Abstract
Cardiovascular diseases such as coronary artery disease, myocardial infarction, and heart failure impact millions of people annually globally and are a major cause of disease and death. This study explores the predictive capabilities of novel metabolic indices (TyG, HOMA-IR, TG/HDL-C, and VAI) for major adverse cardiovascular events (MACE) and analyzes their relationships with diabetes and cardiovascular risks. Using data from the National Health and Nutrition Examination Survey (NHANES) spanning from 2003 to 2018, we applied multiple machine learning algorithms to evaluate nine metabolic indicators including cholesterol levels, triglycerides, insulin, and waist circumference. Through cross-validation to validate model performance, the XGBoost algorithm demonstrated the most accurate performance in predicting cardiovascular outcomes, particularly for diseases like angina and heart failure. Additionally, SHAP value analysis confirmed the critical roles of waist circumference and METS-IR in predicting adverse cardiovascular events. Furthermore, we employed 100 machine learning algorithms to calculate the AUC values of metabolic indicators in predicting AP, CHD, HF, and MI, showing that METS-IR had the greatest contribution in these aspects. This research highlights the significance of metabolic indices in stratifying cardiovascular risks and presents potential avenues for targeted preventive strategies.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13098-025-01753-1.
Keywords: Heart failure, Myocardial infarction, Angina pectoris, Coronary disease, Metabolism
Introduction
Major Adverse Cardiovascular Events (MACE) refers to cardiovascular complications occurring in patients with myocardial injury after non-cardiac surgery (MINS). These complications include coronary disease, myocardial infarction, myocardial ischemia, and heart failure [1]. Angina pectoris is chest pain caused by myocardial ischemia, typically aggravated by physical activity or emotional stress, lasting for more than 30 to 60 s, and relieved by rest and nitroglycerin use. It is estimated that approximately 10 million people in the United States suffer from angina pectoris, with over 500,000 new cases each year [2]. Coronary disease (CHD) is a cardiovascular disease caused by insufficient blood supply to the myocardium due to coronary artery disease and is one of the leading causes of death worldwide. As of 2020, over 10 million people die from coronary disease each year globally [3]. Myocardial infarction (MI) is caused by interrupted blood flow to the myocardial tissue, resulting in tissue death. According to statistics from the American Heart Association, approximately 805,000 Americans experience myocardial infarction each year, with around 605,000 as first-time cases and 200,000 as recurrent myocardial infarctions. Additionally, approximately 122,000 people die from myocardial infarction annually, accounting for 14% of total deaths [4, 5]. Heart failure (HF) is a disease where the heart is unable to pump blood effectively enough to meet the body's needs, leading to symptoms such as fatigue, difficulty breathing, and edema. In the United States, heart failure is a common and increasingly severe disease. It is estimated that approximately 6 million Americans suffer from heart failure, with approximately 1 million people hospitalized for heart failure each year [6].
The development of diabetes is closely related to the occurrence of adverse cardiovascular events. Currently, the main metabolic score for evaluating diabetes is HOMA-IR (Homeostasis Model Assessment-Insulin Resistance Index) [7]. However, four new indicators—VAI (Visceral Adiposity Index), TyG (Triglyceride and Glucose), TG/HDL-C (Ratio of triglyceride to high-density lipoprotein cholesterol), and METS-IR (metabolic score for insulin resistance)—play key roles in predicting the onset of diabetes and are expected to replace HOMA-IR as the key scoring system for diagnosing diabetes [8]. At the same time, in adverse cardiovascular events [9], patients with high levels of VAI have a significantly increased risk of angina [10–12]; when HOMA-IR exceeds 3.59, an increase in HOMA-IR can significantly raise the mortality rate of patients with coronary heart disease combined with hypertension, and the elevation of HOMA-IR also significantly increases the risk of atherosclerosis [13, 14]; studies have also shown that the TyG index is positively correlated with the incidence of myocardial infarction in American adults, serving as a key metabolic score for predicting myocardial infarction occurrence [15]. Recent research has also indicated that a high TyG index may induce exacerbated inflammatory responses and oxidative stress, which play important roles in the progression of heart failure [16]. Meanwhile, TG/HDL-C is significantly associated with the five-year mortality risk of heart failure patients [17]. A high TG/HDL-C ratio reflects lipid metabolism disorders and abnormal blood lipid levels, which may increase the risk of endothelial dysfunction and the occurrence of atherosclerosis [18]. However, there has been no literature reported on which of the five metabolic scores—HOMA-IR, VAI, TyG, TG/HDL-C, and METS-IR—has an impact on adverse cardiovascular events.
In this study, we used data from the National Health and Nutrition Examination Survey (NHANES, 2003–2018) to investigate the relationship between metabolic indicators and scores and angina pectoris, coronary disease, myocardial infarction, and heart failure in major adverse cardiovascular events. We employed six binary classification machine learning algorithms to analyze angina pectoris, coronary disease, myocardial infarction, and heart failure, and selected the most suitable binary classification machine learning algorithm for evaluating various metabolic indicators and score models. Additionally, based on Shapley Additive explanations, we provided explanations for the model that best evaluated major adverse cardiovascular events and sought out the most crucial metabolic indicators and scores in these events.The purpose of this study is to explore the most critical metabolic scores affecting adverse cardiovascular events through a variety of machine learning algorithms.
Method and materials
Study participants
The NHANES study conducted interviews and physical examinations on the US population using a variety of survey strategies. Our study included eight consecutive cycles of NHANES from 2003 to 2018. The exclusion criteria were as follows: (1) participants with uncertain status of angina pectoris, coronary disease, myocardial infarction, and heart failure based on the NHANES questionnaire (indicated by a response of 7 or 9); (2) missing values for the included metabolic indicators (see Supplementary Fig. 1–4 for details); (3) participants under the age of 20 were excluded.
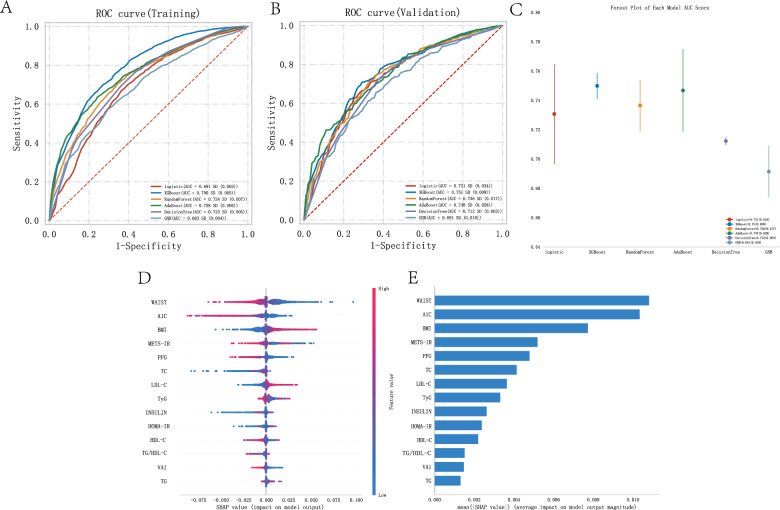
Fig. 1.
Machine learning model evaluation of various metabolic indicators and scores in the angina pectoris population. A AUC results graph of the training set. B AUC results graph of the testing set. C Forest plot of AUC scores. D Summary of the shap model of the XGBoost algorithm. E Importance ranking results of the shap model of the XGBoost algorithm
Data collection
Demographic and social characteristics of the participants were collected from the US population data. These characteristics included gender, age (in years), race/ethnicity, education level (college or above, high school or equivalent, below high school), and poverty income ratio (PIR), etc. In this study, to minimize the impact of factors other than metabolic indicators on major adverse cardiovascular events, we only included four essential demographic and social characteristics: gender, age, race, and PIR.
Our study also included nine metabolic indicators in human blood, including TC (total cholesterol), AIC (glycated hemoglobin), TG (triglycerides), LDL-C (low-density lipoprotein cholesterol), INSULIN (insulin), HDL-C (high-density lipoprotein cholesterol), PFG (fasting plasma glucose), BMI (body mass index), and WAIST (waist circumference). Blood samples were processed, stored, and transported to the Fairview Medical Center laboratory at the University of Minnesota in Minneapolis, Minnesota, for analysis. In addition, we included five metabolic scores closely related to the cardiovascular system in human blood to optimize the predictive and explanatory power of the model: VAI, TyG, TG/HDL-C, METS-IR, and HOMA-IR. We also selected participants who reported being diagnosed or informed by a doctor of angina pectoris, coronary disease, myocardial infarction, and heart failure as the binary outcome variables for machine learning.
Machine learning
In this study, multiple machine learning algorithms were applied to evaluate the predictive models of angina pectoris, coronary disease, myocardial infarction, and heart failure composed of various metabolic indicators and scores. The research data were divided into an 80% training set and a 20% testing set. In the binary logistic regression algorithm, we used maximum likelihood estimation to fit the model, determine the relationship between independent and dependent variables, and predict the classification probability of samples by mapping the output of the linear function into a probability range [19]. We also employed the AdaBoost algorithm as an ensemble learning method, enhancing the accuracy of classification by combining multiple weak learners, such as decision trees. This algorithm focuses more on misclassified samples, thus optimizing the overall model performance [20]. The decision tree algorithm is used to describe the relationship between features and the target variable, obtaining clear classification rules by continuously dividing the feature space. This model is intuitive and suitable for decision-making under specific conditions [21]. XGBoost, as an optimized gradient boosting algorithm, possesses unique advantages in improving model prediction accuracy and controlling overfitting. We used this algorithm for model construction and enhanced the generalization ability of the model through regularization techniques [22]. We also employed the random forest algorithm, constructing multiple decision trees by randomly selecting and sampling features and the sample set, and then combining them into a powerful ensemble model to predict the binary models of angina pectoris, coronary disease, myocardial infarction, and heart failure separately [23]. Additionally, the Gaussian Naive Bayes classification (GNB) algorithm was used to estimate the prior probability of each category, as well as the mean and variance of each feature. By calculating the mean and variance of each feature under different categories, the Gaussian distribution parameters of the features in the binary models of angina pectoris, coronary disease, myocardial infarction, and heart failure can be obtained. These parameters can be used to calculate the posterior probability of a sample belonging to a certain category given a specific feature condition [24].
The training process of all models was conducted using cross-validation methods to ensure the reliability and robustness of the results. Furthermore, we evaluated the classification performance of different models, focusing on metrics such as accuracy, recall, and F1 score, to select the best classification model for further analysis. After combining the discriminative features of each model, we selected the models that were most suitable for identifying angina pectoris, coronary disease, myocardial infarction, and heart failure. We used SHAP values to explain the selected models, incorporating the risk variables related to angina pectoris, coronary disease, myocardial infarction, and heart failure from the participants spanning from 2003 to 2018 in the NHANES questionnaire data.
We also utilized 100 combinations of eight machine learning algorithms: Enet, SVM, glm, RF, GBM, plsRglmmodel, Ridge, and Lasso, to evaluate the predictive effectiveness of the metabolic indicators and scores we established for AP, CHD, HF, and MI. In this study, we employed a clinical sample dataset with AP, CHD, HF, and MI as the target variables. Based on previous research and expert recommendations, we constructed a series of metabolic indicators and scores to predict the occurrence of different cardiovascular events. Subsequently, we built a hundred different predictive models by utilizing various combinations of these machine learning algorithms and trained and optimized the models using the training dataset. Finally, we evaluated the performance of these models using a testing dataset, measuring the AUC (Area Under the Curve). These evaluation results will help determine which metabolic indicators and scores have the best predictive effectiveness for AP, CHD, HF, and MI.
Results
Evaluation of the influence of metabolic indicators and scores on angina pectoris using binary classification machine learning algorithms and machine learning algorithm explanations
In a representative sample of 16,299 US adults, 48.37% were male (= 7,883) and 51.63% were female (= 8,416). Angina pectoris was identified in 435 patients, accounting for 2.67% of the study population. The age of the angina pectoris group ranged from 60 to 77 years, with an average PIR of 1.79. Among the patients, 11% were Mexican American, 7% were Other Hispanic, 62% were Non-Hispanic White, 14% were Non-Hispanic Black, and 6% were Other Race Including Multi Racial. In the blood of the angina pectoris group, the average concentrations were as follows: TC (173 mg/dL), A1 C (5.9 mg/dL), TG (119 mg/dL), LDL-C (97 mg/dL), INSULIN (11.28 uU/mL), HDL-C (1.22 mg/dL), PFG (110 mg/dL), BMI (29.54 kg/m2), WAIST (105.10 cm), VAI (160.47), TyG (8.83), TG/HDL-C (95.88), METS-IR (1.79), and HOMA-IR (3.14) (Table 1). Figure 1A shows the ROC curves of each model based on the training data, providing a comparative analysis of model performance and their AUC values. The AUC for the logistic algorithm was 0.691, XGBoost algorithm was 0.790, RandomForest algorithm was 0.734, AdaBoost algorithm was 0.758, DecisionTree algorithm was 0.723, and GNB algorithm was 0.683. These ROC curves depict the sensitivity and specificity of various metabolic indicators and score models in influencing angina pectoris, serving as key benchmarks for evaluating the effects of different algorithms based on these indicators and scores. Figure 1B extends this evaluation to the validation data, showing the ROC curves that reflect the generalization ability of the models in unseen data. The AUC for the logistic algorithm was 0.731, XGBoost algorithm was 0.751, RandomForest algorithm was 0.736, AdaBoost algorithm was 0.748, DecisionTree algorithm was 0.712, and GNB algorithm was 0.691. The AUC values provide a quantitative assessment of the accuracy of each model, which is crucial for validating their predictive ability in clinical and research settings. Figure 1C compares the AUC scores of different ML models in the validation data using a forest plot. Each point in the plot represents the median AUC value of a model, and the error bars represent the confidence intervals, emphasizing the performance differences between the models. In both the training and validation sets, the XGBoost algorithm achieved the highest AUC value, demonstrating the critical role played by various metabolic indicators and scores in influencing angina pectoris. In Fig. 1D and E, we subsequently built the SHAP models using the XGBoost algorithm, showing that waist circumference had the greatest impact on angina pectoris in the metabolic models. METS-IR had the largest impact among the five metabolic scores (METS-IR, TyG, HOMA-IR, TG/HDL-C, and VAI) on angina pectoris.
Table 1.
Baseline characteristics of participants associated with angina (AP)
| Variable | AP, N = 4351 | CON, N = 15,8641 | p-value2 |
|---|---|---|---|
| AGE, Median (IQR) | 68.00 (60.00–77.00) | 48.00 (34.00–63.00) | < 0.001 |
| PIR, Median (IQR) | 1.79 (1.09–3.14) | 2.16 (1.14–4.10) | < 0.001 |
| TC, Median (IQR) | 173.00 (145.00–210.00) | 190.00 (164.00–218.00) | < 0.001 |
| A1 C, Median (IQR) | 5.90 (5.50–6.60) | 5.50 (5.20–5.80) | < 0.001 |
| TG, Median (IQR) | 119.00 (86.00–165.00) | 103.00 (72.00–151.00) | < 0.001 |
| LDL-C, Median (IQR) | 97.00 (72.50–123.00) | 112.00 (89.00–136.00) | < 0.001 |
| INSULIN, Median (IQR) | 11.28 (6.67–19.95) | 9.38 (5.89–15.44) | < 0.001 |
| HDL-C, Median (IQR) | 1.22 (1.03–1.50) | 1.34 (1.11–1.63) | < 0.001 |
| PFG, Median (IQR) | 110.00 (99.00–130.00) | 100.00 (92.20–110.00) | < 0.001 |
| BMI, Median (IQR) | 29.54 (26.13–34.09) | 27.82 (24.20–32.30) | < 0.001 |
| WAIST, Median (IQR) | 105.10 (96.30–115.85) | 97.50 (87.50–108.40) | < 0.001 |
| VAI, Median (IQR) | 160.47 (101.56–241.89) | 125.80 (76.91–209.69) | < 0.001 |
| TyG, Median (IQR) | 8.83 (8.49–9.27) | 8.57 (8.17–9.01) | < 0.001 |
| TG/HDL-C, Median (IQR) | 95.88 (61.31–148.97) | 75.91 (46.23–125.00) | < 0.001 |
| METS-IR, Median (IQR) | 1.79 (1.69–1.91) | 1.71 (1.61–1.81) | < 0.001 |
| HOMA-IR, Median (IQR) | 3.14 (1.79–6.06) | 2.38 (1.42–4.16) | < 0.001 |
| Gender, n (%) | < 0.001 | ||
| Male | 253 (58) | 7,630 (48) | |
| Female | 182 (42) | 8,234 (52) | |
| Race, n (%) | < 0.001 | ||
| Mexican American | 50 (11) | 2,550 (16) | |
| Other Hispanic | 29 (7) | 1,374 (9) | |
| Non-Hispanic White | 268 (62) | 7,091 (45) | |
| Non-Hispanic Black | 62 (14) | 3,186 (20) | |
| Other Race (Multi-Racial) | 26 (6) | 1,663 (10) |
1Median (IQR) or Frequency (%)
2Wilcoxon rank sum test; Pearson's Chi-squared test
Evaluation of the influence of metabolic indicators and scores on coronary disease using binary classification machine learning algorithms and machine learning algorithm explanations
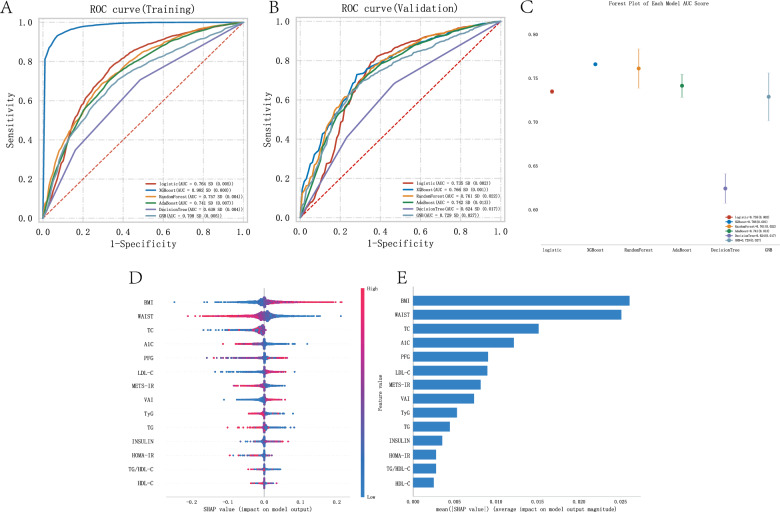
In a representative sample of 16,287 US adults, 48.33% were male (= 7,871) and 51.67% were female (= 8,416). coronary disease was identified in 679 patients, accounting for 3.73% of the study population. The age of the coronary disease group ranged from 63 to 79 years, with an average PIR of 2.15. Among the patients, 9.1% were Mexican American, 6.7% were Other Hispanic, 67% were Non-Hispanic White, 11% were Non-Hispanic Black, and 6.6% were Other Race Including Multi Racial. In the blood of the coronary disease group, the average concentrations were as follows: TC (166 mg/dL), A1 C (5.9 mg/dL), TG (115 mg/dL), LDL-C (90 mg/dL), INSULIN (11.19 uU/mL), HDL-C (1.22 mg/dL), PFG (111 mg/dL), BMI (28.59 kg/m2), WAIST (104.10 cm), VAI (157.53), TyG (8.81), TG/HDL-C (96.49), METS-IR (1.82), and HOMA-IR (3.09) (Table 2). The ROC curves of each model based on the training data, providing a comparative analysis of model performance and their AUC values (Fig. 2A). The AUC for the logistic algorithm was 0.764, XGBoost algorithm was 0.982, RandomForest algorithm was 0.757, AdaBoost algorithm was 0.741, DecisionTree algorithm was 0.638, and GNB algorithm was 0.708. These ROC curves depict the sensitivity and specificity of various metabolic indicators and score models in influencing coronary disease, serving as key benchmarks for evaluating the effects of different algorithms based on these indicators and scores. Figure 2B extends this evaluation to the validation data, showing the ROC curves that reflect the generalization ability of the models in unseen data. The AUC for the logistic algorithm was 0.735, XGBoost algorithm was 0.766, RandomForest algorithm was 0.761, AdaBoost algorithm was 0.742, DecisionTree algorithm was 0.624, and GNB algorithm was 0.729. The AUC values provide a quantitative assessment of the accuracy of each model, which is crucial for validating their predictive ability in clinical and research settings. Figure 2C compares the AUC scores of different ML models in the validation data using a forest plot. Each point in the plot represents the median AUC value of a model, and the error bars represent the confidence intervals, emphasizing the performance differences between the models. In both the training and validation sets, the XGBoost algorithm achieved the highest AUC value, demonstrating the critical role played by various metabolic indicators and scores in influencing coronary disease. In Fig. 2D and E, we subsequently built the SHAP models using the XGBoost algorithm, showing that BMI had the greatest impact on coronary disease in the metabolic models. METS-IR had the largest impact among the five metabolic scores (METS-IR, TyG, HOMA-IR, TG/HDL-C, and VAI) on coronary disease.
Table 2.
Baseline characteristics of participants associated with coronary disease (CHD)
| Variable | CHD, N = 6791 | CON, N = 15,6081 | p-value2 |
|---|---|---|---|
| AGE, Median (IQR) | 70.00 (63.00–79.00) | 48.00 (33.00–63.00) | < 0.001 |
| PIR, Median (IQR) | 2.15 (1.20–3.92) | 2.15 (1.13–4.09) | 0.7 |
| TC, Median (IQR) | 166.00 (141.00–198.00) | 191.00 (165.00–218.00) | < 0.001 |
| A1 C, Median (IQR) | 5.90 (5.50–6.50) | 5.50 (5.20–5.80) | < 0.001 |
| TG, Median (IQR) | 115.00 (84.50–170.00) | 103.00 (72.00–150.00) | < 0.001 |
| LDL-C, Median (IQR) | 90.00 (69.50–115.00) | 112.00 (90.00–137.00) | < 0.001 |
| INSULIN, Median (IQR) | 11.19 (6.68–18.90) | 9.35 (5.89–15.41) | < 0.001 |
| HDL-C, Median (IQR) | 1.22 (1.03–1.50) | 1.34 (1.11–1.66) | < 0.001 |
| PFG, Median (IQR) | 111.00 (99.00–129.00) | 99.60 (92.00–109.00) | < 0.001 |
| BMI, Median (IQR) | 28.59 (25.22–32.61) | 27.82 (24.20–32.30) | 0.001 |
| WAIST, Median (IQR) | 104.10 (94.85–115.00) | 97.30 (87.30–108.20) | < 0.001 |
| VAI, Median (IQR) | 157.53 (93.82–240.34) | 125.38 (76.79–209.08) | < 0.001 |
| TyG, Median (IQR) | 8.81 (8.42–9.27) | 8.57 (8.16–9.00) | < 0.001 |
| TG/HDL-C, Median (IQR) | 96.49 (59.75–149.53) | 75.55 (46.20–124.43) | < 0.001 |
| METS-IR, Median (IQR) | 1.82 (1.71–1.93) | 1.70 (1.61–1.81) | < 0.001 |
| HOMA-IR, Median (IQR) | 3.09 (1.74–5.80) | 2.37 (1.42–4.14) | < 0.001 |
| Gender, n (%) | < 0.001 | ||
| Male | 463 (68) | 7,408 (47) | |
| Female | 216 (32) | 8,200 (53) | |
| Race, n (%) | < 0.001 | ||
| Mexican American | 62 (9.1) | 2,531 (16) | |
| Other Hispanic | 44 (6.7) | 1,360 (8.7) | |
| Non-Hispanic White | 454 (67) | 6,896 (44) | |
| Non-Hispanic Black | 74 (11) | 3,177 (20) | |
| Other Race—Including Multi-Racial | 45 (6.6) | 1,644 (11) |
1Median (IQR) or Frequency (%)
2Wilcoxon rank sum test; Pearson's Chi-squared test
Fig. 2.
Machine learning model evaluation of various metabolic indicators and scores in the coronary disease population. A AUC results graph of the training set. B AUC results graph of the testing set. C Forest plot of AUC scores. D Summary of the shap model of the XGBoost algorithm. E Importance ranking results of the shap model of the XGBoost algorithm
Evaluation of the influence of metabolic indicators and scores on myocardial infarction using binary classification machine learning algorithms and machine learning algorithm explanations
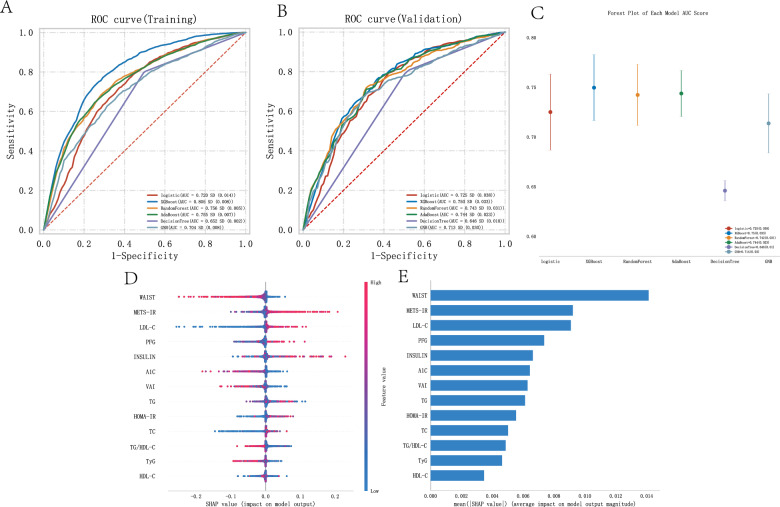
In a representative sample of 16,324 US adults, 48.37% were male (= 7,896) and 51.63% were female (= 8,428). Myocardial infarction was identified in 712 patients, accounting for 4.23% of the study population. The age of the myocardial infarction group ranged from 60 to 77 years, with an average PIR of 1.65. Among the patients, 8.8% were Mexican American, 7.2% were Other Hispanic, 60% were Non-Hispanic White, 17% were Non-Hispanic Black, and 7% were Other Race Including Multi Racial. In the blood of the myocardial infarction group, the average concentrations were as follows: TC (171 mg/dL), A1 C (5.95 mg/dL), TG (114 mg/dL), LDL-C (92 mg/dL), INSULIN (10.82 uU/mL), HDL-C (1.22 mg/dL), PFG (109 mg/dL), BMI (29.05 kg/m2), WAIST (104.45 cm), VAI (149.56), TyG (8.78), TG/HDL-C (92.33), METS-IR (1.80), and HOMA-IR (3.08) (Table 3).Here, the ROC curves of each model based on the training data, providing a comparative analysis of model performance and their AUC values (Fig. 3A). The AUC for the logistic algorithm was 0.734, XGBoost algorithm was 0.794, RandomForest algorithm was 0.729, AdaBoost algorithm was 0.739, DecisionTree algorithm was 0.642, and GNB algorithm was 0.692. These ROC curves depict the sensitivity and specificity of various metabolic indicators and score models in influencing myocardial infarction, serving as key benchmarks for evaluating the effects of different algorithms based on these indicators and scores. Figure 3B extends this evaluation to the validation data, showing the ROC curves that reflect the generalization ability of the models in unseen data. The AUC for the logistic algorithm was 0.748, XGBoost algorithm was 0.751, RandomForest algorithm was 0.746, AdaBoost algorithm was 0.745, DecisionTree algorithm was 0.669, and GNB algorithm was 0.708. The AUC values provide a quantitative assessment of the accuracy of each model, which is crucial for validating their predictive ability in clinical and research settings. Figure 3C compares the AUC scores of different ML models in the validation data using a forest plot. Each point in the plot represents the median AUC value of a model, and the error bars represent the confidence intervals, emphasizing the performance differences between the models. In both the training and validation sets, the XGBoost algorithm achieved the highest AUC value, demonstrating the critical role played by various metabolic indicators and scores in influencing myocardial infarction. In Fig. 3D and E, we subsequently built the SHAP models using the XGBoost algorithm, showing that waist circumference had the greatest impact on myocardial infarction in the metabolic models. METS-IR had the largest impact among the five metabolic scores (METS-IR, TyG, HOMA-IR, TG/HDL-C, and VAI) on myocardial infarction.
Table 3.
Baseline characteristics of participants associated with myocardial infarction (MI)
| Variable | MI, N = 7121 | CON, N = 15,6121 | p-value2 |
|---|---|---|---|
| AGE, Median (IQR) | 69.00 (60.00–77.00) | 48.00 (33.00–63.00) | < 0.001 |
| PIR, Median (IQR) | 1.65 (1.02–3.12) | 2.18 (1.14–4.11) | < 0.001 |
| TC, Median (IQR) | 171.00 (143.75–200.00) | 190.00 (165.00–218.00) | < 0.001 |
| A1 C, Median (IQR) | 5.90 (5.50–6.50) | 5.50 (5.20–5.80) | < 0.001 |
| TG, Median (IQR) | 114.00 (82.00–166.00) | 103.00 (72.00–150.25) | < 0.001 |
| LDL-C, Median (IQR) | 92.00 (72.00–118.00) | 112.00 (90.00–137.00) | < 0.001 |
| INSULIN, Median (IQR) | 10.82 (6.84–19.29) | 9.34 (5.88–15.40) | < 0.001 |
| HDL-C, Median (IQR) | 1.22 (1.03–1.53) | 1.34 (1.11–1.66) | < 0.001 |
| PFG, Median (IQR) | 109.00 (99.00–127.00) | 99.75 (92.00–109.00) | < 0.001 |
| BMI, Median (IQR) | 29.05 (25.19–32.97) | 27.81 (24.20–32.30) | < 0.001 |
| WAIST, Median (IQR) | 104.45 (94.58–115.33) | 97.40 (87.30–108.20) | < 0.001 |
| VAI, Median (IQR) | 149.56 (92.32–241.98) | 125.57 (76.87–209.43) | < 0.001 |
| TyG, Median (IQR) | 8.78 (8.40–9.26) | 8.57 (8.16–9.00) | < 0.001 |
| TG/HDL-C, Median (IQR) | 92.33 (58.79–150.12) | 75.73 (46.21–124.65) | < 0.001 |
| METS-IR, Median (IQR) | 1.80 (1.70–1.92) | 1.70 (1.61–1.81) | < 0.001 |
| HOMA-IR, Median (IQR) | 3.08 (1.81–5.70) | 2.37 (1.42–4.14) | < 0.001 |
| Gender, n (%) | < 0.001 | ||
| Male | 468 (66) | 7,428 (48) | |
| Female | 244 (34) | 8,184 (52) | |
| Race, n (%) | < 0.001 | ||
| Mexican American | 63 (8.8) | 2,540 (16) | |
| Other Hispanic | 51 (7.2) | 1,357 (8.7) | |
| Non-Hispanic White | 430 (60) | 6,937 (44) | |
| Non-Hispanic Black | 123 (17) | 3,130 (20) | |
| Other Race—Including Multi-Racial | 45 (7) | 1,648 (11) |
1Median (IQR) or Frequency (%)
2Wilcoxon rank sum test; Pearson's Chi-squared test
Fig. 3.
Machine learning model evaluation of various metabolic indicators and scores in the myocardial infarction population. A AUC results graph of the training set. B AUC results graph of the testing set. C Forest plot of AUC scores. D Summary of the shap model of the XGBoost algorithm. E Importance ranking results of the shap model of the XGBoost algorithm
Evaluation of the influence of metabolic indicators and scores on heart failure using binary classification machine learning algorithms and machine learning algorithm explanations
In a representative sample of 16,305 US adults, 48.33% were male (= 7,881) and 51.63% were female (= 8,424). Heart failure was identified in 500 patients, accounting for 3.07% of the study population. The age of the heart failure group ranged from 60 to 78 years, with an average PIR of 1.57. Among the patients, 8.4% were Mexican American, 7.2% were Other Hispanic, 58% were Non-Hispanic White, 22% were Non-Hispanic Black, and 4.4% were Other Race Including Multi Racial. In the blood of the heart failure group, the average concentrations were as follows: TC (170 mg/dL), A1 C (5.95 mg/dL), TG (115 mg/dL), LDL-C (93.5 mg/dL), INSULIN (11.4 uU/mL), HDL-C (1.22 mg/dL), PFG (109 mg/dL), BMI (30.1 kg/m2), WAIST (107.4 cm), VAI (163.10), TyG (8.79), TG/HDL-C (94.81), METS-IR (1.82), and HOMA-IR (3.11) (Table 4). Similarly, the ROC curves of each model based on the training data, providing a comparative analysis of model performance and their AUC values (Fig. 4A). The AUC for the logistic algorithm was 0.720, XGBoost algorithm was 0.805, RandomForest algorithm was 0.756, AdaBoost algorithm was 0.755, DecisionTree algorithm was 0.652, and GNB algorithm was 0.704. These ROC curves depict the sensitivity and specificity of various metabolic indicators and score models in influencing heart failure, serving as key benchmarks for evaluating the effects of different algorithms based on these indicators and scores. Figure 4B extends this evaluation to the validation data, showing the ROC curves that reflect the generalization ability of the models in unseen data. The AUC for the logistic algorithm was 0.725, XGBoost algorithm was 0.750, RandomForest algorithm was 0.743, AdaBoost algorithm was 0.744, DecisionTree algorithm was 0.646, and GNB algorithm was 0.713. The AUC values provide a quantitative assessment of the accuracy of each model, which is crucial for validating their predictive ability in clinical and research settings. Figure 4C compares the AUC scores of different ML models in the validation data using a forest plot. Each point in the plot represents the median AUC value of a model, and the error bars represent the confidence intervals, emphasizing the performance differences between the models. In both the training and validation sets, the XGBoost algorithm achieved the highest AUC value, demonstrating the critical role played by various metabolic indicators and scores in influencing heart failure. In Fig. 4D and E, we subsequently built the SHAP models using the XGBoost algorithm, showing that waist circumference had the greatest impact on heart failure in the metabolic models. METS-IR had the largest impact among the five metabolic scores (METS-IR, TyG, HOMA-IR, TG/HDL-C, and VAI) on heart failure.
Table 4.
Baseline characteristics of participants associated with heart failure (HF)
| Variable | HF, N = 5001 | CON, N = 15,8051 | p-value2 |
|---|---|---|---|
| AGE, Median (IQR) | 70.00 (60.00–78.00) | 48.00 (34.00–63.00) | < 0.001 |
| PIR, Median (IQR) | 1.57 (1.02–2.89) | 2.17 (1.14–4.12) | < 0.001 |
| TC, Median (IQR) | 170.00 (142.00–202.00) | 190.00 (164.00–218.00) | < 0.001 |
| A1 C, Median (IQR) | 5.95 (5.50–6.63) | 5.50 (5.20–5.80) | < 0.001 |
| TG, Median (IQR) | 115.00 (81.75–166.25) | 103.00 (72.00–150.00) | < 0.001 |
| LDL-C, Median (IQR) | 93.50 (70.75–121.00) | 112.00 (90.00–136.00) | < 0.001 |
| INSULIN, Median (IQR) | 11.40 (6.40–22.71) | 9.36 (5.89–15.41) | < 0.001 |
| HDL-C, Median (IQR) | 1.22 (1.01–1.50) | 1.34 (1.11–1.63) | < 0.001 |
| PFG, Median (IQR) | 109.00 (98.00–130.00) | 100.00 (92.30–110.00) | < 0.001 |
| BMI, Median (IQR) | 30.10 (25.88–35.46) | 27.80 (24.20–32.24) | < 0.001 |
| WAIST, Median (IQR) | 107.40 (94.93–119.50) | 97.40 (87.50–108.20) | < 0.001 |
| VAI, Median (IQR) | 163.10 (91.54–267.16) | 125.62 (76.96–208.98) | < 0.001 |
| TyG, Median (IQR) | 8.79 (8.37–9.28) | 8.57 (8.17–9.00) | < 0.001 |
| TG/HDL-C, Median (IQR) | 94.81 (57.77–156.31) | 75.86 (46.38–124.59) | < 0.001 |
| METS-IR, Median (IQR) | 1.82 (1.70–1.93) | 1.71 (1.61–1.81) | < 0.001 |
| HOMA-IR, Median (IQR) | 3.11 (1.70–6.97) | 2.37 (1.42–4.15) | < 0.001 |
| Gender, n (%) | < 0.001 | ||
| Male | 278 (56) | 7,603 (48) | |
| Female | 222 (44) | 8,202 (52) | |
| Race, n (%) | < 0.001 | ||
| Mexican American | 42 (8.4) | 2,555 (16) | |
| Other Hispanic | 36 (7.2) | 1,370 (8.7) | |
| Non-Hispanic White | 288 (58) | 7,069 (45) | |
| Non-Hispanic Black | 108 (22) | 3,145 (20) | |
| Other Race—Including Multi-Racial | 26 (4.4) | 1,666 (11) |
1Median (IQR) or Frequency (%)
2Wilcoxon rank sum test; Pearson's Chi-squared test
Fig. 4.
Machine learning model evaluation of various metabolic indicators and scores in the heart failure population. A AUC results graph of the training set. B AUC results graph of the testing set. C Forest plot of AUC scores. D Summary of the shap model of the XGBoost algorithm. E Importance ranking results of the shap model of the XGBoost algorithm
Evaluating the effects of metabolic indicators and Scores on AP, CHD, HF, MI using 100 machine learning algorithms
To further explore the predictive effectiveness of the metabolic indicators and scores we established for AP, CHD, HF, MI, we utilized 100 machine learning algorithms to calculate the AUC values of the metabolic indicators and scores in predicting AP, CHD, HF, MI. The results showed that among the 100 machine learning algorithms, the algorithm with the highest combined AUC values for training and testing sets in predicting AP, after excluding overfitting, was SVM + GBM (Fig. 5A), with a training set AUC value of 0.868 (Fig. 5C) and a testing set AUC value of 0.699 (Fig. 5D). The algorithm with the highest combined AUC values for training and testing sets in predicting CHD was Enet[alpha = 0.3] + GBM (Fig. 5B), with a training set AUC value of 0.852 (Fig. 5E) and a testing set AUC value of 0.751 (Fig. 5F). The algorithm with the highest combined AUC values for training and testing sets in predicting HF was Enet[alpha = 0.8] + GBM (Fig. 5K), with a training set AUC value of 0.830 (Fig. 5G) and a testing set AUC value of 0.776 (Fig. 5H). Finally, the algorithm with the highest combined AUC values for training and testing sets in predicting MI was GBM (Fig. 5L), with a training set AUC value of 0.833 (Fig. 5I) and a testing set AUC value of 0.778 (Fig. 5J). To identify the metabolic scores that have the greatest impact on AP, CHD, HF, and MI, we also performed feature selection using the algorithm with the highest combined AUC values for training and testing sets in predicting AP, CHD, HF, and MI. The results revealed that METS-IR had the highest contribution among the metabolic scores for AP, CHD, HF, and MI.
Fig. 5.
Evaluation of AUC values for AP, CHD, HF, MI prediction using 100 machine learning algorithms. A presents the AUC values for AP prediction using 100 machine learning algorithms. B shows the AUC values for CHD prediction using 100 machine learning algorithms. C displays the AUC value of SVM + GBM algorithm on the training set. D illustrates the AUC value of SVM + GBM algorithm on the testing set. E demonstrates the AUC value of Enet[alpha = 0.3] + GBM algorithm on the training set. F depicts the AUC value of Enet[alpha = 0.3] + GBM algorithm on the testing set. G exhibits the AUC value of Enet[alpha = 0.8] + GBM algorithm on the training set. H showcases the AUC value of Enet[alpha = 0.8] + GBM algorithm on the testing set. I shows the AUC value of GBM algorithm on the training set. J reveals the AUC value of GBM algorithm on the testing set. K represents the AUC value for HF prediction using 100 machine learning algorithms. L indicates the AUC value for MI prediction using 100 machine learning algorithms
Discussion
The results of this study showed that among the six machine learning algorithms used to evaluate the effects of metabolic indicators and scores on angina pectoris, coronary disease, myocardial infarction, and heart failure, the XGBoost algorithm had the highest scores in all four conditions and is the most suitable machine learning algorithm for assessing adverse cardiovascular events. We then established the shap model of the XGBoost algorithm to explain its performance. The results showed that waist circumference was the most important metabolic indicator in angina pectoris, myocardial infarction, and heart failure, while BMI was the most crucial metabolic indicator in coronary disease. In adverse cardiovascular events such as angina pectoris, coronary disease, myocardial infarction, and heart failure, METS-IR played the most critical role in the XGBoost algorithm, with its contribution and importance surpassing the four metabolic scores of TyG, HOMA-IR, TG/HDL-C, and VAI.Furthermore, we utilized 100 machine learning algorithms to calculate the AUC values of the metabolic indicators and scores in predicting AP, CHD, HF, and MI. Subsequently, SVM + GBM, Enet[alpha = 0.3] + GBM, Enet[alpha = 0.8] + GBM, and GBM were used to perform feature selection on the metabolic indicators and scores we established. The results demonstrated that among AP, CHD, HF, and MI, METS-IR was the metabolic score with the highest contribution.
Research has shown that increased BMI significantly increases the risk of coronary disease, and triglycerides play an important mediating role in the effect of BMI on coronary disease [25]. Waist circumference, on the other hand, is an important indicator of obesity and is significantly associated with the risk of angina pectoris or myocardial infarction [26]. In the population of heart failure patients, abdominal obesity determined by waist circumference can increase cardiac compliance and inflammatory response, leading to fluid retention and exacerbating the development of heart failure [27].
Similarly, the relationship between METS-IR and heart failure has been widely studied in the XGBoost algorithm. Studies have shown a significant correlation between long-term elevated METS-IR and the high risk of coronary disease in rural populations [28], and the level of METS-IR in patients with myocardial ischemia (including angina pectoris and myocardial infarction) is significantly higher than that in non-myocardial infarction patients [29].METS-IR as an index to assess the degree of insulin resistance. In endothelial cells, insulin resistance can reduce the tyrosine phosphorylation of IRS-1 and IRS-2, decrease the activity of the PI3 K-Akt signaling pathway, and subsequently affect the activity of nitric oxide synthase and the release of nitric oxide, ultimately leading to endothelial dysfunction, decreased vasodilation capacity, and the promotion of arterial plaque formation and atherosclerosis in coronary disease patients[30, 31]. In addition, activation of the PI3 K/Akt pathway can inhibit apoptosis of myocardial cells, but under the state of insulin resistance, the PI3 K/Akt pathway is inhibited, resulting in a decrease in anti-apoptotic ability and an increased risk of apoptosis in myocardial cells, leading to injury to the coronary disease heart cells and a reduction in myocardial cells [32, 33]; activation of the PI3 K/Akt pathway can also promote cell proliferation and migration, participating in the repair and regeneration process of myocardial tissue[34, 35]. However, under insulin resistance, the function of the PI3 K/Akt pathway is inhibited, which may reduce the proliferation and migration ability of myocardial cells and hinder the repair of necrotic myocardial tissue in coronary disease patients [36, 37]. Furthermore, when insulin signaling is blocked, it can trigger intracellular inflammatory and stress responses, leading to the degradation of IκB (inhibitor) and activation of NF-κB. Activated NF-κB enters the cell nucleus, promoting the transcription of inflammatory factors, cell adhesion molecules, and apoptotic regulatory factors. In endothelial cells, the activation of the NF-κB signaling pathway mediates an increase in the expression of leukocyte adhesion molecules, enhancing the inflammatory response. The enhanced inflammatory response further induces adhesion molecule expression in endothelial cells, leading to leukocyte adhesion and infiltration of inflammatory cells. These inflammatory responses and cell infiltration promote the development of atherosclerosis in patients with myocardial ischemia [38, 39]. Additionally, in a state of insulin resistance, the balance of cell cycle regulatory factors such as cyclin-dependent kinases (CDK) and cell cycle inhibitors (CKI) may be disturbed, leading to abnormal vascular smooth muscle cell cycle and promoting abnormal cell proliferation. At the same time, interference with insulin signaling may induce the production and release of various growth factors such as platelet-derived growth factor (PDGF) and epidermal growth factor (EGF). These cell cycle regulatory factors and growth factors can stimulate the proliferation and migration of vascular smooth muscle cells, promoting the development of atherosclerosis in patients with myocardial ischemia [40, 41].
Similarly, studies have shown a positive correlation between METS-IR and heart failure, with a saturated turning point effect at 40.966, suggesting the potential of METS-IR as a key indicator for predicting heart failure [42]. METS-IR as an index to assess the degree of insulin resistance.In patients with heart failure, insulin resistance leads to a decrease in fatty acid uptake and oxidative capacity, while the uptake capacity of TG increases, resulting in the accumulation of triglycerides (TG) and fatty acids in cardiac cells. At the same time, insulin resistance also disrupts mitochondrial function, inhibits ATP production, and generates excess reactive oxygen species (ROS), damaging mitochondrial DNA and further affecting mitochondrial function [43]. Studies have also shown that oxidative stress and reactive oxygen species (ROS) produced in heart failure exacerbate the development of insulin resistance. The activation of the renin–angiotensin–aldosterone system (RAAS) and sympathetic nervous system (SNS) increases oxidative stress and ROS production, including superoxide ions (O2^-) and hydrogen peroxide (H2O2). ROS can inhibit the function of IRS-1 (insulin receptor substrate-1), leading to the blocking of insulin signaling pathways and exacerbating insulin resistance [44]. Simultaneously, in the insulin-resistant state of heart failure patients, the production of inflammatory factors such as tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) increases. These inflammatory factors can activate inflammatory signaling pathways, such as nuclear factor kappa B (NF-κB) and mitogen-activated protein kinases (MAPKs) [45]. The activity of the NF-κB pathway increases during insulin resistance, leading to increased expression of inflammatory factors, activation of apoptotic pathways, increased apoptosis of myocardial cells, and damage to myocardial function. Insulin resistance also leads to enhanced activity of the MAPK pathway and its branches ERK, JNK, and p38, triggering an inflammatory response, increasing cardiac fibrosis, causing changes in myocardial structure, and reducing myocardial contractile function, exacerbating the development of heart failure [46].
METS-IR, as an index for assessing the degree of insulin resistance, plays a role in heart failure (HF) through complex molecular-level regulation mechanisms. Insulin resistance may influence cardiac cell metabolism and growth by modulating signaling pathways such as AMPK (AMP-activated protein kinase) and mTOR (mammalian target of rapamycin), thereby affecting the contraction and relaxation functions of the heart during the heart failure process [47]. In a state of insulin resistance, the AMPK signaling pathway may be overactivated or inhibited, leading to energy metabolism disorders, which can result in abnormal nucleic acid synthesis and metabolism [48]. Simultaneously, mTOR is activated through two distinct signaling pathways: one is the insulin signaling pathway (PI3 K/Akt/mTOR), and the other is the growth factor signaling pathway (Ras/Raf/MEK/ERK). Activated mTORC1 (mTOR complex 1) promotes protein synthesis, cell cycle progression, and cell proliferation. In a state of insulin resistance, the mTOR signaling pathway may be overregulated or inhibited, leading to abnormal protein synthesis and restricted cell proliferation, ultimately contributing to the development of heart failure [49].Insulin resistance affects myocardial hypertrophy and fibrosis in heart failure patients through the regulation of multiple cellular pathways. In a state of insulin resistance, the activity of the adenosine monophosphate-activated protein kinase (AMPK) signaling pathway decreases, leading to a reduction in the oxidation of fatty acids in myocardial cells and enhancing dependence on glucose uptake, ultimately causing myocardial hypertrophy. In addition, insulin resistance can promote myocardial cell growth by enhancing the activity of extracellular signal-regulated kinases (ERK) family members, exacerbating myocardial hypertrophy in heart failure patients [50, 51].
When analyzing the relationship between METS-IR and coronary heart disease and heart failure, it is necessary to consider possible metabolic abnormalities overlap and comorbidity characteristics. For example, other metabolic diseases such as obesity and diabetes may interact with insulin resistance and cardiovascular diseases, affecting the interpretation and inference of research results. In addition, the lifestyle and genetic factors of individual patients may also have an impact on the results of the study, which needs further in-depth discussion.
In this study, the biomarker model performed excellently, especially in assessing cardiovascular events such as angina, coronary heart disease, myocardial infarction, and heart failure. However, we still need to explore whether this biomarker model can serve as a universal model for the highly diverse metabolic conditions in the general population, including aspects such as stability and adaptability of the model, clinical application and feasibility, as well as potential biases and limitations.
The stability and accuracy of the biomarker model are prerequisites for it to become a universal model. We need to consider the adaptability of this model to different populations and metabolic states. For example, metabolic markers such as BMI and waist circumference may exhibit different correlations in different ethnicities, age groups, and genders. Therefore, the model's ability to personalize adjustments for different metabolic types will directly impact its effectiveness in the general population. In clinical practice, the implementation of the biomarker model needs to consider its practicality and acceptability. Whether medical teams can effectively utilize this model for patient risk assessment and intervention decisions will largely depend on the training level of clinical personnel and the cooperation level of patients. Additionally, the implementation of the model may need to be combined with existing treatment guidelines and healthcare policies to enhance its utility. Despite the outstanding performance of this biomarker model in the current research, we must also be cautious of potential biases and limitations. For example, the model may be influenced by environmental factors, lifestyle, coexisting diseases, etc., which could lead to reduced predictive ability in specific populations. Therefore, in future research, it is crucial to systematically validate the applicability of the model in different backgrounds and clinical conditions to enhance its scientific rigor and clinical significance.
Conclusion
We conducted an in-depth analysis utilizing six machine learning algorithms, with the XGBoost algorithm emerging as the optimal binary classification machine learning algorithm in our study. This algorithm was specifically employed to evaluate the impact of metabolic indicators and scores on adverse cardiovascular events, including angina pectoris, coronary disease, myocardial infarction, and heart failure.
Through the SHAP model explanation of the XGBoost algorithm, we were able to identify waist circumference as the most crucial metabolic indicator influencing angina pectoris, myocardial infarction, and heart failure, while BMI played a pivotal role in coronary disease. Furthermore, in our evaluation of adverse cardiovascular events, including angina pectoris, coronary disease, myocardial infarction, and heart failure, METS-IR emerged as the most critical metabolic score. Remarkably, the contribution and significance of METS-IR surpassed that of other important metabolic scores such as TyG, HOMA-IR, TG/HDL-C, and VAI.
In an effort to comprehensively assess the predictive performance of the metabolic indicators and scores, we employed 100 machine learning algorithms to calculate the AUC values for predicting angina pectoris, coronary heart disease, heart failure, and myocardial infarction. Subsequently, we employed SVM + GBM, Enet[alpha = 0.3] + GBM, Enet[alpha = 0.8] + GBM, and GBM for feature selection analysis on the established metabolic indicators and scores. The results of this analysis unequivocally identified METS-IR as the metabolic score with the highest contribution among angina pectoris, coronary heart disease, heart failure, and myocardial infarction.
This comprehensive evaluation underscores the pivotal role of METS-IR as a key metabolic score in predicting adverse cardiovascular events, further highlighting its importance in the context of cardiovascular risk assessment. These findings provide valuable insights for understanding the intricate relationship between metabolic indicators and cardiovascular outcomes, contributing to the advancement of personalized medicine in cardiovascular care.
Limitation
In this study, we have made some important findings, but there are still some limitations and future prospects that need further exploration.
First, sample selection may affect the reliability of the results. Although this study used data from the National Health and Nutrition Examination Survey (NHANES), the samples were based solely on the American population and may not fully represent patients from other countries or regions. Therefore, future research should consider validation in different populations to enhance the generalizability of the results.
Second, although we employed multiple machine learning algorithms and validated the model performance through cross-validation, the complexity of the model may lead to overfitting issues. Future research should focus on model validation to improve the effectiveness for practical clinical application.
Additionally, our study mainly focused on the association between metabolic indicators and cardiovascular events without fully considering other possible influencing factors such as lifestyle, genetic factors, and environmental factors. These factors may play a crucial role in the development of cardiovascular diseases, so future studies should incorporate more variables in the study design to comprehensively assess their impact on cardiovascular events.
Lastly, while this study emphasizes the importance of metabolic scores like METS-IR in predicting adverse cardiovascular events, future efforts should promote the use of these scores in clinical practice to enable early identification of high-risk patients and develop targeted preventive measures.
Supplementary Information
Acknowledgements
Not applicable
Author contributions
R. Zou, G. Li,T. Ge,C.Luo and B.Zheng take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretations; they also drafted the article. M.Zhang, K. He and J. Hu take responsibility for the statistical analyses and interpretation of the data. X. Fan take responsibility for the full text evaluation and guidance and performed the final approval of the version to be submitted. All authors read and approved the final manuscript.
Funding
National Natural Science Foundation of China (No. 82300315; No. 82374240), Guangdong Province Basic and Applied Basic Research Fund Project (No. 2024 A1515012174; No. 2024 A1515013184; No. 2025A1515012690). National Administration of Traditional Chinese Medicine Research Project (No. 0102023703), Project of the State Key Laboratory of Dampness Syndrome of Traditional Chinese Medicine jointly established by the province and the ministry (No. SZ2022 KF10), Scientific Research Initiation Project of Guangdong Provincial Hospital of Traditional Chinese Medicine (No. 2021 KT1709), Research Project of Guangdong Provincial Bureau of Traditional Chinese Medicine (No. 20241120), Guangdong Provincial Key Laboratory of Research on Emergency in TCM (No. 2023B1212060062; 2023 KT15450). Scientific Research Cultivation Project of Chinese Medicine Guangdong Laboratory (No. HQL2024PZ036), Excellent Young Talents Program of Guangdong Provincial Hospital of Traditional Chinese Medicine (No. SZ2024QN05) and Basic Clinical Collaborative Innovation Program of Guangdong Provincial Hospital of Traditional Chinese Medicine and School of Biomedical Sciences, Guangdong Provincial Hospital of Traditional Chinese Medicine and School of Biomedicine, Chinese University of Hong Kong Basic Clinical Collaborative Innovation Program (No. YN2024HK01), Guangxi Natural Science Foundation (2023GXNSFAA026128), Noncommunicable Chronic Diseases-National Science and Technology Major Project (No. 2024ZD0528206; No. 2024ZD0528200).
Data availability
Data is provided within the manuscript.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guanmou Li, Cheng Luo, Teng Ge and Kunyang He have contributed equally to this work.
Contributor Information
Baoshi Zheng, Email: baoshizhengyx@163.com.
Rongjun Zou, Email: zourj3@mail2.sysu.edu.cn.
Xiaoping Fan, Email: fukui-hanson@hotmail.com.
References
- 1.Sutanto A, Wungu CDK, Susilo H, Sutanto H. Reduction of major adverse cardiovascular events (MACE) after bariatric surgery in patients with obesity and cardiovascular diseases: a systematic review and meta-analysis. Nutrients. 2021. 10.3390/nu13103568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kloner RA, Chaitman B. Angina and its management. J Cardiovasc Pharmacol Ther. 2017;22(3):199–209. [DOI] [PubMed] [Google Scholar]
- 3.Katta N, Loethen T, Lavie CJ, Alpert MA. Obesity and coronary heart disease: epidemiology, pathology, and coronary artery imaging. Curr Probl Cardiol. 2021;46(3): 100655. [DOI] [PubMed] [Google Scholar]
- 4.Salari N, Morddarvanjoghi F, Abdolmaleki A, Rasoulpoor S, Khaleghi AA, Hezarkhani LA, Shohaimi S, Mohammadi M. The global prevalence of myocardial infarction: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2023;23(1):206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gulati R, Behfar A, Narula J, Kanwar A, Lerman A, Cooper L, Singh M. Acute myocardial infarction in young individuals. Mayo Clin Proc. 2020;95(1):136–56. [DOI] [PubMed] [Google Scholar]
- 6.Savarese G, Becher PM, Lund LH, Seferovic P, Rosano GMC, Coats AJS. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc Res. 2023;118(17):3272–87. [DOI] [PubMed] [Google Scholar]
- 7.Wang S, Shi J, Peng Y, Fang Q, Mu Q, Gu W, Hong J, Zhang Y, Wang W. Stronger association of triglyceride glucose index than the HOMA-IR with arterial stiffness in patients with type 2 diabetes: a real-world single-centre study. Cardiovasc Diabetol. 2021;20(1):82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wei J, Liu X, Xue H, Wang Y, Shi Z. Comparisons of visceral adiposity index, body shape index, body mass index and waist circumference and their associations with diabetes mellitus in adults. Nutrients. 2019. 10.3390/nu11071580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang Z, Tan L. Association of insulin resistance-related indicators with cardiovascular disease in Chinese people with different glycemic states. Front Endocrinol. 2025;16:1515559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cheng S, Han Y, Jiang L, Lan Z, Liao H, Guo J. Associations of oxidative balance score and visceral adiposity index with risk of ischaemic heart disease: a cross-sectional study of NHANES, 2005–2018. BMJ Open. 2023;13(7): e072334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bello-Chavolla OY, Almeda-Valdes P, Gomez-Velasco D, Viveros-Ruiz T, Cruz-Bautista I, Romo-Romo A, Sánchez-Lázaro D, Meza-Oviedo D, Vargas-Vázquez A, Campos OA, et al. METS-IR, a novel score to evaluate insulin sensitivity, is predictive of visceral adiposity and incident type 2 diabetes. Eur J Endocrinol. 2018;178(5):533–44. [DOI] [PubMed] [Google Scholar]
- 12.Yang T, Liu Y, Li L, Zheng Y, Wang Y, Su J, Yang R, Luo M, Yu C. Correlation between the triglyceride-to-high-density lipoprotein cholesterol ratio and other unconventional lipid parameters with the risk of prediabetes and Type 2 diabetes in patients with coronary heart disease: a RCSCD-TCM study in China. Cardiovasc Diabetol. 2022;21(1):93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hou XZ, Lv YF, Li YS, Wu Q, Lv QY, Yang YT, Li LL, Ye XJ, Yang CY, Wang MS, et al. Association between different insulin resistance surrogates and all-cause mortality in patients with coronary heart disease and hypertension: NHANES longitudinal cohort study. Cardiovasc Diabetol. 2024;23(1):86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sergi D, Spaggiari R, Dalla Nora E, Angelini S, Castaldo F, Omenetto A, Stifani G, Sanz JM, Passaro A. HOMA-IR and TyG index differ for their relationship with dietary, anthropometric, inflammatory factors and capacity to predict cardiovascular risk. Diabetes Res Clin Pract. 2025;222: 112103. [DOI] [PubMed] [Google Scholar]
- 15.Zheng D, Cao L. Association between myocardial infarction and triglyceride-glucose index: a study based on NHANES database. Glob Heart. 2024;19(1):23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xiao J, Li Y, Gao X, Yan B, Chen F, Zhang C, Wang C, Han T, Zhang Y. Association between triglyceride-glucose index and all cause mortality in critically ill patients with heart failure. Sci Rep. 2025;15(1):16157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhou Z, Liu Q, Zheng M, Zuo Z, Zhang G, Shi R, Wu T. Comparative study on the predictive value of TG/HDL-C, TyG and TyG-BMI indices for 5-year mortality in critically ill patients with chronic heart failure: a retrospective study. Cardiovasc Diabetol. 2024;23(1):213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Poochanasri M, Lertsakulbunlue S, Kookanok C, Rangsin R, Kaewput W, Mungthin M, Samakkarnthai P. Triglyceride to high-density lipoprotein ratio as a predictor for 10-year cardiovascular disease in individuals with diabetes in Thailand. J Health Popul Nutr. 2025;44(1):147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Song X, Liu X, Liu F, Wang C. Comparison of machine learning and logistic regression models in predicting acute kidney injury: a systematic review and meta-analysis. Int J Med Inform. 2021;151: 104484. [DOI] [PubMed] [Google Scholar]
- 20.Tang J, Henderson A, Gardner P. Exploring AdaBoost and Random Forests machine learning approaches for infrared pathology on unbalanced data sets. Analyst. 2021;146(19):5880–91. [DOI] [PubMed] [Google Scholar]
- 21.Elhazmi A, Al-Omari A, Sallam H, Mufti HN, Rabie AA, Alshahrani M, Mady A, Alghamdi A, Altalaq A, Azzam MH, et al. Machine learning decision tree algorithm role for predicting mortality in critically ill adult COVID-19 patients admitted to the ICU. J Infect Public Health. 2022;15(7):826–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hou N, Li M, He L, Xie B, Wang L, Zhang R, Yu Y, Sun X, Pan Z, Wang K. Predicting 30-days mortality for MIMIC-III patients with sepsis-3: a machine learning approach using XGboost. J Transl Med. 2020;18(1):462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hu J, Szymczak S. A review on longitudinal data analysis with random forest. Briefings Bioinform. 2023. 10.1093/bib/bbad002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chambers C, Seethapathi N, Saluja R, Loeb H, Pierce SR, Bogen DK, Prosser L, Johnson MJ, Kording KP. Computer vision to automatically assess infant neuromotor risk. IEEE Trans Neural Syst Rehabil Eng Publ IEEE Eng Med Biol Soc. 2020;28(11):2431–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xu L, Borges MC, Hemani G, Lawlor DA. The role of glycaemic and lipid risk factors in mediating the effect of BMI on coronary heart disease: a two-step, two-sample Mendelian randomisation study. Diabetologia. 2017;60(11):2210–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chi JH, Lee BJ. Association of myocardial infarction and angina pectoris with obesity and biochemical indices in the South Korean population. Sci Rep. 2022;12(1):13769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Powell-Wiley TM, Poirier P, Burke LE, Després JP, Gordon-Larsen P, Lavie CJ, Lear SA, Ndumele CE, Neeland IJ, Sanders P, et al. Obesity and cardiovascular disease: a scientific statement from the American heart association. Circulation. 2021;143(21):e984–1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang S, Zhang X, Keerman M, Guo H, He J, Maimaitijiang R, Wang X, Ma R, Guo S. Impact of the baseline insulin resistance surrogates and their longitudinal trajectories on cardiovascular disease (coronary heart disease and stroke): a prospective cohort study in rural China. Front Endocrinol. 2023;14:1259062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Guo D, Zhang C, Zhang M, Wu Z, Liu X, Zhang Y, Liu L, Sun M, Yang J. Metabolic score for insulin resistance predicts major adverse cardiovascular event in premature coronary artery disease. Aging. 2024;16(7):6364–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhao P, Huang Y, Xiao L, Cui J, Li D, Cao Y, He J, Xu Y, Guo J, Xue H, Chen Y, Li T. Diagnosis of coronary artery disease by acoustic analysis of turbulent murmur caused by coronary artery stenosis: a single center study from China. Cardiovasc Innov Appl. 2023. 10.15212/CVIA.2022.0023. [Google Scholar]
- 31.Liu Y, Sun S, Yu B, Wang Q, Liu H, Shao X, Zhao H. Changes in serum levels of Chitinase protein-40 in children with Kawasaki disease and its clinical significance. BMC Pediatr. 2025;25(1):392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang J, Zhang F, Gao M, Wang Y, Song X, Li Y, Dang Y, Qi X. The systemic immune inflammatory index predicts no-reflow phenomenon after primary percutaneous coronary intervention in older patients with STEMI. Cardiovasc Innov Appl. 2023;7(1):984. [Google Scholar]
- 33.Gao A, Peng B, Gao Y, Yang Z, Li Z, Guo T, Qiu H, Gao R. Evaluation and comparison of inflammatory and insulin resistance indicators on recurrent cardiovascular events in patients undergoing percutaneous coronary intervention: a single center retrospective observational study. Diabetol Metab Syndr. 2025;17(1):157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Heck PM, Dutka DP. Insulin resistance and heart failure. Curr Heart Fail Rep. 2009;6(2):89–94. [DOI] [PubMed] [Google Scholar]
- 35.Han X, Liu X, Zhao X, Wang X, Sun Y, Qu C, Liang J, Yang B. Dapagliflozin ameliorates sepsis-induced heart injury by inhibiting cardiomyocyte apoptosis and electrical remodeling through the PI3K/Akt pathway. Eur J Pharmacol. 2023;955: 175930. [DOI] [PubMed] [Google Scholar]
- 36.Ponnusamy M, Li PF, Wang K. Understanding cardiomyocyte proliferation: an insight into cell cycle activity. Cell Mol Life Sci CMLS. 2017;74(6):1019–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li Y, Xia J, Jiang N, Xian Y, Ju H, Wei Y, Zhang X. Corin protects H(2)O(2)-induced apoptosis through PI3K/AKT and NF-κB pathway in cardiomyocytes. Biomed Pharmacother Biomed Pharmacother. 2018;97:594–9. [DOI] [PubMed] [Google Scholar]
- 38.Reaven G. Insulin resistance and coronary heart disease in nondiabetic individuals. Arterioscler Thromb Vasc Biol. 2012;32(8):1754–9. [DOI] [PubMed] [Google Scholar]
- 39.Wang R, Ma Y, Zhan S, Zhang G, Cao L, Zhang X, Shi T, Chen W. B7–H3 promotes colorectal cancer angiogenesis through activating the NF-κB pathway to induce VEGFA expression. Cell Death Dis. 2020;11(1):55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Choi SS, Kim ES, Koh M, Lee SJ, Lim D, Yang YR, Jang HJ, Seo KA, Min SH, Lee IH, et al. A novel non-agonist peroxisome proliferator-activated receptor γ (PPARγ) ligand UHC1 blocks PPARγ phosphorylation by cyclin-dependent kinase 5 (CDK5) and improves insulin sensitivity. J Biol Chem. 2014;289(38):26618–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liu C, Meng M, Xu B, Xu Y, Li G, Cao Y, Wang D, Qiu J, Yu J, Xu L, et al. Fibroblast growth factor 6 promotes adipocyte progenitor cell proliferation for adipose tissue homeostasis. Diabetes. 2023;72(4):467–82. [DOI] [PubMed] [Google Scholar]
- 42.Su X, Zhao C, Zhang X. Association between METS-IR and heart failure: a cross-sectional study. Front Endocrinol. 2024;15:1416462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Saotome M, Ikoma T, Hasan P, Maekawa Y. Cardiac insulin resistance in heart failure: the role of mitochondrial dynamics. Int J Mol Sci. 2019. 10.3390/ijms20143552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Aroor AR, Mandavia CH, Sowers JR. Insulin resistance and heart failure: molecular mechanisms. Heart Fail Clin. 2012;8(4):609–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Erqou S, Adler AI, Challa AA, Fonarow GC, Echouffo-Tcheugui JB. Insulin resistance and incident heart failure: a meta-analysis. Eur J Heart Fail. 2022;24(6):1139–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yang CD, Pan WQ, Feng S, Quan JW, Chen JW, Shu XY, Aihemaiti M, Ding FH, Shen WF, Lu L, et al. Insulin resistance is associated with heart failure with recovered ejection fraction in patients without diabetes. J Am Heart Assoc. 2022;11(19): e026184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ni W, Jiang R, Xu D, Zhu J, Chen J, Lin Y, Zhou H. Association between insulin resistance indices and outcomes in patients with heart failure with preserved ejection fraction. Cardiovasc Diabetol. 2025;24(1):32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Song G, Liu N, He J, Tang S, Yu Y, Song L. Exploring the role of Myo-inositol in alleviating insulin resistance in polycystic ovary syndrome through the AMPK/GLUT4 pathway. Mol Biol Rep. 2025;52(1):454. [DOI] [PubMed] [Google Scholar]
- 49.Zhu Z, Bai Z, Cui Y, Li X, Zhu X. The potential therapeutic effects of Panax notoginseng in osteoporosis: a comprehensive review. Phytomedicine Int J Phytotherapy Phytopharmacol. 2025;142: 156703. [DOI] [PubMed] [Google Scholar]
- 50.Riehle C, Abel ED. Insulin signaling and heart failure. Circ Res. 2016;118(7):1151–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sato A, Yumita Y, Kagami K, Ishinoda Y, Kimura T, Osaki A, Toya T, Namba T, Endo S, Ido Y, et al. Endothelial extracellular signal-regulated kinase/thromboxane A2/prostanoid receptor pathway aggravates endothelial dysfunction and insulin resistance in a mouse model of metabolic syndrome. J Am Heart Assoc. 2022;11(23): e027538. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data is provided within the manuscript.