ABSTRACT
Mitochondrial transplantation (MT), an innovative regenerative technique widely used to treat diseases caused by mitochondrial dysfunction, shows great promise for clinical application. This procedure can increase the number of mitochondria and improve the function of damaged mitochondria, resulting in increased adenosine triphosphate levels, decreased reactive oxygen species production, improved Ca2+ buffering capacity, modulated inflammatory response, and reduced apoptosis to protect cells, thus promoting tissue repair. In this review, we describe research advances in MT over the last five years, focusing on its application in treating various diseases, including ischaemic injuries (of the kidney, heart, lung, and liver), neurodegenerative disorders, spinal cord injury, sepsis, diabetes mellitus, stroke, and ultraviolet radiation injuries, as well as in procedures such as organ transplantation, focusing on instances where MT demonstrated good efficacy. We also cover the application of engineered mitochondria and mitochondrial combination therapies and present the latest advances in improving MT efficiency, as well as the current clinical applications and shortcomings of MT, aiming to provide a theoretical foundation for enhanced MT utilisation in the future.
Keywords: cardiovascular diseases, ischaemia/reperfusion injury, mitochondrial dysfunction, mitochondrial transplantation, neurodegenerative diseases
Introduction
Mitochondria are membrane-bound organelles that are essential for cellular activities and energy metabolism in eukaryotes. They are responsible for transmitting information within and between cells and regulating various life-sustaining mechanisms, such as adenosine triphosphate (ATP) and lipid synthesis, reactive oxygen species (ROS) production, Ca2+ homeostasis, and various cellular processes (proliferation, autophagy, survival, and apoptosis).1 Mitochondria could protect injured cells by increasing ATP production, decreasing ROS production, and increasing Ca2+ buffering capacity.2 The structural composition of mitochondria is shown in Figure 1. Although physiological ROS levels are vital, serving as second messengers that promote various signalling pathways, excessive ROS could cause oxidative stress (OS) and elevate intracellular Ca2+ levels, preventing mitochondrial movement, which could affect the removal and replenishment of damaged and healthy mitochondria, respectively.3 Proper Ca2+ accumulation in mitochondria improves mitochondrial function by activating oxidative phosphorylation (OXPHOS) and Ca2+-dependent mitochondrial enzymes, stimulating ATP production, and increasing aerobic respiration.4 According to research, mitochondrial dynamics, such as mitochondrial fission, fusion, degradation, and cristae remodelling, are crucial for mitochondrial function and eukaryotic cell survival.5, 6 These dynamics could also contribute to the regulation of mitochondrial morphology, mitochondrial DNA (mtDNA), material exchange, OXPHOS activity, and degradation of damaged mitochondria.7 Mitochondrial cristae remodelling may be related to inter-mitochondrial signalling, and an increase in cristae folds can upregulate ATP synthesis by increasing the surface area available for OXPHOS processes.5 Furthermore, the balance between mitochondrial fusion and fission controls the mitochondrial size, number and shape. Specifically, a decrease in fusion or an increase in fission could increase the population of smaller mitochondria, often appearing as short tubules or spheres; in neurons, such mitochondria are more efficiently translocated in the axon.7 Mitochondrial fission is also associated with biogenesis and autophagy.8 Furthermore, when mitochondrial fission was blocked, mitochondria assumed an elongated shape with limited functionality in promoting neurite extension and supporting axons. Such mitochondria may produce ATP more efficiently7 and inhibit breast cancer metastasis,9 tumour cell stemness,10 and chemoresistance.11 Moreover, mitochondrial fusion could help reduce the burden on stressed mitochondria, enhancing their ability to undergo autophagy and produce more ATP.8
Figure 1. The structure of mitochondria. Created with BioRender.com. ATP: adenosine triphosphate; mtDNA: mitochondrial DNA.
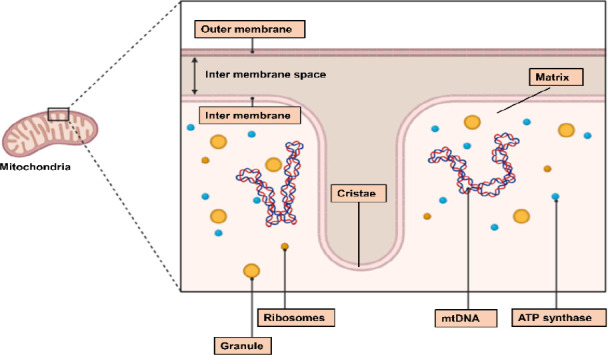
However, changes in the cellular state in pathological environments could cause structural damage to mitochondria, leading to dysfunctional mitochondria, with typical phenotypes of membrane ruffling, swelling, and cristae disorganisation.12 These changes could also cause mitochondrial dysfunction, as evidenced by decreased ATP levels, increased ROS levels, and decreased mitochondrial membrane potential (MMP). The other sign of mitochondrial dysfunction caused by changes in the cellular state is mitochondrial membrane permeability,13 which could lead to functional damage of the cells or their complete death, causing organ dysfunction and even serious health problems such as ageing,14 metabolic disorders,15 cancer,16 and neurodegenerative diseases.17 Additionally, mitochondria have been repeatedly shown to be critically involved in apoptosis,18-20 especially mitochondrial outer membrane permeabilisation has been shown to be associated with a variety of apoptotic responses, such as DNA damage, growth factor deprivation, nutrient deprivation, etc., and is involved in the activation of pro-apoptotic proteins, such as caspases, which are mainly controlled by the Bcl-2 family of proteins.21 Damaged mitochondria are capable of self-repair and cytoprotection via fission, fusion, autophagy, and intercellular transfer mechanisms.22, 23 Furthermore, mitochondria can migrate between cells, offering the possibility of transplanting exogenous healthy mitochondria into cells with damaged mitochondria.23 Currently, MT is widely being explored as an effective novel intervention for the treatment of diseases associated with mitochondrial dysfunction.
In this review, we present research advances in MT and efforts to improve transplantation efficiency, summarising the role played by MT and its therapeutic effects in the treatment of various diseases in the last five years, including ischaemic injuries (of kidney, heart, lung, and liver) and neurological disorders, as well as in procedures such as organ transplantation. We also present strategies for mitochondrial engineering and mitochondrial combination therapies, as well as the current clinical applications of MT, with the aim of providing a theoretical basis for increased use of MT in the future.
Search Methods
We searched PubMed for relevant published studies, using keywords such as ‘mitotherapy’, ‘mitochondria’ OR ‘mitochondrial transplantation’ AND ‘kidney injury’ OR ‘lung injury’ OR ‘liver injury’ OR ‘heart’ OR ‘IRI’ OR ‘pulmonary hypertension’ OR ‘osteoarthritis’ OR ‘muscle damage’ OR ‘neurodegenerative diseases’ OR ‘brain injury’ OR ‘spinal cord injury’ OR ‘nerve injury’. These keywords are combined with other relevant search terms. References from these articles and recommendations of similar articles were used to obtain more relevant articles.
General Overview of Mitochondrial Transplantation
Characterisation of mitochondrial transplantation
Mitochondrial transplantation (MT), also known as mitochondrial therapy (mitotherapy), is an innovative therapeutic approach for treating mitochondria-related diseases. It involves replacing damaged mitochondria with healthy mitochondria isolated or purified from normal cells or tissues with normal respiratory function, thereby increasing mitochondrial population and improving mitochondrial function.24, 25 Research suggests that exogenous MT could replace damaged or unhealthy mitochondria with healthy mitochondria, restoring and fulfilling the function of the former.26 In this context, MT has been widely used in treating various diseases (Table 1).27-56 And transplanted mitochondria were derived from different origins (autologous, allogeneic, and xenogenic sources). Some studies have shown that MT in vivo does not appear to cause any adverse reactions or chronic alloreactivity, allorecognition or damage-associated molecular pattern molecules reaction response in mice.13, 25, 26, 57 Tissue or cell sources that have previously been used for MT research include the placenta,58 liver, skeletal muscle cells, cardiomyocytes, fibroblasts, mesenchymal stem cells (MSCs), astrocytes, adipocytes, platelets, etc.23, 59-64 Mitochondria derived from different sources may be tissue-specific and could play different roles, possibly related to Ca2+ regulation and expression of tissue-specific nuclear-encoded proteins.65 For instance, liver-derived mitochondria exhibit a relatively high OXPHOS coupling index and a high mitochondrial concentration, and the large size and regenerative capacity of the liver in humans are mainly associated with biosynthetic processes.66, 67 Furthermore, the function of transplanted mitochondria may differ based on donor gender. For example, Yu et al.68 used mitochondrial transplants from different mouse genders for melanoma treatment and discovered that both types of mitochondria significantly inhibited tumour cell proliferation, growth, and migratory functions and also promoted tumour cell apoptosis. However, female mouse-derived mitochondria showed a stronger anti-tumour activity. Specifically, they greatly inhibited tumour cell proliferation and also showed a higher protein content, pyruvate dehydrogenase activity, and ATP production capacity than male-derived mitochondria.68, 69 Regarding the survival of transplanted mitochondria, it is advisable to use them promptly after extraction, rather than storing them for long before transplantation. A study comparing the efficacy of freeze-thawed and fresh mitochondrial transplants in treating severe ischaemia/reperfusion injury (IRI) after cardiac arrest discovered that fresh mitochondria had four times the ATP content of freeze-thawed mitochondria and more significantly enhanced the survival rate of the affected rats. This outcome could be due to freezing and thawing yielding relatively nonfunctional mitochondria.70 Research has also shown that, mitochondria transplanted in vivo were found to survive in cardiomyocytes for ≥ 28 days while remaining functional and active.71 And transplanted mitochondria via intravenous injection were shown to persist in the brain, kidneys, and spleen for as up to 24 hours after severe IRI following cardiac arrest, whereas the heart or lungs showed no mitochondrial persistence.69
Table 1. Application of MT in the treatment of different diseases.
| Disease model | Animal | Mitochondrial source | Delivery method | Mitochondrial concentration | Reference |
|---|---|---|---|---|---|
| AKI | Female Yorkshire pigs | Sternocleidomastoid muscle, human cardiac fibroblasts | Renal artery injection, intravascular injection | 1 × 109 mitochondria in 6 mL respiration buffer | 27 |
| Yorkshire pigs | ciPTECs | Renal artery injection | 0.5 mL isolated mitochondria diluted to 10 mL in PBS | 28 | |
| Male Wistar rats | Pectoralis major muscle tissue | Renal artery injection | 7.5 × 106/mL, 400 µL | 29 | |
| Male SD rats | Bilateral soleus muscles | Intratracheal instillation | 100 µg mitochondria in 200 µL PBS | 30 | |
| Male C57BL/6J mice | Gastrocnemius muscle | Pulmonary artery/trachea/nebulisation delivery | 1 × 109 mitochondria in 0.5 mL respiration buffer | 31 | |
| Kidney ischaemia/reperfusion injury | Male Wistar rats | Pectoralis major muscle cells | Renal artery injection | 7.5 × 106/mL, 0.4 mL | 29 |
| LPS-induced lung injury | Male Fisher C57BL/6 mice | ADSCs | Intranasal instillation | 2.5 × 105 AMs/mouse | 32 |
| Liver ischaemia/reperfusion injury | Male Wistar rats | liver | Splenic injection | 7.7 × 106± 1.5 × 106/mL, 100 µL | 33 |
| Adult male Fisher rats | ADSCs | Tail vein injection | 1.2 × 106mitochondria | 34 | |
| Carbon tetrachloride-induced liver injury | Kunming mice | Mouse liver | Intravenous injection | 0.2, and 0.4 mg/kg | 35 |
| Hypoxic pulmonary hypertension | SD rats | Femoral artery smooth muscle cells | Tail vein injection | 2.25 × 108/ mL, ~2 µg | 36 |
| Myocardial ischaemia/reperfusion injury | Male New Zealand white rabbits | Pectoralis major muscle | Subendocardial injection | 9.7 × 106 ± 1.7 × 106 mitochondria/mL, 0.1 mL per injection site, 8 sites in total | 37 |
| Females Yorkshire pigs | Pectoralis major muscle | Subendocardial injection | 9.9 × 107 ± 1.4 × 107 mitochondria/mL; 1.3 × 107 mitochondria per injection site, 8 sites in total | 38 | |
| Intracoronary injection | Single injection 1 × 109 in 6 mL; Serial injections (10 injections of 1 × 109 in 6 mL were delivered every 5 minutes) | 39 | |||
| Right ventricular hypertrophy/right ventricular failure | Immature male Yorkshire piglets | soleus muscle, gastrocnemius muscle | Injection into the right ventricle | 10 × 106/mL, 1 mL | 40 |
| Sepsis | SD rats | L6 muscle cells | Tail vein injection | 50 µg | 41 |
| Male Wistar rats | / | Tail vein injection | 7.5 × 106 mitochondria/mL, 400 µL | 42 | |
| Male C57BL/6 mice | Homologous pectoralis major muscle | Tail vein injection | 2.5–3 × 107 particles/100 µL | 43 | |
| Optic nerve injury | Lister hooded rats | Liver | Vitreous cavity injection | 0.25 mg/mL, 5 µL | 44 |
| SCI | Male SD rats | Soleus muscle | Jugular vein injection | Mitochondria (100 µg) were resuspended in 0.2 mL of PBS | 45 |
| C57BL/6 mice | Mouse BMDMs | Intravenous injection | 2–5 × 106/mL, two injections of 200 µL on the 3rd and 5th days after injury | 46 | |
| Chronic stress-induced apoptosis | Male Wistar rats | Brain | Intra-cerebroventricular injection | 10 µL | 47 |
| Ischaemic stroke | Male SD rats | Astrocytes | Striatum | 100 µg/rat | 48 |
| Acute ischaemic stroke | Male C57BL6 mice | Snap-frozen placenta, freshly isolated skeletal muscle, brain, brown adipose tissues | Intravenous injection | 100 µg protein/100 µL saline/mouse | 49 |
| Sciatic nerve crush injury | SD rats | Baby hamster kidney fibroblast cells (BHK-21 cells) | Sciatic nerve (the epineurium 10 mm distal to the crushed site) | 195 µg of mitochondria diluted in PBS with volume of 100 µL | 50 |
| Peripheral nerve injury | Female SD rats | hUCMSCs | ANA injection | 195 µg, 30 µL per ANA | 51 |
| Bilateral cavernous nerve injury | Male SD rats | ADSCs | Corpus cavernosum | 50 µg/mL, 300 µg | 52 |
| Bacl2 induced skeletal muscle injury | Male C57BL/6J mice | Mouse liver | Gastrocnemius muscle injection | 50 µg/mL | 53 |
| OA | Male Wistar rats | L6 rat cells | Intra-articular space injection in the right knee | 10 µg/50 µL, twice weekly | 54 |
| Acute limb ischaemia | Male C57BL/6J mice | Nonischaemic skeletal muscle | All the muscles of the hindlimb injection | 1 × 106, 1 × 107, 1 × 108, and 1 × 109 mitochondria per gram muscle wet weigh | 55 |
| Duchenne muscular dystrophy | Male C57BL10 mice | Mouse quadriceps muscle | Quadriceps and gastrocnemius muscle injection | 1 µg mitochondrial protein/g body weight in total, per ~50 µL | 56 |
Note: ADSCs: adipose derived mesenchymal stem cells; AKI: acute kidney injury; ALI: acute lung injury; ANA: acellular nerve allografts; BMDM: bone marrow-derived macrophage; ciPTECs: human conditionally immortalised proximal tubular cells; hUCMSCs: human umbilical cord-derived mesenchymal stem cells; LPS: lipopolysaccharide; MT: mitochondrial transplantation; OA: osteoarthritis; PBS: phosphate buffered solution; SCI: spinal cord injury; SD: Sprague-Dawley.
In addition to performing normal mitochondrial functions, transplanted mitochondria could also exert anti-inflammatory effects,72 such as increasing the activity of macrophages and T lymphocytes, stimulating the immune system.73 Furthermore, MT reduces pro-inflammatory cytokine production and lipid accumulation in foamy macrophages, increases macrophage phagocytosis, and achieves anti-inflammatory effects by blocking OXPHOS, nuclear allosterism, and nuclear factor κB (NF-κB) activity; therefore, it may be used to treat inflammatory diseases and ameliorate atherosclerosis.74-76 Additionally, MT in T cells reduces T cell activation and infiltration while promoting Treg cell differentiation in tissues, thereby suppressing inflammatory responses.61
Methods of mitochondrial transplantation
Targeted MT is key to improving MT efficiency. Various MT delivery strategies, including systemic delivery, direct microinjection, vesicle- or liposome-mediated delivery, and cell-mediated delivery using tunneled nanotubes, were shown to potentially affect transplantation efficacy.65 For example, mitochondria-rich extracellular vesicles (EVs) were found to be more effective than MT alone because the lipid bilayer of EVs, which blocks Ca2+-induced mitochondrial damage and fuses directly with the cell membrane, is more conducive to mitochondrial transport into the recipient cell’s cytoplasm.77 However, this method complicates the transportation of large amounts of mitochondria, and the vesicles may also contain other elements besides mitochondria, further making it difficult to discern the role played by MT.65 Furthermore, mitochondrial damage may result from injection.78 On the other hand, magnetic nanoparticles, co-incubation, and Pep-1 (KETWWETWWTEWSQPKKKRKV-cysteamine, one of the family of cell-penetrating peptides)-coupled mitochondria are currently being explored to further improve MT efficiency and some of their impacts on transplantation efficacy have already been identified.79-81 For example, compared to normal MT, cell-penetrating peptide Pep-1-mediated transplantation further increased mitochondrial permeability.82 There are many approaches to transplant mitochondria into cells or tissues (Figure 2).
Figure 2. Approaches for transplanting exogenous mitochondria into cells. Created with BioRender.com.
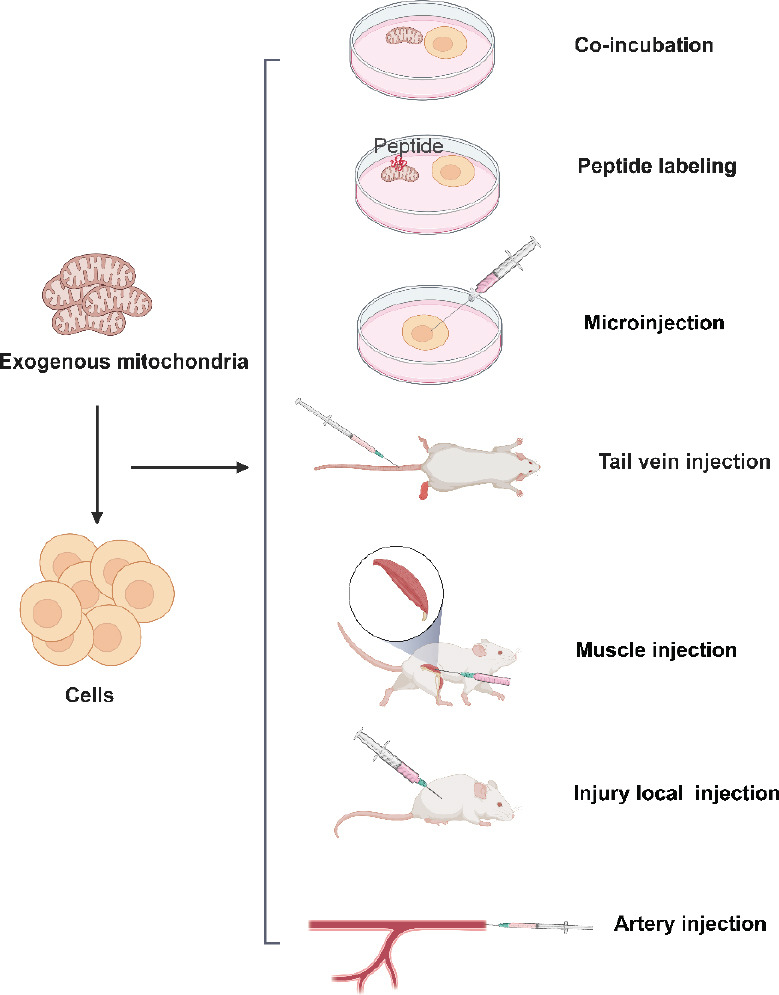
Numerous scholars have recently developed novel MT approaches. For example, Patel et al.57 used a thermally-gelling and erodible hydrogel system to deliver mitochondria to the site of spinal cord injury. This approach protected the mitochondria under physiological conditions in vitro, maintaining their integrity, and also increased the effective uptake rate of neuronal cells, greatly enhancing their metabolic vigour. Additionally, Gäbelein et al.83 proposed a nano-injector that enables the rapid extraction of mitochondria from living single cells and their transplantation into recipient host cells, with a success rate of up to 95%. This approach also maintains mitochondrial activity and integrity and promotes mitochondrial internalisation by cells, thereby increasing MT efficiency. Furthermore, Kim et al.84 developed a centrifugal delivery method that allows mitochondria to cross the cell membrane more easily without causing intracellular damage while also increasing ATP content and improving cellular metabolic activity, allowing for the restoration of mitochondrial function in damaged cells. This method also extracts enough mitochondria from a small number of cells and delivers them efficiently to target cells. On the other hand, Baudo et al.85 used a dextran and triphenylphosphine polymer coupling to functionally isolate mitochondria from cells, resulting in increased mitochondrial internalisation, reduced macrophage glycolysis and decreased pro-inflammatory cytokine expression. Furthermore, MT into transforming growth factor-β-treated fibroblasts via this approach reduced fibrosis by lowering ROS production, extracellular matrix protein expression, and myofibroblast migration and proliferation. Using a similar approach, Wu et al.86 discovered that dextran functionalisation of mitochondria could significantly enhance mitochondrial engraftment efficiency by placing isolated mitochondria in a metabolic dormant state, thus protecting their effective respiratory function.
Advances in Mitochondrial Transplantation in the Treatment of Various Diseases
Kidney, lung, and liver injuries
Kidney injury
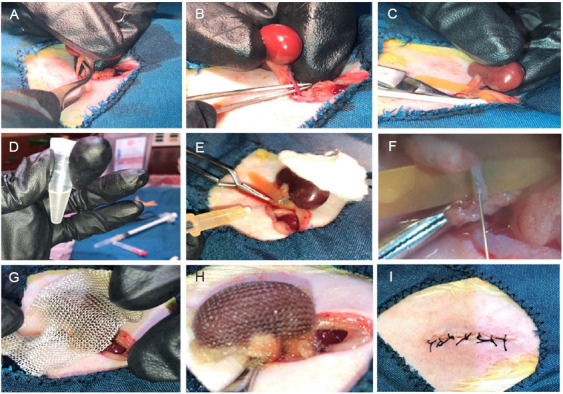
Mitochondrial dysfunction in the kidney can cause acute and chronic renal failure, as well as diabetic nephropathy.87 Since it protects kidney cells and reduces kidney injury by decreasing ROS production, maintains MMP, generates more ATP, and decreases inflammatory responses, MT has been highlighted in multiple studies as a very promising therapeutic stratege for treating kidney injury. For instance, it was reported that the transplantation of mitochondria derived from MSCs leaded to a reduction in the apoptosis of injured renal cells. Moreover, it promoted the proliferation and regeneration of renal tubular cells post-injury, thereby attenuating IRI-induced renal injury.88 Additionally, Preble et al.27 uncovered that directly injecting mitochondria derived from sternocleidomastoid muscle significantly protects the swine kidney from renal IRI, while promoting tissue repair by enhancing the proliferative potential of the injured epithelium. Using a porcine model, Doulamis et al.89 discovered that intra-arterial mitochondrial injection protected the kidneys, reduced proximal tubular necrosis, and downregulated interleukin (IL)-6 (a pro-inflammatory factor), thereby attenuating the inflammatory response. The transplantation of healthy mitochondria also exerts a renoprotective effect by decreasing proximal tubular cytotoxicity induced by gentamicin and famciclovir, decreasing ROS production, inhibiting MMP collapse, and reducing the lipid peroxidation content.26, 90 Moreover, mitochondrial dysfunction has also been identified as a key factor in renal tubular cell death during acute kidney injury. In this regard, multiple studies have shown that MT can effectively treat acute kidney injury. For example, Jabbari et al.29 treated acute kidney injury through the transplantation of muscle-derived mitochondria and then discovered that MT could ameliorate renal injury and restore renal function by decreasing renal tubular cell death and apoptosis during IRI and increasing tubular regeneration. Furthermore, Rossi et al.28 through both in vivo and in vitro experiments, demonstrated that transplanting mitochondria derived from human conditionally immortalised proximal tubular cells enhanced the proliferative capacity of proximal tubular cells and attenuated renal injury by producing more ATP and reducing toxicity and ROS production. Figure 3 shows how exogenous mitochondria are transplanted through the renal artery injection into the kidney in the treatment of IRI-induced renal injury model in rat.29
Figure 3. Application of mitochondrial transplantation in the treatment of ischaemia/reperfusion-induced renal injury model in rat by renal artery injection. (A–C) Ischaemia/reperfusion was induced by ligature of left renal hilum for 45 minutes. (D) The isolated mitochondria derived from pectoralis major muscle cells already prepared for the transplantation. (E, F) The mitochondria were immediately injected into renal artery using a 32 gauge needle. (G–I) After transplantation, the kidney was returned to abdomen and the anterior abdominal wall was closed with sutures. Reprinted from Jabbari et al.29.
Lung injury
MT as a salutary intervention for lung tissue injury, principally through the augmentation of OXPHOS and the attenuation of inflammatory responses. MSC-derived mitochondria, when transplanted into injured lungs, have been shown to increase OXPHOS, inhibit the macrophage secretion of pro-inflammatory cytokines, and increase M2-type macrophages, thereby ameliorating lung injury.29 Furthermore, Pang et al.30 discovered that MT using mitochondria isolated from the soleus muscles of rats reduced inflammatory cell infiltration into acute lung injury-affected lung tissues, which not only enhanced the endothelial cell function of pulmonary blood vessels but also improved gas exchange during the acute phase of acute lung injury, leading to significant restoration of the damaged lung parenchyma.
Additionally, MT also exerts a therapeutic effect against IRI that occurs during lung transplantation. Cloer et al.91 discovered that the transplantation of mitochondria harvested from porcine hearts into lungs affected by IRI reduced pulmonary vascular resistance and OS. Additionally, it suppressed the expression of pro-inflammatory cytokines (IL-6 and IL-1β), and inflammation-related chemokines, thereby attenuating ex vivo lung perfusion-induced lung tissue damage. Moskowitzova et al.31 also discovered that transplantating mitochondria derived from gastrocnemius muscle in mice via vascular transport or nebulisation boosted ATP content in injured lung tissue, replaced damaged mtDNA, and enhanced lung dynamics, reducing lung tissue damage.
In conclusion, these findings suggest that MT not only attenuates cellular damage and inflammation, but may also initiate intrinsic cellular mechanisms that foster tissue repair and regeneration, marking a significant advance in our approach to lung therapy.
Liver injury
Numerous studies have underscored the efficacy of MT in the treatment of liver injuries, including toxin- and IRI-induced liver injury, through mechanisms such as increasing ATP production, decreasing OS, and inhibiting inflammatory responses. For example, by transplanting rat liver-derived mitochondria into the spleen, Lin et al.92 discovered that MT significantly reduced hepatocyte apoptosis after hepatic ischaemia/reperfusion, mitigated mitochondrial damage, and effectively ameliorated OS. Notably, the mitochondria maintained an intact membrane potential for up to 240 minutes after transplantation into the injured liver, signalling ongoing mitochondrial activity. Similarly, Ulger et al.93 found that MT, with mitochondria derived from a rat MSC line, reduced hepatocyte apoptosis and OS induced by p-acetylamino acid toxicity in acute liver-injured rats in vivo, thus attenuating hepatotoxic damage. Additionally, Zhao et al.35 found that mouse liver-derived mitochondria could integrate into hepatocytes through co-incubation in vitro, leading to reduced ROS levels and a bolstered energy supply. Whereas in vivo MT enhanced hepatocytes‘ stress resistance associated with cell survival, significantly treated liver injury induced by carbon tetrachloride (CCl4), increased energy supply to hepatocytes, and attenuated OS, which improved liver function and prevented fibrosis of liver tissue. Interestingly, hepatic IRI was linked to mitochondrial dysfunction in neutrophils.94 In this context, Lu et al.94 discovered that MSC-derived EVs could repair damaged mitochondrial function in neutrophils by transferring mitochondria into the liver, inhibiting the formation of neutrophil extracellular traps and secretion of pro-inflammatory cytokines, thus ameliorating hepatic IRI.
Together, these studies highlight the multifaceted efficacy of MT in the treatment of liver injury. By enhancing cellular energy production, reducing oxidative damage, and modulating inflammatory pathways, MT becomes a noteworthy strategy of interest in the field of mitigation and repair of liver injury.
Cardiovascular disease and heart transplantation
Internalisation of transplanted mitochondria by cardiomyocytes
Transplanted mitochondria, often positioned near cardiomyocytes, seamlessly integrate into these cells in single strands or clusters within 1–4 hours, facilitated by actin-dependent endocytosis. This process aids in the repair of damaged mitochondrial function and mtDNA.37, 38, 71 Current findings suggest that transplanted mitochondria can be internalised by various cardiac cells, including cardiomyocytes and fibroblasts, primarily through an actin-dependent endocytosis mechanism.39 Masuzawa et al.37 noted that cardiomyocytes internalise transplanted mitochondria within 2 hours post-transplantation. These mitochondria retain viability for at least 24 hours, markedly enhancing the oxygen consumption rate and energy production. Moreover, it has been observed that the myocardial absorption rate of mitochondria stands at an average of 76.18 ± 11.85%.95 Sun et al.96 highlighted the potential role of nicotinamide adenine dinucleotide-CD38-cyclic ADP-ribose-Ca2+ signalling in the facilitation of mitochondrial internalisation. It is crucial to note, however, that the necessity of mitochondrial internalisation for cardiac perfusion remains controversial. As long as mitochondria are viable and can sustain their respiratory function, cardiac contractile function will be maintained and myocardial infarct size will be reduced, as cardioprotection occurs rapidly within 10 minutes of reperfusion.97, 98
Cardiac ischaemia/reperfusion injury
MT can significantly modulate cardiac function, promote cardiomyocyte survival, and reduce apoptosis by increasing energy synthesis, promoting damaged mtDNA replacement, and upregulating cytokines.38 In this regard, MT has been widely used in treating heart failure, IRI, and during cardiac transplantation procedures. The cardiac muscle is notable for its substantial daily energy demand, with a healthy heart consuming between 6–30 kg of ATP daily. Mitochondria, accounting for 30% of the volume within cardiomyocytes, fulfill these high energy demands through OXPHOS.99, 100 However, mitochondrial dysfunction in cardiomyocytes occurs as a result of increased ROS production, Ca2+ overload, mitochondrial permeability, and altered mitochondrial dynamics, drastically altering oxygen consumption rate and energy synthesis, ultimately causing cardiomyocyte apoptosis.40, 101 While heart transplantation remains the definitive treatment modality for severe heart failure, the success of transplants is frequently compromised by IRI.102 Interestingly, mitochondria isolated from a variety of sources, including cardiac tissue, arterial blood, and induced pluripotent stem cell-derived cardiomyocytes, have demonstrated a conserved capacity to afford cardioprotection.103 Ali Pour et al.104 revealed that non-autologous MT resulted in a short-term enhancement of bioenergetics, evidenced by improved basal cellular respiration and ATP generation in healthy cardiomyocytes. After a few hours, cardiomyocytes readily internalise transplanted mitochondria, which remain functional and viable, increasing ATP levels and upregulating the cytokines associated with angiogenesis, arteriogenesis, and immunomodulation (e.g., epidermal growth factor, growth-related oncogene, IL-6, monocyte chemoattractant protein 3), thereby preventing apoptosis and enhancing functional recovery post-ischaemia, which in turn provide cardiac protection against IRI.37, 105 Moreover, Blitzer et al.71 explored the efficacy of transplanted mitochondria derived from autologous pectoralis major muscle into a porcine model 2 hours post-IRI and coronary blood flow restoration, and then discovered that the transplanted mitochondria not only remained alive in the pig at 28 days post-surgery but also significantly enhanced cardiomyocyte viability and ameliorated post-ischaemic myocardial function.
MT has been highlighted for its substantial impact in minimising the size of myocardial infarction and preserving the heart in readiness for transplantation. For example, MT has been found to preserve the function and viability of adult porcine myocardium, significantly shrink infarct size, and preserve myocardial function as well as cardiac oxygen consumption in donated hearts after circulatory death.106, 107 Additionally, MT has also facilitated the rapid recovery of lactate, pH, and glucose levels, while improving cerebral microcirculation and neurological functions. These effects have contributed to better survival rates and promoted neurological recovery following resuscitation from cardiac arrest in animal models.108 In another revealing study, Shin et al.109 transplanted mitochondria through coronary arteries and found that MT improved myocardial function, increased coronary blood flow, and reduced size of infarction, thereby highlighting its safety and efficacy of MT. Also of note is the potential of MT in addressing right heart failure. It maintains cardiac contractility by reducing cardiomyocyte apoptosis and fibrosis.40
Pulmonary hypertension
Mitochondrial dysfunction plays a pivotal role in the onset and progression of pulmonary hypertension (PH). Research has indicated that MT holds promising therapeutic potential for PH by bolstering ATP production, mitigating pulmonary vasoconstriction, and dampening inflammatory responses. One illustrative study by Hsu et al.79 discovered that MT significantly enhanced ATP levels in lung tissue, which not only bolstered right ventricular performance but also remodeled pulmonary arterial function. Specifically, it was shown to restore pulmonary artery constriction and pulmonary vascular reactivity, presenting a viable treatment avenue for combating PH. Furthermore, Zhu et al.36 explored the benefits of transplanting mitochondria extracted from pulmonary artery smooth muscles. Their findings indicated that this approach could forestall PH development by inhibiting hypoxia-induced depolarisation of the cell membrane potential, preventing acute hypoxia-induced pulmonary vasoconstriction, and reducing chronic hypoxia-induced pulmonary vascular remodelling. Moreover, they highlighted that exogenously administered mitochondria, when injected intravenously, were capable of reaching the pulmonary artery within 2–24 hours and reached stability by 30 hours. This points towards the rapid integration and functional contribution of transplanted mitochondria, underscoring their potential utility in clinical interventions aimed at treating PH.36
Heart transplantation
Several studies have posited that MT enhances the heart transplantation success rate by improving damaged mitochondrial function and prolonging heart preservation (by increasing cardiomyocyte viability). For example, Moskowitzova et al.64 demonstrated that MT could extend cardiac cold ischaemia time to as much as 29 hours in murine models. This extension is pivotal for enhancing cardiac graft function and mitigating tissue damage incurred during cardiac transplantation, ultimately leading to a reduction in the failure rate of cardiac grafts. Complementing this, Lin et al.110 incubated donor hearts with human platelet-derived mitochondria and discovered that the latter could improve donor’s mitochondrial activity, increase critical enzymes like ATPase, and citrate synthase. Such actions significantly increased cardiomyocyte viability, thereby prolonging preservation time and functional integrity of donor hearts. Post-cardiac transplantation issues, such as immune rejection and delayed graft function, often manifest and are typically linked to programmed cell death processes, including apoptosis and necrosis. Mitochondria released during cellular injury can elicit an endogenous inflammatory response, significantly increasing allogeneic cardiac transplantation rejection. Furthermore, impaired mitochondrial permeability has been associated with the necrotic apoptosis of cardiac endothelial cells. Alternatively, exogenous MT has been found to considerably activate endothelial cells and upregulate the expression of molecules like major histocompatibility complex class II, vascular cell adhesion molecule, and intercellular adhesion molecule.111, 112 Significantly, some studies have showed that allogenic MT did not appear to produce adverse immune reactions between the donated mitochondria and the recipient, signifying a safe and promising path forward in cardiac transplantation therapies.58, 113
Mitochondrial transplantation in combination with other strategies for the treatment of heart disease
While MT holds promise as a therapy for heart disease, it faces certain challenges, including the high extracellular Ca2+ environment’s potential interference with its effectiveness and its inconsistent ability to produce sufficient ATP for sustaining cardiomyocyte contractility.77 These limitations have catalyzed interest in exploring combination therapies that leverage MT alongside other therapeutic avenues to address heart diseases more effectively. Multiple studies have reported that the combined therapeutic approach can ameliorate myocardial infarction and myocardial IRI by modulating autophagy, lowering inflammatory responses, and enhancing mitochondrial function and MMP. For instance, Mokhtari and Badalzadeh114 employed a combined coenzyme Q10 and mitochondrial therapeutic strategy and discovered that it could attenuate myocardial IRI in aged rats by modulating autophagy and downregulating pro-inflammatory factors (tumour necrosis factor (TNF)-α, IL-1β, and IL-6), illustrating the potential of combination therapies in heart disease treatment. Complementing this, Ikeda et al.77 extracted mitochondria-enriched EV grafts derived from human-induced pluripotent stem cell-derived cardiomyocytes and established that these mitochondria could be transferred and integrated into mouse cardiomyocyte mitochondrial networks, thus recuperating damaged mitochondrial function and notably enhancing cardiac function following myocardial infarction. Sun et al.103 delved into another innovative angle by treating mitochondria with Alda-1, a potent agonist of mitochondrial aldehyde dehydrogenase 2. The treatment enhanced mitochondrial survival and MMP, facilitated the fusion of endogenous and exogenous mitochondria, and enhanced the respiratory-mediated mechanical properties of cardiomyocytes. Additionally, it significantly reduced the areas of infarct focus, thereby proving effective in treating myocardial IRI. These studies collectively underscore the promise of MT combination therapies in heart disease management, by not just addressing the limitations of MT when used in isolation but also by amplifying its therapeutic potential through synergistic effects with other treatments.
Osteoarthritis and muscle damage
Osteoarthritis
Mitochondrial dysfunction in chondrocytes, which includes impaired MMP and mitochondrial respiratory chain enzyme complex activity, is crucially involved in the development of osteoarthritis (OA).115 Multiple studies have reported that MT could act as a moderator of the inflammatory response in OA, diminish ROS production, maintain cellular homeostasis, and modulate cell death pathways. Yu et al.116 discovered that MT, using bone marrow derived MSC (BMSC)-derived mitochondria, could confer therapeutic benefits in knee OA by ameliorating chondrocyte mitochondrial dysfunction and stimulating proliferator-activated receptor gamma coactivator-1α signalling to promote mitochondrial biogenesis, thereby ameliorating pathologic cartilage damage, suppressing inflammation, and inhibiting chondrocyte apoptosis. Augmenting these findings, Guo et al.117 successfully transferred mitochondria from BMSCs in vitro into another group of recipient BMSCs from the same batch and discovered that the recipient BMSCs exhibited enhanced proliferation and migratory abilities, as well as potential for osteogenic differentiation. Moreover, Lee et al.54 found that MT from L6 cells protected and promoted chondrocyte regeneration, reduced inflammatory responses and ameliorated the development of OA. This mitigation occurred through the downregulation of pro-inflammatory factors such as IL-1β, monocyte chemoattractant protein-1, TNF-α, and the catabolic factor MMP-13. Furthermore, MT also improved mitochondrial function in injured chondrocytes, activated mitochondrial autophagy, inhibited ROS activity, and reduced necrotic apoptosis in chondrocytes, contributing to the deceleration of OA progression.54 Collectively, these studies present a compelling argument for the therapeutic value of MT in addressing OA.
Muscle injury
MT has been recognised as a potent intervention to counteract muscle damage and accelerate tissue recovery in the context of traumatic muscle injuries. Damaged mitochondria in traumatic muscle injury could result in cell death and increased ROS production, which not only exacerbate the injury but also hinder the healing process of muscle structure and function.53, 118 Mouse liver-derived MT intervenes can ameliorate muscle damage by inhibiting inflammatory responses, regulating apoptosis, and promoting muscle fibre regeneration. It was observed that muscle cells compromised by injury show a selective affinity for incorporating transplanted mitochondria when compared to their uninjured counterparts. This selectivity underscores the injury-specific targeting capacity of MT, which enables direct action at sites requiring therapeutic intervention.53 Beyond muscle repair, MT has been shown to exert a therapeutic effect on tendinopathy, a common musculoskeletal disorder. It has also been reported that in vitro incorporation of human umbilical cord-derived MSC-derived mitochondria into TNF-α-pretreated tendon cells by centrifugation for 24 hours could inhibit apoptosis in tendon cells, restore both the viability of damaged tendon cells and the functionality of damaged mitochondria, alongside suppressed inflammatory responses. While in vivo transplantation of L6 rat myoblast cell line-derived mitochondria for collagenase-induced tendinopathy in rats modulated the metabolic state of tendinopathy, as well as inhibited the expression of pro-inflammatory cytokines IL-1β and IL-6 as well as NF-κB signalling, inhibited apoptosis, and also recycled collagen components of the extracellular matrix, thereby alleviating tendinopathy.119 In sum, through its capacity to directly target and integrate within damaged cells, MT exerts multifaceted therapeutic effects that culminate not only in the reduction of inflammation and regulation of cell death but also in the promotion of tissue recovery and regeneration, be it in muscle injuries or tendinopathies.
Neurological injuries
Neurodegenerative diseases
Neurodegenerative diseases mainly include amyotrophic lateral sclerosis, Parkinson’s disease, multiple sclerosis, Huntington’s disease, and Alzheimer’s disease (AD).120 Ageing is a major risk factor for most neurodegenerative diseases, which have few or no effective treatments.121 Mitochondrial dysfunction in several tissues, which leads to chronic OS and metabolic abnormalities, is a key feature of ageing.122, 123 MT has emerged as a novel therapeutic strategy, offering a beacon of hope. The strategy has shown promise in regulating cellular senescence, which is also a hallmark of ageing and a fundamental process contributing to the pathogenesis of neurodegenerative diseases. For example, Noh et al.124 provided insights into how MT could regulates cellular senescence, attenuates mitochondrial dysfunction and hallmarks of cellular senescence, and also mitigates inflammatory responses by inhibiting NF-κB activity and inflammatory cytokine expression. Javani et al.47 further demonstrated MT in young rats reduced the chronic stress-induced mitochondrial dysfunction in aged rats, decreased neuronal apoptosis in the prefrontal cortex, and increased cell viability in tissues.
Multiple studies have linked mitochondrial dysfunction to the development of neurodegenerative diseases.125 For example, the decrease in MMP and ATP levels that occurs after mitochondrial dysfunction was found to be a driving factor in the development of cognitive deficits in AD.126 Neuronal degeneration in AD has also been strongly associated with mitochondrial damage at synapses in the hippocampus, a high-energy-consuming brain region in which mitochondria are more susceptible to damage.125 And MT has been reported to improve neurodegenerative diseases and ameliorate cognitive dysfunction by increasing the viability of damaged mitochondria and ATP production, decreasing neuronal apoptosis, and protecting neurons from iron death. For example, Nitzan et al.127 discovered that Hela cell-derived mitochondria could effectively ameliorate mitochondrial dysfunction in rats models of AD, improving their cognitive functions by increasing key mitochondrial enzymes like citrate-synthase, cytochrome c oxidase, and enhancing mitochondrial activities, while reducing hippocampal neuronal loss and gliosis. Furthermore, transplanting mitochondria obtained from young mice into the hippocampus of aged mice not only restored mitochondrial function in the latter’s hippocampus, which entails increased ATP levels and enhanced activity of mitochondrial complexes I, II, and IV, but also countered cognitive decline and promoted anti-ageing effects through mechanisms such as the enhancement of Wnt signalling and stimulation of neural progenitor cell proliferation and neurogenesis.128 This is particularly relevant as the dysregulation of the Wnt pathway has been implicated in the development of AD.129 Moreover, Mishra et al.130 and Chen et al.131 have highlighted MT’s neuroprotective role, demonstrating its efficacy in inhibiting neurotoxicity, maintaining neurite extension, and protecting hippocampal neurons from ferroptosis by modulating ROS formation and metabolic activity in a dose-dependent manner.
In summary, MT represents a frontier in regenerative medicine with the potential to address the significant challenge of neurodegenerative diseases. By targeting mitochondrial dysfunction, MT offers a way to not only ameliorate the symptoms but potentially halt or reverse the progression of neurodegenerative diseases, opening up new possibilities for the treatment of these complex disorders.
Cognitive and mood disorders
Neuroinflammation is a defining feature of various neurodegenerative diseases.132 Likewise, mitochondrial dysfunction has a substantial correlation with cognitive impairment.133 Of note, approximately 75% of cancer patients who receive chemotherapy also develop cognitive impairment.134 Therefore, countering this impairment represents a crucial area for therapeutic interventions, and MT has surfaced as a promising candidate. For instance, studies have showed that MT can attenuate brain dysfunction and improve behavioural and cognitive deficits in a sepsis model in rats through the promotion of M1-type microglia polarisation to M2-type, which is associated with brain repair and recovery, as well as the suppression of inflammatory factor release.135 A fascinating aspect of MT is its migratory ability. For instance, nasal transplantation of MSC-derived mitochondria demonstrated their remarkable ability to migrate to the hippocampus and other brain regions. Interestingly, these transplanted mitochondria were promptly absorbed by meningeal cells, especially macrophages. These transplanted mitochondria contributed to metabolic reprogramming to an anti-inflammatory reparative phenotype that promotes recovery of brain function and structure. In addition, nasal-delivered mitochondria can act directly on neurons and glial cells, contributing to the repair of damaged cells and the restoration of synaptic integrity and membrane function within the hippocampus by altering their metabolic programming, ultimately rebalancing oxidative-antioxidant levels and leading to a reversal of cognitive impairment.134 In turn, platelet-derived mitochondria can ameliorate diabetes-induced cognitive deficits by increasing mitochondrial number, restoring mitochondrial function, suppressing OS and neuronal apoptosis, decreasing amyloid-β and tau accumulation in the hippocampus.136, 137 Moreover, MT’s potential extends to psychiatric disorders, such as schizophrenia. Transplantation of mitochondria to schizophrenic lymphoblastoid cells improved mitochondrial functions such as cellular oxygen consumption and MMP and attention deficits in rats.138
MT also shows promise as a therapeutic intervention for mood disorders such as depression. By targeting the physiological underpinnings associated with depression, MT holds potential for alleviating symptoms through a multi-faceted approach. The anti-inflammatory effects of MT are evident in its ability to suppress the activation of astrocytes and microglia, which are significant in neuroinflammation.139 Wang et al.139 found that MT significantly downregulated the expression of neuroinflammatory factors IL-1β, TNF-α, and cyclooxygenase. The transplantation not only quieted the inflammatory response but also promoted the expression of brain-derived neurotrophic factor, crucial for neurogenesis and neuronal survival. Further, it was seen to increase ATP production, enhance OS defenses, and mitigate depression-like behaviours prompted by lipopolysaccharides, a stimulant of the immune response. Supporting the potential of MT for treating mood disorders, Javani et al.140 demonstrated that this approach increased several key markers in the prefrontal cortex of aged rats: MMP, ATP levels, and indoleamine 2,3-dioxygenase levels. They also observed increased dendritic length and spine density, correlating with improvements in anxiety- and depression-like behaviours.
These examples highlight the role of mitochondrial function in mood regulation and the potential of MT in treating disorders like depression. By enhancing mitochondrial function and MMP, as well as inhibiting inflammatory response, MT offers an innovative approach to mood disorder therapies, dovetailing with the expanding understanding of the link between mitochondrial dysfunction and mood disorders.133
Central nervous system injury
Brain injury: Mitochondria play an important role in the central nervous system, especially in generating ATP necessary for neuronal electrical activity.141 It was found that mitochondria can be taken up by various cells in the brain within 24 and 48 hours of MT, including endothelial cells, brain macrophages, pericytes, and oligodendrocytes. Notably, macrophages and pericytes exhibit a higher uptake rate.142 In addition to transplantation, in vitro investigations have demonstrated the uptake of exogenous mitochondria by different brain cell types, such as neurons, astrocytes, and microglia.133 This finding suggests a promising avenue for therapeutic exploration in brain injury management. Notably, MT has exhibited remarkable efficacy in mitigating both traumatic brain injury and IRI, resulting in enhanced outcomes. The potential mechanisms underlying these favorable effects encompass the transfer of functional mitochondrial components, modulation of cellular metabolism, and mitigation of ROS and apoptosis.143 A previous study found that MT using human umbilical cord-derived MSC-derived mitochondria improved mitochondrial function, reduced neuronal apoptosis, and attenuated astrocytosis, as well as microglia activation, which enhanced motor function in traumatic brain injury rats.144 Astrocyte mitochondria can enter the injured cortical neurons and normalise neuronal dendrites and ATP levels after ischaemic stroke.48 Lee et al.145 found that MT enhanced the viability and number of neurons in ischaemic stroke, and reduced the size of the cerebral infarct. Elsewhere, Tseng et al.146 demonstrated that MT improved neuronal damage and functional recovery after ischaemic stroke. Pourmohammadi-Bejarpasi et al.147 also found that MSC-derived MT suppressed neuronal cell apoptosis, inhibited microglia activation, and reduced the infarcted area of the brain, which attenuated acute cerebral ischaemia damage and improved motor function. Moreover, Zhao et al.133 reported that MT not only reduced neuronal apoptosis but also upregulated brain-derived neurotrophic factor expression in astrocytes following traumatic brain injury. Moreover, this intervention enhanced spatial memory and mitigated anxiety symptoms, indicating a multifaceted therapeutic impact on brain injury recovery processes.
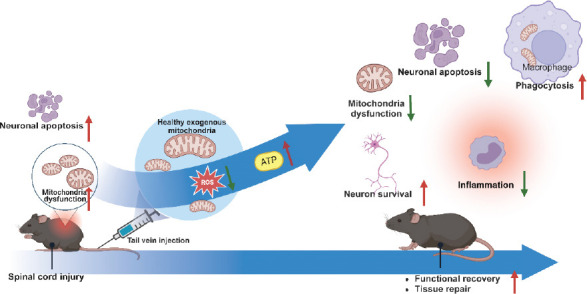
Spinal cord injury: Mitochondrial dysfunction is one of the first step in the development of neuronal damage following spinal cord defect injury.148 In response to such dysfunction, MT has been explored as a novel therapeutic intervention capable of suppressing inflammatory response, OS, and neuronal apoptosis, as well as increase the number of neurons and ATP production to maintain cellular energy production in the injured spinal cord, thereby promoting tissue repair and improving transport function (Figure 4). For example, in a spinal cord ischaemic injury model, Fang et al.45 found that MT could significantly prevent the expression of pro-inflammatory cytokines such as IL-6 and TNF-α in the injured spinal cord. The research also indicated that MT suppressed OS within the spinal endoplasmic reticulum, and inhibited apoptosis and chromatographic catabolism of neurons in the spinal cord, thereby attenuating neurological injury and improving motor function. Research by Huang et al. takes this concept further by utilizing mitochondria derived from M2-type macrophages/microglia to modulate the injury response. In their model, MT not only inhibited polarisation of macrophage towards the pro-inflammatory M1 phenotype but also promoted localized neovascularisation, thereby fostering spinal cord repair and enhancing motor function recovery in mice.149 Finally, it was concluded that mitochondrial dysfunction in M1-type macrophages also generates large amounts of OS injury caused by oxygen free radicals, whereas M2-type macrophages exhibit mitochondrial function.150 Studies have demonstrated that MT combined with other therapeutic strategies can enhance the effectiveness of mitochondria in the treatment of spinal cord injury. Zhu et al.151 combined photobiomodulation (also known as low-level laser therapy) with MT and found that it increased ATP production, reduced OS, and neuronal apoptosis after spinal cord injury, thereby promoting tissue repair and motor function recovery. Moreover, this combination therapy promoted mitochondrial transfer to neurons, with Connex36 playing a mediating role. However, MT seems to lack long-term neuroprotective effects.142 For instance, due to the complex and sensitive structure of the spinal cord, how to deliver mitochondria efficiently and accurately to the site of injury is a key challenge. Moreover, the survival of transplanted mitochondria in host spinal cord tissues, and the ability to integrate effectively into the injured cells are unresolved issues at this stage.
Figure 4. Role of exogenous mitochondrial transplantation in the treatment of spinal cord injury. Created with BioRender.com. ATP: adenosine triphosphate; ROS: reactive oxygen species.
Optic nerve injury: MT is emerging as a promising treatment modality, offering potential therapeutic benefits for optic nerve damage, as well as retinal and macular degeneration. Given that cells in the retina are rich in mitochondria, mitochondrial dysfunction plays a significant role in the pathogenesis of retinal degenerative diseases. Nascimento et al.44 found that MT could improved oxidative metabolism and electrophysiological activity in the retina, thereby enhancing neuronal cell survival in the early stages of optic nerve injury and increase the number of optic nerve axons. Remarkably, transplanted mitochondria remained detectable 28 days post-transplantation. This discovery indicates the potential for long-lasting benefits of MT in treating optic nerve injury. Furthermore, Wu et al.152 found that intravitreal MT could attenuate the progression of retinal degeneration. This process involved the upregulation of growth factor secretion by glial cells and the restoration of functionality in damaged mitochondria within rat models. Additionally, Noh et al.124 provided evidence that MT could exert beneficial effects against age-related macular degeneration by enhancing the function and activity of mitochondria in the retinal pigment epithelium and attenuating cellular signs of ageing. To sum up, these studies indicate that MT holds promise as a treatment strategy for a range of retinal conditions characterised by mitochondrial dysfunction. By improving mitochondrial health in retinal cells, MT not only supports the preservation of these cells but also contributes to the reversal of degenerative processes. This innovative approach could therefore represent a major advancement in the treatment of optic nerve injuries, retinal degeneration, and macular degeneration, offering hope to patients with these challenging conditions.
Peripheral nerve injury: Several studies have documented that MT could promote peripheral nerve repair, chiefly through the attenuation of OS and the reduction in apoptosis of nerve cells. For example, Kuo et al.50 found the protective role of MT against axonal degeneration. Here, incubating mitochondria with nerve explants in vitro was shown to attenuate cytoskeletal loss and OS, thereby contributing to ameliorate the axonal degeneration in injured nerves. In vivo MT promotes neuromuscular reinnervation by enhancing the repair of nerve conduction function after sciatic nerve crush injury, increasing the expression of neurotrophic factors in injured nerves and denervated muscles.50 Further demonstrating the therapeutic potential of MT, Bai et al.51 explored a novel approach for repairing nerve damage by using mitochondria derived from human umbilical cord-derived MSCs. They transplanted these mitochondria into decellularised nerve segments in rat models with sciatic nerve defects. The results were promising: the transplanted segments showed considerable regeneration of axons and vascularisation, leading to notable motor function improvements in the treated animals. Interestingly, the study also revealed that Schwann cells, key players in nerve repair, readily internalised the transplanted mitochondria, which in turn stimulated their proliferation. This suggests that the therapeutic effect might be mediated, at least in part, by this interaction between mitochondria and Schwann cells. Zhai et al.52 reported that transplantation of mitochondria derived from adipose-derived MSCs effectively treated bilateral cavernous nerve injury in rats. This type of MT significantly reduced OS and apoptosis in cavernous smooth muscle cells, increased their number and modulated energy metabolism. This study additionally highlighted MT’s role in strengthening the anti-oxidative defenses of CCSMC in vitro.52 Incidentally, Yao et al.33 explored the outcomes of transplanting exogenous mitochondria into adipose-derived MSCs. The results showed enhanced energy levels within the adipose-derived MSCs and promoted their proliferation, migration, and survival, as well as their secretion of biologically active factors. Crucially, this escalation in performance did not come at the cost of their stemness — a significant consideration for maintaining the cells’ regenerative capabilities.33 This range of restorative effects positions MT as an evolving and promising dimension in the treatment of peripheral neural tissue damage.
Other diseases
Sepsis
Sepsis is a life-threatening organ dysfunction that is caused by dysregulated host response to infection, with an extremely high morbidity and mortality rate.153 Amidst this urgent medical challenge, MT has arisen as a promising therapeutic strategy aimed at rescuing damaged mitochondria and enhancing patient outcomes in sepsis. Early intervention with MT has been shown to offer substantial benefits. Hwang et al. demonstrated that MT, administered 24 hours after after the onset of sepsis, enhanced respiratory function and increased ATP content in muscle and spleen tissues.41 This vital enhancement in cellular energy was accompanied by elevated bacterial clearance from the spleen and blood, reduced apoptosis of spleen cells, and an attenuated hyperinflammatory response during the acute phase of sepsis hyperinflammation by downregulating TNF-α expression in spleen tissue.41 MT‘s advantages extend well into the advanced stages of sepsis, proffering multiple benefits such as heightened OXPHOS in immune cells. This bolstering of OXPHOS is crucial in promoting the polarisation of macrophages from M1 type to M2 type, a transition that enhances overall immunosuppression and ultimately leads to better patient survival rates. In a comparison study conducted by Kim et al.154 mitochondria extracted from muscle cells, hepatocytes, and MSCs were assessed for their ability to fortify mitochondrial function and regulate inflammatory responses. Although all three sources improved mitochondrial function and dampened the inflammatory response in the acute phase by reducing TNF-α expression, while also enhancing immune function in the late phase, mitochondria derived from muscle cells demonstrated the most pronounced effect on survival, suggesting their potential as a preferred mitochondrial donor type. In addition, MT has shown potential to treat sepsis-induced myocardial dysfunction. Mokhtari et al.42] reported that MT improved the function and kinetics of damaged mitochondria, downregulated the expression of mitochondrial fission-related genes, and inhibited the production of inflammatory cytokines TNF-α and IL-1β, thereby improving myocardial dysfunction in a rat model of sepsis induced by cecum ligation and puncture. Importantly, the study noted that multiple MT offered better therapeutic efficacy compared to single transplantation.42 By tackling both the inflammatory and immune dysfunctions characteristic of this complex disease, MT not only improves organ function but also raises crucial promise for improving survival rates among sepsis patients.
Ultraviolet radiation damage and wound healing
Ultraviolet (UV) radiation can alter mitochondrial function and morphology. It has been demonstrated that it alters mitochondrial biogenesis, mitochondrial energy metabolism, mitochondrial fusion and fission, and mitochondrial autophagy, promoting ageing, immunosuppression, and tissue stress,155 underscoring the need for effective interventions to mitigate these detrimental effects. In this context, MT has emerged as a promising therapeutic strategy for counteracting the damage inflicted by UV radiation. By reducing apoptosis and enhancing metabolic activity within the damaged cells, MT offers a ray of hope. A study by Hu et al.156 is crucial in illustrating this potential, revealing that MT could partially reverse the UV irradiation-induced S-phase cell cycle block and the decrease of MMP, and reduce apoptosis. This indicates MT‘s capability to restore cellular function and viability following UV exposure. Evidence from another study further indicated that transplantated mitochondria into peripheral blood mononuclear cells affected by UV irradiation could improved their metabolic activity while reducing apoptosis.157 Beyond its capacity to repair UV-induced mitochondrial damage, MT also plays a crucial role in the promotion of wound healing by inhibiting inflammation and reducing the levels of ROS which delay wound healing.158 A study conducted by Kim et al.159 found that found that platelet-derived mitochondria, when transplantated into human dermal fibroblasts, were successfully internalised by the cells, leading to enhanced fibroblast proliferation and reduced ROS and hydrogen peroxide production, in turn promoted wound healing.
Prospects for Clinical Applications
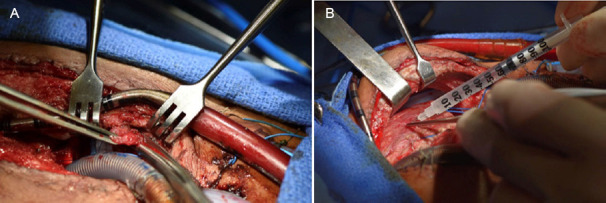
In recent years, MT has attracted significant attention from researchers, especially in the treatment of cardiac IRI. For example, Guariento et al.160 used autologous MT to treat pediatric patients with severe postischaemic reperfusion cardiac shock who required cardiotomy and extracorporeal membrane oxygenation. They found that MT reduced ventricular strain after extracorporeal membrane oxygenation isolation as well as shortened postoperative revascularisation time.160 Emani et al.161 introduced a promising strategy to address myocardial IRI by directly transplanting autologous mitochondria into the heart muscle via epicardial injection. This innovative approach offers several benefits: Firstly, it alleviates adverse immune reactions or inflammation associated with foreign-derived mitochondria. Secondly, it presents minimal short-term complications, enhancing safety for patients. Importantly, the study noted considerable enhancements in ventricular function post-procedure, providing optimism for a potential treatment avenue for this debilitating condition. Figure 5 shows the process of transplanting autologous mitochondria to treat pediatric patients with severe postischaemic reperfusion cardiac shock who required extracorporeal membrane oxygenation.161 However, numerous improvements are needed to accelerate the transfer of MT therapy into clinical application. Firstly, the techniques for mitochondrial extraction, preservation, and transplantation are yet to reach full maturity, and precision in these processes is paramount to ensure that the function of mitochondria remains unaffected during transplantation. Moreover, the survival and stable function of transplanted mitochondria within the host body, and whether they will cause immune reactions or undesirable interactions with host cellular systems need to be rigorously considered. For instance, injected mitochondria are considered to be less able to tolerate high Ca2+ concentrations, among other things.162 Additionally, strict quality control measures are necessary to assess the viability and function of mitochondria before transplantation to ensure their capacity to restore cellular energy metabolism effectively.
Figure 5. Mitochondrial transplantation for paediatric patients who required central extracorporeal membrane oxygenation support for ischaemia/reperfusion associated myocardial dysfunction after cardiac surgical procedure. (A) Biopsy of non-ischaemic skeletal muscle for mitochondrial extraction. (B) Injection of autologous mitochondria into the myocardium using an insulin syringe. Reprinted from Emani et al.161 Copyright 2017 by The American Association for Thoracic Surgery.
Conclusion
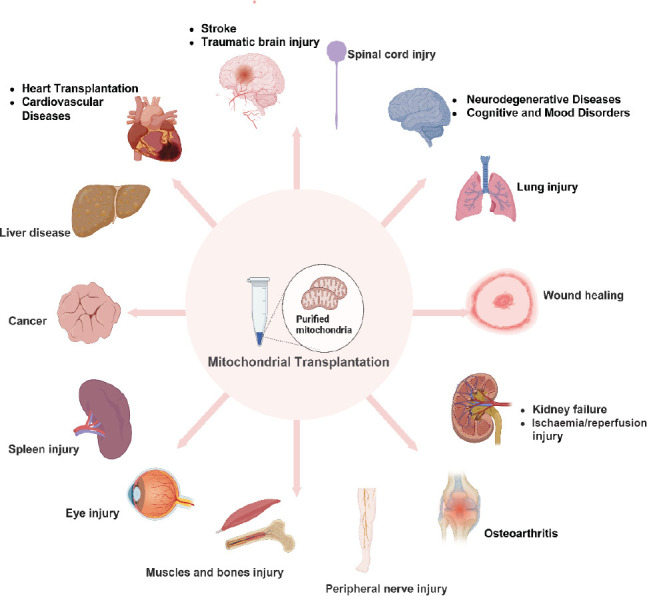
This review on studies performed in the last 5 years shows that mitochondrial dysfunction contributes to occurrence of several diseases, and MT has been widely applied to treat various diseases with promising results. However, our review did not provide an in-depth summary of the mechanisms of MT for the treatment of different diseases, and it is possible that some relevant literature was not searched, making the review incomplete. Despite the many challenges above, MT exhibits substantial clinical potential, which has shown unprecedented therapeutic potential in a number of areas, ranging from treating cardiac and metabolic diseases, neurodegenerative disorders, to organ transplantation (Figure 6). Therefore, MT has the potential to be a new hope for these intractable diseases, and this innovative technique is expected to enhance cellular energy metabolism, repair damaged tissues, and modulate the host immune response, thereby exerting therapeutic effects. In addition, mitochondria lack many surface antigens, such as human leukocyte antigen class 1 class 1 antigens, and therefore exhibit lower immunogenicity than MSCs, which may be an advantage of mitochondria over MSC in favour of clinical translation of allogeneic transplantation.55 The majority of current studies have primarily focused on assessing the effectiveness of MT in disease treatment, while investigations into the associated mechanisms have been limited. Considering the enormous promise that MT demonstrates in both theoretical and experimental research, future studies will inevitably concentrate on overcoming these hurdles and deepening our understanding of MT‘s therapeutic mechanisms, thereby advancing MT technology as a viable and effective treatment modality that can offer significant benefits to patients with a variety of diseases.
Figure 6. Prospects for clinical applications of mitochondrial transplantation. Created with BioRender.com.
Acknowledgement.
We would like to thank MJEditor for providing English editing services during the preparation of this manuscript.
Footnotes
Author contributions: JP performed most of the overall work for this review. YW contributed to the writing framework of the review as co-corresponding authors. XL wrote much of this manuscript. YG, CL and JB contributed to collect literature of the application of MT in different diseases. HC and JZ contributed to edit and correct the review. All authors read and approved the final manuscript.
Financial support: This work was supported by the National Natural Science Foundation of China (No. 32171356).
Conflicts of interest statement: The authors declare no conflict of interest.
References
- 1.Nakamura Y., Park J. H., Hayakawa K. Therapeutic use of extracellular mitochondria in CNS injury and disease. Exp Neurol. 2020;324:113114. doi: 10.1016/j.expneurol.2019.113114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gollihue J. L., Rabchevsky A. G. Prospects for therapeutic mitochondrial transplantation. Mitochondrion. 2017;35:70–79. doi: 10.1016/j.mito.2017.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liao P. C., Tandarich L. C., Hollenbeck P. J. ROS regulation of axonal mitochondrial transport is mediated by Ca2+ and JNK in Drosophila. PLoS One. 2017;12:e0178105. doi: 10.1371/journal.pone.0178105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Llorente-Folch I., Rueda C. B., Pardo B., Szabadkai G., Duchen M. R., Satrustegui J. The regulation of neuronal mitochondrial metabolism by calcium. J Physiol. 2015;593:3447–3462. doi: 10.1113/JP270254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pernas L., Scorrano L. Mito-morphosis:mitochondrial fusion, fission, and cristae remodeling as key mediators of cellular function. Annu Rev Physiol. 2016;78:505–531. doi: 10.1146/annurev-physiol-021115-105011. [DOI] [PubMed] [Google Scholar]
- 6.Archer S. L. Mitochondrial dynamics--mitochondrial fission and fusion in human diseases. N Engl J Med. 2013;369:2236–2251. doi: 10.1056/NEJMra1215233. [DOI] [PubMed] [Google Scholar]
- 7.Chan D. C. Mitochondrial dynamics and its involvement in disease. Annu Rev Pathol. 2020;15:235–259. doi: 10.1146/annurev-pathmechdis-012419-032711. [DOI] [PubMed] [Google Scholar]
- 8.Cheng X. T., Huang N., Sheng Z. H. Programming axonal mitochondrial maintenance and bioenergetics in neurodegeneration and regeneration. Neuron. 2022;110:1899–1923. doi: 10.1016/j.neuron.2022.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhao J., Zhang J., Yu M., Xie Y., Huang Y., Wolff D. W., Abel P. W., Tu Y. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene. 2013;32:4814–4824. doi: 10.1038/onc.2012.494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Peiris-Pagès M., Bonuccelli G., Sotgia F., Lisanti M P. Mitochondrial fission as a driver of stemness in tumor cells:mDIVI1 inhibits mitochondrial function, cell migration and cancer stem cell (CSC) signalling. Oncotarget. 2018;9:13254–13275. doi: 10.18632/oncotarget.24285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Han Y., Cho U., Kim S., Park I. S., Cho J. H., Dhanasekaran D. N., Song Y S. Tumour microenvironment on mitochondrial dynamics and chemoresistance in cancer. Free Radic Res. 2018;52:1271–1287. doi: 10.1080/10715762.2018.1459594. [DOI] [PubMed] [Google Scholar]
- 12.Alexander J. F., Seua A. V., Arroyo L. D., Ray P. R., Wangzhou A., Heiβ-Lückemann L., Schedlowski M., Price T. J., Kavelaars A., Heijnen C. J. Nasal administration of mitochondria reverses chemotherapy-induced cognitive deficits. Theranostics. 2021;11:3109–3130. doi: 10.7150/thno.53474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chaturvedi R. K., Flint Beal M. Mitochondrial diseases of the brain. Free Radic Biol Med. 2013;63:1–29. doi: 10.1016/j.freeradbiomed.2013.03.018. [DOI] [PubMed] [Google Scholar]
- 14.López-Lluch G. Mitochondrial activity and dynamics changes regarding metabolism in ageing and obesity. Mech Ageing Dev. 2017;162:108–121. doi: 10.1016/j.mad.2016.12.005. [DOI] [PubMed] [Google Scholar]
- 15.McInnes J. Mitochondrial-associated metabolic disorders:foundations, pathologies and recent progress. Nutr Metab (Lond) 2013;10:63. doi: 10.1186/1743-7075-10-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Seyfried T. N., Flores R. E., Poff A. M., D'Agostino D. P. Cancer as a metabolic disease:implications for novel therapeutics. Carcinogenesis. 2014;35:515–527. doi: 10.1093/carcin/bgt480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Johri A., Beal M. F. Mitochondrial dysfunction in neurodegenerative diseases. J Pharmacol Exp Ther. 2012;342:619–630. doi: 10.1124/jpet.112.192138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guaragnella N., Palermo V., Galli A., Moro L., Mazzoni C., Giannattasio S. The expanding role of yeast in cancer research and diagnosis:insights into the function of the oncosuppressors p53 and BRCA1/2. FEMS Yeast Res. 2014;14:2–16. doi: 10.1111/1567-1364.12094. [DOI] [PubMed] [Google Scholar]
- 19.Tait S. W., Green D. R. Mitochondria and cell death:outer membrane permeabilization and beyond. Nat Rev Mol Cell Biol. 2010;11:621–632. doi: 10.1038/nrm2952. [DOI] [PubMed] [Google Scholar]
- 20.Wang C., Youle R J. The role of mitochondria in apoptosis*. Annu Rev Genet. 2009;43:95–118. doi: 10.1146/annurev-genet-102108-134850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oberst A., Bender C., Green D. R. Living with death:the evolution of the mitochondrial pathway of apoptosis in animals. Cell Death Differ. 2008;15:1139–1146. doi: 10.1038/cdd.2008.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kaur M. M., Sharma D S. Mitochondrial repair as potential pharmacological target in cerebral ischemia. Mitochondrion. 2022;63:23–31. doi: 10.1016/j.mito.2022.01.001. [DOI] [PubMed] [Google Scholar]
- 23.Hayakawa K., Esposito E., Wang X., Terasaki Y., Liu Y., Xing C., Ji X., Lo E. H. Transfer of mitochondria from astrocytes to neurons after stroke. Nature. 2016;535:551–555. doi: 10.1038/nature18928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tan Y. L., Eng S. P., Hafez P., Abdul Karim N., Law J. X., Ng M. H. Mesenchymal stromal cell mitochondrial transfer as a cell rescue strategy in regenerative medicine:a review of evidence in preclinical models. Stem Cells Transl Med. 2022;11:814–827. doi: 10.1093/stcltm/szac044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hernández-Cruz E. Y., Amador-Martínez I., Aranda-Rivera A. K., Cruz-Gregorio A., Pedraza Chaverri J. Renal damage induced by cadmium and its possible therapy by mitochondrial transplantation. Chem Biol Interact. 2022;361:109961. doi: 10.1016/j.cbi.2022.109961. [DOI] [PubMed] [Google Scholar]
- 26.Seydi E., Rahemi M., Esmaily H., Arjmand A., Pourahmad J. Mitochondrial transplantation attenuates toxicity in rat renal proximal tubular cells caused by Favipiravir. J Pharm Pharmacol. 2023;75:1458–1466. doi: 10.1093/jpp/rgad079. [DOI] [PubMed] [Google Scholar]
- 27.Preble J. M., Pacak C. A., Kondo H., MacKay A. A., Cowan D. B., McCully J. D. Rapid isolation and purification of mitochondria for transplantation by tissue dissociation and differential filtration. J Vis Exp. 2014:e51682. doi: 10.3791/51682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rossi A., Asthana A., Riganti C., Sedrakyan S., Byers L. N., Robertson J., Senger R. S., Montali F., Grange C., Dalmasso A., Porporato P. E., Palles C., Thornton M. E., Da Sacco S., Perin L., Ahn B., McCully J., Orlando G., Bussolati B. Mitochondria transplantation mitigates damage in an in vitro model of renal tubular injury and in an ex vivo model of DCD renal transplantation. Ann Surg. 2023;278:e1313-e1326. doi: 10.1097/SLA.0000000000006005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jabbari H., Roushandeh A. M., Rostami M. K., Razavi-Toosi M. T., Shokrgozar M. A., Jahanian-Najafabadi A., Kuwahara Y., Roudkenar M. H. Mitochondrial transplantation ameliorates ischemia/reperfusion-induced kidney injury in rat. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165809. doi: 10.1016/j.bbadis.2020.165809. [DOI] [PubMed] [Google Scholar]
- 30.Pang Y. L., Fang S. Y., Cheng T. T., Huang C. C., Lin M. W., Lam C. F., Chen K. B. Viable allogeneic mitochondria transplantation improves gas exchange and alveolar-capillary permeability in rats with endotoxin-induced acute lung injuries. Int J Med Sci. 2022;19:1036–1046. doi: 10.7150/ijms.73151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Moskowitzova K., Orfany A., Liu K., Ramirez-Barbieri G., Thedsanamoorthy J. K., Yao R., Guariento A., Doulamis I. P., Blitzer D., Shin B., Snay E. R., Inkster J. A. H., Iken K., Packard A. B., Cowan D. B., Visner G. A., Del Nido P. J., McCully J. D. Mitochondrial transplantation enhances murine lung viability and recovery after ischemia-reperfusion injury. Am J Physiol Lung Cell Mol Physiol. 2020;318:L78-L88. doi: 10.1152/ajplung.00221.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Morrison T. J., Jackson M. V., Cunningham E. K., Kissenpfennig A., McAuley D. F., O'Kane C. M., Krasnodembskaya A. D. Mesenchymal stromal cells modulate macrophages in clinically relevant lung injury models by extracellular vesicle mitochondrial transfer. Am J Respir Crit Care Med. 2017;196:1275–1286. doi: 10.1164/rccm.201701-0170OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yao X., Ma Y., Zhou W., Liao Y., Jiang Z., Lin J., He Q., Wu H., Wei W., Wang X., Björklund M., Ouyang H. In-cytoplasm mitochondrial transplantation for mesenchymal stem cells engineering and tissue regeneration. Bioeng Transl Med. 2022;7:e10250. doi: 10.1002/btm2.10250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sun C. K., Chang C. L., Lin Y. C., Kao Y. H., Chang L. T., Yen C. H., Shao P. L., Chen C. H., Leu S., Yip H. K. Systemic administration of autologous adipose-derived mesenchymal stem cells alleviates hepatic ischemia-reperfusion injury in rats. Crit Care Med. 2012;40:1279–1290. doi: 10.1097/CCM.0b013e31823dae23. [DOI] [PubMed] [Google Scholar]
- 35.Zhao Z., Hou Y., Zhou W., Keerthiga R., Fu A. Mitochondrial transplantation therapy inhibit carbon tetrachloride-induced liver injury through scavenging free radicals and protecting hepatocytes. Bioeng Transl Med. 2021;6:e10209. doi: 10.1002/btm2.10209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhu L., Zhang J., Zhou J., Lu Y., Huang S., Xiao R., Yu X., Zeng X., Liu B., Liu F., Sun M., Dai M., Hao Q., Li J., Wang T., Li T., Hu Q. Mitochondrial transplantation attenuates hypoxic pulmonary hypertension. Oncotarget. 2016;7:48925–48940. doi: 10.18632/oncotarget.10596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Masuzawa A., Black K. M., Pacak C. A., Ericsson M., Barnett R. J., Drumm C., Seth P., Bloch D. B., Levitsky S., Cowan D. B., McCully J. D. Transplantation of autologously derived mitochondria protects the heart from ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol. 2013;304:H966-982. doi: 10.1152/ajpheart.00883.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kaza A. K., Wamala I., Friehs I., Kuebler J. D., Rathod R. H., Berra I., Ericsson M., Yao R., Thedsanamoorthy J. K., Zurakowski D., Levitsky S., Del Nido P. J., Cowan D. B., McCully J. D. Myocardial rescue with autologous mitochondrial transplantation in a porcine model of ischemia/reperfusion. J Thorac Cardiovasc Surg. 2017;153:934–943. doi: 10.1016/j.jtcvs.2016.10.077. [DOI] [PubMed] [Google Scholar]
- 39.Guariento A., Blitzer D., Doulamis I., Shin B., Moskowitzova K., Orfany A., Ramirez-Barbieri G., Staffa S. J., Zurakowski D., Del Nido P. J., McCully J. D. Preischemic autologous mitochondrial transplantation by intracoronary injection for myocardial protection. J Thorac Cardiovasc Surg. 2020;160:e15-e29. doi: 10.1016/j.jtcvs.2019.06.111. [DOI] [PubMed] [Google Scholar]
- 40.Weixler V., Lapusca R., Grangl G., Guariento A., Saeed M. Y., Cowan D. B., Del Nido P. J., McCully J. D., Friehs I. Autogenous mitochondria transplantation for treatment of right heart failure. J Thorac Cardiovasc Surg. 2021;162:e111-e121. doi: 10.1016/j.jtcvs.2020.08.011. [DOI] [PubMed] [Google Scholar]
- 41.Hwang J. W., Lee M. J., Chung T. N., Lee H. A. R., Lee J. H., Choi S. Y., Park Y. J., Kim C. H., Jin I., Kim S. H., Kwak H. B., Heo J. W., Na K., Choi S., Choi Y. S., Kim K. The immune modulatory effects of mitochondrial transplantation on cecal slurry model in rat. Crit Care. 2021;25:20. doi: 10.1186/s13054-020-03436-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mokhtari B., Hamidi M., Badalzadeh R., Mahmoodpoor A. Mitochondrial transplantation protects against sepsis-induced myocardial dysfunction by modulating mitochondrial biogenesis and fission/fusion and inflammatory response. Mol Biol Rep. 2023;50:2147–2158. doi: 10.1007/s11033-022-08115-4. [DOI] [PubMed] [Google Scholar]
- 43.Zhang Z., Yan C., Miao J., Pu K., Ma H., Wang Q. Muscle-derived mitochondrial transplantation reduces inflammation, enhances bacterial clearance, and improves survival in sepsis. Shock. 2021;56:108–118. doi: 10.1097/SHK.0000000000001681. [DOI] [PubMed] [Google Scholar]
- 44.Nascimento-Dos-Santos G., de-Souza-Ferreira E., Lani R., Faria C. C., Araújo V. G., Teixeira-Pinheiro L. C., Vasconcelos T., Gonçalo T., Santiago M. F., Linden R., Galina A., Petrs-Silva H. Neuroprotection from optic nerve injury and modulation of oxidative metabolism by transplantation of active mitochondria to the retina. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165686. doi: 10.1016/j.bbadis.2020.165686. [DOI] [PubMed] [Google Scholar]
- 45.Fang S. Y., Roan J. N., Lee J. S., Chiu M. H., Lin M. W., Liu C. C., Lam C. F. Transplantation of viable mitochondria attenuates neurologic injury after spinal cord ischemia. J Thorac Cardiovasc Surg. 2021;161:e337-e347. doi: 10.1016/j.jtcvs.2019.10.151. [DOI] [PubMed] [Google Scholar]
- 46.Xu J., Shi C., Yuan F., Ding Y., Xie Y., Liu Y., Zhu F., Lu H., Duan C., Hu J., Jiang L. Targeted transplantation of engineered mitochondrial compound promotes functional recovery after spinal cord injury by enhancing macrophage phagocytosis. Bioact Mater. 2024;32:427–444. doi: 10.1016/j.bioactmat.2023.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Javani G., Ghaffari-Nasab A., Farajdokht F., Mohaddes G. Chronic stress-induced apoptosis is mitigated by young mitochondria transplantation in the prefrontal cortex of aged rats. Iran J Basic Med Sci. 2023;26:725–730. doi: 10.22038/IJBMS.2023.69551.15145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Berridge M. V., Schneider R. T., McConnell M. J. Mitochondrial transfer from astrocytes to neurons following ischemic insult:guilt by association? Cell Metab. 2016;24:376-378. doi: 10.1016/j.cmet.2016.08.023. [DOI] [PubMed] [Google Scholar]
- 49.Nakamura Y., Lo E. H., Hayakawa K. Placental mitochondria therapy for cerebral ischemia-reperfusion injury in mice. Stroke. 2020;51:3142–3146. doi: 10.1161/STROKEAHA.120.030152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kuo C. C., Su H. L., Chang T. L., Chiang C. Y., Sheu M. L., Cheng F. C., Chen C. J., Sheehan J., Pan H. C. Prevention of axonal degeneration by perineurium injection of mitochondria in a sciatic nerve crush injury model. Neurosurgery. 2017;80:475–488. doi: 10.1093/neuros/nyw090. [DOI] [PubMed] [Google Scholar]
- 51.Bai J., Yu B., Li C., Cheng H., Guan Y., Ren Z., Zhang T., Song X., Jia Z., Su T., Tao B., Gao H., Yang B., Liang L., Xiong X., Zhou X., Yin L., Peng J., Shang A., Wang Y. Mesenchymal stem cell-derived mitochondria enhance extracellular matrix-derived grafts for the repair of nerve defect. Adv Healthc Mater. 2024;13:e2302128. doi: 10.1002/adhm.202302128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhai J., Chen Z., Chen P., Yang W., Wei H. Adipose derived mesenchymal stem cells-derived mitochondria transplantation ameliorated erectile dysfunction induced by cavernous nerve injury. World J Mens Health. 2024;42:188–201. doi: 10.5534/wjmh.220233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Alway S. E., Paez H. G., Pitzer C. R., Ferrandi P. J., Khan M. M., Mohamed J. S., Carson J. A., Deschenes M. R. Mitochondria transplant therapy improves regeneration and restoration of injured skeletal muscle. J Cachexia Sarcopenia Muscle. 2023;14:493–507. doi: 10.1002/jcsm.13153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lee A. R., Woo J. S., Lee S. Y., Na H. S., Cho K. H., Lee Y. S., Lee J. S., Kim S. A., Park S. H., Kim S. J., Cho M. L. Mitochondrial transplantation ameliorates the development and progression of osteoarthritis. Immune Netw. 2022;22:e14. doi: 10.4110/in.2022.22.e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Orfany A., Arriola C. G., Doulamis I. P., Guariento A., Ramirez-Barbieri G., Moskowitzova K., Shin B., Blitzer D., Rogers C., Del Nido P. J., McCully J. D. Mitochondrial transplantation ameliorates acute limb ischemia. J Vasc Surg. 2020;71:1014–1026. doi: 10.1016/j.jvs.2019.03.079. [DOI] [PubMed] [Google Scholar]
- 56.Dubinin M. V., Mikheeva I. B., Stepanova A. E., Igoshkina A. D., Cherepanova A. A., Semenova A. A., Sharapov V. A., Kireev II., Belosludtsev K N. Mitochondrial transplantation therapy ameliorates muscular dystrophy in mdx mouse model. Biomolecules. 2024;14:316. doi: 10.3390/biom14030316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Patel S. P., Michael F. M., Arif Khan M., Duggan B., Wyse S., Darby D. R., Chaudhuri K., Pham J. T., Gollihue J., DeRouchey J. E., Sullivan P. G., Dziubla T. D., Rabchevsky A. G. Erodible thermogelling hydrogels for localized mitochondrial transplantation to the spinal cord. Mitochondrion. 2022;64:145–155. doi: 10.1016/j.mito.2022.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ramirez-Barbieri G., Moskowitzova K., Shin B., Blitzer D., Orfany A., Guariento A., Iken K., Friehs I., Zurakowski D., Del Nido P. J., McCully J. D. Alloreactivity and allorecognition of syngeneic and allogeneic mitochondria. Mitochondrion. 2019;46:103–115. doi: 10.1016/j.mito.2018.03.002. [DOI] [PubMed] [Google Scholar]
- 59.Spees J. L., Olson S. D., Whitney M. J., Prockop D. J. Mitochondrial transfer between cells can rescue aerobic respiration. Proc Natl Acad Sci U S A. 2006;103:1283–1288. doi: 10.1073/pnas.0510511103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Li X., Zhang Y., Yeung S. C., Liang Y., Liang X., Ding Y., Ip M. S., Tse H. F., Mak J. C., Lian Q. Mitochondrial transfer of induced pluripotent stem cell-derived mesenchymal stem cells to airway epithelial cells attenuates cigarette smoke-induced damage. Am J Respir Cell Mol Biol. 2014;51:455–465. doi: 10.1165/rcmb.2013-0529OC. [DOI] [PubMed] [Google Scholar]
- 61.Court A. C., Le-Gatt A., Luz-Crawford P., Parra E., Aliaga-Tobar V., Bátiz L. F., Contreras R. A., Ortúzar M. I., Kurte M., Elizondo-Vega R., Maracaja-Coutinho V., Pino-Lagos K., Figueroa F. E., Khoury M. Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response. EMBO Rep. 2020;21:e48052. doi: 10.15252/embr.201948052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Boukelmoune N., Chiu G. S., Kavelaars A., Heijnen C. J. Mitochondrial transfer from mesenchymal stem cells to neural stem cells protects against the neurotoxic effects of cisplatin. Acta Neuropathol Commun. 2018;6:139. doi: 10.1186/s40478-018-0644-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Crewe C., Funcke J. B., Li S., Joffin N., Gliniak C. M., Ghaben A. L., An Y. A., Sadek H. A., Gordillo R., Akgul Y., Chen S., Samovski D., Fischer-Posovszky P., Kusminski C. M., Klein S., Scherer P. E. Extracellular vesicle-based interorgan transport of mitochondria from energetically stressed adipocytes. Cell Metab. 2021;33:1853-1868.e11. doi: 10.1016/j.cmet.2021.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Moskowitzova K., Shin B., Liu K., Ramirez-Barbieri G., Guariento A., Blitzer D., Thedsanamoorthy J. K., Yao R., Snay E. R., Inkster J. A. H., Orfany A., Zurakowski D., Cowan D. B., Packard A. B., Visner G. A., Del Nido P. J., McCully J. D. Mitochondrial transplantation prolongs cold ischemia time in murine heart transplantation. J Heart Lung Transplant. 2019;38:92–99. doi: 10.1016/j.healun.2018.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Park A., Oh M., Lee S. J., Oh K. J., Lee E. W., Lee S. C., Bae K. H., Han B. S., Kim W. K. Mitochondrial transplantation as a novel therapeutic strategy for mitochondrial diseases. Int J Mol Sci. 2021;22:4793. doi: 10.3390/ijms22094793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lieser M. J., Park J., Natori S., Jones B. A., Bronk S. F., Gores G. J. Cholestasis confers resistance to the rat liver mitochondrial permeability transition. Gastroenterology. 1998;115:693–701. doi: 10.1016/s0016-5085(98)70149-0. [DOI] [PubMed] [Google Scholar]
- 67.Divakaruni A. S., Brand M. D. The regulation and physiology of mitochondrial proton leak. Physiology (Bethesda) 2011;26:192–205. doi: 10.1152/physiol.00046.2010. [DOI] [PubMed] [Google Scholar]
- 68.Yu Z., Hou Y., Zhou W., Zhao Z., Liu Z., Fu A. The effect of mitochondrial transplantation therapy from different gender on inhibiting cell proliferation of malignant melanoma. Int J Biol Sci. 2021;17:2021–2033. doi: 10.7150/ijbs.59581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Justo R., Boada J., Frontera M., Oliver J., Bermúdez J., Gianotti M. Gender dimorphism in rat liver mitochondrial oxidative metabolism and biogenesis. Am J Physiol Cell Physiol. 2005;289:C372-378. doi: 10.1152/ajpcell.00035.2005. [DOI] [PubMed] [Google Scholar]
- 70.Hayashida K., Takegawa R., Shoaib M., Aoki T., Choudhary R. C., Kuschner C. E., Nishikimi M., Miyara S. J., Rolston D. M., Guevara S., Kim J., Shinozaki K., Molmenti E. P., Becker L. B. Mitochondrial transplantation therapy for ischemia reperfusion injury:a systematic review of animal and human studies. J Transl Med. 2021;19:214. doi: 10.1186/s12967-021-02878-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Blitzer D., Guariento A., Doulamis I. P., Shin B., Moskowitzova K., Barbieri G. R., Orfany A., Del Nido P. J., McCully J. D. Delayed transplantation of autologous mitochondria for cardioprotection in a porcine model. Ann Thorac Surg. 2020;109:711–719. doi: 10.1016/j.athoracsur.2019.06.075. [DOI] [PubMed] [Google Scholar]
- 72.Koch R. E., Josefson C. C., Hill G. E. Mitochondrial function, ornamentation, and immunocompetence. Biol Rev Camb Philos Soc. 2017;92:1459–1474. doi: 10.1111/brv.12291. [DOI] [PubMed] [Google Scholar]
- 73.Desdín-Micó G., Soto-Heredero G., Aranda J. F., Oller J., Carrasco E., Gabandé-Rodríguez E., Blanco E. M., Alfranca A., Cussó L., Desco M., Ibañez B., Gortazar A. R., Fernández-Marcos P., Navarro M. N., Hernaez B., Alcamí A., Baixauli F., Mittelbrunn M. T cells with dysfunctional mitochondria induce multimorbidity and premature senescence. Science. 2020;368:1371–1376. doi: 10.1126/science.aax0860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yu S. H., Kim S., Kim Y., Lee S. E., Park J. H., Cho G., Ha J. C., Jung H., Lim S. M., Han K., Lee H. K., Kang Y. C., Kim C. H. Human umbilical cord mesenchymal stem cell-derived mitochondria (PN-101) attenuate LPS-induced inflammatory responses by inhibiting NFκB signaling pathway. BMB Rep. 2022;55:136–141. doi: 10.5483/BMBRep.2022.55.3.083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jackson M. V., Morrison T. J., Doherty D. F., McAuley D. F., Matthay M. A., Kissenpfennig A., O'Kane C. M., Krasnodembskaya A. D. Mitochondrial transfer via tunneling nanotubes is an important mechanism by which mesenchymal stem cells enhance macrophage phagocytosis in the in vitro and in vivo models of ARDS. Stem Cells. 2016;34:2210–2223. doi: 10.1002/stem.2372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Játiva S., Calle P., Torrico S., Muñoz Á., García M., Martinez I., Sola A., Hotter G. Mitochondrial transplantation enhances phagocytic function and decreases lipid accumulation in foam cell macrophages. Biomedicines. 2022;10:329. doi: 10.3390/biomedicines10020329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ikeda G., Santoso M. R., Tada Y., Li A. M., Vaskova E., Jung J. H., O'Brien C., Egan E., Ye J., Yang P. C. Mitochondria-rich extracellular vesicles from autologous stem cell-derived cardiomyocytes restore energetics of ischemic myocardium. J Am Coll Cardiol. 2021;77:1073–1088. doi: 10.1016/j.jacc.2020.12.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bertero E., Maack C., O'Rourke B. Mitochondrial transplantation in humans:“magical”cure or cause for concern? J Clin Invest. 2018;128:5191-5194. doi: 10.1172/JCI124944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hsu C. H., Roan J. N., Fang S. Y., Chiu M. H., Cheng T. T., Huang C. C., Lin M. W., Lam C. F. Transplantation of viable mitochondria improves right ventricular performance and pulmonary artery remodeling in rats with pulmonary arterial hypertension. J Thorac Cardiovasc Surg. 2022;163:e361-e373. doi: 10.1016/j.jtcvs.2020.08.014. [DOI] [PubMed] [Google Scholar]
- 80.Chang J. C., Liu K. H., Li Y. C., Kou S. J., Wei Y. H., Chuang C. S., Hsieh M., Liu C. S. Functional recovery of human cells harbouring the mitochondrial DNA mutation MERRF A8344G via peptide-mediated mitochondrial delivery. Neurosignals. 2013;21:160–173. doi: 10.1159/000341981. [DOI] [PubMed] [Google Scholar]
- 81.Macheiner T., Fengler V. H., Agreiter M., Eisenberg T., Madeo F., Kolb D., Huppertz B., Ackbar R., Sargsyan K. Magnetomitotransfer:An efficient way for direct mitochondria transfer into cultured human cells. Sci Rep. 2016;6:35571. doi: 10.1038/srep35571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chang J. C., Chang H. S., Wu Y. C., Cheng W. L., Lin T. T., Chang H. J., Chen S. T., Liu C. S. Antitumor actions of intratumoral delivery of membrane-fused mitochondria in a mouse model of triple-negative breast cancers. Onco Targets Ther. 2020;13:5241–5255. doi: 10.2147/OTT.S238143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gäbelein C. G., Feng Q., Sarajlic E., Zambelli T., Guillaume-Gentil O., Kornmann B., Vorholt J. A. Mitochondria transplantation between living cells. PLoS Biol. 2022;20:e3001576. doi: 10.1371/journal.pbio.3001576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kim M. J., Hwang J. W., Yun C. K., Lee Y., Choi Y. S. Delivery of exogenous mitochondria via centrifugation enhances cellular metabolic function. Sci Rep. 2018;8:3330. doi: 10.1038/s41598-018-21539-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Baudo G., Wu S., Massaro M., Liu H., Lee H., Zhang A., Hamilton D. J., Blanco E. Polymer-functionalized mitochondrial transplantation to fibroblasts counteracts a pro-fibrotic phenotype. Int J Mol Sci. 2023;24:10913. doi: 10.3390/ijms241310913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wu S., Zhang A., Li S., Chatterjee S., Qi R., Segura-Ibarra V., Ferrari M., Gupte A., Blanco E., Hamilton D. J. Polymer functionalization of isolated mitochondria for cellular transplantation and metabolic phenotype alteration. Adv Sci (Weinh) 2018;5:1700530. doi: 10.1002/advs.201700530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Galvan D. L., Green N. H., Danesh F. R. The hallmarks of mitochondrial dysfunction in chronic kidney disease. Kidney Int. 2017;92:1051–1057. doi: 10.1016/j.kint.2017.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kubat G. B., Kartal Y., Atalay O., Ulger O., Ekinci O., Celik E., Safali M., Urkan M., Karahan S., Ozler M., Cicek Z., Budak M. T. Investigation of the effect of isolated mitochondria transplantation on renal ischemia-reperfusion injury in rats. Toxicol Appl Pharmacol. 2021;433:115780. doi: 10.1016/j.taap.2021.115780. [DOI] [PubMed] [Google Scholar]
- 89.Doulamis I. P., Guariento A., Duignan T., Kido T., Orfany A., Saeed M. Y., Weixler V. H., Blitzer D., Shin B., Snay E. R., Inkster J. A., Packard A. B., Zurakowski D., Rousselle T., Bajwa A., Parikh S. M., Stillman I. E., Del Nido P. J., McCully J. D. Mitochondrial transplantation by intra-arterial injection for acute kidney injury. Am J Physiol Renal Physiol. 2020;319:F403-F413. doi: 10.1152/ajprenal.00255.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Arjmand A., Shiranirad S., Ameritorzani F., Kamranfar F., Seydi E., Pourahmad J. Mitochondrial transplantation against gentamicin-induced toxicity on rat renal proximal tubular cells:the higher activity of female rat mitochondria. In Vitro Cell Dev Biol Anim. 2023;59:31–40. doi: 10.1007/s11626-022-00743-1. [DOI] [PubMed] [Google Scholar]
- 91.Cloer C. M., Givens C. S., Buie L. K., Rochelle L. K., Lin Y. T., Popa S., Shelton R. V. M., Zhan J., Zimmerman T. R., Jones B. G., Lesesne Z., Hogan S. S., Petersen T. H. Mitochondrial transplant after ischemia reperfusion promotes cellular salvage and improves lung function during ex-vivo lung perfusion. J Heart Lung Transplant. 2023;42:575–584. doi: 10.1016/j.healun.2023.01.002. [DOI] [PubMed] [Google Scholar]
- 92.Lin H. C., Liu S. Y., Lai H. S., Lai I. R. Isolated mitochondria infusion mitigates ischemia-reperfusion injury of the liver in rats. Shock. 2013;39:304–310. doi: 10.1097/SHK.0b013e318283035f. [DOI] [PubMed] [Google Scholar]
- 93.Ulger O., Kubat G. B., Cicek Z., Celik E., Atalay O., Suvay S., Ozler M. The effects of mitochondrial transplantation in acetaminophen-induced liver toxicity in rats. Life Sci. 2021;279:119669. doi: 10.1016/j.lfs.2021.119669. [DOI] [PubMed] [Google Scholar]
- 94.Lu T., Zhang J., Cai J., Xiao J., Sui X., Yuan X., Li R., Li Y., Yao J., Lv G., Chen X., Chen H., Zeng K., Liu Y., Chen W., Chen G., Yang Y., Zheng J., Zhang Y. Extracellular vesicles derived from mesenchymal stromal cells as nanotherapeutics for liver ischaemia-reperfusion injury by transferring mitochondria to modulate the formation of neutrophil extracellular traps. Biomaterials. 2022;284:121486. doi: 10.1016/j.biomaterials.2022.121486. [DOI] [PubMed] [Google Scholar]
- 95.Cowan D. B., Yao R., Akurathi V., Snay E. R., Thedsanamoorthy J. K., Zurakowski D., Ericsson M., Friehs I., Wu Y., Levitsky S., Del Nido P. J., Packard A. B., McCully J D. Intracoronary delivery of mitochondria to the ischemic heart for cardioprotection. PLoS One. 2016;11:e0160889. doi: 10.1371/journal.pone.0160889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Sun C., Liu X., Wang B., Wang Z., Liu Y., Di C., Si J., Li H., Wu Q., Xu D., Li J., Li G., Wang Y., Wang F., Zhang H. Endocytosis-mediated mitochondrial transplantation:Transferring normal human astrocytic mitochondria into glioma cells rescues aerobic respiration and enhances radiosensitivity. Theranostics. 2019;9:3595–3607. doi: 10.7150/thno.33100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lesnefsky E. J., Chen Q., Slabe T. J., Stoll M. S., Minkler P. E., Hassan M. O., Tandler B., Hoppel C. L. Ischemia, rather than reperfusion, inhibits respiration through cytochrome oxidase in the isolated, perfused rabbit heart:role of cardiolipin. Am J Physiol Heart Circ Physiol. 2004;287:H258-267. doi: 10.1152/ajpheart.00348.2003. [DOI] [PubMed] [Google Scholar]
- 98.Chen Q., Moghaddas S., Hoppel C. L., Lesnefsky E. J. Reversible blockade of electron transport during ischemia protects mitochondria and decreases myocardial injury following reperfusion. J Pharmacol Exp Ther. 2006;319:1405–1412. doi: 10.1124/jpet.106.110262. [DOI] [PubMed] [Google Scholar]
- 99.Meerson F. Z., Zaletayeva T. A., Lagutchev S. S., Pshennikova M. G. Structure and mass of mitochondria in the process of compensatory hyperfunction and hypertrophy of the heart. Exp Cell Res. 1964;36:568–578. doi: 10.1016/0014-4827(64)90313-1. [DOI] [PubMed] [Google Scholar]
- 100.Weiss R. G., Gerstenblith G., Bottomley P. A. ATP flux through creatine kinase in the normal, stressed, and failing human heart. Proc Natl Acad Sci U S A. 2005;102:808–813. doi: 10.1073/pnas.0408962102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bisaccia G., Ricci F., Gallina S., Di Baldassarre A., Ghinassi B. Mitochondrial dysfunction and heart disease:critical appraisal of an overlooked association. Int J Mol Sci. 2021;22:614. doi: 10.3390/ijms22020614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Russo M. J., Iribarne A., Hong K. N., Ramlawi B., Chen J. M., Takayama H., Mancini D. M., Naka Y. Factors associated with primary graft failure after heart transplantation. Transplantation. 2010;90:444–450. doi: 10.1097/TP.0b013e3181e6f1eb. [DOI] [PubMed] [Google Scholar]
- 103.Sun X., Chen H., Gao R., Huang Y., Qu Y., Yang H., Wei X., Hu S., Zhang J., Wang P., Zou Y., Hu K., Ge J., Sun A. Mitochondrial transplantation ameliorates doxorubicin-induced cardiac dysfunction via activating glutamine metabolism. iScience. 2023;26:107790. doi: 10.1016/j.isci.2023.107790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ali Pour P., Kenney M. C., Kheradvar A. Bioenergetics Consequences of Mitochondrial Transplantation in Cardiomyocytes. J Am Heart Assoc. 2020;9:e014501. doi: 10.1161/JAHA.119.014501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Roche S., D'Ippolito G., Gomez L. A., Bouckenooghe T., Lehmann S., Montero-Menei C. N., Schiller P. C. Comparative analysis of protein expression of three stem cell populations:models of cytokine delivery system in vivo. Int J Pharm. 2013;440:72–82. doi: 10.1016/j.ijpharm.2011.12.041. [DOI] [PubMed] [Google Scholar]
- 106.Alemany V. S., Nomoto R., Saeed M. Y., Celik A., Regan W. L., Matte G. S., Recco D. P., Emani S. M., Del Nido P. J., McCully J. D. Mitochondrial transplantation preserves myocardial function and viability in pediatric and neonatal pig hearts donated after circulatory death. J Thorac Cardiovasc Surg. 2024;167:e6-e21. doi: 10.1016/j.jtcvs.2023.05.010. [DOI] [PubMed] [Google Scholar]
- 107.Guariento A., Doulamis I. P., Duignan T., Kido T., Regan W. L., Saeed M. Y., Hoganson D. M., Emani S. M., Fynn-Thompson F., Matte G. S., Del Nido P. J., McCully J. D. Mitochondrial transplantation for myocardial protection in ex-situ‒perfused hearts donated after circulatory death. J Heart Lung Transplant. 2020;39:1279–1288. doi: 10.1016/j.healun.2020.06.023. [DOI] [PubMed] [Google Scholar]
- 108.Hayashida K., Takegawa R., Endo Y., Yin T., Choudhary R. C., Aoki T., Nishikimi M., Murao A., Nakamura E., Shoaib M., Kuschner C., Miyara S. J., Kim J., Shinozaki K., Wang P., Becker L. B. Exogenous mitochondrial transplantation improves survival and neurological outcomes after resuscitation from cardiac arrest. BMC Med. 2023;21:56. doi: 10.1186/s12916-023-02759-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Shin B., Saeed M. Y., Esch J. J., Guariento A., Blitzer D., Moskowitzova K., Ramirez-Barbieri G., Orfany A., Thedsanamoorthy J. K., Cowan D. B., Inkster J. A., Snay E. R., Staffa S. J., Packard A. B., Zurakowski D., Del Nido P. J., McCully J. D. A novel biological strategy for myocardial protection by intracoronary delivery of mitochondria:safety and efficacy. JACC Basic Transl Sci. 2019;4:871–888. doi: 10.1016/j.jacbts.2019.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lin Z. J., Kim S., Cui H. X., Han K., Lee H. K., Kim C. H., Kang Y. C., Zhang Y. H. Human platelet mitochondria improve the mitochondrial and cardiac function of donor heart. Pflugers Arch. 2023;475:267–275. doi: 10.1007/s00424-022-02763-y. [DOI] [PubMed] [Google Scholar]
- 111.Lin L., Xu H., Bishawi M., Feng F., Samy K., Truskey G., Barbas A. S., Kirk A. D., Brennan T. V. Circulating mitochondria in organ donors promote allograft rejection. Am J Transplant. 2019;19:1917–1929. doi: 10.1111/ajt.15309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Gan I., Jiang J., Lian D., Huang X., Fuhrmann B., Liu W., Haig A., Jevnikar A. M., Zhang Z. X. Mitochondrial permeability regulates cardiac endothelial cell necroptosis and cardiac allograft rejection. Am J Transplant. 2019;19:686–698. doi: 10.1111/ajt.15112. [DOI] [PubMed] [Google Scholar]
- 113.Doulamis I. P., Guariento A., Duignan T., Orfany A., Kido T., Zurakowski D., Del Nido P. J., McCully J. D. Mitochondrial transplantation for myocardial protection in diabetic hearts. Eur J Cardiothorac Surg. 2020;57:836–845. doi: 10.1093/ejcts/ezz326. [DOI] [PubMed] [Google Scholar]
- 114.Mokhtari B., Badalzadeh R. Mitochondria-targeted combination treatment strategy counteracts myocardial reperfusion injury of aged rats by modulating autophagy and inflammatory response. Mol Biol Rep. 2023;50:3973–3983. doi: 10.1007/s11033-023-08318-3. [DOI] [PubMed] [Google Scholar]
- 115.Liu H., Li Z., Cao Y., Cui Y., Yang X., Meng Z., Wang R. Effect of chondrocyte mitochondrial dysfunction on cartilage degeneration:a possible pathway for osteoarthritis pathology at the subcellular level. Mol Med Rep. 2019;20:3308–3316. doi: 10.3892/mmr.2019.10559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Yu M., Wang D., Chen X., Zhong D., Luo J. BMSCs-derived mitochondria improve osteoarthritis by ameliorating mitochondrial dysfunction and promoting mitochondrial biogenesis in chondrocytes. Stem Cell Rev Rep. 2022;18:3092–3111. doi: 10.1007/s12015-022-10436-7. [DOI] [PubMed] [Google Scholar]
- 117.Guo Y., Chi X., Wang Y., Heng B. C., Wei Y., Zhang X., Zhao H., Yin Y., Deng X. Mitochondria transfer enhances proliferation, migration, and osteogenic differentiation of bone marrow mesenchymal stem cell and promotes bone defect healing. Stem Cell Res Ther. 2020;11:245. doi: 10.1186/s13287-020-01704-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Maezawa T., Tanaka M., Kanazashi M., Maeshige N., Kondo H., Ishihara A., Fujino H. Astaxanthin supplementation attenuates immobilization-induced skeletal muscle fibrosis via suppression of oxidative stress. J Physiol Sci. 2017;67:603–611. doi: 10.1007/s12576-016-0492-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Lee J. M., Hwang J. W., Kim M. J., Jung S. Y., Kim K. S., Ahn E. H., Min K., Choi Y. S. Mitochondrial transplantation modulates inflammation and apoptosis, alleviating tendinopathy both in vivo and in vitro. Antioxidants (Basel) 2021;10:696. doi: 10.3390/antiox10050696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Budge K. M., Neal M. L., Richardson J. R., Safadi F. F. Glycoprotein NMB:an Emerging Role in Neurodegenerative Disease. Mol Neurobiol. 2018;55:5167–5176. doi: 10.1007/s12035-017-0707-z. [DOI] [PubMed] [Google Scholar]
- 121.Hou Y., Dan X., Babbar M., Wei Y., Hasselbalch S. G., Croteau D. L., Bohr V. A. Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol. 2019;15:565–581. doi: 10.1038/s41582-019-0244-7. [DOI] [PubMed] [Google Scholar]
- 122.Kauppila T. E. S., Kauppila J. H. K., Larsson N. G. Mammalian mitochondria and aging:an update. Cell Metab. 2017;25:57–71. doi: 10.1016/j.cmet.2016.09.017. [DOI] [PubMed] [Google Scholar]
- 123.Balaban R. S., Nemoto S., Finkel T. Mitochondria, oxidants, and aging. Cell. 2005;120:483–495. doi: 10.1016/j.cell.2005.02.001. [DOI] [PubMed] [Google Scholar]
- 124.Noh S. E., Lee S. J., Lee T. G., Park K. S., Kim J. H. Inhibition of cellular senescence hallmarks by mitochondrial transplantation in senescence-induced ARPE-19 cells. Neurobiol Aging. 2023;121:157–165. doi: 10.1016/j.neurobiolaging.2022.11.003. [DOI] [PubMed] [Google Scholar]
- 125.Swerdlow R. H. Mitochondria and mitochondrial cascades in Alzheimer's disease. J Alzheimers Dis. 2018;62:1403–1416. doi: 10.3233/JAD-170585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Sharma C., Kim S., Nam Y., Jung U. J., Kim S. R. Mitochondrial dysfunction as a driver of cognitive impairment in Alzheimer's disease. Int J Mol Sci. 2021;22:4850. doi: 10.3390/ijms22094850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Nitzan K., Benhamron S., Valitsky M., Kesner E. E., Lichtenstein M., Ben-Zvi A., Ella E., Segalstein Y., Saada A., Lorberboum-Galski H., Rosenmann H. Mitochondrial transfer ameliorates cognitive deficits, neuronal loss, and gliosis in Alzheimer's disease mice. J Alzheimers Dis. 2019;72:587–604. doi: 10.3233/JAD-190853. [DOI] [PubMed] [Google Scholar]
- 128.Zhang Z., Wei D., Li Z., Guo H., Wu Y., Feng J. Hippocampal mitochondrial transplantation alleviates age-associated cognitive decline via enhancing wnt signaling and neurogenesis. Comput Intell Neurosci. 2022;2022:9325302. doi: 10.1155/2022/9325302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Palomer E., Buechler J., Salinas P. C. Wnt signaling deregulation in the aging and Alzheimer's brain. Front Cell Neurosci. 2019;13:227. doi: 10.3389/fncel.2019.00227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Mishra M., Raik S., Rattan V., Bhattacharyya S. Mitochondria transfer as a potential therapeutic mechanism in Alzheimer's disease-like pathology. Brain Res. 2023;1819:148544. doi: 10.1016/j.brainres.2023.148544. [DOI] [PubMed] [Google Scholar]
- 131.Chen T., Majerníková N., Marmolejo-Garza A., Trombetta-Lima M., Sabogal-Guáqueta A. M., Zhang Y., Ten Kate R., Zuidema M., Mulder P., den Dunnen W., Gosens R., Verpoorte E., Culmsee C., Eisel U. L. M., Dolga A. M. Mitochondrial transplantation rescues neuronal cells from ferroptosis. Free Radic Biol Med. 2023;208:62–72. doi: 10.1016/j.freeradbiomed.2023.07.034. [DOI] [PubMed] [Google Scholar]
- 132.Chen W. W., Zhang X., Huang W. J. Role of neuroinflammation in neurodegenerative diseases (Review) Mol Med Rep. 2016;13:3391–3396. doi: 10.3892/mmr.2016.4948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Zhao J., Qu D., Xi Z., Huan Y., Zhang K., Yu C., Yang D., Kang J., Lin W., Wu S., Wang Y. Mitochondria transplantation protects traumatic brain injury via promoting neuronal survival and astrocytic BDNF. Transl Res. 2021;235:102–114. doi: 10.1016/j.trsl.2021.03.017. [DOI] [PubMed] [Google Scholar]
- 134.Pendergrass J. C., Targum S. D., Harrison J. E. Cognitive impairment associated with cancer:a brief review. Innov Clin Neurosci. 2018;15:36–44. [PMC free article] [PubMed] [Google Scholar]
- 135.Yan C., Ma Z., Ma H., Li Q., Zhai Q., Jiang T., Zhang Z., Wang Q. Mitochondrial transplantation attenuates brain dysfunction in sepsis by driving microglial M2 polarization. Mol Neurobiol. 2020;57:3875–3890. doi: 10.1007/s12035-020-01994-3. [DOI] [PubMed] [Google Scholar]
- 136.Amin S. N. Platelets:the peripheral donor of mitochondria for diabetes-induced cognitive impairment. Clin Sci (Lond) 2021;135:593–595. doi: 10.1042/CS20201297. [DOI] [PubMed] [Google Scholar]
- 137.Ma H., Jiang T., Tang W., Ma Z., Pu K., Xu F., Chang H., Zhao G., Gao W., Li Y., Wang Q. Transplantation of platelet-derived mitochondria alleviates cognitive impairment and mitochondrial dysfunction in db/db mice. Clin Sci (Lond) 2020;134:2161–2175. doi: 10.1042/CS20200530. [DOI] [PubMed] [Google Scholar]
- 138.Robicsek O., Ene H. M., Karry R., Ytzhaki O., Asor E., McPhie D., Cohen B. M., Ben-Yehuda R., Weiner I., Ben-Shachar D. Isolated mitochondria transfer improves neuronal differentiation of schizophrenia-derived induced pluripotent stem cells and rescues deficits in a rat model of the disorder. Schizophr Bull. 2018;44:432–442. doi: 10.1093/schbul/sbx077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Wang Y., Ni J., Gao C., Xie L., Zhai L., Cui G., Yin X. Mitochondrial transplantation attenuates lipopolysaccharide- induced depression-like behaviors. Prog Neuropsychopharmacol Biol Psychiatry. 2019;93:240–249. doi: 10.1016/j.pnpbp.2019.04.010. [DOI] [PubMed] [Google Scholar]
- 140.Javani G., Babri S., Farajdokht F., Ghaffari-Nasab A., Mohaddes G. Mitochondrial transplantation improves anxiety- and depression-like behaviors in aged stress-exposed rats. Mech Ageing Dev. 2022;202:111632. doi: 10.1016/j.mad.2022.111632. [DOI] [PubMed] [Google Scholar]
- 141.Harris J. J., Jolivet R., Attwell D. Synaptic energy use and supply. Neuron. 2012;75:762–777. doi: 10.1016/j.neuron.2012.08.019. [DOI] [PubMed] [Google Scholar]
- 142.Gollihue J. L., Patel S. P., Eldahan K. C., Cox D. H., Donahue R. R., Taylor B. K., Sullivan P. G., Rabchevsky A. G. Effects of mitochondrial transplantation on bioenergetics, cellular incorporation, and functional recovery after spinal cord injury. J Neurotrauma. 2018;35:1800–1818. doi: 10.1089/neu.2017.5605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Xie Q., Zeng J., Zheng Y., Li T., Ren J., Chen K., Zhang Q., Xie R., Xu F., Zhu J. Mitochondrial transplantation attenuates cerebral ischemia-reperfusion injury:possible involvement of mitochondrial component separation. Oxid Med Cell Longev. 2021;2021:1006636. doi: 10.1155/2021/1006636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Bamshad C., Habibi Roudkenar M., Abedinzade M., Yousefzadeh Chabok S., Pourmohammadi-Bejarpasi Z., Najafi-Ghalehlou N., Sato T., Tomita K., Jahanian-Najafabadi A., Feizkhah A., Mohammadi Roushandeh A. Human umbilical cord-derived mesenchymal stem cells-harvested mitochondrial transplantation improved motor function in TBI models through rescuing neuronal cells from apoptosis and alleviating astrogliosis and microglia activation. Int Immunopharmacol. 2023;118:110106. doi: 10.1016/j.intimp.2023.110106. [DOI] [PubMed] [Google Scholar]
- 145.Lee E. H., Kim M., Ko S. H., Kim C. H., Lee M., Park C. H. Primary astrocytic mitochondrial transplantation ameliorates ischemic stroke. BMB Rep. 2023;56:90–95. doi: 10.5483/BMBRep.2022-0108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Tseng N., Lambie S. C., Huynh C. Q., Sanford B., Patel M., Herson P. S., Ormond D. R. Mitochondrial transfer from mesenchymal stem cells improves neuronal metabolism after oxidant injury in vitro:The role of Miro1. J Cereb Blood Flow Metab. 2021;41:761–770. doi: 10.1177/0271678X20928147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Pourmohammadi-Bejarpasi Z., Roushandeh A. M., Saberi A., Rostami M. K., Toosi S. M. R., Jahanian-Najafabadi A., Tomita K., Kuwahara Y., Sato T., Roudkenar M. H. Mesenchymal stem cells-derived mitochondria transplantation mitigates I/R-induced injury, abolishes I/R-induced apoptosis, and restores motor function in acute ischemia stroke rat model. Brain Res Bull. 2020;165:70–80. doi: 10.1016/j.brainresbull.2020.09.018. [DOI] [PubMed] [Google Scholar]
- 148.Sullivan P. G., Krishnamurthy S., Patel S. P., Pandya J. D., Rabchevsky A. G. Temporal characterization of mitochondrial bioenergetics after spinal cord injury. J Neurotrauma. 2007;24:991–999. doi: 10.1089/neu.2006.0242. [DOI] [PubMed] [Google Scholar]
- 149.Huang T., Shen J., Lin J., Zheng X. Effect of M2-like macrophage/microglia-derived mitochondria transplantation in treatment of mouse spinal cord injury. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2022;36:751–759. doi: 10.7507/1002-1892.202201040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Van den Bossche J., O'Neill L. A., Menon D. Macrophage immunometabolism:where are we (going)? Trends Immunol. 2017;38:395-406. doi: 10.1016/j.it.2017.03.001. [DOI] [PubMed] [Google Scholar]
- 151.Zhu Z., Li X., Wang X., Zuo X., Ma Y., Gao X., Liang Z., Zhang Z., Song Z., Ding T., Ju C., Li P., Li K., Zhang J., Quan H., Wang Z., Hu X. Photobiomodulation augments the effects of mitochondrial transplantation in the treatment of spinal cord injury in rats by facilitating mitochondrial transfer to neurons via Connexin 36. Bioeng Transl Med. 2023;8:e10473. doi: 10.1002/btm2.10473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wu S. F., Lin C. Y., Tsai R. K., Wen Y. T., Lin F. H., Chang C. Y., Shen C. I., Lin S. Z., Harn H. J., Chiou T. W., Liu C. S., Chen Y. T., Su H. L. Mitochondrial transplantation moderately ameliorates retinal degeneration in royal college of surgeons rats. Biomedicines. 2022;10:2883. doi: 10.3390/biomedicines10112883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Singer M., Deutschman C. S., Seymour C. W., Shankar-Hari M., Annane D., Bauer M., Bellomo R., Bernard G. R., Chiche J. D., Coopersmith C. M., Hotchkiss R. S., Levy M. M., Marshall J. C., Martin G. S., Opal S. M., Rubenfeld G. D., van der Poll T., Vincent J. L., Angus D. C. The Third International Consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;315:801–810. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Kim Y. S., Lee H. A. R., Lee M. J., Park Y. J., Mun S., Yune C. J., Chung T. N., Bae J., Kim M. J., Choi Y. S., Kim K. The effects of mitochondrial transplantation on sepsis depend on the type of cell from which they are isolated. Int J Mol Sci. 2023;24:10113. doi: 10.3390/ijms241210113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.He H., Xiong L., Jian L., Li L., Wu Y., Qiao S. Role of mitochondria on UV-induced skin damage and molecular mechanisms of active chemical compounds targeting mitochondria. J Photochem Photobiol B. 2022;232:112464. doi: 10.1016/j.jphotobiol.2022.112464. [DOI] [PubMed] [Google Scholar]
- 156.Hu S. S., Li R. Y., Cao X. H., Liu J. J., Wang Z. H., Li Z., Yang M. L., Liu J. W., Hu L. M., Lin C. J., Liu J., Wang C. M. Structural integrity is essential for the protective effect of mitochondrial transplantation against UV-induced cell death. J Photochem Photobiol B. 2022;234:112534. doi: 10.1016/j.jphotobiol.2022.112534. [DOI] [PubMed] [Google Scholar]
- 157.Cabrera F., Ortega M., Velarde F., Parra E., Gallardo S., Barba D., Soto L., Peña G., Pedroza L. A., Jorgensen C., Khoury M., Caicedo A. Primary allogeneic mitochondrial mix (PAMM) transfer/transplant by MitoCeption to address damage in PBMCs caused by ultraviolet radiation. BMC Biotechnol. 2019;19:42. doi: 10.1186/s12896-019-0534-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Schäfer M., Werner S. Oxidative stress in normal and impaired wound repair. Pharmacol Res. 2008;58:165–171. doi: 10.1016/j.phrs.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 159.Kim S., Kim Y., Yu S. H., Lee S. E., Park J. H., Cho G., Choi C., Han K., Kim C. H., Kang Y. C. Platelet-derived mitochondria transfer facilitates wound-closure by modulating ROS levels in dermal fibroblasts. Platelets. 2022;34:2151996. doi: 10.1080/09537104.2022.2151996. [DOI] [PubMed] [Google Scholar]
- 160.Guariento A., Piekarski B. L., Doulamis I. P., Blitzer D., Ferraro A. M., Harrild D. M., Zurakowski D., Del Nido P. J., McCully J. D., Emani S. M. Autologous mitochondrial transplantation for cardiogenic shock in pediatric patients following ischemia-reperfusion injury. J Thorac Cardiovasc Surg. 2021;162:992–1001. doi: 10.1016/j.jtcvs.2020.10.151. [DOI] [PubMed] [Google Scholar]
- 161.Emani S. M., Piekarski B. L., Harrild D., Del Nido P. J., McCully J. D. Autologous mitochondrial transplantation for dysfunction after ischemia-reperfusion injury. J Thorac Cardiovasc Surg. 2017;154:286–289. doi: 10.1016/j.jtcvs.2017.02.018. [DOI] [PubMed] [Google Scholar]
- 162.Bertero E., O'Rourke B., Maack C. Mitochondria do not survive calcium overload during transplantation. Circ Res. 2020;126:784–786. doi: 10.1161/CIRCRESAHA.119.316291. [DOI] [PMC free article] [PubMed] [Google Scholar]


