Abstract
Background
Limited research exists comparing the impacts of robotic-assisted thoracic surgery (RATS) and video-assisted thoracic surgery (VATS) on patients’ physical and mental health-related quality of life (QoL).
Methods
A prospective cohort of stage IA non–small cell lung cancer (NSCLC) patients in the Initiative for Early Lung Cancer Research on Treatment from Mount Sinai Health System had QoL measured before surgery and at 2, 6, and 12 months post-treatment using the Medical Outcomes Study Short-Form 12 (SF-12), with Physical Component Summary (PCS) and Mental Component Summary (MCS); the Functional Assessment of Cancer Therapy-Lung Cancer Subscale (FACT-LCS); and Patient Health Questionnaire-4 (PHQ-4; for depression/anxiety). A locally weighted smoothing curve was fitted to identify the best interval knot for post-treatment QoL trends. A piecewise linear mixed-effects model was developed to estimate differences in baseline, 2-month, and 12-month QoL scores and rates of change, adjusting for age, sex, race, ethnicity, smoking status, pack-years, nodule size/consistency, comorbidities, and surgical extent.
Results
The study cohort comprised 698 patients, including 458 (65.6%) who underwent VATS and 240 (34.4%) who underwent RATS. The RATS group exhibited a more significant initial decline in physical health at 2 months post-surgery but showed significant recovery by 12 months, achieving similar or slightly higher physical scores compared to baseline. No significant differences in mental health scores over time were seen between the groups. Both groups displayed consistent anxiety and depression scores, with significant improvements in anxiety symptoms at the 2-month mark. The RATS group had fewer postoperative complications and conversion to open thoracotomy.
Conclusions
RATS and VATS offer similar long-term QoL outcomes for early-stage NSCLC patients, though RATS patients may experience a sharper initial decline in physical health.
Key Words: quality of life, surgery, robotic-assisted thoracic surgery, video-assisted thoracic surgery, non–small cell lung cancer
Graphical Abstract

Estimated marginal means of PCS for RATS and VATS patients.
Central Message.
Quality of life outcomes for early-stage non–small cell lung cancer patients after robotic-assisted thoracic surgery and video-assisted thoracic surgery are comparable.
Perspective.
Quality of life (QoL) has important implications in treatment decision making for early-stage lung cancer. This study reveals similar long-term trajectories in physical and mental QoL outcomes for both robotic-assisted thoracic surgery (RATS) patients and video-assisted thoracic surgery patients, though RATS patients face steeper initial declines in physical health, reinforcing the need for critical postoperative care within first 2 months after surgery.
The widespread implementation of computed tomography (CT) screening has markedly improved the early detection of lung cancer, facilitating the treatment of the disease at earlier stages and, consequently, improving patient survival rates.1 Concurrently, surgical innovations have expanded treatment options for non–small cell lung cancer (NSCLC), introducing less invasive techniques such as robotic-assisted thoracic surgery (RATS) and video-assisted thoracic surgery (VATS). VATS, known for its minimally invasive nature relative to traditional thoracotomy, has emerged as the preferred standard for less invasive lung resection, particularly wedge resections.2 In contrast, RATS offers greater precision, a broader range of motion, and enhanced ergonomics and is more often used in lobectomies.3
Numerous studies have compared surgical outcomes between RATS and VATS, yielding mixed results. For instance, one group conducted a retrospective propensity-matched analysis of 613 lung cancer surgeries from 2015 to 2020 and found that while RATS had longer operating times compared to VATS, it resulted in less estimated blood loss. However, there were no significant differences in hospital stay, complication rates, or mortality between the 2 methods.4 Another study comparing 23 early-stage NSCLC patients treated by RATS, 41 treated by VATS, and 39 treated by open surgeries found that RATS was associated with shorter hospital stays and more lymph node stations removed.5 Both studies suggested that RATS may offer certain perioperative advantages over VATS; however, the overall benefits and efficiencies of each approach warrant further investigation to delineate their roles in lung cancer treatment. This highlights the need to consider additional factors when deciding between the 2 surgical options, such as patient characteristics, tumor characteristics, available hospital resources, and surgeon's expertise and preference.6, 7, 8 Particularly, the anticipated postoperative recovery trajectory and potential impacts on the patient's quality of life (QoL) and mental health are critical factors guiding the choice, highlighting the importance of a personalized approach to treatment and recovery planning.
Several studies have investigated QoL outcomes associated with these different surgical modalities and have shown mixed results. For example, the RVlob trial in China (ClinicalTrials.gov identifier NCT03134534) indicated that RATS lobectomy and VATS lobectomy patients experienced comparable QoL and postoperative pain up to 48 weeks postsurgery.9 However, another study found higher QoL among RATS patients compared to VATS and open surgery patients.10 Conversely, RATS patients reported less favorable QoL and higher fear of recurrence after lobectomy compared to VATS patients in another study.11 In addition, a previous study reported a slight difference in coping strategies between these 2 groups.12 Despite these findings, direct comparisons of postoperative QoL trajectories between RATS and VATS for early-stage lung cancer patients remain underexplored. Furthermore, many studies have not thoroughly examined the potential differences between physical and mental health QoL outcomes, which are crucial factors in overall patient recovery and satisfaction.
In previous work, we provided insights into differences in QoL in early-stage lung cancer patients between those undergoing surgery and those receiving stereotactic body radiation therapy13 and between those choosing lobectomy versus limited resection within the surgical group.14 Building on this foundation, the present study aimed to examine the postsurgery QoL trajectories, both physical and mental, of early-stage NSCLC patients who underwent RATS and VATS. This investigation is essential for enhancing patient–surgeon discussions and customizing care strategies to improve individual patient outcomes. We hypothesize that there are no significant differences in the postoperative physical and mental QoL outcomes between RATS and VATS patients.
Methods
Study Design and Population
This study was conducted in accordance with the 2013 Declaration of Helsinki. The Institutional Review Board of the Icahn School of Medicine at Mount Sinai approved the study protocol and publication of data (IRB Study-15-01021; January 23, 2025), with informed consent obtained from all participants.
We reviewed all patients enrolled in the Initiative for Early Lung Cancer Research on Treatment (IELCART) since its inception in 2016 who were diagnosed with NSCLC. Eligibility criteria were patients with a first primary clinical stage IA (T1a–1cN0M0) NSCLC, based on the 8th edition of the American Joint Committee on Cancer's Cancer Staging Manual, who had RATS or VATS (including both lobectomies and sublobar resections), had multiple QoL assessments, and received no additional treatments within the first year post-treatment. Patients with metachronous primaries, recurrences, or any additional treatments within 1 year following their initial treatment were excluded from the study.
Sociodemographic and Medical Characteristics
For the study participants, we collected baseline demographics, including age, sex, education level (under college degree or college and above), smoking status, smoking pack-years and self-reported comorbidities, which covered a range of 12 conditions: asthma, emphysema or chronic obstructive pulmonary disease, high blood pressure, high cholesterol, angioplasty or stent, myocardial infarction, stroke, peripheral vascular disease, liver disease, diabetes, kidney disease, and history of cancers other than lung. Body mass index was calculated from recorded height and weight, and race was categorized into 4 groups: white, African American or black, Asian, and other races). Ethnicity (Hispanic vs non-Hispanic) and nodule consistency (solid, part-solid, nonsolid) also were recorded.
QoL and Mental Health Assessment
During the pretreatment clinic visit, interviews were conducted to collect patients’ pretreatment QoL scores. For patients unable to attend the in-person interviews, telephone interviews or mailed questionnaires were completed. QoL scores during follow-up were acquired at clinic visits scheduled at 1, 6, and 12 months after surgery. The QoL instruments used included the Medical Outcomes Study-Short Form 12 (SF-12) and the Functional Assessment of Cancer Therapy-Lung Cancer Subscale (FACT-LCS). The SF-12 aims to assess health-related QoL through 2 subscale scores: the Physical Component Summary (PCS), which assesses physical health QoL, and the Mental Component Summary (MCS), which assesses mental health QoL. The FACT-LCS specifically measures health-related QoL concerning physical symptoms associated with lung cancer and its treatment. Mental health was assessed using the Patient Health Questionnaire-4 (PHQ-4), a brief screener that assesses symptoms of anxiety and depression.
The PCS score and MCS scores were determined using the 12-item Short Form Health Survey version 2 (SF-12v2), which reflects 8 health domains, including physical functioning, role limitations due to physical health, bodily pain, general health perceptions, vitality, social functioning, role limitations due to emotional problems, and mental health, all within the context of the previous 4 weeks.15 The norm-based PCS and MCS scores for the US general population have a mean of 50 with a standard deviation (SD) of 10, with higher scores indicating better physical and mental health. A minimum difference of 3 points is considered clinically significant.
The FACT-Lung (FACT-L) is a multidimensional validated self-report instrument measuring the QoL of patients diagnosed with lung cancer.16 We used only the LCS subset of items that asks about dyspnea, weight loss, mental clarity, coughing, appetite, chest tightness, and difficulty breathing. The scores range from 0 to 28, with higher scores indicating better overall health and fewer symptoms. A change of 2-3 points is considered clinically significant.17
The PHQ-4 is a short self-report questionnaire asking about patients’ anxiety and depression symptoms.18 It includes the Generalized Anxiety Disorder 2-item (GAD-2) and Patient Health Questionnaire-2 item scale (PHQ-2) subscales, each comprising 2 questions. GAD-2 scores can range from 0 to 6, with lower scores indicating fewer anxiety symptoms. A score of 3 or higher is the recommended cutoff point for possible generalized anxiety disorder.19 The PHQ-2 scores can range from 0 to 6, with lower scores indicating fewer symptoms of depression.18 A score of 3 or higher is the suggested cutoff point for clinical depression.20
Statistical Analysis
We summarized the results of sociodemographic and clinical characteristics using mean and SD for continuous and ordinal data and median and interquartile ranges (IQRs) for continuous and ordinal data. For categorical data, we presented frequencies and percentages. To compare RATS and VATS patients, the 2-sample t test was used for normally distributed continuous variables and the Mann-Whitney U test was used for non-normally distributed continuous variables. For comparisons of categorical variables, Pearson's χ2 test (or Fisher's exact test where appropriate) was used.
We used piecewise linear mixed-effects models to analyze the QoL scores, incorporating fixed effects for surgery type and time since surgery (in months), as well as random effects to account for within-subject correlations over time. Using these models, we calculated the estimated marginal means of QoL scores to compare between the RATS and VATS groups. This approach is consistent with our previous studies comparing QoL between stereotactic body radiation therapy and surgery recipients13 and between lobectomy and sublobar resection patients.14 Interactions between surgery type and time were explored to assess the differential impact on QoL trajectory. Two different rates of change were included in the model for each QoL measure, one from time 0 (baseline) to 2 months after treatment and another from 2.1 to 12 months after treatment. Age, sex, race, ethnicity, smoking status and pack-years, maximum nodule size on CT scan, nodule consistency, comorbidity, and surgery extent were adjusted in the final model. A power analysis was conducted to determine the required sample size, ensuring that the study had 80% power to detect differences in QoL outcomes between the groups, given an effect size (Cohen's d) of 0.3. This analysis suggested the need for a minimum of 176 patients per group to achieve statistically robust conclusions at a 5% significance level. All statistical analyses were performed using R statistical software (R Foundation for Statistical Computing).
Results
Patient Demographics
Among the 698 patients, VATS was the more common procedure (n = 458; 65.6%) compared to RATS (n = 240; 34.4%) (P < .001) (Table 1). Patients undergoing RATS were younger on average compared to the VATS recipients (median age, 68.0 years vs 70.0 years; P < .015). The racial composition also differed significantly in the 2 groups; a larger proportion of RATS patients were Asian (18.0% vs 10.5%; P = .008), while the VATS group had a higher percentage of white patients (69.0% vs 58.2%; P = .008). The RATS group included more current smokers (19.7% vs 11.6%; P = .01) and fewer former smokers (55.2% vs 62.2%; P = .01) and had greater smoking pack-years among smokers (median, 33.0 vs 29.0; P = .045). Additionally, RATS patients had larger nodules (median size, 16.0 mm vs 13.0 mm; P < .001), more solid nodules (79.1% vs 72.7%; P = .049), and fewer part-solid nodules (12.6% vs 20.1%; P = .049) nodules compared to VATS patients. Regarding histology, RATS patients had a higher rate of squamous cell carcinoma (14.2% vs 8.7%; P = .032) and a lower rate of adenocarcinoma (74.1% vs 81.2%; P = .032). More lobectomies (62.8% vs 30.3%; P < .001) and fewer sublobar resections (37% vs 69.7%; P < .001) were performed in RATS patients, while more lymph nodes were resected with RATS than with VATS (median, 8.0 vs 2.0; P < .001). The RATS group had fewer postoperative complications (11.3% vs 24.2%; P < .001) and a lower rate of conversion to open thoracotomy (1.3% vs 3.9%; P = .049).
Table 1.
Sociodemographic, clinical, and surgical characteristics
| Characteristic | Overall (N = 698) | RATS (N = 240) | VATS (N = 458) | P value∗ |
|---|---|---|---|---|
| Age, y, median (IQR) | 69.00 (63.00-76.00) | 68.00 (62.00-74.25) | 70.00 (64.00-76.00) | .015 |
| Sex, n (%) | .442 | |||
| Female | 418 (59.89) | 139 (57.92) | 279 (60.92) | |
| Male | 280 (40.11) | 101 (42.08) | 179 (39.08) | |
| Race, n (%) | .008 | |||
| White | 456 (65.33) | 140 (58.33) | 316 (69.00) | |
| Asian | 91 (13.04) | 43 (17.92) | 48 (10.48) | |
| Black | 88 (12.61) | 37 (15.42) | 51 (11.14) | |
| Others | 63 (9.03) | 20 (8.33) | 43 (9.39) | |
| Ethnicity, n (%) | .207 | |||
| Non-Hispanic | 610 (87.39) | 215 (89.58) | 395 (86.24) | |
| Hispanic | 88 (12.61) | 25 (10.42) | 63 (13.76) | |
| Education, n (%) | <.001 | |||
| College or above | 402 (59.12) | 111 (47.84) | 291 (64.96) | |
| No college | 278 (40.88) | 121 (52.16) | 157 (35.04) | |
| Unknown | 18 | 8 | 10 | |
| Smoking status, n (%) | .010 | |||
| Never | 180 (25.79) | 60 (25.00) | 120 (26.20) | |
| Former | 417 (59.74) | 132 (55.00) | 285 (62.23) | |
| Current | 101 (14.47) | 48 (20.00) | 53 (11.57) | |
| Pack-years (among smokers), median (IQR) | 30.00 (14.00-49.00) | 33.00 (15.00-50.00) | 29.00 (12.13-46.89) | .045 |
| BMI, kg/m2, median (IQR) | 25.70 (22.60-29.40) | 25.70 (22.70-28.90) | 25.70 (22.55-29.50) | .661 |
| Unknown, n | 10 | 3 | 7 | |
| Surgery extent, n (%) | <.001 | |||
| Lobectomy or more | 289 (41.40) | 150 (62.50) | 139 (30.35) | |
| Sublobar | 409 (58.60) | 90 (37.50) | 319 (69.65) | |
| Consistency, n (%) | .049 | |||
| Solid | 523 (74.93) | 190 (79.17) | 333 (72.71) | |
| Part-solid | 122 (17.48) | 30 (12.50) | 92 (20.09) | |
| Nonsolid | 52 (7.45) | 20 (8.33) | 32 (6.99) | |
| Others | 1 (0.14) | 0 (0.00) | 1 (0.22) | |
| Nodule size (max diameter), mm, median (IQR) | 14.35 (10.00-19.00) | 15.65 (11.20-19.78) | 13.40 (9.60-18.48) | <.001 |
| Histology, n (%) | .032 | |||
| Adenocarcinoma | 550 (78.80) | 178 (74.17) | 372 (81.22) | |
| Carcinoid-typical | 50 (7.16) | 16 (6.67) | 34 (7.42) | |
| Squamous cell | 74 (10.60) | 34 (14.17) | 40 (8.73) | |
| Others | 24 (3.44) | 12 (5) | 12 (2.62) | |
| Comorbidities, n (%) | .394 | |||
| None | 100 (14.33) | 38 (15.83) | 62 (13.54) | |
| 1-2 | 350 (50.14) | 112 (46.67) | 238 (51.97) | |
| More than 2 | 248 (35.53) | 90 (37.50) | 158 (34.50) | |
| Number LN station, median (IQR) | 2.00 (1.00-4.00) | 4.00 (3.00-5.00) | 1.00 (1.00-3.00) | <.001 |
| Number LN resected, median (IQR) | 3.00 (1.00-7.00) | 8.00 (4.00-12.25) | 2.00 (1.00-4.00) | <.001 |
| Number LN positive, median (IQR) | 0.00 (0.00-0.00) | 0.00 (0.00-0.00) | 0.00 (0.00-0.00) | .078 |
| Distance costal, median (IQR) | 10.00 (2.00-20.00) | 8.65 (0.00-22.15) | 10.50 (2.20-19.15) | .649 |
| Unknown, n | 5 | 2 | 3 | |
| Distance mediastinal, median (IQR) | 36.90 (21.00, 55.00) | 35.55 (18.15-51.20) | 38.20 (23.05-57.90) | .020 |
| Unknown, n | 18 | 6 | 12 | |
| Distance diaphragmatic, median (IQR) | 101.50 (55.15, 143.10) | 92.30 (56.15-138.03) | 105.80 (55.10-144.55) | .385 |
| Unknown, n | 95 | 60 | 35 | |
| Postoperative event, n (%) | 138 (19.77%) | 27 (11.25) | 111 (24.24) | <.001 |
| Conversion to open, n (%) | 21 (3.01%) | 3 (1.25) | 18 (3.93) | .049 |
| Length of stay, d, median (IQR) | 2.83 (1.83, 3.83) | 2.79 (1.83-3.83) | 2.83 (1.83-3.83) | .025 |
| Unknown, n | 75 | 56 | 19 |
RATS, Robot-assisted thoracoscopic surgery; VATS, video-assisted thoracoscopic surgery; IQR, interquartile range; BMI, body mass index; LN, lymph node.
Wilcoxon rank sum test; Pearson χ2 test; Fisher exact test.
SF-12 PCS
Figure 1 captures the trajectory in estimated marginal mean SF-12 PCS scores for the RATS and VATS patients. Table 2 shows the results of a pairwise comparison of the estimated means between the 2 groups. The differences in PCS scores between the 2 groups were not statistically significant at baseline and 2 months and 12 months after surgery. Before surgery, the RATS and VATS groups had the same estimated marginal mean PCS score: 51.08 for RATS and 51.05 for VATS. At 2 months following surgery, there was a decrease in PCS scores; the RATS group declined to 46.27, while the VATS group decreased to 48.70, with the decrease statistically significant for the RATS group (P = .003) but not for the VATS group (P = .08), as detailed in Table 3. By the 12-month mark, both groups demonstrated significant improvements (P < .001), with a mean PCS score of 52.3 for the RATS group and 52.2 for the VATS group.
Figure 1.
Estimated marginal means of the 12-item Short-Form Health Survey (SF-12) Physical Component Score (PCS) score at baseline and 2 months and 12 months postsurgery. QoL, Quality of life; VATS, video-assisted thoracic surgery; RATS, robotic-assisted thoracic surgery.
Table 2.
Least squares means of QoL scores based on adjusted∗ linear piecewise mixed-effects model
| Score | Baseline estimated score |
2-mo estimated score |
12-mo estimated score |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Estimate | SE | P value† | Estimate | SE | P value† | Estimate | SE | P value† | |
| SF-12 PCS | |||||||||
| RATS | 51.08 | 3.04 | - | 46.27 | 3.10 | - | 53.27 | 3.08 | - |
| VATS | 51.05 | 3.04 | - | 48.70 | 3.68 | - | 53.23 | 3.04 | - |
| RATS – VATS | 0.03 | 0.88 | 1 | −2.43 | 1.16 | .29 | 0.04 | 0.98 | 1 |
| SF-12 MCS | |||||||||
| RATS | 55.05 | 2.74 | - | 58.34 | 2.80 | - | 57.65 | 2.77 | - |
| VATS | 56.38 | 2.75 | - | 56.51 | 2.78 | - | 58.39 | 2.75 | - |
| RATS – VATS | −1.32 | 0.78 | .53 | 1.82 | 1.02 | .47 | −0.73 | 0.86 | .96 |
| FACT-LCS | |||||||||
| RATS | 23.46 | 1.11 | - | 22.54 | 1.13 | - | 24.12 | 1.12 | - |
| VATS | 24.16 | 1.11 | - | 23.87 | 1.12 | - | 25.47 | 1.11 | - |
| RATS – VATS | −0.70 | 0.32 | .25 | −1.32 | 0.42 | .02 | −1.35 | 0.36 | .002 |
| GAD-2 | |||||||||
| RATS | 1.78 | 0.45 | - | 0.83 | 0.46 | - | 0.97 | 0.45 | - |
| VATS | 1.78 | 0.45 | - | 1.07 | 0.46 | - | 1.16 | 0.45 | - |
| RATS – VATS | 0.002 | 0.13 | 1 | −0.24 | 0.17 | .73 | −0.19 | 0.14 | .78 |
| PHQ-2 | |||||||||
| RATS | 0.44 | 0.38 | - | 0.34 | 0.39 | - | 0.16 | 0.39 | - |
| VATS | 0.43 | 0.38 | - | 0.42 | 0.39 | - | 0.28 | 0.38 | - |
| RATS – VATS | 0.01 | 0.11 | 1 | −0.07 | 0.14 | 1 | −0.12 | 0.12 | .90 |
QoL, Quality of life; SE, standard error; SF-12, Short-Form 12; PCS, Physical Component Summary; RATS, robot-assisted thoracoscopic surgery; VATS, video-assisted thoracoscopic surgery; MCS, Mental Component Summary; FACT-LCS, Functional Assessment of Cancer Therapy-Lung Cancer Subscale; GAD-2, Generalized Anxiety Disorder 2-item scale; PHQ-2, Patient Health Questionnaire 2-item scale.
Adjusted for age, sex, race, ethnicity, smoking status and pack-years, maximum nodule size on computed tomography scan, nodule consistency, comorbidity, and extent of surgery.
P values were obtained from adjusted linear piecewise mixed-effects model comparing the estimated means of QoL scores between RATS and VATS at each time point, with a threshold of P < .05 for statistical significance.
Table 3.
Comparison of least squares means of QoL scores from baseline to 2 month to 12 month
| Comparison | RATS |
VATS |
||||
|---|---|---|---|---|---|---|
| Estimate | SE | P value∗ | Estimate | SE | P value∗ | |
| SF-12 PCS | ||||||
| 2 mo vs baseline | −4.81 | 1.12 | .003 | −2.35 | 0.87 | .08 |
| 12 mo vs 2 mo | 7.00 | 1.22 | <.001 | 4.52 | 0.87 | <.001 |
| 12 mo vs baseline | 2.19 | 0.88 | .13 | 2.18 | 0.58 | .002 |
| SF-12 MCS | ||||||
| 2 mo vs baseline | 3.29 | 0.97 | .01 | 0.14 | 0.75 | .99 |
| 12 mo vs 2 mo | −0.69 | 1.06 | .99 | 1.87 | 0.75 | .13 |
| 12 mo vs baseline | 2.60 | 0.76 | .009 | 2.01 | 0.50 | <.001 |
| FACT-LCS | ||||||
| 2 mo vs baseline | −0.91 | 0.41 | .22 | −0.28 | 0.32 | .95 |
| 12 mo vs 2 mo | 1.57 | 0.44 | .004 | 1.59 | 0.32 | <.001 |
| 12 mo vs baseline | 0.66 | 0.32 | .29 | 1.31 | 0.21 | <.001 |
| GAD-2 | ||||||
| 2 mo vs baseline | −0.96 | 0.17 | <.001 | −0.71 | 0.13 | <.001 |
| 12 mo vs 2 mo | 0.14 | 0.18 | .97 | 0.09 | 0.13 | .98 |
| 12 mo vs baseline | −0.81 | 0.13 | <.001 | −0.62 | 0.09 | <.001 |
| PHQ-2 | ||||||
| 2 mo vs baseline | −0.09 | 0.13 | .98 | −0.01 | 0.10 | .99 |
| 12 mo vs 2 mo | −0.19 | 0.14 | .75 | −0.14 | 0.10 | .71 |
| 12 mo vs baseline | −0.28 | 0.10 | .06 | −0.15 | 0.07 | .21 |
QoL, Quality of life; RATS, robot-assisted thoracoscopic surgery; VATS, video-assisted thoracoscopic surgery; SE, standard error; SF-12, Short-Form 12; PCS, Physical Component Summary; MCS, Mental Component Summary; FACT-LCS, Functional Assessment of Cancer Therapy-Lung Cancer Subscale; GAD-2, Generalized Anxiety Disorder 2-item scale; PHQ-2, Patient Health Questionnaire-2 item scale.
P values were obtained from comparing the estimated means of QoL scores (2 mo vs baseline, 12 mo vs baseline, and 12 mo vs 2 mo) using the adjusted linear piecewise mixed-effects model for each surgical approach (RATS and VATS). A P value < .05 was considered statistically significant.
SF-12 MCS
Figure 2 depicts the trends in estimated marginal means of SF-12 MCS scores for patients who underwent RATS and VATS. Table 2 shows that the differences in estimated means between the groups were not significant at any time point. Initially, the RATS group started with a mean MCS score of 55.05, and the VATS group had a mean score of 56.38—both above the average for the general population. At the 2-month point, there was a significant improvement (P = .01) in mean value for the RATS group to 58.34, but an insignificant change for the VATS group (+0.14; P = .99). By the 12-month mark, the mean MCS decreased to 57.65 in the RATS group, but this change was neither statistically nor clinically significant, and the score remained well above the general population's average.
Figure 2.
Estimated marginal means of the 12-item Short-Form Health Survey (SF-12) Mental Component Score (MCS) score at baseline and 2 months and 12 months postsurgery. QoL, Quality of life; VATS, video-assisted thoracic surgery; RATS, robotic-assisted thoracic surgery.
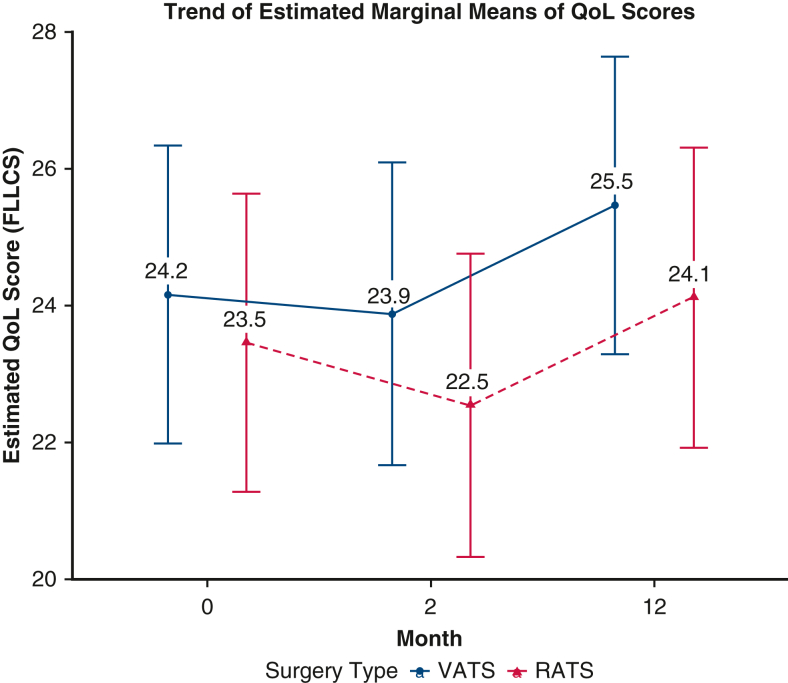
FACT-LCS
Figure 3 shows the trend in estimated marginal mean FACT-LCS scores, reflecting lung cancer–specific symptoms. Initially, the mean score was 23.46 for the RATS group, compared to 24.16 for the VATS group. At 2 months postsurgery, both groups showed a decline in their mean score, but the decreases were not statistically significant, as indicated in Table 3 (P = .22 for RATS; P = .95 for VATS). By the 12-month follow-up, both groups showed statistically significant increases from 2 months (P = .004 for RATS; P < .001 for VATS), with the mean values increasing to 24.12 for the RATS group and to 25.47 for the VATS group. Despite these increases, the estimated mean FACT-LCS scores for the RATS group were statistically significant lower at the 2-month (−1.32; P = .02) and 12-month (−1.35; P = .002) time points (Table 2). However, these differences were smaller than the 2- to 3-point change considered to indicate clinical significance.
Figure 3.
Estimated marginal means of Functional Assessment of Cancer Therapy–Lung Cancer Subscale (FACT-LCS) score at baseline and 2 months and 12 months postsurgery. QoL, Quality of life; FLLCS, Functional Assessment of Cancer Therapy-Lung Cancer Subscale; VATS, video-assisted thoracic surgery; RATS, robotic-assisted thoracic surgery.
GAD-2 and PHQ-2
For anxiety and depression symptoms, as measured by the GAD-2 and PHQ-2 scales, our analysis revealed consistent trends across the 2 groups (Figures 4 and E1). Initially, the RATS and VATS groups had similar mean anxiety scores (both at 1.78) and mean depression scores (RATS, 0.44; VATS, 0.43). At 2 months postsurgery, both groups showed a significant decrease in mean anxiety score (P < .001), to 0.83 for the RATS group and 1.07 for the VATS group. By the 12-month time point, anxiety scores had stabilized, with no significant change from the 2-month scores (RATS, + 0.14; P = .97; VATS, +0.09; P = .98). The PHQ2 depression scores remained consistently low for both groups throughout the 12-month course.
Figure 4.
Estimated marginal means of Generalized Anxiety Disorder 2-item scale (GAD-2) score at baseline and 2 months and 12 months postsurgery. QoL, Quality of life; VATS, video-assisted thoracic surgery; RATS, robotic-assisted thoracic surgery.
Figure E1.
Estimated marginal means of Patient Health Questionnaire-2 item scale (PHQ-2) scores at baseline and 2 months and 12 months postsurgery. QoL, Quality of life; VATS, video-assisted thoracic surgery; RATS, robotic-assisted thoracic surgery.
Discussion
Our study provides a comparison of QoL trajectories for stage IA NSCLC patients undergoing RATS and those undergoing VATS. Physical health declined during the first 2 months after surgery for both RATS and VATS patients, with RATS patients showing a more pronounced initial decline. However, both groups experienced a significant recovery by the 12-month mark and ultimately achieved a higher level of physical health compared to baseline. Similarly, the FACT-LCS scores, which assess specific lung cancer-related symptoms, followed a comparable pattern, with no clinically significant difference between the 2 groups. In contrast to physical health, the mental health scores remained stable over the course of 12 months in both patient groups, consistently remaining above the general population average without significant fluctuations. In terms of anxiety and depression specifically, both groups showed a significant improvement in anxiety symptoms postsurgery, and depression was managed effectively from before surgery to 12 months after surgery. This stability in mental health demonstrates the effectiveness of both surgical techniques in maintaining patient well-being postoperatively and underscores the role of healthcare providers in continuously managing anxiety throughout the recovery process.
The trends in physical health (PCS) and lung cancer–specific symptoms (FACT-LCS) showed that although both the RATS and VATS groups recovered to the same level of physical health at 12 months after surgery, RATS patients experienced a more prominent initial decline within the first 2 months. This could be due to the greater complexity and more challenging anatomic considerations of cases typically chosen for RATS, which often involve larger tumors. Moreover, the more extensive dissection required in RATS might contribute to increased postoperative discomfort or pain. Although the number of incisions is generally similar in RATS and VATS, the specific placement and extent of tissue manipulation might differ, impacting recovery experience. However, we also need to acknowledge that the differences in physical health scores between the 2 groups were not significant at any time point during the recovery period. In addition, we found that the RATS group experienced fewer postoperative complications and lower conversion rates to open thoracotomy, aligning with prior research comparing RATS and VATS.21 These findings suggest a better overall safety profile of the RATS approach during complex procedures, but also emphasize the importance of addressing patients’ needs during the recovery period, especially during the first 2 postoperative months.
The observed disparities in racial composition and smoking status between the RATS and VATS groups suggest that underlying sociocultural factors may influence the choice of surgical approach, which could consequently affect postoperative experiences and patient-reported outcomes. Specifically, the higher proportions of Asian patients and current smokers in the RATS group may reflect specific cultural preferences or perceptions about less-invasive surgeries being associated with faster recovery and less pain, as well as concerns about postoperative complications related to compromised lung function in smokers. Socioeconomic status, often linked with racial differences, also could play a role, as it may dictate access to advanced surgical options like RATS. Furthermore, cultural factors influencing postoperative care and support systems could further influence recovery experience and perceived QoL. Despite the adjustment of race and smoking status in our final models, understanding these multifaceted influences is crucial for healthcare providers to allow them to tailor surgical decisions and postoperative care to optimize patient outcomes.
Our study's strengths lie in its longitudinal approach, capturing QoL at multiple postoperative points. The study's prospective nature significantly minimized recall bias in comparing QoL outcomes, a limitation that has affected many previous studies that relied on retrospective data collection. This enhances the reliability of our findings. The use of a piecewise linear mixed-effects model allowed us to distinctly analyze the change in QoL trajectories before and after the critical 2-month postoperative mark. At the same time, it offers a robust method for handling missing data and measurements taken at irregular time intervals.22 Therefore, we were able to include patients with incomplete QoL data without the risk of introducing bias from excluding them from the analysis.
The choice of treatment in our cohort reflects the shared decisions made by surgeons and patients in clinical practice and, as a result, differences in baseline (preoperative) characteristics may have influenced the postoperative changes observed. It is also essential to recognize that RATS is sometimes used as an alternative to open surgery in more complex cases not suitable for VATS. These potential selection biases might have impacted the homogeneity of the RATS and VATS groups and influenced the QoL outcomes that we reported. In addition, differences in medical knowledge and patient expectations, which were not measured directly in this study, could influence self-reported QoL outcomes. Although we observed higher education levels in the VATS group, unmeasured variables related to patient awareness and expectations remain potential confounders. Despite our efforts to adjust for known confounders in our analysis, there may be residual effects from unmeasured confounders. Another layer of bias arises from the nonblinded nature of surgical approach. Patients aware of their surgical modality may have preconceived notions about the outcomes, which can influence their perception of recovery. Furthermore, our study is limited to the experience within a single health system and focuses on QoL changes within the first year post-surgery. The long-term impacts of VATS and RATS will be evaluated in a subsequent study as longer-term follow-up data are accrued.
In conclusion, both RATS and VATS are valid options for early-stage NSCLC, with both surgical approaches showing favorable outcomes in terms of QoL recovery, both physically and mentally, despite showing initial declines in physical health. Thus, the decision making process for selecting the appropriate surgical method should take into account not only patient demographics and clinical characteristics, but also individual recovery patterns and preferences. Further research is needed to explore the long-term implications of these findings, with a particular focus on understanding the specific sociocultural factors that drive the observed differences in recovery trajectories.
Conflict of Interest Statement
Dr Yankelevitz is a named inventor on a number of patents and patent applications related to the evaluation of chest diseases, including measurements of chest nodules. Dr Yankelevitz has received financial compensation for the licensing of these patents. In addition, he is a consultant and co-owner of Accumetra, a private company developing tools to improve the quality of CT imaging. He is on the advisory board and owns equity in HeartLung, a company that develops software related to CT scans of the chest. He is on the medical advisory board of Median Technology, which is developing technology related to analyzing pulmonary nodules, and is on the medical advisory board of Carestream, a company that develops radiography equipment. He also is on the medical advisory board for LungLife AI. Dr Henschke is an inventor of patents and pending patents owned by Cornell Research Foundation. As of April 2009, she has divested herself of all royalties and other interests arising from these. She is on the medical advisory board for LungLife AI. All other authors reported no conflicts of interest.
The Journal policy requires editors and reviewers to disclose conflicts of interest and to decline handling or reviewing manuscripts for which they may have a conflict of interest. The editors and reviewers of this article have no conflicts of interest.
Footnotes
This work was supported by generous grants from the Simons Foundation International, Ltd. This sponsor had no involvement in the study design, data collection and analysis, data interpretation, or manuscript preparation.
Institutional Review Board approval: MSHS IRB Study-15-01021; approved Jan 23, 2025. All participants provided informed consent.
Contributor Information
Claudia I. Henschke, Email: claudia.henschke@mountsinai.org.
IELCART Investigators:
Raja Flores, Andrew Kaufman, Dong-Seok Lee, Daniel Nicastri, Andrea Wolf, Kimberly Song, Kenneth Rosenzweig, Robert Samstein, Pinaki Dutta, Jorge Gomez, Mary Beth Beasley, Maureen Zakowski, Michael Chung, David F. Yankelevitz, Claudia I. Henschke, Emanuela Taioli, Yeqing Zhu, Natela Paksashvili, Lijing Zhang, Lyu Lyu, Huiwen Chan, Jeffrey Zhu, Sydney Kantor, Lauren Lentini, Daniel Nicastri, Ardeshir Hakami, Arzu Buyuk, Adie Friedman, Ronald Dreifuss, Stacey Verzosa, Karina Aloferdova, Patricia Stacey, Simone De Nobrega, Jeffrey Zhu, Sydney Kantor, Lauren Lentini, Ardeshir Hakami, Jeffrey Zhu, Sydney Kantor, Lauren Lentini, Harvey Pass, Benjamin Cooper, Andre Moreirea, Audrey Sorensen, Leslie Kohman, Robert Dunton, Jason Wallen, Christopher Curtiss, Ernest Scalzetti, Linda Ellinwood, Henry Tannous, Cliff P. Connery, Emilo Torres, Dan Cruzer, Bruce Gendron, Sonya Alyea, Pramila Krumholtz, Ammara Watkins, Elliot Servais, Cameron Stock, Andrea McKee, Edilin Lopez, Howard Hsu, Kaudia Hunter, Jeffrey Lemons, Asa Nixon, Etin-Osa Osa, Paul Lee, Kevin Hyman, Julisa Jurado, David Zeltman, Lawrence Glassman, Rajiv Sharma, Vijay Singh, Rebecca Schwartz, Efstathia Milhelis, Nandanee Karan, Witold Rzyman, Robert Dziedzic, Claudia Henschke, Emanuela Taioli, David Yankelevitz, Rebecca Schwartz, Artit Jirapatnakul, Rowena Yip, Huiwen Chan, Jeffrey Zhu, and Sydney Kantor
Appendix E1
References
- 1.Lancaster H.L., Heuvelmans M.A., Oudkerk M. Low-dose computed tomography lung cancer screening: clinical evidence and implementation research. J Intern Med. 2022;292(1):68–80. doi: 10.1111/joim.13480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gaudet M.A., D’Amico T.A. Thoracoscopic lobectomy for non–small cell lung cancer. Surg Oncol Clin N Am. 2016;25(3):503–513. doi: 10.1016/j.soc.2016.02.005. [DOI] [PubMed] [Google Scholar]
- 3.Solinas M., Novellis P., Veronesi G. Robotic is better than VATS? Ten good reasons to prefer robotic versus manual VATS surgery in lung cancer patients. VATS. 2017;2:60. [Google Scholar]
- 4.Lampridis S., Maraschi A., Le Reun C., Routledge T., Billè A. Robotic versus video-assisted thoracic surgery for lung cancer: short-term outcomes of a propensity matched analysis. Cancers (Basel) 2023;15(8):2391. doi: 10.3390/cancers15082391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Novellis P., Bottoni E., Voulaz E., et al. Robotic surgery, video-assisted thoracic surgery, and open surgery for early stage lung cancer: comparison of costs and outcomes at a single institute. J Thorac Dis. 2018;10(2):790–798. doi: 10.21037/jtd.2018.01.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yu Z., Xie Q., Guo L., et al. Perioperative outcomes of robotic surgery for the treatment of lung cancer compared to a conventional video-assisted thoracoscopic surgery (VATS) technique. Oncotarget. 2017;8(53):91076–91084. doi: 10.18632/oncotarget.19533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Veluswamy R.R., Whittaker Brown S.A., Mhango G., et al. Comparative effectiveness of robotic-assisted surgery for resectable lung cancer in older patients. Chest. 2020;157(5):1313–1321. doi: 10.1016/j.chest.2019.09.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sullivan D.R., Eden K.B., Dieckmann N.F., et al. Understanding patients’ values and preferences regarding early stage lung cancer treatment decision making. Lung Cancer. 2019;131:47–57. doi: 10.1016/j.lungcan.2019.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jin R., Zhang Z., Zheng Y., et al. Health-related quality of life following robotic-assisted or video-assisted lobectomy in patients with non–small cell lung cancer: results from the RVlob randomized clinical trial. Chest. 2023;163(6):1576–1588. doi: 10.1016/j.chest.2022.12.037. [DOI] [PubMed] [Google Scholar]
- 10.Asemota N., Maraschi A., Lampridis S., et al. Comparison of quality of life after robotic, video-assisted, and open surgery for lung cancer. J Clin Med. 2023;12(19):6230. doi: 10.3390/jcm12196230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Williams A.M., Zhao L., Grenda T.R., et al. Higher long-term quality of life metrics after video-assisted thoracoscopic surgery lobectomy compared with robotic-assisted lobectomy. Ann Thorac Surg. 2022;113:1591–1597. doi: 10.1016/j.athoracsur.2020.05.033. [DOI] [PubMed] [Google Scholar]
- 12.Chen Y.P., Zhang Y., Chen X., et al. The effects of different surgical approaches on the psychological status, medical coping mode and quality of life of patients with lung cancer. Front Psychol. 2023;14 doi: 10.3389/fpsyg.2023.1039501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang J., Yip R., Taioli E., et al. Change in quality of life of stage IA non–small cell lung cancer after surgery or radiation therapy. J Thorac Dis. 2024;16(1):147–160. doi: 10.21037/jtd-23-1201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Février E., Yip R., Becker B.J., et al. Change in quality of life of stage IA lung cancer patients after sublobar resection and lobectomy. J Thorac Dis. 2020;12(7):3488–3499. doi: 10.21037/jtd-20-402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ware J.E., Jr. SF-36 health survey update. Spine (Phila Pa 1976) 2000;25(24):3130–3139. doi: 10.1097/00007632-200012150-00008. [DOI] [PubMed] [Google Scholar]
- 16.Cella D.F., Bonomi A.E., Lloyd S.R., Tulsky D.S., Kaplan E., Bonomi P. Reliability and validity of the functional assessment of cancer therapy-lung (FACT-L) quality of life instrument. Lung Cancer. 1995;12(3):199–220. doi: 10.1016/0169-5002(95)00450-f. [DOI] [PubMed] [Google Scholar]
- 17.Cella D., Eton D.T., Fairclough D.L., et al. What is a clinically meaningful change on the functional assessment of cancer therapy-lung (FACT-L) questionnaire? Results from eastern cooperative oncology group (ECOG) study 5592. J Clin Epidemiol. 2002;55(3):285–295. doi: 10.1016/s0895-4356(01)00477-2. [DOI] [PubMed] [Google Scholar]
- 18.Kroenke K., Spitzer R., Williams J.B., Löwe B. An ultra-brief screening scale for anxiety and depression: the PHQ-4. Psychosomatics. 2009;50:613–621. doi: 10.1176/appi.psy.50.6.613. [DOI] [PubMed] [Google Scholar]
- 19.Kroenke K., Spitzer R.L., Williams J.B., Monahan P.O., Löwe B. Anxiety disorders in primary care: prevalence, impairment, comorbidity, and detection. Ann Intern Med. 2007;146(5):317–325. doi: 10.7326/0003-4819-146-5-200703060-00004. [DOI] [PubMed] [Google Scholar]
- 20.Kroenke K., Spitzer R.L., Williams J.B. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41(11):1284–1292. doi: 10.1097/01.MLR.0000093487.78664.3C. [DOI] [PubMed] [Google Scholar]
- 21.Novellis P., Maisonneuve P., Dieci E., et al. Quality of life, postoperative pain, and lymph node dissection in a robotic approach compared to VATS and OPEN for early stage lung cancer. J Clin Med. 2021;10(8):1687. doi: 10.3390/jcm10081687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fitzmaurice G.M., Laird N.M., Ware J.H. 2nd ed. John Wiley & Sons; 2011. Applied Longitudinal Analysis; pp. 410–412. [Google Scholar]