Abstract
Amycolatopsis mediterranei U32 is an industrial strain capable of producing therapeutically useful rifamycin SV. In early days of fermentation studies, nitrate was found to increase the yield of rifamycin along with globally, affecting both carbon and nitrogen metabolism in favor of antibiotic biosynthesis; thus, the nitrate-stimulating effect (NSE) hypothesis was proposed. Although GlnR is likely the master regulator of the pleotropic effect of NSE, the global metabolism affected by NSE has never been systematically examined. In this study, we use mass spectrometry-based metabolomics to quantitatively monitor the metabolomic responses of A. mediterranei U32 to nitrate supplementation. The concentrations of many metabolites involved in central carbon metabolism, including glucose 6-phosphate, glucose 1-phosphate, UDP-glucose, and acetyl-coenzyme A, decrease significantly after the addition of 80 mM potassium nitrate to the medium. We find that the rifamycin SV production yield could be increased by the addition of glucose during the logarithmic growth phase. Moreover, at multiple time points during glucose supplementation in the mid- and late-exponential phases, the yield of rifamycin SV further increases, reaching 354.3%. Quantitative real-time PCR assays of the key genes corresponding to the synthesis of the rifamycin SV precursor combined with data from metabolomics analysis confirm that carbon source deficiency is compensated for after glucose supplementation and that the expression of genes involved in the pathway of 3-amino-5-hydroxybenzoic acid synthesis by UDP-glucose and glutamine is significantly increased. This preliminary exploration of dynamic metabolomic profiles has the potential to increase our understanding of the NSE.
Keywords: Amycolatopsis mediterranei, glucose supplementation, metabolome, nitrate-stimulating effect, rifamycin SV production
Introduction
Rifamycin is an ansamycin antibiotic that effectively inhibits bacterial RNA polymerase via specific binding to its β subunit [1], encoded by rpoB [2], so it has been demonstrated to be a broad-spectrum antibiotic against pathogenic bacterial infections [ 3, 4] and is particularly effective for tuberculosis chemotherapy [5]. A significant increase in rifamycin SV productivity in the industrial strain Amycolatopsis mediterranei U32 via the addition of nitrate into its fermentation medium was reported a few decades ago, and the increase in the specific yield of rifamycin was found to be directly correlated in a dose-dependent manner with the increase in the specific activity of glutamine synthase (GS), which produces glutamine to provide the nitrogen source for the synthesis of the key starter unit of rifamycin, 3-amino-5-hydroxybenzoic acid (AHBA) [ 6, 7] . Thus, the “nitrate-stimulating effect (NSE)” was coined [ 8, 9] and has been demonstrated in the industrial production of other antibiotics, such as rifamycin B [10], lincomycin [ 11– 13] and azalomycin B [14]. Further physiological and biochemical studies also indicated that NSE is likely pleotropic, affecting both nitrogen and carbon primary metabolism toward the production of antibiotics [15].
Notably, the atypical OmpR/PhoB subfamily response regulator GlnR was characterized as an essential regulatory protein for the transcription of the GS encoding the glnA gene in streptomycetes first [16] and then in many other actinomycetes, including strains of A. mediterranei [ 17, 18] and mycobacteria [19]. Research has shown that GlnR is a global transcription factor that is bound to the promoter regions of many metabolic genes [20], globally regulating their expression [21] not only for nitrogen metabolism [ 22– 24] but also for other metabolic processes, such as carbon [ 25– 27] , phosphate [ 28, 29] and even antibiotic biosynthesis [ 18, 30] .
A subsequent genomic study of A. mediterranei U32 revealed its carbon and nitrogen metabolic pathways as well as biosynthesis-related gene clusters, particularly for rifamycin, the rif cluster [31]. The following transcriptome study revealed their expression properties, particularly those related to the biosynthesis of secondary metabolites [32], and thus, this study well integrated the knowledge of NSE to facilitate the acquisition of the molecular mechanism of the GlnR-mediated global regulatory function. Nitrate, which is mediated by functional GlnR, induces the expression of the nasACKBDEF operon [33], and this additional nitrogenous substrate likely maintains the healthy growth status of the cells during fermentation [33]. Moreover, nitrate may also function as a signal to activate the transcription factor GlnR and further affect global metabolism, largely affecting the expression of the corresponding enzymes [7]. In particular, nitrate addition significantly increases the transcription of key genes of the rif cluster and bidirectionally regulates the expression of genes encoding primary metabolic enzymes, i. e., activating the supply but repressing the diverse utilization of the precursors for antibiotic biosynthesis [ 8, 32, 34] . Notably, although the molecular mechanisms of GlnR-mediated global regulation of different promoter regions have been studied in detail [ 7, 21, 32– 34] , most upstream regulatory signals and their functional transduction have yet to be resolved.
Metabolites not only are the products of catalysis and the substrates of assimilation/synthesis but also serve as a veritable reflection of the true physiological states of organisms, with alterations in metabolic profiles directly influencing phenotypic changes [ 35– 37] . The detection and quantification of metabolites can provide insights into the functional activity of cellular metabolic pathways [38]. Integrating transcriptomic and metabolomic analyses enables a comprehensive exploration of the intricate association between gene expression and metabolic regulation [ 39, 40] . In actinomycetes, the genus Saccharopolyspora, which produces antibiotics such as erythromycin and spinosyn A [41], was studied via comparative metabolomics analysis to understand metabolite variation during fermentation [42] and the metabolomic characteristics of high-yield strains [43] and to decipher metabolic pathway differences [44]. Moreover, metabolomics and metabolic flux analysis were used to investigate cell growth, flux distribution and antibiotic production in Streptomyces coelicolor [ 45– 50] . These studies strongly indicate that metabolomic analysis can be applied to gain insights into the metabolic conditions of A. mediterranei strains, including U32, to further elucidate the mechanism of NSE.
In this study, we report a metabolomic analysis of A. mediterranei U32 cultivated in Bennet medium to elucidate the changes in metabolites in response to nitrate. The quantitative analysis revealed that the concentrations of the majority of metabolic intermediates in the Embden-Meyerhof pathway (EMP) and pentose phosphate pathway (PPP) considerably decreased over a short period of batch culture. Optimization of the initial medium composition [51] and feeding strategy [52] are pivotal steps in the commercial production of antibiotics. Through iterative glucose supplementation during the middle and log phases, we observed a promotion of cell growth and a subsequent increase in the rifamycin SV yield. Combination with transcriptional results, the data from a rifamycin SV fermentation factory confirmed that multiple time points of glucose supplementation could increase the yield of rifamycin SV, suggesting a dynamic equilibrium between a rich nitrogen source and a sustainable carbon supply.
Materials and Methods
Strains, media, and culture conditions
A. mediterranei U32 was grown aerobically in Bennet medium (0.1% yeast extract, 1.0% glycerol, 1.0% glucose, 0.2% tryptone, 0.1% beef extract, w/v, pH 7.0) at 30°C [ 8, 53] . The solid plates needed 1.5% (w/v) agar, and the liquid media needed 4% (w/v) zirconia beads (d = 2.5 mm). After 40 h of cultivation, 5% of the cells were inoculated into fresh liquid Bennet medium or Bennet medium supplemented with 80 mM potassium nitrate [30]. The cultures were grown at 30°C in 250-mL shake flasks with 75 mL of media. Samples were collected at the indicated time points.
Measurements of cell growth, glucose concentration, and rifamycin SV production
After growing on Bennet plates for 4 days, A. mediterranei U32 cells were collected and transferred to liquid Bennet medium. The mixture was incubated in a shake flask for approximately 40 h and then incubated with U32 in fresh liquid Bennet medium supplemented with 0 or 80 mM KNO 3, and the final optical density at 600 nm (OD 600) was 0.2. The growth of A. mediterranei U32 cells was determined by measuring the OD 600 values with a NanoDrop 2000C (Thermo Fisher Scientific, Waltham, USA) [54]. The glucose concentration was determined with a glucose assay kit with O-toluidine (Beyotime, Shanghai, China) following the manufacturer’s instructions.
Rifamycin SV was measured via the spectrophotometric method as previously described [ 55, 56] . One milliliter of suspension was taken from the culture broth at 12 h intervals, and the samples were then centrifuged at 8050 g for 5 min at 4°C to collect the supernatant. A total of 20 μL of the supernatant was diluted in 180 μL of acetate buffer containing 0.1% NaNO 2 or 0.1% vitamin C. The optical density was then measured at 447 nm, with the NaNO 2 group used as a blank control and 95% pure rifamycin SV (Sigma-Aldrich, St Louis, USA) used as a standard. Finally, the yield of rifamycin SV was calculated on the basis of the standard curve [30].
Metabolomics
A. mediterranei U32 cells were collected by fast filtration with a sterile nylon membrane filter (0.45-μm pore size) that was mounted on a vacuum device opened to a moderate level. The metabolism was quenched quickly by immersing the filter in liquid nitrogen for 30 s [57]. Metabolites were extracted with 80:20 methanol/water at –80°C [58]. After incubation at –20°C for 20 min, the cells were disrupted in a tissue homogenizer (Bertin, Rockville, USA) for 2 min at 70 Hz. The samples were subsequently centrifuged for 15 min (4°C, 8050 g), after which the supernatants were collected. The pellets were subjected to a second extraction, and the extracts were combined [59].
The cell extracts were analyzed via ultrahigh–performance liquid chromatograph (UHPLC; Acquity; Waters, Milford, USA) coupled to a Q Exactive hybrid quadrupole-orbitrap mass spectrometer (Thermo Fisher Scientific) [60]. The injection volume was 10 μL. Mobile phase A was 20 mM ammonium acetate adjusted to pH 9.2 with ammonium hydroxide, and mobile phase B was acetonitrile. Metabolites were separated with a Luna NH2 column (100 mm ×2 mm, 3-μm particle size; Phenomenex, Torrance, USA) as described previously [61]. The mass spectrometer with a heated electrospray ionization source was operated in positive and negative modes. The key parameters were as follows: ion spray voltage, +3.5 kV/–3.0 kV; capillary temperature, 320°C; probe heater temperature, 350°C; and sheath and auxiliary gas, 35 and 10 arbitrary units, respectively. Mass spectra were acquired via full scan over 70–1000 m/ z at 70,000 resolution. MS/MS spectra were acquired with 30-eV collision energy.
LC-MS data processing and ion annotation based on accurate mass data were performed in Xcalibur 4.0 (Thermo Fisher Scientific) and Compound Discoverer 3.3 (Thermo Fisher Scientific). The identities of the compounds were verified by mass and retention time matching authenticated standards. The relative concentrations of the metabolites were determined via a calibration curve generated with varying concentrations of the chemical standard spiked into the U32 cell extract. Hierarchical clustering analysis of changes in metabolite concentrations was performed via the R package pheatmap (version 1.0.12) [57].
RNA extraction and quantitative real-time PCR (qRT-PCR)
The cells were harvested at different time points with or without glucose supplementation (48, 60, 72, 84, 96, and 108 h) via centrifugation at 8050 g for 5 min at 4°C and then quickly frozen in liquid nitrogen for total RNA isolation. Total RNA was extracted via the ZR Fungal/Bacterial RNA MiniPrep TM (Zymo Research, Orange County, USA) following the manufacturers’ instructions. Reverse transcription was performed with a random hexamer primer using 1 μg of total RNA as the template in a volume of 20 μL via the PrimeScript 1 st Strand cDNA synthesis kit (TaKaRa, Beijing, China), and qRT-PCR was carried out with SYBR Premix Ex Taq II (Tli RNaseH Plus; TaKaRa) in a Step-One Plus real-time PCR system (Applied Biosystems, Foster City, USA). The rpoB gene of A. mediterranei was used as a constitutive reference to normalize gene expression [ 30, 62] . The relative expression levels of the target genes were calculated via the 2 ‒∆∆CT method. At least three independent biological samples were tested in this experiment. All the primers used here are listed in Supplementary Table S1.
Fermentation
Inoculum preparation and fermentation of A. mediterranei U32 were carried out aerobically in seed culture media (2.0% glucose, 0.5% soybean meal, 1.0% peptone, 0.2% yeast extract, 0.2% CaCO 3, and 0.2% NaCl, pH 7.0) and fermentation culture media (10.0% glucose, 0.55% fish meal, 0.75% soybean meal, 0.7% peptone, 0.5% CaCO 3, 0.8% KNO 3, and 0.02% K 2HPO 4, pH 7.0), respectively.
After being activated on Bennet plates for 4 days, A. mediterranei U32 was transferred into 100 mL of liquid seed culture medium and incubated aerobically at 30°C for approximately 48 h. Seed cells were transferred into fermentation culture medium (250 mL) for fermentation in a 1-L conical flask and cultured in a thermostatic shaker at 30°C and 220 rpm.
Statistical analysis
Data are presented as the mean ± SD of n independent experiments, and the number of replicates is provided in the corresponding figure caption. P values were calculated via one-way analysis of variance (ANOVA).
Results
Metabolomic profiling of A. mediterranei revealed an imbalance in carbon and nitrogen metabolism in response to NSE
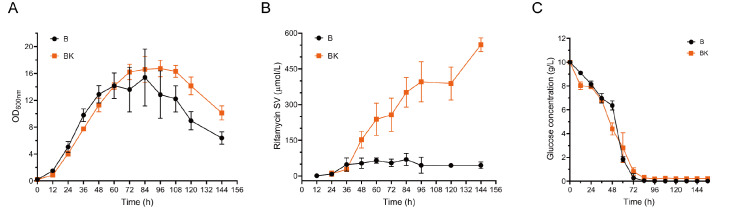
To obtain a comprehensive view of the metabolic responses to NSE, we grew A. mediterranei U32 in Bennet medium with or without 80 mM potassium nitrate (hereafter denoted B and BK media, respectively) and measured the intracellular metabolome under different conditions via liquid chromatography-mass spectrometry (LC-MS). On the basis of cell growth and rifamycin SV production ( Figure 1A,B), three time points (24, 48, and 72 h) corresponding to the exponential growth phase were chosen for metabolomic profiling. We quantified 136 out of the 242 metabolites identified, which are involved in the metabolic pathways of central carbon metabolism, lipid metabolism, nucleotide metabolism, amino acid metabolism, metabolism of cofactors and vitamins, and biosynthesis of secondary metabolites ( Supplementary Figure S1A and Supplementary Table S2).
Figure 1 .
Cell growth, rifamycin SV production and glucose concentration in B and BK
(A) Cell growth, (B) rifamycin SV production, (C) glucose concentration in B and BK; Black line is nitrate-free Bennet medium, red line is Bennet medium with 80 mM KNO3. Error bars represent standard deviations of three biological replicates.
We found that the intracellular concentrations of the metabolites involved in the tricarboxylic acid (TCA) cycle, amino acid metabolism, and nucleotide metabolism were altered ( Figure 2 and Supplementary Figure S1B). Specifically, nitrate addition led to the accumulation of several amino acids, including glutamate, glutamine, and aspartate, and the depletion of alanine. This result is consistent with previous findings that A. mediterranei U32 primarily uses the glutamine synthetase (GS)/glutamine oxoglutarate aminotransferase (GOGAT) pathway for nitrate assimilation and represses the alanine dehydrogenase pathway in the presence of nitrate [24]. Interestingly, we observed a decrease in the concentrations of intermediate metabolites of the EMP and PPP under nitrate supplementation conditions, which included glucose 1-phosphate (G1P), glucose 6-phosphate (G6P), fructose 6-phosphate (F6P), sedoheptulose 7-phosphate (S7P), UDP-glucose (UDPG), and ribose 5-phosphate (R5P) ( Figure 2). The concentration of acetyl-coenzyme A (acetyl-CoA) was also reduced by nitrate supplementation ( Figure 2). The depletion of these carbon metabolites could be due to the activation of the biosynthesis of amino acids and other nitrogenous metabolites. However, as UDPG and acetyl-CoA are precursors for the biosynthesis of rifamycin SV [ 63– 66] , their depletion may impair antibiotic production. Thus, NSE likely resulted in an imbalance in carbon and nitrogen metabolism in A. mediterranei U32. Our results prompt the intriguing question of whether the ability of NSE to cause carbon deficiency could be limited to rifamycin SV production.
Figure 2 .
NSE results in divergent concentration changes of metabolites involved in carbon and nitrogen metabolism
Data shown are the ratio of metabolite concentrations in A.mediterranei U32 grown at 48 h in BK medium versus B medium. Data shown are the means of six independent experiments.
Glucose supplementation increases rifamycin SV production
Considering that the concentrations of intermediate metabolites involved in EMP and PPP in BK were much lower after 48 h, glucose consumption in both B and BK media was monitored by detecting the glucose concentration at various time points after 36 h of incubation ( Figure 1C). The glucose concentration decreased rapidly after 36 h in the BK medium and after 48 h in the B medium and decreased to below 1 g/L under both conditions by 72 h. BK produced significantly more rifamycin SV ( Figure 1B), and the bacteria were carbon deficient in the late-log phase, implying that the supplementation of extra carbon sources in the medium may further increase the synthesis of secondary metabolites.
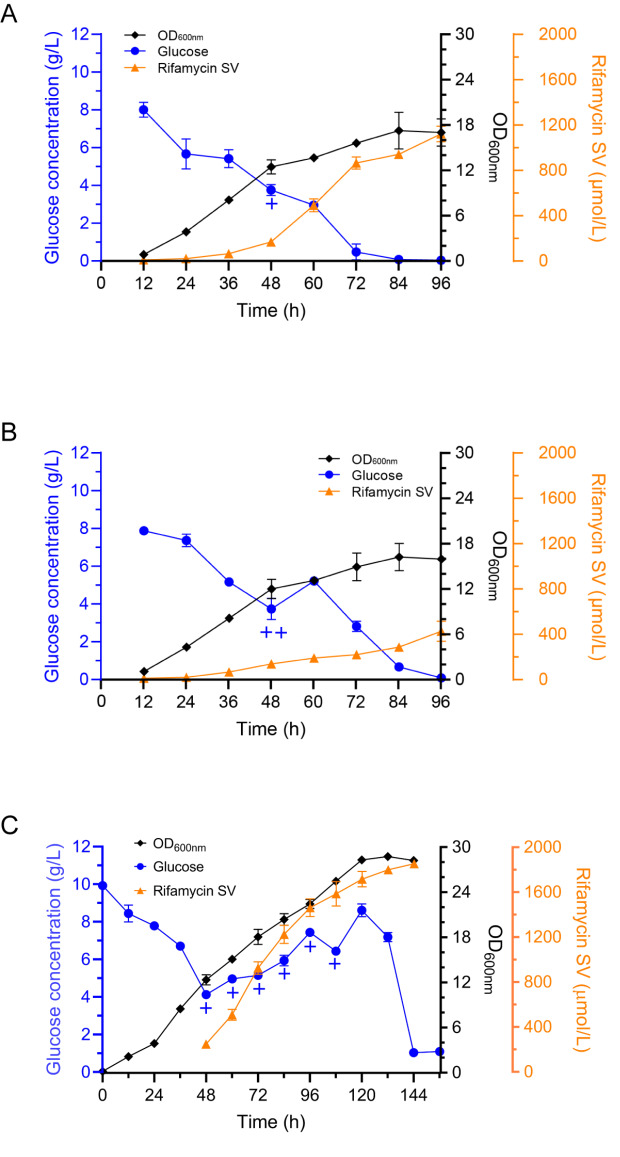
Next, we used both one- and multiple-time point glucose supplementation to investigate the effects on bacterial growth and the synthesis of rifamycin SV. First, we carried out one-time glucose supplementation of 1 mL or 2 mL of 1 M glucose solution at 12, 24, 36, 48, 60 and 72 h and measured the cell growth, glucose concentration and rifamycin yield ( Figure 3A,B and Supplementary Figure S2). For one-time glucose supplementation of 1 mL ( i. e.,0.18 g of glucose), the cell growth showed very little change; only at 72 h could a slight effect be detected (the logarithmic phase was extended to 84 h) ( Supplementary Figure S2I). However, rifamycin SV production was significantly increased at 48, 60 and 72 h ( Figure 3A and Supplementary Figure S2G,I) and reached 1111.1 μM at 96 h when supplemented with 1 mL of glucose at 48 h, which was 280.72% higher than that under the BK condition without glucose supplementation ( Figure 3A). For one-time glucose supplementation of 2 mL, we only observed that the addition at 72 h prolonged the logarithmic period of the bacterium to 108 h ( Supplementary Figure S2J), but the addition at 12 or 24 h accelerated cellular death ( Supplementary Figure S2B,D). However, addition at other time points had a minimal effect on bacterial growth. The yield of rifamycin was essentially maintained between 300 and 400 μM, similar to that of BK without glucose supplementation.
Figure 3 .
Glucose concentration, cell growth and rifamycin SV production of U32 in BK after different glucose supplementation strategies
(A) Adding 1 mL glucose solution once at 48 h. (B) Adding 2 mL glucose solution once at 48 h. (C) Repeatedly adding 1 mL glucose solution at 48, 60, 72, 84, 96 and 108 h. ‘+’ means supplying 1 mL glucose solution, ‘++’ means supplying 2 mL glucose solution. Data are shown as the mean ± SD (n = 3).
Subsequently, at multiple time points, 1 mL of 1 M glucose solution was added to the culture at 48, 60, 72, 84, 96 and 108 h, and we monitored the cell growth, glucose concentration and rifamycin SV yield ( Figure 3C). As expected, accompanying sustained glucose feeding, secondary metabolite synthesis was encouraged, and we noticed a great improvement in cellular growth, with a very long logarithmic phase lasting for 120 h. The rifamycin SV yield was further elevated and reached 1402.5 μM at 96 h, whereas it reached 395.8 μM under the BK condition without sugar supplementation ( Figure 1B). The production of rifamycin SV continuously increased with increasing addition of sugar and reached a yield of 1791.7 μM at 144 h.
In addition, we conducted fermentation with the same glucose supplementation strategy in B medium to illustrate the effect of glucose supplementation itself ( Supplementary Figure S3). Neither cell growth nor rifamycin production remained virtually unchanged. The premise that glucose supplementation increases the yield of rifamycin may be that NSE plays a role.
These results clearly demonstrate that the biosynthetic productivity of secondary metabolites is closely related to the nutrient state of the bacterial fermentation medium and that maintaining a balanced growth state of the bacterium facilitates secondary metabolism.
Dynamic coordination of nitrogen and carbon sources revealed the mechanism of glucose supplementation
To decipher the mechanism of increased rifamycin production induced by glucose supplementation, we employed qRT-PCR to analyze the transcription of genes related to enzymes involved in central carbon metabolism as well as the precursor supply and biosynthesis of rifamycin. In accordance with previous RNA-seq data [32], we chose 21 genes, including 10 genes involved in carbon metabolism ( talA, pgm, nagE, icd, pgi, galU, korA, eno, prsA and sucB), 4 genes involved in nitrogen metabolism ( glnR, glnA, nasA and ald), 3 genes involved in lipid metabolism ( prpE, mcm and AMED_7921), and 4 genes involved in rifamycin SV biosynthesis ( rifL, rifA, rifK and luxR).
As mentioned above, in the presence of NSE, the logarithmic phase of U32 could be prolonged when glucose was also added to the culture medium, leading to the elimination of carbon deficiency ( Figure 3C). Consistently, the transcription of genes related to the EMP and PPP, including pgi, talA, gpm, and korA, was markedly increased by the addition of glucose ( Figure 4A). These results indicate that sugar supplementation enhances the glycolytic pathway in response to bumper carbon source, which may further enhance the TCA cycle to provide more precursors, energy and reducing power for rifamycin production.
Figure 4 .
The transcription of key genes involved in carbohydrate metabolism and rifamycin SV biosynthesis after multiple-time point glucose supplementation
(A) The expressions of genes involved in carbohydrate metabolism. (B) The expressions of genes involved in rifamycin SV biosynthesis. The results showed transcription at 12 h and 24 h with or without glucose supplementation, ‘+’ means adding glucose, the time zero point of sugar addition was used for comparison. The legend shows the time after the first sugar supplementation (48 h), rpoB is used as the reference gene. Error bars represent standard deviations of three biological replicates.
The transcription of genes related to nitrogen assimilation in U32, such as nasA, glnR and glnA, is activated by NSE, while the addition of glucose did not markedly alter the transcription levels of these genes ( Supplementary Figure S4A), which therefore suggested that extracellular carbon availability plays a less important role in nitrogen metabolism in U32.
For rifamycin biosynthesis, ( S)-methylmalonyl-CoA and malonyl-CoA are the extender units. AHBA is the starter unit and is synthesized by a series of enzymes encoded by rifGHIJKLMN via the use of UDPG and glutamine as substrates [64]. We selected rifL and rifK to examine their transcript levels, and the qRT-PCR results revealed that the transcription of both rifL and rifK was greatly increased compared with that observed prior to glucose replenishment ( Figure 4B). In A. mediterranei U32, there are 3 pathways for the biosynthesis of ( S)-methylmalonyl-CoA (the C3 extender unit), i. e., the methylmalonyl-CoA carboxyltransferase (Mct) pathway, the propionyl-CoA carboxylase (Pcc) pathway and the methylmalonyl-CoA mutase (Mcm)-methylmalonyl-CoA epimerase (Mce) pathway, among which the Mcm-Mce pathway is the main pathway [ 34, 67] . The C2 extender unit, malonyl-CoA, is converted from acetyl-CoA by acetyl-CoA carboxylase (Acc) and is a common precursor of fatty acids [ 8, 32] . AMED_2498, the most important of the four mcm genes, consistently displayed decreased expression as a result of NSE, leading to increased accumulation of ( S)-methylmalonyl-CoA ( Figure 5). PrpE (encoding propionyl coenzyme A synthase), mcm, and AMED_7921 (encoding acetyl-CoA carboxylase) were consistently under expressed ( Supplementary Figure S4B). These results are in line with our earlier research on NSE, which showed that nitrate repressed intracellular lipid biosynthesis [ 8, 32] . More importantly, these results highlight that AHBA is the key to being affected by carbon sources for rifamycin precursor biosynthesis. In general, glucose supplementation increases the transcription of genes involved in the EMP and PPP pathways and AHBA synthesis genes, thus increasing rifamycin SV production.
Figure 5 .
Comparison of the expressions of genes and dynamic metabolome after multiple-time point glucose supplementation
Transcription data shown are –ΔΔCt at 60 h (12 h after glucose supplementation); 48 h without glucose supplementation is used as the control group, and rpoB is used as the reference gene. Metabolome data shown are the ratio of both samples above. The solid arrow is a one-step reaction, the dotted arrow is a multi-step reaction, the green arrow is downregulation, and the red arrow is upregulation; Red font with red box means the ratio is greater than 2, pink font with pink box means the ratio is 1.5-2, and black font indicates little change. Data are shown as the mean ± SD (n = 6 independent experiments).
Moreover, we observed that the quantities of metabolites linked to the EMP and PPP either increased or remained unchanged in response to glucose supplementation ( Figure 5). This result indicates that sugar supplementation compensates for the earlier deficiency in carbon sources and thus further enhances rifamycin production ( Figure 5), suggesting that the molecular mechanism of glucose supplementation restored the metabolic balance via the dynamic coordination of nitrogen and carbon sources in response to extracellular nutrients.
Discussion
Noticed and characterized first in A. mediterranei U32 and later demonstrated in a few antibiotic fermentations of actinomycetes, the general rule of NSE was primarily established on the basis of semiquantitative physiological and enzymatic/biochemical analyses. Although studies have revealed that the GlnR regulon likely underscores the pleotropic effect of NSE, regardless of the unclear corresponding signal transduction mechanism, the global dynamics of metabolic homeostasis affected by NSE have never been examined directly. Therefore, this metabolomic study of NSE in A. mediterranei U32 laboratory culture revealed that the addition of nitrate to rich media not only significantly altered both nucleotide and amino acid metabolism, favoring secondary metabolism, as expected ( Supplementary Figure S1A) but also severely reduced the EMP- and PPP-mediated carbohydrate intermediates and energy supply ( Supplementary Figure S1B), resulting in a carbon deficiency physiology that retards the growth of cells and antibiotic production ( Figure 1A,B).
However, if glucose was supplied as a supplemental carbon source during the logarithmic growth phase to rebalance carbon and nitrogen metabolism, the yield of rifamycin SV significantly increased. Transcriptional data revealed that, after resupplementation with glucose, there was a considerable increase in the expression of genes associated with the synthesis of the rifamycin precursor. Combined with the metabolome data following sugar supplementation, we hypothesized that the mechanism of increased rifamycin SV production is that glucose supplementation improves the pathway by which UDPG synthesizes AHBA with glutamine. The assimilation of nitrate into central nitrogen metabolism is beneficial for the generation of the key nitrogenous precursor AHBA of rifamycin SV [ 68, 69] , the synthesis of which requires UDPG and glutamine [64]. Thus, glucose supplementation may directly increase the concentration of UDPG on the one hand and increase the transcription level of EMP in contrast to that in the deficient state; on the other hand, glucose supplementation may restore the acetyl-CoA supply ( Figures 2 and Figure 5). These acetyl-CoAs enter the TCA cycle to participate in the synthesis of glutamine by α-ketoglutarate. Both key precursors of AHBA have strong elevations.
Through multiple-time point glucose supplementation in the mid- and late-exponential phases, the yield of rifamycin SV subsequently further increased to 354.3%. According to Hesham A. El-Enshasy’s research, the use of glucose as a monofeed substrate for A. mediterranei can lead to an increase in the production of rifamycin SV [70]. We analyzed the rifamycin SV yield and reducing sugar consumption data from a rifamycin SV fermentation factory and transformed them into a real-time reducing sugar consumption curve ( Supplementary Figure S5). Multiple time points of glucose supplementation truly increased rifamycin SV production in the presence of nitrate. Thus, this research offers a primary explanation for the standard operation of sugar addition during industrial fermentation ( Figure 5).
In summary, this study, by performing a metabolomic analysis of A. mediterranei U32 in laboratory culture, provides a comprehensive and in-depth view of NSE in combination with data on its transcription, biochemistry and physiology, which were previously accumulated and are currently available. We further characterized a mechanism for high rifamycin SV production in laboratory batch cultures under NSE conditions by utilizing glucose as a supplemental carbon source under conditions of carbon deficiency. A new equilibrium for high rifamycin SV production was reached by adding one- and multiple-time point glucose supplementation so that homeostasis between an enriched nitrogen source and a sustainable carbon supply was established to promote both cell growth and antibiotic production.
Supporting information
Supplementary Data
Supplementary data is available at Acta Biochimica et Biophysica Sinica online.
Supplementary Data
COMPETING INTERESTS
The authors declare that they have no conflict of interest.
Supplementary data is available at Acta Biochimica et Biophysica Sinica online.
Funding Statement
This work was supported by the grants from the National Natural Science Foundation of China (Nos. 31921006 and 92051106) and the National Key R&D Program of China (No. 2018YFA0903700).
References
- 1.Rabussay D, Zillig W. A rifampicin resistent rna‐polymerase from E. coli altered in the β‐subunit . FEBS Lett. . 1969;5:104–106. doi: 10.1016/0014-5793(69)80305-4. [DOI] [PubMed] [Google Scholar]
- 2.Campbell EA, Korzheva N, Mustaev A, Murakami K, Nair S, Goldfarb A, Darst SA. Structural mechanism for rifampicin inhibition of bacterial RNA polymerase. Cell. . 2001;104:901–912. doi: 10.1016/S0092-8674(01)00286-0. [DOI] [PubMed] [Google Scholar]
- 3.Murali Krishna PS, Venkateswarlu G, Venkateswar Rao L. Effect of uracil on rifamycin SV production by Amycolatopsis mediterranei MV35R . Lett Appl Microbiol. . 2000;31:73–76. doi: 10.1046/j.1472-765x.2000.00769.x. [DOI] [PubMed] [Google Scholar]
- 4.Riva S, Silvestri LG. Rifamycins: a general view. Annu Rev Microbiol. . 1972;26:199–224. doi: 10.1146/annurev.mi.26.100172.001215. [DOI] [PubMed] [Google Scholar]
- 5.Corpe RF, Sanchez ES. Rifampin in initial treatment of advanced pulmonary tuberculosis. Chest. . 1972;61:564–567. doi: 10.1378/chest.61.6_Supplement.564. [DOI] [PubMed] [Google Scholar]
- 6.Floss HG, Yu TW. Rifamycinmode of action, resistance, and biosynthesis. Chem Rev. . 2005;105:621–632. doi: 10.1021/cr030112j. [DOI] [PubMed] [Google Scholar]
- 7.Chen YM, Gu WL, Wang W, Jiao RS. Study on the metabolic regulation of biosynthesis of rifamycin by Nocardia mediterranei II. The regulatory effect of nitrate on the metabolic pathway of Nocardia mediterranei. Acta Phytophysiologia Sinica 1980: 291–298
- 8.Jiao RS, Chen YM, Wu MG, Gu WL. Study on the metabolic regulation of biosynthesis of rifamycin by Nocardia mediterranei I. The stimulative effect of nitrate on biosynthesis of tifamycin SV by Nocardia mediterranei. Acta Phytophysiologia Sinica 1979: 395–402
- 9.Jiao RS, Liu CJ, Jin ZK, Zhang XC, Ni LY, Lu ZM. The route of incorporation of nitrogen atom into rifamycin during its biosynthesis. Scientia Sinica Series B. 1984, 27: 380–390 . [PubMed]
- 10.O.m. ET, Salama AA, Hussein MM, El-Sedawy HF. Optimization of industrial production of rifamycin B by Amycolatopsis mediterranei. I. The role of colony morphology and nitrogen sources in productivity . Afr J Biotechnol. . 2004;3:266–272. doi: 10.5897/AJB2004.000-2049. [DOI] [Google Scholar]
- 11.Jin Z, Jiao R. Stimulative effects of nitrate and magnesium salts on biosynthesis of lincomycin by Streptomyces lincolnensis. Chinese biochemical journal 1997, 13: 709–715
- 12.Meng S, Wu H, Wang L, Zhang B, Bai L. Enhancement of antibiotic productions by engineered nitrate utilization in actinomycetes. Appl Microbiol Biotechnol. . 2017;101:5341–5352. doi: 10.1007/s00253-017-8292-7. [DOI] [PubMed] [Google Scholar]
- 13.Li GL, Chiao JS. Nitrate assimilation of Amycolatopsis mediterranei U32 and some properties of its nitrate reductase. Acta Microbiol Sin 1995, 35: 141–148
- 14.Jiang S, Huang WY. Improvement of fermentation conditions for azalomycin B produced by streptomyes hygroscopicus NND-52-C. Chinese Journal of Bioprocess Engineering 2004, 2: 53–57
- 15.Yang YL, Jiang WH, Chiao J, Zhao G. Regulation of rifamycin SV production and glutamine synthetase expression in Amycolatopsis mediterranei U-32 . Actinomycetologica. . 1998;12:141–147. doi: 10.3209/saj.12_141. [DOI] [Google Scholar]
- 16.Wray Jr LV, Atkinson MR, Fisher SH. Identification and cloning of the glnR locus, which is required for transcription of the glnA gene in Streptomyces coelicolor A3(2) . J Bacteriol. . 1991;173:7351–7360. doi: 10.1128/jb.173.22.7351-7360.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yu H, Peng WT, Liu Y, Wu T, Yao YF, Cui MX, Jiang WH, et al. Identification and characterization of glnA promoter and its corresponding Trans-regulatory protein GlnR in the rifamycin SV producing Actinomycete, Amycolatopsis mediterranei U32 . Acta Biochim Biophys Sin. . 2006;38:831–843. doi: 10.1111/j.1745-7270.2006.00238.x. [DOI] [PubMed] [Google Scholar]
- 18.Wang Y, Wang JZ, Shao ZH, Yuan H, Lu YH, Jiang WH, Zhao GP, et al. Three of four GlnR binding sites are essential for GlnR-mediated activation of transcription of the Amycolatopsis mediterranei nas operon . J Bacteriol. . 2013;195:2595–2602. doi: 10.1128/JB.00182-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.He J, Kang X, Wu J, Shao Z, Zhang Z, Wu Y, Yuan H, et al. Transcriptional self-regulation of the master nitrogen regulator GlnR in mycobacteria. J Bacteriol. . 2023;205:e0047922. doi: 10.1128/jb.00479-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tiffert Y, Supra P, Wurm R, Wohlleben W, Wagner R, Reuther J. The Streptomyces coelicolor GlnR regulon: identification of new GlnR targets and evidence for a central role of GlnR in nitrogen metabolism in actinomycetes . Mol Microbiol. . 2008;67:861–880. doi: 10.1111/j.1365-2958.2007.06092.x. [DOI] [PubMed] [Google Scholar]
- 21.Shi J, Feng Z, Xu J, Li F, Zhang Y, Wen A, Wang F, et al. Structural insights into the transcription activation mechanism of the global regulator GlnR from actinobacteria. Proc Natl Acad Sci USA. . 2023;120:e2300282120. doi: 10.1073/pnas.2300282120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang J, Zhao GP. GlnR positively regulates nasA transcription in Streptomyces coelicolor. Biochem Biophys Res Commun. . 2009;386:77–81. doi: 10.1016/j.bbrc.2009.05.147. [DOI] [PubMed] [Google Scholar]
- 23.Wang Y, Cen XF, Zhao GP, Wang J. Characterization of a new GlnR binding box in the promoter of amtB in Streptomyces coelicolor inferred a PhoP/GlnR competitive binding mechanism for transcriptional regulation of amtB . J Bacteriol. . 2012;194:5237–5244. doi: 10.1128/JB.00989-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang Y, Li C, Duan N, Li B, Ding XM, Yao YF, Hu J, et al. GlnR negatively regulates the transcription of the alanine dehydrogenase encoding gene ald in Amycolatopsis mediterranei U32 under nitrogen limited conditions via specific binding to its major transcription initiation site . PLoS One. . 2014;9:e104811. doi: 10.1371/journal.pone.0104811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liao CH, Yao L, Xu Y, Liu WB, Zhou Y, Ye BC. Nitrogen regulator GlnR controls uptake and utilization of non-phosphotransferase-system carbon sources in actinomycetes. Proc Natl Acad Sci USA. . 2015;112:15630–15635. doi: 10.1073/pnas.1508465112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cen XF, Wang JZ, Zhao GP, Wang Y, Wang J. Molecular evidence for the coordination of nitrogen and carbon metabolisms, revealed by a study on the transcriptional regulation of the agl3EFG operon that encodes a putative carbohydrate transporter in Streptomyces coelicolor . Biochem Biophys Res Commun. . 2016;471:510–514. doi: 10.1016/j.bbrc.2016.02.044. [DOI] [PubMed] [Google Scholar]
- 27.Liu XX, Shen MJ, Liu WB, Ye BC. GlnR-mediated regulation of short-chain fatty acid assimilation in Mycobacterium smegmatis . Front Microbiol. . 2018;9:1311. doi: 10.3389/fmicb.2018.01311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sola-Landa A, Rodríguez-García A, Amin R, Wohlleben W, Martín JF. Competition between the GlnR and PhoP regulators for the glnA and amtB promoters in Streptomyces coelicolor . Nucleic Acids Res. . 2013;41:1767–1782. doi: 10.1093/nar/gks1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yao L, Ye BC, Elliot MA. Reciprocal regulation of GlnR and PhoP in response to nitrogen and phosphate limitations in Saccharopolyspora erythraea . Appl Environ Microbiol. . 2016;82:409–420. doi: 10.1128/AEM.02960-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu X, Liu Y, Lei C, Zhao G, Wang J. GlnR Dominates rifamycin biosynthesis by activating the rif cluster genes transcription both directly and indirectly in Amycolatopsis mediterranei . Front Microbiol. . 2020;11:319. doi: 10.3389/fmicb.2020.00319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhao W, Zhong Y, Yuan H, Wang J, Zheng H, Wang Y, Cen X, et al. Complete genome sequence of the rifamycin SV-producing Amycolatopsis mediterranei U32 revealed its genetic characteristics in phylogeny and metabolism. Cell Res. . 2010;20:1096–1108. doi: 10.1038/cr.2010.87. [DOI] [PubMed] [Google Scholar]
- 32.Shao ZH, Ren SX, Liu XQ, Xu J, Yan H, Zhao GP, Wang J. A preliminary study of the mechanism of nitrate-stimulated remarkable increase of rifamycin production in Amycolatopsis mediterranei U32 by RNA-seq . Microb Cell Fact. . 2015;14:75. doi: 10.1186/s12934-015-0264-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shao Z, Gao J, Ding X, Wang J, Chiao J, Zhao G. Identification and functional analysis of a nitrate assimilation operon nasACKBDEF from Amycolatopsis mediterranei U32 . Arch Microbiol. . 2011;193:463–477. doi: 10.1007/s00203-011-0690-0. [DOI] [PubMed] [Google Scholar]
- 34.Zhang W, Yang L, Jiang W, Zhao G, Yang Y, Chiao J. Molecular analysis and heterologous expression of the gene encoding methylmalonyl-coenzyme A mutase from rifamycin SV-producing strain Amycolatopsis mediterranei U32 . Appl Biochem Biotechnol. . 1999;82:209–226. doi: 10.1385/ABAB:82:3:209. [DOI] [PubMed] [Google Scholar]
- 35.Patti GJ, Tautenhahn R, Siuzdak G. Meta-analysis of untargeted metabolomic data from multiple profiling experiments. Nat Protoc. . 2012;7:508–516. doi: 10.1038/nprot.2011.454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Llufrio EM, Cho K, Patti GJ. Systems-level analysis of isotopic labeling in untargeted metabolomic data by X13CMS. Nat Protoc. . 2019;14:1970–1990. doi: 10.1038/s41596-019-0167-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Baker M. Metabolomics: from small molecules to big ideas. Nat Methods. . 2011;8:117–121. doi: 10.1038/nmeth0211-117. [DOI] [Google Scholar]
- 38.Hu L, Liu J, Zhang W, Wang T, Zhang N, Lee YH, Lu H. Functional metabolomics decipher biochemical functions and associated mechanisms underlie small‐molecule metabolism. Mass Spectrometry Rev. . 2020;39:417–433. doi: 10.1002/mas.21611. [DOI] [PubMed] [Google Scholar]
- 39.Maan K, Baghel R, Dhariwal S, Sharma A, Bakhshi R, Rana P. Metabolomics and transcriptomics based multi-omics integration reveals radiation-induced altered pathway networking and underlying mechanism. npj Syst Biol Appl. . 2023;9:42. doi: 10.1038/s41540-023-00305-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu P, Luo J, Zheng Q, Chen Q, Zhai N, Xu S, Xu Y, et al. Integrating transcriptome and metabolome reveals molecular networks involved in genetic and environmental variation in tobacco. DNA Res. . 2020;27:dsaa006. doi: 10.1093/dnares/dsaa006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sayed AM, Abdel‐Wahab NM, Hassan HM, Abdelmohsen UR. Saccharopolyspora: an underexplored source for bioactive natural products . J Appl Microbiol. . 2020;128:314–329. doi: 10.1111/jam.14360. [DOI] [PubMed] [Google Scholar]
- 42.Lu C, Yin J, Zhao F, Li F, Lu W. Metabolomics analysis of the effect of dissolved oxygen on spinosad production by Saccharopolyspora spinosa . Antonie van Leeuwenhoek. . 2017;110:677–685. doi: 10.1007/s10482-017-0835-5. [DOI] [PubMed] [Google Scholar]
- 43.Zhao F, Xue C, Wang M, Wang X, Lu W. A comparative metabolomics analysis of Saccharopolyspora spinosa WT, WH124, and LU104 revealed metabolic mechanisms correlated with increases in spinosad yield . Biosci Biotechnol Biochem. . 2013;77:1661–1668. doi: 10.1271/bbb.130169. [DOI] [PubMed] [Google Scholar]
- 44.Rang J, He H, Yuan S, Tang J, Liu Z, Xia Z, Khan TA, et al. Deciphering the metabolic pathway difference between Saccharopolyspora pogona and Saccharopolyspora spinosa by comparative proteomics and metabonomics . Front Microbiol. . 2020;11:396. doi: 10.3389/fmicb.2020.00396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Obanye AIC, Hobbs G, Gardner DCJ, Oliver SG. Correlation between carbon flux through the pentose phosphate pathway and production of the antibiotic methylenomycin in Streptomyces coelicolor A3(2) . Microbiology. . 1996;142:133–137. doi: 10.1099/13500872-142-1-133. [DOI] [PubMed] [Google Scholar]
- 46.Naeimpoor F, Mavituna F. Metabolic flux analysis in Streptomyces coelicolor under various nutrient limitations . Metab Eng. . 2000;2:140–148. doi: 10.1006/mben.2000.0146. [DOI] [PubMed] [Google Scholar]
- 47.Borodina I, Krabben P, Nielsen J. Genome-scale analysis of Streptomyces coelicolor A3(2) metabolism . Genome Res. . 2005;15:820–829. doi: 10.1101/gr.3364705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Coze F, Gilard F, Tcherkez G, Virolle MJ, Guyonvarch A, Desvaux M. Carbon-flux distribution within Streptomyces coelicolor metabolism: a comparison between the actinorhodin-producing strain M145 and its non-producing derivative M1146 . PLoS One. . 2013;8:e84151. doi: 10.1371/journal.pone.0084151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lim Y, Jung ES, Lee JH, Kim EJ, Hong SJ, Lee YH, Lee CH, et al. Non-targeted metabolomics unravels a media-dependent prodiginines production pathway in Streptomyces coelicolor A3(2) . PLoS One. . 2018;13:e0207541. doi: 10.1371/journal.pone.0207541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Olano C, Lombó F, Méndez C, Salas JA. Improving production of bioactive secondary metabolites in actinomycetes by metabolic engineering. Metab Eng. . 2008;10:281–292. doi: 10.1016/j.ymben.2008.07.001. [DOI] [PubMed] [Google Scholar]
- 51.Bapat PM, Wangikar PP. Optimization of rifamycin B fermentation in shake flasks via a machine‐learning‐based approach. Biotech Bioeng. . 2004;86:201–208. doi: 10.1002/bit.20056. [DOI] [PubMed] [Google Scholar]
- 52.ban Impe JF, Bastin G. Optimal adaptive control of fed-batch fermentation processes. Control Eng Pract. . 1995;3:939–954. doi: 10.1016/0967-0661(95)00077-8. [DOI] [Google Scholar]
- 53.Mejía A, Barrios-gonzález J, Viniegra-gonzález G. Overproduction of rifamycin B by Amycolatopsis mediterranei and its relationship with the toxic effect of barbital on growth . J Antibiot. . 1998;51:58–63. doi: 10.7164/antibiotics.51.58. [DOI] [PubMed] [Google Scholar]
- 54.Ferree MA, Shannon RD. Evaluation of a second derivative UV/visible spectroscopy technique for nitrate and total nitrogen analysis of wastewater samples. Water Res. . 2001;35:327–332. doi: 10.1016/S0043-1354(00)00222-0. [DOI] [PubMed] [Google Scholar]
- 55.Pasqualucci CR, Vigevani A, Radaelli P, Gallo GG. Improved differential spectrophotometric determination of rifamycins. J Pharm Sci. . 1970;59:685–687. doi: 10.1002/jps.2600590522. [DOI] [PubMed] [Google Scholar]
- 56.Yuan H, Zhao W, Zhong Y, Wang J, Qin Z, Ding X, Zhao GP. Two genes, rif15 and rif16, of the rifamycin biosynthetic gene cluster in Amycolatopsis mediterranei likely encode a transketolase and a P450 monooxygenase, respectively, both essential for the conversion of rifamycin SV into B . Acta Biochim Biophys Sin. . 2011;43:948–956. doi: 10.1093/abbs/gmr091. [DOI] [PubMed] [Google Scholar]
- 57.Wang R, Yin Y, Li J, Wang H, Lv W, Gao Y, Wang T, et al. Global stable-isotope tracing metabolomics reveals system-wide metabolic alternations in aging Drosophila. Nat Commun. . 2022;13:3518. doi: 10.1038/s41467-022-31268-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yuan J, Bennett BD, Rabinowitz JD. Kinetic flux profiling for quantitation of cellular metabolic fluxes. Nat Protoc. . 2008;3:1328–1340. doi: 10.1038/nprot.2008.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wang W, Li S, Li Z, Zhang J, Fan K, Tan G, Ai G, et al. Harnessing the intracellular triacylglycerols for titer improvement of polyketides in Streptomyces. Nat Biotechnol. . 2020;38:76–83. doi: 10.1038/s41587-019-0335-4. [DOI] [PubMed] [Google Scholar]
- 60.Zhang H, Liu Y, Nie X, Liu L, Hua Q, Zhao GP, Yang C. The cyanobacterial ornithine–ammonia cycle involves an arginine dihydrolase. Nat Chem Biol. . 2018;14:575–581. doi: 10.1038/s41589-018-0038-z. [DOI] [PubMed] [Google Scholar]
- 61.Dong W, Nie X, Zhu H, Liu Q, Shi K, You L, Zhang Y, et al. Mycobacterial fatty acid catabolism is repressed by FdmR to sustain lipogenesis and virulence. Proc Natl Acad Sci USA. . 2021;118:e2019305118. doi: 10.1073/pnas.2019305118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhang Y, Zhang Y, Li P, Wang Y, Wang J, Shao Z, Zhao G. GlnR positive transcriptional regulation of the phosphate-specific transport system pstSCAB in Amycolatopsis mediterranei U32 . Acta Biochim Biophys Sin. . 2018;50:757–765. doi: 10.1093/abbs/gmy073. [DOI] [PubMed] [Google Scholar]
- 63.Bułyszko I, Dräger G, Klenge A, Kirschning A. Evaluation of the synthetic potential of an AHBA knockout mutant of the rifamycin producer Amycolatopsis mediterranei . Chem Eur J. . 2015;21:19231–19242. doi: 10.1002/chem.201503548. [DOI] [PubMed] [Google Scholar]
- 64.Floss HG, Yu TW, Arakawa K. The biosynthesis of 3-amino-5-hydroxybenzoic acid (AHBA), the precursor of mC7N units in ansamycin and mitomycin antibiotics: a review. J Antibiot. . 2011;64:35–44. doi: 10.1038/ja.2010.139. [DOI] [PubMed] [Google Scholar]
- 65.Guo J, Frost JW. Kanosamine Biosynthesis: A likely source of the aminoshikimate pathway′s nitrogen atom. J Am Chem Soc. . 2002;124:10642–10643. doi: 10.1021/ja026628m. [DOI] [PubMed] [Google Scholar]
- 66.Eads JC, Beeby M, Scapin G, Yu TW, Floss HG. Crystal structure of 3-amino-5-hydroxybenzoic acid (AHBA) synthase , . Biochemistry. . 1999;38:9840–9849. doi: 10.1021/bi990018q. [DOI] [PubMed] [Google Scholar]
- 67.Meier TW, Thomä NH, Leadlay PF. Tritium isotope effects in adenosylcobalamin-dependent methylmalonyl-CoA mutase. Biochemistry. . 1996;35:11791–11796. doi: 10.1021/bi961250o. [DOI] [PubMed] [Google Scholar]
- 68.Tang L, Yoon YJ, Choi CY, Hutchinson CR. Characterization of the enzymatic domains in the modular polyketide synthase involved in rifamycin B biosynthesis by Amycolatopsis mediterranei . Gene. . 1998;216:255–265. doi: 10.1016/S0378-1119(98)00338-2. [DOI] [PubMed] [Google Scholar]
- 69.Yazawa K, Mikami Y, Maeda A, Akao M, Morisaki N, Iwasaki S. Inactivation of rifampin by Nocardia brasiliensis . Antimicrob Agents Chemother. . 1993;37:1313–1317. doi: 10.1128/AAC.37.6.1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.El-Enshasy HA, Beshay UI, El-Diwany AI, Omar HM, El-Kholy AG, El-Najar R. Improvement of rifemycins production by Amycolatopsis mediterranei in batch and fed-batch cultures. Acta Microbiol Pol. 2003, 52: 301–313 . [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.