Abstract
This review provides an overview of the current research on prebiotics and their applications in animal nutrition and health. Various types of prebiotics are introduced and classified, with an analysis of agro-industrial residues as sources. The potential of prebiotics as substrates for the growth and/or activity of probiotic microorganisms, as well as their applications in animal feeds, were examined. This analysis also considered the health-promoting advantages linked to different production techniques and bioactive compounds. The mechanisms by which prebiotics alter gastrointestinal tract microbiota are discussed, specifically in terms of combating pathogenic bacteria and modulating immune cells in the gut. Investigations of the impact of prebiotics in both polygastric and monogastric animals are reported, highlighting their effects on the gut ecosystem and animal performance. This review concludes that agro-industrial residues are valuable sources of prebiotics that possess numerous beneficial health-promoting properties suitable for animal nutrition. These prebiotics can serve as a relevant substitute in the ongoing search for alternative strategies to prevent and control the presence of pathogenic bacteria in animals, particularly considering the restriction on antimicrobial growth promoters. However, further research is necessary to explore the interaction between prebiotics and probiotics in animal care and well-being, to establish systematic management of production and practical applications of these functional feed additives.
Keywords: Prebiotics, Animal nutrition, Feed additives, Gastrointestinal microbiota, Agro-industrial residue
1. Introduction
The practice of using antibiotics as feed additives in animal farming, with the purpose of promoting animal growth and balancing gut flora, has been banned to tackle the dissemination of antibiotic-resistant bacteria and their potential food-borne transmission to humans (Haulisah et al., 2021). However, the elimination of antimicrobial growth promoters has been a challenge for animal nutrition industry, as some adverse consequences have emerged for the production, health, and welfare of animals (Khan et al., 2022). Therefore, significant research efforts have been focused on the development of antibiotic alternatives to maintain or improve animal health and performance. Modulation of the gut microbiota with feed additives such as prebiotics and probiotics have become a need to maximize productivity and maintain animal health and welfare. Among these, prebiotics are a promising alternative. Their inclusion in livestock and poultry feed have shown the capability to improve host health and productivity through the selective stimulation of beneficial gut microbiota (Solís-Cruz et al., 2019). In this regard, agro-industrial waste plays a role as a source of feed ingredients in animal nutrition (Pinotti et al., 2020). In particular, agro-industrial waste is often rich in carbohydrates and fibre, which are considered prebiotics that can promote the proliferation of beneficial microbes in the large intestine. Agro-industrial wastes from fruits (e.g., mango peels, citrus peels, apple pomace) and vegetables (e.g., garlic straw, potato processing waste, onion skins) have been explored to produce prebiotics (Rahmani et al., 2017) that are employed as feed supplements. The term “prebiotics” was first coined by Gibson and Roberfroid (1995) and described as the “non-digested food components that, through stimulation of growth and/or activity of a single type or a limited amount of microorganisms residing in the gastrointestinal tract, improve the health condition of a host” (Gibson and Roberfroid, 1995). Through the years, this description has evolved, and the latest criterion to distinguish a prebiotic was created by the International Scientific Association of Probiotics and Prebiotics (ISAPP) (Gibson et al., 2017). The definition states that a prebiotic is “a substance that is selectively utilized by host microorganisms conferring a health benefit” (Gibson et al., 2017). This description broadens the prebiotic category to encompass not only carbohydrates, but also polyphenols and polyunsaturated fatty acids. The recognition that the applications of prebiotics are not restricted to the human gastrointestinal tract and includes animal nutrition highlights the broader benefits beyond human health.
On the other hand, the discovery of the health benefits of microorganisms can be traced back to 1907 when Russian scientist Elie Metchnikoff observed the higher longevity of Bulgarian farmers who consumed fermented dairy food (Mackowiak, 2013). The term “probiotics” was later coined by Werner Kollath in 1953 to refer to “essential active substances for the healthy development of life” (Gasbarrini et al., 2016). In 2001, the Food and Agriculture Organization (FAO) and the World Health Organization (WHO) officially defined probiotics as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.” The definition was further confirmed by the International Scientific Association for Probiotics and Prebiotics (ISAPP) in 2014 (Hill et al., 2014). The microorganisms commonly used as probiotics belong to various bacterial species, with Bifidobacterium, Lactobacillus, Streptococcus, Bacillus, and Enterococcus the most commonly used (Kim et al., 2020). Over the years, numerous scientific studies have demonstrated the high potential of probiotics to provide health benefits on livestock, leading to a significant increase in research on probiotics in animal feeding (Di Gioia and Biavati, 2018).
The positive effects of prebiotics on health are associated to their capability to modify the gut microflora by selectively stimulating the growth and/or activity of beneficial bacteria, such as Firmicutes, and Bacteroidetes, which produce beneficial metabolites (Uyeno et al., 2015; Yin et al., 2018). Other phyla have specific niches that differ based on the animal species. This can help to control pathogenic bacteria in livestock species, promoting overall health (Azad et al., 2020). Studies have shown that prebiotics, when employed as feed additives in animal nutrition, can lead to significant population proliferation of probiotic bacteria like Bifidobacterium and Lactobacillus. Supplementing generally healthy calves with a prebiotic has revealed to have a positive but not significant effect on the microbial community that is relatively stable in these animals (Heinrichs et al., 2009). Recent findings have shown the potential use of prebiotics as alternatives to antibiotics to enhance animal growth and health without eventual risks to human wellness and the ecosystem (Chiesa et al., 2017; Pan et al., 2019). Studies examining the effects of prebiotics on animal health frequently yield inconsistent results due to the high specificity of individual compounds, variations in dosages, and differences in the timing of the application.
The agriculture industry produces diverse types of wastes, including stems, stalks, leaves, seeds, straws, husks, peels, sugar cane bagasse, and pulp from fruits, cereals, or legumes, among others (Sadh et al., 2018). Various nutrients derived from these wastes, including pectins, cellulose, hemicellulose, and xylans, can support the development of different intestinal microorganisms. In this regard, this review highlights recent findings of prebiotics derived from agro-industrial waste and their effects on animal health.
2. Types of prebiotics
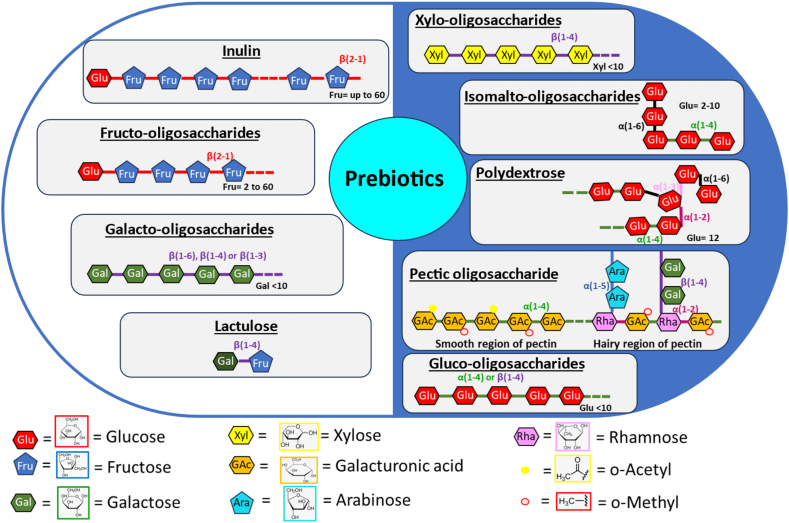
Prebiotics can be classified into several categories based on their development and regulatory status. Inulin, fructo-oligosaccharides (FOS), galacto-oligosaccharides (GOS) and lactulose are generally classified as well-established prebiotics. Xylo-oligosaccharides (XOS), isomalto-oligosaccharides (IMO) among others are classified as “emerging” prebiotics (Cardoso et al., 2021) (Fig. 1). Inulin, XOS, cereal fibre, lacticol, FOS, and GOS are among the most common prebiotics used in animal nutrition (Crittenden and Playne, 2008).
Fig. 1.
Chemical and bonding structures of prebiotics. Different types of prebiotics, including both well-established and emerging prebiotics. The well-established prebiotics include inulin, fructo-oligosaccharides (FOS), galacto-oligosaccharides (GOS), and lactulose. The emerging prebiotics consist of xylo-oligosaccharides (XOS), isomalto-oligosaccharides (IMO), polydextrose, pectic oligosaccharide, and gluco-oligosaccharide.
2.1. Inulin
Inulin is a linear polymer with β-glycosidic bonds (2 → 1) derived from D-fructose, having usually terminal glucose units with β (2 → 1) linkage. Inulin has degree of polymerization of up to 60. It is classified as a fructan and is synthesized by a range of plants. Inulin is a storage carbohydrate present in many plant species (Davani-Davari et al., 2019).
2.2. Fructo-oligosaccharides
Fructo-oligosaccharides are categorized as a type of inulin-derived oligosaccharides, specifically fructans, consisting of 2 to 60 D-fructose units connected by β-(2,1) linkages. They are naturally existing short-chain carbohydrates with a D glucosyl ending (Apolinário et al., 2014). There are three types of FOS with variant polymerization degrees, and also of different origin and structure: 1) linear long-chain fructan, 2) oligomeric fructan (degree of polymerization from 3 to 9); oligofructose consists of fructosyl chains ending with fructose or glucose monomers and 3) short-chain FOS consisting of different fructosyl chains with a terminal glucose monomer (Csernus and Czeglédi, 2020). Fructo-oligosaccharides at an administration dose of 4 g/kg feed (Xu et al., 2003) have been found to promote the growth of probiotic bacteria in male broilers. However, FOS administered at concentrations of 0.5%, 1% and 2% had no effect on growth and productivity of turkey (Juśkiewicz et al., 2006).
2.3. Galacto-oligosaccharides
Galacto-oligosaccharides are galactose-containing oligosaccharides that exhibit diverse variations in chain length, branching, and glycosyl linkages in their chemical structures. Based on the source of β-galactosidase, when lactose acts as the sole substrate, the galactose released during the enzymatic hydrolysis of lactose can be transferred to another lactose molecule via β (1 → 6), β (1 → 4) or β (1 → 3) glycosidic linkage to the galactose moiety (Mei et al., 2022). A novel GOS mixture (Tzortzis et al., 2005) has been shown to promote the growth of Bifidobacterium and Lactobacillus in pigs.
2.4. Lactulose
Lactulose is an artificial disaccharide produced from lactose through the action of β-galactosidase or epimerase. It is composed of fructose and galactose. Lactulose has low absorption rates in the gastrointestinal tract, and the cells lining the intestinal tract lack digestive enzymes for its hydrolysis (Chu et al., 2022).
2.5. Xylo-oligosaccharides
Xylo-oligosaccharides are oligomers of sugar consisting of β-D-xylopyranosyl (xylose) units connected via β (1 → 4)-xylosidic linkages (Santibáñez et al., 2021). Xylo-oligosaccharides are isolated from xylan-containing lignocellulosic materials, such as crop residues, wood, and herbaceous biomass, known to have prebiotic effects. Xylo-oligosaccharides can also be generated by the addition of exogenous enzymes, such as xylanase, to the lignocellulosic residues (Baker et al., 2021).
2.6. Isomalto-oligosaccharides
Isomalto-oligosaccharides are glucose oligomers that exhibit α-D-(1,6)-linkages, occurring as isomaltose, isomaltotriose, isomaltotetraose, isomaltopentaose, and other oligosaccharides with higher branching. IMO are derived from starch through the action of enzymes belonging to various glycosyl hydrolase families. The enzymes involved in the production of IMO exhibit transferase, hydrolytic or branching activity, leading to a mixture of α-linked gluco-oligomers with varying types of glycosidic linkages and degrees of polymerization (2 to 10) (Logtenberg et al., 2021).
2.7. Polydextrose
Polydextrose is a highly branched glucose polymer with random linkages, characterized by an average polymerization degree of 12, with a range from 2 to 120. The molecule encompasses all possible combinations of α- and β-linked 1 → 2, 1 → 3, 1 → 4, and 1 → 6 glycosidic linkages, although the 1 → 6 (both α and β) linkages are the most common. Polydextrose undergoes partial fermentation in the large intestine, favoring increases in the amount of fecal volume, reduction in transit time, and the softening of fecal material, along with a decrease in fecal pH (do Carmo et al., 2016).
2.8. Pectic oligosaccharides
Pectic oligosaccharides are derived from the depolymerization of pectin. Pectins are branched heteropolymers made up of a linear backbone consisting of α (1–4)-linked D-galacturonic acid (GalA) units, which can undergo random acetylation and/or methylation. These linear structures constitute the “smooth region” of pectins, occasionally interrupted by the “hairy regions” characterized by branches composed of neutral sugars. Pectic oligosaccharides have been suggested as a novel category of prebiotics capable of providing several health-promoting effects, such as the promotion of bifidogenic flora, and antioxidant activity (Wongkaew et al., 2022).
2.9. Gluco-oligosaccharides (GlcOS)
Gluco-oligosaccharides refer to the oligosaccharides consisting of exclusively D-glucose units, exhibiting diverse degree of polymerization and glycosidic linkages. The α-gluco-oligosaccharides (α-GlcOS) are hydrolysates of starch (α-[1 → 4] glucan), while β-gluco-oligosaccharides (β-GlcOS) are hydrolysates of (1,3) (1,4)-β-D-glucan. Both provide a valuable prebiotic source that promote the selective proliferation of probiotic bacteria (Zeng et al., 2023).
3. Effects of prebiotics on gut health
The gastrointestinal tract (GIT) harbors a vast array of live microorganisms collectively referred to as gut microbiota, which play crucial roles in both health and disease (Ursell et al., 2012). Nowadays, the prohibition of the utilization of antibiotics as growth promoters has posed a challenge in animal nutrition, resulting in the search for alternative strategies to manage and prevent the colonization of pathogenic bacteria. The alteration of the gut microbiota using novel feed additives (e.g., probiotics and prebiotics) with host-protective functions to promote animal health is a buzzword in the fields of animal feeding (Gaggìa et al., 2010; Markowiak and Śliżewska, 2018). A healthy gut microbiota has numerous important functions in animal health, including aiding in modulating the gastrointestinal environment, strengthening the immune system, and inhibiting the proliferation of pathogenic bacteria (Anadón et al., 2019; Markowiak and Śliżewska, 2018).
3.1. Proposed mechanisms of action
Prebiotics and probiotics are beneficial for gut health as they help establish a balanced gut microbiota in animals (Arsène et al., 2021; Flickinger and Fahey, 2002; Verdonk et al., 2005). They achieve this through several mechanisms, which can be categorized into two main groups: combating pathogenic bacteria and modulating immune cells in the gut.
Prebiotics and probiotics have been observed to exhibit antagonistic activity against pathogenic bacteria (Arsène et al., 2021; Śliżewska and Chlebicz-Wójcik, 2020). In a study by Śliżewska, the population of several Listeria and Salmonella strains decreased significantly after the addition of a synbiotic mix consisting of Lactobacillus strains and inulin. This suggests that synbiotics possess antimicrobial properties towards pathogenic and harmful bacteria in the gut. Further studies have explored this characteristic and identified multiple mechanisms of action against pathogenic bacteria.
Probiotics possess binding sites for adhesion to glycolipid and glycoprotein receptors, such as toll-like receptors in the host, which can block and compete for adhesion sites with various pathogens (Arsène et al., 2021; Pandey et al., 2015). This reduces the likelihood of harmful bacteria entering the bloodstream through adhesion. Some probiotic strains also boost mucus-binding abilities in the gut by having binding protein molecules on their cell surfaces, which enhances their attachment to the mucosal layer. This provides probiotic bacteria a higher chance of competing for nutrients with pathogenic bacteria as they colonize the gut and are less likely to be carried away by the chyme in the intestinal tract (Arsène et al., 2021; Peng et al., 2019). Prebiotics and probiotics can also induce mucin production in the gut, further enhancing their mucus-binding properties. Probiotics are shown to upregulate mucin production from goblet cells in the gut through inflammatory pathways, while prebiotics encourage mucin production to aid the colonization of probiotic bacteria (La Fata et al., 2018; Markowiak and Śliżewska, 2017). The thickening of mucin strengthens the gut barrier function, protecting intestinal cells against the invasion of pathogenic bacteria and regulating the homeostasis of water, antimicrobial, and immune molecules.
Probiotics also produce antibacterial substances, such as short-chain fatty acids (SCFAs) and lactic acid, which help maintain a low pH environment in the gut that inhibits the survival of pathogenic bacteria (Markowiak and Śliżewska, 2018; Peng et al., 2019; Qamar et al., 2016). Some probiotic species produce antibacterial peptides, including bacteriocins and antibiotics, which are effective in inhibiting the proliferation of many harmful bacteria, such as nisin, lacticin, pediocin, and enterocin (Peng et al., 2019). Butyric acid, a type of SCFA, can promote the production of antimicrobial peptides (Chibbar et al., 2017). Probiotics can also disintegrate bacterial toxins and block toxin receptors, metabolize certain toxins, and bind to them, preventing them from coming into contact with intestinal cells and disrupting the gut microbiota (Arsène et al., 2021; Markowiak and Śliżewska, 2017).
Short-chain fatty acids, particularly butyrate, have been shown to inhibit inflammatory pathways. Butyrate uptake into intestinal cells inhibits the ERK/MAPK pathway and downregulates NF-κB dependent gene expression, which reduces the expression of proinflammatory cytokines like TNF-α and IL-6, while increasing the expression of anti-inflammatory IL-10. Butyrate also inhibits HDAC, mediating and reducing inflammation responses in the gut through inhibition of the above two signalling pathways (Nawaz et al., 2018; Parada Venegas et al., 2019). Short-chain fatty acids have also been found to stimulate the expression of anti-inflammatory PGE2 (Cox et al., 2009). Pectin, a mixture of plant polysaccharides, inhibits pro-inflammatory signals in dendritic cells by impeding the TLR signalling pathways, which helps regulate and control inflammation in the gut (Nawaz et al., 2018; Sahasrabudhe et al., 2018).
3.2. Interaction of prebiotics with the host gut microbiota
Prebiotics are short-chain carbohydrates that are resistant to digestion by enzymatic methods but selectively enhance the activity of certain probiotic bacteria in the gut (Anadón et al., 2019; Sarkar, 2007). These carbohydrates include disaccharides, oligosaccharides, polysaccharides, resistant starch, and some sugar polyols, and have a low level of polymerization and a linear shape (Al-Sheraji et al., 2013). They are non-digestible as they have glycosidic bonds that are resistant to digestion, allowing them to be selectively fermented by bacteria in the gut. Mechanistically, as presented in Fig. 2, prebiotics have a promising effect on gut health in both polygastric and monogastric animals (Azad et al., 2020; Zapata et al., 2021). Polygastric animals, such as ruminants, possess a unique digestive system characterized by a specialized fermentation chamber called the rumen. Prebiotics exert their effects in polygastric animals by modulating the microbial population and activity within the rumen. When prebiotics are introduced into the diet of ruminants, they serve as a substrate for specific beneficial microorganisms, promoting their growth and activity. These microorganisms, including fibrolytic bacteria and protozoa, possess the ability to ferment complex carbohydrates and fiber into SCFAs, such as acetate, propionate, and butyrate. During fermentation, combustible gases, lactate, and beneficial SCFAs including acetic, propionic, and butyric acids are produced (Gibson et al., 2017; Sarkar, 2007). Short-chain fatty acids are important for the growth of beneficial bacteria in the gut, preventing dysbiosis, and exerting immunomodulatory effects in the host (Al-Sheraji et al., 2013; Gibson et al., 2017). This modulation of the rumen microbiota by prebiotics enhances the efficiency of nutrient utilization, improves gut health, and reduces the production of methane, a potent greenhouse gas emitted by ruminants (Michalak et al., 2021). In monogastric animals, prebiotics primarily act on the gastrointestinal tract by selectively stimulating the growth and activity of beneficial bacteria. Upon consumption, prebiotics resist digestion in the upper gastrointestinal tract and reach the colon, where they serve as a substrate for specific groups of bacteria, such as Bifidobacteria and Lactobacilli (Gaggìa et al., 2010). These bacteria ferment prebiotics, producing SCFAs. Furthermore, SCFAs have been found to influence the secretion of gut hormones, such as peptide YY (PYY) (Browne and Horgan, 2024; Tellez et al., 2020) and glucagon-like peptide-1 (GLP-1) (Azad et al., 2020; Bachmann et al., 2021), which regulate appetite, satiety, and glucose homeostasis. The fermentation of prebiotics also leads to a decrease in the luminal pH, creating an environment unfavourable for the growth of pathogenic bacteria (Jha et al., 2019). Additionally, prebiotics can enhance the production of mucins, which are protective substances that form a physical barrier in the gut, and stimulate the immune system, contributing to improved gut health and overall well-being in monogastric animals (Azad et al., 2020). Sources of prebiotics typically come from breast milk, plants, microbiological, and enzymatic activities (Sarkar, 2007). Common examples of prebiotics include lactulose, GOS, FOS, inulin, and its hydrolysates, and mannan-oligosaccharides (MOS). In the following paragraphs, three of these prebiotics as examples will be discussed.
Fig. 2.
Health benefits and mechanisms of prebiotics in polygastric and monogastric animals. SCFAs = short-chain fatty acids; PYY = peptide YY; GLP-1 = glucagon-like peptide-1.
The beta glycosidic bonds in FOS render them indigestible until they reach the small intestine (Chen et al., 2016). Fructo-oligosaccharides include 1-ketose, nystose, and 1-fructofuranosylnystose, and belong to the same family of carbohydrates with varying degrees of polymerization and chain length (Martins et al., 2019). Fructo-oligosaccharides were recognized as prebiotic components in the 1990s due to their gut-promoting and selective fermentation properties, evidenced through their ability to stimulate the growth of bifidobacterial strains in the gut (Anadón et al., 2019; Sarkar, 2007). Fructo-oligosaccharides and oligofructose are produced through partial enzymatic hydrolysis of inulin and exhibit similar physical and physiological effects. Like FOS, inulin due to the beta glycosidic bonds also remains undigested in the upper digestive tract, providing physical benefits. In terms of physiological effects, inulin has been shown to improve gut health by altering the composition of fecal metabolites and exerting immunomodulatory and therapeutic effects on the gut in animal (Anadón et al., 2019; Flickinger and Fahey, 2002; Markowiak and Śliżewska, 2018; Meyer and Stasse-Wolthuis, 2009). Like FOS and inulin, both types of GOS, as α-GOS, found in plant sources and β-GOS from lactose (Martins et al., 2019), have galactosidic bonds that render them non-digestible in the upper digestive tract, promoting their fermentation in the colon (Verdonk et al., 2005).
4. Effects of prebiotics on animal health
Nutritional feed additives encourage the establishment of beneficial bacteria. They have been found to strongly influence the progression of the intestinal epithelium while modulating the physiological functions needed to maintain immunity, the digestion of nutrients and the fortitude of the intestinal barrier which favourably affects the growth performance of animals (Al-Baadani et al., 2021). Given that there is a ban on antimicrobial growth promoters, it is necessary to employ different methods to prevent and control the colonization of pathogenic bacteria in animals (Anadón et al., 2019). Using antimicrobial growth promoters in animal feed have aided in the increase of multidrug-resistant microorganisms in livestock and poultry; resulting in animal products that contain antibiotic residues (Bosco, 2019; Forgetta et al., 2012; Mazhar et al., 2021; Mohammadigheisar et al., 2019). The existence of this ban has propelled the scientific community to find alternative products such as prebiotics, not only because of the above mentioned reasons, but also for the well-being of the animals as it is crucial to achieve and maintain good gut health (Al-Baadani et al., 2021; Angwech et al., 2019; Vasanthakumari et al., 2023).
Prebiotics are able to protect the epithelial cells from harmful microorganisms by acting as receptors that assist with the modulation of the host's immune system; controlling inflammation (Ahmadifar et al., 2019; Mohammadi et al., 2022). Insoluble fibres that are isolated from legumes, plants, grains, fruits and cruciferous vegetables are the usual sources of prebiotics; while they may not always be of nutritive value, they carry out functions such as stimulation of the intestine and selection of the microbiota composition Leone and Ferrante, 2023). The most frequently used prebiotics are non-starch polysaccharides (NSP) or oligosaccharides such as alginate oligosaccharide (AlOS), arabinoxylo oligosaccharides (AXOS), FOS, galacto-oligosaccharides (GOS), isomalto-oligosaccharides (IMO), inulin, MOS and xylo-oligosaccharides (XOS) (Azad et al., 2020; Jahan et al., 2022).
According to Azad et al. (2020), inulin, FOS, GOS, trangalacto-oligosaccharides, and lactulose are the prebiotics primarily studied in animal health promotions, used as feed additives given that they are easily fermented in the colon and result in decreased luminal pH and increased SCFA production.
Furthermore, as stated by Patel and Goyal (2012), most prebiotics stimulate bifidogenic and lactic acid bacteria in animals. Prebiotics serve as substrates for fermentation and attach themselves to pathogens. They can increase osmosis in the lumen of the intestine, stimulate the macrophages’ response and the production of SCFAs indirectly, while modulating the immune system. Whenever evaluating the effects prebiotics have on animal health, the following should be taken into consideration: the anatomy and physiology of the animal, how much they rely on the gut microbiota as an energy source, their living environment and dietary composition (Gibson et al., 2017).
4.1. Effects of prebiotics on growth performance and nutrient digestion
Gastrointestinal health is one of the primary objectives to enhance production performance, given its role in crucial biological processes. Sustaining gastrointestinal functionality is indispensable for promoting optimal growth, normal and stable microbiota, effective immune status, and nutrient utilization efficiency (Vasanthakumari et al., 2023). A healthy intestine is necessary to allow proper nutrient digestion and absorption in the host, leading to reduced excretion of metabolites (Peng et al., 2020). The maintenance of intestinal health is attributed to the microbiota that reside within the gastrointestinal tract (Celi et al., 2019).
Beneficial effects of prebiotics on growth performance likely result from the interplay and interaction of several mechanisms, including decreased pathogen colonization, increased SCFA production, alterations in lipid metabolism, and improved mineral absorption (Solís-Cruz et al., 2019). While prebiotics hold the potential to enhance overall intestinal health, their effects on growth performance may be dependent on the broader husbandry management practices employed (Anadón et al., 2019). Nonetheless, nutritional interventions targeting the modulation of gastrointestinal health hold promise as a means to boost a variety of farm animal production parameters, such as growth rates, meat and egg quality, and milk yield.
4.1.1. Monogastric animals
Monogastric animals have one simple, single-chambered stomach of which horses, poultry and pigs are the most popular agricultural species (Ojha et al., 2019). Scientific evidence shows that prebiotics have been used in animal feed for poultry and pigs since the 1990s; with the majority of such research being in poultry (Di Gioia and Biavati, 2018). The prebiotics most commonly used in monogastric animals are FOS, MOS, GOS and inulin; with FOS being more widely investigated than GOS (Park et al., 2013; Rehman et al., 2009).
According to Solís-Cruz et al. (2019), bacteria such as Lactobacillus spp. and Bifidobacterium spp. that form a part of the intestinal microbiota in poultry, confer positive effects on the intestinal physiology and are beneficial to their health. These beneficial bacteria have reportedly increased when chickens were fed diets supplemented with prebiotics—they fermented and metabolized the prebiotics and thus increasing their activity.
Studies by Totton et al. (2012) and Park et al. (2013) found that prebiotics regulated the intestinal microbiota of chickens by selectively stimulating the beneficial bacteria and inhibiting the unwanted bacteria like Salmonella which resulted in an increase of their stool volume. Prebiotics, including FOS and MOS, were found to increase body weight in poultry along with the quantities of beneficial bacteria such as Lactobacillus and Bifidobacterium spp. while decreasing the quantities of pathogenic bacteria like Escherichia coli, Salmonella and Clostridium perfringens (Chambers and Gong, 2011; Lee et al., 2012; Stanley et al., 2014). Angwech et al. (2019) reported that in ovo delivered prebiotics (3.5 mg/embryo on the 12th d of incubation) diminished the severity of intestinal lesions and oocyst excretion-induced infections by Eimeria (parasites capable of causing the disease coccidiosis) in Kuroiler chickens. The chickens were protected from coccidia for the first 56 d of their lives which worked in synergy with an anticoccidial drug to manage the disease post-infection. This resulted in positive effects on the performance of the birds and the quality of their meat. On the other hand, Wan et al. (2018) found that pigs supplemented with AlOS at 100 mg/kg for 2 weeks exhibited increased villus height, villus height-to-crypt depth ratio and goblet cell counts. Similar examples are cited in Table 1.
Table 1.
The effects of feeding prebiotics on animal nutrition and health in monogastric animals.
| Prebiotics | Animal | Dosage | Duration | Effects of prebiotics on gut microbes and immune responses | References |
|---|---|---|---|---|---|
| TOS | Pigs | 35 g/kg | 42 d | Increased the count of stool Bifidobacterium and Lactobacillus. | Smiricky-Tjardes et al. (2003) |
| FOS | Broiler chickens | 4 g/kg | 49 d | Increased daily growth and growth of Bifidobacterium and Lactobacillus. Inhibited the growth of Escherichia coli in gastrointestinal tract. | Xu et al. (2003) |
| FOS | Male turkeys | 2% | 56 d | A reduction of the intestinal pH. | Juśkiewicz et al. (2006) |
| FOS | Weaned piglets | 0.6% | 7 d | Increased intestinal microbes, Bifidobacteria and Lactobacillus in the middle jejunum, along increased IFN-γ levels. Decreased abundance proteobacteria in posterior jejunum and ileum. Decreased levels of IL-4 and IL-10. | Chang et al. (2018) |
| scFOS | Adult pigs | 10 g/d | 28 d | Increased abundance of Bacteroidetes and Prevotella, and decreased abundance of Ruminococcaceae. | Le Bourgot et al. (2019) |
| AOS | Pigs | 100 mg/kg | 14 d | Increased villus height, villus height-to-crypt depth ratio and goblet cells. | Wan et al. (2018) |
| FOS | Salmonella challenged laying hens | 0.5% and 1.0% | 3 wk | Increased the gene expression of TLR-4 and IFN-γ, along with IgA+ cells in the ileal lamina propria. Decreased ceca Salmonella in mature hens. | Adhikari et al. (2018) |
| GOS | Kuroiler chickens | 3.5 mg/embryo | 12th d of incubation until hatched | Reduced severity of intestinal lesions and oocyst excretion induced by natural infection with Eimeria, with positive effects on the chicken's productive traits. | Angwech et al. (2019) |
| MOS + MP | Broiler chickens (Ross 308) | 100 g/t of diet | 35 d | Longer jejunal villus length, positive changes in microbial population and higher level of calcium in blood. Increased serum concentration of calcium. | Karimian and Rezaeipour (2020) |
| Saccharomyces-derived prebiotics | Broiler chickens | 50 and 100 g/t | 42 d | Decreased the prevalence of Camphylobacter. | Froebel et al. (2019) |
| XOS | Pigs | 100 to 500 g/t | 70 d | Decreased relative abundance of Proteobacteria, Citrobacter, along with increased relative abundance of Firmicutes, Lactobacillus during the growing and fattening period. Also, increased SCFAs concentrations in the intestine. | Pan et al. (2019) |
| XOS | Weaned piglets | 0.01% | Weaned | Increased relative abundance of Streptococcus and Turicibacter, along decreased relative abundance of Lactobacillus. Reduced IFN-γ concentration and higher expression of ZO-1 in the ileum. | Yin et al. (2019) |
TOS = trans-galactooligosaccharides; FOS = fructo-oligosaccharides; scFOS = short-chain fructo-oligosaccharides; AlOS = alginate oligosaccharide; GOS = galacto-oligosaccharides; MOS = mannan-oligosaccharides; MP = microbial phytase; XOS = xylo-oligosaccharides; IFN = interferon; IL = interleukin; SCFAs = short-chain fatty acids; ZO = zonula occludens; Ig = immunoglobulin.
Recently, novel precision biotics (PB) have being actively researched for their ability to improve growth performance and control nitrogen emissions (Jacquier et al., 2022; Walsh et al., 2021). This novel class of nutritional feed ingredients function as microbiome metabolic modulators (Jacquier et al., 2022; Walsh et al., 2021). The mechanism of action of PB is through the modulation of metabolic pathway abundance, rather than the modulation of bacterial taxa or overall gut microbial profiles, which differentiates them from existing microbiome-targeting technologies (Bortoluzzi et al., 2023). This targeted modulation promotes beneficial outcomes for both the animal and the environment. Several studies have focused on the impact of a glycan-based PB in the poultry industry. Walsh et al. (2021) and Jacquier et al. (2022) have reported the effects of supplementation of this mixture in broiler chickens. Walsh et al. (2021) evaluated, through meta-analysis, the effects of two structurally distinct, precision glycan microbiome metabolic modulators (MMMs) on the performance of broiler chickens. Microbiome metabolic modulator 1 and MMM2 increased final body weight compared to the negative control by 43 and 48 g/bird, respectively. The different structure of MMMs had an impact on size and performance effects in broiler chickens. Metagenomic analysis confirmed an increase in the abundance of genes belonging to the acrylate pathway, involved in propionate production (fatty acid biosynthesis) in response to both MMMs. Arginine-N-succinyl transferase genes, involved in arginine catabolism, were also upregulated in response to MMM2 (Walsh et al., 2021). Jacquier et al. (2022) conducted a two-part experiment to examine the effects of PB. In the first trial, they tested the main effects of PB dose (0, 250 and 500 g/t) and xylanase supplementation (0 or 100 g/t) in boiler chickens. No significant interaction was detected between PB and xylanase on performance. However, supplementation with PB at 250 and 500 g/t improved feed conversion ratio by 7 and 11 points compared to diets without glycans. The glycan effect was observed to depend on the age of the birds. At 35 d, PB reduced the pH and ammonia concentration in diets with xylanase. The second experiment explored the effect of a single PB dose (500 g/t) compared to a control diet on broiler performance. In both trials, the inclusion of 500 g of glycan PB per tonne feed resulted in a greater proportion of birds without foot pad lesions or defective gaits. The glycan supplement, by targeting the modulation of microbial metabolic pathways related to SCFAs and amino acid metabolism, may have facilitated improved nitrogen utilization in the broilers, thereby reducing ammonia emissions and enhancing litter quality (Jacquier et al., 2022). Additionally, Bortoluzzi et al. (2023) tested the mixture of glycan-based PB to modulate the utilization of proteins and improve the growth performance of broiler chickens. It was observed that supplementation with 3% of PB in the diets had a positive impact on performance, with improved body weight gain and the corrected feed conversion ratio (cFCR). The functional microbiome analysis revealed that PB shifted the microbiome pathways toward a beneficial increase in protein utilization, and desirable pathways related to nitrogen utilization, resulting in improved growth performance (Bortoluzzi et al., 2023).
4.1.2. Polygastric animals
The stomachs of ruminants consists of four compartments: the rumen, reticulum, omasum and the abomasum; with each chamber performing different processes (Stover et al., 2016). Prebiotics effectively alter the activity and composition of the microbiome in the ruminant's gastrointestinal tract because they are substrates that enhance the presence of certain beneficial ruminal microorganisms. However, because ruminants are capable of catabolizing most of the common prebiotic compounds, using prebiotics to promote growth in the ruminant industry has been limited (Bąkowski and Kiczorowska, 2020). Additionally, several nondigestible oligosaccharides naturally found in cell walls of plants are included in ruminant feeds, possibly making the implementation of prebiotics in ruminants unnecessary (Anadón et al., 2019; Di Gioia and Biavati, 2018). Nonetheless, using prebiotics in pre-weaned ruminants (given that they are fed a low-roughage diet) have been found to increase their growth, diminish the chances and severity of diarrhoea and respiratory diseases while aiding in the formation of a desirable intestinal flora, which can improve their performance as they age (Ghosh and Mehla, 2012; Roodposhti and Dabiri, 2012; Uyeno et al., 2015).
Prebiotics such as galactosyl lactose, cello-oligosaccharides, MOS and yeast cell wall extracts are used in pre-weaned calves. It is believed that MOS block the colonization of pathogens in the digestive tract, while FOS along with spray-dried bovine serum reduced the occurrence of enteric disease in calves due to the ability of this sugar to block the adhesion of E. coli and Salmonella to the intestinal epithelium. It was reported that adding galactosyl-lactose (GL) to milk replacer positively impacted the growth and general health of dairy calves. While further investigation is necessary, research has shown that supplementation with MOS, FOS and GL may serve to improve the growth performance of calves in both pre- and post-weaning stages (Bhat and Shaheen, 2017; Uyeno et al., 2015).
In Table 2, Patel et al. (2019) found that Surti buffalo calves who received MOS (10 g/d) experienced increased body weight, average daily gain and feed conversion ratio. While, Alves Costa et al. (2019) found that calves who received MOS (5 g/d for 56 d) experienced significant increases in jejunal villus height and rumen papillae. Similar examples are cited in Table 2.
Table 2.
The effects of feeding prebiotics on animal nutrition and health in polygastric animals.
| Prebiotics | Animal | Dosage | Duration | Effectsof prebiotics on gut microbes and immune responses | References |
|---|---|---|---|---|---|
| MOS | Holstein calves | 4 g/d in milk replacer | 42 d | Improved feed intake and fecal scores. | Heinrichs et al. (2003) |
| MOS (Bio-MOS) | Young calves | 4 g/d in milk replacer | 42 d | Stimulated calf starter intake. Decreased presence of Cl. Perfringens, E. coli and Salmonella spp. | Terré et al. (2007) |
| MOS | Holstein cross calves | 4 g/d | 56 d | Average body weight gain, feed intake and feed conversion efficiency increased. Significant decrease in severity of scours as measured by fecal score and fecal coliform count. | Ghosh and Mehla (2012) |
| scFOS | Veal calves | 3 or 6 g in milk replacer | 168 d | Increased growth performance, carcass quality, faecal concentration of SCFAs and microbial fermentation. | Grand et al. (2013) |
| GOS | Holstein calves | 33.5 mg/g | 56 d | Increased Lactobacillus and Bifidobacterium bacteria. Increased colon crypt depth and jejunal villus length. |
Castro et al. (2016) |
| MOS | Surti buffalo calves | 10 g/d per calf | Increased body weight, average daily gain, feed conversion ratio. | Patel et al. (2019) | |
| MOS | Calves | 5 g/d | 56 d | Significant increases in jejunal villus height and rumen papillae. | Alves Costa et al. (2019) |
| CWS | Calves | 50 g/d | 84 d | Overall improvement in starter intake, postweaning body weight, β-hydroxybutyrate, maintained frame growth and fecal scores. | Senevirathne et al. (2019) |
| Y-MOS | Male Zell lambs | 1.5 g/d in milk replacer | 70 d | Caused feed intake and daily weight gain improvements. Improved immune function (specifically an elevation in serum IgG). | Chashnidel et al. (2020) |
| Inulin | Female calves | 12 g/d in milk replacer | 63 d | Significantly increased faecal concentration of total anaerobic bacteria and Lactobacillus. Increased total aerobic bacteria count in faeces. | Tóth et al. (2020) |
| MOS and β-glucans | Pelibuey × Katahdin crossbred male lambs | 3 g/d per lamb | 93 d | Resulted in enhanced feed efficiency by 6.9% and dietary net energy by 5.9%. | Zapata et al. (2021) |
| Alhagi camelorum fisch polysaccharides from Shanshan county, Xinjiang Uyghur Autonomous region, China | Sheep lambs | 1.5 g/kg | 28 d | Increased levels of GH, INS, IgA and IgM, and cytokines IL-4, IL-10, IL-17, TNF-α and IFN-γ. Various metabolic pathways were modulated; growth was promoted; immunity improved; the risk of gastrointestinal disease was reduced; improved composition of gastrointestinal microbiota observed. | Manafu et al. (2024) |
MOS = mannan-oligosaccharides; scFOS = short-chain fructo-oligosaccharides; GOS = galacto-oligosaccharides; CWS = condensed whey solubles-product containing proprietary prebiotics derived from milk; Y-MOS = mannan oligosaccharides and β-glucans, extracted from cell wall of the yeast (Saccharomyces cerevisiae); SCFAs = short-chain fatty acids; GH = growth hormone; INS = insulin; Ig = immunoglobulin; IFN = interferon; IL = interleukin; TNF = tumor necrosis factor.
5. Agro-industrial residues and processing as sources of prebiotics
Agro-industrial residues are produced in large volumes on an annual basis and represent both an environmental burden and a potential source of value creation. Despite being considered a source of environmental pollution (Das et al., 2018), these organic residues have high nutritional content and bioactive compounds, making them valuable resources for application in animal feed, bioenergy production, and biorefinery processes (Yaashikaa et al., 2022).
Agro-industrial residues are categorized as agricultural (e.g. leaves, stems, seeds) and industrial residues (e.g. food processing waste) (Sadh et al., 2018). Agro-industrial residues are mainly formed of lignocellulosic biomass. Lignocellulose is a source of functional oligosaccharides that can promote the proliferation of probiotic microorganisms, including XOS, arabino-oligosaccharide (AOS), and MOS.
Given their rich nutrient content, agricultural residues provide a valuable source of raw material for prebiotic production.
5.1. Agro-industrial residues as sources of prebiotics
Table 3 summarizes agro-industrial wastes used to obtain prebiotics. These residues come primarily from food processing industries. A list of agro-industrial wastes used in prebiotic production is further discussed.
Table 3.
Examples of prebiotic types, effects and potential substrate sources used for animal health.
| Type of prebiotics | Physiological effects | Agro-industrial residues | References |
|---|---|---|---|
| FOS | Increase in growth performance; improvement in gut microbiota composition; positive immunological effects | Sugarcane bagasse Sugarcane molasses Olive pomace powders Apple pomace, wheat bran, banana peels, guava peels, corn straw, cassava peels, and beetroot peels Jerusalem artichoke tubers Flesh of onion bulbs Flesh and peel of red pitaya, white pitaya, and papaya Coffee silverskin, corn cobs, and cork oak |
de la Rosa et al. (2019) Khatun et al. (2021) Ribeiro et al. (2021) Ganaie et al. (2017) Khuenpet et al. (2017) Mabrok et al. (2018) Khalili et al. (2014) Mussatto and Teixeira (2010) Abd El-Aziz et al. (2022) Csernus and Czeglédi (2020) Quijada et al. (2020) |
| MOS | Improvement of intestinal morphological traits; immune modulator; improvement in gut microbiota composition | Spent coffee grounds brewed coffee Copra meal Soybean meal Sugar palm fruit Palm kernel cake |
Tian et al. (2017) Chantorn et al. (2018) Pangestu et al. (2019) Purnawan et al. (2017) Duan et al. (2019) Youssef et al. (2023) Yang et al. (2022) |
| XOS | Improvement in gut microbiota composition; improvement of nutrient digestibility and energy metabolism; growth performance | Sugarcane bagasse Olive pomace Grape pomace |
Kathiresan et al. (2021) Freitas et al. (2022) Costa et al. (2019) González-Solé et al. (2022) Rao et al. (2024) Dong et al. (2023) |
| GOS | Increase in growth performance; improvement in gut microbiota composition and immune response; reduce the incidence of diarrhea | Whey permeate Milk whey Milk whey permeate Spent coffee grounds Brewed coffee |
Mano et al. (2019) Yañez-Ñeco et al. (2021) Hackenhaar et al. (2021) Tian et al. (2017) Chang et al. (2022) Yu et al. (2023) Tian et al. (2022) |
| Inulin | Increase in growth performance; improvement in gut microbiota composition; enhance immune function | Chicory roots Flesh of onion bulbs |
Chikkerur et al. (2020) Mabrok et al. (2018) Tian et al. (2019) Xia et al. (2021) |
| Polyphenols | Increase in growth and antioxidant performance; enhance immune function | Grape pomace Cranberry pomace Blueberry pomace Apple pomace Mango peels and seed kernels |
Meini et al. (2021) Yang et al. (2023) Costa et al. (2022) Nicolás García et al. (2023) Serra et al. (2021) Waqas et al. (2023) |
| SCFAs | Contribute to energy requirements; pathogen inhibition | Cranberry pomace |
Islam et al. (2020) Wong et al. (2006) Hackmann (2023) |
FOS = fructo-oligosaccharides; MOS = mannan-oligosaccharides; XOS = xylo-oligosaccharides; GOS = galacto-oligosaccharides; SCFAs = short-chain fatty acids.
5.1.1. Sugarcane by-products
Sugarcane bagasse, major by-product of sugarcane industry, reaches 513 million tonnes annually (Toscano Miranda et al., 2021). Sugarcane bagasse is composed of 35% to 50% cellulose, 20% to 25% hemicellulose, 15% to 25% lignin, moisture, and 4% to 6% ash content (Kolawole et al., 2021). Studies have extracted xylan from bagasse and enzymatically hydrolyzed it to generate XOS with prebiotic potential (Kathiresan et al., 2021). Xylo-oligosaccharides from sugarcane bagasse have shown to promote the growth of probiotic bacteria like Lactiplantibacillus plantarum, Lactobacillus brevis, Lactobacillus acidophilus and Lactobacillus viridescens, and exhibit antioxidant and antibacterial activities (Gupta et al., 2022; Kaur et al., 2019). The prebiotic effect of XOS was attributed to their degree of polymerization and the presence of xylobiose and xylotriose (Kaur et al., 2019). Sugarcane bagasse has also been utilized as biomass for the production of fructosyltransferase (Ganaie et al., 2017), an enzyme used to commercially produce FOS.
Another sugarcane by-product, molasses, has been explored for prebiotic production. Enzymatic synthesis of prebiotic oligosaccharides from molasses has been performed using dextransucrase from Leuconostoc mesenteroides MTCC 10508 (Sharma et al., 2016), and whole-cell biocatalyst of Aspergillus pullulans have been used to transform molasses into FOS (Khatun et al., 2021). Overall, these studies have demonstrated the potential of sugarcane by-products, including bagasse and molasses, as valuable sources of prebiotics with attractive health properties for a tailored use. Further research is still needed to fully understand the effects of these derived prebiotics on animal health when used as feed.
5.1.2. Pomace
Pomace, a major food industry residue, is a valuable source of bioactive compounds, including polyphenolics, fibre, vitamins, and carbohydrates. Pomace yield varies by fruit, with grape pomace comprising typically 20% to 30% of the fruit weight, orange pomace 45% to 60%, apple pomace 25% to 35%, olive pomace 30%, and berry pomace 30% to 50% (Gowman et al., 2019; Joshi, 2005; O'shea et al., 2015; Rodríguez-Gutiérrez et al., 2012; Struck et al., 2016). Studies show that pomace can potentially enhance beneficial gut bacteria in monogastric animals like poultry (Mnisi et al., 2022). Olive pomace has been found to promote the production of SCFAs by gut microbiota and exhibits antimicrobial activity (Ribeiro et al., 2021).
Polyphenols with prebiotic activity have also been extracted from grape pomace (Meini et al., 2021). Supplementing broiler chickens’ diet with cranberry pomace has been shown a positive effect on the population of probiotic bacteria and reduce the abundance of pathogens (Islam et al., 2020). The addition of cranberry and blueberry pomace to broiler chicken feed has been found to increase the relative population of Lactobacillus, a beneficial type of bacteria (Yang et al., 2023). Other studies have shown the potential of various fruit pomaces and agro-residues as a suitable source of polyphenols and prebiotic fibres that can benefit animal health and nutrition (Costa et al., 2022; Ganaie et al., 2017; Vlad et al., 2022).
The use of pomace from various sources is now common as a source of biocompounds, due to research revealing its health-promoting properties. Although relevant and positive effects on animal welfare have been disclosed, the potential use of pomace is still at a basic level requiring more investment in fundamental research.
5.1.3. Other residues
The potential prebiotic effects of phenolic compounds derived from mango peels and seed kernels have been discussed (Nicolás García et al., 2023). These compounds offer health benefits, including prebiotic effects on the gut microbiota. Studies have investigated the potential of Jerusalem artichoke (Helianthus tuberosus L.) tubers and chicory roots as sources of prebiotics like inulin and FOS (Chikkerur et al., 2020; Khuenpet et al., 2017). Onions and pitaya fruits are additional sources of FOS and inulin, with the red pitaya showing the highest production of FOS compared to white pitaya and papaya (Khalili et al., 2014; Mabrok et al., 2018).
Studies have evaluated coffee waste materials, like spent coffee grounds and brewed coffee, as sources of oligosaccharides such as GOS and MOS (Nguyen et al., 2019; Tian et al., 2017; Wongsiridetchai et al., 2021). Mannan-oligosaccharides (MOS) derived from spent coffee grounds were found to promote Lactobacillus growth, and inhibit pathogens (Wongsiridetchai et al., 2021). Other agricultural wastes, including potato peels, soybean meal, sugarcane bagasse, and palm products, have also been used to produce MOS with potential prebiotic and antimicrobial effects (Chantorn et al., 2018; Pangestu et al., 2019; Purnawan et al., 2017).
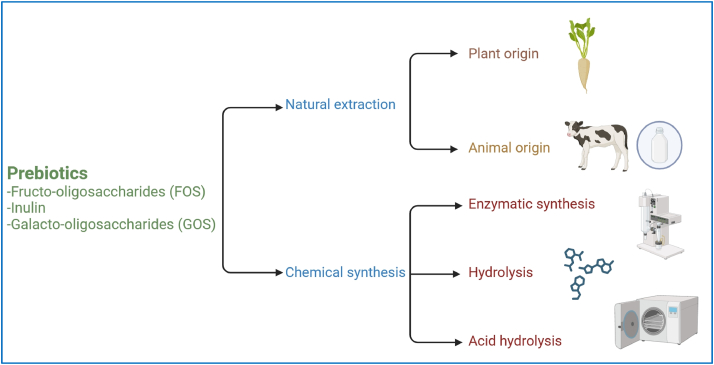
5.2. Prebiotic production from agro-industrial residues
Prebiotic can be produced from agro-industrial waste through enzymatic technologies, fermentation, and acid hydrolysis (Bamigbade et al., 2022; Gonçalves et al., 2023; Patel and Shukla, 2017). Enzymatic hydrolysis uses specific enzymes to gently break down carbohydrates into prebiotic like oligosaccharides, preserving functionality (Radenkovs et al., 2018). Fermentation uses microorganisms to metabolize waste carbohydrates into prebiotic like FOS or GOS (Gonçalves et al., 2023; Patel et al., 2020; Patel and Shukla, 2017). Acid hydrolysis employs acids to break down carbohydrates into prebiotics (Bamigbade et al., 2022), breaking glycosidic bonds and releasing monosaccharides or smaller oligosaccharides, but it requires careful handling due to its corrosive nature. The treatment conditions depend on the type of waste being processed.
As shown in Fig. 3, the two main industrial methods used for producing prebiotics are: 1) natural extraction or hydrolysis from plant or milk sources and 2) chemical synthesis pathways using enzymatic or isomerization reactions (Al-Sheraji et al., 2013). However, natural sources have limited prebiotic content, so most commercial prebiotics are chemically synthesized from raw materials like lactose and sucrose (Davani-Davari et al., 2019). The manufacturing process of FOS and GOS will be discussed in the following paragraphs.
Fig. 3.
The pathways of prebiotic production.
Fructo-oligosaccharides can be produced through enzymatic synthesis or inulin hydrolysis (Martins et al., 2019). Enzymatic synthesis involves the use of fructosyltransferase, such as β-fructofuranosidase from Aspergillus niger. This enzyme cleaves glycosidic bonds and transfers fructose from sucrose to form FOS (Davani-Davari et al., 2019). Free enzymes are typically preferred over those within the microorganism, as they simplify extraction and purification. Glucose oxidase may also be added to prevent glucose build-up, which can inhibit the conversion to FOS (Martins et al., 2019).
Inulin hydrolysis is another approach, and can be performed through enzymatic or acid-based methods. Enzymatic hydrolysis, the most common industrial method, uses endo-inulinase and exo-inulinase enzymes from fungal sources to break down inulin into shorter-chain compounds (Martins et al., 2019). Acid hydrolysis is used when enzymatic methods are not feasible, breaking down inulin in a highly acidic environment to produce FOS (Martins et al., 2019).
Galacto-oligosaccharides can be produced through enzymatic synthesis or hydrolysis, with enzymatic synthesis being the more common industrial method. In enzymatic synthesis, α-galactosidase and β-galactosidase enzymes derived from fungi catalyze transgalactosylation reactions and produce α-GOS and β-GOS, respectively (Martins et al., 2019). These enzymes can be used in both free form and within microorganisms, but they are expensive, so recombinant β-galactosidase is often introduced to enhance yield and simplify processing (Davani-Davari et al., 2019). Lactose and glucose are produced as by-products, which can lower GOS yield and inhibit the reaction. Techniques like chromatography, and precipitation are employed to remove these by-products and optimize the process (Martins et al., 2019). Hydrolysis is mainly used for plant-derived α-GOS, where long polymer chains are hydrolysed to break apart the galactosidic bonds and produce compounds like sucrose, stachyose, and verbascose. Purification and extraction steps are then taken to obtain a higher yield of α-GOS by removing fat, proteins, and fibre (Martins et al., 2019).
Techniques to produce prebiotics from agro-industrial residues are diverse and have been developed on an industrial scale. These methods can secure a production rate compatible with applications in both animal and human nutrition without compromising the need of either. Future work should focus on developing products that integrate or include these prebiotics in applications for animal feeds, fostering animal welfare.
6. Conclusions and future perspectives
In conclusion, with the ban on antimicrobial growth promoters, the utilization of prebiotics derived from agro-industrial residues has emerged as a promising and safe alternative for maintaining animal health and well-being through optimal nutrition. These agro-industrial residues, which may be detrimental to human health and the environment, can be repurposed as valuable resources for prebiotic production.
Prebiotics have been recognised as possessing numerous health-promoting properties that are highly relevant in the management of animal care and well-being through proper nutrition. Prebiotics can be categorized into different groups based on their development and regulatory status, ensuring that their production or extraction from various sources is conducted accordingly. Considering the ban on antimicrobial growth promoters, prebiotics offer an interesting and safe alternative in the treatment of health issues in animal production. Numerous studies have demonstrated the beneficial use of different types of prebiotics in maintaining the health of both polygastric and monogastric animals. Furthermore, research on the interaction between prebiotics and probiotics has proved to be necessary for optimizing animal gut health.
To gain a perspective on the future, it is a prerequisite to bear in mind that the animal gut microbiome and prebiotics have become a prevalent topic in the agriculture and food industry, with intense research in recent years. This research focus is predicted to continue expanding, indicating bright and assured prospects for prebiotics. Current research directions will continue aiming to optimise the harvesting process and benefits of prebiotics, exploring the potential of biofortification, unconventional sources, and discovering new applications for future prebiotics. On the other hand, biofortification that involves editing, eliminating, and inserting specific genetic material into various organisms, will have the potential to greatly enhance the development of prebiotics by simplifying their production process and improving their efficacy. As mentioned earlier, genetically modified yeast currently being used in the production of GOS, will see easier purification and higher yields compared to traditional extraction methods. This technology is expected to be applied in the manufacturing processes of other prebiotic components as well. With biofortification, companies will be able to choose a bacterial strain that requires less stringent growing conditions and will reduce the operational cost of producing prebiotics. Natural bioresources, such as mushroom polysaccharides and marine seaweed that have been shown to display prebiotic effects, will continue to attract research interests on their feasibility, efficacy, and potential as novel prebiotic sources. Although the extraction methods for the functional components in these natural bioresources are still relatively immature and underdeveloped, their potential to become the next best source of prebiotics will doubtlessly thrive.
Credit Author Statement
M. Cristina Ravanal: Writing – review & editing, Writing – original draft, Supervision, Conceptualization. Carolina A. Contador: Writing – review & editing, Writing – original draft. Wing-Tak Wong: Writing – original draft. Qile Zhang: Writing – original draft. Analese Roman-Benn: Writing – original draft. Kong Shun Ah-Hen: Writing – original draft. Pilar E. Ulloa: Writing – original draft. Hon-Ming Lam: Supervision, Funding acquisition, Conceptualization.
Declaration of competing interest
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work, and there is no professional or other personal interest of any nature or kind in any product, service and/or company that could be construed as influencing the content of this paper.
Acknowledgements
This work was supported by the Hong Kong Research Grants Council Area of Excellence Scheme (AoE/M-403/16) and the Lo Kwee-Seong Biomedical Research Fund awarded to Hon-Ming Lam and InEs Género, Project INGE220001 (INGE-2024–08) awarded to María Cristina Ravanal. Any opinions, findings, conclusions or recommendations expressed in this publication do not reflect the views of the Government of the Hong Kong Special Administrative Region or the Innovation Technology Commission.
Footnotes
Peer review under the responsibility of Chinese Association of Animal Science and Veterinary Medicine
References
- Adhikari P., Cosby D.E., Cox N.A., Franca M.S., Williams S.M., Gogal R.M., et al. Effect of dietary fructooligosaccharide supplementation on internal organs Salmonella colonization, immune response, ileal morphology, and ileal immunohistochemistry in laying hens challenged with Salmonella enteritidis. Poult Sci. 2018;97:2525–2533. doi: 10.3382/ps/pey101. [DOI] [PubMed] [Google Scholar]
- Ahmadifar E., Sheikhzadeh N., Roshanaei K., Dargahi N., Faggio C. Can dietary ginger (Zingiber officinale) alter biochemical and immunological parameters and gene expression related to growth, immunity and antioxidant system in zebrafish (Danio rerio)? Aquaculture. 2019;507:341–348. [Google Scholar]
- Al-Baadani H.H., Al-Mufarrej S.I., Al-Garadi M.A., Alhidary I.A., Al-Sagan A.A., Azzam M.M. The use of gum Arabic as a natural prebiotic in animals: a review. Anim Feed Sci Technol. 2021;274 [Google Scholar]
- Abd El-Aziz Aa-O, Abo Ghanima Ma-O, Alsanie W.F., Gaber Aa-OX., Alsenosy Aa-O, Easa A.A., Moawed S.A., Raza Sa-O, Elfadadny Aa-O, Yossef H.A., Ghoneem Wa-O, Shukry Ma-O, Hendawy Aa-OX., Mahrose Ka-O. Fructooligosaccharide supplementation boosts growth performance, antioxidant status, and cecal microbiota differently in two rabbit breeds. Animals. 2022;12:1528. doi: 10.3390/ani12121528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Sheraji S.H., Ismail A., Manap M.Y., Mustafa S., Yusof R.M., Hassan F.A. Prebiotics as functional foods: a review. J Funct Foods. 2013;5:1542–1553. [Google Scholar]
- Alves Costa N., Pansani A.P., de Castro C.H., Basile Colugnati D., Xaxier C.H., Guimarães K.C., et al. Milk restriction or oligosaccharide supplementation in calves improves compensatory gain and digestive tract development without changing hormone levels. PLoS One. 2019;14 doi: 10.1371/journal.pone.0214626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anadón A., Ares I., Martínez-Larrañaga M.R., Martínez M.A. In: Nutraceuticals in veterinary medicine. Gupta R.C., Srivastava A., Lall R., editors. Springer International Publishing; Cham: 2019. Prebiotics and probiotics in feed and animal health; pp. 261–285. [Google Scholar]
- Angwech H., Tavaniello S., Ongwech A., Kaaya A.N., Maiorano G. Efficacy of in ovo delivered prebiotics on growth performance, meat quality and gut health of Kuroiler chickens in the face of a natural coccidiosis challenge. Animals. 2019;9(11) doi: 10.3390/ani9110876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apolinário A.C., de Lima Damasceno B.P., de Macêdo Beltrão N.E., Pessoa A., Converti A., da Silva J.A. Inulin-type fructans: a review on different aspects of biochemical and pharmaceutical technology. Carbohydr Polym. 2014;30:368–378. doi: 10.1016/j.carbpol.2013.09.081. [DOI] [PubMed] [Google Scholar]
- Arsène M.M.J., Davares A.K.L., Andreevna S.L., Vladimirovich E.A., Carime B.Z., Marouf R., Khelifi I. The use of probiotics in animal feeding for safe production and as potential alternatives to antibiotics. Vet World. 2021;14:319–328. doi: 10.14202/vetworld.2021.319-328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azad MaK., Gao J., Ma J., Li T., Tan B., Huang X., Yin J. Opportunities of prebiotics for the intestinal health of monogastric animals. Anim Nutr. 2020;6:379–388. doi: 10.1016/j.aninu.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachmann M., Glatter M., Bochnia M., Greef J.M., Breves G., Zeyner A. Degradation of monosaccharides, disaccharides, and fructans in the stomach of horses adapted to a prebiotic dose of fructooligosaccharides and inulin. J Equine Vet Sci. 2021;105 doi: 10.1016/j.jevs.2021.103731. [DOI] [PubMed] [Google Scholar]
- Baker JT, Duarte ME, Holanda DM, Kim SW. Friend or Foe? Impacts of Dietary Xylans, Xylooligosaccharides, and Xylanases on Intestinal Health and Growth Performance of Monogastric Animals. Animals (Basel) 2021;26:609. doi: 10.3390/ani11030609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bąkowski M, Kiczorowska B. Probiotic microorganisms and herbs in ruminant nutrition as natural modulators of health and production efficiency – a review. Ann Anim Sci. 2020;21:3–28. [Google Scholar]
- Bamigbade G.B., Subhash A.J., Kamal-Eldin A., Nyström L., Ayyash M. An updated review on prebiotics: insights on potentials of food seeds waste as source of potential prebiotics. Molecules. 2022;27:5947. doi: 10.3390/molecules27185947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhat I, Shaheen M. Advances in Synbiotic Therapy in the Management of Gastrointestinal Diseases (Enteric Diseases) in Farm. Animals. 2017;2:147–152. [Google Scholar]
- Bortoluzzi C., Tamburini I., Geremia J. Microbiome modulation, microbiome protein metabolism index, and growth performance of broilers supplemented with a precision biotic. Poult Sci. 2023;102 doi: 10.1016/j.psj.2023.102595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosco NJ. Some Growth Promoters Already Tried to Replace Antimicrobial Growth Promoter in Weaned Pig, a Review. Int Res J Eng Technol. 2019;06:1741. [Google Scholar]
- Browne N., Horgan K. The impact of a proprietary blend of yeast cell wall, short-chain fatty acids, and zinc proteinate on growth, nutrient utilisation, and endocrine hormone secretion in intestinal cell models. Animals. 2024;14:238. doi: 10.3390/ani14020238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardoso B.B., Amorim C., Silvério S.C., Rodrigues L.R. Novel and emerging prebiotics: Advances and opportunities. Adv Food Nutr Res. 2021;95:41–95. doi: 10.1016/bs.afnr.2020.08.001. [DOI] [PubMed] [Google Scholar]
- do Carmo M.M., Walker J.C., Novello D., Caselato V.M., Sgarbieri V.C., Ouwehand A.C., et al. Polydextrose: Physiological Function, and Effects on Health. Nutrients. 2016;8:553. doi: 10.3390/nu8090553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castro J.J., Gomez A., White B.A., Mangian H.J., Loften J.R., Drackley J.K. Changes in the intestinal bacterial community, short-chain fatty acid profile, and intestinal development of preweaned holstein calves. 1. Effects of prebiotic supplementation depend on site and age. J Dairy Sci. 2016;99:9682–9702. doi: 10.3168/jds.2016-11006. [DOI] [PubMed] [Google Scholar]
- Celi P., Verlhac V., Pérez Calvo E., Schmeisser J., Kluenter A.-M. Biomarkers of gastrointestinal functionality in animal nutrition and health. Anim Feed Sci Technol. 2019;250:9–31. [Google Scholar]
- Chambers J, Gong J. The intestinal microbiota and its modulation for Salmonella control in chickens. Food Res Int. 2011;44:3149–3159. doi: 10.1016/j.foodres.2011.08.17. [DOI] [Google Scholar]
- Chang M., Wang F., Ma F., Jin Y., Sun P. Supplementation with galacto-oligosaccharides in early life persistently facilitates the microbial colonization of the rumen and promotes growth of preweaning holstein dairy calves. Anim Nutr. 2022;10:223–233. doi: 10.1016/j.aninu.2022.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang M., Zhao Y., Qin G., Zhang X. Fructo-oligosaccharide alleviates soybean-induced anaphylaxis in piglets by modulating gut microbes. Front Microbiol. 2018;9 doi: 10.3389/fmicb.2018.02769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chantorn S., Piyapittayanun C., Dangpram P. Bioconversion of agricultural wastes to mannooligosaccharides and their prebiotic potential. Chiang Mai J Sci. 2018;45:60–67. [Google Scholar]
- Chashnidel Y., Bahari M., Teimouri Yansari A., Kazemifard M. The effects of dietary supplementation of prebiotic and peptide on growth performance and blood parameters in suckling Zell lambs. Small Rumin Res. 2020;188 [Google Scholar]
- Chen G., Li C., Chen K. Fructooligosaccharides: a review on their mechanisms of action and effects. Stud Nat Prod Chem. 2016;48:209–229. [Google Scholar]
- Chibbar R., Alahmadi A., Dieleman L. Treatment of inflammatory bowel disease in ulcerative colitis. Microbiota Gastrointes Pathophysiol. 2017:343–354. Elsevier. [Google Scholar]
- Chiesa L.M., Nobile M., Panseri S., Arioli F. Antibiotic use in heavy pigs: comparison between urine and muscle samples from food chain animals analysed by hplc-ms/ms. Food Chem. 2017;235:111–118. doi: 10.1016/j.foodchem.2017.04.184. [DOI] [PubMed] [Google Scholar]
- Chikkerur J., Samanta A.K., Kolte A.P., Dhali A., Roy S. Production of short chain fructo-oligosaccharides from inulin of chicory root using fungal endoinulinase. Appl Biochem Biotechnol. 2020;191:695–715. doi: 10.1007/s12010-019-03215-7. [DOI] [PubMed] [Google Scholar]
- Chu N, Ling J, Jie H, Leung K, Poon E. The potential role of lactulose pharmacotherapy in the treatment and prevention of diabetes. Front Endocrinol (Lausanne) 2022;13:956203. doi: 10.3389/fendo.2022.956203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa J.M., Ampese L.C., Ziero H.D.D., Sganzerla W.G., Forster-Carneiro T. Apple pomace biorefinery: integrated approaches for the production of bioenergy, biochemicals, and value-added products – an updated review. J Environ Chem Eng. 2022;10 [Google Scholar]
- Costa J.R., Tonon R.V., Gottschalk L.M.F., Santiago M.C.P.D.A., Mellinger-Silva C., Pastrana L., Pintado M.M., Cabral L.M.C. Enzymatic production of xylooligosaccharides from brazilian syrah grape pomace flour: a green alternative to conventional methods for adding value to agricultural by- products. J Sci Food Agric. 2019;99:1250–1257. doi: 10.1002/jsfa.9297. [DOI] [PubMed] [Google Scholar]
- Cox M.A., Jackson J., Stanton M., Rojas-Triana A., Bober L., Laverty M., Yang X., Zhu F., Liu J., Wang S. Short-chain fatty acids act as antiinflammatory mediators by regulating prostaglandin e2 and cytokines. World J Gastroenterol: WJG. 2009;15:5549. doi: 10.3748/wjg.15.5549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crittenden R., Playne M.J. 2008. Prebiotics.Handbook of probiotics and prebiotics; pp. 533–581. [Google Scholar]
- Csernus B., Czeglédi L. Physiological, antimicrobial, intestine morphological, and immunological effects of fructooligosaccharides in pigs. Arch Anim Breed. 2020;63:325–335. doi: 10.5194/aab-63-325-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das N.G., Huque K.S., Amanullah S.M., Dharmapuri S., Makkar H.P.S. Study of chemical composition and nutritional values of vegetable wastes in Bangladesh. Vet Anim Sci. 2018;5:31–37. doi: 10.1016/j.vas.2018.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davani-Davari D., Negahdaripour M., Karimzadeh I., Seifan M., Mohkam M., Masoumi S.J., Berenjian A., Ghasemi Y. Prebiotics: definition, types, sources, mechanisms, and clinical applications. Foods. 2019;8:92. doi: 10.3390/foods8030092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De La Rosa O., Flores-Gallegos A.C., Muñíz-Marquez D., Nobre C., Contreras-Esquivel J.C., Aguilar C.N. Fructooligosaccharides production from agro-wastes as alternative low-cost source. Trends Food Sci Technol. 2019;91:139–146. [Google Scholar]
- Di Gioia D., Biavati B. In: Probiotics and prebiotics in animal health and food safety. Di Gioia D., Biavati B., editors. Springer International Publishing; Cham: 2018. Probiotics and prebiotics in animal health and food safety: conclusive remarks and future perspectives; pp. 269–273. [Google Scholar]
- Dong L., Zhao L., Li B., Gao Y., Yan T., Lund P., Liu Z., Diao Q. Dietary supplementation with xylooligosaccharides and exogenous enzyme improves milk production, energy utilization efficiency and reduces enteric methane emissions of Jersey cows. J Anim Sci Biotechnol. 2023;14:71. doi: 10.1186/s40104-023-00873-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duan X., Tian G., Chen D., Huang L., Zhang D., Zheng P., Mao X., Yu J., He J., Huang Z., Yu B. Mannan oligosaccharide supplementation in diets of sow and (or) their offspring improved immunity and regulated intestinal bacteria in piglet1. J Anim Sci. 2019;97:4548–4556. doi: 10.1093/jas/skz318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flickinger E.A., Fahey G.C. Pet food and feed applications of inulin, oligofructose and other oligosaccharides. Br J Nutr. 2002;87:S297–S300. doi: 10.1079/BJNBJN/2002552. [DOI] [PubMed] [Google Scholar]
- Forgetta V, Rempel H, Malouin F, Vaillancourt R, Topp E, Dewar K, et al. Pathogenic and multidrug-resistant Escherichia fergusonii from broiler chicken. Poult Sci. 2012;91:512–525. doi: 10.3382/ps.2011-01738. [DOI] [PubMed] [Google Scholar]
- Freitas L., Simões R., Miranda I., Peres F., Ferreira-Dias S. Catalysts. 2022. Optimization of autohydrolysis of olive pomaces to obtain bioactive oligosaccharides: the effect of cultivar and fruit ripening. [Google Scholar]
- Froebel L.K., Jalukar S., Lavergne T.A., Lee J.T., Duong T. Administration of dietary prebiotics improves growth performance and reduces pathogen colonization in broiler chickens. Poult Sci. 2019;98(12):6668–6676. doi: 10.3382/ps/pez537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaggìa F., Mattarelli P., Biavati B. Probiotics and prebiotics in animal feeding for safe food production. Int J Food Microbiol. 2010;141:S15–S28. doi: 10.1016/j.ijfoodmicro.2010.02.031. [DOI] [PubMed] [Google Scholar]
- Ganaie M.A., Soni H., Naikoo G.A., Santos Oliveira L.T., Rawat H.K., Mehta P.K., Narain N. Screening of low cost agricultural wastes to maximize the fructosyltransferase production and its applicability in generation of fructooligosaccharides by solid state fermentation. Int Biodeterior Biodegrad. 2017;118:19–26. [Google Scholar]
- Gasbarrini G., Bonvicini F., Gramenzi A. Probiotics history. J Clin Gastroenterol. 2016;50:S116–S119. doi: 10.1097/MCG.0000000000000697. [DOI] [PubMed] [Google Scholar]
- Ghosh S., Mehla R.K. Influence of dietary supplementation of prebiotics (mannanoligosaccharide) on the performance of crossbred calves. Trop Anim Health Prod. 2012;44:617–622. doi: 10.1007/s11250-011-9944-8. [DOI] [PubMed] [Google Scholar]
- Gibson G.R., Hutkins R., Sanders M.E., Prescott S.L., Reimer R.A., Salminen S.J., Scott K., Stanton C., Swanson K.S., Cani P.D., Verbeke K., Reid G. Expert consensus document: the international scientific association for probiotics and prebiotics (isapp) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 2017;14:491–502. doi: 10.1038/nrgastro.2017.75. [DOI] [PubMed] [Google Scholar]
- Gibson G.R., Roberfroid M.B. Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J Nutr. 1995;125:1401–1412. doi: 10.1093/jn/125.6.1401. [DOI] [PubMed] [Google Scholar]
- Gonçalves D.A., González A., Roupar D., Teixeira J.A., Nobre C. How prebiotics have been produced from agro-industrial waste: an overview of the enzymatic technologies applied and the models used to validate their health claims. Trends Food Sci Technol. 2023;135:74–92. [Google Scholar]
- González-Solé F., Solà-Oriol D., Ramayo-Caldas Y., Rodriguez-Prado M., González Ortiz G., Bedford M.R., Pérez J.F. Supplementation of xylo-oligosaccharides to suckling piglets promotes the growth of fiber-degrading gut bacterial populations during the lactation and nursery periods. Sci Rep. 2022;12 doi: 10.1038/s41598-022-15963-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gowman A.C., Picard M.C., Rodriguez-Uribe A., Misra M., Khalil H., Thimmanagari M., Mohanty A.K. Physicochemical analysis of apple and grape pomaces. Bioresources. 2019;14:3210–3230. [Google Scholar]
- Grand E., Respondek F., Martineau C., Detilleux J., Bertrand G. Effects of short-chain fructooligosaccharides on growth performance of preruminant veal calves. J Dairy Sci. 2013;96:1094–1101. doi: 10.3168/jds.2011-4949. [DOI] [PubMed] [Google Scholar]
- Gupta M., Bangotra R., Sharma S., Vaid S., Kapoor N., Dutt H.C., Bajaj B.K. Bioprocess development for production of xylooligosaccharides prebiotics from sugarcane bagasse with high bioactivity potential. Ind Crop Prod. 2022;178 [Google Scholar]
- Hackenhaar C.R., Spolidoro L.S., Flores E.E.E., Klein M.P., Hertz P.F. Batch synthesis of galactooligosaccharides from co-products of milk processing using immobilized β-galactosidase from bacillus circulans. Biocatal Agric Biotechnol. 2021;36 [Google Scholar]
- Hackmann T.J. New biochemical pathways for forming short-chain fatty acids during fermentation in rumen bacteria. JDS Commun. 2023;5:230–235. doi: 10.3168/jdsc.2023-0427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haulisah N.A., Hassan L., Bejo S.K., Jajere S.M., Ahmad N.I. High levels of antibiotic resistance in isolates from diseased livestock. Front Vet Sci. 2021;8 doi: 10.3389/fvets.2021.652351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinrichs A.J., Jones C.M., Elizondo-Salazar J.A., Terrill S.J. Effects of a prebiotic supplement on health of neonatal dairy calves. Livest Sci. 2009;125:149–154. [Google Scholar]
- Heinrichs A.J., Jones C.M., Heinrichs B.S. Effects of mannan oligosaccharide or antibiotics in neonatal diets on health and growth of dairy calves. J Dairy Sci. 2003;86:4064–4069. doi: 10.3168/jds.S0022-0302(03)74018-1. [DOI] [PubMed] [Google Scholar]
- Hill C., Guarner F., Reid G., Gibson G.R., Merenstein D.J., Pot B., et al. Expert consensus document: the international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11:506–514. doi: 10.1038/nrgastro.2014.66. [DOI] [PubMed] [Google Scholar]
- Islam M.R., Hassan Y.I., Das Q., Lepp D., Hernandez M., Godfrey D.V., Orban S., Ross K., Delaquis P., Diarra M.S. Dietary organic cranberry pomace influences multiple blood biochemical parameters and cecal microbiota in pasture-raised broiler chickens. J Funct Foods. 2020;72 [Google Scholar]
- Jacquier V., Walsh M.C., Schyns G., Claypool J., Blokker B., Bortoluzzi C., Geremia J. Animals. 2022. Evaluation of a precision biotic on the growth performance, welfare indicators, ammonia output, and litter quality of broiler chickens. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jahan AA, González Ortiz G, Moss AF, Bhuiyan MM, Morgan NK. Role of supplemental oligosaccharides in poultry diets. Worlds Poult Sci J. 2022;78:615–639. [Google Scholar]
- Jha R., Fouhse J.M., Tiwari U.P., Li L., Willing B.P. Dietary fiber and intestinal health of monogastric animals. Front Vet Sci. 2019;6 doi: 10.3389/fvets.2019.00048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi V. Solid state fermentation of apple pomace for the production of value added products vk joshi and devender attri fermentation technology laboratory, department of postharvest technology dr. Ys parmar university of horticulture and forestry, nauni-173 230, solan. Pollution in Urban Industrial Environment. 2005:180. [Google Scholar]
- Juśkiewicz J., Jankowski J., Zduńczyk Z., Mikulski D. Performance and gastrointestinal tract metabolism of turkeys fed diets with different contents of fructooligosaccharides. Poult Sci. 2006;85:886–891. doi: 10.1093/ps/85.5.886. [DOI] [PubMed] [Google Scholar]
- Karimian R.A., Rezaeipour V. Effects of dietary mannan-oligosaccharides and phytase supplementation alone or in combination on growth performance, serum metabolites, cecal microbiota activity and intestinal morphology in broiler chickens. Poult Sci J. 2020;8(1):27–32. [Google Scholar]
- Kathiresan N., Gopal L., Karuppiah V., Naveenethan R., Abraham D., Thangavel K. 2021. Utilization of agricultural waste for the production of xylooligosaccharides using response surface methodology and their in vitro prebiotic efficacy. [Google Scholar]
- Kaur R., Uppal S.K., Sharma P. Production of xylooligosaccharides from sugarcane bagasse and evaluation of their prebiotic potency in vitro. Waste Biomass Valoriz. 2019;10:2627–2635. [Google Scholar]
- Khalili R.M.A., Kota K., Mahmud J.S., Abidin Z.U. 2014. Isolation and characterization of oligosaccharides composition in organically grown red pitaya, white pitaya and papaya. [Google Scholar]
- Khan R.U., Naz S., Raziq F., Qudratullah Q., Khan N.A., Laudadio V., Tufarelli V., Ragni M. Prospects of organic acids as safe alternative to antibiotics in broiler chickens diet. Environ Sci Pollut Control Ser. 2022;29:32594–32604. doi: 10.1007/s11356-022-19241-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khatun M.S., Hassanpour M., Mussatto S.I., Harrison M.D., Speight R.E., O’hara I.M., Zhang Z. Transformation of sugarcane molasses into fructooligosaccharides with enhanced prebiotic activity using whole-cell biocatalysts from aureobasidium pullulans frr 5284 and an invertase-deficient saccharomyces cerevisiae 1403-7a. Bioresour Bioproc. 2021;8:85. doi: 10.1186/s40643-021-00438-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khuenpet K., Jittanit W., Sirisansaneeyakul S., Srichamnong W. Inulin powder production from jerusalem artichoke (helianthus tuberosus l.) tuber powder and its application to commercial food products. J Food Process Preserv. 2017;41 [Google Scholar]
- Kim H., Jeon B., Kim W.J., Chung D.-K. Effect of paraprobiotic prepared from kimchi-derived lactobacillus plantarum k8 on skin moisturizing activity in human keratinocyte. J Funct Foods. 2020;75 [Google Scholar]
- Kolawole J.T., Babafemi A.J., Fanijo E., Chandra Paul S., Combrinck R. State-of-the-art review on the use of sugarcane bagasse ash in cementitious materials. Cement Concr Compos. 2021;118 [Google Scholar]
- La Fata G., Weber P., Mohajeri M.H. Probiotics and the gut immune system: indirect regulation. Probiotics Antimicrob proteins. 2018;10:11–21. doi: 10.1007/s12602-017-9322-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Bourgot C., LFerret-Bernard S., Apper E., Taminiau B., Cahu A., Le Normand L., et al. Perinatal short-chain fructooligosaccharides program intestinal microbiota and improve enteroinsular axis function and inflammatory status in high-fat diet-fed adult pigs. FASEB Journal. 2019;33:301–313. doi: 10.1096/fj.201800108R. [DOI] [PubMed] [Google Scholar]
- Lee W-H, Pathanibul P., Quarterman J., Jo J-H., Han NS., Miller MJ., et al. Whole cell biosynthesis of a functional oligosaccharide, 2′-fucosyllactose, using engineered Escherichia coli. Microb Cell Fact. 2012;11:48. doi: 10.1186/1475-2859-11-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leone F., Ferrante V. Effects of prebiotics and precision biotics on performance, animal welfare and environmental impact. A review. Sci Total Environ. 2023;901:165951. doi: 10.1016/j.scitotenv.2023.165951. [DOI] [PubMed] [Google Scholar]
- Logtenberg MJ, Akkerman R, Hobé RG, Donners KMH, Van Leeuwen SS, Hermes GDA, et al. Structure-Specific Fermentation of Galacto-Oligosaccharides, Isomalto-Oligosaccharides and Isomalto/Malto-Polysaccharides by Infant Fecal Microbiota and Impact on Dendritic Cell Cytokine Responses. Mol Nutr Food Res. 2021;65 doi: 10.1002/mnfr.202001077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mabrok H.B., Soliman M.I., Mohammad M.A., Hussein L. 2018. Hplc profiles of onion fructooligosaccharides and inulin and their prebiotic effects on modulating key markers of colon function, calcium metabolism and bone mass in rat model. [Google Scholar]
- Mackowiak P.A. Recycling metchnikoff: probiotics, the intestinal microbiome and the quest for long life. Front Public Health. 2013;1:52. doi: 10.3389/fpubh.2013.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manafu Z., Zhang Z., Malajiang X., Abula S., Guo Q., Wu Y., Wusiman A., Bake B. Effects of alhagi camelorum fisch polysaccharide from different regions on growth performance and gastrointestinal microbiota of sheep lambs. Front Pharmacol. 2024;15 doi: 10.3389/fphar.2024.1379394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mano M.C.R., Paulino B.N., Pastore G.M. Whey permeate as the raw material in galacto-oligosaccharide synthesis using commercial enzymes. Food Res Int. 2019;124:78–85. doi: 10.1016/j.foodres.2018.09.019. [DOI] [PubMed] [Google Scholar]
- Markowiak P., Śliżewska K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients. 2017;9:1021. doi: 10.3390/nu9091021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markowiak P., Śliżewska K. The role of probiotics, prebiotics and synbiotics in animal nutrition. Gut Pathog. 2018;10:21. doi: 10.1186/s13099-018-0250-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martins G.N., Ureta M.M., Tymczyszyn E.E., Castilho P.C., Gomez-Zavaglia A. Technological aspects of the production of fructo and galacto-oligosaccharides. Enzymatic synthesis and hydrolysis. Front Nutr. 2019;6:78. doi: 10.3389/fnut.2019.00078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazhar SH, Li X, Rashid A, Su J, Xu J, Brejnrod AD, et al. Co-selection of antibiotic resistance genes, and mobile genetic elements in the presence of heavy metals in poultry farm environments. Sci Total Environ. 2021;755:142702. doi: 10.1016/j.scitotenv.2020.142702. [DOI] [PubMed] [Google Scholar]
- Mei Z, Yuan J, Li D. Biological activity of galacto-oligosaccharides: A review. Front Microbiol. 2022;13:993052. doi: 10.3389/fmicb.2022.993052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meini M.-R., Cabezudo I., Galetto C.S., Romanini D. Production of grape pomace extracts with enhanced antioxidant and prebiotic activities through solid-state fermentation by Aspergillus niger and Aspergillus oryzae. Food Biosci. 2021;42 [Google Scholar]
- Meyer D, Stasse-Wolthuis M. The bifidogenic effect of inulin and oligofructose and its consequences for gut health. Eur J Clin Nutr. 2009;63:1277–1289. doi: 10.1038/ejcn.2009.64. [DOI] [PubMed] [Google Scholar]
- Michalak M., Wojnarowski K., Cholewińska P., Szeligowska N., Bawej M., Pacoń J. Selected alternative feed additives used to manipulate the rumen microbiome. Animals. 2021;11:1542. doi: 10.3390/ani11061542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mnisi C.M., Mhlongo G., Manyeula F. Fruit pomaces as functional ingredients in poultry nutrition: a review. Front Anim Sci. 2022;3 [Google Scholar]
- Mohammadi G, Karimi AA, Hafezieh M, Dawood MAO. Abo-Al-Ela HG. Pistachio hull polysaccharide protects Nile tilapia against LPS-induced excessive inflammatory responses and oxidative stress, possibly via TLR2 and Nrf2 signaling pathways. Fish Shellfish Immunol. 2022;121:276–284. doi: 10.1016/j.fsi.2021.12.042. [DOI] [PubMed] [Google Scholar]
- Mohammadigheisar M, Shirley RB, Barton J, Welsher A, Thiery P, Kiarie E. Growth performance and gastrointestinal responses in heavy Tom turkeys fed antibiotic free corn−soybean meal diets supplemented with multiple doses of a single strain Bacillus subtilis probiotic (DSM29784)1. Poult Sci. 2019;98:5541–5550. doi: 10.3382/ps/pez305. [DOI] [PubMed] [Google Scholar]
- Mussatto S.I., Teixeira J.A. Increase in the fructooligosaccharides yield and productivity by solid-state fermentation with aspergillus japonicus using agro-industrial residues as support and nutrient source. Biochem Eng J. 2010;53:154–157. [Google Scholar]
- Nawaz A., Bakhsh Javaid A., Irshad S., Hoseinifar S.H., Xiong H. The functionality of prebiotics as immunostimulant: evidences from trials on terrestrial and aquatic animals. Fish Shellfish Immunol. 2018;76:272–278. doi: 10.1016/j.fsi.2018.03.004. [DOI] [PubMed] [Google Scholar]
- Nguyen Q.A., Cho E.J., Lee D.-S., Bae H.-J. Development of an advanced integrative process to create valuable biosugars including manno-oligosaccharides and mannose from spent coffee grounds. Bioresour Technol. 2019;272:209–216. doi: 10.1016/j.biortech.2018.10.018. [DOI] [PubMed] [Google Scholar]
- Nicolás García M., Borrás Enríquez A.J., González Escobar J.L., Calva Cruz O.D.J., Pérez Pérez V., Sánchez Becerril M. Phenolic compounds in agro-industrial waste of mango fruit: impact on health and its prebiotic effect – a review. Pol J Food Nutr Sci. 2023;73:5–23. [Google Scholar]
- Ojha BK, Singh PK, Shrivastava N. Chapter 7 - Enzymes in the Animal Feed Industry. In: Kuddus MBT-E in FB, editor., Academic Press; 2019. pp. 93–109. [Google Scholar]
- O’shea N., Ktenioudaki A., Smyth T., Mcloughlin P., Doran L., Auty M., Arendt E., Gallagher E. Physicochemical assessment of two fruit by-products as functional ingredients: apple and orange pomace. J Food Eng. 2015;153:89–95. [Google Scholar]
- Pan J., Yin J., Zhang K., Xie P., Ding H., Huang X., Blachier F., Kong X. Dietary xylo-oligosaccharide supplementation alters gut microbial composition and activity in pigs according to age and dose. AMB Express. 2019;9:134. doi: 10.1186/s13568-019-0858-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pandey K.R., Naik S.R., Vakil B.V. Probiotics, prebiotics and synbiotics-a review. J Food Sci Technol. 2015;52:7577–7587. doi: 10.1007/s13197-015-1921-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pangestu R., Rahmani N., Palar R., Lisdiyanti P. IOP conference series: earth and environmental science. IOP Publishing; 2019. The effect of biomass particle size and chemical structure on the enzymatic hydrolysis reaction of galactomannan from sugar palm fruit by β-mannanase from kitasatospora sp. Ky576672. [Google Scholar]
- Parada Venegas D., De La Fuente M.K., Landskron G., González M.J., Quera R., Dijkstra G., Harmsen H.J., Faber K.N., Hermoso M.A. Short chain fatty acids (scfas)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front Immunol. 2019;277 doi: 10.3389/fimmu.2019.00277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park SH, Hanning I, Perrota A, Bench BJ, Alm E, Ricke SC. Modifying the gastrointestinal ecology in alternatively raised poultry and the potential for molecular and metabolomic assessment1 1Presented as part of the Next Generation Sequencing: Applications for Food Safety and Poultry Production Symposium at the P. Poult Sci. 2013;92:546–561. doi: 10.3382/ps.2012-02734. [DOI] [PubMed] [Google Scholar]
- Patel D., Ratre P., Singh R., Chaudhary S. vol. 8. 2019. pp. 265–269. (Effect of prebiotic and probiotic supplementation on growth performance and body measurement in preruminant Surti buffalo calves). [Google Scholar]
- Patel P., Patel B., Amaresan N., Joshi B., Shah R., Krishnamurthy R. Isolation and characterization of lactococcus garvieae from the fish gut for in vitro fermentation with carbohydrates from agro-industrial waste. Biotechnol Rep. 2020;28 doi: 10.1016/j.btre.2020.e00555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel S, Goyal A. The current trends and future perspectives of prebiotics research: a review. 3 Biotech. 2012;2:115–125. [Google Scholar]
- Patel S., Shukla S. In: Fermented foods in health and disease prevention. Frias J., Martinez-Villaluenga C., Peñas E., editors. Academic Press; Boston: 2017. Chapter 30 - fermentation of food wastes for generation of nutraceuticals and supplements; pp. 707–734. [Google Scholar]
- Peng M., Patel P., Nagarajan V., Bernhardt C., Carrion M., Biswas D. Elsevier; 2019. Feasible options to control colonization of enteric pathogens with designed synbiotics.Dietary interventions in gastrointestinal diseases; pp. 135–149. [Google Scholar]
- Peng M., Tabashsum Z., Anderson M., Truong A., Houser A.K., Padilla J., Akmel A., Bhatti J., Rahaman S.O., Biswas D. Effectiveness of probiotics, prebiotics, and prebiotic-like components in common functional foods. Compr Rev Food Sci Food Saf. 2020;19:1908–1933. doi: 10.1111/1541-4337.12565. [DOI] [PubMed] [Google Scholar]
- Pinotti L., Manoni M., Fumagalli F., Rovere N., Luciano A., Ottoboni M., Ferrari L., Cheli F., Djuragic O. Animals. 2020. Reduce, reuse, recycle for food waste: a second life for fresh-cut leafy salad crops in animal diets. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purnawan A., Yopi Y., Irawadi T.T. Production of manooligomannan from palm kernel cake by mannanase produced from streptomyces cyaenus. Biosaintifika: J Biol Educ. 2017;9:73–80. [Google Scholar]
- Qamar T.R., Syed F., Nasir M., Rehman H., Zahid M.N., Liu R.H., Iqbal S. Novel combination of prebiotics galacto-oligosaccharides and inulin-inhibited aberrant crypt foci formation and biomarkers of colon cancer in wistar rats. Nutrients. 2016;8:465. doi: 10.3390/nu8080465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quijada N.M., Bodas R., Lorenzo J.M., Schmitz-Esser S., Rodríguez-Lázaro D., Hernández M. Biomolecules. 2020. Dietary supplementation with sugar beet fructooligosaccharides and garlic residues promotes growth of beneficial bacteria and increases weight gain in neonatal lambs. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radenkovs V., Juhnevica-Radenkova K., Górnaś P., Seglina D. Non-waste technology through the enzymatic hydrolysis of agro-industrial by-products. Trends Food Sci Technol. 2018;77:64–76. [Google Scholar]
- Rahmani N., Kashiwagi N., Lee J., Niimi-Nakamura S., Matsumoto H., Kahar P., Lisdiyanti P., Yopi Prasetya B., Ogino C., Kondo A. Mannan endo-1,4-β-mannosidase from kitasatospora sp. Isolated in Indonesia and its potential for production of mannooligosaccharides from mannan polymers. AMB Express. 2017;7:100. doi: 10.1186/s13568-017-0401-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao Z., Li Y., Yang X., Guo Y., Zhang W., Wang Z. Diet xylo-oligosaccharide supplementation improves growth performance, immune function, and intestinal health of broilers. Anim Nutr. 2024;17:165–176. doi: 10.1016/j.aninu.2024.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehman H, Vahjen W, Kohl-Parisini A, Ahmad I, Zentek J. Influence of fermentable carbohydrates on the intestinal bacteria and enteropathogens in broilers. Worlds Poult Sci J. 2009;65:75–90. [Google Scholar]
- Ribeiro T.B., Costa C.M., Bonifácio - Lopes T., Silva S., Veiga M., Monforte A.R., Nunes J., Vicente A.A., Pintado M. Prebiotic effects of olive pomace powders in the gut: in vitro evaluation of the inhibition of adhesion of pathogens, prebiotic and antioxidant effects. Food Hydrocoll. 2021;112 [Google Scholar]
- Rodríguez-Gutiérrez G., Lama-Muñoz A., Ruiz-Méndez M., Rubio-Senent F., Fernández-Bolaños J. In: Olive oil—constituents, quality, health properties and bioconversions. Boskou D., editor. 2012. New olive-pomace oil improved by hydrothermal pre-treatments; pp. 249–266. [Google Scholar]
- Roodposhti PM, Dabiri N. Effects of probiotic and prebiotic on average daily gain, fecal shedding of Escherichia coli, and immune system status in newborn female calves. Asian-Australasian J Anim Sci. 2012;25:1255–1261. doi: 10.5713/ajas.2011.11312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadh P.K., Duhan S., Duhan J.S. Agro-industrial wastes and their utilization using solid state fermentation: a review. Bioresour Bioproc. 2018;5:1. [Google Scholar]
- Sahasrabudhe N.M., Beukema M., Tian L., Troost B., Scholte J., Bruininx E., Bruggeman G., Van Den Berg M., Scheurink A., Schols H.A. Dietary fiber pectin directly blocks toll-like receptor 2–1 and prevents doxorubicin-induced ileitis. Front Immunol. 2018;9:383. doi: 10.3389/fimmu.2018.00383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santibáñez L, Henríquez C, Corro-Tejeda R, Bernal S, Armijo B, Salazar O. Xylooligosaccharides from lignocellulosic biomass: A comprehensive review. Carbohydr Polym. 2021;1:117118. doi: 10.1016/j.carbpol.2020.117118. [DOI] [PubMed] [Google Scholar]
- Sarkar S. Potential of prebiotics as functional foods–a review. Nutr Food Sci. 2007;37:168–177. [Google Scholar]
- Senevirathne N.D., Anderson J.L., Metzger L. Growth performance, nutrient utilization, and health of dairy calves supplemented with condensed whey solubles. J Dairy Sci. 2019;102:8108–8119. doi: 10.3168/jds.2019-16314. [DOI] [PubMed] [Google Scholar]
- Serra V., Salvatori G., Pastorelli G. Animals. 2021. Dietary polyphenol supplementation in food producing animals: effects on the quality of derived products. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma M., Patel S.N., Lata K., Singh U., Krishania M., Sangwan R.S., Singh S.P. A novel approach of integrated bioprocessing of cane molasses for production of prebiotic and functional bioproducts. Bioresour Technol. 2016;219:311–318. doi: 10.1016/j.biortech.2016.07.131. [DOI] [PubMed] [Google Scholar]
- Śliżewska K., Chlebicz-Wójcik A. The in vitro analysis of prebiotics to be used as a component of a synbiotic preparation. Nutrients. 2020;12:1272. doi: 10.3390/nu12051272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smiricky-Tjardes M.R., Grieshop C.M., Flickinger E.A., Bauer L.L., Fahey G.C., Jr. Dietary galactooligosaccharides affect ileal and total-tract nutrient digestibility, ileal and fecal bacterial concentrations, and ileal fermentative characteristics of growing pigs1. J Anim Sci. 2003;81:2535–2545. doi: 10.2527/2003.81102535x. [DOI] [PubMed] [Google Scholar]
- Solís-Cruz B., Hernandez-Patlan D., Hargis B., Tellez G. The poultry industry. 2019. Use of prebiotics as an alternative to antibiotic growth promoters; pp. 1–22. [Google Scholar]
- Stanley D, Hughes RJ, Moore RJ. Microbiota of the chicken gastrointestinal tract: influence on health, productivity and disease. Appl Microbiol Biotechnol. 2014;98:4301–4310. doi: 10.1007/s00253-014-5646-2. [DOI] [PubMed] [Google Scholar]
- Stover MG, Watson RR, Collier RJ. In: Preedy Prebiotics, and Synbiotics VRBT-P. Watson RR, editor. Academic Press; 2016. Chapter 2 - Pre- and Probiotic Supplementation in Ruminant Livestock Production; pp. 25–36. [Google Scholar]
- Struck S., Plaza M., Turner C., Rohm H. Berry pomace–a review of processing and chemical analysis of its polyphenols. Int J Food Sci Technol. 2016;51:1305–1318. [Google Scholar]
- Tellez G., Arreguin-Nava M.A., Maguey J.A., Michel M.A., Latorre J.D., Merino-Guzman R., Hernandez-Velasco X., Moore P.A., Hargis B.M., Tellez-Isaias G. Effect of bacillus–direct-fed microbial on leaky gut, serum peptide yy concentration, bone mineralization, and ammonia excretion in neonatal female Turkey poults fed with a rye-based diet. Poult Sci. 2020;99:4514–4520. doi: 10.1016/j.psj.2020.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terré M., Calvo M.A., Adelantado C., Kocher A., Bach A. Effects of mannan oligosaccharides on performance and microorganism fecal counts of calves following an enhanced-growth feeding program. Anim Feed Sci Technol. 2007;137:115–125. [Google Scholar]
- Tian K., Liu J., Sun Y., Wu Y., Chen J., Zhang R., He T., Dong G. Effects of dietary supplementation of inulin on rumen fermentation and bacterial microbiota, inflammatory response and growth performance in finishing beef steers fed high or low-concentrate diet. Anim Feed Sci Technol. 2019;258 [Google Scholar]
- Tian L., Zeng Q., Dong W., Guo Q., Wu Z., Pan X., Li G., Liu Y. Addressing the source contribution of pm 2 . 5 on mortality : an evaluation study of its impacts on excess mortality in China. Environ Res Lett. 2017;12 [Google Scholar]
- Tian S., Wang J., Gao R., Wang J., Zhu W. Early-life galacto-oligosaccharides supplementation alleviates the small intestinal oxidative stress and dysfunction of lipopolysaccharide-challenged suckling piglets. J Anim Sci Biotechnol. 2022;13:70. doi: 10.1186/s40104-022-00711-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toscano Miranda N., Lopes Motta I., Maciel Filho R., Wolf Maciel M.R. Sugarcane bagasse pyrolysis: a review of operating conditions and products properties. Renew Sustain Energy Rev. 2021;149 [Google Scholar]
- Tóth S., Kovács M., Bóta B., Szabó-Fodor J., Bakos G., Fébel H. Effect of mannanoligosaccharide (mos) and inulin supplementation on the performance and certain physiological parameters of calves reared on milk replacer. J Appl Anim Res. 2020;48:228–234. [Google Scholar]
- Totton SC, Farrar AM, Wilkins W, Bucher O, Waddell LA, Wilhelm BJ, et al. The effectiveness of selected feed and water additives for reducing Salmonella spp. of public health importance in broiler chickens: A systematic review, meta-analysis, and meta-regression approach. Prev Vet Med. 2012;106:197–213. doi: 10.1016/j.prevetmed.2012.07.007. [DOI] [PubMed] [Google Scholar]
- Tzortzis G., Goulas A.K., Gee J.M., Gibson G.R. A novel galactooligosaccharide mixture increases the bifidobacterial population numbers in a continuous in vitro fermentation system and in the proximal colonic contents of pigs in vivo1. J Nutr. 2005;135:1726–1731. doi: 10.1093/jn/135.7.1726. [DOI] [PubMed] [Google Scholar]
- Ursell L.K., Metcalf J.L., Parfrey L.W., Knight R. Defining the human microbiome. Nutr Rev. 2012;70:S38–S44. doi: 10.1111/j.1753-4887.2012.00493.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uyeno Y., Shigemori S., Shimosato T. Effect of probiotics/prebiotics on cattle health and productivity. Microb Environ. 2015;30:126–132. doi: 10.1264/jsme2.ME14176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasanthakumari B.L., Gedye K.R., Abdollahi M.R., Di Benedetto M., Sanchez D.G., Wealleans A., Ravindran V. A new monocomponent xylanase improves performance, ileal digestibility of energy and nutrients, intestinal morphology, and intestinal microbiota in young broilers. J Appl Poultry Res. 2023;32 [Google Scholar]
- Verdonk JMaJ., Shim S.B., Van Leeuwen P., Verstegen M.W.A. Application of inulin-type fructans in animal feed and pet food. Br J Nutr. 2005;93:S125–S138. doi: 10.1079/bjn20041355. [DOI] [PubMed] [Google Scholar]
- Vlad C.C., Păcularu-Burada B., Vasile A.M., Milea Ș.A., Bahrim G.-E., Râpeanu G., Stănciuc N. Antioxidants. 2022. Upgrading the functional potential of apple pomace in value-added ingredients with probiotics. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh M.C., Jacquier V., Schyns G., Claypool J., Tamburini I., Blokker B., Geremia J.M. A novel microbiome metabolic modulator improves the growth performance of broiler chickens in multiple trials and modulates targeted energy and amino acid metabolic pathways in the cecal metagenome. Poult Sci. 2021;100 doi: 10.1016/j.psj.2020.10.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan J., Zhang J., Chen D.W., Yu B., Mao X., Zheng P., et al. Alginate oligosaccharide-induced intestinal morphology, barrier function and epithelium apoptosis modifications have beneficial effects on the growth performance of weaned pigs. J Anim Sci Biotechnol. 2018;9 doi: 10.1186/s40104-018-0273-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waqas M., Salman M., Sharif M.S. Application of polyphenolic compounds in animal nutrition and their promising effects. J Anim Feed Sci. 2023;32:233–256. [Google Scholar]
- Wong J.M.W., De Souza R., Kendall C.W.C., Emam A., Jenkins D.J.A. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol. 2006;40 doi: 10.1097/00004836-200603000-00015. [DOI] [PubMed] [Google Scholar]
- Wongkaew M, Tangjaidee P, Leksawasdi N, Jantanasakulwong K, Rachtanapun P, Seesuriyachan P, et al. Mango Pectic Oligosaccharides: A Novel Prebiotic for Functional Food. Front Nutr. 2022;24:798543. doi: 10.3389/fnut.2022.798543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wongsiridetchai C., Jonjaroen V., Sawangwan T., Charoenrat T., Chantorn S. Evaluation of prebiotic mannooligosaccharides obtained from spent coffee grounds for nutraceutical application. Lebensm Wiss Technol. 2021;148 [Google Scholar]
- Xia Y., Miao J., Zhang Y., Zhang H., Kong L., Seviour R., Kong Y. Dietary inulin supplementation modulates the composition and activities of carbohydrate-metabolizing organisms in the cecal microbiota of broiler chickens. PLoS One. 2021;16 doi: 10.1371/journal.pone.0258663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z.R., Hu C.H., Xia M.S., Zhan X.A., Wang M.Q. Effects of dietary fructooligosaccharide on digestive enzyme activities, intestinal microflora and morphology of male broilers. Poult Sci. 2003;82:1030–1036. doi: 10.1093/ps/82.6.1030. [DOI] [PubMed] [Google Scholar]
- Yaashikaa P.R., Senthil Kumar P., Varjani S. Valorization of agro-industrial wastes for biorefinery process and circular bioeconomy: a critical review. Bioresour Technol. 2022;343 doi: 10.1016/j.biortech.2021.126126. [DOI] [PubMed] [Google Scholar]
- Yañez-Ñeco C.V., Cervantes F.V., Amaya-Delgado L., Ballesteros A.O., Plou F.J., Arrizon J. Synthesis of β(1 → 3) and β(1 → 6) galactooligosaccharides from lactose and whey using a recombinant β-galactosidase from pantoea anthophila. Electron J Biotechnol. 2021;49:14–21. [Google Scholar]
- Yang C., Das Q., Rehman M.A., Yin X., Shay J., Gauthier M., Lau C.H., Ross K., Diarra M.S. Microorganisms. 2023. Microbiome of ceca from broiler chicken vaccinated or not against coccidiosis and fed berry pomaces. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang C., Zhang T., Tian Q., Cheng Y., Gebeyew K., Liu G., Tan Z., He Z. Supplementing mannan oligosaccharide reduces the passive transfer of immunoglobulin g and improves antioxidative capacity, immunity, and intestinal microbiota in neonatal goats. Front Microbiol. 2022;12 doi: 10.3389/fmicb.2021.795081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin J, Li F, Kong X, Wen C, Guo Q, Zhang L, et al. Dietary xylo-oligosaccharide improves intestinal functions in weaned piglets. Food Funct. 2019;10:2701–2709. doi: 10.1039/c8fo02485e. [DOI] [PubMed] [Google Scholar]
- Yin J., Li Y., Han H., Liu Z., Zeng X., Li T., Yin Y. Long-term effects of lysine concentration on growth performance, intestinal microbiome, and metabolic profiles in a pig model. Food Funct. 2018;9:4153–4163. doi: 10.1039/c8fo00973b. [DOI] [PubMed] [Google Scholar]
- Youssef I.M., Khalil H.A., Jaber F.A., Alhazzaa R.A., Alkholy S.O., Almehmadi A.M., Alhassani W.E., Al-Shehri M., Hassan H., Hassan M.S., Abd El Halim H.S., El-Hack M.E.A., Youssef K.M., Abo-Samra M.A. Influence of dietary mannan-oligosaccharides supplementation on hematological characteristics, blood biochemical parameters, immune response and histological state of laying hens. Poult Sci. 2023;102 doi: 10.1016/j.psj.2023.103071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X., Ma F., Dai H., Liu J., Hashem N.M., Sun P. Animals. 2023. Effects of different galacto-oligosaccharide supplementation on growth performance, immune function, serum nutrients, and appetite-related hormones in holstein calves. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zapata O., Cervantes A., Barreras A., Monge-Navarro F., González-Vizcarra V.M., Estrada-Angulo A., Urías-Estrada J.D., Corona L., Zinn R.A., Martínez-Alvarez I.G., Plascencia A. Effects of single or combined supplementation of probiotics and prebiotics on ruminal fermentation, ruminal bacteria and total tract digestion in lambs. Small Rumin Res. 2021;204 [Google Scholar]
- Zeng M, van Pijkeren JP, Pan X. Gluco-oligosaccharides as potential prebiotics: Synthesis, purification, structural characterization, and evaluation of prebiotic effect. Compr Rev Food Sci Food Saf. 2023;22:2611–2651. doi: 10.1111/1541-4337.13156. [DOI] [PubMed] [Google Scholar]