Abstract
Lung cancer surgery has advanced significantly over the decades, profoundly impacting patient outcomes and surgical practices. This review examines the major historical milestones in lung cancer surgery—from early pneumonectomies to the establishment of lobectomy as the standard treatment for resectable non-small cell lung cancer (NSCLC) and the subsequent evolution toward sublobar resections (including segmentectomy and wedge resection) for early-stage NSCLC. Growing evidence for the efficacy of sublobar resections has redefined the surgical approach for early-stage lung cancer by shifting the focus from lobectomy to less invasive procedures. However, despite the demonstrated non-inferiority of sublobar resections, concerns about locoregional recurrence continue to pose a major challenge. Addressing this issue is essential for optimizing surgical outcomes in early-stage NSCLC. One promising innovation to mitigate recurrence is the novel asymmetrical linear stapler (NALS), which represents a significant advancement in stapling technology for minimally invasive lung cancer surgery. This review traces the evolution of lung cancer surgery from the 1960s to 2024, focusing on key milestones and the role of NALS in addressing current challenges.
Keywords: Thoracic surgery, Lung neoplasms
Introduction
Lung cancer, one of the most common and deadly cancers worldwide, has a long and evolving surgical history [1]. Surgical interventions have played a central role in lung cancer treatment, evolving from early 20th-century methods to modern minimally invasive techniques. This article traces the milestones and developments in lung cancer surgery, highlighting the progression of surgical techniques and their impact on patient outcomes.
The pioneering evolution of lung cancer surgery: milestones in early thoracic innovation
The history of lung cancer surgery is a remarkable journey marked by continual innovation and driven by early pioneers in thoracic surgery. Below are key milestones that contributed to the development of modern lung cancer surgery.
1861: Jules-Émile Péan’s tumor excision
The French surgeon Jules-Émile Péan performed one of the earliest recorded lung operations by excising a lung tumor. His approach involved suturing the pleura to the lung before tumor removal, marking a significant milestone in the emerging field of thoracic surgery, even though the limitations in anesthesia and infection control at the time affected the procedure’s safety [2].
1891: Theodore Tuffier’s partial lung resection
The French surgeon Theodore Tuffier performed the first successful partial lung resection for tuberculosis. This advancement was significant, given the widespread prevalence of tuberculosis in the 19th century, and Tuffier’s approach provided hope for the surgical treatment of infected lung tissue [3].
1895: William MacEwen’s first pneumonectomy
The Scottish surgeon Dr. William MacEwen performed the first complete pneumonectomy, removing an entire lung to treat tuberculosis. The survival of his patient marked a breakthrough, demonstrating that patients could endure major lung resections and laying the groundwork for future advances in lung surgery [4].
1901: Lothar Heidenhain’s anatomic lobectomy
In 1901, the German surgeon Dr. Lothar Heidenhain performed the first documented anatomic lobectomy to treat bronchiectasis. This procedure, which involved removing a specific lobe of the lung rather than the entire organ, laid the foundation for more precise lung procedures in the future [3].
1912: Hugh Morriston Davies’ lobectomy for cancer
In London, Hugh Morriston Davies achieved another significant advance by performing the first lobectomy specifically for lung cancer. This milestone demonstrated that cancerous lung tissue could be surgically excised, thereby contributing to evolving treatment strategies for lung cancer [5].
1913: Intratracheal anesthesia by S. J. Meltzer and J. Auer
S. J. Meltzer and J. Auer introduced a pivotal breakthrough in anesthesia by developing intratracheal anesthesia. This innovation made chest surgery, including major lung resections, safer and more feasible by allowing better control of the patient’s airway and ventilation during surgery [6].
1933: Evarts A. Graham’s first reported pneumonectomy for lung cancer
In a major milestone for lung cancer surgery, Dr. Evarts A. Graham performed the first reported pneumonectomy—the complete removal of one lung—for a patient with lung cancer. This groundbreaking operation demonstrated that complete lung resection could serve as a curative option for cancer patients, thereby establishing a new approach to treating lung malignancies [7].
1955: Paulson and Shaw’s sleeve resections and tracheal resections
The surgeons Dr. Donald L. Paulson and Dr. Robert R. Shaw extended their experimental work on sleeve and tracheal resections from animal models to humans, revolutionizing thoracic surgery. Sleeve resections emerged as a vital technique for preserving lung function while excising cancerous sections of the airway, thereby providing more options for patients with central lung tumors [8]. These milestones reflect the early challenges and breakthroughs in lung cancer surgery, highlighting the ingenuity of early surgeons and the steady improvements in surgical techniques, anesthesia, and postoperative care that have contributed to the development of modern thoracic surgery.
Early days of standard surgical treatment for lung cancer: open thoracotomy and pneumonectomy
Early in the 20th century, surgical treatment for lung cancer began when surgeons had limited knowledge and tools for addressing thoracic malignancies. The first reported pneumonectomy, the complete removal of one lung, was performed by Evarts A. Graham in 1933 on a patient with lung cancer. Although this radical surgery was a significant breakthrough, it was associated with high morbidity and mortality [7].
Open thoracotomy, which required large incisions to access the chest cavity, remained the standard for decades. Later, lobectomy—the removal of one lobe of the lung—was introduced as a less extensive alternative to complete lung removal, preserving more lung function while achieving similar oncological outcomes in selected patients.
The advent of lobectomy
By the mid-20th century, lobectomy gained prominence as surgeons recognized that removing only the affected lobe could be as effective as a pneumonectomy for early- stage lung cancer. This approach marked a significant step forward in balancing oncological control with quality of life preservation. By the 1960s, lobectomy became the preferred surgical approach for most patients with non-small cell lung cancer (NSCLC), particularly in early-stage cases.
This strategy was supported by William Cahan’s 1960 article in the Journal of Thoracic and Cardiovascular Surgery, which highlighted the advancements achieved by performing lobectomy rather than the traditional pneumonectomy in lung cancer treatment. Cahan’s work emphasized the importance of a radical approach to ensure complete resection of both the tumor and the surrounding lymph nodes. The study discussed technical steps and clinical implications based on an experience with 48 radical lobectomies performed between 1951 and 1958 at the Thoracic Surgical Service of the Memorial Center. Radical lobectomy was defined as the en bloc excision of one or two lobes of an entire lung along with a regional mediastinal and hilar lymph node dissection [9].
Key contributions of this study include: (1) Advocating the removal of both cancerous tissue and regional lymph nodes to improve long-term outcomes, thereby establishing radical lobectomy as the standard practice for localized lung cancer. (2) Highlighting the importance of achieving complete tumor resection to reduce the risk of local recurrence. (3) Reporting improved postoperative survival rates in patients who underwent this comprehensive procedure compared to those who received less aggressive surgery.
Cahan’s research contributed to a more aggressive surgical philosophy, shaping the future of thoracic oncology and refining the surgical standards for lung cancer treatment.
Emergence of segmentectomy and sublobar resections (1995–2024)
The advent of sublobar resection in lung cancer surgery represents a significant evolution in the treatment of early-stage NSCLC. Traditionally, lobectomy—the removal of an entire lobe of the lung—has been the standard of care for operable early-stage lung cancer, as established by surgeons such as William Cahan. However, research over time has demonstrated that sublobar resection may be an effective alternative for certain patients, particularly those with small, peripheral tumors or compromised lung function.
Early controversy and trials (including LCSG 821)
The Lung Cancer Study Group (LCSG) 821 established lobectomy as the preferred treatment for early-stage NSCLC by demonstrating higher local recurrence rates associated with sublobar resections. However, this long-standing preference has been challenged over the years as additional research into sublobar techniques has emerged [10].
Advances in imaging and screening
The introduction of low-dose computed tomography screening has enabled the detection of smaller, earlier-stage lung tumors, many of which appear as ground-glass opacities (GGOs). This advancement has renewed interest in sublobar resection, particularly for tumors measuring ≤2 cm, where preserving lung function is a critical consideration.
Technological advances in surgery
Technological advances in minimally invasive surgery, including video-assisted thoracoscopic surgery (VATS) and robotic-assisted thoracic surgery (RATS), have enhanced the safety and effectiveness of sublobar resection techniques by improving local control and reducing postoperative complications.
Landmark trials (JCOG 0802, JCOG 1211, CALGB 140503, and 2024 Society of Thoracic Surgeons database analysis)
JCOG 0802 trial (Japanese Clinical Oncology Group, 2022)
The JCOG 0802 trial was a randomized controlled study that compared segmentectomy—a form of sublobar resection—to lobectomy in patients with small peripheral NSCLC (tumors ≤2 cm). Although overall survival was improved in the segmentectomy group, the local recurrence rate was higher than in the lobectomy group, with rates of approximately 10.5% versus 5.4%, respectively. This finding underscores the challenge of achieving equivalent oncological outcomes with sublobar resection in terms of local control and long-term survival [11].
JCOG 1211 (Japanese Clinical Oncology Group, 2023)
This trial compared segmentectomy with wedge resection in patients with peripheral, GGO-dominant stage IA NSCLC (≤2 cm). The study confirmed that segmentectomy provided superior local control and overall survival compared to wedge resection, reinforcing segmentectomy as the more oncologically sound choice for small peripheral tumors [12].
CALGB 140503
This large randomized trial compared lobectomy with sublobar resection (either segmentectomy or wedge resection) in patients with stage IA NSCLC (≤2 cm). The study demonstrated that sublobar resection was non-inferior to lobectomy regarding overall survival and disease-free survival. Additionally, it highlighted the functional benefits of sublobar resections, including better preservation of lung function and fewer complications. Although the local recurrence rate was slightly higher in the sublobar resection group (approximately 7.0% compared to 4.0% for lobectomy), this difference was not statistically significant [13].
2024 Society of Thoracic Surgeons database analysis
This extensive database analysis compared sublobar techniques, namely segmentectomy versus wedge resection, in patients with stage IA NSCLC (tumors less than 3 cm). The analysis found that wedge resection was equivalent to segmentectomy for clinical stage IA NSCLC and suggested that wedge resection is a viable option for high-risk patients who may not tolerate lung resection well. This study provides real-world insights into the efficacy of sublobar techniques in a broad clinical context [14].
Current guidelines and use
The findings from JCOG 0802, JCOG 1211, CALGB 140503, and the 2024 Society of Thoracic Surgeons (STS) database analysis have significantly influenced current surgical guidelines. Sublobar resection is now widely regarded as the preferred technique for small, early-stage tumors, particularly those measuring ≤2 cm or ≤3 cm, given its comparable oncologic outcomes to lobectomy. Although wedge resection remains an option for selected cases—especially in patients with GGO-dominant tumors—segmentectomy is favored when feasible because of its superior local control and overall survival, as demonstrated in the JCOG trials. However, the CALGB 140503 trial showed no difference in long-term survival or recurrence-free survival between the lobectomy and sublobar resection groups, even though the number of patients undergoing wedge resection was higher than those undergoing segmentectomy in the sublobar group. CALGB 140503 may be the first randomized trial to provide evidence that wedge resection is a viable option for early NSCLC tumors less than 2 cm [13]. Additionally, the STS 2024 database analysis offers further rationale for using sublobar resection—particularly wedge resection—in selected cases of clinical stage IA NSCLC (≤3 cm) [14].
The evolution of sublobar resection for early-stage NSCLC has been shaped by multiple studies, beginning with the LCSG 821 trial, which favored lobectomy due to concerns about recurrence rates. However, recent trials—including JCOG 0802, JCOG 1211, and CALGB 140503—as well as the 2024 STS database analysis, have demonstrated that sublobar resection can yield oncologic outcomes comparable to lobectomy for stage IA NSCLC (≤3 cm) while preserving lung function more effectively. Consequently, sublobar resection, including wedge resection, has emerged as a key option in the modern treatment of early-stage lung cancer, offering comparable oncologic control with reduced morbidity and better lung function preservation [12-14].
The minimally invasive revolution: VATS and robotic-assisted surgery
One of the most transformative periods in lung cancer surgery began with the advent of minimally invasive techniques in the 1990s and 2000s. VATS was introduced as an alternative to open thoracotomy. Instead of requiring a large incision, VATS utilizes small incisions through which a camera and surgical instruments are inserted, enabling surgeons to perform lobectomy and other resections with significantly less trauma. VATS rapidly gained popularity due to advantages such as reduced postoperative pain, shorter hospital stays, and faster recovery times compared to open surgery. Moreover, the oncological outcomes of VATS lobectomy have been shown to be equivalent to those of traditional open lobectomy in early-stage lung cancer, establishing it as the standard approach in many centers.
Building on the success of VATS, RATS emerged in the 2000s. RATS employs robotic systems that offer enhanced dexterity, precision, and visualization. Similar to VATS, robotic surgery facilitates minimally invasive lung cancer resections, but it also provides improved control in complex cases. Although robotic surgery is more expensive than VATS, its use is growing due to its potential benefits for certain patient populations.
Current unmet needs for sublobar resection
Sublobar resection, including both wedge resection and segmentectomy, has been explored as a lung-sparing surgical approach for early-stage NSCLC, particularly in patients with limited lung function or small peripheral tumors. Although these procedures offer the advantage of preserving more lung tissue compared to lobectomy—the standard treatment for early-stage NSCLC—the risk of local recurrence remains a concern, underscoring the unmet need to optimize outcomes for patients undergoing sublobar resections.
Local recurrence rate: insights from JCOG 0802 and CALGB 140503 trials
Two major clinical trials, JCOG 0802 and CALGB 140503, have provided valuable insights into local recurrence rates and the efficacy of sublobar resection compared to lobar resection [11,13]. In the JCOG 0802 trial, the local recurrence rates were 10.5% for sublobar resection compared to 5.4% for lobectomy, while in the CALGB 140503 trial, the rates were 7.0% versus 4.0%, respectively.
Although sublobar resection offers promising outcomes in terms of survival and lung function preservation, the higher local recurrence rates compared to lobectomy represent a significant unmet need in lung cancer surgery. The challenge is to identify patients who can safely benefit from sublobar resection without an increased risk of local relapse. Refining patient selection criteria, improving surgical techniques, and incorporating adjunct therapies—such as targeted radiation or adjuvant chemotherapy—may help address this gap. The goal is to provide less invasive procedures while maintaining the same level of oncological safety and local control as lobectomy, particularly in early-stage disease.
While the results of the JCOG 0802 and CALGB 140503 trials are valuable, they also highlight the need for ongoing research to refine sublobar resection techniques and optimize outcomes for patients with early-stage NSCLC.
New frontiers: lung-sparing techniques and hybrid approaches
In the past decade, lung cancer surgery has continued to evolve toward more personalized and conservative approaches. Lung-sparing procedures, including refined segmentectomy techniques, are under ongoing investigation to assess their safety and efficacy. Moreover, the development of hybrid approaches—which combine surgical resection with adjunct therapies such as stereotactic body radiotherapy, targeted therapies, and ablation treatments—has opened new possibilities for treating patients with localized and early-stage disease [15,16].
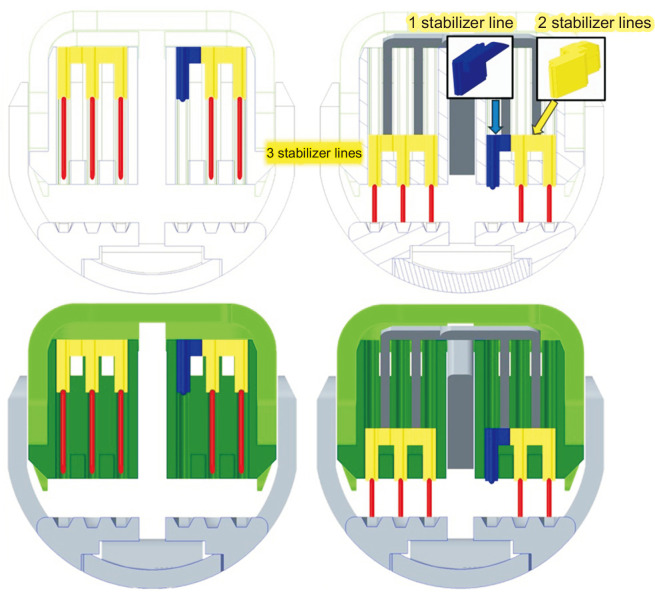
Additionally, innovations like the novel asymmetrical linear stapler are enhancing surgical precision and safety during lung resections, particularly in complex central tumors. These advancements reflect a broader trend toward minimizing invasiveness while maximizing oncological outcomes [17,18] (Fig. 1).
Fig. 1.
Cross-sectional view of a novel asymmetrical linear stapler. Red line: titanium fastener; Blue unit: stabilizer unit for surgical margin tissue protection; yellow unit: pusher for titanium fastener.
Conclusion
From the pioneering pneumonectomy to the advent of minimally invasive techniques, the history of lung cancer surgery illustrates remarkable progress in both technology and surgical philosophy. Today, the focus on less invasive, lung-sparing procedures and personalized treatment plans represents a shift toward balancing long-term survival with quality of life. As technology and research continue to advance, further refinements in lung cancer surgery are likely, offering renewed hope to patients confronting this challenging disease.
The evolution of lung cancer surgery underscores the importance of continuous innovation and the careful application of new techniques, ensuring that each advancement is aimed at improving outcomes and enhancing patient care.
Funding Statement
Funding This research was supported by the Post TIPS Program, funded by the Ministry of SMEs and Startups (MSS, Republic of Korea).
Article information
Author contributions
All the work was done by Min-Woong Kang.
Conflict of interest
Min-Woong Kang is the inventor of the novel asymmetrical linear stapler and he works as the CEO of Meditulip Company, which developed the novel asymmetrical linear stapler. Except for that, no potential conflict of interest relevant to this article was reported.
References
- 1.Leiter A, Veluswamy RR, Wisnivesky JP. The global burden of lung cancer: current status and future trends. Nat Rev Clin Oncol. 2023;20:624–39. doi: 10.1038/s41571-023-00798-3. https://doi.org/10.1038/s41571-023-00798-3. [DOI] [PubMed] [Google Scholar]
- 2.Chang JH. Historical aspects. In: Myers NA, Angerpointner TA, editors. Paediatric thoracic surgery. Springer-Verlag Berlin Heidelberg; 1991. p. 5-29.
- 3.Khaitan PG, D'Amico TA. Milestones in thoracic surgery. J Thorac Cardiovasc Surg. 2018;155:2779–89. doi: 10.1016/j.jtcvs.2017.12.149. https://doi.org/10.1016/j.jtcvs.2017.12.149. [DOI] [PubMed] [Google Scholar]
- 4.Macewen W. The Cavendish lecture on some points in the surgery of the lung. Br Med J. 1906;2:1–7. doi: 10.1136/bmj.2.2375.1. https://doi.org/10.1136/bmj.2.2375.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Davies HM. Recent advances in the surgery of the lung and pleura. Br J Surg. 1913;1:228–58. doi: 10.1002/bjs.1800010211. https://doi.org/10.1002/bjs.1800010211. [DOI] [Google Scholar]
- 6.Meltzer SJ, Auer J. Continuous respiration without respiratory movements. J Exp Med. 1909;11:622–5. doi: 10.1084/jem.11.4.622. https://doi.org/10.1084/jem.11.4.622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brewer LA 3rd, author. Historical notes on lung cancer before and after Graham's successful pneumonectomy in 1933. Am J Surg. 1982;143:650–9. doi: 10.1016/0002-9610(82)90030-7. https://doi.org/10.1016/0002-9610(82)90030-7. [DOI] [PubMed] [Google Scholar]
- 8.Paulson DL, Shaw RR. Preservation of lung tissue by means of bronchoplastic procedures. Am J Surg. 1955;89:347–55. doi: 10.1016/0002-9610(55)90074-X. https://doi.org/10.1016/0002-9610(55)90074-x. [DOI] [PubMed] [Google Scholar]
- 9.Cahan WG. Radical lobectomy. J Thorac Cardiovasc Surg. 1960;39:555–72. doi: 10.1016/S0022-5223(20)31797-9. [DOI] [PubMed] [Google Scholar]
- 10.Ginsberg RJ, Rubinstein LV. Randomized trial of lobectomy versus limited resection for T1 N0 non-small cell lung cancer. Lung Cancer Study Group. Ann Thorac Surg. 1995;60:615–23. doi: 10.1016/0003-4975(95)00537-U. https://doi.org/10.1016/0003-4975(95)00537-u. [DOI] [PubMed] [Google Scholar]
- 11.Saji H, Okada M, Tsuboi M, et al. Segmentectomy versus lobectomy in small-sized peripheral non-small-cell lung cancer (JCOG0802/WJOG4607L): a multicentre, open-label, phase 3, randomised, controlled, non-inferiority trial. Lancet. 2022;399:1607–17. doi: 10.1016/S0140-6736(21)02333-3. https://doi.org/10.1016/S0140-6736(21)02333-3. [DOI] [PubMed] [Google Scholar]
- 12.Aokage K, Suzuki K, Saji H, et al. Segmentectomy for ground- glass-dominant lung cancer with a tumour diameter of 3 cm or less including ground-glass opacity (JCOG1211): a multicentre, single-arm, confirmatory, phase 3 trial. Lancet Respir Med. 2023;11:540–9. doi: 10.1016/S2213-2600(23)00041-3. https://doi.org/10.1016/S2213-2600(23)00041-3. [DOI] [PubMed] [Google Scholar]
- 13.Altorki N, Wang X, Kozono D, et al. Lobar or sublobar resection for peripheral stage IA non-small-cell lung cancer. N Engl J Med. 2023;388:489–98. doi: 10.1056/NEJMoa2212083. https://doi.org/10.1056/NEJMoa2212083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Towe CW, Grau-Sepulveda MV, Hartwig MG, et al. The Society of Thoracic Surgeons database analysis: comparing sublobar techniques in stage IA lung cancer. Ann Thorac Surg. 2024;118:665–71. doi: 10.1016/j.athoracsur.2024.03.008. https://doi.org/10.1016/j.athoracsur.2024.03.008. [DOI] [PubMed] [Google Scholar]
- 15.Chang JY, Senan S, Paul MA, et al. Stereotactic ablative radiotherapy versus lobectomy for operable stage I non-small-cell lung cancer: a pooled analysis of two randomised trials. Lancet Oncol. 2015;16:630–7. doi: 10.1016/S1470-2045(15)70168-3. https://doi.org/10.1016/S1470-2045(15)70168-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vogl TJ, Nour-Eldin NA, Albrecht MH, et al. Thermal ablation of lung tumors: focus on microwave ablation. Rofo. 2017;189:828–43. doi: 10.1055/s-0043-109010. https://doi.org/10.1055/s-0043-109010. [DOI] [PubMed] [Google Scholar]
- 17.Kang SK, Bok JS, Cho HJ, Kang MW. Novel asymmetrical linear stapler (NALS) for pathologic evaluation of true resection margin tissue. J Thorac Dis. 2018;10(Suppl 14):S1631–6. doi: 10.21037/jtd.2018.03.158. https://doi.org/10.21037/jtd.2018.03.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jung YC, Piao WX, Kim JY, et al. Novel asymmetrical linear stapler: safety test and pathological assessment in a porcine model. J Surg Res. 2024;304:58–66. doi: 10.1016/j.jss.2024.10.003. https://doi.org/10.1016/j.jss.2024.10.003. [DOI] [PubMed] [Google Scholar]