Abstract
Computed tomography coronary angiography provides a non-invasive evaluation of coronary artery disease that includes phenotyping of atherosclerotic plaques and the surrounding perivascular adipose tissue (PVAT). Image analysis techniques have been developed to quantify atherosclerotic plaque burden and morphology as well as the associated PVAT attenuation, and emerging radiomic approaches can add further contextual information. PVAT attenuation might provide a novel measure of vascular health that could be indicative of the pathogenetic processes implicated in atherosclerosis such as inflammation, fibrosis or increased vascularity. Bidirectional signalling between the coronary artery and adjacent PVAT has been hypothesized to contribute to coronary artery disease progression and provide a potential novel measure of the risk of future cardiovascular events. However, despite the development of more advanced radiomic and artificial intelligence-based algorithms, studies involving large datasets suggest that the measurement of PVAT attenuation contributes only modest additional predictive discrimination to standard cardiovascular risk scores. In this Review, we explore the pathobiology of coronary atherosclerotic plaques and PVAT, describe their phenotyping with computed tomography coronary angiography, and discuss potential future applications in clinical risk prediction and patient management.
Introduction
Atherosclerosis is a leading cause of cardiovascular mortality worldwide and a major public health concern. Atherosclerosis is a systemic disease with a complex pathophysiology1. Typically, atherosclerosis is a continuous process that can start in childhood and is affected by multiple modifiable and non-modifiable risk factors2,3. Rapid progression and disruption of an atherosclerotic plaque can result in plaque rupture or erosion, leading to acute occlusion of vessels in the heart (myocardial infarction), brain (ischaemic stroke) or lower extremities (acute limb ischaemia).
Although contemporary clinical practice is primarily focused on obstructive stenoses of the coronaries, the majority (up to 80%) of clinical coronary atherothrombotic events are caused by non-obstructive lesions, which are more prevalent than obstructive lesions4. Indeed, retrospective studies have consistently shown that the presence of severe stenoses does not add to the risk of adverse coronary events beyond assessments of plaque burden5–9. Although 90% of atherosclerotic plaque ruptures are clinically silent, identifying plaques at the highest risk of rupture and their overall burden could improve risk stratification at the patient level and guide medical management10. With a growing armamentarium of medical therapies, non-invasive identification of high-risk lesions remains an unmet clinical need11–13.
Cardiovascular imaging and advanced techniques for quantitative analysis allow the non-invasive evaluation of coronary atherosclerosis, including the plaque and its surrounding perivascular adipose tissue (PVAT)14. Phenotyping of atherosclerotic plaque includes characterization of the plaque burden and morphology as well as the PVAT volume and PVAT signal attenuation on computed tomography (CT)15. Vascular inflammation is one of the contributors to atherosclerosis but its complex relationship with clinical risk factors and the magnitude of the effect on the vasculature remain to be precisely defined16. Bidirectional signalling occurs between the blood vessel and the adjacent PVAT17. In this Review, we explore the pathobiology of coronary atherosclerotic plaques and PVAT, describe their phenotyping with CT coronary angiography (CTCA), and discuss potential future applications in clinical risk prediction and patient management.
Definition of PVAT
Epicardial adipose tissue and PVAT are fat depots around the heart and arteries, respectively, and differ in their specific anatomical locations and surrounding structures. Epicardial adipose tissue is located in the visceral layer of the pericardium, covers the surface of the heart and receives its blood supply from the coronary circulation18. PVAT surrounds most arteries, including the coronary arteries, although PVAT is not present around the cerebral or pulmonary arteries. PVAT is defined as being within a radial distance from the outer wall of the vessel that is equal to the vessel diameter and can be considered to be the fourth layer of the artery wall (also referred to as the tunica adiposa)19–21 (Fig. 1).
Fig. 1 |. Anatomy and definitions of thoracic fat.
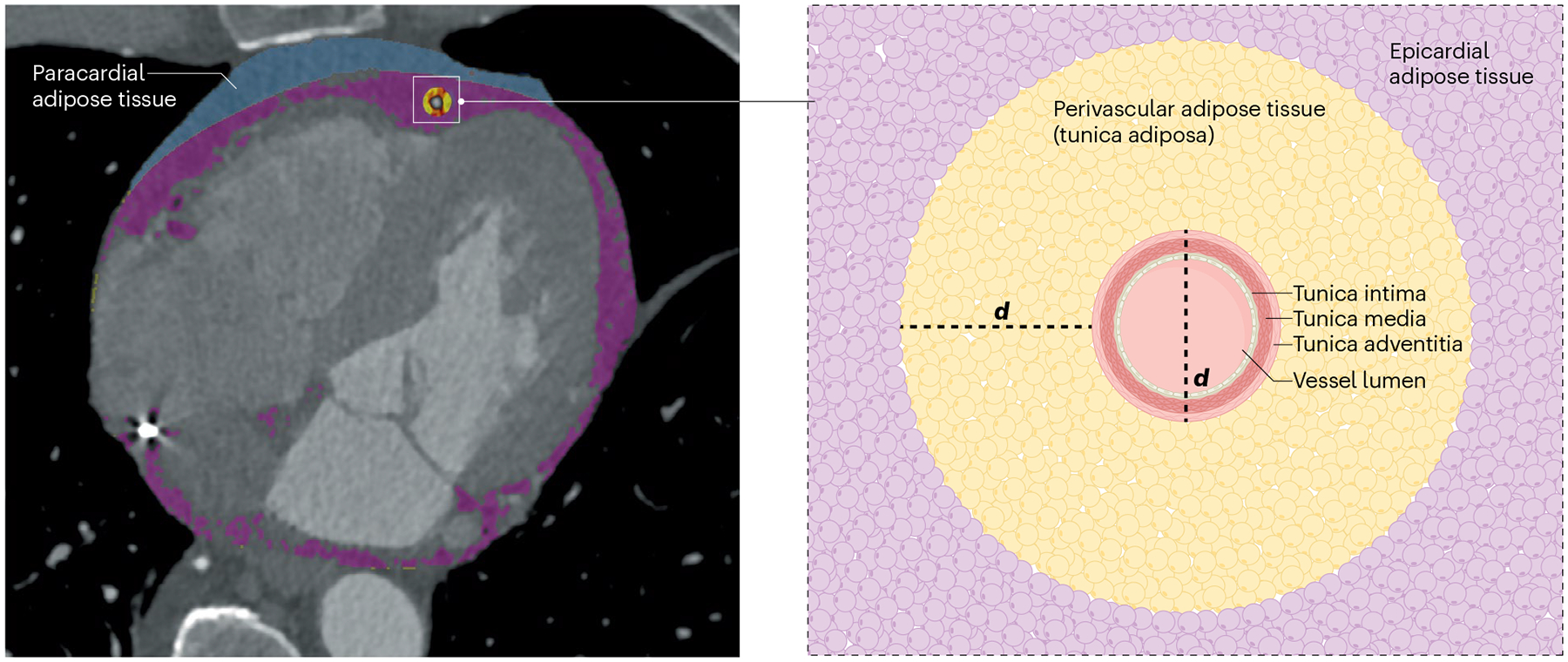
Paracardial adipose tissue (blue), epicardial adipose tissue (purple) and perivascular adipose tissue (yellow) are thoracic fat depots that can be measured by cardiac computed tomography. Epicardial adipose tissue is located between the myocardium and the visceral layer of the pericardium. Pericardial adipose tissue includes the paracardial and epicardial fat together. Perivascular adipose tissue is defined as being within a radial distance from the outer vessel wall that is equal to vessel diameter (d) and can be considered to be the fourth layer of the artery wall (also referred to as the tunica adiposa).
Adult human PVAT consists primarily of white adipose tissue, with brown adipocytes being only rarely observed in PVAT22. White and brown adipose tissues have different morphologies and secretomes, reflecting their distinct metabolic roles. White adipocytes store energy as triglycerides and have lower metabolic activity than brown adipocytes. By contrast, brown adipocytes are more metabolically active, specialize in energy dissipation through heat production and are more abundant in humans at younger ages23. However, in response to various physiological influences, such as diet24, exercise25 and medication26, white adipocytes in PVAT and adipose depots can undergo browning, with further differentiation into beige adipocytes that have a similar metabolic activity to that of brown adipocytes27 (Fig. 2). Browning of white adipocytes is a complex process regulated by various agents (including proteins and microRNAs) that affect a variety of pathways, depending on the triggering factor28,29.
Fig. 2 |. The PVAT secretome: anti-inflammatory and pro-inflammatory profiles.

a, Under physiological conditions, the secretome of the beige adipocytes in the perivascular adipose tissue (PVAT) includes anti-inflammatory and vasorelaxant compounds. b, Various risk factors, including ageing, obesity, smoking and comorbidities such as diabetes mellitus, result in whitening of adipocytes in the PVAT. This process contributes to the progression of atherosclerosis because the secretome of the PVAT changes to include pro-inflammatory and vasoconstrictive compounds.
Sex-specific differences in PVAT
The differences in cardiovascular risk between men and women are multifactorial, and the ectopic fat depots are potential contributors. In the Framingham Heart Study30, women had a higher volume of thoracic periaortic fat than men. Additionally, postmenopausal women were more likely than men of the same age to accumulate excess thoracic fat30. The declining oestrogen level after menopause was also associated with an increased volume of pericardial adipose tissue31–33. Several studies have used CTCA to compare the characteristics of PVAT between men and women, consistently showing increased attenuation in men, which is indicative of greater adipocyte differentiation and lower lipid content34,35. However, the absolute values of PVAT attenuation varied significantly between the cohorts, underscoring the confounding effect of the scan protocol. Interestingly, higher PVAT attenuation was strongly associated with measures of atherosclerotic plaque vulnerability such as thin-cap fibroatheroma, macrophage accumulation and greater lipid index on optical coherence tomography in women but not in men36.
Atherosclerosis and PVAT: insights from preclinical studies
Atherogenesis
Atherosclerosis starts with dysfunction of the structures and pathways that are otherwise responsible for maintaining vascular homeostasis, and endothelial dysfunction is a hallmark of disease initiation37,38. Various stimuli adversely affect endothelial function, primarily through inflammation and oxidative stress. Disrupted vascular homeostasis promotes atherosclerosis by increasing leukocyte adhesion to endothelial cells, amplifying cytokine secretion and increasing permeability of the vessel wall to lipoprotein particles39. PVAT affects these processes through paracrine signalling and often has opposing effects, depending on the different disease substrates. In healthy individuals, PVAT releases vasorelaxant molecules such as adiponectin, hydrogen sulfide and palmitic acid methyl ester40. However, obesity and insulin resistance can decrease the production of these mediators by the adipocytes41–43. Similarly, decreased production of major atheroprotective neurotransmitters, such as nitric oxide and hydrogen sulfide, occurs in hypertrophic adipocytes of PVAT in mice with obesity44,45 (Fig. 3). Obesity, diabetes mellitus or smoking disturb the redox balance in PVAT, resulting in the overproduction of reactive oxygen species46.
Fig. 3 |. Bidirectional interactions between PVAT and the vessel wall in atherosclerosis.

The vessel wall and the perivascular adipose tissue (PVAT) are hypothesized to interact during the formation of atherosclerotic plaques. Adipokines released by the PVAT can affect atherosclerotic plaque formation (outside-to-inside signalling). The same adipocytes might also change in response to the local inflammation of the diseased vessel segment (inside-to-outside signalling). a, Various factors impair endothelial function, primarily through inflammation and oxidative stress. Decreased production of major atheroprotective neurotransmitters, such as nitric oxide and hydrogen sulfide, by endothelial cells is a hallmark of early atherosclerosis. b, Atherosclerotic plaques form through the accumulation of LDL particles in the arterial intima, the infiltration of inflammatory immune cells into the artery wall and the migration of vascular smooth muscle cells to the intima. These cells take up lipoproteins and transform into foam cells. In response to changes in the vessel wall, PVAT starts secreting pro-inflammatory cytokines that promote further infiltration of foam cell precursors into the intimal layer. c, Atherosclerosis progression leads to the formation of a vulnerable plaque with a large thrombogenic necrotic core covered by a thin layer of fibrous tissue. Excess accumulation of necrotic cells and cholesterol crystals contributes to growth of the necrotic core. Moreover, a pro-inflammatory environment downregulates the expression of genes responsible for collagen synthesis by vascular smooth muscle cells, contributing to plaque vulnerability and decreasing the thickness of the fibrous cap.
Atherosclerotic plaque formation involves the accumulation in the arterial intima of lipoprotein–cholesterol particles from the plasma and the recruitment of pro-inflammatory immune cells into the artery wall. Dysfunctional endothelium allows the infiltration of apolipoprotein B-containing lipoproteins from the plasma into the intima, where they undergo oxidation47–50. These modified LDL-cholesterol particles subsequently trigger local inflammation51, resulting in monocyte recruitment to the arterial wall52,53 and vascular smooth muscle cell migration to the intima54 as well as the transition of these cells into foam cells following the uptake of lipoproteins55–57. In parallel with atherosclerotic plaque development, the phenotype of PVAT also changes, with the adipose tissue becoming a source of pro-inflammatory cytokines, which propagates the infiltration of the progenitors of foam cells into the intimal space58. Post-mortem evaluations of human samples show an increased presence of immune cells — especially macrophages and B lymphocytes — in the PVAT that surrounds sections of the coronary artery with atherosclerosis59,60. Infiltrating macrophages produce cytokines such as IL-6, IFNγ and tumour necrosis factor61. Macrophages are also involved in T cell activation through antigen presentation62. In mice, T cell-dependent inflammation in the adventitia correlates with atherosclerotic lesion size, and the depletion of these cells can prevent atherosclerosis development63,64. Finally, the role of B cells in atherosclerosis is not fully understood but they might contribute to atherosclerotic plaque formation through antigen presentation and the production of antibodies65.
Atherosclerotic plaque development continues with increasing subendothelial deposition of lipids66. Initial fatty streaks transform slowly into lipid-rich necrotic cores, and medial vascular smooth muscle cells migrate to the intima of the artery to form a fibrous cap, which protects the potentially prothrombotic load of the atherosclerotic plaque from rupture67,68. Fibrous cap formation is mediated by growth factors released by foam cells and PVAT69,70. Inflamed PVAT can potentiate fibrous cap formation through the release of IL-6, leptin and tumour necrosis factor71. Furthermore, in vivo studies in pigs have demonstrated that the chronic application of inflammatory cytokines, such as IL-1β or CC-motif chemokine 2, to the adventitia of the coronary arteries induces fibrous cap formation and vascular remodelling72,73. By contrast, matrix metalloproteinases released after cell death in the necrotic core reduce the size of the fibrous cap74. Depending on the inflammatory status of the local environment and other factors, atherosclerotic plaques either progress and become high-risk, vulnerable lesions or regress and are associated with a low risk of clinical sequelae. In advanced lesions, increased oxidative stress75, activation of receptors involved in cell death signalling, inhibition of survival pathways and nutrient deprivation76 result in macrophage death and impaired efferocytosis77. Excess accumulation of apoptotic cells and cholesterol crystals78 contributes to necrotic core growth and atherosclerotic plaque vulnerability74,79,80.
A typical high-risk atherosclerotic plaque is characterized by a large thrombogenic necrotic core covered by a thin layer of fibrous tissue (the fibrous cap)81. Proteolysis of the collagen forming the extracellular matrix of fibrous caps has been associated with the macrophage secretome through mechanical tests on strips of human aorta, with a good correlation between higher macrophage density and greater extensibility and decreased maximum stress of the fibrous cap82. Additionally, in mice with obesity, exosomes secreted by the subcutaneous and visceral adipose tissue were shown to induce pro-inflammatory macrophage polarization, thereby promoting a pro-inflammatory environment83. T cells infiltrate the adjacent PVAT and become an additional source of chemokines that create a positive feedback mechanism for increased recruitment of monocytes and T cells84–86. Moreover, IFNγ secreted by activated T cells downregulates the expression of genes responsible for collagen synthesis in vascular smooth muscle cells87. In mouse femoral arteries, macrophages in the media and adventitia (but not in the intima) seem to be important for extensive atherosclerotic remodelling via extracellular matrix degradation and inhibition of vascular smooth muscle cells88.
Bidirectional signalling
Given its direct proximity to and continuity with the coronary wall adventitia, PVAT can interact with the vasculature in a bidirectional manner through paracrine and vasocrine signalling pathways (Fig. 3). PVAT secretes bioactive components that can diffuse directly into the vessel wall, producing phenotypic changes in a process known as outside-to-inside signalling89,90. Adipocytes are a source of adipokines, neurotransmitters, reactive oxygen species and fatty acid metabolites. Their secretome is further complemented by cytokines released from immune cells infiltrating from the vasa vasorum91. Under physiological conditions, the secretome of PVAT has anti-inflammatory and anti-constrictive effects on the vessel wall92,93. However, chronic diseases can negatively affect the homeostatic function of the PVAT and change the secretome to a pro-inflammatory and vasoconstrictive state that ultimately promotes atherosclerosis94,95.
Conversely, PVAT can function as a sensor of vascular oxidation and inflammation. The composition and biology of local PVAT surrounding diseased coronary artery segments can change in response to pro-inflammatory cytokines and lipid peroxidase products released by the diseased vessel segment, a concept known as inside-to-outside signalling18,89,96. Histology studies have shown that oxidative stress increases the expression of ADIPOQ, which encodes adiponectin, in pericoronary adipocytes97. The potential effect of the vessel wall on adjacent adipose tissue was investigated by comparing the CT signal attenuation and secretome of epicardial fat depots from regions of human coronary artery with myocardial bridging compared with proximal, non-bridging regions98. The analysis showed a higher concentration of pro-inflammatory cytokines in the non-bridging segment of a coronary artery that had direct contact with the PVAT99,100. Production of pro-inflammatory adipokines might promote expansion of the vasa vasorum and increase the local concentration of T cells and macrophages that augment the inflammatory environment through a positive feedback loop101. Therefore, local perivascular inflammation might indicate atherosclerotic plaque instability, with increased release of atheromatous emboli and microcirculatory dysfunction that negatively affect long-term outcomes after cardiovascular events91. However, much is unknown about these effects in humans because of the low availability of fresh coronary artery tissue with its associated PVAT. Indeed, the aforementioned studies have primarily explored these associations in mice.
Comprehensive evaluation by CTCA
A single imaging modality that assesses both atherosclerosis and the PVAT could provide a comprehensive evaluation of the coronary arteries. Of the available clinical imaging techniques, CTCA has the best spatial and temporal resolution to visualize and phenotype both coronary atherosclerosis and the PVAT. With CTCA, tissue characterization and phenotyping are based on coronary artery segmentation and differences in X-ray attenuation. For atherosclerotic plaques, X-ray photon attenuation provides a method for differentiating between calcified and non-calcified fibrotic plaques and lipid-rich, potentially necrotic plaques (low attenuation: <45 Hounsfield units (HU))102. By contrast, because of its very low tissue density, coronary PVAT has a much lower attenuation on CT, with typical values ranging from −30 HU to −190 HU, with mean coronary PVAT attenuation ranging between −60 HU and −80 HU (ref. 89).
Atherosclerotic plaque phenotyping
Over the past two decades, the coronary artery calcium (CAC) score measured from non-contrast CT scans has been used to evaluate coronary atherosclerotic plaques103. Although the CAC score identifies the presence and burden of calcified atherosclerotic plaques and is predictive of adverse cardiovascular events in asymptomatic individuals, the score does not provide information on luminal stenosis, the extent of atherosclerosis or plaque composition104. Indeed, the CAC score misses atherosclerosis in a substantial proportion of individuals, with non-calcified atherosclerosis being present in approximately 16% of individuals who have a CAC score of zero105,106. By detecting both calcified and non-calcified atherosclerotic plaques, CTCA addresses these shortcomings and provides a more comprehensive and in-depth phenotyping of coronary atheroma.
The presence of coronary atherosclerosis and the number of coronary vessels affected are established and robust predictors of future coronary events107,108. Certain qualitative features of the atherosclerotic plaque, such as positive remodelling, lack of calcification, spotty calcification and the napkin-ring sign, are indicative of a higher risk of adverse coronary events109,110. This qualitative assessment is complemented by volumetric and attenuation-based quantitative analyses, which provide measures of total, calcified, non-calcified, and low-attenuation plaque volume and burden. These quantitative measures have low interobserver variation, high scan–rescan reproducibility and close correlation with assessment by invasive imaging such as intravascular ultrasonography102,111. In a case–control study, quantitative plaque assessment, including measurement of low-attenuation plaque, outperformed assessments of stenosis severity or overall plaque burden for the identification of patients at high risk of adverse coronary events112. In a post hoc analysis of the SCOT-HEART trial9 involving patients with stable chest pain, low-attenuation plaque burden was predictive of fatal or non-fatal myocardial infarction independently of clinical risk score, CAC score and stenosis severity. Patients with a low-attenuation plaque burden of >4% were nearly five times more likely to have a subsequent myocardial infarction than patients with a low-attenuation plaque burden of ≤4%9. Although women with stable chest pain generally had less atherosclerotic plaque of any type than men, low-attenuation plaque burden was still predictive of an increased risk of adverse outcomes regardless of sex113. A novel deep learning system for automated quantification of atherosclerotic plaques has been developed and externally validated using the SCOT-HEART trial population114. Deep learning-based quantification of atherosclerotic plaque volume could be performed in <6 s and was predictive of the risk of myocardial infarction114.
Atherosclerotic plaques can be serially assessed with repeat scanning to evaluate disease progression (Fig. 4). In a study of 449 patients who underwent serial CTCA scans, acute coronary syndrome occurred in 14.3% of those with evidence of atherosclerotic plaque progression but in only 0.3% of those without progression115. However, a remaining challenge is to predict those in whom coronary artery atherosclerotic plaque will progress and to intervene before major adverse cardiac events (MACE) occur, without the need for repeated imaging.
Fig. 4 |. Associations between 18F-NaF uptake and PVAT in coronary artery disease.
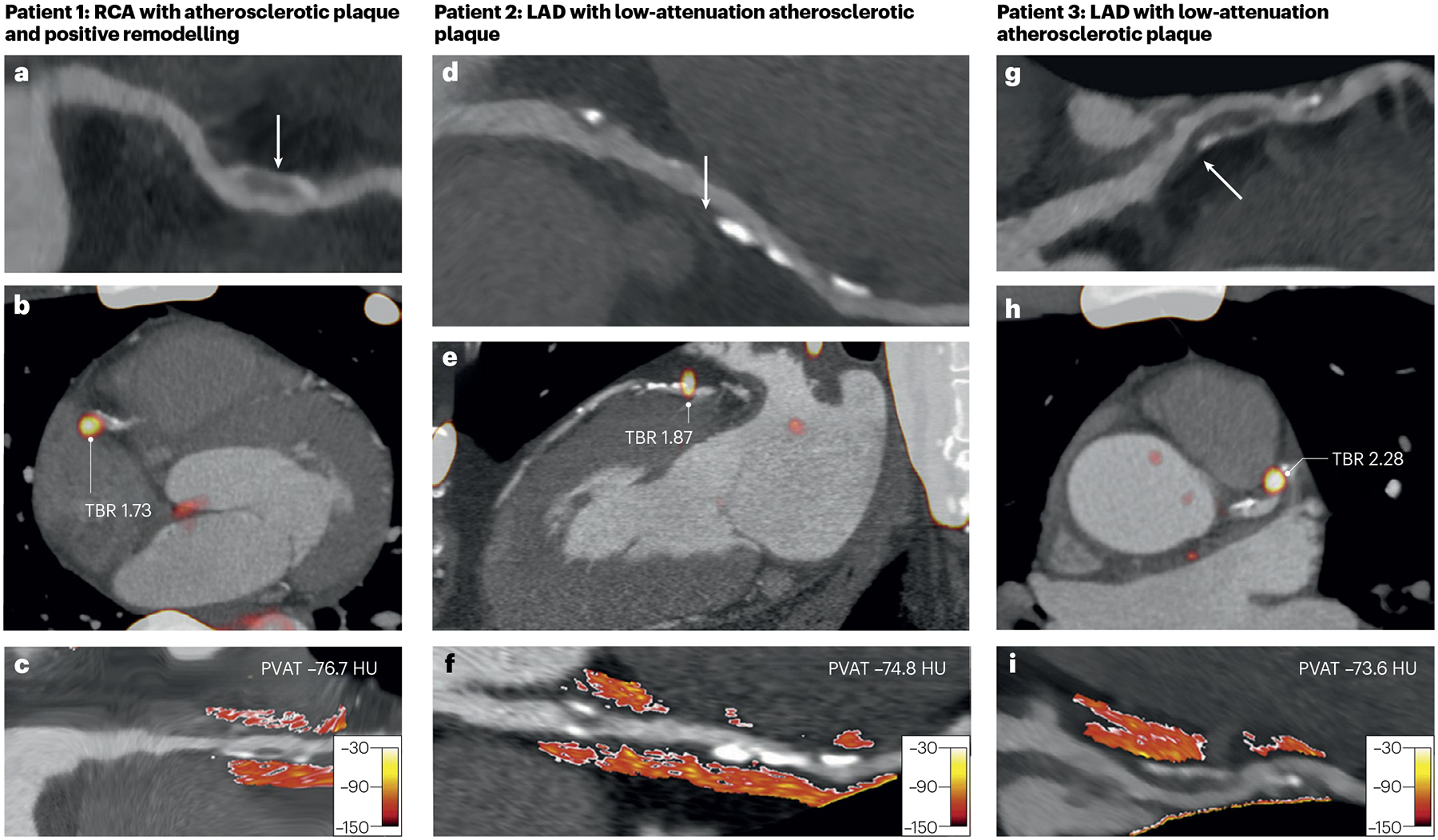
Increased atherosclerotic plaque vulnerability measured by 18F-NaF uptake (a marker of active calcification) on positron emission tomography and by attenuation of perivascular adipose tissue (PVAT) on computed tomography coronary angiography. Patient 1 is a man aged 53 years with right coronary artery (RCA) atherosclerotic plaque with positive remodelling (arrow) (part a), focal 18F-NaF uptake with an elevated target-to-background ratio (TBR) of 1.73 (part b), and increased PVAT attenuation (mean PVAT density −76.7 Hounsfield units (HU)) (part c). Patient 2 is a man aged 66 years with left anterior descending coronary artery (LAD) atherosclerotic plaque with low attenuation (arrow) (part d), focal 18F-NaF uptake with an elevated TBR of 1.87 (part e) and increased PVAT attenuation (mean PVAT density −74.8 HU) (part f). Patient 3 is a man aged 54 years with LAD atherosclerotic plaque with low attenuation (arrow) (part g), focal 18F-NaF uptake with an elevated TBR of 2.28 (part h) and increased PVAT attenuation (mean PVAT density −73.6 HU) (part i). Adapted with permission from ref. 133, Elsevier.
PVAT phenotyping
Changes in the lipid content of PVAT can be detected by alterations in its attenuation on CT. The CT attenuation of PVAT with normal, large, lipid-rich adipocytes is approximately −190 HU, whereas the CT attenuation of PVAT with denser, more aqueous adipocytes rises towards −30 HU (ref. 89). In practice, the CT attenuation of PVAT is measured by calculating the average attenuation within a circumferential volume of interest in the PVAT surrounding each of the coronary arteries14,116. Several measures at increasing radial distance from the outer wall of the coronary artery can be made and, under physiological conditions, CT attenuation shows a gradient from less negative (less lipophilic content) closer to the vessel wall to more negative (more lipophilic content) further away from the vessel wall89.
The Hounsfield scale of CT attenuation inherently limits PVAT measurements by CT because various technical factors, such as the reconstruction algorithm, CT model variations and tube voltage, can influence measures of PVAT attenuation117,118. Indeed, increasing the tube voltage from 70 kV to 120 kV can change the PVAT attenuation by 10%119, which restricts between-scan comparability, especially when using different scan protocols. This limitation is important given the relatively small differences in PVAT attenuation observed between patients with or without atherosclerotic disease as well as those with or without adverse clinical outcomes: usually a mean difference of only 4–6 HU (refs. 120–122). Test–retest variation in PVAT attenuation also remains a concern because no studies have been published. In addition, the effects of atherosclerotic plaque artefacts, such as those caused by extensive calcium, or the presence of intracoronary stents should be considered as potential confounders when measuring PVAT attenuation123.
Beyond these technical considerations, other factors can further complicate the accurate and consistent measurement of PVAT. Motion artefacts can affect image quality and the accuracy of PVAT measurements. Differences in intraluminal contrast enhancement can alter the apparent density of PVAT, and sites closer to the aorta might be affected by contrast blooming89. For this reason, the first 10 mm of the right coronary artery are excluded from analyses, and PVAT around this artery is measured in a segment between 10 mm and 50 mm away from its ostium. Anatomical variation in fat distribution around the coronary arteries, with very little adipose tissue surrounding distal vessels, limits the consistent measurement of PVAT, and differences in PVAT have been reported both at different sites along a vessel and between different arteries124. In general, vessels with a diameter <1.5 mm are excluded from analyses of PVAT but developments in imaging (such as photon-counting CT) might allow the evaluation of PVAT of smaller vessels125. Careful consideration of anatomical and technical variables when measuring PVAT in clinical and research settings is needed, and methods for PVAT measurement need to be standardized across scanner types and settings.
The interpretation of PVAT attenuation as a risk marker should also be considered. Much of the reported association between PVAT attenuation and inflammation is inferential. Ex vivo and in vitro culture techniques of human tissues have predominantly used thoracic aortic and epicardial adipose tissue rather than coronary PVAT89. Although the in vitro addition of vasoactive factors or inflammatory cytokines can cause changes in adipocytes and adipose tissue density and increase CT attenuation, inflammation is not necessarily driving the increase in CT attenuation in vivo. Other pathobiological processes that influence PVAT density can also affect PVAT attenuation. Indeed, fibrosis and increased vascularity of adipose tissue have been suggested to modify PVAT attenuation to a similar if not greater extent than inflammation126. Therefore, issues of association, cause and effect, and epiphenomena remain unresolved, such that whether in vivo changes in PVAT attenuation are indicative of local inflammation in the coronary arteries is uncertain.
The field of PVAT assessment has progressed rapidly, and various different metrics are now being reported in relation to PVAT attenuation. In addition to the measurement of PVAT attenuation itself, a proprietary fat attenuation index score has been developed that adjusts PVAT attenuation for technical scan parameters (such as tube voltage), anatomical factors (such as the perivascular distribution of fat) and demographics (age and sex)126. Radiomics analysis — a technique that extracts quantitative data from medical images to describe detailed tissue characteristics — incorporates textural features of the PVAT into the analysis and generates a fat radiomic profile, which seems to improve the risk prediction of non-fatal myocardial infarction or death from coronary heart disease compared with atherosclerotic plaque analysis126–128. In addition, radiomics might facilitate the non-invasive transcriptomic analysis of various tissues, including PVAT and atherosclerotic plaques129,130. Finally, proprietary artificial intelligence-based risk prediction algorithms have been developed that incorporate the fat attenuation index score, atherosclerotic plaque burden and clinical cardiovascular risk factors (such as diabetes, smoking, hyperlipidaemia and hypertension)122. Therefore, these risk scores are a combination of many metrics of clinical cardiovascular risk and coronary artery disease. Of note, these derivatives of PVAT attenuation have not been validated as markers of coronary inflammation.
Coronary atherosclerosis and PVAT in humans
As described above, the anatomical contiguity of PVAT with the vasculature raises the potential for local interactions with the coronary arteries. Several clinical studies have investigated associations between PVAT attenuation and atherosclerotic disease. PVAT attenuation is higher in coronary arteries with atherosclerotic plaque, obstructive coronary artery disease and in patients with acute presentations of coronary artery disease89. Moreover, in ten patients with myocardial infarction, PVAT attenuation was higher at the site of ten stented culprit lesions than in seven stented non-culprit lesions89. In general, these findings have been confirmed by other investigators who also found higher PVAT attenuation in patients with atherosclerosis, obstructive coronary artery disease, and high-risk or culprit atherosclerotic plaques14,125,131,132. In a post hoc analysis of the SCOT-HEART trial121, PVAT attenuation of the right coronary artery was higher in patients with obstructive disease than in those with non-obstructive disease, although the absolute differences were small (−75 HU versus −76 HU; P = 0.002), and PVAT attenuation had only weak associations with the burden of total plaque, non-calcified plaque, or low-attenuation plaque and no association with the CAC score or calcium burden.
The correlation between PVAT attenuation and other imaging measures of vascular inflammation and high-risk atherosclerotic plaque features has been investigated. In 41 patients with high-risk atherosclerotic plaque features, higher PVAT attenuation was associated with focal 18F-NaF uptake in the coronary artery, supporting a possible link between vascular inflammation, atherosclerotic plaque activity and microcalcification133. PVAT attenuation was also weakly associated (r = 0.30–0.35; P ≤ 0.004) with coronary inflammation as assessed by 68Ga-DOTATATE uptake (a measure of vascular macrophage activation) on positron emission tomography134. These small, observational studies support a link between PVAT attenuation and atherosclerotic plaque inflammation, although the associations are fairly weak.
PVAT and risk prediction
Myocardial infarction
PVAT attenuation should intuitively be a strong predictor of myocardial infarction given its association with coronary artery disease and its severity14,135. Furthermore, if PVAT attenuation is a marker of coronary pathobiology and inflammation, then this association should be even stronger because myocardial infarction is triggered by inflamed and unstable coronary atherosclerotic plaques. Several studies have demonstrated associations between PVAT attenuation and subsequent myocardial infarction but these associations are modest after adjustment for known clinical risk factors or the extent of coronary artery disease9,120,121,136. Larger studies have not reported the prognostic performance of PVAT attenuation for the single end point of myocardial infarction122.
Given that the overall independent risk prediction is modest, attempts have been made to use PVAT attenuation scores with or without radiomics and artificial intelligence-based risk scores to increase the predictive power of atherosclerotic plaque characteristics and PVAT attenuation. PVAT radiomics phenotype is different in patients with acute myocardial infarction compared with patients with stable or no coronary artery disease, and a radiomics-based model outperformed PVAT attenuation alone in identifying patients with a recent acute myocardial infarction128. Interestingly, the radiomics features with the highest discriminatory power for acute myocardial infarction were textural and geometric features not intensity-based metrics such as CT attenuation128. In another study, 1,391 radiomics features (based on volume, attenuation and texture) of PVAT were assessed to characterize a radiomics signature of high-risk PVAT, which was used to identify individuals at increased cardiovascular risk120. Although this model did not correlate with the presence of high-risk atherosclerotic plaque features, higher PVAT attenuation values were observed in the 44 patients with acute myocardial infarction than in the 44 matched control individuals, and a positive association was observed with the composite end point of non-fatal myocardial infarction or cardiac death120.
Cardiac death
PVAT attenuation does seem to have a closer correlation with all-cause and cardiac death than with myocardial infarction. Data from the CRISP-CT study120 showed that, in combination with a number of other clinical and imaging factors, higher PVAT attenuation improved risk prediction of all-cause and especially cardiac death. However, the incremental value of PVAT attenuation was modest (increase in the c-statistic of 0.049) and there seemed to be interactions with high-risk atherosclerotic plaque features120. Standard cardiovascular risk factors and atherosclerotic plaque characteristics accounted for most of the predictive power.
The ORFAN study122 evaluated the prognostic performance of the proprietary fat attenuation index and artificial intelligence-based risk scores in 3,393 patients from two centres in the UK. Both scores were predictive of cardiac death over 10 years in patients with or without coronary atherosclerosis122. However, the QRISK3 score (the standard clinical risk score in the UK) also showed excellent predictive power for cardiac death in this population (c-statistic = 0.831), and the sequential addition of coronary artery stenosis and an overall artificial intelligence-based risk score to the QRISK3 score produced only modest incremental improvements in discrimination (increases in the c-statistic of 0.007 and 0.016, respectively)122. The predictive power of the artificial intelligence-based risk score alone was not reported, and whether more sophisticated measures of atherosclerotic plaque burden would have provided similar improvements in risk stratification is uncertain. The study also used an unusual definition for the composite end point of MACE, which combined cardiac death, myocardial infarction and new-onset heart failure: the last of which has an indirect and non-specific relationship to atherothrombotic coronary artery disease. New-onset heart failure accounted for 44% of the events in this composite end point122. The performance of each score for the composite end point of MACE was less powerful than for the single end point of cardiac death, with the QRISK3 score having a c-statistic of 0.784 for MACE and 0.831 for cardiac death alone122. The sequential addition of coronary artery stenosis and an artificial intelligence-based risk score (including clinical risk factors, PVAT score and coronary plaque burden) had modest effects, increasing the c-statistic by 0.005 and 0.016, respectively122.
This dilutional effect on prognostic performance when transitioning from cardiac death to MACE is perhaps counterintuitive for a metric that focuses on coronary inflammation. Interestingly, two other studies have investigated the association between PVAT attenuation and long-term mortality, with durations of follow-up that were similar to that of the ORFAN study (over 9 years) but with contradictory results (Table 1). One study involving 293 individuals reported that increased PVAT attenuation (≥−73.5 HU) around the proximal right coronary artery was predictive of MACE (coronary revascularization, non-fatal myocardial infarction and/or cardiac death)137, whereas another study used a larger dataset of 483 patients and found no association between PVAT attenuation and MACE (all-cause death, myocardial infarction or unstable angina pectoris)138. Importantly, neither of these studies included new-onset heart failure, which was a component of the primary composite end point in the ORFAN study122.
Table 1 |.
Studies investigating the association between PVAT attenuation and MACE
| Study | Patient population | Number of patients | Primary outcome | Main findings |
|---|---|---|---|---|
| CRISP-CT (2018)120 | Patients undergoing clinically indicated CTCA, with derivation (n = 1,872) and validation (n = 2,040) cohorts | 3,912 | All-cause and cardiac death | PVAT attenuation around the proximal RCA was predictive of cardiac death (adjusted HR 2.06, 95% CI 1.50–2.83 per 1 SD increase; P < 0.0001) and all-cause death (adjusted HR 1.84, 95% CI 1.45–2.33; P < 0.0001) at a median follow-up of 54 months in the validation cohort. High PVAT attenuation (≥−70.1 HU) was associated with an increased risk of cardiac death (adjusted HR 5.62, 95% CI 2.90–10.88; P < 0.0001) and all-cause death (adjusted HR 3.69, 95% CI 2.26–6.02; P < 0.0001). High PVAT attenuation was associated with an increased risk of myocardial infarction (adjusted HR 5.08, 95% CI 1.89–13.61; P = 0.0012) |
| Goeller et al. (2021)137 | Patients with atypical chest pain and at low-to-intermediate risk of obstructive coronary artery disease | 293 | MACE (coronary revascularization, non-fatal myocardial infarction and/or cardiac death) | High PVAT attenuation (≥−73.5 HU) around the proximal RCA was predictive of MACE (adjusted OR 2.01, 95% CI 1.02–3.97) at mean follow-up of 9.6 years |
| SCOT-HEART (2022)121 | Patients with suspected angina pectoris | 1,697 | Fatal or non-fatal myocardial infarction | PVAT attenuation around the proximal RCA was predictive of fatal or non-fatal myocardial infarction (adjusted HR 1.54, 95% CI 1.02–2.12) at a median follow-up of 4.7 years. High PVAT attenuation (≥−70.5 HU) was associated with an increased risk of fatal or non-fatal myocardial infarction (HR 2.45, 95% CI 1.23–4.80) |
| CORE320 (2022)154 | Patients aged 45–85 years with suspected or known coronary artery disease and clinically referred for invasive coronary angiography | 344 | MACE (stroke, myocardial infarction or death) | PVAT attenuation around the proximal RCA was not predictive of MACE (HR 0.682, 95% CI 0.435–1.070). PVAT attenuation around the proximal RCA did not improve prediction of MACE beyond the use of established risk factors |
| Wen et al. (2023)155 | Patients with acute chest pain and low-to-intermediate risk of obstructive coronary artery disease | 1,313 | MACE (cardiovascular death, non-fatal myocardial infarction, unplanned revascularization and hospitalization for unstable angina) | PVAT attenuation around the proximal RCA was predictive of MACE (adjusted HR 1.033, 95% CI 1.010–1.057) at median follow-up of 38 months |
| Chen et al. (2024)156 | Clinically indicated CTCA, with derivation (n = 425) and validation (n = 183) cohorts | 608 | MACE (cardiovascular death, non-fatal myocardial infarction, unplanned revascularization and hospitalization for unstable angina) | Averaged PVAT attenuation around atherosclerotic lesions was predictive of MACE (adjusted HR 1.108, 95% CI 1.059–1.159) at a median follow-up of 58 months in the validation cohort. Averaged PVAT attenuation around atherosclerotic lesions was associated with a higher c-statistic for all-cause and cardiac death (0.680, 95% CI 0.608–0.751) than PVAT attenuation around the proximal RCA (0.586, 95% CI 0.506–0.667) in the validation cohort |
| ORFAN (2024)122 | Study cohort B (clinically indicated CTCA) | 3,393 | MACE (myocardial infarction, new-onset heart failure and cardiac death) | Increased fat attenuation index score in all three coronary arteries had an additive effect on the risk of cardiac death (adjusted HR 29.8, 95% CI 13.9–63.9) or MACE (adjusted HR 12.6, 95% CI 8.5–18.6) |
| Yang et al. (2024)157 | Patients with chest pain and a low-to-intermediate risk of obstructive coronary artery disease | 514 | MACE (unstable angina requiring hospitalization, coronary revascularization, non-fatal myocardial infarction and all-cause death) | PVAT attenuation around the proximal RCA was predictive of MACE (adjusted HR 1.026, 95% CI 1.002–1.05) at a median follow-up of 31 months |
| van Rosendael et al. (2024)138 | Symptomatic patients with an intermediate risk of coronary artery disease | 922 | MACE (all-cause death, myocardial infarction or unstable angina pectoris) | PVAT attenuation was not associated with the risk of MACE at a median follow-up of 9.5 years |
CTCA, computed tomography coronary angiography; HU, Hounsfield units; MACE, major adverse cardiovascular events; PVAT, perivascular adipose tissue; RCA, right coronary artery.
PVAT and monitoring treatment response
The growing availability of imaging biomarkers to assess atherosclerotic disease presents a unique opportunity to improve therapeutic decision-making and identify targets for precision medicine139. Many studies are now focusing on the broader cardiometabolic effects of anti-atherosclerotic drugs, including their potential effect on PVAT attenuation. Atherosclerotic plaque stabilization or regression is the ultimate goal of medical therapy for atherosclerosis140 and can be achieved through changes in plaque composition and phenotype141,142 (Fig. 5).
Fig. 5 |. Atherosclerotic plaque regression and stabilization.
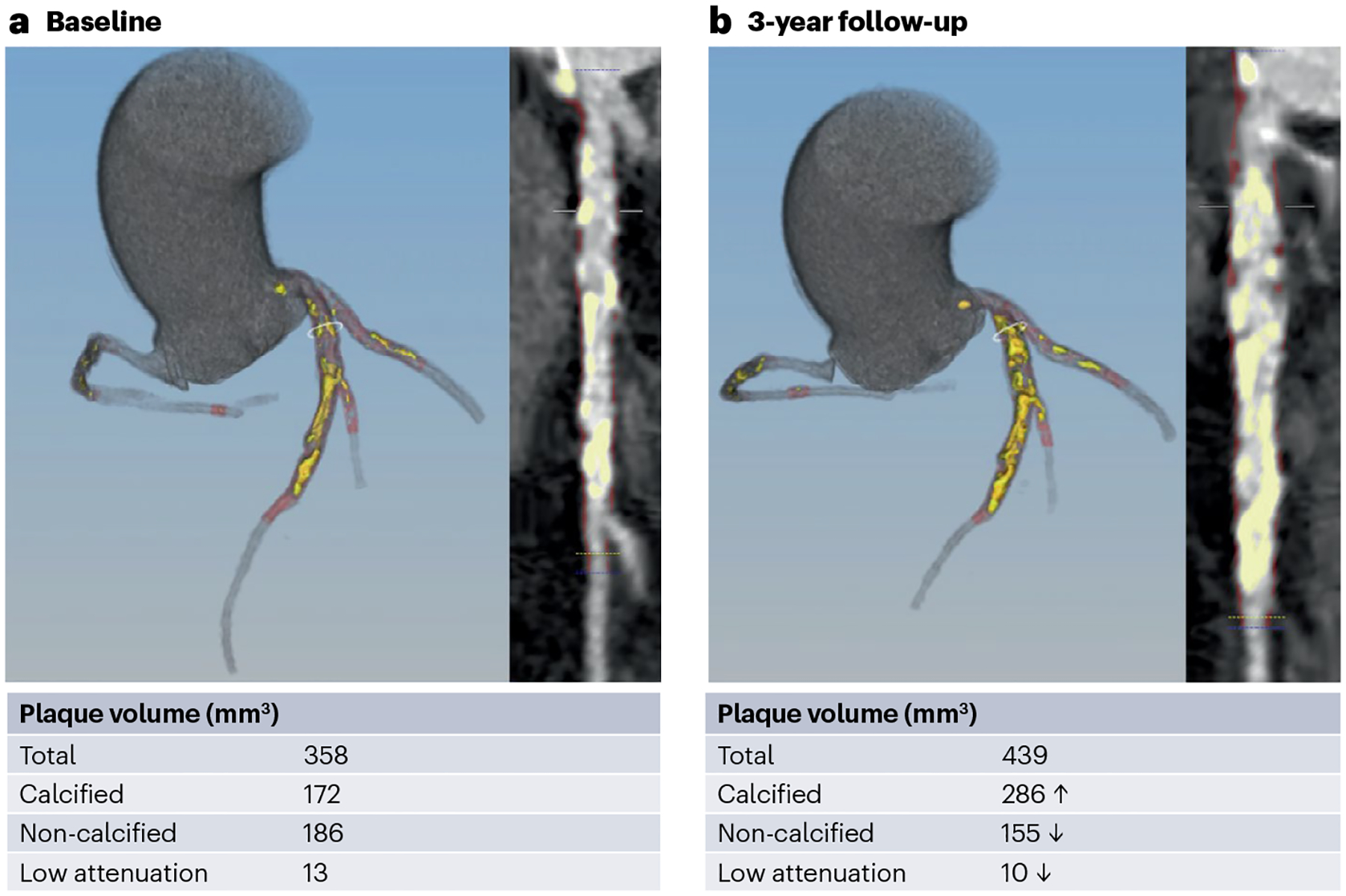
Treatment of a patient with 20 mg of rosuvastatin led to a 44% decrease in plasma LDL-cholesterol levels between baseline (part a) and the 3-year follow-up (part b). Computed tomography coronary angiography shows that the amount of non-calcified and low-attenuation atherosclerotic plaque decreased, whereas the amount of calcified atherosclerotic plaque increased, thereby increasing plaque stability. The mean attenuation of the perivascular adipose tissue decreased by 2 Hounsfield units.
Statin therapy is the cornerstone of lipid-lowering therapy and promotes atherosclerotic plaque stabilization through a decrease in non-calcific plaque volume and an increase in calcific atheroma143, which has been associated with improvements in patient outcomes144,145. Similar effects have been reported with further reductions in plasma LDL concentrations after the addition of ezetimibe146 or proprotein convertase subtilisin–kexin type 9 inhibitors147. Of note, statin therapy has also been reported to have beneficial effects on PVAT attenuation148. In 180 patients with chest pain and an intermediate risk of coronary artery disease, statin therapy over 1 year reduced the mean CT attenuation in PVAT surrounding non-calcified (from −68 ± 9 HU to −72 ± 8 HU; P < 0.001) and mixed (from −71 ± 9 HU to −73 ± 9 HU; P = 0.014) atherosclerotic plaques but had no significant effect on CT attenuation in PVAT surrounding calcified atherosclerotic plaques (from −71 ± 10 HU to −72 ± 10 HU; P = 0.258)148. However, these findings again illustrate that changes in PVAT attenuation are fairly small, which creates difficulties when translating them for use as an imaging biomarker to guide management decisions in clinical practice. Whether the next generation of lipid-modifying therapies, such as targeted therapies to lower plasma lipoprotein(a) levels, will similarly influence atherosclerotic plaque composition or PVAT attenuation needs to be determined.
In a longitudinal study of 134 consecutive patients with moderate to severe psoriasis, biologic therapy was associated with a decrease in coronary PVAT attenuation149. Although this study did not examine the association between PVAT attenuation and coronary atherosclerotic plaque and did not include hard cardiovascular end points, the findings suggest that PVAT attenuation might have a role as a biomarker of inflammation that could be used to track the response to anti-inflammatory treatments, subject to further validation.
Limitations and future directions
Although much research has been conducted in atherosclerosis and PVAT, we still have much to learn in terms of the underlying pathobiology. The causative pathways and confirmation of bidirectional signalling are yet to be definitively proven. Moreover, while the inflammatory hypothesis of PVAT attenuation on CT remains an attractive concept, the increasingly complex radiomic metrics that are being developed suggest that other pathways are also likely to contribute, and central questions of cause and effect need to be addressed.
Phenotyping of atherosclerotic plaques and PVAT can be used to improve prognostication beyond the use of clinical risk scores and measures of lesion severity. However, their incremental value is modest, and similar increases in prognostication have been reported with the addition of CAC scoring to clinical risk scores (which increases the c-statistic by 0.05 compared with the Framingham risk score alone150). If these complex measures are to be adopted into routine clinical practice, they will need to be highly automated and demonstrate a step change in prognostication that adds value to simple measures of cardiovascular risk. Furthermore, the multitude of different approaches and scores need to be standardized and take account of the different CT scanner vendors, imaging protocols and scan parameters.
Many cardiovascular risk scores predict death better than myocardial infarction, which probably reflects the dominance of age in most models151,152. Phenotyping of atherosclerotic plaques and PVAT attenuation heralded the promise of providing additive and independent risk prediction, especially for acute myocardial infarction. Although some success has been achieved with each, the strengths of the association are modest, remain to be fully validated and arguably are at odds with the hypothesis of local coronary arterial inflammation.
Finally, phenotyping of atherosclerotic plaques and PVAT attenuation might provide more dynamic measures of vascular health that could function as surrogate biomarkers to test emerging novel therapeutic interventions. Early observational data are promising in this regard149,153; however, larger prospective trials are needed to validate these imaging biomarkers. Large trials will be needed for measures such as low-attenuation plaque volume and PVAT attenuation because variability in these parameters is high and the change in response to treatment is small. These limitations might negate the benefits shown in small proof-of-concept studies into these surrogate biomarkers14.
Conclusions
CTCA enables the non-invasive evaluation of atherosclerotic plaques and PVAT of the coronary arteries. Image analysis techniques can now quantify the burden and morphology of the atherosclerotic plaque and the PVAT attenuation. Phenotyping of atherosclerotic plaques improves the prediction of future myocardial infarction beyond the use of cardiovascular risk scores and current indices of cardiovascular risk. PVAT attenuation might be useful for the prediction of atherosclerotic disease progression and cardiac events, although current data suggest that this measure provides only modest additional predictive discrimination when combined with standard cardiovascular risk scores. Further large, prospective trials are needed to validate these imaging biomarkers for the prediction of future myocardial infarction and to monitor atherosclerotic disease progression and treatment response.
Key points.
Computed tomography coronary angiography (CTCA) provides a non-invasive method to evaluate coronary artery disease that allows the phenotyping of atherosclerotic plaques and surrounding perivascular adipose tissue (PVAT).
Bidirectional signalling between the coronary arteries and the adjacent PVAT might contribute to the progression of atherosclerosis.
Certain atherosclerotic plaque characteristics (such as positive remodelling, non-calcified plaque, spotty calcification and the napkin-ring sign) are indicative of an increased risk of adverse coronary events; quantitative plaque assessment might help to identify patients at high risk, beyond traditional assessments of stenosis severity.
Despite advances in radiomic and artificial intelligence-based algorithms, studies indicate that the use of PVAT signal attenuation on CTCA only modestly improves predictive discrimination beyond the use of standard cardiovascular risk scores.
Measuring PVAT attenuation by CTCA is affected by various technical factors (such as reconstruction algorithms, scanner variations and tube voltage), which can influence the consistency and accuracy of the measurements, complicating their use in clinical practice.
Acknowledgements
K.G. is supported by the Foundation for Polish Science and a Polish National Agency for Academic Exchange. J.G. is supported by the Research Foundation Flanders (FWO) grant for long stay abroad (V414524N, V428223N) and the European Association of Cardiovascular Imaging (EACVI) Research Grant 2022. N.N. is supported by the National Health and Medical Research Council of Australia (APP1197028). D.E.N. is supported by the British Heart Foundation (CH/09/002, RG/F/22/110093, RE/24/130012). K.G., J.G. and D.D. are supported by the National Institute of Health/National Heart, Lung, and Blood Institute grants (1R01HL148787-01A1, 1R01HL151266 and 1R01HL175875), and a grant from the Miriam and Sheldon G. Adelson Medical Research Foundation.
Footnotes
Competing interests
P.J.S., D.B. and D.D. may receive royalties from Cedars-Sinai Medical Center and have equity in APQ Health. The other authors declare no competing interests.
References
- 1.Vaduganathan M, Mensah GA, Turco JV, Fuster V & Roth GA The global burden of cardiovascular diseases and risk. J. Am. Coll. Cardiol 80, 2361–2371 (2022). [DOI] [PubMed] [Google Scholar]
- 2.Ibanez B et al. Progression of early subclinical atherosclerosis (PESA) study. J. Am. Coll. Cardiol 78, 156–179 (2021). [DOI] [PubMed] [Google Scholar]
- 3.Libby P et al. Atherosclerosis. Nat. Rev. Dis. Prim 5, 56 (2019). [DOI] [PubMed] [Google Scholar]
- 4.Arbab-Zadeh A, Nakano M, Virmani R & Fuster V Acute coronary events. Circulation 125, 1147–1156 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Falk E, Shah PK & Fuster V Coronary plaque disruption. Circulation 92, 657–671 (1995). [DOI] [PubMed] [Google Scholar]
- 6.Wang JC, Normand SL, Mauri L & Kuntz RE Coronary artery spatial distribution of acute myocardial infarction occlusions. Circulation 110, 278–284 (2004). [DOI] [PubMed] [Google Scholar]
- 7.Stone GW et al. A prospective natural-history study of coronary atherosclerosis. N. Engl. J. Med 364, 226–235 (2011). [DOI] [PubMed] [Google Scholar]
- 8.Puri R, Nicholls SJ, Ellis SG, Tuzcu EM & Kapadia SR High-risk coronary atheroma: the interplay between ischemia, plaque burden, and disease progression. J. Am. Coll. Cardiol 63, 1134–1140 (2014). [DOI] [PubMed] [Google Scholar]
- 9.Williams MC et al. Low-attenuation noncalcified plaque on coronary computed tomography angiography predicts myocardial infarction: results from the Multicenter SCOT-HEART Trial (Scottish Computed Tomography of the HEART). Circulation 141, 1452–1462 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Narula J et al. Histopathologic characteristics of atherosclerotic coronary disease and implications of the findings for the invasive and noninvasive detection of vulnerable plaques. J. Am. Coll. Cardiol 61, 1041–1051 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang T et al. Longitudinal assessment of coronary plaque regression related to sodium-glucose cotransporter-2 inhibitor using coronary computed tomography angiography. Cardiovasc. Diabetol 23, 267 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liu S et al. Effect of PCSK9 antibodies on coronary plaque regression and stabilization derived from intravascular imaging in patients with coronary artery disease: a meta-analysis. Int. J. Cardiol 392, 131330 (2023). [DOI] [PubMed] [Google Scholar]
- 13.Vaidya K et al. Colchicine therapy and plaque stabilization in patients with acute coronary syndrome: a CT coronary angiography study. JACC Cardiovasc. Imaging 11, 305–316 (2018). [DOI] [PubMed] [Google Scholar]
- 14.Goeller M et al. Pericoronary adipose tissue computed tomography attenuation and high-risk plaque characteristics in acute coronary syndrome compared with stable coronary artery disease. JAMA Cardiol 3, 858–863 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kwiecinski J et al. Noninvasive coronary atherosclerotic plaque imaging. JACC Cardiovasc. Imaging 16, 1608–1622 (2023). [DOI] [PubMed] [Google Scholar]
- 16.Qi X-Y et al. Perivascular adipose tissue (PVAT) in atherosclerosis: a double-edged sword. Cardiovasc. Diabetol 17, 134 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tan N, Dey D, Marwick TH & Nerlekar N Pericoronary adipose tissue as a marker of cardiovascular risk. J. Am. Coll. Cardiol 81, 913–923 (2023). [DOI] [PubMed] [Google Scholar]
- 18.Kotanidis CP & Antoniades C Perivascular fat imaging by computed tomography (CT): a virtual guide. Br. J. Pharmacol 178, 4270–4290 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hillock-Watling C & Gotlieb AI The pathobiology of perivascular adipose tissue (PVAT), the fourth layer of the blood vessel wall. Cardiovasc. Pathol 61, 107459 (2022). [DOI] [PubMed] [Google Scholar]
- 20.Oikonomou EK & Antoniades C The role of adipose tissue in cardiovascular health and disease. Nat. Rev. Cardiol 16, 83–99 (2019). [DOI] [PubMed] [Google Scholar]
- 21.Brown NK et al. Perivascular adipose tissue in vascular function and disease: a review of current research and animal models. Arterioscler. Thromb. Vasc. Biol 34, 1621–1630 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Koenen M, Hill MA, Cohen P & Sowers JR Obesity, adipose tissue and vascular dysfunction. Circ. Res 128, 951–968 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Frontini A & Cinti S Distribution and development of brown adipocytes in the murine and human adipose organ. Cell Metab 11, 253–256 (2010). [DOI] [PubMed] [Google Scholar]
- 24.Pérez-Martí A et al. A low-protein diet induces body weight loss and browning of subcutaneous white adipose tissue through enhanced expression of hepatic fibroblast growth factor 21 (FGF21). Mol. Nutr. Food Res 61, 1600725 (2017). [DOI] [PubMed] [Google Scholar]
- 25.Otero-Díaz B et al. Exercise induces white adipose tissue browning across the weight spectrum in humans. Front. Physiol 9, 1781 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lucchini FC et al. ASK1 inhibits browning of white adipose tissue in obesity. Nat. Commun 11, 1642 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kalinovich AV, de Jong JM, Cannon B & Nedergaard J UCP1 in adipose tissues: two steps to full browning. Biochimie 134, 127–137 (2017). [DOI] [PubMed] [Google Scholar]
- 28.Fischer C et al. A miR-327–FGF10–FGFR2-mediated autocrine signaling mechanism controls white fat browning. Nat. Commun 8, 2079 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Machado SA et al. Browning of the white adipose tissue regulation: new insights into nutritional and metabolic relevance in health and diseases. Nutr. Metab 19, 61 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Britton KA et al. Prevalence, distribution, and risk factor correlates of high thoracic periaortic fat in the Framingham Heart Study. J. Am. Heart Assoc 1, e004200 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.El Khoudary SR et al. Postmenopausal women with greater paracardial fat have more coronary artery calcification than premenopausal women: the Study of Women’s Health Across the Nation (SWAN) Cardiovascular Fat Ancillary Study. J. Am. Heart Assoc 6, e004545 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ahmad AA, Randall MD & Roberts RE Sex differences in the role of phospholipase A(2) —dependent arachidonic acid pathway in the perivascular adipose tissue function in pigs. J. Physiol 595, 6623–6634 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang D et al. Endothelial dysfunction and enhanced contractility in microvessels from ovariectomized rats. Hypertension 63, 1063–1069 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yuvaraj J et al. Pericoronary adipose tissue attenuation on coronary computed tomography angiography associates with male sex and Indigenous Australian status. Sci. Rep 13, 15509 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.van Rosendael SE et al. Vessel and sex differences in pericoronary adipose tissue attenuation obtained with coronary CT in individuals without coronary atherosclerosis. Int. J. Cardiovasc. Imaging 38, 2781–2789 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kinoshita D et al. Sex-specific association between perivascular inflammation and plaque vulnerability. Circ. Cardiovasc. Imaging 17, e016178 (2024). [DOI] [PubMed] [Google Scholar]
- 37.Matsuzawa Y & Lerman A Endothelial dysfunction and coronary artery disease: assessment, prognosis, and treatment. Coron. Artery Dis 25, 713–724 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rajendran P et al. The vascular endothelium and human diseases. Int. J. Biol. Sci 9, 1057–1069 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ross R Atherosclerosis — an inflammatory disease. N. Engl. J. Med 340, 115–126 (1999). [DOI] [PubMed] [Google Scholar]
- 40.Cheng CK, Bakar HA, Gollasch M & Huang Y Perivascular adipose tissue: the sixth man of the cardiovascular system. Cardiovasc. Drugs Ther 32, 481–502 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Akoumianakis I & Antoniades C The interplay between adipose tissue and the cardiovascular system: is fat always bad? Cardiovasc. Res 113, 999–1008 (2017). [DOI] [PubMed] [Google Scholar]
- 42.Akoumianakis I, Tarun A & Antoniades C Perivascular adipose tissue as a regulator of vascular disease pathogenesis: identifying novel therapeutic targets. Br. J. Pharmacol 174, 3411–3424 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Antonopoulos AS et al. Adiponectin as a link between type 2 diabetes and vascular NADPH oxidase activity in the human arterial wall: the regulatory role of perivascular adipose tissue. Diabetes 64, 2207–2219 (2015). [DOI] [PubMed] [Google Scholar]
- 44.Mani S et al. Decreased endogenous production of hydrogen sulfide accelerates atherosclerosis. Circulation 127, 2523–2534 (2013). [DOI] [PubMed] [Google Scholar]
- 45.Kauser K, da Cunha V, Fitch R, Mallari C & Rubanyi GM Role of endogenous nitric oxide in progression of atherosclerosis in apolipoprotein E-deficient mice. Am. J. Physiol. Heart Circ. Physiol 278, H1679–H1685 (2000). [DOI] [PubMed] [Google Scholar]
- 46.Thomas C, Mackey MM, Diaz AA & Cox DP Hydroxyl radical is produced via the Fenton reaction in submitochondrial particles under oxidative stress: implications for diseases associated with iron accumulation. Redox Rep 14, 102–108 (2009). [DOI] [PubMed] [Google Scholar]
- 47.Rippe B, Rosengren BI, Carlsson O & Venturoli D Transendothelial transport: the vesicle controversy. J. Vasc. Res 39, 375–390 (2002). [DOI] [PubMed] [Google Scholar]
- 48.Jang E, Robert J, Rohrer L, von Eckardstein A & Lee WL Transendothelial transport of lipoproteins. Atherosclerosis 315, 111–125 (2020). [DOI] [PubMed] [Google Scholar]
- 49.Sheedy FJ et al. CD36 coordinates NLRP3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nat. Immunol 14, 812–820 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Witztum JL & Steinberg D The oxidative modification hypothesis of atherosclerosis: does it hold for humans? Trends Cardiovasc. Med 11, 93–102 (2001). [DOI] [PubMed] [Google Scholar]
- 51.Pober JS & Sessa WC Evolving functions of endothelial cells in inflammation. Nat. Rev. Immunol 7, 803–815 (2007). [DOI] [PubMed] [Google Scholar]
- 52.Ropraz P, Imhof BA, Matthes T, Wehrle-Haller B & Sidibé A Simultaneous study of the recruitment of monocyte subpopulations under flow in vitro. J. Vis. Exp 141, e58509 (2018). [DOI] [PubMed] [Google Scholar]
- 53.Gerhardt T & Ley K Monocyte trafficking across the vessel wall. Cardiovasc. Res 107, 321–330 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Choi HY et al. ATP-binding cassette transporter A1 expression and apolipoprotein A-I binding are impaired in intima-type arterial smooth muscle cells. Circulation 119, 3223–3231 (2009). [DOI] [PubMed] [Google Scholar]
- 55.Allahverdian S, Chehroudi AC, McManus BM, Abraham T & Francis GA Contribution of intimal smooth muscle cells to cholesterol accumulation and macrophage-like cells in human atherosclerosis. Circulation 129, 1551–1559 (2014). [DOI] [PubMed] [Google Scholar]
- 56.Chinetti-Gbaguidi G et al. Human atherosclerotic plaque alternative macrophages display low cholesterol handling but high phagocytosis because of distinct activities of the PPARγ and LXRα pathways. Circ. Res 108, 985–995 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Maitra U, Parks JS & Li L An innate immunity signaling process suppresses macrophage ABCA1 expression through IRAK-1-mediated downregulation of retinoic acid receptor alpha and NFATc2. Mol. Cell Biol 29, 5989–5997 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fitzgibbons TP & Czech MP Epicardial and perivascular adipose tissues and their influence on cardiovascular disease: basic mechanisms and clinical associations. J. Am. Heart Assoc 3, e000582 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Verhagen SN, Vink A, van der Graaf Y & Visseren FL Coronary perivascular adipose tissue characteristics are related to atherosclerotic plaque size and composition. A post-mortem study. Atherosclerosis 225, 99–104 (2012). [DOI] [PubMed] [Google Scholar]
- 60.Farias-Itao DS et al. B lymphocytes and macrophages in the perivascular adipose tissue are associated with coronary atherosclerosis: an autopsy study. J. Am. Heart Assoc 8, e013793 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Oh DY, Morinaga H, Talukdar S, Bae EJ & Olefsky JM Increased macrophage migration into adipose tissue in obese mice. Diabetes 61, 346–354 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Shirai T, Hilhorst M, Harrison DG, Goronzy JJ & Weyand CM Macrophages in vascular inflammation — from atherosclerosis to vasculitis. Autoimmunity 48, 139–151 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Moos MP et al. The lamina adventitia is the major site of immune cell accumulation in standard chow-fed apolipoprotein E-deficient mice. Arterioscler. Thromb. Vasc. Biol 25, 2386–2391 (2005). [DOI] [PubMed] [Google Scholar]
- 64.Ketelhuth DF & Hansson GK Adaptive response of T and B cells in atherosclerosis. Circ. Res 118, 668–678 (2016). [DOI] [PubMed] [Google Scholar]
- 65.Tay C et al. B-cell-specific depletion of tumour necrosis factor alpha inhibits atherosclerosis development and plaque vulnerability to rupture by reducing cell death and inflammation. Cardiovasc. Res 111, 385–397 (2016). [DOI] [PubMed] [Google Scholar]
- 66.Du X et al. Insulin resistance reduces arterial prostacyclin synthase and eNOS activities by increasing endothelial fatty acid oxidation. J. Clin. Invest 116, 1071–1080 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Watson MG, Byrne HM, Macaskill C & Myerscough MR A two-phase model of early fibrous cap formation in atherosclerosis. J. Theor. Biol 456, 123–136 (2018). [DOI] [PubMed] [Google Scholar]
- 68.Chamié D, Wang Z, Bezerra H, Rollins AM & Costa MA Optical coherence tomography and fibrous cap characterization. Curr. Cardiovasc. Imaging Rep 4, 276–283 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bennett MR, Sinha S & Owens GK Vascular smooth muscle cells in atherosclerosis. Circ. Res 118, 692–702 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Manka D et al. Transplanted perivascular adipose tissue accelerates injury-induced neointimal hyperplasia: role of monocyte chemoattractant protein-1. Arterioscler. Thromb. Vasc. Biol 34, 1723–1730 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Miao CY & Li ZY The role of perivascular adipose tissue in vascular smooth muscle cell growth. Br. J. Pharmacol 165, 643–658 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Miyata K et al. Rho-kinase is involved in macrophage-mediated formation of coronary vascular lesions in pigs in vivo. Arterioscler. Thromb. Vasc. Biol 20, 2351–2358 (2000). [DOI] [PubMed] [Google Scholar]
- 73.Shimokawa H et al. Chronic treatment with interleukin-1 beta induces coronary intimal lesions and vasospastic responses in pigs in vivo. The role of platelet-derived growth factor. J. Clin. Invest 97, 769–776 (1996). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Johnson JL et al. Relationship of MMP-14 and TIMP-3 expression with macrophage activation and human atherosclerotic plaque vulnerability. Mediators Inflamm 2014, 276457 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Giacco F & Brownlee M Oxidative stress and diabetic complications. Circ. Res 107, 1058–1070 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Creager MA, Lüscher TF, Cosentino F & Beckman JA Diabetes and vascular disease: pathophysiology, clinical consequences, and medical therapy: part I. Circulation 108, 1527–1532 (2003). [DOI] [PubMed] [Google Scholar]
- 77.Adkar SS & Leeper NJ Efferocytosis in atherosclerosis. Nat. Rev. Cardiol 21, 762–779 (2024). [DOI] [PubMed] [Google Scholar]
- 78.Mulay SR & Anders HJ Crystallopathies. N. Engl. J. Med 374, 2465–2476 (2016). [DOI] [PubMed] [Google Scholar]
- 79.Nishikawa T et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 404, 787–790 (2000). [DOI] [PubMed] [Google Scholar]
- 80.García-García HM et al. Relationship between cardiovascular risk factors and biomarkers with necrotic core and atheroma size: a serial intravascular ultrasound radiofrequency data analysis. Int. J. Cardiovasc. Imaging 28, 695–703 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Virmani R, Burke AP, Farb A & Kolodgie FD Pathology of the vulnerable plaque. J. Am. Coll. Cardiol 47, C13–C18 (2006). [DOI] [PubMed] [Google Scholar]
- 82.Lendon CL, Davies MJ, Born GV & Richardson PD Atherosclerotic plaque caps are locally weakened when macrophages density is increased. Atherosclerosis 87, 87–90 (1991). [DOI] [PubMed] [Google Scholar]
- 83.Xie Z et al. Adipose-derived exosomes exert proatherogenic effects by regulating macrophage foam cell formation and polarization. J. Am. Heart Assoc 7, e007442 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Itani HA et al. Activation of human T cells in hypertension: studies of humanized mice and hypertensive humans. Hypertension 68, 123–132 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wu L et al. Activation of invariant natural killer T cells by lipid excess promotes tissue inflammation, insulin resistance, and hepatic steatosis in obese mice. Proc. Natl Acad. Sci. USA 109, E1143–E1152 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wu H et al. T-cell accumulation and regulated on activation, normal T cell expressed and secreted upregulation in adipose tissue in obesity. Circulation 115, 1029–1038 (2007). [DOI] [PubMed] [Google Scholar]
- 87.Amento EP, Ehsani N, Palmer H & Libby P Cytokines and growth factors positively and negatively regulate interstitial collagen gene expression in human vascular smooth muscle cells. Arterioscler. Thromb 11, 1223–1230 (1991). [DOI] [PubMed] [Google Scholar]
- 88.Yamashita A et al. Medial and adventitial macrophages are associated with expansive atherosclerotic remodeling in rabbit femoral artery. Histol. Histopathol 23, 127–136 (2008). [DOI] [PubMed] [Google Scholar]
- 89.Antonopoulos AS et al. Detecting human coronary inflammation by imaging perivascular fat. Sci. Transl. Med 9, eaal2658 (2017). [DOI] [PubMed] [Google Scholar]
- 90.Antoniades C Dysfunctional’ adipose tissue in cardiovascular disease: a reprogrammable target or an innocent bystander? Cardiovasc. Res 113, 997–998 (2017). [DOI] [PubMed] [Google Scholar]
- 91.Mulligan-Kehoe MJ The vasa vasorum in diseased and nondiseased arteries. Am. J. Physiol. Heart Circ. Physiol 298, H295–H305 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Greenstein AS et al. Local inflammation and hypoxia abolish the protective anticontractile properties of perivascular fat in obese patients. Circulation 119, 1661–1670 (2009). [DOI] [PubMed] [Google Scholar]
- 93.Aghamohammadzadeh R et al. Effects of bariatric surgery on human small artery function: evidence for reduction in perivascular adipocyte inflammation, and the restoration of normal anticontractile activity despite persistent obesity. J. Am. Coll. Cardiol 62, 128–135 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Calabro P, Samudio I, Willerson JT & Yeh ET Resistin promotes smooth muscle cell proliferation through activation of extracellular signal-regulated kinase 1/2 and phosphatidylinositol 3-kinase pathways. Circulation 110, 3335–3340 (2004). [DOI] [PubMed] [Google Scholar]
- 95.Chen C et al. Resistin decreases expression of endothelial nitric oxide synthase through oxidative stress in human coronary artery endothelial cells. Am. J. Physiol. Heart Circ. Physiol 299, H193–H201 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Grant RW & Stephens JM Fat in flames: influence of cytokines and pattern recognition receptors on adipocyte lipolysis. Am. J. Physiol. Endocrinol. Metab 309, E205–E213 (2015). [DOI] [PubMed] [Google Scholar]
- 97.Kawanami D et al. Direct reciprocal effects of resistin and adiponectin on vascular endothelial cells: a new insight into adipocytokine-endothelial cell interactions. Biochem. Biophys. Res. Commun 314, 415–419 (2004). [DOI] [PubMed] [Google Scholar]
- 98.McLaughlin T et al. Relationship between coronary atheroma, epicardial adipose tissue inflammation, and adipocyte differentiation across the human myocardial bridge. J. Am. Heart Assoc 10, e021003 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ohyama K et al. Association of coronary perivascular adipose tissue inflammation and drug-eluting stent-induced coronary hyperconstricting responses in pigs: 18F-fluorodeoxyglucose positron emission tomography imaging study. Arterioscler. Thromb. Vasc. Biol 37, 1757–1764 (2017). [DOI] [PubMed] [Google Scholar]
- 100.Takaoka M et al. Endovascular injury induces rapid phenotypic changes in perivascular adipose tissue. Arterioscler. Thromb. Vasc. Biol 30, 1576–1582 (2010). [DOI] [PubMed] [Google Scholar]
- 101.Kim HW, Shi H, Winkler MA, Lee R & Weintraub NL Perivascular adipose tissue and vascular perturbation/atherosclerosis. Arterioscler. Thromb. Vasc. Biol 40, 2569–2576 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Matsumoto H et al. Standardized volumetric plaque quantification and characterization from coronary CT angiography: a head-to-head comparison with invasive intravascular ultrasound. Eur. Radiol 29, 6129–6139 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Cho I et al. Prognostic value of coronary computed tomographic angiography findings in asymptomatic individuals: a 6-year follow-up from the prospective multicentre international CONFIRM study. Eur. Heart J 39, 934–941 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Commandeur F et al. Machine learning to predict the long-term risk of myocardial infarction and cardiac death based on clinical risk, coronary calcium, and epicardial adipose tissue: a prospective study. Cardiovasc. Res 116, 2216–2225 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Han D et al. Prognostic significance of subtle coronary calcification in patients with zero coronary artery calcium score: from the CONFIRM registry. Atherosclerosis 309, 33–38 (2020). [DOI] [PubMed] [Google Scholar]
- 106.Osborne-Grinter M et al. Association of coronary artery calcium score with qualitatively and quantitatively assessed adverse plaque on coronary CT angiography in the SCOT-HEART trial. Eur. Heart J. Cardiovasc. Imaging 23, 1210–1221 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kato S, Azuma M, Horita N & Utsunomiya D Prognostic significance of CAD-RADS for patients with suspected coronary artery disease: a systematic review and meta-analysis. Radiol. Adv 1, umae007 (2024). [Google Scholar]
- 108.Maclean E, Cronshaw R, Newby DE, Nicol E & Williams MC Prognostic utility of semi-quantitative coronary computed tomography angiography scores in the SCOT-HEART trial. J. Cardiovasc. Comput. Tomogr 17, 393–400 (2023). [DOI] [PubMed] [Google Scholar]
- 109.Motoyama S et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J. Am. Coll. Cardiol 50, 319–326 (2007). [DOI] [PubMed] [Google Scholar]
- 110.Williams MC et al. Coronary artery plaque characteristics associated with adverse outcomes in the SCOT-HEART study. J. Am. Coll. Cardiol 73, 291–301 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Tanisawa H et al. Quantification of low-attenuation plaque burden from coronary CT angiography: a head-to-head comparison with near-infrared spectroscopy intravascular US. Radiol. Cardiothorac. Imaging 5, e230090 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Chang HJ et al. Coronary atherosclerotic precursors of acute coronary syndromes. J. Am. Coll. Cardiol 71, 2511–2522 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Williams MC et al. Sex-specific computed tomography coronary plaque characterization and risk of myocardial infarction. JACC Cardiovasc. Imaging 14, 1804–1814 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lin A et al. Deep learning-enabled coronary CT angiography for plaque and stenosis quantification and cardiac risk prediction: an international multicentre study. Lancet Digit. Health 4, e256–e265 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Motoyama S et al. Plaque characterization by coronary computed tomography angiography and the likelihood of acute coronary events in mid-term follow-up. J. Am. Coll. Cardiol 66, 337–346 (2015). [DOI] [PubMed] [Google Scholar]
- 116.Tzolos E et al. Repeatability of quantitative pericoronary adipose tissue attenuation and coronary plaque burden from coronary CT angiography. J. Cardiovasc. Comput. Tomogr 15, 81–84 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Chen X et al. Pericoronary adipose tissue attenuation assessed by dual-layer spectral detector computed tomography is a sensitive imaging marker of high-risk plaques. Quant. Imaging Med. Surg 11, 2093–2103 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Tan N, Dey D, Marwick TH & Nerlekar N Pericoronary adipose tissue as a marker of cardiovascular risk: JACC review topic of the week. J. Am. Coll. Cardiol 81, 913–923 (2023). [DOI] [PubMed] [Google Scholar]
- 119.Ma R et al. Towards reference values of pericoronary adipose tissue attenuation: impact of coronary artery and tube voltage in coronary computed tomography angiography. Eur. Radiol 30, 6838–6846 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Oikonomou EK et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): a post-hoc analysis of prospective outcome data. Lancet 392, 929–939 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Tzolos E et al. Pericoronary adipose tissue attenuation, low-attenuation plaque burden, and 5-year risk of myocardial infarction. JACC Cardiovasc. Imaging 15, 1078–1088 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Chan K et al. Inflammatory risk and cardiovascular events in patients without obstructive coronary artery disease: the ORFAN multicentre, longitudinal cohort study. Lancet 403, 2606–2618 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Stuijfzand WJ et al. Value of hybrid imaging with PET/CT to guide percutaneous revascularization of chronic total coronary occlusion. Curr. Cardiovasc. Imaging Rep 8, 26 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Bao W et al. A preliminary coronary computed tomography angiography-based study of perivascular fat attenuation index: relation with epicardial adipose tissue and its distribution over the entire coronary vasculature. Eur. Radiol 32, 6028–6036 (2022). [DOI] [PubMed] [Google Scholar]
- 125.Goeller M et al. Relationship between changes in pericoronary adipose tissue attenuation and coronary plaque burden quantified from coronary computed tomography angiography. Eur. Heart J. Cardiovasc. Imaging 20, 636–643 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Oikonomou EK et al. A novel machine learning-derived radiotranscriptomic signature of perivascular fat improves cardiac risk prediction using coronary CT angiography. Eur. Heart J 40, 3529–3543 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Oikonomou EK et al. Standardized measurement of coronary inflammation using cardiovascular computed tomography: integration in clinical care as a prognostic medical device. Cardiovasc. Res 117, 2677–2690 (2021). [DOI] [PubMed] [Google Scholar]
- 128.Lin A et al. Myocardial infarction associates with a distinct pericoronary adipose tissue radiomic phenotype: a prospective case-control study. JACC Cardiovasc. Imaging 13, 2371–2383 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Kotanidis CP et al. Constructing custom-made radiotranscriptomic signatures of vascular inflammation from routine CT angiograms: a prospective outcomes validation study in COVID-19. Lancet Digit. Health 4, e705–e716 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Buckler AJ et al. Virtual transcriptomics: noninvasive phenotyping of atherosclerosis by decoding plaque biology from computed tomography angiography imaging. Arterioscler. Thromb. Vasc. Biol 41, 1738–1750 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Lu MT et al. Epicardial and paracardial adipose tissue volume and attenuation — association with high-risk coronary plaque on computed tomographic angiography in the ROMICAT II trial. Atherosclerosis 251, 47–54 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Okubo R et al. Pericoronary adipose tissue ratio is a stronger associated factor of plaque vulnerability than epicardial adipose tissue on coronary computed tomography angiography. Heart Vessel 32, 813–822 (2017). [DOI] [PubMed] [Google Scholar]
- 133.Kwiecinski J et al. Peri-coronary adipose tissue density is associated with 18F-sodium fluoride coronary uptake in stable patients with high-risk plaques. JACC Cardiovasc. Imaging 12, 2000–2010 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Wall C et al. Pericoronary and periaortic adipose tissue density are associated with inflammatory disease activity in Takayasu arteritis and atherosclerosis. Eur. Heart J. Open 1, oeab019 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Lin A et al. Pericoronary adipose tissue computed tomography attenuation distinguishes different stages of coronary artery disease: a cross-sectional study. Eur. Heart J. Cardiovasc. Imaging 22, 298–306 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Yuvaraj J et al. Pericoronary adipose tissue attenuation is associated with high-risk plaque and subsequent acute coronary syndrome in patients with stable coronary artery disease. Cells 10, 1143 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Goeller M et al. Pericoronary adipose tissue CT attenuation and its association with serum levels of atherosclerosis-relevant inflammatory mediators, coronary calcification and major adverse cardiac events. J. Cardiovasc. Comput. Tomogr 15, 449–454 (2021). [DOI] [PubMed] [Google Scholar]
- 138.van Rosendael SE et al. Pericoronary adipose tissue for predicting long-term outcome. Eur. Heart J. Cardiovasc. Imaging 25, 1351–1359 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Mézquita AJV et al. Clinical quantitative coronary artery stenosis and coronary atherosclerosis imaging: a consensus statement from the Quantitative Cardiovascular Imaging Study Group. Nat. Rev. Cardiol 20, 696–714 (2023). [DOI] [PubMed] [Google Scholar]
- 140.Mori H et al. Coronary artery calcification and its progression: what does it really mean? JACC Cardiovasc. Imaging 11, 127–142 (2018). [DOI] [PubMed] [Google Scholar]
- 141.van Rosendael AR et al. Association of high-density calcified 1K plaque with risk of acute coronary syndrome. JAMA Cardiol 5, 282–290 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Aldana-Bitar J, Bhatt DL & Budoff MJ Regression and stabilization of atherogenic plaques. Trends Cardiovasc. Med 34, 340–346 (2023). [DOI] [PubMed] [Google Scholar]
- 143.Lee SE et al. Effects of statins on coronary atherosclerotic plaques: the PARADIGM study. JACC Cardiovasc. Imaging 11, 1475–1484 (2018). [DOI] [PubMed] [Google Scholar]
- 144.Andelius L, Mortensen MB, Nørgaard BL & Abdulla J Impact of statin therapy on coronary plaque burden and composition assessed by coronary computed tomographic angiography: a systematic review and meta-analysis. Eur. Heart J. Cardiovasc. Imaging 19, 850–858 (2018). [DOI] [PubMed] [Google Scholar]
- 145.Shin S et al. Impact of intensive LDL cholesterol lowering on coronary artery atherosclerosis progression: a serial CT angiography study. JACC Cardiovasc. Imaging 10, 437–446 (2017). [DOI] [PubMed] [Google Scholar]
- 146.Tsujita K et al. Impact of dual lipid-lowering strategy with ezetimibe and atorvastatin on coronary plaque regression in patients with percutaneous coronary intervention: the Multicenter Randomized Controlled PRECISE-IVUS trial. J. Am. Coll. Cardiol 66, 495–507 (2015). [DOI] [PubMed] [Google Scholar]
- 147.Pérez de Isla L et al. Alirocumab and coronary atherosclerosis in asymptomatic patients with familial hypercholesterolemia: the ARCHITECT study. Circulation 147, 1436–1443 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Dai X et al. Serial change of perivascular fat attenuation index after statin treatment: insights from a coronary CT angiography follow-up study. Int. J. Cardiol 319, 144–149 (2020). [DOI] [PubMed] [Google Scholar]
- 149.Elnabawi YA et al. Association of biologic therapy with coronary inflammation in patients with psoriasis as assessed by perivascular fat attenuation index. JAMA Cardiol 4, 885–891 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Kavousi M et al. Evaluation of newer risk markers for coronary heart disease risk classification: a cohort study. Ann. Intern. Med 156, 438–444 (2012). [DOI] [PubMed] [Google Scholar]
- 151.Khan SS et al. Development and validation of the American Heart Association’s PREVENT equations. Circulation 149, 430–449 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Fox KA et al. Prediction of risk of death and myocardial infarction in the six months after presentation with acute coronary syndrome: prospective multinational observational study (GRACE). BMJ 333, 1091 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Yu M-M et al. Evolocumab attenuate pericoronary adipose tissue density via reduction of lipoprotein(a) in type 2 diabetes mellitus: a serial follow-up CCTA study. Cardiovasc. Diabetol 22, 121 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Chatterjee D et al. Perivascular fat attenuation for predicting adverse cardiac events in stable patients undergoing invasive coronary angiography. J. Cardiovasc. Comput. Tomogr 16, 483–490 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Wen D et al. Lack of incremental prognostic value of pericoronary adipose tissue computed tomography attenuation beyond coronary artery disease reporting and data system for major adverse cardiovascular events in patients with acute chest pain. Circ. Cardiovasc. Imaging 16, 536–544 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Chen M et al. Lesion-specific pericoronary adipose tissue CT attenuation improves risk prediction of major adverse cardiovascular events in coronary artery disease. Br. J. Radiol 97, 258–266 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Yang W et al. Prognostic value of non-alcoholic fatty liver disease and RCA pericoronary adipose tissue CT attenuation in patients with acute chest pain. Acad. Radiol 31, 1773–1783 (2024). [DOI] [PubMed] [Google Scholar]


