Abstract
Spontaneous resorption of lumbar disk herniation (LDH) presents a promising avenue for the non-surgical management of herniated disks. Here we present a 40-year-old female with severe L5/S1 herniation who experienced spontaneous resorption confirmed by MRI. The patient adhered to a comprehensive non-operative treatment regimen comprising NSAIDs, heat therapy, massage therapy, acupuncture, and kinesitherapy. Over two years, the patient showed substantial clinical improvement, with significant pain reduction and MRI evidence of disk resorption, resulting in a marked decrease in their visual analog scale (VAS) pain scores. Through a systematic review of existing literature, we identify that spontaneous resorption is associated with mechanisms such as inflammation, neovascularization, macrophage infiltration, matrix degradation, disruption of immune privilege, apoptosis and autophagy, and disc dehydration. Clinical predictors for spontaneous resorption of LDH include the size, type and composition of the herniation, rim enhancement on contrast-enhanced MRI, and involvement of the posterior longitudinal ligament (PLL). Future research should focus on elucidating the molecular mechanisms of resorption, regulation of inflammatory response, macrophage polarization, matrix degradation, immune privilege and neovascularization, developing advanced imaging techniques to predict resorption potential, and exploring personalized treatment strategies based on machine learning and deep learning prediction models.
Keywords: Lumbar disk herniation, Spontaneous resorption, Non-surgical management, Conservative treatment, Case report
Lumbar disc herniation (LDH) is a common cause of low back pain, radiculopathy, and functional disability. Recent studies have increasingly demonstrated the phenomenon of spontaneous resorption, where herniated disc material shrinks or disappears without surgical intervention [1, 2]. Spontaneous resorption of lumbar disk herniation (LDH) is a phenomenon that has garnered attention due to its implications for non-surgical management in patients with herniated disks [3, 4]. Here, we a patient that underscore the possibility and potential of this non-invasive approach, highlighting the clinical progression, therapeutic measures, and radiological evidence confirming spontaneous disk resorption.
Case presentation
All the procedures of this study were approved by the ethics committee of the Sixth Affiliated Hospital of Xinjiang Medical University, all the procedures followed the guidelines of the declaration of Helsinki.
In 2021, a 40-year-old female presented to the department of spine surgery with a one-year history of severe lower back pain radiating to both legs. She had no history of injuries, chronic diseases, or prior surgeries. On presentation, her Visual Analog Scale (VAS) score for lower back pain was eight. Physical examination revealed that vertebral flexion, extension, and lateral flexion markedly aggravated pain in the buttocks and the rear lateral surface of both legs down to the ankles.
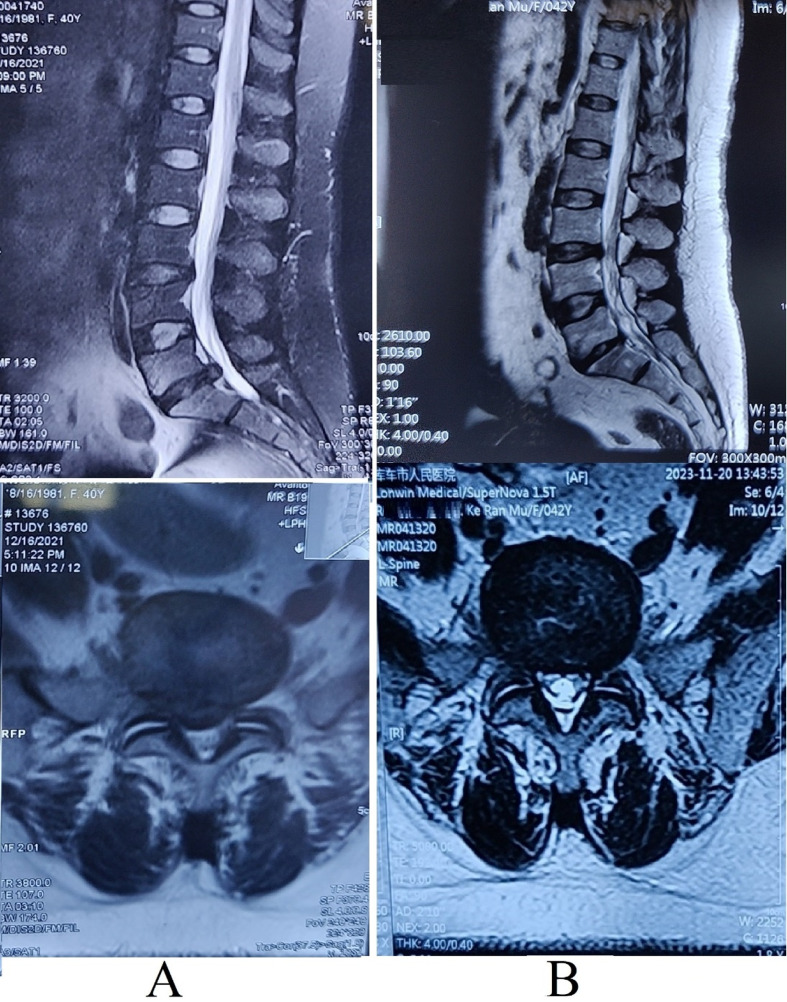
MRI scans at that time confirmed the presence of a large herniated disk at the L5/S1 level (Fig. 1, Panel A). Despite a recommendation for surgical intervention to remove the herniated disk, the patient declined surgery. Instead, she opted for a comprehensive non-operative treatment protocol. This regimen included non-steroidal anti-inflammatory drugs (NSAIDs), heat therapy, massage therapy, acupuncture, and kinesitherapy aimed at strengthening the lower back muscles.
Fig. 1.
MRI Scans of L5/S1 Herniated Disk Before and After Non-Operative Treatment. Panel A: Initial MRI scan revealing a large herniated disk at the L5/S1 level (indicated by the arrow). Panel B: Follow-up MRI scan at 24 months showing spontaneous resorption of the herniated disk at L5/S1 (indicated by the arrow). The patient declined surgical intervention and instead pursued a comprehensive non-operative treatment protocol, including NSAIDs, heat therapy, massage therapy, acupuncture, and kinesitherapy. Over two years, her symptoms resolved completely, with no recurrence of back or leg pain and significant improvement in functional mobility
Over the subsequent two years, the patient’s back pain gradually alleviated, and the radiating pain in her legs completely subsided. At 24 months, she reported no recurrence of back or leg pain, with her VAS pain score for lower back pain reduced to one. Additionally, vertebral flexion, extension, and lateral flexion no longer aggravated pain in the buttocks or legs. Follow-up MRI demonstrated spontaneous resorption of the herniated disk at L5/S1 (Fig. 1, Panel B).
Discussion
Lumbar disc herniation is a prevalent condition in spinal surgery, characterized by low back pain, radicular leg pain, and neurological deficits. Traditional treatment approaches include conservative management and surgical intervention. However, studies have revealed that spontaneous resorption of herniated disc material can occur without surgery, providing a rationale for conservative treatment and altering clinical management strategies [5–34] (see Table 1).
Table 1.
The incidence of spontaneous resorption of LDH
| Study | Country | Study design | %(T/R) SRLDH | Age |
|---|---|---|---|---|
| Teplick et al., 1985 [5] | USA | Retrospective | 20.0 (55/11) | NA |
| Saal et al., 1990 [6] | USA | Prospective | 81.8 (11/9) | NA |
| Bozzao et al., 1992 [7] | Italy | Prospective | 66.7 (21/14) | 20–64 |
| Bush et al., 1992 [8] | UK | Prospective | 64.0 (111/71) | 17–72 |
| Maigne et al., 1992 [9] | France | Prospective | 81.3 (48/39) | 26–75 |
| Ellenberg et al., 1993 [10] | USA | Prospective | 78.6 (14/11) | 28–67 |
| Gallucci et al., 1995 [11] | Italy | Prospective | 73.3 (15/11) | 27–62 |
| Matsubara et al., 1995 [12] | Japan | Prospective | 64.0 (32/20) | 16–52 |
| Yukawa et al., 1996 [13] | Japan | Retrospective | 53.3 (30/16) | 14–69 |
| Komori et al., 1998 [14] | Japan | Prospective | 66.7 (48/32) | 20–75 |
| Takada et al., 2001 [15] | Japan | Prospective | 88.1 (42/37) | 16–64 |
| Ahn et al., 2002 [16] | Korea | Prospective | 76.5 (17/13) | 19–73 |
| Splendiani et al., 2004 [17] | Italy | Prospective | 34.7 (72/25) | 21–68 |
| Autio et al., 2006 [18] | Finland | Retrospective | 91.9 (74/68) | 19–78 |
| Erly et al., 2006 [19] | USA | Retrospective | 69.4 (36/25) | NA |
| Jensen et al., 2006 [20] | Denmark | Prospective | 46.8 (139/65) | 18–65 |
| Cribb et al., 2007 [21] | UK | Retrospective | 93.3 (15/14) | 24–73 |
| Benson et al., 2010 [22] | UK | Prospective | 100 (28/28) | 25–62 |
| Iwabuchi et al., 2010 [23] | Japan | Prospective | 61.8 (34/21) | NA |
| Barzouhi et al., 2013 [24] | Netherlands | RCT | 92.6 (95/88) | 18–65 |
| Yu et al., 2014 [25] | China | Prospective | 50.6 (83/42) | 16–60 |
| Hong et al., 2016 [26] | Korea | Retrospective | 85.7 (28/24) | 26–78 |
| Demirel et al., 2017 [27] | Turkey | RCT | 90.0 (20/18) | NA |
| Lee et al., 2017 [28] | Korea | Retrospective | 96.2(505/486) | NA |
| Kesikburun et al. 2019 [29] | Turkey | Prospective | 90.0 (40/36) | 39.7–66.7 |
| Dai et al., 2020 [30] | China | Prospective | 69.7 (66/46) | 25–67 |
| Ma et al., 2021 [31] | China | Retrospective | 46.2(409/189) | 14–70 |
| Sucuoglu et al., 2021 [32] | Turkey | Prospective | 89.1 (55/49) | 25–67 |
| Hournung 2023 [33] | China | Retrospective | 100 (93/90) | 20–51 |
| Budrovac 2023 [34] | Croatia | RCT | 66.7 (30/20) | 18–65 |
%(T/R) SRLDH: Percentage (resorption/total) of spontaneous resorption of LDH
Mechanisms of spontaneous resorption in lumbar disc herniation
Inflammation
The inflammatory response is one of the core mechanisms of spontaneous resorption of LDH. When herniated disc tissue enters the epidural space, it triggers an immune response, leading to the infiltration of inflammatory cells and the release of inflammatory mediators. These inflammatory mediators include tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6). They accelerate the degradation and absorption of the herniated tissue by activating matrix metalloproteinases (MMPs) and promoting neovascularization [35–37]. TNF-α, IL-1β, IL-6, and IL-8 are released in the early stages of the inflammatory response, further promoting the infiltration of inflammatory cells and the persistence of the inflammatory response. These inflammatory mediators play a significant role in the spontaneous resorption of LDH. TNF-α and IL-1β can stimulate disc cells to produce more inflammatory mediators and chemokines, such as monocyte chemoattractant protein-1 (MCP-1), thereby attracting more inflammatory cells (e.g., macrophages) to the site of the herniated tissue. The release of inflammatory mediators creates a positive feedback loop, further amplifying the inflammatory response. For example, TNF-α can induce the expression of MCP-1, which in turn attracts more macrophages to the herniated tissue, leading to the release of more TNF-α and IL-1β. In the later stages of the inflammatory response, the release of anti-inflammatory mediators (such as IL-4 and IL-10) gradually suppresses the inflammatory response, promoting tissue repair and absorption.
Neovascularization
Neovascularization is a critical mechanism in the resorption process. New blood vessels form around the herniated disc tissue, providing a pathway for inflammatory cells and degrading enzymes to enter the herniated tissue, thereby promoting the absorption process [38]. Vascular endothelial growth factor (VEGF) and other angiogenic factors are upregulated in herniated disc tissue, facilitating the formation of new blood vessels and accelerating the resorption process [39, 40]. Studies have shown that VEGF is highly expressed in herniated disc tissue, promoting the proliferation and migration of vascular endothelial cells, leading to the formation of new blood vessels. Basic fibroblast growth factor (bFGF) and platelet-derived growth factor (PDGF) also contribute to the formation of new blood vessels. These growth factors stimulate the proliferation and migration of vascular endothelial cells, promoting angiogenesis.
The functions of neovascularization are primarily reflected in the following two aspects: (1) Infiltration of inflammatory cells: New blood vessels provide a pathway for inflammatory cells (e.g., macrophages) to enter the herniated tissue, thereby sustaining the inflammatory response and promoting the degradation of the herniated tissue. (2) Supply of nutrients and oxygen: New blood vessels deliver essential nutrients and oxygen to the herniated tissue, supporting the activity of inflammatory cells and degrading enzymes, thus facilitating the resorption process.
Macrophage infiltration
Macrophages play a critical role in the spontaneous resorption of LDH. When herniated disc tissue enters the epidural space, it triggers the infiltration and activation of macrophages. Macrophages promote the degradation and absorption of herniated tissue by phagocytizing cellular debris and secreting inflammatory mediators.
In the recruitment of macrophages, chemokines and inflammatory mediators play a major role. Chemokines such as MCP-1 and MIP-1α secreted by disc cells and inflammatory cells, attract macrophages to the site of the herniated tissue. Inflammatory mediators such as TNF-α and IL-1βalso promote the recruitment and activation of macrophages. These inflammatory mediators stimulate disc cells and inflammatory cells to produce more chemokines, thereby attracting additional macrophages.
The primary functions of macrophages include phagocytosis and the secretion of inflammatory mediators and matrix-degrading enzymes: (1) Macrophages can phagocytize cellular debris and degradation products in the herniated tissue, thereby reducing the volume of the herniated mass. (2) Macrophages secrete various inflammatory mediators such as TNF-α, IL-1β, and IL-6, which further promote the inflammatory response and matrix degradation. (3) Macrophages secrete matrix metalloproteinases (MMPs), which degrade collagen and proteoglycans in the disc tissue, facilitating the absorption of the herniated tissue.
Matrix degradation
Matrix degradation is the final step in the spontaneous resorption of LDH. Matrix metalloproteinases (MMPs) are key enzymes in matrix degradation, capable of breaking down collagen and proteoglycans in the disc tissue, thereby promoting the absorption of herniated tissue. Studies have shown that herniated disc tissue contains a high density of macrophages, which secrete enzymes such as matrix metalloproteinases (MMPs) that facilitate the degradation and remodeling of the disc material [41–43].
MMP-3 and MMP-7 play a significant role in the spontaneous resorption of LDH. These matrix metalloproteinases degrade collagen and proteoglycans in the disc tissue, promoting the breakdown and absorption of herniated tissue. The activity of MMPs is regulated by various factors, including inflammatory mediators (such as TNF-α and IL-1β) and growth factors (such as VEGF and bFGF). These factors stimulate disc cells and inflammatory cells to produce more MMPs, thereby accelerating matrix degradation.
The progression and resolution of the inflammatory response significantly influence the regulation of matrix degradation. In the early stages of the inflammatory response, the release of inflammatory mediators promotes the production and activity of MMPs, accelerating matrix degradation. In the later stages, the release of anti-inflammatory mediators inhibits the production and activity of MMPs, slowing down matrix degradation. Additionally, the formation and function of new blood vessels play a crucial role in regulating matrix degradation. New blood vessels provide a pathway for inflammatory cells and degrading enzymes to enter the herniated tissue, thereby promoting matrix degradation.
Disruption of immune privilege
Normal disc tissue possesses immune privilege, meaning the immune system does not mount a strong immune response against it. However, when herniated disc tissue enters the epidural space, this immune privilege is disrupted, triggering an immune response. The primary mechanisms of immune privilege include the blood-nucleus pulposus barrier and the Fas/FasL system.
Normal disc tissue is isolated from the immune system by the blood-nucleus pulposus barrier, which maintains immune privilege. This barrier, composed of disc cells and extracellular matrix, prevents the entry of immune cells and inflammatory mediators. When herniated disc tissue enters the epidural space, it disrupts the blood-nucleus pulposus barrier, exposing the tissue to the immune system. This exposure triggers an immune response, leading to the infiltration of inflammatory cells and the release of inflammatory mediators.
The Fas/FasL system also plays a critical role in maintaining the immune privilege of disc tissue. FasL, expressed by disc cells, induces apoptosis in immune cells and vascular endothelial cells, thereby preventing immune cell infiltration and neovascularization. Dysregulation of the Fas/FasL system leads to immune cell infiltration and the formation of new blood vessels, promoting inflammation and matrix degradation.
Apoptosis and autophagy
In herniated disc tissue, the incidence of apoptosis is relatively high. Apoptosis is primarily induced by inflammatory mediators such as TNF-α and IL-1β, and growth factors such as VEGF and bFGF, which promote the degradation and absorption of herniated tissue. The regulation of apoptosis involves multiple signaling pathways, including the p38 MAPK pathway. Activation of the p38 MAPK pathway can induce apoptosis in disc cells, accelerating the absorption of herniated tissue.
Autophagy is a cellular self-degradation process that removes damaged components and metabolic waste within cells. In herniated disc tissue, the incidence of autophagy is relatively high, contributing to the degradation and absorption of herniated tissue. The regulation of autophagy involves multiple signaling pathways, including the PI3K/AKT pathway. Activation of the PI3K/AKT pathway can inhibit autophagy, thereby slowing the absorption of herniated tissue.
Disc dehydration
Disc dehydration is another proposed mechanism of spontaneous resorption. The herniated disc material gradually loses water content, leading to shrinkage and eventual retraction into the annulus fibrosus. MRI studies have shown that the signal intensity of herniated disc material on T2-weighted images decreases over time, indicating a reduction in water content [44–46]. However, the role of disc dehydration in resorption remains controversial and requires further investigation [47–49].
Clinical predictors of spontaneous resorption
Size of the herniation
Larger disc herniations are more likely to undergo spontaneous resorption. Erly et al. found that larger herniations exhibit higher T2-weighted signal intensity on MRI, which correlates positively with resorption [50]. However, other studies have reported no significant relationship between herniation size and resorption, suggesting that this predictor requires further validation [51–53].
Type of herniation
The type of LDH herniation is closely related to the incidence of spontaneous resorption. Based on MRI imaging findings, LDH can be classified into bulging, protrusion, extrusion, and sequestration. Studies have shown that extrusion and sequestration types of LDH have higher rates of spontaneous resorption, while bulging and protrusion types have lower rates. This may be because the nucleus pulposus tissue in extrusion and sequestration types is more likely to come into contact with the bloodstream, triggering immune and inflammatory responses that promote resorption. Ahn et al. found that all sequestrated herniations exhibited partial or complete resorption [54]. A systematic review by Chiu et al. confirmed that the resorption rate for sequestrated herniations is as high as 96% [30].
Composition of herniated disk
The composition of the herniated disc also influences the incidence of spontaneous resorption. Studies have shown that patients with herniated tissue containing a higher proportion of nucleus pulposus are more likely to experience spontaneous resorption, whereas herniated tissue containing more cartilaginous endplate is less likely to be absorbed. This may be because nucleus pulposus tissue has a higher water content, making it more prone to vascularization and dehydration, thereby facilitating resorption.
Rim enhancement on contrast-enhanced MRI
The degree of rim enhancement on contrast-enhanced MRI is a significant predictor of spontaneous resorption. Autio et al. found that herniations with greater rim enhancement thickness were more likely to undergo resorption [55]. Rim enhancement reflects the extent of neovascularization and inflammation, making it a useful predictor of resorption [56–58].
Involvement of the posterior longitudinal ligament (PLL)
The involvement of the PLL is also associated with spontaneous resorption. Ahn et al. found that herniations penetrating the PLL were more likely to resorb [59]. Seo et al. corroborated this finding, suggesting that penetration of the PLL exposes the herniated material to the epidural vascular supply, facilitating resorption [60–62].
Modic changes
Modic changes, which are MRI findings associated with disc degeneration, have been linked to resorption. Shan et al. found that herniations without Modic changes were more likely to undergo resorption [63]. However, Kawaguchi et al. reported no association between Modic changes and resorption, indicating that this predictor requires further investigation [64–66].
Clinical predictive factors
In addition to imaging examinations, certain clinical features can also serve as reference indicators for predicting spontaneous resorption of LDH: (1) Duration of Symptoms: Patients with a shorter duration of symptoms are more likely to experience spontaneous resorption. This may be because the inflammatory response in patients with a shorter symptom duration is more active, promoting the resorption process. (2) Pain Severity: Patients with milder pain are more likely to experience spontaneous resorption. This may be because the inflammatory response in patients with milder pain is more moderate, facilitating the resorption process. (3) Neurological Deficits: Patients with milder neurological deficits are more likely to experience spontaneous resorption. This may be because the degree of compression from the herniated disc is less severe in patients with milder neurological deficits, favoring the resorption process.
Treatment strategies
Conservative treatment is the first-line approach for lumbar disc herniation and includes pharmacological therapy, physical therapy such as mechanical traction [67–69], and rehabilitation. Studies have shown that conservative management can effectively alleviate symptoms and promote spontaneous resorption of herniated disc material [70–72]. A meta-analysis by Wang et al. recommended a follow-up period of 4 to 10.5 months to assess the potential for resorption [73].
Future research directions
Regulation of inflammatory response
The inflammatory response is one of the core mechanisms of spontaneous resorption in LDH [74]. Future research should further explore the regulatory mechanisms of the inflammatory response to promote spontaneous resorption.
1) Targeted regulation of inflammatory mediators: TNF-α and IL-1β are key mediators of the inflammatory response. Future research could develop targeted drugs against these inflammatory mediators to regulate the intensity and duration of the inflammatory response. For example, anti-TNF-α antibodies and IL-1β receptor antagonists may help control the inflammatory response and promote spontaneous resorption. Anti-inflammatory mediators (such as IL-4 and IL-10) play a crucial role in the resolution of the inflammatory response. Future research could explore how exogenous anti-inflammatory mediators (such as recombinant proteins or gene therapy) can facilitate the resolution of the inflammatory response, thereby accelerating spontaneous resorption.
2) Regulation of inflammatory signaling pathways: NF-κB Signaling Pathway: The NF-κB signaling pathway plays a critical role in regulating the inflammatory response. Future research could investigate how to modulate the NF-κB signaling pathway, either by inhibition or activation, to regulate the inflammatory response and promote spontaneous resorption.
MAPK Signaling Pathway: The MAPK signaling pathway (e.g., p38 MAPK) plays an important role in regulating the inflammatory response and apoptosis. Future research could explore how to modulate the MAPK signaling pathway to promote spontaneous resorption.
Regulation of neovascularization
Neovascularization is another important mechanism in the spontaneous resorption of LDH. Future research should further explore the regulatory mechanisms of neovascularization to promote spontaneous resorption.
1) Application of angiogenic factors: VEGF is a key mediator of neovascularization. Future research could investigate how exogenous VEGF (such as recombinant proteins or gene therapy) can be used to promote the formation of new blood vessels, thereby accelerating spontaneous resorption. bFGF and PDGF also play roles in promoting neovascularization. Future research could explore how the combined application of these growth factors can enhance the formation of new blood vessels, facilitating spontaneous resorption.
2) Regulation of angiogenesis inhibitors: In some cases, excessive neovascularization may lead to the persistence of the inflammatory response and tissue damage. Future research could investigate how to modulate anti-angiogenic factors (such as TIMP-3) to balance the formation of new blood vessels, thereby promoting spontaneous resorption.
Regulation of macrophage polarization
Macrophages play a critical role in the spontaneous resorption of LDH. Future research should further explore the regulatory mechanisms of macrophage polarization to promote spontaneous resorption.
1) Balance of M1/M2 macrophages: M1 macrophages primarily secrete pro-inflammatory factors, promoting inflammatory responses and matrix degradation. Future research could investigate how to regulate the activity of M1 macrophages to facilitate spontaneous resorption. M2 macrophages mainly secrete anti-inflammatory factors, promoting tissue repair and absorption. Future research could explore how to modulate the activity of M2 macrophages to enhance spontaneous resorption.
2) Molecular mechanisms of macrophage polarization: Regulation of Signaling Pathways: Macrophage polarization involves multiple signaling pathways, including the PI3K/AKT and STAT pathways. Future research could investigate how to regulate these signaling pathways to promote macrophage polarization, thereby accelerating spontaneous resorption. Epigenetic regulation: Epigenetic mechanisms (such as DNA methylation and histone modification) play a significant role in macrophage polarization. Future research could explore how epigenetic regulation can be utilized to promote macrophage polarization, thus facilitating spontaneous resorption.
Regulation of matrix degradation
Matrix degradation is the final step in the spontaneous resorption of LDH. Future research should further explore the regulatory mechanisms of matrix degradation to promote spontaneous resorption.
1) Regulation of MMPs: MMP-3 and MMP-7 are key enzymes in matrix degradation. Future research could investigate how to regulate the activity of these enzymes to promote matrix degradation, thereby accelerating spontaneous resorption. In some cases, excessive matrix degradation may lead to tissue damage. Future research could explore how to balance matrix degradation by applying MMP inhibitors, thus promoting spontaneous resorption.
2) Molecular mechanisms of matrix degradation: Inflammatory mediators can stimulate the production and activity of MMPs, while growth factors can also stimulate the production and activity of MMPs. Future research could investigate how to regulate these inflammatory mediators and growth factors to promote matrix degradation, thereby accelerating spontaneous resorption.
Regulation of immune privilege
Normal disc tissue possesses immune privilege, and future research should further explore the regulatory mechanisms of immune privilege to promote spontaneous resorption.
1) Regulation of the blood-nucleus pulposus barrier: In some cases, disrupting the blood-nucleus pulposus barrier may help trigger an immune response, thus promoting spontaneous resorption. Future research could investigate how to regulate the disruption of the blood-nucleus pulposus barrier to facilitate spontaneous resorption.
2) Regulation of the Fas/FasL system: The Fas/FasL system plays a critical role in maintaining the immune privilege of disc tissue. In some cases, inhibiting the Fas/FasL system may help trigger an immune response, thereby promoting spontaneous resorption. Future research could investigate how to regulate the inhibition of the Fas/FasL system to facilitate spontaneous resorption.
Regulation of apoptosis and autophagy
Apoptosis and autophagy play important roles in the spontaneous resorption of LDH. Future research should further explore the regulatory mechanisms of these processes to promote spontaneous resorption. Although theoretically, inducing apoptosis and autophagy could promote the degradation and absorption of herniated tissue, in some cases, inhibiting apoptosis and autophagy may help protect disc tissue, thereby facilitating spontaneous resorption. Future research could explore how to regulate the inhibition of apoptosis and autophagy to promote spontaneous resorption.
Pain genetics
Research on pain genetics offers new directions for pain management. Studies have shown that polymorphisms in the OPRM1 gene are associated with pain intensity following lumbar disc herniation [75–77]. Future research should investigate the relationship between pain-related genes and resorption, with the goal of developing personalized pain management strategies [78–79].
Advances in imaging techniques
Advances in imaging technologies, such as chemical exchange saturation transfer (CEST), T1ρ imaging, T2 mapping, ultrashort echo time MRI (UTE-MRI), and sodium MRI, offer new tools for studying disc degeneration and herniation [80–82]. These techniques can reveal biochemical changes in disc tissue, aiding in the prediction of resorption potential [83].
Prediction models based on machine learning and deep learning
With the advancement of artificial intelligence technology, the application of machine learning and deep learning in the medical field has gradually increased. Machine learning is a technology that trains models using data to make predictions and decisions. Deep learning, a subset of machine learning, simulates the learning process of the human brain through multi-layer neural networks, enabling automatic feature extraction from data and complex pattern recognition. In the medical field, machine learning and deep learning are primarily used for disease diagnosis, prognosis prediction, and treatment optimization. For example, machine learning-based image analysis can assist doctors in disease diagnosis, and deep learning-based prediction models can help doctors evaluate patient prognosis.
In recent years, machine learning and deep learning-based prediction models for postoperative recovery in LDH have become a research hotspot. These models analyze patients’ clinical and imaging data to predict postoperative recovery outcomes, thereby providing a basis for clinical decision-making. Future research should further optimize these models, focusing on the following three aspects to improve prediction accuracy: 1) Optimization of data quality: The first step in building an LDH postoperative recovery prediction model is data collection. Typically, researchers collect clinical data from patients, including age, gender, body mass index (BMI), medical history, preoperative examination results, and more. Additionally, imaging data, such as the type, size, and location of disc herniation shown on MRI, are important input variables. Future research should further optimize data standardization methods to improve model prediction accuracy and expand sample sizes to enhance the model’s generalization ability.
2) Optimization of model algorithms: After data preprocessing, researchers use various machine learning and deep learning algorithms to build prediction models. Commonly used machine learning algorithms include Random Forests, Support Vector Machines (SVM), and Logistic Regression. Commonly used deep learning algorithms include Multilayer Perceptron (MLP) and Convolutional Neural Networks (CNN). Future research should explore more machine learning and deep learning algorithms and further optimize model hyperparameters to improve prediction accuracy, and apply those prediction models for personalized treatment and real-time monitoring to dynamically adjust treatment plans, thereby improving treatment outcomes.
Conclusion
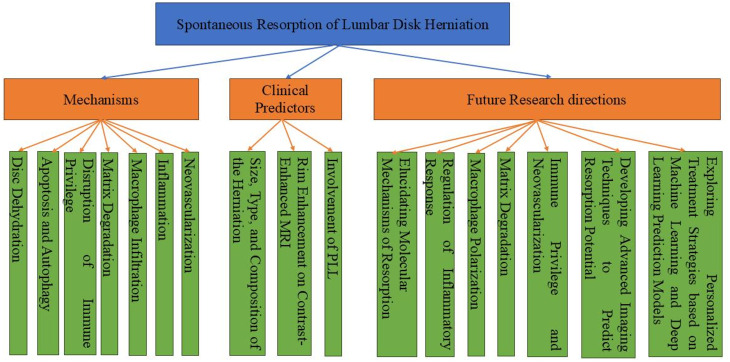
Spontaneous resorption in lumbar disc herniation is a complex, multifactorial process involving mechanisms such as inflammation, neovascularization, disc dehydration, and mechanical traction. Clinical predictors of resorption include the size of the herniation, sequestration, rim enhancement on MRI, inflammatory mediators, and involvement of the posterior longitudinal ligament. Future research should focus on elucidating the molecular mechanisms of resorption, developing advanced imaging techniques to predict resorption potential, and exploring personalized treatment strategies based on machine learning and deep learning prediction models (Fig. 2).
Fig. 2.
Mechanisms, clinical predictors and future research trends of spontaneous resorption of lumbar disk herniation
Author contributions
YZ and ZJ; Conceptualization, methodology, software, formal analysis, investigation, resources, data curation. AA writing—original draft preparation, visualization, supervision, validation, AM: writing—review and editing, project administration, funding acquisition. All authors have read and agreed to the published version of the manuscript.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Fujiwara A, Kobayashi N, Saiki K, Kitagawa T, Tamai K, Saotome K. Association of the resorption of herniated lumbar discs with matrix metalloproteinase-3 (MMP-3) immunohistochemical expression in macrophages. Spine. 2003;28(6):637–44.12671347 [Google Scholar]
- 2.Henriksen M, Keller C, Rahme H. Non-operative management of acute lumbar disc herniation: a case study. BMJ Case Rep, 2009, bcr0820075003.
- 3.El Barzouhi A. Magnetic resonance imaging in follow-up assessment of sciatica. N Engl J Med, 2013. [DOI] [PubMed]
- 4.Peul WC. Surgery versus prolonged Conservative treatment for sciatica. N Engl J Med. 2007. [DOI] [PubMed]
- 5.Teplick JG, Haskin ME. Spontaneous regression of herniated nucleus pulposus. AJR Am J Roentgenol. 1985;145(2):371–5. 10.2214/ajr.145.2.371. [DOI] [PubMed] [Google Scholar]
- 6.Saal JA, Saal JS, Herzog RJ. The natural history of lumbar intervertebral disc extrusions treated nonoperatively. Spine (Phila Pa 1976). 1990;15(7):683–6. 10.1097/00007632-199007000-00013. [DOI] [PubMed] [Google Scholar]
- 7.Bozzao A, Gallucci M, Masciocchi C, Aprile I, Barile A, Passariello R. Lumbar disk herniation: MR imaging assessment of natural history in patients treated without surgery. Radiology. 1992;185(1):135–41. 10.1148/radiology.185.1.1523297. [DOI] [PubMed] [Google Scholar]
- 8.Bush K, Cowan N, Katz DE, Gishen P. The natural history of sciatica associated with disc pathology. A prospective study with clinical and independent radiologic follow-up. Spine (Phila Pa 1976). 1992;17(10):1205–12. 10.1097/00007632-199210000-00013. [DOI] [PubMed] [Google Scholar]
- 9.Maigne JY, Rime B, Deligne B. Computed tomographic follow-up study of forty-eight cases of nonoperatively treated lumbar intervertebral disc herniation. Spine (Phila Pa 1976). 1992;17(9):1071–4. 10.1097/00007632-199209000-00010. [DOI] [PubMed] [Google Scholar]
- 10.Ellenberg MR, Ross ML, Honet JC, Schwartz M, Chodoroff G, Enochs S. Prospective evaluation of the course of disc herniations in patients with proven radiculopathy. Arch Phys Med Rehabil. 1993;74(1):3–8. [PubMed] [Google Scholar]
- 11.Gallucci M, Bozzao A, Orlandi B, Manetta R, Brughitta G, Lupattelli L. Does Postcontrast MR enhancement in lumbar disk herniation have prognostic value? J Comput Assist Tomogr. 1995;19(1):34–8. 10.1097/00004728-199501000-00006. [DOI] [PubMed] [Google Scholar]
- 12.Matsubara Y, Kato F, Mimatsu K, Kajino G, Nakamura S, Nitta H. Serial changes on MRI in lumbar disc herniations treated conservatively. Neuroradiology. 1995;37(5):378–83. 10.1007/BF00588017. [DOI] [PubMed] [Google Scholar]
- 13.Yukawa Y, Kato F, Matsubara Y, Kajino G, Nakamura S, Nitta H. Serial magnetic resonance imaging follow-up study of lumbar disc herniation conservatively treated for average 30 months: relation between reduction of herniation and degeneration of disc. J Spinal Disord. 1996;9(3):251–6. 10.1097/00002517-199606000-00012. [PubMed] [Google Scholar]
- 14.Komori H, Okawa A, Haro H, Muneta T, Yamamoto H, Shinomiya K. Contrast-enhanced magnetic resonance imaging in Conservative management of lumbar disc herniation. Spine (Phila Pa 1976). 1998;23(1):67–73. 10.1097/00007632-199801010-00015. [DOI] [PubMed] [Google Scholar]
- 15.Takada E, Takahashi M, Shimada K. Natural history of lumbar disc hernia with radicular leg pain: spontaneous MRI changes of the herniated mass and correlation with clinical outcome. J Orthop Surg (Hong Kong). 2001;9(1):1–7. 10.1177/230949900100900102. [DOI] [PubMed] [Google Scholar]
- 16.Ahn SH, Park HW, Byun WM, et al. Comparison of clinical outcomes and natural morphologic changes between sequestered and large central extruded disc herniations. Yonsei Med J. 2002;43(3):283–90. 10.3349/ymj.2002.43.3.283. [DOI] [PubMed] [Google Scholar]
- 17.Splendiani A, Puglielli E, De Amicis R, Barile A, Masciocchi C, Gallucci M. Spontaneous resolution of lumbar disk herniation: predictive signs for prognostic evaluation. Neuroradiology. 2004;46(11):916–22. 10.1007/s00234-004-1232-0. [DOI] [PubMed] [Google Scholar]
- 18.Autio RA, Karppinen J, Niinimäki J, et al. Determinants of spontaneous resorption of intervertebral disc herniations. Spine (Phila Pa 1976). 2006;31(11):1247–52. 10.1097/01.brs.0000217681.83524.4a. [DOI] [PubMed] [Google Scholar]
- 19.Erly WK, Munoz D, Beaton R. Can MRI signal characteristics of lumbar disk herniations predict disk regression? J Comput Assist Tomogr. 2006;30(3):486–9. 10.1097/00004728-200605000-00022. [DOI] [PubMed] [Google Scholar]
- 20.Jensen TS, Albert HB, Soerensen JS, Manniche C, Leboeuf-Yde C. Natural course of disc morphology in patients with sciatica: an MRI study using a standardized qualitative classification system. Spine (Phila Pa 1976). 2006;31(14). 10.1097/01.brs.0000221992.77779.37. [DOI] [PubMed]
- 21.Cribb GL, Jaffray DC, Cassar-Pullicino VN. Observations on the natural history of massive lumbar disc herniation. J Bone Joint Surg Br. 2007;89(6):782–4. 10.1302/0301-620X.89B6.18712. [DOI] [PubMed] [Google Scholar]
- 22.Benson RT, Tavares SP, Robertson SC, Sharp R, Marshall RW. Conservatively treated massive prolapsed discs: a 7-year follow-up. Ann R Coll Surg Engl. 2010;92(2):147–53. 10.1308/003588410X12518836438840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Iwabuchi M, Murakami K, Ara F, Otani K, Kikuchi SI. The predictive factors for the resorption of a lumbar disc herniation on plain MRI. Fukushima J Med Sci. 2010;56(2):91–7. 10.5387/fms.56.91. [DOI] [PubMed] [Google Scholar]
- 24.el Barzouhi A, Vleggeert-Lankamp CLAM, Lycklama à Nijeholt GJ, et al. Magnetic resonance imaging in follow-up assessment of sciatica. N Engl J Med Published Online. 2013. 10.1056/NEJMoa1209250. [DOI] [PubMed] [Google Scholar]
- 25.Yu PF, Jiang H, Liu JT, et al. Traditional Chinese medicine treatment for ruptured lumbar disc herniation: clinical observations in 102 cases. Orthop Surg. 2014;6(3):229–35. 10.1111/os.12120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hong SJ, Kim DY, Kim H, Kim S, Shin KM, Kang SS. Resorption of massive lumbar disc herniation on MRI treated with epidural steroid injection: A retrospective study of 28 cases. Pain Physician. 2016;19(6):381–8. 10.36076/ppj/2016.19.381. [PubMed] [Google Scholar]
- 27.Demirel A, Yorubulut M, Ergun N. Regression of lumbar disc herniation by physiotherapy. Does nonsurgical spinal decompression therapy make a difference? Double-blind randomized controlled trial. J Back Musculoskelet Rehabil. 2017;30(5):1015–22. 10.3233/BMR-169581. [DOI] [PubMed] [Google Scholar]
- 28.Lee J, Kim J, Shin JS, et al. Long-Term course to lumbar disc resorption patients and predictive factors associated with disc resorption. Evid Based Complement Alternat Med. 2017;2147408. 10.1155/2017/2147408. [DOI] [PMC free article] [PubMed]
- 29.Kesikburun B, Eksioglu E, Turan A, Adiguzel E, Kesikburun S, Cakci A. Spontaneous regression of extruded lumbar disc herniation: correlation with clinical outcome. Pak J Med Sci. 2019;35(4):974–80. 10.12669/pjms.35.4.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dai F, Dai YX, Jiang H, Yu PF, Liu JT. Non-surgical treatment with XSHHD for ruptured lumbar disc herniation: a 3-year prospective observational study. BMC Musculoskelet Disord. 2020;21(1):690. 10.1186/s12891-020-03723-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ma Z, Yu P, Jiang H, et al. Conservative treatment for giant lumbar disc herniation: clinical study in 409 cases. Pain Physician. 2021;24(5):E639–48. 10.36076/ppj.2021.24.E639. [PubMed] [Google Scholar]
- 32.Sucuoğlu H, Barut AY. Clinical and radiological Follow-Up results of patients with sequestered lumbar disc herniation: A prospective cohort study. Med Princ Pract. 2021;30(3):244–52. 10.1159/000515308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hornung AL, Barajas JN, Rudisill SS, et al. Prediction of lumbar disc herniation resorption in symptomatic patients: a prospective, multi-imaging and clinical phenotype study. Spine J. 2023;23(2):247–60. 10.1016/j.spinee.2022.10.003. [DOI] [PubMed] [Google Scholar]
- 34.Budrovac D, Radoš I, Hnatešen D, Haršanji-Drenjančević I, Tot OK, Katić F, Lukić I, Škiljić S, Nešković N, Dimitrijević I. Effectiveness of epidural steroid injection depending on discoradicular contact: A prospective randomized trial. Int J Environ Res Public Health. 2023;20(4):3672. 10.3390/ijerph20043672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhong M, Liu JT, Jiang H, Mo W, Yu PF, Li XC, Xue RR. Incidence of spontaneous resorption of lumbar disc herniation: a meta-analysis. Pain Physician. 2017;21(1):545–52. [PubMed] [Google Scholar]
- 36.Maher C, Underwood M, Buchbinder R. Non-specific low back pain. Lancet. 2017;389(10070):736–47. [DOI] [PubMed] [Google Scholar]
- 37.Jiang H. Incidence of spontaneous resorption of lumbar disc herniation: a meta-analysis. Pain Physician. 2017;21(11):E45–52. [PubMed] [Google Scholar]
- 38.Hyama A, Mochida J, Sakai D. Stem cell applications in intervertebral disc repair. Cell Mol Biol. 2008;54(1):24–32. [PubMed] [Google Scholar]
- 39.Autio RA, Karppinen J, Niinimäki J, Ojala R, Kurunlahti M, Haapea M, Vanharanta H, Tervonen O. Determinants of spontaneous resorption of intervertebral disc herniations. Spine (Phila Pa 1976). 2006;31(11):1247–52. [DOI] [PubMed] [Google Scholar]
- 40.Kobayashi S, Meir A, Kokubo Y, Uchida K, Takeno K, Miyazaki T, Yayama T, Kubota M, Nomura E, Mwaka E, Baba H. Ultrastructural analysis on lumbar disc herniation using surgical specimens: role of neovascularization and macrophages in hernias. Spine (Phila Pa 1976). 2009;34(7):655–62. [DOI] [PubMed] [Google Scholar]
- 41.Ahn SH, Ahn MW, Byun WM. Effect of the transligamentous extension of lumbar disc herniations on their regression and the clinical outcome of sciatica. Spine (Phila Pa 1976). 2000;25(4):475–80. [DOI] [PubMed] [Google Scholar]
- 42.Yukawa Y, Kato F, Matsubara Y, Kajino G, Nakamura S, Nitta H. Serial magnetic resonance imaging follow-up study of lumbar disc herniation conservatively treated for average 30 months: relation between reduction of herniation and degeneration of disc. J Spinal Disord. 1996;9(3):251–6. [PubMed] [Google Scholar]
- 43.Benzoni HT, Rathmell JP, Wu CL, Turk PC, Argoff CE, Hurley RW. Practical management of pain. 5th ed. Amsterdam, The Netherlands: Elsevier Inc.; 2013. [Google Scholar]
- 44.Takada T, Nishida K, Doita M, Myamoto H, Kurosaka M. Interleukin-6 production is upregulated by interaction between disc tissue and macrophages. Spine (Phila Pa 1976). 2004;29(10):1089–92. discussion 1093. [DOI] [PubMed] [Google Scholar]
- 45.Kang JD, Stefanovic-Rack M, McIntyre LA, Georgescu HI, Evans CT. Toward a biochemical Understanding of human intervertebral disc degeneration and herniation. Contributions of nitric oxide, interleukins, prostaglandin E2, and matrix metalloproteinases. Spine (Phila Pa 1976). 1997;22(10):1065–73. [DOI] [PubMed] [Google Scholar]
- 46.Koike Y, Uzuki M, Kokubun S, Sawai T. Angiogenesis and inflammatory cell infiltration in lumbar disc herniation. Spine (Phila Pa 1976). 2003;28(17):1928–33. [DOI] [PubMed] [Google Scholar]
- 47.Rothoeff R, Woertgen C, Holzschuh M, Brehme K, Ritschoff J, Bravanski A. Macrophage tissue infiltration, clinical symptoms, and signs in patients with lumbar disc herniation. A clinicopathological study on 179 patients. Acta Neurochir. 1998;41(20):2142–65. [DOI] [PubMed] [Google Scholar]
- 48.Yoshida M, Nakamura T, Sei A, Kikuchi T, Takagi K. Matsukawa. A intervertebral disc cells produce tumor necrosis factor Α, interleukin-1β, and monocyte chemoattractant protein-1 immediately after herniation: an experimental study using a new hernia model. Spine (Phila Pa 1976). 2005;30(11):51–3. [DOI] [PubMed] [Google Scholar]
- 49.Burke JG, Watson RWG, McCormack D, Dowling FE, Walsh MG, Fitzpatrick JM. Spontaneous production of monocyte chemoattractant protein-1 and interleukin-8 by the human lumbar intervertebral disc. Spine (Phila Pa 1976). 2002;27(13):1402–7. [DOI] [PubMed] [Google Scholar]
- 50.Ohba T, Haro H. TWEAK and TSLP in disc degeneration and spontaneous hernia resorption. JOR Spine. 2020;31(1):e1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ozaki S, Muro T, Ito S, Mizushima M. Neovascularization of the outermost area of herniated lumbar intervertebral discs. J Orthop Sci. 1999;44(2):86–92. [DOI] [PubMed] [Google Scholar]
- 52.Shami MF, Setton LA, Jarvis W, So S, Chen J, Jiroj I, Bullock E, Isaacs RE, Brown C, Richardson WJ. Proinflammatory cytokine expression profile in degenerated and herniated human intervertebral disc tissues. Arthritis Rheum. 2010;62(7):1974–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Haro H, Karo T, Komori H, Osada M, Shinomiya K. Vascular endothelial growth factor (VEGF)-induced angiogenesis in herniated disc resorption. J Orthop Res. 2002;20(3):409–15. [DOI] [PubMed] [Google Scholar]
- 54.Minamide A, Hashizume H, Yoshida M, Kawakami M, Hayashi N, Tamaki T. Effects of basic fibroblast growth factor on spontaneous resorption of herniated intervertebral discs. An experimental study in the rabbit. Spine (Phila Pa 1976). 1999;24(10):940–5. [DOI] [PubMed] [Google Scholar]
- 55.Hemmi T, Salvo K, Nakano S, Kanematsu Y, Kajikawa T, Katoh S, Goel VK. Natural history of extruded lumbar intervertebral disc herniation. J Med Invest. 2002;49(1–2):40–3. [PubMed] [Google Scholar]
- 56.Slavin AV, Raja A, Thompson J, Wagner EC. Spontaneous regression of a large lumbar disc herniation: report of an illustrative case. Surg Neurol. 2001;56(5):333–6. discussion 337. [DOI] [PubMed] [Google Scholar]
- 57.Kim SG, Yang JC, Kim TW, Park RH. Spontaneous regression of extruded lumbar disc herniation: three cases report. Korean J Spine. 2013;10(2):98–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Bozzzo A, Gallucci M, Masciocchi C, Aprile I, Barile A, Passariello R. Lumbar disk herniation: MR imaging assessment of natural history in patients treated without surgery. Radiology. 1992;185(1):135–41. [DOI] [PubMed] [Google Scholar]
- 59.Teplick J, Haskin M. Spontaneous regression of herniated nucleus pullposus. Am J Roentgenol. 1985;146(2):371–5. [DOI] [PubMed] [Google Scholar]
- 60.Onel D, Tuadzi M, Sari H, Demirk C. Computed tomographic investigation of the effect of traction on lumbar disc herniation. Spine (Phila Pa 1976). 1989;41(1):82–90. [DOI] [PubMed] [Google Scholar]
- 61.Seo JY, Roh YH, Kim YH, Ha KX. Three-dimensional analysis of volumetric changes in herniated discs of the lumbar spine: dose spontaneous resorption of herniated discs always occur? Eur Spine J. 2016;25(5):1393–402. [DOI] [PubMed] [Google Scholar]
- 62.Komori H, Shinomiya K, Nakai O, Yamaura I, Takeda S, Furuya K. The natural history of herniated nucleus pullposus with radiculopathy. Spine (Phila Pa 1976). 1996;21(2):25–9. [DOI] [PubMed] [Google Scholar]
- 63.Ahn SH, Park HW, Byun WW, Ahn MW, Jang SH, Bae JH, Kim YK. Comparison of clinical outcomes and natural morphologic changes between sequestered and large central extruded disc herniations. Yonsei Med J. 2002;43(3):283–90. [DOI] [PubMed] [Google Scholar]
- 64.Oriel T, Orz Y, Attia W, Almursen K. Spontaneous resorption of sequestered intervertebral disc herniation. World Neurosurg. 2012;77(1):146–52. [DOI] [PubMed] [Google Scholar]
- 65.Chiu CC, Chuang TY, Chang KH, Wu CH, Lin PW, Hsu WY. The probability of spontaneous regression of lumbar herniated disc: a systematic review. Clin Rehabil. 2015;29(2):184–95. [DOI] [PubMed] [Google Scholar]
- 66.Erly WK, Munoz D, Beaton R. Can MRI signal characteristics of lumbar disk herniation: predict disk regression? J Comput Assist Tomogr. 2006;30(3):486–9. [DOI] [PubMed] [Google Scholar]
- 67.Haro H, Crawford HC, Fingleton B, Shinomiya K, Spengler DM, Mattisian LM. Matrix metalloproteinase-2-dependent release of tumor necrosis factor-alpha in a model of herniated disc resorption. J Clin Invest. 2000;105(2):145–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Karo T, Haro H, Komori H, Shinomiya K. Sequential dynamics of inflammatory cytokine, angiogenesis including factor and matrix degrading enzymes during spontaneous resorption of the herniated disc. J Orthop Res. 2004;22(4):895–900. [DOI] [PubMed] [Google Scholar]
- 69.Hegewald AA, Neumann K, Kalwitz G, Freymann D, Erides M, Schmiedeer K, Kaps C, Thorne C. The chemokines CXCL10 and XCL1 recruit human annulus fibrosus cells. Spine (Phila Pa 1976). 2012;37(2):101–7. [DOI] [PubMed] [Google Scholar]
- 70.Splendiani A, Puglielli E, De Annicis R, Barile A, Masciocchi C, Gallucci M. Spontaneous resolution of lumbar disk herniation: predictive signs for prognostic evaluation. Neuromatology. 2004;46(11):916–22. [DOI] [PubMed] [Google Scholar]
- 71.Shin Z, Fan Z, Xie Q, Suyou L, Liu J, Wang C, Zhao F. Spontaneous resorption of lumbar disc herniation is less likely when modic changes are present. Spine (Phila Pa 1976). 2014;39(9):736–44. [DOI] [PubMed] [Google Scholar]
- 72.Kawaguchi K, Harimaya K, Matsumoto Y, Hayashida M, Okada S, Jida K, Kato G, Tsuchiya K, Dorf T, Oda Y, Iwanoto Y, Nakashima Y. Effect of cartilaginous endplates on extruded disc resorption in lumbar disc herniation. PLoS ONE. 2018;13(4). [DOI] [PMC free article] [PubMed]
- 73.Wang Y, Dai G, Jiang L, Liao S. The incidence of regression after the non-surgical treatment of symptomatic lumbar disc herniation: a systematic review and meta-analysis. BMC Musculoskelet Disord. 2020;21(1):530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kato T, Haro H, Komori H, et al. Sequential dynamics of inflammatory cytokine, angiogenesis inducing factor and matrix degrading enzymes during spontaneous resorption of the herniated disc. J Orthop Res. 2004;22(4):895–900. [DOI] [PubMed] [Google Scholar]
- 75.Olsen MB, Jacobsen LM, Schistad EI, et al. Pain intensity the first year after lumbar disc herniation is associated with the A118G polymorphism in the opioid receptor mu 1 gene: evidence of a sex and genotype interaction. J Neurosci. 2012;32(29):9831–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Chidambaran V, Zhang X, Geisler K, et al. Enrichment of genomic pathways based on differential DNA methylation associated with chronic postsurgical pain and anxiety in children: a prospective, pilot study. J Pain. 2019;20(7):771–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bennett DL, Clark AJ, Huang J, et al. The role of voltage-gated sodium channels in pain signaling. Physiol Rev. 2019;99(2):1079–151. [DOI] [PubMed] [Google Scholar]
- 78.Martirosyan NL, Patel AA, Carotenuto A, et al. Genetic alterations in intervertebral disc disease. Front Surg. 2016;3:59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bjørnland T, Moen A, Schistad EI, et al. Genes associated with persistent lumbar radicular pain; a systematic review. BMC Musculoskelet Disord. 2016;17(1):500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Samartzis D, Borthakur A, Belfer I, et al. Novel diagnostic and prognostic methods for disc degeneration and low back pain. Spine J. 2015;15(9):1919–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wang YX. Radiological imaging in degenerative disc disease: current status and future directions. Quant Imaging Med Surg. 2017;7(5):547–57. [Google Scholar]
- 82.Zuo J, Saadat E, Romero A, et al. Assessment of intervertebral disc degeneration with magnetic resonance imaging and spectroscopy. Front Biosci (Landmark Ed). 2011;16:1612–22. [Google Scholar]
- 83.Borthakur A, Maurer PM, Fenty M, et al. T1ρ MRI and discography in the assessment of lumbar disc degeneration. Eur Spine J. 2011;20(5):737–44.10.1007/s00586-010-1640-2.21190045 [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.