Abstract
Due to its association with low-quality milk and a decrease in milk production in bovines, mastitis is a major cause of economic loss. Additionally, mastitis can be harmful to suckling newborns and can cause damage to the mammary gland. In mastitic mammary secretions there is a substantial increase in somatic cells, specifically neutrophils. In this study we examined the ability of mastitic and nonmastitic mammary secretions to cause in vitro neutrophil chemotaxis using a microchemotaxis assay. Also, the role of the inflammatory chemokine interleukin-8 (IL-8) in neutrophil recruitment during mastitis was addressed in these in vitro experiments. We found that both nonmastitic and mastitic mammary secretions were chemotactic, not chemokinetic, for neutrophils. The neutrophil chemotactic activity in mastitic, but not nonmastitic, mammary secretions was blocked by anti-IL-8 antibodies. Molecular mass separation of the active components showed that the chemotactic activity of the mastitic secretions was present in the 10-kDa-or-less fraction and was blocked by anti-IL-8 antibodies. These results indicate that IL-8 plays a major role in neutrophil recruitment during mastitis. An understanding of its role will be of help in designing strategies for immunomodulatory therapies for mastitis.
Mastitis is detrimental to both the suckling newborn and the mammary gland. For the bovine dairy industry, mastitis is also a major cause of economic loss due to its association with decreased milk production and low-quality milk (4). One hallmark feature of mastitis is the substantial increase in somatic cells found in mammary secretions (5, 17). Somatic cells include lymphocytes, a small percentage of epithelial cells, macrophages, and neutrophils (21). The increase in somatic cells, specifically neutrophils, is thought to serve as a mechanism against an increase in the infection of the gland (26). The migration of neutrophils from the peripheral blood, through the mammary tissue, and into the mammary secretions is called chemotaxis (22). Briefly, chemotaxis is a highly regulated process in which selectins, integrins, and chemoattractants interact to generate cell migration (31). Selectins are adhesion molecules on leukocyte cell membranes that have an N-terminal domain homologous to Ca2+-dependent lectins and are responsible for the attachment of leukocytes to vessel walls (2). Integrins are responsible for leukocyte-endothelial cell interactions which precede the migration into tissue (15). Lastly, chemoattractants are soluble mediators released at or near the site of chemotaxis. They function to regulate integrins as well as to bind leukocytes and modulate migration (22). The cytokine interleukin-8 (IL-8) is one such chemotactic factor.
IL-8 is a chemokine that is produced by numerous cell types including lymphocytes (9), neutrophils (33), monocytes/macrophages (27), and epithelial cells (8), including human mammary gland epithelial cells (19). Also, many different tumor cell lines are able to produce IL-8 (34). Additionally, human milk mononuclear cells that have been stimulated by lipopolysaccharide (LPS) are shown to produce IL-8 (30). IL-8 has several biological activities, including recruiting and activating neutrophils (10), inducing neutrophil degranulation (27), stimulating phagocytosis of opsonized particles (7), and recruiting T lymphocytes (12, 16). IL-8 does appear to be specific to neutrophils and T cells in that eosinophils and monocytes do not respond to it (27). In addition, IL-8 has been detected in human mammary secretions. Human maternal cells in breast milk express mRNA for IL-8 (32), and in bovine mammary secretions, IL-8 was detected in mammary secretions from glands that had been challenged with Escherichia coli (28, 29).
In this study we examined whether nonmastitic and mastitic mammary secretions were chemotactic for neutrophil chemotaxis and if IL-8 was responsible. Our results show both mastitic and nonmastitic secretions were chemotactic rather than chemokinetic for neutrophils. The neutrophil chemotactic activity in mastitic, but not nonmastitic, mammary secretions was blocked by anti-IL-8 antibodies.
MATERIALS AND METHODS
Reagents.
All reagents were obtained from Sigma Chemical Co., St. Louis, Mo., unless otherwise noted.
Anti-human IL-8 antiserum produced in chickens that was found to cross-react with bovine IL-8 (23) was kindly provided by Donald L. Kreutzer (Departments of Pathology and Surgery, Vision Immunology Center, University of Connecticut).
Mammary secretions.
Normal lactation-stage mammary secretions were collected from individual quarters of four Holstein cows as described previously (1) and from a mastitic Holstein cow (four quarters) housed at the Kellog Dairy Center at the University of Connecticut. Samples were grouped as nonmastitic or mastitic based on an increase in somatic cell counts (>7.5 × 105 cells/ml), bacteriological studies, and clinical signs of inflammation of the mammary gland (i.e., swelling, redness, and heat) or milk (i.e., clots and flakes) (1). The causative agent of mastitis in the mastitic samples was Staphylococcus aureus.
Isolation of responder cells.
Bovine blood was collected via venipuncture into an EDTA vacutainer tube (Fisher Scientific, Pittsburgh, Pa.). Whole blood was centrifuged at 400 × g for 20 min. The plasma and buffy coat layers were aspirated, and the erythrocyte pellet, which contained neutrophils, was subjected to hypotonic lysis to remove the erythrocytes. Neutrophils were recovered (500 × g, 5 min), washed three times with RPMI medium, and resuspended at a final concentration of 2 × 106 cells/ml in RPMI medium. Typically, the viability was greater than 98% as shown by the trypan blue exclusion test.
Preparation of whey.
Mammary secretions were centrifuged at 500 × g for 20 min to remove fat and cells. Samples were then centrifuged at 100,000 × g at 4°C for 30 min, and the supernatants (whey) were stored at −20°C until use. Before use, the clarified whey was passed through a 0.45-μm-pore-sized filter. Protein concentrations of the samples were determined as described previously (1) by the bicinchoninic acid method (14).
Molecular mass fractionation of mastitic whey.
Mastitic whey was first treated with rennin (23.6 U/mg; 1 U will coagulate 10 ml of whey) at 30°C for 30 min to separate casein. Treated whey was centrifuged at 100,000 × g for 1 h and the casein-free supernatant was collected. Purity was assessed by the absence of a casein band on a sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel (data not shown).
Casein-free mastitic whey was further separated according to molecular mass by using a filter system. Briefly, casein-free whey was placed into an Amicon apparatus (Amicon Corp., Beverly, Mass.) with a 10-kDa cutoff membrane. With constant stirring at 4°C the whey was passed through the membrane by using N2 gas as a source of pressure. Material that was retained was saved as the fraction greater than 10 kDa, and the effluent was collected and the process was repeated using a 1-kDa cutoff filter. The material that did not flow through was collected and labeled as the less-than-10-kDa fraction.
The casein-free whey, the fraction less than 10 kDa, and the fraction greater than 10 kDa were all dialyzed by using a 3.5-kDa cutoff dialysis membrane against distilled water overnight at 4°C.
Fractionated samples were lyophilized and stored desiccated at −70°C until use.
Chemotactic assays.
Neutrophil migration-inducing activity in mammary secretions was assayed by a microchemotaxis technique with a 48-well modified Boyden chamber (Neuroprobe, Cabin John, Md.) (2, 3). A polycarbonate membrane (pore size, 5 μm; Poretics Corp., Livermore, Calif.) was used. A total of 105 neutrophils in a volume of 50 μl was added in the top wells of the chamber. The bottom wells contained 30 μl of the sample to be measured for chemotactic activity. Cell migration proceeded in an incubator with humidified air with 5% CO2 for 30 min. The filters were then removed, the side facing the top well was rinsed with phosphate-buffered saline and wiped clean to eliminate any nonmigrating cells. Filters were fixed with methanol (30 s), allowed to air dry, stained with Diff-Quick (Fisher Scientific), mounted with the side facing the bottom well oriented upward onto a glass slide (75 by 50 mm) (Corning Glass, Corning, N.Y.), and rubbed with immersion oil. Neutrophils were counted under a light microscope with a 40× objective and a 10× ocular. Neutrophil migration was expressed as the chemotactic index (CI), which is the number of neutrophils which migrated towards a sample/the number of neutrophils which migrated towards a control medium.
RPMI medium was used as a negative control, and to ensure that the assay was working properly LPS (GIBCO, Grand Island, N.Y.)-stimulated bovine macrophage culture supernatant was used as a positive control in chemotaxis assays. Briefly, 2 × 106 blood monocytes were isolated by Ficoll-Hypaque (specific density, 1.084) and adherence to plastic. Adherent macrophages were stimulated with LPS (25 μg/ml) in RPMI medium with 10% fetal bovine serum at 37°C for 24 h. The supernatant was collected and stored at −20°C. Positive controls yielded an average CI of 3.7 ± 0.81.
Checkerboard analysis.
Checkerboard analysis (35) to differentiate between chemotaxis and chemokinesis was performed by placing 30 μl of various dilutions of secretions in the lower wells of the Boyden chamber and by also placing 50 μl of various dilutions in the upper wells with 105 responder cells. A migration assay was carried out as described above.
Data analysis.
Data were expressed as means ± standard errors of the means. A two-tailed Student’s t test was used to determine significance (P < 0.05).
RESULTS
Chemotactic activity of mammary secretions.
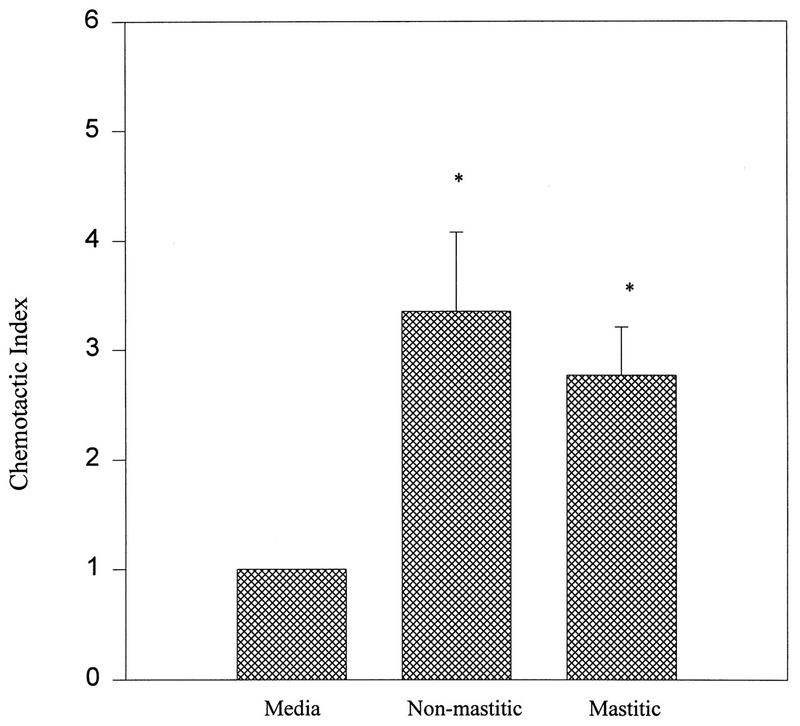
Figure 1 shows the chemotactic activity of mastitic and nonmastitic mammary secretions. Nonmastitic secretions had a CI of 3.4 ± 0.7 and mastitic secretions had a CI of 2.8 ± 0.4. Both nonmastitic and mastitic secretions had CIs that were significantly higher than that of the control medium.
FIG. 1.
Chemotactic activities of nonmastitic and mastitic mammary secretions. The data are from a representative experiment with at least three separate nonmastitic or mastitic mammary secretions. Samples were run at physiological concentrations. Data are mean CI values (number of neutrophils which migrated towards sample/number of neutrophils which migrated towards a control medium) ± standard errors of the means. ∗, P < 0.05 compared to the control medium (media).
Checkerboard analysis of mammary secretions.
Checkerboard analysis was performed to determine whether neutrophil migration was due to either chemotaxis or chemokinesis. Dilutions of mammary secretions were added to the upper and lower wells of the Boyden chamber and neutrophil migration was quantitated. As shown in Fig. 2A (nonmastitic) and B (mastitic), neutrophil migration generally increased as the concentration gradient between the upper and lower chambers increased. This indicates that the migratory activity in the mammary secretions was chemotactic rather than chemokinetic.
FIG. 2.
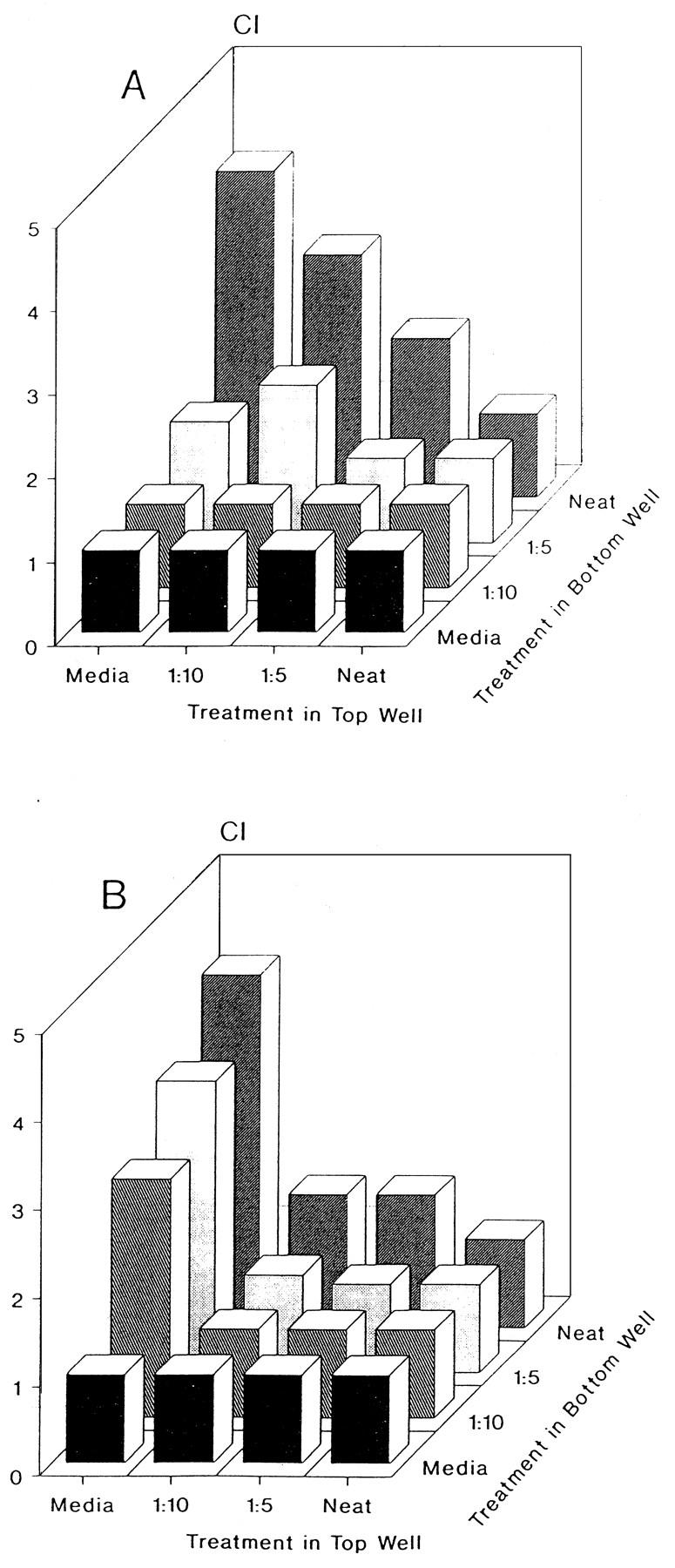
Checkerboard analysis of nonmastitic (A) and mastitic (B) mammary secretions. Various concentration gradients were established by placing different dilutions (Neat, undiluted) of samples in the bottom and top (along with responder cells) wells of a Boyden chamber. The data indicate that migration is dependent on an increasing concentration gradient. Data are expressed as the means of triplicate samples. Media, medium alone (used as a negative control).
Effects of anti-IL-8 antibodies on neutrophil chemotactic activities of nonmastitic and mastitic mammary secretions.
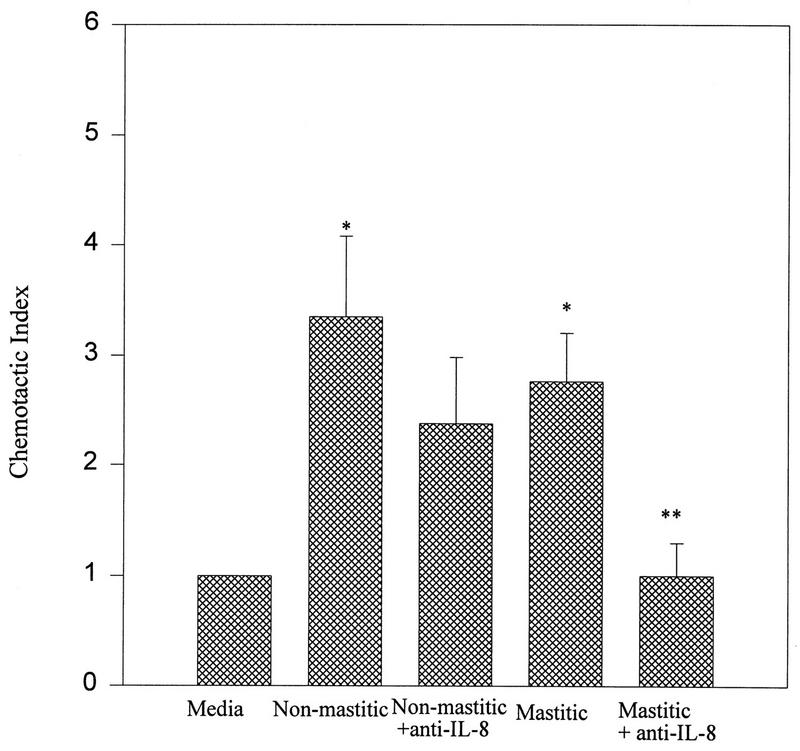
Since IL-8 is a potent inducer of neutrophil migration we used antibodies specific for IL-8 (23) to determine if IL-8 was involved in neutrophil chemotaxis towards mammary secretions. Figure 3 shows that anti-IL-8 antibodies significantly blocked the chemotactic activity of mastitic mammary secretions (P < 0.05). In contrast, anti-IL-8 antibodies did not have a significant effect on the chemotactic activity of nonmastitic mammary secretions.
FIG. 3.
Effects of anti-IL-8 antibodies on neutrophil chemotaxis towards nonmastitic and mastitic mammary secretions. Data are representative of at least three separate samples and are expressed as means ± standard errors of the means. ∗, P < 0.05 compared to the control medium (media); ∗∗, P < 0.05 compared to mastitic secretions.
Chemotactic activity of molecular mass fractions of mastitic mammary secretions.
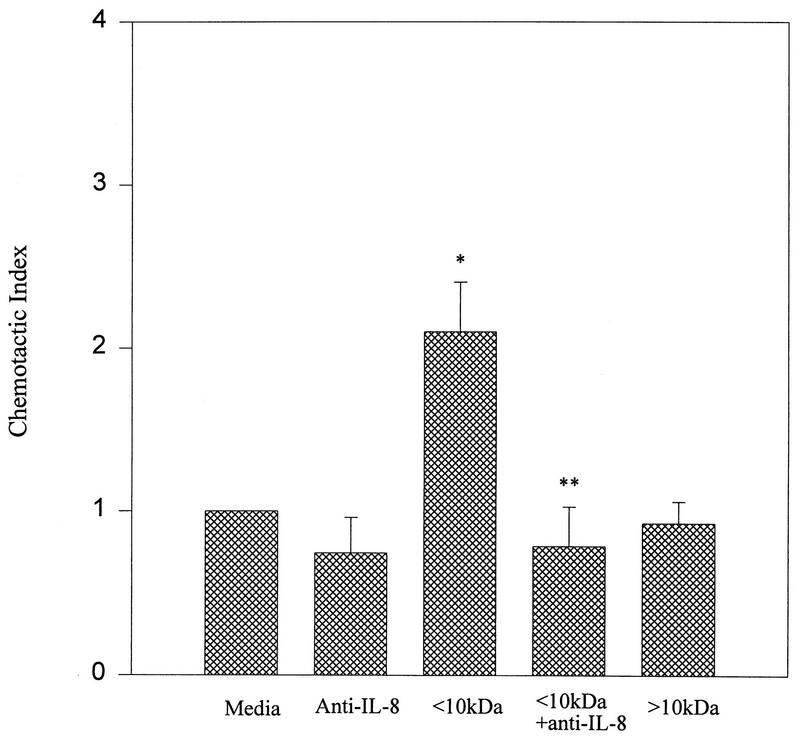
Bovine IL-8 has previously been shown to have a molecular mass of 7.8 kDa (11). We therefore fractionated mastitic samples to determine if the chemotactic activity falls within the anticipated fraction. Prior to this determination, and to facilitate molecular mass fractionation, casein (a milk protein found in significant amounts) had been precipitated by rennin and the casein-free whey was analyzed for chemotactic activity. Casein-free whey was found to retain its chemotactic activity, and this activity could be eliminated by anti-IL-8 antibodies (data not shown). The fraction of mastitic casein-free whey greater than 10 kDa did not cause significant chemotaxis, whereas the fraction smaller than 10 kDa did cause significant chemotaxis that could be abrogated by anti-IL-8 antibodies (Fig. 4).
FIG. 4.
Chemotactic activities of mastitic milk molecular mass fractions. Data are expressed as means of triplicate samples ± standard errors of the means. ∗, P < 0.05 compared to the control medium (media); ∗∗, P < 0.05 compared to the <10-kDa fraction. The <10-kDa fraction was used at a concentration of 0.1 mg/ml (1 mg/ml was inhibitory to chemotaxis). The >10-kDa fraction was used at a 1-mg/ml concentration, and further dilution had no effect on chemotaxis.
Protein concentrations.
In order to determine if an increase in IL-8 activity was due to an increase in protein concentration, the protein concentrations of the whey samples were determined. Table 1 shows that the protein concentrations of nonmastitic and mastitic secretions were not significantly different.
TABLE 1.
Protein concentrations of nonmastitic and mastitic mammary secretionsa
| Sample | Protein concn (mg/ml) |
|---|---|
| 1M | 10.17 |
| 4M | 12.49 |
| 5M | 12.40 |
| 7M | 15.22 |
| 2NM | 12.08 |
| 3NM | 12.33 |
| 6NM | 14.80 |
| 8NM | 16.65 |
Mammary secretions were collected from individual quarters and were classified as nonmastitic or mastitic based on criteria described in Materials and Methods. The secretion was assessed for protein concentration to determine if increased chemotactic activity was related to increased protein concentration. M, mastitic; NM, nonmastitic.
DISCUSSION
Chemotaxis of neutrophils is a complex process involving attachment of neutrophils to vessel walls, transmigration through the vasculature endothelium, and migration into the tissue site (for a review, see reference 22). Soluble mediators of chemotaxis, chemoattractants, help to regulate and control migration. IL-8 is one such mediator. It is an 8-kDa protein (11) which has been shown to induce leukocyte migration in vitro and to mediate inflammation in vivo (10). Its role in recruiting neutrophils during inflammation prompted us to examine the role of IL-8 in inflammation of the mammary gland (mastitis).
Mastitis is a major cause of economic loss to the dairy industry (4). It is characterized by increases in the somatic cell count and viable bacteria in the mammary secretions as well as by clinical signs. Others have shown that cytokines play an important regulatory role in mastitis (6, 20, 28, 29). In this study we determined whether nonmastitic and mastitic secretions could cause in vitro neutrophil chemotaxis and, if so, whether IL-8 was involved.
As shown in Fig. 1 both nonmastitic and mastitic secretions induced neutrophil migration in vitro. A checkerboard analysis indicated that the activity was chemotactic and not chemokinetic (Fig. 2). Since both nonmastitic and mastitic secretions caused neutrophil chemotaxis, we next determined if IL-8 was involved in either case. By using anti-human IL-8 antibodies produced in chickens (which cross-react to bovine IL-8 [23]) to block chemotaxis, it was determined that mastitic secretions, but not nonmastitic secretions, contained IL-8. The anti-IL-8 antibodies blocked nearly 100% of the chemotactic activities in mastitic secretions, whereas they had only a 25% inhibitory effect (which was not significant) on chemotaxis caused by normal secretions. Additionally, when the mastitic samples were fractionated according to molecular mass, the fraction less than 10 kDa was found to contain chemotactic activity that could be abrogated by anti-IL-8 antibodies, whereas the fraction greater than 10 kDa did not contain any chemotactic activity. Currently, nonmastitic mammary secretions are being further analyzed to characterize properties of the chemoattractants found in them. Interestingly, at high concentrations the fraction less than 10 kDa did not show chemotactic activity (data not shown). This may have been due to a previously identified low-molecular-weight inhibitor that inhibits chemotaxis (18). This inhibitory effect on chemotaxis was, in our study, alleviated by serial dilutions.
Numerous cell types have the ability to produce IL-8, including leukocytes (9, 26, 33) and epithelial cells (8). Both of these cell types may produce IL-8 during mastitis. There is a breakdown of the mammary gland epithelium during mastitis which may allow leaking of IL-8 and other proteins into the secretions. Also, leukocytes in mammary secretions could be activated by other proinflammatory cytokines, such as tumor necrosis factor alpha and IL-1 (25, 32), to produce IL-8. Tumor necrosis factor alpha has been shown to be present in bovine (24) and human (25) mammary secretions, and its concentration in these secretions increases during mastitis (29).
IL-8 was not found to be involved in in vitro chemotaxis of neutrophils induced by nonmastitic mammary secretions. More work is being done to assess the chemotactic factors found in normal secretions and to determine why they were not present in mastitic secretions. This lack of non-IL-8-induced chemotaxis in mastitic secretions may be due in part to the inflammatory environment of the gland allowing for the production of factors that negatively affect the chemotactic activity of neutrophils towards chemotactic factors by mechanisms such as the binding of cytokine and/or chemokine receptors or the inhibiting of the production of chemokines by cells. For example, transforming growth factor β (1), IL-10 (13), and IL-4 (which inhibits T-cell migration) have been shown, in some circumstances, to inhibit chemotaxis (12).
ACKNOWLEDGMENTS
This work was supported by USDA AMD 9404477 and the Storrs Agricultural Experimental Station NE-112 project.
We are grateful to Arnold Nieminen at the Kellog Dairy Center (University of Connecticut) for assistance in collecting milk samples and Alexander Pantschenko for help in obtaining blood samples. We thank Lynn Hinckley and her staff at the Diagnostic Testing Services (University of Connecticut) for assistance with the bacteriological studies.
REFERENCES
- 1.Ayoub I A, Bendal R B, Yang T J. Increase in the proportion of CD4+ T-lymphocytes and the levels of transforming growth factor-β in the milk of mastitic cows. Immunol Infect Dis. 1996;6:145–150. [Google Scholar]
- 2.Bevilacque M P. Endothelial-leukocyte adhesion molecules. Annu Rev Immunol. 1993;11:767–804. doi: 10.1146/annurev.iy.11.040193.004003. [DOI] [PubMed] [Google Scholar]
- 3.Boyden S. Chemotactic effect of mixtures of antibody and antigen on polymorphonuclear leukocytes. J Exp Med. 1962;115:453–459. doi: 10.1084/jem.115.3.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Christ W L, Harmon R J, Newman L E. How much does mastitis cost? Udder Top. 1983;6:6–9. [Google Scholar]
- 5.Daley M J, Oldham E R, Williams T J, Coyle P A. Bovine mastitis. I. Quantitative and qualitative properties of host polymorphonuclear cells during Staphylococcus aureus mastitis infection. Am J Vet Res. 1991;52:474–479. [PubMed] [Google Scholar]
- 6.Daley M J, Williams T J, Coyles P A, Furda G, Dougherty R, Hayes P. Prevention and treatment of Staphylococcus aureus infections with recombinant cytokines. Cytokine. 1993;5:276–284. doi: 10.1016/1043-4666(93)90015-w. [DOI] [PubMed] [Google Scholar]
- 7.Detmers P A, Powell D E, Walz A, Clark-Lewis I, Baggionlini M, Cohn Z A. Differential effects of neutrophil-activating peptide/IL-8 and its homologues on leukocyte adhesion and phagocytosis. J Immunol. 1991;147:4211–4217. [PubMed] [Google Scholar]
- 8.Elner V M, Streiter R M, Elner S G, Baggionlini M, Lindley I, Kunkel S L. Neutrophil chemotactic factor (IL-8) gene expression by cytokine-treated retinal pigment epithelial cells. Am J Pathol. 1990;136:745–750. [PMC free article] [PubMed] [Google Scholar]
- 9.Gregory H, Young J, Schroder J M, Mirowitz U, Christophers E. Structure determination of a human lymphocyte derived neutrophil activating peptide (LYNAP) Biochem Biophys Res Commun. 1988;151:883–890. doi: 10.1016/s0006-291x(88)80364-4. [DOI] [PubMed] [Google Scholar]
- 10.Harda A, Sekido N, Akahoshi T, Wada T, Mukaida N, Matsushima K. Essential involvement of interleukin-8 (IL-8) in acute inflammation. J Leukocyte Biol. 1994;56:559–564. [PubMed] [Google Scholar]
- 11.Hassfurther R L, Canning P C, Geib R W. Isolation and characterization of an interleukin-8-like peptide in the bovine species. Vet Immunol and Immunopathol. 1994;42:117–126. doi: 10.1016/0165-2427(94)90001-9. [DOI] [PubMed] [Google Scholar]
- 12.Jinquan T, Deleyran B, Gesser B, Maare H, Delevran M, Larsen C G, Thestrup-Pederson K. Regulation of human T-lymphocyte chemotaxis in vitro by T-cell derived cytokines IL-2, IFN-γ, IL-4, IL-10, and IL-13. J Immunol. 1995;154:3742–3752. [PubMed] [Google Scholar]
- 13.Kasama T, Streiter R M, Lukacs N W, Burdick M D, Kunkel S L. Regulation of neutrophil-derived chemokine expression by IL-10. J Immunol. 1994;152:3559–3569. [PubMed] [Google Scholar]
- 14.Keller R P, Neville M C. Determination of total protein in human milk: comparison of methods. Clin Chem. 1986;32:120–123. [PubMed] [Google Scholar]
- 15.Larson R S, Springer T A. Structure and function of leukocyte integrins. Immunol Rev. 1990;114:181–217. doi: 10.1111/j.1600-065x.1990.tb00565.x. [DOI] [PubMed] [Google Scholar]
- 16.Leonard E J, Skeel A, Yoshimura T, Noer K, Kutvivt S, VanEpps D. Leukocyte specificity and binding of human neutrophil attractant/activation protein-1. J Immunol. 1990;144:1323–1330. [PubMed] [Google Scholar]
- 17.Newbould F H S, Neave F K. The response of the bovine mammary gland to an infusion of Staphylococci. J Dairy Res. 1965;32:163–170. [Google Scholar]
- 18.Ormrod D J, Miller T E. A low molecular weight component derived from the milk of hyperimmunised cows suppresses inflammation by inhibiting neutrophil emigration. Agents Actions. 1992;37:70–79. doi: 10.1007/BF01987892. [DOI] [PubMed] [Google Scholar]
- 19.Palkowitz K H, Royner C L, Garofalo R, Rudolff H E, Schmalstieg F C, Jr, Goldman A S. Production of interleukin-6 and interleukin-8 by human mammary gland epithelial cells. J Reprod Immunol. 1994;26:57–64. doi: 10.1016/0165-0378(93)00867-s. [DOI] [PubMed] [Google Scholar]
- 20.Persson K, Larson I, Hallen Sandgren C. Effects of certain inflammatory mediators on bovine neutrophil migration in vivo and in vitro. Vet Immunol Immunopathol. 1993;37:99–112. doi: 10.1016/0165-2427(93)90058-c. [DOI] [PubMed] [Google Scholar]
- 21.Philpot W N, Nickerson S C. Mastitis: counterattack. Naperville, Ill: Babson Bros., Co.; 1991. pp. 1–133. [Google Scholar]
- 22.Picker L J. Control of lymphocyte homing. Curr Opin Immunol. 1994;6:394–396. doi: 10.1016/0952-7915(94)90118-x. [DOI] [PubMed] [Google Scholar]
- 23.Qi J, Kreutzer D L. Fibrin activation of vascular endothelial cells: induction of IL-8 expression. J Immunol. 1995;155:867–876. [PubMed] [Google Scholar]
- 24.Rewinski M J, Yang T J. Lactation stage-dependent changes in levels of tumor necrosis factor/cachectin in milk. Am J Reprod Immunol. 1994;31:170–176. doi: 10.1111/j.1600-0897.1994.tb00863.x. [DOI] [PubMed] [Google Scholar]
- 25.Rudloff H E, Schmalstieg F C, Mustaha A A, Palowitz K H, Liu S K, Goldman A S. Tumor necrosis factor-α in human milk. Pediatr Res. 1992;31:29–33. doi: 10.1203/00006450-199201000-00005. [DOI] [PubMed] [Google Scholar]
- 26.Sandholm M, Honkanen-Buzalski T, Kaartinen L, Pyorala S. The bovine udder and mastitis. Jyväskylä, Finland: Gummerus Kirjapaino, Oy; 1995. [Google Scholar]
- 27.Schroder J M, Morwietz U, Morita E, Christophers E. Purification and partial biochemical characterization of a human monocyte-derived, neutrophil-activating peptide that lacks interleukin-1 activity. J Immunol. 1987;139:3474–3483. [PubMed] [Google Scholar]
- 28.Shuster D E, Lee E K, Kehrli M E. Bacterial growth, inflammatory cytokine production, and neutrophil recruitment during coliform mastitis in cows within ten days after calving, compared with cows at midlactation. Am J Vet Res. 1996;57:1569–1575. [PubMed] [Google Scholar]
- 29.Shuster D E, Kehrli M E, Rainard P, Paape M. Complement fragment C5a and inflammatory cytokines in neutrophil recruitment during intramammary infection with Escherichia coli. Infect Immun. 1997;65:3286–3292. doi: 10.1128/iai.65.8.3286-3292.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Skansen-Saphir U A, Lindfors A, Anderson U. Cytokine production in mononuclear cells of human milk studied at the single-cell level. Pediatr Res. 1993;34:213–216. doi: 10.1203/00006450-199308000-00023. [DOI] [PubMed] [Google Scholar]
- 31.Springer T A. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell. 1994;76:301–304. doi: 10.1016/0092-8674(94)90337-9. [DOI] [PubMed] [Google Scholar]
- 32.Srivastava M D, Srivastava A, Brouhard B, Saneto R, Groh-Wargo S, Kubit J. Cytokines in human milk. Res Commun Mol Pathol Pharmacol. 1996;93:263–287. [PubMed] [Google Scholar]
- 33.Strieter R M, Kasahara K, Allen R, Showell H J, Standiford T J, Kunkel S L. Human neutrophils exhibit disparate chemotactic factor gene expression. Biochem Biophys Res Commun. 1990;173:725–730. doi: 10.1016/s0006-291x(05)80095-6. [DOI] [PubMed] [Google Scholar]
- 34.VanDamme J. Interleukin-8 and related chemotactic cytokines. In: Thomson A, editor. The cytokine handbook. New York, N.Y: Academic Press; 1994. pp. 185–208. [Google Scholar]
- 35.Zigmund S, Hirsch J. Leukocyte locomotion and chemotaxis: new methods for evaluation and demonstration of a cell derived chemotactic factor. J Exp Med. 1973;137:387–410. doi: 10.1084/jem.137.2.387. [DOI] [PMC free article] [PubMed] [Google Scholar]