Abstract
As ageing is linked to the development of neurodegenerative diseases (NDs), such as Alzheimer’s Disease and Parkinson’s Disease, it is important to disentangle the independent effect of age-related changes from those due to disease processes. To do so, changes to central nervous system (CNS) cells as a function of advanced age need better characterisation. Microglia are of particular interest due to their proposed links with the development and progression of NDs through control of the CNS immune response. Therefore, understanding the extent to which microglial dysfunction is related to phyisological ageing, rather than a disease process, is critical. As microglia age, they are believed to take on a pro-inflammatory phenotype with a distinct dystrophic morphology. Nevertheless, while established hallmarks of ageing have been investigated across a range of other cell types, such as macrophages, a detailed consideration of functional changes that occur in aged microglia remains elusive. Here, we describe the dynamic phenotypes of microglia and evaluate the current state of understanding of microglial ageing, focusing on the recently updated twelve hallmarks of ageing. Understanding how these hallmarks present in microglia represents a step towards better characterisation of microglial ageing, which is essential in the development of more representative models of NDs.
Graphical Abstract
Microglial changes across different stages of life. Microglia have diverse functions throughout life; however, the characterisation of ageing hallmarks in microglia has been inconsistent
Keywords: Microglia, Ageing, Morphology, Neuroinflammation, Inflammageing, Immunosenescence
Introduction
Brain Changes with Age
As our population ages, healthy ageing and the ability to maintain an independent lifestyle in the later years of life are becoming increasingly relevant (Dlima et al. 2024). Globally, the number of individuals over the age of 60 is projected to double by 2050 (Rudnicka et al. 2020). This is significant, as ageing is the number one risk factor for developing neurodegenerative diseases (ND), such as Alzheimer’s Disease (AD) and Parkinson’s Disease (PD) (Hou et al. 2019), leading to dramatic expected increases in prevalence for these diseases (Hou et al. 2019; Pallas et al. 2008). In line with this, the number of global AD cases increased 160.84% from 1990–2019 (Nichols et al. 2019) and the number of PD cases rose 155.5% within the same period (Ou et al. 2021). Notably, both advanced age and NDs share alterations in several common underlying physiological processes, impacting the function of central nervous system (CNS) cells. López-Otín and colleagues (López-Otín et al. 2023) outlined 12 hallmarks of ageing, many of which show overlapping patterns of change with NDs. In fact, recent reviews have focused on these hallmarks in AD (Zhao and Huai 2023; Liu et al. 2024) and PD (Coleman and Martin 2022). Specifically, these 12 hallmarks include genomic instability, telomere attrition, epigenetic changes, loss of proteostasis, disabled macroautophagy, dysregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, chronic inflammation and dysbiosis. These are defined further in Table 1, with direction provided for further reading on the presentation of each of these in age-related NDs.
Table 1.
Definitions of the 12 hallmarks of ageing and their presentation in NDs
| Hallmark | Definition and main features as described by López-Otín and colleagues (López-Otín et al. 2023) | Presentation in age-related NDs | |
|---|---|---|---|
| AD | PD | ||
| Genomic instability |
The accumulation of genetic damage with age • Loss of DNA repair mechanisms • Accumulation of DNA damage • Increased frequency of DNA double-strand breaks |
↑DNA damage and impaired DNA repair mechanisms in post-mortem AD brains Reviewed: (Hou et al. 2017) |
↑DNA damage in post-mortem PD brains and experimental PD models Recent review: (Wang et al. 2023) |
| Telomere attrition |
The progressive shortening of telomeres • ↓telomerase activation • ↓telomerase reverse transcriptase (TERT) and telomerase RNA component (TERC) • Loss of telomere maintenance mechanisms |
↓peripheral telomere length in AD patients and telomere maintenance protects against AD pathology Recent review: (Kuan et al. 2023) |
Conflicting reports on telomere dynamics in PD. Lengthening and shortening observed in leukocytes but no changes observed in substantia nigra Recent review: (Vellingiri et al. 2023) Meta-analysis showing no association between PD and telomere length: (Forero et al. 2016) |
| Epigenetic changes |
Changes in epigenetic pathways • Histone modifications • DNA methylation • Chromatin remodelling • Altered gene expression without changes to DNA sequences |
Post-mortem AD brains show ↑DNA methylation and abnormal localisation of histone modifications Recent review: (Gao et al. 2022) |
Alterations in DNA methylation in human PD samples and experimental models Recent review: (Sharma et al. 2024) |
| Loss of proteostasis |
De-regulation of protein homeostasis • ↑ oxidative protein damage • Failure of protein quality control • Misfolded and incomplete proteins • Changes in proteasome capacity |
Ubiquitination and protein synthesis mechanism dysfunction is associated with AD risk and pathology in experimental AD models Recent review: (Cozachenco et al. 2023) |
Proteasome components are inhibited by Lewy body pathology and loss of proteasome is associated with PD pathology in the substantia nigra Review: (Lehtonen et al. 2019) |
| Disabled macroautophagy |
Impaired autophagic recycling of damaged organelles and proteins • ↓ recycling of cytoplasmic components |
Dysfunction associated with AD pathology Recent review: (Liu and Li 2019) |
Activation of macroautophagic pathways associated with neuronal degeneration and PD pathology Recent review: (Nechushtai et al. 2023) |
| Deregulated nutrient sensing |
Dysregulation of metabolic pathways involved in nutrient sensing • ↑activity of mTOR pathway • Dysregulated Insulin sensing • ↓AMPK and sirtuin pathways |
Signalling pathways are impaired in human AD and in experimental AD models mTOR pathway reviewed: (Perluigi et al. 2021) insulin pathways reviewed: (Gabbouj et al. 2019) AMPK pathway reviewed: (Cai et al. 2012) |
Signalling pathways are impaired in human PD samples and in experimental PD models mTOR pathway reviewed: (Lan et al. 2017) Insulin pathway reviewed: (Castilla-Cortázar et al. 2020) |
| Mitochondrial dysfunction |
Dysfunctional mitochondrial homeostasis and ↓function • ↓mitochondrial turnover • ↑ROS production • ↑mitochondrial membrane permeability |
Mitochondrial damage and dysfunction present in experimental AD models |
Mitochondria in post-mortem PD tissue and experimental models show reduced activity and impaired function Recent review: (Henrich et al. 2023) |
| Cellular senescence |
A state of irreversible growth arrest to prevent proliferation of damaged cells • Cell cycle arrest (↑p16INK4A, ↑ p21Cip1/Waf1) • Formation of senescence-associated heterochromatin foci (SAHF) • ↑ Inflammatory profile |
Neuronal and glial senescence associated with AD pathology (Bussian et al. 2018; Herdy et al. 2022)(Bhat et al. 2012) Recent review: (Liu 2022) |
Glial and neuronal senescence evident in experimental PD models and post-mortem PD brain tissue (Chinta et al. 2018; Shen et al. 2024) Recent review: (Miller et al. 2022) |
| Stem cell exhaustion |
Decline in number and function of stem cells • ↓Tissue renewal/impaired repair |
Altered neurogenesis (Haughey et al. 2002) AD pathology decreases proliferation in human neural stem cells (Lee et al. 2013) |
MPTP induces a premature senescent phenotype human neural stem cells (Zhu et al. 2015) |
| Altered intercellular communication |
Changes in signalling between cells • Altered activity of neural and hormonal pathways • Changes in extracellular molecules and cytokines • Changes in junction mediated communication |
Alterations in neuronal signalling Reviewed: (Gadhave et al. 2021) |
Alterations in substantia nigra dopaminergic signalling Recent review: (Dong-Chen et al. 2023) |
| Chronic inflammation |
Low-grade, persistent inflammation • ↑inflammatory responses ↓immune function • Overexpression of pro-inflammatory cytokines • Activation of immune cells |
↑inflammatory markers and glial activation in experimental and post-mortem brain tissue Recent review: (Wong-Guerra et al. 2023) |
↑ peripheral inflammatory cytokines in PD patients ↑microglial activation in post-mortem tissue Recent review: (Tansey et al. 2022) |
| Dysbiosis |
Imbalance of the gut microbiome • ↓microbial diversity and ↑harmful microbes |
↓microbial diversity, ↑gut-brain axis dysregulation Recent review: (Liu et al. 2020) |
Altered composition of gut microbiome Recent systematic review: (Li et al. 2023b) |
The pathological commonalities between ageing hallmarks and NDs underscores the need to disentangle the independent effects of physiological age-related changes from those due to disease processes. Understanding each of these hallmarks on a molecular level is crucial for improving our ability to replicate the cellular environment that occurs in NDs. However, these changes may present differently in different CNS cell types. For example, cell cycle arrest is a defining characteristic of cellular senescence, a hallmark of ageing (López-Otín et al. 2023); however, in post-mitotic cells, such as neurons, cell cycle arrest does not function as a marker of an aged cell (Sapieha and Mallette 2018). Conversely, microglia and astrocytes are capable of division (Joya and Martín 2021) and, thus, have the potential to express this element of cellular senescence. Given the role of glial cells in supporting neuronal health (Rahman et al. 2022), characterising ageing in these support cells is critical. While both microglia and astrocytes have important immune functions in the CNS (see reviews (Borst et al. 2021), (Giovannoni and Quintana 2020)), microglia are considered the primary drivers of the immune response (Ginhoux et al. 2013) and are the focus of the current review.
Microglial Function Across the Lifespan
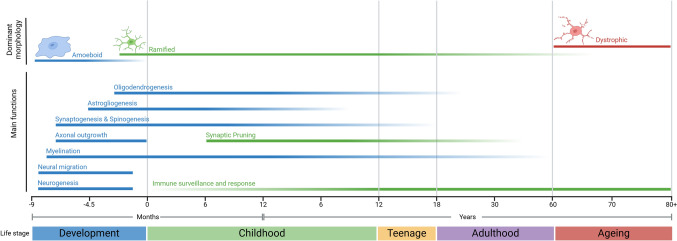
Microglia are the tissue resident macrophages in the CNS, accounting for 5–20% of the total cell population in the brain (Hugh Perry 1998). Microglia arise from progenitor cells in the yolk sac during early development (Cuadros et al. 1993; Alliot et al. 1999), where they are responsible for facilitating cell migration, neurogenesis and gliogenesis (Menassa and Gomez-Nicola 2018). Conversely, while they have responsibilities in CNS maintenance, including synaptic pruning and phagocytosis of apoptotic cells (Borst et al. 2021), their major role in the adult CNS is the initiation of an adequate immune response to injury or infection (Ginhoux et al. 2013). Due to their dual role in both homeostasis and immune functions, microglia are highly dynamic cells, with constantly changing morphologies and functional phenotypes throughout the lifespan (Fig. 1).
Fig. 1.
Microglial roles and morphologies during different stages of life. During development and early childhood, microglia have major roles in supporting the development of the CNS. Before birth, microglia display an amoeboid morphology. Ramified morphology begins to become the dominant morphology at the time of birth and remains so until late adulthood. During childhood, microglia maintain developmental roles through synaptic pruning, but transition to their major immune role around adolescence. During ageing, microglia acquire a dystrophic morphology; however, the functional changes at this stage remain unclear. Figure created in BioRender
During development and early childhood, microglia are amoeboid and migratory cells (Menassa et al. 2022), responsible for neurogenesis (Matsui and Mori 2018; Cunningham et al. 2013), neuronal migration (Ueno and Yamashita 2014), synaptic connections (Miyamoto et al. 2016) and axon growth (Squarzoni et al. 2014)). Additionally, microglia support the differentiation of astrocytes and oligodendrocytes from progenitor cells (Reemst et al. 2016; Borst et al. 2021). This ensures successful ongoing brain development and facilitates CNS plasticity during critical developmental periods (Mallya et al. 2019). Throughout the late teenage/young adult years, microglia further facilitate synaptic pruning to maintain network plasticity (Paolicelli et al. 2011; Mastenbroek et al. 2024). As individuals progress through early and middle adulthood, microglia maintain a surveillant non-inflammatory phenotype, with much of their population displaying ramified morphology (Harry and Kraft 2012).
Conversely, as individuals move into older adulthood (i.e. 60 + years of age), microglia typically display a dystrophic morphology (Greenwood and Brown 2021), also called “senescent microglia”, which features marked de-ramification with remaining processes fragmented or swollen (Neumann et al. 2023). These cells have been mainly characterised in aged human post-mortem tissue (Lopes et al. 2008; Streit et al. 2004) and studied to a lesser extent in aged mice (d'Avila et al. 2018; St-Pierre et al. 2022). Such microglial dystrophy has been proposed as more reflective of a disease-associated phenotype, rather than ageing itself (Streit et al. 2009; Shahidehpour et al. 2021). However, the idea that microglia develop an abnormal morphology, characterised mainly by process swelling and fragmentation, during ageing remains the general consensus (Wendimu and Hooks 2022; Paolicelli et al. 2022; Augusto-Oliveira et al. 2025). Beyond morphological change, it is well documented that microglia have a decreased ability to perform homeostatic functions and exhibit an altered immune response with advancing age (Niraula et al. 2017). This includes a more reactive phenotype, with microglia existing in a primed state featuring heightened immune responses, with longer time required to resolve the response following immune pathway activation (Norden et al. 2015). In line with this, when compared to microglia in 3-to 4-mo male Sprague–Dawley rats, those from 22-month-old (mo) rats showed higher numbers of microglia featuring an “activated” morphology (short, thick processes and small cell body) following intracerebral haemorrhage induced by collagenase injection to the striatum (Wasserman et al. 2008). Further, microglia from these aged rats demonstrated a prolonged inflammatory response following injury (Wasserman et al. 2008). Similarly, 18-to 20-mo male BALB/c mice demonstrated a hyperactive microglial response to lipopolysaccharide (LPS) stimulation compared to 3-to 4-mo mice (Henry et al. 2009).
To better understand microglial diversity, the literature has proposed various systems of classification and subtyping based on transcriptomic and proteomic analyses, as well as morphological distinctions. Transcriptomics provides a valuable tool to distinguish populations of microglia during ageing and disease (Stratoulias et al. 2019). This analysis has been used to identify specific subtypes of microglia using single-cell RNA sequencing in tissue collected from both human post-mortem samples and mouse AD models. Keren-Shual and colleagues (2017) identified a disease-associated microglia (DAM) subtype which exists in close proximity to markers of disease pathology and has a unique signature (Keren-Shaul et al. 2017). This signature is characterised by increased phagocytosis and activation of pathways linked to increased AD risk (including APOE, TREM-2 and CTSD) (Keren-Shaul et al. 2017). The DAM subtype has also been linked to other neurodegenerative conditions, including Amyotrophic Lateral Sclerosis (ALS) (Keren-Shaul et al. 2017; Jauregui et al. 2023) and PD (Schonhoff et al. 2023). Outside of disease, age-related transcriptomic profiles of microglia have also been identified using single-cell RNA sequencing. For example, Olah et al. (2018) identified a set of genes expressed primarily by microglia in the aged human dorsolateral prefrontal cortex, which was termed the HuMi_Aged dataset. When compared to another transcriptomic dataset from middle-aged microglia (Zhang et al. 2016), genes such as nuclear factor κ B subunit 1 (NFKB1), interleukin 1β (IL1 β), triggering receptor expressed on myeloid cells 1(TREM-1), Toll-like receptor 4 (TLR4) and cluster of differentiation 83 (CD83) were found to be downregulated with age. In contrast, genes including translocator protein (TSPO), cluster of differentiation 14 (CD14), presenilin enhancer gamma-secretase subunit (PEN-2), and cathepsin D (CTSD) were upregulated (Olah et al. 2018). This suggests that microglia undergo a shift in their genetic profile with age, involving alterations in immune signalling and responses. Further, transcriptional analysis of microglia across the mouse lifespan (E14 to P540) has been conducted by Hammond et al. (2019). This study did not identify specific clusters of microglia, but instead observed the expansion of inflammatory and interferon responding groups of microglia at 18-mo of age. Interestingly, however, despite identifying these enriched clusters, they made up only a small overall number of microglia, indicating that while numbers of pro-inflammatory cells may increase with age, the majority of the microglial population, at least in in mice, does not show a drastic transcriptomic shift with increased age (Hammond et al. 2019). This disparity between microglial changes in human and mice highlights the need for cross-species studies to understand the translatability of results, although a discussion of this is outside of the scope of the current review (for review see: (Barron et al. 2021)).
Similarly, proteomic analysis has contributed greatly to our understanding of microglia. Microglia isolated from the brains of patients undergoing treatment for medial temporal lobe epilepsy (n = 3) underwent proteomic analysis using cytometry by time of flight (CyTOF) (Böttcher et al. 2019). This analysis uncovered distinct patterns of protein expression across brain regions, namely an increased expression of markers of microglial activation (CD68, CD86, CD45, HLD-DR) in the thalamus and subventricular zone compared to the cerebellum, temporal lobe and frontal lobe. Further, this study confirmed the expression of proteins P2RY12, TMEM119, TREM2, EMR1 and CD64 as part of a signature that could be used to distinguish microglia from other myeloid cell types (Böttcher et al. 2019). In another study, proteomic analysis of microglia isolated from the brains of the APP NL-F mouse model of AD revealed upregulation of antigen presenting and interferon response proteins, and downregulation of proteins involved in homeostasis and cell migration, at 6- and 12-mo compared to WT controls. This study provides a snapshot not only of how microglial protein signatures change with disease, but also with age, as these alterations were not observed in this model at 3 mo (Sebastian Monasor et al. 2020). During ageing, the microglial proteome in mice shifts to feature decreased expression of homeostatic proteins (P2RY12 and TMEM119 (Grubman et al. 2021)) and increased expression of lysosomal proteins (LYZ (Grubman et al. 2021), HLA-A and CST7 (Keane et al. 2021)), proteins involved in phagocytosis (LPL (Keane et al. 2021)) and microglial priming (ITGAX, SPP1 (Keane et al. 2021)), and antigen presenting proteins (HLA-DQB1 and CD74 (Grubman et al. 2021)). Overall, the use of proteomics to understand microglial function across health and disease is limited compared to transcriptomics (Davis and Lloyd 2024). These technologies, particularly when applied at the single-cell level, offer powerful insights into how microglial phenotypes change with age and disease. However, while both methods are gaining popularity in the field, they are not yet widely applied, partially due to their high cost and need for specialised infrastructure (Williams et al. 2022). As a result, more traditional approaches remain commonly employed, although these methods are not without their limitations. For example, morphological classification of microglia is widespread, despite its inherent subjectivity (Young and Morrison 2018), and difficulties associated with directly comparing between studies due to inconsistent terminology (Paolicelli et al. 2022). Nevertheless, the use of morphological classification, in conjunction with immunohistochemical or immunofluorescent staining for functional markers, continues to be the most used method to assess microglial phenotypes in in vivo pre-clinical studies.
Molecular Mechanisms and Morphology of Microglial Phenotypes
Various morphological classifications for microglia are utilised across the literature, which often have overlapping functions and markers. Studies have observed changes in cellular markers and secretory profiles, alongside shifts in microglial morphology, which has allowed researchers insight into potential functions of morphological phenotypes (e.g. rod microglia (Bachstetter et al. 2017)). These morphological classifications and observed corresponding functions, markers and secreted factors are summarised in Fig. 2. Importantly, although morphology is a common method of classifying microglia, morphological change does not always correlate with an overall shift in function. Therefore, when assessing changes in microglial populations, it is best practice to pair morphological analysis with the use of known functional markers, such as MHCII, to assess immune activation (Jurga et al. 2020). It should be further noted that microglial classification throughout the literature is inconsistent, with morphological terms being used interchangeably such as bushy and hyper-ramified (see: Ziebell et al. 2015; Reddaway et al. 2023)). Further, less characterised morphologies, such as rod microglia are vaguely described, with little understanding of their functional phenotype (Giordano et al. 2021).
Fig. 2.
Microglial classifications used throughout the literature and associated morphologies, functions and markers. Green: Phenotypes present under physiological conditions, Yellow: Phenotypes present following induction of immune response (i.e. response to injury/infection), Red: Phenotypes present under dysregulated or pathological conditions a Ramified microglia are in a state of CNS surveillance, with low baseline levels of inflammatory cytokine release. b Hyper-ramified microglia functionally have either a heightened or an impaired response to stimuli. c Reactive microglia initiate the immune response. d Bushy microglia are thought to be transitioning towards immune activation and have some phagocytic and inflammatory functions. e Rod microglia are associated with degeneration and injury, with potential roles in structural support of neurons. f Amoeboid microglia function as macrophages, with main roles in phagocytosis and secretion of pro-inflammatory factors. g Dystrophic microglia are associated with neurodegeneration and ageing and have a diminished capacity to perform normal homeostatic functions. *Markers expressed on microglial cells within each classification. #Factors measured in conjunction with microglia for each classification. Images from BioRender
In response to injury or infection, the microglial immune response is initiated. This immune response involves alterations in expression of surface markers, secretion of signalling factors and is often accompanied by a shift in morphology. The classical definition of microglial activation is displaying a mobile, amoeboid morphology (Fig. 2f), increased phagocytosis and increased release of pro-inflammatory cytokines (Giulian and Baker 1986). The activation of a microglial immune response has often been shown to occur in the presence of protein aggregation, as occurs during ageing and in age-related NDs (Scheiblich et al. 2021; Barger and Harmon 1997; Gao et al. 2023). This activation occurs through a number of pathways involving pattern recognition receptors (PRRs), which allow the recognition of pathogenic signals such as damage-associated molecular patterns (DAMPs), and pathogen-associated molecular patterns (PAMPs) (Glezer et al. 2007; Kigerl et al. 2014). These pathways are summarised in Fig. 3a; however, microglia can also undergo inflammatory activation driven by intracellular DNA-sensing mechanisms which are summarised in Fig. 3b.
Fig. 3.
Pathways of microglial responses. a PRR response pathways in microglia. PRRs, such as 1. TLRs, 2. RLRs, and 3. NLRs, recognise PAMPs/DAMPs and trigger signalling cascades, including 4. Exposure of NF-κB subunits by degradation if IκB and translocation of NF-κB to the nucleus. These cascades cause the downstream release of inflammatory cytokines. b DNA-driven immune responses in microglia. 1. cGAS binds to free DNA in the cytosol (mtDNA, nuclear DNA, foreign DNA), which activates the cGAS-STING pathway, causing the transcription of pro-inflammatory genes and release of pro-inflammatory cytokines. Figure was created using BioRender
Historically, microglia have been referred to as resting or activated. However, these definitions are now considered inaccurate, as microglia are in fact constantly “active” and perform many functions under homeostatic conditions (Paolicelli et al. 2022). Notably, studies often use the term “microglial activation” to refer to microglia that are reacting to a stimulus or undergoing the induction of an immune response. The major phenotypes that this terminology encompasses include those that are present under homeostatic conditions, during an immune response, and under disease conditions. As shown in Fig. 2, various morphological classes of microglia can exist under both homeostatic and responsive states. When discussing previous publications where microglia have been referred to as activated, the current paper endeavours to specify the phenotype observed with reference to those outlined in Fig. 2. Where previous observations are vague or unclear, the terminology used by original authors has been used here, with additional information regarding the type of “activation” provided where possible.
Considering the aged background on which NDs such as AD and PD occur, it is reasonable to infer that age-related alterations in microglia are a major contributor to disease pathophysiology in NDs (Gamage et al. 2023; Zhang et al. 2023). Given this link, the importance of including age as a factor in the pre-clinical modelling of NDs cannot be understated. However, there is a clear lack of consideration of age in current pre-clinical studies of NDs (reviewed: Carr et al. 2024; Sun et al. 2020)). In order to better model the age-related changes occurring in CNS cells, there needs to be broader understanding of how the CNS immune response changes with advancing age (Angelova and Brown 2019), particularly the role of microglia in this process.
Understanding Molecular Changes in Microglia Through the Hallmarks of Ageing
As discussed above, changes in microglial inflammatory pathways and morphology with ageing and disease have been previously described. However, there has been little characterisation of microglia with reference to the known hallmarks of ageing. The hallmarks of ageing (López-Otín et al. 2023) represent specific patterns of change in critical systems which can distinguish an aged phenotype and have been observed in both AD and PD (Table 1). These hallmarks have been described in peripheral macrophages (Guimarães et al. 2021) but are less well investigated specifically in microglia, with studies instead focusing on how microglial production and release of pro-inflammatory factors and microglial immune responses change with age. Below, we review the studies to date that have explored the presentation of these hallmarks in microglia with advancing age, with a focus on how these changes may be relevant in the context of neurodegeneration.
Genomic Instability
Genomic instability as a hallmark of ageing is defined by an accumulation of damaged DNA and loss of or alterations in DNA repair pathways (Chen et al. 2022). In microglia, this has been studied both preclinically and in human post-mortem tissue. In 24-mo C57BL/6 male and female mice, microglia have been shown to accumulate damaged DNA fragments as identified by γ-H2A.X staining and an increase in the percentage of pSTING + /CD11b + cells compared to 2-mo animals (Arvanitaki et al. 2024). These changes indicate activation of the DNA-driven immune response as a contributing factor to the acquisition of a pro-inflammatory phenotype in microglia. Similarly, the Ercc1Δ/− mouse is deficient in DNA repair mechanisms and displays features of accelerated ageing. In Ercc1Δ/− mice at 16-weeks-old following peripheral LPS injection, microglia displayed hypertrophied processes and an increase in cell body size, along with increased proliferation, phagocytosis and ROS production compared to wild-type (WT) controls (Raj et al. 2014). In addition, Ercc1Δ/−mice showed evidence of microglial priming, indicated by an enhanced inflammatory response following peripheral LPS challenge, a result which has also been replicated in Ercc1Δ/−mice at older ages (4-, 8- and 14-mo) (Zhang et al. 2021). While DNA damage itself was not explicitly measured in microglia in either of these studies, these results implicate a loss of DNA repair mechanisms in driving microglial ageing and pro-inflammatory microglial responses.
Conversely, in human post-mortem tissue from 14 healthy controls (6M:8F) aged 65–96 years old, dystrophic microglia did not show positive γ-H2A.X staining (Neumann et al. 2023), but were strongly correlated with increased ferritin expression, a marker also known to increase with ageing. Ferritin protein, most prominently expressed in microglia in the brain, can store excess iron and increased expression of ferritin in microglia can be used as an indicator of iron overload in the brain (Lopes et al. 2008; Streit et al. 2018). Iron overload has been linked to increased γ-H2A.X in hiPSCs (Han et al. 2020) with the role of ferritin in microglia proposed to be to mediate the damaging effects of excess iron in the brain (Neumann et al. 2023). However, the accumulation of iron-containing ferritin protein has been suggested as a contributing factor to the dystrophic morphology observed in aged microglia (Neumann et al. 2023). While healthy control brains did not show increased DNA damage markers with age when assessed by γ-H2A.X staining, another human post-mortem study in older adults (mean age 94.07 years ± 0.95) identified a subset of genes preferentially expressed by aged microglia, termed the HuMi_Aged dataset (Olah et al. 2018). DNA damage-associated pathways were enriched in this dataset, along with telomere maintenance and chromosomal maintenance pathways (Olah et al. 2018). These results together suggest that while γ-H2A.X expression is not associated with aged microglia in humans, alterations in DNA damage pathways are indeed present in aged human microglia. However, the specific changes which occur due to alterations in these pathways are yet to be identified. These results should also be interpreted with caution, as they are derived from only two studies, with the former containing a relatively small sample size and no young control cohort. This represents a wider issue in the field, with young healthy brain tissue difficult to obtain (Bell et al. 2008), making human post-mortem studies challenging and leaving remaining questions about how well age-related changes observed in pre-clinical rodent models reflect those seen in the in human brain. While large animal models, which are arguably more translational (Sorby-Adams et al. 2018), can be used to investigate microglial alterations, to our knowledge, none have yet been used to investigate changes that occur in the microglial genome with age. This necessitates further research into this area to uncover the extent to which typical markers of senescence, such as accumulated DNA damage, can be observed in aged microglia across species.
Telomere Shortening
Telomere shortening is one of the most widely recognised and accepted hallmarks of ageing (Fasching 2018). Telomeres, protective DNA sequences which cap the end of chromosomes, function to stabilise the ends of chromosomes and protect genomic material from degradation (Vaiserman and Krasnienkov 2021). However, with each cell division, telomeres become progressively shorter due to the inability of DNA polymerase to fully replicate the end of the C-rich lagging strand (Vaiserman and Krasnienkov 2021). This effect is referred to as the end replication problem, where telomeres will shorten until reaching a critical length where they can no longer protect the chromosome (Vaiserman and Krasnienkov 2021). This triggers cell cycle arrest and cells either enter a senescent state or undergo apoptosis. Telomere length has been widely utilised as a biomarker of ageing (Sanders and Newman 2013). However, there is debate as to whether these pre-clinical results have translational relevance for human ageing, where there have been conflicting results regarding the association of telomere length and age. While a full discussion of this debate is beyond the scope of this review, detailed information regarding the validity of telomere shortening as a marker of ageing can be found: (Mather et al. 2011; Vaiserman and Krasnienkov 2021).
The evidence for telomere length as a marker of ageing has come primarily from peripheral blood leukocytes and fibroblasts. A 2022 systematic review and meta-analysis of 27 studies reported that longer leukocyte telomeres were associated with lower levels of age-related structural change (brain volume loss) and cognitive decline in non-demented cases (Gampawar et al. 2022). Telomere alterations in the brain itself have been less well investigated. Nevertheless, some studies in isolated rat microglia demonstrate telomere shortening with age. The first study to investigate glial telomeres was conducted by Flanary and Streit (Flanary and Streit 2004), who found telomeres in microglia from neonatal rat cortices shorten over 32 days in culture when measured using flow cytometry fluorescent in situ hybridization (flow-FISH). Telomerase activity in these microglia declined steadily until day 24, but increased dramatically from then until the end of the 32-day culture period (Flanary and Streit 2004). Similarly, in a 2007 study by the same authors, microglia isolated from 30-mo male Fisher-344 rats were found to have shorter telomeres than microglia isolated from 3-mo rats when measured using flow-FISH (Flanary et al. 2007). These aged microglia also had decreased telomerase activity compared to young microglia, indicating both a decline in telomere length and telomere maintenance with age in rodent microglia (Flanary et al. 2007). Of note, the authors also investigated microglial telomere length in human post-mortem tissue from AD patients in this study. Microglia were isolated from the superior frontal gyrus/frontal pole region of 4 AD (86 years old ± 2.54) and one non-dementia case (86 years old), and microglial telomere length and telomerase activity were measured. Telomeres in AD microglia were significantly shorter than those seen in the non-dementia case; however, no changes in telomerase activity were observed, indicating that decreases in telomerase activity are not necessary for shortening of telomeres to occur (Flanary et al. 2007). Notably, however, there was no investigation of microglial telomere length compared to young human tissue, likely due to limitations associated with obtaining post-mortem tissue from young donors, as discussed above.
Several other studies have also investigated telomere shortening in the context of NDs. For example, one study utilised a cross between the APP23 transgenic mouse model of AD and the Telomerase RNA component (Terc) knockout model of telomere shortening (G3Terc−/−APP23+) to investigate AD pathology on the background of shortened telomeres (Rolyan et al. 2011). Interestingly, at 12-mo G3Terc−/−APP23+ mice had reduced Aβ pathology in the frontal cortex, as well as improved spatial memory compared to APP23+ mice. In addition, G3Terc−/−APP23+ mice showed no increase in microglial immune activation (MHCII+ microglia) in the vicinity of Aβ plaques (Rolyan et al. 2011). Similarly, a cross between a pre-clinical model of PD, the Thy-1[A30P] α-synuclein transgenic mouse model, and Terc−/− mice (αSYNtg/tgG3Terc−/−) showed decreased microglial Il-1β gene expression and reductions in expression of MHCII in the brainstem of 17-mo αSYNtg/tgG3Terc−/− mice compared to αSYNtg/tg mice (Scheffold et al. 2016). Taken together, these results surprisingly seem to suggest that telomere shortening is protective against the induction of microglial immune responses to disease pathology. However, contrary to what was observed in the AD mouse model, accelerated disease phenotype and early death were seen in PD mice. This suggests that loss of telomere maintenance may inhibit a protective response by microglia, ultimately leading to worsened disease phenotypes, at least in older animals. Further research is required to unravel the mechanisms that underlie these processes. In addition, as telomere shortening was artificially induced via Terc knockout in both of these previous studies, it is important to investigate how this may differ from changes that occur with physiological ageing.
Epigenetic Changes
Epigenetic changes encompass a range of heritable changes that cause alterations in gene expression, without directly changing DNA sequences (la Torre et al. 2023). The changes most studied with regard to ageing include chromatin remodelling, histone modifications and DNA methylation (see review: (la Torre et al. 2023)). Alterations in all these processes have been reported to occur in microglia with advancing age, with DNA methylation and histone deacetylase pathways identified as two of the enriched pathways in the HiMi_Aged dataset discussed above (Olah et al. 2018), suggesting specific changes in these pathways in human-aged microglia. Chromatin remodelling refers to alterations in the structure and organisation of chromatin and includes altered chromatin accessibility (CA), a measure of how easily genes may be accessed for transcription (Li et al. 2023a). One study, which conducted mapping of CA by ATAC-seq in the microglia of female and male C57BL/6J mice at 3-, 14- and 24-mo, described a number of genes differentially expressed with age that were termed age-dependent microglia genes (ADEM genes) (Li et al. 2023a). Genes which positively correlated with age were termed P-ADEM genes (notably genes related to phagocytosis, IFN signalling, ROS production and antigen presentation), while genes that negatively correlated with age were designated as N-ADEM genes (including chemokine suppression and iron transportation genes). P-ADEM genes had a higher CA than N-ADEM genes in promoter regions, indicating that alterations in CA of ADEM genes may dictate the emergence of specific microglial phenotypes during ageing (Li et al. 2023a). For example, the increased CA of ROS production genes (Cybb and Hp) and IFN signalling genes (Ifitm3, Ifi204, Cxcl16, Xaf1, Gas6 and Tgtp2) suggests a shift towards a more pro-inflammatory phenotype in microglia, potentially leading to increased microglia driven inflammation with age.
Histone modifications have also been shown to occur in microglia with advancing age. In line with this, an age-related decrease in the histone demethylase Jumonji domain-containing protein-3 (JMJD3) has been observed in microglia of male C57BL/6 mice aged 16–18-mo compared to 2- to 3-mo mice (Tang et al. 2014). JMJD3 is suggested to be necessary for microglia to initiate an anti-inflammatory response (Tang et al. 2014). Thus, its decrease with age is potentially representative of a shift towards a pro-inflammatory phenotype with advancing age. This study also found an increased level of the histone H3K27me3 in the midbrain of aged mice. H3K27me3 is the trimethylation of lysine 27 on histone H3, a histone modification which is associated with reduced transcriptional activity, which can be reduced by JMJD3. Thus, this increase in H3K27me3 may be a consequence of the decrease in JMJD3 seen in microglia with advanced age and may further contribute to imbalances in microglial responses.
Finally, DNA methylation refers to the addition of a methyl group to a DNA molecule, typically associated with silencing of genes. In microglia isolated from young adult (4- to 6-mo) and aged (24- to 26-mo) C57BL/6 mice exposed to LPS, an increase in IL-1β expression was observed with age (Matt et al. 2016). When probed further, IL-1β promoter DNA in aged microglia from older animals showed decreased methylation compared to that seen in younger animals (Matt et al. 2016). This suggests that, with age, silencing of pro-inflammatory cytokine genes is reduced following an inflammatory stimulus, potentially leading to an overactive microglial response. Overall, changes in CA and DNA methylation in microglia suggest a shift towards pro-inflammatory secretory phenotype, with histone modifications contributing to a reduced likelihood of taking on an anti-inflammatory phenotype. Taken together, this provides compelling evidence that microglia undergo a number of epigenetic changes with advancing age which cause an enhanced pro-inflammatory response. This response may be further exacerbated in the presence of ND-associated pathological proteins, such as Aβ in AD or α-synuclein in PD.
Overall, characterisation of epigenetic changes in microglia during ageing is sparce and specific pathways should be further investigated to understand the functional consequences of these changes. For example, Olah et al. (2018) identified DNA methylation and histone deacetylases as enriched in aged human microglia. Notably, however, this study did not investigate the specifics of either of these modifications and key questions remain. What genes are affected by the altered DNA methylation? Does this preferentially impact functional pathways in microglia, such as protein degradation or inflammatory pathways? Do either of these epigenetic alterations contribute to changes in overall microglial phenotypes with age? Investigating the functional consequences of these changes in microglia will improve our understanding of microglial ageing and which changes can be specifically targeted to improve outcomes with age and in NDs.
Loss of Proteostasis
López-Otín and colleagues describe the hallmark loss of proteostasis as dysregulation of the intracellular mechanisms responsible for the degradation of proteins (López-Otín et al. 2023). A decline of proteostasis is often related to the aggregation and accumulation of proteins, a hallmark of neurodegenerative diseases such as AD and PD. Thus, the study of microglial proteostasis with age is critical in order to understand the mechanisms by which pathology may be targeted. In the HuMi_Aged dataset, the ER-phagosome pathway was enriched and upregulated in aged microglia (Olah et al. 2018) when compared to previous data generated from middle-aged microglia (Olah et al. 2018; Zhang et al. 2016). The ER-phagosome pathway facilitates communication of the endoplasmic reticulum (ER) with phagosomes and supports the maturation of phagosomes (Ghavami and Fairn 2022). This enrichment may, therefore, be reflective of a shift towards increased immune surveillance and increased antigen presentation. Indeed, Olah et al. (2018) also found enrichment of antigen processing-cross presenting pathways in their analysis. In addition, this enhanced activity of the ER-phagosome pathway may also be acting as a compensatory mechanism to combat proteostatic stress as a result of accumulation of misfolded proteins in the ER such as collagen 1 (De Leonibus et al. 2024), but no studies have yet investigated this mechanism in microglia. Further, proteomic analysis of microglia isolated from young (3- to 5-mo) and aged (20- to 24-mo) mice revealed differential expression in a number of proteins, including downregulation of proteasome complex subunits PSMB4, PSMA6 and PSMC1 (Flowers et al. 2017). Downregulation of these subunits would likely impair the ability of microglia to degrade damaged or misfolded proteins, leading to ineffective protein clearance in aged tissues. In AD, for example, microglia are important for degradation of Aβ (Qiu et al. 1997). However, if their capacity for proteasomal degradation is impaired, this may lead to accumulation of pathological Aβ within microglia, further disrupting proteostasis and protein degradation ability (Pomilio et al. 2020). In APP/PS1 mice also deficient in the immunoproteasome component LMP7, microglial immunoreactivity was decreased at 8-mo when compared to APP/PS1 mice with LMP7 of the same age. Interestingly, however, this change in microglial state did not alter the presence of Aβ pathology in these mice at any age (Wagner et al. 2017). It is important to note that this was only assessed at 8-mo, relatively young compared to other studies on microglial ageing, and results may, therefore, differ in older animals. Accordingly, tamoxifen-induced deletion of COP1 E3 ubiquitin ligase at ~ 2-mo of age resulted in increased expression of microglial genes involved in IFN signalling and APOE, a major AD risk factor gene, at 22-mo transgenic C57BL/6N mice, compared to WT mice (Ndoja et al. 2020). This result suggests that, in addition to accumulation of damaged or misfolded proteins, the loss of negative regulatory mechanisms, such as COP1 mediated ubiquitination, can promote pro-inflammatory signalling within microglia, which may contribute to sustained microglial immune activation during ageing.
Such effects may also be sex specific, at least to some extent. For example, in 18-mo-old C57BL/6 mice, microglia showed sex-dependent alterations in proteostasis. Microglia in female mice demonstrated a decreased ability to phagocytose Aβ42, while microglia from male mice showed a decrease in proteolytic gene expression, but no change in phagocytic abilities (Thomas et al. 2022). Of note, microglia from both sexes at 22-mo of age had decreased expression of the phagocytic receptor TREM2 (Thomas et al. 2022), suggesting a potential alteration in recognition of pathological proteins in aged microglia. Overall, there is mounting evidence that there is a de-regulation of the protein degradation pathways within microglia with advancing age. This may have significant implications for the pathogenesis of diseases like AD and PD, where an inability of microglia to combat pathological protein aggregation may contribute to the spread of pathology throughout the brain (Gao et al. 2023).
Disabled Macroautophagy
Macroautophagy is defined as the recycling of large cargo and cellular contents by lysosomes, with cargo packaged into specialised vesicles called autophagosomes (Nieto-Torres and Hansen 2021). With age, the efficiency of cellular autophagy declines, resulting in a decreased turnover of organelles, protein and lipid degradation and reduced ability to dispose of pathogens (Nieto-Torres and Hansen 2021). In microglia, age-related autophagic changes are understudied, with most research to date focused on these changes in neurons. However, there has been some investigation of changes with age in microglial degradation pathways. Disruptions in macroautophagy have been investigated in vitro via knockout of ATG7, a key autophagic gene, in both primary microglia and BV2-microglia-like cells. ATG7 knockout has been shown to increase expression of Nod-Like Receptor Pyrin Domain-3 (NLRP3) inflammasome components CASP-1 and ASC following administration and uptake of Aβ (Cho et al. 2014). Thus, disruption of microglial autophagy may cause expression of pro-inflammatory factors and priming of microglia for expression of the NLRP3 inflammasome upon NLRP3 activation.
Although this mechanism has not been specifically investigated in the context ageing, disruption of Aβ receptor turnover in microglia from aged animals, combined with evidence of macroautophagic disruption in peripheral macrophages, strengthens the possibility that dysregulated receptor turnover occurs with age and impairs microglial responses. For example, in bone marrow derived macrophages isolated from 23-mo C57BL/6 mice, there was reduced expression of autophagic genes (ATG6 and ATG7) and impaired autophagic flux measured by LC3 (Stranks et al. 2015). Of note, like the above results seen in ATG7 deficient primary microglia, ATG7 deficient macrophages in this study demonstrated a pro-inflammatory phenotype. This suggests a role of dysregulated autophagy in macrophage-driven inflammation and solidifies the theory that similar processes may occur in microglia with age, potentially predisposing these cells to an inadequate response to ND pathology.
While distinct from macroautophagy, microglia also rely on distinct endocytic pathways to process extracellular material, and these pathways have been shown to change with age. LC3-associated endocytosis (LANDO) is critical in the turnover of Aβ receptors (including TLR4 and TREM2) on microglia, ensuring correct recognition of the Aβ protein (Heckmann et al.). Microglia isolated from 24-mo mice lacking the WD domain of Atg16L, which is necessary for LANDO recycling of Aβ receptors, demonstrated decreases in uptake of Aβ in culture compared to aged-matched WT controls, suggesting a role for Aβ receptor turnover in dysfunctional proteostasis with age (Heckmann et al. 2020). This suggests that turnover of microglial surface receptors is crucial to ensure that microglia can respond adequately to pathological proteins, and loss of these mechanisms with age may limit the ability of microglia to respond to pathology. Further investigation is necessary to determine whether macroautophagic and endocytic pathways interact in aged microglia and how potential changes in these pathways may influence microglial ageing.
Dysregulated Nutrient Sensing
Ageing is associated with an overall decline in the availability of nutrients, due, in large part, to alterations in food and nutrition habits of older adults, influenced by reduced appetite, difficulties accessing food and altered taste and smell (Robinson 2018). Deficiencies in vitamin B12, Calcium, Iron and Vitamin D are common, as older adults rely more on heavily processed foods with low nutritional value (Rémond et al. 2015). In addition, changes in metabolism including insulin resistance and reduced mitochondrial efficiency, can influence nutrient processing at a cellular level (Gao et al. 2014). In response to these changes, key nutrient signalling pathways are affected, including the mechanistic target of rapamycin (mTOR), the 5’AMP-activated protein kinase (AMPK) (Jewell and Guan 2013; Khan et al. 2025) and insulin/Insulin-like growth factor-1 (IGF-1) signalling pathways. These disruptions affect cellular homeostasis, insulin responses and energy production across multiple tissues, including the brain, where these pathways and associated signalling cascades are critical in maintaining CNS homeostasis (Doust et al. 2022). The critical role of microglial metabolic plasticity in maintaining immune function under energetic stress has been highlighted by Bernier et al. (2020), who demonstrated that microglia from C57BL/6 mice (4-mo) are capable of rapidly adapting to glucose deprivation by switching to glutaminolysis (Bernier et al. 2020). This switch was reliant on mTOR-dependent signalling, highlighting the importance of effective nutrient sensing pathways in maintaining microglial function under stressful environments, such as are present during ageing and disease. In fact, the capacity of microglia to sustain bioenergetic processes has been shown to decrease with age. Compared to microglia from young mice (2–4-mo), microglia from aged mice (20–24-mo) demonstrate increased glucose conversion to glycogen and reduced mitochondrial respiration (Minhas et al. 2021), resulting in an energy deficient state. Importantly, this state was able to be reversed in microglia by inhibition of EP2 signalling, which restored metabolic function, suggesting that impairments in microglial nutrient sensing deficiencies are potentially targetable through metabolic reprogramming (Minhas et al. 2021). These results suggest that nutrient availability and metabolic flexibility in microglia are crucial for maintaining CNS homeostasis, but can become compromised during ageing, where dietary habits shift and nutrient deficiencies are common.
Changes in dietary habits can have profound consequences on microglial function specifically. For example, insulin resistance, which is commonly observed in older adults (Kolb et al. 2023), is hypothesised to drive microglia towards a pro-inflammatory state by the induction of hyperinsulinemia (Doust et al. 2022). The resulting increase in cytokine production may further inhibit insulin signalling in the brain. Interestingly, however, a previous study showed that microglia in the hippocampus of 22-mo Wistar rats did not mount an immune response following intracerebroventricular insulin administration for 5 days (Haas et al. 2020). Comparatively, increased numbers of IBA1 and CD68 positive microglia, as well as increased expression of COX-2 and IL-1β, were observed in 3-mo rats in response to the same treatment protocol within that study. This suggests that during ageing, the responsiveness of microglia to insulin signalling is diminished.
Interestingly, insulin/IGF-1 signalling has been linked with longevity in multiple species, including C. elegans, Drosophila melanogaster and laboratory mouse strains (see review: (van Heemst 2010)). In aged APPSWE/PS1ΔE9 mice (24-mo), a model of AD, IGF-1 mRNA and TNF mRNA levels were both increased in the hippocampus compared to age-matched WT mice. The authors theorised that this was due to an increase in IGF-1 mRNA-expressing microglia in the subgranular zone of the dentate gyrus (Myhre et al. 2019), but this was somewhat speculative. This would suggest that increased IGF-1 signalling is a key component of microglial pro-inflammatory responses in AD during advanced ageing, but comparison to microglia in young animals is needed to confirm this conclusion.
Another signalling pathway, the mTOR pathway, acts as a cellular energy sensor, responding to amino acids, glucose, oxygen and growth factors, including IGF-1 (Garza-Lombó et al. 2018). Dysregulated mTOR signalling with age, perhaps stemming from persistently elevated levels of IGF-1 or other growth factors, can lead to overactive nutrient sensing and contribute to the chronic, low-grade inflammation observed during ageing (reviewed: (Stallone et al. 2019)). This was observed in C57BL/6J mice, where mTOR activation was significantly increased at 23-mo of age compared to 6-mo of age in microglia isolated from female mice, as measured using phospho-flow cytometry (Keane et al. 2021). Studies in model organisms, such as Saccharomyces cerevisiae and C. elegans, have also supported the link between mTOR activity and ageing, where reduced mTOR activity has been shown to extend the lifespan (Fabrizio et al. 2001; Vellai et al. 2003). Despite this, studies investigating alterations in the mTOR pathway in aged microglia in these model organisms remain sparce.
The AMPK pathway serves as a central regulator of energy homeostasis, maintaining balance by counteracting mTOR activity (Garza-Lombó et al. 2018). AMPK responds to energy depletion (low ATP) by inhibiting anabolic processes and activating catabolic pathways (Garza-Lombó et al. 2018). These functions highlight the role of AMPK as a critical controller of ageing processes by modulating metabolism, oxidative stress and inflammation (Salminen and Kaarniranta 2012). Activity of the AMPK pathway has been shown to increase with age in the brain, with phosphorylation of AMPK in young male C57BL/6 mice (2- to 3-mo) lower than that seen in aged 16- to 18-mo mice, suggesting more AMPK activity in the brain during healthy ageing (Liu et al. 2012). However, in this same study, activation of AMPK following reversible middle cerebral artery occlusion was decreased in aged mice compared to young mice (Liu et al. 2012), indicating altered responsiveness of AMPK during ageing, potentially contributing to worsened outcomes following injury or pathology. Interestingly, the anti-inflammatory effects of AMPK activation in microglia have been previously demonstrated. Specifically, activation of the AMPK pathway by administration of 80µM of ENERGI-F704, a small molecule drug which causes phosphorylation and activation of AMPK, reduced the pro-Il-6 and TNF response of BV2 cells following LPS stimulation (Chen et al. 2014). This suggests an ameliorating effect, and potentially beneficial role of this pathway, in reducing microglial inflammation (Chen et al. 2014). Despite this, however, it is not yet known if and how levels of AMPK pathway activation are altered with age in microglia.
In order to understand the full effect of nutrient pathways on microglia in ageing, future research should seek to characterise changes in the responsiveness of microglia to nutrients and what downstream pathways are impacted by these changes. The apparent impact of insulin resistance on microglial responsiveness, coupled with evidence of altered IGF-1 signalling in ageing and AD, highlights a potential area of investigation for future studies. Profiling microglial expression of components of the insulin/IGF-1 pathway during ageing, potentially using single-cell transcriptomics, will increase our understanding of what mechanisms may drive these functional changes. Additionally, investigating other pathways in these cells, such as AMPK and its major components (e.g. mTOR and LKB1), will further elucidate the impact of nutrient sensing on microglial functions with ageing.
Mitochondrial Dysfunction
Mitochondria are responsible for regulating a host of different cellular processes but are mainly involved in energy production and oxidative balance. They are a critical component of the free radical theory of ageing, due to their high production of and vulnerability to ROS. Additionally, damaged or dysfunctional mitochondria are involved in inflammatory processes, particularly by activation of the DNA-driven immune response by mtDNA damage or by free mtDNA released from damaged mitochondria (Fig. 3b). With age, mitochondrial function declines, leading to increased production of ROS and ROS-induced inflammatory responses (Amorim et al. 2022). Additionally, deficiencies in nutrient availability, as outlined in the above section, can have profound effects on mitochondrial metabolism and energy production. For example, Minhas et al. (2021) observed that altered glucose handling by PGE2-EP2 signalling in aged microglia shifted glucose towards glycogen storage rather than mitochondrial use, resulting in decreased mitochondrial respiration. Additionally, vitamin deficiencies inhibit the metabolic functioning of mitochondria and contribute to observed mitochondrial decay during ageing (Ames et al. 2005). Due to the high energy requirement for continuous immune functioning, microglia heavily rely on mitochondrial energy for maintaining function (Fairley et al. 2021) and are greatly affected by mitochondrial impairment and dysfunction (Ryu et al. 2003).
Mitochondrial transcription factor A (TFAM) is a promotor of mtDNA transcription and is critical in maintaining correct mtDNA transcription and regulating degradation of damaged mtDNA (Xu et al. 2019). A prior study investigated changes in this with age through overexpression of TFAM in male C57BL/6 mice at 2-mo and 24-mo. In this study, oxidative stress markers 8-oxo-deoxyguanosin (8-oxo-dG) and hydroxy-2-noneal (HNE) increased with age in microglia from WT mice, whereas no changes were reported in microglia from TFAM-overexpressing mice (Hayashi et al. 2008). This suggests that TFAM may play a protective role in maintaining mitochondrial and oxidative homeostasis in ageing microglia. Additionally, this study assessed whole brain malondialdehyde (MDA) levels, a marker of lipid peroxidation, and mitochondrial complex 1 activity (Hayashi et al. 2008). Aged WT mice demonstrated an increase in MDA levels, coupled with a decrease in complex 1 activity with age, suggesting age-related mitochondrial dysfunction and heightened oxidative stress. In contrast, TFAM-overexpressing mice showed no changes with age in either measure. While not specifically assessed in microglia, the increased 8-oxo-dG and HNE in aged microglia in these tissues suggest that microglial TFAM dysregulation may contribute to a broader oxidative stress observed in the ageing brain (Hayashi et al. 2008).
As well as damaged mtDNA accumulation with age, mtDNA can also move into the cytosol, caused during ageing by altered mitophagy, leading to the escape of both damaged and undamaged mtDNA from mitochondria and stimulating the DNA-driven immune response (Pérez-Treviño et al. 2020). In line with this, microglia from 20-mo C57BL/6J mice have shown an increased amount of cytosolic mtDNA compared to microglia from 2- to 3-mo mice (Gulen et al. 2023). This stimulated the cGAS-STING pathway in these animals, leading to the acquisition of a pro-inflammatory phenotype (Gulen et al. 2023). Taken together, these studies suggest that, with age, a loss of TFAM control of mtDNA leads to dysregulated ROS production, with dysfunctional mitophagy further contributing to the accumulation of mtDNA within the cytosol of microglia. Combined, these mechanisms increase the pro-inflammatory nature of aged microglia and increased ROS production likely causes further damage to mitochondria, feeding the cycle of continued inflammation and oxidative stress which is characteristic of both ageing and NDs. To date, however, limited studies have sought to characterise age-related changes in specific aspects of mitochondrial biology in microglia, with this an important avenue for future research.
Cellular Senescence
Cellular senescence is the term used to refer to cells which have undergone an irreversible cell cycle arrest (Gorgoulis et al. 2019) and is commonly associated with ageing, as senescent cells are known to accumulate in aged tissues (López-Otín et al. 2023). There are many features of senescence (reviewed: (González-Gualda et al. 2021)), but the characteristic cell cycle arrest is the most defining feature. Indeed, microglia demonstrate changes in the senescence-associated markers of cell cycle arrest, p16INK4A and p21Cip1/Waf1, with advancing age. In 18-mo male C57BL/6 mice, expression of p16INK4A, but not p21Cip1/Waf1, was observed in microglia (Ritzel et al. 2019). This study also reported an increase in DNA damage (H2A.X stain) and lipofuscin accumulation in microglia in these mice. Lipofuscin progressively accumulates within aged cells and is considered to be involved in a number of age-related pathologies (Jung et al. 2007). Indeed, an increase in lipofuscin accumulation has been demonstrated within microglia in 24-mo C57BL/6J mice of both sexes, although this was low in comparison to the levels that accumulated within neurons (Stillman et al. 2023). This indicates that while microglia do exhibit age-associated buildup of lipofuscin, they may possess more efficient clearance mechanisms than neurons; however, this still appears to become impaired with age.
Lipofuscin accumulation is often investigated in conjunction with senescence-associated β-Galactosidase (SA-β-Gal), as the presence of both has been shown to be related to lysosomal dysfunction with age (Georgakopoulou et al. 2013). In microglia, SA-β-Gal has been utilised to identify senescent microglia for further analysis. For example, to assess the proximity of senescent microglia to Aβ plaques, microglia in 8-mo 5XFAD mice (a model of AD pathology) and C57BL/6J WT mice were compared. Sagittal brain sections were stained for SA-β-Gal before undergoing 3,3’-diaminobenzidine (DAB) staining with IBA1 for microglial detection. Co-localisation of DAB (IBA1) with SA-β-Gal pigment demonstrated that the microglia surrounding pathological plaques were SA-β-Gal positive (Shin et al. 2024). SA-β-Gal staining has also been used to identify senescent microglia in vitro, identifying senescence in BV2 microglia-like cells following three or six treatments with the endotoxin LPS (Yu et al. 2012). Further, these BV2 cells demonstrated the acquisition of another well described marker of senescence, senescence-associated heterochromatin foci (SAHF). SAHF are regions of highly condensed chromatin formed in response to stresses, such as DNA damage and telomere dysfunction (Aird and Zhang 2013). These formations are seen in LPS-induced ageing in BV2 cells (Yu et al. 2012; Borgonetti and Galeotti 2023), but have not been assessed in in vivo studies looking at microglia change with advancing age.
Another marker of cellular senescence previously assessed in microglia is the acquisition of the senescence-associated secretory phenotype (SASP), characterised by an increase in the production and release of pro-inflammatory cytokines and chemokines (Rim et al. 2024). Microglia are known for their increased secretion of pro-inflammatory factors in NDs and indeed take on a similar phenotype during normal ageing. Microglia in 18-mo transgenic p7.2fms-EGFP mice on a C57BL6/6XCBA background have shown an increased release of pro- (IL-6, TNF, IL-1β) and anti- (IL-10) inflammatory cytokines compared to mice at 2-mo of age (Sierra et al. 2007). Additionally, microglia from the spinal cords of C57BL/6J male mice at 22-mo of age show an increase in inflammatory cytokine release (TNF, IL-1β), coupled with increased ROS-induced oxidative stress and microglial phagocytic ability (Ritzel et al. 2015). However, microglia are inherently inflammatory cells and many of the secreted factors associated with the SASP overlap with those secreted by microglia during the immune response (Ng et al. 2023). Additionally, as these cells are often studied in the context of diseases with an inflammatory component, using the SASP as a single marker of senescence in these cells is discouraged.
Across the literature, a stringent panel of markers for cellular senescence is not uniformly applied and many studies rely heavily on only one or two markers to distinguish senescent cells. This is problematic, as markers such as SA-β-Gal, the SASP and cell cycle inhibitors (p16 and p21) are not always specific to ageing, and often appear upregulated in other conditions (Ng et al. 2023). This could lead to the mislabelling of populations of cells as senescent. Issues with the definition of senescence as it relates to microglia have been previously discussed by (Ng et al. 2023), with a rigorous approach using multiple markers encouraged to define populations of senescent microglia. This integrated approach has been utilised by Hartmann et al. (2023) when establishing a measure of age in cultured fibroblasts. This approach examined telomere length, cell cycle inhibitors, SA-β-Gal and secretion of cytokines to create a scored age for cells in culture marked against actual chronological age of the donor (Hartmann et al. 2023). Future studies should endeavour to use a similar approach when defining senescence in microglia.
Stem Cell Exhaustion
As a hallmark of ageing, stem cell exhaustion describes the decline in regenerative potential of adult stem cells. This is defined by López-Otín and colleagues (López-Otín et al. 2013, 2023) as the reduction of cell cycle activity of hematopoietic stem cells, contributing to the lack of capacity for tissue regeneration with age. Microglia originate from yolk sac progenitor cells (Cuadros et al. 1993; Alliot et al. 1999) and the microglial population self-renews regularly throughout the lifespan (Askew et al. 2017; Manjally and Tay 2022; Réu et al. 2017; Ajami et al. 2007). The mechanisms of microglial renewal in the adult brain are debated (Huang et al. 2018; Elmore et al. 2014; Bruttger et al. 2015). Elmore et al. (2014) demonstrated rapid repopulation of microglia in the brains of male 18-mo WT C57BL/6 mice following depletion of the microglial population by administration of 290mg/kg of PLX3397 for 28 days. Only 3 days following cessation of treatments, IBA1 positive microglia began to emerge and by 14 days microglia with ramified morphologies were present throughout the brain in equal amounts to that of age-matched untreated mice (Elmore et al. 2014). The newly formed microglia at 3 days expressed Ki67, indicating proliferation, and were positive for the neuroectodermal development marker nestin. Notably, subsequent analysis revealed that microglial repopulation was not due to infiltration of circulating monocytes, with fate mapping using BrdU labelling of proliferative non-microglial cells revealing that these cells went on to become microglia. These cells were also positive for nestin, suggesting that they may represent a population of microglial progenitor cells (Elmore et al. 2014).
Nonetheless, evidence suggests that this method of microglial renewal occurs mainly following mass depletion of the microglial population, with previous studies in mice demonstrating that microglia are generally long lived, with half the microglial population lasting the entire lifespan of an animal (Füger et al. 2017). In support of this, a study using 3-mo and 22-mo male C57BL/6 mice demonstrated that depleting microglia by PLX3397 administration for 7 days and allowing rapid repopulation from these hypothesised progenitor cells led to a microglial population in 22-mo ‘repopulated’ mice which resembled that seen in 3-mo controls (Elmore et al. 2018). This suggests a different ageing profile compared to microglia which have undergone slow turnover throughout the lifespan (Réu et al. 2017). Similarly, a study in human post-mortem tissue from young (20- to 35-years-old) and old (58- to 79-years-old) adults showed no changes in the number of microglia positive for the proliferative marker Ki67 between groups. This supports the hypothesis of slow turnover across the lifespan, rather than a rapid regeneration of microglia from a progenitor population (Askew et al. 2017).
Across the lifespan, the microglial population may be depleted by insults such as infections, injuries or neurodegenerative pathology. Indeed, the presence of amyloid pathology in a triple-transgenic CD11b-CreERT2;R26-tdTomato;APP/PS1 mouse model of AD leads to steady loss of microglia and compensatory proliferation (Füger et al. 2017). Interestingly, utilising a cranial window for visualisation of fluorescently labelled microglia, newly appearing cells were observed to be preceded by increased cell body volume of the presumed origin cell, suggesting proliferation from existing microglia, rather than a progenitor population (Füger et al. 2017). Overall, the literature as a whole supports that microglial renewal occurs across the lifespan through proliferation from mature cells, with the potential for regeneration from apparent progenitor cells following significant depletions in the microglial population (Hughes and Bergles 2014; Elmore et al. 2014, 2018). Whether this progenitor population exists in human brains, however, has not yet been fully established, and key questions remain around if the regenerative capacity of such cells is altered with age in mice.
Altered Intercellular Signalling
Changes in intercellular communication are a hallmark of ageing, with alterations in cell signalling pathways potentially leading to an increased pro-inflammatory environment and loss of homeostatic processes (López-Otín et al. 2023). During ageing, there is dysregulation in several mechanisms of intercellular communication, including changes in pro-inflammatory signalling pathways. For microglia in particular, intrinsic changes in pathways such as the NF-κB/NLRP3, Fyn kinase and MAPK pathways lead to altered release of key signalling molecules, including cytokines. The NLRP3 inflammasome is expressed by both microglia (Cho et al. 2014) and astrocytes (Lu et al. 2014) in the CNS, but is primarily associated with microglia (Chiarini et al. 2023). Under physiological conditions, NLRP3 is expressed at low levels. However, during the inflammatory response, microglia have increased activation of the NLRP3 inflammasome complex (Fig. 3a) (Song et al. 2017). This leads to the release of IL-1β, in turn activating neighbouring cells, including other microglia, which further perpetuate the inflammatory response (Kamo et al. 2013). With age, the NLRP3 inflammasome becomes overactive, indicating a potential contribution to the chronic pro-inflammatory state seen in microglia with advancing age (Liang et al. 2024). In line with this, in 18-mo old male BALB/c mice, multiple NLRP3 pathway proteins, including ASC, caspase-1, IL-18 and NLRC4, were increased in the hippocampus and cortex compared to 3-mo mice (Mejias et al. 2018). Additionally, there was an increase in pyroptosis in these regions, indicating a role for increased NLRP3 signalling in pyroptotic cell death. Of note, microglial activation measured by assessment of microglial morphology in the dentate gyrus has been shown to decrease in 23-mo NLRP3−/− mice compared to age-matched WT controls (Youm et al. 2013). Additionally, this measure decreased relative to 2-mo NLRP3−/− mice, suggesting that NLRP3 activation may play a larger role in microglial dynamics with advancing age (Youm et al. 2013).
NF-κB signalling is involved in activation of the NLRP3 inflammasome by causing the transcription of pro-IL-1β, pro-caspase-1 and NLRP3 (Liu et al. 2017) (Fig. 3a). Age-related increases in NF-κB signalling and nuclear translocation lead to an increase in both NLRP3 inflammasome formation and secretion of pro-inflammatory cytokines (Liu et al. 2017). Figure 3a.4 shows the mechanisms by which NF-κB is translocated to the nucleus. IκB is normally bound to NF-κB in the cytoplasm and prevents NF-κB activation. IκB phosphorylation and degradation by the proteasome exposes NF-κB localisation subunits, including P50 and P65, leading to translocation of NF-κB to the nucleus, where it then triggers various gene expression (Liu et al. 2017). Alterations in these NF-κB subunits, specifically ablation of the p50 subunit in C57BL/6J mice (p50−/−), has been shown to increase the inflammatory response of microglia to LPS administration (Taetzsch et al. 2019). This response was exacerbated in p50−/− mice at 16–18-mo compared to both p50+/+ age-matched controls and young p50−/− mice (3-weeks-old), suggesting that NF-κB disruption may be particularly detrimental in the context of ageing (Taetzsch et al. 2019).
Fyn kinase and MAPK pathways are upstream regulators of both NLRP3 and NF-κB, with increased Fyn and p38 MAPK signalling promoting pro-inflammatory signals within microglia (Bachstetter et al. 2011; Panicker et al. 2015). Previous literature indicates that activation of both pathways may be impacted by ageing, although in different ways. One study investigated Fyn kinase signalling in 12-mo C57BL/6J mice following traumatic stress induced by dorsomyotomy and exploratory laparotomy (Zhao et al. 2012). This study demonstrated reductions in Fyn signalling in both pre- and post-synaptic density protein fractions in the frontal cortex of aged mice compared to 2-mo mice three days following surgical procedure (Zhao et al. 2012). This suggests that Fyn signalling following traumatic stress is further altered in an aged environment. Conversely, in male C57BL/6J mice at 20-mo of age, phosphorylated-p38 MAPK protein levels in the brain were increased compared to 2-mo mice (Li et al. 2011). While neither Fyn nor MAPK pathways have yet been studied specifically in microglia during ageing, given these are key immune pathways in microglia, it is reasonable to hypothesise that dysregulation of these pathways may be responsible for the upregulation in pro-inflammatory signalling seen in microglia with increased age. Understanding these pathways in microglia is critical for identifying microglial signalling pathways which contribute to dysfunction with ageing.
Chronic Inflammation
Evidence for chronic inflammation in the aged brain has previously been extensively reviewed (Andronie-Cioara et al. 2023; Jin et al. 2022; Moyse et al. 2022). Given their critical role in regulating the inflammatory response, microglia are likely a major driver of this response. Notably, microglia in aged mice sustain the inflammatory response for longer following brain injury than their young counterparts. For example, following a controlled cortical impact (CCI), microglia in the hippocampus of male 24-mo C57BL/6 mice showed increased relative expression of Cd11b and IBA1 (markers of the microglial immune response) following injury, which remained heightened at 14 days post-injury (Sandhir et al. 2008). Comparatively, these markers in adult mice at 5- to 6-mo were expressed at lower levels at all timepoints and returned to baseline expression levels by 14 days post-injury (Sandhir et al. 2008). Additionally, a separate study has shown that aged C57BL/6 male mice display a propensity to acquire a pro-inflammatory phenotype at 24-months of age following CCI. These mice demonstrated a decreased ability to mount an anti-inflammatory response compared to 3-mo mice (Kumar et al. 2013). Similar results have also been seen in stroke models, where, following intracerebral haemorrhage induced by collagenase injection to the striatum, male Sprague–Dawley rats at 22-mo showed a slower microglial response and prolonged microglial immune activation around the hematoma compared to 3- to 4-mo rats following injury (Wasserman et al. 2008). This study additionally demonstrated morphological differences in microglia of naïve animals with age, with microglia from 24-mo rats demonstrating shorter and thicker processes, characteristic of bushy morphology, compared to the ramified morphology in 3- to 4-mo rats.
The population of microglia is also altered with age; however, there appear to be clear sex differences in these changes. For example, in the hippocampus of male C57BL/6 mice, microglial number was stable throughout the lifespan, with no changes observed from young (4- to 5-mo) to middle-aged (13- to 14-mo) to aged (27- to 28-mo) mice in the dentate gyrus, CA1 or Hilus (Long et al. 1998). Additionally, no changes in microglial density in these regions were observed, nor were there differences in the number of astrocytes present, indicating that glial numbers are stable with age in males, at least within hippocampal regions. Conversely, in a separate study by the same authors, in the CA1 region of female C57BL/6 mice, microglial numbers increased steadily across young (3- to 4-mo), middle-aged (13- to 14-mo) and aged (20- to 24-mo) cohorts (Mouton et al. 2002). Importantly, at each timepoint, the microglial counts in female mice were significantly higher than CA1 counts in the equivalent male cohort from the previous study. These differences are also seen in human post-mortem brains, where the number of microglia in the female neocortex (age range: 18–93 years) has been shown to increase with age, despite no such changes observed in these regions in males (age range: 19–87 years) (Pelvig et al. 2008). Taken together, these results indicate an effect of biological sex on microglial dynamics with age and thus highlight the importance of including sex-balanced cohorts in ageing research.
Beyond population dynamics, the phenotype of microglia is also altered with increased age. In line with this, the number of microglia initiating an immune response, as measured by MHCII positivity, increased with age in both mouse and non-human primate tissue. In male C57BL/6 mice, the number of Cd11b/MHCII positive microglia at 22- to 24-mo was increased compared to in 3- to 5-mo mice (Garner et al. 2018). Similarly, when investigated in macaque brains, microglial MHCII expression increased at 10- to 18-years-old compared to at 2- to 5-years-old (Sheffield and Berman 1998), indicating that these changes are not only seen in rodent brains but also in the brains of higher order mammals. In humans, microglial immune activation with age has been studied by utilising the positron emission tomography (PET) ligand (R)-[11c]PK111955, which specifically binds to inflammatory microglia expressing the 18 kDa translocator protein (TSPO) (Shah et al. 1994). One study using this ligand in a cohort of 33 subjects (22M:11F) aged 19- to 79-years-old found an association between increasing age and enhanced ligand binding throughout the brain. This data suggests that activation of microglial inflammatory pathways is associated with increasing age in healthy living subjects (Schuitemaker et al. 2012). Additionally, in the HuMi_Aged dataset, interferon signalling pathways were enriched and upregulated compared to microglia from a middle-aged dataset (Olah et al. 2018; Zhang et al. 2016). Taken together, these data support a pattern of increased inflammatory microglia with age, regardless of species and sex. This altered microglial phenotype may contribute to the ongoing chronic inflammation seen in age-related NDs.
Age-related intrinsic changes within microglia are thought to drive the chronic inflammatory environment observed in the aged brain. Nuclear and mtDNA damage is thought to play a key role in this through the DNA-driven immune response (Pérez-Treviño et al. 2020). In line with this, in 20-mo C57BL/6J mice, STING activity was shown to be upregulated in microglia, suggesting heightened cytosolic DNA sensing in these cells (Gulen et al. 2023). Indeed, the increased stimulation of the cGAS-STING pathway, described in the mitochondrial dysfunction section above, is thought to drive the shift towards a pro-inflammatory phenotype with age, contributing to ongoing inflammation in microglia (Gulen et al. 2023). These studies support the idea that microglia are a driver of the chronic inflammation observed with advancing age, which potentially underlies and likely exacerbates ND pathology.
Dysbiosis
Dysbiosis as a hallmark of ageing refers to the imbalance of bacteria in the gut (López-Otín et al. 2023). A 2022 review identified two groups of taxa associated with ageing (Ghosh et al. 2022). The first group consisted of mainly pro-inflammatory taxa and was associated with unhealthy ageing, defined by shortened lifespans, whereas the second group consisted of beneficial taxa and was associated with healthy ageing. Notably, levels of beneficial taxa were decreased in unhealthy ageing (Ghosh et al. 2022). This suggests that longevity and healthy ageing may be associated with higher levels of beneficial taxa in the gut, while pro-inflammatory taxa may lead to an accelerated ageing process. Links between changes in the composition of the gut microbiome and CNS conditions are becoming increasingly apparent (Abdel-Haq et al. 2019), and the gut–brain axis is an important pathway of regulation of glial function (Loh et al. 2024).
Microglia appear to be the most vulnerable CNS cell type to changes in the gut microbiome (Huang et al. 2023). For example, in 2-mo Kun Ming mice, microglial gene expression in response to altered gut microbiome was altered more than in any other cell type (Huang et al. 2023). Specifically, differential expression of inflammasome genes, genes associated with mitochondrial dysfunction and oxidative phosphorylation genes was noted in both the prefrontal cortex and hippocampus (Huang et al. 2023). Given this, it is reasonable to hypothesis that microglial function and maturation may be influenced by age-related changes in the gut microbiota. In line with this, indirect modulation of microglia via interactions of pro-inflammatory bacteria with peripheral immune cells has been demonstrated in aged mice. Peripheral administration of LPS in 20-mo male BALB/c mice induced a hyperactive microglial response, as measured by increased mRNA expression of IL-1β, IL-10 and TLR2 in isolated microglia (Henry et al. 2009). Given that LPS is often released as a signalling byproduct by microbes in the gut (Mohr et al. 2022) and that increased gut permeability in older age allows LPS to cross into blood circulation (Thevaranjan et al. 2017), this study may implicate age-related pro-inflammatory changes in the gut as a potential moderator of microglial response. To date, however, there are limited studies on the impact of specific age-related microbial alterations in the gut-brain axis on microglia. Nevertheless, the evidence that microglia can be disproportionately affected by changes in the gut suggests that this could be a potential area for intervention to reduce aberrant age-related changes in microglia.
Beyond Immune Activation: Unresolved Questions and Future Directions for Microglial Ageing
Overall, the dystrophic morphology of aged microglia is accompanied by the acquisition of a senescent state with increased pro-inflammatory signalling. The aged phenotype of microglia as related to the twelve hallmarks of ageing is outlined in Fig. 4. Microglia display a decreased capacity for homeostatic functions, including phagocytosis, and an increased production and release of pro-inflammatory factors, resulting in increased pro-inflammatory signalling. In general, this decreased capacity of microglia to maintain homeostatic immune functions may contribute to the vulnerability of the brain to age-related diseases, including the development of NDs (Koellhoffer et al. 2017). However, these changes may not be the sole cause of NDs, and indeed, age-related changes in neurons also appear to be major drivers of disease pathophysiology (Höhn et al. 2020).
Fig. 4.
Features of adult (left) and aged (right) microglia. Left: Normal young adult microglia are highly motile with a ramified morphology and can perform homeostatic and immune functions without inducing hostile environmental conditions. Right: Aged microglia display enlarged dystrophic morphology and present with many of the classic hallmarks of ageing
While the studies discussed above highlight significant features of aged microglia, this is still a relatively understudied area. Research on ageing in peripheral macrophages has garnered more attention, with the hallmarks of ageing in macrophages reviewed recently (Guimarães et al. 2021). However, microglia have not received equivalent focus, a notable gap which is likely due to the challenges of accurately replicating aged CNS cells in research models (Carr et al. 2024). This is also reflective of the fact that ageing more generally is often not considered in pre-clinical modelling studies of ND, a critical consideration to improve translational potential (Carr et al. 2024). Tables 2 and 3 summarise studies conducted to date which have investigated each hallmark of ageing in microglia in either experimental models or human tissue, respectively.
Table 2.
Evidence of the hallmarks of ageing in microglia from laboratory models
| Hallmark | Features seen in aged microglia | ||||
|---|---|---|---|---|---|
| Model | Sex | Ages compared Age of observed change is underlined |
Changes observed | Ref | |
| Genomic instability | C57BL/6 mice | Not specified | 24 mo compared to 2 mo |
↑accumulation of DNA damage measured by γ-H2A.X staining ↑ percentage of microglia positive for pSTING and CD11b |
(Arvanitaki et al. 2024) |
| Ercc1Δ/−mice on C57BL6J/FVB background | Male and female | 16wo compared to age/sex matched genetic controls |
Hypertrophy of processes and ↑ cell body size ↑proliferation, ↑phagocytosis, ↑ROS production |
(Raj et al. 2014) | |
| Ercc1Δ/−mice induced by tamoxifen administration at 6- to 8-wo on C57BL6J/FVB background | Male and female | 2-, 6-, and 12-months following tamoxifen administration (approx. 4-, 6-, and 14-mo) compared to age/sex matched genetic controls | ↓microglial number at 6- and 12-month timepoints | (Zhang et al. 2021) | |
| Telomere attrition | Microglia isolated from cortex of neonatal rats | Not specified | Aged by replicative senescence over 32 days in culture |
↓telomere length measured by flow-FISH Steady decline and then rapid increase in telomerase activity during culture |
(Flanary and Streit 2004) |
| Microglial isolated from Fisher-344 rats | Male | 30-mo compared to 3-mo |
↓telomere length measured by flow-FISH ↓telomerase activity |
(Flanary et al. 2007) | |
| Terc knockout mice with AD pathology: G3Terc−/−APP23+ mice | Not specified | 12-mo compared to age-matched genetic controls |
↓Aβ pathology in frontal cortex Improved spatial memory No microglial immune activation near Aβ plaques |
(Rolyan et al. 2011) | |
| Terc knockout mice with PD pathology: αSYNtg/tgG3Terc−/− mice | Not specified | 75-wo compared to age-matched genetic controls | ↓microglial IL-1β and MHCII in the brainstem | (Scheffold et al. 2016) | |
| Epigenetic changes | Microglia isolated from C57BL/6J mice following LPS exposure | Not specified | 24- to 26-mo compared to 4- to 6-mo | ↓ methylation of IL-1β promoter DNA leading to increased IL-1β production | (Matt et al. 2016) |
| C57BL/6J mice | Male | 16- to 18-mo compared to 2- to 3-mo |
↑ iNOS, IL-6 and TNF ↑ H3K27me3 in the midbrain ↓ JMJD3 |
(Tang et al. 2014) | |
| C57BL/6J mice | Male and female | 24-mo compared to 3- and 14-mo |
↑ chromatin accessibility of phagocytosis, IFN signalling, ROS production and antigen presentation genes ↓ chromatin accessibility of chemokine suppression and iron transportation genes |
(Li et al. 2023a) | |
| Loss of proteostasis | Microglia isolated from C57BL/6N mice | Not specified | 20- to 24-mo compared to 3- to 5-mo | ↓ proteasomal subunits PSMB4, PSMA6, PSMC1 | (Flowers et al. 2017) |
| APP/PS1 mice lacking the LMP7 immunoproteasome component | Not specified | 8-mo compared to age-matched genetic controls |
↓ microglial immunoreactivity No change in Aβ pathology |
(Wagner et al. 2017) | |
| Deletion of E3 ubiquitin ligase COP1 in C57BL/6N mice by tamoxifen administration | Male and female | 22-mo compared to young WT mice |
↑ microglial IFN signalling gene expression ↑APOE expression |
(Ndoja et al. 2020) | |
| C57BL/6 mice | Male and female |
Sex differences at 18-mo compared to 3-mo Both sexes differences at 22-mo compared to 3-mo |
Female mice: ↓ microglial phagocytic action on Aβ42 Male mice: ↓ proteolytic gene expression Both sexes: ↓ expression of phagocytic receptor TREM2 |
(Thomas et al. 2022) | |
| Disabled macroautophagy | Microglia isolated from mice with deletion of WD domain of Atg16L on a mixed 129/C57BL/6 background | Male and female | 24-mo compared to age-matched genetic controls | ↓ uptake of Aβ | (Heckmann et al. 2020) |
| Bone marrow derived macrophages isolated from C56BL/6 mice** | Male | 23-mo compared to 6- to 8-wo |
↓expression of autophagic genes ATF7 and ATG-6 Impaired autophagic flux |
(Stranks et al. 2015) | |
| Deregulated nutrient sensing | Microglia isolated from C57BL/6N mice | Not specified | 20- to 24-mo compared to 3- to 5-mo |
↓ mTORC2-dependent AKT D473 phosphorylation ↑IL-6, IL-1β, TNFα at baseline and in response to TNFα |
(Flowers et al. 2017) |
| Wistar rats treated with insulin for 5 days | Male | 22-mo compared to 3-mo | ↓IBA1 and CD68 positive microglia in the hippocampus and no induction of COX-2/IL-1β immune response | (Haas et al. 2020) | |
| Microglia isolated from C57BL/6J mice | Female | 23-mo | mTOR dysregulation | (Keane et al. 2021) | |
| APPSWE/PS1ΔE9 mice | Female | 24-mo compared to age-matched genetic controls |
↑IGF-1 and TNF mRNA in hippocampus ↑IGF-1 mRNA-expressing microglia in subgranular zone of dentate gyrus |
(Myhre et al. 2019) | |
| Mitochondrial dysfunction | WT C57BL/6 mice and C57BL/6 mice overexpressing TFAM | Male |
24-mo WT compared to 2-mo WT 24-mo TFAM compared to 2-mo WT |
24-mo WT: ↑lipid peroxidation 24-mo TFAM: ↓ lipid peroxidation |
(Hayashi et al. 2008) |
| Microglia isolated from C57BL/6 mice | Not specified | 20- to 24-mo compared to 2- to 4-mo |
↑PGE2-EP2 signalling leading to enhanced glycogen storage and ↓mitochondrial respiration Inhibition of EP2 signalling restored metabolic function *changes also relevant to dysregulated nutrient sensing due to altered glucose handling via PGE2-EP2 signalling |
(Minhas et al. 2021) | |
| Microglia isolated from C57BL/6 mice | Not specified | 20-mo compared to 8- to 12-wo | ↑cytosolic mtDNA | (Gulen et al. 2023) | |
| Cellular senescence | C57BL/6 mice | Male | 18-mo compared to 3-mo |
↑ expression of p16INK4A but no change in p21Cip1/Waf1 Accumulated lipofuscin ↑ DNA damage measured by γ-H2A.X staining |
(Ritzel et al. 2019) |
| C57BL/6 mice | Male and Female | 24-mo | Accumulated lipofuscin | (Stillman et al. 2023) | |
| 5XFAD mice and C57BL/6J WT mice | Male and female | 8-mo compared to 4-mo | ↑presence of SA-β-Gal+ microglia proximal to Aβ plaques | (Shin et al. 2024) | |
| BV2 microglia-like cells | N/A | Cells aged by repetitive (6x) LPS administration | ↑SA-β-Gal and ↑SAHF formation | (Yu et al. 2012) | |
| Transgenic p7.2fms-EGFP mice on a C57BL6/6XCBA background | Male and female | 18-mo compared to 2-mo | ↑pro- ((IL-6, TNFα, IL-1β) and anti- (IL-10) inflammatory cytokines | (Sierra et al. 2007) | |
| Microglia isolated from spinal cords of C57BL/6J mice | Male | 22-mo compared to 3-mo |
↑TNFα, IL-1β release ↑ ROS-induced oxidative stress ↑microglia phagocytic ability |
(Ritzel et al. 2015) | |
| Stem cell exhaustion | Cd11b-CreERT2 transgenic mice on a C57BL/6J x C3Hej background | Male and female | 15-mo compared to 4-mo | Microglial cells are long lived and stable across life | (Füger et al. 2017) |
| C57BL/6 mice | Not specified | 18- to 20-mo compared to 4- to 6-mo | No changes in microglial number in hippocampus or cortex but ↑ in the thalamus | (Askew et al. 2017) | |
| Altered intercellular communication | BALB/c mice | Male | 18-mo compared to 3-mo |
↑ NLRP3 pathway proteins in hippocampus and cortex ↑pyroptosis in hippocampus and cortex |
(Mejias et al. 2018) |
| NLRP3−/− mice | Male and female | 23-mo compared to 2-mo and to aged-matched genetic controls |
Loss of NLRP3 caused ↓ in microglial immune activation in dentate gyrus ↓in IL-1β and TNF mRNA expression |
(Youm et al. 2013) | |
| C57BL/6J mice deficient in the p50 NF-κB subunit | Not specified | 16- to 18-mo compared to 3-wo and age-matched genetic controls | ↑inflammatory response of microglia to LPS administration | (Taetzsch et al. 2019) | |
| C57BL/6J mice | Not specified | 12-mo compared to 2-mo | ↓Fyn in pre- and post-synaptic protein fractions from the frontal cortex following traumatic stress | (Zhao et al. 2012) | |
| C57BL/6J mice | Male | 20-mo compared to 2-mo | ↑phospho-p38 MAPK in brain tissue | (Li et al. 2011) | |
| Chronic inflammation | C57BL/6 mice following a moderate controlled cortical impact (CCI) | Male | 21- to 24-mo compared to 5- to 6-mo |
↑ inflammatory response in the hippocampus following injury ↑ time to resolve response |
(Sandhir et al. 2008) |
| C57BL/6 mice following CCI | Male | 24-mo compared to 3-mo | ↑pro- and ↓ anti-inflammatory markers of microglia | (Kumar et al. 2013) | |
| Sprague–Dawley rats following collagenase-induced intracerebral haemorrhage | Male | 22-mo compared to 3- to 4-mo |
Slower microglial response to injury Prolonged immune activation around hematoma |
(Wasserman et al. 2008) | |
| C57BL/6J mice | Male | 27- to 28-mo compared to 13- to 14-mo and 4- to 5-mo | No change in microglial number in hippocampal regions across ageing | (Long et al. 1998) | |
| C57BL/6J mice | Female | 20- to 24-mo compared to 13- to 14-mo and 3- to 4-mo |
Increase in microglial numbers with age in CA1 region Increased numbers at each age group compared to equivalent cohort in males (study above (253)) |
(Mouton et al. 2002) | |
| C57BL/6 mice | Male | 22- to 24-mo compared to 3- to 5-mo | ↑ MHCII positive microglia | (Garner et al. 2018) | |
| Macaca nemestrina | Females | 10- to 18-yo compared to 2- to 5-yo | ↑ MHCII positive microglia | (Sheffield and Berman 1998) | |
| C57BL/6J mice | Not specified | 20-mo compared to 3-mo | ↑ STING activity | (Gulen et al. 2023) | |
| Dysbiosis | BALB/c mice | Male | 18- to 20-mo compared to 3- to 4-mo |
Peripheral administration of LPS leads to hyperactive immune response ↑mRNA expression of IL-1β, IL-10 and TLR2 |
(Henry et al. 2009) |
**studies conducted in macrophages NOT microglia
Table 3.
Evidence of the hallmarks of ageing in microglia from human samples
| Hallmark | Features seen in aged microglia | ||||
|---|---|---|---|---|---|
| Model | Sex | Ages compared Age of observed change is underlined |
Changes observed | Ref | |
| Genomic instability | Human post-mortem tissue from the: temporal cortex, hippocampus, entorhinal cortex, substantia nigra, superior colliculi, locus coeruleus and inferior colliculi | 6M:8F | 65–96 years old (80.42 ± 9.07) | Dystrophic microglia associated with ↑ferritin but not γ-H2A.X staining | (Neumann et al. 2023) |
| Microglia isolated from human dorsolateral pre-frontal cortex (HuMi_Aged dataset) | 10F | 94.07 years ± 0.95 compared to published middle age microglial transcriptomic dataset (53 years ± 5.29) |
Identified 1060 upregulated, and 1174 downregulated genes with ageing ↑chromosomal maintenance pathways |
(Olah et al. 2018) | |
| Telomere attrition | Microglia isolated from human superior frontal gyrus/frontal pole region |
AD = 3M:1F ND = M |
4 AD cases (86-years-old ± 2.54) One non-dementia case (86-years-old) |
↓telomere length measured by flow-FISH No change in telomerase activity |
(Flanary et al. 2007) |
| Epigenetic alterations | HuMi_Aged dataset | 10F | 94.07 years ± 0.95 |
Analysis of REACTOME pathways revealed enrichment of DNA methylation pathways Upregulation of DNA methylation and histone deacetylase pathways compared to microglia from middle age (53 years old) dataset |
(Olah et al. 2018) |
| Loss of proteostasis | HuMi_Aged dataset | 10F | 94.07 years ± 0.95 | ER-phagosome pathway enriched in HuMi dataset and upregulated compared to middle-aged microglia (53 years old) | (Olah et al. 2018) |
| Disabled macroautophagy | No studies investigating human microglia during ageing | ||||
| Deregulated nutrient sensing | No studies investigating human microglia during ageing | ||||
| Mitochondrial dysfunction | No studies investigating human microglia during ageing | ||||
| Cellular senescence | HuMi_Aged dataset | 10F | 94.07 years ± 0.95 | Enrichment of telomere induced senescence pathway and SASP pathways | (Olah et al. 2018) |
| Stem cell exhaustion | Human post-mortem tissue | Not specified | 58- to 79-yo compared to 20- to 35-yo | No changes in number of proliferating (Ki67+) microglia | (Askew et al. 2017) |
| Altered intercellular communication | No studies investigating human microglia during ageing | ||||
| Chronic inflammation | Human post-mortem neocortex | Males and females |
Age range females 18–93 years Age range males 19–87 years |
Number of microglia increased with age in females but not males | (Pelvig et al. 2008) |
| In vivo human brain (PET imaging) | Males and females (22M:11F) | 19 to 79-years-old | ↑ (R)-[11c]PK111955 ligand binding associated with ↑ age | (Schuitemaker et al. 2012) | |
| Microglia isolated from human right parietal cortex | N = 39, sex not specified | 34 to 102-years-old | Top 100 genes most affected by ageing were enriched for immune response pathways | (Galatro et al. 2017) | |
| HuMi_Aged dataset | 10F | 94.07 years ± 0.95 | Interferon signalling pathways enriched in HuMi dataset and upregulated compared to middle-aged microglia (53 years old) | (Olah et al. 2018) | |
| Dysbiosis | No studies investigating human microglia during ageing | ||||
Much of the literature discussed in the current review is based on investigations in small animal models, with limited research conducted in aged microglia in human samples to date. Notably, for several hallmarks (i.e. disabled macroautophagy, dysregulated nutrient sensing, mitochondrial dysfunction, altered intercellular communication and dysbiosis), we could not identify any human studies investigating microglia-specific changes with age. Further, while one transcriptional study by Olah et al. (2018) identified changes related to epigenetic alterations, loss of proteostasis and cellular senescence, beyond this singular study, there was also a lack of research examining these hallmarks in human microglia specifically. The limited literature on human microglia with age is likely due to the difficulties associated with obtaining human tissue (Rush et al. 2024). Unlike in laboratory models, where brain tissue can be collected at controlled timepoints, studying human microglia relies on post-mortem samples, which can be difficult to obtain, particularly for healthy older adults. Additionally, variables which may influence data, including post-mortem interval and pre-existing pathology, particularly in aged individuals, further complicates this analysis. To combat these challenges, future research should make use of in vivo imaging techniques, including positron emission tomography (PET, which has been utilised to assess human brain ageing and neuroinflammation ((R)-[11c]PK111955 (Schuitemaker et al. 2012), [18F]FDG (Xue et al. 2024; Doering et al. 2023), 18F-AV-1451 (Schöll et al. 2016)). Although such tracers differ in their specificity to microglia (reviewed: (Beaino et al. 2021)), they nevertheless present opportunities to elucidate age-related brain changes without reliance on post-mortem tissues.
In addition, there is an underappreciation of sex-specific differences in microglial ageing and immune activation. Previous literature has highlighted sex differences in microglia with age and in neuroinflammation more generally. For instance, in female Fischer 344 rats, microglial number in the dentate gyrus increases with age at 18-mo compared to in rats at 3-mo, but no difference is observed in male rats (Perkins et al. 2018). Similarly, in human post-mortem tissue, no changes in the number of microglia in the neocortex has been observed with age in males (19 to 87 years), but increased numbers have been seen in females (18 to 93 years) (Pelvig et al. 2008). These studies suggest sex-specific differences in microglial dynamics, which have been further explored and reviewed (Cyr and de Rivero Vaccari 2023; Lynch 2022) and have been suggested as a driving mechanism for broader sex differences seen in ND susceptibility (Lynch 2022). This is of concern, as only 20% of pre-clinical neuroscience studies in rodents use both sexes and 25% do not report the sex of subjects (Beery 2018). Future research should, therefore, endeavour to consistently include and report data for both female and male animals, in accordance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines (Percie du Sert et al. 2020) in order to better understand the influence of sex on age-related changes in microglia.
Beyond a general lack of knowledge regarding microglial expression of key hallmarks of ageing, the current review highlights the inconsistencies in classifying microglia, a well-documented issue in the field (Paolicelli et al. 2022; Reddaway et al. 2023; Green et al. 2022). Previous reviews have discouraged the use of outdated terms, such as resting and activated (Paolicelli et al. 2022) and outlined the lack of consensus regarding morphological classifications of microglia (Reddaway et al. 2023). While extensive discussion of this issue is beyond the scope of the current review, in order to work towards a cohesive understanding of aged microglia and move the field forward, this issue must be addressed. When classing populations of microglia, future studies should refer to functional classifications, rather than relying solely on morphological profile. Further, as outlined by (Paolicelli et al. 2022), it is critical that the wider scientific community collaborate to establish a unified approach to microglial analysis and classification potentially through the use of complex analyses, such as single-cell proteomics and transcriptomics.
Conclusions
As the major immune cells of the brain, microglia have a critical role in maintaining brain health and responding to immune challenges. Ageing has a profound impact on microglial function and intrinsic changes within these cells are suggested to contribute to vulnerability of the brain to age-related disorders, including NDs. Currently, the literature defines aged microglia as pro-inflammatory and dystrophic, with a similar phenotype to that seen in disease. However, studies on microglial morphology may benefit from greater consistency of terminology and clear definition of microglial phenotypes. In addition, studies have largely focused on pro-inflammatory processes linked to microglia in NDs or following injury. However, understanding the changes which occur in microglia with physiological ageing alone is critical in understanding the contribution of the ageing process to disease susceptibility and progression.
Here, a consideration of alterations in the 12 hallmarks of ageing (López-Otín et al. 2023) within microglia highlights clear gaps in our knowledge of how these processes change in microglia with increasing age. Many of these processes have been investigated in the context of disease (e.g. NDs), but studies have only rarely investigated the impact of ageing alone on these hallmarks. For example, both chronic inflammation and changes in proteostasis within microglia have been extensively studied in the context of ND. Conversely, other hallmarks not as clearly associated with ND pathophysiology, such as gut dysbiosis and stem cell exhaustion, have been investigated in aged microglia in only a handful of studies to date. Thus, the full phenotype of aged microglia remains poorly characterised and needs to be better understood to improve our ability to capture these changes in research models. As such, a comprehensive characterisation of microglia at different points across the lifespan is needed to understand how these cells contribute to vulnerability to disease, which may lead to the identification of novel therapeutic targets to reduce detrimental effects related to microglial ageing.
Author Contributions
All authors contributed to the development of this manuscript. Conceptualisation and formulation of the first manuscript draft and production of figures was conducted by LC. SM provided input on previous versions of the manuscript. LCP assisted in comceptualisation and provided input on previous versions of the manuscript. All authors read and approved the final version of the manuscript.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. LC was supported by an Australian Government Research Training Program Scholarship provided by the Department of Education and Training, Canberra, Australia. Figures were created using BioRender.
Data Availability
No datasets were generated or analysed during the current study.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Abdel-Haq R, Schlachetzki JCM, Glass CK, Mazmanian SK (2019) Microbiome-microglia connections via the gut-brain axis. J Exp Med 216(1):41–59. 10.1084/jem.20180794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aird KM, Zhang R (2013) Detection of senescence-associated heterochromatin foci (SAHF). Methods Mol Biol 965:185–196. 10.1007/978-1-62703-239-1_12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ajami B, Bennett JL, Krieger C, Tetzlaff W, Rossi FMV (2007) Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat Neurosci 10(12):1538–1543. 10.1038/nn2014 [DOI] [PubMed] [Google Scholar]
- Alliot F, Godin I, Pessac B (1999) Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res Dev Brain Res 117(2):145–152. 10.1016/s0165-3806(99)00113-3 [DOI] [PubMed] [Google Scholar]
- Ames BN, Atamna H, Killilea DW (2005) Mineral and vitamin deficiencies can accelerate the mitochondrial decay of aging. Mol Aspects Med 26(4):363–378. 10.1016/j.mam.2005.07.007 [DOI] [PubMed] [Google Scholar]
- Amorim JA, Coppotelli G, Rolo AP, Palmeira CM, Ross JM, Sinclair DA (2022) Mitochondrial and metabolic dysfunction in ageing and age-related diseases. Nat Rev Endocrinol 18(4):243–258. 10.1038/s41574-021-00626-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andronie-Cioara FL, Ardelean AI, Nistor-Cseppento CD, Jurcau A, Jurcau MC, Pascalau N, Marcu F (2023) Molecular mechanisms of neuroinflammation in aging and Alzheimer’s disease progression. Int J Mol Sci. 10.3390/ijms24031869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angelova DM, Brown DR (2019) Microglia and the aging brain: are senescent microglia the key to neurodegeneration? J Neurochem 151(6):676–688. 10.1111/jnc.14860 [DOI] [PubMed] [Google Scholar]
- Arvanitaki ES, Goulielmaki E, Gkirtzimanaki K, Niotis G, Tsakani E, Nenedaki E, Rouska I, Kefalogianni M, Xydias D, Kalafatakis I, Psilodimitrakopoulos S, Karagogeos D, Schumacher B, Stratakis E, Garinis GA (2024) Microglia-derived extracellular vesicles trigger age-related neurodegeneration upon DNA damage. Proc Natl Acad Sci 121(17):e2317402121. 10.1073/pnas.2317402121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Askew K, Li K, Olmos-Alonso A, Garcia-Moreno F, Liang Y, Richardson P, Tipton T, Chapman MA, Riecken K, Beccari S, Sierra A, Molnár Z, Cragg MS, Garaschuk O, Perry VH, Gomez-Nicola D (2017) Coupled proliferation and apoptosis maintain the rapid turnover of microglia in the adult brain. Cell Rep 18(2):391–405. 10.1016/j.celrep.2016.12.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Augusto-Oliveira M, Arrifano GdP, Leal-Nazaré CG, Chaves-Filho A, Santos-Sacramento L, Lopes-Araujo A, Tremblay M-È, Crespo-Lopez ME (2025) Morphological diversity of microglia: Implications for learning, environmental adaptation, ageing, sex differences and neuropathology. Neurosci Biobehav Rev 172:106091. 10.1016/j.neubiorev.2025.106091 [DOI] [PubMed] [Google Scholar]
- Bachstetter AD, Xing B, de Almeida L, Dimayuga ER, Watterson DM, Van Eldik LJ (2011) Microglial p38α MAPK is a key regulator of proinflammatory cytokine up-regulation induced by toll-like receptor (TLR) ligands or beta-amyloid (Aβ). J Neuroinflammation 8(1):79. 10.1186/1742-2094-8-79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachstetter AD, Ighodaro ET, Hassoun Y, Aldeiri D, Neltner JH, Patel E, Abner EL, Nelson PT (2017) Rod-shaped microglia morphology is associated with aging in 2 human autopsy series. Neurobiol Aging 52:98–105. 10.1016/j.neurobiolaging.2016.12.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barger SW, Harmon AD (1997) Microglial activation by Alzheimer amyloid precursor protein and modulation by apolipoprotein E. Nature 388(6645):878–881. 10.1038/42257 [DOI] [PubMed] [Google Scholar]
- Barron HC, Mars RB, Dupret D, Lerch JP, Sampaio-Baptista C (2021) Cross-species neuroscience: closing the explanatory gap. Philos Trans R Soc Lond B Biol Sci 376(1815):20190633. 10.1098/rstb.2019.0633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beaino W, Janssen B, Vugts DJ, de Vries HE, Windhorst AD (2021) Towards PET imaging of the dynamic phenotypes of microglia. Clin Exp Immunol 206(3):282–300. 10.1111/cei.13649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beery AK (2018) Inclusion of females does not increase variability in rodent research studies. Curr Opin Behav Sci 23:143–149. 10.1016/j.cobeha.2018.06.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell JE, Alafuzoff I, Al-Sarraj S, Arzberger T, Bogdanovic N, Budka H, Dexter DT, Falkai P, Ferrer I, Gelpi E, Gentleman SM, Giaccone G, Huitinga I, Ironside JW, Klioueva N, Kovacs GG, Meyronet D, Palkovits M, Parchi P, Patsouris E, Reynolds R, Riederer P, Roggendorf W, Seilhean D, Schmitt A, Schmitz P, Streichenberger N, Schwalber A, Kretzschmar H (2008) Management of a twenty-first century brain bank: experience in the BrainNet Europe consortium. Acta Neuropathol 115(5):497–507. 10.1007/s00401-008-0360-8 [DOI] [PubMed] [Google Scholar]
- Bernier L-P, York EM, Kamyabi A, Choi HB, Weilinger NL, MacVicar BA (2020) Microglial metabolic flexibility supports immune surveillance of the brain parenchyma. Nat Commun 11(1):1559. 10.1038/s41467-020-15267-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhat R, Crowe EP, Bitto A, Moh M, Katsetos CD, Garcia FU, Johnson FB, Trojanowski JQ, Sell C, Torres C (2012) Astrocyte senescence as a component of Alzheimer’s disease. PLoS ONE 7(9):e45069. 10.1371/journal.pone.0045069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhatia S, Rawal R, Sharma P, Singh T, Singh M, Singh V (2022) Mitochondrial dysfunction in Alzheimer’s disease: opportunities for drug development. Curr Neuropharmacol 20(4):675–692. 10.2174/1570159x19666210517114016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borgonetti V, Galeotti N (2023) Microglia senescence is related to neuropathic pain-associated comorbidities in the spared nerve injury model. Pain 164(5):1106–1117. 10.1097/j.pain.0000000000002807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borst K, Dumas AA, Prinz M (2021) Microglia: Immune and non-immune functions. Immunity 54(10):2194–2208. 10.1016/j.immuni.2021.09.014 [DOI] [PubMed] [Google Scholar]
- Böttcher C, Schlickeiser S, Sneeboer MAM, Kunkel D, Knop A, Paza E, Fidzinski P, Kraus L, Snijders GJL, Kahn RS, Schulz AR, Mei HE, Hol EM, Siegmund B, Glauben R, Spruth EJ, de Witte LD, Priller J, Psy NBB (2019) Human microglia regional heterogeneity and phenotypes determined by multiplexed single-cell mass cytometry. Nat Neurosci 22(1):78–90. 10.1038/s41593-018-0290-2 [DOI] [PubMed] [Google Scholar]
- Bruttger J, Karram K, Wörtge S, Regen T, Marini F, Hoppmann N, Klein M, Blank T, Yona S, Wolf Y, Mack M, Pinteaux E, Müller W, Zipp F, Binder H, Bopp T, Prinz M, Jung S, Waisman A (2015) Genetic cell ablation reveals clusters of local self-renewing microglia in the mammalian central nervous system. Immunity 43(1):92–106. 10.1016/j.immuni.2015.06.012 [DOI] [PubMed] [Google Scholar]
- Bussian TJ, Aziz A, Meyer CF, Swenson BL, van Deursen JM, Baker DJ (2018) Clearance of senescent glial cells prevents tau-dependent pathology and cognitive decline. Nature 562(7728):578–582. 10.1038/s41586-018-0543-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Z, Yan LJ, Li K, Quazi SH, Zhao B (2012) Roles of AMP-activated protein kinase in Alzheimer’s disease. Neuromolecular Med 14(1):1–14. 10.1007/s12017-012-8173-2 [DOI] [PubMed] [Google Scholar]
- Carr LM, Mustafa S, Care A, Collins-Praino LE (2024) More than a number: incorporating the aged phenotype to improve in vitro and in vivo modeling of neurodegenerative disease. Brain Behav Immun 119:554–571. 10.1016/j.bbi.2024.04.023 [DOI] [PubMed] [Google Scholar]
- Castilla-Cortázar I, Aguirre GA, Femat-Roldán G, Martín-Estal I, Espinosa L (2020) Is insulin-like growth factor-1 involved in Parkinson’s disease development? J Transl Med 18(1):70. 10.1186/s12967-020-02223-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CC, Lin JT, Cheng YF, Kuo CY, Huang CF, Kao SH, Liang YJ, Cheng CY, Chen HM (2014) Amelioration of LPS-induced inflammation response in microglia by AMPK activation. Biomed Res Int 2014:692061. 10.1155/2014/692061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen M, Linstra R, van Vugt MATM (2022) Genomic instability, inflammatory signaling and response to cancer immunotherapy. Biochimica et Biophysica Acta (BBA) Reviews on Cancer. 1877 (1): 188661. 10.1016/j.bbcan.2021.188661 [DOI] [PubMed]
- Chiarini A, Gui L, Viviani C, Armato U, Dal Prà I (2023) NLRP3 inflammasome’s activation in acute and chronic brain diseases-an update on pathogenetic mechanisms and therapeutic perspectives with respect to other inflammasomes. Biomedicines. 10.3390/biomedicines11040999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinta SJ, Woods G, Demaria M, Rane A, Zou Y, McQuade A, Rajagopalan S, Limbad C, Madden DT, Campisi J, Andersen JK (2018) Cellular senescence is induced by the environmental neurotoxin paraquat and contributes to neuropathology linked to Parkinson’s disease. Cell Rep 22(4):930–940. 10.1016/j.celrep.2017.12.092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho M-H, Cho K, Kang H-J, Jeon E-Y, Kim H-S, Kwon H-J, Kim H-M, Kim D-H, Yoon S-Y (2014) Autophagy in microglia degrades extracellular β-amyloid fibrils and regulates the NLRP3 inflammasome. Autophagy 10(10):1761–1775. 10.4161/auto.29647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coleman C, Martin I (2022) Unraveling Parkinson’s disease neurodegeneration: does aging hold the clues? J Parkinsons Dis 12:2321–2338. 10.3233/jpd-223363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cozachenco D, Ribeiro FC, Ferreira ST (2023) Defective proteostasis in Alzheimer’s disease. Ageing Res Rev 85:101862. 10.1016/j.arr.2023.101862 [DOI] [PubMed] [Google Scholar]
- Cuadros MA, Martin C, Coltey P, Almendros A, Navascués J (1993) First appearance, distribution, and origin of macrophages in the early development of the avian central nervous system. J Compar Neurol 330(1):113–129. 10.1002/cne.903300110 [DOI] [PubMed] [Google Scholar]
- Cunningham CL, Martínez-Cerdeño V, Noctor SC (2013) Microglia regulate the number of neural precursor cells in the developing cerebral cortex. J Neurosci 33(10):4216. 10.1523/jneurosci.3441-12.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cyr B, de Rivero Vaccari JP (2023) Sex differences in the inflammatory profile in the brain of young and aged mice. Cells. 10.3390/cells12101372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- d’Avila JC, Siqueira LD, Mazeraud A, Azevedo EP, Foguel D, Castro-Faria-Neto HC, Sharshar T, Chrétien F, Bozza FA (2018) Age-related cognitive impairment is associated with long-term neuroinflammation and oxidative stress in a mouse model of episodic systemic inflammation. J Neuroinflammation 15(1):28. 10.1186/s12974-018-1059-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis E, Lloyd AF (2024) The proteomic landscape of microglia in health and disease. Front Cell Neurosci. 10.3389/fncel.2024.1379717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Leonibus C, Maddaluno M, Ferriero R, Besio R, Cinque L, Lim PJ, Palma A, De Cegli R, Gagliotta S, Montefusco S, Iavazzo M, Rohrbach M, Giunta C, Polishchuk E, Medina DL, Di Bernardo D, Forlino A, Piccolo P, Settembre C (2024) Sestrin2 drives ER-phagy in response to protein misfolding. Dev Cell 59(16):2035-2052.e2010. 10.1016/j.devcel.2024.07.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng Y, Lu J, Sivakumar V, Ling EA, Kaur C (2008) Amoeboid microglia in the periventricular white matter induce oligodendrocyte damage through expression of proinflammatory cytokines via MAP kinase signaling pathway in hypoxic neonatal rats. Brain Pathol 18(3):387–400. 10.1111/j.1750-3639.2008.00138.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dlima SD, Hall A, Aminu AQ, Akpan A, Todd C, Vardy ERLC (2024) Frailty: a global health challenge in need of local action. BMJ Glob Health 9(8):e015173. 10.1136/bmjgh-2024-015173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doering E, Antonopoulos G, Hoenig M, van Eimeren T, Daamen M, Boecker H, Jessen F, Düzel E, Eickhoff S, Patil K, Drzezga A (2023) MRI or 18F-FDG PET for brain age gap estimation: links to cognition, pathology, and alzheimer disease progression. J Nucl Med. 10.2967/jnumed.123.265931 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong-Chen X, Yong C, Yang X, Chen-Yu S, Li-Hua P (2023) Signaling pathways in Parkinson’s disease: molecular mechanisms and therapeutic interventions. Signal Transduct Target Ther 8(1):73. 10.1038/s41392-023-01353-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doust YV, Sumargo N, Ziebell JM, Premilovac D (2022) Insulin resistance in the brain: evidence supporting a role for inflammation, reactive microglia, and the impact of biological sex. Neuroendocrinology 112(11):1027–1038. 10.1159/000524059 [DOI] [PubMed] [Google Scholar]
- Elmore MR, Najafi AR, Koike MA, Dagher NN, Spangenberg EE, Rice RA, Kitazawa M, Matusow B, Nguyen H, West BL, Green KN (2014) Colony-stimulating factor 1 receptor signaling is necessary for microglia viability, unmasking a microglia progenitor cell in the adult brain. Neuron 82(2):380–397. 10.1016/j.neuron.2014.02.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elmore MRP, Hohsfield LA, Kramár EA, Soreq L, Lee RJ, Pham ST, Najafi AR, Spangenberg EE, Wood MA, West BL, Green KN (2018) Replacement of microglia in the aged brain reverses cognitive, synaptic, and neuronal deficits in mice. Aging Cell 17(6):e12832. 10.1111/acel.12832 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabrizio P, Pozza F, Pletcher SD, Gendron CM, Longo VD (2001) Regulation of longevity and stress resistance by Sch9 in yeast. Science 292(5515):288–290. 10.1126/science.1059497 [DOI] [PubMed] [Google Scholar]
- Fairley LH, Wong JH, Barron AM (2021) Mitochondrial regulation of microglial immunometabolism in Alzheimer’s disease. Front Immunol 12:624538. 10.3389/fimmu.2021.624538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fasching CL (2018) Telomere length measurement as a clinical biomarker of aging and disease. Crit Rev Clin Lab Sci 55(7):443–465. 10.1080/10408363.2018.1504274 [DOI] [PubMed] [Google Scholar]
- Flanary BE, Streit WJ (2004) Progressive telomere shortening occurs in cultured rat microglia, but not astrocytes. Glia 45(1):75–88. 10.1002/glia.10301 [DOI] [PubMed] [Google Scholar]
- Flanary BE, Sammons NW, Nguyen C, Walker D, Streit WJ (2007) Evidence that aging and amyloid promote microglial cell senescence. Rejuvenation Res 10(1):61–74. 10.1089/rej.2006.9096 [DOI] [PubMed] [Google Scholar]
- Flowers A, Bell-Temin H, Jalloh A, Stevens SM, Bickford PC (2017) Proteomic analysis of aged microglia: shifts in transcription, bioenergetics, and nutrient response. J Neuroinflammation 14(1):96. 10.1186/s12974-017-0840-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forero DA, González-Giraldo Y, López-Quintero C, Castro-Vega LJ, Barreto GE, Perry G (2016) Telomere length in Parkinson’s disease: a meta-analysis. Exp Gerontol 75:53–55. 10.1016/j.exger.2016.01.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Füger P, Hefendehl JK, Veeraraghavalu K, Wendeln A-C, Schlosser C, Obermüller U, Wegenast-Braun BM, Neher JJ, Martus P, Kohsaka S, Thunemann M, Feil R, Sisodia SS, Skodras A, Jucker M (2017) Microglia turnover with aging and in an Alzheimer’s model via long-term in vivo single-cell imaging. Nat Neurosci 20(10):1371–1376. 10.1038/nn.4631 [DOI] [PubMed] [Google Scholar]
- Gabbouj S, Ryhänen S, Marttinen M, Wittrahm R, Takalo M, Kemppainen S, Martiskainen H, Tanila H, Haapasalo A, Hiltunen M, Natunen T (2019) Altered insulin signaling in Alzheimer’s disease brain – special emphasis on PI3K-Akt pathway. Front Neurosci. 10.3389/fnins.2019.00629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gadhave K, Kumar D, Uversky VN, Giri R (2021) A multitude of signaling pathways associated with Alzheimer’s disease and their roles in AD pathogenesis and therapy. Med Res Rev 41(5):2689–2745. 10.1002/med.21719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galatro TF, Holtman IR, Lerario AM, Vainchtein ID, Brouwer N, Sola PR, Veras MM, Pereira TF, Leite REP, Möller T, Wes PD, Sogayar MC, Laman JD, den Dunnen W, Pasqualucci CA, Oba-Shinjo SM, Boddeke E, Marie SKN, Eggen BJL (2017) Transcriptomic analysis of purified human cortical microglia reveals age-associated changes. Nat Neurosci 20(8):1162–1171. 10.1038/nn.4597 [DOI] [PubMed] [Google Scholar]
- Gamage R, Rossetti I, Niedermayer G, Münch G, Buskila Y, Gyengesi E (2023) Chronic neuroinflammation during aging leads to cholinergic neurodegeneration in the mouse medial septum. J Neuroinflammation 20(1):235. 10.1186/s12974-023-02897-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gampawar P, Schmidt R, Schmidt H (2022) Telomere length and brain aging: a systematic review and meta-analysis. Ageing Res Rev 80:101679. 10.1016/j.arr.2022.101679 [DOI] [PubMed] [Google Scholar]
- Gao AW, Cantó C, Houtkooper RH (2014) Mitochondrial response to nutrient availability and its role in metabolic disease. EMBO Mol Med 6(5):580–589. 10.1002/emmm.201303782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao X, Chen Q, Yao H, Tan J, Liu Z, Zhou Y, Zou Z (2022) Epigenetics in Alzheimer’s disease. Front Aging Neurosci. 10.3389/fnagi.2022.911635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao C, Jiang J, Tan Y, Chen S (2023) Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal Transduct Target Ther 8(1):359. 10.1038/s41392-023-01588-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garner KM, Amin R, Johnson RW, Scarlett EJ, Burton MD (2018) Microglia priming by interleukin-6 signaling is enhanced in aged mice. J Neuroimmunol 324:90–99. 10.1016/j.jneuroim.2018.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garza-Lombó C, Schroder A, Reyes-Reyes EM, Franco R (2018) mTOR/AMPK signaling in the brain: Cell metabolism, proteostasis and survival. Curr Opin Toxicol 8:102–110. 10.1016/j.cotox.2018.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georgakopoulou EA, Tsimaratou K, Evangelou K, Fernandez Marcos PJ, Zoumpourlis V, Trougakos IP, Kletsas D, Bartek J, Serrano M, Gorgoulis VG (2013) Specific lipofuscin staining as a novel biomarker to detect replicative and stress-induced senescence. A method applicable in cryo-preserved and archival tissues. Aging (Albany NY). 5(1): 37–50. 10.18632/aging.100527 [DOI] [PMC free article] [PubMed]
- Ghavami M, Fairn GD (2022) Endoplasmic reticulum-Phagosome contact sites from the cradle to the grave. Front Cell Dev Biol 10:1074443. 10.3389/fcell.2022.1074443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh TS, Shanahan F, O’Toole PW (2022) The gut microbiome as a modulator of healthy ageing. Nat Rev Gastroenterol Hepatol 19(9):565–584. 10.1038/s41575-022-00605-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ginhoux F, Lim S, Hoeffel G, Low D, Huber T (2013) Origin and differentiation of microglia. Front Cell Neurosci. 10.3389/fncel.2013.00045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giordano KR, Denman CR, Dubisch PS, Akhter M, Lifshitz J (2021) An update on the rod microglia variant in experimental and clinical brain injury and disease. Brain Commun. 10.1093/braincomms/fcaa227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giovannoni F, Quintana FJ (2020) The role of astrocytes in CNS inflammation. Trends Immunol 41(9):805–819. 10.1016/j.it.2020.07.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giulian D, Baker TJ (1986) Characterization of ameboid microglia isolated from developing mammalian brain. J Neurosci 6(8):2163–2178. 10.1523/jneurosci.06-08-02163.1986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glezer I, Simard AR, Rivest S (2007) Neuroprotective role of the innate immune system by microglia. Neuroscience 147(4):867–883. 10.1016/j.neuroscience.2007.02.055 [DOI] [PubMed] [Google Scholar]
- González-Gualda E, Baker AG, Fruk L, Muñoz-Espín D (2021) A guide to assessing cellular senescence in vitro and in vivo. FEBS J 288(1):56–80. 10.1111/febs.15570 [DOI] [PubMed] [Google Scholar]
- Gorgoulis V, Adams PD, Alimonti A, Bennett DC, Bischof O, Bishop C, Campisi J, Collado M, Evangelou K, Ferbeyre G, Gil J, Hara E, Krizhanovsky V, Jurk D, Maier AB, Narita M, Niedernhofer L, Passos JF, Robbins PD, Schmitt CA, Sedivy J, Vougas K, von Zglinicki T, Zhou D, Serrano M, Demaria M (2019) Cellular senescence: defining a path forward. Cell 179(4):813–827. 10.1016/j.cell.2019.10.005 [DOI] [PubMed] [Google Scholar]
- Green TRF, Rowe RK (2024) Quantifying microglial morphology: an insight into function. Clin Exp Immunol 216(3):221–229. 10.1093/cei/uxae023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green TRF, Murphy SM, Rowe RK (2022) Comparisons of quantitative approaches for assessing microglial morphology reveal inconsistencies, ecological fallacy, and a need for standardization. Sci Rep 12(1):18196. 10.1038/s41598-022-23091-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwood EK, Brown DR (2021) Senescent microglia: the key to the ageing brain? Int J Mol Sci. 10.3390/ijms22094402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grubman A, Choo XY, Chew G, Ouyang JF, Sun G, Croft NP, Rossello FJ, Simmons R, Buckberry S, Landin DV, Pflueger J, Vandekolk TH, Abay Z, Zhou Y, Liu X, Chen J, Larcombe M, Haynes JM, McLean C, Williams S, Chai SY, Wilson T, Lister R, Pouton CW, Purcell AW, Rackham OJL, Petretto E, Polo JM (2021) Transcriptional signature in microglia associated with Aβ plaque phagocytosis. Nat Commun 12(1):3015. 10.1038/s41467-021-23111-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guimarães GR, Almeida PP, de Oliveira SL, Rodrigues LP, de Carvalho JL, Boroni M (2021) Hallmarks of aging in macrophages: consequences to skin inflammaging. Cells. 10.3390/cells10061323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gulen MF, Samson N, Keller A, Schwabenland M, Liu C, Glück S, Thacker VV, Favre L, Mangeat B, Kroese LJ, Krimpenfort P, Prinz M, Ablasser A (2023) cGAS–STING drives ageing-related inflammation and neurodegeneration. Nature 620(7973):374–380. 10.1038/s41586-023-06373-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haas CB, de Carvalho AK, Muller AP, Eggen BJL, Portela LV (2020) Insulin activates microglia and increases COX-2/IL-1β expression in young but not in aged hippocampus. Brain Res 1741:146884. 10.1016/j.brainres.2020.146884 [DOI] [PubMed] [Google Scholar]
- Hammond TR, Dufort C, Dissing-Olesen L, Giera S, Young A, Wysoker A, Walker AJ, Gergits F, Segel M, Nemesh J, Marsh SE, Saunders A, Macosko E, Ginhoux F, Chen J, Franklin RJM, Piao X, McCarroll SA, Stevens B (2019) Single-cell RNA sequencing of microglia throughout the mouse lifespan and in the injured brain reveals complex cell-state changes. Immunity 50(1):253-271.e256. 10.1016/j.immuni.2018.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Z, Xu Z, Chen L, Ye D, Yu Y, Zhang Y, Cao Y, Djibril B, Guo X, Gao X, Zhang W, Yu M, Liu S, Yan G, Jin M, Huang Q, Wang X, Hua B, Feng C, Yang F, Ma W, Liu Y (2020) Iron overload inhibits self-renewal of human pluripotent stem cells via DNA damage and generation of reactive oxygen species. FEBS Open Bio 10(5):726–733. 10.1002/2211-5463.12811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harry GJ, Kraft AD (2012) Microglia in the developing brain: a potential target with lifetime effects. Neurotoxicology 33(2):191–206. 10.1016/j.neuro.2012.01.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartmann C, Herling L, Hartmann A, Köckritz V, Fuellen G, Walter M, Hermann A (2023) Systematic estimation of biological age of in vitro cell culture systems by an age-associated marker panel. Front Aging. 10.3389/fragi.2023.1129107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haughey NJ, Liu D, Nath A, Borchard AC, Mattson MP (2002) Disruption of neurogenesis in the subventricular zone of adult mice, and in human cortical neuronal precursor cells in culture, by amyloid β-peptide. NeuroMol Med 1(2):125–135. 10.1385/NMM:1:2:125 [DOI] [PubMed] [Google Scholar]
- Hayashi Y, Yoshida M, Yamato M, Ide T, Wu Z, Ochi-Shindou M, Kanki T, Kang D, Sunagawa K, Tsutsui H, Nakanishi H (2008) Reverse of age-dependent memory impairment and mitochondrial DNA damage in microglia by an overexpression of human mitochondrial transcription factor a in mice. J Neurosci 28(34):8624–8634. 10.1523/jneurosci.1957-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heckmann BL, Teubner BJW, Boada-Romero E, Tummers B, Guy C, Fitzgerald P, Mayer U, Carding S, Zakharenko SS, Wileman T, Green DR (2020) Noncanonical function of an autophagy protein prevents spontaneous Alzheimer’s disease. Sci Adv. 10.1126/sciadv.abb9036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellwig S, Brioschi S, Dieni S, Frings L, Masuch A, Blank T, Biber K (2016) Altered microglia morphology and higher resilience to stress-induced depression-like behavior in CX3CR1-deficient mice. Brain Behav Immun 55:126–137. 10.1016/j.bbi.2015.11.008 [DOI] [PubMed] [Google Scholar]
- Henrich MT, Oertel WH, Surmeier DJ, Geibl FF (2023) Mitochondrial dysfunction in Parkinson’s disease – a key disease hallmark with therapeutic potential. Mol Neurodegener 18(1):83. 10.1186/s13024-023-00676-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry CJ, Huang Y, Wynne AM, Godbout JP (2009) Peripheral lipopolysaccharide (LPS) challenge promotes microglial hyperactivity in aged mice that is associated with exaggerated induction of both pro-inflammatory IL-1beta and anti-inflammatory IL-10 cytokines. Brain Behav Immun 23(3):309–317. 10.1016/j.bbi.2008.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herdy JR, Traxler L, Agarwal RK, Karbacher L, Schlachetzki JCM, Boehnke L, Zangwill D, Galasko D, Glass CK, Mertens J, Gage FH (2022) Increased post-mitotic senescence in aged human neurons is a pathological feature of Alzheimer’s disease. Cell Stem Cell 29(12):1637-1652.e1636. 10.1016/j.stem.2022.11.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Höhn A, Tramutola A, Cascella R (2020) Proteostasis failure in neurodegenerative diseases: focus on oxidative stress. Oxid Med Cell Longev 2020:5497046. 10.1155/2020/5497046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hou Y, Song H, Croteau DL, Akbari M, Bohr VA (2017) Genome instability in Alzheimer disease. Mech Ageing Dev 161:83–94. 10.1016/j.mad.2016.04.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL, Bohr VA (2019) Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 15(10):565–581. 10.1038/s41582-019-0244-7 [DOI] [PubMed] [Google Scholar]
- Huang Y, Xu Z, Xiong S, Sun F, Qin G, Hu G, Wang J, Zhao L, Liang YX, Wu T, Lu Z, Humayun MS, So KF, Pan Y, Li N, Yuan TF, Rao Y, Peng B (2018) Repopulated microglia are solely derived from the proliferation of residual microglia after acute depletion. Nat Neurosci 21(4):530–540. 10.1038/s41593-018-0090-8 [DOI] [PubMed] [Google Scholar]
- Huang Y, Wu J, Zhang H, Li Y, Wen L, Tan X, Cheng K, Liu Y, Pu J, Liu L, Wang H, Li W, Perry SW, Wong ML, Licinio J, Zheng P, Xie P (2023) The gut microbiome modulates the transformation of microglial subtypes. Mol Psychiatry 28(4):1611–1621. 10.1038/s41380-023-02017-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hugh Perry V (1998) A revised view of the central nervous system microenvironment and major histocompatibility complex class II antigen presentation. J Neuroimmunol 90(2):113–121. 10.1016/S0165-5728(98)00145-3 [DOI] [PubMed] [Google Scholar]
- Hughes EG, Bergles DE (2014) Hidden progenitors replace microglia in the adult brain. Neuron 82(2):253–255. 10.1016/j.neuron.2014.04.010 [DOI] [PubMed] [Google Scholar]
- Jauregui C, Blanco-Luquin I, Macías M, Roldan M, Caballero C, Pagola I, Mendioroz M, Jericó I (2023) Exploring the disease-associated microglia state in amyotrophic lateral sclerosis. Biomedicines. 10.3390/biomedicines11112994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jewell JL, Guan KL (2013) Nutrient signaling to mTOR and cell growth. Trends Biochem Sci 38(5):233–242. 10.1016/j.tibs.2013.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin R, Chan AKY, Wu J, Lee TMC (2022) Relationships between inflammation and age-related neurocognitive changes. Int J Mol Sci. 10.3390/ijms232012573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joya A, Martín A (2021) Evaluation of glial cell proliferation with non-invasive molecular imaging methods after stroke. Neural Regen Res 16(11):2209–2210. 10.4103/1673-5374.310681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung T, Bader N, Grune T (2007) Lipofuscin: formation, distribution, and metabolic consequences. Ann N Y Acad Sci 1119:97–111. 10.1196/annals.1404.008 [DOI] [PubMed] [Google Scholar]
- Jurga AM, Paleczna M, Kuter KZ (2020) Overview of general and discriminating markers of differential microglia phenotypes. Front Cell Neurosci 14:198. 10.3389/fncel.2020.00198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamo N, Ke B, Ghaffari AA, Shen XD, Busuttil RW, Cheng G, Kupiec-Weglinski JW (2013) ASC/caspase-1/IL-1β signaling triggers inflammatory responses by promoting HMGB1 induction in liver ischemia/reperfusion injury. Hepatology 58(1):351–362. 10.1002/hep.26320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keane L, Antignano I, Riechers SP, Zollinger R, Dumas AA, Offermann N, Bernis ME, Russ J, Graelmann F, McCormick PN, Esser J, Tejera D, Nagano A, Wang J, Chelala C, Biederbick Y, Halle A, Salomoni P, Heneka MT, Capasso M (2021) mTOR-dependent translation amplifies microglia priming in aging mice. J Clin Invest. 10.1172/jci13272734651589 [Google Scholar]
- Kenkhuis B, Somarakis A, de Haan L, Dzyubachyk O, Ijsselsteijn ME, de Miranda NFCC, Lelieveldt BPF, Dijkstra J, van Roon-Mom WMC, Höllt T, van der Weerd L (2021) Iron loading is a prominent feature of activated microglia in Alzheimer’s disease patients. Acta Neuropathol Commun 9(1):27. 10.1186/s40478-021-01126-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keren-Shaul H, Spinrad A, Weiner A, Matcovitch-Natan O, Dvir-Szternfeld R, Ulland TK, David E, Baruch K, Lara-Astaiso D, Toth B, Itzkovitz S, Colonna M, Schwartz M, Amit I (2017) A unique microglia type associated with restricting development of Alzheimer’s disease. Cell 169(7):1276-1290.e1217. 10.1016/j.cell.2017.05.018 [DOI] [PubMed] [Google Scholar]
- Khan MZ, Zugaza JL, Torres Aleman I (2025) The signaling landscape of insulin-like growth factor 1. J Biol Chem 301(1):108047. 10.1016/j.jbc.2024.108047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kigerl KA, de Rivero Vaccari JP, Dietrich WD, Popovich PG, Keane RW (2014) Pattern recognition receptors and central nervous system repair. Exp Neurol 258:5–16. 10.1016/j.expneurol.2014.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koellhoffer EC, McCullough LD, Ritzel RM (2017) Old maids: aging and its impact on microglia function. Int J Mol Sci. 10.3390/ijms18040769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolb H, Kempf K, Martin S (2023) Insulin and aging - a disappointing relationship. Front Endocrinol (Lausanne) 14:1261298. 10.3389/fendo.2023.1261298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuan X-Y, Fauzi NSA, Ng KY, Bakhtiar A (2023) Exploring the causal relationship between telomere biology and Alzheimer’s disease. Mol Neurobiol 60(8):4169–4183. 10.1007/s12035-023-03337-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar A, Stoica BA, Sabirzhanov B, Burns MP, Faden AI, Loane DJ (2013) Traumatic brain injury in aged animals increases lesion size and chronically alters microglial/macrophage classical and alternative activation states. Neurobiol Aging 34(5):1397–1411. 10.1016/j.neurobiolaging.2012.11.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- la Torre A, Lo Vecchio F, Greco A (2023) Epigenetic mechanisms of aging and aging-associated diseases. Cells. 10.3390/cells12081163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ladeby R, Wirenfeldt M, Garcia-Ovejero D, Fenger C, Dissing-Olesen L, Dalmau I, Finsen B (2005) Microglial cell population dynamics in the injured adult central nervous system. Brain Res Rev 48(2):196–206. 10.1016/j.brainresrev.2004.12.009 [DOI] [PubMed] [Google Scholar]
- Lan AP, Chen J, Zhao Y, Chai Z, Hu Y (2017) mTOR signaling in Parkinson’s disease. Neuromolecular Med 19(1):1–10. 10.1007/s12017-016-8417-7 [DOI] [PubMed] [Google Scholar]
- Lee IS, Jung K, Kim IS, Park KI (2013) Amyloid-β oligomers regulate the properties of human neural stem cells through GSK-3β signaling. Exp Mol Med 45(11):e60. 10.1038/emm.2013.125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehtonen Š, Sonninen TM, Wojciechowski S, Goldsteins G, Koistinaho J (2019) Dysfunction of cellular proteostasis in Parkinson’s disease. Front Neurosci 13:457. 10.3389/fnins.2019.00457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leuner K, Müller WE, Reichert AS (2012) From mitochondrial dysfunction to amyloid beta formation: novel insights into the pathogenesis of Alzheimer’s disease. Mol Neurobiol 46(1):186–193. 10.1007/s12035-012-8307-4 [DOI] [PubMed] [Google Scholar]
- Li Z, Li J, Bu X, Liu X, Tankersley CG, Wang C, Huang K (2011) Age-induced augmentation of p38 MAPK phosphorylation in mouse lung. Exp Gerontol 46(8):694–702. 10.1016/j.exger.2011.04.005 [DOI] [PubMed] [Google Scholar]
- Li X, Li Y, Jin Y, Zhang Y, Wu J, Xu Z, Huang Y, Cai L, Gao S, Liu T, Zeng F, Wang Y, Wang W, Yuan T-F, Tian H, Shu Y, Guo F, Lu W, Mao Y, Mei X, Rao Y, Peng B (2023a) Transcriptional and epigenetic decoding of the microglial aging process. Nature Aging 3(10):1288–1311. 10.1038/s43587-023-00479-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Liang H, Hu Y, Lu L, Zheng C, Fan Y, Wu B, Zou T, Luo X, Zhang X, Zeng Y, Liu Z, Zhou Z, Yue Z, Ren Y, Li Z, Su Q, Xu P (2023b) Gut bacterial profiles in Parkinson’s disease: A systematic review. CNS Neurosci Ther 29(1):140–157. 10.1111/cns.13990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang R, Qi X, Cai Q, Niu L, Huang X, Zhang D, Ling J, Wu Y, Chen Y, Yang P, Liu J, Zhang J, Yu P (2024) The role of NLRP3 inflammasome in aging and age-related diseases. Immun Ageing 21(1):14. 10.1186/s12979-023-00395-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Münch AE, Chung W-S, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Buckwalter MS, Rowitch DH, Dawson VL, Dawson TM, Stevens B, Barres BA (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541(7638):481–487. 10.1038/nature21029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu RM (2022) Aging, cellular senescence, and Alzheimer’s disease. Int J Mol Sci. 10.3390/ijms23041989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Li L (2019) Targeting autophagy for the treatment of Alzheimer’s disease: challenges and opportunities. Front Mol Neurosci. 10.3389/fnmol.2019.00203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu F, Benashski SE, Persky R, Xu Y, Li J, McCullough LD (2012) Age-related changes in AMP-activated protein kinase after stroke. Age (Dordr) 34(1):157–168. 10.1007/s11357-011-9214-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu T, Zhang L, Joo D, Sun S-C (2017) NF-κB signaling in inflammation. Signal Transduct Target Ther 2(1):17023. 10.1038/sigtrans.2017.23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Gao J, Zhu M, Liu K, Zhang HL (2020) Gut microbiota and dysbiosis in Alzheimer’s disease: implications for pathogenesis and treatment. Mol Neurobiol 57(12):5026–5043. 10.1007/s12035-020-02073-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Tan Y, Zhang Z, Yi M, Zhu L, Peng W (2024) The interaction between ageing and Alzheimer’s disease: insights from the hallmarks of ageing. Transl Neurodegeneration 13(1):7. 10.1186/s40035-024-00397-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loh JS, Mak WQ, Tan LKS, Ng CX, Chan HH, Yeow SH, Foo JB, Ong YS, How CW, Khaw KY (2024) Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases. Signal Transduct Target Ther 9(1):37. 10.1038/s41392-024-01743-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long JM, Kalehua AN, Muth NJ, Calhoun ME, Jucker M, Hengemihle JM, Ingram DK, Mouton PR (1998) Stereological analysis of astrocyte and microglia in aging mouse hippocampus. Neurobiol Aging 19(5):497–503. 10.1016/S0197-4580(98)00088-8 [DOI] [PubMed] [Google Scholar]
- Lopes KO, Sparks DL, Streit WJ (2008) Microglial dystrophy in the aged and Alzheimer’s disease brain is associated with ferritin immunoreactivity. Glia 56(10):1048–1060. 10.1002/glia.20678 [DOI] [PubMed] [Google Scholar]
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153(6):1194–1217. 10.1016/j.cell.2013.05.039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G (2023) Hallmarks of aging: an expanding universe. Cell 186(2):243–278. 10.1016/j.cell.2022.11.001 [DOI] [PubMed] [Google Scholar]
- Lu M, Sun X-L, Qiao C, Liu Y, Ding J-H, Hu G (2014) Uncoupling protein 2 deficiency aggravates astrocytic endoplasmic reticulum stress and nod-like receptor protein 3 inflammasome activation. Neurobiol Aging 35(2):421–430. 10.1016/j.neurobiolaging.2013.08.015 [DOI] [PubMed] [Google Scholar]
- Lynch MA (2022) Exploring sex-related differences in microglia may be a game-changer in precision medicine. Front Aging Neurosci 14:868448. 10.3389/fnagi.2022.868448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallya AP, Wang HD, Lee HNR, Deutch AY (2019) Microglial pruning of synapses in the prefrontal cortex during adolescence. Cereb Cortex 29(4):1634–1643. 10.1093/cercor/bhy061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manjally AV, Tay TL (2022) Attack of the clones: microglia in health and disease. Front Cell Neurosci. 10.3389/fncel.2022.831747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maras PM, Hebda-Bauer EK, Hagenauer MH, Hilde KL, Blandino P, Watson SJ, Akil H (2022) Differences in microglia morphological profiles reflect divergent emotional temperaments: insights from a selective breeding model. Transl Psychiatry 12(1):105. 10.1038/s41398-022-01821-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mastenbroek LJM, Kooistra SM, Eggen BJL, Prins JR (2024) The role of microglia in early neurodevelopment and the effects of maternal immune activation. Seminars Immunopathol 46(1):1. 10.1007/s00281-024-01017-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mather KA, Jorm AF, Parslow RA, Christensen H (2011) Is telomere length a biomarker of aging? A review. J Gerontol A Biol Sci Med Sci 66(2):202–213. 10.1093/gerona/glq180 [DOI] [PubMed] [Google Scholar]
- Matsui TK, Mori E (2018) Microglia support neural stem cell maintenance and growth. Biochem Biophys Res Commun 503(3):1880–1884. 10.1016/j.bbrc.2018.07.130 [DOI] [PubMed] [Google Scholar]
- Matt SM, Lawson MA, Johnson RW (2016) Aging and peripheral lipopolysaccharide can modulate epigenetic regulators and decrease IL-1β promoter DNA methylation in microglia. Neurobiol Aging 47:1–9. 10.1016/j.neurobiolaging.2016.07.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mejias NH, Martinez CC, Stephens ME, de Rivero Vaccari JP (2018) Contribution of the inflammasome to inflammaging. J Inflamm 15(1):23. 10.1186/s12950-018-0198-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menassa DA, Gomez-Nicola D (2018) Microglial dynamics during human brain development. Frontiers in Immunol. 10.3389/fimmu.2018.01014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menassa DA, Muntslag TAO, Martin-Estebané M, Barry-Carroll L, Chapman MA, Adorjan I, Tyler T, Turnbull B, Rose-Zerilli MJJ, Nicoll JAR, Krsnik Z, Kostovic I, Gomez-Nicola D (2022) The spatiotemporal dynamics of microglia across the human lifespan. Dev Cell 57(17):2127-2139.e2126. 10.1016/j.devcel.2022.07.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller SJ, Campbell CE, Jimenez-Corea HA, Wu GH, Logan R (2022) Neuroglial senescence, α-synucleinopathy, and the therapeutic potential of senolytics in Parkinson’s disease. Front Neurosci 16:824191. 10.3389/fnins.2022.824191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minhas PS, Latif-Hernandez A, McReynolds MR, Durairaj AS, Wang Q, Rubin A, Joshi AU, He JQ, Gauba E, Liu L, Wang C, Linde M, Sugiura Y, Moon PK, Majeti R, Suematsu M, Mochly-Rosen D, Weissman IL, Longo FM, Rabinowitz JD, Andreasson KI (2021) Restoring metabolism of myeloid cells reverses cognitive decline in ageing. Nature 590(7844):122–128. 10.1038/s41586-020-03160-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto A, Wake H, Ishikawa AW, Eto K, Shibata K, Murakoshi H, Koizumi S, Moorhouse AJ, Yoshimura Y, Nabekura J (2016) Microglia contact induces synapse formation in developing somatosensory cortex. Nat Commun 7(1):12540. 10.1038/ncomms12540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohr AE, Crawford M, Jasbi P, Fessler S, Sweazea KL (2022) Lipopolysaccharide and the gut microbiota: considering structural variation. FEBS Lett 596(7):849–875. 10.1002/1873-3468.14328 [DOI] [PubMed] [Google Scholar]
- Mouton PR, Long JM, Lei D-L, Howard V, Jucker M, Calhoun ME, Ingram DK (2002) Age and gender effects on microglia and astrocyte numbers in brains of mice. Brain Res 956(1):30–35. 10.1016/S0006-8993(02)03475-3 [DOI] [PubMed] [Google Scholar]
- Moyse E, Krantic S, Djellouli N, Roger S, Angoulvant D, Debacq C, Leroy V, Fougere B, Aidoud A (2022) Neuroinflammation: a possible link between chronic vascular disorders and neurodegenerative diseases. Front Aging Neurosci. 10.3389/fnagi.2022.827263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myhre CL, Thygesen C, Villadsen B, Vollerup J, Ilkjær L, Krohn KT, Grebing M, Zhao S, Khan AM, Dissing-Olesen L, Jensen MS, Babcock AA, Finsen B (2019) Microglia express insulin-like growth factor-1 in the hippocampus of aged APP(swe)/PS1(ΔE9) transgenic mice. Front Cell Neurosci 13:308. 10.3389/fncel.2019.00308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakanishi H, Zhang J, Koike M, Nishioku T, Okamoto Y, Kominami E, von Figura K, Peters C, Yamamoto K, Saftig P, Uchiyama Y (2001) Involvement of nitric oxide released from microglia-macrophages in pathological changes of cathepsin D-deficient mice. J Neurosci 21(19):7526–7533. 10.1523/jneurosci.21-19-07526.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ndoja A, Reja R, Lee S-H, Webster JD, Ngu H, Rose CM, Kirkpatrick DS, Modrusan Z, Chen Y-JJ, Dugger DL, Gandham V, Xie L, Newton K, Dixit VM (2020) Ubiquitin ligase COP1 suppresses neuroinflammation by degrading c/EBPβ in microglia. Cell 182(5):1156-1169.e1112. 10.1016/j.cell.2020.07.011 [DOI] [PubMed] [Google Scholar]
- Nechushtai L, Frenkel D, Pinkas-Kramarski R (2023) Autophagy in Parkinson’s disease. Biomolecules. 10.3390/biom13101435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumann P, Lenz DE, Streit WJ, Bechmann I (2023) Is microglial dystrophy a form of cellular senescence? An analysis of senescence markers in the aged human brain. Glia 71(2):377–390. 10.1002/glia.24282 [DOI] [PubMed] [Google Scholar]
- Ng PY, McNeely TL, Baker DJ (2023) Untangling senescent and damage-associated microglia in the aging and diseased brain. FEBS J 290(5):1326–1339. 10.1111/febs.16315 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nichols E, Szoeke CEI, Vollset SE, Abbasi N, Abd-Allah F, Abdela J, Aichour MTE, Akinyemi RO, Alahdab F, Asgedom SW, Awasthi A, Barker-Collo SL, Baune BT, Béjot Y, Belachew AB, Bennett DA, Biadgo B, Bijani A, Bin Sayeed MS, Brayne C, Carpenter DO, Carvalho F, Catalá-López F, Cerin E, Choi J-YJ, Dang AK, Degefa MG, Djalalinia S, Dubey M, Duken EE, Edvardsson D, Endres M, Eskandarieh S, Faro A, Farzadfar F, Fereshtehnejad S-M, Fernandes E, Filip I, Fischer F, Gebre AK, Geremew D, Ghasemi-Kasman M, Gnedovskaya EV, Gupta R, Hachinski V, Hagos TB, Hamidi S, Hankey GJ, Haro JM, Hay SI, Irvani SSN, Jha RP, Jonas JB, Kalani R, Karch A, Kasaeian A, Khader YS, Khalil IA, Khan EA, Khanna T, Khoja TAM, Khubchandani J, Kisa A, Kissimova-Skarbek K, Kivimäki M, Koyanagi A, Krohn KJ, Logroscino G, Lorkowski S, Majdan M, Malekzadeh R, März W, Massano J, Mengistu G, Meretoja A, Mohammadi M, Mohammadi-Khanaposhtani M, Mokdad AH, Mondello S, Moradi G, Nagel G, Naghavi M, Naik G, Nguyen LH, Nguyen TH, Nirayo YL, Nixon MR, Ofori-Asenso R, Ogbo FA, Olagunju AT, Owolabi MO, Panda-Jonas S, Passos VMdA, Pereira DM, Pinilla-Monsalve GD, Piradov MA, Pond CD, Poustchi H, Qorbani M, Radfar A, Reiner RC Jr, Robinson SR, Roshandel G, Rostami A, Russ TC, Sachdev PS, Safari H, Safiri S, Sahathevan R, Salimi Y, Satpathy M, Sawhney M, Saylan M, Sepanlou SG, Shafieesabet A, Shaikh MA, Sahraian MA, Shigematsu M, Shiri R, Shiue I, Silva JP, Smith M, Sobhani S, Stein DJ, Tabarés-Seisdedos R, Tovani-Palone MR, Tran BX, Tran TT, Tsegay AT, Ullah I, Venketasubramanian N, Vlassov V, Wang Y-P, Weiss J, Westerman R, Wijeratne T, Wyper GMA, Yano Y, Yimer EM, Yonemoto N, Yousefifard M, Zaidi Z, Zare Z, Vos T, Feigin VL, Murray CJL (2019) Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: a systematic analysis for the global burden of disease study 2016. The Lancet Neurology 18(1):88–106. 10.1016/S1474-4422(18)30403-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieto-Torres JL, Hansen M (2021) Macroautophagy and aging: the impact of cellular recycling on health and longevity. Mol Aspects Med 82:101020. 10.1016/j.mam.2021.101020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niraula A, Sheridan JF, Godbout JP (2017) Microglia priming with aging and stress. Neuropsychopharmacology 42(1):318–333. 10.1038/npp.2016.185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norden DM, Muccigrosso MM, Godbout JP (2015) Microglial priming and enhanced reactivity to secondary insult in aging, and traumatic CNS injury, and neurodegenerative disease. Neuropharmacology 96(Pt A):29–41. 10.1016/j.neuropharm.2014.10.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olah M, Patrick E, Villani A-C, Xu J, White CC, Ryan KJ, Piehowski P, Kapasi A, Nejad P, Cimpean M, Connor S, Yung CJ, Frangieh M, McHenry A, Elyaman W, Petyuk V, Schneider JA, Bennett DA, De Jager PL, Bradshaw EM (2018) A transcriptomic atlas of aged human microglia. Nat Commun 9(1):539. 10.1038/s41467-018-02926-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ou Z, Pan J, Tang S, Duan D, Yu D, Nong H, Wang Z (2021) Global trends in the incidence, prevalence, and years lived with disability of Parkinson’s disease in 204 countries/territories from 1990 to 2019. Front Public Health 9:776847. 10.3389/fpubh.2021.776847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pallas M, Camins A, Smith MA, Perry G, Lee H-g, Casadesus G (2008) From aging to Alzheimer’s disease: unveiling “The Switch” with the senescence-accelerated mouse model (SAMP8). J Alzheimers Dis 15:615–624. 10.3233/JAD-2008-15408 [DOI] [PubMed] [Google Scholar]
- Panicker N, Saminathan H, Jin H, Neal M, Harischandra DS, Gordon R, Kanthasamy K, Lawana V, Sarkar S, Luo J, Anantharam V, Kanthasamy AG, Kanthasamy A (2015) Fyn kinase regulates microglial neuroinflammatory responses in cell culture and animal models of Parkinson’s disease. J Neurosci 35(27):10058–10077. 10.1523/jneurosci.0302-15.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paolicelli RC, Bolasco G, Pagani F, Maggi L, Scianni M, Panzanelli P, Giustetto M, Ferreira TA, Guiducci E, Dumas L, Ragozzino D, Gross CT (2011) Synaptic pruning by microglia is necessary for normal brain development. Science 333(6048):1456–1458. 10.1126/science.1202529 [DOI] [PubMed] [Google Scholar]
- Paolicelli RC, Sierra A, Stevens B, Tremblay M-E, Aguzzi A, Ajami B, Amit I, Audinat E, Bechmann I, Bennett M, Bennett F, Bessis A, Biber K, Bilbo S, Blurton-Jones M, Boddeke E, Brites D, Brône B, Brown GC, Butovsky O, Carson MJ, Castellano B, Colonna M, Cowley SA, Cunningham C, Davalos D, De Jager PL, de Strooper B, Denes A, Eggen BJL, Eyo U, Galea E, Garel S, Ginhoux F, Glass CK, Gokce O, Gomez-Nicola D, González B, Gordon S, Graeber MB, Greenhalgh AD, Gressens P, Greter M, Gutmann DH, Haass C, Heneka MT, Heppner FL, Hong S, Hume DA, Jung S, Kettenmann H, Kipnis J, Koyama R, Lemke G, Lynch M, Majewska A, Malcangio M, Malm T, Mancuso R, Masuda T, Matteoli M, McColl BW, Miron VE, Molofsky AV, Monje M, Mracsko E, Nadjar A, Neher JJ, Neniskyte U, Neumann H, Noda M, Peng B, Peri F, Perry VH, Popovich PG, Pridans C, Priller J, Prinz M, Ragozzino D, Ransohoff RM, Salter MW, Schaefer A, Schafer DP, Schwartz M, Simons M, Smith CJ, Streit WJ, Tay TL, Tsai L-H, Verkhratsky A, von Bernhardi R, Wake H, Wittamer V, Wolf SA, Wu L-J, Wyss-Coray T (2022) Microglia states and nomenclature: a field at its crossroads. Neuron 110(21):3458–3483. 10.1016/j.neuron.2022.10.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelvig DP, Pakkenberg H, Stark AK, Pakkenberg B (2008) Neocortical glial cell numbers in human brains. Neurobiol Aging 29(11):1754–1762. 10.1016/j.neurobiolaging.2007.04.013 [DOI] [PubMed] [Google Scholar]
- Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl U, Emerson M, Garner P, Holgate ST, Howells DW, Karp NA, Lazic SE, Lidster K, MacCallum CJ, Macleod M, Pearl EJ, Petersen OH, Rawle F, Reynolds P, Rooney K, Sena ES, Silberberg SD, Steckler T, Würbel H (2020) The ARRIVE guidelines 20: updated guidelines for reporting animal research. PLOS Biol. 10.1371/journal.pbio.300041032663221 [Google Scholar]
- Pérez-Treviño P, Velásquez M, García N (2020) Mechanisms of mitochondrial DNA escape and its relationship with different metabolic diseases. Biochimica et Biophysica Acta (BBA) Molecular Basis of Disease. 1866 (6): 165761. 10.1016/j.bbadis.2020.165761 [DOI] [PubMed]
- Perkins AE, Piazza MK, Deak T (2018) Stereological analysis of microglia in aged male and female fischer 344 rats in socially relevant brain regions. Neuroscience 377:40–52. 10.1016/j.neuroscience.2018.02.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perluigi M, Di Domenico F, Barone E, Butterfield DA (2021) mTOR in Alzheimer disease and its earlier stages: Links to oxidative damage in the progression of this dementing disorder. Free Radic Biol Med 169:382–396. 10.1016/j.freeradbiomed.2021.04.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pomilio C, Gorojod RM, Riudavets M, Vinuesa A, Presa J, Gregosa A, Bentivegna M, Alaimo A, Alcon SP, Sevlever G, Kotler ML, Beauquis J, Saravia F (2020) Microglial autophagy is impaired by prolonged exposure to β-amyloid peptides: evidence from experimental models and Alzheimer’s disease patients. Geroscience 42(2):613–632. 10.1007/s11357-020-00161-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qiu WQ, Ye Z, Kholodenko D, Seubert P, Selkoe DJ (1997) Degradation of amyloid β-protein by a metalloprotease secreted by microglia and other neural and non-neural cells. J Biol Chem 272(10):6641–6646. 10.1074/jbc.272.10.6641 [DOI] [PubMed] [Google Scholar]
- Rahman MM, Islam MR, Yamin M, Islam MM, Sarker MT, Meem AFK, Akter A, Emran TB, Cavalu S, Sharma R (2022) Emerging role of neuron-Glia in neurological disorders: at a glance. Oxid Med Cell Longev 2022:3201644. 10.1155/2022/3201644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raj DDA, Jaarsma D, Holtman IR, Olah M, Ferreira FM, Schaafsma W, Brouwer N, Meijer MM, de Waard MC, van der Pluijm I, Brandt R, Kreft KL, Laman JD, de Haan G, Biber KPH, Hoeijmakers JHJ, Eggen BJL, Boddeke HWGM (2014) Priming of microglia in a DNA-repair deficient model of accelerated aging. Neurobiol Aging 35(9):2147–2160. 10.1016/j.neurobiolaging.2014.03.025 [DOI] [PubMed] [Google Scholar]
- Reddaway J, Richardson PE, Bevan RJ, Stoneman J, Palombo M (2023) Microglial morphometric analysis: so many options, so little consistency. Front Neuroinform. 10.3389/fninf.2023.1211188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reemst K, Noctor SC, Lucassen PJ, Hol EM (2016) The indispensable roles of microglia and astrocytes during brain development. Front Human Neurosci. 10.3389/fnhum.2016.00566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rémond D, Shahar DR, Gille D, Pinto P, Kachal J, Peyron M-A, Santos CND, Walther B, Bordoni A, Dupont D, Tomás-Cobos L, Vergères G (2015) Understanding the gastrointestinal tract of the elderly to develop dietary solutions that prevent malnutrition. Oncotarget. 10.18632/oncotarget.4030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Réu P, Khosravi A, Bernard S, Mold JE, Salehpour M, Alkass K, Perl S, Tisdale J, Possnert G, Druid H, Frisén J (2017) The lifespan and turnover of microglia in the human brain. Cell Rep 20(4):779–784. 10.1016/j.celrep.2017.07.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rim C, You M-J, Nahm M, Kwon M-S (2024) Emerging role of senescent microglia in brain aging-related neurodegenerative diseases. Transl Neurodegen 13(1):10. 10.1186/s40035-024-00402-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ritzel RM, Patel AR, Pan S, Crapser J, Hammond M, Jellison E, McCullough LD (2015) Age- and location-related changes in microglial function. Neurobiol Aging 36(6):2153–2163. 10.1016/j.neurobiolaging.2015.02.016 [DOI] [PubMed] [Google Scholar]
- Ritzel RM, Lai Y-J, Crapser JD, Patel AR, Schrecengost A, Grenier JM, Mancini NS, Patrizz A, Jellison ER, Morales-Scheihing D, Venna VR, Kofler JK, Liu F, Verma R, McCullough LD (2018) Aging alters the immunological response to ischemic stroke. Acta Neuropathol 136(1):89–110. 10.1007/s00401-018-1859-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ritzel RM, Doran SJ, Glaser EP, Meadows VE, Faden AI, Stoica BA, Loane DJ (2019) Old age increases microglial senescence, exacerbates secondary neuroinflammation, and worsens neurological outcomes after acute traumatic brain injury in mice. Neurobiol Aging 77:194–206. 10.1016/j.neurobiolaging.2019.02.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson SM (2018) Improving nutrition to support healthy ageing: what are the opportunities for intervention? Proc Nutr Soc 77(3):257–264. 10.1017/s0029665117004037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolyan H, Scheffold A, Heinrich A, Begus-Nahrmann Y, Langkopf BH, Hölter SM, Vogt-Weisenhorn DM, Liss B, Wurst W, Lie DC, Thal DR, Biber K, Rudolph KL (2011) Telomere shortening reduces Alzheimer’s disease amyloid pathology in mice. Brain 134(7):2044–2056. 10.1093/brain/awr133 [DOI] [PubMed] [Google Scholar]
- Rudnicka E, Napierała P, Podfigurna A, Męczekalski B, Smolarczyk R, Grymowicz M (2020) The World Health Organization (WHO) approach to healthy ageing. Maturitas 139:6–11. 10.1016/j.maturitas.2020.05.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rush A, Weil C, Siminoff L, Griffin C, Paul CL, Mahadevan A, Sutherland G (2024) The experts speak: challenges in banking brain tissue for research. Biopreservation and Biobanking 22(2):179–184. 10.1089/bio.2024.29135.ajr [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryu JK, Nagai A, Kim J, Lee MC, McLarnon JG, Kim SU (2003) Microglial activation and cell death induced by the mitochondrial toxin 3-nitropropionic acid: in vitro and in vivo studies. Neurobiol Dis 12(2):121–132. 10.1016/S0969-9961(03)00002-0 [DOI] [PubMed] [Google Scholar]
- Salminen A, Kaarniranta K (2012) AMP-activated protein kinase (AMPK) controls the aging process via an integrated signaling network. Ageing Res Rev 11(2):230–241. 10.1016/j.arr.2011.12.005 [DOI] [PubMed] [Google Scholar]
- Sanders JL, Newman AB (2013) Telomere length in epidemiology: a biomarker of aging, age-related disease, both, or neither? Epidemiol Rev 35(1):112–131. 10.1093/epirev/mxs008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandhir R, Onyszchuk G, Berman NE (2008) Exacerbated glial response in the aged mouse hippocampus following controlled cortical impact injury. Exp Neurol 213(2):372–380. 10.1016/j.expneurol.2008.06.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sapieha P, Mallette FA (2018) Cellular senescence in postmitotic cells: beyond growth arrest. Trends Cell Biol 28(8):595–607. 10.1016/j.tcb.2018.03.003 [DOI] [PubMed] [Google Scholar]
- Scheffold A, Holtman IR, Dieni S, Brouwer N, Katz S-F, Jebaraj BMC, Kahle PJ, Hengerer B, Lechel A, Stilgenbauer S, Boddeke EWGM, Eggen BJL, Rudolph K-L, Biber K (2016) Telomere shortening leads to an acceleration of synucleinopathy and impaired microglia response in a genetic mouse model. Acta Neuropathol Commun 4(1):87. 10.1186/s40478-016-0364-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheiblich H, Bousset L, Schwartz S, Griep A, Latz E, Melki R, Heneka MT (2021) Microglial NLRP3 inflammasome activation upon TLR2 and TLR5 ligation by distinct α-synuclein assemblies. J Immunol 207(8):2143–2154. 10.4049/jimmunol.2100035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schöll M, Lockhart Samuel N, Schonhaut Daniel R, O’Neil James P, Janabi M, Ossenkoppele R, Baker Suzanne L, Vogel Jacob W, Faria J, Schwimmer Henry D, Rabinovici Gil D, Jagust William J (2016) PET imaging of Tau deposition in the aging human brain. Neuron 89(5):971–982. 10.1016/j.neuron.2016.01.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schonhoff AM, Figge DA, Williams GP, Jurkuvenaite A, Gallups NJ, Childers GM, Webster JM, Standaert DG, Goldman JE, Harms AS (2023) Border-associated macrophages mediate the neuroinflammatory response in an alpha-synuclein model of Parkinson disease. Nat Commun 14(1):3754. 10.1038/s41467-023-39060-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuitemaker A, van der Doef TF, Boellaard R, van der Flier WM, Yaqub M, Windhorst AD, Barkhof F, Jonker C, Kloet RW, Lammertsma AA, Scheltens P, van Berckel BNM (2012) Microglial activation in healthy aging. Neurobiol Aging 33(6):1067–1072. 10.1016/j.neurobiolaging.2010.09.016 [DOI] [PubMed] [Google Scholar]
- Sebastian Monasor L, Müller SA, Colombo AV, Tanrioever G, König J, Roth S, Liesz A, Berghofer A, Piechotta A, Prestel M, Saito T, Saido TC, Herms J, Willem M, Haass C, Lichtenthaler SF, Tahirovic S (2020) Fibrillar Aβ triggers microglial proteome alterations and dysfunction in Alzheimer mouse models. Elife 9:e54083. 10.7554/eLife.54083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah F, Hume SP, Pike VW, Ashworth S, McDermott J (1994) Synthesis of the enantiomers of [N-methyl-11C]PK 11195 and comparison of their behaviours as radioligands for PK binding sites in rats. Nucl Med Biol 21(4):573–581. 10.1016/0969-8051(94)90022-1 [DOI] [PubMed] [Google Scholar]
- Shahidehpour RK, Higdon RE, Crawford NG, Neltner JH, Ighodaro ET, Patel E, Price D, Nelson PT, Bachstetter AD (2021) Dystrophic microglia are associated with neurodegenerative disease and not healthy aging in the human brain. Neurobiol Aging 99:19–27. 10.1016/j.neurobiolaging.2020.12.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma R, Bisht P, Kesharwani A, Murti K, Kumar N (2024) Epigenetic modifications in Parkinson’s disease: a critical review. Eur J Pharmacol 975:176641. 10.1016/j.ejphar.2024.176641 [DOI] [PubMed] [Google Scholar]
- Sheffield LG, Berman NEJ (1998) Microglial expression of MHC class II increases in normal aging of nonhuman primates. Neurobiol Aging 19(1):47–55. 10.1016/S0197-4580(97)00168-1 [DOI] [PubMed] [Google Scholar]
- Shen Q-q, Jv X-h, Ma X-z, Li C, Liu L, Jia W-t, Qu L, Chen L-l, Xie J-x (2024) Cell senescence induced by toxic interaction between α-synuclein and iron precedes nigral dopaminergic neuron loss in a mouse model of Parkinson’s disease. Acta Pharmacol Sin 45(2):268–281. 10.1038/s41401-023-01153-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin HJ, Kim IS, Choi SG, Lee K, Park H, Shin J, Kim D, Beom J, Yi YY, Gupta DP, Song GJ, Chung W-S, Lee CJ, Kim DW (2024) Rejuvenating aged microglia by p16ink4a-siRNA-loaded nanoparticles increases amyloid-β clearance in animal models of Alzheimer’s disease. Mol Neurodegener 19(1):25. 10.1186/s13024-024-00715-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sierra A, Gottfried-Blackmore AC, McEwen BS, Bulloch K (2007) Microglia derived from aging mice exhibit an altered inflammatory profile. Glia 55(4):412–424. 10.1002/glia.20468 [DOI] [PubMed] [Google Scholar]
- Smith KL, Kassem MS, Clarke DJ, Kuligowski MP, Bedoya-Pérez MA, Todd SM, Lagopoulos J, Bennett MR, Arnold JC (2019) Microglial cell hyper-ramification and neuronal dendritic spine loss in the hippocampus and medial prefrontal cortex in a mouse model of PTSD. Brain Behav Immun 80:889–899. 10.1016/j.bbi.2019.05.042 [DOI] [PubMed] [Google Scholar]
- Solito E, Sastre M (2012) Microglia function in Alzheimer’s disease. Front Pharmacol. 10.3389/fphar.2012.00014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sołtys Z, Ziaja M, Pawliński R, Setkowicz Z, Janeczko K (2001) Morphology of reactive microglia in the injured cerebral cortex. Fractal analysis and complementary quantitative methods. J Neurosci Res 63(1):90–97 [DOI] [PubMed] [Google Scholar]
- Song L, Pei L, Yao S, Wu Y, Shang Y (2017) NLRP3 Inflammasome in Neurological Diseases, from Functions to Therapies. Frontiers in Cellular Neuroscience 11 [DOI] [PMC free article] [PubMed]
- Sorby-Adams AJ, Vink R, Turner RJ (2018) Large animal models of stroke and traumatic brain injury as translational tools. American J Physiol Regulat Integ Compar Physiol 315(2):R165–R190. 10.1152/ajpregu.00163.2017 [DOI] [PubMed] [Google Scholar]
- Squarzoni P, Oller G, Hoeffel G, Pont-Lezica L, Rostaing P, Low D, Bessis A, Ginhoux F, Garel S (2014) Microglia modulate wiring of the embryonic forebrain. Cell Rep 8(5):1271–1279. 10.1016/j.celrep.2014.07.042 [DOI] [PubMed] [Google Scholar]
- Stallone G, Infante B, Prisciandaro C, Grandaliano G (2019) mTOR and aging: an old fashioned dress. Int J Mol Sci. 10.3390/ijms20112774 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stillman JM, Mendes Lopes F, Lin JP, Hu K, Reich DS, Schafer DP (2023) Lipofuscin-like autofluorescence within microglia and its impact on studying microglial engulfment. Nat Commun 14(1):7060. 10.1038/s41467-023-42809-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- St-Pierre M-K, Carrier M, González Ibáñez F, Šimončičová E, Wallman M-J, Vallières L, Parent M, Tremblay M-È (2022) Ultrastructural characterization of dark microglia during aging in a mouse model of Alzheimer’s disease pathology and in human post-mortem brain samples. J Neuroinflammation 19(1):235. 10.1186/s12974-022-02595-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stranks AJ, Hansen AL, Panse I, Mortensen M, Ferguson DJ, Puleston DJ, Shenderov K, Watson AS, Veldhoen M, Phadwal K, Cerundolo V, Simon AK (2015) Autophagy controls acquisition of aging features in macrophages. J Innate Immun 7(4):375–391. 10.1159/000370112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stratoulias V, Venero JL, Tremblay M, Joseph B (2019) Microglial subtypes: diversity within the microglial community. EMBO J. 10.15252/embj.2019101997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streit WJ, Sammons NW, Kuhns AJ, Sparks DL (2004) Dystrophic microglia in the aging human brain. Glia 45(2):208–212. 10.1002/glia.10319 [DOI] [PubMed] [Google Scholar]
- Streit WJ, Braak H, Xue Q-S, Bechmann I (2009) Dystrophic (senescent) rather than activated microglial cells are associated with tau pathology and likely precede neurodegeneration in Alzheimer’s disease. Acta Neuropathol 118(4):475–485. 10.1007/s00401-009-0556-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streit WJ, Braak H, Del Tredici K, Leyh J, Lier J, Khoshbouei H, Eisenlöffel C, Müller W, Bechmann I (2018) Microglial activation occurs late during preclinical Alzheimer’s disease. Glia 66(12):2550–2562. 10.1002/glia.23510 [DOI] [PubMed] [Google Scholar]
- Sun M, McDonald SJ, Brady RD, Collins-Praino L, Yamakawa GR, Monif M, O’Brien TJ, Cloud GC, Sobey CG, Mychasiuk R, Loane DJ, Shultz SR (2020) The need to incorporate aged animals into the preclinical modeling of neurological conditions. Neurosci Biobehav Rev 109:114–128. 10.1016/j.neubiorev.2019.12.027 [DOI] [PubMed] [Google Scholar]
- Taetzsch T, Benusa S, Levesque S, Mumaw CL, Block ML (2019) Loss of NF-κB p50 function synergistically augments microglial priming in the middle-aged brain. J Neuroinflammation 16(1):60. 10.1186/s12974-019-1446-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tam WY, Ma CHE (2014) Bipolar/rod-shaped microglia are proliferating microglia with distinct M1/M2 phenotypes. Sci Rep 4(1):7279. 10.1038/srep07279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang Y, Li T, Li J, Yang J, Liu H, Zhang XJ, Le W (2014) Jmjd3 is essential for the epigenetic modulation of microglia phenotypes in the immune pathogenesis of Parkinson’s disease. Cell Death Differ 21(3):369–380. 10.1038/cdd.2013.159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tansey MG, Wallings RL, Houser MC, Herrick MK, Keating CE, Joers V (2022) Inflammation and immune dysfunction in Parkinson disease. Nat Rev Immunol 22(11):657–673. 10.1038/s41577-022-00684-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thevaranjan N, Puchta A, Schulz C, Naidoo A, Szamosi JC, Verschoor CP, Loukov D, Schenck LP, Jury J, Foley KP, Schertzer JD, Larché MJ, Davidson DJ, Verdú EF, Surette MG, Bowdish DME (2017) Age-associated microbial dysbiosis promotes intestinal permeability, systemic inflammation, and macrophage dysfunction. Cell Host Microbe 21(4):455-466.e454. 10.1016/j.chom.2017.03.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas AL, Lehn MA, Janssen EM, Hildeman DA, Chougnet CA (2022) Naturally-aged microglia exhibit phagocytic dysfunction accompanied by gene expression changes reflective of underlying neurologic disease. Sci Rep 12(1):19471. 10.1038/s41598-022-21920-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueno M, Yamashita T (2014) Bidirectional tuning of microglia in the developing brain: from neurogenesis to neural circuit formation. Curr Opin Neurobiol 27:8–15. 10.1016/j.conb.2014.02.004 [DOI] [PubMed] [Google Scholar]
- Vaiserman A, Krasnienkov D (2021) Telomere length as a marker of biological age: state-of-the-art, open issues, and future perspectives. Front Gene. 10.3389/fgene.2020.630186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Heemst D (2010) Insulin, IGF-1 and longevity. Aging Dis 1(2):147–157 [PMC free article] [PubMed] [Google Scholar]
- Vellai T, Takacs-Vellai K, Zhang Y, Kovacs AL, Orosz L, Müller F (2003) Influence of TOR kinase on lifespan in C elegans. Nature 426:620–620. 10.1038/426620a [DOI] [PubMed] [Google Scholar]
- Vellingiri B, Balasubramani K, Iyer M, Raj N, Elangovan A, Song K, Yeo H-C, Jayakumar N, Kinoshita M, Thangarasu R, Narayanasamy A, Dayem AA, Prajapati VK, Gopalakrishnan AV, Cho S-G (2023) Role of telomeres and telomerase in Parkinson’s disease—a new theranostics? Adv Biol 7(12):2300097. 10.1002/adbi.202300097 [DOI] [PubMed] [Google Scholar]
- Wagner LK, Gilling KE, Schormann E, Kloetzel PM, Heppner FL, Krüger E, Prokop S (2017) Immunoproteasome deficiency alters microglial cytokine response and improves cognitive deficits in Alzheimer’s disease-like APPPS1 mice. Acta Neuropathol Commun 5(1):52. 10.1186/s40478-017-0453-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waller R, Baxter L, Fillingham DJ, Coelho S, Pozo JM, Mozumder M, Frangi AF, Ince PG, Simpson JE, Highley JR (2019) Iba-1-/CD68+ microglia are a prominent feature of age-associated deep subcortical white matter lesions. PLoS ONE 14(1):e0210888. 10.1371/journal.pone.0210888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang ZX, Li YL, Pu JL, Zhang BR (2023) DNA damage-mediated neurotoxicity in Parkinson’s disease. Int J Mol Sci. 10.3390/ijms24076313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasserman JK, Yang H, Schlichter LC (2008) Glial responses, neuron death and lesion resolution after intracerebral hemorrhage in young vs aged rats. European J Neurosci 28(7):1316–1328. 10.1111/j.1460-9568.2008.06442.x [DOI] [PubMed] [Google Scholar]
- Wendimu MY, Hooks SB (2022) Microglia phenotypes in aging and neurodegenerative diseases. Cells. 10.3390/cells11132091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams CG, Lee HJ, Asatsuma T, Vento-Tormo R, Haque A (2022) An introduction to spatial transcriptomics for biomedical research. Genome Med 14(1):68. 10.1186/s13073-022-01075-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong-Guerra M, Calfio C, Maccioni RB, Rojo LE (2023) Revisiting the neuroinflammation hypothesis in Alzheimer’s disease: a focus on the druggability of current targets. Front Pharmacol. 10.3389/fphar.2023.1161850 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu W, Boyd RM, Tree MO, Samkari F, Zhao L (2019) Mitochondrial transcription factor a promotes DNA strand cleavage at abasic sites. Proc Natl Acad Sci 116(36):17792–17799. 10.1073/pnas.1911252116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue L, Fu Y, Gao X, Feng G, Qian S, Wei L, Li L, Zhuo C, Zhang H, Tian M (2024) [18F]FDG PET integrated with structural MRI for accurate brain age prediction. Eur J Nucl Med Mol Imaging 51(12):3617–3629. 10.1007/s00259-024-06784-w [DOI] [PubMed] [Google Scholar]
- Youm Y-H, Grant Ryan W, McCabe Laura R, Albarado Diana C, Nguyen Kim Y, Ravussin A, Pistell P, Newman S, Carter R, Laque A, Münzberg H, Rosen Clifford J, Ingram Donald K, Salbaum JM, Dixit Vishwa D (2013) Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab 18(4):519–532. 10.1016/j.cmet.2013.09.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young K, Morrison H (2018) Quantifying microglia morphology from photomicrographs of immunohistochemistry prepared tissue using imageJ. J Vis Exp. 10.3791/57648 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu H-M, Zhao Y-M, Luo X-G, Feng Y, Ren Y, Shang H, He Z-Y, Luo X-M, Chen S-D, Wang X-Y (2012) Repeated lipopolysaccharide stimulation induces cellular senescence in BV2 cells. NeuroImmunoModulation 19(2):131–136. 10.1159/000330254 [DOI] [PubMed] [Google Scholar]
- Zhang Y, Sloan SA, Clarke LE, Caneda C, Plaza CA, Blumenthal PD, Vogel H, Steinberg GK, Edwards MS, Li G, Duncan JA 3rd, Cheshier SH, Shuer LM, Chang EF, Grant GA, Gephart MG, Barres BA (2016) Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 89(1):37–53. 10.1016/j.neuron.2015.11.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Heng Y, Kooistra SM, van Weering HRJ, Brummer ML, Gerrits E, Wesseling EM, Brouwer N, Nijboer TW, Dubbelaar ML, Boddeke EWGM, Eggen BJL (2021) Intrinsic DNA damage repair deficiency results in progressive microglia loss and replacement. Glia 69(3):729–745. 10.1002/glia.23925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, Xiao D, Mao Q, Xia H (2023) Role of neuroinflammation in neurodegeneration development. Signal Transduct Target Ther 8(1):267. 10.1038/s41392-023-01486-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao J, Huai J (2023) Role of primary aging hallmarks in Alzheimer´s disease. Theranostics 13(1):197–230. 10.7150/thno.79535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H, Zhao X, Cao X, Wu G (2012) Age-Dependent Neuroimmune Modulation of IGF-1R in the Traumatic Mice. Immunity & Ageing 9(1):12. 10.1186/1742-4933-9-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu L, Dong C, Sun C, Ma R, Yang D, Zhu H, Xu J (2015) Rejuvenation of MPTP-induced human neural precursor cell senescence by activating autophagy. Biochem Biophys Res Commun 464(2):526–533. 10.1016/j.bbrc.2015.06.174 [DOI] [PubMed] [Google Scholar]
- Ziebell JM, Taylor SE, Cao T, Harrison JL, Lifshitz J (2012) Rod microglia: elongation, alignment, and coupling to form trains across the somatosensory cortex after experimental diffuse brain injury. J Neuroinflammation 9:247. 10.1186/1742-2094-9-247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziebell JM, Adelson PD, Lifshitz J (2015) Microglia: dismantling and rebuilding circuits after acute neurological injury. Metab Brain Dis 30(2):393–400. 10.1007/s11011-014-9539-y [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.