Abstract
Since their discovery in 2007, there has been growing awareness of the importance of fungal extracellular vesicles (EVs) for fungal physiology, host–pathogen interactions and virulence. Fungal EVs are nanostructures comprising bilayered membranes and molecules of various types that participate in several pathophysiological processes in fungal biology, including secretion, cellular communication, immunopathogenesis and drug resistance. However, many questions remain regarding the classification of EVs, their cellular origin, passage across the cell wall, experimental models for functional and compositional analyses, production in vitro and in vivo and biomarkers for EVs. Here, we discuss gaps in the literature of fungal EVs and identify key questions for the field. We present the history of fungal EV discovery, discuss five major unanswered questions in fungal EV biology and provide future perspectives for fungal EV research. We primarily focus our discussion on human fungal pathogens, but also extend it to include knowledge of other fungi, such as plant pathogens. With this Perspective we hope to stimulate new approaches and expand studies to understand the biology of fungal EVs.
Extracellular vesicles (EVs) are produced by all major classes of organism studied to date1. Fungal EVs—as in other organisms—are bilayered membrane structures comprising several types of biomolecules, including proteins, polysaccharides, RNA, pigments and small molecules (Fig. 1).
Fig. 1 |. Composition of fungal EVs.
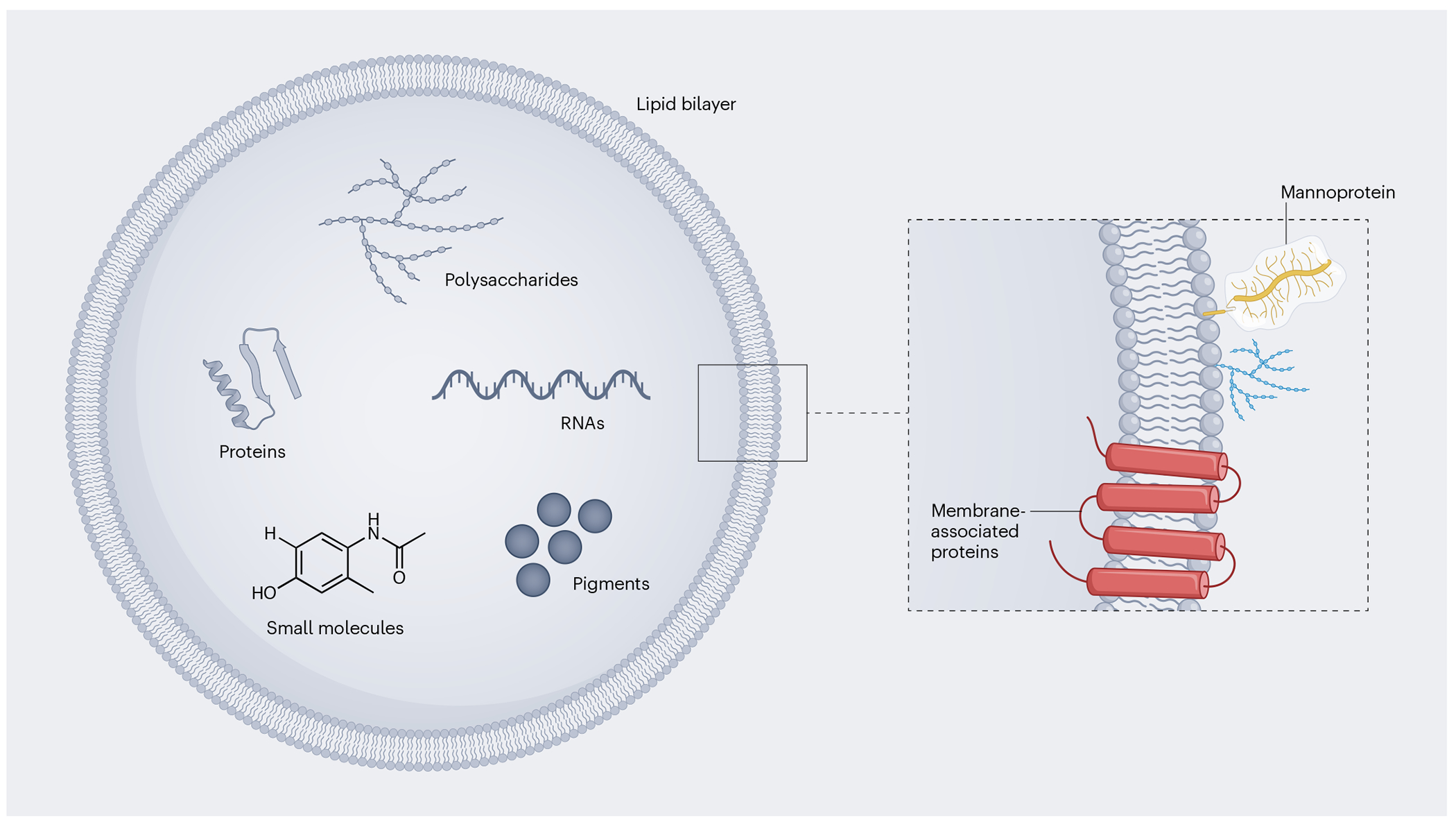
Fungal EVs can carry a wide variety of biomolecules, including proteins, polysaccharides, RNA, pigments and small molecules. The membrane structure of fungal EVs (inset) is still poorly understood, although microscopic evidence suggests a bilayered lipid membrane adorned with proteins and glycans. The presence of integral membrane proteins, as well as outer polysaccharides and mannoproteins decorating the vesicular surface, has been described in fungal EVs5. Although other membrane components remain poorly characterized, it has been suggested that fungal EVs can be coated with lectins and pathogen-associated molecular patterns109–111.
The International Society for Extracellular Vesicles defines EVs as particles released from cells, surrounded by a lipid bilayer and unable to replicate independently2. The most common subtypes of eukaryotic EVs are exosomes and microvesicles1. Exosomes originate from the endosomal system and microvesicles arise through blebbing from the plasma membrane. Microvesicles are typically larger than exosomes. In most studies reporting on EVs, a broad population of vesicles is probably being investigated rather than a specific subtype, unless the subcellular origin of the EVs can be demonstrated. Based on the International Society for Extracellular Vesicles’ recommendations, classification of fungal EVs into exosomes or microvesicles is not recommended. Although there is evidence that fungal EVs can include these and other subtypes depending on the organism and analytical conditions3–5, the precise subcellular origin of fungal EVs has not yet been determined. We recommend the general term fungal EVs, since it aligns with the definition of lipid-bilayered particles released from cells with no ability to replicate independently.
In eukaryotes, EV formation generally starts intracellularly or at the plasma membrane level, resulting in vesicle release to the extracellular milieu6. However, for cells with complex cell walls, such as prokaryotic bacteria and archaea, as well as eukaryotic fungi and plants, understanding the production and release of EVs to the outer environment is challenging. This is because their transit through the cell wall is yet to be defined. The cell wall is a dynamic but dense layer mostly comprising polysaccharides and glycoproteins (in plants and fungi)7 or hybrid glycans (in Gram-positive bacteria and mycobacteria)8. In all of these cases, the cell wall is a hydrophilic structure with viscoelastic properties that must be compliant to the trafficking of membrane-containing structures.
The release of EVs across the cell wall has been associated with physiological processes in fungi such as secretion and prion propagation, as well as immunological and immunopathological fungus–host interactions9. Notably, fungal EVs trigger interferon signalling10. However, essential questions regarding the physiological relevance of fungal EVs and their functions during host–pathogen interactions remain unanswered11. In this Perspective we explore the properties of fungal EVs and discuss the knowledge gaps and future research directions in this field.
History of fungal EVs
Box 1 describes the accumulation of microscopic evidence for the occurrence of fungal EVs over time. Fungal EVs were discovered and formally defined in 200712; however, there were hints of their existence long before that. To our knowledge, the first visual suggestion of fungal EVs dates back to 1958 in the wood-decay fungus Polystictus versicolor13, now known as Coriolus versicolor (the turkey tail mushroom). In this model, Girbardt described cellular components that were defined as different-shaped structures located outside the plasma membrane13. Using the current terminology, these structures resemble vesicles being released into the periplasmic space after fusion of multivesicular body (MVB)-like organelles with the plasma membrane. MVBs originate from endosome maturation and contain multiple internal vesicles formed by the inward folding of their membrane. Similar MVB-like structures fused with the plasma membrane were observed by freeze-fracture transmission electron microscopy (TEM) for the encapsulated pathogen Cryptococcus neoformans in 1973 during murine infection14. In this study, Takeo and colleagues describe a “bag-like paramural body showing multivesicles”, with some of the “paramural bodies found to be multivesicular systems”. These observations were associated with a large number of vesicles during infection compared with in vitro growth and a “high-secretion activity in C. neoformans grown in the parasitic state”. However, Takeo et al. restricted their observations to the cell wall and inner capsule and did not venture into the space outside the capsule, which presumably was thought to provide a barrier to extracellular transit.
BOX 1. Timeline of microscopic evidence for the occurrence of fungal EVs.
MVB-like structures associated with the plasma membrane were reported in 1958 for C. versicolor13. Microvesicle-like structures were reported in 1972 for A. nidulans15. MVB-like structures associated with the cell wall were detected in 1973 for C. neoformans using freeze-fracture electron microscopy14. Isolated chitosomes produced by Mucor rouxii were described in 1976 (top image) and shown to catalyse the synthesis of cell wall fibres (bottom image)19. EV-like structures associated with the outer cell wall were reported in 1990 for C. albicans20 and at the cell wall–plasma membrane interface in 2000 for C. neoformans16. EVs were first isolated from fungal supernatants in 2007 (top image), when they were also detected in the inner layers of the cell wall (bottom image)12. With the advent of cryo-EM, fungal EVs were analysed in detail in vitro and in vivo. EVs of Golovinomyces orontii were observed in the periplasmic space and interfacial matrix between a G. orontii haustorium and an Arabidopsis thaliana epidermal cell in 2011112. EVs were detected (arrowhead) within the apical cell wall of an N. crassa hypha in 202056 (the white asterisk indicates a Spitzenkörper macrovesicle). In 2021, cryo-electron tomography revealed two vesicle populations with either no surface decoration (top image) or fibrillar decoration (bottom image) in C. neoformans5. In 2022, EVs of Colletotrichum lindemuthianum were observed in the periplasmic space (black arrowheads in top image) and interfacial matrix between infection hyphae and Phaseolus vulgaris cells (black asterisk in bottom image)24. Note that several other studies reported EVs by electron microscopy after 2007. Owing to space limitations, we have only included images generated by cryo-EM. For C. neoformans (2021), the images to the right show a magnified view of the parts of the images to the left indicated by a dashed box. CW, cell wall; FP, fungal plasma membrane; FW, fungal wall; PP, plant plasma membrane. Photographs adapted with permission from (top left to bottom right): C. versicolor, ref. 13, Springer Nature Limited; A. nidulans, ref. 15, Society for General Microbiology; C. neoformans (1973), ref. 14, American Society for Microbiology; M. rouxii (top and bottom), ref. 19, National Academy of Sciences; C. albicans, ref. 20, American Society for Microbiology; C. neoformans (2000), ref. 16, American Society for Microbiology; C. neoformans (2007, top and bottom), ref. 12, American Society for Microbiology; C. neoformans (2021, top and bottom), ref. 5 under a Creative Commons license CC BY 4.0; C. lindemuthianum (top and bottom), ref. 24 under a Creative Commons license CC BY 4.0. Photograph of N. crassa courtesy of Rob Roberson.

Other mechanisms resulting in EV release were also documented before 2007. Vesicles possibly budding out of the plasma membrane were reported in 1972 for the filamentous fungus Aspergillus nidulans15 and in 2000 for C. neoformans16. In A. nidulans, small vesicles were observed near the plasma membrane, in addition to outer membrane blebbing15, resembling the shedding process currently associated with microvesicle formation1. However, it cannot be ruled out that the structures reported in this study were membrane tubules rather than vesicles or blebs, as previously reported in fungal cells undergoing plasmolysis17—a process through which cells lose water in a hypertonic solution. Interestingly, the authors of the A. nidulans study15 suggested that the vesicles may be involved in wall formation. Membrane shedding resulting in EVs associated with cell wall synthesis was demonstrated decades later in the fungal pathogen Aspergillus fumigatus18, but earlier studies by Bracker and colleagues19 in the 1970s demonstrated vesicle-like structures named chitosomes in association with the synthesis of cell wall fibres. It remains unknown whether chitosomes are related to EVs, but their morphology and possible involvement with cell wall synthesis are consistent with this hypothesis.
In C. neoformans16, the detection of glycosphingolipids on the cell wall was associated with possible vesicle transport across this structure16. Glycolipid-containing vesicles were observed at the cell surface and were shown to move across the periplasmic space and deposit cell membrane constituents on the cell wall16. Structures resembling EVs in association with the plasma membrane–cell wall interface, in addition to the outer layers of the cell wall, were also reported in 1990 for the opportunistic fungal pathogen Candida albicans20. In this study, external EV-like structures were described as “emerged blebs enclosed by a discernible double membrane”.
Although there might be additional earlier studies suggesting the existence of fungal EVs that we do not discuss here, these reports underscore the notion that EVs were visualized but not characterized as a distinct transport system before 2007. These studies mostly relied on TEM and were accompanied by concerns that vesicle-like structures could be artefacts of microscopy and lipid aggregation, whereas the cell wall was considered a barrier impenetrable to such large structures, contradicting the existence of EVs21.
In 2007, a study on the trans-cell-wall transport of polysaccharides in C. neoformans demonstrated membrane-containing structures associated with the fungal cell wall, leading to the hypothesis that these membranous particles could be isolated from culture supernatants12. Ultracentrifugation protocols in association with electron microscopy revealed that these particles could be isolated from culture fluids of C. neoformans and they contained polysaccharides that were used by the fungus for assembly of the cell surface12,21. Initial investigations with human fungal pathogens were extended to a wide range of species, including plant fungal pathogens. For instance, in Botrytis cinerea, TEM of infectious hyphae and hyphae grown in vitro revealed EVs of various sizes and densities22. In Ustilago maydis, EV-enriched fractions contained messenger RNAs that were upregulated during infection23.
Post-2007 research on fungal EVs was based on cryo-fixation techniques to preserve membranes in their near-native state. Cryoelectron microscopy (cryo-EM) can be used for imaging isolated fungal EVs and has revealed new features, including fibrillar decorations on the EV surface. These decorations mainly correspond to mannoproteins of still unknown functions5. Plunge freezing or high-pressure freezing followed by freeze substitution and resin embedding are suitable for cryo-EM imaging of EVs in thicker samples, such as cultured hyphae and infected plant tissues.
What is the origin of EVs?
The exact subcellular origin of fungal EVs has not yet been identified. There are multiple possible origins of fungal EVs, as illustrated in Fig. 2 and discussed below.
Fig. 2 |. Potential mechanisms of EV biogenesis in fungi.

Exosome biogenesis starts with plasma membrane invagination (i) and the subsequent endocytosis of external substances (represented as blue, irregular particles). This process leads to the formation of endosomes, followed by degradation of the ingested molecules. Endosomes can generate intraluminal vesicles (red spheres) by inward folding of their membranes, forming MVBs. MVBs can fuse with the plasma membrane and release intraluminal vesicles extracellularly. Alternatively, microvesicle-like EVs can form by membrane budding (ii). In fungi, these vesicles are deposited in the periplasmic space between the plasma membrane and cell wall, where they are directed to the outer space by as yet unknown mechanisms.
Endosomal origin of fungal EVs
The functions of eukaryotic MVBs in sorting and trafficking molecules, in addition to exosome formation, are widely known. In fungi, as in other eukaryotes, morphological studies suggest that MVBs fuse with the plasma membrane, releasing intraluminal vesicles into the periplasmic space (Fig. 2)14,24. From here, the fungal EVs could traverse the cell wall to the extracellular space. Interestingly, a C. neoformans mutant lacking P4-ATPase Apt1p—one of the eukaryotic flippases responsible for regulating lipid asymmetry in biological membranes—exhibits aberrant MVBs and produces EVs with altered dimension and composition25,26. This suggests that flippases are indirect regulators of EV formation in fungi, implying that this class of enzymes could be targeted using genetic and/or pharmacological tools for functional studies of fungal EVs.
Using a proteomics approach, Oliveira et al.27 concluded that Saccharomyces cerevisiae mutants lacking proteins responsible for MVB formation still produce EVs but have altered protein composition. In fact, only a few studies point to direct connections between the functionality of MVBs and EV formation in fungi. The subunits of the endosomal sorting complexes required for transport (ESCRT) are necessary for EV formation in eukaryotes28. In a C. albicans model of biofilm formation, 16 out of 21 mutants defective in orthologues of ESCRT proteins (Vps27, Hse1, Vps23, Vps28, Mvb12, Srn2, Vps22, Vps25, Vps36, Vps2, Vps20, Snf7, Vps24, Bro1, Doa4 and Vps4) showed slightly reduced EV production29. Together, these observations suggest that fungal cells produce EVs that could be related to mammalian exosomes.
Plasma membrane origin of fungal EVs
Plasma membrane blebbing in association with the cell wall has been repeatedly reported in fungi, including S. cerevisiae30, Schizosaccharomyces pombe31, C. neoformans16 and C. albicans20. Indeed, some of the essential eukaryotic components for constructing the plasma membrane play a crucial role in the formation of fungal EVs. In C. albicans, genes associated with phospholipid biosynthesis influence both the morphology and cargo of EVs32. Deletion of AIM25, a gene encoding a presumed scramblase, impacts the generation of cryptococcal EVs33. An Δaim25 mutant strain produced EVs that exhibited larger dimensions and differences in small RNA cargo compared with those from the wild-type strain33.
A major challenge in studying the formation of EVs at the plasma membrane level in fungi arises owing to the presence of a thick and complex cell wall. In 1998, Osumi31 noted structures resembling microvesicles in protoplasts (cells with an artificially degraded cell wall) of S. pombe. More recently, Rizzo et al.18 used A. fumigatus protoplasts to investigate the release of EVs from the plasma membrane. More recently, the integration of fluorescence microscopy, super-resolution fluorescence microscopy, high-resolution scanning electron microscopy and TEM has made it possible to image plasma membrane projections and the release of particles resembling EVs in greater detail18.
The idea that fungal EVs can originate at the level of the plasma membrane is supported by early studies that were not specifically investigating EV biology. In C. neoformans, plasma membrane projections forming vesicle-like structures in contact with the cell wall were identified two decades ago16. Similarly, in S. cerevisiae, plasma membrane invaginations were observed to lead to the formation of vesicular structures containing cytoplasmic material that were subsequently transported into the periplasmic space34. This observation aligns with the identification of cytoplasmic proteins in fungal EVs5,24,35–37. Of note, these plasma membrane invaginations resemble eisosomes, which comprise plasma membrane protein complexes that mark the site of endocytosis in some eukaryotes38. As reported for C. albicans, the eisosome component Sur7 is present in EVs24,39,40. However, further studies are necessary to investigate whether the EVs derived from cytoplasmic substrates in S. cerevisiae are associated with eisosome formation. Recent studies indicate that eiosomes do not directly participate in endocytosis, but rather play diverse roles in fungal lipid homeostasis, plasma membrane organization, cell wall synthesis regulation and stress responses41,42. However, the molecular mechanisms necessary for EV formation at the plasma membrane are largely unknown.
How do EVs cross the cell wall bidirectionally?
In cell-wall-containing organisms, such as plants, fungi and bacteria, crossing the cell wall is necessary for molecules to reach the extracellular space. In addition, several fungal species produce extracellular matrices (three-dimensional frameworks comprising carbohydrates, proteins, lipids and nucleic acids)43, which may pose a further barrier to EV movement. For decades, it has been known that cell wall-containing organisms efficiently export various molecules, as exemplified by the ability of yeasts to secrete biologically active invertases44. In fungi, the cell wall is recognized for its viscoelastic nature and its ability to withstand hydrostatic turgor pressure45,46. This property results from a combination of rigid chitin chains with flexible β-1,3-glucans47, which are cross-linked among themselves and with other cell wall components. This semi-permeable barrier enables fungal cells to regulate their volume and facilitate or restrict the diffusion of macromolecules. In C. neoformans, the cell wall contains small pores at the nanometre scale, even in the presence of dense melanization48. EV passage through the cell wall therefore requires the interaction of these membrane structures with fungal cell wall components.
Despite our understanding of the fungal cell wall, the mechanisms through which large molecules traverse it remain poorly understood. When fungal EVs were initially described, it was suggested that vesicular export might facilitate transport across the cell wall12. The detection of various extracellular macromolecules in fungal EVs, including pigments, polysaccharides, proteins, RNAs and small molecules, supported this hypothesis (reviewed in refs. 49,50). A possible association between vesicles and the cell wall agrees with studies conducted in the 1960s and 1970s, which detected lipids in cell wall fractions of fungi51,52. More recent studies demonstrated that fungal EVs indeed contribute with a substantial portion of cell wall lipids comprising the cell wall53,54. Additionally, observations using different microscopy methods revealed vesicle-like structures associated with the cell wall, spanning both inner and outer layers12,24,36,55. Although TEM data indicate the passage of EVs through yeast cell walls, there is limited evidence for EVs transiting the cell walls of filamentous fungi. This is surprising if one assumes that EVs are abundantly secreted by filamentous fungi. A rare example of EV passage through the cell wall was provided in Neurospora crassa in 202056. The study showed that membrane-bound spherical-to-ovoid inclusions are present within the apical cell57.
The presence of cell-wall-associated vesicles in fungi, along with the description of cell wall pores57, lends support to the existence of mechanisms to facilitate vesicle passage through the fungal cell wall. Several hypotheses have been proposed to explain this phenomenon. For example, proteomics studies have identified dozens of cell wall hydrolytic enzymes in fungal EVs5,37,58,59. Thus, as suggested for bacteria, EVs might catalyse their own passage through the wall by breaking down some of its structural components in cases where polysaccharide-degrading enzymes are associated with the EV surface60. Corroborating this hypothesis, EVs released by the fungal pathogen Histoplasma capsulatum carry substantial amounts of active chitinase-1 (Cts1)61. Inhibiting the enzymatic activity of Cts1 with methylxanthines significantly decreased the release of EVs by H. capsulatum yeasts, directly impacting fungal virulence61. Cts1 possesses a glycosylphosphatidylinositol attachment site, suggesting its presence in the external layer of the plasma membrane and possibly in EV membranes62. This observation, along with the fact that chitinase activity was detectable in intact EVs, suggests that Cts1 is localized on the surface of these EVs61. Although this study aids our understanding of the release of EVs by H. capsulatum, the chitinolytic activity detected in EVs from C. albicans, Candida parapsilosis and S. cerevisiae was very low or even absent, suggesting differences between species. In addition, it remains unclear whether the presence of hydrolases in EVs correlates with cell wall degradation.
Given that fungal EVs can traverse the cell wall, an important question arises: do EVs contribute to cell wall remodelling? As previously discussed, the passage of EVs through the cell wall of H. capsulatum may be influenced by chitinases61, but other enzymes capable of hydrolysing structural compounds, such as glucanases and proteases, have also been characterized as EV components35. The direct effect of these enzymes on the cell wall network has not been thoroughly investigated. Nevertheless, proteomic analysis of fungal EVs revealed the presence of enzymes associated with glycan and polysaccharide synthesis, suggesting that EVs may contribute to cell wall synthesis and remodelling18,59,61. Indeed, protoplasts of A. fumigatus grown under conditions stimulating cell wall regeneration contained a fibrillar material closely associated with EV-like compartments18. Notably, EVs isolated from cell wall regeneration conditions exclusively contained N-acetyl-galactosaminyl residues and exhibited a higher glucose content than EVs isolated from non-regenerating conditions18. These monosaccharides are the building units of the cell wall polysaccharides galactosaminogalactan and glucan, respectively, suggesting an association between EVs and cell wall synthesis. Work on S. cerevisiae also showed that the higher susceptibility of a mutant strain lacking chitin-synthase 3 to caspofungin was rescued with the addition of EVs from the wild-type strain and by strains overexpressing this enzyme59. Understanding the role of fungal EVs in the cell wall may provide insights into the plasticity of this structure and shed light on the mechanisms for the release of proteins and other molecules.
EVs could also be spontaneously released from the periplasmic space during cell division, a process requiring the hydrolysis of cell wall layers to facilitate cell separation. Another possibility is that EVs present in the extracellular space might be propelled extracellularly during cell wall synthesis. This assumes that they would be deposited onto cell wall layers, subsequently becoming more external with cell wall thickening. Finally, the idea that the cell wall is more flexible than initially thought63 aligns with the hypothesis that vesicle traffic could be driven by motor-like proteins, such as chitin synthases with myosin-like domains64 and myosin-related proteins detected in association with EVs by proteomic approaches65.
In certain fungal species, the cell wall is not the final barrier for EV release. As mentioned earlier, members of the Cryptococcus genus are coated by a thick capsule and EVs promote capsular formation12. However, both wild-type and acapsular strains of Cryptococcus produce EVs, although the capsule affects vesicular properties5,58,66. A comparison between the dimensions of EVs produced by a wild-type strain and those produced by an acapsular mutant demonstrated that the majority of vesicles from mutant cells had a diameter of around 50 nm, with most in the range of 20–90 nm and only 4.9% larger than 90 nm66. Vesicles from wild-type cells were much more variable in size, ranging from 20 nm to over 200 nm, with 50.6% displaying a diameter larger than 90 nm66. Since cryptococcal EVs contain highly hydrophilic capsular polysaccharides12, we hypothesize that the cargo component could cause alterations in dimensions, since hydrophilic compounds would contribute with the intra-vesicular retention of water.
Several studies have reported the presence of free EVs in cryptococcal supernatants. These observations imply the existence of uncharacterized mechanisms for EVs to traverse the capsule. The structure of the cryptococcal capsule is more porous than that of the fungal cell wall67, suggesting that the capsular network is more permissive to vesicle passage, a property that can be influenced by multiple factors. For instance, capsules from C. neoformans cells cultivated for 15 days were significantly less permeable to dextrans than cells cultivated for two days68, suggesting that ageing affects capsular permeability. It remains unknown whether similar effects are observed for cryptococcal EVs. It has been reported that the binding of antibodies to the capsule changes its permeability, implying that the host’s immune response might also affect EV passage through the capsule69.
There are further potential mechanisms for EV release across the cell wall, some of which we propose here. EVs could be expelled by regulated processes resulting in programmed cell bursting. It is also possible that in some situations EVs do not actually cross the cell wall. After secretion to the periplasmic space, EVs could burst to release their contents, which could then diffuse across the fungal cell wall. Furthermore, turgor pressure could be important for EV passage, but so far these mechanisms have not been explored experimentally.
Previously, it was unclear whether cells of the same fungal species could communicate with each other by taking up EVs, particularly because the cell wall was considered chemically incompatible with the import of membranous structures. Demonstration that EVs serve as vehicles for prion transmission in yeast70,71 supported the concept that fungal cells could take up vesicles. Further support came from the finding that lipid formulations of the antifungal amphotericin B, such as AmBisome, reach the plasma membrane of C. albicans in their intact form, suggesting that the fungal cell wall is permissive to vesicular structures, potentially through rapid remodelling46. C. neoformans was equally permissive to liposome penetration unless the cell wall was melanized46. Recent solid-state NMR studies have shown that amphotericin B forms extra membranous asymmetric head-to-tail dimer stacks, which act as sponges that extract ergosterol from the cell membrane72. This model does not require the AmBisome liposomal carrier to be transported to the fungal cell membrane, but it must be delivered through the layers of the cell wall. Previous studies46 showed that the 60-nm-diameter AmBisome liposome can even transport non-elastic 15 nm colloidal gold particles in the lumen of the liposome through the inner and outer cell wall layers despite the predicted porosity of the cell wall being restrictive to particles above 5.8 nm in diameter46.
EVs have also been shown to deliver their components intact to the fungal cytoplasm in human and plant pathogens. In the grey mould B. cinerea, small RNAs are protected and transported into the plant cells by EVs to induce cross-kingdom RNA interference of host genes73. In the human pathogen Cryptococcus gattii, an avirulent strain regained its virulence by the addition of EVs produced by a virulent isolate of the same species74. In a plant infection model, Arabidopsis EVs containing small RNA or messenger RNA were taken up by B. cinerea, resulting in disease control and attenuated pathogenicity75,76. Notably, in the same model, fungal small RNAs were exported in EVs to enter plant cells73. These results suggest the existence of mechanisms for the passage of fungal EVs through the plant cell wall. An exception are fungi that penetrate the wall and produce haustoria or intracellular hyphae in living plant cells.
Plant cells incorporate fungal EVs through clathrin-mediated endocytosis73, but whether similar mechanisms occur in fungal cells remains to be explored. The fact that EVs released by C. albicans yeasts can impede their own filamentation77 suggests that EVs can traverse the cell wall to reach the intracellular space, as suggested for Paracoccidioides brasiliensis78. Considering that the fungal cell wall can allow the passage of bacterial macromolecules79 and even bacteria80, these findings may not be surprising. However, the mechanisms by which fungal cells take up EVs remain unknown.
Are EVs produced in vivo?
Demonstrating conclusively that fungal EVs are produced during in vivo infection and have roles in fungal pathobiology is challenging. Standard methods for EV isolation from human or animal fluids will inevitably co-isolate host vesicles, thereby complicating the use of conventional techniques to ascertain the presence of fungal EVs. For example, nanoparticle tracking analysis or TEM cannot distinguish between host and fungal EVs. The detection of fungus-specific molecules (for example, ergosterol) is also problematic, as there remains a possibility that these fungal components were incorporated by host cells and subsequently released within EVs. Thus, there is a challenging need for precise methodologies to definitively demonstrate that fungal EVs are produced during infection.
Most studies characterizing fungal EVs at the structural and functional levels are based on the isolation of EVs from culture supernatants obtained in vitro. Fungal EVs are produced and efficiently detected in fungal cultures developed under diverse conditions, including variable composition of media, temperature, time of growth and fluidity of the cultivation medium (solid and liquid)9. EV cargo is strongly regulated by nutritional cues, such as protein availability, reducing agents and vitamins81,82, indicating that EV production and release are regular physiological events during fungal growth. However, the lack of biomarkers specific to EVs from fungal pathogens of humans makes it challenging to demonstrate their production during infection. In contrast, for bacterial and protozoan EVs, specific biomarkers have supported the design of diagnostics for parasitic and bacterial diseases, including tuberculosis83–85.
An EV biomarker, pounchless 1 (PLS1), was successfully identified in the plant-pathogenic fungus B. cinerea73. PLS1 is a transmembrane tetraspanin protein that labels secreted fungal EVs. Both mammals and plants also utilize tetraspanin proteins (such as mammalian CD63 and CD9 and plant tetraspanins 8 and 9) as important biomarkers for their EVs, specifically exosomes or exosome-like EVs. Using a PLS1–YFP B. cinerea strain to infect transgenic Arabidopsis plants expressing tetraspanin 8-CFP, EVs from both the plant and the fungal pathogen were observed in the extracellular apoplastic fluid. Since tetraspanins are transmembrane proteins located on the EV surface, it is possible to purify fungal EVs using an affinity capture method, as has been successfully demonstrated in plants86.
Indirect evidence supporting the idea that EVs from human fungal pathogens are produced in vivo was presented in early studies. For instance, components from EVs produced by C. neoformans, H. capsulatum and P. brasiliensis were recognized by sera obtained from infected human patients36,37,58. Vesicle-like structures were microscopically detected in infected tissues in a C. neoformans murine infection model12, as well as in Pneumocystis carinii-infected rat tissues87, but the origin of these vesicles (host or fungi) could not be distinguished. In the plant infection model (citrus fruits), EVs released by Penicillium digitatum were detected in vivo as carriers of fungal toxins, including tryptoquialanines and fungisporin88.
The recent use of mass spectrometry and serological tests using fungal EVs for the detection of fungal disease has provided direct evidence for fungal EV production during human infection89,90. In urine samples from patients with invasive aspergillosis, EVs bearing galactofuranose have been detected89,91. Notably, galactofuranose, the five-membered ring form of galactose, is abundantly produced by Aspergillus species, but has never been found in mammals92. These results strongly indicate that fungal EVs are produced during infection, although the possibility that fungal antigens were endocytosed by host cells and exported in human EVs cannot be ruled out. Similar findings were recently reported for other fungal pathogens, as EVs isolated from the serum and urine samples of patients with infections caused by C. albicans, C. neoformans and P. brasiliensis contained fungal molecules90. These data highlight the need for universal EV biomarkers that could be applicable to other models.
Biomarkers, especially those accessible on the EV surface, will also be invaluable for the affinity purification of fungal EVs from infected tissues, where host EVs probably predominate. In the case of plant-pathogenic fungi, many virulence-associated genes are only expressed during host interaction, at specific infection stages or in specialized fungal cell types. The corresponding proteins would not be found in EVs produced by cultured mycelia, so analysis of EVs purified from infected plants will be essential to understand their role in the interaction. Notable progress has been made in the field of plant infections, where the SNARE (Snc1 and Sso2), 14-3-3 (Bmh1) and tetraspanin (BcPLS1) proteins were characterized as fungal EV biomarkers24,73. These findings encourage the development of similar studies in human fungal pathogens.
What are the functions of subpopulations of fungal EVs?
Most studies investigate a broad population of EVs with undetermined subcellular origin93. This directly impacts functional studies, including those focused on determining biogenesis, drug resistance, biofilm formation and bioactive molecule incorporation by recipient cells, since it is unknown which EV subpopulation is responsible for each biological effect. This also applies to the determination of EV composition since molecules associated with different vesicle subpopulations will be detected5,35,94. For instance, in immunological studies proposing EVs as vaccine candidates, the observed protective effects could be attenuated by the presence of EV subpopulations with opposite immune functions, so identifying the EV population responsible for a particular phenotype is complex. Furthermore, in studies analysing the composition of EVs, it may be unclear which components belong to each vesicle subpopulation. This is further complicated by the fact that fungal EVs are highly diverse within fungal isolates belonging to the same species3. This highlights the need for better methods to separate and analyse subpopulations of fungal EVs. One approach is to import and adapt methods that have been successfully used for other eukaryotes or bacteria, including gradient centrifugation, genetic and/or chemical screenings for altered EV production, chromatographic separation, affinity chromatography and others. Progress has been made by employing these approaches for separating EV fractions of different densities3–5, but major uncertainties persist, since the reasons for the variable properties of fungal EVs are still unknown. Experimental evidence based on gradient centrifugation approaches suggests that fungal EV subpopulations may have variable antibody reactivity and morphology, which could be explored for the development of new analytic approaches3,94.
What are the genetic regulators of fungal EVs?
A limited number of fungal mutant strains deficient in EV production have been identified4,27,29,95. A screen identifying EV regulators4 revealed several transcription factors (Hap2, Gat5, Bzp2 and Liv4), which are positive regulators of EV production in C. neoformans. These transcription factors, as well as the other components of the heterotrimeric HAP complex (a key regulator of mitochondrial function; Hap3 and Hap5 proteins)96 regulated EV production in C. neoformans4. Although the Δhap2 mutant strain produced less than 5% of the wild-type EV levels, it did not display any major growth defect, suggesting that a wild-type level of EV production is dispensable in this fungus under the conditions tested. The exact mechanisms by which these transcription factors regulate EV production are still unknown. We anticipate that the combination of omics strategies with biochemistry, in vivo and genetic analyses will help to answer some of the current questions concerning the production, composition, diversity and biological roles of fungal EVs.
Future perspectives
As the recognition of the biological importance of EVs produced by fungi grows, more questions emerge regarding their cellular origin, formation and in vivo function(s). By identifying the key knowledge gaps in the field, we hope to stimulate the development of new experimental approaches to generate insights into fungal EVs.
The challenges in identifying the genetic regulators of fungal EVs are significant, especially in filamentous or dimorphic fungi. The production of EVs by filamentous fungi was first described in 201497 and further confirmed in other investigations18,22,88,98–106. However, most of the research on fungal EVs has been conducted in yeast cells. This reflects our limited understanding of how EVs are produced by multicellular fungi. Despite these challenges, progress has been made in this area. Mutant collections are now available for several filamentous fungi and recent advances have been made in the genetic manipulation of EV-producing dimorphic pathogens belonging to the Sporothrix107 and Paracoccidioides108 genera, which are known for their genetic complexity. We anticipate that the coming years will witness important advances in understanding how EVs are formed in different morphological stages of fungi.
Fungal EVs can be leveraged for diverse purposes, such as vaccine development and drug design, but deeper knowledge is required for applications to be successful. For example, to design future drugs and vaccines using fungal EVs as targets or prototypes, it is necessary to clarify the pathways involved in their formation and to enhance methods for their separation and functional analysis. Understanding how EVs are produced during infection will aid in designing new interventions. High-resolution microscopy methods combined with genetic approaches may reveal how EVs cross the fungal cell wall. Additionally, identifying biomarkers will enable the creation of widely applicable tools for fundamental biological models, fungal disease diagnosis and microbial pathogenesis.
Acknowledgements
M.L.R. was funded by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under award number R01AI183314, as well as Brazilian National Council for Scientific and Technological Development (CNPq) grants 404365/2023-0 and 304998/2022-2 and the Program for Research Stimulation of the Carlos Chagas Institute of the Oswaldo Cruz Foundation. H.J. was funded by grants from the National Institutes of Health (R35GM136379), National Science Foundation (IOS 2020731), US Department of Agriculture (2021-67013-34258) and CIFAR Fungal Kingdom fellowship. L.N. was funded by CNPq grants 408843/2021-7 and 310778/2020-4 and FAPERJ grants E-26/201.602/2022 and E-26/211.300/2021. J.D.N. and D.Z.-M. were funded in part by AI171093 and AI156104. R.J.O. and T.-T.-H.C. were supported by funding from grant ANR-17-CAPS-0004-01. The BIOGER unit benefits from support from Saclay Plant Sciences-527 SPS (ANR-17-EUR-0007). T.G. was funded by the Portuguese Foundation for Science and Technology under projects UIDB/04539/2020, UIDP/04539/2020 and LA/P/0058/2020. I.V.E. is funded by grants from the Institut Pasteur and French National Research Agency. I.V.E. is an Azrieli Global Scholar in the CIFAR Program Fungal Kingdom: Threats and Opportunities. T.P.F. was funded by FAPESP 2022/02992-0. G.J. is funded by grants from the Institut Pasteur and French National Research Agency (Projet ResistEV AAPG2021 CE35 and Project FEVCOM AAPG2023 CE35). R.P. has received funding from FAPESP 2022/11123-5, CNPq and CAPES. M.K. was supported by the Centre National de la Recherche Scientifique. A.C. was supported in part by National Institutes of Health grants AI052733-16, AI152078-01 and HL059842-19. N.A.R.G. acknowledges support from Wellcome Trust Investigator, Collaborative, Equipment, Strategic and Biomedical Resource awards (101873, 200208, 215599 and 224323). N.A.R.G. also thanks the Medical Research Council (MR/M026663/2) and MRC Centre for Medical Mycology (MR/N006364/2) for support, as well as the National Institute for Health and Care Research Exeter Biomedical Research Centre. The views expressed are those of the authors and not necessarily those of the National Institute for Health and Care Research or Department of Health and Social Care. J.R. is funded by CNPq grant 404365/2023-0 and FAPERJ grant E-26/210.468/2024. G.J., R.C.M. and M.L.R. are members of the Pasteur International unit Fungal Extracellular Vesicles. Original versions of Figs. 1 and 2 drawn by Wagner Nagib.
Footnotes
Competing interests
The authors declare no competing interests.
References
- 1.Raposo G & Stoorvogel W Extracellular vesicles: exosomes, microvesicles, and friends. J. Cell Biol 200, 373–383 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Welsh JA et al. Minimal information for studies of extracellular vesicles (MISEV2023): from basic to advanced approaches. J. Extracell. Vesicles 13, e12451 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Reis FCG et al. Analysis of cryptococcal extracellular vesicles: experimental approaches for studying their diversity among multiple isolates, kinetics of production, methods of separation, and detection in cultures of titan cells. Microbiol. Spectr 9, e00125 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rizzo J et al. Coregulation of extracellular vesicle production and fluconazole susceptibility in Cryptococcus neoformans. mBio 14, e0087023 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rizzo J et al. Cryptococcus extracellular vesicles properties and their use as vaccine platforms. J. Extracell. Vesicles 10, e12129 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Colombo M, Raposo G & Thery C Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell Dev. Biol 30, 255–289 (2014). [DOI] [PubMed] [Google Scholar]
- 7.Latgé J The cell wall: a carbohydrate armour for the fungal cell. Mol. Microbiol 66, 279–290 (2007). [DOI] [PubMed] [Google Scholar]
- 8.Kumar S, Mollo A, Kahne D & Ruiz N The bacterial cell wall: from lipid II flipping to polymerization. Chem. Rev 122, 8884–8910 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rodrigues ML, May RC & Janbon G The multiple frontiers in the study of extracellular vesicles produced by fungi. Microbes Infect. 26, 105233 (2023). [DOI] [PubMed] [Google Scholar]
- 10.Brown Harding H et al. Candida albicans extracellular vesicles trigger type I IFN signalling via cGAS and STING. Nat. Microbiol 9, 95–107 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rodrigues ML, Godinho RMC, Zamith-Miranda D & Nimrichter L Traveling into outer space: unanswered questions about fungal extracellular vesicles. PLoS Pathog. 11, 1005240 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rodrigues ML et al. Vesicular polysaccharide export in Cryptococcus neoformans is a eukaryotic solution to the problem of fungal trans-cell wall transport. Eukaryot. Cell 6, 48–59 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Girbardt M About the substructure of Polystictus versicolor L. Arch. Mikrobiol 28, 255–269 (1958). [PubMed] [Google Scholar]
- 14.Takeo K, Uesaka I, Uehira K & Nishiura M Fine structure of Cryptococcus neoformans grown in vivo as observed by freeze-etching. J. Bacteriol 113, 1449–1454 (1973). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gibson RK & Peberdy JF Fine structure of protoplasts of Aspergillus nidulans. J. Gen. Microbiol 72, 529–538 (1972). [DOI] [PubMed] [Google Scholar]
- 16.Rodrigues ML et al. Human antibodies against a purified glucosylceramide from Cryptococcus neoformans inhibit cell budding and fungal growth. Infect. Immun 68, 7049–7060 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bachewich CL & Heath IB Differential cytoplasm–plasma membrane–cell wall adhesion patterns and their relationships to hyphal tip growth and organelle motility. Protoplasma 200, 71–86 (1997). [Google Scholar]
- 18.Rizzo J et al. Characterization of extracellular vesicles produced by Aspergillus fumigatus protoplasts. mSphere 5, e00476–20 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bracker CE, Ruiz-Herrera J & Bartnicki-Garcia S Structure and transformation of chitin synthetase particles (chitosomes) during microfibril synthesis in vitro. Proc. Natl Acad. Sci. USA 73, 4570–4574 (1976). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Anderson J, Mihalik R & Soll DR Ultrastructure and antigenicity of the unique cell wall pimple of the Candida opaque phenotype. J. Bacteriol 172, 224–235 (1990). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Coelho C & Casadevall A Answers to naysayers regarding microbial extracellular vesicles. Biochem. Soc. Trans 47, 1005–1012 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.De Vallée A et al. Extracellular vesicles of the plant pathogen Botrytis cinerea. J. Fungi 9, 495 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kwon S et al. mRNA inventory of extracellular vesicles from Ustilago maydis. J. Fungi 7, 562 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rutter BD et al. The development of extracellular vesicle markers for the fungal phytopathogen Colletotrichum higginsianum. J. Extracell. Vesicles 11, e12216 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rizzo J et al. Role of the Apt1 protein in polysaccharide secretion by Cryptococcus neoformans. Eukaryot. Cell 13, 715–726 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rizzo J et al. The putative flippase Apt1 is required for intracellular membrane architecture and biosynthesis of polysaccharide and lipids in Cryptococcus neoformans. Biochim. Biophys. Acta Mol. Cell. Res 1865, 532–541 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Oliveira DL et al. Characterization of yeast extracellular vesicles: evidence for the participation of different pathways of cellular traffic in vesicle biogenesis. PLoS ONE 5, e11113 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Henne WM, Buchkovich NJ & Emr SD The ESCRT pathway. Dev. Cell 21, 77–91 (2011). [DOI] [PubMed] [Google Scholar]
- 29.Zarnowski R et al. Candida albicans biofilm-induced vesicles confer drug resistance through matrix biogenesis. PLoS Biol. 16, e2006872 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Oliveira DL et al. Biogenesis of extracellular vesicles in yeast. Commun. Integr. Biol 3, 533–535 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Osumi M The ultrastructure of yeast: cell wall structure and formation. Micron 29, 207–233 (1998). [DOI] [PubMed] [Google Scholar]
- 32.Wolf JM, Espadas J, Luque-Garcia J, Reynolds T & Casadevall A Lipid biosynthetic genes affect Candida albicans extracellular vesicle morphology, cargo, and immunostimulatory properties. Eukaryot. Cell 14, 745–754 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reis FCG et al. A novel protocol for the isolation of fungal extracellular vesicles reveals the participation of a putative scramblase in polysaccharide export and capsule construction in Cryptococcus gattii. mSphere 4, e00080–19 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rodrigues ML, Franzen AJ, Nimrichter L & Miranda K Vesicular mechanisms of traffic of fungal molecules to the extracellular space. Curr. Opin. Microbiol 16, 414–420 (2013). [DOI] [PubMed] [Google Scholar]
- 35.Rodrigues ML, Nakayasu ES, Almeida IC & Nimrichter L The impact of proteomics on the understanding of functions and biogenesis of fungal extracellular vesicles. J. Proteomics 97, 177–186 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vallejo MC et al. The pathogenic fungus Paracoccidioides brasiliensis exports extracellular vesicles containing highly immunogenic α-galactosyl epitopes. Eukaryot. Cell 10, 343–351 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Albuquerque PC et al. Vesicular transport in Histoplasma capsulatum: an effective mechanism for trans-cell wall transfer of proteins and lipids in ascomycetes. Cell Microbiol. 10, 1695–1710 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Walther TC et al. Eisosomes mark static sites of endocytosis. Nature 439, 998–1003 (2006). [DOI] [PubMed] [Google Scholar]
- 39.Dawson CS et al. Protein markers for Candida albicans EVs include claudin-like Sur7 family proteins. J. Extracell. Vesicles 9, 1750810 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.McKenna JA et al. SUR7 deletion in Candida albicans impacts extracellular vesicle features and delivery of virulence factors. J. Extracell. Biol 2, e82 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Douglas LM, Wang HX, Li L & Konopka JB Membrane compartment occupied by Can1 (MCC) and eisosome subdomains of the fungal plasma membrane. Membranes 1, 394–411 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Moseley JB Eisosomes. Curr. Biol 28, R376–R378 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mitchell KF, Zarnowski R & Andes DR The extracellular matrix of fungal biofilms. Adv. Exp. Med. Biol 931, 21–35 (2016). [DOI] [PubMed] [Google Scholar]
- 44.Dworschack RG & Wickerham LJ Production of extracellular invertase by the yeast, Saccharomyces uvarum NRRL Y-972. Arch. Biochem. Biophys 76, 449–456 (1958). [DOI] [PubMed] [Google Scholar]
- 45.Kaminskyj SGW, Garrill A & Heath IB The relation between turgor and tip growth in Saprolegnia ferax: turgor is necessary, but not sufficient to explain apical extension rates. Exp. Mycol 16, 64–75 (1992). [Google Scholar]
- 46.Walker L et al. The viscoelastic properties of the fungal cell wall allow traffic of ambisome as intact liposome vesicles. mBio 9, e02383–17 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gow NAR, Latge J & Munro CA The fungal cell wall: structure, biosynthesis, and function. Microbiol. Spectr 5, 1–25 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Eisenman HC et al. Microstructure of cell wall-associated melanin in the human pathogenic fungus Cryptococcus neoformans. Biochemistry 44, 3683–3693 (2005). [DOI] [PubMed] [Google Scholar]
- 49.Rodrigues ML & Nimrichter L From fundamental biology to the search for innovation: the story of fungal extracellular vesicles. Eur. J. Cell Biol 101, 151205 (2022). [DOI] [PubMed] [Google Scholar]
- 50.Puccia R Current status on extracellular vesicles from the dimorphic pathogenic species of Paracoccidioides. Curr. Top. Microbiol. Immunol 432, 19–33 (2021). [DOI] [PubMed] [Google Scholar]
- 51.Cox RA & Best GK Cell wall composition of two strains of Blastomyces dermatitidis exhibiting differences in virulence for mice. Infect. Immun 5, 449–453 (1972). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kanetsuna F, Carbonell LM, Moreno RE & Rodriguez J Cell wall composition of the yeast and mycelial forms of Paracoccidioides brasiliensis. J. Bacteriol 97, 1036–1041 (1969). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Longo LVG et al. Characterization of cell wall lipids from the pathogenic phase of Paracoccidioides brasiliensis cultivated in the presence or absence of human plasma. PLoS ONE 8, e63372 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Vallejo MC et al. Correction: lipidomic analysis of extracellular vesicles from the pathogenic phase of Paracoccidioides brasiliensis. PLoS ONE 7, e39463 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wolf JM, Espadas-Moreno J, Luque-Garcia JL & Casadevall A Interaction of Cryptococcus neoformans extracellular vesicles with the cell wall. Eukaryot. Cell 13, 1484–1493 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Roberson RW Subcellular structure and behaviour in fungal hyphae. J. Microsc 280, 75–85 (2020). [DOI] [PubMed] [Google Scholar]
- 57.De Souza Pereira R & Geibel J Direct observation of oxidative stress on the cell wall of Saccharomyces cerevisiae strains with atomic force microscopy. Mol. Cell. Biochem 201, 17–24 (1999). [DOI] [PubMed] [Google Scholar]
- 58.Rodrigues ML et al. Extracellular vesicles produced by Cryptococcus neoformans contain protein components associated with virulence. Eukaryot. Cell 7, 58–67 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhao K et al. Extracellular vesicles secreted by Saccharomyces cerevisiae are involved in cell wall remodelling. Commun. Biol 2, 305 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lee E-Y et al. Gram-positive bacteria produce membrane vesicles: proteomics-based characterization of Staphylococcus aureus-derived membrane vesicles. Proteomics 9, 5425–5436 (2009). [DOI] [PubMed] [Google Scholar]
- 61.Valdez AF et al. Traversing the cell wall: the chitinolytic activity of Histoplasma capsulatum extracellular vesicles facilitates their release. J. Fungi 9, 1052 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Goughenour KD, Whalin J, Slot JC & Rappleye CA Diversification of fungal chitinases and their functional differentiation in Histoplasma capsulatum. Mol. Biol. Evol 38, 1339–1355 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gow NAR, Casadevall A & Fang W Top five unanswered questions in fungal cell surface research. Cell Surface 10, 100114 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fernandes C, Gow NAR & Gonçalves T The importance of subclasses of chitin synthase enzymes with myosin-like domains for the fitness of fungi. Fungal Biol. Rev 30, 1–14 (2016). [Google Scholar]
- 65.Parreira VDSC, Santos LGC, Rodrigues ML & Passetti F ExVe: the knowledge base of orthologous proteins identified in fungal extracellular vesicles. Comput. Struct. Biotechnol. J 19, 2286–2296 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tefsen B et al. Deletion of the CAP10 gene of Cryptococcus neoformans results in a pleiotropic phenotype with changes in expression of virulence factors. Res. Microbiol 165, 399–410 (2014). [DOI] [PubMed] [Google Scholar]
- 67.Doering TL How sweet it is! Cell wall biogenesis and polysaccharide capsule formation in Cryptococcus neoformans. Annu. Rev. Microbiol 63, 223–247 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Cordero RJB et al. Chronological aging is associated with biophysical and chemical changes in the capsule of Cryptococcus neoformans. Infect. Immun 79, 4990–5000 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Mukherjee J, Cleare W & Casadevall A Monoclonal antibody mediated capsular reactions (Quellung) in Cryptococcus neoformans. J. Immunol. Methods 184, 139–143 (1995). [DOI] [PubMed] [Google Scholar]
- 70.Kabani M, Pilard M & Melki R Glucose availability dictates the export of the soluble and prion forms of Sup35p via periplasmic or extracellular vesicles. Mol. Microbiol 114, 322–332 (2020). [DOI] [PubMed] [Google Scholar]
- 71.Kabani M & Melki R Sup35p in its soluble and prion states is packaged inside extracellular vesicles. mBio 6, e01017–15 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lewandowska A et al. Fungicidal amphotericin B sponges are assemblies of staggered asymmetric homodimers encasing large void volumes. Nat. Struct. Mol. Biol 28, 972–981 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.He B et al. Fungal small RNAs ride in extracellular vesicles to enter plant cells through clathrin-mediated endocytosis. Nat. Commun 14, 4383 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Bielska E et al. Pathogen-derived extracellular vesicles mediate virulence in the fatal human pathogen Cryptococcus gattii. Nat. Commun 9, 1556 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cai Q et al. Plants send small RNAs in extracellular vesicles to fungal pathogen to silence virulence genes. Science 360, 1126–1129 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wang S et al. Plant mRNAs move into a fungal pathogen via extracellular vesicles to reduce infection. Cell Host Microbe 32, 93–105.e6 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Honorato L et al. Extracellular vesicles regulate biofilm formation and yeast-to-hypha differentiation in Candida albicans. mBio 13, e0030122 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Octaviano CE, Abrantes NE & Puccia R Extracellular vesicles from Paracoccidioides brasiliensis can induce the expression of fungal virulence traits in vitro and enhance infection in mice. Front. Cell Infect. Microbiol 12, 834653 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hooykaas PJJ et al. Agrobacterium-mediated transformation of yeast and fungi. Curr. Top. Microbiol. Immunol 418, 349–374 (2018). [DOI] [PubMed] [Google Scholar]
- 80.Moebius N, Üzüm Z, Dijksterhuis J, Lackner G & Hertweck C Active invasion of bacteria into living fungal cells. eLife 3, e03007 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Cleare LG et al. Media matters! Alterations in the loading and release of Histoplasma capsulatum extracellular vesicles in response to different nutritional milieus. Cell Microbiol. 22, e13217 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Marina CL et al. Nutritional conditions modulate C. neoformans extracellular vesicles’ capacity to elicit host immune response. Microorganisms 8, 1815 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Salgueiro VC et al. Extracellular vesicles in mycobacteria: new findings in biogenesis, host–pathogen interactions, and diagnostics. mBio 15, e0255223 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Yang J, Shin T-S, Kim JS, Jee Y-K & Kim Y-K A new horizon of precision medicine: combination of the microbiome and extracellular vesicles. Exp. Mol. Med 54, 466–482 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Menezes SA & Tasca T Extracellular vesicles in parasitic diseases—from pathogenesis to future diagnostic tools. Microbes Infect. 26, 105310 (2024). [DOI] [PubMed] [Google Scholar]
- 86.He B et al. RNA-binding proteins contribute to small RNA loading in plant extracellular vesicles. Nat. Plants 7, 342–352 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sayson SG, Ashbaugh A & Cushion MT Extracellular vesicles from Pneumocystis carinii-infected rats impair fungal viability but are dispensable for macrophage functions. Microbiol. Spectr 12, e0365323 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Costa JH et al. Phytotoxic tryptoquialanines produced in vivo by Penicillium digitatum are exported in extracellular vesicles. mBio 12, e03393–20 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Datta K, Marr KA & Staab JF Methods and compositions using extracellular vesicles for the detection of disease and disorders. Australian Patent AU2020258381A1 (2020).
- 90.De Rezende CP et al. Extracellular vesicles produced during fungal infection in humans are immunologically active. Preprint at bioRxiv 10.1101/2024.03.20.585987 (2024). [DOI] [Google Scholar]
- 91.Datta K et al. Characterisation of mAb476-reactive galactofuranose-bearing Aspergillus antigens excreted in urine. In Proc. 29th European Congress of Clinical Microbiology & Infectious Diseases O1127 (ESCMID, 2019). [Google Scholar]
- 92.Tefsen B, Ram AF, van Die I & Routier FH Galactofuranose in eukaryotes: aspects of biosynthesis and functional impact. Glycobiology 22, 456–469 (2012). [DOI] [PubMed] [Google Scholar]
- 93.Minimal Information for Studies of Extracellular Vesicles (MISEV2023): from Basic to Advanced Approaches (International Society for Extracellular Vesicles, 2024); https://www.isev.org/misev [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Piffer AC et al. Protocol for separation of fungal extracellular vesicles using ultracentrifugation from solid medium cultures. STAR Protoc. 5, 103069 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Schekman R Lasker Basic Medical Research Award. SEC mutants and the secretory apparatus. Nat. Med 8, 1055–1058 (2002). [DOI] [PubMed] [Google Scholar]
- 96.Panepinto J et al. Sec6-dependent sorting of fungal extracellular exosomes and laccase of Cryptococcus neoformans. Mol. Microbiol 71, 1165–1176 (2009). [DOI] [PubMed] [Google Scholar]
- 97.Mao Y & Chen C The Hap complex in yeasts: structure, assembly mode, and gene regulation. Front. Microbiol 10, 1645 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Silva BMA et al. Characterization of Alternaria infectoria extracellular vesicles. Med. Mycol 52, 202–210 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Freitas MS et al. Aspergillus fumigatus extracellular vesicles display increased Galleria mellonella survival but partial pro-inflammatory response by macrophages. J. Fungi 9, 541 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Souza JAM et al. Characterization of Aspergillus fumigatus extracellular vesicles and their effects on macrophages and neutrophils functions. Front. Microbiol 10, 2008 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Brauer VS et al. Extracellular vesicles from Aspergillus flavus induce M1 polarization in vitro. mSphere 5, e00190–20 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Las-Casas LO et al. Pathogenicity and growth conditions modulate Fonsecaea extracellular vesicles’ ability to interact with macrophages. Front. Cell Infect. Microbiol 12, 879018 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Medina-Castellanos E, Salgado-Bautista DA, Martínez-Andrade JM, Cadena-Nava RD & Riquelme M Nanosized extracellular vesicles released by Neurospora crassa hyphae. Fungal Genet. Biol 165, 103778 (2023). [DOI] [PubMed] [Google Scholar]
- 104.De Paula RG et al. Extracellular vesicles carry cellulases in the industrial fungus Trichoderma reesei. Biotechnol. Biofuels 12, 146 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Hill EH & Solomon PS Extracellular vesicles from the apoplastic fungal wheat pathogen Zymoseptoria tritici. Fungal Biol. Biotechnol 7, 13 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Černoša A et al. Isolation and characterization of extracellular vesicles from biotechnologically important fungus Aureobasidium pullulans. Fungal Biol. Biotechnol 9, 16 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Garcia-Ceron D et al. Extracellular vesicles from Fusarium graminearum contain protein effectors expressed during infection of corn. J. Fungi 7, 977 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hatinguais R et al. CRISPR-based tools for targeted genetic manipulation in pathogenic Sporothrix species. Microbiol. Spectr 11, e0507822 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Chaves AFA et al. Updates in Paracoccidioides biology and genetic advances in fungus manipulation. J. Fungi 7, 116 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Da Silva RP et al. Extracellular vesicles from Paracoccidioides pathogenic species transport polysaccharide and expose ligands for DC-SIGN receptors. Sci. Rep 5, 14213 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Gow NAR & Lenardon MD Architecture of the dynamic fungal cell wall. Nat. Rev. Microbiol 21, 248–259 (2023). [DOI] [PubMed] [Google Scholar]
- 112.Micali CO, Neumann U, Grunewald D, Panstruga R & O’Connell R Biogenesis of a specialized plant–fungal interface during host cell internalization of Golovinomyces orontii haustoria. Cell Microbiol 13, 210–226 (2011). [DOI] [PubMed] [Google Scholar]


