Abstract
Background
As chronic obstructive pulmonary disease (COPD) progresses, it can limit physical activity, resulting in reduced mobility. And patients with COPD have a higher incidence of cognitive impairment compared to normal individuals. The aim of this study was to investigate the effect of breathing training in COPD patients’ gait and cognitive function by changing the body position.
Study Design and Methods
Eighty-three COPD patients were recruited and randomized (1:1:1) into a head-down strong abdominal breathing training group (HDBT), a head-down training group (HDT), and a strong abdominal breathing training group (BT). Cognitive function and gait performance were assessed after the intervention by comparing patients’ stride frequency, stride length, step speed and Montreal cognitive assessment(MoCA) scores.
Results
63 patients completed this study. After 12 weeks, in terms of Obstacle walking tasks gait, the HDBT group significantly improved the performance of COPD patients in obstacle walking tasks in terms of stride length (73.29±0.64, P<0.001), step speed (97.73±0.47, P=0.018) performance. At the same time, we also found that there was no statistical difference in the gait performance of each group in the walking task (P > 0.05). In terms of cognition, cognitive function scores(MoCA) were significantly higher in HDBT (P<0.001) HDT (P<0.001), and BT (P<0.001) compared to baseline.
Conclusion
A 12-week HDBT program elevated cognitive function and improved stride length and speed metrics during an obstacle walking task in patients with stable COPD.
Clinical Trial Registration Number
chictr2400080452(30/01/2024).
Keywords: chronic obstructive pulmonary disease, pulmonary rehabilitation, diaphragm, head-down position, gait analysis, dual-tasking
Plain Language Summary
Why was this study done?
People with chronic obstructive pulmonary disease (COPD) often face challenges with movement and thinking skills as their condition worsens. Everyday tasks like walking around obstacles can become harder, and memory or focus may decline. This study tested whether simple breathing exercises—with or without body position changes—could improve walking ability and cognitive health in people with stable COPD.
What did the researchers do?
Researchers divided 63 adults with COPD into three groups for 12 weeks:
Group 1 practiced breathing techniques while lying head-down.
Group 2 did head-down positioning without breathing exercises.
Group 3 did breathing exercises in normal sitting/standing positions.
They measured changes in walking patterns (step length, speed) during regular and obstacle-filled paths, along with cognitive tests.
What did they find?
The head-down breathing group showed clearer improvements in stepping over obstacles compared to other groups.
All groups scored better on cognitive tests after 12 weeks.
Simple walking (without obstacles) showed no meaningful changes across groups.
What do these results mean?
Combining breathing practice with position changes may help people with COPD move more confidently in challenging situations like avoiding tripping hazards. The cognitive improvements across all groups suggest breathing exercises alone—whether lying down or upright—could support brain health in COPD. While longer studies are needed, these non-drug approaches show potential for helping people stay active and mentally sharp. Patients and healthcare teams might consider adding similar exercises to daily routines.
Introduction
Chronic obstructive pulmonary disease (COPD) is a prevalent, preventable, and treatable condition marked by persistent airflow limitation. This disease primarily results from prolonged exposure to inhaled particles, such as cigarette smoke and environmental pollutants, in addition to genetic, developmental, and socio-environmental factors.1 In patients with COPD, as the disease progresses, the stiffness of neck and shoulder soft tissues and joints restricts the physical activities of patients, including head extension, high tension of neck muscles,2 thoracic kyphosis, and shoulder internal rotation, weakened contractile properties of diaphragm fibers, reduced elasticity, loss of serial sarcomeres, destruction of muscle fibrin, and morphological changes of diaphragm.3 The diaphragm plays a crucial role in sustaining normal ventilation. In patients with COPD, systemic inflammation and hyperinflation can result in mechanical alterations that impair diaphragmatic function.4 These alterations place the diaphragm in a mechanically disadvantaged position, diminishing its capacity to generate airflow and pressure, increasing the work of breathing, and ultimately leading to diaphragmatic fatigue.5 Furthermore, these alterations can lead to symptoms such as chest tightness, weakness of inspiratory muscles, elevated respiratory resistance, and dyspnea, and may also impair physical activity and walking capacity.6 At the same time, COPD patients are often accompanied by cognitive impairment, which is mostly manifested by impaired language learning, language processing, and language memory.7,8 Long-term hypoxemia and hypercapnia, as well as chronic inflammatory reactions, may damage brain function, resulting in memory loss, inattention, and executive dysfunction.9 Greater cognitive resources are required when performing cognitive tasks, resulting in increased blood flow to areas of high neural activity and a corresponding increase in oxygenated hemoglobin (O2Hb) content.10 At the same time, studies have shown that the stride time variability of COPD patients is greater.11 This will have a significant impact on the quality of life and independence of patients with COPD. In addition, chronic pain, activity limitations, and psychological stress can also exacerbate the risk of cognitive decline.12 Therefore, it is particularly important for COPD patients to improve diaphragm function and pay attention to cognitive function. In rehabilitation training, with the emergence of handstand equipment, more and more people use this equipment for rehabilitation treatment. A study on head-down training in older people shows that the change of body position has a certain impact on cerebral blood flow, and can also reduce the psychological pressure in older people’s life and improve their sleep quality.13 In another study on the magnitude of diaphragmatic excursion of head down to healthy people, it was concluded that healthy subjects obtained the best change of hemidiaphragm excursion by using a 30 °head down tilt position,14 and the application of breathing training could also improve the cognitive function of COPD patients.15 Therefore, the aim of this study was to investigate the effects of head-down strong abdominal breathing training on gait and cognitive performance in patients with stable COPD. So as to provide clinical treatment ideas for the rehabilitation of cognitive function and gait stability of COPD patients. We hypothesized that 12 weeks of head-down strong abdominal breathing training would improve cognitive function and gait performance in COPD patients.
Methods
Design
In this study, 83 COPD patients (GOLD I) were recruited and randomly assigned to head down strong abdominal breathing training group (HDBT), strong abdominal breathing training group (BT), head down training group (HDT) according to the ratio of 1:1:1. 20 people were lost during the training. All subjects received rehabilitation training (PR) for 60 min three times a week for 12 weeks.16,17 Cognitive function and gait performance were measured in each group before and after the experiment.
Subjects
The subjects of this study were patients with mild COPD at a stable stage. The recruitment information was publicized by posting posters in the community, on-site explanations, and WeChat group publicity. Those who are interested in participating in this experiment will be uniformly arranged to have lung function screening in the Department of Respiratory and Critical Care Medicine, Navy Anqing Hospital of the People’s Liberation Army. The screening personnel underwent specialized training by the chief physician of the respiratory department and adhered strictly to the established COPD diagnostic criteria during the screening process.
Inclusion Criteria
COPD was diagnosed according to the global onset of obstructive pulmonary disease (GOLD) criteria (1 s forced expiratory volume (FEV1) / forced vital capacity (FVC) <70%), aged between 55–80 years, and at GOLD stage I.
Exclusion Criteria
(1) During the first 4 weeks, there was one exacerbation of chronic obstructive pulmonary disease (COPD), a history of chronic heart failure, stroke, or other high-risk cardiopulmonary or orthopedic conditions, including critical aortic stenosis, early post-myocardial infarction, or lower limb amputation;
(2) Diagnosis of lung cancer or history of thoracoabdominal surgery;
(3) Body mass index (BMI) ≥ 30;
(4) Hospitalized patients;
(5) Visual or auditory impairments, as well as color vision deficiencies;
(6) Administered medications with the potential to induce cognitive impairment;
(7) Underwent pulmonary rehabilitation within the past 12 months, or is currently engaged in an exercise program;
Randomization and Concealment
Randomization
83 patients with COPD were recruited in this study. All participants were randomly divided into three groups. The random sequence generator (www.random.org) generated the sequence and sealed the sequence in an envelope before randomly distributing it to the participants. Then 83 subjects were divided into HDBT, HDT, and BT groups according to the ratio of 1:1:1.
Allocation Concealment
Place the distribution order in sealed opaque envelopes numbered sequentially. Open the corresponding envelope, and the subjects will conduct the test according to the order in the envelope.
Adherence and Adverse Events
Compliance
Throughout the intervention, patients were monitored weekly via telephone to assess their feelings, feedback, or suggestions regarding their participation in the study, with responses provided accordingly. Compliance with the trial protocol was also documented, including reasons for failure to meet inclusion criteria (eg, age, incorrect diagnosis, etc). Instances where subjects did not complete the assigned intervention, received alternative interventions, or did not adhere to the experimental protocol at all were also recorded. Additionally, the number of subjects who were unable to complete the trial and the associated reasons were documented.18 However, relevant data collected from patients who dropped out midway through the study were not included in the data analysis in the results section of the study.
Adverse Events
Any adverse events during the trial, including dizziness, headache, nausea, etc.
Experimental Plan
One week before the experiment, patients focused on learning head-down strong abdominal breathing exercises and training. This ensured that participants could perform the required actions correctly during the formal trial. During the formal experiment, the lung rehabilitation training of each group will be carried out according to the experimental operation requirements and the principle from easy to difficult. For the HDBT and HDT groups, subjects were asked to empty their bowels before each training session and to perform rehabilitation exercises at least 1 h after meals. The intervention consisted of 60-minute sessions, conducted three times a week. Each session included 5 minutes of warm-up adaptation exercises at the beginning, followed by 50 minutes of head-down strong abdominal breathing training or head-down training, and concluded with 5 minutes of stretching exercises for relaxation. For the group involving abdominal breathing (HDBT, BT), the respiratory rate should be set at 20–30 beats/min (metronome prompt) to maintain the target heart rate, and the target heart rate = (220 age) × (0.65–0.85).19–21 Head-down training (see Figure 1) was performed on an inverted and upside-down fitness device (Patent No. 201821510570.5) with an inclination angle of 0–30 °. For the BT group, strong abdominal breathing exercises were performed in a seated position. The intervention period is 12 weeks. During the intervention period, if patients cannot continue to carry out relevant training, they can appropriately increase the short rest. The cumulative exercise duration of a single intervention can reach the standard.
Figure 1.
Head down training.
HDBT
First, lay the patient’s body on the handstand trainer and present a tilted state with the head lower than the feet. The selection of tilt angle is based on the actual acceptability of patients and adjusted in the range of 0 ° −30 °. Secondly, strong abdominal breathing is breathing by consciously increasing the respiratory rate based on abdominal breathing. Finally, the handstand trainer was combined with strong abdominal breathing training.
Exercise plan (number of groups * duration * interval between groups): Week 1–2: 10 groups *3 minutes *2 minutes. Weeks 3–6: 5 groups *8 minutes *2 minutes. 7–12 weeks: 1 group *50 minutes. Among them, the frequency of each breath is 20–30 times/min (metronome prompt).
Motion sensation: when breathing in the abdominal style, you will feel the fluctuation of the abdomen. At the same time, because in the head-down state, you may feel the pressure on the chest when breathing in. In addition, to maintain the balance and stability of the body, this may cause fatigue or soreness in the muscles of the upper and lower limbs.
The main muscles involved: are the diaphragm, abdominal muscles (including rectus abdominis, external oblique abdominis, internal oblique abdominis, and transverse abdominis), intercostal muscles, and auxiliary respiratory muscles.
HDT
Place the patient’s body on the handstand trainer and tilt the head below the feet. The selection of tilt angle is based on the actual acceptability of patients and adjusted in the range of 0 ° −30 °.
Exercise plan (number of groups * duration * interval between groups): Week 1–2: 10 groups *3 minutes *2 minutes. Weeks 3–6: 5 groups *8 minutes *2 minutes. 7–12 weeks: 1 group *50 minutes.
Motion sensation: in the head-down state, you may feel the chest compression increase during inspiration. At the same time, in head-down training, the upper and lower limbs may need to provide additional support to maintain the balance and stability of the body. This may cause fatigue or soreness in the muscles of the upper and lower limbs.
Main muscles involved: mainly involving core muscles (abdominal muscles, back muscles), upper limb muscles (deltoid muscles, rotator cuff muscles, biceps brachii and triceps brachii), lower limb muscles (quadriceps femoris, triceps crus, etc), and other auxiliary muscle groups.
BT
With the body in a sitting position, place one hand (usually the right hand) near the belly button of the abdomen to sense the ups and downs of the abdomen. Inhale slowly and deeply through the nose, and feel the abdomen gradually bulge outward as you inhale. When inhaling, you should relax your shoulders and chest to avoid the interference of chest breathing. The inspiratory time can be controlled at about 2 seconds, and then exhale slowly and evenly through the mouth or nose, while feeling the abdomen gradually concave inward with exhalation. When exhaling, abdominal muscle contraction is applied to push the diaphragm upward, and all exhaust gases are discharged from the lungs. The exhalation time can be controlled at about 3 seconds to ensure that the time ratio of inspiration and expiration is 1:2 or 1:3.
Exercise plan (number of groups * training time): 1 group *50 minutes, lasting for 12 weeks
Motion sensation: the body gradually heats up, the breathing deepens and accelerates, and there is slight soreness in the legs and feet.
Main muscles involved: quadriceps of the thigh, triceps of the calf, gluteus maximus of the hip, etc.
Relevant Outcome Measures
Relevant outcome indicators were cognitive scores at baseline and after 12 weeks and gait during task walking. Patients’ gait performance was assessed by changes in stride frequency, stride length, and step speed during the task, while cognitive function was evaluated with the Montreal Cognitive Assessment (MoCA) under resting conditions.11 MoCA is one of the validated ways to evaluate cognitive function.22 It is important to identify the effectiveness of interventions.
Gait Test
The gait test in this study was tested through a walking task in two contexts. The first type of task is a walking task (walking back and forth three times on a 10-meter-long trail, a total of 30 meters10), and the second type of task is the obstacle walking task (walking back and forth three times on a 10-meter-long trail, a total of 30 meters, at the same time, barriers about 15cm high were placed on the center of the 10-meter-long trail).23 Gait speed is measured based on the walking speed during home activities. Measurement of gait indexes gait analysis system24 (sab-gait three-dimensional gait analyzer (Beijing Cyberfit Information Technology Co., Ltd). was used to record the patients’ stride frequency, stride length, and gait speed indexes during task walking at baseline and after 12 weeks of PR. To reduce experimenter-related bias, the same assessor tested all subjects and provided consistent instructions. Each subject performed each task twice: once for familiarization and once for assessment.
Safety
The safety of the head-down tilt and abdominal breathing protocols was validated through preliminary feasibility testing and subsequent protocol optimization. However, in order to prevent accidents from occurring, relevant safety arrangements were still made during the experiment implementation phase. Safety officers were designated to oversee and assist participants throughout the experiment. Any adverse events were promptly documented and reported to the principal investigator for evaluation of their relevance to the trial. In cases of serious adverse events, clinicians from the Navy Anqing Hospital of the Chinese People’s Liberation Army were notified to ensure timely medical intervention. For participants who withdrew from the study, the reasons for withdrawal were recorded in the case report form. Researchers made efforts to maintain contact with these participants and complete assessments when feasible, and the end-of-trial form was subsequently filled out.
Ethical Approval and Informed Consent
This experiment was carried out according to the declaration of Helsinki and was approved by the ethics committee of Anqing Normal University (No: anu2023002), and was approved by the Chinese clinical experiment registry (https://www.chictr.org.cn/ Clinical trial registration number: chictr2400080452) (30/01/2024).
All participants provided written informed consent after receiving a detailed explanation of the study procedures: low head tilt breathing exercises, abdominal exercises, and cognitive function assessment. The study protocol emphasized voluntary participation, unrestricted right to withdraw, pre-intervention health screening, and anonymous data storage. Risks were minimal (non-invasive methods, standardized protocols). Ethical compliance followed institutional guidelines and the Declaration of Helsinki.
Statistical Analysis
Sample Size Calculation
The determination of sample size is mainly based on the main outcome indicators of the test. G-Power software was used to conduct variance analysis to calculate the sample size, in which the effect size was 0.25, α was 0.05, and power was 0.8. The patients also needed to be divided into three groups, and the calculated sample size was 72. With an anticipated sample loss of 10%–15%, we aim to recruit 83 COPD patients for the study.
Statistical Methods
The data were statistically and analytically analyzed using SPSS v.27.0. Categorical data were expressed as counts (% and 95% confidence interval (CI)) and continuous data were expressed as mean (SD) or median (25th–75th percentile) according to the distribution. Normal Gaussian distributions of the data were verified using the Shapiro–Wilk test. In comparing the differences between the three groups of subjects after the intervention, ANOVA or non-parametric tests were used for comparison and analysis. When the ANOVA F ratio was significant (P<0.05), multiple comparisons were performed using the LSD significant difference post-procedure to determine paired differences. Following a statistically significant Kruskal–Wallis H-test, post hoc pairwise comparisons were performed using Mann–Whitney U-tests with Bonferroni correction to control for family-wise error rate. P<0.05 indicates statistical significance.
Results
Baseline Characteristic
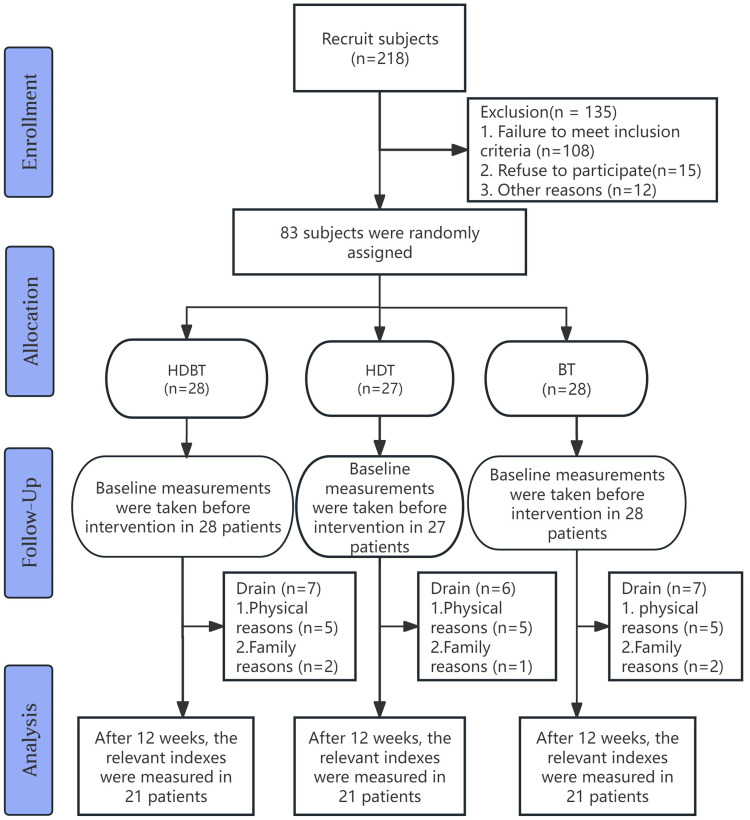
Among 218 screened patients, 83 met the inclusion criteria. Twenty participants withdrew during the study, yielding 63 completed cases (HDBT:21, HDT:21, BT:21). Baseline characteristics were balanced across groups (P>0.05; Table 1), and the study flowchart is provided in Figure 2.
Table 1.
Demographic and Baseline Characteristics
| Characteristics | HDBT (n=21) | HDT (n=21) | BT (n =21) | P value |
|---|---|---|---|---|
| Demographics | ||||
| Women, n,(%) | 16(76.2) | 14(66.7) | 14(66.7) | 0.74 |
| Age, years,±SD | 66.71±6.64 | 66.43±6.79x | 67.29±6.85 | 0.916 |
| BMI,kg /m2 | 26.56±1.00 | 26.65±1.06 | 25.94±1.13 | 0.069 |
| Pulmonary Function | ||||
| FVC,(L) | 2.39±0.45 | 2.21±0.43 | 2.25±0.41 | 0.345 |
| FEV1,(L) | 1.49±0.31 | 1.38±0.27 | 1.39±0.289 | 0.401 |
| FEV1/FVC (%) | 62.16±3.16 | 62.79±2.25 | 61.63±3.28 | 0.445 |
| Blood Gas Parameters | ||||
| PO2,mmHg | 75.49±1.14 | 75.75±1.22 | 75.60±1.22 | 0.776 |
| PCO2,mmHG | 50.82±2.26 | 51.15±2.42 | 50.70±2.61 | 0.826 |
| Gait performance | ||||
| Stride frequency,(walking task) | 1.40±0.11 | 1.37±0.11 | 1.38±0.11 | 0.721 |
| Stride length,(cm, walking task) | 73.42±3.18 | 72.91±3.06 | 73.74±3.01 | 0.683 |
| Step speed,(cm/s, walking task) | 102.51±4.81 | 100.06±7.16 | 101.37±7.17 | 0.475 |
| Stride frequency,(obstacle walking task) | 1.36±0.08 | 1.35±0.07 | 1.36±0.07 | 0.742 |
| Stride length,(cm, obstacle walking task) | 70.12±2.99 | 71.11±2.55 | 70.83±2.70 | 0.487 |
| Step speed,(cm/s, obstacle walking task) | 95.40±4.04 | 95.70±3.04 | 96.40±2.68 | 0.611 |
| Cognitive function scores | ||||
| MoCA,score | 18(18–19) | 19(18–20) | 19(19–19) | 0.85 |
Notes: Data are presented as percentage, mean ± SD, or median (interquartile range) unless otherwise indicated.BMI=body-mass index. FEV1=forced expiratory volume in 1 s. FVC=forced vital capacity.PCO2=partial pressure of carbon dioxide. Level of significance was set at P <0.05.
Abbreviation: PO2, partial pressure of oxygen.
Figure 2.
Experimental flow chart.
Gait Performance After 12 weeks of Intervention Training
After 12 weeks of the PR program. Comparisons between the three groups showed significant between-group differences in stride frequency (p=0.017), stride length (p=0.003), and step speed (p=0.033) during the obstacle walking task. In the walking task, there was no significant difference in the comparison between the three groups (Table 2). Specifically, in the stride frequency metric for obstacle walking. HDBT demonstrated significantly higher frequency compared to BT (p=0.005), but no differences were observed between HDBT vs HDT (p=0.19) or HDT vs BT (p=0.112). In the stride length indicator for obstacle walking, HDBT showed greater stride length than both HDT (p=0.002) and BT (p=0.005), whereas HDT and BT did not differ (p=0.82). In the step speed indicator for obstacle walking, HDBT exhibited faster speed compared to HDT (p=0.015), and HDT was slower than BT (p=0.041), but HDBT vs BT showed no difference (p=0.682). The comparison of gait performance before and after PR showed that there was no statistical difference in all gait indicators of the three groups during the walking task. In the obstacle walking task, the HDBT group concentrated on the pre-post comparison of stride length and step speed, and there were statistical differences. In the HDT group, there were statistically significant differences in step speed (P = 0.049). In contrast, stride length (p=0.01) and step speed (p=0.043) in obstacle walking task in the BT group showed similar significant differences as in the HDBT group. Unfortunately, By comparing before and after the intervention we found that there was no statistically significant difference between the three groups in terms of stride frequency, stride length and step speed in the walking task (P>0.05). See Table 3 for details.
Table 2.
Gait and Cognitive Index Characteristics Among Groups After 12 weeks
| Characteristics | HDBT (n=21) | HDT (n=21) | BT (n =21) | P value |
|---|---|---|---|---|
| Gait performance | ||||
| Stride frequency,(walking task) | 1.40±0.04 | 1.39±0.04 | 1.37±0.05 | 0.166 |
| Stride length,(cm, walking task) | 73.67±1.10 | 73.38±1.08 | 74.24±1.21 | 0.051 |
| Step speed,(cm/s, walking task) | 103.01±2.79 | 102.29±2.99 | 101.97±2.60 | 0.470 |
| Stride frequency,(obstacle walking task) | 1.33±0.01 | 1.34±0.02 | 1.35±0.01 | 0.017*a |
| Stride length,(cm, obstacle walking task) | 73.29±0.64 | 72.42±1.06 | 72.49±0.89 | 0.003**b |
| Step speed,(cm/s, obstacle walking task) | 97.73±0.47 | 97.00±1.08 | 97.61±1.13 | 0.033*c |
| Cognitive function scores | ||||
| MoCA,score | 24(23–24) | 23(22.5–23) | 23(22–23) | 0.015*d |
Notes: Data are presented as percentage, mean±SD, or median (interquartile range) unless otherwise indicated.Level of significance was set at P <0.05.*p < 0.05, **p < 0.01. a HDBT vs HDT: P=0.19; HDBT vs BT: P=0.005**; HDT vs BT: P=0.112. b HDBT vs HDT: P=0.002**; HDBT vs BT: P=0.005**; HDT vs BT: P= 0.82. c HDBT vs HDT: P=0.015*; HDBT vs BT: P=0.682; HDT vs BT: P=0.041*. d HDBT vs HDT: P=0.048*; HDBT vs BT: P= 0.028*; HDT vs BT: P=0.851.
Table 3.
Gait and Cognitive Index Characteristics Between Groups Before and After PR
| Characteristics | HDBT (n=21) | P value | HDT (n=21) | P value | BT (n =21) | P value | |||
|---|---|---|---|---|---|---|---|---|---|
| Before PR | After PR | Before PR | After PR | Before PR | After PR | ||||
| Gait performance | |||||||||
| Stride frequency,(walking task) | 1.40±0.11 | 1.40±0.04 | 0.953 | 1.37±0.11 | 1.39±0.04 | 0.471 | 1.38±0.11 | 1.37±0.05 | 0.922 |
| Stride length,(cm, walking task) | 73.42±3.18 | 73.67±1.10 | 0.734 | 72.91±3.06 | 73.38±1.08 | 0.482 | 73.74±3.01 | 74.24±1.21 | 0.464 |
| Step speed,(cm/s, walking task) | 102.51±4.81 | 103.01±2.79 | 0.708 | 100.06±7.16 | 102.29±2.99 | 0.211 | 101.37±7.17 | 101.97±2.60 | 0.761 |
| Stride frequency,(obstacle walking task) | 1.36±0.08 | 1.33±0.01 | 0.087 | 1.35±0.07 | 1.34±0.02 | 0.622 | 1.36±0.07 | 1.35±0.01 | 0.292 |
| Stride length,(cm, obstacle walking task) | 70.12±2.99 | 73.29±0.64 | <0.001** | 71.11±2.55 | 72.42±1.06 | 0.057 | 70.83±2.70 | 72.49±0.89 | 0.010* |
| Step speed,(cm/s, obstacle walking task) | 95.40±4.04 | 97.73±0.47 | 0.018* | 95.70±3.04 | 97.00±1.08 | 0.049* | 96.40±2.68 | 97.61±1.13 | 0.043* |
| Cognitive function scores | |||||||||
| MoCA,score | 18(18–19) | 24(23–24) | <0.001** | 19(18–20) | 23(22.5–23) | <0.001** | 19(19–19) | 23(22–23) | <0.001** |
Notes: Data are presented as percentage, mean±SD, or median (interquartile range) unless otherwise indicated.Level of significance was set at P <0.05.*p < 0.05, **p < 0.01.
Assessment of Cognitive Function Before and After Intervention
To further prove the impact of our PR scheme on the cognitive function of COPD patients, We combined patients’ MoCA scores before and after the intervention to assess changes in cognitive functioning. As can be seen in Table 1, there was no statistical difference between the three groups in terms of cognitive function scores at baseline. As can be concluded from Table 2, after 12 weeks of intervention, the difference between the three groups was statistically significant (p < 0.05). Specifically, HDBT had higher cognitive scores than HDT (p=0.048) and BT (p=0.028), while HDT and BT were comparable (p=0.851). Table 3 reveals the changes in cognitive function scores of the three groups in terms of pre- and post-PR comparisons. Cognitive functioning improved significantly in all three groups after the intervention (P<0.001).
Discuss
Our preliminary study based on the rehabilitation effect of head-down abdominal breathing training on COPD patients showed that the 12-week HDBT program elevated cognitive function and improved stride length and speed metrics during an obstacle walking task in patients with stable COPD. This is almost consistent with our research hypothesis. In addition, head-down strong abdominal breathing training did not significantly change the gait of a walking task.
The emergence of this result may improve cognitive tasks and gait performance through synergistic mechanisms. First, head-down tilt improves cerebral hypoxia through gravitational blood redistribution25 and supports prefrontal lobe function; second, abdominal breathing optimizes oxygen delivery by increasing diaphragmatic efficiency26 and alleviates hypoxemia during dual-task walking; and then the tilt angle (0°-30°) stimulates vestibular-intuitional adaptations and improves dynamic balance.27
Head-down strong abdominal breathing training is very rare in the field of rehabilitation. Of course, our conception of this scheme is not without theoretical basis. First of all, head-down training is a kind of resistance training against gravity. As a medical exercise rehabilitation treatment method, it first appeared in 1694 and is mainly used for clinical diagnosis, treatment, intervention measures, and other research.28 Head-down training has also been involved in recent studies. For example, two weeks of head-down training can effectively protect the cardiopulmonary health of adults aged 55–65 years,29 and head-down training can significantly improve cerebral blood flow30 and cerebral oxygenation.31 Abdominal breathing is one of the ways of breathing, and its effects on lung function26 and cognitive function32 have been confirmed. The head-down tilt angle in this trial was determined through a dual evidence-based approach, synthesizing maximal safe angles reported in healthy populations (up to −30°14 and −90°13 under controlled conditions) and down-regulating these thresholds according to the compromised cardiopulmonary reserve characteristic of elderly COPD patients. A dynamically adjusted protocol (0°to −30°) was implemented to balance intervention efficacy with respiratory mechanics tolerance, incorporating real-time biofeedback to individualize angle progression.
In this study, the exploration of gait is mainly analyzed from the frequency, stride length, and step speed. Due to the lack of direct comparison with previous studies on gait in head-down training. We only analyze from the perspective of abdominal breathing. When we increase the frequency of abdominal breathing, we also change some indicators of gait, which is consistent with Tassani et al’s study on abdominal breathing.33 However, the analysis of gait under tasks should also be comprehensively analyzed in combination with gait variability, step width,11,34 and the interaction of head down and strong abdominal breathing. In future exploration, more indicators can be combined to comprehensively explore and analyze the deep-seated mechanism. In addition, the gait stability of COPD patients is correlated with the occurrence of fall risk, and the study found that COPD patients are 55% more likely to fall than non-COPD patients.35 There is a close link between COPD disease itself and fall risk. This also shows that our study on the gait of COPD patients has certain practical significance.
In terms of cognitive function, the MoCA was chosen in this study as a single measure of cognitive function in COPD patients, and its applicability needs to be discussed objectively. The MoCA covers multidimensional cognitive domains (executive function, visuospatial, etc). and has significant sensitivity in identifying cognitive function,36,37 and its scores are negatively correlated with pulmonary function parameters (eg, FEV₁%pred), supporting clinical correlation validity. However, MoCA has limited assessment of complex attention and language function, and education dependence may introduce bias in older, less educated populations. Nonetheless, its brevity (<10 min) significantly reduces the testing burden in patients with respiratory limitation, and there are similar studies exploring the status of altered cognitive functioning in patients by applying MoCA only.38,39 Although multimodal assessments (eg, neuroimaging) can improve accuracy, MoCA remains a reasonable choice for the trade-off between validity and practicability based on the feasibility and patient tolerability goals of this study. The results of this study showed the efficacy of head-down strong abdominal breathing exercise in significantly improving cognitive function in COPD patients. However, this result is inconsistent with the conclusion of BASNER et al. Head-down bed rest for 30–60 days in pilots does not improve the cognitive function of patients.40 This may be explained by the correlation with the subject population and the addition of breathing exercises in this study. Another study found no significant improvement in step speed or stride time variability during dual-task performance following pulmonary rehabilitation.11 Considering that cognitive task performance can be detrimental to patients with COPD and that there are currently no definitive pulmonary rehabilitation measures to improve this, it is important to explore in greater depth the implementation options to identify measures to improve cognitive performance in these patients. Therefore, implementation programs should be explored in greater depth to identify rehabilitation measures to improve cognitive performance in these patients.
Although the curative effect of abdominal breathing has been generally recognized, we want to appropriately increase the respiratory rate (respiratory rate is 20–30 times/min, metronome prompt) based on routine training intensity, and continuously and strongly intervene in the respiratory muscles, to explore the curative effect of this way. The final result is consistent with our expectations. Through this PR program, COPD patients also gained benefits under strong abdominal breathing exercises. However, it needs to be considered whether this training method of increasing respiratory rate and changing body position applies in patients with moderate to severe COPD, which still lacks theoretical and practical basis. The condition of moderate and severe patients is more serious, which can be further explored by reducing the intensity of head-down strong abdominal breathing exercises in the future.
We specially selected patients with mild COPD in the stable stage, because our rehabilitation program was applied to COPD patients for the first time, taking into account the possible adverse reactions. Such as increased heart rate, psychological discomfort, dizziness, and nausea.13,41 However, one case of adverse event was still reported during the experimental implementation phase. One subject complained of significant dizziness on day 2 of formal training, which was determined to be an intervention-related adverse event (AE). The research team immediately initiated a contingency plan to complete a hemodynamic assessment (blood pressure: 112/74 mmHg; heart rate: 88 bpm) and systematic cause screening. A structured interview revealed that the patient had a history of severe sleep deprivation the night before (stayed up until 3 am). Taking into account the recommendations of the GOLD Guidelines on the safety of pulmonary rehabilitation, the team adopted a dual-track intervention strategy: ① strengthened health behavioral education, explicitly requiring the patient to maintain a regular work and rest schedule; ② adjusted the training program to reduce the angle of the head-down tilt from 15 ° to 8 ° in order to reduce the risk of postural hypotension. After the above interventions, the patient successfully completed the subsequent rehabilitation program and no further AE was reported.
Home pulmonary rehabilitation has been selected and recognized by many patients for its characteristics of providing flexibility,42 promoting self-management,43 and personalized rehabilitation plans.44,45 HDBT can also be used as a practical family therapy, which is very suitable for people who find it inconvenient to travel or have limited time to recover in the outpatient clinic. Although supervised pulmonary rehabilitation programs provide a lot of help to solve the common symptoms of COPD, they are not suitable for office workers with limited rehabilitation time and patients who often stay indoors. These data support the role of HDBT in managing those who are unable or unwilling to participate in such programs. In addition, the cost of head down training device is relatively cheap compared with other professional rehabilitation devices, which is easy to implement in the general population.
There are still some deficiencies in our study. First, the proportion of men and women is not balanced enough. This is because this study, as a preliminary study, chose the intervention study in the community considering the acceptance of patients, adverse reactions, attrition rate, etc. There are many uncontrollable factors in the community, such as health concepts, living habits, etc. There is a certain degree of imbalance in the proportion of men and women who finally participate in this study. Future research can be further studied by balancing the gender ratio. Secondly, the research on gait is not deep enough. For example, further analysis of the gait cycle, range of joint motion, and gait variability can make the evidence of relevant results more powerful. Finally, the measurement of cognitive function can be further explored in combination with fMRI,46 fNIRS,47 and other instruments.
Conclusion
A 12-week HDBT program elevated cognitive function and improved stride length and speed metrics during an obstacle walking task in patients with stable COPD. These data support the role of HDBT in managing patients who are unable or unwilling to participate in current pulmonary rehabilitation programs. The duration of the effect and the impact on health status need further follow-up investigation.
Acknowledgments
We thank Prof. Mingyun Sun for suggesting the idea of this study. We thank Dr. Ruichen Jiang, Prof. Mingyun Sun, and Prof. Heping Xiang for funding this study. We would like to thank Professor Ming Yun Sun for suggesting the idea of this research.
Funding Statement
This study was funded by the Professor’s Fund, Anqing Normal University, China (No. 81902307), the Natural Science Research Program for Higher Education Institutions in Anhui Province (KJ2021A0653), and the University Outstanding Youth Research Project of Anhui Province (Social Sciences) (No. 2022AH030101).
Data Sharing Statement
Due to data privacy regulations, individual participant data collected in this study will not be publicly accessible. However, data can be obtained from the corresponding author on reasonable request (Mingyun Sun).
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that the study was conducted in the absence of any business or financial relationship that could be interpreted as a potential conflict of interest.
References
- 1.Kahnert K, Jörres RA, Behr J. et al. The diagnosis and treatment of COPD and its comorbidities. Dtsch Arztebl Int. 2023;120(25):434–444. doi: 10.3238/arztebl.m2023.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boontha N, Chen S, Lin JJ. Impairment of scapular control in individuals with chronic obstructive pulmonary disease (COPD): systematic review and meta-analysis. Physiother Theory Pract. 2023;39(9):1816–1831. doi: 10.1080/09593985.2022.2060885 [DOI] [PubMed] [Google Scholar]
- 3.Chun EM, Han SJ, Modi HN. Analysis of diaphragmatic movement before and after pulmonary rehabilitation using fluoroscopy imaging in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2015;10:193–199. doi: 10.2147/COPD.S74438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hua-Rong Z, Liang C, Rong L, et al. Ultrasonographic evaluation of diaphragm function in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. Medicine. 2022;101(51):e32560. doi: 10.1097/MD.0000000000032560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhang J, Zhang C, Yan L, et al. Shear wave elastography of the diaphragm in acute exacerbation of chronic obstructive pulmonary disease: a prospective observational study. Medicine. 2023;102(11):e33329. doi: 10.1097/MD.0000000000033329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shimada A, Kawata N, Sato H, et al. Dynamic quantitative magnetic resonance imaging assessment of areas of the lung during free-breathing of patients with chronic obstructive pulmonary disease. Acad Radiol. 2022;29 Suppl 2:S215–s225. doi: 10.1016/j.acra.2021.03.034 [DOI] [PubMed] [Google Scholar]
- 7.Chang SS, Chen S, Mcavay GJ, et al. Effect of coexisting chronic obstructive pulmonary disease and cognitive impairment on health outcomes in older adults. J Am Geriatr Soc. 2012;60(10):1839–1846. doi: 10.1111/j.1532-5415.2012.04171.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jianhua Wang, Guifang Hu, Wang L. Factors associated with chronic obstructive pulmonary disease combined with respiratory failure affecting sleep and cognitive function and its mechanism. J Lung Dis. 2017;10(06):713–717. [Google Scholar]
- 9.Cleutjens FA, Janssen DJ, Ponds RW, et al. Cognitive-pulmonary disease. Biomed Res Int. 2014;2014:697825. doi: 10.1155/2014/697825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hassan SA, Campos MA, Kasawara KT, et al. Changes in oxyhemoglobin concentration in the prefrontal cortex during cognitive-motor dual tasks in people with chronic obstructive pulmonary disease. COPD. 2020;17(3):289–296. doi: 10.1080/15412555.2020.1767561 [DOI] [PubMed] [Google Scholar]
- 11.Heraud N, Alexandre F, Gueugnon M, et al. Impact of chronic obstructive pulmonary disease on cognitive and motor performances in dual-task WALKING. COPD. 2018;15(3):277–282. doi: 10.1080/15412555.2018.1469607 [DOI] [PubMed] [Google Scholar]
- 12.Iacobucci GJ, Visnjevac O, POURAFKARI L, et al. Ketamine: an update on cellular and subcellular mechanisms with implications for clinical practice. Pain Physician. 2017;20(2):E285–e301. [PubMed] [Google Scholar]
- 13.Zhangjie GUO, Peng LIU, Hongmei YANG, et al. Effects of head down tilt training on carotid blood flow. Psychol Stress Anxiety Sensitivity Elderly. 2022;25(Supplement_1):A83–A84. [Google Scholar]
- 14.Schewitz J, Roos R, Aswegen HVAN, et al. The effect of two passive head-down tilt positions on diaphragm excursion in healthy adults: a preliminary study. Physiother Theory Pract. 2016;32(3):223–231. doi: 10.3109/09593985.2015.1137664 [DOI] [PubMed] [Google Scholar]
- 15.Lu Y, Li P, Li N, et al. Effects of home-based breathing exercises in subjects with COPD. Respir Care. 2020;65(3):377–387. doi: 10.4187/respcare.07121 [DOI] [PubMed] [Google Scholar]
- 16.Cirak Yburan, Yelvar GDYILMAZ, Elbasi NDURUSTKAN. Effectiveness of 12-week inspiratory muscle training with manual therapy in patients with COPD: a randomized controlled study. Clin Respir J. 2022;16(4):317–328. doi: 10.1111/crj.13486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Demeyer H, Louvaris Z, FREI A, et al. Physical activity is increased by a 12-week semiautomated telecoaching programme in patients with COPD: a multicentre randomised controlled trial. Thorax. 2017;72(5):415–423. doi: 10.1136/thoraxjnl-2016-209026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Song F, Ding K, SUN M, et al. Effect of 12-week head-down strong abdominal breathing on cognitive function in patients with stable chronic obstructive pulmonary disease: a single-centre randomised controlled trial protocol. Trials. 2024;25(1):351. doi: 10.1186/s13063-024-08193-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Probst VS, Troosters T, Pitta F, et al. Cardiopulmonary stress during exercise training in patients with COPD. Eur Respir J. 2006;27(6):1110–1118. doi: 10.1183/09031936.06.00110605 [DOI] [PubMed] [Google Scholar]
- 20.Shookster D, Lindsey B, Cortes N, et al. Accuracy of commonly used age-predicted maximal heart rate equations. Int J Exerc Sci. 2020;13(7):1242–1250. doi: 10.70252/XFSJ6815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tanaka H, Monahan KD, Seals DR. Age-predicted maximal heart rate revisited. J Am Coll Cardiol. 2001;37(1):153–156. doi: 10.1016/S0735-1097(00)01054-8 [DOI] [PubMed] [Google Scholar]
- 22.Li F, Harmer P, ECKSTROM E, et al. Clinical effectiveness of cognitively enhanced tai ji quan training on global cognition and dual-task performance during walking in older adults with mild cognitive impairment or self-reported memory concerns: a randomized controlled trial. Ann Intern Med. 2023;176(11):1498–1507. doi: 10.7326/M23-1603 [DOI] [PubMed] [Google Scholar]
- 23.Zhenlan L. A Study on the Effect of Exercise Intervention on Gait in Mild-to-Moderate Parkinson’s Disease Patients During Different Task Activities. Shanghai Institute of Physical Education; 2021. D. doi: 10.27315/d.cnki.gstyx.2021.000037 [DOI] [Google Scholar]
- 24.Lina WANG, Zhiqin CHEN, Wenmin DONG. Effect of lower limb rehabilitation robot training combined with mirror therapy on gait control in hemiplegic patients with stroke. J Naval Med. 2023;44(11):1163–1167. [Google Scholar]
- 25.Ogoh S, Sato K, ABREU SDE, et al. Arterial and venous cerebral blood flow responses to long-term head-down bed rest in male volunteers. Exp Physiol. 2020;105(1):44–52. doi: 10.1113/EP088057 [DOI] [PubMed] [Google Scholar]
- 26.Abdullahi A, Wong TW, Ng SS. Efficacy of diaphragmatic breathing exercise on respiratory, cognitive, and motor function outcomes in patients with stroke: a systematic review and meta-analysis. Front Neurol. 2023;14:1233408. doi: 10.3389/fneur.2023.1233408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Diaz-Artiles Ana, Karmali Faisal. Vestibular precision at the level of perception, eye movements, posture, and neurons. Neuroscience. 2021;468:282–320. doi: 10.1016/j.neuroscience.2021.05.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Papadakis M, Trompoukis C. Surgery in antiquity: the origin of the trendelenburg position revisited. Acta Chir Belg. 2021;121(3):222–223. doi: 10.1080/00015458.2020.1865622 [DOI] [PubMed] [Google Scholar]
- 29.Hedge ET, Mastrandrea CJ, HUGHSON RL. Loss of cardiorespiratory fitness and its recovery following two weeks of head-down bed rest and the protective effects of exercise in 55- to 65-yr-old adults. J Appl Physiol. 2023;134(4):1022–1031. doi: 10.1152/japplphysiol.00726.2022 [DOI] [PubMed] [Google Scholar]
- 30.Zhao ZA, Zhang NN, Tao L, et al. Effect of head-down tilt on clinical outcome and cerebral perfusion in ischemic stroke patients: a case series. Front Neurol. 2022;13:992885. doi: 10.3389/fneur.2022.992885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mekari S, Murphy RJL, Mackinnon ARS, et al. The impact of a short-period head-down tilt on executive function in younger adults. Sci Rep. 2022;12(1):20888. doi: 10.1038/s41598-022-25123-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ma X, Yue ZQ, Gong ZQ, et al. The effect of diaphragmatic breathing on attention, negative affect and stress in healthy adults. Front Psychol. 2017;8:874. doi: 10.3389/fpsyg.2017.00874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tassani S, Chaves P, Beardsley M, et al. Breathing, postural stability, and psychological health: a study to explore triangular links. Front Bioeng Biotechnol. 2024;12:1347939. doi: 10.3389/fbioe.2024.1347939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Matsuura T, Sakashita K, Grushnikov A, et al. Statistical analysis of dual-task gait characteristics for cognitive score estimation. Sci Rep. 2019;9(1):19927. doi: 10.1038/s41598-019-56485-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hakamy ALI, Bolton Charlottee, Gibson Jacke, et al. Risk of fall in patients with COPD. Thorax. 2018;73(11):1079–1080. doi: 10.1136/thoraxjnl-2017-211008 [DOI] [PubMed] [Google Scholar]
- 36.Kang JM, Cho YS, Park S, et al. Montreal cognitive assessment reflects cognitive reserve. BMC Geriatr. 2018;18(1):261. doi: 10.1186/s12877-018-0951-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nasreddine ZS, phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, moca: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699. doi: 10.1111/j.1532-5415.2005.53221.x [DOI] [PubMed] [Google Scholar]
- 38.Frain JA, Chen L. Examining the effectiveness of a cognitive intervention to improve cognitive function in a population of older adults living with hiv: a pilot study. Ther Adv Infect Dis. 2018;5(1):19–28. doi: 10.1177/2049936117736456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ye B, Wei D, Pan L. Montreal cognitive assessment of cognitive dysfunction after basal ganglia stroke. Acta Neurol Belg. 2022;122(4):881–884. doi: 10.1007/s13760-022-01967-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Basner M, Stahn AC, Nasrini J, et al. Effects of head-down tilt bed rest plus elevated CO 2 on cognitive performance. J Appl Physiol. 2021;130(4):1235–1246. doi: 10.1152/japplphysiol.00865.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Davis JE, Horwood KE, Dejong GK. Effects of exercise during head-down bed rest on postural control. Aviat Space Environ Med. 1997;68(5):392–395. [PubMed] [Google Scholar]
- 42.Rachel J, PHYSIOTHERAPY GARROD. The pros and cons of pulmonary rehabilitation at home. Physiotherapy. 1998;84(12):603–607. [Google Scholar]
- 43.Holland AE, Mahal A, Hill CJ, et al. Benefits and Costs of Home-Based Pulmonary Rehabilitation in Chronic Obstructive Pulmonary Disease - a Multi-Centre Randomised Controlled Equivalence Trial. Vol. 13. BMC Pulm Med; 2013:57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Resqueti Vregiane, Gorostiza A, Gáldiz JB, et al. Benefits of a home-based pulmonary rehabilitation program for patients with severe chronic obstructive pulmonary disease][J. Arch Bronconeumol. 2007;43(11):599–604. doi: 10.1157/13111345 [DOI] [PubMed] [Google Scholar]
- 45.Spencer LM, Alison JA, Mckeough ZJ. Maintaining benefits following pulmonary rehabilitation: a randomised controlled trial. Eur Respir J. 2010;35(3):571–577. doi: 10.1183/09031936.00073609 [DOI] [PubMed] [Google Scholar]
- 46.Zhu C, Liao Y, Ding Y, et al. Functional magnetic resonance imaging is used to evaluate memory, language, and cognitive function in patients with medication-resistant temporal lobe epidemic. Minerva Surg. 2022;77(5):506–508. doi: 10.23736/S2724-5691.21.09267-4 [DOI] [PubMed] [Google Scholar]
- 47.Li Y, Li X, Zhaung W, et al. Relationship between cognitive function and brain activation in major depressive disorder patients with and without insomnia: a functional near-infrared spectroscopy (Fnirs) study. J Psychiatr Res. 2024;169:134–141. doi: 10.1016/j.jpsychires.2023.11.002 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Due to data privacy regulations, individual participant data collected in this study will not be publicly accessible. However, data can be obtained from the corresponding author on reasonable request (Mingyun Sun).