (a marker of macrophages and monocytes) and ZO-1, CD14 vs. CD206 (a marker of macrophages and immature dendritic cells), and CD206 vs. CD369 (a marker for dendritic cells, monocytes, and macrophages) (Figs. 2B, 2C). No colocalization between CD68 and ZO-1, CD68, and phalloidin or CD14 and ZO-1 were observed (Figs. 2A, 2B). These results suggested that there were monocyte-like cells or macrophages in the tested cataractous anterior epithelium.
Figure 2.
PG and macrophage are colocalized in the anterior capsular tissue of patients with cataracts. (A–C) Immunofluorescence staining in whole mount the anterior capsular cataractous epithelium with antibodies against CD68 and E-cadherin, CD68, and ZO-1 or CD68/phalloidin (A), CD14 vs. ZO-1 (B) or CD14 vs. CD206, CD369 vs. CD206 (C). The nucleus was stained with DAPI. Scale bar, 10 µm. (D) The immunofluorescence staining in whole mount the anterior capsular cataractous epithelium with antibodies against RgpA and CD14 (top) or RpgA and CD68 (bottom). Scale bar, 10 µm. con, control.
Next, we determined whether the macrophages are the host of PG in the cataractous anterior capsular epithelium. The anterior capsular tissues from 10 patients with ARC were stained in whole mount with the antibodies against RgpA and CD68 or RgpA and CD14. The results showed that the RgpA-positive cells were completely overlapped with CD14+ or CD68+ cells (Fig. 2D). Altogether, we postulated that the macrophages or monocyte-like cells host PG in the cataractous anterior epithelium.
PG-Infected Macrophages Are Associated With Oxidative Stress in Lens In Vivo or Oxidative Stress, Apoptosis, and EMT of Lens Epithelial Cells In Vitro
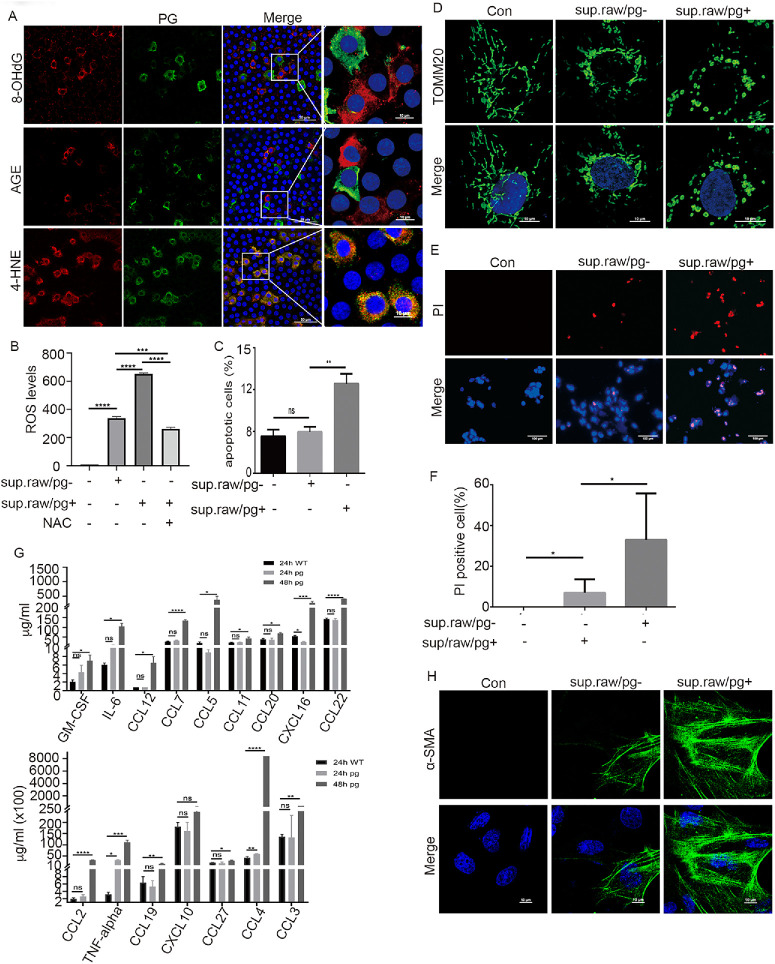
Upregulation of oxidative stress is associated closely with cataract occurrence. We then tested the association of PG+ macrophages and superoxide products in the anterior capsular epithelium of ten ARC patients by using double immunostaining PG and expression of oxidative biomarkers of 8-Hydroxydeovexyguanosine (8-OHdG), advanced glycation end products (AGEs) and 4-hydroxynonenal. The results showed that the expression of 8-OHdG and AGEs was up-regulated in the tested cataractous anterior capsular epithelial cells (Fig. 3A), but weak in the PG-positive macrophages (Supplementary Fig. S2A). Interestingly, the PG-positive macrophages were surrounded by the AGE or 8-OHdG-positive lens epithelial cells (Fig. 3A and Supplementary Fig. S2A). In contrast, 4-hydroxynonenal is solely positive in PG-positive cells (Fig. 3A). Those results suggested that PG infection increased lipid peroxidation in PG-positive macrophages and oxidative stress in those cataractous anterior epithelial cells closed to PG-positive macrophages.
Figure 3.
Induction of oxidative stress, apoptosis, and EMT of lens epithelial cells by PG-infected macrophages. (A) Whole mount immunofluorescence staining cataractous anterior capsular epithelium with antibody against RgpA together with 8-OHdG, AGE and 4-hydroxynonenal (4-HNE). The nuclear was stained by DAPI. Scale bar, 10 µm. (B) Flow cytometry measures the ROS level in primary rat lens epithelial cells that were treated with the supernatants of Raw264.7/PG- or Raw264.7/PG+ cells. Data are mean ± SD, n = 3. ***P < 0.001, ****P < 0.0001. The ROS inhibitor NAC was used as a control (con). (C) Flow cytometry measuring the apoptosis of primary rat lens epithelial cells that were treated with supernatants of Raw264.7/PG− or Raw264.7/PG+ cells. Data are mean ± SD, n = 3. *P < 0.05. (D) Immunofluorescent staining the mitochondria with anti-TOMM20 antibody in primary rat lens epithelial cells that were treated in same way as did in (C). Scale bar, 10 µm. (E) Immunostaining primary rat lens epithelial cells with PI and Hoechst 33433 that were treated in same way as in (C). Scale bar, 100 µm. (F) Quantitation of PI-positive cells vs. the Hoechst-33433–positive cells in (E). Data are mean ± SD, n = 5. *P < 0.05. (G) Luminex multiplex analysis of the expression of chemokines and cytokines in the supernatants of Raw264.7/PG− vs. Raw264.7/PG+ cells. Data are mean ± SD, n = 3. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. (H) Immunofluorescent staining the expression of α-SMA in the primary rat lens epithelial cells that were treated in same way as in (C). The nuclei were stained with DAPI. Scale bar, 10 µm.
Next, we studied the regulatory effect of PG-infected macrophages on lens epithelial cells in vitro. To do this, monocyte Raw264.7 cells were infected with PG bacteria as described in the Methods. PG-infected Raw264.7 (Raw264.7/PG+) were cultured in the complete media for 48 hours followed by incubating in serum-free media for another 24 hours. After this, the supernatants of Raw264.7/PG+ were collected, and the left cells were subjected to immunofluorescence staining with anti-RgpA antibody (Supplementary Figs. S2B, S2C). The results showed that PG could invade into and survive in Raw264.7 cells for at least 72 hours (Supplementary Figs. S2B, S2C). We treated the primary rat lens epithelial cells with the supernatants of Raw264.7/PG- or Raw264.7/PG+ for 72 hours. The primary rat lens epithelial cells cultured in normal media were used as controls. The results showed that the supernatants of both Raw264.7 and Raw264.7/PG+ cells could significantly elevate ROS level in rat primary lens epithelial cells, but the induction of ROS by supernatants of Raw264.7/PG+ cells is higher than that of Raw264.7/PG− cells (Figs. 3B, Supplementary S2D). The increased ROS level in rat primary lens epithelial cells was significantly suppressed by N-acetylcysteine, an inhibitor of ROS (Figs. 3B, Supplementary S2D). Consistent with the results in Figure 3A, the supernatants of Raw264.7/PG+ elevated the expression levels of 8-OHdG and AGE in rat primary lens epithelial cells (Supplementary Fig. S2E). In addition, we found that both supernatants of Raw264.7/PG− and Raw264.7/PG+ cells induced mitochondrial fragmentation, apoptosis, and necrosis of primary rat lens epithelial cells with high efficiency of supernatants of Raw264.7/PG+ (Figs. 3C–F, Supplementary S2F). These results suggest that the supernatant of Raw264.7/PG+ induced oxidative stress and apoptosis of rat primary lens epithelial cells in vitro. To determine harmful factors, such as, inflammatory factors in the supernatants of Raw264.7/PG+ cells, we analyzed the profiles of inflammatory factors in the supernatants of Raw264.7/PG+ cells vs. Raw264.7/PG− cells cultured at 24 and 48 hours. The results showed that Raw264.7/PG+ cells expressed high levels of proapoptotic factors and cytokines, such as TNFα, IL-6, CCL4, CCL-7, CCL-5, MCP-1, and MCP-5. compared with Raw264.7/PG− cells (Fig. 3G). These results suggested that the cytokines produced by PG-infected macrophages are associated with the induction of oxidative stress and apoptosis of lens epithelial cells. In addition to inducing apoptosis, oxidative stress also induces EMT of lens epithelial cells.31 As expected, the expression of α smooth muscle actin (α-SMA), an EMT marker, was up-regulated in primary rat lens epithelial cells that were treated with supernatants of Raw264.7/PG− and Raw164.7/PG+ cells, with a strong effect of the supernatants of Raw64.7/PG+ cells (Fig. 3H). These results indicate that PG-infected macrophages exert multiple regulatory effects on lens epithelial cells, including oxidative stress, apoptosis, and EMT.
PG-Positive Macrophages Enhance Traumatic ASCs in Rat Model
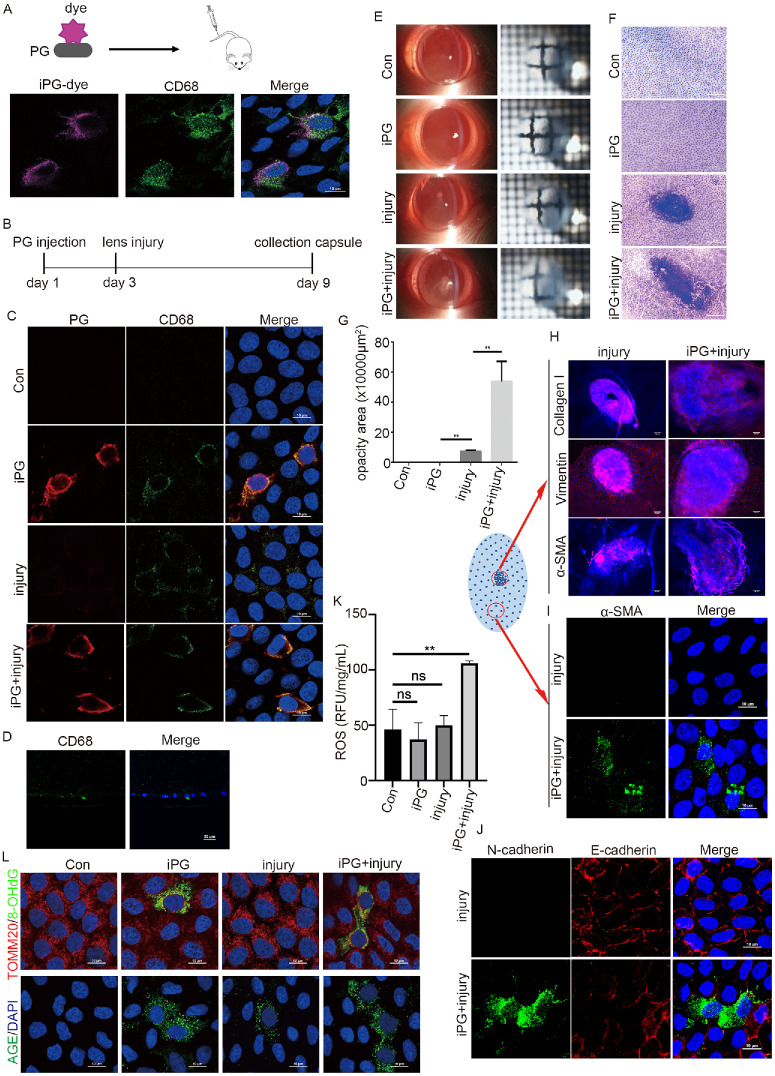
Macrophages were chemoattracted to diverse tissues participating in multiple processes, such as tissue remodeling, inflammation, wound healing, and fibrosis. Previous studies indicated that wounded cornea could promote immune cells migrating along zonule fibers into the equator lens epithelium.32 Our results indicated that PG-positive macrophages exist in cataractous anterior epithelium (Fig. 2). Accordingly, we proposed that macrophages/PG+ play a role during cataract development. To test this in an animal model, we first tested the infiltration of PG bacteria into lens via macrophages. The PG bacteria were labeled with CellVue Claret generating fluorescent PG-dye bacteria (Fig. 4A, top). We injected 107 PG-dye bacteria via the tail vein into rats (Fig. 4A, top). After 5 days, the lens anterior epithelium were stained with CD68 vs. fluorescent bacteria PG dye in whole mount. As the results showed that the fluorescent PG-dye were predominantly colocalized with CD68+ macrophages in the rat lens anterior epithelium (Fig. 4A). These results suggested that PG could migrate into the lens epithelium via monocytes. To determine the regulatory effect of PG injected via the tail vein on cataract progression, the ASC model was recruited. The 6-week-old Wistar rats (with an equal number of males and females) were divided into four groups with six rats in each group. Group 1 rats were given a tail vein injection of PBS (control), group 2 rats were injected with 107 PG bacteria, group 3 rats were given traumatic injury of lens at the central anterior part to generate ASC, and group 4 rats were given a tail vein injection of PG followed by traumatic injury. After injury, the rats were allowed to recovered for 7 days (Fig. 4B). Anterior capsules were collected for analysis of PG and ASC progression. The immunofluorescence in whole-mount results showed that CD68+ macrophages were observed in the anterior epithelium of groups 2, 3, and 4 rats, but not in group 1 rats (Fig. 4C). The PG-positive macrophages were observed in the samples of group 2 and 4 rats, less in group 3 rats, and not in group 1 rats (Fig. 4C). The existence of CD68+ macrophages in the anterior epithelium of ASC in the PG + injury group was also confirmed by using immunohistochemistry (Fig. 4D). These results showed that lens injury facilitated the recruitment of macrophages, including PG-positive macrophages, into the lens anterior epithelium. The results of slit-lamp and grind transparency assays indicated that PG infection increased ASC volume and opacity (Fig. 4E), and the increased ASC with multiple layer of cells was confirmed by hematoxylin and eosin staining (Figs. 4F, 4G). The pathological characteristic of ASC is upregulation of fibrosis. The immunofluorescent staining results showed that injection of PG enhanced the expression of collagen I, vimentin, and α-SMA in the ASC tissues (Fig. 4H), and the expression of α-SMA and N-cadherin, and decreased E-cadherin expression in the peripheral lens anterior epithelium surrounding SAC tissues (Figs. 4I, 4J). These results suggested that the invaded GP-positive monocytes could enhance the EMT and fibrosis induced by traumatic injury, because PG infection could increase oxidative stress in lens epithelial cells (Fig. 3). We then measured the ROS in the tissues of ASC models with spectrophotometer, and the results showed that PG could increase ROS levels significantly in ASC tissues compared with that in ASC alone or other two controls (Fig. 4K). Moreover, the expression of 8-OHdG and AGE was increased in the anterior epithelium of rats injected with PG alone (group 2) or PG + ASC (group 4) (Fig. 4L). These results suggest that PG infection promotes cataract development in ASC rat models at least via increasing the oxidative stress and EMT of lens epithelial cells.
Figure 4.
PG infection enhances traumatic anterior capsular cataracts in rats. (A) Immunofluorescence staining CD68-macrophage vs. PG dye in the anterior epithelium of Wistar rats that received a tail vein injection of PG dye. The nucleus was stained with DAPI. Scale bar, 10 µm. (Top) Schematic map of PG labeled by CellVue Claret (PG dye) and PG dye tail vein injection. (B) Schematic map of tail vein injection of PG into ASC rat models. (C) Immunostaining with antibodies against PG and CD68 in the rat lens capsules of the WT group, PG injected group (iPG), lens injury group (injury), and PG injected plus injury group (iPG + injury). The nucleus was stained with DAPI. Scale bar, 10 µm. (D) Immunohistochemistry staining the cryosection of rat lens of iPG + injury group with anti-CD68. The nucleus was stained with DAPI. Scale bar, 20 µm. (E) The images of slit-lamp microscopy and grind transparency of rat lens that were treated in (C). (F) Hematoxylin and eosin staining capsular anterior epithelium of rats that were treated in (C). Scale bar, 200 µm. (G) Quantitation of the ASC area in (F). Data are mean ± SD, n = 6. **P < 0.01. (H and I) Immunofluorescent staining in whole mount expression of collagen I, vimentin, and α-SMA in the ASC tissues (H) or in the peripheral lens epithelium close to ASC tissues of rat lens of the injury group or iPG + injury group, the nuclei were stained with DAPI. Scale bar, 50 µm. (J) Immunofluorescent staining in whole mount the expression of N-cadherin and E-cadherin in the peripheral lens epithelium surrounding ASC tissues of lens from injury group or iPG + injury group, the nuclei were stained with DAPI, Scale bar, 10 µm. (K) Enzyme labeling measures the ROS level in the rat lens of WT group, iPG group, injury group, and iPG + injury group. Data are mean ± SD, n = 3. **P < 0.01. (L) Immunofluorescence staining in whole mount the rat lens capsules of the WT group, iPG group, injury group, and iPG + injury group with antibodies against TOMM20 (red) and 8-OHdG (green) (top) or AGE alone (bottom). The nuclei were stained by DAPI (blue). Scale bar, 10 µm. con, control; ns, not significant.
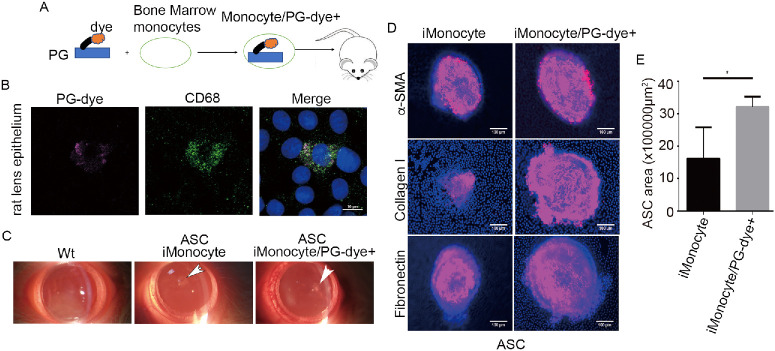
To avoid the direct cytotoxicity of PG injected via the tail vein, we performed a rat allogeneic monocytes transplantation assay in ASC rats. BMMs were isolated from rats followed by infecting with PG dye (Fig. 5A), generating BMM/PG-dye+ cells (Supplementary Fig. S3). The BMM/PG-dye+ cells were injected via the tail vein into normal rats. BMMs without PG infection were used as controls. The results of whole mount staining indicated that BMM/PG-dye+ cells could invade into the rat lens anterior epithelium (Fig. 5B). To determine the roles of migrated BMM/PG-dye+ in ASC development, rats were injected via the tail vein with BMMs or BMM/PG-dye+. After 2 days of recovery, the rats underwent an ASC operation. The results of the slit-lamp microscope showed that the injection of BMM/PG-dye+ increase ASC size (Figs. 5C, 5D). The results of immunofluorescent staining in whole mount indicated that injection of BMM/PG-dye+ cells up-regulated the expression of α-SMA, collagen I, and fibronectin in ASC tissues compared with that of injected BMM alone (Figs. 5D, 5E). These results were consistent with results presented in Figure 4. Taken together, the macrophages were hosts for PG in the anterior capsular epithelium, promoting cataract development.
Figure 5.
Transplantation of PG monocytes promote cataract progression in rat ASC model. (A) The schematic map of tail vein injection of BMMs that were pre-infected with PG dye. (B) Whole mount immunofluorescence staining of transplanted monocyte/PGdye+ cells in the lens anterior capsular epithelium of rats with anti-CD68 antibody. The nuclei were stained with DAPI, Scale bar, 10 µm. (C) Slit lamp microscopy observed ASC of rats that were received allogeneic monocytes (immunocytes) or immunocyte/PGdye+. (D) Immunofluorescent staining in whole mount the expression of collagen-1, α-SMA, and fibronectin in ASC tissues from rats treated in (C). (E) The quantitation of ASC area of rats in (D) in image J. Data are mean ± SD, n = 20. *P < 0.05.
In addition to ASC model, we tested the regulatory effect of PG infection in a galactose-induced rat cataract model. Forty 4-week-old Wistar rats were divided into three groups: group 1 rats were injected intraperitoneally with PBS; group 2 rats were injected with a high concentration of galactose (9 g/kg, intraperitoneal injection, 12-hour intervals) for 30 days; and group 3 rats were injected via the tail vein with 107 PG once, followed by giving galactose treatment for 30 days. The cataracts were observed with slit-lamp microscopy. The results showed that the rats in groups 2 and 3 developed cortical cataracts at day 15 or 16, and the opacity of cataractous lens became severe with prolonged galactose treatment. In contrast, PG injection did not enhance the incidence or severity of galactose-induced cataracts (Supplementary Fig. S4A). The results of immunofluorescence in whole mount showed that there were a number of CD68+ cells in the anterior epithelium of PG-injected galactose-induced cataracts (Supplementary Fig. S4B). No CD68+ macrophages were observed in the anterior epithelium of control or galactose-induced cataracts. These results suggested that PG-positive macrophages were able to invade into the lens epithelium under osmotic stress condition, but its promotion activity on cataract development was not observed.
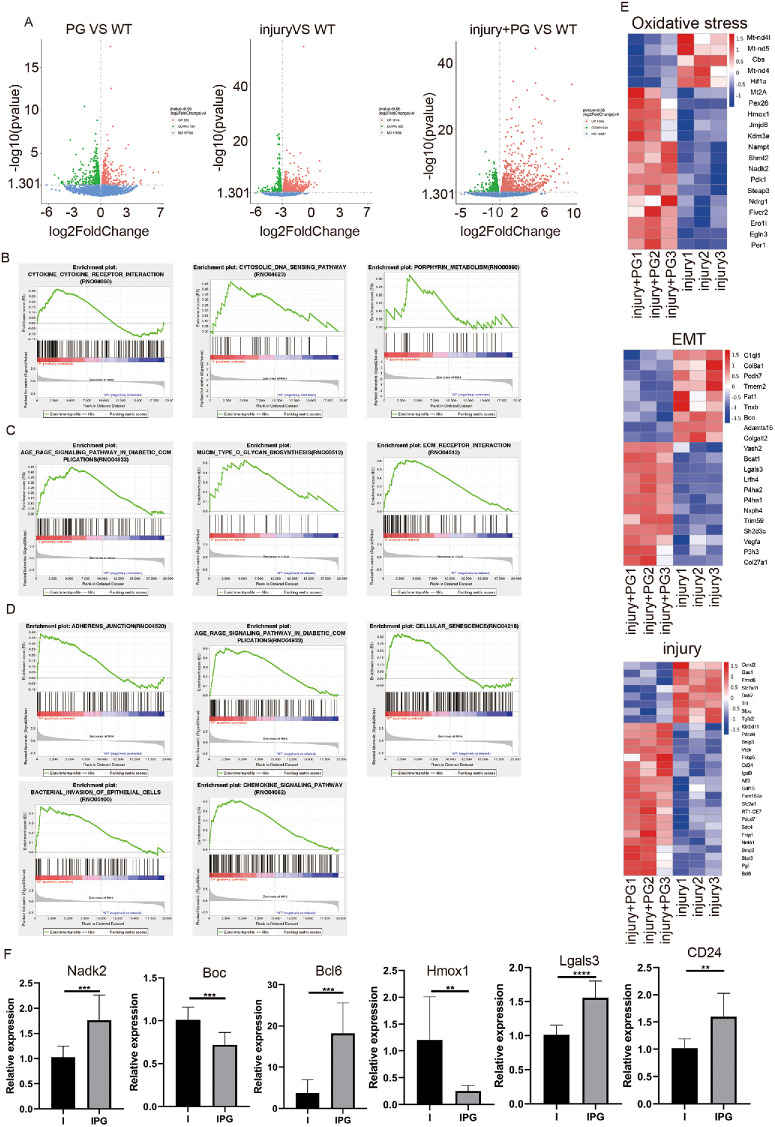
Transcriptome Analysis of Gene Expression Profile in a PG-infected ASC Model
To comprehensively understand the gene change in PG-promoted ASC, the lens anterior epithelium of four groups of rats that were treated in Figures 4D and 4E were applied to RNA deep sequencing. Based on our RNA sequencing results, compared with the WT group, there were 559 up-regulated genes and 724 down-regulated genes in the lens with PG-injection alone; 800 down-regulated genes and 1010 up-regulated genes in ASC lens; 938 down-regulated genes and 1595 up-regulated genes in the PG + ASC lenses (Fig. 6A). To better explore the biological functions of differentially expressed genes, KEGG analysis showed that compared with PBS-injected rat lens, PG-injected rat lens affected predominantly the signal pathways that are associated with cytokine receptor interaction, the DNA sensing pathway, and porphyrin metabolism (Fig. 6B); ASC alone mainly affected the signals associated with AGE–receptor for AGE (RAGE) signaling pathway, mucin-type O-glycan biosynthesis, and ECM receptor interaction (Fig. 6C). ASC + PG lensed changed the signals associated with adherens junction, AGE–RAGE signaling pathway and cellular senescence, bacterial invasion of epithelial cells, and chemokine signaling pathway (Fig. 6D). According to the analysis results of KEGG, we found that the differences in differentially expressed genes between the ASC lens and the ASC + PG lensed mainly focused on the following signal pathways: oxidative stress-related proteins, apoptosis and injury-related proteins, and EMT-related proteins (Fig. 6E). Furthermore, we conducted qPCR to validate the expression of these genes. As expected, compared with the ASC lens, NAD kinase 2, B-cell chronic lymphocytic leukemia/lymphoma 6, lectin, galactosidase-binding, soluble 3, and CD24 were up-regulated in ASC + PG lens tissues. The antioxidative stress genes, such as BOC cell adhesion associated-oncogene regulated and heme oxygenase (decycling) 1 were down-regulated in ASC + PG lensed compared with that in ASC lens (Fig. 6F). Taken together, PG infection aggravates the development of traumatic ASCs by increasing oxidative stress, apoptosis, and cell EMT.
Figure 6.
Transcriptome profile analysis of gene expression in rat anterior capsular tissues. (A) Volcano maps and Venn diagram analysis of differentially expressed genes in the anterior capsular tissues of rats from the iPG group vs. WT group; injury group vs. WT group, or iPG + injury group vs. WT group. (B) GSEA analysis of KEGG enrichment in lens of iPG group vs. WT. (C) GSEA analysis of KEGG enrichment in lens of injury group vs. WT. (D) GSEA analysis of KEGG enrichment in lens of iPG + injury group vs. WT. (E) Heat maps exhibition of differentially expressed genes of oxidative stress, injury, cellular matrix, inflammation, differentiation, and EMT in lens anterior capsular tissues of lens of the injury group vs. the iPG + injury group. Three samples of each group were applied for analysis. (F) qPCR analysis of differentially expressed genes in oxidative stress, injury, cellular matrix, inflammation, differentiation, and EMT in (E). Data are mean ± SD, n = 3. *P < 0.05, **P < 0.01, ***P < 0.001.
Discussion
Cataracts are a leading cause of visual loss in aged people. The etiology of cataracts is not fully understood and is likely to be multifactorial, such as light, hyperglycemic, oxidative damage, smoking, alcohol, and so on.33 Recently, prevalent studies suggest that patients with periodontitis have a high incidence of cataracts.24 In this paper, we found that PG infection is a novel risk factor for cataracts. PGs 16s rRNA gene is positive in 43.3% AH and 82% to 94% anterior capsular tissues of patients with cataracts (Fig. 1). Moreover, we found that macrophages but not lens epithelial cells are PG-hosted cells in cataractous anterior epithelium (Figs. 2, 4, 5). To test the roles of PG infection on cataracts, we recruited rat allogeneic monocyte–transplantation assay, and found that tail vein injected PG-infected BMMs can migrate, invade, and reside in the anterior lens epithelium of injured rat lens (Fig. 5), which then enforces the pathological progression of injury-induced ASC by elevating production of inflammatory cytokines and/or inducing oxidative stress and EMT to surrounding epithelial cells (Figs. 3–5). The study results in vitro suggested that PG-infected Raw264.7 cells can produce harmful factors, such as inflammatory cytokines, which can induce oxidative stress, apoptosis and EMT of primary rat lens epithelial cells (Fig. 3). Those results suggest that translocation of PG-infected macrophages into injured lens enforces cataract development.
PG has been identified in diverse tissues with chronic diseases, such as Alzheimer's disease neurons, atherosclerosis, arthritis tissues, and tumors.34–37 Common methods of identifying infection of intracellular PG are PCR amplification of genomic DNA of PG16s rRNA and immunofluorescent staining for the expression of gingipain proteins, such as RgpA or RgpB. Using PCR, we found that approximately 43.3% of AH from patients with cataracts are shown PG positive. To avoid blood cell contamination of AH, we acquired AH with a syringe before cataract surgery. Because PG is normally living inside host cells, this leads us to wonder how PG exists in AH. The results in Figure 4 indicated that PG positive cells were CD68+ macrophages in lens epithelium, and PG-positive macrophages can infiltrate into lens the epithelium in a rat ASC model (Fig. 4). Thus, we postulate that macrophages may be the host of PG in AH. Remarkably, HMCs and DCs increase the rates of PG-positive AH. Previous studies indicate that diabetes enhances PG-infected periodontitis.27 Diabetes is a common cause of lens damage, which may increase migration of PG-positive macrophages into AH, and this proposal may also apply to HMCs.
In addition to AH-PG, we surprisingly find that the genomic DNA of PGs 16s rRNA is positive in the anterior capsular tissues of 82% HMC, 94% of DC, 93% of ARCs, and 92% of congenital cataract patients. These results suggest that PG could invade the intracellular of cells in lens anterior epithelium of cataracts. However, unlike PG-infected endothelial cells in atherosclerosis,38 or PG-infected neurons in patients with Alzheimer's disease,16 we found that these PG-positive cells in the lens epithelia are macrophages rather than lens epithelial cells (Figs. 1, 2). We define the macrophages in cataractous lens anterior epithelium by using antibodies against CD68, CD14, CD206, and CD369. However, the nuclear morphology of macrophages looks similar to lens epithelial cells. It is unclear whether the invaded macrophages have to change their shape to adapt to lens epithelial cells. We validate that monocytes act as transmitters of PG to lens epithelium by transplanting PG-positive BMMs into a rat ASC model (Fig. 5). Interestingly, we found that PGs 16s rRNA genome is tested positive in 92% of anterior epithelium of patients with congenital cataracts aged from 2 to 10 years (Fig. 1). This finding implies that PG infection is also a potential risk factor of congenital cataracts. However, how PG is transmitted to the congenital cataractous lens remains unclear. It is reported that pregnant women with periodontitis have a greater chance of transmitting PG via the blood stream through the placenta to the fetus and impairing fetal development. Breast feeding mothers with periodontitis have a high chance of transmitting PG to their infant.39 According to these data and our results, we propose that congenital cataracts attract PG-positive macrophages to invade into the cataractous lens, which then facilitates the progression of congenital cataracts. However, the regulatory roles of PG infection on the congenital cataracts need more experiments as validation.
PGs invade directly and survive in several cell types, such as fibroblast cells, monocytes, macrophages, endothelial cells, and chondrocytes. Blood cells, such as monocytes, macrophages, and red blood cells, are the predominant means to transmit PG to distant tissues.5,40 Macrophages are reported to migrate along the ciliary zonules into and reside in equatorial the epithelium, participating in lens development, lens wound repair, and fibrosis.29,41 Consistent with these reports, our data showed that monocytes or macrophages are vehicles that bring PG into the lens anterior epithelium under stressful conditions (Figs. 4, 5). No PG bacteria were detected in lens epithelial cells, suggesting that PG was not transmitted from macrophages to lens epithelial cells. Altogether, we assume that the migration of macrophages from the ciliary body is the source of PG in cataractous lenses.
To determine the role of PG during cataract occurrence, we recruited two models: One is the galactose-induced cataract model, and the other is traumatic-induced ASC. In the galactose-induced rat cataract model, PG injected via the tail vein did not show a triggering effect on galactose-induced cataracts. In the ASC model, however, PG promoted ASC development (Figs. 4, 5). The mechanisms underlying this different outcome remain unclear. However, both models facilitated macrophages invading the cataractous lens epithelium (Figs. 5, Supplementary S4). In the ASC model, PG infection upregulates the genes’ expression associated with AGE-RAGE pathway, ECM-receptor interaction pathways and chemokine-signal pathways, and down-regulates the genes’ expression associated with antioxidative pathways (Fig. 6). According to those results, we postulate that PG infection acts at least as a promoter during cataract progression, although the roles of PG on cataract initiation have not been investigated. The pathological promotion of PG infection has been reported in diverse age-related diseases, such as Alzheimer’s disease, cancer, atherosclerosis, heart diseases, and arthritis.6,16 These results demonstrate that chronic infection of PG is an etiological factor for cataract development.
Conclusions
PG infection is associated closely with ARCs as well as congenital cataracts. PG exists in macrophages in the anterior epithelium of cataracts, and promotes cataract development. Prophylaxis of PG should be a candidate strategy to delay cataract development.
Supplementary Material
Acknowledgments
Supported by grants from the National Nature Science Foundation of China (No. 81900843 and U1604171), the Science and Technology Development Project of Henan Province (No. 222102310673, 222102310297, 242102310398, and 232102310210), and the Young Talents Project of Sinopharm Dongfeng General Hospital (No. 2022Q23).
Disclosure: D. Zhang, None; J. Qu, None; C. Ke, None; X. Kong, None; M. Liu, None; I. Nawaz Khan, None; S. Huang, None; H. Tian, None; T. Xie, None; K. Qiu, None; J. Li, None; M. Wang, None; H. Li, None; F. Yuan, None; W. Guo, None; M. Cao, None; J. Zhang, None; K. Zhu, None; J. Luo, None; F. Zhang, None; X. Cui, None; H. Mu, None; Y. Hu, None
Supplementary Material
Supplementary Video S1. Immunofluorescence staining in whole mount of anterior capsular epithelium of cataractous patients with antibodies against RgpA (green) and ZO-1 (red). The picture was taken in Z-scan. The nuclei were stained with DAPI. Scale bar: 10 µm.
Supplementary Video S2. Immunofluorescence staining in whole mount of anterior capsular epithelium of cataractous patients with antibodies against CD68 (green) and ZO-1 (red). The picture was taken in Z-scan. The nuclei were stained with DAPI. Scale bar: 10 µm.
References
- 1. Asbell PA, Dualan I, Mindel J, Brocks D, Ahmad M, Epstein S.. Age-related cataract. Lancet. 2005; 365: 599–609. [DOI] [PubMed] [Google Scholar]
- 2. Liu YC, Wilkins M, Kim T, Malyugin B, Mehta JS.. Cataracts. Lancet. 2017; 390: 600–612. [DOI] [PubMed] [Google Scholar]
- 3. Ang MJ, Afshari NA.. Cataract and systemic disease: a review. Clin Exp Ophthalmol. 2021; 49: 118–127. [DOI] [PubMed] [Google Scholar]
- 4. Rewri P, Sharma M, Vats DP, Singhal A.. Seroprevalence, risk associations, and cost analysis of screening for viral infections among patients of cataract surgery. Indian J Ophthalmol. 2018; 66: 394–399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. York A. P. gingivalis drives carcinoma progression. Nat Rev Microbiol. 2020; 18: 674. [DOI] [PubMed] [Google Scholar]
- 6. Maresz KJ, Hellvard A, Sroka A, et al.. Porphyromonas gingivalis facilitates the development and progression of destructive arthritis through its unique bacterial peptidylarginine deiminase (PAD). PLoS Pathog. 2013; 9: e1003627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Nakahara T, Hyogo H, Ono A, et al.. Involvement of Porphyromonas gingivalis in the progression of non-alcoholic fatty liver disease. J Gastroenterol. 2018; 53: 269–280. [DOI] [PubMed] [Google Scholar]
- 8. Li C, Yu R, Ding Y.. Association between Porphyromonas Gingivalis and systemic diseases: Focus on T cells-mediated adaptive immunity. Front Cell Infect Microbiol. 2022; 12: 1026457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Giacona MB, Papapanou PN, Lamster IB, et al.. Porphyromonas gingivalis induces its uptake by human macrophages and promotes foam cell formation in vitro. FEMS Microbiol Lett. 2004; 241: 95–101. [DOI] [PubMed] [Google Scholar]
- 10. Lee K, Roberts JS, Choi CH, Atanasova KR, Yilmaz Ö.. Porphyromonas gingivalis traffics into endoplasmic reticulum-rich-autophagosomes for successful survival in human gingival epithelial cells. Virulence. 2018; 9: 845–859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Lenzo JC, O'Brien-Simpson NM, Cecil J, Holden JA, Reynolds EC. Determination of active phagocytosis of unopsonized porphyromonas gingivalis by macrophages and neutrophils using the pH-sensitive fluorescent dye pHrodo. Infect Immun. 2016; 84: 1753–1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Andrian E, Grenier D, Rouabhia M.. Porphyromonas gingivalis-epithelial cell interactions in periodontitis. J Dent Res. 2006; 85: 392–403. [DOI] [PubMed] [Google Scholar]
- 13. Inaba H, Kuboniwa M, Bainbridge B, et al.. Porphyromonas gingivalis invades human trophoblasts and inhibits proliferation by inducing G1 arrest and apoptosis. Cell Microbiol. 2009; 11: 1517–1532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Singh S, Yadav PK, Singh AK.. In-silico structural characterization and phylogenetic analysis of Nucleoside diphosphate kinase: a novel antiapoptotic protein of Porphyromonas gingivalis. J Cell Biochem. 2023; 124: 545–556. [DOI] [PubMed] [Google Scholar]
- 15. Muñoz-Medel M, Pinto MP, Goralsky L, et al.. Porphyromonas gingivalis, a bridge between oral health and immune evasion in gastric cancer. Front Oncol. 2024; 14: 1403089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Dominy SS, Lynch C, Ermini F, et al.. Porphyromonas gingivalis in Alzheimer's disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci Adv. 2019; 5: eaau3333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kobayashi T, Okada M, Ito S, et al.. Assessment of interleukin-6 receptor inhibition therapy on periodontal condition in patients with rheumatoid arthritis and chronic periodontitis. J Periodontol. 2014; 85: 57–67. [DOI] [PubMed] [Google Scholar]
- 18. Botero JE, Posada-Lopez A, Mejia-Vallejo J, Pineda-Tamayo RA, Bedoya-Giraldo E.. Effects of nonsurgical periodontal therapy in patients with rheumatoid arthritis: a prospective before and after study. Colomb Med (Cali). 2021; 52: e2095051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Chiu CJ, Chang ML, Kantarci A, Van Dyke TE, Shi W.. Exposure to Porphyromonas gingivalis and modifiable risk factors modulate risk for early diabetic retinopathy. Transl Vis Sci Technol. 2021; 10: 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Arjunan P, Swaminathan R, Yuan J, et al.. Exacerbation of AMD phenotype in lasered CNV murine model by dysbiotic oral pathogens. Antioxidants (Basel). 2021;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Gasmi Benahmed A, Kumar Mujawdiya P, Noor S, Gasmi A. Porphyromonas gingivalis in the development of periodontitis: impact on dysbiosis and inflammation. Arch Razi Inst. 2022; 77: 1539–1551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Karesvuo P, Gursoy UK, Pussinen PJ, et al.. Alveolar bone loss associated with age-related macular degeneration in males. J Periodontol. 2013; 84: 58–67. [DOI] [PubMed] [Google Scholar]
- 23. Arjunan P, Swaminathan R, Yuan J, et al.. Invasion of human retinal pigment epithelial cells by Porphyromonas gingivalis leading to vacuolar/cytosolic localization and autophagy dysfunction in-vitro. Sci Rep. 2020; 10: 7468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Yeh LJ, Shen TC, Sun KT, Lin CL, Hsia NY.. Periodontitis and subsequent risk of cataract: results from real-world practice. Front Med (Lausanne). 2022; 9: 721119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Stepp MA, Menko AS.. Immune responses to injury and their links to eye disease. Transl Res. 2021; 236: 52–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hong HL, Flurin L, Greenwood-Quaintance KE, et al.. 16S rRNA Gene PCR/sequencing of heart valves for diagnosis of infective endocarditis in routine clinical practice. J Clin Microbiol. 2023; 61: e0034123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Ojima M, Takeda M, Yoshioka H, et al.. Relationship of periodontal bacterium genotypic variations with periodontitis in type 2 diabetic patients. Diabetes Care. 2005; 28: 433–434. [DOI] [PubMed] [Google Scholar]
- 28. Cheng C, Nowak RB, Fowler VM.. The lens actin filament cytoskeleton: diverse structures for complex functions. Exp Eye Res. 2017; 156: 58–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. DeDreu J, Le PM, Menko AS.. The ciliary zonules provide a pathway for immune cells to populate the avascular lens during eye development. Exp Biol Med (Maywood). 2022; 247: 2251–2273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Logan CM, Bowen CJ, Menko AS.. Induction of immune surveillance of the dysmorphogenic lens. Sci Rep. 2017; 7: 16235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Wei Z, Caty J, Whitson J, et al.. Reduced glutathione level promotes epithelial-mesenchymal transition in lens epithelial cells via a Wnt/beta-catenin-mediated pathway: relevance for cataract therapy. Am J Pathol. 2017; 187: 2399–2412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. DeDreu J, Bowen CJ, Logan CM, et al.. An immune response to the avascular lens following wounding of the cornea involves ciliary zonule fibrils. FASEB J. 2020; 34: 9316–9336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Gupta VB, Rajagopala M, Ravishankar B.. Etiopathogenesis of cataract: an appraisal. Indian J Ophthalmol. 2014; 62: 103–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Ermini F, Low VF, Song JJ, et al.. Ultrastructural localization of Porphyromonas gingivalis gingipains in the substantia nigra of Parkinson's disease brains. NPJ Parkinsons Dis. 2024; 10: 90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Rao A, Kumar BK.. Role of periodontal pathogens in atherosclerotic plaque development and progression: An overview. Acta Microbiol Immunol Hung. 2023; 70: 272–277. [DOI] [PubMed] [Google Scholar]
- 36. Tan Q, Ma X, Yang B, et al.. Periodontitis pathogen Porphyromonas gingivalis promotes pancreatic tumorigenesis via neutrophil elastase from tumor-associated neutrophils. Gut Microbes. 2022; 14: 2073785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Totaro MC, Cattani P, Ria F, et al.. Porphyromonas gingivalis and the pathogenesis of rheumatoid arthritis: analysis of various compartments including the synovial tissue. Arthritis Res Ther. 2013; 15: R66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Xie M, Tang Q, Nie J, et al.. BMAL1-downregulation aggravates porphyromonas gingivalis-induced atherosclerosis by encouraging oxidative stress. Circ Res. 2020; 126: e15–e29. [DOI] [PubMed] [Google Scholar]
- 39. Ye C, Katagiri S, Miyasaka N, et al.. The periodontopathic bacteria in placenta, saliva and subgingival plaque of threatened preterm labor and preterm low birth weight cases: a longitudinal study in Japanese pregnant women. Clin Oral Investig. 2020; 24: 4261–4270. [DOI] [PubMed] [Google Scholar]
- 40. Pritchard AB, Crean S, Olsen I, Singhrao SK.. Periodontitis, microbiomes and their role in Alzheimer's disease. Front Aging Neurosci. 2017; 9: 336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Li Y, Li Z, Quan Y, et al.. Macrophage recruitment in immune-privileged lens during capsule repair, necrotic fiber removal, and fibrosis. iScience. 2021; 24: 102533. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.