Abstract
Background
In patients with asthma uncontrolled by a medium or high-strength (MS/HS) inhaled corticosteroid (ICS) plus long-acting β2-agonist (LABA), according to Global Initiative for Asthma (GINA) guidelines, a maintenance therapy option is the addition of a long-acting muscarinic antagonist (LAMA) via single-inhaler triple therapy (SITT). Evidence has previously been published on the cost-effectiveness of a SITT extra fine formulation of beclomethasone, formoterol and glycopyrronium bromide (BDP/FOR/GLY) vs. dual ICS/LABA combination, using data from two 52-week clinical trials (TRIMARAN and TRIGGER). However, there is limited evidence on the comparative cost-effectiveness of SITTs. The current analysis evaluated the cost-effectiveness of BDP/FOR/GLY versus other SITTs, in the UK setting.
Methods
Markov cohort state-transition model was developed to investigate the cost-effectiveness of BDP/FOR/GLY Medium Strength (MS) vs. fluticasone, umeclidinium, and vilanterol (FF/UMEC/VI) MS and, BDP/FOR/GLY High Strength vs. FF/UMEC/VI HS and vs. indacaterol acetate, glycopyrronium bromide, and mometasone (IND/GLY/MF) HS. A network meta-analysis was performed to estimate comparative efficacy of BDP/FOR/GLY against other SITTs. The model analyzed cost, quality-adjusted life-years (QALYs), and incremental cost-effectiveness ratio (ICER), net monetary benefit (NMB), and was developed from the perspective of England National Health Service (NHS) and Prescribed Specialized Services expenditure (2022 costs). Uncertainty of the inputs was estimated using one-way and probabilistic sensitivity analyses.
Results
BDP/FOR/GLY MS was projected to be a dominant treatment alternative against FF/UMEC/VI MS (£5,121 less costly, gained 0.065 additional QALYs). Similarly, BDP/FOR/GLY HS was a dominant treatment alternative against FF/UMEC/VI HS (£143, 0.003 additional QALYs) and IND/GLY/MF HS (£692 less costly, gained 0.023 additional QALYs). BDP/FOR/GLY MS and HS had 77.1%, 51.3%, and 61.2% likelihoods to be cost-effective vs. FF/UMEC/VI MS, FF/UMEC/VI HS, and IND/GLY/MF HS at the defined willingness-to-pay (WTP) threshold of £20,000 per QALY gained, respectively.
Conclusions
BDP/FOR/GLY MS and HS were a dominant treatment alternative compared with FF/UMEC/VI, both MS and HS, and IND/GLY/MF HS in patients with asthma uncontrolled by ICS/LABA.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13561-025-00640-9.
Keywords: Asthma, Single-inhaler triple therapy, Network meta-analysis, Cost-effectiveness, Exacerbations, England
Introduction
Asthma is a chronic inflammatory disease of the respiratory tract that presents with bronchial hyperresponsiveness and variable airflow obstruction [1]. It is characterized by variable symptoms of wheeze, shortness of breath, chest tightness and/or cough, and by variable expiratory airflow [2]. More than 300 million people are affected by asthma globally, and it is likely that by 2025 a further 100 million may be affected [3]. In the United Kingdom (UK), over 8 million people or approximately 12% of the population, have been diagnosed with asthma [4].
The long-term goals of asthma treatment are to control symptoms, and to minimize future risk of asthma associated with exacerbations, persistent airflow limitation, mortality, and asthma medication related adverse events [5]. Inhaled corticosteroids (ICS) with the addition of a second controller (e.g. a long-acting β2-agonist [LABA]) have been the mainstay of treatment of asthma [2, 6]. Current treatments allow achieving control in many patients with asthma, but most asthma patients are still sub-optimally managed and remain uncontrolled [7]. Uncontrolled asthma is defined by poor symptom control (frequent symptoms or reliever use, activity limited by asthma, or night waking due to asthma) and/or the occurrence of frequent or severe asthma exacerbations [2, 8, 9]. A cohort study from the UK reported that 35% and 46% of patients who initiated medium strength (MS) or high strength (HS) ICS-LABA remained uncontrolled, respectively. Similarly, 64% and 70% of patients with asthma who were already adherent to MS or HS ICS-LABA remain uncontrolled, respectively [10]. Uncontrolled asthma is associated with higher clinical burden (greater risk of symptoms, exacerbations, comorbidities and side-effects with corticosteroid use) [8, 11] and mortality than those with controlled asthma [12, 13]. The survival probability at 2 years for patients with uncontrolled asthma (92.0%) was lower than in the general population cohort (96.6%, relative risk of death: 2.35) and tended to be lower than in the controlled asthmatic cohort (94.3%; p = 0.07) [14]. In the UK, the direct and indirect costs of asthma treatment were projected to be £6.2 billion/year, and twice more in patients with uncontrolled compared to well-controlled asthma [15]. Uncontrolled asthma can also negatively impact patients’ health-related quality-of-life (HRQoL) (limitations due to physical health problems, vitality, social functioning and poor emotional wellbeing) [8, 16].
The widely used Global Initiative for Asthma (GINA) prevention and management strategy focuses on long-term treatment for symptom control and risk reduction. The strategy is based on a stepwise approach; step 1 (low dose ICS treatment for mild asthma) to step 5 (high dose ICS treatment for severe asthma) [2]. The GINA 2024 report [2] recommends the use of long-acting muscarinic antagonists (LAMA) as an add-on therapy for patients (aged ≥ 18 years) at Step 4–5, in those uncontrolled on MS or HS ICS/LABA. This can be achieved either by using separate inhalers or single-inhaler triple therapy (SITT) [17]. Several phase III clinical trials assessed the safety and efficacy of SITT in patients with uncontrolled asthma on double therapy [18–21]. Two clinical trials in patients with asthma that were uncontrolled despite treatment with a combination of MS ICS/LABA (TRIMARAN) or HS ICS/LABA (TRIGGER) assessed the efficacy and safety of SITT extrafine formulation i.e., combination of beclomethasone, formoterol, and glycopyrronium bromide (BDP/FOR/GLY, Trimbow®) versus BDP/FOR. In these two clinical trials, Trimbow® improved lung function and reduced exacerbations compared to double therapy (BDP/FOR) [21]. Other SITTs approved by the Food and Drug Administration (FDA) or European Medicines Agency (EMA) for patients with uncontrolled asthma are fluticasone, umeclidinium, and vilanterol HS (FF/UMEC/VI, Trelegy Ellipta®), and indacaterol acetate, glycopyrronium bromide, and mometasone HS (IND/GLY/MF, Enerzair Breezhaler®). Their phase III clinical trials (ARGON, CAPTAIN, and IRIDIUM) also demonstrated significantly higher efficacy versus double therapies in terms of lung function [18–21], but only favorable trends compared to double therapies were observed on moderate-to-severe exacerbations [22].
To date, despite evidence demonstrating the cost-effectiveness of SITTs over double therapies in patients with uncontrolled asthma [23, 24], there is limited evidence on the comparative cost-effectiveness of SITTs. This analysis aims to address this gap by comparing EMA or FDA-approved SITTs for the treatment of patients with uncontrolled asthma. Specifically, we evaluated the cost-effectiveness, in the UK setting, of BDP/FOR/GLY versus other SITTs (approved by EMA or FDA for the treatment of uncontrolled asthma).
Materials and methods
Model structure
A Markov cohort state-transition model based on previously published cost-effectiveness model in asthma [25] was developed to investigate the relative cost-effectiveness of BDP/FOR/GLY MS versus FF/UMEC/VI MS and BDP/FOR/GLY HS versus FF/UMEC/VI HS and IND/GLY/MF HS, in patients with asthma, uncontrolled by ICS/LABA. The structure used by Zafari et al. (2016) [25] was deemed the most suitable given the study objective, as it enabled a clearer differentiation of exacerbation states by categorizing patient health states by exacerbation-free and exacerbations with a varying degree of severity. This preserves a link to the data derived from the clinical trials [21] in directly modelling treatment effects, as well as simulating health state transitions within an established modelling paradigm. Additionally, patients within these recurrent exacerbation states were analyzed separately to account for variations in treatments, clinical outcomes, costs, and quality of life, thereby enhancing the credibility of the patient’s modeled clinical trajectory.
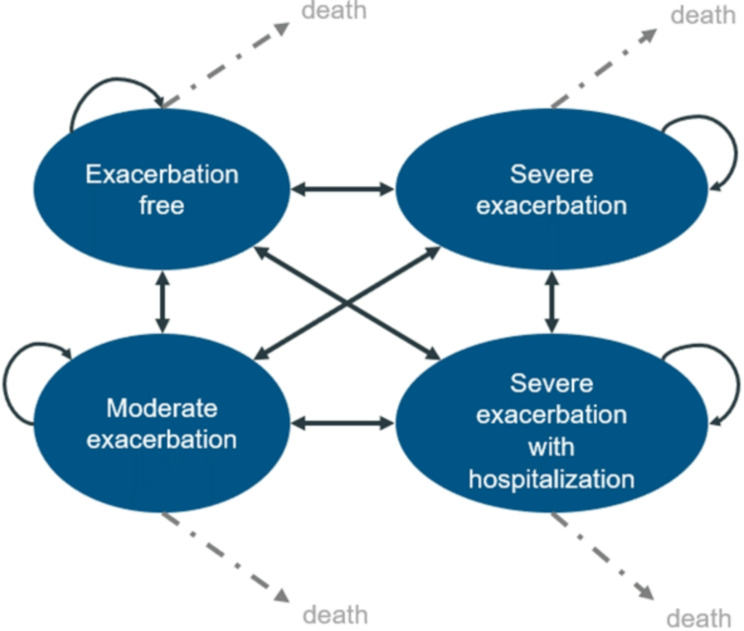
This decision-analytic model reflected the clinical course in asthma through five mutually exclusive health states: exacerbation-free state, three exacerbation states (‘moderate exacerbation’, ‘severe exacerbation without hospitalization’ and ‘severe exacerbation with hospitalization’), and death (Fig. 1). Asthma exacerbation was defined as per TRIGGER and TRIMARAN clinical studies [21] as acute or sub-acute episode of progressive worsening of at least one symptom of asthma (e.g., shortness of breath, cough, wheezing, or chest tightness). Moderate exacerbations were defined in accordance with the American Thoracic Society (ATS) and European Respiratory Society (ERS) joint statement [26]. Severe asthma exacerbation with hospitalization were defined as asthma-related hospitalization with/without prior emergency room (ER) visit, with/without intensive care unit (ICU) stay, and severe exacerbation without hospitalization were defined similarly but without an inpatient stay. These definitions were discussed with medical and health economic experts to ensure that they represent routine clinical practice in the National Health Service (NHS) England and were in line with other published asthma models [27–33].
Fig. 1.
Schematic of states in the Markov transition model
Treatment effect in the model was driven by the reduction in the frequency of asthma exacerbations, improved recovery from exacerbations, and precluding patients from worsening their exacerbation status. The model’s measurement of treatment effectiveness was based on the time a patient stays in the respective exacerbation health states for the modeled time horizon. Extrapolation of clinical treatment efficacy data was based on the average of transition probabilities at data collection time points within the 52-week follow-up period of the pivotal studies. Since the frequency of adverse events was low and comparable between the study groups in both trials [21], adverse events were excluded in the model. This is also in line with previous economic evaluations of severe asthma patients [27–33].
The cycle length was set at one week, with the lifetime horizon assumed to be 40 years.
Model inputs
Target population
The patient population included in the model was aligned with the approved indication for BDP/FOR/GLY [34] i.e., adult patients who experienced one or more exacerbations in the previous year and requiring regular treatment of asthma- not adequately controlled with MS or HS dose combination therapy (ICS/LABA). Cohort characteristics were informed from the TRIGGER and TRIMARAN [21].
Interventions and comparators
The model assessed the cost-effectiveness of BDP/FOR/GLY compared to other SITTs approved by the FDA or EMA for the treatment of uncontrolled asthma based on clinical practice currently adopted in England. BDP/FOR/GLY MS was compared to FF/UMEC/VI MS (only approved by the FDA, not EMA). BDP/FOR/GLY HS was compared with FF/UMEC/VI HS and IND/GLY/MF HS.
Clinical effectiveness
Transitions between health states for BDP/FOR/GLY MS and HS were informed by the TRIMARAN and TRIGGER studies (Table 1) [21]. A network meta-analysis (NMA) was performed and published previously to estimate the comparative efficacy of BDP/FOR/GLY SITT for patients with uncontrolled asthma despite treatment with MS or HS ICS/LABA [35]. The transition matrices for each comparator were calculated by multiplying the BDP/FOR/GLY MS and HS transition matrixes by the rate ratios (RRs) from this NMA [36]. The RRs were informed by the relative treatment efficacy of annualized rates of moderate exacerbations, and moderate-to-severe exacerbations from the NMA (Table 2) [35]. In the absence of RR outputs for severe exacerbation with hospitalization, the severe exacerbation RR was applied to severe exacerbation health states with/without hospitalization (Table 1). Transitions beyond the trials’ time horizon were extrapolated based on an average of transitions probabilities in week 24–40 and week 41–52 and remained constant over the model’s time horizon. All-cause mortality was applied to all health states but no excess asthma-related mortality was considered in the base case.
Table 1.
Summary of model inputs
| Transition probabilities | ||||||||
|---|---|---|---|---|---|---|---|---|
| BDP/FOR/GLY MS | BDP/FOR/GLY HS | |||||||
| To | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization |
| From | Week 0–4 | Week 0–4 | ||||||
| No exacerbation | 80.8% | 16.8% | 2.2% | 0.2% | 81.6% | 16.3% | 2.1% | 0.0% |
| Moderate exacerbation | 100.0% | 0.0% | 0.0% | 0.0% | 100.0% | 0.0% | 0.0% | 0.0% |
| Severe exacerbation | 100.0% | 0.0% | 0.0% | 0.0% | 100.0% | 0.0% | 0.0% | 0.0% |
| Severe exacerbation + hospitalization | 100.0% | 0.0% | 0.0% | 0.0% | 100.0% | 0.0% | 0.0% | 0.0% |
| To | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization |
| From | Week 5–12 | Week 5–12 | ||||||
| No exacerbation | 80.6% | 15.1% | 3.7% | 0.7% | 79.1% | 16.3% | 4.4% | 0.2% |
| Moderate exacerbation | 45.1% | 52.7% | 2.2% | 0.0% | 44.8% | 44.8% | 9.2% | 1.1% |
| Severe exacerbation | 66.7% | 8.3% | 25.0% | 0.0% | 72.7% | 9.1% | 18.2% | 0.0% |
| Severe exacerbation + hospitalization | 0.0% | 100.0% | 0.0% | 0.0% | 25.0% | 25.0% | 25.0% | 25.0% |
| To | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization |
| From | Week 13–26 | Week 13–26 | ||||||
| No exacerbation | 77.4% | 18.7% | 3.7% | 0.2% | 78.8% | 14.5% | 6.4% | 0.3% |
| Moderate exacerbation | 44.0% | 50.0% | 6.0% | 0.0% | 48.6% | 40.5% | 9.9% | 0.9% |
| Severe exacerbation | 52.4% | 33.3% | 14.3% | 0.0% | 31.0% | 27.6% | 37.9% | 3.4% |
| Severe exacerbation + hospitalization | 66.7% | 0.0% | 33.3% | 0.0% | 50.0% | 0.0% | 50.0% | 0.0% |
| To | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization |
| From | Week 27–40 | Week 27–40 | ||||||
| No exacerbation | 86.1% | 10.9% | 2.9% | 0.0% | 82.6% | 12.9% | 4.0% | 0.5% |
| Moderate exacerbation | 48.6% | 44.3% | 7.1% | 0.0% | 49.1% | 40.0% | 10.0% | 0.9% |
| Severe exacerbation | 30.8% | 42.3% | 26.9% | 0.0% | 54.2% | 29.2% | 16.7% | 0.0% |
| Severe exacerbation + hospitalization | 0.0% | 0.0% | 0.0% | 100.0% | 66.7% | 33.3% | 0.0% | 0.0% |
| To | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization |
| From | Week 41–52 | Week 41–52 | ||||||
| No exacerbation | 88.7% | 9.5% | 1.5% | 0.3% | 89.0% | 7.7% | 3.3% | 0.0% |
| Moderate exacerbation | 45.6% | 47.4% | 7.0% | 0.0% | 52.3% | 43.0% | 3.7% | 0.9% |
| Severe exacerbation | 50.0% | 17.9% | 28.6% | 3.6% | 58.8% | 17.6% | 23.5% | 0.0% |
| Severe exacerbation + hospitalization | 100.0% | 0.0% | 0.0% | 0.0% | 100.0% | 0.0% | 0.0% | 0.0% |
| To | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization |
| From | Average | Average | ||||||
| No exacerbation | 87.4% | 10.2% | 2.2% | 0.1% | 85.8% | 10.3% | 3.7% | 0.3% |
| Moderate exacerbation | 47.1% | 45.8% | 7.1% | 0.0% | 50.7% | 41.5% | 6.9% | 0.9% |
| Severe exacerbation | 40.4% | 30.1% | 27.7% | 1.8% | 56.5% | 23.4% | 20.1% | 0.0% |
| Severe exacerbation + hospitalization | 50.0% | 0.0% | 0.0% | 50.0% | 83.3% | 16.7% | 0.0% | 0.0% |
| Health state costs (per cycle) | ||||||||
| Cost | No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | ||||
| Inpatient cost | £0.06 | £0.00 | £4.88 | £1,278.21 | ||||
| Outpatient cost | £17.07 | £48.60 | £66.99 | £192.02 | ||||
| Home visits | £1.65 | £3.54 | £11.49 | £5.37 | ||||
| Laboratory tests | £7.02 | £40.49 | £20.34 | £32.70 | ||||
| Co-medication | £6.00 | £8.41 | £5.94 | £53.69 | ||||
|
Total (95% CI) |
£31.80 (£25.44-£38.17) |
£101.04 (£80.83-£121.25) |
£109.65 (£87.72-£131.57) |
£1,561.99 (£1,249.59-1,874.39) |
||||
| Health state utilities | ||||||||
| No exacerbation | Moderate exacerbation | Severe exacerbation | Severe exacerbation + hospitalization | |||||
|
Mean (95% CI) |
0.89 (0.80–0.98) |
0.65 (0.58–0.71) |
0.57 (0.51–0.63) |
0.33 (0.30–0.36) |
||||
CI: Confidence interval; BDP/FOR/GLY: Beclomethasone, Formoterol, and Glycopyrronium bromide; HS: High strength; MS: Moderate strength
Table 2.
Comparative efficacy based on network meta-analysis
| Outcome | RR (95% CI) | Interpretation |
|---|---|---|
| MS BDP/FOR/GLY vs. MS FF/UMEC/VI - Severe Asthma Exacerbations | 0.65 (0.49 to 0.89) | BDP/FOR/GLY MS is superior |
| MS BDP/FOR/GLY vs. MS FF/UMEC/VI - Moderate & Severe Asthma Exacerbations | 1.01 (0.86 to 1.2) | Numerically favourable for FF/UMEC/VI |
| HS BDP/FOR/GLY vs. HS FF/UMEC/VI - Severe Asthma Exacerbations | 0.97 (0.7 to 1.32) | Numerically favourable for BDP/FOR/GLY |
| HS BDP/FOR/GLY vs. HS FF/UMEC/VI - Moderate & Severe Asthma Exacerbations | 1.00 (0.84 to 1.2) | Treatments are equivalent |
|
HS BDP/FOR/GLY vs. HS IND/GLY/MF - Severe Asthma Exacerbations |
0.91 (0.7 to 1.19) | Numerically favourable for BDP/FOR/GLY |
|
HS BDP/FOR/GLY vs. HS IND/GLY/MF - Mod. & Severe Asthma Exacerbations |
0.99 (0.85 to 1.16) | Numerically favourable for BDP/FOR/GLY |
CI: Confidence interval; HS: High strength; MS: Medium strength; NMA: Network meta-analysis. BDP/FOR/GLY: Beclomethasone, Formoterol, and Glycopyrronium bromide; FF/UMEC/VI: Fluticasone, Umeclidinium, and Vilanterol; IND/GLY/MF; Indacaterol acetate, Glycopyrronium bromide, and Mometasone; RR: Rate ratio
Health state utilities
Previously validated exacerbation-related EuroQoL 5 dimensions (EQ-5D) quality-of-life data were used for the model. The mean utility values from Lloyd and colleagues [37] for the exacerbation-free, severe exacerbation without hospitalization, and severe exacerbation with hospitalization were 0.890, 0.570 and 0.330, respectively. Willson et al., [38] reported that since utility estimates for mild exacerbation were not found in literature, an assumption was made that this value was the average between the values for uncontrolled asthma and for a severe exacerbation without hospitalization. This estimated utility value of a ME (0.649) was very similar to those reported in the literature for outpatient-managed exacerbations (0.65–0.67) (Table 1).
Healthcare resource utilization
Healthcare resource utilization (HCRU) associated with any of the model health states was informed by Willson et al., study (2014) [38], which used a survey of 15 HCPs from England, five general practitioners (GPs), five respiratory nurses and five asthma specialists, conducted in early 2013.
The HCRU of exacerbation states was directly applicable from the Willson et al., 2014 [38]. HCRU of ‘exacerbation-free’ health state was estimated as an average of HCRU for ‘controlled asthma’, ‘partially controlled asthma’ and ‘uncontrolled asthma’ health state from Willson et al., 2014 [38]. Distribution of patients among these states was informed based on ACQ-6 of BDP/FOR/GLY studies [21]. Total costs per cycle for each health state were estimated by weighing unit costs with the corresponding probability of resource utilization within each health state (Table 1).
Unit costs
The model takes the perspective of the England NHS and Prescribed Specialised Services (PSS) expenditure, in which only healthcare costs were assessed. Costs were expressed in 2022 prices (latest available at the time). The unit costs used to value resource use were obtained from published sources such as the Unit Costs of Health and Social Care 2022 [39], the National Schedule of Reference Costs 2021/22 [40], and the British National Formulary (BNF) 2024 [41].
The unit costs included intervention, comparator therapy, co-medication for asthma therapy costs. Inpatient (ER admission and ambulance use) and outpatient (GP, nurse, asthma specialists) costs were sourced from the NHS reference costs and PSS, respectively. Other unit costs (home visits [GP, nurse], laboratory tests and procedures [flu vaccination, spirometry, desensitization] and hospitalizations [ER visit, inpatient admission, ICU, ambulance]) were sourced from the published literature and were inflated to reflect 2022 prices (see Additional file 1).
Discounting
Consistent with the National Institute for Health and Care Excellence guidance (NICE) [42], an annual discount rate of 3.5% was applied to both costs and health effects (life-years [LYs] and quality-adjusted life-years [QALYs]) after the first year.
Analysis method
In addition to the base case analysis (as described in the ‘model structure’ section), sensitivity analyses were carried out to assess the robustness of the conclusions when the values of key variables, which are uncertain, are altered.
Sensitivity analysis
The base case analysis of the cost-effectiveness analysis (CEA) used the mean expected values of all input variables (deterministic model).
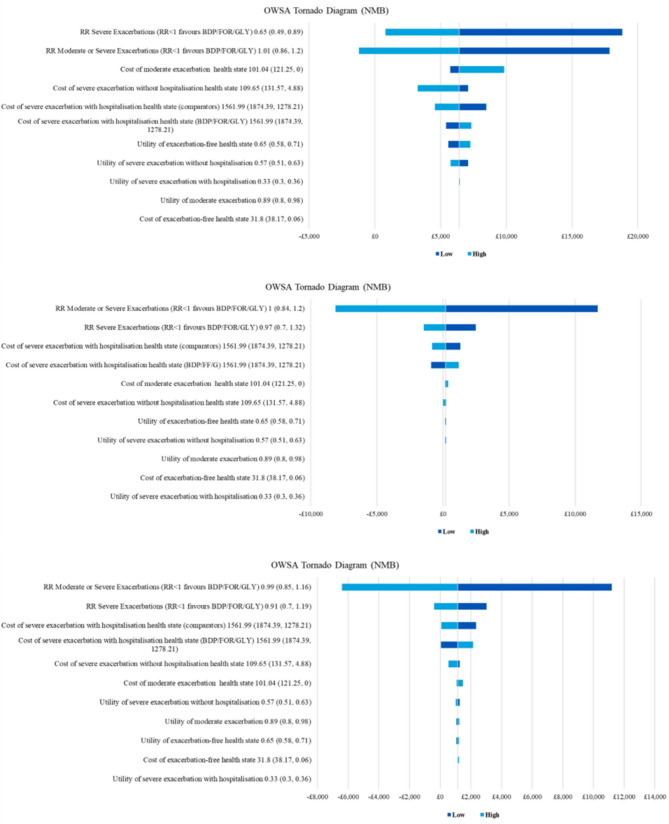
Sensitivity analyses were carried out to assess the robustness of the conclusions when the values of key variables, which are uncertain, are altered. One-way sensitivity analysis (OWSA) was conducted for each treatment comparison to test the impact on cost-effectiveness of uncertainty in the input parameters, based on the upper and lower 95% CIs of each value. In deterministic OWSA, variables such as RR of severe and moderate exacerbation, total costs per health state, and utilities were tested. The OWSA tornado is presented in terms of net monetary benefit (NMB) to avoid misinterpretation of results, as comparison in many cases results in negative incremental cost-effectiveness ratio (ICER) for BDP/FOR/GLY (e.g., more effective, less costly).
Probabilistic sensitivity analysis (PSA) was conducted to estimate the impact of joint uncertainty around key model input parameters on the probability of BDP/FOR/GLY being cost-effective, with 5,000 iterations. In the PSA, model input variables were regarded as stochastic, and their values were sampled from relevant distributions. Results of the PSA were presented in the form of scatter plots and cost-effectiveness acceptability curves (CEAC). For the PSA, uncertainty in RRs was modeled using convergence diagnostics and output analysis (CODA) values from the NMA rather than assuming a lognormal distribution informed by the summary statistic outputs of the PSA. The uncertainty of cost estimates was captured by uncertainties in resource use, assuming fixed unit costs for the resources. The lognormal distribution was used to represent the uncertainty in parameters describing outpatient visits, home visits and laboratory services, while parameters related to the use of co-medications and inpatient admissions were sampled using beta distribution. Health related utilities were simulated with the beta distribution.
Scenario analysis
Three alternative scenarios were considered to assess the impact of uncertainty in various parameters on the cost-effectiveness of BDP/FOR/GLY. Scenario 1 explored the impact of assuming that exacerbation probabilities in the extrapolated period are based on the last visit from TRIGGER and TRIMARAN studies. Scenario 2 analyzed the assumption that patients with a severe exacerbation that do not require hospitalization always visit accident and emergency department (A&E) (i.e., probability of A&E = 1), and that inpatient costs for a moderate exacerbation are equal to inpatient costs for the exacerbation-free health state (i.e., £0.06 per cycle, instead of no cost). Lastly, Scenario 3 explored the mortality risk due to severe exacerbation with hospitalization.
Results
Deterministic base case
BDP/FOR/GLY MS vs. FF/UMEC/VI MS
In the base case, BDP/FOR/GLY MS was found to be £5,121 less costly and showed marginal improvement in QALYs (0.065) compared to FF/UMEC/VI MS. With higher QALYs and lower total costs, BDP/FOR/GLY resulted in being a dominant treatment alternative against FF/UMEC/VI MS at a willingness-to-pay (WTP) threshold of £20,000 per QALY gained (Table 3). (Add Table 3here)
Table 3.
Base case cost-effectiveness analysis (medium strength and high strength)
| Therapy | Costs | LYs | QALYs | ICER (cost/QALY) | NMB (QALYs) |
|---|---|---|---|---|---|
| Medium strength | |||||
| BDP/FOR/GLY MS | £59,912 | 18.38 | 15.31 | ||
| FF/UMEC/VI MS | £65,032 | 18.38 | 15.24 | ||
| Incremental | -£5,121 | - | 0.065 | BDP/FOR/GLY Dominant | £6,420 |
| High strength | |||||
| BDP/FOR/GLY HS | £60,202 | 18.38 | 15.30 | - | - |
| FF/UMEC/VI HS | £60,345 | 18.38 | 15.30 | - | - |
| Incremental | -£143 | - | 0.003 | BDP/FOR/GLY Dominant | £195 |
| IND/GLY/MF HS | £60,894 | 18.38 | 15.28 | ||
| Incremental | -£692 | - | 0.023 | BDP/FOR/GLY Dominant | £1,150 |
HS: High strength; ICER: Incremental cost-effectiveness ratio; LY: Life-years; MS: Medium strength; NMB: Net monetary benefit; QALY: quality-adjusted life year. BDP/FOR/GLY: Beclomethasone, Formoterol, and Glycopyrronium bromide; FF/UMEC/VI: Fluticasone, Umeclidinium, and Vilanterol; IND/GLY/MF; Indacaterol acetate, Glycopyrronium bromide, and Mometasone
BDP/FOR/GLY HS vs. FF/UMEC/VI HS and IND/GLY/MF HS
BDP/FOR/GLY HS was found to be £143 less costly and marginally improved the QALYs (0.003) compared to FF/UMEC/VI HS. Similarly, BDP/FOR/GLY HS was £692 less costly and marginally improved the QALYs (0.023) compared to IND/GLY/MF HS. With higher QALYs and lower total costs, BDP/FOR/GLY HS was projected to be a dominant treatment alternative against FF/UMEC/VI HS and IND/GLY/MF HS at a WTP threshold of £20,000 per QALY gained (Table 3).
Sensitivity analysis
BDP/FOR/GLY MS vs. FF/UMEC/VI MS
The highest impact on the Net Monetary Benefit (NMB) value was seen for RR for severe and moderate/severe exacerbations, ranging from £802 to £18,834. However, BDP/FOR/GLY MS remains cost-effective in most univariate analyses (NMB>£0) except for upper values the RR of moderate or severe exacerbations. The model was much less sensitive to the costs and utilities of health states, however variables tested in the OWSA were limited (Fig. 2a).
Fig. 2.
Tornado diagrams (NBM). a. BDP/FOR/GLY MS vs. FF/UMEC/VI MS. b. BDP/FOR/GLY HS vs. FF/UMEC/VI HS. c. BDP/FOR/GLY HS vs. IND/GLY/MF HS. HS: High strength; MS: Medium strength; NBM: Net monetary benefit. BDP/FOR/GLY: Beclomethasone, Formoterol, and Glycopyrronium bromide; FF/UMEC/VI: Fluticasone, Umeclidinium, and Vilanterol; IND/GLY/MF; Indacaterol acetate, Glycopyrronium bromide, and Mometasone; RR: Rate ratio
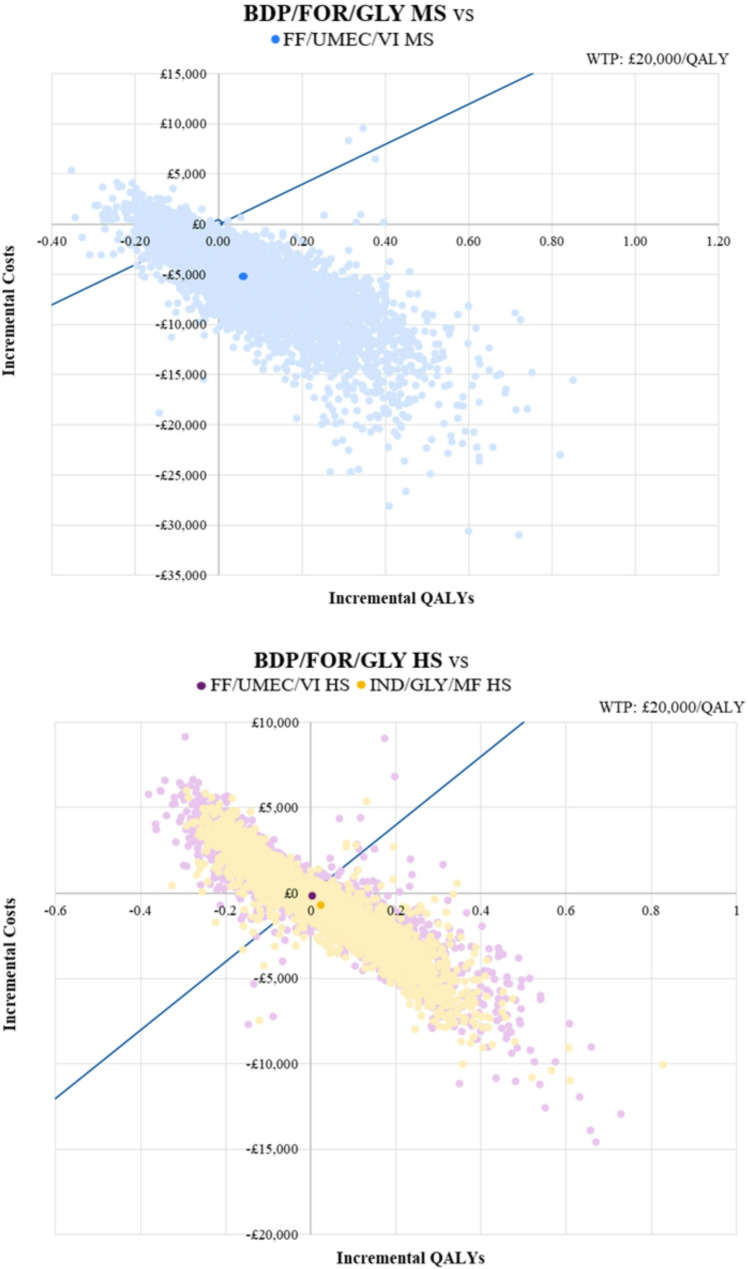
Differences in costs and health outcomes between the two drugs resulting from each simulation are presented in a cost-effectiveness plane and cost-effectiveness acceptability curve. The PSA analysis shows that most of observations fall in the South-East (70.8%) and South-West (24.3%) quadrants of the scatterplot (Fig. 3a). The probability of BDP/FOR/GLY MS is cost-effective in 77.1% of cases as compared to FF/UMEC/VI MS, at the defined WTP threshold of £20,000 per QALY gained (see Additional file 2).
Fig. 3.
Cost-effectiveness plane. a) BDP/FOR/GLY MS vs. FF/UMEC/VI MS. b) BDP/FOR/GLY HS vs. FF/UMEC/VI HS and IND/GLY/MF HS. HS: High strength; MS: Medium strength; PSA: Probabilistic sensitivity analysis QALY: quality-adjusted life year; WTP: Willingness-to-pay. BDP/FOR/GLY: Beclomethasone, Formoterol, and Glycopyrronium bromide; FF/UMEC/VI: Fluticasone, Umeclidinium, and Vilanterol; IND/GLY/MF; Indacaterol acetate, Glycopyrronium bromide, and Mometasone
BDP/FOR/GLY HS vs. FF/UMEC/VI HS
The cost-effectiveness of BDP/FOR/GLY HS versus FF/UMEC/VI HS was most sensitive to the RR for moderate or severe exacerbations varying from -£8,138 to £11,710. The OWSA showed substantial variation around the base case estimate of NMB (for a WTP £20,000 per QALY gained), with the decision on the cost-effectiveness of BDP/FOR/GLY being affected in four univariate analyses (Fig. 2b).
The PSA analysis shows that most of observations fall in the South-East (45.4%) and North-West (44.9%) quadrants of the scatterplot (Fig. 3b). The probability of BDP/FOR/GLY HS is cost-effective in 51.3% of cases as compared to FF/UMEC/VI HS, at the defined WTP threshold of £20,000 per QALY gained (see Additional file 2).
BDP/FOR/GLY HS vs. IND/GLY/MF HS
The cost-effectiveness of BDP/FOR/GLY HS versus IND/GLY/MF HS was most sensitive to the RR for moderate or severe exacerbations with results varying from -£6,411 and £11,196. Results of the OWSA showed that there was substantial variation around the base case NMB estimate (for a WTP of £20,000 per QALY gained), which was related to the treatment effect of BDP/FOR/GLY HS versus IND/GLY/MF HS (Fig. 2c). (Add Fig. 2c here)
The PSA analysis shows that most of observations fall in the South-East (55.1%) and North-West (34.1%) quadrants of the scatterplot (Fig. 3b). The probability of BDP/FOR/GLY HS is cost-effective in 61.2% of cases as compared to IND/GLY/MF HS, at the defined WTP threshold of £20,000 per QALY gained (see Additional file 2).
Scenario analyses
The scenario analyses showed the base case deterministic results are robust to structural and input uncertainty. In all three scenarios MS and HS BDP/FOR/GLY remained dominant versus FF/UMEC/VI MS, FF/UMEC/VI HS, and IND/GLY/MF HS (Table 4).
Table 4.
Incremental cost, life-years and QALYs, and icers for the three scenario analyses
| Scenarios | Incremental Costs | Incremental QALYs | ICER (cost/QALY) | NMB (QALYs) |
|---|---|---|---|---|
| Scenario 1: Exploration assumption | ||||
| BDP/FOR/GLY MS vs. FF/UMEC/VI MS | -£3,919 | 0.019 | BDP/FOR/GLY Dominant | £4,302 |
| BDP/FOR/GLY HS vs. FF/UMEC/VI HS | -£40 | 0.002 | BDP/FOR/GLY Dominant | £78 |
| BDP/FOR/GLY HS vs. IND/GLY/MF HS | -£343 | 0.018 | BDP/FOR/GLY Dominant | £711 |
| Scenario 2: Probability of ER visit in severe exacerbation without hospitalization | ||||
| BDP/FOR/GLY MS vs. FF/UMEC/VI MS | -£7,500 | 0.065 | BDP/FOR/GLY Dominant | £8,800 |
| BDP/FOR/GLY HS vs. FF/UMEC/VI HS | -£281 | 0.003 | BDP/FOR/GLY Dominant | £333 |
| BDP/FOR/GLY HS vs. IND/GLY/MF HS | -£1,145 | 0.023 | BDP/FOR/GLY Dominant | £1,603 |
| Scenario 3: Including asthma excess mortality | ||||
| BDP/FOR/GLY MS vs. FF/UMEC/VI MS | -£1,502 | 0.820 | BDP/FOR/GLY Dominant | £17,909 |
| BDP/FOR/GLY HS vs. FF/UMEC/VI HS | -£43 | 0.026 | BDP/FOR/GLY Dominant | £566 |
| BDP/FOR/GLY HS vs. IND/GLY/MF HS | -£343 | 0.100 | BDP/FOR/GLY Dominant | £2,337 |
HS: High strength; ICER: Incremental cost-effectiveness ratio; LY: Life years; MS: Medium strength; NMB: Incremental net monetary benefit QALY: quality-adjusted life year. BDP/FOR/GLY: Beclomethasone, Formoterol, and Glycopyrronium bromide; FF/UMEC/VI: Fluticasone, Umeclidinium, and Vilanterol; IND/GLY/MF; Indacaterol acetate, Glycopyrronium bromide, and Mometasone
Discussion
Several therapies are available for asthma management, despite that a significant proportion of patients with moderate-to-severe asthma do not achieve adequate control [2]. Nonadherence to treatment, the use of multiple inhalers, and errors in inhaler handling are some of the factors associated with uncontrolled asthma; therefore, improving treatment adherence could improve asthma control. In patients who are already receiving triple therapy via different devices, switching to fixed dose combination (FDC) in a single inhalation device (SITTs) could confer benefits by reducing critical errors in the management of inhalers, improving therapeutic adherence, and lowering costs, while maintaining the same clinical efficacy [43]. Furthermore, to achieve symptom control, it is essential to access both the lung’s large and small airways. The dimensions and structure of the bronchial lumen make it challenging for treatments to accomplish this. Inhaled extrafine SITTs are capable of treating the entire bronchial tree including the small airways more effectively than other conventional therapies [44].
Recently, a few SITTs have been marketed and the GINA recommend adding a LAMA in asthma patients who still experience symptoms or exacerbations, despite treated with MS or HS ICS/LABA [2]. Multiple phase III clinical trials (TRIMARAN, TRIGGER, IRIDIUM, ARGON, and CAPTAIN) have confirmed therapeutic benefits of SITTs in this patient population [18–21]. TRIMARAN and TRIGGER were the first trials to evaluate the efficacy and safety of an extrafine SITT, Trimbow® (BDP/FOR/GLY), in the management of moderate-to-severe asthma versus double therapy, Fostair® (beclomethasone dipropionate/formoterol fumarate). The clinical outcomes of these two trials were used to assess the cost-effectiveness of BDP/FOR/GLY versus Fostair® by Orlovic et al., [23]. This was the first CEA of BDP/FOR/GLY in uncontrolled asthma patients based on a NHS England perspective. The current analysis builds on the previous study by Orlovic et al., [23] and utilizes outcomes from the recently published NMA [35] to assess the cost-effectiveness of BDP/FOR/GLY compared to other SITTs (FF/UMEC/VI MS and HS, and IND/GLY/MF HS).
In the base case, BDP/FOR/GLY MS was projected to be a dominant treatment alternative against FF/UMEC/VI MS i.e., BDP/FOR/GLY lead to increased QALYs (more effective at producing health benefits in terms of reduced exacerbations), and net cost savings. Similarly, BDP/FOR/GLY HS was projected to be a dominant treatment alternative against FF/UMEC/VI MS and HS, and IND/GLY/MF HS. The OWSA results showed that there was substantial variation around the NMB estimate (for a WTP of £20,000 per QALY gained) related to the relative treatment effect of BDP/FOR/GLY versus other comparators and costs associated with moderate-to-severe exacerbations. The various scenarios also illustrated the impact of uncertainty on the CEA, although importantly both MS and HS BDP/FOR/GLY remained dominant compared with MS and HS SITTs in all three scenarios. In the PSA, BDP/FOR/GLY MS had a 71% likelihood to be a dominant strategy versus FF/UMEC/VI MS at a WTP threshold of £20,000 per QALY gained. Conversely, the probability of BDP/FOR/GLY HS being dominated by FF/UMEC/VI and IND/GLY/MF HS at a WTP threshold of £20,000 per QALY gained was 45% and 34% respectively. The uncertainty can be attributed to the relative treatment efficacy informed by indirect treatment comparison methods.
These findings are in line with results of CEA by Orlovic et al., [23] which used a similar model to that used in our analysis. As with our base case analysis, the model by Orlovic et al., [23] did not explicitly account for the costs of adverse events or loss of productivity. Another study by Mtibaa et al., [45] compared the cost-effectiveness of IND/GLY/MF SITT with ICS/LABA + TIO in Canada. The study population were patients whose asthma remained uncontrolled despite treatment with HS ICS/LABA. In the base case analysis, SITT was found to be less costly and more effective and had the highest probability of being cost-effective at WTP threshold compared with ICS/LABA + TIO.
The results of our study must be interpreted with caution considering a few limitations. Major limitations of this analysis, which are inherited by most of the CEAs, is the use of clinical efficacy inputs from trial data. Assumptions had to be made when extrapolating the data beyond the trial duration, which was a 52-week period, including that the treatment effect would continue to be observed. To assess the cost-effectiveness of BDP/FOR/GLY over a lifetime horizon, extrapolations of the trial data were required to predict movements between health states, introducing a degree of uncertainty into the model, which could not be validated against the trial data. Health state resource utilization was deduced from published literature, which may have provided outdated information, and was therefore validated by one England-based clinical expert. Nonetheless, base case deterministic results are robust to structural and input uncertainties, which was demonstrated by running scenario analysis, addressing these limitations. Additional limitations of the study are due to the Markov model not considering any improvements in either lung function or overall asthma control and to the assumption that all patients persisted with treatments throughout the lifetime horizon at the adherence rates observed in the clinical trials. The impact of poor adherence, treatment discontinuation and treatment switching in real-world were not investigated in the model due to a lack of robust evidence. These limitations can be addressed by ongoing research to confirm the long-term treatment effects and adherence of BDP/FOR/GLY in moderate-to-severe asthma patients [46]. An ongoing multicenter, multicountry, prospective study TriMaximize aims to provide the characteristics, therapy pathways and health-related outcomes of patients with moderate-to-severe asthma prescribed with BDP/FOR/GLY extrafine SITT in real-world practice [46]. Lastly the results for FF/UMEC/VI MS and HS are based on the list price for FF/UMEC/VI in chronic obstructive pulmonary disorder (COPD) as it was not yet approved in asthma. FF/UMEC/VI HS was assumed to be at flat price with FF/UMEC/VI MS and hence should be interpreted with caution.
Conclusion
In the base case analysis, BDP/FOR/GLY MS and HS were dominant compared with FF/UMEC/VI MS and HS and IND/GLY/MF HS in patients with asthma uncontrolled by ICS/LABA, at a standard WTP threshold of £20,000 per QALY gained from the NHS England perspective. Furthermore, sensitivity and scenario analyses conducted by varying different parameters confirmed the robustness of the base case results. The comparisons between BDP/FOR/GLY HS versus FF/UMEC/VI HS and IND/GLY/MF HS were characterized by some uncertainty, related to the relative treatment efficacy informed by the indirect treatment comparison methods. The results of our study could provide a reference for the economic impact of BDP/FOR/GLY from the publicly funded health payer perspective in England and may be generalizable to other health care systems with comparable pricing structures.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1: Unit costs (medication and HCRU). Description of data: Input values including unit costs of medications and health care resource utilization
Supplementary Material 2: Cost-effectiveness acceptability curves. Description of data: Probabilistic sensitivity analysis results in the form of cost-effectiveness acceptability curves
Acknowledgements
This study was supported by Chiesi Farmaceutici S.p.A., Via Palermo 26A, 43122, Parma, Italy.IV, SD, AM and MD are employees of Chiesi Farmaceutici SpA, the funder of this work. YS, DT and IG were employees of IQVIA at the time of development of this study, and IQVIA received consulting fees from Chiesi Farmaceutici S.p.A. to support the manuscript development. All the authors agreed to the manuscript’s final version.
Author contributions
YS and DT contributed towards conception of the study, acquisition of data, analysis, and interpretation of data, they drafted the manuscript and reviewed it critically for intellectual content. IV, SD, AM, MD and IG contributed to the conception of the study and the methodology, they critically reviewed the manuscript and provided constructive feedback to its design and layout. All the authors agreed to the manuscript’s final version. IV submitted the study and has taken responsibility for the overall content. All the authors agreed to the manuscript’s final version.
Funding
This study was supported by Chiesi Farmaceutici S.p.A., Via Palermo 26 A, 43122, Parma, Italy.
Data availability
The data supporting the conclusions of this article is included within the article and/or based on publication currently ‘in press’ and available if requested by the editorial office.
Declarations
Competing interests
The authors declare no competing interests.
Previous presentations
The paper is original, nor all neither any of its parts have been reproduced in other publications.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ukena D, Fishman L, Niebling WB. Bronchial asthma: diagnosis and long-term treatment in adults. Dtsch Arztebl Int. 2008;105(21):385–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.GINA. Global strategy for asthma management and prevention. 2024.
- 3.World Health Organization. Asthma: key facts. 2023 30 Nov 2023]; Available from: https://www.who.int/news-room/fact-sheets/detail/asthma
- 4.National Institute for Health and Care Excellence (NICE). Prevalence and background information on asthma. 2023 30 Nov 2023]; Available from: https://www.nice.org.uk/cks-uk-only#:~:text=Prognosis-,What is the prevalence of asthma%3F,have been diagnosed with asthma
- 5.UptoDate. An overview of asthma management. 2023 4 Dec 2023]; Available from: https://www.uptodate.com/contents/an-overview-of-asthma-management
- 6.Alangari AA. Corticosteroids in the treatment of acute asthma. Ann Thorac Med. 2014;9(4):187–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lingner H, et al. What patients really think about asthma guidelines: barriers to guideline implementation from the patients’ perspective. BMC Pulm Med. 2017;17(1):13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Busse WW, Kraft M. Current unmet needs and potential solutions to uncontrolled asthma. Eur Respiratory Rev. 2022;31(163):210176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chung KF, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343–73. [DOI] [PubMed] [Google Scholar]
- 10.Buhl R, et al. One-year follow up of asthmatic patients newly initiated on treatment with medium- or high-dose inhaled corticosteroid-long-acting β(2)-agonist in UK primary care settings. Respir Med. 2020;162:105859. [DOI] [PubMed] [Google Scholar]
- 11.Kostakou E et al. Acute severe asthma in adolescent and adult patients: current perspectives on assessment and management. J Clin Med. 2019;8(9). [DOI] [PMC free article] [PubMed]
- 12.Royal College of Physicians. Why asthma still kills: The National Review of Asthma Deaths (NRAD). 2014.
- 13.Gullach AJ, et al. Sudden death in young persons with uncontrolled asthma - a nationwide cohort study in Denmark. BMC Pulm Med. 2015;15(1):35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Roche N, et al. Real-life impact of uncontrolled severe asthma on mortality and healthcare use in adolescents and adults: findings from the retrospective, observational RESONANCE study in France. BMJ Open. 2022;12(8):e060160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pavord ID, et al. The impact of poor asthma control among asthma patients treated with inhaled corticosteroids plus long-acting β(2)-agonists in the United Kingdom: a cross-sectional analysis. NPJ Prim Care Respir Med. 2017;27(1):17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.CDC. Uncontrolled asthma among adults. 2019 30 Nov 2023]; Available from: https://www.cdc.gov/asthma/asthma_stats/uncontrolled-asthma-adults-2019.htm
- 17.Agusti A, et al. Single inhaler triple therapy (SITT) in asthma: systematic review and practice implications. Allergy. 2022;77(4):1105–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gessner C, et al. Fixed-dose combination of indacaterol/glycopyrronium/mometasone furoate once-daily versus salmeterol/fluticasone twice-daily plus tiotropium once-daily in patients with uncontrolled asthma: a randomised, phase IIIb, non-inferiority study (ARGON). Respir Med. 2020;170:106021. [DOI] [PubMed] [Google Scholar]
- 19.Kerstjens HAM, et al. Once-daily, single-inhaler mometasone-indacaterol-glycopyrronium versus mometasone-indacaterol or twice-daily fluticasone-salmeterol in patients with inadequately controlled asthma (IRIDIUM): a randomised, double-blind, controlled phase 3 study. Lancet Respir Med. 2020;8(10):1000–12. [DOI] [PubMed] [Google Scholar]
- 20.Lee LA, et al. Efficacy and safety of once-daily single-inhaler triple therapy (FF/UMEC/VI) versus FF/VI in patients with inadequately controlled asthma (CAPTAIN): a double-blind, randomised, phase 3A trial. Lancet Respir Med. 2021;9(1):69–84. [DOI] [PubMed] [Google Scholar]
- 21.Virchow JC, et al. Single inhaler extrafine triple therapy in uncontrolled asthma (TRIMARAN and TRIGGER): two double-blind, parallel-group, randomised, controlled phase 3 trials. Lancet. 2019;394(10210):1737–49. [DOI] [PubMed] [Google Scholar]
- 22.Papi A, et al. Inhaled long-acting muscarinic antagonists in asthma - a narrative review. Eur J Intern Med. 2021;85:14–22. [DOI] [PubMed] [Google Scholar]
- 23.Orlovic M, et al. Cost-effectiveness of single-inhaler extrafine beclometasone dipropionate/formoterol fumarate/glycopyrronium in patients with uncontrolled asthma in England. Respir Med. 2022;201:106934. [DOI] [PubMed] [Google Scholar]
- 24.Buendia JA, Patino DG. Cost-utility of triple versus dual inhaler therapy in moderate to severe asthma. BMC Pulm Med. 2021;21(1):398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zafari Z, et al. Cost-effectiveness of bronchial thermoplasty, omalizumab, and standard therapy for moderate-to-severe allergic asthma. PLoS ONE. 2016;11(1):e0146003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Virchow JC, et al. Defining moderate asthma exacerbations in clinical trials based on ATS/ERS joint statement. Respir Med. 2015;109(5):547–56. [DOI] [PubMed] [Google Scholar]
- 27.Campbell JD, Spackman DE, Sullivan SD. The costs and consequences of omalizumab in uncontrolled asthma from a USA payer perspective. Allergy. 2010;65(9):1141–8. [DOI] [PubMed] [Google Scholar]
- 28.Paltiel AD, et al. Cost-effectiveness of inhaled corticosteroids in adults with mild-to-moderate asthma: results from the asthma policy model. J Allergy Clin Immunol. 2001;108(1):39–46. [DOI] [PubMed] [Google Scholar]
- 29.van Nooten F, et al. Cost-effectiveness of omalizumab for uncontrolled allergic asthma in the Netherlands. J Med Econ. 2013;16(3):342–8. [DOI] [PubMed] [Google Scholar]
- 30.Wu AC, et al. Cost-effectiveness of Omalizumab in adults with severe asthma: results from the asthma policy model. J Allergy Clin Immunol. 2007;120(5):1146–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Oba Y, Salzman GA. Cost-effectiveness analysis of omalizumab in adults and adolescents with moderate-to-severe allergic asthma. J Allergy Clin Immunol. 2004;114(2):265–9. [DOI] [PubMed] [Google Scholar]
- 32.Dewilde S, et al. The economic value of anti-IgE in severe persistent, IgE-mediated (allergic) asthma patients: adaptation of INNOVATE to Sweden. Curr Med Res Opin. 2006;22(9):1765–76. [DOI] [PubMed] [Google Scholar]
- 33.Brown R, et al. Cost-effectiveness of omalizumab in patients with severe persistent allergic asthma. Allergy. 2007;62(2):149–53. [DOI] [PubMed] [Google Scholar]
- 34.European Medicines Agency. Trimbow (beclometasone / formoterol / glycopyrronium bromide). 2019 23 Nov 2023]; Available from: https://www.ema.europa.eu/en/documents/overview/trimbow-epar-medicine-overview_en.pdf
- 35.Braido F, Vlachaki I, Nikolaidis GF, et al. Single inhaler with beclometasone, formoterol, and glycopyrronium versus triple therapies in adults with uncontrolled asthma: a systematic review and meta-analysis. Sci Rep. 2025;15:4191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Orlovic M, et al. EE556 beclometasone/formoterol/glycopyrronium vs. Fixed and open combinations of inhaled corticosteroids, long-acting B2-agonist, and long-Acting muscarinic antagonist in adults with uncontrolled asthma: a network meta-analysis. Value Health. 2023;26:S159–60. [Google Scholar]
- 37.Lloyd A, Price D, Brown R. The impact of asthma exacerbations on health-related quality of life in moderate to severe asthma patients in the UK. Prim Care Respir J. 2007;16(1):22–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Willson J, et al. Cost effectiveness of tiotropium in patients with asthma poorly controlled on inhaled glucocorticosteroids and long-acting β-agonists. Appl Health Econ Health Policy. 2014;12(4):447–59. [DOI] [PubMed] [Google Scholar]
- 39.Personal Social Sevices Research Unit. Unit costs of health and social care. 2022 19 Jan 2024]; Available from: https://www.pssru.ac.uk/unitcostsreport/
- 40.National Health Service-UK. 2021/22 National cost collection data publication. 2023 19 Jan 2024]; Available from: https://www.england.nhs.uk/publication/2021-22-national-cost-collection-data-publication/
- 41.British National Formulary (BNF). Medicines complete. 2024 07 Feb 2024]; Available from: https://about.medicinescomplete.com/publication/british-national-formulary/
- 42.National Institute for Health and Care Excellence (NICE). Guide to the methods of technology appraisal 2013. 2013 23 Nov 2023]; Available from: https://www.nice.org.uk/process/pmg9/chapter/foreword [PubMed]
- 43.Al-Moamary MS, et al. When single-inhaler triple therapy is a preferred option in asthma management? Ann Thorac Med. 2022;17(4):185–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Díaz-García R, Flores-Ramírez G, Ramírez-Oseguera RT. Effect of extrafine formulation of BDP/FF inhaler on asthma control, small airway function and airway inflammation among Mexican asthmatic patients. A retrospective analysis. Respir Med. 2020;165:105932. [DOI] [PubMed] [Google Scholar]
- 45.Mtibaa M, et al. Cost-effectiveness of once-daily, single-inhaler indacaterol acetate/ glycopyrronium bromide/ mometasone furoate in patients with uncontrolled moderate-to-severe asthma in Canada. ClinicoEconomics Outcomes Res. 2021;13(null):957–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gessner C, et al. Impact of extrafine formulation single-inhaler triple therapy on lung function after three months of treatment in patients with asthma: the trimaximize study. Eur Respir J. 2023;62(suppl 67):PA2379. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material 1: Unit costs (medication and HCRU). Description of data: Input values including unit costs of medications and health care resource utilization
Supplementary Material 2: Cost-effectiveness acceptability curves. Description of data: Probabilistic sensitivity analysis results in the form of cost-effectiveness acceptability curves
Data Availability Statement
The data supporting the conclusions of this article is included within the article and/or based on publication currently ‘in press’ and available if requested by the editorial office.