Abstract
Objective: To assess the effects of silver needle thermal conduction therapy on spinal neurotransmitter activation and inflammation reduction in the myofascial trigger point musculature of a rat model with myofascial pain syndrome (MPS). Methods: The MPS rat model was induced by a combination of impact and exercise fatigue. MPS rats were then subjected to silver needle thermal conduction therapy for 14 days. Thermal and mechanical pain thresholds were assessed using the thermal hyperalgesia and mechanical hyperalgesia tests. Transverse and longitudinal sections of the right medial thigh muscle were prepared for histological analysis using HE staining. Western blot and immunohistochemistry were used to measure the expression of calcitonin gene-related peptide (CGRP), substance P (SP), and 5-hydroxytryptamine (5-HT) in the L4-6 segment of the spinal cord. Additionally, the levels of inflammatory cytokines, including interleukin 1-β (IL-1β) and tumor necrosis factor α (TNF-α), in the right medial femoral muscle were assessed via ELISA. Results: MPS rats exhibited a decreased thermal and mechanical pain threshold compared to control rats. Muscle fiber atrophy and deformation were observed in the MPS rats. Levels of IL-1β and TNF-α were elevated in the right medial femoral muscle, while CGRP and SP expression were increased, and 5-HT expression was decreased in the spinal cord. Silver needle thermal conduction therapy effectively reversed these pathological changes in the MPS rats. Conclusions: Silver needle thermal conduction therapy alleviated MPS in rats by modulating spinal neurotransmitter expression and reducing muscle inflammation.
Keywords: Myofascial pain syndrome, silver needle thermal conduction therapy, chronic pain, acupuncture effect, thermal effect
Introduction
Myofascial pain syndrome (MPS) is a common condition characterized by localized or referred pain, typically associated with musculoskeletal disorders [1]. It is defined by the presence of myofascial trigger points (MTrPs) - palpable, discrete, localized nodules within taut bands of skeletal muscle, which can cause pain when compressed [2]. Approximately 85% of the population experiences myofascial pain at least once in their lifetime [3]. The prevalence of MPS varies depending on clinical context, ranging from 21% in general orthopedic patients to 30% in general medical clinic patients, and up to 93% in specialized pain management centers [4]. MPS encompasses both acute and chronic pain and is often under-recognized as a significant cause of disability in clinical settings [5].
While the precise mechanisms underlying MPS and MTrPs remain incompletely understood, current theories suggest a link between peripheral nociception and central sensitization [6]. The primary objective of MPS treatment is to alleviate pain and restore function by targeting active MTrPs [7]. A variety of therapeutic approaches have been employed for MPS, including ischemic compression [8], therapeutic ultrasound [9], muscle energy techniques [10], stretching [10], manipulation [11], acupuncture [12], dry needling [13], wet needling [14], and oral medications. Among these, dry needling has gained increasing popularity as a non-surgical treatment due to its straightforward application and low risk profile [15]. Acupuncture targeting primary MTrPs has also proven to be effective, safe, and cost-efficient for managing myofascial pain [16].
Dry needling involves the insertion of fine needles into MTrPs within the skin and muscles to mechanically disrupt the affected tissues, without the use of anesthetics [17]. This technique, which closely resembles traditional acupuncture methods for treating “Ah shi” points, induces localized muscle twitch responses akin to the “de qi” sensation in traditional Chinese medicine (TCM). Notably, studies have shown a significant correlation between MTrPs and acupuncture points [18]. However, the relationship between dry needling and acupuncture remains controversial. Many dry needling practitioners do not consider it a form of acupuncture, as it is based on Western neuroanatomy rather than the ancient theories of TCM [19].
Despite this controversy, silver needle thermal (SNT) conduction therapy - an approach within modern Chinese acupuncture-moxibustion - shares striking similarities with dry needling. SNT conduction therapy was developed by integrating the “large needle”, “warming needle”, and “long needle” concepts from the “Traditional Chinese pivot nine needles” system, combining their advantages. Unlike traditional acupuncture needles, silver needles are made of 85% silver, with small amounts of copper, chromium, and nickel. These needles are thicker and longer (approximately 1.1 mm in diameter) than standard acupuncture needles, offering increased flexibility and durability. The handles are typically 5 cm long, and the needle bodies come in five lengths: 6 cm, 8 cm, 10 cm, 12 cm, and 15 cm, allowing for deeper penetration than traditional acupuncture needles.
Silver needle acupuncture was first developed by LU Yin-hua in the 1940s in Shanghai. In the 1970s, Xuan Zhe-ren, an orthopedic expert, expanded this technique, particularly for severe lumbocrural lumbosacral pain, by transforming it into a thermal conduction therapy. Unlike traditional acupuncture, which targets acupoints along meridians, SNT conduction therapy involves inserting needles based on the distribution of tender points rather than specific acupoints. After needle insertion, a 1.5 cm moxa-ball is placed at the needle’s end and ignited to produce slow, sustained heat. This therapy, rooted in soft tissue surgical anatomy and the distribution patterns of tender points, has shown promising results in providing both immediate pain relief and long-term analgesic effects for MPS patients [20]. Interestingly, its long-term effectiveness in treating MPS seems to surpass that of dry needling. However, studies investigating SNT conduction therapy for MPS remain limited, and the underlying molecular mechanisms are still unclear. Therefore, this study aims to assess the therapeutic effects of SNT conduction therapy in an animal model of MPS and explore its potential molecular mechanisms.
Materials and methods
Animals and ethical statement
Adult male Sprague-Dawley rats (weight: 300-350 g; Guizhou Medical University Animal Center, Guiyang, China) were used in this study. The rats were individually housed in galvanized wire mesh cages under controlled environmental conditions (12-hour light/dark cycle, 21-25°C, 50-55% humidity). All rats were cared for according to the guidelines of the International Society of Animal Pain Studies to minimize pain and discomfort. Animal experiments were approved by the Animal Care and Use Committee.
Experimental design
Healthy adult male Sprague-Dawley rats were randomly assigned to three groups (n=8 per group): the normal control (NC) group, the MPS model (MPS) group, and the SNT group. Rats in the NC group were maintained under the same environmental conditions as the other groups but without any experimental interventions. Chronic MPS was induced in the MPS group through a combination of impact and exercise fatigue. The SNT group rats received SNT therapy. All rats were housed under identical conditions and euthanized simultaneously at the end of the study. After 14 days of SNT therapy, rats underwent thermal hyperalgesia and mechanical hyperalgesia tests. Rats were euthanized via intraperitoneal injection of sodium pentobarbital (150 mg/kg), and their spinal cords (L4-L6 segments) and right medial femoral muscles were collected and stored immediately at -80°C.
Establishment of the chronic MPS model
The MPS model was established in two phases: an intervention period (8 weeks) and a recovery period (4 weeks). Prior to the experiment, rats were acclimatized to exercise on a multi-channel running treadmill (WI32812, Dongxiyi Technology Co., Ltd., Beijing) for 3 days. During the intervention period, rats were anesthetized with an intraperitoneal injection of 30 mg/kg 1% sodium pentobarbital (Sigma-Aldrich, USA). They were then positioned at the bottom of a striking device. A wooden striker with a kinetic energy of 2.352 J was dropped from a height of 20 cm onto the right medial femoral muscle. On day 2, rats underwent centrifugal treadmill training with the treadmill set to a -16°C downhill running mode at a speed of 16 m/min. Both MPS and SNT groups participated in this training. During the recovery period, no interventions were applied, and rats were allowed to engage in normal daily activities with regular feeding for four weeks. Thermal and mechanical pain thresholds were measured to confirm the establishment of the MPS model.
SNT therapy
Rats in the SNT group were anesthetized with 30 mg/kg 1% sodium pentobarbital via abdominal injection. The biceps femoris muscle in the hindlimb was palpated to locate taut nodules, approximately 3-4 mm in diameter, which were targeted for treatment. After disinfection, a 10 cm, 0.6 mm diameter silver needle was inserted into the starting and ending points, as well as the tension zone of the right medial femoral muscle, until the needle tip reached the femoral surface. The SNT conduction instrument was used to heat the needle to a temperature of 110°C. During the procedure, the highest skin temperature at the needle injection point was 34°C, and the heating duration was 15 minutes. After needle removal, the puncture site was disinfected with 75% alcohol and covered with aseptic dressings. The rats were treated continuously for 14 days. The exact procedure for SNT therapy is illustrated in Figure 1.
Figure 1.
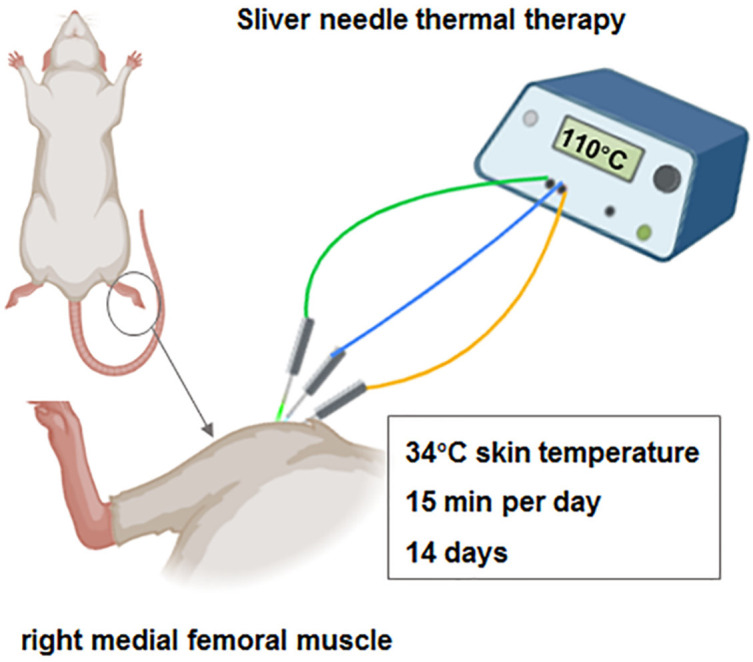
The sketch map for the treatment method of silver needle thermal therapy.
Thermal hyperalgesia test
The thermal hyperalgesia test was used to assess the paw withdrawal threshold. A hot plate was set to 52 ± 0.2°C, and rats were placed on the plate. The time taken for the rats to lift their posterior paw or begin licking it was recorded. The maximum observation time was 45 seconds to prevent tissue damage. Each rat underwent the test three times, with at least a 15-minute rest period between trials. The average paw withdrawal latency was calculated for each rat.
Mechanical hyperalgesia test
To evaluate the persistence of chronic pain, each rat was first placed in an individual plexiglass chamber (26 × 20 × 14 cm3) for a 20-minute acclimation period before testing. After the thermal pain threshold assessment, a two-hour interval was observed to ensure the reliability of subsequent measurements. Mechanical hyperalgesia was assessed using calibrated von Frey filaments (Stoelting, Wood Dale, IL, USA). The paw withdrawal mechanical threshold was recorded.
Hematoxylin and Eosin (HE) staining
Histological changes in the affected right medial femoral muscle were evaluated using HE staining. Tissue samples were fixed in 4% paraformaldehyde and embedded in paraffin for 12 hours. Transverse and longitudinal sections (5 μm) of the right medial thigh muscle were prepared. The sections were dewaxed in xylene, rehydrated with decreasing concentrations of ethanol, and stained with HE staining solution (Solarbio, Beijing, China). Muscle fiber morphology and arrangement were examined under an optical microscope at 100× magnification. The transverse sectional area of muscle fibers in transverse sections and the length of muscle fibers in longitudinal sections were quantified using ImageJ software.
Western blot assay
Spinal cord tissues were lysed in RIPA buffer. Protein concentrations were determined using a bicinchoninic acid assay. Next, 20 μg of total protein were separated by 10% SDS-PAGE and transferred to a PVDF membrane. After blocking with 5% skim milk, membranes were incubated overnight at 4°C with primary antibodies against calcitonin gene-related peptide (CGRP), SP, and 5-HT (1:500 dilution, Abcam, UK). The membranes were then incubated with specific secondary antibodies for 1 hour at room temperature. Bands were detected using enhanced chemiluminescence (ECL) reagents, and the intensity of the bands was quantified using ImageJ software. GAPDH was used as a loading control.
Immunohistochemistry assay
After dewaxing and rehydration, spinal cord tissue sections were treated with sodium citrate solution (10 mM, pH 6.0) and heated in a microwave. Endogenous peroxidase activity was blocked with 3% H2O2. The sections were incubated overnight at 4°C with primary antibodies against CGRP, SP, and 5-HT (Abcam, UK), followed by incubation with secondary antibodies for 1 hour at room temperature. The sections were then developed using a DAB kit (Beyotime, China), and tissue images were obtained using a light microscope.
Enzyme-linked immunosorbent assay (ELISA)
The levels of IL-1β and TNF-α in the right medial femoral muscle were measured using commercial ELISA kits (Solarbio, Beijing, China), following the manufacturer’s instructions.
Statistical analysis
Data were analyzed using SPSS 18.0 software and are presented as the mean ± standard deviation (mean ± SD). One-way analysis of variance (ANOVA) was used to compare differences between groups, and Student’s t-test was used for pairwise comparisons. Statistical significance was set at P<0.05.
Results
Effect of SNT conduction therapy on thermal and mechanical hyperalgesia
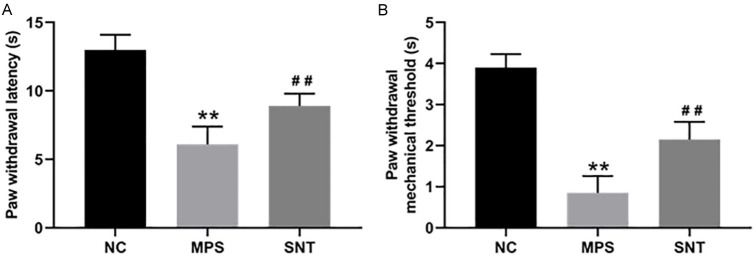
The MPS rat model was established and subsequently treated with SNT conduction therapy. The treatment protocol for SNT therapy is shown in Figure 1. As shown in Figure 2A and 2B, the paw withdrawal latency and mechanical threshold were significantly reduced in the MPS rats compared to the control rats, indicating successful establishment of the MPS model. However, in the SNT group, both the paw withdrawal latency and mechanical threshold were significantly increased compared to the MPS group.
Figure 2.
The thermal and mechanical hyperalgesia of rats in each group. A. The paw withdrawal latency of rats in each group. B. The paw withdrawal mechanical threshold of rats in each group. N=8. Data are presented as mean ± SD. **P<0.01, compared with the NC group. ##P<0.01, compared with the MPS group. NC, normal control; MPS, myofascial pain syndrome; SNT, silver needle thermal therapy.
Effect of SNT conduction therapy on pathomorphological changes in muscles
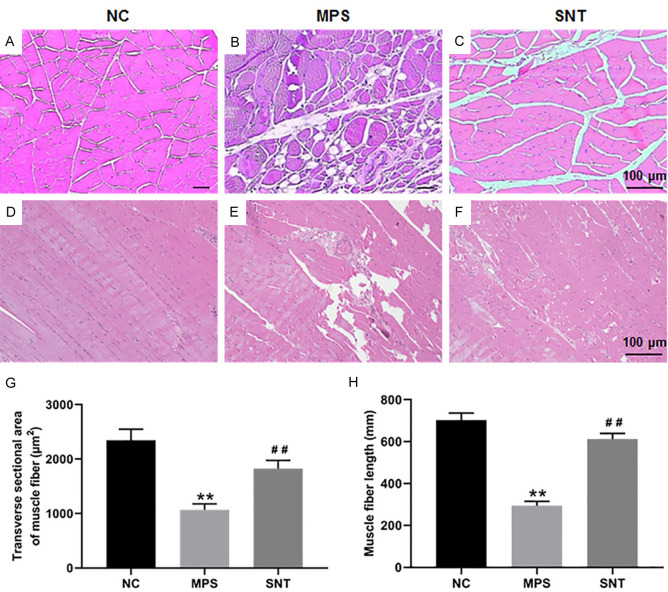
In the NC group, both the transverse section (Figure 3A) and longitudinal section (Figure 3D) of muscle tissue exhibited a uniform and regular structure, with tightly arranged muscle cells and uniform intermuscular gaps. The muscle fibers were neatly and densely arranged. In the MPS group, the transverse section showed multiple circular or elliptical muscle fibers of varying sizes, indicating muscle fiber atrophy and deformation (Figure 3B). The longitudinal section revealed disordered muscle fiber arrangement and atrophied myofilaments (Figure 3E). In the SNT group, muscle fibers displayed mild atrophy and denaturation, but the structures appeared similar to those in the NC group (Figure 3C, 3F). We found that both the transverse sectional area of muscle fibers (Figure 3G) and muscle fiber length (Figure 3H) were reduced in the MPS group compared to the NC group. However, SNT treatment reversed these effects.
Figure 3.
Hematoxylin and Eosin (HE) staining of the transverse and longitudinal sectional sections of right medial thigh muscle tissues in each group. A-C. The transverse sectional sections of right medial thigh muscle tissues in each group. D-F. The longitudinal sectional sections of right medial thigh muscle tissues in each group. G. Transverse sectional area of muscle fibers in each group. H. The length of muscle fibers in longitudinal sections in each group. Data are presented as mean ± SD. **P<0.01, compared with the NC group. ##P<0.01, compared with the MPS group. NC, normal control; MPS, myofascial pain syndrome; SNT, silver needle thermal therapy.
Effect of SNT conduction therapy on IL-1β and TNF-α levels in muscles
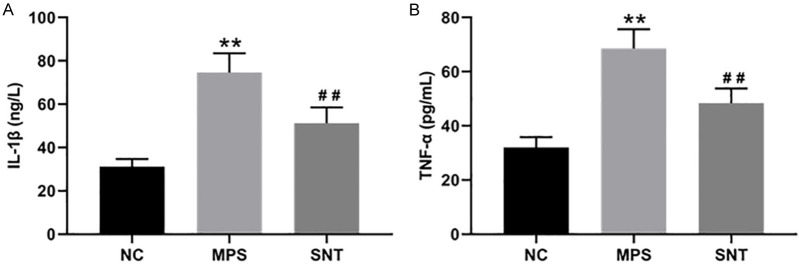
The levels of IL-1β (Figure 4A) and TNF-α (Figure 4B) in the muscles of MPS rats were significantly elevated compared to the NC group. However, SNT treatment reduced the levels of both IL-1β and TNF-α in the muscles of MPS rats.
Figure 4.
The expression of interleukin 1-β (IL-1β) and tumor necrosis factor α (TNF-α) in the muscles of the rats in each group. A. The expression of IL-1β in the muscles of the rats in each group was examined using an ELISA kit. B. The expression of TNF-α in the muscles of the rats in each group was examined using an ELISA kit. N=8. Data are presented as mean ± SD. **P<0.01, compared with the NC group. ##P<0.01, compared with the MPS group. NC, normal control; MPS, myofascial pain syndrome; SNT, silver needle thermal therapy.
Effect of SNT conduction therapy on CGRP, SP, and 5-HT in the spinal cord
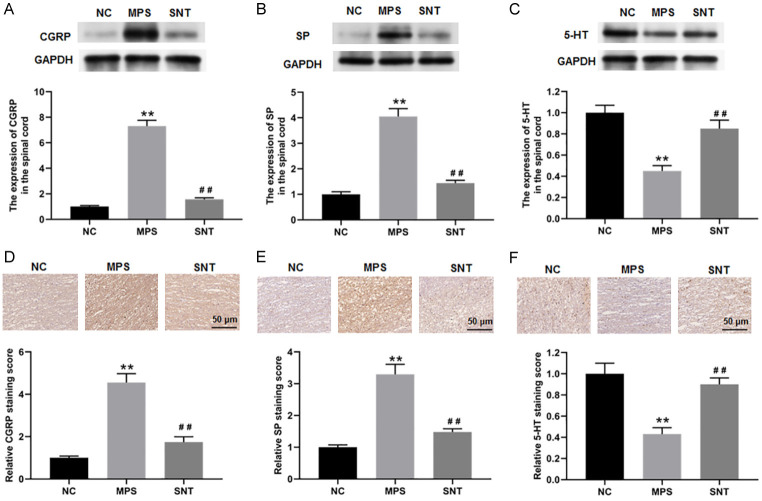
Western blot analysis revealed that the expression levels of CGRP (Figure 5A) and SP (Figure 5B) were significantly increased, while the 5-HT level (Figure 5C) was decreased in the spinal cord of the MPS group compared to the NC group. However, SNT therapy reduced CGRP and SP expression and increased 5-HT expression in the spinal cord of MPS rats. Immunohistochemical results also confirmed these findings (Figure 5D-F).
Figure 5.
The expression of calcitonin gene-related peptide (CGRP), substance P (SP), and 5-hydroxytryptamine (5-HT) in the spinal cord of rats in each group. The protein expression of CGRP, SP, and 5-HT in the muscles of the rats in each group was examined using (A-C) Western blot analysis and (D-F) immunohistochemistry. N=8. Data are presented as mean ± SD. **P<0.01, compared with the NC group. ##P<0.01, compared with the MPS group. NC, normal control; MPS, myofascial pain syndrome; SNT, silver needle thermal therapy.
Discussion
MPS commonly manifests as soft tissue pain, but the exact pathogenesis remains unclear. Emerging evidence suggests that the central nervous system may play a role in the progression of MPS. Specifically, pain originating from MTrPs has been shown to induce central senfective treatment for MPS. This acupuncture-based therapy provides immediate pain relief and long-term analgesic effects. It has been shown to improve blood circulation, loosen muscle tissue, and reduce inflammation, thus offering promising therapeutic outcomes for soft tissue pain management [22].
Inflammation and the increased release of pain-inducing substances are key factors in the exacerbation of MPS. Studies have reported significantly elevated levels of inflammatory mediators, such as IL-1β and TNF-α, in the MTrP regions of MPS patients. Furthermore, pain severity in these patients correlates with the local concentrations of these substances. Both IL-1β and TNF-α are potent pro-inflammatory cytokines involved in immune responses, inflammation, and tissue repair. TNF-α can directly affect primary afferent neurons, inducing hyperalgesia [23]. Exercise-induced muscle injury is associated with elevated TNF-α levels [24], while IL-1β has been shown to contribute to thermal hyperalgesia in rats [25]. Our study showed that the expression of IL-1β and TNF-α was significantly increased in the muscle tissues of MPS rats. After treatment with SNT conduction therapy, the thermal pain threshold in these rats significantly improved. This was accompanied by a decrease in the expression of IL-1β and TNF-α in the muscle tissues of the SNT group. These results suggest that SNT conduction therapy may alleviate pain in MPS by reducing IL-1β and TNF-α expression.
CGRP is primarily found in C and Aδ sensory fibers, which are widely distributed throughout the body and around blood vessels. These fibers have dual functions in both sensory (pain perception) and efferent (motor action) roles. While CGRP is known for its potent vasodilatory effects, its role in pain modulation remains controversial. Hyperalgesia, characterized by increased sensitivity to painful stimuli, can result from peripheral nerve sensitization (peripheral sensitization) or spinal cord dorsal horn sensitization (central sensitization). The role of CGRP in hyperalgesia remains debated, with varying results across studies. Although peripheral CGRP injections have shown modest effects, CGRP is believed to play a more significant role in central nervous system pain pathways [26]. Intrathecal administration of CGRP has been shown to induce mechanical hyperalgesia, and electrophysiological studies have demonstrated sensitization of spinal dorsal horn neurons by CGRP [27]. In our study, MPS rats exhibited a significantly lower thermal pain threshold compared to the NC group, and CGRP content in the lumbar spinal cord was markedly higher. This suggests that elevated CGRP levels in the spinal cord may contribute to pain perception. After SNT conduction therapy, rats in the SNT group exhibited a significantly higher thermal pain threshold than those in the MPS group. Additionally, CGRP expression in the spinal cord was reduced in the SNT group, indicating that SNT conduction therapy may increase the pain threshold and alleviate pain by inhibiting CGRP expression in the spinal cord. This mechanism may help explain the efficacy of silver needle thermal conduction therapy.
When peripheral inflammation or nociceptive signals are present, SP, the primary afferent neurotransmitter, is involved in transmitting nociceptive signals to the central nervous system. SP induces mast cell degranulation, releasing histamine and serotonin, and upregulates proinflammatory cytokines (e.g., TNF-α, IL-6) as well as anti-inflammatory cytokines (e.g., IL-4, IL-10). These signaling molecules collectively enhance excitatory synaptic transmission and suppress inhibitory synaptic transmission in the spinal cord [28]. Previous studies have shown an association between SP and pain/inflammation near active MTrPs in the upper trapezius muscle [29]. In our study, we observed a significant increase in SP expression in the MPS group compared to the NC group, suggesting that the aseptic inflammation in the muscle fascia of MPS rats stimulates peripheral nociceptive receptors, leading to the release of SP, which is then transmitted to the spinal cord via the dorsal root ganglion, ultimately causing pain. Interestingly, rats treated with SNT conduction therapy exhibited reduced SP expression in the spinal cord compared to the MPS group. This suggests that SNT may inhibit the afferent transmission of nociceptive signals, thereby exerting an analgesic effect.
5-HT is involved in pain modulation through both facilitatory and inhibitory descending pathways. Recent research suggests that the 5-HT descending inhibitory system plays a critical role in pain regulation [30]. When nociceptive receptors are stimulated, descending 5-HT neurons release 5-HT, which binds to corresponding receptors to inhibit neuronal excitability and prevent pain signal transmission to the spinal cord [31]. In rat models of spinal nerve ligation-induced pain, decreased 5-HT levels and impaired receptor function have been linked to dysfunction in the 5-HT inhibitory pathway, contributing to central sensitization and neuropathic pain [32]. Studies using intramuscular heating needles have shown that thermal stimulation at 43°C alleviates both mechanical and heat hyperalgesia in rats by activating 5-HT mechanisms and enhancing descending inhibition [33]. In this study, no significant differences in 5-HT expression were observed between the MPS and NC groups. However, we found a significant increase in 5-HT expression in the spinal cord of rats after two weeks of SNT conduction therapy. This suggests that SNT may promote 5-HT release in the spinal cord through long-term stimulation of the descending inhibitory pathway, thereby producing a lasting analgesic effect by reducing nociceptive signal transmission and central sensitization.
This study has broad potential applications. Preliminary experimental evidence supports the clinical use of this therapy, and future clinical trials should be conducted to assess its efficacy and safety in human MPS patients. Additionally, given that MPS often coexists with other chronic pain conditions such as fibromyalgia and chronic back pain, studying the efficacy of silver needle therapy in these diseases may broaden its applicability. This study still has limitations. First, the small sample size of rats may limit the generalizability of our findings. Second, the MPS model established through a combination of impact and exercise fatigue may not fully replicate the complex etiology and progression of human MPS. Third, the precise mechanisms underlying the therapeutic effects of SNT therapy remain unclear, and our research did not explore how heat conduction influences neurotransmitter and inflammatory factor expression. Future research should address these limitations by expanding the sample size and investigating the molecular mechanisms involved in silver needle thermal therapy.
In conclusion, SNT conduction therapy has shown promising effects in managing MPS. We propose that it inhibits the expression of inflammatory factors in affected muscles, reducing peripheral sensitization. Additionally, it decreases the expression of CGRP and SP while increasing 5-HT expression in the spinal cord, contributing to a sustained analgesic effect through central descending inhibition. This therapy offers a potential therapeutic strategy for MPS management.
Acknowledgements
This study was supported by Guizhou Provincial Health Commission (No. gzwjkj2020-1-137); National base cultivation project of Guizhou Medical University (No. 19nsp060); National Natural Science Foundation of China, Regional Foundation of China (No. 82060811); Foundation of Guizhou Science and Technology Integration -ZK[2021]; Guizhou Province Science and Technology Planning Project; National Natural Science Foundation of China, Regional Foundation of China (No. 82160226); Guizhou Province Science and Technology Plan Project (Grants No: Qianke He Foundation -ZK[2024] Key 036); Guizhou Provincial Science and Technology Projects (Grants No: Qianke He Foundation -2k[2024]-211); 82060811, National Natural Science Foundation of China, Regional Foundation of China; Foundation of Guizhou Science and Technology Integration -ZK[2021], Guizhou Province Science and Technology Planning Project; 82160226, National Natural Science Foundation of China, Regional Foundation of China; and Guizhou Province Science and Technology Plan Project (Grants No: Qianke He Foundation -ZK[2023] Key 044).
Disclosure of conflict of interest
None.
Abbreviations
- MPS
Myofascial pain syndrome
- CGRP
calcitonin gene-related peptide
- SP
substance P
- 5-HT
5-hydroxytryptamine
- IL-1β
interleukin 1-β
- TNF-α
tumor necrosis factor α
References
- 1.Galasso A, Urits I, An D, Nguyen D, Borchart M, Yazdi C, Manchikanti L, Kaye RJ, Kaye AD, Mancuso KF, Viswanath O. A comprehensive review of the treatment and management of myofascial pain syndrome. Curr Pain Headache Rep. 2020;24:43. doi: 10.1007/s11916-020-00877-5. [DOI] [PubMed] [Google Scholar]
- 2.Duarte FCK, Chien R, Ghazinour G, Murnaghan K, West DWD, Kumbhare DA. Myofascial pain as an unseen comorbidity in osteoarthritis: a scoping review. Clin J Pain. 2023;39:188–201. doi: 10.1097/AJP.0000000000001102. [DOI] [PubMed] [Google Scholar]
- 3.Paoletta M, Moretti A, Liguori S, Toro G, Gimigliano F, Iolascon G. Efficacy and effectiveness of extracorporeal shockwave therapy in patients with myofascial pain or fibromyalgia: a scoping review. Medicina (Kaunas) 2022;58:1014. doi: 10.3390/medicina58081014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Urits I, Charipova K, Gress K, Schaaf AL, Gupta S, Kiernan HC, Choi PE, Jung JW, Cornett E, Kaye AD, Viswanath O. Treatment and management of myofascial pain syndrome. Best Pract Res Clin Anaesthesiol. 2020;34:427–448. doi: 10.1016/j.bpa.2020.08.003. [DOI] [PubMed] [Google Scholar]
- 5.Appasamy M, Lam C, Alm J, Chadwick AL. Trigger point injections. Phys Med Rehabil Clin N Am. 2022;33:307–333. doi: 10.1016/j.pmr.2022.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fernández-de-Las-Peñas C, Nijs J. Trigger point dry needling for the treatment of myofascial pain syndrome: current perspectives within a pain neuroscience paradigm. J Pain Res. 2019;12:1899–1911. doi: 10.2147/JPR.S154728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barbero M, Schneebeli A, Koetsier E, Maino P. Myofascial pain syndrome and trigger points: evaluation and treatment in patients with musculoskeletal pain. Curr Opin Support Palliat Care. 2019;13:270–276. doi: 10.1097/SPC.0000000000000445. [DOI] [PubMed] [Google Scholar]
- 8.Zhang Q, Fu C, Huang L, Xiong F, Peng L, Liang Z, Chen L, He C, Wei Q. Efficacy of extracorporeal shockwave therapy on pain and function in myofascial pain syndrome of the trapezius: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2020;101:1437–1446. doi: 10.1016/j.apmr.2020.02.013. [DOI] [PubMed] [Google Scholar]
- 9.Mansoori SS, Moustafa IM, Ahbouch A, Harrison DE. Optimal duration of stretching exercise in patients with chronic myofascial pain syndrome: a randomized controlled trial. J Rehabil Med. 2021;53:jrm00142. doi: 10.2340/16501977-2781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Baltazar M, Russo JAO, De Lucca V, Mitidieri AMS, da Silva APM, Gurian MBF, Poli-Neto OB, Rosa ESJC. Therapeutic ultrasound versus injection of local anesthetic in the treatment of women with chronic pelvic pain secondary to abdominal myofascial syndrome: a randomized clinical trial. BMC Womens Health. 2022;22:325. doi: 10.1186/s12905-022-01910-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Corum M, Aydin T, Medin Ceylan C, Kesiktas FN. The comparative effects of spinal manipulation, myofascial release and exercise in tension-type headache patients with neck pain: a randomized controlled trial. Complement Ther Clin Pract. 2021;43:101319. doi: 10.1016/j.ctcp.2021.101319. [DOI] [PubMed] [Google Scholar]
- 12.Lu F, Ren P, Zhang Q, Shao X. Research trends of acupuncture therapy on myofascial pain syndrome from 2000 to 2022: a bibliometric analysis. J Pain Res. 2023;16:1025–1038. doi: 10.2147/JPR.S401875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Taşkesen F, Cezairli B. The effectiveness of the masseteric nerve block compared with trigger point injections and dry needling in myofascial pain. Cranio. 2023;41:96–101. doi: 10.1080/08869634.2020.1820686. [DOI] [PubMed] [Google Scholar]
- 14.Zha M, Chaffee K, Alsarraj J. Trigger point injections and dry needling can be effective in treating long COVID syndrome-related myalgia: a case report. J Med Case Rep. 2022;16:31. doi: 10.1186/s13256-021-03239-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yehoshua I, Rimon O, Mizrahi Reuveni M, Peleg R, Adler L. Dry needling for the treatment of acute myofascial pain syndrome in general practitioners’ clinics: a cohort study. BMC Prim Care. 2022;23:339. doi: 10.1186/s12875-022-01951-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xiong J, Zhou X, Luo X, Gong X, Jiang L, Luo Q, Zhang S, Jiang C, Pu T, Liu J, Zhang J, Li B, Chi H. Acupuncture therapy on myofascial pain syndrome: a systematic review and meta-analysis. Front Neurol. 2024;15:1374542. doi: 10.3389/fneur.2024.1374542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Menéndez-Torre Á, Pintado-Zugasti AM, Zaldivar JNC, García-Bermejo P, Gómez-Costa D, Molina-Álvarez M, Arribas-Romano A, Fernández-Carnero J. Effectiveness of deep dry needling versus manual therapy in the treatment of myofascial temporomandibular disorders: a systematic review and network meta-analysis. Chiropr Man Therap. 2023;31:46. doi: 10.1186/s12998-023-00489-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee S, Lee IS, Chae Y. Similarities between Ashi acupoints and myofascial trigger points: exploring the relationship between body surface treatment points. Front Neurosci. 2022;16:947884. doi: 10.3389/fnins.2022.947884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chang WH, Tu LW, Pei YC, Chen CK, Wang SH, Wong AM. Comparison of the effects between lasers applied to myofascial trigger points and to classical acupoints for patients with cervical myofascial pain syndrome. Biomed J. 2021;44:739–747. doi: 10.1016/j.bj.2020.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fan AY, Xu J, Li YM. Evidence and expert opinions: dry needling versus acupuncture (II): The American Alliance for Professional Acupuncture Safety (AAPAS) White Paper 2016. Chin J Integr Med. 2017;23:83–90. doi: 10.1007/s11655-017-2800-6. [DOI] [PubMed] [Google Scholar]
- 21.Lv X, Wo C, Yao J, Lu W, Yu Z, Qin Y, Wang Y, Zhang Z, Wu Y, Huang Y, Wang L. Silver needle thermal therapy relieves pain, repairs the damaged myofascial fiber, and reduces the expression of 5-HT3 receptors in the spinal cord of rats with myofascial pain syndrome. Neurol India. 2022;70:S288–S295. doi: 10.4103/0028-3886.360917. [DOI] [PubMed] [Google Scholar]
- 22.Fernández-de-Las-Peñas C, Nijs J, Cagnie B, Gerwin RD, Plaza-Manzano G, Valera-Calero JA, Arendt-Nielsen L. Myofascial pain syndrome: a nociceptive condition comorbid with neuropathic or nociplastic pain. Life (Basel) 2023;13:694. doi: 10.3390/life13030694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.de Magalhães SF, Manzo LP, de Faria FM, de Oliveira-Fusaro MC, Nishijima CM, Vieira WF, Bonet IJM, Dos Santos GG, Tambeli CH, Parada CA. Inflammatory pain in peripheral tissue depends on the activation of the TNF-α type 1 receptor in the primary afferent neuron. Eur J Neurosci. 2021;53:376–389. doi: 10.1111/ejn.14985. [DOI] [PubMed] [Google Scholar]
- 24.Wei S, Qiu CY, Jin Y, Liu TT, Hu WP. TNF-α acutely enhances acid-sensing ion channel currents in rat dorsal root ganglion neurons via a p38 MAPK pathway. J Neuroinflammation. 2021;18:92. doi: 10.1186/s12974-021-02151-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fernández-Lázaro D, Mielgo-Ayuso J, Seco Calvo J, Córdova Martínez A, Caballero García A, Fernandez-Lazaro CI. Modulation of exercise-induced muscle damage, inflammation, and oxidative markers by curcumin supplementation in a physically active population: a systematic review. Nutrients. 2020;12:501. doi: 10.3390/nu12020501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yuan Y, Zhao Y, Shen M, Wang C, Dong B, Xie K, Yu Y, Yu Y. Spinal NLRP3 inflammasome activation mediates IL-1β release and contributes to remifentanil-induced postoperative hyperalgesia by regulating NMDA receptor NR1 subunit phosphorylation and GLT-1 expression in rats. Mol Pain. 2022;18:17448069221093016. doi: 10.1177/17448069221093016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Iyengar S, Ossipov MH, Johnson KW. The role of calcitonin gene-related peptide in peripheral and central pain mechanisms including migraine. Pain. 2017;158:543–559. doi: 10.1097/j.pain.0000000000000831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Paige C, Plasencia-Fernandez I, Kume M, Papalampropoulou-Tsiridou M, Lorenzo LE, David ET, He L, Mejia GL, Driskill C, Ferrini F, Feldhaus AL, Garcia-Martinez LF, Akopian AN, De Koninck Y, Dussor G, Price TJ. A female-specific role for calcitonin gene-related peptide (CGRP) in rodent pain models. J Neurosci. 2022;42:1930–1944. doi: 10.1523/JNEUROSCI.1137-21.2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lauritano D, Mastrangelo F, D’Ovidio C, Ronconi G, Caraffa A, Gallenga CE, Frydas I, Kritas SK, Trimarchi M, Carinci F, Conti P. Activation of mast cells by neuropeptides: the role of pro-inflammatory and anti-inflammatory cytokines. Int J Mol Sci. 2023;24:4811. doi: 10.3390/ijms24054811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Green DP, Limjunyawong N, Gour N, Pundir P, Dong X. A mast-cell-specific receptor mediates neurogenic inflammation and pain. Neuron. 2019;101:412–420. e413. doi: 10.1016/j.neuron.2019.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shah JP, Danoff JV, Desai MJ, Parikh S, Nakamura LY, Phillips TM, Gerber LH. Biochemicals associated with pain and inflammation are elevated in sites near to and remote from active myofascial trigger points. Arch Phys Med Rehabil. 2008;89:16–23. doi: 10.1016/j.apmr.2007.10.018. [DOI] [PubMed] [Google Scholar]
- 32.Yamaguchi C, Yamamoto D, Fujimaru Y, Asano T, Takaoka A. Acetaminophen exerts an analgesic effect on muscular hyperalgesia in repeated cold-stressed rats through the enhancement of the descending pain inhibitory system involving spinal 5-HT(3) and noradrenergic α(2) receptors. Biol Pharm Bull. 2021;44:1067–1074. doi: 10.1248/bpb.b21-00178. [DOI] [PubMed] [Google Scholar]
- 33.Li CJ, Zhang LG, Liu LB, An MQ, Dong LG, Gu HY, Dai YP, Wang F, Mao CJ, Liu CF. Inhibition of spinal 5-HT3 receptor and spinal dorsal horn neuronal excitability alleviates hyperalgesia in a rat model of Parkinson’s disease. Mol Neurobiol. 2022;59:7253–7264. doi: 10.1007/s12035-022-03034-8. [DOI] [PubMed] [Google Scholar]