Abstract
Organic acids are valuable platform chemicals for future biorefining applications. Such applications involve the conversion of low-cost renewable resources to platform sugars, which are then converted to platform chemicals by fermentation and further derivatized to large-volume chemicals through conventional catalytic routes. Organic acids are toxic to many of the microorganisms, such as Escherichia coli, proposed to serve as biorefining platform hosts at concentrations well below what is required for economical production. The toxicity is two-fold including not only pH based growth inhibition but also anion-specific effects on metabolism that also affect growth. E. coli maintain viability at very low pH through several different tolerance mechanisms including but not limited to the use of decarboxylation reactions that consume protons, ion transporters that remove protons, increased expression of known stress genes, and changing membrane composition. The focus of this mini-review is on organic acid toxicity and associated tolerance mechanisms as well as several examples of successful organic acid production processes for E. coli.
Review
Biorefining Platforms
Biorefining promises the development of efficient processes for the conversion of renewable sources of carbon and energy into large volume commodity chemicals. It has been estimated that such bioprocesses already account for 5% of the 1.2 trillion dollar US chemical market [1], with some projecting future values of up to 50% of the total US chemical market generated through biological means. While the attractiveness of such bioprocesses has been recognized for some time [2,3], recent advances in biological engineering and associated sciences [4-15], several biorefining success stories [16-18], and instability in the price and future availability of oil [19], have collectively reinvigorated interest in the large scale production of chemicals through biological routes. Nevertheless, many challenges still remain for the economical bio-production of commodity chemicals. Such challenges encompass the need to not only inexpensively convert biomass into usable sources of carbon and energy but also to engineer microbes to produce relevant chemicals at high titers and productivities while minimizing the generation of byproducts that might foul downstream processes [1,20,21]. One model for addressing the latter of such challenges involves the generation of platform organisms that can be easily engineered and re-engineered to produce a variety of building block chemicals that are amenable to conversions to higher value products via traditional catalytic routes (see Figure 1). Although chemical pretreatment of raw materials impairs viability of platform organisms, this review will focus on product toxicity issues associated with the production of organic acids in E. coli (for further information on sugar extraction from raw materials see Zaldavar, et al. [22] and Knauf, et al. [23]).
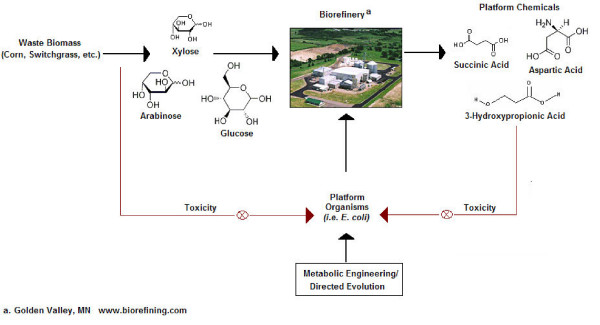
Figure 1.
Conceptual model of toxicity in biorefining applications. Sugars are extracted from waste biomass for use as feedstock for platform organisms in a biorefinery. Metabolically engineered microorganisms convert sugars into valuable platform chemicals that are then further derivatized to large-volume chemicals. Product and feedstock toxicity are observed, thus limiting productivity of biorefining applications.
The US Department of Energy (USDOE) recently released a prioritized list of building block chemicals for future biorefining endeavors. Priority was assigned based on the projected value of the platform chemical and potential derivatives as well as what technological developments were required for the production of the chemical and associated derivatives [21]. The report emphasized the importance of organic acids to the future of biorefining efforts (eight of the top twelve chemicals were organic acids, see Table 1 in additional file 1). The USDOE is not the first to recognize the importance of organic acids. In fact, there is a rich literature describing microbial production of organic acids [17,20,24,25], including several successful commercial bioprocesses [26-28]. Product toxicity is one of the primary challenges in the development of organic acid bioprocesses based on the use of platform host organisms, such as E. coli. In particular, while E. coli is known to survive very high concentrations of acids (pH = 2) when passing through the mammalian stomach, E. coli are surprisingly acid sensitive in exponential phase when cultured planktonically [29,30]. Moreover, undissociated organic acids, which pass freely through the outer and plasma membranes of E. coli [31,32], dissociate upon entry into the slightly alkaline cytoplasm releasing protons that lower internal pH (pHi) and anions that specifically inhibit different aspects of metabolism resulting in impaired growth [33-35]. Titers and productivities of 50–100 g/L and 2–3 g/L·hr are expected for the economical manufacturing of most building block acids by fermentation. The pKa values range from 3–5 for these organic acids, which would result in a pH reduction to around 2.0 for titers of 50 g/L. This highlights a key challenge in the metabolic engineering of organic acid production hosts. That is, high titers result in the addition of protons to the culture, which either result in a decreased pH or the addition of large volumes of base titrant. At low pH, organic acids are undissociated, thus they pass freely through the membrane and inhibit growth. At high pH, the process is less efficient due to base requirements and because export of the organic acid cannot proceed by free diffusion alone (for a more detailed discussion of organic acid export issues see Van Maris et al. [36]). What is desired, therefore, is a platform organism that not only produces high levels of organic acid chemicals but also is tolerant to any associated toxicity.
Many microbes are capable of producing platform chemicals by aerobic and anaerobic fermentation processes [22]. L-lactic acid has traditionally been produced by lactic acid bacteria. Although many lactic acid bacteria strains have been studied extensively [37], the ability to produce optically pure L-lactic acid is hampered by the presence of both L and D lactate dehydrogenase genes [38]. Pure L-lactic acid must therefore be produced via another pathway, as the racemic acid product is not useful for downstream conversion into polylactic acid. A number of other microorganisms have been used for industrial fermentation of several of the building block organic acids identified in Table 1. Large scale production of amino acids has been accomplished in Corynebacteriumglutamicum [39], succinic acid has been produced by Actinobacillus succinogenes [40], and itaconic acid production has been carried out with Aspergillus terrus [41]. While successful, the future application of these organisms as platform hosts is limited when compared with E. coli. E. coli is advantageous as a platform host because it is the most well characterized model organism, it has been used in recombinant processes for over 20 years, there are a wide variety of good genetic tools, and it is sensitive to many antibiotics used in genetic engineering efforts [42]. Moreover, the completion of the E. coli genome sequence has already enabled many functional genomics studies and proven useful in metabolic engineering efforts [43]. Finally, E. coli grows quickly in minimal media and maintains the ability to metabolize both 5 and 6 carbon sugars, which is a specific advantage over the use of industrially relevant yeast strains [22]. This mini-review will describe the basic mechanisms underlying organic acid toxicity and associated tolerance pathways in E. coli followed by a short discussion of several metabolic engineering strategies employed for the production of organic acids in E. coli.
Organic Acid Toxicity in E. coli
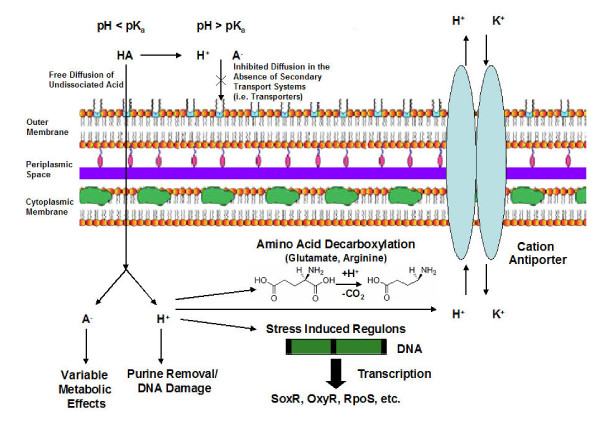
One of the primary factors contributing to the toxicity of organic acids is their ability to diffuse across E. coli cellular membranes when undissociated as opposed to the restricted passage of dissociated protons and anions (see Figure 2) [31,32]. Diffusion of dissociated acids is limited to secondary transport, which is known to involve H+/monocarboxylic acid symporters. However, the detailed mechanism and specificities of the transporters remain unknown [31]. E. coli maintain a cytoplasmic pH (pHi = 7.5) that is most often higher than that of the external media and typically well above the pKa of organic acids [44,45]. As a result, organic acids exist in the dissociated form within the cytoplasm. Thus, diffusing organic acids entering into the cytoplasm will dissociate and disrupt the pHi and anion pool of the cytoplasm. The resulting increase in internal acidity can affect the integrity of purine bases [46] and result in denaturing of essential enzymes inside the cell [35], both of which negatively affect cell viability.
Figure 2.
An overview of organic acid toxicity and tolerance mechanisms in E. coli. Diffusion of undissociated acid molecules can occur freely in acidic medium but is limited to transport systems at neutral or basic pH. The toxic effects associated with organic acids are the result of both anion specific affects on metabolism as well as increased internal proton concentrations. Affects on internal pH are mitigated by transport of protons out of the membrane, consumption of protons by decarboxylation reactions, and, more generally, induction of stress regulons. Anion specific tolerance mechanisms are not well characterized.
Organic acid anions affect cell growth in a variety of manners. Increased anion concentration has been shown to lead to an increased transport of potassium ions into the cell, which increases turgor pressure [47,48]. To maintain a constant turgor pressure and cell volume, glutamate is transported out of the cell [48]. This transport activity concomitantly disrupts the osmolarity of the cytoplasm, which in turn lowers the cell's growth potential and viability. In addition to this general anion effect, there are also effects specific to each organic acid. It has been proposed that enzymes involved in protein synthesis are sensitive to a combination of two unrelated mechanisms, including the acidification of pHi and the formation of an anionic pool [35]. Although this finding implies that the organic inhibition due to the anion pool could be acid specific, the details describing this dual inhibition mechanism remain unclear. Kirkpatrick et al. reported proteins exhibiting increased expression in response to extracellular acetate [33]. Among these are the OppA transporter, RpoS regulon, several amino acid uptake proteins, DNA binding proteins, and extreme-acid preiplasmic chaperones. Interestingly, when formate was introduced in place of acetate the expression of the previously mentioned proteins was repressed, indicating that the response was anion specific. This finding introduces new challenges in addressing organic acid tolerance. Specifically, it highlights the need to engineer both pH and as well as specific anion tolerance into host organisms.
Finally, production of organic acids might include intermediates that are themselves toxic. For example, 3-hydroxypropionic acid (3HP) is closely related to the antimicrobial compound Reuterin. Reuterin describes the hydroxypropionaldehyde (HPA) system including HPA, HPA dimer, and HPA hydrate. Reuterin is inhibitory to several bacteria, including E. coli, at concentrations as low as 0.03–0.05 g/L [49-51]. It is thought that the toxicity could be the result of inhibition of DNA synthesis [52]. It has been postulated that the reactivity of the aldehyde group of HPA causes DNA damage similarly to formaldehyde, which is the aldehyde analog of formic acid [49]. Intermediate toxicity can be managed either by optimization of the production pathway in the host or by engineering tolerance to the intermediate itself.
Organic Acid Tolerance in E. coli
E. coli has a remarkable ability to remain viable under a broad range of pH conditions. This ability is essential for its survival in the mammalian digestive system where pH can vary between pH = 2–8. Several different acid tolerance mechanisms have been identified in E. coli. While each mechanism is capable of providing some degree of tolerance, they are regulated differently and confer varying levels of tolerance.
Although most acid tolerance systems are activated in stationary phase, acid tolerance as low as pH = 3 has been observed in exponential phase E. coli grown under aerobic conditions, which is advantageous from a productivity standpoint [30]. Although the underlying tolerance mechanism is not known, such tolerance can be reliably activated by adapting cells at sublethal pH values between 4.3 and 5.8 [53]. E. coli that exhibit growth phase tolerance remain viable at pH values on the same order as stationary phase tolerance, however the percent survival is significantly lower. Lin et al. reported 1% survival of the original culture following acid adaption at pH 4.3 followed by acid challenge at pH 3.3 compared to 0.0001% survival for unadapted cultures. This is compared to stationary-phase cultures, which exhibited up to 50% survival.
Three stationary phase acid resistance systems have been studied in the most detail [29,30]. These systems confer the highest levels of tolerance and are believed to be responsible for stationary phase E. coli survival when passing through the mammalian stomach. Acid resistance system 1 (AR1) is activated in slightly acidic media (pH 5.5) in the absence of extracellular glucose or amino acids. E. coli grown aerobically under these conditions retain viability under acid challenges as low as pH = 2.5 [54]. This system is also referred to as the oxidative or glucose-repressed system, since the expression of this system is thought to be regulated either directly or indirectly by RpoS and cyclicAMP receptor protein (CRP) [55,56]. Acid resistance system 2 (AR2) is activated in E. coli grown in aerobic conditions in acidic complex media. This system requires the presence of extracellular glucose and glutamate and is dependent upon genes encoding glutamate decarboxylase (gadAB) and a glutamate:GABA antiporter (gabC) [30]. Under such conditions, E. coli have been demonstrated to exhibit acidic resistance up to a pH of 2. The mechanism involves the expenditure of excess cytoplasmic protons during amino acid decarboxylation reactions (see Figure 2), thus raising the internal pH [54,55]. Acid resistance system 3 (AR3) parallels the mechanisms of AR2 with several slight deviations [30,54,55]. AR3 is activated under anaerobic conditions, in complex media with added glucose. It also involves amino acid decarboxylation reactions to lower the internal pH, but requires extracellular arginine in place of glutamate. AR3 also requires increased expression of arginine decarboxylase and an arginine: agmatine antiporter for increased acid tolerance.
Finally, several general acid tolerance mechanisms that regulate the physical properties of the membrane or the effectiveness of ion transport have been identified. These active responses, or those that occur as a result of the cell's ability to sense pH changes, are independent of growth and are induced by pH shifts as small as 0.2 pH units [57]. The first response is the ability of the microorganism to adjust membrane properties, such as lipid content, thus effectively changing the proton permeability [57]. Another cellular response to acid shock is the induction of genes responsible for repairing and preventing lethal cellular damage. Specifically, increased expression of the oxyR and soxR regulatory genes has been observed by transcriptional profiling of acid tolerant phenotypes [45,58]. These systems regulate the removal of damaging oxidizing agents, thus preventing further DNA damage under acidic stress [46]. Finally, acid tolerance can be achieved by adjusting the ionic transporter efficiency, effectively regulating the anion and cation balance as a means of maintaining a constant internal pH [47].
Organic Acid Production in E. coli
Metabolic and genetic engineering, directed evolution, and classic strain selection have all been employed in the development of E. coli strains that produce building block organic acids, including lactic-acid, succinic acid, and 3HP [17,25,59,60]. Improved titers have been achieved due to optimization of fermentation conditions and relavant pathways utilized. However, titer limitations exist when fermentation is carried out in unbuffered media, which allows the pH to acidify due to increased acid concentration. Alternatively large amounts of base titrant are required to raise the pH of the media during the organic acid production leaving the final acid molecule in the undissociated form. Following production under these conditions, large volumes of acid must be added to recover the acid in the protonated form. Metabolic and genetic engineering of acid tolerance into production strains, making fermentation at a pH less than the pKa of the acid produced possible, would circumvent the need for the additional consumption of acid and base titrants, and thus lower the overall production cost. Similarly, engineering strain fitness to increase productivity at a decreased pH would improve productivity and reduce base consumption.
Lactic acid production is one of the most successful examples to date of the engineering of large volume chemical production in E. coli. E. coli was selected as a favorable host strain due to its ability to consume both pentose and hexose sugars and to generate optically pure L-lactic acid, which is the desired product for downstream polylactic acid (PLA) production [61,62]. An effective lactic acid producing strain of E. coli was created by induced expression of the L-specific lactic acid dehydrogenase (LDH) gene from Streptococcus bovis. High titers (50–75 g/L) were observed under controlled pH (pH = 7) and anaerobic conditions. Titers were drastically decreased (10–20 g/L) as the pH was allowed to drop with increasing acid production [59]. However, allowing the pH to fall below the pKa of lactic acid also resulted in decreased concentration of the acid in the undissociated form, which facilitated the subsequent isolation of the protonated acid. Interestingly, the choice of host strain made a significant difference in lactic acid production [59]. Those constructed from an E. coli B strain showed a titer of almost twice that produced from K12 derivatives. The increased production was attributed primarily to differences in the native growth characteristics rather than increased acid tolerance.
Economically competitive titers of succinic acid have also been achieved in E. coli. Strains were engineered to limit flux to other anaerobic byproducts normally formed during fermentation [60]. Specifically, succinic acid production was optimized by redirecting the metabolic flux at the pyruvate node away from lactate and formate through inactivation of the pyruvate-formatelyase and lactate dehydrogenase [60,63]. The maximum yield in succinic acid production was approximately 50 g/L in pH controlled cultures. However, similar to lactic acid studies, succinic acid production was significantly repressed when pH was not kept at neutral levels.
A final example of metabolic engineering organic acid production in E. coli was reported by Cargill in 2001 [17]. Suthers and Cameron engineered a 2-step glycerol to 3HP pathway in E. coli. Glycerol was first converted to 3HPA via a glycerol dehydratase enzyme (dhaB – isolated from Klebsiella pneumoniae). 3HPA was then converted to 3HP via an aldehyde dehydrogenase (ald). This first pathway was not ideal for several reasons including a very low reported titer (0.2 g/L), the use of the more expensive glycerol as opposed to glucose, and the generation of the highly toxic 3-HPA (reuterin) compound. Selifinova et al. later proposed five additional pathways for the production of 3-HP directly from glucose in E. coli [36]. Results for each of such pathways have yet to be reported. One issue that has yet to be addressed is how to fulfill the desire to produce 3-HP at a pH below the pKa = 4.51 of 3-HP, which would lessen the dependency on large volumes of base titrant to retain neutral pH at high titers.
Metabolic engineering of E. coli organic acid tolerance represents an important future opportunity. As discussed above, E. coli possess several systems for surviving pH as low as 2.0, which is much lower than what is required for an economical biorefining process. Since induction of these systems is well characterized and the relevant genes are known in many cases, future efforts might be better focused on the development of multi-stage fermentations that allow for generation of biomass prior to induction of acid tolerance and, ultimately, acid production. Future genetic engineering efforts might focus on engineering tolerance against the less well characterized metabolic effects associated with increased organic acid anion concentrations. For example, the addition of acetate, benzoate, and propionate to culture media at a concentration of 8 mM has been observed to inhibit growth of E. coli up to 50% [35]. The acetate inhibition is thought to be caused by limited methionine pools combined with increasing concentrations of homocysteine, a toxic intermediate, due to inactivation of a key enzyme in the methionine synthesis pathway, which can be countered by the addition of methionine to the media. This finding established that growth inhibition is the result of both of lowered pH and specific anionic effects, which decreases the activity of key enzymes. Thus, engineering tolerance to specific organic acid anion effects by increased expression of inhibited enzymes could aid in increasing overall process productivity.
Conclusion
Organic acids are a valuable sector of the industrial chemical market, which have already been successfully produced through microbial fermentation. However, product titers have been variable, ranging from less than 1 g/L to concentrations cost competitive with current petrochemical production processes. These fermentation processes have been limited in E. coli due to product and intermediate toxicity. Toxicity is directly measured by growth inhibition, which specifically decreases productivity. This review highlighted what is known about organic-acid toxicity and tolerance mechanisms in E. coli. Specifically, E. coli are growth inhibited by the increase in both proton and associated anion concentrations that are characteristic of organic-acid production processes. While several acid-tolerance mechanisms have been characterized in E. coli, anion specific mechanisms require additional study. Thus, future metabolic engineering efforts that seek to improve understanding of these issues within the context of organic-acid biorefining applications should prove useful.
Supplementary Material
Table 1: Organic acids for platform biorefining applications. (* see references [64,65])
Contributor Information
Tanya Warnecke, Email: tanya.warnecke@colorado.edu.
Ryan T Gill, Email: rtg@colorado.edu.
References
- Bachmann R, Riese J. From promise to profit. Industrial Biotechnology. 2005;1:9–15. [Google Scholar]
- Leeper SA, Ward TE, Andrews GF. Production of Organic Chemicals via Bioconversion: A Review for Potential. US DOE's Idaho Operations Office; 1991. [Google Scholar]
- Herrera S. Industrial biotechnology - a chance at redemption. Nature Biotechnology. 2004;22:671–675. doi: 10.1038/nbt0604-671. [DOI] [PubMed] [Google Scholar]
- Crameri A, Raillard S, Bermudez E, Stemmer WPC. DNA shuffling of a family of genes from diverse species accelerates directed evolution . Nature. 1994;370:389–390. doi: 10.1038/34663. [DOI] [PubMed] [Google Scholar]
- Patnaik R, Louie S, Gavrilovic V, Perry K, Stemmer WPC, Ryan CM, del Cardayré S. Genome shuffling of Lactobacillus for improved acid tolerance. Nature Biotechnology. 2002;20:707–712. doi: 10.1038/nbt0702-707. [DOI] [PubMed] [Google Scholar]
- Canada KA, Iwashita S, Shim H, Wood TK. Directed evolution of toluene ortho-monooxygenase for enhanced 1-naphthol synthesis and chlorinated ethene degradation. Journal Of Bacteriology. 2002;184:344–349. doi: 10.1128/JB.184.2.344-349.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farmer WR, Liao JC. Improving lycopene production in Escherichia coli by engineering metabolic control. Nature Biotechnology. 2000;18:533–537. doi: 10.1038/75398. [DOI] [PubMed] [Google Scholar]
- Fodor SPA, Read JL, Pirrung MC, Stryer L, Lu AT, Solas D. Light-Directed, Spatially Addressable Parallel Chemical Synthesis. Science. 1991;251:767–773. doi: 10.1126/science.1990438. [DOI] [PubMed] [Google Scholar]
- Gill RT, Wildt S, Yang YT, Ziesman S, Stephanopoulos G. Genome-wide screening for trait conferring genes using DNA microarrays. Proceedings Of The National Academy Of Sciences Of The United States Of America. 2002;99:7033–7038. doi: 10.1073/pnas.102154799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin VJJ, Pitera DJ, Withers ST, Newman JD, Keasling JD. Engineering a mevalonate pathway in Escherichia coli for production of terpenoids. Nature Biotechnology. 2003;21:796–802. doi: 10.1038/nbt833. [DOI] [PubMed] [Google Scholar]
- Ohnishi J, Mitsuhashi S, Hayashi M, Ando S, Yokoi H, Ochiai K, Ikeda M. A novel methodology employing Corynebacterium glutamicum genome information to generate a new L-lysine-producing mutant. Applied Microbiology And Biotechnology. 2002;58:217–223. doi: 10.1007/s00253-001-0883-6. [DOI] [PubMed] [Google Scholar]
- Schmidt-Dannert C, Umeno D, Arnold FH. Molecular breeding of carotenoid biosynthetic pathways. Nature Biotechnology. 2000;18:750–753. doi: 10.1038/77319. [DOI] [PubMed] [Google Scholar]
- Bailey JE. Towards a science of metabolic engineering. Science. 1991;252:1668–1675. doi: 10.1126/science.2047876. [DOI] [PubMed] [Google Scholar]
- Bailey JE, Sburlati A, Hatzimanikatis V, Lee K, Renner WA, Tsai PS. Inverse metabolic engineering: A strategy for directed genetic engineering of useful phenotypes. Biotechnology And Bioengineering. 2002;79:568–579. doi: 10.1002/bit.10441. [DOI] [PubMed] [Google Scholar]
- Stephanopoulos G, Vallino JJ. Network Rigidity And Metabolic Engineering In Metabolite Overproduction. Science. 1991;252:1675–1681. doi: 10.1126/science.1904627. [DOI] [PubMed] [Google Scholar]
- Higley DP, Sun Y. Acid-dyeable polymer compositions. US patent 938760 . 2004.
- Suthers PF, Cameron DC. Production of 3-Hydroxypropionic acid in recombinant organisms. PCT WO 01-16346. 2001.
- Gatenby AA, Haynie SL, Nagarajan Method for the production of 1,3-propanediol by recombinant organisms. WO 9821339. 1998.
- Deffeyes KS. Hubbert's Peak: The Impending World Oil Shortage. Princeton, NJ , Princeton University Press; 2001. [Google Scholar]
- Chotani G, Dodge T, Hsu A, Kumar M, LaDuca R, Trimbur D, Weyler W, Sanford K. The commercial production of chemicals using pathway engineering. Biochimica Et Biophysica Acta-Protein Structure And Molecular Enzymology. 2000;1543:434–455. doi: 10.1016/s0167-4838(00)00234-x. [DOI] [PubMed] [Google Scholar]
- Werpy T, Petersen G. Volume 1: Results of Screening for Potential Candidates from Sugars and Synthetic Gas. Oak Ridge, TN , U.S. Department of Energy; 2004. Top Value Added Chemicals from Biomass. [Google Scholar]
- Zaldivar J, Nielsen J, Olsson L. Fuel ethanol production from lignocellulose: a challenge for metabolic engineering and process integration. Applied Microbiology And Biotechnology. 2001;56:17–34. doi: 10.1007/s002530100624. [DOI] [PubMed] [Google Scholar]
- Knauf M, Moniruzzaman M. Lignocellulosic biomass processing: A perspective. International Sugar Journal. 2004;106:147–150. [Google Scholar]
- Trends in Life Sciences: US Market . Washington DC , International Access Corporation (IAC); 2002. [Google Scholar]
- Selifonova OV, Jessen H, Gort SJ, Selmer T, Buckel W. 3-Hydroxypropionic acid and other organic compounds. PCT WO 02/42418. 2002.
- Ngheim NP, Donnelly MI, Millard CS, Stols L. Method for the production of dicarboxylic acids . 1999.
- Reichstein T. Process for the manufacture of levoascorbic acid (vitamin C). US patent 2,265,121. 1941.
- Skory CD. Fungal lactate dehydrogenase gene and constructs for the expression thereof. US Patent 535381 . 2000.
- Richard H, Foster JW. Escherichia coli glutamate- and arginine-dependent acid resistance systems increase internal pH and reverse transmembrane potential. Journal Of Bacteriology. 2004;186:6032–6041. doi: 10.1128/JB.186.18.6032-6041.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richard HT, Foster JW. Acid resistance in Escherichia coli. (Advances In Applied Microbiology).Advances In Applied Microbiology, Vol 52. 2003;52:167–186. doi: 10.1016/s0065-2164(03)01007-4. [DOI] [PubMed] [Google Scholar]
- Poole RC, Halestrap AP. Transport Of Lactate And Other Monocarboxylates Across Mammalian Plasma-Membranes. American Journal Of Physiology. 1993;264:C761–C782. doi: 10.1152/ajpcell.1993.264.4.C761. [DOI] [PubMed] [Google Scholar]
- Walter A, Gutknecht J. Monocarboxylic Acid Permeation Through Lipid Bilayer-Membranes. Journal Of Membrane Biology. 1984;77:255–264. doi: 10.1007/BF01870573. [DOI] [PubMed] [Google Scholar]
- Kirkpatrick C, Maurer LM, Oyelakin NE, Yoncheva YN, Maurer R, Slonczewski JL. Acetate and formate stress: Opposite responses in the proteome of Escherichia coli. Journal Of Bacteriology. 2001;183:6466–6477. doi: 10.1128/JB.183.21.6466-6477.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roe AJ, McLaggan D, Davidson I, O'Byrne C, Booth IR. Perturbation of anion balance during inhibition of growth of Escherichia coli by weak acids. Journal Of Bacteriology. 1998;180:767–772. doi: 10.1128/jb.180.4.767-772.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roe AJ, O'Byrne C, McLaggan D, Booth IR. Inhibition of Escherichia coli growth by acetic acid: a problem with methionine biosynthesis and homocysteine toxicity. Microbiology-Sgm. 2002;148:2215–2222. doi: 10.1099/00221287-148-7-2215. [DOI] [PubMed] [Google Scholar]
- van Maris AJA, Konings WN, van Dijken JP, Pronk JT. Microbial export of lactic and 3-hydroxypropanoic acid: implications for industrial fermentation processes. Metabolic Engineering. 2004;6:245–255. doi: 10.1016/j.ymben.2004.05.001. [DOI] [PubMed] [Google Scholar]
- Narayanan N, Roychoudhury PK, Srivastava A. L (+)lactic acid fermentation and its product polymerization. Electronic Journal Of Biotechnology. 2004;7:167–U2. [Google Scholar]
- Saitoh S, Ishida N, Onishi T, Tokuhiro K, Nagamori E, Kitamoto K, Takahashi H. Genetically engineered wine yeast produces a high concentration of L-lactic acid of extremely high optical purity. Applied And Environmental Microbiology. 2005;71:2789–2792. doi: 10.1128/AEM.71.5.2789-2792.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda M, Nakagawa S. The Corynebacterium glutamicum genome: features and impacts on biotechnological processes. Applied Microbiology And Biotechnology. 2003;62:99–109. doi: 10.1007/s00253-003-1328-1. [DOI] [PubMed] [Google Scholar]
- Zeikus JG, Jain MK, Elankovan P. Biotechnology of succinic acid production and markets for derived industrial products. Applied Microbiology And Biotechnology. 1999;51:545–552. [Google Scholar]
- Willke T, Vorlop KD. Biotechnological production of itaconic acid. Applied Microbiology And Biotechnology. 2001;56:289–295. doi: 10.1007/s002530100685. [DOI] [PubMed] [Google Scholar]
- Nelson DL, Cox MM. Lehninger Principles of Biochemistry. 3rd. New York, NY , Worth Publisher; 2000. [Google Scholar]
- Harrington CA, Rosenow C, Retief J. Monitoring gene expression using DNA microarrays. Current Opinion In Microbiology. 2000;3:285–291. doi: 10.1016/s1369-5274(00)00091-6. [DOI] [PubMed] [Google Scholar]
- Goulbourne E, Matin M, Zychlinsky E, Matin A. Mechanism Of Delta-pH Maintenance In Active And Inactive Cells Of An Obligately Acidophilic Bacterium. Journal Of Bacteriology. 1986;166:59–65. doi: 10.1128/jb.166.1.59-65.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurer LM, Yohannes E, Bondurant SS, Radmacher M, Slonczewski JL. pH regulates genes for flagellar motility, catabolism, and oxidative stress in Escherichia coli K-12. Journal Of Bacteriology. 2005;187:304–319. doi: 10.1128/JB.187.1.304-319.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi SH, Baumler DJ, Kaspar CW. Contribution of dps to acid stress tolerance and oxidative stress tolerance in Escherichia coli O157 : H7. Applied And Environmental Microbiology. 2000;66:3911–3916. doi: 10.1128/aem.66.9.3911-3916.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kroll RG, Booth IR. The Relationship Between Intracellular Ph, The Ph Gradient And Potassium-Transport In Escherichia-Coli. Biochemical Journal. 1983;216:709–716. doi: 10.1042/bj2160709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaggan D, Naprstek J, Buurman ET, Epstein W. Interdependence Of K+ And Glutamate Accumulation During Osmotic Adaptation Of Escherichia-Coli. Journal Of Biological Chemistry. 1994;269:1911–1917. [PubMed] [Google Scholar]
- Sung HW, Chen CN, Chang Y, H.F. L. Biocompatibility Study of Biological Tissues Fixed by a Natural Compound (Reuterin) Produced by Lactobacillus Reuteri. Biomaterials. 2002;23:3203–3214. doi: 10.1016/s0142-9612(02)00072-8. [DOI] [PubMed] [Google Scholar]
- Ganzle MG. Reutericyclin: biological activity, mode of action, and potential applications. Applied Microbiology And Biotechnology. 2004;64:326–332. doi: 10.1007/s00253-003-1536-8. [DOI] [PubMed] [Google Scholar]
- Luthi-Peng Q, Dileme FB, Puhan Z. Effect of glucose on glycerol bioconversion by Lactobacillus reuteri. Applied Microbiology And Biotechnology. 2002;59:289–296. doi: 10.1007/s00253-002-1002-z. [DOI] [PubMed] [Google Scholar]
- Rasch M. The influence of temperature, salt and pH on the inhibitory effect of reuterin on Escherichia coli. International Journal of Food Microbiology. 2002;72:225–231. doi: 10.1016/s0168-1605(01)00637-7. [DOI] [PubMed] [Google Scholar]
- Goodson M, Rowbury RJ. Habituation to normally lethal acidity by prior growth of Escherichia coli at a sub-lethal acid pH value. Lett Appl Microbiol. 1989;8:77–79. [Google Scholar]
- Lin JS, Smith MP, Chapin KC, Baik HS, Bennett GN, Foster JW. Mechanisms of acid resistance in enterohemorrhagic Escherichia coli. Applied And Environmental Microbiology. 1996;62:3094–3100. doi: 10.1128/aem.62.9.3094-3100.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castanie-Cornet MP, Penfound TA, Smith D, Elliott JF, Foster JW. Control of acid resistance in Escherichia coli. Journal Of Bacteriology. 1999;181:3525–3535. doi: 10.1128/jb.181.11.3525-3535.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin JS, Lee IS, Frey J, Slonczewski JL, Foster JW. Comparative-Analysis Of Extreme Acid Survival In Salmonella-Typhimurium, Shigella-Flexneri, And Escherichia-Coli. Journal Of Bacteriology. 1995;177:4097–4104. doi: 10.1128/jb.177.14.4097-4104.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booth IR. The regulation of intracellular pH in bacteria. Novartis Found Symposium. 1999;221:19–37. doi: 10.1002/9780470515631.ch3. [DOI] [PubMed] [Google Scholar]
- Storz G, Imlay JA. Oxidative Stress. Current Opinions in Microbiology. 1999;2:188–194. doi: 10.1016/s1369-5274(99)80033-2. [DOI] [PubMed] [Google Scholar]
- Dien BS, Nichols NN, Bothast RJ. Recombinant Escherichia coli engineered for production of L-lactic acid from hexose and pentose sugars. Journal Of Industrial Microbiology & Biotechnology. 2001;27:259–264. doi: 10.1038/sj.jim.7000195. [DOI] [PubMed] [Google Scholar]
- Vemuri GN, Eiteman MA, Altman E. Effects of growth mode and pyruvate carboxylase on succinic acid production by metabolically engineered strains of Escherichia coli. Applied And Environmental Microbiology. 2002;68:1715–1727. doi: 10.1128/AEM.68.4.1715-1727.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang DE, Jung HC, Rhee JS, Pan JG. Homofermentative production of D- or L-lactate in metabolically engineered Escherichia coli RR1. Applied And Environmental Microbiology. 1999;65:1384–1389. doi: 10.1128/aem.65.4.1384-1389.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gruber PR, Hall ES, Kolstad JJ, Iwen ML, Benson RD, Borchardt RL. Continuous process for manufacture of lactide polymers with controlled optical purity. US Patent 5142023. Cargill, Inc.; 1992. [Google Scholar]
- Donnelly MI, Millard CS, Clark DP, Chen MJ, Rathke JW. A novel fermentation pathway in an Escherichia coli mutant producing succinic acid, acetic acid, and ethanol. Applied Biochemistry And Biotechnology. 1998;70-2:187–198. doi: 10.1007/BF02920135. [DOI] [PubMed] [Google Scholar]
- Marz U. RGA-103R Worldwide Markets for Fermentation Ingredients
- Paster M, Pellegrino JL, Carole TM. Industrial Bioproducts: Today and Tomorrow. Washington DC , US Department of Energy, Office of Energy Efficiency and Renewable Energy & Office of the Biomass Program; 2003. Energetics Incorporated. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table 1: Organic acids for platform biorefining applications. (* see references [64,65])