Abstract
1. The occurrence of haemoglobin in invertebrate nerves is surveyed. Haemoglobin was observed in the nerves and ganglia of the marine nematode Amphiporus sp. and of the polychaet annelid Halosydna sp. 2. Haemoglobins from the nerve and ganglia of the polychaet annelid Aphrodite aculeata L. and from the nerve of the gastropod mollusc Aplysia californica have been partially purified. The haem in each case was identified as iron protoporphyrin IX. 3. The minimum molecular weight of Aphrodite nerve haemoglobin deduced from the haem content and amino acid analysis is 17090, in agreement with the molecular weight 15600±1000 determined by sedimentation equilibrium. 4. The molecular weight of Aplysia nerve haemoglobin was determined by sedimentation equilibrium to be 16400±1000. 5. The oxygen dissociation curves are hyperbolic. Half-saturation is achieved at 1·1mm. Hg for Aphrodite nerve haemoglobin and at 4·0mm. Hg for Aplysia nerve haemoglobin. The coefficients for partition between carbon monoxide and oxygen are: Aphrodite nerve haemoglobin, 167; Aplysia nerve haemoglobin, 116. 6. The ferrous haemoglobins combine with cyanide. 7. We conclude that the intracellular haemoglobins of muscle and nerve are similar.
Full text
PDF


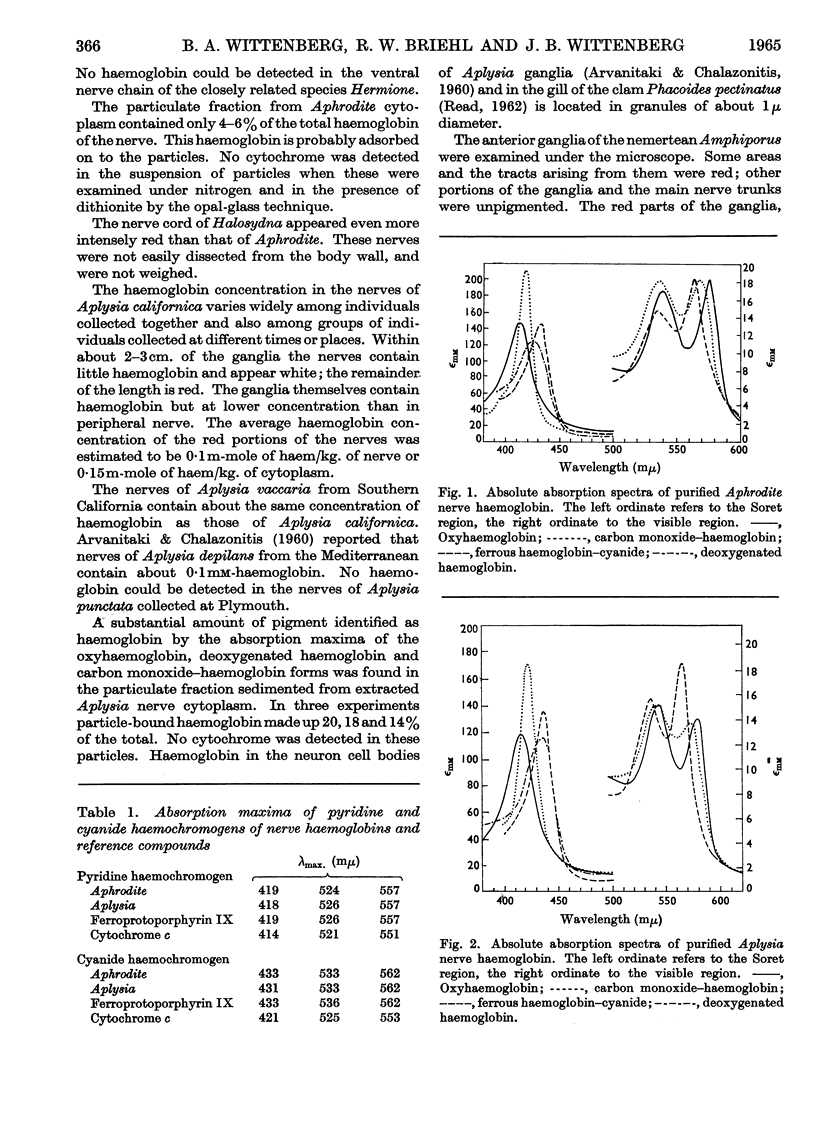
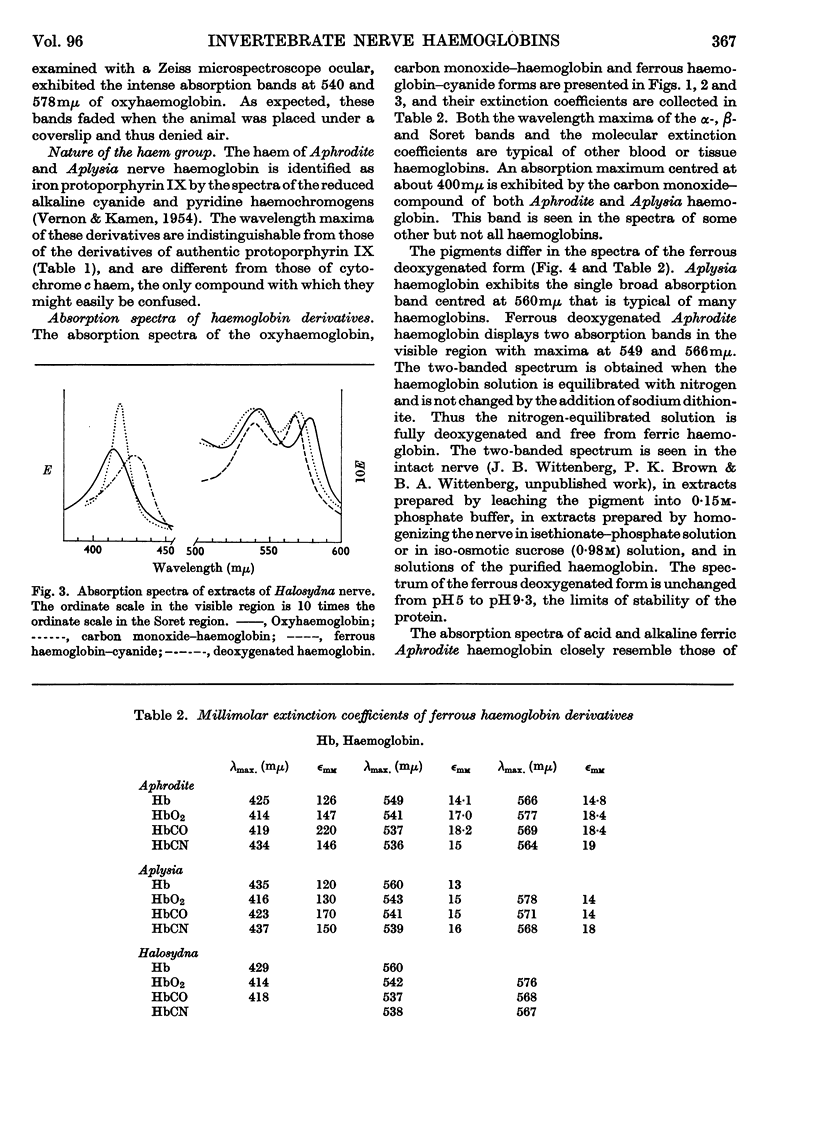
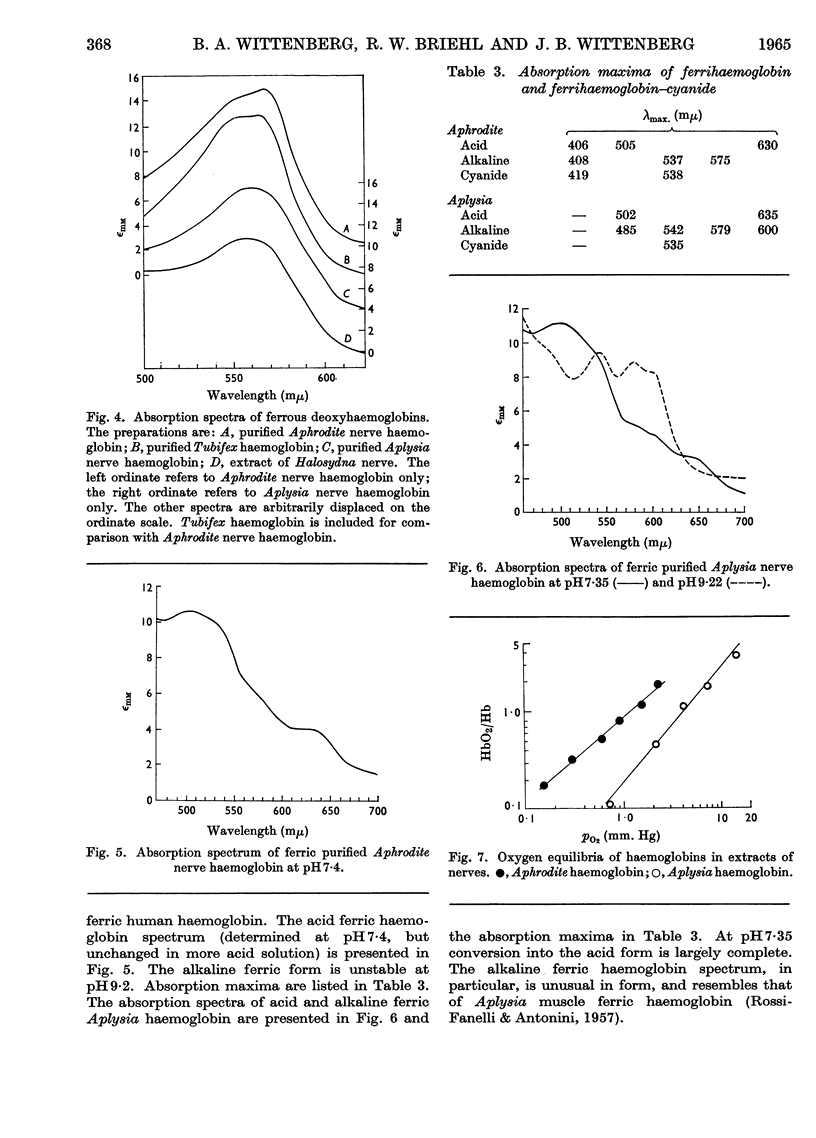
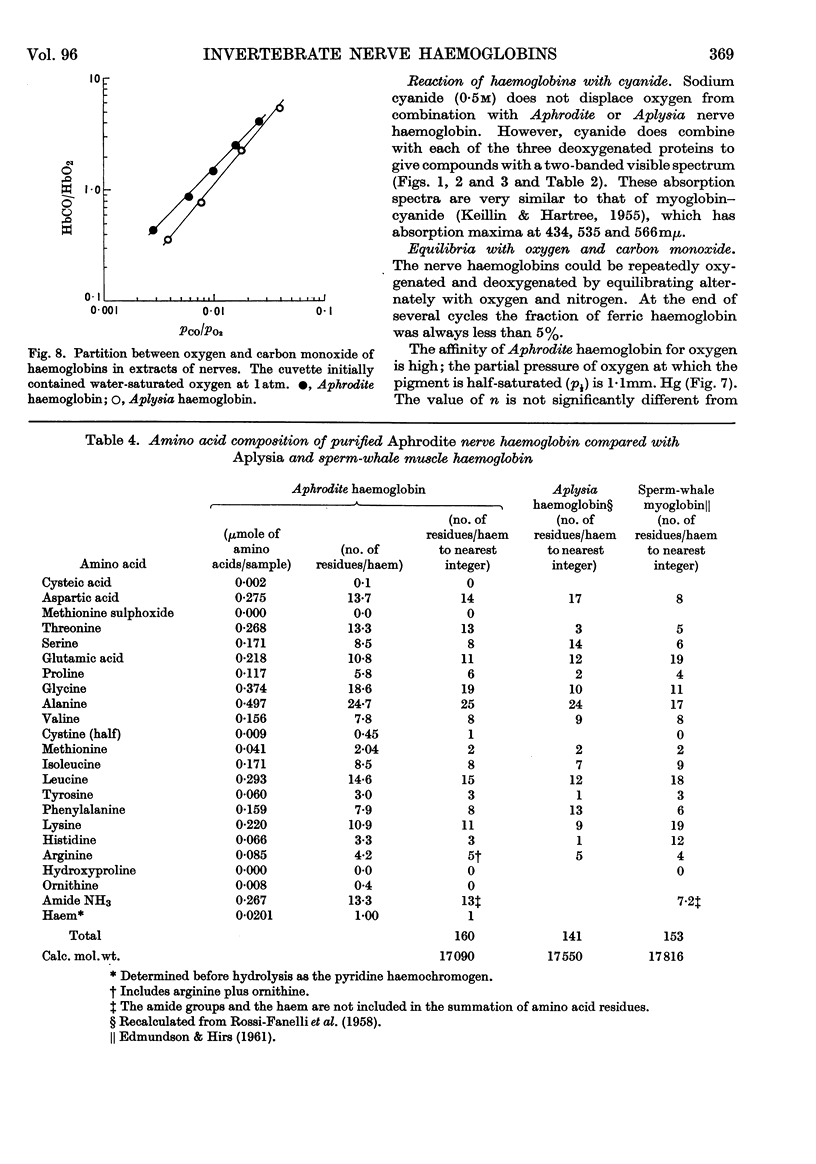


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ALLEN D. W., GUTHE K. F., WYMAN J., Jr Further studies on the oxygen equilibrium of hemoglobin. J Biol Chem. 1950 Nov;187(1):393–410. [PubMed] [Google Scholar]
- BAKER P. F., HODGKIN A. L., SHAW T. I. Replacement of the axoplasm of giant nerve fibres with artificial solutions. J Physiol. 1962 Nov;164:330–354. doi: 10.1113/jphysiol.1962.sp007025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- COGGESHALL R. E., FAWCETT D. W. THE FINE STRUCTURE OF THE CENTRAL NERVOUS SYSTEM OF THE LEECH, HIRUDO MEDICINALIS. J Neurophysiol. 1964 Mar;27:229–289. doi: 10.1152/jn.1964.27.2.229. [DOI] [PubMed] [Google Scholar]
- EDMUNDSON A. B., HIRS C. H. The amino-acid sequence of sperm whale myoglobin. Chemical studies. Nature. 1961 May 20;190:663–665. doi: 10.1038/190663a0. [DOI] [PubMed] [Google Scholar]
- FOX H. M. Haemoglobin in the Crustacea. Nature. 1957 Jan 19;179(4551):148–148. doi: 10.1038/179148a0. [DOI] [PubMed] [Google Scholar]
- FOX H. M. The effect of oxygen on the concentration of haem in invertebrates. Proc R Soc Lond B Biol Sci. 1955 Jan 27;143(911):203–214. doi: 10.1098/rspb.1955.0005. [DOI] [PubMed] [Google Scholar]
- KEILIN D., HARTREE E. F. Cyanide compounds of ferroperoxidase and myoglobin and their reversible photodissociation. Biochem J. 1955 Sep;61(1):153–171. doi: 10.1042/bj0610153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KENNEDY D. Neural photoreception in a lamellibranch mollusc. J Gen Physiol. 1960 Nov;44:277–299. doi: 10.1085/jgp.44.2.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MANWELL C. Heme-heme interactions in the oxygen equilibrium of some invertebrate myoglobins. Arch Biochem Biophys. 1960 Aug;89:194–201. doi: 10.1016/0003-9861(60)90042-4. [DOI] [PubMed] [Google Scholar]
- McMEEKIN T. L., MARSHALL K. Specific volumes of proteins and the relationship to their amino acid contents. Science. 1952 Aug 8;116(3006):142–143. doi: 10.1126/science.116.3006.142. [DOI] [PubMed] [Google Scholar]
- ROSSI-FANELLI A., ANTONINI E. A new type of myoglobin isolated and crystallized from the muscles of Aplysiae. Biokhimiia. 1957 Jan-Feb;22(1-2):336–344. [PubMed] [Google Scholar]
- SCHELER W. [Light absorption and oxygen binding curve of hemoglobin from Tubifex tubifex]. Biochem Z. 1960;332:366–375. [PubMed] [Google Scholar]
- STRITTMATTER P., BURCH H. B. THE HEME PROTEIN IN GANGLIA OF SPISULA SOLIDISSIMA. Biochim Biophys Acta. 1963 Nov 15;78:562–563. doi: 10.1016/0006-3002(63)90928-4. [DOI] [PubMed] [Google Scholar]
- VERNON L. P., KAMEN M. D. Hematin compounds in photosynthetic bacteria. J Biol Chem. 1954 Dec;211(2):643–662. [PubMed] [Google Scholar]
- YPHANTIS D. A. EQUILIBRIUM ULTRACENTRIFUGATION OF DILUTE SOLUTIONS. Biochemistry. 1964 Mar;3:297–317. doi: 10.1021/bi00891a003. [DOI] [PubMed] [Google Scholar]


