Abstract
Background
Isavuconazole is a recent broad-spectrum triazole indicated for the treatment of invasive aspergillosis and mucormycosis when amphotericin B is inappropriate. However, limited information exists on its clinical use.
Objective
We set up a retrospective multicentre study to describe the clinical practice of isavuconazole including indications, exposure, and hepatic safety.
Methods
From January 2021 to June 2023, all patients who received isavuconazole and had at least one therapeutic drug monitoring (TDM) measurement, were included. To identify independent predictors of isavuconazole trough concentrations (Cmin), linear regression analyses were performed. Causal relationship between the occurrence of liver injury and isavuconazole was also analysed.
Results
Most of the included patients (n = 102) were admitted into haematology units (41.1% [n = 42]) or intensive care units (ICU) (30.4% [n = 31]). Aspergillosis (47.0% [n = 48]), mucormycosis (25.6% [n = 26]), and off-label empirical treatments (18.6% [n = 19]), were the three most common indications. About half of the patients (46.1% [n = 47]) had an optimal exposure, while 42.2% (n = 43) were underexposed, and 11.7% (n = 12) were overexposed. Albumin level on the day of TDM was a significant factor associated with an increase in isavuconazole Cmin (p = 0.010). Among the 11 patients who had liver test abnormalities, isavuconazole was discontinued in six (n = 6) patients and liver injury was attributable to isavuconazole in two (n = 2) patients.
Conclusions
This multicentre analysis highlighted the common use of isavuconazole as an off-label indication, as well as the frequent underexposure of patients to isavuconazole. Albumin on the day of TDM appeared to be an important factor driving isavuconazole exposure, especially in ICU patients.
Key Points
| In this study of isavuconazole real-life use performed in 102 patients, off-label use of the drug for empirical therapy or prophylaxis was frequent (24.5%). |
| Isavuconazole tolerance was overall good, but liver function test disturbances were reported in 11.1% of patients. |
| Drug exposure was within target range in only 46% of patients. |
Introduction
Invasive fungal infections (IFIs) are responsible for more than 1.5 million deaths every year worldwide [1]. Triazole antifungals were the first orally bioavailable broad-spectrum antifungal agents introduced in the 1970s for the treatment of IFI [2]. The primary mechanism of action of triazoles is through the specific inhibition of a cytochrome P450 (CYP) 14-α-sterol demethylase enzyme resulting in depletion of cellular ergosterol, a critical component of fungal cell membranes [2]. Isavuconazole is the most recent of the clinically available triazoles indicated in adults for the treatment of invasive aspergillosis, as well as for mucormycosis in patients for whom amphotericin B is inappropriate [3].
Isavuconazole has a broad-spectrum antifungal activity against yeasts, dimorphic fungi, and moulds including the Mucorales [4]. It has a long terminal half-life allowing once-daily dosing after the initial loading doses. It also has more predictable pharmacokinetics than voriconazole making it highly attractive for clinical use [5]. Moreover, isavuconazole has an improved safety profile, particularly when compared to voriconazole with a more favourable drug–drug interaction profile and lack of QTc interval prolongation [6, 7]. However, like all triazole antifungals, isavuconazole can induce hepatotoxicity including elevated liver chemistry tests or hepatitis [3].
There are limited real-life data in the literature describing the clinical use of isavuconazole [8]. The main objective of our multicentre study was to describe the clinical practice of isavuconazole in terms of indications, exposure according to isavuconazole trough concentration, and hepatic safety. The secondary objectives were to identify variables associated with isavuconazole trough concentration and to analyse the causal relationship between the occurrence of liver injury and isavuconazole treatment.
Materials and Methods
Study Design and Inclusion/Exclusion Criteria
This study was a 30-month (January 2021–June 2023) retrospective multicentre descriptive study using computerised patients’ files of Lyon and Clermont-Ferrand University hospitals. Patients who received oral or intravenous isavuconazole and had at least one measured isavuconazole concentration in therapeutic drug monitoring (TDM) during their stay were included in the study. There was no age limit for inclusion. Patients who received isavuconazole, but did not have a TDM, were not included.
Therapeutic Drug Monitoring
Serum isavuconazole concentrations were determined in the pharmacology laboratory of Lyon and Clermont-Ferrand hospitals using a validated liquid chromatography/tandem mass spectrometry (LC/MS/MS) method. The lower limit of quantification was 0.2 mg/L and 0.12 mg/L for Lyon and Clermont-Ferrand hospitals, respectively. Isavuconazole trough concentration (Cmin) was sampled 24 ± 4 h after isavuconazole administration. Target isavuconazole Cmin ranged from 2 to 4.5 mg/L [9]. Patients were categorised into three groups based on results observed on the first TDM occasion: patients who had optimal isavuconazole concentrations (Cmin = 2–4.5 mg/L), supratherapeutic concentrations (Cmin > 4.5 mg/L), and subtherapeutic concentrations (Cmin < 2 mg/L). Cmin was normalised to a dose of 100 mg by dividing Cmin by the dose and then multiplying the result by 100. During the study period, major diagnostic, therapeutic, and infection control standards, remained unchanged.
Study Variables and their Description
The following data were collected using electronic health records: age, gender, department, liver disease, biological parameters before isavuconazole initiation and on the day of TDM (total proteins, albumin, aspartate aminotransferase [AST], alanine aminotransferase [ALT], gamma-glutamyl transferase [GGT], alkaline phosphatase [ALP], total bilirubin), isavuconazole dosing regimen (date, route, dose), isavuconazole serum Cmin, previous exposure to any azoles (< 6 months), hepatotoxic co-medications, and mycological diagnosis. The following outcomes regarding liver safety were collected: occurrence of liver test abnormalities during treatment, time to onset from isavuconazole initiation, and potential isavuconazole discontinuation in case of liver test abnormalities. To ensure reproducibility and completeness of data extraction, an Excel spreadsheet (Microsoft Corp., Redmond, WA, USA) compiling all variables to be extracted was used. AD, ALB, and CC, were in charge of data collection. Data extraction was double-checked by ALB. Disagreements over data extraction were resolved by discussion. Data quality was checked by ALB and PP for completeness, plausibility, and integrity before synthesis. The checklist of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement hosted by the Enhancing the QUAlity and Transparency Of health Research (EQUATOR) network was used as a methodological support.
Liver Safety and Causality Assessment
Liver test abnormalities were defined as the occurrence of any abnormal liver test above the upper limit of normal (ULN) during isavuconazole treatment whereas liver tests were normal before treatment. Drug-induced liver injury (DILI) was defined according to the criteria proposed by an international Expert Working Group [10]. Accordingly, any of the following DILI criteria were used: at least a ≥ 5-fold elevation above the ULN for ALT, a ≥ 2-fold elevation above the ULN for ALP (particularly with accompanying elevations in concentrations of 5′-nucleotidase or GGT in the absence of known bone pathology driving the rise in ALP level), or a ≥ 3-fold elevation in ALT concentration and simultaneous elevation of bilirubin concentration exceeding 2-fold ULN.
Causality assessment of liver injury was performed according to the WHO-Uppsala Monitoring Centre (UMC) assessment criteria [11]. Six causality categories, i.e., certain, probable/likely, possible, unlikely, conditional, and unassessable, were used. A drug-related event can only be considered when one at the first three levels is found. The essential distinctions between “Probable” and “Possible” are that in the latter case, there may be another equally likely explanation for the event and/or there is no information or uncertainty with regard to what has happened after stopping.
Statistical Analysis
Categorical variables were presented with numbers and percentages whereas continuous variables were presented with medians ± interquartile range (Q1–Q3). For categorical variables, comparisons between groups were performed using the Chi-squared test or the Fisher’s exact test, as appropriate. For continuous variables, comparisons between groups were performed by the non-parametric Mann-Whitney U test.
To identify independent predictors of isavuconazole Cmin, univariable and multivariable linear regression analyses were performed. All variables with a p-value < 0.05 in univariable analysis were included in the multivariable analysis. Among them, variables contributing to multicollinearity were excluded from the multivariable analysis. Multicollinearity was studied using the Kruskal-Wallis rank sum test or the Pearson's Chi-squared test, respectively, for continuous and categorical variables. The method used for handling missing data was case deletion, considering that the assumption of missing completely at random was satisfied. Estimates with their respective p-values are presented.
A univariable Pearson correlation analysis was used to identify the potential relationship between isavuconazole Cmin and albumin on the day of TDM. A related scatter plot was displayed to show the relationship between the two numerical variables. Receiver operating characteristic (ROC) curve analyses were used to predict an albumin cut-off and an isavuconazole time to TDM needed to achieve an optimal isavuconazole exposure > 2 mg/L.
The R-4.0.2 software (R Foundation for Statistical Computing, Vienna, Austria) was used for statistical analyses. A p-value < 0.05 was considered statistically significant.
Ethics Statement
This was a non-interventional study with no additional procedure. Data were collected during routine patient care. This study was conducted in accordance with the Declaration of Helsinki and national and institutional standards. This study was approved as an evaluation of professional practices by the Quality department of Lyon University Hospital. Due to the retrospective nature of the study, formal consent was not required. Electronic records were under the auspice of the French National Committee for Data Protection and Freedom of Information (N°23–138).
Results
Baseline Characteristics
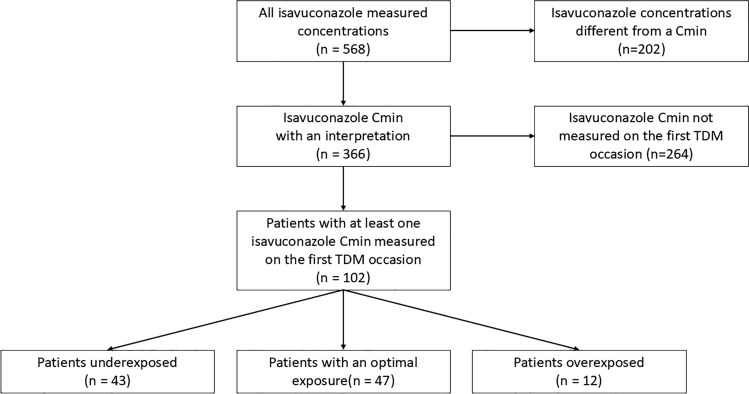
During the 30-month study period, 182 isavuconazole concentrations from Centre A and 386 from Centre B were collected (Fig. 1). Among them, 149 isavuconazole concentrations from Centre A and 217 from Centre B were eligible and matched the definition of isavuconazole Cmin. In total, 202 isavuconazole concentrations were discarded as they did not match the definition of isavuconazole Cmin. The 366 eligible isavuconazole Cmin were collected from 102 patients (57 from Centre A and 45 from Centre B) including 66 males and 36 females with a median age of 60.0 years. Baseline characteristics of the 102 included patients on the first TDM occasion are detailed in Table 1.
Fig. 1.
Study flow-chart. Cmin trough concentration, TDM therapeutic drug monitoring
Table 1.
Baseline characteristics of patients on the first isavuconazole therapeutic drug monitoring
| Variable, n (%) or median (Q1–Q3) | Patients (n = 102) |
|---|---|
| Clinical features | |
| Sex, M/F | 66/36 |
| Age (y) | 60.0 (52.0–68.0) |
| Underlying liver disease | 10 (9.8) |
| Hospitalisation units | |
| Haematology | 42 (41.1) |
| ICU | 31 (30.4) |
| Infectious disease | 12 (11.8) |
| Pneumology | 5 (4.9) |
| Others | 10 (9.8) |
| Biological parameters before isavuconazole initiation | |
| Total proteins (g/L) (n = 101) | 62.0 (55.4–70.0) |
| Albumin (g/L) (n = 77) | 25.6 (21.4–29.8) |
| AST (U/L) (n = 99) | 25 (17–47.5) |
| ALT (U/L) (n = 99) | 25 (14–49.5) |
| GGT (U/L) (n = 99) | 90 (45.5–221) |
| ALP (U/L) (n = 99) | 117 (87.5–181.5) |
| Total bilirubin (µmol/L) (n = 99) | 8 (5–13) |
| Biological parameters on the day of TDM | |
| Total proteins (g/L) (n = 94) | 63.0 (54.3–69.8) |
| Albumin (g/L) (n = 74) | 27.1 (22.0–35.2) |
| AST (U/L) (n = 100) | 24 (17.8–37.0) |
| ALT (U/L) (n = 100) | 26.5 (14.0–41.3) |
| GGT (U/L) (n = 100) | 92.5 (48.5–218.75) |
| ALP (U/L) (n = 99) | 123.0 (96.5–188.0) |
| Total bilirubin (µmol/L) (n = 91) | 9 (6–13) |
| Treatments | |
| Oral route | 74 (72.5) |
| Isavuconazole dose (mg/day) | 200 (200–600) |
| Isavuconazole duration (days) (n = 81) | 88 (25-217) |
| Isavuconazole trough concentration on the first occasion (mg/L) | 2.2 (1.3–3.6) |
| Time to TDM from isavuconazole initiation (day) | 8.5 (4.0–26.8) |
| Exposure | |
| Within therapeutic range (2–4.5 mg/L) | 47 (46.1) |
| Underexposure (< 2 mg/L) | 43 (42.2) |
| Overexposure (> 4.5 mg/L) | 12 (11.7) |
| Previous exposure to azoles (< 6 months) | 63 (52.0) |
| Hepatotoxic co-medication | 41 (40.2) |
| Mycological diagnosis | |
| Aspergillosis | 48 (47.0) |
| Mucormycosis | 26 (25.6) |
| Aspergillosis and mucormycosis co-infection | 3 (2.9) |
| Empirical treatment | 19 (18.6) |
| Prophylaxis | 6 (5.9) |
| Liver safety (n = 99) | |
| Abnormal liver test during treatment | 11 (11.1) |
| GGT | 6 (6.0) |
| GGT and ALP | 5 (5.0) |
| GGT (U/L) (n = 11) | 264.0 (166.0–413.5) |
| ALP (U/L) (n = 11) | 228.0 (185.0–334.0) |
| Time to occurrence from isavuconazole initiation (days) | 20 (6–48) |
| Isavuconazole discontinuation in case of abnormal liver test | 6 (6.0) |
| Case of isavuconazole induced liver injury | 2 (2.0) |
ALP alkaline phosphatase, ALT alanine aminotransferase, AST aspartate aminotransferase, GGT gamma-glutamyl transferase, ICU intensive care unit, TDM therapeutic drug monitoring
Most patients were hospitalised in haematology units (41.1% [n = 42]) or intensive care units (ICUs) (30.4%, [n = 31]). Most received isavuconazole orally (72.5% [n = 74]) for a median duration of 88 days (25–217 days). Of note, haematological patients had a longer median isavuconazole duration compared to other patients (130 and 35.5 days, respectively [p = 0.015]). Aspergillosis (47.0% [n = 48]), followed by mucormycosis (25.6% [n = 26]) and empirical treatment (18.6% [n = 19]), were the three most common indications for isavuconazole prescriptions. The median time to first TDM was 8.5 days (4.0–26.8). Median isavuconazole Cmin was 2.2 mg/L (1.3–3.6 mg/L). About half of the patients (46.1% [n = 47]) had an optimal exposure, 42.2% of them (n = 43) were underexposed, and 11.7% (n = 12) were overexposed. Exposure to azoles within the previous six months and hepatotoxic co-medications were common in this population, accounting, respectively, for 52.0% (n = 63) and 40.2% (n = 41) of patients.
Factors Associated with Isavuconazole Concentrations
Variables associated with isavuconazole dose-normalised Cmin are shown in Table 2. According to univariable linear regression analysis, haematology unit (p = 0.007), albumin on the day of TDM (p < 0.001), total proteins on the day of TDM (p = 0.028), time to TDM from isavuconazole initiation (p = 0.003), and oral route (p = 0.015), were significantly associated with an increase in isavuconazole dose-normalised Cmin. Of note, similar results were obtained when using isavuconazole Cmin without normalisation to the dose (data not shown).
Table 2.
Univariable and multivariable analyses of variables potentially associated with isavuconazole dose-normalised trough concentration
| Characteristics | Univariable analysis | Multivariable analysis | ||
|---|---|---|---|---|
| β coefficient | p value | β coefficient | p value | |
| Clinical features | ||||
| Sex, M/F | 0.2866 | 0.126 | ||
| Age (y) | 0.0007 | 0.897 | ||
| Underlying liver disease | − 0.1038 | 0.731 | ||
| Haematology unit | 0.4897 | 0.007 | ||
| Biological parameters before isavuconazole initiation | ||||
| Total proteins (g/L) (n = 101) | 0.0116 | 0.164 | ||
| Albumin (g/L) (n = 77) | 0.0463 | 0.001 | ||
| AST (U/L) (n = 99) | − 0.0003 | 0.486 | ||
| ALT (U/L) (n = 99) | − 0.0001 | 0.584 | ||
| GGT (U/L) (n = 99) | 0.0003 | 0.417 | ||
| ALP (U/L) (n = 99) | 0.0002 | 0.698 | ||
| Total bilirubin (µmol/L) (n = 99) | 0.0001 | 0.981 | ||
| Biological parameters on the day of TDM | ||||
| Total proteins (g/L) (n = 94) | 0.0199 | 0.028 | ||
| Albumin (g/L) (n = 74) | 0.0424 | 0.001 | 0.0339 | 0.009 |
| AST (U/L) (n = 100) | − 0.0001 | 0.536 | ||
| ALT (U/L) (n = 100) | − 0.0003 | 0.494 | ||
| GGT (U/L) (n = 100) | 0.000001 | 0.997 | ||
| ALP (U/L) (n = 99) | − 0.0001 | 0.859 | ||
| Total bilirubin (µmol/L) (n = 91) | − 0.0002 | 0.961 | ||
| Treatments | ||||
| Oral route | 0.4828 | 0.015 | 0.0536 | 0.792 |
| Isavuconazole duration (day) (n = 81) | 0.0013 | 0.058 | ||
| Time to TDM from isavuconazole initiation (day) | 0.0021 | 0.003 | 0.0014 | 0.212 |
| Previous exposure to azoles (< 6 months) | 0.2229 | 0.217 | ||
| Hepatotoxic co-medication | − 0.0310 | 0.866 | ||
| Liver safety (n = 99) | ||||
| Abnormal liver test during treatment | − 0.0261 | 0.928 | ||
| Isavuconazole discontinuation | 0.2730 | 0.379 | ||
| Isavuconazole induced liver injury | 0.2730 | 0.379 | ||
ALP alkaline phosphatase, ALT alanine aminotransferase, AST aspartate aminotransferase, GGT gamma-glutamyl transferase, TDM therapeutic drug monitoring
Collinearity and correlation analysis showed that haematological unit was associated with albumin (p < 0.001) and oral route (p < 0.001), while albumin correlated with total proteins (p < 0.001). Of note, most patients in haematological units had an albumin on the day of TDM above albumin median 27 g/L (73.5% [25/34]) and received isavuconazole orally (92.9% [39/42]). According to the multivariable linear regression analyses excluding factors of multicollinearity (i.e., haematological unit and total proteins), albumin on the day of TDM was significantly associated with isavuconazole Cmin (p = 0.010): a 10-point increase in albumin level was associated with an average 0.3 mg/L increase in isavuconazole Cmin (Table 2).
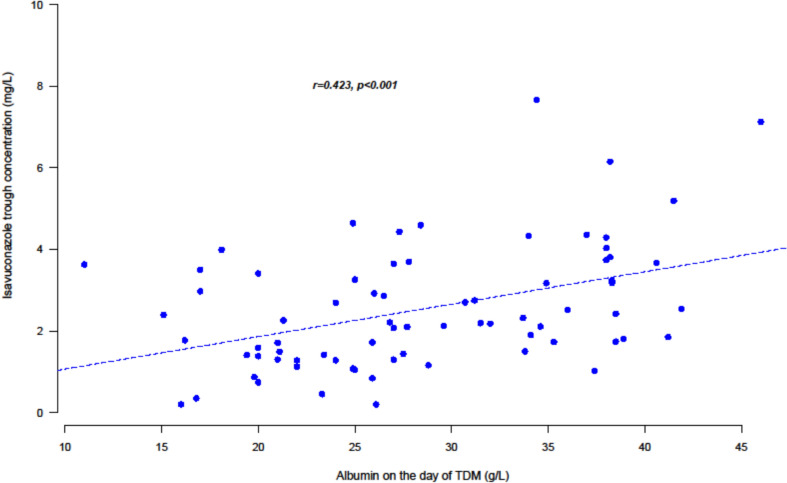
Figure 2 shows the linear correlation between isavuconazole Cmin concentrations and albumin on the day of TDM, which was statistically significant (r = 0.423; p < 0.001). Interestingly, patients with albumin above a median value of 27 g/L on the day of TDM (n = 37) had higher median isavuconazole Cmin compared to patients with albumin ≤ 27 g/L (2.70 and 1.59, respectively [p < 0.001]) and most of them had an optimal exposure (62.2% [23/37]), whereas isavuconazole daily doses were not significantly different among those groups (p > 0.1). Of note, patients from haematological units had significantly higher albuminaemia on the day of TDM compared to ICU patients (median 34.1 g/L and 23.3 g/L, respectively; p < 0.001) and isavuconazole exposure was optimal for 59.5% of them compared to 32.3% for ICU patients (p = 0.001).
Fig. 2.
Univariable Pearson correlation analysis illustrating the relationship between isavuconazole concentrations and albumin on the day of therapeutic drug monitoring
Figure 3 shows the ROC curve analysis for the binary classification of isavuconazole optimal exposure (defined as Cmin > 2 mg/L) based on albumin level. The area under the curve (AUC) was statistically significant and the albumin cut-off was 26.5 mg/L. This value was associated with a sensitivity (Se) of 0.74, a specificity (Sp) of 0.68, a predictive positive value (PPV) of 0.76, and a negative predictive value (NPV) of 0.66.
Fig. 3.
Receiver operating characteristic (ROC) curve analyses to predict albumin cut-off needed to achieve an optimal isavuconazole exposure > 2 mg/L. The AUC was statistically significant and the albumin cut-off was 26.5 mg/L (Se = 0.74; Sp = 0.68; PPV = 0.76; NPV = 0.66). AUC area under the curve, NPV negative predictive value, PPV predictive positive value, Se sensitivity, Sp specificity, TDM therapeutic drug monitoring
The same analysis for the time to TDM is shown in Figure 4. The time to TDM cut-off associated with an optimal exposure to isavuconazole was 9 days (Se = 0.71; Sp = 0.77; PPV = 0.80; NPV = 0.67).
Fig. 4.
Receiver operating characteristic (ROC) curve analyses to predict time to TDM needed to achieve an optimal isavuconazole exposure > 2 mg/L. The AUC was statistically significant and the time to TDM cut-off associated with an optimal exposure to isavuconazole was 9 days (Se = 0.71; Sp = 0.77; PPV = 0.80; NPV = 0.67). AUC area under the curve, NPV negative predictive value, PPV predictive positive value, Se sensitivity, Sp specificity, TDM therapeutic drug monitoring
Liver Safety and Causality Assessment
Among the 99 patients with available data on liver tests, 11 (11.1%) experienced at least one abnormal liver test after a median of 20 (6–48) days while liver tests were normal at baseline. Six (n = 6) patients had isolated elevation of GGT and five (n = 5) patients had an elevation of ALP combined to GGT, indicative of a cholestatic pattern. Whereas ALP and GGT levels were significantly increased for these 11 patients compared to other patients on the day of TDM (ALP = 228 vs 175 [p = 0.010] and GGT = 264 vs 170 [p = 0.006], respectively), it was not the case at baseline before isavuconazole initiation (p > 0.1). Abnormal liver tests led to isavuconazole discontinuation in 54.5% (6/11) of patients: four of them had abnormal ALP combined with GGT and two had abnormal GGT. Of note, the patients with abnormal liver tests did not have higher Cmin compared to other patients (2.60 and 2.57 mg/L, respectively [p > 0.1]).
Among the 11 patients, 2 met the clinical chemistry criteria for drug-induced liver injury [10]. The liver injury was considered as probably caused by isavuconazole in two patients who experienced an increase in ALP of 3.3- and 3.6-fold the ULN with accompanying elevations in GGT after 25 and 20 days of treatment, respectively. Severity of liver injury was classified as mild considering bilirubin concentration was below 2-fold the ULN. Liver test returned to normal in both patients 48 days and 26 days after isavuconazole discontinuation. In these two patients, the association between abnormal liver test and isavuconazole exposure was qualified as “probable” because the liver test abnormalities, including ALP and GGT, had a reasonable time relationship to isavuconazole intake, was unlikely attributed to disease or other drugs, and resolved after isavuconazole discontinuation. One patient was admitted to the haematology unit and one in an ICU. They were treated orally for an invasive aspergillosis at a daily dose of 200 mg over 127 days for the first patient, and intravenously for a mucormycosis at a daily dose of 200 mg during 21 days for the second patient. They both received concomitant potentially hepatotoxic medication for more than 3 months (cyclosporin for a myelodysplasia and tacrolimus for a lung transplant, respectively) that was continued throughout the study, and the first patient was previously exposed to azoles (voriconazole) 3 months before. The first patient was overexposed to isavuconazole (7.7 mg/L) and had an albuminemia of 34.4 g/L, whereas the second patient was underexposed to isavuconazole (1.3 mg/L) and had an albuminemia of 24 g/L.
Discussion
This multicentre study provided an overview of isavuconazole clinical practice including indications, exposure, and hepatic safety. Most patients who had an isavuconazole TDM were hospitalised in haematology units and were treated for an aspergillosis or a mucormycosis. Isavuconazole was indeed previously reported to have a favorable clinical response and a good safety profile in 200 patients with haematologic malignancies presenting an invasive fungal disease, whether used as first-line therapy or after the failure of other azole and non-azole prophylaxis or therapies [12]. In haematological patients, isavuconazole used as an empirical therapy was also a common practice representing in our study 23.8% of the prescriptions, although this is an off-label practice, which is not recommended by any international or national recommendations. This practice was previously reported in a large US transplant and cancer centre where isavuconazole was used empirically in 55% of the patients [13]. The off-label use of isavuconazole as a prophylactic treatment was less common than empirical use, representing about 6% of the prescriptions in our study. A recent retrospective study, which aimed to evaluate the mycological efficacy of isavuconazole as an antifungal prophylaxis during induction therapy of acute myeloid leukaemia or post-haematopoietic cell transplantation, reported no difference in antifungal efficacy of isavuconazole compared to posaconazole or voriconazole [14]. However, further clinical evaluation is necessary to support this practice.
We reported prolonged duration of isavuconazole treatment, mostly in haematological patients, with a median of 130 days compared to 35.5 days for other patients (p = 0.015). Prolonged treatment was demonstrated to be safe in 50 patients with haematologic malignancy receiving isavuconazole ≥ 6 months [15]; thus, the good safety profile of long-term isavuconazole may not prompt clinicians to early re-evaluation for discontinuation or transition to another antifungal. In non-neutropenic patients, a retrospective multicentric study reported comparable results regarding isavuconazole safety: only 5.9% of patients developed toxicity, mainly hepatic [16]. In our study, abnormal liver tests (ALP combined to GGT or isolated GGT elevation) occurred in 11.1% patients with a median of 20 days after isavuconazole initiation, but led to isavuconazole discontinuation in 6 out of 11 (54.5%) patients. Elevated liver tests including transaminase increase, ALP increase, bilirubin increase, and GGT increase, are reported in the summary of product characteristics of isavuconazole as the most common treatment-related adverse reactions with a prevalence of 7.9% [3]. In a retrospective study aiming to investigate isavuconazole outcomes and safety, a high proportion of patients (29%) experienced hepatic test disturbance occurring with a median 20 days from the start of isavuconazole therapy [17]. The ALP and GGT rise was the predominant hepatic test disturbance [17]. The rate of isavuconazole discontinuation for attributed hepatotoxicity was only 5% [17]. According to isavuconazole summary of product characteristics, the elevations in hepatic enzymes rarely required discontinuation of isavuconazole, but monitoring of hepatic enzymes should be considered, as clinically indicated [3].
Among the 11 patients who had an abnormal liver test, only two had probable isavuconazole-induced liver injury. The ALP and GGT elevations resolved after isavuconazole discontinuation in both patients. After 127 days of isavuconazole, one of the two patients was overexposed to isavuconazole with a Cmin of 7.7 mg/L. It was reported that the incidence of isavuconazole overexposure increased with time on therapy due to isavuconazole prolonged half-life and accumulation over time [18]. In this study, the risk of Cmin < 1.0 mg/L was about 1%, and that of Cmin > 5.13 mg/L was 27.7 and 39.2% at 28 and 60 days, respectively [19].
The data concerning concentration-effect relationships of isavuconazole are still scarce and conflicting. A quartile analysis of concentration data from the SECURE trial showed no relationships between concentration and either efficacy or adverse effects [19]. By contrast, in a single-centre study performed in adult patients, high concentrations have been associated with a higher proportion of adverse effects, mainly gastrointestinal [20]. In children, a single-centre study suggested that low isavuconazole area under the concentration-time curve (AUC) was associated with mortality, while trough concentration > 5 mg/L was associated with a higher rate of AST/ALT elevation [21]. In the present study, occurrence of abnormal liver test during isavuconazole treatment was not associated with an increase in isavuconazole Cmin according to the univariable analysis. By contrast, the influence of drug exposure on both efficacy and hepatotoxicity has been more clearly established for voriconazole [22, 23]. Low voriconazole exposure has frequently been associated with a higher incidence of treatment failure [21]. High voriconazole concentrations were associated with a higher incidence of neurotoxic adverse events including visual or auditory hallucinations, confusion or encephalopathy, suggesting a strong relationship between voriconazole concentration and neurotoxicity [21].
Regarding TDM results, we observed an underexposure to isavuconazole in almost half of the patients, especially in patients hospitalised in the intensive care unit (ICU). This difference in isavuconazole exposure between ICU and non-ICU populations was previously observed in patients treated with standard-dose isavuconazole: the ICU population had significantly lower isavuconazole blood levels compared to the non-ICU population [9]. In another study including adult ICU patients, only 35.8% of simulated patients reached an exposure target defined as a total AUC > 60 mg·h/L at Day 14 after standard dosing [24]. Renal replacement therapy (RRT) and extracorporeal membrane oxygenation (ECMO) requirement were identified as factors for sub-therapeutic drug levels in critically ill patients [25]. Another factor that may explain underexposure to isavuconazole in ICU patients was albumin level. In our study, we found that albumin on the day of TDM was significantly associated with isavuconazole Cmin. Protein level was previously reported to be associated with isavuconazole Cmin in a monocentric study of isavuconazole TDM: multivariate analyses showed that the dose, ASAT, and protein levels, were independently associated with isavuconazole Cmin [26]. In our study, patients hospitalised in haematology units had higher albumin levels and higher isavuconazole Cmin compared with patients in ICU, despite similar isavuconazole dosage. Our ROC analysis showed that an albumin cut-off of 26.5 mg/L was a significant predictor of isavuconazole Cmin > 2 mg/L. The influence of albumin on isavuconazole Cmin could be explained by high binding (> 99%) to human plasma proteins, predominantly to albumin [3]. Hypoalbuminemia could lead to an increase in volume of distribution and/or total body clearance. Surprisingly, albumin has not been reported as a significant covariate in population PK studies of isavuconazole to date [27]. Jansen and colleagues who analysed both total and unbound isavuconazole concentrations reported no significant correlation between isavuconazole unbound fraction and serum albumin concentration [24]. Further research is necessary to confirm the influence of albumin concentration on isavuconazole exposure.
The risk of underexposure in critically ill patients underlies the need for TDM, especially for patients under RRT or ECMO and patients with hypoalbuminemia. According to recent insights of the Society of Infectious Diseases Pharmacists [28], critically ill patients were mentioned as patients who should benefit from isavuconazole TDM. Other groups include patients receiving isavuconazole through alternative methods of administration (e.g., opened capsules via enteral feeding tubes), patients having drug–drug interactions, those having extreme weights, refractory/resistant infections, or other factors anticipated to alter pharmacokinetics. Children were also reported to have a high proportion of Cmin outside the therapeutic range, especially those receiving ECMO [29].
Time-to-first TDM is important to consider when interpreting isavuconazole concentration. We observed a trend of increasing Cmin with time and a cut-off of 9 days was associated with increased probability of Cmin > 2 mg/L. Bolcato et al. also reported a positive association between duration of therapy and dose normalised Cmin [26]. A linear increase Cmin of 0.032 mg/L for each day of isavuconazole treatment was reported in a retrospective study on isavuconazole blood levels demonstrating that Cmin is increasing with treatment duration [20]. This association reflects isavuconazole slow accumulation due to its prolonged half-life with average values of 110 h for oral route and 115 h for intravenous route [3]. This slow accumulation has implications for TDM interpretation. On the one hand, low concentrations measured too early (< 10 days of therapy) may not reflect steady-state exposure and may not justify dosage adjustment. On the other hand, a too low initial exposure is still not optimal and raises concerns about the adequacy of the loading dose, which is only recommended on the first two days of therapy.
As isavuconazole has a good safety profile, a higher and/or prolonged loading dose may be considered to achieve therapeutic concentrations earlier, especially in patients with risk of underexposure. Interestingly, median time to TDM was longer in patients hospitalised in haematology compared to ICU patients (14.5 days and 4.0 days, respectively), which may also explain the difference in exposure observed between the two populations.
This study has several limitations including the retrospective design and potential underreporting of adverse effects. The observed exposures were likely influenced by the TDM schedule, which was not predefined. Another limitation of our study was the determination of isavuconazole total concentration that represents both bound and unbound concentration. Unbound Cmin may be indeed a more relevant index of isavuconazole exposure, but it should also be acknowledged that its use is limited by the lack of pharmacokinetic/pharmacodynamic targets for this unbound fraction [24].
Conclusion
This multicentre analysis of isavuconazole clinical practice highlighted the common off-label indication of isavuconazole that needs to be better assessed through clinical studies and stewardship programs. Albumin on the day of TDM appeared to be an important factor influencing isavuconazole exposure. Underexposure to isavuconazole was frequent in our population, which may justify isavuconazole TDM, especially in ICU patients with low albuminemia. Time between treatment initiation and TDM should be considered for the interpretation of isavuconazole concentration to avoid a misinterpretation of a low isavuconazole concentrations.
Acknowledgements
We thank the Committee for Medicinal Products and Medical Devices, the Anti-infective Committee, and the regional group for antifungal stewardship. We thank the Quality department and the QUASPERFE committee for their methodological support in the evaluation of professional practices. From Clermont-Ferrand University hospital, we thank Dr Florent Ferrer from the Pharmacology unit, Lina Ben Riyene from the Pharmacy unit, Dr Charlotte Oris and Clément Aubry from the Biochemistry unit, and the intensive care unit. From Hospices Civils de Lyon, we thank Vincent Cottin, Eva Chatron and Arthur Petat from the Pneumology unit, and Sandrine Roux from the Infectious Disease department.
Declarations
Funding
Open access funding provided by Hospices Civils de Lyon.
Conflicts of interest
Sylvain Goutelle is an Editorial Board member of Clinical Drug Investigation. Sylvain Goutelle was not involved in the selection of peer reviewers for the manuscript nor any of the subsequent editorial decisions
Ethics Approval
This was a non-interventional study with no additional procedure. Data were collected during routine patient care. This study was conducted in accordance with the Declaration of Helsinki and national and institutional standards. This study was approved as an evaluation of professional practices by the Quality department of Lyon university hospital.
Consent to Participate
Due to the retrospective nature of the study, formal consent was not required. Electronic records were under the auspice of the French National Committee for Data Protection and Freedom of Information (N°23–138).
Consent for Publication
Not applicable.
Availability of Data and Material
The data that support the findings of this study are not openly available due to protect study participant privacy.
Code Availability
Not applicable.
Authors’ Contributions
ALB, SG, and LF, designed the work. AD, AH, ALB, CC, CM, CMC, HLW, NM, PP, PR, and ACL, contributed to data acquisition. ALB, AM, FP, PP, SG, and TV, contributed to data analysis. ALB and AD wrote the original draft. All authors reviewed the manuscript critically and approved the final version. All authors agreed to be accountable for all aspects of the work.
Contributor Information
Anne-Lise Bienvenu, Email: anne-lise.bienvenu@chu-lyon.fr.
Sylvain Goutelle, Email: sylvain.goutelle@chu-lyon.fr, Email: anne-lise.bienvenu@chu-lyon.fr.
References
- 1.Brown GD, Ballou ER, Bates S, Bignell EM, Borman AM, Brand AC, et al. The pathobiology of human fungal infections. Nat Rev Microbiol. 2024;22:687–704. [DOI] [PubMed]
- 2.Wu Y-J. Chapter 1 - Heterocycles and medicine: a survey of the heterocyclic drugs approved by the U.S. FDA from 2000 to present. In: Gribble GW, Joule JA, editors. Progress in heterocyclic chemistry [Internet]. Elsevier; 2012 [cited 2024 Jul 18]. p 1–53. Available from: https://www.sciencedirect.com/science/article/pii/B9780080968070000014. Accessed March 22, 2025.
- 3.Cresemba | European Medicines Agency [Internet]. [cited 2023 Dec 14]. Available from: https://www.ema.europa.eu/en/medicines/human/EPAR/cresemba. Accessed March 22, 2025.
- 4.Lewis JS, Wiederhold NP, Hakki M, Thompson GR. New perspectives on antimicrobial agents: isavuconazole. Antimicrob Agents Chemother. 2022;66:e00177-e222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Desai A, Schmitt-Hoffmann A-H, Mujais S, Townsend R. Population pharmacokinetics of isavuconazole in subjects with mild or moderate hepatic impairment. Antimicrob Agents Chemother. 2016;60:3025–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Maertens JA, Raad II, Marr KA, Patterson TF, Kontoyiannis DP, Cornely OA, et al. Isavuconazole versus voriconazole for primary treatment of invasive mould disease caused by Aspergillus and other filamentous fungi (SECURE): a phase 3, randomised-controlled, non-inferiority trial. Lancet. 2016;387:760–9. [DOI] [PubMed] [Google Scholar]
- 7.Lewis R, Niazi-Ali S, McIvor A, Kanj SS, Maertens J, Bassetti M, et al. Triazole antifungal drug interactions—practical considerations for excellent prescribing. J Antimicrob Chemother. 2024;79:1203–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ergün M, Jansen AME, Hilbrands LB, de Kort E, Kunst H, Reijers MHE, et al. Isavuconazole as prophylaxis and therapy for invasive fungal diseases: a real-life observational study. J Antimicrob Chemother. 2024;79:1801–10 [DOI] [PMC free article] [PubMed]
- 9.Mikulska M, Melchio M, Signori A, Ullah N, Miletich F, Sepulcri C, et al. Lower blood levels of isavuconazole in critically ill patients compared with other populations: possible need for therapeutic drug monitoring. J Antimicrob Chemother. 2024;79:835–45. [DOI] [PubMed] [Google Scholar]
- 10.Aithal GP, Watkins PB, Andrade RJ, Larrey D, Molokhia M, Takikawa H, et al. Case definition and phenotype standardization in drug-induced liver injury. Clin Pharmacol Ther. 2011;89:806–15. [DOI] [PubMed] [Google Scholar]
- 11.The use of the WHO-UMC system for standardised case causality assessment [Internet]. [cited 2024 Jul 18]. Available from: https://www.who.int/publications/m/item/WHO-causality-assessment. Accessed March 22, 2025.
- 12.Dagher H, Hachem R, Chaftari A-M, Jiang Y, Ali S, Deeba R, et al. Real-World Use of Isavuconazole as Primary Therapy for Invasive Fungal Infections in High-Risk Patients with Hematologic Malignancy or Stem Cell Transplant. J Fungi (Basel). 2022;8:74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hassouna H, Athans V, Brizendine KD. Real-world use-Isavuconazole at a large academic medical center. Mycoses. 2019;62:534–41. [DOI] [PubMed] [Google Scholar]
- 14.Scott SA, Perry C, Mahmoudjafari Z, Martin GA, Boyd S, Thompson J, et al. Incidence of breakthrough fungal infections on isavuconazole prophylaxis compared to posaconazole and voriconazole. Transplant Infectious Dis. 2023;25: e14045. [DOI] [PubMed] [Google Scholar]
- 15.DiPippo AJ, Kontoyiannis DP. Lack of toxicity with long-term isavuconazole use in patients with hematologic malignancy. Clin Infect Dis. 2019;69:1624–7. [DOI] [PubMed] [Google Scholar]
- 16.Monzó-Gallo P, Lopera C, Badía-Tejero AM, Machado M, García-Rodríguez J, Vidal-Cortés P, et al. Safety and effectiveness of isavuconazole in real-life non-neutropenic patients. Int J Infect Dis. 2024;144: 107070. [DOI] [PubMed] [Google Scholar]
- 17.Couchepin J, Reinhold I, Kronig I, Guidi M, Buclin T, Schreiber PW, et al. Isavuconazole for the treatment of fungal infections: a real-life experience from the Fungal Infection Network of Switzerland (FUNGINOS). Open Forum Infect Dis. 2024;11:ofaw223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cojutti PG, Carnelutti A, Lazzarotto D, Sozio E, Candoni A, Fanin R, et al. Population pharmacokinetics and pharmacodynamic target attainment of isavuconazole against aspergillus fumigatus and aspergillus flavus in adult patients with invasive fungal diseases: should therapeutic drug monitoring for isavuconazole be considered as mandatory as for the other mold-active azoles? Pharmaceutics. 2021;13:2099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kaindl T, Andes D, Engelhardt M, Saulay M, Larger P, Groll AH. Variability and exposure–response relationships of isavuconazole plasma concentrations in the Phase 3 SECURE trial of patients with invasive mould diseases. J Antimicrob Chemother. 2019;74:761–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Furfaro E, Signori A, Di Grazia C, Dominietto A, Raiola AM, Aquino S, et al. Serial monitoring of isavuconazole blood levels during prolonged antifungal therapy. J Antimicrob Chemother. 2019;74:2341–6. [DOI] [PubMed] [Google Scholar]
- 21.Elhence H, Mongkolrattanothai K, Mohandas S, Neely MN. Isavuconazole pharmacokinetics and pharmacodynamics in children. Pharmaceutics. 2022;15:75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dolton MJ, McLachlan AJ. Voriconazole pharmacokinetics and exposure–response relationships: assessing the links between exposure, efficacy and toxicity. Int J Antimicrob Agents. 2014;44:183–93. [DOI] [PubMed] [Google Scholar]
- 23.Wang Y, Wang T, Xie J, Yang Q, Zheng X, Dong W, et al. Risk factors for voriconazole-associated hepatotoxicity in patients in the intensive care unit. Pharmacotherapy. 2016;36:757–65. [DOI] [PubMed] [Google Scholar]
- 24.Jansen AME, Mertens B, Spriet I, Verweij PE, Schouten J, Wauters J, et al. Population pharmacokinetics of total and unbound isavuconazole in critically ill patients: implications for adaptive dosing strategies. Clin Pharmacokinet. 2023;62:1701–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bertram R, Naumann H-T, Bartsch V, Hitzl W, Kinzig M, Haarmeyer G-S, et al. Clinical and demographic factors affecting trough levels of isavuconazole in critically ill patients with or without COVID-19. Mycoses. 2023;66:1071–8. [DOI] [PubMed] [Google Scholar]
- 26.Bolcato L, Thiebaut-Bertrand A, Stanke-Labesque F, Gautier-Veyret E. Variability of isavuconazole trough concentrations during longitudinal therapeutic drug monitoring. JCM. 2022;11:5756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen N, Wang X, Li Y, Yang P, Huang M, Lu X. Population pharmacokinetics of isavuconazole in adult: a systematic review. IDR. 2023;16:7559–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McCreary EK, Davis MR, Narayanan N, Andes DR, Cattaneo D, Christian R, et al. Utility of triazole antifungal therapeutic drug monitoring: insights from the Society of Infectious Diseases Pharmacists: Endorsed by the Mycoses Study Group Education and Research Consortium. Pharmacotherapy. 2023;43:1043–50. [DOI] [PubMed] [Google Scholar]
- 29.Fernández Ledesma B, Mendoza-Palomar N, Melendo Pérez S, Fernández-Polo A, Renedo Miró B, Pau Parra A, et al. Isavuconazole use and TDM in real-world pediatric practice. Antimicrob Agents Chemother. 2023;67: e0082923. [DOI] [PMC free article] [PubMed] [Google Scholar]