Abstract
Introduction
Postpartum haemorrhage (PPH) remains a leading global cause of maternal deaths, with over 70,000 annual deaths. Low- and middle-income countries disproportionately bear this burden, often due to compromised cold chains for oxytocin and insufficient staff training.
Methods
This review synthesized thirty publications on heat stable carbetocin (HSC) published from January 2018–March 2025. Eligible studies include randomized trials, systematic and narrative reviews, cost or economic evaluations, and implementation studies. Study findings were categorized into clinical efficacy and safety of HSC, cost-effectiveness, and adoption barriers and facilitators from field implementation, and sustainability strategies.
Results
Several large-scale trials, especially the CHAMPION trial, demonstrated that HSC is clinically non-inferior to oxytocin. In a pilot study from Nigeria (n = 18,364 deliveries), 56% of women received HSC for PPH prevention and PPH incidence dropped to 0.8%. Cost models from India estimated that HSC could prevent about 5,500 additional PPH cases and save five maternal lives per 100,000 births when priced comparably to oxytocin. Programs in Kenya and conflict-affected South Sudan achieved >90% coverage of HSC by implementing WHO policy updates, pooled procurement, simulation drills for frontline health workers, and appointing “PPH champions”. Awareness of uterotonics and PPH danger signs among postpartum women increased from 48% to 81% after community-based intervention in Kenya. The thermostability of HSC (up to three years without refrigeration at a temperature of ≤30°C) addresses gaps in cold chain associated with oxytocin and reduces wastage from degradation. Integrating HSC with tranexamic acid and other postpartum haemorrhage bundle elements further improved maternal outcomes.
Conclusion
Heat-stable carbetocin offers a viable, cost-effective uterotonic strategy in LMIC settings. Consistent training, supportive supervision, regulatory oversight, and domestic funding, including private sector investment, are critical to achieving widespread adoption of HSC in LMIC. Expanding HSC across resource-limited settings could substantially reduce PPH-related deaths and accelerate maternal survival goals.
Keywords: heat-stable carbetocin, postpartum haemorrhage, low- and middle-income countries, humanitarian setting, EMOTIVE, tranexamic acid
Introduction
Postpartum haemorrhage (PPH) remains the leading cause of maternal mortality worldwide and accounts for significant proportion of all maternal deaths. Despite the existence of effective prophylactic interventions, nearly fourteen million women experience PPH annually, and over 70,000 of these cases lead to death of which 99% occur in low- and lower-middle income countries.1–3 Low- and middle-income countries (LMICs) are disproportionately affected as a result of fragile health systems, limited cold-chain infrastructure, staff shortages, and fragmented obstetric services which undermine the efficacy of standard uterotonics such as oxytocin. In many LMIC settings, staff shortages do not just reflect low provider numbers but also insufficient refresher opportunities, which can weaken PPH management protocols.4–8 Carbetocin, an oxytocin analogue developed by Ferring Pharmaceuticals as Pabal, received its first approval in Canada in June 1997 and initially indicated only for prevention of PPH following caesarean sections. Conventional carbetocin, like oxytocin, typically requires cold-chain storage (2–8°C) to maintain potency, a requirement often unmet in tropical and conflict-affected facilities.3–6 Carbetocin acts within 1–2 minutes, has safety comparable to oxytocin, and its longer half-life facilitates sustained uterine contraction. Heat-stable carbetocin (HSC) is a long-acting analogue of oxytocin which addresses this vulnerability since it remains stable at ambient temperatures (up to 30°C) for at least three years, even at 75% humidity.5,6 This thermostability is critical for health facilities in LMIC to store a reliable uterotonic without depending on constant refrigeration or uninterrupted power supply. In 2018, following the Carbetocin HAemorrhage PreventION (CHAMPION) trial,9 the guideline development group of WHO updated the uterotonic recommendations for PPH prevention to include carbetocin. The WHO 2018 uterotonic guidance recommends that for all births, an effective uterotonic should be used in the third stage of labour to prevent PPH. The update specifically notes that heat-stable carbetocin can be used as a first-line option in settings where its cost is comparable and it is available, especially where maintaining a cold chain for oxytocin is problematic. In 2019, HSC was added to the WHO Model List of Essential Medicines (core list of reproductive health medicines) for PPH prevention. Following this development, the International Federation of Gynecology and Obstetrics (FIGO) and International Confederation of Midwives also issued a joint statement (FIGO/ICM 2021) endorsing uterotonics for active management of third stage of labour and acknowledging HSC as an important alternative to oxytocin for PPH prevention when cold-chain or quality of drugs is a concern. These developments signalled to countries and procurement agencies that HSC is an evidence-based intervention ready for adoption. In October 2023, WHO released a comprehensive “Roadmap to Combat Postpartum Haemorrhage 2023–2030”,6 The roadmap further solidifies the role of HSC in the global strategy for PPH reduction. This roadmap, developed with multiple stakeholders, outlines key goals for scaling up proven interventions and innovations. One of its central recommendations is to optimize uterotonic prophylaxis for all births in countries with high burden of PPH by 2030. It explicitly highlights the use of heat-stable uterotonics as the preferred option in environments where refrigeration cannot be assured, and this is the reality in many LMIC facilities.6 The guideline also underscores the synergistic role of tranexamic acid (TXA) in managing PPH and suggests a complementary pathway where HSC prevents PPH while TXA (with other bundle elements) addresses established post partum haemorrhage.4–6 The WHO also cautioned that HSC should only be used for PPH prevention after delivery of the baby (not for induction or augmentation of labour, and not for treatment of PPH) and that introducing it must be accompanied by provider education to prevent misuse.
Despite its promise, large-scale adoption of HSC faces challenges such as unfamiliar protocols, uncertain financing, and fragmented supply chains, among others.8 Many policymakers require documented cost-effectiveness and robust field evidence before updating national guidelines or essential medicines lists. Providers are also hesitant in switching from the ubiquitous oxytocin given the inadequate training or confusion over dosage and timing.8 This review consolidates the latest data on clinical efficacy, safety, cost effectiveness, and field implementation experiences of HSC in humanitarian and stable settings. It also highlights policy frameworks, especially the WHO Roadmap, that can accelerate adoption. Examples from Nigeria, Kenya, India, Ghana, Uganda, South Sudan, and other settings illustrate key strategies to integrate HSC into routine PPH prevention. The discussion includes synergy with TXA and other PPH bundle elements and recommended steps for policymakers. The review aims to build a convincing argument for LMICs to scale up HSC as part of a comprehensive postpartum haemorrhage management bundle.
Methods
Literature Search Strategy
A structured literature search (January 2018–March 2025) was conducted in PubMed, Dimensions, Google Scholar, and the WHO Global Health Library. Keywords included “heat-stable carbetocin”, “carbetocin”, “oxytocin”, “LMICs”, “maternal mortality”, “PPH prevention”, and “cold-chain uterotonics”. The search prioritized randomized controlled trials (RCTs), observational studies, systematic reviews, policy documents, and cost or budget-impact evaluations, which referenced HSC or specified heat stability of carbetocin.
Eligibility Criteria
Studies are eligible if they (1) analysed HSC or specifically mentioned that carbetocin required no cold-chain storage in the context of postpartum haemorrhage, (2) evaluated cost or supply considerations, or (3) documented real-world experiences of HSC introduction in LMICs. Articles that addressed carbetocin without differentiating it from heat-stable formulations were included only if relevant details were provided. The review excluded papers, which focus solely on other uterotonics (eg, ergometrine or misoprostol) without mentioning HSC or PPH interventions.
Study Selection and Flow Diagram
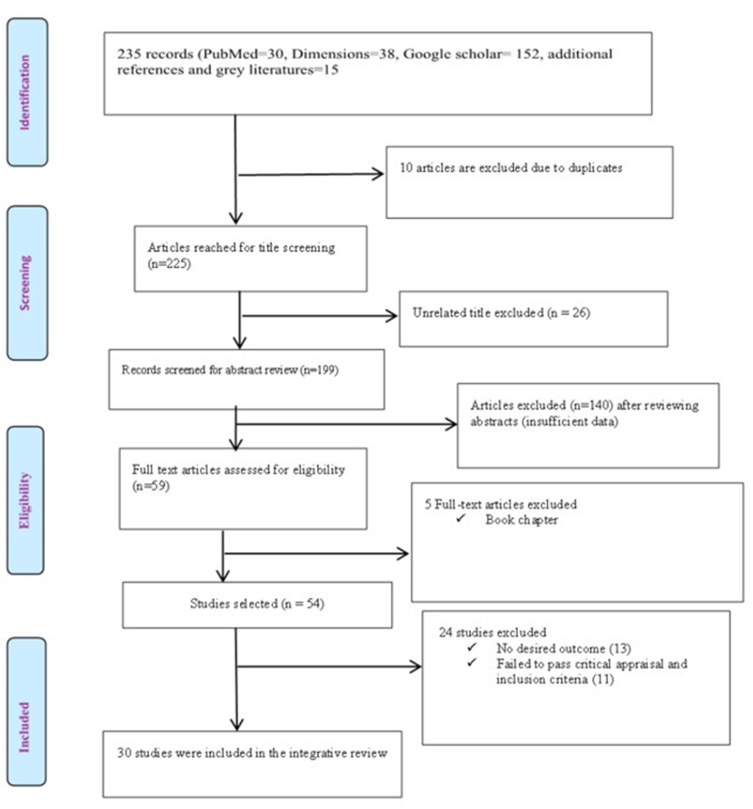
Two researchers (VAA and QESA) independently screened abstracts and full-text articles for inclusion. Disagreements were resolved through discussion. Figure 1 is the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flowchart that presents the selection process, with reasons for exclusion at each stage (eg, insufficient detail on HSC, irrelevance to PPH).
Figure 1.
PRISMA Flow chart of the study selected.
Data Extraction and Synthesis
The review team (VAA, QESA) extracted key data on study design, location, target population, interventions (HSC alone or combined with other measures such as tranexamic acid), sample size, clinical outcomes, and policy or cost findings. Findings were organized into thematic categories: (1) clinical efficacy and safety, (2) cost-effectiveness, (3) implementation experiences, barriers and strategies, (4) Use of HSC/EMOTIVE in Humanitarian and Fragile Settings and Lessons Learned, and 5) Knowledge Gaps and Sustainability. Table 1 summarizes these references.
Table 1.
Overview of Included Studies
| S/N | Source | Setting | Design | Population/Target Audience | Intervention/Main Content | Key Findings |
|---|---|---|---|---|---|---|
| 1 | Salihu et al, 20241 | High-burden LMICs | Editorial / Special Collection Overview | Not applicable | Provided an overview of postpartum haemorrhage (PPH) implementation research in LMICs. Underscored prevention and treatment gaps. | Emphasized the urgency of tackling PPH mortality and the need for coordinated evidence-based strategies in resource-limited settings. |
| 2 | Fievet et al, 20242 | Global perspective | Commentary / Review on Maternal Complications | Not specified (pregnancy/childbirth focus) | Explored leading maternal health threats, including PPH and challenges of quality uterotonic. | Highlighted PPH as a major cause of maternal mortality and stressed supply-chain barriers for effective uterotonics. |
| 3 | Ameh & Althabe, 20223 | Global, emphasis on postpartum haemorrhage | Commentary / Editorial | Not applicable | Discussed policy, practice, and research to improve global PPH management, aligned with WHO recommendations. | Stressed bridging policy and practice for PPH care and called for strengthened uterotonic availability and updated guidelines. |
| 4 | Amode et al, 20244 | Nigeria (3 states, secondary/tertiary facilities) | Mixed-methods implementation research | 18,364 deliveries + providers | Examined uterotonics use patterns and HSC acceptability/safety for preventing PPH. | Showed HSC prophylaxis in 56% of deliveries with a low PPH incidence (0.8%); providers preferred HSC for easier storage and fewer side effects. |
| 5 | Tran et al, 20245 | South Sudan, basic obstetric facilities | Implementation study (stepwise) | 3,142 women (Feb 2022–Feb 2023) | Assessed HSC + TXA integration for PPH prevention/treatment through capacity-strengthening (training, kits, algorithms). | Achieved >99% uterotonic coverage; shifted primarily from oxytocin to >80% HSC; introduced TXA for treatment but staff had some confusion over medication roles in crisis settings. |
| 6 | WHO, 20236 | Global Policy Framework (2023–2030) | Policy document (Roadmap) | Ministries of Health, donors, providers | Presented a strategic roadmap to reduce PPH morbidity/mortality, emphasized supply-chain improvements, systematic training, and robust data collection. | Called for universal uterotonics (including HSC) and early detection/treatment (TXA) within strengthened health systems; defined specific PPH prevention/management goals by 2030. |
| 7 | Bautista et al, 20247 | Ghana | Economic modeling | National-level data | Modelled economic burden of PPH from substandard uterotonics, assessed costs to families and the healthcare system. | Revealed a about $18.8 million annual burden tied to poor-quality uterotonics; estimated about 20,000 PPH cases could be averted via improved drug quality. |
| 8 | Ng’ang’a et al, 20228 | Sub-Saharan Africa (various) | Advocacy initiative/commentary | Policy-level stakeholders | Examined slow inclusion of WHO-recommended PPH meds (TXA, HSC) in national guidelines/EMLs. | Reported fragmented processes as a key hurdle and called for policy alignment, enhanced regulation, and multi-stakeholder engagement. |
| 9 | Widmer et al, 20189 | Twenty-three sites in ten countries | Randomized, double-blind, noninferiority trial | 29,645 women (vaginal birth) | Compared HSC vs oxytocin for PPH prevention; both drugs remained cold to maintain blinding. | Found HSC non-inferior to oxytocin for ≥500 mL blood loss or additional uterotonics; non-inferiority for ≥1000 mL was not demonstrated (low event rate), supporting role of HSC in certain contexts. |
| 10 | Vernekar et al, 202210 | Single facility, India (WHO CHAMPION sub-study) | Randomized controlled trial (nested) | 1,799 women (vaginal delivery) | Compared change in postpartum haemoglobin: HSC (100 µg IM) vs oxytocin (10 IU IM). | Recorded a slightly lower post-delivery haemoglobin in the HSC group; overall difference was small, and no major adverse events occurred. |
| 11 | Ghosh et al, 202411 | Derived from CHAMPION trial data | Secondary analysis of RCT data | Women at risk for PPH | Investigated macrosomia, high parity, or prior PPH as factors affecting HSC vs oxytocin prophylaxis. | Demonstrated that prophylactic efficacy of carbetocin remained consistent across these risk subgroups, indicating broad HSC applicability. |
| 12 | Malm et al, 201812 | Not country-specific (lab-based) | Development & stability research | Focus on HSC formulation for LMICs | Studied a heat-stable carbetocin to withstand ≥30°C. | Concluded that HSC retained ≥95% purity over three years at 30°C (and shorter stints at 50–60°C), confirming feasibility for non-refrigerated LMIC distribution. |
| 13 | Ai et al, 202113 | A systematic review, multiple RCTs | Systematic review + meta-analysis | 17 RCTs on carbetocin side effects | Focused on side effects (eg, vomiting, diarrhea) for carbetocin vs oxytocin. | Found lower risk of vomiting and a slightly higher rate of diarrhea with carbetocin, indicating an overall manageable side-effect profile for PPH prophylaxis. |
| 14 | Yaliwal et al, 202414 | Single center, India (elective C-sections) | Single-blinded, randomized, parallel-group trial | 212 pregnant women (full-term) | Compared 50 µg vs 100 µg carbetocin on cardiovascular effects in caesarean delivery. | Reported no significant differences in heart rate, blood pressure, or cardiac biomarkers, implying similar cardiovascular safety at both doses. |
| 15 | Al Zubaidi & Alhaidari, 202115 | Iraq, emergency caesarean section | Randomized controlled trial | 300 participants (2:1 ratio) | Compared HSC (100 µg IV) vs oxytocin (10 IU) for PPH prevention in emergency C-sections. | Reduced additional uterotonic use and effectively controlled bleeding with minimal hemodynamic changes, confirming non-inferiority to oxytocin. |
| 16 | Chikkamath et al, 202116 | Subanalysis of WHO CHAMPION data | Secondary analysis of extensive RCT data | 10,040 participants, vaginal birth (no PPH interventions) | Examined third-stage labour duration (TSL) and postpartum blood loss in prophylactic uterotonics users. | Observed a steep rise in blood-loss if TSL ≤ 10 min; patterns were similar for oxytocin and HSC, and prolonged TSL correlated with increased bleeding. |
| 17 | Rosmini et al, 202417 | Humanitarian settings (S. Sudan, Uganda) | Mixed methods training initiative (LDHF) | 126 midwives and providers (9 sessions) | Implemented low-dose/high-frequency refresher training on PPH prevention/treatment (HSC + TXA). | Showed significant gains in knowledge, skill retention, and confidence. Encouraged midwifery-led drills and ongoing support in fragile settings. |
| 18 | Ruto et al, 202418 | Public sector hospitals in Kenya (39) | Mixed methods (pre-post + qualitative) | 171 hCPs, 19 policymakers | Investigated HSC introduction for PPH prevention, gathered policy and provider insights. | Found 98.8% HSC coverage by end-line. Providers commended minimal cold-chain dependence; policymakers called for budget allocation to sustain usage. |
| 19 | Tran et al, 202319 | Basic maternity in S. Sudan, Uganda | Qualitative study (FGDs, interviews) | 64 healthcare staff (mostly midwives) | Explored an HSC/TXA package for PPH prevention in fragile humanitarian contexts, with focus on provider feedback. | Revealed providers regarded HSC/TXA as effective and user-friendly, noting confusion with older uterotonics and highlighting the usefulness of supportive tools (WhatsApp, better record-keeping). |
| 20 | Tancred et al, 202420 | Uganda, Ghana (referral facilities) | Mixed-methods readiness assessment | Hospitals able to provide C-sections and transfusions | Explored facility readiness for PPH care, focusing on staff competence and possible new meds (HSC, TXA). | Found decent readiness but cited limited staffing, scarce blood transfusions, and incomplete TXA/HSC adoption; recommended minimal upgrades for better PPH care. |
| 21 | Rushwan et al, 202421 | LMICs, postpartum haemorrhage | Feasibility study (implementation research) | Various LMICs implementing lesser-used PPH meds | Summarized real-world feasibility, acceptability, and proper usage of novel PPH medicines, including HSC and TXA. | Concluded these medicines integrated well into routine care with sufficient training and oversight; emphasized the importance of user acceptance and correct usage. |
| 22 | Muthamia et al, 202422 | 10 Kenyan counties, 39 facilities | Mixed methods (baseline/end-line + qualitative) | 191 postpartum women, 20 IDIs | Evaluated postpartum women’s knowledge of danger signs and uterotonics, along with respectful care, after a community-based intervention. | Documented a rise in uterotonic awareness (48% → 81%) and minimal disrespect/abuse. Study underscored the value of provider communication and adequate ANC visits. |
| 23 | Gülmezoglu et al, 202423 | LMICs, maternal medicines | Commentary / Thematic analysis | Not specifically stated | Discussed the journeys of HSC and TXA, stressed the need for national ownership, end-to-end planning, and robust supply chains in maternal-health drug introductions. | Argued that sustained introduction of maternal meds requires holistic strategies from R&D to in-country distribution, pointed to ongoing challenges in HSC/TXA access. |
| 24 | Cook et al, 202324 | India (public sector births) | Decision-tree cost-effectiveness model | Modelled 100,000 births scenario | Compared cost and outcomes of HSC vs oxytocin/misoprostol for routine PPH prophylaxis. | Estimated prevention of about 5,468 additional PPH cases and about $171,700 saved per 100k births; projected about $11.4 million in yearly savings at about 19% coverage. |
| 25 | Theunissen et al, 202125 | India, Kenya, Nigeria, Uganda | Cost of PPH vs no-PPH analysis | 2,966 women (PPH cases + random controls) | Investigated hospital-care cost differentials for women experiencing PPH vs those without, considering uterotonic type. | Showed PPH care cost 21–309% more than non-PPH care, with wide facility/country variations; underlined the economic case for higher-quality uterotonics. |
| 26 | Alwadhi et al, 202426 | Public health facilities in Dewas, India | Retrospective study (PPP model) | 18,497 women admitted for delivery | Evaluated a public-private partnership introducing HSC in India’s public system, reviewed feasibility, and correct usage. | Achieved 99.9% timely uterotonic coverage; only 3.43% did not receive HSC prophylaxis. PPH incidence was 0.7%, with zero adverse events; concluded PPP was feasible for wider scale-up of HSC. |
| 27 | Hofmeyr, 202327 | Global overview; postpartum haemorrhage context | Narrative review of emerging PPH treatment | Not specific population | Summarized novel or improvised PPH management approaches (eg, microneedle patches, compression sutures), focusing on low-resource settings. | Found many under-researched ideas and called for rigorous studies to determine effectiveness. |
| 28 | Maged et al, 202428 | Multi-country | Systematic review and meta-analysis | Low-risk women undergoing caesareans | Compared carbetocin vs oxytocin in preventing PPH for caesarean deliveries. | Reported that carbetocin reduced extra uterotonic use, transfusions, and haemoglobin drop with no significant increase in adverse events. |
| 29 | Trivedi et al, 202529 | Tertiary hospital, Jharkhand (India) | Double-blind RCT | 42 women undergoing C-section | Compared IV carbetocin vs IM oxytocin for PPH prevention in caesarean deliveries. | Demonstrated reduced mean blood loss and haemoglobin drop in the carbetocin group; safety profiles for both interventions were equivalent. |
| 30 | Ajima et al, 202530 | Kano, Lagos, Niger (Nigeria) | Mixed methods + HCD | Policymakers, health workers, supply actors | Introduced HSC for PPH through supply-chain strengthening, human-centered design, and a market-shaping approach. | Documented improved HSC uptake across 87 facilities; DRF financing, training, and stakeholder engagement supported stable uterotonic access. |
Quality Assessment
No formal risk-of-bias scoring was performed. Conflicting results were cross-checked with large-scale trials (eg, WHO CHAMPION) and relevant WHO guidelines. Potential publication bias, which favour successful pilot experiences, was considered a limitation when interpreting results.
Results
Overview of Included Studies
The search yielded thirty publications that met the inclusion criteria (see Table 1). Twenty-six studies reported clinical performance, policy integration, or cost implications of HSC in LMICs. Five studies reported the role of carbetocin in caesarean deliveries and provided new insights on hemodynamic stability and reduction in blood loss.14,15,28,30,31 Ten studies4,5,17–22,26,30 reported real world implementation experience with HSC; three were economic modelling/cost studies.7,24,25
The Burden of PPH and Gaps in Conventional Uterotonics
PPH disproportionately affects LMICs due to weaknesses at multiple system levels. Many peripheral clinics lack functional cold-chain storage, which undermines the potency of oxytocin. Substandard or falsified oxytocin vials circulate widely, and this often leads to unpredictable effect on PPH prevention.7 Frequent staff turnover also limited routine supervision, and inadequate supply forecasting, which further compounds these problems.3,8 Although misoprostol is temperature-stable, it can show inconsistent performance or poor quality if sourced improperly and this dampens provider confidence. Programs that encourage in-facility deliveries still confront high risks of postpartum haemorrhage if the uterotonics have degraded. Studies have shown that better quality of uterotonic correlates with fewer acute PPH complications and broader cost savings for health systems.2,7
Clinical Efficacy and Safety of Heat-Stable Carbetocin in vaginal and caeserean delivery
The CHAMPION trial remains the most groundbreaking large-scale RCT comparing intramuscular HSC with intramuscular oxytocin.9 CHAMPION compared a single intramuscular dose of heat-stable carbetocin to the standard oxytocin injection given immediately after vaginal delivery for PPH prevention. The results demonstrated that HSC is clinically non-inferior to oxytocin in preventing primary PPH. About 14% of women in both groups experienced blood loss ≥500 mL or needed additional uterotonics.9 Also, rates of severe PPH (≥1000 mL blood loss) need for blood transfusions or surgical interventions, and adverse maternal outcomes were statistically similar between HSC and oxytocin groups.9 Both drugs had low rates of side effects such as hypotension or tachycardia, and no unexpected safety concerns were found. These findings established that HSC offers equivalent efficacy in the prevention of PPH with a comparable safety profile to the gold-standard uterotonic. Furthermore, a 2018 Cochrane review compared 196 uterotonic trials (over 130,000 participants).29 The study found that carbetocin was among the top three most effective agents at preventing ≥500 mL blood loss, comparable to combinations like oxytocin + ergometrine and superior to oxytocin alone in terms of efficacy.29 Carbetocin was the only highly efficacious uterotonic not associated with an increase in significant side effects relative to oxytocin.29 Ergometrine-containing regimens, while effective, caused more hypertension and other side effects, whereas carbetocin provided a favourable balance of high efficacy and safety. Oxytocin typically degrades if exposed to temperatures above 8°C for prolonged periods, hence the requirement for an unbroken cold chain. HSC, by contrast, preserves ≥95% potency under ambient conditions (up to 30°C) for at least three years.12 Field reports in LMICs highlight that actual storage conditions, characterized by power outages and unreliable transportation, often nullify the potency of oxytocin by the time it is eventually administrated.5,8 The thermostability of HSC protects against such degradation, which therefore minimizes the risk of postpartum hemorrhage due to weak uterotonic doses. The thermostability of HSC also means that providers can maintain stocks without strict refrigeration or daily temperature monitoring and can therefore save time for other maternal health and related services.17 It is therefore possible that the head-to-head results of the CHAMPION trial in ideal cold-chain conditions might underestimate the true advantage of HSC in the field since oxytocin in routine use often loses potency due to heat exposure.23 Therefore, the efficacy of HSC equivalence to oxytocin in trials translates to a potential efficacy edge in practice where cold chain is weak since it reliably provides the uterotonic effect intended, whereas oxytocin ampoules may be inactive by the time they reach labour wards in some LMIC facilities. Other secondary analysis of CHAMPION trial and side effect studies reported slightly lower post-delivery haemoglobin, consistent prophylactic efficacy of carbetocin, feasibility for non-refrigerated LMIC distribution, manageable side-effect profile including cardiovascular effects of HSC when used for PPH prophylaxis.10–16
Moreover, recent studies now provide in-depth insights into the role of heat-stable carbetocin in preventing excessive bleeding during caesarean sections.14,15,28,31 An updated systematic review and meta-analysis showed that carbetocin decreased the requirement for additional uterotonics and blood transfusions among women with minimal risk undergoing caesarean delivery.28 Another randomized study in India observed that carbetocin reduced intraoperative blood loss and shortened the duration of maternal hemodynamic compromise during caesarean section.31 This was supported by another prospective trial from Poland, which found that carbetocin produced more stable hemodynamic than oxytocin, leading to fewer fluctuations in blood pressure and heart rate after drug administration.32 The extended duration of action of carbetocin compared to oxytocin makes it suitable for single-dose prophylaxis during caesarean sections without the need for maintenance infusions. Table 2 below provides a comparative summary of key uterotonics used in LMIC settings, highlights their clinical properties, routes of administration, and WHO recommendations for postpartum haemorrhage prevention and treatment.
Table 2.
Key Characteristics and WHO Recommendations for Common Uterotonics
| Uterotonic | Typical Prophylaxis Dosage | Heat Stability | Onset | Approx. Duration of Action | Route(s) | Side Effects | Requires Cold Chain | WHO Recommendation |
|---|---|---|---|---|---|---|---|---|
| Heat-Stable Carbetocin | 100 µg IM or IV (single dose) | Stable up to 30°C for ≥3 years | 1–2 minutes | 60 minutes | IM / IV | Mild hypotension, nausea/vomiting (rare) | No | Prevention of PPH for both vaginal and caesarean deliveries where cost is comparable and cold chain is unreliable. Not recommended for induction/augmentation or treatment of PPH. |
| Oxytocin | 10 IU IM or IV (single dose) | Unstable if >8°C for prolonged periods | 1–2 minutes | 30 minutes | IM / IV | Hypotension, tachycardia, water intoxication | Yes | Gold-standard prophylaxis and treatment in all settings if quality and cold chain can be guaranteed. Also used for induction and augmentation. |
| Misoprostol | 400–600 µg oral (prophylaxis) | Thermostable | 2–5 minutes (oral) | 60+ minutes (oral) | Oral / Rectal / Sublingual | Shivering, fever, gastrointestinal upset | No | Prevention (when injectables not available) and treatment of PPH. Not used for induction/augmentation. Recommended for vaginal births. |
| Ergometrine | 0.2 mg IM or IV (single dose) | Requires 2–8°C (degrades in higher temperatures) | 2–7 minutes | Up to 180 minutes (3 hours) | IM / IV | Hypertension, nausea, vomiting | Yes | Prevention or treatment of PPH but less favoured due to hypertensive side effects. Not used for induction/augmentation. |
| Oxytocin + Ergometrine | eg, (5 IU Oxytocin + 0.5 mg Ergometrine) IM | Same as Oxytocin/Ergometrine requirements | 1–2 minutes (Oxytocin) | Up to 180 min (ergometrine portion) | IM | Hypertension, more adverse effects than oxytocin alone | Yes | Prevention of PPH only when combined for additional efficacy, not for induction/augmentation. Use with caution in hypertensive women. |
Cost-Effectiveness of HSC in LMIC
Another critical consideration for health systems in LMIC is whether adopting heat-stable carbetocin is affordable and cost-effective compared to existing uterotonics like oxytocin. Several economic evaluations and cost modelling studies in recent years have examined HSC introduction in contexts with weak cold-chain infrastructure. Overall, the evidence suggests that HSC can be highly cost-effective and even cost-saving in LMICs, particularly when the health system costs of averted PPH complications are factored in the model.
In 2023, a study evaluated the cost-effectiveness and budget impact of HSC for PPH prophylaxis in India’s public health system.24 The study used a decision-tree model across 100,000 births and compared three strategies, namely: prophylactic oxytocin, misoprostol, or heat-stable carbetocin.24 The model incorporated real-world factors such as varying levels of care (primary to tertiary facilities), differing PPH risks for vaginal vs caesarean births, and downstream costs of managing PPH and its complications. The findings showed that adopting HSC in place of oxytocin could reduce overall costs to the health system and improve outcomes. Per 100,000 births, the use of HSC could prevent an additional 5,500 cases of PPH and avert about five maternal deaths, which translates to 244 more DALYs (disability-adjusted life years) saved, compared to oxytocin.24 These health gains stem from better maintenance of efficacy (no losses from cold-chain failures) of HSC leading to fewer cases of haemorrhage and emergency interventions. Moreover, the averted costs of treating PPH (surgeries, transfusions, extended care) outweighed the higher unit price of HSC in this model. Given the subsidized price assumed for India’s public sector, HSC dominated oxytocin and was more cost-effective and cost-saving.24 The authors conclude that scaling up HSC for all births in India would increase the efficiency of health spending and contribute to achieving reduction in maternal mortality.
In other LMIC settings, similar analyses echo these findings. Another economic evaluation from Nigeria modelled the introduction of HSC in public hospitals (where PPH contributes about 22% of maternal deaths).4 The authors found that HSC would reduce PPH incidence and related maternal deaths versus the current mix of oxytocin/misoprostol. The cost per life saved and cost per DALY averted were well below conventional willingness-to-pay thresholds, making HSC a highly cost-effective intervention in Nigeria.4 Nigeria faces a severe problem of substandard or falsified oxytocin in circulation due to broken cold chains and poor regulation.4 By replacing a portion of these ineffective doses with reliable HSC, the health system could avoid expensive emergency referrals and interventions for uncontrolled haemorrhage. Budget impact projections in Nigeria show that while procurement costs for HSC are higher, the overall five-year maternal health budget could translate to net savings from fewer PPH complications.4
However, cost-effectiveness is a function of the price point of HSC. In contexts where HSC is priced significantly higher than oxytocin without subsidy, the value proposition may be less favourable.33 For example, an earlier analysis in the Philippines found that at a price of USD $18 per dose, carbetocin was not cost-effective compared to oxytocin for PPH prevention for both vaginal delivery and caesarean delivery, given the country’s cost-per-QALY threshold. The incremental cost per QALY gained in that scenario exceeded what the Philippine health system typically finds acceptable.33 Sensitivity analyses showed that the cost-effectiveness of HSC was highly sensitive to the drug price. A substantial reduction in the price of HSC or higher than assumed efficacy would lead to significant cost-effectiveness. This highlights an important implementation point, which is affordability. Global efforts (through UNFPA, MSD for Mothers, Ferring Pharmaceuticals, etc.) have focused on ensuring HSC is offered to LMICs at an affordable price close to that of oxytocin. Since 2019, registration of HSC was kickstarted in over ninety countries at a cost similar to that of oxytocin, as part of an access agreement.34 If such price parity is achieved, the economic barrier to HSC adoption will be minimal, and the substantial downstream savings (from averted maternal morbidity and mortality) will make it an extremely attractive investment for health systems.7,25
Implementation Challenges and Strategies in LMICs
Translating the promise of HSC into field reality in low-resource settings requires confronting several implementation challenges. These include policy and regulatory barriers, supply chain and procurement bottlenecks, training needs of healthcare workers, quality improvement and peer learning, as well as monitoring and evaluation. A study conducted in Ghana and Uganda found a moderate readiness but staff shortages and inadequate infrastructure for adoption of HSC was a barrier.20 Experiences from countries like Nigeria, Kenya, India, Uganda, and South Sudan illustrate the barriers and highlight strategies to overcome these challenges, from policy adoption down to facility-level practice.
Policy and Regulatory Barrier
The first challenge is policy and regulatory hurdles in getting HSC approved and included in national protocols. Many LMICs have been slow to update their national clinical guidelines and Essential Medicines Lists to include HSC, even after the recommendation by WHO.4,8 Some of the factors include limited awareness of the new evidence among policymakers, bureaucratic processes for drug registration, and uncertainty about cost and supply. For instance, by 2021 only a handful of high-burden African countries had added HSC to their PPH guidelines. Advocacy efforts by organizations like WACI Health in fourteen sub-Saharan countries found that upstream knowledge gaps were a major barrier to policy action. National stakeholders were often not fully aware of the 2018 WHO uterotonic guideline or the addition of HSC to the WHO Essential Medicines List.8 To address this, Ministries of Health in Nigeria, Kenya, Uganda, and other countries have been engaged through workshops and policy dialogues to disseminate the global guidance and build buy-in for HSC. These efforts have started to yield results. Nigeria, for example, convened a national expert meeting in 2022 to consider HSC and updated its National PPH guidelines in 2024 to include HSC, Tranexamic acid, calibrated drapes and PPH bundle elements.35 Kenya and India adopted carbetocin early, being part of the CHAMPION trial and have included carbetocin as a recommended option in maternal health guideline in certain circumstances where the quality of oxytocin is a concern.22,26
Procurement and Supply Chain
Following policy approval, ensuring HSC availability requires robust procurement and distribution planning. The supply chains for maternal health commodities in LMIC often face funding constraints, difficulties in forecasting, and bottlenecks in last-mile delivery. Introducing HSC warrants the addition of a new product into the supply system and entails training logisticians to forecast demand, securing financing for procurement (potentially at a higher unit cost initially), and managing distribution. Since HSC is thermostable, it eases some logistics (no need for refrigerated transport or storage, which is a major advantage) but must still be transported to all maternity clinics. Some countries have strategized by leveraging global procurement mechanisms. For examples, a consortium of partners (Clinton Health Access Initiative and UNFPA) is helping countries like South Sudan and Uganda to procure HSC through pooled orders to achieve lower prices and guaranteed quality.5,25 The additional benefit of HSC is reduced wastage. Unlike oxytocin, which might expire quickly due to cold-chain compromises, HSC maintains viability, which translates to fewer discarded vials and less frequent need for resupply. However, to safeguard against counterfeit products, governments must work closely with regulatory agencies to import only WHO-prequalified or Stringent Regulatory Authority-approved HSC (from trusted manufacturers) to prevent substandard versions from entering the market.21,25 A practical example of this integrated approach comes from Nigeria, where the Smiles for Mothers program successfully introduced HSC in Kano, Lagos, and Niger states.30 The program aligned human-centered design with a market-shaping value chain to address supply chain bottlenecks through policy reforms, Drug Revolving Fund-backed procurement plans, and comprehensive training for health workers and logisticians.30 Ultimately, the Smile for Mothers program demonstrated that coordinated, locally adapted strategies can facilitate sustainable access to HSC in low-resource settings.
Training of Frontline Healthcare Workers
Frontline providers are responsible for the administration of HSC, and their knowledge and attitudes can make or break the intervention.4,18,26 The roll-out of HSC in pilot settings revealed initial confusion among staff about differentiating HSC from oxytocin and tranexamic acid.5,17,19 In South Sudan and Uganda, midwives noted that having multiple new drugs for PPH (HSC for prevention, TXA for treatment) was initially confusing in record-keeping and protocols, especially if they had previously used only oxytocin.5 Therefore, training and clear protocols are vital. Strategies that have worked include updating the active management of third stage of labor (AMTSL) protocols and checklists and reinforcing this through drills. The experience with multifaceted PPH interventions in Nigerian hospitals underscores that training must be ongoing.36 In that study, a combination of on-site drills, distribution of PPH management charts, and periodic supportive supervision led to improved adherence to PPH protocols. As part of introducing heat-stable carbetocin (HSC), programs have adopted the low-dose, high-frequency (LDHF) training approach, which consists of short, focused training or mentorship sessions delivered repeatedly on-site.5,17,19 For example, a pragmatic cluster-randomized trial in Uganda showed that combining peer-assisted learning (PAL) with LDHF simulation training significantly improved provider skills and clinical performance in preventing PPH. The intervention increased timely uterotonic administration from 8% to 50% and reduced the incidence of PPH by 17%.37 In Kenya, post-partum women's knowledge of PPH danger signs and awareness of uterotonic increased from 48% to 81% following a community-based intervention.22 Similarly, a multicounty pilot study in Kenya, Nigeria, South Africa, and Tanzania demonstrated that integrating calibrated drapes for accurate blood loss measurement within simulation-based trainings and clinical drills led to improved adherence to PPH management guidelines, with significant increase in the use of critical commodities like oxytocin and tranexamic acid.38 Moreover, formative research in Kenya, Nigeria, and South Africa highlighted that repeated simulation trainings, supported by clinical champions and structured audit-feedback, strengthened the implementation of the E-MOTIVE care bundle by addressing knowledge gaps, building provider confidence, and fostering effective teamwork.39
Quality Improvement and Peer Practice Sessions
Changing routine practices in busy maternity units can be challenging. Even after stocking HSC and training providers, ensuring consistent administration of the drug to every woman after birth remains an issue. Providers may unintentionally forget or delay uterotonic administration amidst the busy environment of childbirth, or they may hesitate to use a new drug due to misconceptions or uncertainties.39 To address these human factors, facilities have introduced practical quality improvement strategies such as double-check systems and accountability measures. For instance, in Kenya, midwives in charge routinely reviewed delivery records provided gentle reminders to staff who missed documenting prophylactic uterotonics, thereby reinforcing consistent use.18 In Nigeria, empowering respected staff as “PPH champions”—typically experienced obstetricians or senior midwives has proven successful. These champions actively encourage peers to embrace HSC and other new and lesser used PPH commodities, offer support in real-time problem-solving and foster a supportive environment for new practices.40 In addition, incorporating HSC administration into routine emergency obstetric simulation drills has empowered staff to practice together in realistic scenarios led by peers, reinforcing skills, building confidence, and embedding HSC into the team’s daily workflow. These thoughtful and culturally sensitive strategies are critical in bridging the gap between knowledge and consistent practice.
Monitoring and Data Systems
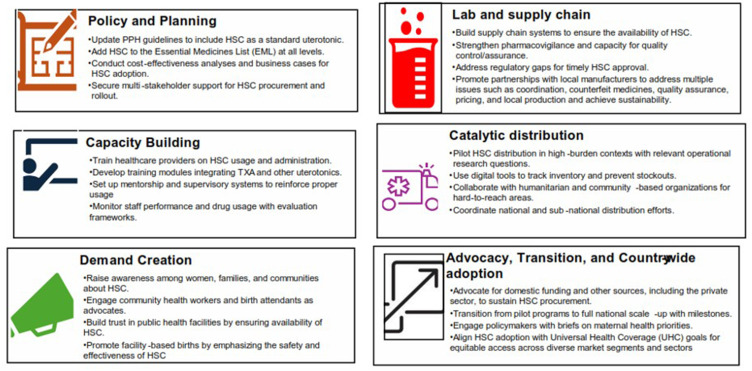
In many countries in LMIC, the health management information systems (HMIS) or facility registers initially have columns for oxytocin or misoprostol usage, but not for carbetocin. This could potentially lead to under-reporting or misclassification. Maternity staff might still tick “oxytocin” even if HSC was given, due to old forms and registers. Revising facility registers, PPH case report forms, and national HMIS to capture “Carbetocin given – Yes/No” is necessary for tracking coverage and impact. Countries piloting HSC and other PPH commodities, like India and Nigeria, have begun to adapt their recording tools accordingly.8 In South Sudan, it was observed that inadequate record systems were a major barrier to measuring the uptake of HSC until new documentation was introduced.4 In addition, pharmacovigilance systems need to be activated to collect any reports of adverse reactions to HSC. Prioritizing M&E including periodic data review meetings can help implementers to track key indicators such as PPH incidence, rates of uterotonic use by type, and any “missed opportunities” that could galvanize mid-course corrections in the implementation strategy.8,19,25 Figure 2 summarizes the Framework for the Introduction and Scale-Up of Heat-Stable Carbetocin and other lesser used PPH commodities in LMICs.
Figure 2.
Framework for the Introduction and Scale-Up of Heat-Stable Carbetocin and other lesser used PPH commodities in LMICs.
Use of HSC/EMOTIVE Bundle in Humanitarian and Fragile Settings and Lessons Learned
In humanitarian and fragile settings, such as conflict zones, refugee camps, and areas with extreme resource limitations, the challenges of PPH management are amplified. These settings often lack reliable electricity (precluding cold storage), have severe shortages of skilled health workers (with maternal care often delivered by midwives or nurses), and face disrupted supply chains. Heat-stable carbetocin offers promise in such contexts, as does the utilization of innovative training and task-sharing models to maximize its impact. Recent implementation studies shed light on how HSC (for prevention) and TXA (for treatment) were introduced in basic maternity facilities in South Sudan and Uganda, countries that host large numbers of displaced persons and typifying humanitarian conditions.5,19
Providers in South Sudan’s camps and Ugandan refugee settlements reported that having HSC available markedly increased their confidence in preventing PPH.5 Previously, they relied on oxytocin, which was of questionable quality due to storage issues in clinics with no refrigerators or erratic fuel for generators. With HSC, midwives could carry a supply in their kit bags without being apprehensive of spoilage and administer it in remote health posts or mobile clinics immediately after delivery. Midwives noted its “rapid and reliable action” and observed fewer incidents of heavy bleeding in women who received HSC, reinforcing their trust in the drug.5 This finding is vital in humanitarian settings, where referral to higher-level care is extremely difficult and preventing haemorrhage at the community or primary facility level may be the only chance to save a woman’s life. Moreover, staff were positive that HSC comes in a single-dose ampoule (100 micrograms in 1 mL), which simplifies coordination during an emergency with no need to assemble an IV drip or worry about dosing multiple tablets as with misoprostol. In fragile settings where human resources are stretched, such simplicity and effectiveness are a major asset. Task-Sharing is also key in this setting. Because doctors and obstetricians are scarce, task-sharing lifesaving interventions with midwives and other cadres is crucial. The introduction of HSC and TXA in South Sudan was achieved entirely through midwives at basic emergency obstetric care centres as no obstetrician was on site. Midwives were empowered to administer HSC for every delivery and to give TXA for PPH without waiting for a physician’s approval.5,19 To ensure these providers were equipped for these new roles, a tailored low-dose high-frequency (LDHF) training program was implemented. Midwives underwent an initial 1–2-day refresher workshop on PPH prevention and treatment, where they practiced with anatomical models and case scenarios using HSC and TXA. This was followed by on-the-job mentorship, where experienced trainers or supervisors conduct monthly visit to each facility to reinforce skills, answer questions, and observe drill scenarios.5,17 A “community of practice” WhatsApp group was also established so that midwives in different clinics could share experiences, ask questions, and receive real-time guidance from experts. These novel approaches (LDHF plus virtual mentoring) mitigated the challenge of limited training opportunities, institutionalized and sustained the correct use of HSC/TXA despite frequent staff turnover.
There are several enablers that explain the success of these interventions. First, both national and local health authorities in these projects gave explicit support to using HSC and TXA (often through official circulars and inclusion in kit lists), which empowered providers to act. The second enabler is the provision of PPH kits. Facilities were supplied with ready-to-use PPH emergency kits, which included HSC ampoules, TXA vials, syringes, IV fluids, and simple tools like a timing clock and an alarm bell.17 These kits are standardized and easily accessible in the delivery room. Midwives reported that having a designated “PPH box” with all components prepared them psychologically to manage PPH more aggressively.5 The third enabler is the availability of visibly displayed Job aids and protocols. Algorithms for PPH management (highlighting when to give HSC, TXA, etc.) were posted on walls and this reinforced the steps during emergencies. The last enabler is community engagement, recognizing that the presence of effective interventions like HSC/TXA made survival more likely, thereby building trust in the community and health system. In these settings, community health workers were sensitized to encourage facility deliveries and to assist with emergency transport. Notwithstanding, the program implementers encountered certain challenges. Newly recruited staff were initially unfamiliar with HSC, so the program adopted ongoing LDHF mentorship to ensure rapid onboarding. Supply disruptions due to insecurity or funding issues sometimes caused commodity shortages in clinics, which highlights the importance of strong coordination with humanitarian supply agencies (eg, UNFPA, NGOs). Initial confusion between medications like TXA and oxytocin was resolved through targeted training and clearly labelled color-coded drug kits. Moreover, extremely low-resource settings often lacked essential equipment like blood pressure cuffs, IV lines, or the capacity for safe blood transfusions. This indicates that HSC and TXA must be integrated into a broader package of emergency obstetric care and PPH bundle. The E-MOTIVE strategy, a WHO-endorsed bundle focusing on Early detection, Massage, Oxytocic drugs, Tranexamic acid, IV fluids, and Escalation incorporates a two-stage approach: prophylactic uterotonics (eg, HSC) to prevent PPH, and administration of TXA plus other bundle measures once PPH is diagnosed. The integrated bundled approach recommended by WHO aims to empower clinicians to prevent and detect haemorrhage early and simultaneously respond with multiple interventions. Despite these barriers, introducing HSC in humanitarian settings has proven transformative and has led to significant improvement in maternal outcomes.
Knowledge Gaps and Sustainability
Despite substantial evidence that HSC is non-inferior to oxytocin, further operational research is needed on best approaches to large-scale adoption in primary care facilities without experienced obstetricians. Future trials could evaluate community-level distribution or synergy with simplified postpartum follow-up using digital tools. Additional cost-effectiveness analyses remain critical in settings that lack external subsidies or stable financing. HSC is not a standalone fix for PPH but an integral part of comprehensive maternal health improvement. Programs must also address antenatal anaemia, ensure blood transfusion capacity for severe cases, and strengthen referral systems. Governments should embed HSC in routine budgets rather than relying solely on donor-funded initiatives. Bulk purchasing or multi-country frameworks can drive the per-vial price closer to that of oxytocin, which will reinforce the cost-effectiveness of HSC. Manufacturers, distributors, and pharmacists play decisive roles in making HSC widely accessible. Clear price agreements, local regulatory enforcement, and targeted training can accelerate the uptake of HSC. Partnerships with entities like Ferring Pharmaceuticals and MSD for mothers who already invest in technology transfers and price negotiations to expand HSC footprint in LMICs will be crucial. Implementation science must investigate how HSC adoption interacts with staff turnover, drug supply disruptions, and variable policy environments. Rigorous monitoring of outcomes (eg, PPH incidence, maternal complications, costs averted) will inform evidence-based policy refinements. Implementation science must also address any potential long-term safety signals or rare adverse events through robust pharmacovigilance systems.
Strengths and Limitations
This integrative review incorporated diverse data sources, which include randomized trials, cost-effectiveness models, and field implementation reports. This provides a multi-dimensional perspective on HSC. The global scope, spanning humanitarian to stable urban contexts, strengthens the transferability of lessons. However, the authors did not perform a formal risk-of-bias assessment. Also, some pilot projects reported short follow-up durations, and publication bias may favour studies showing successful HSC outcomes. References on conventional carbetocin occasionally lack clarity on whether the reported carbetocin was the heat-stable form. Nonetheless, the included body of evidence offers robust support for the clinical merit of HSC and underscores policy enablers for adoption.
Conclusion
Heat-stable carbetocin addresses a major gap in PPH prevention by eliminating cold-chain requirements, which often compromise the effectiveness of oxytocin in many LMIC settings. Multiple studies found that HSC is clinically non-inferior, and cost analyses suggest that once HSC pricing approaches that of oxytocin, it becomes highly cost-effective or even cost-saving. Field experiences from Nigeria, Kenya, India, and South Sudan demonstrate that training, supportive supervision, effective regulation, and pooled procurement are essential to overcome bottlenecks in supply and workforce readiness. Expanding HSC uptake is achievable if national policymakers update guidelines, secure funding, and incorporate HSC into comprehensive maternal health packages that include tranexamic acid and other essential components of the PPH care bundle. These steps align with the WHO 2023–2030 roadmap, which promotes universal uterotonic coverage, routine use of TXA, and system-wide strengthening. Programs must also address ancillary factors like antenatal anaemia, blood transfusion capability, and strong referral pathways for refractory cases. Integrating heat-stable carbetocin in routine practice in LMIC can significantly reduce maternal deaths from PPH, accelerate progress toward Sustainable Development Goals, and build more resilient maternal health systems. Ongoing research and implementation science are required to refine best practices, ensure equitable access, and achieve the vision of no preventable maternal death during childbirth.
Acknowledgments
The authors want to acknowledge Universitas Padjadjaran, Bandung, West Java, Indonesia, for supporting the publication fees.
Funding Statement
The authors would like to acknowledge the contribution of the Scientific Excellence Research Grant to Qorinah Estiningtyas Sakilah Adnani from Universitas Padjadjaran, Bandung, Indonesia (grant number: 908/UN6.3.1/PT.00/2025).
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Salihu HM, Umar AA, Azuine RE, Azuine RE. Special collection: implementation research evidence for prevention and treatment of postpartum hemorrhage in high-burden low- and middle-income countries. Int J MCH AIDS. 2024;13(Suppl 1):S1–S3. PMID: 39629310; PMCID: PMC11583812. doi: 10.25259/IJMA_35_2024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fievet L, Hewgill S, Li W, Ourumis E, Paleologou A, Pecore R. A leading threat to maternal health: complications related to pregnancy and childbirth. Reprod Health. 2024;20:116. doi: 10.1186/s12978-023-01657-1 [DOI] [Google Scholar]
- 3.Ameh C, Althabe F. Improving postpartum hemorrhage care: policy, practice, and research. Int J Gynecol Obstet. 2022;158(s1):11–13. doi: 10.1002/ijgo.14271 [DOI] [PubMed] [Google Scholar]
- 4.Amode OA, Negedu OV, Joseph JT, et al An Implementation research study on uterotonics use patterns and heat-stable carbetocin acceptability and safety for prevention of postpartum hemorrhage in Nigeria. Int J MCH AIDS. 2024;13(Suppl 1):S38–S45. PMID: 39629305; PMCID: PMC11583818. doi: 10.25259/IJMA_1_2024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tran NT, Mugerwa K, Ochan AW, et al Postpartum hemorrhage in humanitarian settings: heat-stable carbetocin and tranexamic acid implementation study in South Sudan. Int J MCH AIDS. 2024;13(Suppl 1):S55–S63. PMID: 39629301; PMCID: PMC11583825. doi: 10.25259/IJMA_8_2023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.A Roadmap to Combat Postpartum Haemorrhage Between 2023 and 2030. Geneva: World. Health Organization; 2023. [Google Scholar]
- 7.Bautista K, Lee YA, Higgins CR, et al Modeling the economic burden of postpartum hemorrhage due to substandard uterotonics in Ghana. PLOS Glob Public Health. 2024;4(6):e0003181. PMID: 38900726; PMCID: PMC11189185. doi: 10.1371/journal.pgph.0003181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ng’ang’a J, Chitimbe T, Mburu R, et al Challenges in updating national guidelines and essential medicines lists in Sub-Saharan African countries to include WHO-recommended postpartum hemorrhage medicines. Int J Gynecol Obstet. 2022;158(s1):11–13. doi: 10.1002/ijgo.14269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Widmer M, Piaggio G, Nguyen TMH, et al WHO CHAMPION Trial Group. Heat-stable carbetocin versus oxytocin to prevent hemorrhage after vaginal birth. N Engl J Med. 2018;379(8):743–752. PMID: 29949473. doi: 10.1056/NEJMoa1805489 [DOI] [PubMed] [Google Scholar]
- 10.Vernekar SS, Goudar SS, Metgud M, et al Effect of heat stable carbetocin vs oxytocin for preventing postpartum haemorrhage on post delivery hemoglobin—a randomized controlled trial. J Matern Fetal Neonatal Med. 2022;35(25):8744–8751. PMID: 34763599. doi: 10.1080/14767058.2021.2001799 [DOI] [PubMed] [Google Scholar]
- 11.Ghosh R, Owa OO, Santos N, et al Heat stable carbetocin or oxytocin for prevention of postpartum hemorrhage among women at risk: a secondary analysis of the CHAMPION trial. Int J Gynecol Obstet. 2024;164(1):124–130. doi: 10.1002/ijgo.14938 [DOI] [PubMed] [Google Scholar]
- 12.Malm M, Madsen I, Kjellström J. Development and stability of a heat-stable formulation of carbetocin for the prevention of postpartum haemorrhage for use in low and middle-income countries. J Pept Sci. 2018;24(6):e3082. PMID: 29700898. doi: 10.1002/psc.3082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ai W, Zeng Y, Ma Y, et al Side-effects of carbetocin to prevent postpartum hemorrhage: a systematic review and meta-analysis of randomized controlled trials. Pharmacol Res Perspect. 2021;9(2):e00745. PMID: 33723868. doi: 10.1002/prp2.745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yaliwal RG, Patil NG, Bidri S, Bidri S. A comparative study of the cardiovascular effects of a lower dose of heat-stable carbetocin versus the standard dose in the prevention of postpartum hemorrhage during elective cesarean delivery: a single-blinded, randomized, parallel-group trial. Cureus. 2024;16(7):e65049. PMID: 39165461. doi: 10.7759/cureus.65049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Al Zubaidi S, Alhaidari T. Heat stable carbetocin vs. oxytocin for the prevention of post-partum hemorrhage in emergency caesarean delivery: a randomized controlled trial. J Perinat Med. 2021;50(2):150–156. PMID: 34535047. doi: 10.1515/jpm-2021-0206 [DOI] [PubMed] [Google Scholar]
- 16.Chikkamath SB, Katageri GM, Mallapur AA, et al Duration of third stage labour and postpartum blood loss: a secondary analysis of the WHO CHAMPION trial data. Reprod Health. 2021;18(1):230. PMID: 34775959. doi: 10.1186/s12978-021-01284-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rosmini A, Mugerwa K, Ochan AW, et al Empowering midwives in humanitarian settings: integrating heat-stable carbetocin and tranexamic acid into postpartum hemorrhage training. Int J MCH AIDS. 2024;13(Suppl 1):S72–S80. doi: 10.25259/IJMA_10_2023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ruto D, Muthamia M, Njeri E, et al Introduction of heat-stable carbetocin for postpartum hemorrhage prevention in public sector hospitals in Kenya: provider experience and policy insights. Int J MCH AIDS. 2024;13(Suppl 1):S28–S37. doi: 10.25259/IJMA_4_2024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tran NT, Mugerwa K, Ochan AW, et al Postpartum hemorrhage in humanitarian settings: implementation insights from using heat-stable carbetocin and tranexamic acid. Int J MCH AIDS. 2023;13(Suppl 1):S64–S71. doi: 10.25259/IJMA_7_2023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tancred T, Weeks AD, Mubangizi V, et al Assessment of structural and process readiness for postpartum haemorrhage care in Uganda and Ghana: a mixed methods study. BJOG. 2024;132(4):433–443. doi: 10.1111/1471-0528.17953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rushwan S, Tufa T, Gülmezoglu M, et al Feasibility, acceptability, and appropriate use of novel and lesser-used medicines for prevention and treatment of postpartum hemorrhage: evidence from implementation research. Int J MCH AIDS. 2024;13(Suppl 1):S4–S8. doi: 10.25259/IJMA_17_2023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Muthamia M, Nyaga F, Kiio M, et al Postpartum women’s knowledge on pregnancy-related danger signs, uterotonics and their perceptions on respectful maternity care in 10 selected counties, Kenya. Res Square. 2024. doi: 10.21203/rs.3.rs-3823253/v1 [DOI] [Google Scholar]
- 23.Gülmezoglu M, Chinery L, Rushwan S, et al A tale of two medicines: the need for ownership, end-to-end planning and execution for development and introduction of maternal health medicines. Int J MCH AIDS. 2024;13(Suppl 1):S9–S14. doi: 10.25259/IJMA_21_2024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cook JR, Saxena K, Taylor C, et al Cost-effectiveness and budget impact of heat-stable carbetocin compared to oxytocin and misoprostol for the prevention of postpartum hemorrhage (PPH) in women giving birth in India. BMC Health Serv Res. 2023;23(1):267. doi: 10.1186/s12913-023-09263-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Theunissen F, Cleps I, Goudar S, et al Cost of hospital care of women with postpartum haemorrhage in India, Kenya, Nigeria and Uganda: a financial case for improved prevention. Reprod Health. 2021;18(1):18. doi: 10.1186/s12978-020-01063-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Alwadhi V, Mishra A, Nagendra A, et al Introduction of heat-stable carbetocin through a public-private partnership model in India: a retrospective study to determine feasibility and its optimal use in public health settings. Int J MCH AIDS. 2024;13(Suppl 1):S81–S88. doi: 10.25259/IJMA_11_2023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hofmeyr GJ. Novel concepts and improvisation for treating postpartum haemorrhage: a narrative review of emerging techniques. Reprod Health. 2023;20(116). doi: 10.1186/s12978-023-01657-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Maged AM, El-Goly NA, Turki D, Bassiouny N. El-Demiry N. A systematic review and meta-analysis of randomized trials comparing carbetocin to oxytocin in preventing postpartum hemorrhage after cesarean delivery in low-risk women. J Obstet Gynaecol Res. 2024. doi: 10.1111/jog.16194 [DOI] [PubMed] [Google Scholar]
- 29.Gallos ID, Papadopoulou A, Man R, et al Uterotonic agents for preventing postpartum haemorrhage: a network meta-analysis. Cochrane Database Syst Rev. 2018;12(12):CD011689. doi: 10.1002/14651858 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ajima E, Nwaokorie C, Kangkum N, et al Establishing sustainable access to quality uterotonics in Kano, Lagos and Niger States—a supply chain perspective. Int J Health Plann Manage. 2025;40(3):701–715. doi: 10.1002/hpm.3910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Trivedi K, Sinha T, Boipai P, et al The role of oxytocin versus carbetocin in prevention of postpartum hemorrhage in cesarean section: a randomized control trial. Ann Afr Med. 2025;24(2):378–383. doi: 10.4103/aam.aam_218_24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zagrodnik E, Ziętek M, Machałowski T, Dołęgowska B, Szczuko M, Szczuko M. Carbetocin is more effective in stabilizing hemodynamic parameters compared to oxytocin during cesarean section. Biomedicines. 2025;13(3):685. doi: 10.3390/biomedicines13030685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Briones JR, Talungchit P, Thavorncharoensap M, Chaikledkaew U. Economic evaluation of carbetocin as prophylaxis for postpartum hemorrhage in the Philippines. BMC Health Serv Res. 2020;20(1):975. doi: 10.1186/s12913-020-05834-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gallos ID, Coomarasamy A. Carbetocin: worth the extra expense?. Best Pract Res Clin Obstet Gynaecol. 2019;61:55–65. doi: 10.1016/j.bpobgyn.2019.04.001 [DOI] [PubMed] [Google Scholar]
- 35.Federal Ministry of Health and Social Welfare (FMoH&SW). Postpartum facilitator training manual. 2024. Available from: https://www.health.gov.ng/Source/55/Family-Health. Accessed May 26, 2025.
- 36.Okonofua FE, Ekezue B, Ntoimo LFC, et al Effects of multifaceted interventions to prevent and manage primary postpartum haemorrhage in referral hospitals: a quasi-experimental study in Nigeria. BMJ Glob Health. 2022;7(4):e007779. doi: 10.1136/bmjgh-2021-007779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Evans CL, Bazant E, Atukunda I, et al Peer-assisted learning after onsite, low-dose, high-frequency training and practice on simulators to prevent and treat postpartum hemorrhage and neonatal asphyxia: a pragmatic trial in 12 districts in Uganda. PLoS One. 2018;13(12):e0207909. doi: 10.1371/journal.pone.0207909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Forbes G, Akter S, Miller S, et al Development and piloting of implementation strategies to support delivery of a clinical intervention for postpartum hemorrhage in four sub-Saharan Africa countries. Glob Health Sci Pract. 2024;12(5):e2300387. doi: 10.9745/GHSP-D-23-00387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Forbes G, Akter S, Miller S, et al Factors influencing postpartum haemorrhage detection and management and the implementation of a new postpartum haemorrhage care bundle (E-MOTIVE) in Kenya, Nigeria, and South Africa. Implement Sci. 2023;18(1):1. doi: 10.1186/s13012-022-01253-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Smiles for Mothers Consortium, Federal Ministry of Health (FMOH). Smiles for mothers national scale-up event: to disseminate integrated pph response best practices from Kano, Lagos, and Niger States, Nigeria (Event Report). 2023. Available from: https://scidar.org/wp-content/uploads/2023/07/National-Scale-up-Event-Report.pdf. Accessed May 26, 2025.