Abstract
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin disease that significantly affects elderly patients, particularly those with multiple comorbidities. Tralokinumab, an IL- 13-neutralizing monoclonal antibody, is approved at 300 mg every 2 weeks (Q2 W), with the option to optimize to a once-monthly (Q4 W) regimen in patients achieving optimal disease control. This study evaluates its long-term efficacy and safety in elderly patients (> 65 years) and explores predictors of treatment response and optimization.
Methods
A retrospective multicenter study was conducted across four Spanish hospitals, including patients > 65 years old with moderate-to-severe AD treated with tralokinumab. The primary endpoints were treatment efficacy and safety at weeks 16, 24, and 52, while secondary endpoints included identifying predictors of treatment response and successful dose optimization.
Results
A total of 24 patients (mean age 75.3 ± 8.4 years, 45.8% male) were included. A significant reduction in EASI (Eczema Area and Severity Index), Pruritus NRS (Numerical Rating Scale), DLQI (Dermatology Life Quality Index), and BSA (Body Surface Area) scores was observed at weeks 16, 24, and 52 (p < 0.05). Dose optimization to 300 mg Q4 W was achieved in 25% of patients. Male sex (p = 0.042) and higher baseline EASI (p = 0.004) were associated with poorer early response, whereas a shorter time to systemic treatment initiation (p = 0.023) increased the likelihood of dose optimization. No serious adverse events or neoplasm progression were reported.
Conclusion
Tralokinumab demonstrated sustained efficacy and safety in elderly patients with moderate-to-severe AD, even with multiple comorbidities. Dose optimization to Q4 W was feasible in a subset of patients. Early systemic treatment initiation was linked to better outcomes, emphasizing the need for proactive disease management.
Keywords: Atopic dermatitis, Tralokinumab, Elderly patients, Optimization
Key Summary Points
| Comorbidities pose significant challenges in managing moderate-to-severe atopic dermatitis (AD) in elderly patients. |
| Expert consensus supports prioritizing biologics in elderly patients with AD because of their safer profile. |
| Tralokinumab maintains efficacy in elderly patients, even in those with significant comorbidities. |
| Flexible dosing with tralokinumab improves convenience while maintaining long-term efficacy. |
Introduction
Atopic dermatitis (AD) is a chronic and debilitating inflammatory skin condition. In recent years, biologics and JAK inhibitors have emerged as key systemic therapies to control flares and potentially reach a Minimal Disease Activity (MDA) status in individuals with moderate-to-severe AD [1]. For patients who achieve optimal outcomes with biological therapy, reducing the dosing frequency (optimization) may be a viable consideration [2, 3]. This approach holds particular significance for elderly individuals (> 65 years), who are often on multiple medications and have several comorbidities [2, 3]. Tralokinumab is a fully human IgG4 high-affinity monoclonal antibody that specifically neutralizes IL- 13 and is currently approved for the treatment of moderate-to-severe AD in adults and adolescents > 12 years old [4, 5]. Approved dosing regimens include 300 mg every 2 weeks (Q2 W) as well as a reduced dosing frequency every 4 weeks (Q4 W), which clinicians may consider for patients who achieve clear or almost clear skin at Week 16 with the initial Q2 W regimen [4]. The efficacy and safety of tralokinumab have been demonstrated in real-world series [6, 7] and ECZTRA trials [4, 5, 8]; however, real-world data remain limited, particularly in multi-medicated elderly patients [7]. The aim of the study is to evaluate the efficacy and safety of tralokinumab in patients > 65 years, identifying potential predictors of early response and treatment optimization to Q4 W.
Methods
A retrospective, non-interventional multicenter study was conducted at the dermatology departments of four Spanish hospitals from June 2022 to December 2024. It included elderly patients (> 65 years) with moderate-to-severe AD treated with tralokinumab in a daily practice setting. The collected data are presented in Table 1. Disease severity was measured using the EASI score (Eczema Area and Severity Index), BSA (body surface area), Pruritus NRS (numerical rating scale), and DLQi (Dermatology Life Quality Index). IGA (Investigator’s Global Assessment) data were excluded because of missing information. EASI 75 and EASI 90 response rates were also determined. The primary endpoint was to analyze the efficacy and safety of tralokinumab at weeks 16, 24, and 52 (when available). Secondary objectives included analyzing the influence of variables such as age, sex, BMI, number of comorbidities, atopic comorbidities, duration of AD, time from diagnosis to systemic therapy initiation, and baseline EASI on the EASI response at week 16 as well as the possibility of treatment optimization to 300 mg Q4 W. The normality of variables was assessed using the Shapiro-Wilk test. A univariate analysis was performed to evaluate the influence of the previously mentioned independent variables (age, sex, BMI, number of comorbidities, atopic comorbidities, duration of AD, time from diagnosis to systemic therapy initiation) on the EASI response at week 16 and the possibility of optimizing tralokinumab. Statistical significance was defined as p < 0.05. The study was conducted in compliance with national regulations and ethical principles governing biomedical research in Spain. According to the national legislation (Law 14/2007 on Biomedical Research and the EU General Data Protection Regulation [GDPR] 2016/679), research involving anonymized data may be exempt from requiring Institutional Review Board (IRB) approval. In this case, due to the full anonymization of the data, an exemption was granted in accordance with the applicable regulations. The study ensured compliance with all relevant ethical and data protection guidelines.
Table 1.
Demographic data
| N = 24 | |
|---|---|
| Age (years) (mean, SD) | 75.3 ± 8.4 |
| Females, number (%) | 13 (54.2) |
| Males, number (%) | 11 (45.8) |
| BMI (kg/m2) | 28.4 ± 4.1 |
| Number of comorbidities (mean, SD) | 3.3 ± 1.8 |
| Number of concomitant drugs | 4.8 ± 3.2 |
| Atopic comorbidities (asthma, rhinitis, eosinophilic esophagitis, and/or conjunctivitis) | 20.8% |
| Duration of AD (years) (mean, SD) | 5.9 ± 3.6 |
| Time to initiation of systemic therapy (years) (mean, SD) | 3.8 ± 3 |
| Involvement of special areas (%) | |
| Hands | 25 |
| Face and/or neck | 12.5 |
| Genital | 0 |
| Mean exposure time to tralokinumab (days) (mean, SD) | 413.8 ± 194.8 |
| Previous systemic treatments (%) | |
| Methotrexate | 12.5 |
| Cyclosporin | 8.3 |
| Azathioprine | 0 |
| Concomitant treatments (%) | 45.8% |
| Dose optimization to 300 mg Q4 W (%) | 25% |
BMI body mass index, SD standard deviation, Q4 W once monthly
Results
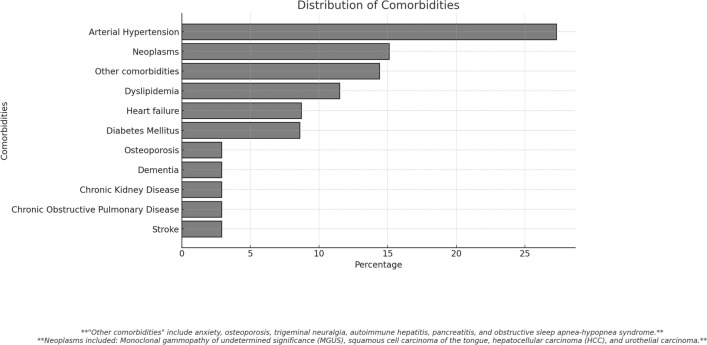
A total of 24 patients were included in the study. The mean age of the patients was 75.3 ± 8.4 years, and 45.8% were male. Patients had an average of 3.3 ± 1.8 comorbidities and took 4.8 ± 3.2 concomitant medications. The sociodemographic characteristics of the cohort are detailed in Table 1. Regarding the predominant AD subtypes, 41.6% of the patients presented with generalized lesions, followed by nummular eczema (29.2%) and prurigo (29.2%). Additionally, 33.3% had involvement of special areas. Six patients presented hand eczema, two of them with the dyshidrotic subtype, and achieved optimal control. Patch testing was performed in 84% of the patients, yielding positive results in only two cases: one patient reacted to a mix of fragrances II and Lyral and the other to potassium dichromate. Only one patient had received prior biologic therapy with dupilumab. Concomitant treatments included topical corticosteroids (TCS) in seven patients, systemic corticosteroids (SCS) in three patients, and cyclosporine in one patient. Arterial hypertension (defined as blood pressure ≥ 140/90 mmHg) was the most common comorbidity, reported in 27.3% of patients, followed by neoplasms (15.1%) and dyslipidemia (11.5%). AD comorbidities (asthma, rhinitis, eosinophilic esophagitis, and/or conjunctivitis) were reported in 20.8% of patients. The comorbidity profile is detailed in Fig. 1.
Fig. 1.
Comorbidity profile of the patients
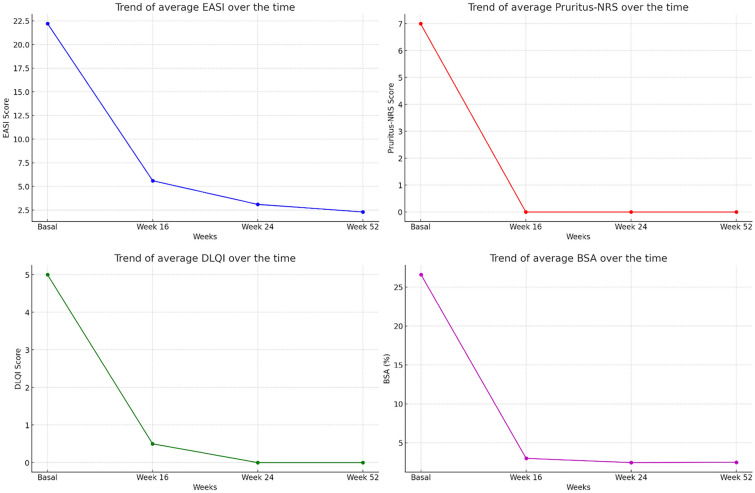
A significant reduction in EASI, pruritus NRS, DLQI, and BSA scores was observed at weeks 16, 24, and 52 (Table 2 and Fig. 2). Dose optimization to 300 mg Q4 W was achieved in 25% of the patients. Another patient was initially started on an optimized dose (Q4 W), experienced a loss of response, and later achieved complete clearance and pruritus control on a 300 mg Q2 W regimen. Male sex (p = 0.042) and a higher baseline EASI score (p = 0.004) were associated with higher EASI values at week 16, while the prurigo subtype of AD approached significance (p = 0.068). A shorter time to initiation of systemic therapy (p = 0.023) was associated with a higher likelihood of dose optimization, while a lower BMI (p = 0.099) showed a trend toward significance. Other variables studied were not significantly associated with an increased likelihood of achieving an optimized dose. No mild or severe adverse events were observed during the follow-up period. No neoplasm progression was reported. Three patients with moderate asthma did not experience any worsening of their condition during the follow-up period.
Table 2.
Response to tralokinumab treatment during the follow-up period
| Basal | Week 16 (N = 24) | Week 24 (N = 22) | Week 52 (N = 16) | |
|---|---|---|---|---|
| EASI (mean ± SD) | 22.2 ± 7.4 | 5.6 ± 8.9 | 3.1 ± 7.7 | 2.3 ± 6.0 |
| Pruritus-NRS (median ± IQR) | 7 ± 1 | 0 ± 2.5 | 0 ± 1 | 0 ± 0.8 |
| DLQI (median ± IQR) | 5 ± 3.8 | 0.5 ± 2.8 | 0 ± 1 | 0 ± 0.8 |
| BSA (mean ± SD) | 26.6 ± 17.1 | 3.0 ± 5.8 | 2.46 ± 7.42 | 2.5 ± 7.5 |
| EASI 75 response (%) | 69.6 | 88.2 | 90.9 | |
| EASI 90 response (%) | 47.8 | 64.7 | 81.8 |
SD standard deviation, IQR interquartile range, BSA body surface area, NRS numerical rating scale, EASI Eczema Area and Severity Index, DLQI dermatology life quality index
Fig. 2.
Tralokinumab effectiveness during the follow-up period
Discussion
Comorbidities present significant challenges to managing moderate-to-severe AD in patients over 65 years old [7]. Acceptance of advanced therapies for this population is often limited among both dermatologists and family members. However, expert consensus emphasizes the importance of prioritizing biological therapies in elderly patients with AD because of their safer profile and minimal drug interactions [9]. Post hoc analyses from the phase III ECZTRA 1 and 2 trials have demonstrated that tralokinumab maintains comparable efficacy in older adults and younger cohorts [7]. Efficacy in terms of reducing the EASI, DLQI, and P-NRS was sustained in the medium and long term, even in patients with significant comorbidities, as observed in our cohort. Potestio et al. [7] further support these findings in elderly patients with diverse comorbidities. In our series, the time to achieve an optimal response was independent of AD subtype, although patients with prurigo-like lesions experienced a slightly slower response. As reported by Hagino et al. [10], in our study, factors such as a higher baseline EASI score and male gender could also be associated with a slower initial response at week 16. The possibility of dose flexibility supported by the labeled information could be a distinguishing factor [2–4]. Data from the ECZTRA trials suggest that patients achieving a stable response at Week 16, defined as an IGA score of 0/1 and pruritus NRS < 3 on tralokinumab Q2 W, can safely transition to a once-monthly regimen (Q4 W), maintaining efficacy rates above 72% through Week 52 [8]. This approach improves treatment convenience and minimizes drug-drug interactions, which is particularly important in patients on multiple medications. Furthermore, patients who lost clinical response after switching to Q4 W were able to regain it within a median of 4.4 weeks upon reverting to Q2 W dosing [2], a fact observed in one of our patients. This finding aligns with the results from Weidinger et al. [3], where 94.6% of patients who experienced a loss of response on a Q4 W regimen successfully regained it after returning to the Q2 W dose. Regarding safety, no severe adverse events or reactivations of pre-existing neoplasms were reported in our cohort, including patients with a history of malignancy. An earlier initiation of advanced systemic therapy was associated with better outcomes in our series. These findings highlight the importance of early and proactive management of AD in older patients. The main limitations of the study include its retrospective design, the lack of a control group, and the small sample size.
Conclusion
Based on our results, tralokinumab appears to be a safe and effective therapeutic option for patients > 65 years old with moderate-to-severe AD, including those with multiple comorbidities. Treatment flexibility through reduced dosing frequency to Q4 W is feasible and maintains efficacy in patients with stable disease control.
Acknowledgements
We thank the participants of the study.
Author Contributions
F.J. Melgosa Ramos, S.Santos Alarcón, J.M. Sánchez Motilla, and P. Mercader García have equally contributed to the conception, design, data collection, interpretation, writing-draft preparation, and review of the present work. The final version to be published has been approved by all the authors.
Funding
No funding or sponsorship was received for this study or publication of this article.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Conflict of Interest
Francisco Javier Melgosa Ramos has received honoraria and/or travel grants and/or has acted as an advisory board member for Novartis, Abbvie, Janssen Cilag, UCB, Lilly, LEO Pharma, L’Oreal, Sanofi, Almirall, and Amgen. Sergio Santos Alarcón has received honoraria and/or travel grants and/or has acted as an advisory board member for Almirall, Abbvie, Adium Pharma, Amgen, Pfizer, Novartis, Janssen-Cilag, Lilly, Leo Pharma, UCB Pharma, Pierre Fabre, Isdin, Sanofi, and Viñas. Jose María Sánchez Motilla has received honoraria and/or travel grants and/or has acted as an advisory board member for Sanofi, Leo Pharma, Abbie, Pfizer, Lilly, Almirall, and Novartis. Pedro Mercader has received honoraria and/or travel grants and/or has acted as an advisory board member for Sanofi, Leo Pharma, Lilly, Almirall, and Abbvie.
Ethical Approval
The study was conducted in compliance with national regulations and ethical principles governing biomedical research in Spain. According to the national legislation (Law 14/2007 on Biomedical Research and the EU General Data Protection Regulation [GDPR] 2016/679), research involving anonymized data may be exempt from requiring Institutional Review Board (IRB) approval. In this case, due to the full anonymization of the data, an exemption was granted in accordance with the applicable regulations. The patient in this manuscript has given informed consent to the publication of their case details.
References
- 1.Silverberg JI, Gooderham M, Katoh N, Aoki V, Pink AE, Binamer Y, et al. Combining treat-to-target principles and shared decision-making: International expert consensus-based recommendations with a novel concept for minimal disease activity criteria in atopic dermatitis. J Eur Acad Dermatol Venereol. 2024;38(11):2139–48. [DOI] [PubMed] [Google Scholar]
- 2.Carrascosa JM. Optimizing dosing in atopic dermatitis therapy: Looking for the cream of the crop. Br J Dermatol. 2024. 10.1093/bjd/ljae514. [DOI] [PubMed] [Google Scholar]
- 3.Weidinger S, Bewley A, Hong HC, Silvestre JF, Peris K, Wollenberg A, et al. Predicting success with reduced dosing frequency of tralokinumab in patients with moderate-to-severe atopic dermatitis. Br J Dermatol. 2024. 10.1093/bjd/ljae439. [DOI] [PubMed] [Google Scholar]
- 4.Simpson EL, Pink AE, Blauvelt A, Gooderham M, Armstrong AW, Worm M, Katoh N, Peris K, Puig L, Barbarot S, Mark T, Steffensen LA, Tindberg AM, Wollenberg A. Tralokinumab efficacy over 1 year in adults with moderate-to-severe atopic dermatitis: pooled data from two phase III trials. Am J Clin Dermatol. 2023;24(6):939–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Paller AS, Flohr C, Cork M, Bewley A, Blauvelt A, Hong HC, et al. Efficacy and safety of tralokinumab in adolescents with moderate to severe atopic dermatitis: the phase 3 ECZTRA 6 randomized clinical trial. JAMA Dermatol. 2023;159(6):596–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pereyra-Rodríguez JJ, Herranz P, Ruiz-Villaverde R, Elosua-González M, Galán-Gutiérrez M, Figueras-Nart I, et al. Treatment of severe atopic dermatitis with tralokinumab in clinical practice: short-term effectiveness and safety results. Clin Exp Dermatol. 2023;48(9):991–7. [DOI] [PubMed] [Google Scholar]
- 7.Potestio L, Patruno C, Dastoli S, Brescia C, Napolitano M. Tralokinumab for the treatment of adult atopic dermatitis in special populations. J Asthma Allergy. 2024;17:791–9. 10.2147/JAA.S474411.PMID:39161703;PMCID:PMC11330743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blauvelt A, Langley RG, Lacour JP, Toth D, Laquer V, Beissert S, et al. Long-term 2-year safety and efficacy of tralokinumab in adults with moderate-to-severe atopic dermatitis: Interim analysis of the ECZTEND open-label extension trial. J Am Acad Dermatol. 2022;87(4):815–24. [DOI] [PubMed] [Google Scholar]
- 9.Adam DN, Gooderham MJ, Beecker JR, Hong CH, Jack CS, Jain V, et al. Expert consensus on the systemic treatment of atopic dermatitis in special populations. J Eur Acad Dermatol Venereol. 2023;37(6):1135–48. [DOI] [PubMed] [Google Scholar]
- 10.Hagino T, Saeki H, Fujimoto E, Kanda N. Identification of early and late responders to Anti-IL-13 antibody tralokinumab in atopic dermatitis: a real-world japanese study. Dermatitis. 2024. 10.1089/derm.2024.0460. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.