Abstract
Background
The human monoclonal antibody belimumab (BEL) has emerged as a promising treatment for systemic lupus erythematosus (SLE), particularly for reducing the need for glucocorticoids and minimizing organ damage. The optimal timing of BEL initiation has been unclear; emerging evidence suggests that early intervention with BEL, particularly within the first 5 years of diagnosis, may yield better outcomes by modulating disease progression and reducing flare frequency. Understanding the relationship between disease duration and BEL efficacy is essential for the development of tailored strategies.
Patients and methods
We analyzed patients with SLE treated at our hospital and associated facilities who were diagnosed according to the 1997 ACR or 2012 SLICC criteria and who began BEL treatment between December 2017 and August 2021. Patients who were followed for ≥ 12 months after BEL initiation were included. We investigated the changes in the patients' Safety of Estrogens in Lupus National Assessment–Systemic Lupus Erythematosus Disease Activity Index (SELENA-SLEDAI) scores at 3, 6, 9, and 12 months after the introduction of BEL, comparing patients with disease durations ≤ 5 years to those with > 5 years. A mixed-effects model was adjusted for the patients' ages, prednisolone dosages, initial SELENA-SLEDAI scores, Systemic Lupus International Collaborating Clinics (SLICC) damage index (SDI), hydroxychloroquine use, and lupus nephritis. Clinical manifestations including arthritis, skin lesions, and hematological abnormalities were monitored to assess the broader impacts of BEL.
Results
One hundred eleven patients were initially registered; among them, 97 patients were included in the final analysis. The study population (mean age, 41 years; mean SELENA-SLEDAI, 7 points; 51% using hydroxychloroquine) included 19 patients with a ≤ 5-year SLE duration and 78 with SLE durations > 5 years. The baseline SELENA-SLEDAI scores were higher in the ≤ 5-year group (p = 0.047), indicating more active disease. Patients with ≤ 5 years of disease had significantly greater improvements in SELENA-SLEDAI scores at 6, 9, and 12 months (p < 0.05).
Conclusions
These results highlight the importance of early BEL initiation in SLE, demonstrating that patients with shorter disease durations achieve more substantial improvements in disease activity with early BEL treatment. Our findings also reveal the potential benefits of early BEL intervention and suggest that incorporating the disease duration into treatment decisions may optimize patient outcomes.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13075-025-03581-0.
Keywords: Belimumab, Systemic lupus erythematosus, SELENA-SLEDAI, Disease duration, Early intervention, B cell therapy
Introduction
Systemic lupus erythematosus (SLE) is a complex autoimmune disease characterized by the production of autoantibodies, widespread inflammation, and diverse clinical manifestations affecting multiple organ systems. Central to the pathogenesis of SLE is the role of activated B cells, which not only produce autoantibodies but also secrete pro-inflammatory cytokines and present antigens to T cells, thereby perpetuating the autoimmune response [1, 2]. Recent studies have highlighted the critical involvement of memory B cells in the pathogenesis of SLE, emphasizing their role in sustaining autoantibody production and disease flares. The increase in the proportion of double-negative (DN) B cells and the extrafollicular B-cell responses has been linked to higher disease activity and glucocorticoid dependence in SLE patients [3].
Belimumab (BEL), a fully human monoclonal antibody targeting B-lymphocyte stimulator (BLyS), has emerged as a pivotal treatment in the management of SLE. Also known as BAFF (B-cell activating factor), BLyS plays a crucial role as a cytokine in the B-cell survival pathway, enhancing B-cell proliferation and antibody production [4]. By selectively binding to soluble BLyS, BEL inhibits its activity, thereby modulating the survival and function of B cells, particularly impacting the autoreactive clones [4]. This mechanism supports the use of BEL in the treatment of SLE, with the aim of curbing the aberrant B-cell activity that is central to the disease pathogenesis.
An early initiation of BEL during the initial phase of SLE has been suggested to suppress the progression of this disease [5]. The 2023 European League Against Rheumatism (EULAR) recommendations further advocate for the early use of biologics, including BEL, to improve disease control and minimize glucocorticoid use, underscoring the importance of timely intervention [6–11]. In particular, in patients with longer disease duration, changes in B-cell subsets [12] may limit the efficacy of BEL. Although the efficacy of BEL has been described, the influence of the duration and severity of SLE on treatment outcomes have not been established.
We thus conducted the present study to (i) clarify the association between the timing of the introduction of BEL and the treatment outcomes in patients with SLE and (ii) evaluate the benefits of early intervention with BEL. By investigating this relationship, we aimed to provide insights that may help clinicians optimize the timing of BEL therapy for maximal benefit.
Patients and methods
Patients
Patients diagnosed with SLE based on the 1997 American College of Rheumatology (ACR) Diagnostic Revision Criteria [13] or the 2012 Systemic Lupus International Collaborating Clinics (SLICC) Classification Criteria [14] at Nagasaki University Hospital or its affiliated hospitals in Nagasaki Prefecture and who started treatment with BEL between December 2017 and August 2021 were enrolled. The patients who were followed up for ≥ 12 months after the initiation of BEL treatment were included. Some of the patients provided written informed consent for the use of their data, and the opt-out strategy was used by the remainder of the patients. Patients who declined to give informed consent were excluded. The study was reviewed and approved by the Medical Ethics Committee of Nagasaki University Hospital (approval no. 19102116–6).
The evaluation of the patients' SELENA-SLEDAI scores over time
We compared the decrease in the patients' Safety of Estrogens in Lupus National Assessment-Systemic Lupus Erythematosus Disease Activity Index (SELENA-SLEDAI) scores [15] at different time points (3, 6, 9, and 12 months) after the introduction of BEL between two patient groups categorized based on their disease duration at the introduction of BEL (≤ 5 years and > 5 years).
Data collection
The patients' baseline characteristics were collected at the time of BEL initiation. The demographic data included the patient's age at their diagnosis of SLE, age at BEL initiation, sex, disease duration, and body mass index (BMI). Clinical characteristics such as comorbidities including Sjögren's syndrome (SS) and antiphospholipid syndrome (APS) were recorded, and disease manifestations such as rash, alopecia, arthritis, lupus nephritis (LN), and neuropsychiatric SLE (NPSLE) were assessed. Laboratory data included complete blood counts, i.e., the white blood cell (WBC) count, lymphocyte count, hemoglobin, and platelet count. Immunological parameters were also measured: complement 3 (C3), complement 4 (C4), total hemolytic complement (CH50), immunoglobulin (Ig)G, IgA, IgM, anti-double-stranded DNA antibody (anti-dsDNA), and anti-Smith (Sm) antibodies. The therapeutic data collected at the time of each patient's BEL initiation included the daily dose of glucocorticoids (prednisolone-equivalent), the use/non-use of hydroxychloroquine (HCQ), and immunosuppressive therapies such as tacrolimus (TAC), mycophenolate mofetil (MMF), cyclosporine (CyA), mizoribine (MZR), azathioprine (AZP), and methotrexate (MTX). We also collected data on organ damage using the Systemic Lupus International Collaborating Clinics (SLICC) damage index (SDI) [16] at baseline.
Statistical analyses
The patients' baseline characteristics are summarized as the median and interquartile range (IQR) or the number and percentage. We divided the patients into two groups based on disease duration: ≤ 5 years and > 5 years. Clinical characteristics were summarized and compared between these groups with the Wilcoxon signed-rank test or Fisher's exact test. We used a mixed-effects model [17] to compare the primary endpoint, i.e., SELENA-SLEDAI scores, between the groups at each time point. The model included the following adjustment variables: age at BEL initiation, prednisolone (PSL) dose, SELENA-SLEDAI score at BEL initiation, SDI at BEL initiation, hydroxychloroquine (HCQ) use/non-use, and the presence of LN. Dropouts at any time point were considered missing data. Although probability (p)-values are reported, no adjustments were made for multiplicity; caution is thus needed in interpretations of our findings, and the data should be considered reference values only. All statistical analyses were performed using JMP® Pro17.2 software (SAS Institute, Cary, NC).
Results
Patient characteristics
A total of 97 patients were included in the final analysis (Fig. 1). Of the initially registered 111 patients, one was excluded due to the absence of a baseline SELENA-SLEDAI score, and 13 patients were excluded because they could not be followed up for ≥ 12 months after their BEL introduction. The demographic and disease-related features of the 97 patients are summarized in Table 1. The majority of the patients were female (91.7%). The median age at the onset of SLE was 24.0 years (IQR 18.2–34.8 years), the median age at the introduction of BEL was 41 years (IQR 32–48.5 years), and the median disease duration of SLE was 150 months (IQR 66.5–268 months). At the time of BEL initiation, 19 of the 97 patients (19.6%) had a disease duration < 5 years, and the other 78 patients (80.4%) had a disease duration ≥ 5 years. The median SELENA-SLEDAI score of all 97 patients before the introduction of BEL was 5.0 (IQR 4.0–9.5). The median dose of glucocorticoids (prednisolone-equivalent) was 7.5 mg/day (IQR 5.0–12.5), and 49 (50.5%) patients were treated with the antimalarial hydroxychloroquine (HCQ). Forty-one (42.2%) patients exhibited LN, and 12 (12.4%) patients exhibited neuropsychiatric SLE (NPSLE). Of the 41 patients with LN, renal biopsy data from 36 patients were available, with histological classification including 3 cases of class II, 5 of class III, 3 of mixed class III and V, 11 of class IV, and 14 of class V. Of the 97 enrolled patients, 14 (14.4%) discontinued BEL: 12 (15.3%) in the > 5-year group and two (10.5%) in the ≤ 5-year group (Fig. 2). The common reasons for discontinuation of BEL were adverse events (n = 7) and disease progression (n = 4) (Table 2).
Fig. 1.
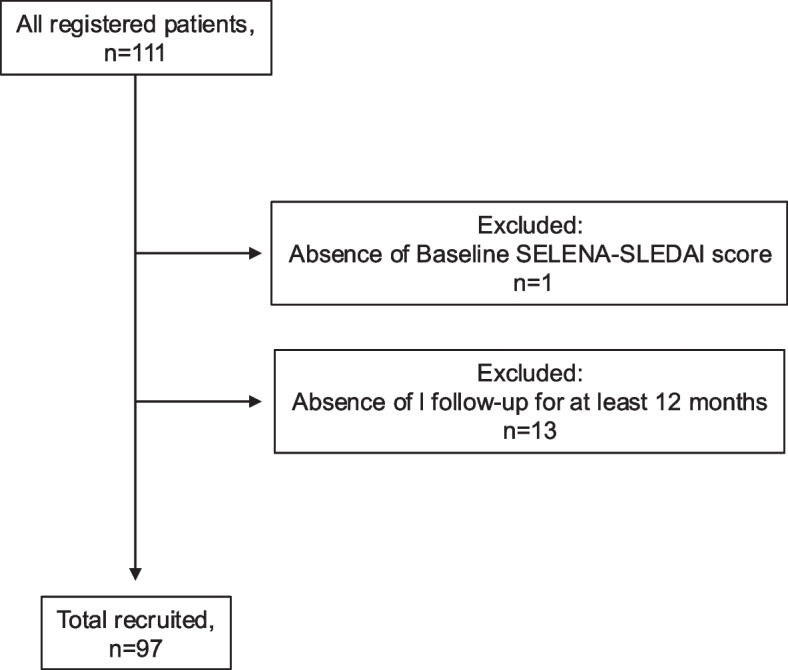
Flow diagram of the study's patient registration and inclusion
Table 1.
The baseline characteristics of the study's 97 patients with systemic lupus erythematosus (SLE)
| Variable | Median | IQR | Variable | Median | IQR |
|---|---|---|---|---|---|
| Age at diagnosis, yrs | 24 | (18.2–34.8) | CH50, U/mL | 30 | (21.8–40.1) |
| Age at BEL introduction, yrs | 41 | (32–48.5) | C3, mg/dL | 77.7 | (61.7–92.1) |
| Female, n (%) | 89/97 (91.7) | C4, mg/dL | 12.7 | (8.4–18.8) | |
| Disease duration, mos | 150 | (66.5–.268) | Sjögren syndrome, n (%) | 20/97 (20.6) | |
| BMI | 21.9 | (18.9–24.6) | APS, n (%) | 14/97 (14.4) | |
| Rash, n (%) | 39/97 (40.2) | Lupus nephritis, n (%) | 41/97 (42.3) | ||
| Alopecia, n (%) | 32/97 (32.9) | Neuropsychiatric SLE, n (%) | 12/97 (12.4) | ||
| Arthritis, n (%) | 49/97 (50.5) | SELENA-SLEDAI, points | 5.0 | (4.0–9.5) | |
| WBC, /mL | 5320 | SDI, points | 0 | (0–1.0) | |
| Lymphocyte count, /mL | 1010 | (3905–7100) | Glucocorticoid, mg/day | 7.5 | (5.0–12.5) |
| Hemoglobin, g/dL | 12 | (639–1405) | Antimalarial, n (%) | 49/97 (50.5) | |
| Platelets, × 104/mL | 22.7 | (10.9–13.2) | Immunosuppressant, n (%) | 80/97 (82.5) | |
| Anti-dsDNA, U/mL | 13.6 | (17.6–27.4) | TAC, n (%) | 61/97 (62.9) | |
| Anti-Sm, n, %* | 27/97 (27.8) | (3.8–32.4) | MMF, n (%) | 18/97 (18.6) | |
| IgG, mg/dL | 1390 | CyA, n (%) | 8/97 (8.2) | ||
| IgA, mg/dL | 260 | (1040–1700) | MZR, n (%) | 6/97 (6.2) | |
| IgM, mg/dL | 73 | (185–373) | AZP, n (%) | 3/97 (3.1) | |
| MTX, n (%) | 2/97 (2.1) | ||||
AZP Azathioprine, CyA Cyclosporine, IQR Interquartile range, MMF Mycophenolate mofetil, mos. Months, MTX Methotrexate, MZR Mizoribine, SDI Systemic Lupus International Collaborating Clinics damage index, TAC Tacrolimus, WBC White blood cell
Fig. 2.
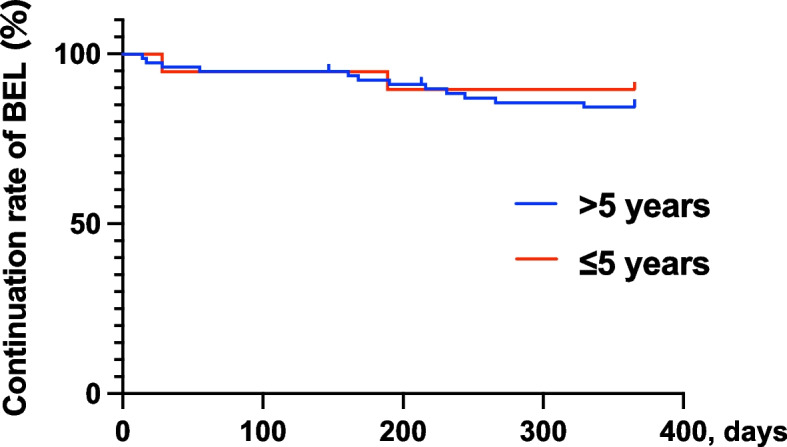
The rates of belimumab (BEL) continuation among the 97 patients with SLE (those with a disease duration ≤ 5 years or > 5 years at the time of belimumab initiation)
Table 2.
Causes of belimumab discontinuation
| Cause of discontinuation | ≤ 5 yrs n = 19 |
> 5 yrs n = 78 |
|---|---|---|
| Total | 2 (10.5%) | 12 (15.4%) |
| Insufficiency | 1 (5.3%) | 3 (3.9%) |
| Skin rash | 1 (5.3%) | 1 (1.3%) |
| Infections | 0 (0.0%) | 2 (2.6%) |
| Dizziness | 0 (0.0%) | 1 (1.3%) |
| Generalized pruritus | 0 (0.0%) | 1 (1.3%) |
| Fever | 0 (0.0%) | 1 (1.3%) |
| Desire for pregnancy | 0 (0.0%) | 1 (1.3%) |
| Pregnancy | 0 (0.0%) | 1 (1.3%) |
| Patient's preference | 0 (0.0%) | 1 (1.3%) |
We then divided the 97 patients into two groups based on their disease duration at the introduction of BEL (≤ 5 years and > 5 years) (Table 3). Lupus nephritis (LN) was significantly more frequent in the later-initiation group (p = 0.042), and the SELENA-SLEDAI scores were significantly lower in the later-initiation group (p = 0.047). In addition, the SDI scores were significantly higher in the later-initiation group (p = 0.032), reflecting more accumulated organ damage in patients with longer disease duration.
Table 3.
Comparison with disease duration
| Variable | ≤ 5 yrs (n = 19) | > 5 yrs (n = 78) | p-value | Variable | ≤ 5 yrs | > 5 yrs (n = 78) | p-value | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | IQR | Median | IQR | Median | IQR | Median | IQR | ||||
| Age at diagnosis, yrs | 38.5 | 21.4–43.2 | 23.5 | 17.9–32 | 0.030 | CH50, U/mL | 27 | 18.0–41.7 | 30.1 | 23.8–40.1 | 0.276 |
| Age at BEL introduction, yrs | 40 | 23–45) | 43 | 35–52 | 0.037 | C3, mg/dL | 65.5 | 55.9–96.0 | 78 | 63.4–90.8 | 0.496 |
| Female, n (%) | 18/19 (94.7) | 71/78 (91.0) | 0.580 | C4, mg/dL | 12.6 | 5.7–15.8 | 13.1 | 9.1–19.5 | 0.380 | ||
| Disease duration, mos | 24 | 13–47) | 189.5 | 110.8–280.8 | < 0.0001 | Sjögren syndrome, n (%) | 6/19 (31.6) | 14/78 (17.9) | 0.212 | ||
| BMI | 23.2 | 21.6–25.9 | 21.5 | 18.7–24.5 | 0.067 | APS, n (%) | 2/19 (10.53) | 12/78 (15.4) | 0.731 | ||
| Rash, n (%) | 10/19 (52.6) | 29/78 (37.2) | 0.297 | Lupus nephritis, n (%) | 4/19 (21.1) | 37/78 (47.4) | 0.042 | ||||
| Alopecia, n (%) | 8/19 (42.1) | 24/78 (30.8) | 0.417 | Neuropsychiatric SLE, n (%) | 0/19 (0.0) | 12/78 (15.4) | 0.115 | ||||
| Arthritis, n (%) | 13/19 (68.4) | 36/78 (46.2) | 0.124 | SELENA-SLEDAI | 8 | 4.0–12 | 4 | 3.8–8.0 | 0.047 | ||
| WBC, /mL | 4400 | 2900–5460 | 5400 | SDI | 0 | 0–0 | 1 | 0–2.0 | 0.032 | ||
| Lymphocyte count, /mL | 770 | 580–1070 | 1110 | 4100–7150 | 0.015 | Glucocorticoid, mg/day | 9.5 | 5.0–15.0 | 7.5 | 5.0–12.5 | 0.668 |
| Hemoglobin, g/dL | 12.1 | 11.1–13.2 | 11.9 | 687–1505 | 0.054 | Antimalarial, n (%) | 11/19 (57.9) | 38/78 (48.7) | 0.610 | ||
| Platelets, × 104/mL | 21.4 | 17.4–26.3 | 23.6 | 10.9–13.2 | 0.959 | Immunosuppressant, n (%) | 12/19 (63.2) | 68/78 (87.2) | 0.038 | ||
| Anti-dsDNA, U/mL | 19.8 | 2.7–54.0 | 11.1 | 17.9–27.9 | 0.400 | TAC, n (%) | 7/19 (36.8) | 54/78 (69.2) | 0.016 | ||
| Anti-Sm, n (%) | 5/19 (26.3) | 22/78 (28.2) | 1.000 | 4.8–30.9 | 0.359 | MMF, n (%) | 3/19 (5.25) | 7/78 (8.97) | 1.000 | ||
| IgG, mg/dL | 1707 | 968–2081 | 1376 | CyA, n (%) | 1/19 (5.25) | 7/78 (8.97) | 1.000 | ||||
| IgA, mg/dL | 227 | 179–312 | 282 | 1045–1622 | 0.248 | MZR, n (%) | 0/19(0.00) | 6/78 (7.69) | 0.059 | ||
| IgM, mg/dL | 79 | 54–222 | 72 | 185–381 | 0.496 | AZP, n (%) | 2/19(10.5) | 1/78 (1.28) | 0.097 | ||
| MTX, n (%) | 0/19(0.00) | 2/78 (2.56) | 1.000 | ||||||||
AZP Azathioprine, CyA Cyclosporine, IQR Interquartile range, MMF Mycophenolate mofetil, mos Months, MTX Methotrexate, MZR Mizoribine, SDI Systemic Lupus International Collaborating Clinics damage index, TAC Tacrolimus, WBC White blood cell
*p < 0.05. P-values were determined with the nonparametric Wilcoxon rank-sum test and Fisher's exact test
Differences in the decrease in the SELENA-SLEDAI score between the earlier and later initiations of BEL
Adjusting for the following variables in the mixed-effects model: age at BEL initiation, PSL dose, SELENA-SLEDAI score at BEL initiation, SDI at BEL initiation, HCQ use/non-use, and the presence of LN, the earlier-initiation group showed significantly greater improvement in SELENA-SLEDAI scores compared to the later-initiation group (Fig. 3). The trends in serum complement levels, anti-dsDNA antibody levels, and PSL dosage are shown in Supplementary Fig. S1.
Fig. 3.
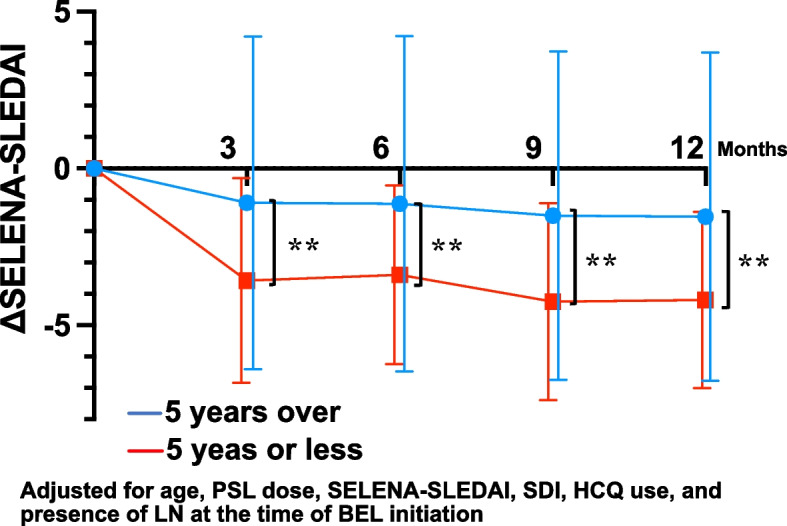
Changes in the patients' SELENA-SLEDAI scores after the introduction of belimumab (BEL) treatment in patients with different durations of SLE. The reduction in SELENA-SLEDAI scores at 3, 6, 9, and 12 months in the two groups (disease duration ≤ 5 years and > 5 years at the time of belimumab introduction) was compared using a mixed-effects model included the following adjustment variables: age at BEL initiation, prednisone (PSL) dose, SELENA-SLEDAI score at BEL initiation, Systemic Lupus International Collaborating Clinics damage index (SDI) at BEL initiation, hydroxychloroquine (HCQ) use/non-use, and the presence of lupus nephritis (LN). Error bar: standard deviation. **p < 0.01
Discussion
The results of this study highlight the significant benefits of initiating BEL treatment within the first 5 years of a patient's diagnosis of SLE. Our analyses revealed that the patients with shorter disease durations achieved greater improvements in SELENA-SLEDAI scores than those with longer disease durations. This finding aligns with emerging evidence suggesting that early intervention with BEL can modify the disease trajectory more effectively, potentially preventing long-term organ damage and reducing patients' reliance on glucocorticoids [5, 18, 19].
The biological rationale behind this observation may stem from differences in B-cell subpopulations and the expression of BAFF (B-cell activating factor) receptors across various stages of disease progression [3, 20]. The expression of BAFF receptor decreases as B cells differentiate, with memory B cells and switched memory B cells exhibiting significantly lower receptor levels [12]. In patients with prolonged disease durations, the predominance of these memory B cells may reduce the efficacy of BEL, which primarily targets immature and autoreactive B cells by inhibiting BAFF signaling. This hypothesis is supported by a report indicating that patients with longer disease durations have higher counts of memory/switched memory B cells [21]. Consequently, the limited action of BEL on these mature B-cell populations might explain the attenuated response in long-standing SLE cases, implying that in patients with a longer disease duration, the accumulations of memory B cells and DN B cells may limit the efficacy of BLyS inhibition. This suggests that new treatment strategies may be necessary to achieve optimal disease control in these patients, such as a dual inhibition of BLyS and APRIL (a proliferation-inducing ligand; also known as 'tumor necrosis factor ligand superfamily member 13'), CD20-targeting therapies, or combination approaches with chimeric antigen receptor (CAR)-T cell therapies [22].
Shorter disease duration has been associated with higher response rates to BEL. A study of an Italian cohort observed that patients with a disease duration ≤ 2 years had significantly greater improvement at 6 months and 24 months [5]. This finding suggested that initiating BEL within 2 years of the SLE diagnosis may enhance the likelihood of achieving favorable outcomes, supporting the importance of early biologic intervention. Consistent with this, a recent Chinese study further demonstrated the clinical benefits of very early BEL initiation—specifically within 6 months of disease onset [23]. In that study, patients receiving BEL in the early phase of SLE showed a significantly higher rate of achieving lupus low disease activity status (LLDAS) at Week 24, along with more frequent glucocorticoid tapering and improved patient-reported outcomes compared to those receiving standard care alone. Notably, even among patients with lupus nephritis, nearly half achieved complete renal response within 24 weeks. These findings reinforce the importance of timely BEL initiation to optimize clinical outcomes. In parallel, real-world data from the United States also support early introduction of BEL, showing that patients who began BEL prior to immunosuppressant use had lower flare rates, shorter glucocorticoid use duration, and delayed onset of organ damage compared to those who received BEL after immunosuppressants [24]. These benefits were evident regardless of baseline SLE severity, highlighting the broader applicability of early BEL intervention in diverse clinical settings. Together, these international studies corroborate our findings in the present Japanese cohort, where early BEL use was associated with better outcomes, thereby validating the clinical value of prompt BEL initiation across multiple populations.
Following the similar results observed in the Italian cohort, our present Japanese cohort demonstrated comparable findings, further validating the benefits of early BEL initiation in diverse populations. An investigation with sub-analyses of patient cohorts with joint and skin involvement indicated that 17% of the patients with joint symptoms and 18.5% of those with skin symptoms had a disease duration ≤ 2 years at baseline [18]. While the direct relationship between disease duration and treatment efficacy was not the primary focus of that investigation, the results emphasize the potential advantages of early BEL introduction.
This disparity in response underscores the importance of targeting B cell-driven autoimmunity at an earlier, more reversible stage of SLE. In the early phase of the disease, autoreactive B cells are more likely to be naïve or transitional, making them susceptible to BAFF inhibition. In contrast, as SLE progresses, the immune system becomes increasingly dominated by antigen-experienced memory B cells, which are less dependent on BAFF for survival [12, 22]. This shift in B-cell subpopulations may diminish the capacity of BEL to exert its therapeutic effects, highlighting the necessity of early treatment initiation.
Data from clinical trials and real-world studies indicate that early BEL introduction not only enhances disease control but also mitigates organ damage by curtailing inflammation and flare frequency [5, 18]. Reduced glucocorticoid exposure through early biologic use is a key factor in minimizing long-term complications, reinforcing the value of prompt intervention.
One of the key strengths of the present study lies in its comprehensive analysis of patients across different disease stages, providing insights into the benefits of early BEL initiation. By incorporating data from a real-world setting, our findings reflect practical clinical outcomes and contribute to the growing body of evidence supporting BEL's use beyond controlled trial environments. Our findings also emphasize the importance of integrating disease duration as a critical factor in treatment planning, which may influence future guidelines and therapeutic approaches. Another notable strength of this study is the long-term follow-up period, which allowed us to capture sustained responses and assess the durability of BEL efficacy over time. This extended observation period helps address the gaps in previous studies that focused primarily on short-term outcomes. Our inclusion of patients with varying levels of immunosuppressant exposure (i.e., immunosuppressant-naïve and previously treated patients) enhances the generalizability of our findings to broader patient populations, offering valuable insights into BEL's role as a first-line biological option.
Despite these strengths, several study limitations must be acknowledged. This retrospective study introduced potential biases in patient selection and data collection. Prospective studies, such as multicenter observational cohorts, could provide further validation of our findings while addressing the inherent limitations of a retrospective design. The absence of a priori sample size calculation may have reduced the statistical power to detect subtle effects and limited the generalizability of the findings. The sample size for the patients with shorter disease durations (n = 19) was much smaller than that of the long-standing SLE patients (n = 78), potentially affecting the statistical power of our findings. Detailed information on the clinical manifestations that led to the initiation of Belimumab was not consistently available in the medical records, which limited our ability to analyze treatment indications in depth. Similarly, data on the clinical activity of lupus nephritis at the time of treatment initiation were not systematically collected, making it difficult to assess the relationship between disease activity and treatment response. Additionally, while we accounted for various confounding factors, the influence of unmeasured variables such as genetic predisposition or environmental triggers cannot be ruled out. Future prospective studies with standardized data collection and predefined power analyses are warranted to validate our results.
Further considerations include varying treatment protocols across institutions and the recent approval of hydroxychloroquine (HCQ) for the treatment of SLE in Japan, which could influence evaluations of the advantages of early BEL treatment. As HCQ becomes more widely used, its combination with BEL in early-stage SLE patients may yield different therapeutic outcomes, necessitating updated analyses that reflect these evolving treatment landscapes.
Another limitation of this study is its reliance on SELENA-SLEDAI as the primary measure of disease activity. While widely used, the SELENA-SLEDAI may not fully capture subtle clinical improvements or specific organ involvement that could influence treatment responses. Incorporating additional indices such as the Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI) [25] or musculoskeletal assessments may provide a more comprehensive evaluation of BEL's efficacy. The relatively low baseline SLEDAI values in many of the patients may indicate milder disease activity or well-controlled disease at the time of Belimumab initiation. This reduces the ability to generalize our findings to patients with high disease activity, and future studies should include patients with a broader range of disease severities to better assess the efficacy of Belimumab in more active disease states.
Lastly, our study population was drawn from a limited geographical region, which may restrict the applicability of the results to other clinical settings. Future studies should aim to validate these findings through larger, multicenter cohorts to ensure broader relevance and strengthen the evidence supporting early BEL intervention.
Conclusions
This study suggests that initiating BEL treatment during the early phase of SLE may be associated with improved disease control, greater reductions in glucocorticoid use, and potentially lower long-term organ damage. These findings are consistent with the 2023 EULAR recommendations [6], highlighting the importance of early biologic therapy in optimizing SLE treatment outcomes. Targeting B-cell activation during the initial stages of SLE may enhance the therapeutic efficacy and improve long-term prognoses. However, given the retrospective and observational nature of this study, along with the possibility of confounding by indication, our results should be interpreted with caution. Further prospective and larger multicenter studies are essential to confirm our findings and refine treatment strategies for diverse patient populations.
Supplementary Information
Acknowledgements
Not applicable.
Abbreviations
- ACR
American College of Rheumatology
- APS
Antiphospholipid syndrome
- BAFF
B-cell activating factor
- BEL
Belimumab
- BMI
Body mass index
- C3
Complement 3
- C4
Complement 4
- CH50
Total hemolytic complement
- CLASI
Cutaneous Lupus Erythematosus Disease Area and Severity Index
- CyA
Cyclosporine
- dsDNA
Double-stranded DNA
- EULAR
European League Against Rheumatism
- HCQ
Hydroxychloroquine
- IQR
Interquartile range
- LN
Lupus nephritis
- MMF
Mycophenolate mofetil
- MTX
Methotrexate
- MZR
Mizoribine
- NPSLE
Neuropsychiatric systemic lupus erythematosus
- PSL
Prednisolone
- SDI
Systemic Lupus International Collaborating Clinics damage index
- SELENA-SLEDAI
Safety of Estrogens in Lupus National Assessment–Systemic Lupus Erythematosus Disease Activity Index
- SLE
Systemic lupus erythematosus
- SLICC
Systemic Lupus International Collaborating Clinics
- Sm
Smith (antibody)
- SS
Sjögren's syndrome
- TAC
Tacrolimus
Authors’ contributions
Drs. KI and MU had full access to all of the data in the study. They took responsibility for the integrity and accuracy of the data analysis. Study conception and design: KK, KI, SS, and AK. Data acquisition: KK, KI, MU, TS, TS, YN, AO, YH, KF, TA, TM, MF, NM, and AK. Analysis and interpretation of data: KK, KI, MU, and SS. All authors read and approved the final manuscript.
Funding
This work was supported by grants from the Japan Society for the Promotion of Science (JSPS) KAKENHI (no. 23K07895 to KI and nos. 21K20905 and 22K16362 to MU), a JCR Grant for Promoting Research for FRONTIER (to KI), and the Takeda Science Foundation (to KI).
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
This study was performed in accord with the Declaration of Helsinki and was approved by the Investigation and Ethics Committee at Nagasaki University Hospital (approval no. 19102116–6). The patients gave their informed consent to be subjected to the protocol.
Consent for publication
Some of the patients provided written informed consent for the use and publications of their data, and the opt-out strategy was used by the remainder of the patients.
Competing interests
KI has received speaker honoraria from AbbVie GK, Asahi Kasei Pharma Corporation, AstraZeneca K.K., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Eli Lilly Japan K.K., GlaxoSmithKline K.K. (GSK), Janssen Pharmaceutical K.K., Mitsubishi Tanabe Pharma Corporation, Ono Pharmaceutical Co., Ltd., and Taisho Pharmaceutical Co., Ltd. MU has received honoraria from AbbVie GK, Asahi Kasei Pharma Corporation, AstraZeneca K.K., and GlaxoSmithKline K.K. (GSK). TS has received honoraria from AstraZeneca K.K. and GlaxoSmithKline K.K. (GSK). AK has received honoraria from AbbVie GK, Asahi Kasei Pharma Corporation, Boehringer Ingelheim, Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Eli Lilly Japan K.K., Ono Pharmaceutical Co., Ltd., Pfizer, and Taisho Pharmaceutical Co., Ltd.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Kunihiro Ichinose, Email: kichinose@med.shimane-u.ac.jp.
Masataka Umeda, Email: masatakau0807@nagasaki-u.ac.jp.
References
- 1.Tsokos GC. Systemic lupus erythematosus. N Engl J Med. 2011;365(22):2110–21. [DOI] [PubMed] [Google Scholar]
- 2.Tsokos GC. Autoimmunity and organ damage in systemic lupus erythematosus. Nat Immunol. 2020;21:605–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gatto M, Zen M, Iaccarino L, Doria A. New therapeutic strategies in systemic lupus erythematosus management. Nat Rev Rheumatol. 2019;15(1):30–48. [DOI] [PubMed] [Google Scholar]
- 4.Mockel T, Basta F, Weinmann-Menke J, Schwarting A. B cell activating factor (BAFF): Structure, functions, autoimmunity and clinical implications in Systemic Lupus Erythematosus (SLE). Autoimmun Rev. 2021;20(2):102736. [DOI] [PubMed] [Google Scholar]
- 5.Gatto M, Saccon F, Zen M, Regola F, Fredi M, Andreoli L, Tincani A, Urban ML, Emmi G, Ceccarelli F, et al. Early disease and low baseline damage as predictors of response to belimumab in patients with systemic lupus erythematosus in a real-life setting. Arthritis Rheumatol. 2020;72(8):1314–24. [DOI] [PubMed] [Google Scholar]
- 6.Fanouriakis A, Kostopoulou M, Andersen J, Aringer M, Arnaud L, Bae SC, Boletis J, Bruce IN, Cervera R, Doria A, et al. EULAR recommendations for the management of systemic lupus erythematosus: 2023 update. Ann Rheum Dis. 2024;83(1):15–29. [DOI] [PubMed] [Google Scholar]
- 7.Navarra SV, Guzman RM, Gallacher AE, Hall S, Levy RA, Jimenez RE, Li EK, Thomas M, Kim HY, Leon MG, et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet. 2011;377(9767):721–31. [DOI] [PubMed] [Google Scholar]
- 8.Furie R, Petri M, Zamani O, Cervera R, Wallace DJ, Tegzova D, Sanchez-Guerrero J, Schwarting A, Merrill JT, Chatham WW, et al. A phase III, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits B lymphocyte stimulator, in patients with systemic lupus erythematosus. Arthritis Rheum. 2011;63(12):3918–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Furie R, Rovin BH, Houssiau F, Malvar A, Teng YKO, Contreras G, Amoura Z, Yu X, Mok CC, Santiago MB, et al. Two-year, randomized, controlled trial of belimumab in lupus nephritis. N Engl J Med. 2020;383(12):1117–28. [DOI] [PubMed] [Google Scholar]
- 10.Sakai H, Miyazaki Y, Nakayamada S, Kubo S, Hanami K, Fukuyo S, Yamaguchi A, Miyagawa I, Ueno M, Tanaka H, et al. Efficacy, safety, and optimal intervention of belimumab for proliferative lupus nephritis patients in real-world settings: LOOPS registry. Rheumatology (Oxford). 2024;64(4):1930–9. [DOI] [PubMed] [Google Scholar]
- 11.Sishi L, Zhang J, You X, Chen B, Liang Y, Zhou Y, Ding X, Lv Y, Zhang H, Su B, et al. Efficacy and safety of belimumab in patients with lupus nephritis: a real-world retrospective observational study. Rheumatology (Oxford). 2023;64(2):614–22. [DOI] [PubMed] [Google Scholar]
- 12.Dalakas MC. B cells as therapeutic targets in autoimmune neurological disorders. Nat Clin Pract Neurol. 2008;4(10):557–67. [DOI] [PubMed] [Google Scholar]
- 13.Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997;40(9):1725. [DOI] [PubMed] [Google Scholar]
- 14.Petri M, Orbai AM, Alarcon GS, Gordon C, Merrill JT, Fortin PR, Bruce IN, Isenberg D, Wallace DJ, Nived O, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64(8):2677–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mikdashi J, Nived O. Measuring disease activity in adults with systemic lupus erythematosus: the challenges of administrative burden and responsiveness to patient concerns in clinical research. Arthritis Res Ther. 2015;17(1):183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ghazali WSW, Daud SMM, Mohammad N, Wong KK. Slicc damage index score in systemic lupus erythematosus patients and its associated factors. Medicine (Baltimore). 2018;97(42):e12787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gomes DGE. Should I use fixed effects or random effects when I have fewer than five levels of a grouping factor in a mixed-effects model? PeerJ. 2022;10:e12794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zen M, Gatto M, Depascale R, Regola F, Fredi M, Andreoli L, Franceschini F, Urban ML, Emmi G, Ceccarelli F, et al. Early and late response and glucocorticoid-sparing effect of belimumab in patients with systemic lupus erythematosus with joint and skin manifestations: results from the Belimumab in Real Life Setting Study-Joint and Skin (BeRLiSS-JS). J Pers Med. 2023;13(4):691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tani C, Zucchi D, Cardelli C, Elefante E, Signorini V, Schiliro D, Cascarano G, Gualtieri L, Valevich A, Puccetti G, et al. Analysis of belimumab prescription and outcomes in a 10-year monocentric cohort: is there an advantage with early use? RMD Open. 2024;10(2):e003981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee DSW, Rojas OL, Gommerman JL. B cell depletion therapies in autoimmune disease: advances and mechanistic insights. Nat Rev Drug Discov. 2021;20(3):179–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhu L, Yin Z, Ju B, Zhang J, Wang Y, Lv X, Hao Z, He L. Altered frequencies of memory B cells in new-onset systemic lupus erythematosus patients. Clin Rheumatol. 2018;37(1):205–12. [DOI] [PubMed] [Google Scholar]
- 22.Dorner T, Lipsky PE. The essential roles of memory B cells in the pathogenesis of systemic lupus erythematosus. Nat Rev Rheumatol. 2024;20(12):770–82. [DOI] [PubMed] [Google Scholar]
- 23.Lu C, He N, Dou L, Yu H, Li M, Leng X, Zeng X. Belimumab in early systemic lupus erythematosus: A propensity score matching analysis. Immun Inflamm Dis. 2024;12(8):e1362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rubin B, Chen Y, Worley K, Rabideau B, Wu B, Chang R, DerSarkissian M. Improved health outcomes in patients with systemic lupus erythematosus following early belimumab initiation without prior immunosuppressant use: a real-world descriptive study. Rheumatol Ther. 2024;11(4):947–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Albrecht J, Taylor L, Berlin JA, Dulay S, Ang G, Fakharzadeh S, Kantor J, Kim E, Militello G, McGinnis K, et al. The CLASI (Cutaneous Lupus Erythematosus Disease Area and Severity Index): an outcome instrument for cutaneous lupus erythematosus. J Invest Dermatol. 2005;125(5):889–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.


