ABSTRACT
Objective
Potentially untrustworthy medical research is often identified after publication. We evaluated the effectiveness and efficiency of post‐publication review of such studies in women's health.
Design
Cohort study.
Sample
Potentially untrustworthy papers published in women's health journals.
Methods
We wrote to the editors and publishers about potentially untrustworthy papers in women's health and requested an investigation according to the procedure established by the Committee of Publication Ethics (COPE).
Main Outcome Measure
Study characteristics, investigation outcome classed as retraction, expression of concern (EoC), correction or no wrongdoing found, and time to decision. We also report the case completion rate per journal and publisher.
Results
Between 7th November 2017 and 30th April 2024, we wrote to editors and publishers of 891 potentially untrustworthy papers published in 206 different journals. At present, 263 (30%) of 891 papers received an outcome, with 227 (86%) labelled as problematic [152 (58%) retracted; 75 (29%) EoC]. For articles with a decision, it took a median time of 38 months for editors and publishers to decide, with 13% of the flagged cases reaching a decision within 12 months.
Conclusions
The current PPR process is inefficient and ineffective in assessing and removing untrustworthy data from the medical literature.
Keywords: post‐publication review, trustworthiness, untrustworthy data, Women's health
1. Introduction
Clinical research is the cornerstone of evidence‐based medicine and the foundation of modern healthcare. While the design and quality of studies can be discussed, it has, until now, been taken at face value that studies are at least based on reliable data. In recent years, it has become apparent that this assumption is incorrect [1, 2, 3, 4]. This is worrisome since these studies, specifically randomised clinical trials (RCTs), continue to inform medical guidelines and clinical practice.
Carlisle reported that among > 500 RCTs submitted to the journal Anaesthesia, 73 (14%) contained false data and 43 (8%) were deemed fatally flawed ‘zombie trials’ [1]. Based on this, Ioannidis estimated that there could be hundreds of thousands of problematic RCTs across all disciplines that go unchallenged and can even be harmful [3].
Alfirevic et al. using the Cochrane Pregnancy and Childbirth Group's trustworthiness screening tool (CPC‐TST) to assess 374 RCTs included in 18 Cochrane reviews, concluded that 95 RCTs (25%) merited exclusion for this reason, resulting in changes in 14 (80%) of the reviews, and important differences in the conclusions and/or implications in six (33%) [5]. A recent update of the International Evidence‐based Guideline for Polycystic Ovary Syndrome (PCOS) excluded 45% of eligible RCTs for not meeting trustworthiness criteria [6]. The estimate that at least 25% of RCTs used to inform clinical practice are untrustworthy seems justified [1, 3, 7].
Pre‐publication prevention is ideal, but a different approach is needed for already published studies. The Committee on Publication of Ethics (COPE), a charitable organisation that advocates for research integrity and ethical conduct, has established guidelines for post‐publication review and COPE member journals are bound by these [8]. If data integrity breaches are flagged, journals and publishers must investigate and, in case of wrongdoing, issue a formal public notice [9]. This serves as a critical juncture for investigating, correcting and, if needed, retracting potentially problematic data from the published literature.
We studied the effectiveness and efficiency of this process by assessing the response rate of editors and publishers of women's health journals to concerns raised about papers with potentially problematic data.
2. Method
2.1. Study Design
Between 7th November 2017 and 30th April 2024, we collated potentially untrustworthy clinical research papers, mainly on women's health. The papers were initially identified in the process of establishing systematic reviews and clinical guidelines. We also systematically assessed the work of authors in which problems had been identified, and we followed newly published RCTs. We used online databases, including PubMed, PubPeer, Google Scholar and the Retraction Watch database, to track author's previous work [10, 11]. Each identified RCT was cross‐checked against items in the checklist to assess Trustworthiness in Randomised Clinical Trials (TRACT) [12]. This checklist assesses trustworthiness based on seven domains (governance, author group, plausibility of intervention usage, timeframe, drop‐out rate, baseline characteristics and outcomes) [12]. Each domain has signalling questions answered as either no concerns, some concerns/no information, or major concerns [12]. Since the TRACT checklist was developed over the trajectory of this study, early assessments were less formal. Observational studies were also assessed using applicable checklist items. If concerns were raised in several domains, or if an article showed a single finding highly suggestive of data integrity breach, the article was flagged to the editors and publishers.
We have previously published examples of problematic papers [13, 14]. Our concerns were classified into eight categories: double publication, ethics concerns, fabricated data, plagiarism, similar outcome group characteristics, statistical discrepancies, study protocol deviations and trial registry discrepancies.
We informed journal editors and publishers via e‐mail, highlighting the concerns in detail and requested that the papers be assessed according to COPE guidelines [8]. We requested confirmation of receipt and resent if no response was received or we had no updates after 6 months.
2.2. Data Collection
All data were collected in a Google spreadsheet accessible to team members. We tabulated author(s), publication year, journal, publisher, institute, type of concern raised, type of study (RCT/observational), article link, initial e‐mail date sent to editors and publishers, and date of confirmation of receipt. Progress was denoted as retraction, Expression of Concern (EoC), correction, investigation concluded no action required or pending investigation. Initially, the last author (BWM) registered all findings; from 2023, the first author (SS) systematically verified all original e‐mails sent to editors and publishers, including the response date and outcome.
2.3. Statistical Analysis
We tabulated the type of outcomes received for each paper as retraction, EoC, correction or investigation concluded no action required and pending investigation. The median response time to issue an outcome was estimated using Kaplan–Meier curves. We note that there are several instances where the Kaplan–Meier curve provides an estimate of the median time to outcome despite < 50% of the papers in the sample being issued with an outcome. This is a consequence of including information from papers from which an outcome had not yet been issued. We performed subgroup analysis by publishers, journals and countries. Journals were categorised into five quartile rankings Q1, Q2, Q3, Q4 and unclassified according to the Scimago Journal and Country Ranking database [15]. We also calculated the time to response between the initial date of correspondence with editors and/or publishers and the final decision date. For each journal and publisher, we calculated the median time to response for those papers using Kaplan–Meier curves.
All statistical analysis was conducted using R statistical software (version 4.4.0). The analysis included outcomes as of 30th April 2024.
3. Results
Our initial database contained 1159 potentially untrustworthy papers (Figure 1). We excluded 268 papers where we had not yet written to editors (N = 218), or we considered the paper to be too old/insufficient evidence for concern at this point (N = 50).
FIGURE 1.
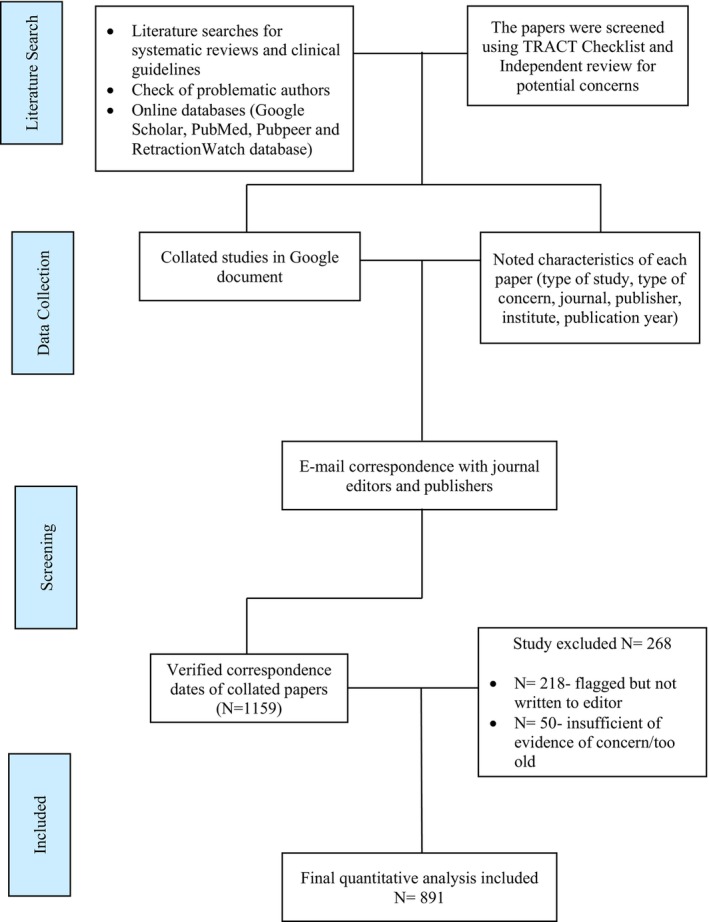
Flowchart of the number of papers analysed.
3.1. Baseline Characteristics
Table 1 presents the publication year, type of study and region of the studied papers. Most were published after 2010. More than half were RCTs.
TABLE 1.
Baseline characteristics of papers.
| Total N = 891 | Investigation concluded | Pending investigation N = 628 | ||||
|---|---|---|---|---|---|---|
| Retraction N = 152 | Expression of concern N = 75 | Correction N = 6 | Investigation concluded no action N = 30 | |||
| Publication year | ||||||
| < 2000 | 12 | 1 (8%) | 3 (25%) | 0 (0%) | 2 (17%) | 6 (50%) |
| 2000–2010 | 145 | 27 (19%) | 19 (13%) | 0 (0%) | 5 (3%) | 94 (65%) |
| 2010–2020 | 527 | 89 (17%) | 47 (9%) | 4 (1%) | 11 (2%) | 376 (71%) |
| 2020‐Present | 207 | 35 (17%) | 6 (3%) | 2 (1%) | 12 (6%) | 152 (73%) |
| Type of study | ||||||
| Observational | 362 | 40 (11%) | 25 (7%) | 1 (0.4%) | 5 (1%) | 291 (80%) |
| RCT | 529 | 112 (21%) | 50 (9.5%) | 5 (0.9%) | 25 (4.7%) | 337 (64%) |
| Country of origin | ||||||
| Middle East | 789 | 141 (18%) | 72 (9%) | 6 (1%) | 19 (2%) | 551 (70%) |
| Europe | 55 | 6 (11%) | 2 (4%) | 0 (0%) | 3 (5%) | 44 (80%) |
| Asia | 40 | 4 (10%) | 1 (2%) | 0 (0%) | 8 (20%) | 27 (68%) |
| Other (USA, Brazil and Tunisia) | 7 | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 7 (100%) |
| Year 1st email sent | ||||||
| 2017 | 2 | 2 (100%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| 2019 | 19 | 5 (26%) | 12 (64%) | 0 (0%) | 1 (5%) | 1 (5%) |
| 2020 | 62 | 34 (54%) | 10 (16%) | 0 (0%) | 2 (4%) | 16 (26%) |
| 2021 | 187 | 64 (34%) | 18 (10%) | 2 (1%) | 7 (4%) | 96 (51%) |
| 2022 | 351 | 39 (11%) | 27 (8%) | 4 (1%) | 8 (2%) | 273 (78%) |
| 2023 | 199 | 8 (4%) | 8 (4%) | 0 (0%) | 12 (6%) | 171 (86%) |
| 2024 (until April '24) | 71 | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 71 (100%) |
Note: % Calculated from the total number of cases.
The number of cases flagged increased over time. The majority of cases were authored from countries in the Middle East (Egypt = 674, Iran = 95, Saudi Arabia = 10, and Turkey = 8), followed by Europe (Italy = 43, Germany = 9) and Asia (India = 26, China = 10).
3.2. Outcome of Editorial Investigations
As of 30th April 2024, 263/891 papers (29%) had received a final decision: retraction (152/263, 58%) and EoC (75/263, 29%) made up 86% of the decisions. There were 30 papers (11%) where, according to the investigation, no editorial action was required, while six papers had an erratum and subsequent corrections. The remaining 628 papers (71%) were yet to receive a decision.
3.3. Summary of Concern of Retracted and Expression of Concern Papers
Among the 227 papers that were retracted or received an EoC, 134 papers (69%) received said outcomes due to the presence of false data (retracted due to false data, N = 87; EoC due to false data, N = 47). Moreover, 43/227 (19%) papers received a retraction (N = 26) or an EoC (N = 17) due to plagiarism. One paper was retracted because of a lack of ethics approval and another because of the double publication.
3.4. Outcome Response Time
The median time editors and publishers took to issue any outcome (retraction, EoC, correction, or investigation concluded no action) was 38 months, with only 13% of papers receiving conclusive outcomes within the first 12 months (Figure 2).
FIGURE 2.
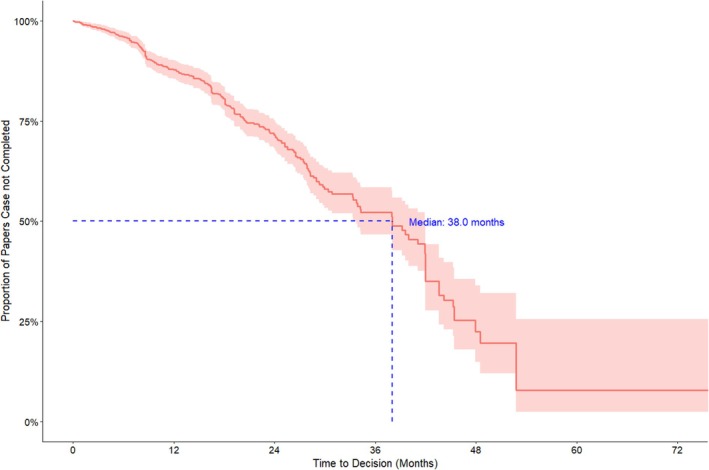
Kaplan‐Meier curve of time to case completion (all journals and publishers) up to 60 months, with right censoring for cases still“pending investigation” at the study end date.
3.5. Outcomes by Journal
We wrote to 206 journals published by 80 publishers or societies. The full case completion rates by journal, discipline and journal base region are provided in Table S1. The flagged papers spanned 31 disciplines, predominantly women's health‐related studies.
Of 210 journals, 54 issued responses, while the remaining 156 journals (each with between 1 and 49 concerns, mostly flagged due to the presence of potential false data) had not finalised any conclusions. Among the Q1 journals, Fertility and Sterility (57 cases) resolved 39% (22/57) of cases, with a median time to conclusion of 31 months (Table 2). Moreover, 76% of resolved cases by Fertility and Sterility were retractions (N = 14) or EoC (N = 2), the highest of all Q1 journals. The International Journal of Gynaecology and Obstetrics had the most papers flagged from all Q1 journals (N = 67), with a case completion rate of 33% and 57% of the completed cases resulting in a retraction (median time to response of 34 months) The European Journal of Contraception and Reproductive Health Care had the highest case completion rate (19/24, 75%), with 18 out of 19 completed cases receiving retractions after a median time to decision of 22 months [16, 17].
TABLE 2.
Journal case completion rate.
| Journal N = 206 | Q ranking | Publisher | Number of flagged papers N = 891 | Case completion rate (%) | Retraction N = 152 | Expression of concern N = 75 | Correction N = 6 | Investigation concluded no action N = 30 | Pending investigation N = 628 | Median time to response (Months) *assuming 30 days per month |
|---|---|---|---|---|---|---|---|---|---|---|
| Journal of Maternal‐Fetal and Neonatal Medicine | Q2 | Taylor & Francis | 78 (8.8%) | 29 (37%) | 11 (38%) | 18 (62%) | 0 (0%) | 0 (0%) | 49 (63%) | 28 |
| International Journal of Gynecology and Obstetrics | Q1 | Wiley Blackwell | 67 (7.5%) | 22 (33%) | 13 (57%) | 0 (0%) | 0 (0%) | 10 (43%) | 44 (66%) | 34 |
| Archives of Gynecology and Obstetrics | Q2 | Springer | 60 (6.7%) | 31 (52%) | 15 (48%) | 16 (52%) | 0 (0%) | 0 (0%) | 29 (48%) | 38 |
| Fertility Sterility | Q1 | Elsevier | 57 (6.4%) | 22 (37%) | 14 (64%) | 2 (9%) | 1 (5%) | 4 (18%) | 36 (63%) | 31 |
| European Journal of Obstetrics and Gynecology and Reproductive Biology | Q2 | Elsevier | 38 (4.3%) | 16 (42%) | 14 (88%) | 2 (12%) | 0 (0%) | 0 (0%) | 22 (58%) | 28 |
| Gynecological Endocrinology | Q2 | Taylor & Francis | 27 (3.0%) | 7 (26%) | 5 (71%) | 2 (29%) | 0 (0%) | 0 (0%) | 20 (74%) | 45 |
| European Journal of Contraception & Reproductive Health Care | Q2 | Taylor & Francis | 24 (2.7%) | 19 (79%) | 18 (95%) | 0 (0%) | 1 (5%) | 0 (0%) | 6 (25%) | 22 |
| Journal of Obstetrics and Gynaecology | Q3 | Taylor & Francis | 23 (2.6%) | 12 (52%) | 12 (100%) | 0 (0%) | 0 (0%) | 0 (0%) | 11 (48%) | 28 |
| Journal of Obstetrics and Gynaecology Research | Q2 | Wiley Blackwell | 21 (2.4%) | 5 (24%) | 2 (50%) | 1 (25%) | 1 (25%) | 0 (0%) | 17 (81%) | 42 |
| Reproductive Biomedicine Online | Q1 | Elsevier | 20 (2.2%) | 5 (25%) | 5 (100%) | 0 (0%) | 0 (0%) | 0 (0%) | 15 (75%) | 44 |
Note: % Calculated from the number of completed cases.
For European‐based journals, the case completion rate was 34%, with 59% of completed cases ending in retraction and 28% in an EoC. For US‐based journals, the case completion rate was 32%, with 52% of completed cases ending in retraction and 32% in an EoC. None of the Middle East‐based journals reached a decision on the 46 cases flagged.
3.6. Outcomes by Publisher
The four publishers with the most cases raised were Elsevier, Taylor & Francis, Springer, and Wiley‐Blackwell (Table 3). Case completion rates varied between 30% and 42%. Combined retraction and EoC rates for decisions taken were high, varying between (70/71, 99%) for Taylor & Francis and (68%) for Wiley‐Blackwell. Full case completion rates of all publishers are listed in Table S2. Karger had the highest case completion rate (7/9, 78%), with a median time of 7 months to resolve a case. Publisher Termedia did not resolve any of the 11 flagged cases.
TABLE 3.
Publisher case completion rate.
| Status | Informed papers N = 891 | Case completion (%) | Retraction N = 152 | Expression of concern N = 75 | Correction N = 6 | Investigation concluded no action N = 30 | Pending investigation N = 628 | Median time to response (Months), *assuming 30 days per month |
|---|---|---|---|---|---|---|---|---|
| Elsevier | 204 (23%) | 61 (30%) | 43 (70%) | 10 (16%) | 2 (3%) | 6 (10%) | 143 (70%) | 42 |
| Taylor & Francis | 170 (19%) | 70 (41%) | 50 (71%) | 20 (28%) | 0 (0%) | 1 (2%) | 100 (58%) | 28 |
| Springer | 154 (17%) | 50 (33%) | 26 (52%) | 18 (36%) | 2 (4%) | 4 (8%) | 103 (67%) | 25 |
| Wiley Blackwell | 125 (14%) | 37 (30%) | 17 (46%) | 8 (22%) | 1 (3%) | 11 (30%) | 88 (70%) | 34 |
| Wolters Kluwer | 45 (5.1%) | 22 (49%) | 5 (23%) | 14 (63%) | 0 (0%) | 3 (14%) | 23 (51%) | 18 |
| Oxford University Press | 13 (1.5%) | 3 (23%) | 1 (33%) | 0 (0%) | 0 (0%) | 2 (67%) | 10 (77%) | NA |
| Termedia | 11 (1.2%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 11 (100%) | NA |
| Karger | 9 (1.0%) | 7 (78%) | 2 (29%) | 4 (57%) | 0 (0%) | 1 (14%) | 2 (22%) | 7 |
Note: The percentage in the “Informed papers” column is the percentage of the column total. The percentage in the “Case completion” and “Pending investigation” columns are the percentages that case completion and pending investigation represent of the “Informed papers” – these two percentages add up to 100%. The percentages in the remaining four columns (“Retraction”, “Expression of concern”, “Correction” and “Investigation concluded no action”) are percentages of the number of case completions and add up to 100%.
4. Discussion
4.1. Main Findings
In this series of almost 900 papers, we found delays in academic publishers' responses to flagged problematic papers. A decision was reached on less than a third of flagged cases over a median time of over 3 years, and only 13% was resolved within a year. About 85% of the decisions were retractions or EoC.
4.2. Strengths and Limitations
Our survey is large and up to date. The high rate of retractions and EoC among resolved cases suggests that our methods for identifying problematic papers are robust. Although we flagged > 900 potentially problematic papers, mostly in women's health, this probably represents only a small fraction of the problematic papers circulating in the literature if we assume a rate of 5%–10% of all research papers and 25% of RCTs [3]. Our estimates do not apply to predatory journals where it is often difficult to contact editors and publishers [18]. We did not assess papers published in known predatory journals.
4.3. Comparison With Previous Research
Our results echo previous studies' concerns about the extent of untrustworthy data in women's health and other disciplines [1]. Chambers et al. reported that article retractions within obstetrics and gynaecology were increasing, with the most frequently cited reasons being plagiarism, fabricated results, article duplication and compromised peer review [2]. Moylan and Kowalczuk studied retraction messages and concluded that checklists and templates for various classes of retraction notices would increase transparency in retraction messages [19]. A recent Nature article reported that the retraction rate for European‐based biomedical papers has quadrupled between 2000 and 2021, with the majority stemming from data and image manipulation [20]. This is partly due to whistle‐blowers flagging untrustworthy papers and the increased use of digital tools such as plagiarism detectors [20]. Retractions are only the tip of the iceberg of problematic papers.
We found that in > 85% of cases, our concerns were confirmed by retraction or EoC, which, in combination with the slow and often absent response, indicates that the number of retractions is not a marker for the true number of problematic studies. We do not think the high combined retraction/EoC rate is inflated, as a few straightforward cases were resolved quicker, while some remain unaddressed. Tables S3A,B describe 6 simple cases from authors of different countries and different journals where, in our opinion, at least an expression of concern should not take more than 2 weeks, but no action has been taken so far.
Others have also raised concerns about the ineffectiveness of the post‐publication assessment [21, 22]. Bolland et al. who reported similar slow response rates to us, proposed a new process for dealing with post‐publication assessment integrity involving the establishment of an independent panel that assesses publication integrity and transparently and timely report the outcomes of those assessments [21].
4.4. Interpretation
There is variation between journals. The European Journal of Contraception and Health Care (Taylor and Francis) concluded investigation of 19 papers within 2 years, resulting in 18 retractions [16]. Meanwhile, Contraception (Elsevier) did not solve any of their 11 flagged cases and may not have ever started an investigation. It is unlikely that authors send all their problematic papers to one journal and trustworthy research to others.
The delays we observed matter especially for RCTs, which are included in systematic reviews and meta‐analyses and, via them, determine recommendations in clinical guidelines. Untrustworthy RCT data harms patients, and delay in removing it allows the harm to persist. Recent guidelines on the use of foetal pillow during a Caesarean section were revised after retraction of the RCT, dominating the meta‐analysis 5 years after initial concerns had been raised [23, 24]. In another example, the effectiveness of progesterone for recurrent miscarriage has been called into question following the retractions of large RCTs demonstrating its effectiveness [25, 26, 27]. The American Society for Reproductive Medicine (ASRM) recently decided to exclude an RCT from their thyroid guideline, which reversed previous recommendations to prescribe levothyroxine supplementation in infertile women with subclinical hypothyroidism [28]. The involved journal had published a correction [29]. However, the ASRM practice committee decided to exclude the study. We did not assess this study in the series reported here, but another paper of the same author group received an EoC after we reported signs of data copying [30].
Our findings have implications for publishers. They have both a moral duty to speed up their processes and an interest in doing so. If patients are harmed by treatment based on problematic papers after the publisher has been alerted, publishers could be liable [31]. Delays also affect the public's trust in science. In 2023, a United Kingdom (UK) parliament House of Commons Committee recommended that publishers assess and provide a response within 2 months. This standard is not being met [32].
Publishers might consider the following innovations. Firstly, to use existing manuscript processing software to address post‐publication concerns. This would mean less risk of losing versions, datasets and correspondence than email. Fertility and Sterility have recently introduced an online portal where concerns about potentially problematic papers can be registered directly [33]. Editors should collaborate to review problematic authors who have published across multiple journals. We have published examples of how this might be done [13, 14, 34, 35]. Editors, reviewers and other stakeholders also need training in detecting problematic data because many delays and incorrect decisions are based on ignorance and denial [36]. For some publishers, patient safety and the trustworthiness of science might be of less importance than reputational or legal issues and profit.
Adjustments to the COPE guidelines might help. Current guidelines have no timelines and recommend that the authors' institute(s) should investigate, despite their inherent conflict of interest. Editors and publishers also have a conflict of interest, and a third party evaluating concerns systematically and timely is likely to be an improvement [19]. Issuing an early EoC following the journal's and publisher's initial investigation would allow potentially untrustworthy papers to be publicly flagged prevent the use of the study in guidelines and allow more time for a detailed investigation to be performed without the risk of harm to patients.
Of course, some of the measures proposed above require extra effort and resources, but the alternative—having fake data inform clinical practice—is, at least for us, not acceptable. Also, delayed response results in problematic papers informing meta‐analysis, which requires extra effort to repair [23]. BJOG itself initially refused to investigate an RCT on sildenafil for tocolysis, with the paper being retracted 21 months after the initial concern was raised [37]. This paper is now included in a meta‐analysis suggesting the benefit of sildenafil in pregnancy [38]. Similar to other meta‐analyses, one of which was retracted 5 years and 52 citations after it was published [39, 40]. In reality, sildenafil in pregnancy is not effective and maybe even harmful for the baby [41].
5. Conclusion
The publication of untrustworthy papers in scientific literature undermines the credibility and reliability of clinical practice and research. We have highlighted limitations in the current post‐publication review system for problematic papers with both inordinate delays and many investigations never taking place. We urge editors and publishers to work in unison and with reasonable timelines with a priority for patient safety to improve the current post‐publication review system and counteract the rise in false data in scientific literature.
Author Contributions
The project's aim was discussed and developed with the assistance of B.M.W. and J.T. All authors were involved in data interpretation and revision of the paper. B.W.M., J.T. and J.N. reviewed published papers. B.M.W. and J.T. corresponded with editors and publishers. S.S. screened, verified investigation outcomes, and prepared the manuscript. L.S.A. conducted the statistical analysis. All aspects of the project were conducted with the guidance of BMW.
Ethics Statement
No ethical approval was required as data was collected from publicly available information.
Conflicts of Interest
B.W.M. is supported by a National Health Medical Research Council (NHMRC) Practitioner Fellowship (GNT1082548). B.W.M. reports consultancy, travel support and research funding from Merck and consultancy for Organon and Norgine.
Supporting information
Table S1. Supporting Information.
Table S2. Supporting Information.
Data S1. Supporting Information.
Data S2. Supporting Information.
Acknowledgements
We would like to thank all editors and publishers who corresponded with the team. Open access publishing facilitated by Monash University, as part of the Wiley ‐ Monash University agreement via the Council of Australian University Librarians.
Funding: The authors received no specific funding for this work.
Data Availability Statement
An anonymous dataset is added to the manuscript.
References
- 1. Carlisle J. B., “False Individual Patient Data and Zombie Randomised Controlled Trials Submitted to Anaesthesia,” Anaesthesia 76 (2021): 472–479. [DOI] [PubMed] [Google Scholar]
- 2. Chambers L., Michener C., and Falcone T., “Plagiarism and Data Falsification Are the Most Common Reasons for Retracted Publications in Obstetrics and Gynaecology,” BJOG 126 (2019): 1134–1140. [DOI] [PubMed] [Google Scholar]
- 3. Ioannidis J. P. A., “Hundreds of Thousands of Zombie Randomised Trials Circulate Among Us,” Anaesthesia 76 (2021): 444–447. [DOI] [PubMed] [Google Scholar]
- 4. Saiz L. C., Erviti J., and Garjón J., “When Authors Lie, Readers Cry and Editors Sigh,” BMJ Evidence‐Based Medicine 23 (2018): 92–95. [DOI] [PubMed] [Google Scholar]
- 5. Weeks J., Cuthbert A., and Alfirevic Z., “Trustworthiness Assessment as an Inclusion Criterion for Systematic Reviews—What Is the Impact on Results?,” Cochrane Evidence Synthesis and Methods 1 (2023): e12037. [Google Scholar]
- 6. Mousa A., Flanagan M., Tay C. T., et al., “Research Integrity in Guidelines and evIDence Synthesis (RIGID): A Framework for Assessing Research Integrity in Guideline Development and Evidence Synthesis,” eClinicalMedicine 74 (2024): 102717, 10.1016/j.eclinm.2024.102717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Mol B. W. and Ioannidis J. P. A., “How Do We Increase the Trustworthiness of Medical Publications?,” Fertility and Sterility 120 (2023): 412–414. [DOI] [PubMed] [Google Scholar]
- 8. Committee on Publication Ethics , “About COPE,” 2023, https://publicationethics.org/about/our‐organisation.
- 9. COPE Council , “COPE Flowcharts and Infographics—Handling of Post‐Publication Critiques,” 2021, https://publicationethics.org/node/50816.
- 10. PubPeer , “PubPeer—The Online Journal Club,” 2024, https://blog.pubpeer.com/.
- 11. “The RetractionWatch Database,” 2018, Center for Scientific Integrity New York, http://retractiondatabase.org/RetractionSearch.aspx.
- 12. Mol B. W., Lai S., Rahim A., et al., “Checklist to Assess Trustworthiness in RAndomised Controlled Trials (TRACT Checklist): Concept Proposal and Pilot,” Research Integrity and Peer Review 8 (2023): 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Liu S., Singh N., and Mol B. W., “The Integrity of Seven Randomized Trials Evaluating Treatments for Premature Ejaculation,” Andrologia 54 (2022): e14573. [DOI] [PubMed] [Google Scholar]
- 14. Bordewijk E. M., Wang R., Askie L. M., et al., “Data Integrity of 35 Randomised Controlled Trials in women' Health,” European Journal of Obstetrics, Gynecology, and Reproductive Biology 249 (2020): 72–83. [DOI] [PubMed] [Google Scholar]
- 15. SCImago , “SJR—SCImago Journal & Country Rank,” 2024, http://www.scimagojr.com.
- 16. Roumen F., Grandi G., and Bitzer J., “Retraction of Peer‐Reviewed Articles, a Difficult but Crucial Choice: Our Experience From the European Journal of Contraception & Reproductive Health Care,” European Journal of Contraception & Reproductive Health Care 29 (2024): 83–84. [DOI] [PubMed] [Google Scholar]
- 17. Laban M., Abd Alhamid M., Ibrahim E. A., Elyan A., and Ibrahim A., “Endometrial Histopathology, Ovarian Changes and Bleeding Patterns Among Users of Long‐Acting Progestin‐Only Contraceptives in Egypt,” European Journal of Contraception & Reproductive Health Care 17 (2012): 451–457. [DOI] [PubMed] [Google Scholar]
- 18. Richtig G., Berger M., Köller M. C., et al., “Predatory Journals: Perception, Impact and Use of Beall's List by the Scientific Community‐A Bibliometric Big Data Study,” PLoS One 18 (2023): e0287547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Moylan E. C. and Kowalczuk M. K., “Why Articles Are Retracted: A Retrospective Cross‐Sectional Study of Retraction Notices at BioMed Central,” BMJ Open 6 (2016): e012047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Else H., “Biomedical Paper Retractions Have Quadrupled in 20 Years—Why?,” Nature 630 (2024): 280–281. [DOI] [PubMed] [Google Scholar]
- 21. Bolland M. J., Grey A., Avenell A., and Klein A., “Correcting the Scientific Record—A Broken System?,” Accountability in Research 28 (2021): 265–279. [DOI] [PubMed] [Google Scholar]
- 22. Williams A. C. C., Hearn L., Moore R. A., et al., “Effective Quality Control in the Medical Literature: Investigation and Retraction vs Inaction,” Journal of Clinical Epidemiology 157 (2023): 156–157. [DOI] [PubMed] [Google Scholar]
- 23.“Retraction: Management of Impacted Fetal Head at Caesarean Birth,” BJOG 131, no. 10 (2024): 1434. [DOI] [PubMed] [Google Scholar]
- 24. Seal S. L., Dey A., Barman S. C., Kamilya G., Mukherji J., and Onwude J. L., “Retraction: Randomized Controlled Trial of Elevation of the Fetal Head With a Fetal Pillow During Cesarean Delivery at Full Cervical Dilatation,” International Journal of Gynaecology and Obstetrics 162 (2023): 1129. [DOI] [PubMed] [Google Scholar]
- 25. Ismail A. M. A. A., Ali M. K., and Amin A. F., “Statement of Retraction: Peri‐Conceptional Progesterone Treatment in Women With Unexplained Recurrent Miscarriage: A Randomized Double‐Blind Placebo‐Controlled Trial,” Journal of Maternal‐Fetal and Neonatal Medicine 33 (2020): 1073. [DOI] [PubMed] [Google Scholar]
- 26. Kumar A. B. N., Prasad S., Aggarwal S., et al., “Retraction Notice to Oral Dydrogesterone Treatment During Early Pregnancy to Prevent Recurrent Pregnancy Loss and Its Role in Modulation of Cytokine Production: A Double‐Blind, Randomized, Parallel, Placebo‐Controlled Trial,” Fertility and Sterility 119 (2023): 518. [DOI] [PubMed] [Google Scholar]
- 27. Chong K., Li W., Roberts I., and Mol B. W., “Making Miscarriage Matter,” Lancet 398 (2021): 743–744. [DOI] [PubMed] [Google Scholar]
- 28. Medicine PCotASfR , “Subclinical Hypothyroidism in the Infertile Female Population: A Guideline,” Fertility and Sterility 104 (2015): 545–553. [DOI] [PubMed] [Google Scholar]
- 29. Abdel Rahman A. H., Aly Abbassy H., and Abbassy A. A., “Improved In Vitro Fertilization Outcomes After Treatment of Subclinical Hypothyroidism in Infertile Women,” Endocrine Practice 16 (2010): 792–797. [DOI] [PubMed] [Google Scholar]
- 30. Galal A. F. A. H., AbdElrahman A. H., Abdelhafez M. S., and Badawy A., “Expression of Concern—Accelerated Coasting Does Not Affect Oocyte Maturation,” Gynecologic and Obstetric Investigation 88 (2023): 391. [DOI] [PubMed] [Google Scholar]
- 31. Bolland M., Avenell A., and Grey A., “Publication Integrity: What Is It, Why Does It Matter, How It Is Safeguarded and How Could We Do Better?,” Journal of the Royal Society of New Zealand 55 (2024): 1–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Science, Innovation and Technology Committee , Reproducibility and Research Integrity (United Kingdom Parliment, 2023). [Google Scholar]
- 33. Fertility and Sterility , “Submission Form for Allegation of Breach in Data Integrity,” 2024, https://www.surveymonkey.com/r/academic_complaint.
- 34. Liu Y., Thornton J. G., Li W., van Wely M., and Mol B. W., “Concerns About Data Integrity of 22 Randomized Controlled Trials in Women's Health,” American Journal of Perinatology 40 (2023): 279–289. [DOI] [PubMed] [Google Scholar]
- 35. Bordewijk E. M., Li W., Gurrin L. C., Thornton J. G., van Wely M., and Mol B. W., “An Investigation of Seven Other Publications by the First Author of a Retracted Paper due to Doubts About Data Integrity,” European Journal of Obstetrics, Gynecology, and Reproductive Biology 261 (2021): 236–241. [DOI] [PubMed] [Google Scholar]
- 36. Wilkinson J., Heal C., Antoniou G. A., et al., “Assessing the Feasibility and Impact of Clinical Trial Trustworthiness Checks via an Application to Cochrane Reviews: Stage 2 of the INSPECT‐SR Project,” medRxiv (2024), 2024.2011.2025.24316905. [Google Scholar]
- 37. Maher M. A., Sayyed T. M., and El‐Khadry S. W., “Nifedipine Alone or Combined With Sildenafil Citrate for Management of Threatened Preterm Labour: A Randomised Trial,” BJOG 126 (2019): 729–735. [DOI] [PubMed] [Google Scholar]
- 38. Manouchehri E., Makvandi S., Razi M., Sahebari M., and Larki M., “Efficient Administration of a Combination of Nifedipine and Sildenafil Citrate Versus Only Nifedipine on Clinical Outcomes in Women With Threatened Preterm Labor: A Systematic Review and Meta‐Analysis,” BMC Pediatrics 24 (2024): 106. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 39.“Retraction: The Effects of Sildenafil in Maternal and Fetal Outcomes in Pregnancy: A Systematic Review and Meta‐Analysis,” PLoS One 19 (2024): e0310291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Rakhanova Y., Almawi W. Y., Aimagambetova G., and Riethmacher D., “The Effects of Sildenafil Citrate on Intrauterine Growth Restriction: A Systematic Review and Meta‐Analysis,” BMC Pregnancy and Childbirth 23 (2023): 409. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 41. Pels A., Ganzevoort W., Kenny L. C., et al., “Interventions Affecting the Nitric Oxide Pathway Versus Placebo or no Therapy for Fetal Growth Restriction in Pregnancy,” Cochrane Database of Systematic Reviews 7 (2023): Cd014498. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Supporting Information.
Table S2. Supporting Information.
Data S1. Supporting Information.
Data S2. Supporting Information.
Data Availability Statement
An anonymous dataset is added to the manuscript.


