Abstract
Vascular dementia is a highly heterogeneous neurodegenerative disorder induced by a variety of factors. Currently, there are no definitive treatments for the cognitive dysfunction associated with vascular dementia. However, early detection and preventive measures have proven effective in reducing the risk of onset and improving patient prognosis. Nitric oxide plays an integral role in various physiological and pathological processes within the central nervous system. In recent years, nitric oxide has been implicated in the regulation of synaptic plasticity and has emerged as a crucial factor in the pathophysiology of vascular dementia. At different stages of vascular dementia, nitric oxide levels and bioavailability undergo dynamic alterations, with a marked reduction in the later stages, which significantly contributes to the cognitive deficits associated with the disease. This review provides a comprehensive review of the emerging role of nitric oxide in the physiological and pathological processes underlying vascular dementia, focusing on its effects on synaptic dysfunction, neuroinflammation, oxidative stress, and blood‒brain barrier integrity. Furthermore, we suggest that targeting the nitric oxide soluble guanylate cyclase-cyclic guanosine monophosphate pathway through specific therapeutic strategies may offer a novel approach for treating vascular dementia, potentially improving both cognitive function and patient prognosis. The review contributes to a better understanding of the multifaceted role of nitric oxide in vascular dementia and to offering insights into future therapeutic interventions.
Keywords: apoptosis, blood‒brain barrier, cell death, dementia, mitochondrial dysfunction, neuroprotection, nitric oxide synthase, nitric oxide, oxidative stress, vascular dementia
Introduction
According to statistics from the World Health Organization for 2024, nearly 10 million individuals are diagnosed with dementia annually, with approximately 60% of these cases occurring in low- and middle-income countries.1 The estimated global economic burden of dementia is approximately $818 billion per year, representing over 1% of the world’s gross domestic product.2 This total cost encompasses direct medical expenses, social care costs, and informal care expenses, which include the loss of income for caregivers.
Dementia is an umbrella term that encompasses various diseases. In addition to Alzheimer’s disease (AD), vascular dementia (VD) accounts for approximately 12% to 20% of dementia cases.3 In recent years, AD has garnered significant attention within the realm of international cerebrovascular disease research due to its complex pathogenesis and the ambiguity surrounding its pathogenic factors. Nevertheless, VD continues to be one of the leading causes of adult disability worldwide. The increasing prevalence of cerebrovascular diseases has led to a corresponding increase in the incidence of VD, which is closely linked to various cerebrovascular risk factors. Despite this escalating burden, therapeutic options that can significantly enhance cognitive function and improve long-term outcomes for patients with VD remain limited. This situation imposes considerable strain on medical and healthcare expenditures.
The pathophysiology and etiology of VD are closely linked to stroke.4 The incidence of VD within 1 year post-stroke is approximately 33%, and the cumulative incidence over 5 years remains significant at approximately 31%.5 However, the threshold of brain damage necessary to precipitate cognitive and behavioral impairments remains uncertain. This highlights the need for further research to elucidate the mechanisms underlying VD and its relationship with cerebrovascular events.
VD is a type of dementia primarily caused by cerebrovascular disease or impaired cerebral blood flow, and it is recognized as the most prevalent form of dementia following AD.6 Vascular cognitive impairment encompasses a spectrum that ranges from mild cognitive impairment to dementia, often coexisting with neurodegenerative disorders such as AD. The interplay between vascular cognitive impairment and AD can exacerbate cognitive decline, resulting in cumulative effects on cognitive aging.7
VD can be classified into four main types based on its pathological underpinnings: post-stroke dementia, subcortical ischemic VD, multiple infarction (cortical) dementia, and mixed dementia.8 Several risk factors have been identified that increase the likelihood of developing VD, including advanced age, smoking, a family history of dementia, a history of recurrent strokes, and hypertension.9
The prevention and management of VD are closely linked to the effective management of stroke.10 Moreover, the deterioration of VD is associated with decreased bioavailability of nitric oxide (NO) and endothelial dysfunction, both of which contribute to disease progression.11,12 Over time, patients with VD typically experience a gradual decline in their condition, which can lead to a significant reduction in quality of life or even death due to complications.
Given these considerations, in-depth research into the role of medical gases, particularly NO, in the prevention and treatment of VD may yield novel therapeutic strategies. As a medical gas, NO has garnered considerable interest from both clinical and experimental researchers due to its physiological roles across multiple systems.
This article aims to summarize the current research trends surrounding NO and NO synthase (NOS), beginning with an exploration of the sources, biosynthetic pathways, and functional alterations of NO in pathological contexts. Recent advancements in understanding the role of NO in the pathogenesis of VD, including its contributions to maintaining endothelial homeostasis, regulating neuroinflammation, participating in oxygen metabolism, and modulating synaptic plasticity, are emphasized.
Search Strategy
A computer-based online search was performed on the PubMed database to retrieve articles published up to July 31, 2024. To maximize the specificity and sensitivity of the search, a combination of the following terms was used: vascular dementia, nitric oxide, neuroinflammation, oxidative stress, nitric oxide synthase, medical gas research, synaptic dysfunction, Alzheimer’s disease, blood‒brain barrier, and vascular cognitive impairment. Further screening was conducted through titles and abstracts, and only studies exploring the relationship between nitric oxide and the pathogenesis and prognosis of VD were included to investigate the effect of NO on VD treatment. There are no language or research type restrictions. Studies on the relationships between partial NO and stroke treatment and patient prognosis have also been included in the exploration. The focus is on articles published within the past 10 years.
Advancements in Nitric Oxide
At the beginning of the 20th century, NO was predominantly viewed as an environmental pollutant and a chemical toxin. However, advancements in scientific research have fundamentally transformed our understanding of the role of NO in human physiology. In the 1970s, Katsuki et al.13 made a groundbreaking discovery that nitroglycerin, upon entering the bloodstream, is converted into NO, a transient gas that induces vasodilation, thereby increasing blood flow and increasing the cardiac supply. In the 1980s, Furchgott and Zawadzki14 identified a class of molecules known as endothelium-derived relaxation factors, which are responsible for inducing vasodilation in endothelial cells. These factors exist for extremely short durations, typically less than one second. These molecules were originally termed endothelium-derived relaxation factors. In 1986, Ignarro et al.15 established that these vasodilators were, in fact, NO. They further demonstrated that endothelial tissues are capable of synthesizing NO to regulate blood pressure.
NO is a unique molecule that can dissolve in both water and lipids, allowing it to disperse freely across cell membranes.16 It is characterized by high reactivity and a short half-life, making it prone to deactivation in the presence of oxygen and superoxide ions. However, it remains relatively stable under conditions facilitated by superoxide dismutase and in acidic environments.17 In 1992, NO was recognized as the “Molecule of the Year,” highlighting its previously underestimated significance in human health.17,18,19,20 The pivotal nature of NO in cardiovascular health was further validated in 1998 when three prominent scientists were awarded the Nobel Prize in Physiology or Medicine for their discovery of NO as a key signaling molecule in the cardiovascular system, elucidating its role in maintaining vascular health and regulating blood pressure. This revelation not only revolutionized our understanding of NO but also revealed its diverse mechanisms in promoting health.
NO, also referred to as nitrogen oxide, is synthesized primarily in the body through both enzymatic and nonenzymatic pathways,21 with the enzymatic pathways being particularly pronounced in vascular endothelial cells and neurons.22 In a significant study conducted by Davidson et al.23 in 1998, the efficacy of inhaled NO in treating persistent pulmonary arterial hypertension in full-term neonates was confirmed. In 1999, inhalation therapy with NO was subsequently incorporated into international treatment guidelines for acute hypoxic respiratory failure in newborns. Further research in 2006 by Puikuan et al.24 demonstrated that nebulized nitroglycerin inhalation therapy could alleviate pulmonary arterial hypertension resulting from elevated pulmonary blood flow, thereby reinforcing the therapeutic potential of NO in managing pulmonary arterial hypertension. More recently, in 2017, researchers from the School of Basic Sciences at Shenzhen University and the University of Massachusetts School of Medicine elucidated the mechanism by which NO inhibits neutrophil inflammation in the lungs, thereby suppressing tuberculosis replication.25 This finding underscores the critical role of NO produced by inducible NOS (iNOS) in host defense mechanisms (Figure 1).
Figure 1.
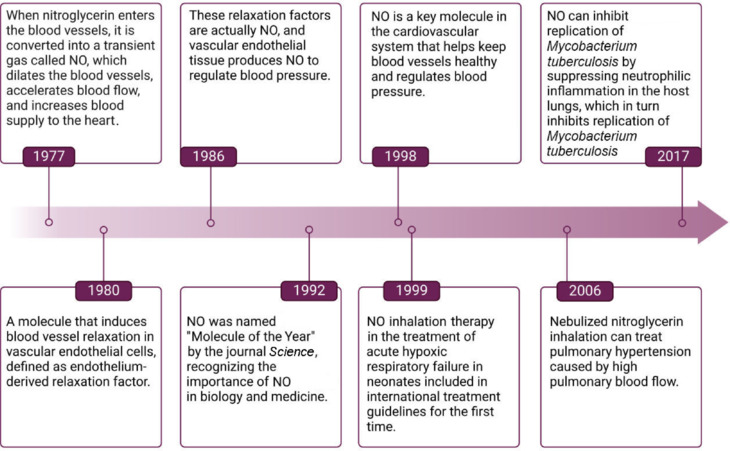
Historical milestones in the development of nitric oxide.
Created with BioRender.com. NO: Nitric oxide.
These advancements not only increase our understanding of the physiological functions of NO but also offer new perspectives and strategies for its application in the treatment of VD and other related diseases.
Physiological Role of Nitric Oxide
NO is a pivotal bioactive molecule that plays a significant role in regulating various cellular functions and physiological processes, serving as a crucial mediator of intercellular communication.26 The mechanisms through which NO exerts its effects in both physiological and pathological contexts are multifaceted: (1) Vasodilation and blood pressure regulation: NO is instrumental in maintaining vascular tension and regulating blood flow and blood pressure through its vasodilatory effects, which are essential for cardiovascular health.27 (2) Mitochondrial autophagy and cellular health: NO may facilitate the clearance of aged mitochondria, thereby promoting the health of intracellular mitochondria. This process is mediated through its influence on mitochondrial autophagy-related proteins, such as PINK1 and Parkin.28,29,30 (3) Macrophage activation and antibacterial activity: Endotoxins stimulate macrophages to produce significant amounts of iNOS, which synthesizes NO from L-arginine. NO interacts with superoxide ions to form peroxynitrite (ONOO⁻), thereby enhancing the antibacterial activity of macrophages.31,32 (4) Platelet function and thrombosis: NO binds to and activates soluble guanylate cyclase in platelets, leading to the downregulation of the platelet membrane glycoproteins IIb/IIIa. This action reduces platelet adhesion to endothelial cells, decreases the risk of thrombosis, and modulates leukocyte adhesion and smooth muscle cell proliferation.33,34 (5) DNA stability and antioxidant effects: NO indirectly affects DNA stability and enhances cellular antioxidant, anti-inflammatory, and antiviral responses through the modulation of intracellular redox states and the regulation of various signaling pathways.35,36,37 (6) Neurotransmitter function and synaptic regulation: As a neurotransmitter synthesized in the central nervous system, NO plays a critical role in synaptic transmission. It is released from the postsynaptic compartment and diffuses into presynaptic terminals through the synaptic cleft, promoting the release of glutamate and influencing long-term potentiation at synapses. This process is involved in pain modulation, memory formation, and neuronal activity38,39,40,41,42 (Figure 2). (7) Regulation of the peripheral nervous system and respiratory system: In the peripheral nervous system, NO acts as a mediator of vasodilation. As a noncholinergic and nonadrenergic neurotransmitter, it relaxes airway smooth muscle, dilates airways, and contributes to bacterial clearance in airway defense. The measurement of NO in exhaled breath has advanced clinical applications, promoting noninvasive technologies for assessing airway inflammation.43 (8) Gastrointestinal function regulation: In the gastrointestinal tract, NO regulates various secretory functions, including gastric acid and pepsinogen production, by acting on parietal cells, chief cells, mucous cells, and gastrointestinal epithelial cells. This regulation is considered part of the nonadrenergic and noncholinergic effects on gastrointestinal function.33 These diverse roles of NO underscore its importance in maintaining physiological homeostasis and highlight its potential therapeutic applications across a range of diseases (Figure 2).
Figure 2.
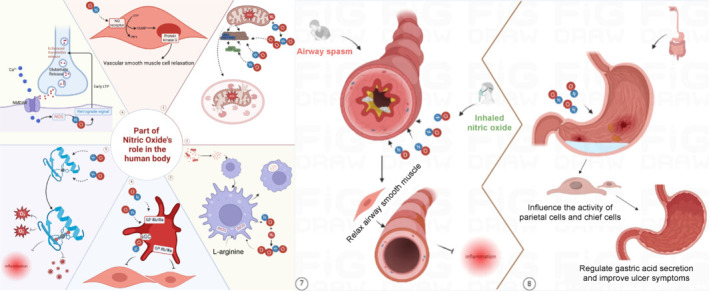
Physiological role of nitric oxide.
① NO activates the sGC cGMP pathway to promote vascular smooth muscle cell relaxation. ② PINK1 accumulates on damaged mitochondrial membranes, and NO and peroxynitrite are formed by the binding of NO with superoxide ions produced by damaged mitochondria, regulating the activity of PINK1 and Parkin to promote autophagy in aging mitochondria and maintain cellular health. ③ Activation of macrophages by endotoxins leads to the production of inducible NOS, which interacts with arginine to produce NO. NO and peroxynitrite stimulate macrophages to further regulate immune responses and promote phagocytosis of endotoxins by macrophages. ④ NO has been demonstrated to bind and activate sGC in platelets, leading to a reduction in the expression levels of GP IIb/IIIa and a subsequent decrease in platelet adhesion to endothelial cells. ⑤ NO indirectly affects DNA stability and enhances cellular antioxidant, anti-inflammatory, and antiviral responses through the modulation of intracellular redox states and the regulation of various signaling pathways. ⑥ NO released from the postsynaptic compartment enters presynaptic terminals through the synaptic cleft, which further promotes the release of glutamate and affects the long-term enhancement of synapses by releasing retrograde signals.⑦ NO relaxes airway smooth muscle, dilates airways, and contributes to bacterial clearance in airway defense. ⑧ NO regulates various secretory functions, including gastric acid and pepsinogen production, by acting on parietal cells, chief cells, mucous cells, and gastrointestinal epithelial cells. Created with BioRender.com. Ca2+: Calcium ion; cGMP: cyclic guanosine monophosphate; GP IIb/IIIa: platelet glycoprotein IIb/IIIa complex; GTP: guanosine triphosphate; iNOS: inducible nitric oxide synthase; LTP: long-term potentiation; NMDAR: N-methyl-D-aspartate receptor receptor; NO: nitric oxide; NOS: nitric oxide synthase; Parkin: E3 ubiquitin ligase; PINK1: PTEN-induced kinase 1; PP1: protein phosphatase 1; ROS: reactive oxygen species; sGC: soluble guanylate cyclase; Zn2+: zinc ion.
Source of Nitric Oxide
L-arginine and L-citrulline are two amino acids that play crucial roles in promoting the production of NO within the body.44 Specifically, the kidneys convert L-citrulline into L-arginine, which serves as a direct precursor for NO synthesis. In clinical practice, NO supplements, including L-arginine and L-citrulline, have been recommended to increase NO levels in patients with erectile dysfunction. These supplements positively influence vascular health and blood flow by promoting NO production.
Clinical administration of these amino acids can be achieved through various methods, including oral ingestion, injection, inhalation, topical application, and sublingual administration. Exogenous inhalation of NO has been utilized clinically for more than two decades and has demonstrated significant therapeutic effects on conditions such as pulmonary hypertension and neonatal hypoxemic respiratory failure. The safety and efficacy of inhaled NO have been well established,45 and it is currently employed in the treatment of acute respiratory distress syndrome, severe pneumonia, high-altitude pulmonary edema, acute pulmonary embolism, heart failure, and severe postoperative hypoxemia. However, its effectiveness in treating VD remains limited. Researchers have suggested that the intake of dietary nitrates and nitrites may hold therapeutic potential for various diseases, including stroke.46 These compounds can release NO within the body and are also associated with medications such as organic nitrates, nitrites, and nitroglycerin. Importantly, however, excessive consumption of nitrates and nitrites during endogenous nitrification may increase the risk of cancer,47 particularly in the context of VD treatment.
In the field of nanomedicine, researchers have developed iron sulfide nanoclusters capable of catalyzing the conversion of sodium nitrite to NO under moderate electric fields. These electrocatalytic nanoclusters, which function as drug carriers, not only increase the solubility and stability of pharmaceuticals but also improve their targeting capabilities.48 Recently, biomimetic nanomedicine delivery strategies utilizing cell membrane vesicles, live cells, and extracellular vesicles have garnered significant attention. For example, mesenchymal stem cells possess homing capabilities, enabling them to migrate to sites of injury or inflammation, where they can release therapeutic drugs or genes. They are utilized for the delivery of anti-inflammatory agents or gene therapy vectors.49 It is anticipated that targeted nanomedicines related to NO will have broad application prospects in the treatment of VD.
In addition to advancements in NO supplementation and nanocatalysis, lifestyle factors such as diet and exercise as sources of NO production in the body should not be overlooked. Strategies such as smoking cessation, dietary salt reduction, dietary modification, and regular physical activity are essential for maintaining optimal cerebrovascular function.50 Although no specific “NO food” exists, certain foods are known to increase NO levels in the body. These foods typically contain nitrates, which are converted into nitrites and subsequently into NO. A randomized, double-blind, parallel-group study involving 40 healthy adults demonstrated that daily consumption of 5.5 mmol of nitrate (equivalent to 450 mL of beetroot juice) resulted in increased cerebral blood flow, which may correlate with improvements in cognitive function.51 Notably, approximately 80% of dietary nitrate intake is derived from vegetables.
The effects of nitrate on human health are multifaceted and influenced by its source, intake level, and individual health status. Moderate nitrate intake, particularly from vegetable consumption, is associated with positive health outcomes, whereas excessive intake, especially from processed meats, may increase health risks.52 Thus, it is advisable to adopt a balanced diet and pay attention to the quality of drinking water. Research indicates that low doses of alcohol can activate endothelial NOS (eNOS) and upregulate its expression, thereby promoting the release of NO from endothelial cells.53 Conversely, long-term consumption of high concentrations of alcohol may impair endothelial function, leading to a reduction in eNOS expression.
In addition to vegetables and alcohol, fruits and chocolate also serve as significant dietary sources of nitrates, contributing to increased NO levels in the body. Studies have shown that the consumption of nitrates, whether in the form of sodium salts or as part of vegetables, can reduce oxygen consumption and enhance athletic performance. These findings show the potential benefits of nitrates in maintaining health and promoting physical fitness.54
Both acute and chronic physical exercise are associated with increased nitrite concentrations in the body,55 which parallels the concentrations achieved through dietary nitrate intake.56 Furthermore, research suggests that exercise can increase antioxidant activity, which helps mitigate the degradation of NO caused by free radicals. Based on these findings, a reasonable intake of dietary nitrates combined with regular physical exercise significantly improves the bioavailability of NO and may have beneficial effects on cognitive function.
The arginine NO pathway serves as the primary route for the biosynthesis of NO in the body. In this pathway, NOS catalyzes the oxidation of guanidine nitrogen in L-arginine, resulting in the production of NO and L-citrulline.57 The NOS system has garnered significant attention from researchers due to its role in regulating cerebral blood flow and neuronal activity. There are three subtypes of NOS: neuronal NOS (nNOS), eNOS, and iNOS, which are expressed in response to injury.
NO has both neurotoxic and neuroprotective effects, with these outcomes dependent on the specific NOS subtype produced by cells and the timing of its expression.57 NO derived from iNOS and nNOS may contribute to neurotoxicity, as it can directly damage protein structures, impair mitochondrial function, and act as a free radical that induces apoptosis. Conversely, NO produced by eNOS primarily exerts a neuroprotective effect in the context of VD through its vasodilatory properties.58
Following acute and chronic brain injuries, NO generated by the NOS system plays a critical role in maintaining systemic circulation and cerebral blood flow. It achieves this by promoting vasodilation and inhibiting platelet aggregation and leukocyte adhesion, thereby mitigating the progression of vascular cognitive impairment. The neuroprotective effects of eNOS-derived NO are largely mediated through the regulation of the vascular bed and peripheral nerve tissue.59
Extensive research has focused on the role of NO in subarachnoid hemorrhage, a form of acute hemorrhagic brain injury. Long-term VD following subarachnoid hemorrhage is characterized by delayed ischemic neurological deficits and cerebral vasospasm. These phenomena are associated with complex pathophysiological processes, and some studies indicate that dysfunction of NOS near the cerebral vascular bed contributes to cerebral vasospasm, delayed ischemic neurological deficits, and the clearance of deoxyhemoglobin.60,61,62 This relationship suggests that the NO concentration is linked to cerebral vasospasm, with some analyses suggesting that NO activation may improve the vascular diameter, thereby exerting a neuroprotective effect.63,64,65 However, there is a lack of definitive scientific evidence to support a positive correlation between changes in NO concentration and vascular protection.
NO plays a pivotal role in both physiological and pathological changes, particularly in relation to VD and other cognitive dysfunction disorders. Research has demonstrated that the expression of nNOS in neuronal cells is crucial for regulating neuronal plasticity, memory formation, and neurotransmitter release.66 The NO produced by nNOS is essential for modulating synaptic plasticity and has a neuroprotective effect on vascular cognitive disorders, such as VD.67 In contrast, iNOS is primarily expressed by astrocytes and microglia in the central nervous system, and its expression is regulated at both the transcriptional and translational levels.68 These findings underscore the complex and dualistic roles of NO in the brain, highlighting its potential as both a therapeutic target and a biomarker in the context of VD and related cognitive impairments. Experimental evidence indicates that cells expressing iNOS accumulate around areas of brain damage, suggesting an inflammatory link between glial cell-derived NO and neuronal injury.69 Studies have shown that the overexpression or dysregulation of iNOS is associated with the onset and progression of various diseases, including sepsis, cancer, and neurodegenerative disorders.70 Following inflammatory injury or ischemic events, the expression of iNOS in microglia is activated, positioning iNOS as an immunogenic form of NOS.71,72
The inhibition of iNOS has been shown to enhance synaptic plasticity and ameliorate memory deficits induced by traumatic stress.73 Recent research has demonstrated that the iNOS inhibitor aminoguanidine significantly alleviates endothelial dysfunction and cognitive impairment in rat models of VD.74 Consequently, iNOS inhibitors are being explored as potential therapeutics for neural targeting, with the prospect of playing a critical role in the treatment of brain injuries.
Given the significant role of iNOS in nerve injury and related pathologies, the development of iNOS inhibitors represents a promising avenue for future research aimed at addressing cognitive dysfunction. These findings underscore the potential of targeting the iNOS pathway as a therapeutic strategy for improving cognitive outcomes in various neurological conditions.75
Modulatory Roles of Nitric Oxide in Vascular Dementia
Improving vascular tension and endothelial homeostasis
Vascular tension, defined as the resistance of the vascular wall to blood flow, is primarily regulated by the contraction state of vascular smooth muscle cells.76 Abnormal regulation of vascular tension due to cerebrovascular disease can adversely affect cerebral blood flow and exacerbate cognitive dysfunction.77 Therefore, precise regulation of vascular tension is essential for maintaining healthy cerebral blood flow and for delaying cognitive decline in patients with VD.78
NO is predominantly produced by eNOS in vascular endothelial cells. NO activates soluble guanylate cyclase, leading to an increase in intracellular cyclic guanosine monophosphate (cGMP) levels.79 This cascade results in the relaxation of vascular smooth muscle cells and subsequent vasodilation, thereby maintaining endothelial homeostasis. The blood‒brain barrier (BBB) consists of continuous endothelial cells lining the microvessels of the brain.80 Disruption of the BBB facilitates the progression of cognitive impairment following brain injury,81 with endothelial dysfunction being a critical mechanism underlying VD. Damaged endothelial cells can reduce blood flow and increase the permeability of the cerebral vascular barrier.82 Recent studies have shown that the nNOS inhibitor 2-aminopyridine can inhibit NO production and enhance cell membrane permeability, thereby facilitating the passage of certain substances across the BBB.83 In the context of VD, NO deficiency may lead to reduced synthesis of brain-derived neurotrophic factor, which inhibits the protective functions of astrocytes,84 ultimately resulting in BBB damage and the release of cytotoxic substances in the brain. This creates a vicious cycle that exacerbates the cognitive impairments associated with VD. Both the precursor and mature forms of brain-derived neurotrophic factors are critical in the pathophysiological mechanisms of cognitive impairment, as they play vital roles in maintaining neuronal survival, promoting plasticity, and regulating neurotransmitter systems.85 A review of recent research highlights that under physiological conditions, endogenous NO primarily regulates microvascular permeability,86 with various vasoactive neurotransmitters acting through this pathway. Conversely, under pathological conditions, elevated NO concentrations may compromise the barrier function of vascular endothelial cells, increase permeability, and contribute to secondary brain edema87 (Figure 3). The immune inflammatory response following brain injury may increase the production of matrix metalloproteinases and myeloperoxidase, which are significant contributors to BBB disruption.88 Future research should focus on elucidating the threshold at which increased BBB permeability occurs due to changes in NO concentration, as well as investigating the causal relationships between NO and BBB-related factors, such as matrix metalloproteinases.
Figure 3.
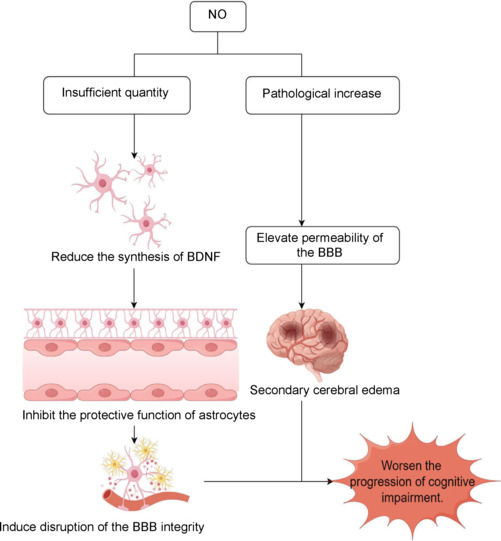
Effects of different concentrations of NO on the endothelial function of cerebral blood vessels.
In the context of VD, NO deficiency may lead to reduced synthesis of brain-derived neurotrophic factor, which inhibits the protective functions of astrocytes, ultimately resulting in BBB damage and the release of cytotoxic substances in the brain. Under pathological conditions, elevated NO concentrations may compromise the barrier function of vascular endothelial cells, increase permeability, and contribute to secondary brain edema. Created with Figdraw. BBB: Blood‒brain barrier; BDNF: brain-derived neurotrophic factor; NO: nitric oxide; VD: vascular dementia.
Alleviating neuroinflammation
Inflammation plays a pivotal role in cerebrovascular diseases, particularly following cerebral ischemia.89,90 The inflammatory response is a significant contributor to secondary damage to the brain as well as other organs and is integral to the pathogenesis of various neurological injuries.88,91,92 VD is frequently associated with pronounced neuroinflammation, which is a key factor in cognitive impairment.93 Patients with VD often exhibit cerebrovascular lesions or impaired cerebral blood flow. Prolonged chronic hypoperfusion can induce ischemia and hypoxia, leading to excessive activation of neuroinflammation, thereby triggering or exacerbating cognitive decline.
Neuroinflammation involves the activation of immune responses by microglia and astrocytes in the central nervous system.94,95 Microglia play crucial roles in detecting environmental changes, responding to harmful stimuli, and clearing debris and apoptotic neurons.10 In the context of VD, the activation status of astrocytes and microglia serves as an important indicator of neurodegeneration.96 Moderate concentrations of NO can inhibit the activity of astrocytes and microglia, thereby reducing neuroinflammation and neuronal damage associated with VD.97 However, persistent inflammation can lead to the activation of microglia, resulting in the production of NO, which inhibits the proliferation of neural stem cells. This inhibitory effect may occur through the nitration of epidermal growth factor receptors, which blocks normal signal transduction mediated by the extracellular signal-regulated kinase/mitogen-activated protein kinase pathway, ultimately hindering neural stem cell proliferation.98 Maksoud et al.99 demonstrated that the slow-release NO donor NOC-18 can inhibit microglial proliferation by activating protein kinase G. In a persistent state of neuroinflammation, inflammatory cytokines such as tumor necrosis factor-α and -β can activate iNOS via the Toll-like receptor 4/nuclear factor kappa B pathway, leading to the production of excess NO. This NO can react with reactive oxygen species (ROS) to form peroxynitrite (ONOO−), a potent oxidant and nitrating agent that mediates oxidative and nitrative stress. Pathologically elevated concentrations of NO can result in a cascade of pathological damage through oxidative/nitrative/nitrosative stress and the NO/cGMP signaling pathway.100
Recent findings suggest that the NO/cGMP signaling pathway may underlie the increased expression of glial fibrillary acidic protein in human glioblastoma U-373MG cells, suggesting a potential new target for therapeutic interventions in neurological diseases. However, in the absence of cGMP-protein kinase G signaling, NO may accelerate the progression of the microglial cell cycle.101 These results highlight the complexity of the mechanisms by which NO regulates neuroinflammation, making it challenging to differentiate between the physiological neuroprotective effects of moderate NO and the pathological damage caused by excessive NO production during neuroinflammatory processes.
In VD, although iNOS activity is elevated, the overall expression of NO significantly decreases. Under the influence of various inflammatory factors and ROS, NO expression increases in astrocytes and microglia. Exogenous NO can inhibit the activation of astrocytes and microglia, thereby suppressing neuroinflammation through the soluble guanylate cyclase-cGMP-protein kinase G pathway.102 Consequently, downregulating NO production by inhibiting iNOS in microglia may represent an effective strategy for mitigating neuroinflammation.
Inhibiting oxidative stress
Oxidative stress is a fundamental pathophysiological process in brain injury diseases and significantly influences the alleviation or progression of cognitive impairment.103 This condition triggers the production and accumulation of ROS and reactive nitrogen species, leading to a cascade of oxidative reactions, including the generation of hydrogen peroxide (H2O2), lipid peroxidation, and damage to DNA and RNA, as well as mitochondrial dysfunction. These processes further exacerbate neuronal and glial cell damage. Consequently, antioxidant stress strategies, such as VD, are vital for preventing and treating cognitive impairments and for promoting neurological recovery.
The extent of oxidative stress is a determinant of the course and prognosis of cognitive impairments, including VD. Mitochondrial dysfunction related to oxidative stress represents a core pathophysiological mechanism underlying cognitive decline and dementia in patients with VD.104 In low-oxygen environments, the regional accumulation of NO within cells is critical for glycolysis,105 as NO helps maintain normal ATP levels by inhibiting mitochondrial respiration and enhancing glycolytic activity.106
Conversely, under high-oxygen conditions, hydroxyl radicals can react with DNA components, resulting in DNA damage that may lead to strand breakage or cross-linking. Such DNA damage activates downstream pathways, including those mediated by 3-phosphate inositol kinase-related kinase, which facilitate the recognition of DNA damage and promote the nuclear translocation of apoptosis-inducing factors, ultimately leading to the degradation of DNA repair enzymes. Mitochondria are significant sources of ROS; however, their capacity for DNA repair is limited, and they lack histone protection, rendering mitochondrial DNA particularly vulnerable to ROS-induced damage. Additionally, ROS can inflict damage on endothelial cells and impair NO production, contributing to the neurotoxic effects associated with pathologically high concentrations of NO107 (Figure 4).
Figure 4.
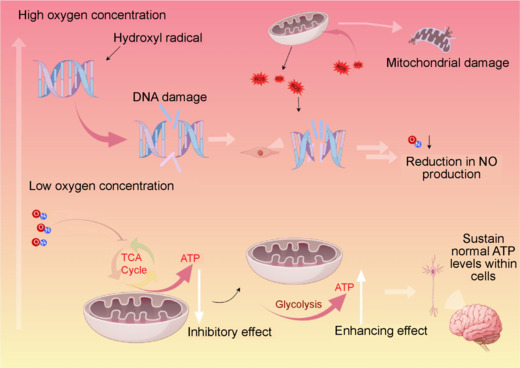
The modulatory effects of NO on metabolic processes under varying oxygen concentrations.
In low-oxygen environments, the regional accumulation of NO within cells is critical for glycolysis, as NO helps maintain normal ATP levels by inhibiting mitochondrial respiration and enhancing glycolytic activity. Under conditions of elevated oxygen concentrations, hydroxyl radicals can interact with DNA constituents, leading to DNA damage and the generation of significant amounts of ROS within mitochondria. However, the capacity for DNA repair is limited, and the absence of histone protection renders mitochondrial DNA particularly vulnerable to ROS-induced damage. Furthermore, ROS can inflict DNA damage in endothelial cells, subsequently diminishing NO production. Created with Figdraw. ATP: Adenosine triphosphate; NO: nitric oxide; ROS: reactive oxygen species; TCA cycle: tricarboxylic acid cycle.
Throughout the course of elevated ROS concentrations, the interplay between NO levels and the inhibition of NO-mediated cellular protection by oxidative stress remains complex and uncertain, potentially accelerating the deterioration of neurological function. Therefore, precise regulation of NO concentrations and oxidative stress levels is crucial for maintaining neurological function and effectively treating cognitive impairments.
On the other hand, nitrosative stress is closely linked to oxidative stress and involves the interplay between the formation and clearance pathways of ROS and reactive nitrogen species.108 ROS, including superoxide anion (O2−), H2O2, and hydroxyl radicals, interact at elevated ROS levels to generate a series of potent oxidative free radicals and nitro compounds, such as ONOO− and peroxynitrous acid (HOONO).109 These compounds can lead to the formation of 3-nitrotyrosine from various biomolecules, including lipids, DNA, RNA, and proteins, resulting in cellular damage or apoptosis—a phenomenon commonly referred to as nitrosative stress.110 The mechanisms of cell death associated with nitrosative stress include the nitration of target protein tyrosines, mitochondrial dysfunction, and disruption of cell membranes. In the context of ischemic brain injury, blood flow obstruction leads to a rapid decrease in the NO concentration. As the condition stabilizes and VD develops following brain injury, blood flow gradually recovers, resulting in an increase in NO production, which is primarily mediated by nNOS. Research utilizing nNOS gene knockout mice and specific nNOS inhibitors has supported the notion111 that NO synthesized through nNOS is significantly associated with glutamate-induced calcium overload in ischemic neurons.112 Within 1 hour after reperfusion, NO levels return to physiological concentrations. However, defects in the nNOS gene or inhibition of nNOS can reduce the area of the ischemic penumbra and the extent of neuronal necrosis. Moreover, inhibiting nNOS has been shown to decrease the production of oxygen free radicals and nitrosative stress,113 mitigate excitotoxicity, and downregulate the expression of caspase-3 in ischemic stroke.114
These findings underscore the complex role of nNOS and its derived NO in the pathophysiology of ischemic brain injury and highlight the potential therapeutic implications of targeting nNOS to alleviate nitrosative stress and its associated cellular damage.
Regulating synaptic plasticity
Neural cells establish functional neural circuits through specialized communication structures known as synapses, which are essential for the transmission and storage of information.115,116 Synapses can undergo specific structural and functional changes in response to continuous neuronal activity, a phenomenon referred to as synaptic plasticity.117,118 This process is critical for the development of the nervous system as well as for learning and memory. Synaptic dysfunction is recognized as one of the primary factors contributing to cognitive decline.
NO is a key player in neuronal communication and significantly influences synaptic plasticity, thereby playing an important role in the pathogenesis of VD. The mechanisms and effects of NO are multifaceted, and NO is essential for enhancing learning and memory functions in the context of VD. As a neurotransmitter, NO modulates long-term potentiation of synaptic transmission, a fundamental process underlying learning and memory.119 The production and regulation of NO are closely linked to the release of neurotransmitters associated with cognitive functions, such as acetylcholine.120
Research indicates that NO participates in synaptic plasticity and memory formation through the NO-cGMP-cyclic adenosine monophosphate response element-binding signaling pathway.121 Additionally, NO plays a role in regulating cerebral blood flow, which is crucial for delivering oxygen and nutrients to the brain, thereby influencing cognitive function.75 In terms of neural development, NO is involved in regulating developmental processes and gene expression, which are vital for the formation and consolidation of learning and memory.122 A study suggested a potential interaction between NO and nicotinic acetylcholine receptors during learning and memory processes in rats. For example, the administration of the NO precursor L-arginine or the α7 nicotinic acetylcholine receptor agonist choline chloride into the lateral ventricle has been shown to enhance learning and memory behaviors in these animals.123 Furthermore, NO influences learning and memory by interacting with N-methyl-D-aspartate glutamate receptors124 (Figure 5). When N-methyl-D-aspartate receptors are overactivated, they stimulate the production of NO, which can then undergo S-nitrosylation reactions and attach to various proteins. This process may result in premature death of neuronal cells, a hallmark of neurodegenerative diseases.
Figure 5.
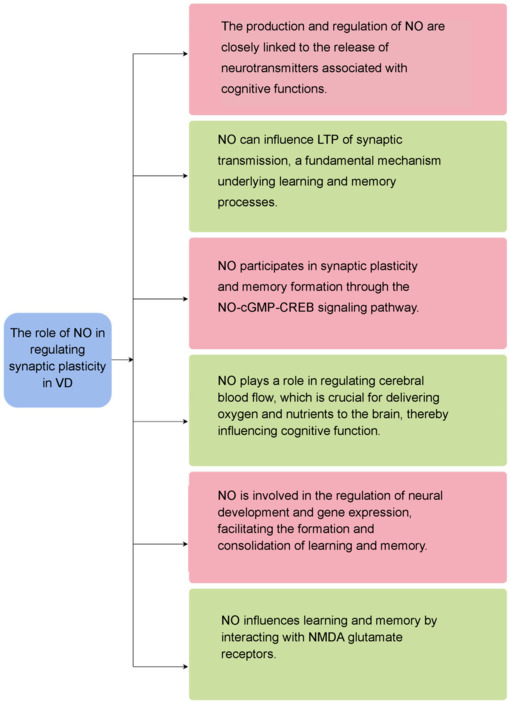
Role of NO in the modulation of synaptic plasticity in the context of VD.
Created with Figdraw. cAMP: Cyclic adenosine monophosphate; cGMP: cyclic guanosine monophosphate; CREB: cAMP response element-binding protein; LTP: long-term potentiation; NMDA: N-methyl-D-aspartate; NO: nitric oxide; VD: vascular dementia.
Excessive NO can have neurotoxic effects, directly damaging protein structures, impairing mitochondrial function, and inducing apoptosis as a free radical.125,126,127 As neurodegenerative diseases progress, NO deficiency often occurs in critical brain regions, such as the substantia nigra pars compacta, hippocampus, and striatum. Conversely, moderate levels of NO have been shown to exert beneficial therapeutic effects on synaptic plasticity in VD.
In summary, NO plays a dual role in cognitive impairment related to VD, where both deficiency and excess NO can lead to synaptic dysfunction and neuronal damage.128 Given its regulatory role in synaptic plasticity, NO represents a potential target for therapeutic interventions aimed at treating VD.129
Research Advances in the Application of Nitric Oxide Synthase Inhibitors for Vascular Dementia
The experimental application of NOS inhibitors in the management of VD represents a burgeoning area of research. Regulating NOS activity has emerged as a promising therapeutic strategy for addressing the pathophysiological mechanisms underlying VD. NOS inhibitors can be categorized into nonselective and selective agents: for example, L-NAME (Nω-nitro-L-arginine methyl ester) and L-NNA (Nω-nitro-L-arginine) are known to inhibit all isoforms of NOS, whereas 7-nitroindole predominantly targets nNOS, and L-NIO (Nω-nitroso-L-arginine) primarily inhibits iNOS.
Numerous studies employing animal models of VD have been conducted to assess the therapeutic efficacy of NOS inhibitors.130,131 The evidence from these investigations indicates that NOS inhibitors may enhance cognitive function in such models. For example, animals administered L-NAME demonstrated improved spatial memory performance, as evidenced by enhanced outcomes in the Morris water maze test. The neuroprotective effects of NOS inhibitors are hypothesized to stem from their ability to mitigate the excessive production of NO during the progression of VD, thereby alleviating oxidative stress and neuroinflammation.132
Selective NOS inhibitors may further contribute to the amelioration of cognitive deficits by modulating cerebral blood flow, which in turn improves the delivery of oxygen and nutrients to brain tissue.133 Conversely, the use of nonselective NOS inhibitors may be associated with adverse effects, including hypertension and disturbances in cardiovascular function.134 Consequently, selective NOS inhibitors are posited to possess greater therapeutic potential.
The effectiveness and safety profile of NOS inhibitors are contingent upon factors such as dosage and administration route. Therefore, it is imperative for experimental research to optimize these parameters to achieve maximal therapeutic outcomes. While short-term studies have highlighted the potential benefits of NOS inhibitors, further investigations are warranted to explore the selective inhibition, dosage optimization, and long-term safety of extended treatment regimens.
Limitations
First, due to time, resource, and knowledge limitations, investigators may not be able to cover all relevant literature, which may result in some important studies or ideas being missed in the review. Second, only PubMed articles were searched, and no other biomedical databases were included. Third, the effect of NO on related pathways in terms of molecular structure was not discussed.
Conclusion
NO, as a gaseous signaling molecule, plays a crucial role in the onset and progression of cognitive impairment diseases, including VD. Currently, numerous treatment strategies targeting the NOS/NO pathway in VD are under investigation, showing promise for therapeutic applications. Research on NO has emerged as a potential game changer in the field of VD, with a particular focus on NO-related targeted nanomedicines, which are gaining attention for their specificity and effectiveness.
One effective strategy for protecting neurotransmitter transmission between synapses involves the selective inhibition of iNOS to reduce NO accumulation in damaged brain areas. Therefore, distinguishing the roles of the three subtypes of NOS isoenzymes and adopting a combination therapy approach appears to be a more favorable treatment option. Future research should concentrate on developing satisfactory and rational combination therapies, thoroughly elucidating the relationship and mechanisms of action between endogenous NO and VD in both physiological and pathological states. Such insights may provide new avenues for the development of effective treatment modalities for VD.
The exploration of the correlation between medical gases and diseases aims to enhance clinical practice. This review details the interaction between NO and VD, acknowledging that while its potential mechanisms have not been fully elucidated, further clinical trials are necessary to validate its efficacy. We recommend that future clinical trials expand their sample sizes and include follow-up assessments regarding the speed and severity of cognitive impairment progression. Additionally, variables such as NO-related targeted nanomedicines and selective iNOS inhibitors should be incorporated to develop more accurate drug delivery systems. This approach aims to improve treatment efficacy while minimizing side effects and achieving precise control over the release of NO-related targeted nanomedicines (Table 1).135,136,137,138,139,140,141
Table 1.
Common nitric oxide synthase inhibitors
| Inhibitor | Function introduction | Reference |
|---|---|---|
| 1400W | It is a slow binding, highly selective iNOS inhibitor with a Kd value not exceeding 7 nM. Research has shown that 1400W can reduce the generation of NO, thereby alleviating oxidative stress and neuronal apoptosis, and improving spatial memory impairment in rats caused by acute hypoxia reoxygenation. | 135 |
| 2-Iminobiotin | It is a reversible NO synthase inhibitor that has effects on mouse iNOS and rat nNOS, with Ki values of 21.8 and 37.5 μM, respectively. 2-iminobiotin and low temperature environment can protect human nerve cells from hypoxia induced cell damage. | 136 |
| 7-NI | It is a reversible competitive inhibitor of NOS that efficiently and selectively inhibits nNOS and iNOS. Research has shown that 7-NI can affect cognitive function in rats, including passive avoidance and elevated maze memory performance. | 137 |
| SMT | It is an efficient, selective, and competitive inhibitor of iNOS. The inhibitory effect of SMT on iNOS in vitro is 10 to 30 times stronger than that of NG-methyl-L-arginine or other known NOS inhibitors, and its effect on iNOS activity can be reversed by excessive L-arginine. | 138 |
| Madecassic acid | Madecasic acid is the first natural triterpene isolated from Centella asiatica, which has a variety of anti-inflammatory and anti-diabetes effects, blocking the activation of NF-κB in macrophages and leading to inhibition of iNOS, COX-2, TNF alpha, IL-1beta and IL-6. | 139 |
| L-NAME | L-NAME is a non-selective inhibitor of nitric oxide synthase that inhibits nNOS, The Ki values of eNOS and iNOS are 15, 39 nM and 4.4 μM. | 140 |
| DPI | DPI is an inhibitor of NADPH oxidase and an effective and irreversible inhibitor of iNOS/eNOS. DPI also acts as an activator of TRPA1 and selectively inhibits intracellular reactive oxygen species. | 141 |
7-NI: 7-Nitroindazole; COX-2: cyclooxygenase-2; DPI: diphenyleneiodonium chloride; eNOS: endothelial NOS; IL: interleukin; iNOS: inducible NOS; NADPH: nicotinamide adenine dinucleotide phosphate; NF-κB: nuclear factor kappa B; nNOS: neuronal NOS; NO: nitric oxide; NOS: NO synthase; SMT: S-methylisothiourea sulfate; TNF: tumor necrosis factor; TRPA1: transient receptor potential ankyrin 1.
Enhancing the therapeutic efficacy of NO in the treatment of diseases associated with cognitive dysfunction, such as VD, also holds the potential to advance precision medicine in other fields, including the treatment of airway inflammation in the respiratory system, intestinal mucosal protection in the digestive system, and targeted interventions for osteoarthritis in the musculoskeletal system. By precisely regulating the bioavailability and timing of NO, more personalized and efficient treatment strategies can be developed for these diverse conditions.
Funding Statement
Funding: This work was supported by the China Postdoctoral Science Foundation, No. 2022M712689, and the Natural Science Foundation of the Jiangsu Higher Education Institutions of China, No. 22KJB1800029, and Jiangsu Province Science and Technology Deputy Project Fund, No. FZ20240964 (all to TX).
Footnotes
Conflicts of interest: The authors declare no conflicts of interest.
Data availability statement:
Not available.
References
- 1.Koyanagi A, Veronese N, Stubbs B, et al. Food insecurity is associated with mild cognitive impairment among middle-aged and older adults in South Africa: findings from a nationally representative survey. Nutrients. 2019;11:749. doi: 10.3390/nu11040749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kakutani S, Watanabe H, Murayama N. Green tea intake and risks for dementia, Alzheimer’s disease, mild cognitive impairment, and cognitive impairment: a systematic review. Nutrients. 2019;11:1165. doi: 10.3390/nu11051165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yan N, Zhang JJ. The emerging roles of ferroptosis in vascular cognitive impairment. Front Neurosci. 2019;13:811. doi: 10.3389/fnins.2019.00811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bu X, Zhang Y, Bazzano LA, et al. Effects of early blood pressure reduction on cognitive function in patients with acute ischemic stroke. Int J Stroke. 2016;11:1009–1019. doi: 10.1177/1747493016660094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Whiteley WN, Gupta AK, Godec T, et al. Long-term incidence of stroke and dementia in ASCOT. Stroke. 2021;52:3088–3096. doi: 10.1161/STROKEAHA.120.033489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Llorens F, Hermann P, Villar-Piqué A, et al. Cerebrospinal fluid lipocalin 2 as a novel biomarker for the differential diagnosis of vascular dementia. Nat Commun. 2020;11:619. doi: 10.1038/s41467-020-14373-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shabir O, Pendry B, Lee L, et al. Assessment of neurovascular coupling and cortical spreading depression in mixed mouse models of atherosclerosis and Alzheimer’s disease. Elife. 2022;11:e68242. doi: 10.7554/eLife.68242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Skrobot OA, Black SE, Chen C, et al. Progress toward standardized diagnosis of vascular cognitive impairment: guidelines from the vascular impairment of cognition classification consensus study. Alzheimers Dement. 2018;14:280–292. doi: 10.1016/j.jalz.2017.09.007. [DOI] [PubMed] [Google Scholar]
- 9.Forrester SN, McManus DD, Saczynski JS, Pierre-Louis IC, Bamgbade BA, Kiefe CI. A cross-sectional analysis of racial differences in accelerated aging and cognitive function among patients with atrial fibrillation: The SAGE-AF study: Forrester, Accelerated aging and cognitive function. EClinicalMedicine. 2021;39:101060. doi: 10.1016/j.eclinm.2021.101060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wei C, Zhu Z, Zheng JN, et al. Chinese medicine, Succinum, ameliorates cognitive impairment of carotid artery ligation rats and inhibits apoptosis of HT22 hippocampal cells via regulation of the GSK3β/β-catenin pathway. Front Pharmacol. 2022;13:867477. doi: 10.3389/fphar.2022.867477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Johnson NF, Gold BT, Brown CA, et al. Endothelial function is associated with white matter microstructure and executive function in older adults. Front Aging Neurosci. 2017;9:255. doi: 10.3389/fnagi.2017.00255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jenkins NDM, Rogers EM, Banks NF, et al. Childhood psychosocial stress is linked with impaired vascular endothelial function, lower SIRT1, and oxidative stress in young adulthood. Am J Physiol Heart Circ Physiol. 2021;321:H532–H541. doi: 10.1152/ajpheart.00123.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Katsuki S, Arnold W, Mittal C, Murad F. Stimulation of guanylate cyclase by sodium nitroprusside, nitroglycerin and nitric oxide in various tissue preparations and comparison to the effects of sodium azide and hydroxylamine. J Cyclic Nucleotide Res. 1977;3:23–35. [PubMed] [Google Scholar]
- 14.Furchgott RF, Zawadzki JV. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980;288:373–376. doi: 10.1038/288373a0. [DOI] [PubMed] [Google Scholar]
- 15.Ignarro LJ, Adams JB, Horwitz PM, Wood KS. Activation of soluble guanylate cyclase by NO-hemoproteins involves NO-heme exchange. Comparison of heme-containing and heme-deficient enzyme forms. J Biol Chem. 1986;261:4997–5002. [PubMed] [Google Scholar]
- 16.Furchgott RF. Endothelium-derived relaxing factor: discovery, early studies, and identification as nitric oxide. Biosci Rep. 1999;19:235–251. doi: 10.1023/a:1020537506008. [DOI] [PubMed] [Google Scholar]
- 17.Culotta E, Koshland DE., Jr NO news is good news. Science. 1992;258:1862–1865. doi: 10.1126/science.1361684. [DOI] [PubMed] [Google Scholar]
- 18.Ignarro LJ. Nitric oxide: a unique endogenous signaling molecule in vascular biology (Nobel lecture) Angew Chem Int Ed Engl. 1999;38:1882–1892. doi: 10.1002/(SICI)1521-3773(19990712)38:13/14<1882::AID-ANIE1882>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 19.Carpenter AW, Schoenfisch MH. Nitric oxide release: part II. Therapeutic applications. Chem Soc Rev. 2012;41:3742–3752. doi: 10.1039/c2cs15273h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Furchgott RF. Endothelium-derived relaxing factor: discovery, early studies, and identifcation as nitric oxide (Nobel lecture) Angew Chem Int Ed Engl. 1999;38:1870–1880. doi: 10.1002/(SICI)1521-3773(19990712)38:13/14<1870::AID-ANIE1870>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 21.Wei L, Zhang J, Wei S, et al. Nitric oxide enhanced salt stress tolerance in tomato seedlings, involving phytohormone equilibrium and photosynthesis. Int J Mol Sci. 2022;23:4539. doi: 10.3390/ijms23094539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Foresi N, Correa-Aragunde N, Parisi G, Caló G, Salerno G, Lamattina L. Characterization of a nitric oxide synthase from the plant kingdom: NO generation from the green alga Ostreococcus tauri is light irradiance and growth phase dependent. Plant Cell. 2010;22:3816–3830. doi: 10.1105/tpc.109.073510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Davidson D, Barefield ES, Kattwinkel J, et al. Inhaled nitric oxide for the early treatment of persistent pulmonary hypertension of the term newborn: a randomized, double-masked, placebo-controlled, dose-response, multicenter study. The I-NO/PPHN Study Group. Pediatrics. 1998;101:325–334. doi: 10.1542/peds.101.3.325. [DOI] [PubMed] [Google Scholar]
- 24.Puikuan K, Chunyu Z, Jin F, Chaoshu T, Junbao D. Inhalation of nebulized nitroglycerin, a nitric oxide donor, for the treatment of pulmonary hypertension induced by high pulmonary blood flow. Heart Vessels. 2006;21:169–179. doi: 10.1007/s00380-005-0876-y. [DOI] [PubMed] [Google Scholar]
- 25.Mishra BB, Lovewell RR, Olive AJ, et al. Nitric oxide prevents a pathogen-permissive granulocytic inflammation during tuberculosis. Nat Microbiol. 2017;2:17072. doi: 10.1038/nmicrobiol.2017.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hafez S, Khan MB, Awad ME, Wagner JD, Hess DC. Short-term acute exercise preconditioning reduces neurovascular injury after stroke through induced eNOS activation. Transl Stroke Res. 2020;11:851–860. doi: 10.1007/s12975-019-00767-y. [DOI] [PubMed] [Google Scholar]
- 27.Tartar JL, Kalman D, Hewlings S. A prospective study evaluating the effects of a nutritional supplement intervention on cognition, mood states, and mental performance in video gamers. Nutrients. 2019;11:2326. doi: 10.3390/nu11102326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Martinez A, Lectez B, Ramirez J, et al. Quantitative proteomic analysis of Parkin substrates in Drosophila neurons. Mol Neurodegener. 2017;12:29. doi: 10.1186/s13024-017-0170-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lin M, Xian H, Chen Z, et al. MCM8-mediated mitophagy protects vascular health in response to nitric oxide signaling in a mouse model of Kawasaki disease. Nat Cardiovasc Res. 2023;2:778–792. doi: 10.1038/s44161-023-00314-x. [DOI] [PubMed] [Google Scholar]
- 30.Kim BG, Song Y, Lee MG, et al. Macrophages from mice administered Rhus verniciflua stokes extract show selective anti-inflammatory activity. Nutrients. 2018;10:1926. doi: 10.3390/nu10121926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.MacKinney A, Woska E, Spasojevic I, Batinic-Haberle I, Zennadi R. Disrupting the vicious cycle created by NOX activation in sickle erythrocytes exposed to hypoxia/reoxygenation prevents adhesion and vasoocclusion. Redox Biol. 2019;25:101097. doi: 10.1016/j.redox.2019.101097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pandey A, Estepp JH, Raja R, Kang G, Ramkrishna D. Mathematical modeling of hydroxyurea therapy in individuals with sickle cell disease. Pharmaceutics. 2022;14:1065. doi: 10.3390/pharmaceutics14051065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pacher P, Beckman JS, Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol Rev. 2007;87:315–424. doi: 10.1152/physrev.00029.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ochijewicz D, Rdzanek A, Przybyłowski T, et al. Influence of apnea hypopnea index and the degree of airflow limitation on endothelial function in patients undergoing diagnostic coronary angiography. Biology (Basel) 2022;11:457. doi: 10.3390/biology11030457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Coma R, Ribes M, Gili J, Zabala M. Seasonality in coastal benthic ecosystems. Trends Ecol Evol. 2000(15):448–453. doi: 10.1016/s0169-5347(00)01970-4. [DOI] [PubMed] [Google Scholar]
- 36.Søvsø MB, Hermansen SB, Færk E, et al. Diagnosis and mortality of emergency department patients in the North Denmark region. BMC Health Serv Res. 2018;18:548. doi: 10.1186/s12913-018-3361-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Stamler JS, Meissner G. Physiology of nitric oxide in skeletal muscle. Physiol Rev. 2001;81:209–237. doi: 10.1152/physrev.2001.81.1.209. [DOI] [PubMed] [Google Scholar]
- 38.Jiang S, Dandu C, Geng X. Clinical application of nitric oxide in ischemia and reperfusion injury: A literature review. Brain Circ. 2020;6:248–253. doi: 10.4103/bc.bc_69_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Azarkish F, Armin F, Parvar AAA, Dehghani A. The influence of renal ischemia-reperfusion injury on remote organs: the histological brain changes in male and female rats. Brain Circ. 2021;7:194–200. doi: 10.4103/bc.bc_3_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Picón-Pagès P, Garcia-Buendia J, Muñoz FJ. Functions and dysfunctions of nitric oxide in brain. Biochim Biophys Acta Mol Basis Dis. 2019;1865:1949–1967. doi: 10.1016/j.bbadis.2018.11.007. [DOI] [PubMed] [Google Scholar]
- 41.Nobili S, Lucarini E, Murzilli S, Vanelli A, Di Cesare Mannelli L, Ghelardini C. Efficacy evaluation of plant products in the treatment of erectile dysfunction related to diabetes. Nutrients. 2021;13:4520. doi: 10.3390/nu13124520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jagielski P, Wnęk D, Łuszczki E, et al. Proposition of a new POLA index to assess the immunomodulatory properties of the diet and its relationship with the gut microbiota, using the example of the incidence of COVID-19 in a group of people without comorbidities. Nutrients. 2022;14:4227. doi: 10.3390/nu14204227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Derwall M, Ebeling A, Nolte KW, et al. Inhaled nitric oxide improves transpulmonary blood flow and clinical outcomes after prolonged cardiac arrest: a large animal study. Crit Care. 2015;19:328. doi: 10.1186/s13054-015-1050-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lundberg JO, Weitzberg E, Gladwin MT. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat Rev Drug Discov. 2008;7:156–167. doi: 10.1038/nrd2466. [DOI] [PubMed] [Google Scholar]
- 45.Habermeyer M, Roth A, Guth S, et al. Nitrate and nitrite in the diet: how to assess their benefit and risk for human health. Mol Nutr Food Res. 2015;59:106–128. doi: 10.1002/mnfr.201400286. [DOI] [PubMed] [Google Scholar]
- 46.Chen Y, Chen C, Zhang X, et al. Platinum complexes of curcumin delivered by dual-responsive polymeric nanoparticles improve chemotherapeutic efficacy based on the enhanced anti-metastasis activity and reduce side effects. Acta Pharm Sin B. 2020;10:1106–1121. doi: 10.1016/j.apsb.2019.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hachinski V, Einhäupl K, Ganten D, et al. Preventing dementia by preventing stroke: The Berlin Manifesto. Alzheimers Dement. 2019;15:961–984. doi: 10.1016/j.jalz.2019.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wightman EL, Haskell-Ramsay CF, Thompson KG, et al. Dietary nitrate modulates cerebral blood flow parameters and cognitive performance in humans: a double-blind, placebo-controlled, crossover investigation. Physiol Behav. 2015;149:149–158. doi: 10.1016/j.physbeh.2015.05.035. [DOI] [PubMed] [Google Scholar]
- 49.Shi Y, Hu G, Su J, et al. Mesenchymal stem cells: a new strategy for immunosuppression and tissue repair. Cell Res. 2010;20:510–518. doi: 10.1038/cr.2010.44. [DOI] [PubMed] [Google Scholar]
- 50.Wu S, Liu Y, Cui X, et al. Assessment of potential nitrite safety risk of leafy vegetables after domestic cooking. Foods. 2021;10:2953. doi: 10.3390/foods10122953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lundberg JO, Gladwin MT, Weitzberg E. Strategies to increase nitric oxide signalling in cardiovascular disease. Nat Rev Drug Discov. 2015;14:623–641. doi: 10.1038/nrd4623. [DOI] [PubMed] [Google Scholar]
- 52.Hendgen-Cotta UB, Luedike P, Totzeck M, et al. Dietary nitrate supplementation improves revascularization in chronic ischemia. Circulation. 2012;126:1983–1992. doi: 10.1161/CIRCULATIONAHA.112.112912. [DOI] [PubMed] [Google Scholar]
- 53.Lauer T, Heiss C, Balzer J, et al. Age-dependent endothelial dysfunction is associated with failure to increase plasma nitrite in response to exercise. Basic Res Cardiol. 2008;103:291–297. doi: 10.1007/s00395-008-0714-3. [DOI] [PubMed] [Google Scholar]
- 54.Lundberg JO, Govoni M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic Biol Med. 2004;37:395–400. doi: 10.1016/j.freeradbiomed.2004.04.027. [DOI] [PubMed] [Google Scholar]
- 55.Mader MM, Böger R, Appel D, et al. Intrathecal and systemic alterations of L-arginine metabolism in patients after intracerebral hemorrhage. J Cereb Blood Flow Metab. 2021;41:1964–1977. doi: 10.1177/0271678X20983216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hu Y, Xiang J, Su L, Tang X. The regulation of nitric oxide in tumor progression and therapy. J Int Med Res. 2020;48:300060520905985. doi: 10.1177/0300060520905985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Coucha M, Abdelsaid M, Li W, et al. Nox4 contributes to the hypoxia-mediated regulation of actin cytoskeleton in cerebrovascular smooth muscle. Life Sci. 2016;163:46–54. doi: 10.1016/j.lfs.2016.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Li H, Horke S, Förstermann U. Vascular oxidative stress, nitric oxide and atherosclerosis. Atherosclerosis. 2014;237:208–219. doi: 10.1016/j.atherosclerosis.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 59.Huang Z, Huang PL, Ma J, et al. Enlarged infarcts in endothelial nitric oxide synthase knockout mice are attenuated by nitro-L-arginine. J Cereb Blood Flow Metab. 1996;16:981–987. doi: 10.1097/00004647-199609000-00023. [DOI] [PubMed] [Google Scholar]
- 60.Fadel PJ. Nitric oxide and cardiovascular regulation: beyond the endothelium. Hypertension. 2017;69:778–779. doi: 10.1161/HYPERTENSIONAHA.117.08999. [DOI] [PubMed] [Google Scholar]
- 61.Pluta RM, Hansen-Schwartz J, Dreier J, et al. Cerebral vasospasm following subarachnoid hemorrhage: time for a new world of thought. Neurol Res. 2009;31:151–158. doi: 10.1179/174313209X393564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Martin CO, Rymer MM. Hemorrhagic stroke: aneurysmal subarachnoid hemorrhage. Mo Med. 2011;108:124–127. [PMC free article] [PubMed] [Google Scholar]
- 63.Tseng MY, Czosnyka M, Richards H, Pickard JD, Kirkpatrick PJ. Effects of acute treatment with pravastatin on cerebral vasospasm, autoregulation, and delayed ischemic deficits after aneurysmal subarachnoid hemorrhage: a phase II randomized placebo-controlled trial. Stroke. 2005;36:1627–1632. doi: 10.1161/01.STR.0000176743.67564.5d. [DOI] [PubMed] [Google Scholar]
- 64.Pluta RM, Oldfield EH. Analysis of nitric oxide (NO) in cerebral vasospasm after aneursymal bleeding. Rev Recent Clin Trials. 2007;2:59–67. doi: 10.2174/157488707779318062. [DOI] [PubMed] [Google Scholar]
- 65.Pluta RM. Dysfunction of nitric oxide synthases as a cause and therapeutic target in delayed cerebral vasospasm after SAH. Acta Neurochir Suppl. 2008;104:139–147. doi: 10.1007/978-3-211-75718-5_28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pluta RM. Dysfunction of nitric oxide synthases as a cause and therapeutic target in delayed cerebral vasospasm after SAH. Neurol Res. 2006;28:730–737. doi: 10.1179/016164106X152052. [DOI] [PubMed] [Google Scholar]
- 67.Ponomarenko A, Tyrtyshnaia A, Ivashkevich D, Ermolenko E, Dyuizen I, Manzhulo I. Synaptamide modulates astroglial activity in mild traumatic brain injury. Mar Drugs. 2022;20:538. doi: 10.3390/md20080538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hu XD, Yang P, Cao Y, Yang LS. Expression of iNOS and nNOS in acute carbon monoxide poisoning delayed encephalopathy and its correlation with neuronal degeneration and necrosis. Zhonghua Jizhen Yixue Zazhi. 2020;29:1310–1317. [Google Scholar]
- 69.Lee M, Rey K, Besler K, Wang C, Choy J. Immunobiology of nitric oxide and regulation of inducible nitric oxide synthase. Results Probl Cell Differ. 2017;62:181–207. doi: 10.1007/978-3-319-54090-0_8. [DOI] [PubMed] [Google Scholar]
- 70.Saini R, Singh S. Inducible nitric oxide synthase: an asset to neutrophils. J Leukoc Biol. 2019;105:49–61. doi: 10.1002/JLB.4RU0418-161R. [DOI] [PubMed] [Google Scholar]
- 71.Ghasemi M, Fatemi A. Pathologic role of glial nitric oxide in adult and pediatric neuroinflammatory diseases. Neurosci Biobehav Rev. 2014;45:168–182. doi: 10.1016/j.neubiorev.2014.06.002. [DOI] [PubMed] [Google Scholar]
- 72.Cinelli MA, Do HT, Miley GP, Silverman RB. Inducible nitric oxide synthase: Regulation, structure, and inhibition. Med Res Rev. 2020;40:158–189. doi: 10.1002/med.21599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.García-Ortiz A, Serrador JM. Nitric oxide signaling in T cell-mediated immunity. Trends Mol Med. 2018;24:412–427. doi: 10.1016/j.molmed.2018.02.002. [DOI] [PubMed] [Google Scholar]
- 74.Zhu HY, Hong FF, Yang SL. The roles of nitric oxide synthase/nitric oxide pathway in the pathology of vascular dementia and related therapeutic approaches. Int J Mol Sci. 2021;22:4540. doi: 10.3390/ijms22094540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Garry PS, Ezra M, Rowland MJ, Westbrook J, Pattinson KT. The role of the nitric oxide pathway in brain injury and its treatment--from bench to bedside. Exp Neurol. 2015;263:235–243. doi: 10.1016/j.expneurol.2014.10.017. [DOI] [PubMed] [Google Scholar]
- 76.Munshaw S, Bruche S, Redpath AN, et al. Thymosin β4 protects against aortic aneurysm via endocytic regulation of growth factor signaling. J Clin Invest. 2021;131:e127884. doi: 10.1172/JCI127884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Liu Y, Dong J, Song Q, et al. Correlation between cerebral venous oxygen level and cognitive status in patients with Alzheimer’s disease using quantitative susceptibility mapping. Front Neurosci. 2020;14:570848. doi: 10.3389/fnins.2020.570848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zhang J, Liu L, Sun H, et al. Cerebral microbleeds are associated with mild cognitive impairment in patients with hypertension. J Am Heart Assoc. 2018;7:e008453. doi: 10.1161/JAHA.117.008453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sélley E, Kun S, Szijártó IA, et al. Exenatide induces aortic vasodilation increasing hydrogen sulphide, carbon monoxide and nitric oxide production. Cardiovasc Diabetol. 2014;13:69. doi: 10.1186/1475-2840-13-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Li S, Qiu N, Ni A, Hamblin MH, Yin KJ. Role of regulatory non-coding RNAs in traumatic brain injury. Neurochem Int. 2024;172:105643. doi: 10.1016/j.neuint.2023.105643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chen XY, Wan SF, Yao NN, et al. Inhibition of the immunoproteasome LMP2 ameliorates ischemia/hypoxia-induced blood-brain barrier injury through the Wnt/β-catenin signalling pathway. Mil Med Res. 2021;8:62. doi: 10.1186/s40779-021-00356-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Li Y, Ma T, Zhu X, et al. Zinc improves neurological recovery by promoting angiogenesis via the astrocyte-mediated HIF-1α/VEGF signaling pathway in experimental stroke. CNS Neurosci Ther. 2022;28:1790–1799. doi: 10.1111/cns.13918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Do HT, Li H, Chreifi G, Poulos TL, Silverman RB. Optimization of blood-brain barrier permeability with potent and selective human neuronal nitric oxide synthase inhibitors having a 2-aminopyridine scaffold. J Med Chem. 2019;62:2690–2707. doi: 10.1021/acs.jmedchem.8b02032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zhu HY, Hong FF, Yang SL. The roles of nitric oxide synthase/nitric oxide pathway in the pathology of vascular dementia and related therapeutic approaches. Int J Mol Sci. 2021:22. doi: 10.3390/ijms22094540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Jeong YH, Kim TI, Oh YC, Ma JY. Chrysanthemum indicum prevents hydrogen peroxide-induced neurotoxicity by activating the TrkB/Akt signaling pathway in hippocampal neuronal cells. Nutrients. 2021;13:3690. doi: 10.3390/nu13113690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chen Y, Chen Z, Duan J, et al. H(2)O(2)-responsive VEGF/NGF gene co-delivery nano-system achieves stable vascularization in ischemic hindlimbs. J Nanobiotechnology. 2022;20:145. doi: 10.1186/s12951-022-01328-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Xu C, Lu HX, Wang YX, Chen Y, Yang SH, Luo YJ. Association between smoking and the risk of acute mountain sickness: a meta-analysis of observational studies. Mil Med Res. 2016;3:37. doi: 10.1186/s40779-016-0108-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lin Y, Hou C, Wang C, et al. Research progress on digestive disorders following traumatic brain injury. Front Immunol. 2024;15:1524495. doi: 10.3389/fimmu.2024.1524495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Yang X, Xiong T, Li S. Role of long noncoding RNAs in angiogenesis-related cerebrovascular disorders and regenerative medicine: a narrative review. Regen Med Rep. 2024;1:156–171. [Google Scholar]
- 90.Li H, Wang Z. Blood biomarkers for clinical applications in Alzheimer’s disease: A narrative review. NeuroMarkers. 2025;2:100078. [Google Scholar]
- 91.Zhang B, Zhao J, Wang Z, Guo P, Liu A, Du G. Identification of multi-target anti-AD chemical constituents from traditional chinese medicine formulae by integrating virtual screening and in vitro validation. Front Pharmacol. 2021;12:709607. doi: 10.3389/fphar.2021.709607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Zhang J, Li Z, Chandrasekar A, et al. Fast maturation of splenic dendritic cells upon TBI is associated with FLT3/FLT3L signaling. Front Immunol. 2022;13:824459. doi: 10.3389/fimmu.2022.824459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Rosenberg GA. Extracellular matrix inflammation in vascular cognitive impairment and dementia. Clin Sci (Lond) 2017;131:425–437. doi: 10.1042/CS20160604. [DOI] [PubMed] [Google Scholar]
- 94.Wang XX, Zhang B, Xia R, Jia QY. Inflammation, apoptosis and autophagy as critical players in vascular dementia. Eur Rev Med Pharmacol Sci. 2020;24:9601–9614. doi: 10.26355/eurrev_202009_23048. [DOI] [PubMed] [Google Scholar]
- 95.Fröhlich A, Olde Heuvel F, Rehman R, et al. Neuronal nuclear calcium signaling suppression of microglial reactivity is mediated by osteoprotegerin after traumatic brain injury. J Neuroinflammation. 2022;19:279. doi: 10.1186/s12974-022-02634-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lee JW, Nam H, Kim LE, et al. TLR4 (toll-like receptor 4) activation suppresses autophagy through inhibition of FOXO3 and impairs phagocytic capacity of microglia. Autophagy. 2019;15:753–770. doi: 10.1080/15548627.2018.1556946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Iadecola C. The pathobiology of vascular dementia. Neuron. 2013;80:844–866. doi: 10.1016/j.neuron.2013.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Carreira BP, Morte MI, Santos AI, et al. Nitric oxide from inflammatory origin impairs neural stem cell proliferation by inhibiting epidermal growth factor receptor signaling. Front Cell Neurosci. 2014;8:343. doi: 10.3389/fncel.2014.00343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Maksoud MJE, Tellios V, Xiang YY, Lu WY. Nitric oxide signaling inhibits microglia proliferation by activation of protein kinase-G. Nitric Oxide. 2020;94:125–134. doi: 10.1016/j.niox.2019.11.005. [DOI] [PubMed] [Google Scholar]
- 100.Lacza Z, Pankotai E, Busija DW. Mitochondrial nitric oxide synthase: current concepts and controversies. Front Biosci (Landmark Ed) 2009;14:4436–4443. doi: 10.2741/3539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Sticozzi C, Belmonte G, Frosini M, Pessina F. Nitric oxide/cyclic GMP-dependent calcium signalling mediates IL-6- and TNF-α-induced expression of glial fibrillary acid protein. J Mol Neurosci. 2021;71:854–866. doi: 10.1007/s12031-020-01708-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Correia SS, Liu G, Jacobson S, et al. The CNS-penetrant soluble guanylate cyclase stimulator CYR119 attenuates markers of inflammation in the central nervous system. J Neuroinflammation. 2021;18:213. doi: 10.1186/s12974-021-02275-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Gao Y, Xu Y, Yin J. Selenomethionine ameliorates cognitive impairment, decreases hippocampal oxidative stress and attenuates dysbiosis in D-galactose-treated mice. Antioxidants (Basel) 2022;11:111. doi: 10.3390/antiox11010111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ouyang M, Zhang Q, Shu J, et al. Capsaicin ameliorates the loosening of mitochondria-associated endoplasmic reticulum membranes and improves cognitive function in rats with chronic cerebral hypoperfusion. Front Cell Neurosci. 2022;16:822702. doi: 10.3389/fncel.2022.822702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Caballano-Infantes E, Díaz I, Hitos AB, et al. Stemness of human pluripotent cells: hypoxia-like response induced by low nitric oxide. Antioxidants (Basel) 2021;10:1408. doi: 10.3390/antiox10091408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Beltran-Povea A, Caballano-Infantes E, Salguero-Aranda C, et al. Role of nitric oxide in the maintenance of pluripotency and regulation of the hypoxia response in stem cells. World J Stem Cells. 2015;7:605–617. doi: 10.4252/wjsc.v7.i3.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Broustas CG, Duval AJ, Chaudhary KR, Friedman RA, Virk RK, Lieberman HB. Targeting MEK5 impairs nonhomologous end-joining repair and sensitizes prostate cancer to DNA damaging agents. Oncogene. 2020;39:2467–2477. doi: 10.1038/s41388-020-1163-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Chen HS, Chen X, Li WT, Shen JG. Targeting RNS/caveolin-1/MMP signaling cascades to protect against cerebral ischemia-reperfusion injuries: potential application for drug discovery. Acta Pharmacol Sin. 2018;39:669–682. doi: 10.1038/aps.2018.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Feng J, Chen X, Lu S, et al. Naringin attenuates cerebral ischemia-reperfusion injury through inhibiting peroxynitrite-mediated mitophagy activation. Mol Neurobiol. 2018;55:9029–9042. doi: 10.1007/s12035-018-1027-7. [DOI] [PubMed] [Google Scholar]
- 110.Sun L, Yang L, Fu Y, et al. Capacity of HSYA to inhibit nitrotyrosine formation induced by focal ischemic brain injury. Nitric Oxide. 2013;35:144–151. doi: 10.1016/j.niox.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 111.Ito Y, Ohkubo T, Asano Y, et al. Nitric oxide production during cerebral ischemia and reperfusion in eNOS- and nNOS-knockout mice. Curr Neurovasc Res. 2010;7:23–31. doi: 10.2174/156720210790820190. [DOI] [PubMed] [Google Scholar]
- 112.Zhou L, Zhu DY. Neuronal nitric oxide synthase: structure, subcellular localization, regulation, and clinical implications. Nitric Oxide. 2009;20:223–230. doi: 10.1016/j.niox.2009.03.001. [DOI] [PubMed] [Google Scholar]
- 113.Gürsoy-Ozdemir Y, Bolay H, Saribaş O, Dalkara T. Role of endothelial nitric oxide generation and peroxynitrite formation in reperfusion injury after focal cerebral ischemia. Stroke. 2000;31:1974–1980. doi: 10.1161/01.str.31.8.1974. discussion 1981. [DOI] [PubMed] [Google Scholar]
- 114.Sun M, Zhao Y, Gu Y, Xu C. Inhibition of nNOS reduces ischemic cell death through down-regulating calpain and caspase-3 after experimental stroke. Neurochem Int. 2009;54:339–346. doi: 10.1016/j.neuint.2008.12.017. [DOI] [PubMed] [Google Scholar]
- 115.Choi YR, Kim HS, Yoon SJ, et al. Nutritional status and diet style affect cognitive function in alcoholic liver disease. Nutrients. 2021;13:185. doi: 10.3390/nu13010185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Huo A, Wang J, Li Q, et al. Molecular mechanisms underlying microglial sensing and phagocytosis in synaptic pruning. Neural Regen Res. 2024;19:1284–1290. doi: 10.4103/1673-5374.385854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Wang Y, Pan Q, Tian R, et al. Repeated oxytocin prevents central sensitization by regulating synaptic plasticity via oxytocin receptor in a chronic migraine mouse model. J Headache Pain. 2021;22:84. doi: 10.1186/s10194-021-01299-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Li S, Olde Heuvel F, Rehman R, et al. Interleukin-13 and its receptor are synaptic proteins involved in plasticity and neuroprotection. Nat Commun. 2023;14:200. doi: 10.1038/s41467-023-35806-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Frey U, Morris RG. Synaptic tagging and long-term potentiation. Nature. 1997;385:533–536. doi: 10.1038/385533a0. [DOI] [PubMed] [Google Scholar]
- 120.Huang Y, Jiao B, Zhu B, et al. Nitric oxide in the spinal cord is involved in the hyperalgesia induced by tetrahydrobiopterin in chronic restraint stress rats. Front Neurosci. 2021;15:593654. doi: 10.3389/fnins.2021.593654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Noriega-Prieto JA, Maglio LE, Ibáñez-Santana S, Fernández de Sevilla D. Endocannabinoid and nitric oxide-dependent IGF-I-mediated synaptic plasticity at mice barrel cortex. Cells. 2022;11:1641. doi: 10.3390/cells11101641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Cassel JC, Schweizer T, Lazaris A, et al. Cognitive deficits in aged rats correlate with levels of L-arginine, not with nNOS expression or 3,4–DAP-evoked transmitter release in the frontoparietal cortex. Eur Neuropsychopharmacol. 2005;15:163–175. doi: 10.1016/j.euroneuro.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 123.Jing ZH, Wei XM, Wang SH, Chen YF, Liu LX, Qi WX. The synergetic effects of nitric oxide and nicotinic acetylcholine receptor on learning and memory of rats. Sheng Li Xue Bao. 2014;66:307–314. [PubMed] [Google Scholar]
- 124.Shi ZQ, Sunico CR, McKercher SR, et al. S-nitrosylated SHP-2 contributes to NMDA receptor-mediated excitotoxicity in acute ischemic stroke. Proc Natl Acad Sci U S A. 2013;110:3137–3142. doi: 10.1073/pnas.1215501110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Liu H, Li J, Zhao F, Wang H, Qu Y, Mu D. Nitric oxide synthase in hypoxic or ischemic brain injury. Rev Neurosci. 2015;26:105–117. doi: 10.1515/revneuro-2014-0041. [DOI] [PubMed] [Google Scholar]
- 126.Xu XX, Shi RX, Fu Y, et al. Neuronal nitric oxide synthase/reactive oxygen species pathway is involved in apoptosis and pyroptosis in epilepsy. Neural Regen Res. 2023;18:1277–1285. doi: 10.4103/1673-5374.357906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Ding JS, Zhang Y, Wang TY, et al. Therapeutic applications of hydrogen sulfide and novel donors for cerebral ischemic stroke: a narrative review. Med Gas Res. 2023;13:7–9. doi: 10.4103/2045-9912.350863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Zhang X, Chen Z, Xiong Y, Zhou Q, Zhu LQ, Liu D. The emerging role of nitric oxide in the synaptic dysfunction of vascular dementia. Neural Regen Res. 2025;20:402–415. doi: 10.4103/NRR.NRR-D-23-01353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Oliveira C, Benfeito S, Fernandes C, Cagide F, Silva T, Borges F. NO and HNO donors, nitrones, and nitroxides: Past, present, and future. Med Res Rev. 2018;38:1159–1187. doi: 10.1002/med.21461. [DOI] [PubMed] [Google Scholar]
- 130.Xu LH, Fang JP, Huang WG, et al. Marrow graft rejection by repeated transfusions of allogeneic donor spleen cells. Bone Marrow Transplant. 2007;40:691–698. doi: 10.1038/sj.bmt.1705791. [DOI] [PubMed] [Google Scholar]
- 131.Chen X, Shu S, Bayliss DA. HCN1 channel subunits are a molecular substrate for hypnotic actions of ketamine. J Neurosci. 2009;29:600–609. doi: 10.1523/JNEUROSCI.3481-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Schulte B, Schmidt CS, Kuhnigk O, et al. Structural barriers in the context of opiate substitution treatment in Germany--a survey among physicians in primary care. Subst Abuse Treat Prev Policy. 2013;8:26. doi: 10.1186/1747-597X-8-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Dougherty DD, Corse AK, Chou T, et al. Open-label study of duloxetine for the treatment of obsessive-compulsive disorder. Int J Neuropsychopharmacol. 2015;18:pyu062. doi: 10.1093/ijnp/pyu062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Kitamura Y, Araki H, Shibata K, Gomita Y, Tanizaki Y. 5-HT(1A) receptor full agonist, 8–OH-DPAT, exerts antidepressant-like effects in the forced swim test in ACTH-treated rats. Eur J Pharmacol. 2003;481:75–77. doi: 10.1016/j.ejphar.2003.09.008. [DOI] [PubMed] [Google Scholar]
- 135.Zhao XY, Lempke SL, Urbán Arroyo JC, et al. iNOS is necessary for GBP-mediated T. gondii clearance in murine macrophages via vacuole nitration and intravacuolar network collapse. Nat Commun. 2024;15:2698. doi: 10.1038/s41467-024-46790-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Sup SJ, Green BG, Grant SK. 2-Iminobiotin is an inhibitor of nitric oxide synthases. Biochem Biophys Res Commun. 1994;204:962–968. doi: 10.1006/bbrc.1994.2554. [DOI] [PubMed] [Google Scholar]
- 137.Babbedge RC, Bland-Ward PA, Hart SL, Moore PK. Inhibition of rat cerebellar nitric oxide synthase by 7-nitro indazole and related substituted indazoles. Br J Pharmacol. 1993;110:225–228. doi: 10.1111/j.1476-5381.1993.tb13796.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Shi Y, Cao Y, Han X, Xie L, Xiao K. iNOS inhibitor S-methylisothiourea alleviates smoke inhalation-induced acute lung injury by suppressing inflammation and macrophage infiltration. Int Immunopharmacol. 2024;126:111097. doi: 10.1016/j.intimp.2023.111097. [DOI] [PubMed] [Google Scholar]
- 139.Fu X, He S, Wang L, et al. Madecassic acid ameliorates the progression of osteoarthritis: an in vitro and in vivo study. Drug Des Devel Ther. 2022;16:3793–3804. doi: 10.2147/DDDT.S383632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Jin S, Teng X, Xiao L, et al. Hydrogen sulfide ameliorated L-NAME-induced hypertensive heart disease by the Akt/eNOS/NO pathway. Exp Biol Med (Maywood) 2017;242:1831–1841. doi: 10.1177/1535370217732325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Wang Y, Luo J, Guan X, Zhao Y, Sun L. Bacillus cereus cereolysin O induces pyroptosis in an undecapeptide-dependent manner. Cell Death Discov. 2024;10:122. doi: 10.1038/s41420-024-01887-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not available.


