Abstract
Objective
Although diabetes mellitus (DM) is considered an important prognostic factor for hysterectomy outcomes, the relationship between DM and postoperative complications remains unclear. The aim of this study was to investigate whether DM is associated with an increased risk of complications following hysterectomy.
Methods
We systematically searched PubMed, Embase, and the Cochrane Library for relevant articles published on or before March 15, 2024. Pooled odds ratios (ORs) and 95% confidence intervals (CIs) were calculated via random effects meta-analysis. The primary outcome was the risk of complications posthysterectomy, including postoperative infection and hospital readmission. Additionally, we conducted subgroup and sensitivity analyses to explore the main sources of heterogeneity and assess the stability of the results.
Results
A total of 19 cohort studies comprising 375,531 participants met the inclusion criteria. This meta-analysis revealed that DM was significantly associated with postoperative infection (OR 2.01, 95% CI 1.46–2.77). Additionally, DM was significantly associated with low postoperative survival (5 years) (OR 4.43, 95% CI 2.98–6.58), readmission (OR 1.59, 95% CI 1.42–1.79), embolism (OR 1.31, 95% CI 1.18–1.46), renal failure (OR 3.88, 95% CI 2.74–5.51) and reintubation (OR 3.23, 95% CI 1.64–6.35), whereas DM was not associated with an extended length of stay (OR 1.39, 95% CI 0.93–2.10), myocardial infarction (OR 1.86, 95% CI 0.58–5.98) or blood transfusion (OR 1.36, 95% CI 0.88–2.11) in patients following hysterectomy.
Conclusions
DM increases the risk of postoperative infection following hysterectomy. Special attention should be given to diabetic patients to reduce the incidence of complications after hysterectomy.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12905-025-03835-8.
Keywords: Diabetes mellitus, Hysterectomy, Postoperative complication, infection, Meta-analysis
Introduction
Diabetes is one of the most common chronic diseases worldwide. Approximately 38.4 million Americans (11.6% of the U.S. population) have diabetes, and another 97.6 million adults aged 18 years or older are prediabetic (38.0% of the U.S. adult population) [1]. Chronic hyperglycemia can result in insulin secretion disorders or varying degrees of insulin resistance, leading to coronary heart disease, stroke, peripheral vascular disease, end-stage renal disease, retinopathy, neuropathy, and other related conditions [2]. Currently, approximately 10–15% of surgical patients have diabetes, and the relationship between diabetes and adverse postoperative outcomes has been extensively documented [3–6].
Hysterectomy is one of the most common surgical procedures performed on women [7, 8]. In the United States, more than 600,000 hysterectomies are performed annually, with an overall rate of 5.4 hysterectomies per 1,000 women [9]. As the number of outpatient hysterectomies increases each year, the overall number of hysterectomies is likely to continue to increase [10]. Studies have shown that, compared with nondiabetic patients, patients with diabetes have a significantly greater risk of adverse events, including postoperative infection, delayed wound healing, and cardiac complications such as myocardial infarction, renal insufficiency, reoperation, mortality, and an extended length of stay [11–14]. Bing et al. [15] reported a notable increase in posthysterectomy mortality among diabetic patients compared with their nondiabetic counterparts. However, Slavchev et al. [16] reported that there is no significant correlation between diabetes and posthysterectomy mortality. The discrepancies in the findings of these studies may arise from the relatively limited sample sizes or significant heterogeneity among them. The relationship between diabetes and posthysterectomy complications is still unclear. Hence, the primary objective of the present study was to conduct a meta-analysis to explore the potential influence of diabetes on patient outcomes after hysterectomy.
Methods
Standard protocol approvals, registrations, and patient consent
Our research study was registered on PROSPERO, an international systematic review registration website (Registration number: CRD42024550273), and reported in accordance with the Cochrane Handbook and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Given the nature of this study, informed consent and institutional review board approval were not needed.
Search strategy
Two independent researchers systematically searched the PubMed, Embase, and Cochrane Library electronic databases from inception to March 15, 2024, without restrictions on language or publication date. Medical subject heading (MeSH) terms were used to search PubMed and the Cochrane Library, whereas Embase subject heading (Emtree) terms were used for the Embase database. These terms were combined with free-text words, including synonyms and closely related terms, pertaining to hysterectomy, diabetes, and postoperative complications. A detailed search strategy and specific terms (“Hysterectomy” or “Genital Diseases, Female” or “Genital Diseases, Female/surgery” or “Minimally Invasive Surgical Procedures” or “Robotic Surgical Procedures” or “Laparoscopy”) and (“Diabetes Mellitus” or “Diabetes Mellitus, Type 1” or “Diabetes Mellitus, Type 2”) and (“Mortality” or “Length of Stay” or “Morbidity” or “Postoperative Complications” or “Surgical Wound Infection” or “Venous Thrombosis” or “Urinary Tract Infections”) were used (eTable 1). To ensure comprehensive coverage, we conducted manual searches of the reference lists of previous systematic reviews and meta-analyses to identify any potentially missed articles. In cases where multiple articles reported on the same cohort, we included only the most recent publication or the one with the largest sample size for analysis.
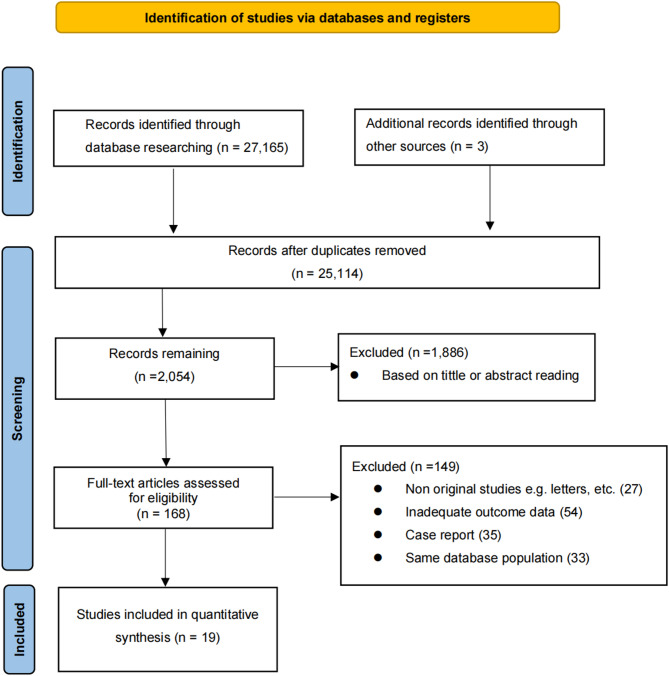
We utilized ENDNOTE X9 software to initially screen titles and abstracts, removing duplicates and citations that did not meet the inclusion criteria. The researchers subsequently independently assessed the full texts of the identified studies to determine if they met the inclusion criteria. In cases of discrepancy between the two reviewers, a third-party evaluator was consulted for evaluation. A flow diagram illustrating the filtering process is depicted in Fig. 1.
Fig.1.
Flowchart of the study selection
Selection criteria
The selection criteria for this study followed the population, intervention, comparison, outcome and study design (PICOS) approach.
Potential studies were considered eligible for inclusion if they met the following preestablished inclusion/exclusion criteria:
Study design: Prospective or retrospective observational studies.
Participants: Patients diagnosed with diabetes who underwent hysterectomy.
Outcomes and measures of associations: The primary outcome was postoperative infection, while secondary outcomes included other complications such as postoperative low survival rate, readmission, myocardial infarction, embolism, renal failure, re-catheterization, prolonged hospitalization, and blood transfusion. Effect indicators included odds ratio (OR), relative risk (RR), hazard ratio (HR), and 95% confidence interval (CI).
We excluded duplicate publications, conference papers, studies using the same population, case reports, reviews, and studies that did not provide sufficient data to calculate the odds ratio for postoperative complications in diabetic patients.
Data extraction
Data extraction was conducted by two reviewers via a predesigned independent study data extraction table. Discrepancies between the reviewers were resolved through discussion or consultation with a third party. The extracted data included the first author, publication year, geographic region, observation period, cohort sample size, definition of diabetes, measurement of diabetes, reported outcomes after hysterectomy, reported ORs, follow-up period, and adjusted variables.
Quality assessment
Each eligible study was independently evaluated via the Newcastle‒Ottawa Scale (NOS) [17], which comprises three quality parameters: the selection of cohorts, the comparability between groups, and the evaluation of outcomes. The total score for each study ranged from 0 to 9, with NOS scores of 0–7 and 8–9 indicating low and high quality, respectively (Table 1).
Table 1.
Methodological quality score of the included studies based on the Newcastle–Ottawa scale (NOS) tool
| Author | Year | Study Design | Selection | Comparability | Exposure/Outcome | Total Score | Risk of Bias |
|||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Representativeness of cohort * | Selection of control cohort * | Ascertainment of exposure * | Outcome not present at start * | Comparability of cohorts ** | Assessment of outcome * | Length of follow-up * | Adequacy of follow-up * | Total score 9* | ||||
| Franchi et al. | 2001 | Cross-sectional study | * | * | * | * | ** | * | * | * | 9 | Low |
| Molina-Cabrillana et al. | 2008 | Prospective study | * | * | * | * | ** | * | 7 | High | ||
| Lake et al. | 2013 | Cross-sectional study | * | * | * | * | ** | * | * | * | 9 | Low |
| Mahdi et al. | 2014 | Prospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Catanzarite et al. | 2015 | Retrospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Jennings et al. | 2015 | Prospective study | * | * | * | * | * | * | * | * | 8 | Low |
| Tuomi et al. | 2016 | Retrospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Jiamset et al. | 2016 | Retrospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Laughlin-Tommaso et al. | 2016 | Retrospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Penn et al. | 2017 | Retrospective study | * | * | * | * | * | * | * | * | 8 | Low |
| Agrawal et al. | 2018 | Prospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Corrigan et al. | 2019 | Prospective study | * | * | * | * | * | * | * | * | 8 | Low |
| Liang et al. | 2020 | Prospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Slavchev et al. | 2021 | Retrospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Ringel et al. | 2021 | Retrospective cohort study | * | * | * | * | * | * | * | * | 8 | Low |
| Mauney et al. | 2022 | Cross-sectional study | * | * | * | * | ** | * | * | * | 9 | Low |
| Bing et al. | 2022 | Retrospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Boyles et al. | 2022 | Retrospective study | * | * | * | * | ** | * | * | * | 9 | Low |
| Wang et al. | 2022 | Retrospective study | * | * | * | * | ** | * | 7 | High | ||
Evaluation of the strength of evidence
All analyses were conducted via STATA software (version 12.0; Stata, University Station, Texas, USA). A random effects meta-analysis was employed due to anticipated between-study heterogeneity. Fully adjusted effect estimates (ORs) of the associations between diabetes and surgical outcomes were used to derive combined risk estimates, illustrated graphically with forest plots. Interstudy heterogeneity was evaluated via the Cochrane Q test and I2 test, with heterogeneity considered statistically significant at I2 ≥ 50% or P < 0.05. To explore sources of heterogeneity, subgroup analyses were performed on the basis of study design (prospective or retrospective cohort), geographic region (United States or other countries), and infection type (surgical site infection (SSI), urinary tract infection (UTI), sepsis, or pneumonia). Publication bias was assessed through visual inspection of funnel plot symmetry, and Duvall and Tweedy’s pruning and filling methods were employed to adjust risk estimates to evaluate the impact of potential publication bias. A sensitivity analysis involving the sequential omission of a single study was conducted to assess the stability of the results. All the statistical tests were two-tailed, and P < 0.05 was considered to indicate statistical significance.
Results
Literature search
The initial search strategy yielded a total of 27,165 articles, and 3 relevant studies were included through manual searches. A total of 2,690 duplicate records were excluded. After screening, 2,054 articles were initially identified, of which 1,889 were excluded after title and abstract review. Upon full-text review, 149 articles were excluded because they lacked available or sufficient data for analysis, resulting in the inclusion of 19 studies. Nineteen studies [15, 16, 18–34] involving 375,531 participants (average sample size 19,765) met the inclusion criteria for the present meta-analysis (Fig. 1).
Study characteristics
The baseline characteristics of the patients included in this study are summarized in Table 2. All the included studies were published between 2003 and 2022, with seventeen studies [15, 16, 20–34] being published in 2013 or later. The meta-analysis included studies conducted across three continents, comprising 11 studies from the United States [20–23, 25, 26, 28–30, 33, 34], 4 studies from Europe [16, 18, 19, 24], and 4 studies from Asia [15, 27, 31, 32]. Nineteen studies adjusted for critical experimental variables [15, 16, 18, 20–22, 24, 27, 29, 30, 32–34], such as age, timing of surgery, and body mass index (BMI), through Cox regression or multivariate analysis.
Table 2.
Characteristics of studies included in meta-analysis
| First author | Year | Study Design | Region | Observation Period | Sample size | Measurements of diabetes |
|---|---|---|---|---|---|---|
| Franchi et al. | 2001 | Cross-sectional study | Italy | 1989–1999 | 455 | Diagnosed and treated by their primary care physician |
| Molina-Cabrillana et al. | 2008 | Prospective study | Spain | 2000–2004 | 1,540 | NR |
| Lake et al. | 2013 | Cross-sectional study | USA | 2005–2009 | 13,822 | ICD-9-CM diagnoses codes |
| Mahdi et al. | 2014 | Retrospective study | USA | 2005–2011 | 28,366 | ICD-9-CM diagnoses codes |
| Catanzarite et al. | 2015 | Retrospective study | USA | 2011–2012 | 21,228 | ICD-9-CM diagnoses codes |
| Jennings et al. | 2015 | Prospective study | USA | 2011–2012 | 36,941 | ICD-9-CM diagnoses codes |
| Tuomi et al. | 2016 | Retrospective study | Finland | 2007–2013 | 912 | NR |
| Jiamset et al. | 2016 | Retrospective study | Thailand | 2001–2014 | 444 | Diagnosed and treated by their primary care physician |
| Laughlin-Tommaso et al. | 2016 | Retrospective study | USA | 1965–2002 | 7632 | Electronic codes of the Mayo Clinic Study of Uterine Disease and Health |
| Penn et al. | 2017 | Retrospective study | USA | 2013–2015 | 2,068 | ICD-9-CM diagnoses codes |
| Agrawal et al. | 2018 | Prospective study | USA | 2006–2015 | 157,589 | ICD-9-CM diagnoses codes |
| Corrigan et al. | 2019 | Prospective study | USA | 2007–2016 | 56,640 | ICD-9-CM diagnoses codes |
| Liang et al. | 2020 | Prospective study | China | 2004–2011 | 431 | HbA1c > 7.0% |
| Slavchev et al. | 2021 | Retrospective study | Bulgaria | 2004–2012 | 104 | Diagnosed and treated by their primary care physician |
| Ringel et al. | 2021 | Retrospective cohort study | USA | 2012–2017 | 41,286 | HbA1c ≥ 6.5% |
| Mauney et al. | 2022 | Cross-sectional study | USA | 2015–2017 | 5,098 | ICD-9-CM diagnoses codes |
| Bing et al. | 2022 | Retrospective study | China | 2011–2021 | 48 | Diagnosed and treated by their primary care physician |
| Boyles et al. | 2022 | Retrospective study | USA | 2015–2019 | 739 | NR |
| Wang et al. | 2022 | Retrospective study | China | 2013–2021 | 188 | Diagnosed and treated by their primary care physician |
| First author | Diabetes definition | Type of hysterectomy | Outcomes | Follow-up period | Adjusted variables |
|---|---|---|---|---|---|
| Franchi et al. | Diagnosed and treated by their primary care physician | Abdominal hysterectomy | Incisional hernia | 5 Years | Severe wound infection; Body mass index > 27 kg/m2; Age > 62 y; Fascial closure with interrupted suture |
| Molina-Cabrillana et al. | NR | Abdominal hysterectomy; Vaginal hysterectomy | SSI | NR | NR |
| Lake et al. | Identified from ICD-9 discharge diagnoses | Laparotomy; Total vaginal hysterectomy; Laparoscopy | Cellulitis(SSI) | 1 Month | Route of hysterectomy; Operative time > 75th percentile duration; American Society of Anesthesiologists class 3; Body mass index |
| Mahdi et al. | Identified from ICD-9 discharge diagnoses | Abdominal hysterectomy; Laparoscopic hysterectomy; | SSI | 1 Month | Smoking; Respiratory risk factors; Overweight; American Society of Anesthesiologists class > 3; Blood transfusion; Operative time > 180 min; Creatinine concentration > 2 mg/dL; Serum albumin concentration < 3 mg/dL |
| Catanzarite et al. | Identified from ICD-9 discharge diagnoses | Laparoscopic-assisted vaginal hysterectomy; Laparoscopic hysterectomy; Vaginal hysterectomy | Readmission | 1 Month | Age; Obesity; Smoking; Steroid use; Dyspnea; Hypertension; COPD; Prior surgery > 30 days; Bleeding disorders; ASA class 3 or 4; Type of hysterectomy; Inpatient status; LOS |
| Jennings et al. | Identified from ICD-9 discharge diagnoses | Total laparoscopic; Laparoscopic-assisted vaginal; Supracervical hysterectomies | Readmission | 1 Month | NR |
| Tuomi et al. | NR | Minimally invasive hysterectomy; Traditional laparoscopy hysterectomy | SSI | 1 Month | Body mass index; Current smoking; Leukocytosis; Minimally invasive surgery; Operative time; Estimated blood loss |
| Jiamset et al. | Diagnosed and treated by their primary care physician | Radical hysterectomy | Hemorrhage > 1,500 mL; Blood transfusion; Vascular injury; Postoperative morbidity Febrile morbidity; Urinary tract infection; Vaginal stump infection; Wound infection;5-Year overall survival | 5 Years | Age; Node metastasis |
| Laughlin-Tommaso et al. | Identified from the Mayo Clinic Study of Uterine Disease and Health | NR | Cardiovascular diseases | 3 Years | Hyperlipidemia; Hypertension; Obesity; Metabolic syndrome; Polycystic ovary syndrome |
| Penn et al. | Identified from ICD-9 discharge diagnoses | NR | Readmission | 1 Month | NR |
| Agrawal et al. | Identified from ICD-9 discharge diagnoses | Minimally invasive hysterectomy; Abdominal hysterectomy; Vaginal hysterectomy | Extended LOS | 1 Month | Year of operation; Age; Race; Body mass index; Functional status; Chronic obstructive pulmonary disease; Bleeding disorder; Intraoperative conditions; Postoperative conditions |
| Corrigan et al. | Identified from ICD-9 discharge diagnoses | Total laparoscopic hysterectomy; Laparoscopic-assisted vaginal hysterectomy; Laparoscopic supracervical hysterectomy | Superficial surgical site wound infection; Reintubation; Failure to wean; Myocardial infarction; Extended LOS; Pneumonia; Renal insufficiency; Renal failure; Urinary tract infection; Myocardial infarction; Transfusion; Sepsis | 1 Month | NR |
| Liang et al. | According to American Diabetes Association diagnostic criteria or a patient-reported history of diabetes | NR | 5-year survival | 5 Years |
Age; BMI; Serum creatinine; Tumor stage Histology; Tumor differentiation; Tumor size; Deep stromal invasion; LVSI; Hypertension; Cardiovascular disease |
| Slavchev et al. | Diagnosed and treated by their primary care physician | Radical hysterectomy; Total hysterectomy | 5-year survival | 5 Years | Age; Ascites; Type of surgery; Residual tumor size; LN metastasis; Performance status |
| Ringel et al. | Managed for > 2 weeks before surgery | NR | Readmission; ED visit; Reoperation; Acute renal injury; Pneumonia; Superficial surgical site infection; Deep surgical site infection; Organ space surgical site infection; UTI; Cardiac dysfunction; DVT; PE; Severe sepsis; Unplanned intubation; MI; Stroke; Cardiac arrest; Death | 1 Month | NR |
| Mauney et al. | Identified from ICD-9 discharge diagnoses | Peripartum hysterectomy | VTE | 6 Weeks | Hysterectomy; Advanced maternal age; Obesity; Pregnancy-related hypertension; Primary hypercoagulable state; Tobacco use; Multifetal gestation; Peripartum infection |
| Bing et al. | Diagnosed and treated by their primary care physician | NR | Mortality | 5 Years | Age; NLR > 3.0995; MLR > 0.2386; PLR > 154.3309; Tumor stage |
| Boyles et al. | NR | Abdominal hysterectomy | SSI | 1 Month | BMI > 40; Bowel resection; ASA class 3 or 4; Closing tray protocol |
| Wang, D. | Diagnosed and treated by their primary care physician | Abdominal hysterectomy | SSI | NR | NR |
Abbreviations: ASA = American Society of Anesthesiologists; BMI = Body mass index; COPD = Chronic obstructive pulmonary disease; DM = Diabetes mellitus; DVT = Deep venous thrombosis; ED = Emergency department; HbA1c = Glycosylated hemoglobin; IDDM = Insulin-dependent diabetes mellitus; LOS = Length of stay; LVSI = Lymphatic vascular space involvement; LN = Lymph nodes; MI = Myocardial infarction; MLR = Monocyte/lymphocyte ratio; NIDDM = Non-insulin-dependent diabetes mellitus; NLR = Neutrophil/lymphocyte ratio; PE = Pulmonary embolism; PLR = Platelet/lymphocyte ratio; SSI = Surgical site infections; UTI = Urinary tract infection; VTE = Venous thromboembolism
Primary outcome
Postoperative infection
Nine studies [19–21, 24, 26, 28, 29, 31, 32] investigated the association between diabetes mellitus (DM) and the risk of postoperative infection. Overall, the diabetic group presented a significantly greater risk of postoperative infection than did the nondiabetic group (OR 2.01, 95% CI 1.46–2.77), with significant heterogeneity observed (I2 = 63.7%) (eFigure 1). Sensitivity analysis was employed to assess the stability of the findings, revealing that the study by Corrigan et al. [26] exhibited high heterogeneity (eFigure 20). Subgroup analysis on the basis of infection type revealed that diabetes was linked to increased risks of SSI (OR 2.01, 95% CI 1.46–2.77), pneumonia (OR 2.87, 95% CI 1.69–4.89) and UTI (OR 1.47, 95% CI 1.23–1.74). Among the other subgroup analyses, those based on an increased risk of sepsis, those conducted in North America, and the different study types also revealed significant associations. In addition, subgroup analyses of studies conducted in Asian and European countries did not reveal a significant association between diabetes and postoperative infection (eFigure 2). We found that the heterogeneity was significantly reduced in the study design, infection type, and study area subgroup analyses, suggesting that the heterogeneity could be attributed to these variables (Table 3).
Table 3.
Analyses for the association between diabetes and hysterectomy complications
| Outcomes | OR | 95% CI | I², % | P value | Egger’s test P value |
|---|---|---|---|---|---|
| Postoperative infection | 2.01 | 1.46–2.77 | 63.7 | 0.005 | 0.348 |
| Low postoperative survival (5 years) | 4.43 | 2.98–6.58 | 0.0 | 0.422 | 0.462 |
| Readmission | 1.59 | 1.42–1.79 | 0.0 | 0.776 | - |
| Embolism | 1.31 | 1.18–1.46 | 0.0 | 0.412 | - |
| Renal failure | 3.88 | 2.74–5.51 | 0.0 | 0.939 | - |
| Reintubation | 3.23 | 1.64–6.35 | 46.9 | 0.170 | - |
| Extended LOS | 1.39 | 0.93–2.10 | 89.4 | 0.002 | - |
| MI | 1.86 | 0.58–5.98 | 79.4 | 0.008 | - |
| Blood transfusion | 1.36 | 0.88–2.11 | 0.0 | 0.821 | - |
Abbreviations: LOS = Length of stay; MI = Myocardial infarction
Secondary outcomes
Five studies [15, 16, 27, 28, 32] investigated the association between diabetes mellitus and low postoperative survival (5 years) and revealed a significant correlation. Diabetes was found to be significantly associated with low postoperative survival (5 years) (OR 3.56, 95% CI 1.94–6.53), albeit with substantial heterogeneity (I2 = 63.7%) (eFigure 3). The stability of the results was assessed through sensitivity analysis, revealing that the study by Slavchev et al. [16] exhibited high heterogeneity (eFigure 21). Upon exclusion of this study from the pooled analysis, the low postoperative survival rate (5 years) remained significantly greater in the diabetic group than in the nondiabetic group (OR 4.43, 95% CI 2.98–6.58) (eFigure 4). Three studies [22, 23, 28] assessed the association between diabetes and readmission risk (OR 1.59, 95% CI 1.42–1.79), with no heterogeneity in the results (I2 = 0.0%) (eFigure 5). We also found a significant association between diabetes and venous thromboembolism (VTE) (OR 1.31, 95% CI 1.18–1.46) (eFigure 6), renal failure (OR 3.88, 95% CI 2.74–5.51) (eFigure 7), and reintubation (OR 3.23, 95% CI 1.64–6.35) (eFigure 8). However, we did not find a significant association between diabetes and extended length of stay (OR 1.39, 95% CI 0.93–2.10) (eFigure 9), myocardial infarction (OR 1.86, 95% CI 0.58–5.98) (eFigure 10), or blood transfusion (OR 1.36, 95% CI 0.88–2.11) (eFigure 11). Publication bias was not assessed for these outcomes because of the limited number of studies available for a meaningful assessment of publication bias (< 10 studies for each outcome) (Table 4).
Table 4.
Subgroup analyses for the association between diabetes and infection after hysterectomy
| Variables | OR | 95% CI | I2 (%) | No. studies | P for subgroup differences | |
|---|---|---|---|---|---|---|
| Study design | ||||||
| Retrospective | 2.23 | 1.21–4.12 | 67.7 | 5 | p < 0.001 | |
| Prospective | 1.78 | 1.28–2.47 | 0.0 | 3 | p = 0.010 | |
| Infection type | ||||||
| SSI | 2.01 | 1.46–2.77 | 63.7 | 9 | p < 0.001 | |
| Pneumonia | 2.87 | 1.69–4.89 | 20.2 | 2 | p < 0.001 | |
| UTI | 1.47 | 1.23–1.74 | 0.0 | 3 | p < 0.001 | |
| Sepsis | 2.79 | 1.36–5.74 | 61.1 | 2 | p = 0.005 | |
| Regions | ||||||
| America | 2.66 | 2.04–3.47 | 0.0 | 4 | p < 0.001 | |
| Others | 1.37 | 0.76–2.49 | 42.3 | 4 | p = 0.295 |
Bias and sensitivity analysis
The assessment of publication bias for postoperative complications across 19 studies followed the previously described statistical analysis procedures. Bias tests were also conducted for infection, low postoperative survival, readmission risk, renal failure, reintubation, extended length of stay, myocardial infarction, and blood transfusion (eFigures 11–19). The results revealed no significant publication bias. Furthermore, a sensitivity analysis was performed to evaluate the robustness of the results for each study factor combination (eFigures 20–27).
Discussion
Principal findings
This meta-analysis, which pooled data from 19 cohort studies, revealed that diabetes was correlated with adverse postoperative outcomes following hysterectomy. These included an elevated risk of postoperative infection, readmission, poor postoperative survival (5 years), embolism, renal failure, and reintubation. Across most studied subgroups, these risks remained heightened, and sensitivity analyses corroborated the main results. Nevertheless, no significant associations were detected between diabetes and an extended length of stay, myocardial infarction, or blood transfusion.
Potential mechanisms
The pathogenesis underlying the increased risk of complications in diabetic patients post hysterectomy remains unknown. Studies indicate that individuals with diabetes exhibit markedly elevated rates of infection, including respiratory, urinary tract, gastrointestinal, and cutaneous mucosal infections, compared with their nondiabetic counterparts [35–37]. Impaired white blood cell function, microvascular changes, hyperglycemia, and other factors contribute to compromised immune function and cellular immune dysfunction, thereby increasing the risk of postoperative infection [36]. Studies have demonstrated that diabetes can hinder the adhesion, chemotaxis, and phagocytosis of polymorphonuclear leukocytes [35]. Additionally, the antioxidant system, in which polymorphonuclear white blood cells participate in bactericidal activity, may also be impaired [38]. A hyperglycemic environment can inhibit glucose-6-phosphate dehydrogenase (G6PD), increase the apoptosis of polymorphonuclear leukocytes, and diminish their transport through the endothelium, thus impeding their antibacterial function [39]. Furthermore, other studies have indicated that monocytes in diabetic patients secrete lower levels of interleukin-1 (IL-1) and IL-6 following lipopolysaccharide stimulation [40]. An increase in glycosylation can inhibit the production of IL-10 by myeloid cells and interferon and tumor necrosis factor by T cells. It also reduces the expression of the class I major histocompatibility complex (MHC) on myeloid cells [41]. These cytokines play crucial roles in pathogen resistance and adaptive immune responses by stimulating antibody production and effector T-cell development [42]. Studies by Greenhalgh et al. [43] have indicated that diabetes, due to impaired innate and adaptive immune responses to invasive pathogens, increases patients’ susceptibility to infections such as respiratory tract infections, urinary tract infections, and skin and mucosal infections. It has been reported that patients with diabetes, particularly those with insulin-dependent diabetes, have a significantly higher risk of postoperative infections, as hyperglycemia can impair the immune system and create a favorable environment for the proliferation of certain bacteria [44]. Consequently, in individuals with diabetes, the production of these cytokines may be inhibited, thereby attenuating the immune response to invading pathogens and increasing susceptibility to infection. These factors likely underlie the significantly increased risk of postoperative infection observed in diabetic patients.
Comparisons with previous literature
Currently, most hysterectomies are performed using minimally invasive techniques, including laparoscopic-assisted vaginal hysterectomy and robot-assisted hysterectomy for endometrial cancer [45]. Corrado et al. [46] found that minimally invasive surgery can reduce operation time and the incidence of postoperative complications in patients with early high-risk endometrial cancer, allowing for quicker recovery and hospital discharge. Additionally, minimally invasive surgery does not negatively impact patient survival or recurrence rates. Liu et al.‘s study [47] indicates that diabetes is also a risk factor preventing same-day discharge after minimally invasive hysterectomy for both malignant and benign gynecological conditions. Regarding the relationship between hysterectomy and diabetes, the study by Chian et al. [48] demonstrates that the incidence of diabetes is 40% higher in women who have undergone hysterectomy compared to the control group. On the issue of the impact of uterine manipulators on the oncological outcomes of minimally invasive treatment of endometrial cancer, the study by Scutiero et al. [45] found no association between the use of uterine manipulators and postoperative infection or recurrence of endometrial cancer. Therefore, extensive prospective cohort studies are needed in the future to validate the relationships among diabetes, surgical instruments, and hysterectomy.
Implications for clinical practice and future studies
This study evaluated the risk of various complications following hysterectomy in diabetic patients and provides insights for future clinical practice. Early preoperative intervention and effective diabetes management during the perioperative period are crucial for mitigating postoperative complications. Despite the undeniable clinical importance of perioperative management in diabetic patients, a standardized preoperative treatment approach for diabetic patients has yet to be established. Moreover, some obstetricians and gynecologists may not fully appreciate the association between diabetes and complications post hysterectomy. In the long-term, these findings will aid clinicians in better assessing the postoperative complications of hysterectomy in diabetic patients, offering a foundation for large prospective cohort studies and providing novel evidence to guide clinical management. Intensive preoperative glycemic control is beneficial for reducing the risk of postoperative complications in diabetic patients.
Strengths and limitations
This study has several strengths. First, to the best of our knowledge, this is the largest and most comprehensive meta-analysis of high-quality cohort studies related to this subject. It provides the latest evidence on the potential relationship between diabetes and complications following hysterectomy. Second, we conducted a thorough literature search using MeSH/Emtree terms and free-text words across three main databases - PubMed, Cochrane Library, and Embase - and developed a comprehensive search strategy without date or language restrictions. This approach ensures that all relevant original studies meeting the inclusion criteria are identified, thereby minimizing publication bias and enhancing the reproducibility of our results. Third, we adhered to the PRISMA guidelines and used the NOS scale to ensure complete, informative, and transparent reporting of the included studies. Fourth, we employed various methods, including sensitivity analyses and subgroup analyses, to thoroughly test the stability of our results.
There are still some potential limitations to our study. First, due to the lack of individual patient data, we were unable to determine whether the severity of diabetes, patients taking or discontinuing diabetes medications, HbA1c levels, the most recent HbA1c value, original diagnosis, and different surgical modalities of hysterectomy might have significantly impacted the outcomes. Future studies may focus on the impact of these factors on patient outcomes. Second, our conclusions are largely based on data from observational cohort studies, so we cannot infer a causal relationship between diabetes and post-hysterectomy infection, readmissions, and embolism. Third, follow-up times are not fully comparable, representing a potential confounding factor in assessing oncological outcomes. Fourth, the limited number of included studies and the fact that most studies are retrospective may lead to unmeasured confounding factors and bias. Future studies with higher levels of evidence, such as large prospective cohort studies, are needed to confirm these findings.
Conclusion
The present study indicated that diabetes may serve as a risk factor for postoperative infection following hysterectomy. However, the potential of diabetes as a predictor for other postoperative complications warrants further investigation through large prospective cohort studies.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
Not applicable.
Author contributions
Yuhuan Zeng: Study concept and design, Acquisition of data, Analysis and interpretation of data, Drafting of the manuscript, Critical revision of the manuscript for important intellectual content Yuanhu Lei: Study concept and design, Acquisition of data, Analysis and interpretation of data, Drafting of the manuscript, Critical revision of the manuscript for important intellectual content Min Li: Analysis and interpretation of data, Critical revision of the manuscript for important intellectual contentWei Liu: Critical revision of the manuscript for important intellectual contentJie Zhou: Critical revision of the manuscript for important intellectual content Jie Zhong: Critical revision of the manuscript for important intellectual contentGuangming Song: Critical revision of the manuscript for important intellectual content Yukun Li: Critical revision of the manuscript for important intellectual content Xiangfang Tan: Study concept and design, Critical revision of the manuscript for important intellectual content, Study supervision Qunfeng Zhang: Study concept and design, Critical revision of the manuscript for important intellectual content, Study supervisionSigned by all authors as follows: Yuhuan Zeng, Yuanhu Lei, Min Li, Wei Liu, Jie Zhou, Jie Zhong, Guangming Song, Yukun Li, Xiangfang Tan, Qunfeng Zhang.
Funding
The authors declare that no funds, grants or other support were received during the preparation of this manuscript.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Informed consent
Not applicable.
Consent to participate
Not applicable.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yuhuan Zeng, Yuanhu Lei and Min Li contributed equally to this work.
Contributor Information
Xiangfang Tan, Email: tanxiangfang02@163.com.
Qunfeng Zhang, Email: xiaofeng29@163.com.
References
- 1.National Diabetes Statistics Report. Estimates of Diabetes and Its Burden in the United States. In. Edited by Centers for Disease C, Prevention. Atlanta, GA; 2023.
- 2.Harding JL, Pavkov ME, Magliano DJ, Shaw JE, Gregg EW. Global trends in diabetes complications: a review of current evidence. Diabetologia. 2019;62(1):3–16. [DOI] [PubMed] [Google Scholar]
- 3.Dhatariya K, Levy N, Hall GM. The impact of glycaemic variability on the surgical patient. Curr Opin Anaesthesiol. 2016;29(3):430–7. [DOI] [PubMed] [Google Scholar]
- 4.Sebranek JJ, Lugli AK, Coursin DB. Glycaemic control in the perioperative period. Br J Anaesth. 2013;111(Suppl 1):i18–34. [DOI] [PubMed] [Google Scholar]
- 5.Kwon S, Thompson R, Dellinger P, Yanez D, Farrohki E, Flum D. Importance of perioperative glycemic control in general surgery: a report from the surgical care and outcomes assessment program. Ann Surg. 2013;257(1):8–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.DiNardo M, Donihi AC, Forte P, Gieraltowski L, Korytkowski M. Standardized glycemic management and perioperative glycemic outcomes in patients with diabetes mellitus who undergo same-day surgery. Endocr Pract. 2011;17(3):404–11. [DOI] [PubMed] [Google Scholar]
- 7.Burgess A, Fish M, Goldberg S, Summers K, Cornwell K, Lowe J. Surgical-Site infection prevention after hysterectomy: use of a consensus bundle to guide improvement. J Healthc Qual. 2020;42(4):188–94. [DOI] [PubMed] [Google Scholar]
- 8.Moore BJ, Steiner CA, Davis PH, Stocks C, Barrett ML. Trends in hysterectomies and oophorectomies in hospital inpatient and ambulatory settings, 2005–2013. Healthcare cost and utilization project (HCUP) statistical briefs. edn. Rockville (MD): Agency for Healthcare Research and Quality (US); 2006. [PubMed] [Google Scholar]
- 9.Harvey SV, Pfeiffer RM, Landy R, Wentzensen N, Clarke MA. Trends and predictors of hysterectomy prevalence among women in the united States. Am J Obstet Gynecol. 2022;227(4):e611611–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Morgan DM, Kamdar NS, Swenson CW, Kobernik EK, Sammarco AG, Nallamothu B. Nationwide trends in the utilization of and payments for hysterectomy in the United States among commercially insured women. Am J Obstet Gynecol 2018, 218(4):425.e421-425.e418. [DOI] [PMC free article] [PubMed]
- 11.Chuah LL, Papamargaritis D, Pillai D, Krishnamoorthy A, le Roux CW. Morbidity and mortality of diabetes with surgery. Nutr Hosp. 2013;28(Suppl 2):47–52. [DOI] [PubMed] [Google Scholar]
- 12.Rollins KE, Varadhan KK, Dhatariya K, Lobo DN. Systematic review of the impact of HbA1c on outcomes following surgery in patients with diabetes mellitus. Clin Nutr. 2016;35(2):308–16. [DOI] [PubMed] [Google Scholar]
- 13.Zhang X, Hou A, Cao J, Liu Y, Lou J, Li H, Ma Y, Song Y, Mi W, Liu J. Association of diabetes mellitus with postoperative complications and mortality after Non-Cardiac surgery: A Meta-Analysis and systematic review. Front Endocrinol (Lausanne). 2022;13:841256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tan DJH, Yaow CYL, Mok HT, Ng CH, Tai CH, Tham HY, Foo FJ, Chong CS. The influence of diabetes on postoperative complications following colorectal surgery. Tech Coloproctol. 2021;25(3):267–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bing RS, Tsui WL, Ding DC. The association between diabetes mellitus, high monocyte/lymphocyte ratio, and survival in endometrial cancer: A retrospective cohort study. Diagnostics (Basel) 2022, 13(1). [DOI] [PMC free article] [PubMed]
- 16.Slavchev S, Kornovski Y, Yordanov A, Ivanova Y, Kostov S, Slavcheva S. Survival in advanced epithelial ovarian cancer associated with cardiovascular comorbidities and type 2 diabetes mellitus. Curr Oncol. 2021;28(5):3668–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25(9):603–5. [DOI] [PubMed] [Google Scholar]
- 18.Franchi M, Ghezzi F, Buttarelli M, Tateo S, Balestreri D, Bolis P. Incisional hernia in gynecologic oncology patients: a 10-year study. Obstet Gynecol. 2001;97(5 Pt 1):696–700. [DOI] [PubMed] [Google Scholar]
- 19.Molina-Cabrillana J, Valle-Morales L, Hernandez-Vera J, López-Carrió I, García-Hernández JA, Bolaños-Rivero M. Surveillance and risk factors on hysterectomy wound infection rate in Gran canaria, Spain. Eur J Obstet Gynecol Reprod Biol. 2008;136(2):232–8. [DOI] [PubMed] [Google Scholar]
- 20.Lake AG, McPencow AM, Dick-Biascoechea MA, Martin DK, Erekson EA. Surgical site infection after hysterectomy. Am J Obstet Gynecol. 2013;209(5):e490491–499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mahdi H, Goodrich S, Lockhart D, DeBernardo R, Moslemi-Kebria M. Predictors of surgical site infection in women undergoing hysterectomy for benign gynecologic disease: a multicenter analysis using the National surgical quality improvement program data. J Minim Invasive Gynecol. 2014;21(5):901–9. [DOI] [PubMed] [Google Scholar]
- 22.Catanzarite T, Vieira B, Qin C, Milad MP. Risk factors for unscheduled 30-day readmission after benign hysterectomy. South Med J. 2015;108(9):524–30. [DOI] [PubMed] [Google Scholar]
- 23.Jennings AJ, Spencer RJ, Medlin E, Rice LW, Uppal S. Predictors of 30-day readmission and impact of same-day discharge in laparoscopic hysterectomy. Am J Obstet Gynecol. 2015;213(3):e344341–347. [DOI] [PubMed] [Google Scholar]
- 24.Tuomi T, Pasanen A, Leminen A, Bützow R, Loukovaara M. Incidence of and risk factors for surgical site infections in women undergoing hysterectomy for endometrial carcinoma. Acta Obstet Gynecol Scand. 2016;95(4):480–5. [DOI] [PubMed] [Google Scholar]
- 25.Penn CA, Kamdar NS, Morgan DM, Uppal S. Preoperatively predicting non-home discharge after surgery for gynecologic malignancy. Gynecol Oncol. 2017;145:15–6. [DOI] [PubMed] [Google Scholar]
- 26.Corrigan KE, Vargas MV, Robinson HN, Gu A, Wei C, Tyan P, Singh N, Tappy EE, Moawad GN. Impact of diabetes mellitus on postoperative complications following laparoscopic hysterectomy for benign indications. Gynecol Obstet Invest. 2019;84(6):583–90. [DOI] [PubMed] [Google Scholar]
- 27.Liang SH, Shen YC, Wu JY, Wang LJ, Wu MF, Li J. Impact of poor preoperative glycemic control on outcomes among patients with cervical Cancer undergoing a radical hysterectomy. Oncol Res Treat. 2020;43(1–2):10–8. [DOI] [PubMed] [Google Scholar]
- 28.Ringel NE, Morgan DM, Kamdar N, Gutman RE. Hysterectomy complications relative to HbA(1c) levels: identifying a threshold for surgical planning. J Minim Invasive Gynecol. 2021;28(10):1735–e17421731. [DOI] [PubMed] [Google Scholar]
- 29.Boyles G, DeMari J, Barrington D, Baek J, Busho A, Akinduro J, Cohn D, Nagel C. Less is more: abdominal closure protocol does not reduce surgical site infection after hysterectomy. Gynecol Oncol. 2022;165:S6. [DOI] [PubMed] [Google Scholar]
- 30.Mauney L, Jr. Barth WH, Clapp MA. Association between peripartum hysterectomy and venous thromboembolism. Am J Obstet Gynecol. 2022;226(1):e119111–119111. [DOI] [PubMed]
- 31.Wang D, Chen Y, Deng J, Xiao G, Li Y, Lin L, You Y. A retrospective study from 2 tertiary hospitals in China to evaluate the risk factors for surgical site infections after abdominal hysterectomy in 188 patients. Med Sci Monit. 2022;28:e936198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jiamset I, Hanprasertpong J. Impact of diabetes mellitus on oncological outcomes after radical hysterectomy for early stage cervical cancer. J Gynecol Oncol. 2016;27(3):e28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Laughlin-Tommaso SK, Khan Z, Weaver AL, Schleck CD, Rocca WA, Stewart EA. Cardiovascular risk factors and diseases in women undergoing hysterectomy with ovarian conservation. Menopause. 2016;23(2):121–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Agrawal S, Chen L, Tergas AI, Hou JY, St Clair CM, Ananth CV, Neugut AI, Hershman DL, Wright JD. Characteristics associated with prolonged length of stay after hysterectomy for benign gynecologic conditions. Am J Obstet Gynecol 2018, 219(1):89.e81-89.e15. [DOI] [PubMed]
- 35.Casqueiro J, Casqueiro J, Alves C. Infections in patients with diabetes mellitus: A review of pathogenesis. Indian J Endocrinol Metab. 2012;16(Suppl 1Suppl1):S27–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Geerlings SE, Hoepelman AI. Immune dysfunction in patients with diabetes mellitus (DM). FEMS Immunol Med Microbiol. 1999;26(3–4):259–65. [DOI] [PubMed] [Google Scholar]
- 37.Muller LM, Gorter KJ, Hak E, Goudzwaard WL, Schellevis FG, Hoepelman AI, Rutten GE. Increased risk of common infections in patients with type 1 and type 2 diabetes mellitus. Clin Infect Dis. 2005;41(3):281–8. [DOI] [PubMed] [Google Scholar]
- 38.Muchová J, Liptáková A, Országhová Z, Garaiová I, Tison P, Cársky J, Duracková Z. Antioxidant systems in polymorphonuclear leucocytes of type 2 diabetes mellitus. Diabet Med. 1999;16(1):74–8. [DOI] [PubMed] [Google Scholar]
- 39.Peleg AY, Weerarathna T, McCarthy JS, Davis TM. Common infections in diabetes: pathogenesis, management and relationship to glycaemic control. Diabetes Metab Res Rev. 2007;23(1):3–13. [DOI] [PubMed] [Google Scholar]
- 40.Berbudi A, Rahmadika N, Tjahjadi AI, Ruslami R. Type 2 diabetes and its impact on the immune system. Curr Diabetes Rev. 2020;16(5):442–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Price CL, Hassi HO, English NR, Blakemore AI, Stagg AJ, Knight SC. Methylglyoxal modulates immune responses: relevance to diabetes. J Cell Mol Med. 2010;14(6b):1806–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. 2014;6(10):a016295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Greenhalgh DG. Wound healing and diabetes mellitus. Clin Plast Surg. 2003;30(1):37–45. [DOI] [PubMed] [Google Scholar]
- 44.Marchant MH Jr., Viens NA, Cook C, Vail TP, Bolognesi MP. The impact of glycemic control and diabetes mellitus on perioperative outcomes after total joint arthroplasty. J Bone Joint Surg Am. 2009;91(7):1621–9. [DOI] [PubMed] [Google Scholar]
- 45.Scutiero G, Vizzielli G, Taliento C, Bernardi G, Martinello R, Cianci S, Riemma G, Scambia G, Greco P. Influence of uterine manipulator on oncological outcome in minimally invasive surgery of endometrial cancer: A systematic review and meta-analysis. Eur J Surg Oncol. 2022;48(10):2112–8. [DOI] [PubMed] [Google Scholar]
- 46.Corrado G, Ciccarone F, Cosentino F, Legge F, Rosati A, Arcieri M, Turco LC, Certelli C, Federico A, Vizza E, et al. Role of minimally invasive surgery versus open approach in patients with early-stage uterine carcinosarcomas: a retrospective multicentric study. J Cancer Res Clin Oncol. 2021;147(3):845–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liu J, Chen Y, Tan X, Chen H. Factors influencing same-day discharge after minimally invasive hysterectomy for malignant and non-malignant gynecological diseases: a systematic review and meta-analysis. Front Oncol. 2023;13:1307694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chiang CH, Chen W, Tsai IJ, Hsu CY, Wang JH, Lin SZ, Ding DC. Diabetes mellitus risk after hysterectomy: A population-based retrospective cohort study. Med (Baltim). 2021;100(4):e24468. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.