Abstract
Introduction
The plasma lipidome has emerged as an important indicator for assessing host metabolic and immune status in sepsis. While previous studies have largely examined specific lipid class changes in adults sepsis, comprehensive investigations into plasma lipidomic alterations in pediatric sepsis are limited. This study aimed to characterize the plasma lipidome in pediatric sepsis using a metabolomics-based exploratory approach, providing insights into pathophysiological mechanisms and potential biomarkers.
Methods
A retrospective study was conducted on pediatric patients with sepsis admitted to the pediatric intensive care unit (PICU). Untargeted lipidomics analysis using ultra-performance liquid chromatography coupled with Orbitrap mass spectrometry (UPLC-Orbitrap) was performed to compare metabolomic profiles between non-infected control patients and sepsis patients.
Results
Compared to controls, plasma lipid levels in sepsis patients decreased by 33.3%, increased by 20.2%, and remained unchanged in 46.5% of cases. A total of 1,257 differential lipids were identified in sepsis patients, with 24 lipids showing significant associations with pSOFA scores. In the recovery and deterioration subgroups, 186 differential lipids were identified, with triglyceride (TG) representing the highest proportion at 16.4%. Notably, 15 lipids with significant statistical differences were identified as differential lipid species through a comparison of those associated with pSOFA scores and those linked to sepsis prognosis. Fatty acid (FA) levels were significantly elevated in the sepsis group compared to controls, with arachidonic acid (FA(20:4)) showing the most significant increase (P < 0.001).
Conclusion
Alterations in plasma lipid profiles among children with sepsis reflect disease severity, systemic inflammatory responses, and sepsis prognosis. These findings underscore the prognostic potential of lipidomics and its value in understanding sepsis pathophysiology.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11306-025-02255-x.
Keywords: Pediatric sepsis, Plasma lipidome, Untargeted lipidomics, Fatty acid
Introduction
Sepsis remains a major global health challenge, contributing to approximately 3.3 million childhood deaths annually (Sanchez-Pinto et al., 2024). Despite significant advancements in healthcare that have led to improved clinical outcomes and a steady decline in mortality rates (Bauer et al., 2020), severe sepsis continues to be the leading cause of death in Pediatric Intensive Care Units (PICU) (Barber et al., 2023; Weiss & Fitzgerald, 2024). Alarmingly, one-third of these fatalities are attributed to refractory shock (Zhang & Ning, 2021). While advancements in antibiotic therapies and infection control strategies have provided some progress, early recognition and timely effective treatment of sepsis remain critical clinical challenges. The search for potential biomarkers has been continued from several years to guide critical care physicians in distinguishing the source and etiology of sepsis to predict the disease progression into septic shock.
Metabolomics in sepsis and septic shock offers a powerful tool for early detection and identification of infection by using biomarkers (Pandey, 2024b). A study using a targeted LC-MS/MS-based metabolomics approach found that plasma levels of low unsaturated long-chain phosphatidylcholines (PC) and lysophosphatidylcholines (LPC) were significantly lower in non-survivors of sepsis compared to survivors, suggesting that lipid homeostasis may influence mortality associated with septic shock patients (Siddiqui et al., 2020). Cambiaghi et al. conducted an integrated analysis of proteomic and metabolomic data associated with patients with septic shock and found that the metabolomic profile of non-survivors included long-chain polyunsaturated fatty acids, such as PC aa C42:6, PC aa C40:6, and LPC species. Over time, an imbalance in plasmalogen levels was also observed between non-survivors and survivors (Cambiaghi et al., 2018). Another study compared metabolic profiles based on infection types and demonstrated that LPC a C18:0, and sphingomyelin (SM) C16:1 were associated with non-survivors in sepsis with community-acquired pneumonia, intra-abdominal infections, and bloodstream infections, respectively (Pandey, 2024a).
Pediatric Sequential Organ Failure Assessment (pSOFA) scores has demonstrated a strong correlation with in-hospital mortality, establishing it as a valuable tool for evaluating disease severity in pediatric sepsis patients (Balamuth et al., 2022). In addition to pSOFA scores, the 2015 Chinese pediatric Sepsis Guidelines recommend using white blood cell (WBC), C-reactive protein (CRP), procalcitonin (PCT) and lactate levels as evaluation indicators (“[Expert consensus for the diagnosis and management of septic shock (infectious shock) in children (2015)],” 2015). However, the prognostic utility of some of these markers is limited. For example, WBC count and CRP levels exhibit low specificity for predicting sepsis outcomes, as no significant differences have been observed between the survival and non-survival groups (Schlapbach et al., 2018). The pathophysiology of sepsis involves an overwhelming and dysregulated inflammatory response, characterized by excessive release of inflammatory factors that damage endothelial cells and activate the coagulation system. This cascade results in substantial platelet (PLT) consumption, placing the body in a hypercoagulable state and increasing the risk of disseminated intravascular coagulation (DIC). Consequently, monitoring plasma inflammatory factors and PLT levels is critical for assessing sepsis severity and prognosis (Giustozzi et al., 2021). Arachidonic acid (ARA), an important precursor of inflammatory mediators, plays a central role in the systemic inflammatory response (SIRS) associated with sepsis. During the acute phase of sepsis, accelerated ARA metabolism leads to the production of pro-inflammatory mediators such as prostaglandins and leukotrienes, contributing to an amplified immune response (Wang et al., 2019). Elevated levels of ARA have been observed in cases of strong inflammatory responses, highlighting its potential as a marker of disease activity.
Despite significant advancements in understanding lipid metabolism in adult sepsis, studies on pediatric sepsis remain scarce (Chouchane et al., 2024; Li et al., 2021; Tian et al., 2022). Most prior research has focused on specific lipid types, often neglecting a comprehensive analysis of the entire lipidome. We hypothesize that specific lipidomic changes are closely associated with sepsis severity and prognosis in pediatric patients, potentially serving as valuable biomarkers for disease progression and clinical outcomes. Therefore, in this study, we analyzed the plasma lipidom of pediatric sepsis patients to characterize lipid alterations and their association with disease severity, immune response, and prognosis.
Materials and methods
Study population
A total of 51 pediatric patients diagnosed with sepsis were recruited from the PICU at the Children’s Hospital of Fudan University between June 2022 and May 2024. Eligible participants were aged 28 days to 18 years and met the diagnostic criteria for sepsis. Exclusion criteria included known lipid metabolism disorders, hyperlipidemia, parenteral nutrition, and severe hepatic dysfunction. Based on outcomes within 28 days of sepsis diagnosis, patients were stratified into the recovery group and the deterioration group. Outpatient subjects from the same period, free of infections or acute illnesses and matched by gender and age, served as controls. The following demographic and clinical variables were recorded for all participants: age, sex, body mass index (BMI), pSOFA scores, pediatric critical illness score (PCIS), pediatric multiple organ dysfunction score (pMODS), presence of septic shock, mechanical ventilation, severe pneumonia, DIC, respiratory failure, and comorbidities such as renal disease, cardiac disease, hypertension, and neurological disorders. Pathogen identification was also performed for all septic patients.
The study was approved by Ethics Committee of Children’s Hospital of Fudan University ([2024] No. 116), and informed consent was obtained from all participants’ parents or guardians. From each participant, 2 mL peripheral blood was drawn under fasting and anticoagulated with EDTA. After centrifugation, the sample was immediately stored in the refrigerator of − 80 °C for the subsequent UHPLC-MS/MS analysis.
Sample preparation and lipid extraction
Lipids were extracted according to MTBE method. Briefly, a 200 µL volume of water was added to sample and vortexed for 5 s. Subsequently, 240 µL of precooling methanol was added and the mixture vortexed for 30 s. After that, 800 µL of MTBE was added and the mixture was ultrasound 20 min at 4 ℃ followed by sitting still for 30 min at room temperature. The solution was centrifuged at 14,000 g for 15 min at 10℃ and the upper organic solvent layer was obtained and dried under nitrogen.
LC-MS/MS method for lipid analysis
Reverse phase chromatography was selected for LC separation using CSH C18 column (1.7 μm, 2.1 mm× 100 mm, Waters). The lipid extracts were re-dissolved in 200 µL 90% isopropanol/acetonitrile, centrifuged at 14,000 g for 15 min, finally 3 µL of sample was injected. Solvent A was acetonitrile–water (6:4, v/v) with 0.1% formic acid and 0.1 mM ammonium formate and solvent B was acetonitrile–isopropanol (1:9, v/v) with 0.1% formic acid and 0.1 mM ammonium formate. The initial mobile phase was 30% solvent B at a flow rate of 300 µL/min. It was held for 2 min, and then linearly increased to 100% solvent B in 23 min, followed by equilibrating at 5% solvent B for 10 min.
Mass spectra was acquired by Q-Exactive Plus in positive and negative mode, respectively. ESI parameters were optimized and preset for all measurements as follows: Source temperature, 300 °C; Capillary Temp, 350 °C, the ion spray voltage was set at 3000 V, S-Lens RF Level was set at 50% and the scan range of the instruments was set at m/z 200–1800.
Identification by lipid search
“Lipid Search” is a search engine for the identification of lipid species based on MS/MS math. LipidSearch contains more than 30 lipid classes and more than 1,500,000 fragment ions in the database. Both mass tolerance for precursor and fragment were set to 5 ppm.
Statistical analysis
Continuous variables with normal distributions were compared using Student’s t-test, while non-normally distributed data were analyzed using the nonparametric Mann-Whitney U test to determine differences between groups (the Benjamini-Hochberg (BH) correction was applied to all multiple comparisons). LipidSearch (Thermo Scientific™) (Taguchi & Ishikawa, 2010) was used to analyze the metabolites using the orthogonal partial least squares (PLS) model. The lipidomics data were sum-normalized using R software (https://www.r-project.org/). Correlations between variables were tested using Spearman’s rank correlation test, and scatter plots were created for visualization using GraphPad Prism version 10.1 (GraphPad Software, San Diego, CA, USA). Lipid class levels calculated by aggregating all lipid species values within a class for each subject. All statistical tests were two-tailed, with P < 0.05 considered statistically significant.
Results
Clinical characteristics
A total of 48 pediatric sepsis patients and 48 control patients were included in the final analysis (Fig. S1). Among the sepsis cohort, 37 patients achieved recovery, while 11 experienced clinical deterioration, which included 8 deaths and 3 voluntary discharges. Within the sepsis group: Septic shock was observed in 24 patients, with a significantly higher occurrence in the deterioration group (n = 9, 81.8%). Mechanical ventilation was required for 26 patients, with 90.9% (n = 10) belonging to the deterioration group. Disseminated intravascular coagulation (DIC) developed in 18 patients, of whom 81.8% (n = 9) were in the deterioration group. Respiratory failure occurred in 29 patients, and notably, all patients in the deterioration group (n = 11, 100%) experienced this complication. Moreover, cardiac dysfunction was identified in 17 patients, with 90.9% (n = 10) from the deterioration group. pSOFA scores was notably higher in the deterioration group [15.0 (13.0, 16.0)] compared to the recovery group [6.5 (4.2, 11.0)]. Additionally, Gram-positive bacteria were the most commonly identified pathogens in the sepsis group (Table 1).
Table 1.
Baseline demographic and clinical characteristics
| Control (n = 48) | Sepsis_Recovery (n = 37) | Sepsis_Deterioration (n = 11) | P | |
|---|---|---|---|---|
| Baseline | ||||
| Sex (male) | 29 (60.4) | 20 (54.1) | 6 (54.6) | 0.536 |
| Age (years) | 5.5 (3.1) | 6.3 (4.0) | 6.2 (4.2) | 0.270 |
| BMI | 16.2 [14.3, 18.1] | 15.2 [13.2, 17.6] | 16.3 [13.8, 17.7] | 0.166 |
| PICU stay | - | 18.1 (19.4) | 9.50 (10.2) | 0.165 |
| Clinical features | ||||
| pSOFA score | - | 6.5 [4.2, 11.0] | 15.0 [13.0, 16.0] | < 0.001 |
| PCIS score | - | 78.5 (8.5) | 71.3 (9.5) | 0.020 |
| pMODS score | - | 5.0 [4.0, 6.0] | 6.0 [4.0, 8.0] | 0.099 |
| PRISMIII | - | 7.0 [5.0, 10.5] | 19.0 [13.0, 21.0] | < 0.001 |
| Septic shock | - | 15 (40.5) | 9 (81.8) | 0.016 |
| OMV | - | 16 (43.2) | 10 (90.9) | 0.005 |
| Severe pneumonia | - | 12 (32.4) | 7 (63.6) | 0.085 |
| DIC | - | 9 (24.3) | 9 (81.8) | < 0.001 |
| Respiratory failure | - | 18 (48.6) | 11 (100) | 0.002 |
| Positive bacteria | - | 20 (54.1) | 6 (54.5) | 0.977 |
| Pathogens | 14 (37.8) | 5 (45.5) | 0.732 | |
| Comorbidities | ||||
| Cardiac insufficiency | - | 7 (18.9) | 10 (90.9) | < 0.001 |
| Renal insufficiency | - | 9 (24.3) | 6 (54.5) | 0.074 |
| Hypertension | - | 2 (5.4) | 0 (0) | 1.000 |
| NSD | - | 20 (54.1) | 6(54.5) | 0.977 |
Data are presented as median [interquartile range], mean (SD) or n (%). The Pathogens refer to the proportion of bacteria. Controls, non-infected, outpatient control. pSOFA, pediatric sequential organ failure assessment; PCIS, pediatric critical illness score; pMODS, pediatric multiple organ dysfunction score; PRISMIII, pediatric risk of mortality III; OMV, on mechanical ventilation; DIC, disseminated intravascular coagulation; NSD, nervous system disease
Alterations in the plasma lipidome of sepsis patients
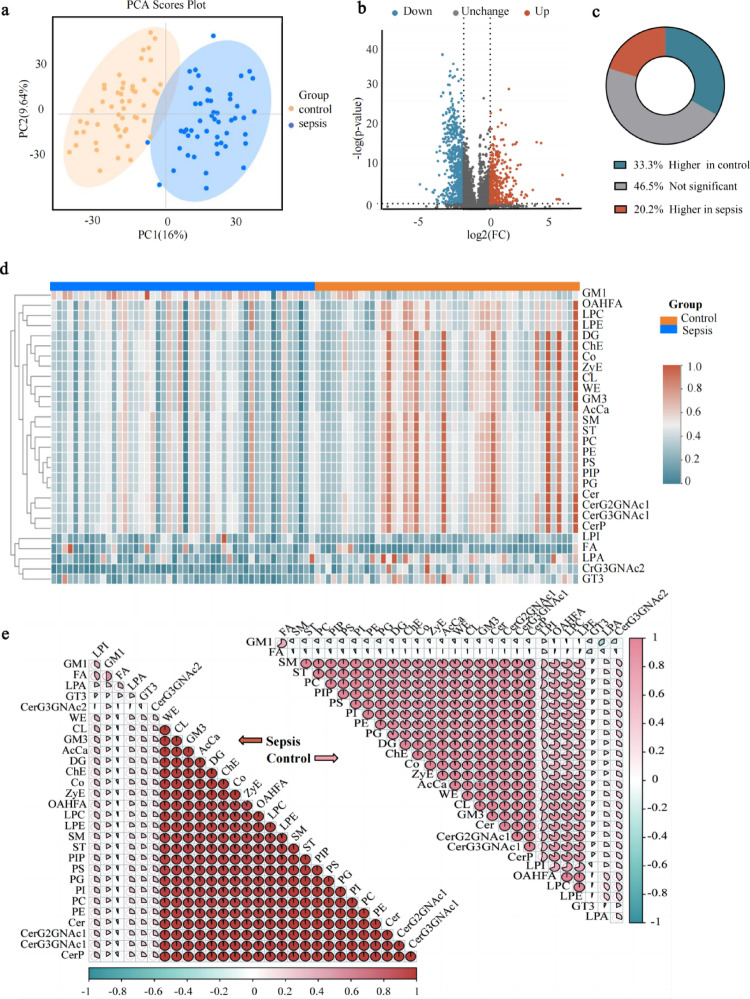
UHPLC-MS/MS technology was used to perform targeted detection of plasma lipid metabolites in the sepsis and control groups. After baseline filtering, peak recognition, extraction, retention time correction, peak alignment, and sum normalization, a total of 44 types of lipid classes and 4143 lipid species were obtained (Table S1). The QC results demonstrated good reproducibility in both positive and negative ion modes (Fig. S2). Significant differences in plasma lipid levels were observed between the sepsis and control groups (Fig. 1a). The majority of lipid species was significantly different between the groups, mostly including lipids with a decreased abundance in sepsis patients (33.3% decreased, 20.2% increased, 46.5% unchanged when compared to controls) (Fig. 1b, c). Specifically, lipid classes such as lysophosphatidic acid (LPA), trihexosyl di-N-acetylhexosyl ceramide (CerG3GNAc2) and trisialo trihexosyl (GT3) were significantly decreased, whereas monosialo tetrahexosyl ceramide (GM1) and free fatty acids (FA) exhibited a significant increase in the sepsis group (Fig. S3). Unsupervised hierarchical clustering analysis further underscored these differences, showing distinct patterns of lipid abundance between the sepsis and control groups (Fig. 1d).
Fig. 1.
Alterations in the Plasma Lipidome of Sepsis Patients. a PCA score plot of the control and sepsis groups. X-axis and Y-axis show the first and second principal component with the percentage of explained variance respectively, and the ellipse represents the 95% confidence interval. b Volcano plot of differential lipids based on univariate analysis.The x-axis represents the log2-transformed fold change values, the y-axis represents the log10-transformed P-values, and the points represent lipid species. c Pie chart of differential lipids based on univariate analysis. d Unsupervised hierarchical clustering heatmap of lipid classes in the control and sepsis groups (Only the lipids with differences were analyzed). e Correlation heatmap of lipid classes in the control and sepsis groups (Only the lipids with differences were analyzed)
Correlation analysis of lipid species
Correlation analysis revealed a high degree of positive correlation among lipid classes such as phosphatidylinositol (4) phosphate (PIP), phosphatidylserine (PS), phosphatidylglycerol (PG), phosphatidylinositol (PI), phosphatidylcholine (PC), phosphatidylethanolamine (PE), diglyceride (DG), cholesteryl ester (ChE), coenzyme Q (Co), zymosteryl ester (ZyE), ceramides (Cer), dihexosyl N-acetylhexosyl ceramide (CerG3GNAc1), ceramide phosphate (CerP), cardiolipin (CL), and TG in both groups. Interestingly, in the control group, LPA and LPC exhibited weaker correlations with other lipids compared to the sepsis group. Conversely, certain lipids, including GM1, FA, LPA, GT3, CerG3GNAc2, phosphatidylinositol (4, 5) bisphosphate (PIP2), and sialylated ester (SiE), demonstrated weak or negligible correlations in both groups (Fig. 1e).
Changes in plasma lipidome closely associated with disease severity
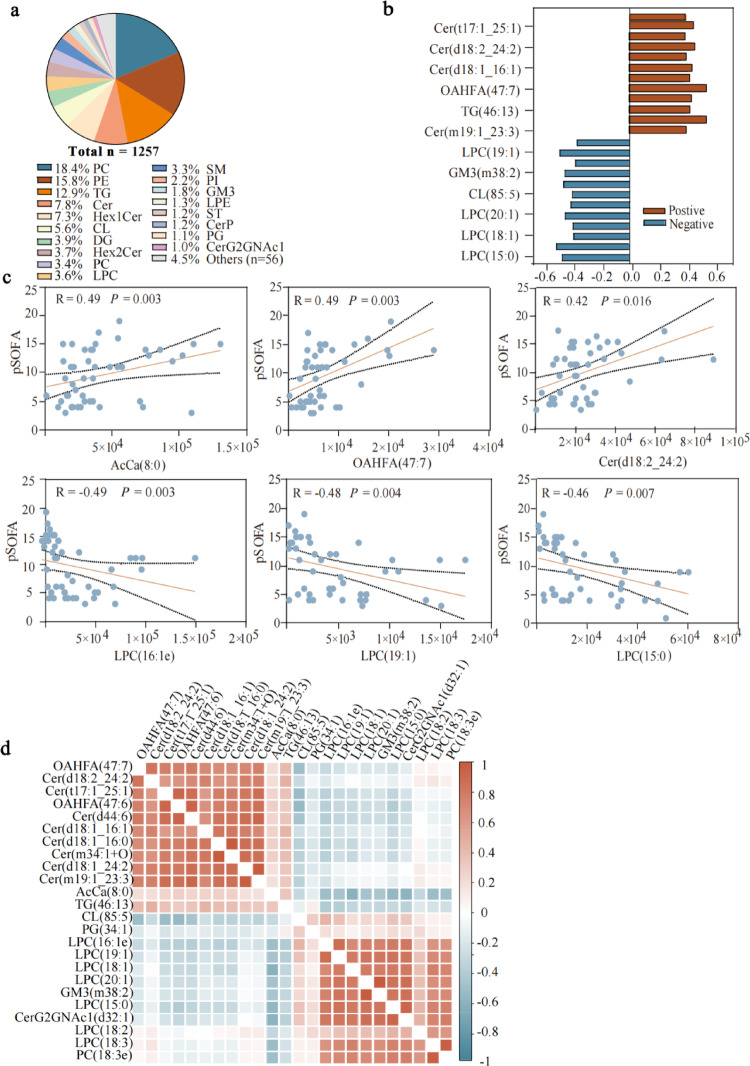
The PLS-DA model revealed a clear separation of plasma lipid profiles between the two groups, as demonstrated in the score plot (Fig. S4a). The model demonstrated strong performance with an R²Y of 0.975 and a Q²Y of 0.913, indicating excellent explanatory power and satisfactory predictive ability (R²Y ≥ 0.88) (Fig. S4b). By the cutoff of fold change > 1.5 or < 0.67, and VIP ≥ 1.0, a total of 1,257 differential lipids were identified in patients with sepsis compared to controls, including 231 PC (18.4%), 199 PE (15.8%), 163 TG (12.9%), 99 Cer (7.8%), and 91 hexosyl ceramides (Hex1 Cer) (7.3%) (Fig. 2a). Of these, 24 lipids demonstrated significant associations with pSOFA scores (Fig. 2b, Table S2). Among these, lipids positively correlated with pSOFA scores included acyl Carnitine (AcCa) (8:0) (R = 0.49, P = 0.003), (O-acyl)−1-hydroxy fatty acid (OAHFA) (47:7) (R = 0.49, P = 0.003), and Cer (18:2_24:2) (R = 0.42, P = 0.016). Conversely, lipid negatively correlated with pSOFA scores were LPC (16:1e) (R = −0.49, P = 0.003), LPC (19:1) (R = −0.48, P = 0.004) and LPC (15:0) (R = −0.46, P = 0.007) (Fig. 2c). Further correlation analysis among these 24 differential lipids showed that Cer was positively correlated with OAHFA and negatively correlated with TG, LPC, CL, and GM3 (Fig. 2d).
Fig. 2.
Changes in plasma lipidome closely associated with disease severity. a Pie chart of differential lipids of the control and sepsis groups based on PLS-DA results. b Bar plot of lipids correlated with pSOFA scores. c Scatter plot of lipids correlated with pSOFA scores. d Correlation heatmap of lipids correlated with pSOFA scores
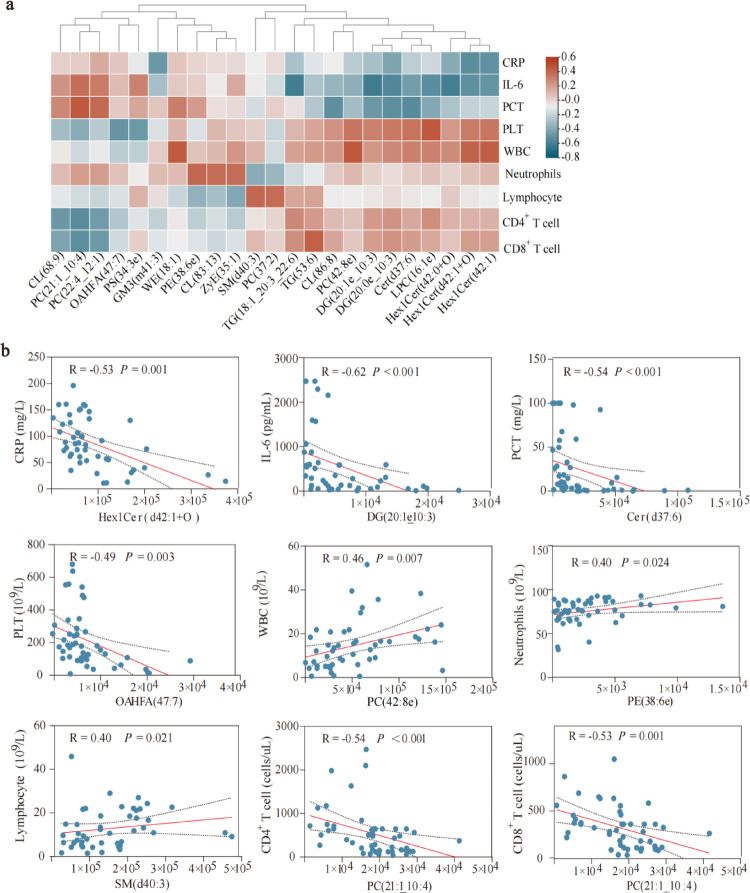
Host response to infection linked to changes in the plasma lipidome
No significant differences were observed in levels of WBC, CRP, IL-6, PCT and lactate between the recovery and deterioration groups among sepsis patients. However, the recovery group exhibited a significant increase in the absolute counts of CD3+, CD4+ and CD8+ T cells (Table 2). We performed a correlation analysis between 1,257 differential lipids and various inflammatory and cytokine markers (BH-adjusted p < 0.05). A heatmap was generated using lipids with statistically significant correlations. Specifically, 11 lipids were positively correlated with PLT, WBC, lymphocytes, neutrophils, CD4+ T, and CD8+ T cells, and negatively correlated with CRP, IL-6, and PCT. Conversely, four lipids showed positive correlations with CRP, IL-6, PCT, and neutrophils, and negative associations with WBC, PLT, lymphocytes, CD4+ T, and CD8+ T cells (Fig. 3a). Significant correlations were found between the following lipids and inflammatory markers: Hex1 Cer and CRP (d42:1 + O) (R = −0.53, P = 0.001), DG and IL-6 (20:1e_10:3) (R = −0.62, P < 0.001), Cer and PCT (d37:6) (R = −0.54, P < 0.001), OAHFA and PLT (47:7) (R = −0.49, P = 0.003), PC and WBC (42:8e) (R = 0.46, P = 0.007), PE and neutrophils (38:6e) (R = 0.40, P = 0.024), SM and lymphocytes (d40:3) (R = 0.40, P = 0.021), PC and CD4+ T cells, CD8+ T cells (21:1_10:4) (R = −0.54, P < 0.001) (R = −0.53, P = 0.001) (Fig. 3b).
Table 2.
Laboratory test results
| Routine Lab | Recovery (n = 37) | Deterioration (n = 11) | P |
|---|---|---|---|
| WBC, ×109/L | 15.5 (12.0) | 11.9 (7.3) | 0.354 |
| Platelet, ×109/L | 188.0 [111.5, 322.5] | 96.0 [42.0, 169.0] | 0.012 |
| Neutrophil, ×109/L | 80.4 [70.0, 88.2] | 79.1 [69.1, 91.5] | 0.844 |
| Lymphocyte, ×109/L | 10.9 [6.5, 20.2] | 12.4 [4.4, 21.7] | 0.713 |
| CD3+ T cell, cells/µL | 1078.0 [398.8, 1195.4] | 371.8 [147.7, 544.4] | 0.022 |
| CD4+ T cell, cells/µL | 562.9 [216.3, 681.9] | 166.8 [71.9, 289.9] | 0.004 |
| CD8+ T cell, cells/µL | 355.1 [162.0, 469.5] | 179.5 [90.3, 252.4] | 0.020 |
| CD19+ T cell, cells/µL | 486.9 [173.9, 660.0] | 394.2 [98.5, 497.3] | 0.144 |
| CRP, mg/L | 86.3 (45.6) | 88.5 (55.9) | 0.894 |
| PCT, mg/L | 7.9 [0.9, 42.1] | 10.0 [4.2, 28.3] | 0.677 |
| Lactate, mmol/L | 1.1 [0.9, 1.8] | 2.6 [0.7, 7.8] | 0.102 |
| IL-6, pg/mL | 286.7 [53.7, 605.2] | 357.6 [69.8, 887.8] | 0.548 |
| Total bilirubin, µmol/L | 6.6 [4.9, 12.0] | 17.2 [6.8, 93.0] | 0.019 |
| ALT, U/L | 17.6 [10.9, 29.7] | 54.0 [20.0, 421.5] | 0.008 |
| AST, U/L | 39.3 [23.4, 61.8] | 328.0 [44.7, 827.3] | 0.002 |
| D-dimers, µg/mL | 2.6 [1.4, 7.1] | 5.7 [2.7, 17.7] | 0.079 |
| Blood glucose, mmol/L | 6.7 [5.4, 7.7] | 8.4 [6.1, 11.3] | 0.062 |
| Vasoactive drugs | 13 (35.1) | 10 (90.9) | 0.001 |
Data are presented as median [interquartile range], mean (SD) or n (%). WBC, white blood cell; CRP, c-reactive protein; PCT, procaicitonin; IL-6, interleukin-6; ALT, alanine aminotransferase; AST, aspartate aminotransferase
Fig. 3.
Host response to infection linked to changes in the plasma lipidome. a Correlation heatmap of differential lipids and laboratory indicators. b Scatter plot of lipids correlated with laboratory indicators
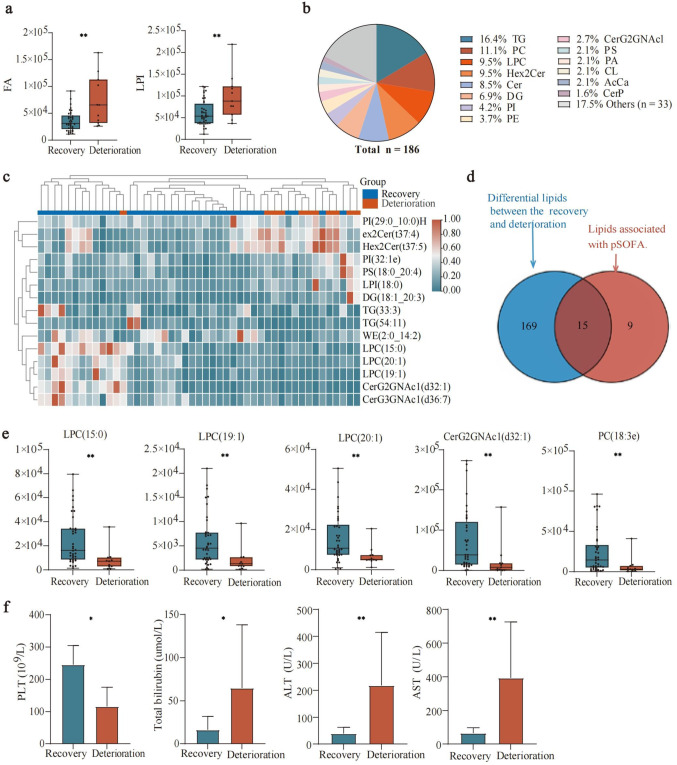
Alterations in the plasma lipidome of sepsis patients associated with a worse prognosis
Lipid class analysis revealed significant differences in FA and Lysophosphatidylinositol (LPI) levels (P < 0.05) between the recovery and deterioration groups, with both lipid classes elevated in the deterioration group (Fig. 4a). All lipids were compared between the sepsis deterioration and recovery groups, identifying a total of 186 differential lipids, with TGs accounting for the highest proportion at 16.4% (Fig. 4b). We performed unsupervised hierarchical clustering analysis on the top 15 lipids and found that PI (29:0_10:0), Hex2 Cer (t37:4), and Hex2 Cer (t35:5) exhibited an increasing trend in the deterioration group, while LPC (15:0), LPC (20:1), LPC (19:1), N-acetylglucosamine ceramide (CerG2GNAc1) (d32:1), and CerG3GNAc1 (d36:7) showed an increasing trend in the recovery group (Fig. 4c). Furthermore, 15 lipids with significant statistical differences were identified as differential lipid species through a comparison of those associated with pSOFA scores and those associated with sepsis prognosis (Fig. 4d). The top five differential lipids were LPC (15:0), LPC (19:1), LPC (20:1), CerG2GNAc1 (d32:1), and PC (18:3e) (Fig. 4e). In addition, clinical parameters demonstrated that patients in the deterioration group exhibited significantly lower platelet counts and higher levels of total bilirubin, alanine aminotransferase (ALT), and aspartate aminotransferase (AST) compared to the recovery group (Fig. 4f).
Fig. 4.
Alterations in the plasma lipidome of sepsis patients associated with a worse prognosis. a Boxplot of lipid classes in the sepsis recovery and deterioration groups. b Pie chart of differential lipids between the sepsis recovery and deterioration groups. c Unsupervised hierarchical clustering heatmap of differential lipids between the sepsis recovery and deterioration groups. d, e In all, 15 lipids with the significant statistical difference between the lipids associated with pSOFA score and those associated with sepsis prognosis were found in the Venn intersection of the comparison (The bar chart displays only the top five differential lipids). f Bar chart of laboratory indicators in the sepsis recovery and deterioration groups. Statistically significant differences are indicated, with *P < 0.05, **P < 0.01, ***P < 0.001
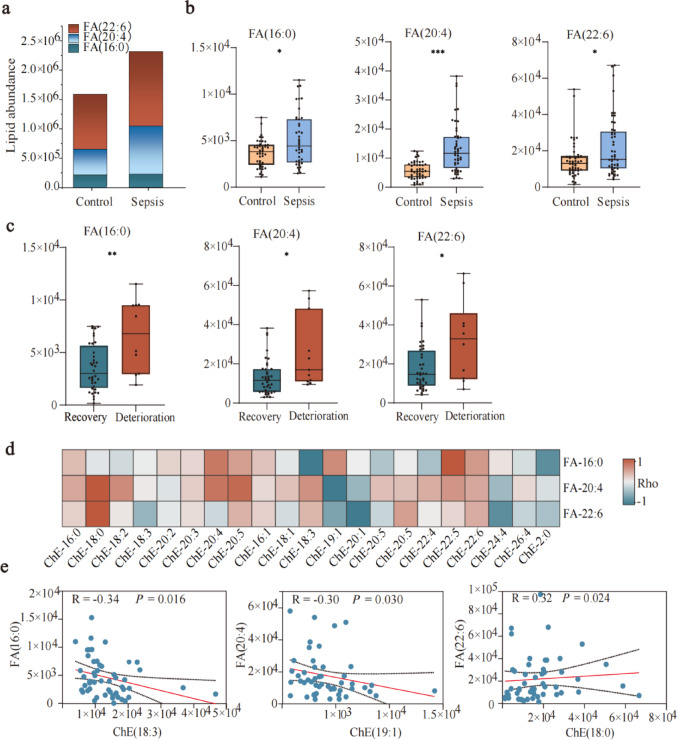
Changes in the fatty acid profile of sepsis patients
Analysis of the fatty acid profile revealed elevated levels of FA (16:0), FA (20:4), and FA (22:6) in the sepsis group compared to controls, with the most pronounced increase observed in FA (20:4) (P < 0.001) (Fig. 5a, b). Further subgroup analysis revealed distinct patterns of these fatty acids in relation to disease prognosis. Specifically, levels of FA (20:4), FA (16:0), and FA (22:6) were significantly higher in the deterioration group compared to the recovery group. FA (16:0) showed the most substantial elevation among the three fatty acids in patients with a worse prognosis (P < 0.01) (Fig. 5c). In addition, correlation analysis between FA and ChE demonstrated differing relationships. FA (16:0) and ChE (18:3), as well as FA (20:4) and ChE (19:1), were negatively correlated, whereas FA (22:6) and ChE (18:0) exhibited a positive correlation (Fig. 5d, e).
Fig. 5.
Changes in the fatty acid profile of sepsis patients. a Stacked bar chart of fatty acids comparing the control and sepsis groups. b Boxplots of fatty acids in the control and sepsis groups. c Boxplots of fatty acids in the recovery and deterioration groups. d Correlation heatmap of fatty acids and cholesterol esters in sepsis patients. e Scatter plot of fatty acids and cholesterol esters correlation. Statistically significant differences are indicated, with *P < 0.05, **P < 0.01, ***P < 0.001
Discussion
We conducted untargeted metabolomics analysis to explore lipid alterations in pediatric sepsis and found that most blood lipids were significantly decreased in sepsis patients compared to the control group at PICU admission. It was found specific lipids—like LPC and Cer—are strongly associated with immune regulation and inflammatory responses also to being related to higher pSOFA scores and a poorer outcome for sepsis.
Previous studies have demonstrated that most lipid species, including sphingolipids, PC, and DG, are significantly reduced in sepsis patients compared to controls (Han, 2016). Consistent with these results, we also found that a variety of LPA, CerG3GNAc2, and GT3 decreased significantly in the plasma of sepsis patients. LPA is a small glycerophospholipid molecule that exerts anti-inflammatory effects by reducing the release of inflammatory cytokines and complement activation, and its production is mediated by autotaxin, a plasma lysophospholipase D that hydrolyzes LPC to promote LPA’s anti-inflammatory action on macrophages (Mecatti et al., 2018). LPA prevents endotoxin-induced organ damage through G protein-coupled receptors and PPAR-γ, playing a protective role in renal ischemia, wound healing, and colitis (Murch et al., 2007). Therefore, the significant decrease in LPA during sepsis may exacerbate organ dysfunction. While these lipid alterations indeed reflect a state of systemic imbalance, the exact mechanisms by which they interact with the body and their specific roles remain unclear.
Other studies in adults have found that a significant feature of the lipid profile of CAP-sepsis is an increase in TG (Chouchane et al., 2024). However, in our study, we found that GM1 and FA were increased, while no significant differences in TG levels were observed between the sepsis group and the control group. Additionally, although TG had the highest proportion among the differential lipids between the recovery and deterioration groups in sepsis, the difference was not significant. A longitudinal cohort study found that the levels of most lipid subclasses, including TGs, increased with age. However, this trend disappeared after adjusting for BMI. Furthermore, large and small TGs exhibited distinct patterns, highlighting their different functional roles across the TGs spectrum (Hornburg et al., 2023). For instance, small TGs show distinct associations with certain cytokines and chemokines and are rapidly depleted during early respiratory viral infections (RVI), followed by a rapid recovery to baseline levels. Their depletion during infection suggests a crucial role in energy metabolism and signaling to support early inflammation. High plasma levels of TG relate to better survival in sepsis patients (Lee et al., 2015), possibly by dampening systemic damage caused by pathogens: TG-rich lipoproteins can bind and neutralize bacterial lipopolysaccharide, and enhance its clearance. Therefore, we suggest further investigation into the functions of small and large TGs, as well as inflammation-related changes in TG levels in pediatric sepsis.
The observed lipid alterations also parallel findings from previous studies on Cer levels. For instance, elevated Cer levels have been positively correlated with SOFA scores, reflecting disease severity and poor outcomes (Wu et al., 2019). Cers are not a single lipid entity but rather a family of signaling lipids that serve as central metabolites of the sphingolipid family. They form the lipid backbone to which a diverse array of headgroup structures is conjugated, making them essential components of organelles and cell membranes. Additionally, ceramides play a crucial role in signal transduction, regulating key physiological processes such as growth, differentiation, proliferation, migration, apoptosis, and cell death (Caterino et al., 2021). Cers also act as signaling molecules that regulate cellular processes such as endoplasmic reticulum (ER) stress, apoptosis, and insulin sensitivity, which in turn may affect whole-body physiology (Rossi et al., 2024).
Chang et al. reported significantly decreased LPC concentrations in sepsis-induced ARDS patients compared to non-ARDS controls, with higher LPC levels observed in patients with direct ARDS compared to those with indirect ARDS (Chang et al., 2023). Similarly, our study results showed that compared to the control group, LPC levels were decreased in sepsis patients and negatively correlated with pSOFA scores. LPC is a product of PC degradation mediated by phospholipase A2 (PLA2) and is closely associated with inflammation. Although LPC can be produced in the circulation when PLA2 cleaves PC, they can be converted back to PC by the enzyme lysophosphatidylcholine acyltransferase (LPCAT) in the presence of Acyl-CoA (Wang et al., 2012). Exogenous LPC induces pro-inflammatory effects such as upregulated gene expression for smooth muscle/fibroblast-directed growth factors and adhesion molecules in endothelial cells, increased release of IL-1β, IL-6, and TNF-α from adipocytes, enhanced secretion of interferon-γ from peripheral blood mononuclear leucocytes, and increased activation of B cells and macrophages (Law et al., 2019). Furthermore, it serves as a ligand for lymphocytes and can be depleted by conversion into anti-inflammatory LPA. Recent study suggested that LPC (16:0) predicts 28 day and 90-day mortality better than other LPC subtypes. Impaired metabolic homeostasis is considered a key factor contributing to the persistently low LPC levels in sepsis patients (Lee et al., 2020). These lower LPC levels have been associated with excessive inflammatory responses and worse clinical outcomes (Montague et al., 2022), underscoring the critical role of LPC in the pathophysiology of sepsis. Our findings further support the potential of LPC as a key biomarker for sepsis, with utility in diagnosis, prognosis prediction, and the identification of therapeutic targets.
In this study, we observed elevated FA levels in sepsis patients compared to controls, with further increases seen in patients experiencing sepsis deterioration. In adult patients, studies have also reported a close association between plasma polyunsaturated fatty acid concentrations and cholesterolemia, including low ChE and high FA levels (Rival et al., 2013). Previous studies have demonstrated that omega-3 FAs exert beneficial effects on health by lowering plasma cholesterol levels and serving as precursors for pro-resolving mediators, such as resolvins, protectins, and maresins (Hornburg et al., 2023). Furthermore, docosahexaenoic acid (DHA, FA 22:6) has been shown to regulate the composition of the gut microbiome, promote the production of catabolic mediators and anti-inflammatory factors, inhibit NF-κB activation, and influence the function of membrane lipid rafts (Zhongqiu & Jingjing, 2022). In contrast, omega-6 polyunsaturated fatty acids, such as arachidonic acid (ARA, FA 20:4), are generally associated with promoting inflammation. ARA, a component of cell membrane phospholipids, is released by phospholipase A2 and converted into thromboxanes, prostaglandins, and leukotrienes, initiating an inflammatory cascade by promoting leukocyte recruitment and thrombosis (Barber et al., 2023). Fish oil lipid emulsions are rich in omega-3 polyunsaturated fatty acids (PUFAs) and possess unique anti-inflammatory and immunomodulatory properties. Multiple studies have confirmed that they can reduce inflammatory cytokine levels in surgical patients, lower the incidence of infections, shorten hospital stays, and help preserve organ function (Zhang, 2023). Regulating the balance between omega-3 and omega-6 fatty acids can reduce excessive inflammation, and lipid supplementation may offer a novel adjunctive therapy to improve clinical outcomes in critically ill children.
Despite the significance of these findings, our study has several limitations. First, blood samples were collected at a single time point, specifically upon PICU admission. Although efforts were made to obtain samples as early as possible on the first day of diagnosis, variability in the timing of disease onset and clinical presentation may have introduced confounding factors. Second, the relatively small sample size and single-center design limit the generalizability of our findings. The absence of an independent validation cohort further constrains the robustness of our conclusions. Third, we were unable to perform dynamic monitoring of lipid levels over time, which restricts a comprehensive understanding of lipidomic alterations during the progression and resolution of sepsis. To address these limitations, future studies should employ larger, multi-center cohorts with longitudinal sampling to validate and expand upon our findings. A more detailed temporal analysis of lipidomic profiles may provide greater insights into the dynamic role of lipid metabolism in immune responses and sepsis outcomes.
In summary, this study is the first to exploratorily analyze the changes in the plasma lipidome of children during sepsis, revealing the overall and specific lipid alterations associated with sepsis severity and prognosis. Identifying lipid species associated with sepsis severity suggests that lipid profiling could aid in early risk stratification and prognosis prediction in the PICU, with LPC and FA emerging as promising biomarkers for distinguishing between mild and severe cases. Additionally, lipids such as DHA (FA 22:6) and LPC, known for their roles in inflammation resolution and immune modulation, could serve as potential therapeutic targets. Lipid-based interventions, such as lipid supplementation or metabolic modulation, may provide new avenues for adjunctive therapy. Ultimately, lipidomic analysis may contribute to a more personalized approach to sepsis treatment, enabling tailored interventions based on individual metabolic and lipidomic profiles. Future larger-scale, multicenter longitudinal studies are needed to further elucidate the dynamic changes in the lipid profile and its mechanistic role in the pathophysiology of sepsis.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The authors would like to thank the participants and their guardians, the collaborating clinicians, and other clinical staff. We would also like to acknowledge the Shanghai Zhongke New Life Biotechnology Co., Ltd for the support of sample analysis and relative quantification of lipids.
Author contributions
RA, JB, YC, and ZC collected and analyzed the patient data; WY, HZ, GS, RA, and YC performed the statistical analysis; KW, YW, and TL collected and processed clinical samples; YL, WC, RA, and JB wrote the manuscript; CZ, GL, and HC contributed to conceptualization, manuscript writing and editing, statistical analysis, and visualization. All authors have read and approved the final manuscript.
Funding
The study was supported by grants from the National Key R&D Program of China (2021YFC2701800 to G Lu), National Natural Science Foundation of China (82202374 to C Zhang), Natural Science Foundation of Shanghai (22ZR1408500 to C Zhang), Shanghai Municipal Science and Technology Major Project (ZD2021 CY001 to G Lu), Municipal Health System Key Supporting Discipline Project (2023ZDFC0103 to W Chen), Natural Science Foundation of Anhui (2308085MH267 to G Lu), Ningbo Medical and Health Brand Discipline (PPXK2024-06 to H Chen).
Data availability
The datasets used for the analysis in the current study are available from the corresponding author on reasonable request.
Declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
Ethical approval for the study was provided by Ethics Committee of Children’s Hospital of Fudan University ([2024] No. 116). Written informed consent was obtained from all parents or their surrogates of studied children.
Consent for publication
All authors consent to the publication of this manuscript.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reyihangu Awuti, Jiayi Bai and Ye Cheng contributed equally to this work.
Contributor Information
Hehe Chen, Email: hehebb0425@163.com.
Guoping Lu, Email: lgp@fudan.edu.cn.
Caiyan Zhang, Email: zhangcy17@fudan.edu.cn.
References
- Balamuth, F., Scott, H. F., Weiss, S. L., Webb, M., Chamberlain, J. M., Bajaj, L., Depinet, H., Grundmeier, R. W., Campos, D., Deakyne Davies, S. J., Simon, N. J., Cook, L. J., & Alpern, E. R. (2022). Validation of the pediatric sequential organ failure assessment score and evaluation of third international consensus definitions for Sepsis and septic shock definitions in the pediatric emergency department. JAMA Pediatr, 176(7), 672–678. 10.1001/jamapediatrics.2022.1301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barber, G., Tanic, J., & Leligdowicz, A. (2023). Circulating protein and lipid markers of early sepsis diagnosis and prognosis: A scoping review. Current Opinion in Lipidology, 34(2), 70–81. 10.1097/mol.0000000000000870 [DOI] [PubMed] [Google Scholar]
- Bauer, M., Gerlach, H., Vogelmann, T., Preissing, F., Stiefel, J., & Adam, D. (2020). Mortality in sepsis and septic shock in Europe, North America and Australia between 2009 and 2019- results from a systematic review and meta-analysis. Critical Care, 24(1), 239. 10.1186/s13054-020-02950-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cambiaghi, A., Díaz, R., Martinez, J. B., Odena, A., Brunelli, L., Caironi, P., Masson, S., Baselli, G., Ristagno, G., Gattinoni, L., de Oliveira, E., Pastorelli, R., & Ferrario, M. (2018). An innovative approach for the integration of proteomics and metabolomics data in severe septic shock patients stratified for mortality. Scientific Reports, 8(1), 6681. 10.1038/s41598-018-25035-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caterino, M., Gelzo, M., Sol, S., Fedele, R., Annunziata, A., Calabrese, C., Fiorentino, G., D’Abbraccio, M., Dell’Isola, C., Fusco, F. M., Parrella, R., Fabbrocini, G., Gentile, I., Andolfo, I., Capasso, M., Costanzo, M., Daniele, A., Marchese, E., Polito, R., Russo, R., Missero, C., Ruoppolo, M., & Castaldo, G. (2021). Dysregulation of lipid metabolism and pathological inflammation in patients with COVID-19. Scientific Reports, 11(1), 2941. 10.1038/s41598-021-82426-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang, Y., Yoo, H. J., Kim, S. J., Lee, K., Lim, C. M., Hong, S. B., Koh, Y., & Huh, J. W. (2023). A targeted metabolomics approach for sepsis-induced ARDS and its subphenotypes. Critical Care, 27(1), 263. 10.1186/s13054-023-04552-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chouchane, O., Schuurman, A. R., Reijnders, T. D. Y., Peters-Sengers, H., Butler, J. M., Uhel, F., Schultz, M. J., Bonten, M. J., Cremer, O. L., Calfee, C. S., Matthay, M. A., Langley, R. J., Alipanah-Lechner, N., Kingsmore, S. F., Rogers, A., van Weeghel, M., Vaz, F. M., & van der Poll, T. (2024). The plasma lipidomic landscape in patients with Sepsis due to Community-acquired pneumonia. American Journal of Respiratory and Critical Care Medicine, 209(8), 973–986. 10.1164/rccm.202308-1321OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- Expert consensus for the diagnosis and management of septic shock (infectious shock) in children (2015). Zhonghua Er Ke Za Zhi, 53(8), 576–580. [PubMed]
- Giustozzi, M., Ehrlinder, H., Bongiovanni, D., Borovac, J. A., Guerreiro, R. A., Gąsecka, A., Papakonstantinou, P. E., & Parker, W. A. E. (2021). Coagulopathy and sepsis: Pathophysiology, clinical manifestations and treatment. Blood Reviews, 50, 100864. 10.1016/j.blre.2021.100864 [DOI] [PubMed] [Google Scholar]
- Han, X. (2016). Lipidomics for studying metabolism. Nature Reviews. Endocrinology, 12(11), 668–679. 10.1038/nrendo.2016.98 [DOI] [PubMed] [Google Scholar]
- Hornburg, D., Wu, S., Moqri, M., Zhou, X., Contrepois, K., Bararpour, N., Traber, G. M., Su, B., Metwally, A. A., Avina, M., Zhou, W., Ubellacker, J. M., Mishra, T., Schüssler-Fiorenza Rose, S. M., Kavathas, P. B., Williams, K. J., & Snyder, M. P. (2023). Dynamic lipidome alterations associated with human health, disease and ageing. Nat Metab, 5(9), 1578–1594. 10.1038/s42255-023-00880-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law, S. H., Chan, M. L., Marathe, G. K., Parveen, F., Chen, C. H., & Ke, L. Y. (2019). An updated review of lysophosphatidylcholine metabolism in human diseases. International Journal of Molecular Sciences. 10.3390/ijms20051149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, E. H., Shin, M. H., Park, J. M., Lee, S. G., Ku, N. S., Kim, Y. S., Park, M. S., Pyun, J. C., & Chung, K. S. (2020). Diagnosis and mortality prediction of sepsis via lysophosphatidylcholine 16: 0 measured by MALDI-TOF MS. Scientific Reports, 10(1), 13833. 10.1038/s41598-020-70799-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, S. H., Park, M. S., Park, B. H., Jung, W. J., Lee, I. S., Kim, S. Y., Kim, E. Y., Jung, J. Y., Kang, Y. A., Kim, Y. S., Kim, S. K., Chang, J., & Chung, K. S. (2015). Prognostic implications of serum lipid metabolism over time during Sepsis. Biomed Research International, 2015, 789298. 10.1155/2015/789298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, Q., Cui, Y., Xu, B., Wang, Y., Lv, F., Li, Z., Li, H., Chen, X., Peng, X., Chen, Y., Wu, E., Qu, D., Jian, Y., & Si, H. (2021). Main active components of Jiawei gegen Qinlian Decoction protects against ulcerative colitis under different dietary environments in a gut microbiota-dependent manner. Pharmacological Research, 170, 105694. 10.1016/j.phrs.2021.105694 [DOI] [PubMed] [Google Scholar]
- Mecatti, G. C., Messias, F., Sant’Anna Paiola, M. C., Angolini, R. M. F., da Silva Cunha, C. F., Eberlin, I. B., M. N., & de Oliveira Carvalho, P. (2018). Lipidomic profiling of plasma and erythrocytes from septic patients reveals potential biomarker candidates. Biomark Insights, 13, 1177271918765137. 10.1177/1177271918765137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montague, B., Summers, A., Bhawal, R., Anderson, E. T., Kraus-Malett, S., Zhang, S., & Goggs, R. (2022). Identifying potential biomarkers and therapeutic targets for dogs with sepsis using metabolomics and lipidomics analyses. PLoS One, 17(7), e0271137. 10.1371/journal.pone.0271137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murch, O., Collin, M., & Thiemermann, C. (2007). Lysophosphatidic acid reduces the organ injury caused by endotoxemia-a role for G-protein-coupled receptors and peroxisome proliferator-activated receptor-gamma. Shock (Augusta, Ga.), 27(1), 48–54. 10.1097/01.shk.0000235086.63723.7e [DOI] [PubMed] [Google Scholar]
- Pandey, S. (2024a). Advances in metabolomics in critically ill patients with Sepsis and septic shock. Clin Exp Emerg Med. 10.15441/ceem.24.211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pandey, S. (2024b). Sepsis, management & advances in metabolomics. Nanotheranostics, 8(3), 270–284. 10.7150/ntno.94071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rival, T., Cinq-Frais, C., Silva-Sifontes, S., Garcia, J., Riu, B., Salvayre, R., Genestal, M., & Caspar-Bauguil, S. (2013). Alteration of plasma phospholipid fatty acid profile in patients with septic shock. Biochimie, 95(11), 2177–2181. 10.1016/j.biochi.2013.08.006 [DOI] [PubMed] [Google Scholar]
- Rossi, A., Ruoppolo, M., Fedele, R., Pirozzi, F., Rosano, C., Auricchio, R., Melis, D., Strisciuglio, P., Oosterveer, M. H., Derks, T. G. J., Parenti, G., & Caterino, M. (2024). A specific serum lipid signature characterizes patients with glycogen storage disease type Ia. Journal of Lipid Research, 65(10), 100651. 10.1016/j.jlr.2024.100651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez-Pinto, L. N., Bennett, T. D., DeWitt, P. E., Russell, S., Rebull, M. N., Martin, B., Akech, S., Albers, D. J., Alpern, E. R., Balamuth, F., Bembea, M., Chisti, M. J., Evans, I., Horvat, C. M., Jaramillo-Bustamante, J. C., Kissoon, N., Menon, K., Scott, H. F., Weiss, S. L., Wiens, M. O., Zimmerman, J. J., Argent, A. C., Sorce, L. R., Schlapbach, L. J., Watson, R. S., Biban, P., Carrol, E., Chiotos, K., De Oliveira, F., Hall, C., Inwald, M. W., Ishimine, D., Levin, P., Lodha, M., Nadel, R., Nakagawa, S., Peters, S., Randolph, M. J., Ranjit, A. G., Souza, S., Tissieres, D. C., P., & Wynn, J. L. (2024). Development and validation of the Phoenix criteria for pediatric Sepsis and septic shock. Jama, 331(8), 675–686. 10.1001/jama.2024.0196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlapbach, L. J., Straney, L., Bellomo, R., MacLaren, G., & Pilcher, D. (2018). Prognostic accuracy of age-adapted SOFA, SIRS, PELOD-2, and qSOFA for in-hospital mortality among children with suspected infection admitted to the intensive care unit. Intensive Care Medicine, 44(2), 179–188. 10.1007/s00134-017-5021-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siddiqui, M. A., Pandey, S., Azim, A., Sinha, N., & Siddiqui, M. H. (2020). Metabolomics: An emerging potential approach to Decipher critical illnesses. Biophysical Chemistry, 267, 106462. 10.1016/j.bpc.2020.106462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taguchi, R., & Ishikawa, M. (2010). Precise and global identification of phospholipid molecular species by an orbitrap mass spectrometer and automated search engine lipid search. Journal of Chromatography A, 1217(25), 4229–4239. 10.1016/j.chroma.2010.04.034 [DOI] [PubMed] [Google Scholar]
- Tian, T., Mao, Q., Xie, J., Wang, Y., Shao, W. H., Zhong, Q., & Chen, J. J. (2022). Multi-omics data reveals the disturbance of glycerophospholipid metabolism caused by disordered gut microbiota in depressed mice. Journal of Advanced Research, 39, 135–145. 10.1016/j.jare.2021.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, L., Shen, W., Kazachkov, M., Chen, G., Chen, Q., Carlsson, A. S., Stymne, S., Weselake, R. J., & Zou, J. (2012). Metabolic interactions between the lands cycle and the Kennedy pathway of glycerolipid synthesis in Arabidopsis developing seeds. The Plant Cell, 24(11), 4652–4669. 10.1105/tpc.112.104604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, T., Fu, X., Chen, Q., Patra, J. K., Wang, D., Wang, Z., & Gai, Z. (2019). Arachidonic acid metabolism and kidney inflammation. International Journal of Molecular Sciences. 10.3390/ijms20153683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss, S. L., & Fitzgerald, J. C. (2024). Pediatric Sepsis diagnosis, management, and Sub-phenotypes. Pediatrics. 10.1542/peds.2023-062967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, X., Hou, J., Li, H., Xie, G., Zhang, X., Zheng, J., Wang, J., Gao, F., Yao, Y., Liu, H., & Fang, X. (2019). Inverse correlation between plasma Sphingosine-1-Phosphate and ceramide concentrations in septic patients and their utility in predicting mortality. Shock (Augusta, Ga.), 51(6), 718–724. 10.1097/shk.0000000000001229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, L. (2023). Interpretation of the Chinese guidelines for parenteral and enteral nutrition in adult patients (2023 Edition). China Journal of Clinical Nutrition. (in Chinese).
- Zhang, Y. Y., & Ning, B. T. (2021). Signaling pathways and intervention therapies in sepsis. Signal Transduct Target Ther, 6(1), 407. 10.1038/s41392-021-00816-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhongqiu, L., & Jingjing, N. (2022). Pay attention to nutritional treatment of sepsis. Zhejiang Medicine,44(04), 339–344. 10.12056/j.issn.1006. in Chinese. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used for the analysis in the current study are available from the corresponding author on reasonable request.