Abstract
Background and Objectives:
The LevaLap 1.0 (Core Access Surgical Technologies, Atlanta, GA) was designed to promote safer, more stable, and more predictable abdominal access when using the Veress needle for insufflation. We report on the first postmarket clinical study (PMCF) assessing experience with the use of the LevaLap 1.0 during gynecologic laparoscopic surgery.
Methods:
Prospective multicenter study, including women ≥18 years old, excluding pregnancy, access site surgery in prior 10 days, abdominal hernia, contraindication to Veress needle or laparoscopy use, body mass index (BMI) >30 kg/m2, and inability/unwillingness to provide consent.
Results:
A total of 158 subjects were included, involving nine surgeons. Mean age was 43.6 ± 14.6 years and mean BMI: 24.7 ± 3.8 kg/m2. Access site was 83.5% transumbilical, 15.2% periumbilical, and 1.2% other. Using the device 96.8% (95% confidence interval [CI]: 92.8–99.0%, n = 152/157) of patients’ access was successfully achieved at first attempt and 99.4% (95% CI: 96.5–100.0, n = 156/157) within the first 2 attempts. One minor device-related adverse event was reported: a circular redness on the skin at the site of device application, resolving spontaneously. Surgeons noted easier access in 59.5%, increased confidence in 68.3%, increased access control in 67.1%, and increased access efficiency in 66.4% of cases.
Conclusion:
This PMCF study indicates that the use of the LevaLap 1.0 resulted in easier, greater control and greater efficiency during abdominal access using the Veress needle. In 96.8% access was achieved at first attempt and in 99.4% within the first 2 attempts. The use of the LevaLap 1.0 may facilitate abdominal access when using the Veress needle for insufflation.
Keywords: Abdominal access, Laparoscopy, Postmarket clinical follow-up, Surgical complications, Veress needle
INTRODUCTION
Over 15 million laparoscopic procedures are performed every year,1 a volume that is expected to continue to increase,1,2 as the benefits of minimally invasive surgery (MIS) and robotic surgery over open procedures continue to be recognized. While overall complication rates are low, about 50% of all complications occur at the time of abdominal access or port placement.3 These complications can be severe, and even life threatening, with injuries to large vessels and the gastrointestinal tract.3–5
The incidence of abdominal access-related complications is likely to continue to increase due to a number of factors: first, simply due to the increasing number of procedures being performed and, second, due to the greater number of practitioners performing the procedure.6–8 While the procedure is becoming democratized, with a great fraction of the surgeon population performing MIS, this also means that the procedures are no longer restricted to high-volume high-experience surgeons. In fact, it is likely that the rise in the number of procedures being performed will be driven primarily by an increase in the number of surgeons performing MIS, many of whom will have lower surgical volumes and, consequently, less experience than dedicated MIS surgeons, past and present.
Consistent with this hypothesis, a national retrospective cohort study in the Netherlands observed that the broad implementation of advanced MIS procedures resulted in an increasing number of these procedures.9 However, per surgeon volumes remained low and even decreased for some procedures. Other nations have also observed that while the number of laparoscopic surgeries has increased, the number of cases per surgeon decreased.10 Finally, the number of MIS surgeries in which residents participate may be insufficient to ensure their competency.11,12
Consequently, there is an urgent need to develop approaches and technologies that will facilitate and decrease the variability of abdominal access for laparoscopic surgery for all surgeons, regardless of physical attributes, sex, or surgical volume. A technology that could increase a more predictable abdominal access may be useful for surgeons; the objectives of this technology should be to increase the reproducibility, safety, and speed of the procedure. The LevaLap 1.0 is such a device, facilitating and creating a stable and more predictable stable platform when using the Veress needle for initial abdominal insufflation during laparoscopic surgery. Although there have been attempts to develop technologies with a similar mechanism of action,13,14 such technologies were not technologically ready to be commercialized. The LevaLap 1.0 is a newly developed device with optimized ergonomic design.
In a prior study, we observed that, among other benefits, the LevaLap 1.0 increases the distance between the access site and retroperitoneal vessels by >5 cm,15 potentially reducing the risk of injury to these critical structures during laparoscopic access. The LevaLap 1.0 facilitates Veress needle insertion regardless of variations in surgeon upper body and hand strength or the use of potentially disfiguring towel clamps and other grasping instruments.
The post-market clinical follow-up (PCMF) is an important part of postmarket surveillance, one that now applies to almost all medical devices under the new European Medical Device Regulation effective May 2021.16 In the United States the Food and Drug Administration (FDA) regulates postmarket surveillance activities for medical devices through 21 Code of Federal Regulations (CFR) Part 822.17 Undertaking a PMCF is not only required in certain jurisdictions (e.g., the EU) but is considered best practice in the device industry.18 We should note that the PCMF is not a comparative study (e.g., randomized clinical trial). The PMCF solely aims to confirm the safety and performance of the device under normal use, identify any new side effects or risks, monitor known side effects and contraindications, ensure the benefit-risk ratio remains acceptable, and identify any misuse or off-label use of the device.19
In this first multicenter PMCF study, we report the success rate of laparoscopic access with the LevaLap 1.0, the associated complication rate, and the operator experience and impression, under normal use. The primary objective of the study was to describe, in patients indicated for a gynecological abdominal procedure suitable for laparoscopic intervention using the LevaLap 1.0 device, the success of laparoscopy access at the first attempt. Secondary objectives of the study included describing the adverse events (AEs) pertaining to the access procedure or pertaining to the access procedure up to 2-weeks postprocedure, the proportion of first-access attempts that were free of procedure-related complications, any device deficiencies, the time required to deploy the LevaLap 1.0 device and create the initial additional peritoneal safe space, and the use of the LevaLap 1.0 device in real-life circumstances.
MATERIALS AND METHODS
Device
The LevaLap 1.0 (Core Access Surgical Technologies [CAST], Atlanta, GA) device is a sterile clear plastic (styrene-butadiene copolymers) bell with a vacuum port that connects to a standard operating room (OR) vacuum system (Figures 1 and 2). At its superior edge it contains an injection septum constructed of thermoplastic elastomers and designed to allow the insertion of the Veress needle without loss of vacuum. The device facilitates the placement of the Veress insufflation needle into the abdomen by raising the abdominal wall away from the retroperitoneal vessels using standard OR wall suction. It also provides a more predictable and stable surface or platform for Veress needle insertion.
Figure 1.

The LevaLap 1.0 device (Core Access Surgical Technologies). The device measures 10 cm in height and 15 cm in diameter. Reproduced with permission from Orsi et al.15
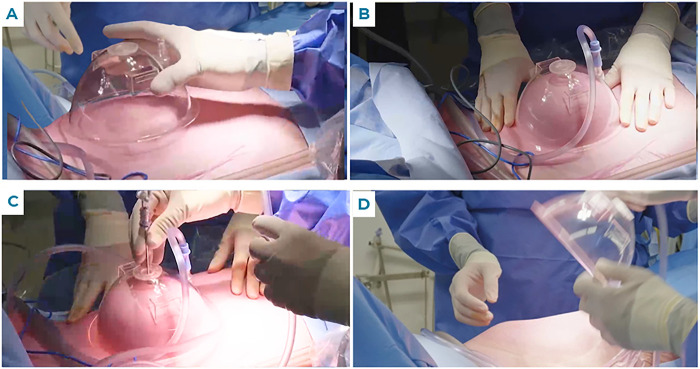
Figure 2.
Utilizing the LevaLap 1.0 device. (A) The LevaLap 1.0 is placed on the abdominal wall at Veress needle insertion site. (B) Standard OR suction is applied to the LevaLap 1.0 port to lift the abdominal wall. (C) Once the abdominal wall has been lifted and is in apposition to the LevaLap 1.0 interior, the Veress needle is inserted, generally at 90° to the abdominal wall, to create the pneumoperitoneum. (D) The Veress needle is retracted, suction is released, and the LevaLap 1.0 removed.
After placing the LevaLap 1.0 on the patient’s abdomen over the surgical entry site vacuum is applied through the vacuum port and the negative pressure lifts the patient’s abdominal wall into the curve of the bell chamber. The level of negative pressure (vacuum) was set by the individual surgeon to ensure the abdominal wall rose and made contact with the injection septum of the device. The Veress needle is then placed directly into the abdomen through the injection septum. As the device brings the anterior abdominal wall closer to the cup of the device, a normal length Veress needle is used. The soft material of the injection septum absorbs and distributes needle pressure, allows needle puncture without leakage of the negative pressure, and permits inserting the needle vertically (90°F to the abdominal wall). After appropriate intraabdominal placement of the Veress needle is verified by the usual safety measures, the abdomen is insufflated using carbon dioxide (CO2). Once abdominal distention is complete, the Veress needle is retracted, suction to the device is released, and the LevaLap 1.0 is withdrawn from the operating field, leaving the abdominal region ready for surgery.
Study Protocol
The PMCF is a single-arm, prospective, multicenter study of women undergoing gynecologic laparoscopic surgery. Inclusion criteria included age 18 years and older and indicated for laparoscopic gynecological abdominal procedures. Exclusion criteria included: (1) pregnancy, (2) surgery over the chosen access site within the previous 10 days, (3) confirmed symptomatic abdominal hernia, (4) when use of a Veress needle and/or laparoscopic techniques are contraindicated, (5) body mass index (BMI) >30 kg/m2, and (6) inability or refusal to sign the patient informed consent form, or unable to give a verbal or written nonopposition, depending on the local regulations. However, surgeons were able to request exceptions to the protocol, and as they became more comfortable with the use of the device some of them ventured beyond the recommended 30 kg/m2 BMI with success.
Each surgeon was trained on the use and deployment of the LevaLap 1.0 and on the completion of a uniform standardized case report form (CRF) prior to the start of the clinical investigation. Specifically, each participating investigator was required to use the LevaLap 1.0 device in at least seven cases prior to recruiting subjects for the PMCF study. This helped minimize surgeon’s learning curve, especially since 1 of the secondary outcomes relates to the measurement of time required to achieve adequate device insufflation to create adequate pneumoperitoneum.
Study subjects underwent surgery using the LevaLap 1.0 device to facilitate abdominal access using the Veress needle for insufflation and were followed for 2 weeks. Using the CRF patient data was collected by each surgeon including age, BMI, inclusion and exclusion criteria, and indication for the procedure. Immediately following the procedure, the surgeon completed a series of question relating to the procedure including the site of access (transumbilical, periumbilical, Palmer’s point, and other), any safety tests that were done to verify correct positioning of the Veress needle inside the abdominal cavity, the time when the LevaLap 1.0 device was positioned on the skin (T0) and the time when pneumoperitoneum was first successfully created (TF), the number of attempts it took to achieve successful abdominal access and any reason for failure, the maximum negative pressure required for the abdominal wall to rise and make contact with septum, and the preset intra-abdominal pressure used at the beginning of the procedure before the insertion of the first trocar.
Each surgeon also completed a series of questions related to the use of the device. Questions included the ease of use and the ease of access to create the pneumoperitoneum with the device, whether the device provided the surgeon with increased confidence, increased access control, and increased access efficiency. Surgeons were also queried regarding whether they would recommend the device to a fellow surgeon, what they felt were the advantages of the device, the number of cases that would be required to feel experienced with the use of the device, and the number of cases needed to added value of the device, and whether the access procedure benefited from the use of the device. Various questions relating to device handing were completed by the surgeon and a Device Deficiency Form was required in the event there was felt to be inadequacy of the device with respect to its identity, quality, durability, reliability, usability, safety, or performance. Finally, the surgeon recorded any AEs, either immediate or identified during the 2-week follow-up period, including the nature and severity of the event, whether anticipated or unanticipated, and whether it was felt to be related to the laparoscopic access procedure or to the LevaLap 1.0 device itself.
Participating Centers
Eight centers participated in this prospective study, and the study was approved by the ethics committee of each participating center and each patient was required to give their signed and dated informed consent or verbal nonopposition, depending on the local regulations. To reduce data variability, each participating center was required to contribute at least 15 cases each to the study and all participating surgeons were required to contribute at least 5 cases each to the study. The study was estimated to take 4–6 months, including patient recruitment and follow-up periods. We should note that while most participating surgeons were high volume experienced operators, a few were less experienced.
Study Compliance
The study was set up by ClinSearch (Malakoff, France), an independent service provider company acting under contract. That company proceeded with the appropriate regulatory applications and ethics approval was granted when required. The study was managed in accordance with the applicable regulations in force in each European Union member state where the study was set up. The remote quality control was carried out according to ClinSearch’s procedures.
Data Protection
Data for this study was recorded on paper CRFs and submitted by investigators to the data collection room (DCR) electronically. The CRF form is built into a Trial Interactive room provided by TransPerfect Life Sciences (New York, NY). This validated system meets the FDA’s Title 21 CFR part 11, ensuring the validation of the system, the traceability of the audit trail, the retention, protection, reproducibility and recovery of trial data, control of access to information and electronic signature, among others. Data was transcribed into the system by qualified personnel and then verified through a document workflow within the DCR. Electronic queries were raised against the documents provided if questions arise. Investigators were obliged to respond electronically if questions arose and provide clarification or corrected document(s) as necessary. The requests with their responses were managed through the DCR. If a correction was made, the corrected information was entered into the form, superseding the original information entered, if applicable. An immutable audit trail permitted the identification of the modification. Data integrity was protected through rigorous controls, processes, and system validation documented in TransPerfect’s Quality Management System. An additional final independent quality control was performed by ClinSearch to validate the data at the end of the study.
Endpoints, Sample Size Calculations, and Statistical Analysis
Endpoints
The primary endpoint of the study was the percentage of procedures in which the pneumoperitoneum was achieved at the first attempt. Secondary endpoints included various safety outcomes, such as the proportion and description of AEs that were found to be related to the access procedure, either during the procedure or up to 2 weeks postprocedure, and the proportion and description of all serious AEs (SAEs) that occurred from the time of the procedure up to the 2-weeks follow-up. Also included were the percentage of procedures in which the pneumoperitoneum was achieved at the first attempt without any complications (procedure-related AEs), the proportion and description of all device deficiencies that occur during the procedure, and the measurement of the time required to obtain a “satisfactory” pneumoperitoneum. Various conditions during the use of the LevaLap 1.0 device were also considered secondary endpoints including the mean maximal vacuum pressure required to raise the abdominal wall to contact the septum, the mean preset intra-abdominal pressure used, and the proportion of procedures where the placement position of the LevaLap 1.0 device was changed between access attempts, including specifics related to the new location. Finally, there were several exploratory outcomes.
Determination of sample size
Since the study was a postmarket follow-up observational descriptive study, the sample size was not based on formal statistical hypothesis testing but on estimating the precision of the 95% confidence interval (CI) of the primary endpoint. A sample size of 122, recruited over 5–7 centers, was determined as the minimum needed to ensure a precision of ±5% of the 95% CI for the primary outcome. This calculation assumed that the percentage of procedures in which the pneumoperitoneum was achieved at the first attempt would fall between the least and most favorable published scenarios, i.e., from 86.9%20 to 93.6%.21
Statistical analysis
Continuous variables were described using mean and standard deviation, or median and range (or interquartile range [IQR]), while categorical variables were presented as the number and percentage of patients in each category. The 95% CIs of the proportion of successful procedures were calculated using the exact binomial method. The time required to successfully create a satisfactory pneumoperitoneum using LevaLap 1.0. was calculated as the difference between when the device was positioned on the skin (T0) and when pneumoperitoneum was first successfully created (TF). If multiple attempts were needed, TF was recorded at the end of the final successful attempt; patients who did not achieve success by the third attempt were excluded from this calculation.
RESULTS
Although the planned sample size was 122, the actual number of participants was greater due to the higher than expected number of centers participating. Consequently, the total number of subjects enrolled in the study was 163 across eight centers. Five cases from three surgeons were excluded from further analysis as these surgeons contributed outlier low numbers of cases to the study (2 surgeons undertook 1 case each in the protocol and the third surgeon did three cases). A total of 158 subjects were included in the analysis (Table 1), operated upon by nine surgeons, each performing ≥5 cases (5–22 cases/surgeon). Mean patient age was 43.6 ± 14.6 years (18–84 years) and mean BMI was 24.7 ± 3.8 kg/m2 (17.8–37 kg/m2). Access site was chosen by the surgeon, and was transumbilical in 83.5%, periumbilical in 15.2%, and other in 1.2% (i.e., Palmer’s point in 1 subject for a pelvic tumor rising to just below the umbilicus, and Lee-Huang point [middle upper abdomen] in another subject due to an 11 cm leiomyoma) of study subjects. Indications for surgery are listed in Table 2.
Table 1.
Number of Patients by Site and Surgeon
| Site Name | N per Center (%) | N per Surgeon (%) |
|---|---|---|
| Frauenklinik an der Elbe | 22 (13.9) | 22 (13.9) |
| Saarland University Medical Center | 20 (12.7) | 5 (3.2) |
| 15 (9.5) | ||
| University of Freiburg | 19 (12.0) | 19 (12.0) |
| Odens University Hospital | 19 (12.0) | 19 (12.0) |
| Hospital Beclere | 22 (13.9) | 22 (13.9) |
| Hospital Lariboisiere | 17 (10.8) | 17 (10.8) |
| Clinique Prive de lYvette | 19 (12.0) | 19 (12.0) |
| Oslo University Hospital | 20 (12.7) | 20 (12.7) |
| Total | 158 (100.0) | 158 (100.0) |
Table 2.
Indications for Laparoscopic Procedures among Study Subjects
| Indication | N | % * |
|---|---|---|
| Adnexal | 40 | 20.8 |
| Endometriosis | 46 | 23.9 |
| Infertility | 22 | 11.5 |
| Oncological | 9 | 4.7 |
| Prolapse | 11 | 5.7 |
| Uterine | 62 | 32.3 |
| Other** | 2 | 1 |
The sum of percentages exceeds 100% because patients could have multiple indications. There was a total of 192 indications for 158 patients: 125 patients (79.1%) had a single indication, 32 patients (20.3%) had 2 indications, and 1 patient (0.6%) had three indications.
One myoma and 1 tubo-ovarian abscess.
Primary Endpoint
In 153 of 158 (96.8%) of study subjects pneumoperitoneum was achieved at the first attempt (95% CI: 92.8–99.0%). In 5 study subjects (3.2%), abdominal access with the Veress needle required more than 1 attempt. A second attempt led to successful outcomes in 4 of these 5 cases, leading to a 99.4% success rate within the first 2 attempts (95% CI: 96.5–100.0).
The reasons given for requiring more than 1 attempt included adhesions-related issues (n = 3), “a deeper positioning of the needle solved the matter” (n = 1), and “failure to obtained vacuum due to skin closed vacuum opening” (n = 1). In the study subject where the inability to achieve successful abdominal access was due to a failure to obtain adequate device vacuum due to skin being excessively lax and stoppering the vacuum port, the inability to achieve abdominal access persisted even after a third attempt. There was no difference in median BMI between the 153 subjects in which abdominal access was able to be achieved at the first attempt and the 5 requiring more than 1 attempt (25.0 vs 24.5 kg/m2, respectively).
Secondary Endpoints
Safety outcomes with the LevaLap 1.0
No SAEs were reported, and no device deficiencies were observed during the access procedures across all patients. One device-related AE, graded as mild, was reported during access attempts, i.e., a circular redness on the skin was observed during the procedure at the site of device application, which lasted a few hours and was completely resolved the next morning. No intervention was undertaken, and the issue resolved without sequelae the following morning. One additional AE, also graded as mild, was reported which was felt to be related to the laparoscopic procedure, but not device use or the abdominal access, i.e., study subject developed a minor urinary tract infection 2 days postintervention, which successfully resolved using medication within 2 days without sequelae. No other AEs were reported.
Time required to successfully create pneumoperitoneum with the LevaLap 1.0
The median time required to successfully create a satisfactory pneumoperitoneum was 2 minutes (IQR: 2–3 minutes; range: 1–6 minutes) (Figure 3).
Figure 3.
Time to achieve satisfactory pneumoperitoneum with the LevaLap 1.0 (n = 158).
Pressure required to successfully raise the abdominal wall with the LevaLap 1.0
The maximal negative pressure (vacuum) required for the abdominal wall to rise and make contact with the injection septum was available in 64.5% (102/158) of study subjects. Among these, the mean negative pressure required to raise the abdomen was 195.4 ± 118.0 mmHg (median = 300.00 mmHg; range: 135–530 mmHg).
Surgeon impression of the LevaLap 1.0 device use
In 59.5% of cases surgeons reported that the device increased ease of access, in 68.3% (strongly agree/agree) increased confidence, in 67.1% (strongly agree/agree) increased access control, and in 66.4% (strongly agree/agree) improved access efficiency. Surgeons reported that in 98.7% (strongly agree/agree) of cases the device was easy to use. Surgeons felt that the device positively ‘did’ or ‘maybe did’ benefit access in 88.0% of procedures and in 77.9% of cases surgeons reported they would recommend the use of the device to fellow surgeons. Principal advantages of the device as reported by the surgeons included ease of use (ranked first or second in 70.9% of cases), increased patient safety (ranked first or second in 62.1% of cases), increased access control (ranked first or second in 48.1% of cases), and time saved (ranked first or second in 18.9% of cases). Surgeons felt that a median number of 55–10 cases was needed to feel experienced with the device and a median number of 103–20 cases was needed to determine the added value of the device.
Surgeon impression of the LevaLap 1.0 device handling
On a per case basis, surgeons noted that in 2.5% there was some damage to device upon delivery, that in 5.1% they did not feel the device packaging was appropriate and practical, and that in 2.5% the instructions were not easy to follow. Surgeons also noted that in 1.3% of cases the negative pressure features and applications were not effective in raising the abdomen, in 0.6% the injection septum was not easy to pierce, in 0.6% overall the device features were not easy to utilize.
DISCUSSION
This prospective PMCF study reports that the LevaLap 1.0 provides pneumoperitoneum in nearly all patients within the first 2 attempts, without significant AEs. Furthermore, surgeons reported that the device increased ease of access and control and increased surgeon’s confidence in most patients. Most surgeons felt that the device benefited the abdominal access procedure in almost 90% of patients and in almost 80% of cases the surgeon would recommend the device to a colleague.
Specific data regarding the patient’s prior surgical history was not available, although a number of the surgeons used the device in patients with prior laparotomy (via midline or Pfannenstiel incisions). However, we should note that the decision to use or not use the LevaLap 1.0 in patients with prior laparotomy primarily relates to the use of the Veress needle for abdominal entry rather than to the use of the device. While in this study most access was umbilical (∼80%) in 1 case the device was used at Palmers Point, with the surgeon confirming proper apposition of the device and skin. In clinical use, we are aware that the device has been extensively used at Palmer’s point and there have been no complaints received regarding problems with proper sealing of the device and skin.
In general, an increase in the number of attempts required to successfully access the abdomen and achieve pneumoperitoneum is associated with increased risk.22 In approximately 8% of all laparoscopies, surgeons require 2 or more access attempts with the Veress needle to achieve successful intra-abdominal access. When this happens, the risk of complications increases to 16%–16% on the second attempt, 44–64% on the third attempt, and more than 85% after the third attempt.20,21 With the use of the LevaLap 1.0 successful abdominal access at first attempt was achieved in 96.8% of patients. This compares favorably with data from Richardson and Sutton20 who reported that without the use of the device under consideration, pneumoperitoneum was achieved at the first attempt in only 86.9% of cases. Using the LevaLap 1.0 successful abdominal access was achieved by the second attempt in fully 99.4% of subjects.
While some surgeons have advocated open access techniques as a way to reduce complications,23 and while these may be of value in patients known to have significant intraabdominal adhesions, in general, no significant differences in overall complication rates have been found for closed compared with open techniques for primary abdominal insufflation.24–27 Likewise, there is no evidence that suggests that radially expanding trocars reduce major complications during primary or secondary port placement.27 Thus, considering speed, simplicity, cost and complications rates, the closed (Veress needle) technique remains a favored method for gaining abdominal access.
As mentioned above, the number of surgeons performing MIS via laparoscopy continues to increase,6–8 although many perform a lesser number of surgeries than MIS surgeons did previously.9,10 This suggests that a device, such as the LevaLap 1.0, that provides a standardized and more predictable abdominal access and is easy to use (a median of 5 cases were deemed necessary for surgeons to attain the requisite experience) would be of benefit to these surgeons. Standardization and predictability in this group of surgeons is of utmost importance28,29 and the role of the Levalap 1.0 in improving training should be tested in future studies. Nonetheless, and as the present data notes, the many benefits of the LevaLap 1.0 are also experienced by high-volume surgeons. In addition, globally the diversity of surgeons is rising, including an increase in the number of female surgeons.30,31 Consequently, a device that reduces the need for specific upper body strength or hand sizes in achieving abdominal access rapidly and safely would be beneficial.32 Again, the LevaLap 1.0 provides these advantages, regardless of surgeon build, height, or strength.
Overall, surgeons felt the device was easy to use and was beneficial in several ways (e.g., increased ease of access, access control, access efficiency, and surgeon’s confidence) in the vast majority of cases performed. Considering that approximately 70% of the surgeons were experienced operators who were very accustomed to Veress needle insertion, the fact that in between 50% and 70% of cases they observed improvements in access ease, confidence, efficiency, and control is noteworthy. Furthermore, in only 1 patient with a mild AE, a circular redness at the site of the device application, with the issue resolving without intervention or sequelae the morning following the surgical procedure. We should note that the patient was quite thin (19.6 kg/m2) and the preset pressure used to elevate the abdomen using the LevaLap 1.0 device, as determined by the surgeon, was on the higher side (450 mmHg). Nonetheless, the device was used in several other thin patients with the same or higher vacuum pressures without issue.
In summary, the LevaLap 1.0 enables a safer and more successful abdominal entry by facilitating access at the first attempt, as repeated attempts increase the risk of complications. The LevaLap 1.0 also helps ensure a stable, predictable platform for Veress needle insertion, increasing the speed, control, efficiency, and confidence when undertaking laparoscopic surgery. The device increases surgeon’s control, precision, and speed during Veress needle placement by standardizing laparoscopic access so that the operator experiences the same feel case after case, regardless of their physical or hand size and upper body strength, and without the need for a second assistant or the use of potentially disfiguring grasping or towel clamps.
Footnotes
Coprincipal investigators.
Contributor Information
Xavier Deffieux, Service de Gynécologie, Antoine-Béclère Hospital, Clamart, France. (Dr. Deffieux).
Jean-Louis Benifla, Department of Gynecology and Obstetrics, Lariboisière Hospital, Paris, France. (Drs. Benifla and Huchon).
Ingolf Juhasz-Böss, Department of Obstetrics and Gynaecology at the Medical Center, University Hospital of Freiburg, Freiburg im Breisgau, Germany (Drs. Juhasz-Böss, Moritz Breitbach, and Taran).
Moritz Breitbach, Department of Obstetrics and Gynaecology at the Medical Center, University Hospital of Freiburg, Freiburg im Breisgau, Germany (Drs. Juhasz-Böss, Moritz Breitbach, and Taran).
Olaf Buchweitz, Frauenklinik an der Elbe, Hamburg, Germany. (Drs. Buchweitz and Hackethal).
Nassir Habib, Clinique de l'Yvette, Longjumeau, France. (Dr. Habib).
Kirsten Hald, Division of Gynecology and Obstetrics, Oslo University Hospital, Oslo, Norway. (Drs. Hald and Michelsen-Wahl).
Bashar Haj Hamoud, Department of Gynecology, Obstetrics, and Reproductive Medicine, Saarland University Medical Center, Homburg, Germany. (Drs. Haj Hamoud, Nigdelis, and Solomayer).
Cyrille Huchon, Department of Gynecology and Obstetrics, Lariboisière Hospital, Paris, France. (Drs. Benifla and Huchon).
Vibeke Lysdal, Department of Obstetrics and Gynecology, Odense University Hospital, Odense, Denmark. (Drs. Lysdal and Rudnicki).
Meletios P. Nigdelis, Department of Gynecology, Obstetrics, and Reproductive Medicine, Saarland University Medical Center, Homburg, Germany. (Drs. Haj Hamoud, Nigdelis, and Solomayer).
Martin Rudnicki, Department of Obstetrics and Gynecology, Odense University Hospital, Odense, Denmark. (Drs. Lysdal and Rudnicki).
Erich-Franz Solomayer, Department of Gynecology, Obstetrics, and Reproductive Medicine, Saarland University Medical Center, Homburg, Germany. (Drs. Haj Hamoud, Nigdelis, and Solomayer).
Florin-Andrei Taran, Department of Obstetrics and Gynaecology at the Medical Center, University Hospital of Freiburg, Freiburg im Breisgau, Germany (Drs. Juhasz-Böss, Moritz Breitbach, and Taran).
Henrik Michelsen-Wahl, Division of Gynecology and Obstetrics, Oslo University Hospital, Oslo, Norway. (Drs. Hald and Michelsen-Wahl).
Ricardo Azziz, Departments of Obstetrics and Gynecology and Medicine, Heersink School of Medicine, University of Alabama at Birmingham, Birmingham, AL. (Dr. Azziz); Department of Healthcare Organization and Policy, School of Public Health, University of Alabama at Birmingham, Birmingham, AL. (Dr. Azziz); Department of Health Policy, Management and Behavior, School of Public Health, University at Albany, SUNY, Rensselaer, NY. (Dr. Azziz).
Vincenzo Bagnardi, Department of Statistics and Quantitative Methods, University of Milan-Bicocca, Milan, Italy. (Dr. Bagnardi).
Andreas Hackethal, Frauenklinik an der Elbe, Hamburg, Germany. (Drs. Buchweitz and Hackethal).
References:
- 1.Market and Markets™. Laparoscopic instruments market by product (laparoscope, insufflator, suction/irrigation systems, access & energy devices), application (gynecology, general, urology, colorectal, bariatric, pediatric), end user (hospital, ASC) & region – global forecast to 2026. Northbrook, IL: Market and Markets™; 2021. Available at: https://www.marketsandmarkets.com/Market-Reports/laparoscopic-instruments-market-130019497.html. Accessed July 21, 2024. [Google Scholar]
- 2.iData Research. Over 13 million laparoscopic procedures are performed globally every year. Burnaby, Canada: iData Research; 2020. Available at: https://idataresearch.com/over-13-million-laparoscopic-procedures-are-performed-globally-every-year/. Accessed July 20, 2024. [Google Scholar]
- 3.Magrina JF. Complications of laparoscopic surgery. Clin Obstet Gynecol. 2002;45(2):469–480. [DOI] [PubMed] [Google Scholar]
- 4.Pryor A, Mann WJ, Jr, Bates AT. Complications of laparoscopic surgery. In: UpToDate, Marks J, Falcone T. (Section Eds). Waltham, MA: UpToDate. Accessed July 20, 2024. [Google Scholar]
- 5.Fuller J, Ashar BS, Carey-Corrado J. Trocar-associated injuries and fatalities: an analysis of 1399 reports to the FDA. J Minim Invasive Gynecol. 2005;12(4):302–307. [DOI] [PubMed] [Google Scholar]
- 6.St John A, Caturegli I, Kubicki NS, Kavic SM. The rise of minimally invasive surgery: 16 year analysis of the progressive replacement of open surgery with laparoscopy. JSLS. 2020;24(4):e2020. 00076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mattingly AS, Chen MM, Divi V, Holsinger FC, Saraswathula A. Minimally invasive surgery in the United States, 2022: understanding its value using new datasets. J Surg Res. 2023;281:33–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.You SS, Kell M, Oresanya L, Kuo LE, Dauer E. Complex minimally invasive surgery in general surgery training: keeping up with the times. Global Surg Educ. 2025;4:1. [Google Scholar]
- 9.Tummers FHMP, Hoebink J, Driessen SRC, Jansen FW, Twijnstra ARH. Decline in surgeon volume after successful implementation of advanced laparoscopic surgery in gynecology: an undesired side effect? Acta Obstet Gynecol Scand. 2021;100(11):2082–2090. [DOI] [PubMed] [Google Scholar]
- 10.Lin CH, Long CY, Huang KH, Lo TS, Wu MP. Surgical trend and volume effect on the choice of hysterectomy benign gynecologic conditions. Gynecol Minim Invasive Ther. 2021;10(1):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Qureshi A, Vergis A, Jimenez C, et al. MIS training in Canada: a national survey of general surgery residents. Surg Endosc. 2011;25(9):3057–3065. [DOI] [PubMed] [Google Scholar]
- 12.Klebanoff JS, Marfori CQ, Vargas MV, Amdur RL, Wu CZ, Moawad GN. Ob/gyn resident self-perceived preparedness for minimally invasive surgery. BMC Med Educ. 2020;20(1):185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lemyre M, Watson J, Hsu S, Kazanegra R, Nezhat C. An innovative method of access for laparoscopic surgery. J Minim Invasive Gynecol. 2008;15(6):96S. [Google Scholar]
- 14.Bettaiah R, Kamath SS, Ghanti R. LapCap: a novel device to reduce entry complications. J Minim Invasive Gynecol. 2018;25(4):566–568. [DOI] [PubMed] [Google Scholar]
- 15.Orsi F, Maiettini D, Bagnardi V, Azziz R. Prospective cohort study quantifying the effect of the LevaLap 1.0 on the distance between the abdominal wall and intra-abdominal viscera. J Minim Invasive Gynecol. 2023;30(9):748–756. [DOI] [PubMed] [Google Scholar]
- 16.Regulation (EU). 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices, amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009 and repealing Council Directives 90/385/EEC and 93/42/EEC. Available at: http://data.europa.eu/eli/reg/2017/745/oj.
- 17.U.S. Food & Drug Administration. CFR - Code of Federal Regulations Title 21, August 30, 2024. Available at: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=822.
- 18.Medical Device Clinical Evaluation Working Group, International Medical Device Regulators Forum. Post-Market Clinical Follow-Up Studies. March 25, 2021. Available at: https://www.imdrf.org/sites/default/files/docs/imdrf/final/technical/imdrf-tech-210325-wng65.pdf.
- 19.Medical Device Coordination Group. MDCG 2020-7, Post-Market Clinical Follow-Up (PMCF) Plan Template, A guide for manufacturers and notified bodies. April 2020. Available at: https://health.ec.europa.eu/system/files/2020-09/md_mdcg_2020_7_guidance_pmcf_plan_template_en_0.pdf.
- 20.Richardson RE, Sutton CJG. Complications of first entry: a prospective laparoscopy audit. Gynaecol Endoscopy. 1999;8(6):327–334. [Google Scholar]
- 21.Ahmad G, O’Flynn H, Duffy JMN, Phillips K, Watson A. Laparoscopic entry techniques. Cochrane Database Syst Rev. 2012(2):CD006583. [DOI] [PubMed] [Google Scholar]
- 22.Krishnakumar S, Tambe P. Entry complications in laparoscopic surgery. J Gynecol Endosc Surg. 2009;1(1):4–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bonjer HJ, Hazebroek EJ, Kazemier G, Giuffrida MC, Meijer WS, Lange JF. Open versus closed establishment of pneumoperitoneum in laparoscopic surgery. Br J Surg. 1997;84(5):599–602. [PubMed] [Google Scholar]
- 24.Jansen FW, Kolkman W, Bakkum EA, de Kroon CD, Trimbos-Kemper TC, Trimbos JB. Complications of laparoscopy: an inquiry about closed- versus open-entry technique. Am J Obstet Gynecol. 2004;190(3):634–638. [DOI] [PubMed] [Google Scholar]
- 25.Nuzzo G, Giuliante F, Tebala GD, Vellone M, Cavicchioni C. Routine use of open technique in laparoscopic operations. J Am Coll Surg. 1997;184(1):58–62. [PubMed] [Google Scholar]
- 26.Deziel DJ, Millikan KW, Economou SG, Doolas A, Ko ST, Airan MC. Complications of laparoscopic cholecystectomy: a national survey of 4,292 hospitals and an analysis of 77,604 cases. Am J Surg. 1993;165(1):9–14. [DOI] [PubMed] [Google Scholar]
- 27.Thepsuwan J, Huang K-G, Wilamarta M, Adlan A-S, Manvelyan V, Lee C-L. Principles of safe abdominal entry in laparoscopic gynecologic surgery. Gynecol Minim Invasive Ther. 2013;2(4):105–109. [Google Scholar]
- 28.Shore EM, Lefebvre GG, Grantcharov TP. Gynecology resident laparoscopy training: present and future. Am J Obstet Gynecol. 2015;212(3):298–301, 298.e1. [DOI] [PubMed] [Google Scholar]
- 29.Wright KN, Truong M, Siedhoff MT. Residency training in gynecologic surgery: where do we go from here? J Gynecol Surg. 2022;38(6):375–378. [Google Scholar]
- 30.Linscheid LJ, Holliday EB, Ahmed A, et al. Women in academic surgery over the last four decades. PLoS One. 2020;15(12):e0243308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Saadé S, Delafontaine A, Cattan J, Celanie D, Saiydoun G. Attractiveness and gender dynamics in surgical specialties: a comparative analysis of French medical graduates (2017-2022). BMC Med Educ. 2024;24(1):197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Armijo PR, Flores L, Pokala B, Huang CK, Siu KC, Oleynikov D. Gender equity in ergonomics: does muscle effort in laparoscopic surgery differ between men and women? Surg Endosc. 2022;36(1):396–401. [DOI] [PubMed] [Google Scholar]