Abstract
Background
The role of routine beta-blocker (BB) use after uncomplicated acute myocardial infarction (AMI) treated with contemporary therapies is not well established.
Objectives
The authors conducted a retrospective cohort study using linked registry and administrative data to evaluate whether early BB discontinuation (a prescription ending within 180 days of discharge) is associated with clinical outcomes.
Methods
We included patients who survived at least 180 days after AMI from 2008 to 2017 with new BB prescription and left ventricular ejection fraction ≥50%. The primary outcome was a composite of recurrent AMI, myocardial revascularization, or all-cause mortality within 5 years. Secondary outcomes were each of the components of the composite. Cox proportional hazard models were used to evaluate the association between early BB discontinuation and outcomes.
Results
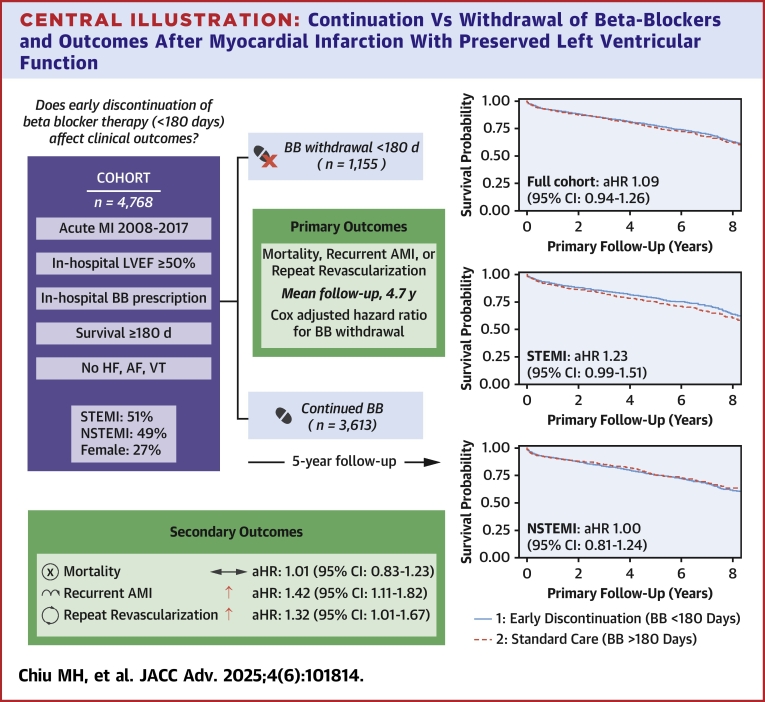
Among the 4,768 included patients, 1,155 (24.2%) discontinued BB within 180 days of AMI discharge. During a median follow-up time of 57 months, 964 patients (20.2%) experienced the primary outcome. Early BB discontinuation was not associated with an increased risk of the primary outcome (adjusted HR: 1.09; 95% CI: 0.94-1.26), or with all-cause mortality (HR: 1.04; 95% CI: 0.86-1.26). However, early BB discontinuation was associated with an increased risk for recurrent AMI and a higher rate of repeat revascularization.
Conclusions
In patients with preserved left ventricular ejection fraction after AMI, discontinuation of BB within 180 days was not associated with a significantly increased risk of a composite outcome of death, recurrent AMI, or revascularization but was associated with increased risk of recurrent AMI and need for repeat revascularization.
Key words: acute myocardial infarction, beta-blocker, mortality
Central Illustration
Guideline-recommended therapy for patients who have suffered an acute myocardial infarction (AMI) includes antiplatelet therapy, statins, inhibitors of the renin-angiotensin-aldosterone system, and beta-blockers (BBs).1,2 BBs are a cornerstone of treatment for ischemic heart disease in the acute phase because they inhibit the physiological sympathetic activation, reduce myocardial workload, reduce infarct size, and have antiarrhythmic and antiremodeling effects.3, 4, 5, 6 BB also have a well-defined role in long-term therapy of patients with persistent reduction in left ventricular (LV) systolic function. However, the role of longer-term BB treatment when AMI course is not complicated by heart failure, LV systolic dysfunction, recurrent ischemia, or arrhythmia is not well established. Most guidelines recommend BB use in the absence of contraindications with grade 1B evidence in ST-segment elevation myocardial infarction (STEMI) and grade 2A in non–ST-segment elevation myocardial infarction (NSTEMI).1,7,8 The recommendation for BB use after AMI predominately comes from studies that predate routine implementation of a contemporary strategy of early reperfusion and medical therapy.9, 10, 11 Recent guidelines report uncertainty in the strength of the evidence for long-term use of BB post revascularization with preserved LV ejection fraction (LVEF).8,12
Observational studies that have evaluated the duration of BB use and outcomes after AMI have several limitations, including incomplete ascertainment or heterogeneous definitions of post-myocardial infarction (MI) LV dysfunction, inclusion of patients with independent indications or contraindications to BB, lack of longitudinal prescription data, and relatively short durations of follow-up.13, 14, 15, 16 Two recently published open-label randomized clinical trials have added to this literature but have not completely resolved the dilemma. The REDUCE AMI (Randomized Evaluation of Decreased Usage of Beta-Blockers after Acute Myocardial Infarction) trial found that initiating BBs in MI survivors with preserved LVEF at the time of discharge did not reduce the incidence of death from any cause or recurrent AMI,17 whereas the ABYSS (Assessment of Beta-Blocker Interruption 1 Year after an Uncomplicated Myocardial Infarction on Safety and Symptomatic Cardiac Events Requiring Hospitalization) trial failed to find that interrupting BBs a mean of 2.9 years after MI was noninferior to continuing them, for a composite primary endpoint of death, nonfatal MI, nonfatal stroke, or hospitalization for cardiovascular reasons.18 We sought to assess the association between discontinuation of newly prescribed BB within 180 days after AMI and long-term cardiovascular outcomes among a well-characterized, population-based cohort of AMI survivors without definite indications for long-term BB therapy.
Methods
Study design and data sources
This was a retrospective cohort study using data from the APPROACH (Alberta Provincial Project for Outcome Assessment in Coronary Heart disease) registry and linked administrative health databases from Alberta, Canada.19,20 APPROACH is a population-based initiative that prospectively collects demographic information, clinical characteristics, comorbidities, details of coronary anatomy and revascularization procedures, and ascertains all-cause mortality through linkage with Alberta Vital Statistics for all patients receiving coronary angiography in the province of Alberta.19 During the study period, all cardiac catheterization and revascularization procedures were performed at one of three tertiary care urban cardiac centers. Data from the APPROACH registry were linked using each patient's unique personal health identifier with echocardiography databases from the largest urban hospital sites in Alberta to enrich ascertainment of LV function. The cohort was also linked with Alberta's Pharmaceutical Information Network, which captures >96% of drug dispensations for patients of any age from community pharmacies in Alberta, as well as the Alberta hospital discharge abstract database, which records all hospital codes for diagnoses and procedures to ascertain nonfatal outcomes. The University of Calgary's Conjoint Health Research Ethics Board approved the study, including a waiver of individual patient consent.
Cohort formation
The cohort included patients with a coronary angiography procedure recorded in APPROACH between January 2008 and March 2017 with AMI as the procedure indication, age ≥18 years at the time of angiography, LVEF ≥50%, receipt of a new prescription for BB at hospital discharge, and survival >180 days after hospital discharge. Patients were excluded if they had a preadmission prescription for BB or did not receive a prescription for a BB at discharge, or if they had in-hospital events typically associated with a requirement for, or a contraindication to, BB therapy, including ventricular arrhythmias, heart failure, atrial fibrillation or flutter, atrioventricular or sinus node dysfunction, or asthma.
Characterization of left ventricular function
LVEF was obtained from the APPROACH registry and hospital echocardiography databases, and we included all measures from the date of admission to 180 days after index discharge. LV function was determined by ventriculogram at the time of angiography and/or with transthoracic echocardiogram results obtained in hospital laboratories. Patients were excluded if the last LVEF measure in the ascertainment period was <50% or if no LVEF was available.
Classification of beta-blocker therapy
Outpatient drug prescription data prior to and following the index hospitalization were determined using the Pharmaceutical Information Network database. Dispensations for all commercially available BBs from 1 year prior to admission to 6 years after discharge from the index hospitalization were determined for all patients. BB prescriptions dispensed between 7 days before to 30 days after discharge were considered as new if they did not have a dispensation in the 1 year prior to admission. BB dosing was assessed at index discharge, 180 days, and 365 days, and classified as low, medium, or high according to equivalent mean metoprolol SR daily dose (low = <50 mg, med = −50 to ≤125 mg, and high = ≥125 mg). Patients were classified as early BB discontinuation if their last BB prescription ended within 180 days of discharge and as standard BB treatment if treatment persisted beyond 180 days. Since early BB prescription post-MI was universally recommended in the absence of contraindications during the study period, the 180-day threshold was used for our landmark analysis to ensure that all patients were deemed BB-eligible by their attending cardiologists during the index AMI hospitalization and most would have had the opportunity for post-AMI cardiology follow-up, with the assumption that prescriptions extending beyond that time were intended to continue long-term. This design strives to emulate a clinical trial of short- vs long-duration BB use post-MI similar in design to the ABYSS trial and avoids some pitfalls of observational research including immortal time bias and failure to align start of follow-up, specification of eligibility, and treatment assignment.21
Outcomes
The primary outcome was a composite of mortality, recurrent AMI, or repeat coronary revascularization. Recurrent AMI was defined as a hospital discharge for STEMI, NSTEMI, or AMI unspecified based on a validated approach using the most-responsible diagnosis from the Discharge Abstract Database (Supplemental Table 1).20 Repeat coronary revascularization was defined as any percutaneous coronary intervention or coronary artery bypass grafting procedures, identified from APPROACH. Secondary outcomes were the individual components of this composite.
Statistical analyses
Patient characteristics were compared using Student's t-test for continuous variables and a chi-squared test for categorical variables. Patient follow-up started at 180 days after index hospital discharge and continued until December 2017 or the date of first primary or secondary outcome event. Patients were censored at the time of death for consideration of secondary outcomes. Time to event curves were plotted using the Kaplan-Meier method. The associations between BB treatment duration and the primary and secondary outcomes were examined using Cox proportional hazards models with adjustment for age, sex, AMI subtype (STEMI or NSTEMI), comorbidities (prior MI, prior coronary artery bypass grafting, prior percutaneous coronary intervention, extent of angiographic coronary artery disease [CAD], major noncardiac comorbidities, and prescriptions for other secondary prevention medications [angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, statin, P2Y12 inhibitor]) assessed 180 days after index discharge. These covariates were included in models because they are known prognostic factors for major adverse cardiac events as well as goal-directed therapy with known mortality benefit. Acetylsalicylic acid is available over the counter in Alberta and thus not accurately measured in the Pharmaceutical Information Network database. The proportional hazards assumption was verified by visual inspection of Kaplan-Meier survival curves. In a post hoc analysis, we reported results stratified according to AMI subtype (STEMI or NSTEMI, excluding the 0.96% of patients with unclassified AMI subtype) and assessed statistical interaction with the Wald test. Two-sided P values <0.05 were used to denote statistical significance. SAS Version 9.4 software (SAS Institute) package was used for analyses.
Results
Cohort formation and characteristics
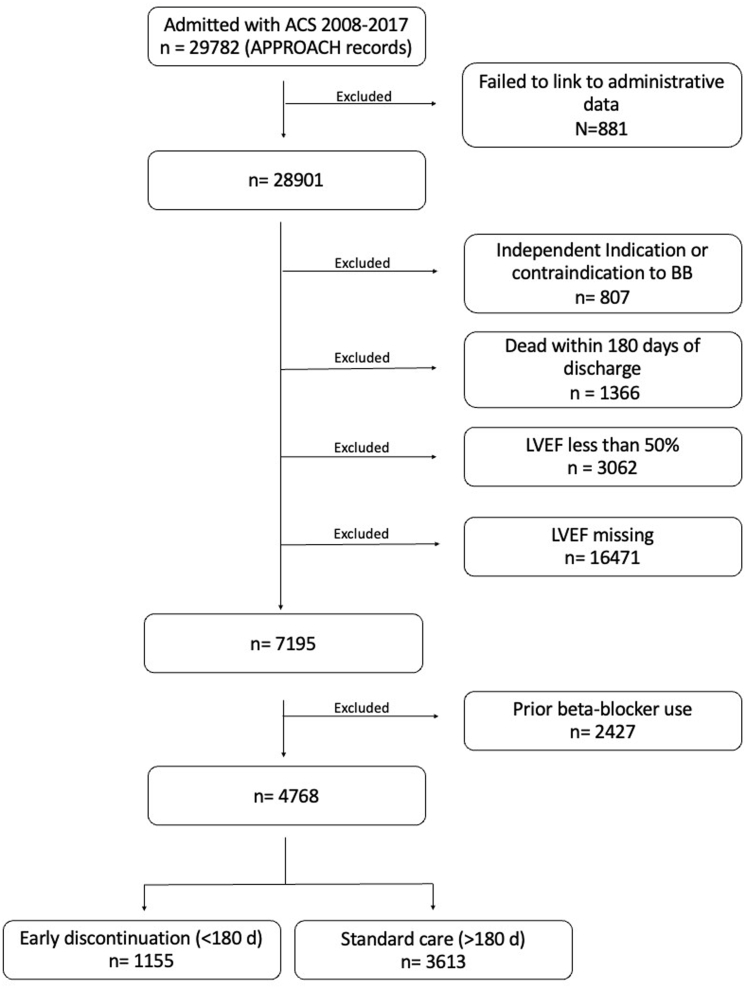
Among the 29,782 potentially eligible patients, 4,768 met all eligibility criteria, including 1,155 (24.2%) in the early BB discontinuation group and 3,613 (75.8%) in the standard BB treatment group (Figure 1). The most frequent reason for exclusion was lack of an available LVEF value. Table 1 summarizes the baseline characteristics of the 2 study groups, and Supplemental Table 2 compares the characteristics of the included cohort with those excluded based on missing in-hospital LVEF value. Patients in the early BB discontinuation group were younger (mean age 59.8 vs 61.5 years; P < 0.001), but the distributions of AMI subtype, sex, and comorbidities were similar between the 2 groups. Patients in the early BB discontinuation group had less extensive CAD and were less likely to undergo percutaneous coronary intervention in-hospital (61.4% vs 71.2%, P < 0.001), but LVEF was similar in the 2 groups. The early BB discontinuation group was also less likely to be prescribed an angiotensin-converting enzyme inhibitor/angiotensin receptor blocker or statins at hospital discharge (P < 0.001). Patients in the 2 groups had a similar distribution of BB medications and dosing at hospital discharge (Supplemental Table 3). All patients in the standard BB treatment group who were alive at 1 year after discharge had an active BB prescription on dispensing records covering this time point. In both groups, the majority of patients were prescribed metoprolol and classified as a medium strength BB dosing.
Figure 1.
Cohort Formation
ACS = acute coronary syndrome; APPROACH = Alberta Provincial Project for Outcome Assessment in Coronary Heart Disease; BB = beta-blocker; LVEF = left ventricular ejection fraction.
Table 1.
Baseline Characteristics of Patients in the Early Vs Standard Beta-Blocker Group
| Early BB Discontinuation (n = 1,155) | Continued BB Treatment (n = 3,613) | P Value | |
|---|---|---|---|
| Characteristic | |||
| Age (y), mean (SD) | 59.8 (13.0) | 61.5 (11.7) | <0.001 |
| Female | 303 (26.2) | 996 (27.6) | 0.38 |
| STEMIa | 562 (48.7) | 1,866 (51.6) | 0.21 |
| NSTEMI | 581 (50.3) | 1,713 (47.4) | |
| Cardiovascular history | |||
| Prior MI | 32 (2.8) | 73 (2.0) | 0.13 |
| Prior PCI | 10 (0.9) | 42 (1.2) | 0.40 |
| Prior CABG | 19 (1.6) | 66 (1.8) | 0.68 |
| Comorbidities | |||
| Cerebrovascular disease | 30 (2.6) | 110 (3.0) | 0.43 |
| Peripheral vascular disease | 91 (7.9) | 274 (7.6) | 0.74 |
| Diabetes mellitus | 213 (18.4) | 750 (20.8) | 0.09 |
| Dyslipidemia | 626 (54.2) | 2,052 (56.8) | 0.12 |
| Smoking current | 400 (34.6) | 1,126 (31.2) | 0.08 |
| Smoking former | 186 (16.1) | 629 (17.4) | 0.08 |
| Chronic pulmonary disease | 106 (9.2) | 276 (7.6) | 0.09 |
| Renal disease | 33 (2.9) | 72 (2.0) | 0.08 |
| Liver disease | 16 (1.4) | 31 (0.9) | 0.11 |
| Malignancy | 37 (3.2) | 92 (2.5) | 0.23 |
| Dementia | 0 (0.0) | 3 (0.1) | 0.33 |
| Extent of CAD | |||
| Nonobstructive | 159 (13.77) | 202 (5.59) | <0.001 |
| 1- or 2-vessel | 651 (56.36) | 2,197 (60.81) | |
| 3-vessel and/or left main | 343 (29.70) | 1,213 (33.57) | |
| Missing | 2 (0.17) | 1 (0.03) | |
| In-hospital events | |||
| PCI | 709 (61.4) | 2,571 (71.2) | <0.0001 |
| CABG | 130 (11.3) | 425 (11.8) | 0.64 |
| Medical therapy alone | 316 (27.4) | 617 (17.0) | <0.0001 |
| Echocardiography | |||
| Predischarge echo | 825 (71.4) | 2,629 (72.8) | 0.38 |
| Predischarge LVEF (%) | 61.4 | 61.7 | 0.53 |
| Follow-up echo within 365 d | 184 (15.9) | 497 (13.8) | 0.07 |
| Median time to echo days, median (IQR) | 94.0 (29.5, 147.0) | 98.0 (47.0, 160.0) | 0.18 |
| Mean follow-up LVEF (%), mean (SD) | 60.4 (12.4) | 60.3 (9.7) | 0.95 |
| Active secondary prevention prescriptions 180 d after index discharge | |||
| ACE inhibitor or ARB | 934 (80.9) | 3,338 (92.4) | <0.0001 |
| Statin | 988 (85.5) | 3,491 (96.6) | <0.0001 |
| P2Y12 inhibitor | 683 (59.1) | 2,822 (78.1) | <0.0001 |
Values are n (%) unless otherwise indicated.
ACE = angiotensin-converting enzyme; ARB = angiotensin receptor blocker; BB = beta-blocker; CABG = coronary artery bypass grafting; CAD = coronary artery disease; LVEF= left ventricular ejection fraction; MI = myocardial infarction; NSTEMI = non–ST-segment elevation myocardial infarction; PCI = percutaneous coronary intervention; STEMI = ST-segment elevation myocardial infarction.
<1% of patients were not classified by AMI subtype as STEMI or NSTEMI.
Clinical outcomes
Over a median follow-up time of 56.6 months, 251 patients (4.51 events per 100 person-years) in the early BB discontinuation group and 713 patients (4.28 per 100 patient-years) in the standard BB treatment group experienced the composite primary outcome (Central Illustration). In the overall cohort, there was no significant association between early BB discontinuation and the primary outcome in the multivariable-adjusted Cox model (HR: 1.1; 95% CI: 1.0-1.3; P = 0.28) (Table 2 and Central Illustration). There was no significant interaction between treatment and AMI presentation type (interaction P = 0.11); with consistent associations between early BB discontinuation and primary outcome in the STEMI subgroup (adjusted HR: 1.2; 95% CI: 1.0-1.5; P = 0.08) and the NSTEMI subgroup (adjusted HR: 1.0; 95% CI: 0.8-1.3; P = 0.9,) in the fully adjusted Cox models.
Central Illustration.
Continuation Vs Withdrawal of Beta-Blockers and Outcomes After Myocardial Infarction With Preserved Left Ventricular Function
AF = atrial fibrillation; aHR = adjusted HR associated with beta-blocker withdrawal; AMI = acute myocardial infarction; HF = heart failure; MI = myocardial infarction; NSTEMI = non–ST-segment elevation myocardial infarction; STEMI = ST-segment elevation myocardial infarction; VT = ventricular tachycardia; other abbreviations as in Figure 1.
Table 2.
Association Between Early BB Discontinuation and the Primary and Secondary Clinical Outcomes
| Early BB Discontinuation (n = 1,155) Crude Events, Incidence Rate |
Standard Care (n = 3,613) Crude Events, Incidence Rate |
Adjusted HR (95% CI) | P Value for Interaction | |
|---|---|---|---|---|
| Primary composite outcome | ||||
| Entire cohort | 251, 4.52 per 100 py | 713, 4.29 per 100 py | 1.09 (0.94-1.27) P = 0.27 | |
| STEMI | 131, 4.94 per 100 py | 337, 3.96 per 100 py | 1.23 (0.99-1.51) P = 0.06 | 0.18 |
| NSTEMI | 118, 4.17 per 100 py | 365, 4.61 per 100 py | 1.00 (0.80-1.24) P = 0.99 | |
| Secondary outcomes | ||||
| All-cause mortality | ||||
| Entire cohort | 156, 2.49 per 100 py | 400, 2.14 per 100 py | 1.01 (0.83-1.23) P = 0.91 | |
| STEMI | 72, 2.37 per 100 py | 169, 1.77 per 100 py | 1.01 (0.75-1.36) P = 0.94 | 0.68 |
| NSTEMI | 82, 2.6 per 100 py | 225, 2.51 per 100 py | 0.94 (0.72-1.23) P = 0.67 | |
| Recurrent AMI | ||||
| Entire cohort | 88, 1.52 per 100 py | 232, 1.33 per 100 py | 1.42 (1.11-1.82) P = 0.006 | |
| STEMI | 50, 1.81 per 100 py | 113, 1.26 per 100 py | 1.62 (1.16-2.28) P = 0.005 | 0.32 |
| NSTEMI | 37, 1.26 per 100 py | 116, 1.39 per 100 py | 1.23 (0.84-1.81) P = 0.28 | |
| Repeat revascularization | ||||
| Entire cohort | 99, 1.71 per 100 py | 291, 1.70 per 100 py | 1.32 (1.05-1.67) P = 0.02 | |
| STEMI | 60, 2.16 per 100 py | 156, 1.78 per 100 py | 1.45 (1.07-1.96) P = 0.01 | 0.13 |
| NSTEMI | 38, 1.29 per 100 py | 131, 1.59 per 100 py | 1.24 (0.85-1.79) P = 0.26 |
All models are adjusted for age, sex, acute myocardial infarction (AMI) subtype, prior MI, prior CABG, prior PCI, cerebrovascular disease, peripheral vascular disease, heart failure, hypertension, diabetes, smoking status, renal disease, liver disease, malignancy, dyslipidemia, diabetes, extent of CAD, Duke Jeopardy score, and prescriptions for other secondary prevention medications (ACEi or ARB, statin, P2Y12 inhibitors). We excluded 3 patients who had missing data for extent of CAD.
AMI = acute myocardial infarction; py = persons-years; other abbreviations as in Table 1.
There was a significantly higher hazard of recurrent AMI in the early BB discontinuation group in the overall cohort (adjusted HR: 1.4; 95% CI: 1.1-1.8; P = 0.007). This association was stronger in the STEMI subgroup (adjusted HR: 1.6; 95% CI: 1.2-2.3) than in the NSTEMI subgroup (adjusted HR: 1.2; 95% CI: 0.8-1.7), though the interaction test was not significant (P = 0.26) (Table 2). There was also a significantly higher hazard of repeat revascularization in the entire cohort (adjusted HR: 1.3; 95% CI: 1.1-1.7; P = 0.01) driven by the STEMI subgroup (adjusted HR: 1.5; 95% CI: 1.1-2.0; P = 0.01). There was no statistically significant association between early BB discontinuation and all-cause mortality, and no significant interaction between treatment group and STEMI or NSTEMI presentation. Supplemental Tables 4 to 7 report the full multivariable adjusted models for the primary and secondary outcomes.
Discussion
In this cohort of survivors of uncomplicated AMI free of heart failure, arrhythmia, or LV dysfunction, 24% of patients had early discontinuation of BBs within 180 days. Continuation of BBs past 180 days was not associated with a reduction in the composite endpoint of death, recurrent AMI, or repeat revascularization in the subsequent 5 years. However, we observed an association between early BB discontinuation and increased risks of both recurrent AMI and repeat coronary revascularization procedures. No association was found between early BB discontinuation and all-cause mortality. Our study helps to address some of the limitations of previous observational studies, by restricting inclusion to new users of BB after AMI who are eligible for this treatment without other evidence-based indications or contraindications, and focusing on the timeframe when decisions about the duration of BB therapy post-MI are most relevant (ie, 6 months after the index AMI). Our finding of similar rates of death regardless of BB continuation or withdrawal is in keeping with the REDUCE-AMI and ABYSS triaIs.17,18 However, both of these trials reported no difference in recurrent AMI between treatment groups, contrary to our findings. The differences may be due to a larger number of follow-up AMI events in our study leading to increased power to detect an effect, or to unrecognized confounding, or to chance. The ABYSS trial, which was similar in design to this analysis, did suggest a higher rate of cardiovascular hospitalizations in the BB interruption group, though the rate of repeat revascularization procedures was relatively low and similar between the groups. One important difference between the ABYSS trial and this analysis is that in ABYSS, the mean time between the last AMI and randomization was 2.9 years, suggesting that the trial enrolled a population with chronic stable CAD. In addition, our observations may be related to the lower use of percutaneous coronary intervention during the index hospitalization (61% to 71%) in our study compared with the randomized trials.
The current American Heart Association guideline recommendation continuation of BB in patients with normal LV function after STEMI and NSTEMI as Class I and Class IIa recommendations, respectively.1,7 The European Society of Cardiology guidelines provide a Class IIa recommendation.22 However, the evidence for these guideline recommendations was generated prior to routine reperfusion and revascularization and modern pharmacological therapies for AMI, including statins and dual antiplatelet therapies. The 2023 American Heart Association/American College of Cardiology guideline for the management of patients with chronic CAD acknowledged the uncertain benefit of continuing BB beyond 1 year in patients with a previous MI but without a history of or current LVEF ≤50%, angina, arrhythmias, or uncontrolled hypertension (Class 2b, Level of Evidence: B–nonrandomized).23
Our study adds to the evidence suggesting a re-evaluation of the role of long-term BB therapy in uncomplicated AMI in the current management era and suggests that not all patients benefit from long-term BB following AMI. Previous observational studies have explored the impacts of BB on clinical outcomes in the context of contemporary AMI treatment, with conflicting results. A retrospective study of 91,895 patients identified between 2007 and 2013 found no difference in mortality with an average treatment effect coefficient of 0.07; 95% CI: −0.60 to 0.76; P = 0.827 for acute coronary syndrome (ACS) patients with no significant difference between STEMI and NSTEMI patients.13 A study of 3,177 patients from the Korea Acute Myocardial Infarction Registry database found no survival benefits of BB treatment at 2 years in patients with an LVEF >50% at 1 year (P = 0.004).24 A 2015 metanalysis including 40,873 patients found no significant association between BB therapy and rates of cardiac death, ACS, or readmission in patients with preserved LV function.25 A cohort study of 179,810 survivors of AMI without heart failure or severe LV dysfunction showed no mortality benefit at 1 year with BB use and no differences between NSTEMI and STEMI cohorts.13 The lack of benefit was also observed in a 969 patient STEMI cohort published in 2021.14 Additionally a recent retrospective study including 2,519 patients without heart failure with LVEF >50% found no difference in all-cause mortality, rehospitalization, recurrent MI, or admission for heart failure at short term (1 year) and long-term (median 3.61 years).26
Conversely, a large retrospective study of 28,970 patients suggested early discontinuation of BB had an increased risk of all-cause mortality, recurrent MI, hospitalization, and heart failure.15 A 2021 Cochrane review including 25 trials suggested that BB may reduce the risk of mortality, recurrent ACS, and angina in post-AMI patients without heart failure, but the authors noted low to moderate certainty of the results.16 The majority of recent studies reporting benefits of longer-term BB therapy have defined their cohorts based on the absence of heart failure but without a requirement to document normal LVEF. Overall, studies on this topic have yielded mixed results and do not support a clear consensus to continue BB in the long-term in stable post-MI patients with preserved LV systolic function. The optimal duration of BB use also remains unclear. Studies that report a mortality benefit have varying results from 30 days27 to over 1 year of BB use.15,28,29 Many of these studies report limitations in monitoring longitudinal prescriptions. Benefits of BB can be potentially underestimated if prescriptions are not filled or if patients are nonadherent. Our study of 4,768 patients provides longitudinal prescription data over the follow-up period.
An interesting finding of our analysis is the potential for increased risks of recurrent AMI and revascularization with BB discontinuation, in particular among people with a STEMI as their index event. Patients in the early discontinuation group in the STEMI subgroup of our cohort had higher frequency of recurrent AMI and need for repeat revascularization with percutaneous coronary intervention compared to the BB continuation group (adjusted HR: 1.7; 95% CI: 1.2-2.3; P = 0.004) and adjusted HR: 1.5; 95% CI: 1.1-2.0; P = 0.01, respectively), suggesting that they may be a group with a higher risk of recurrent ischemia who might benefit from BB. It is possible that incomplete revascularization was a more common contributing factor to recurrent AMI in patients with STEMI. Additionally, patients with NSTEMI tend to have more comorbidities, atherosclerotic disease, and history of prior MI and coronary revascularization than those with STEMI suggesting they have a different phenotype that responds differently to BB.30 It is unlikely that less intensive use of other medical therapies explains differences between patients with STEMI and NSTEMI, as this was not observed in our study.31 Neither the REDUCE AMI nor ABYSS trials observed differential effects of BB therapy on their primary outcome according to AMI subtype.17,18
Our results support the need for further randomized control trials to better understand the role of BB use following AMI in patients with preserved LV function. Such clinical trials should be designed to resolve remaining questions regarding treatment duration, dosage, and effect modification by different types of ACS. Multiple ongoing clinical trials are aiming to help resolve these questions, including SMART-DECISION (Long-term Beta-blocker Therapy After Acute Myocardial Infarction; NCT04769362), REBOOT-CNIC (TREatment With Beta-blockers After myOcardial Infarction withOut Reduced Ejection fracTion; NCT03596385), REDUCE-SWEDEHEART (Evaluation of Decreased Usage of Betablockers After Myocardial Infarction in the SWEDEHEART Registry; NCT03278509), BETAMI (BEtablocker Treatment After Acute Myocardial Infarction in Patients Without Reduced Left Ventricular Systolic Function; NCT03646357), and DANBLOCK (Danish Trial of Beta Blocker Treatment After Myocardial Infarction Without Reduced Ejection Fraction; NCT03778554).32 Future guideline iterations incorporating the results of the currently published and ongoing observational and randomized studies should provide increased confidence in recommendations informing questions of the need for and optimal duration of BB therapy in patients with uncomplicated AMI.
Study Limitations
This was a retrospective observational study making it susceptible to bias owing to residual confounding from unmeasured covariates and treatment selection biases. We required documentation of preserved LVEF for study inclusion, which could have introduced selection bias. However, this design choice was integral to our research question. Our population was limited to the province of Alberta, Canada and may lack generalizability to different practice patterns, ethnic diversity and health care systems. Our study intentionally excluded patients who died within 180 days of their index AMI in order to address a specific question related to the long-term continuation of BB therapy in patients for whom it was newly prescribed post-AMI. This design does not permit an assessment of the impacts of duration of BB therapy shorter than 180 days and potentially adds survival bias. Whether BBs were discontinued on the advice of a physician, side effects, nonadherence, or for other reasons is unknown, and the reason for discontinuation could be related to subsequent outcomes. Typically, patients adherent to medications have higher health literacy and/or socioeconomic status with overall better health outcomes.33 In our study a significant difference was observed between the 2 groups in the extent of CAD and use of percutaneous coronary intervention. Though we adjusted for these variables, it is conceivable that incomplete revascularization was a driver of recurrent AMI in the early BB discontinuation group. As with all subgroup analyses, the stratified analyses based on the STEMI and NSTEMI should be considered exploratory and may represent a chance finding.
Conclusions
In survivors of uncomplicated AMI with preserved LVEF, discontinuation of BB before 180 days was not associated with increased risk the composite outcome of death, recurrent AMI, or repeat coronary revascularization. However, early BB discontinuation was associated with increased risks of recurrent AMI and repeat revascularization, with a suggestion that these associations may be stronger in those with STEMI compared with NSTEMI as the index presentation. Randomized trials testing durations of BB therapy after AMI should be designed to examine differences between patients with STEMI and NSTEMI and focus on patients with preserved LVEF.
Perspectives.
COMPETENCY IN MEDICAL KNOWLEDGE: In survivors of uncomplicated AMI with preserved LV function, whether long-term BB therapy improves outcomes has been unclear. In this observational study, discontinuation of BBs before 180 days was not associated with increased risk the composite outcome of death, recurrent AMI, or repeat coronary revascularization over 5 years of follow-up. However, BB discontinuation was associated with increased risks of recurrent MI and repeat revascularization, with a suggestion that these associations may be stronger in those with STEMI compared with NSTEMI as the index presentation.
COMPETENCY IN PATIENT CARE AND PROCEDURAL SKILLS: The need for ongoing BB therapy should be individualized in follow-up for patients with preserved LV function following MI. In the absence of angina, heart failure symptoms, arrhythmias, many patients may be able to safely discontinue this therapy.
TRANSLATIONAL OUTLOOK: Randomized trials testing durations of BB therapy after MI should be designed to examine differences between patients with STEMI and NSTEMI and focus on patients with preserved LV function.
Funding support and author disclosures
Funding was provided by the Libin Cardiac Institute's Clinical Research Development Fund. AbSPORU is supported by CIHR, Alberta Innovates, the University Hospital Foundation, and in-kind contributions from AHS, WCHRI, ACHRI, and the Universities of Alberta and Calgary. The authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Acknowledgments
The authors acknowledge Maria Dalton, MSc, for assistance with figure preparation. The APPROACH registry relies on the efforts of Alberta's cardiovascular health professionals for maintaining exceptional data completeness and quality.
Footnotes
The authors attest they are in compliance with human studies committees and animal welfare regulations of the authors’ institutions and Food and Drug Administration guidelines, including patient consent where appropriate. For more information, visit the Author Center.
Appendix
For supplemental tables, please see the online version of this paper.
Supplemental data
References
- 1.O’Gara P.T., Kushner F.G., Ascheim D.D., et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction. Circulation. 2013;127(4):e362–e425. doi: 10.1161/CIR.0b013e3182742cf6. [DOI] [PubMed] [Google Scholar]
- 2.Amsterdam E.A., Wenger N.K., Brindis R.G., et al. 2014 AHA/ACC guideline for the management of patients with non–ST-elevation acute coronary syndromes. J Am Coll Cardiol. 2014;64(24):e139–e228. doi: 10.1016/j.jacc.2014.09.017. [DOI] [PubMed] [Google Scholar]
- 3.Yusuf S., Peto R., Lewis J., Collins R., Sleight P. Beta blockade during and after myocardial infarction: an overview of the randomized trials. Prog Cardiovasc Dis. 1985;27(5):335–371. doi: 10.1016/s0033-0620(85)80003-7. [DOI] [PubMed] [Google Scholar]
- 4.Olsson G., Rehnqvist N. Evaluation of antiarrhythmic effect of metoprolol treatment after acute myocardial infarction: relationship between treatment responses and survival during a 3-year follow-up. Eur Heart J. 1986;7(4):312–319. doi: 10.1093/oxfordjournals.eurheartj.a062069. [DOI] [PubMed] [Google Scholar]
- 5.Group B.R. A randomized trial of propranolol in patients with acute myocardial infarction. JAMA. 1982;247:1707. doi: 10.1001/jama.1982.03320370021023. [DOI] [PubMed] [Google Scholar]
- 6.Group I.-C. Randomised trial of intravenous atenolol among 16,027 cases of suspected acute myocardial infarction: ISIS-1. Lancet. 1986;2(8498):57–66. [PubMed] [Google Scholar]
- 7.Amsterdam E.A., Wenger N.K., Brindis R.G., et al. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: executive summary: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. J Am Coll Cardiol. 2014;64(24):2645–2687. doi: 10.1016/j.jacc.2014.09.017. [DOI] [PubMed] [Google Scholar]
- 8.Byrne R.A., Rossello X., Coughlan J.J., et al. 2023 ESC Guidelines for the management of acute coronary syndromes: developed by the task force on the management of acute coronary syndromes of the European Society of Cardiology (ESC) Eur Heart J. 2023;44(38):3720–3826. doi: 10.1093/eurheartj/ehad191. [DOI] [PubMed] [Google Scholar]
- 9.Freemantle N., Cleland J., Young P., Mason J., Harrison J. β Blockade after myocardial infarction: systematic review and meta regression analysis. BMJ. 1999;318(7200):1730–1737. doi: 10.1136/bmj.318.7200.1730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hjalmarson Å., Herlitz J., Malek I., et al. Effect on mortality of metoprolol in acute myocardial infarction: a double-blind randomised trial. Lancet. 1981;318(8251):823–827. doi: 10.1016/s0140-6736(81)91101-6. [DOI] [PubMed] [Google Scholar]
- 11.Chen Z., Pan H., Chen Y., et al. COMMIT (Clopidogrel and Metoprolol in Myocardial Infarction Trial) collaborative group early intravenous then oral metoprolol in 45,852 patients with acute myocardial infarction: randomised placebo-controlled trial. Lancet. 2005;366(9497):1622–1632. doi: 10.1016/S0140-6736(05)67661-1. [DOI] [PubMed] [Google Scholar]
- 12.Lawton J.S., Tamis-Holland J.E., Bangalore S., et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on clinical practice guidelines. J Am Coll Cardiol. 2022;79(2):e21–e129. doi: 10.1016/j.jacc.2021.09.006. [DOI] [PubMed] [Google Scholar]
- 13.Dondo T.B., Hall M., West Robert M., et al. β-Blockers and mortality after Acute Myocardial Infarction in patients without heart failure or ventricular dysfunction. J Am Coll Cardiol. 2017;69(22):2710–2720. doi: 10.1016/j.jacc.2017.03.578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Velásquez-Rodríguez J., Bruña V., Vicent L., et al. Influence of left ventricular systolic function on the long-term benefit of beta-blockers after ST-segment elevation myocardial infarction. Rev Port Cardiol (Engl Ed) 2021;40(4):285–290. doi: 10.1016/j.repc.2020.07.017. [DOI] [PubMed] [Google Scholar]
- 15.Kim J., Kang D., Park H., et al. Long-term β-blocker therapy and clinical outcomes after acute myocardial infarction in patients without heart failure: nationwide cohort study. Eur Heart J. 2020;41(37):3521–3529. doi: 10.1093/eurheartj/ehaa376. [DOI] [PubMed] [Google Scholar]
- 16.Safi S., Sethi N.J., Korang S.K., et al. Beta-blockers in patients without heart failure after myocardial infarction. Cochrane Database Syst Rev. 2021;11:CD012565. doi: 10.1002/14651858.CD012565.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yndigegn T., Lindahl B., Mars K., et al. Beta-blockers after myocardial infarction and preserved ejection fraction. N Engl J Med. 2024;390(15):1372–1381. doi: 10.1056/NEJMoa2401479. [DOI] [PubMed] [Google Scholar]
- 18.Silvain J., Cayla G., Ferrari E., et al. Beta-blocker interruption or continuation after myocardial infarction. N Engl J Med. 2024;391(14):1277–1286. doi: 10.1056/NEJMoa2404204. [DOI] [PubMed] [Google Scholar]
- 19.Ghali W.A., Knudtson M.L. Overview of the Alberta provincial Project for outcome assessment in coronary heart disease. On behalf of the APPROACH investigators. Can J Cardiol. 2000;16(10):1225–1230. [PubMed] [Google Scholar]
- 20.Southern D.A., James M.T., Wilton S.B., et al. Expanding the impact of a longstanding Canadian cardiac registry through data linkage: challenges and opportunities. Int J Popul Data Sci. 2018;3(3):9. doi: 10.23889/ijpds.v3i3.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hernan M.A., Sauer B.C., Hernandez-Diaz S., Platt R., Shrier I. Specifying a target trial prevents immortal time bias and other self-inflicted injuries in observational analyses. J Clin Epidemiol. 2016;79:70–75. doi: 10.1016/j.jclinepi.2016.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Steg P.G., James S.K., Atar D., et al. ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2012;33(20):2569–2619. doi: 10.1093/eurheartj/ehs215. [DOI] [PubMed] [Google Scholar]
- 23.Virani S.S., Newby L.K., Arnold S.V., et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA guideline for the management of patients with chronic coronary disease: a report of the American Heart Association/American College of Cardiology Joint Committee on clinical practice guidelines. Circulation. 2023;148(9):e9–e119. doi: 10.1161/CIR.0000000000001168. [DOI] [PubMed] [Google Scholar]
- 24.Park C.S., Yang H.-M., Ki Y.-J., et al. Left ventricular ejection fraction 1 Year after acute myocardial infarction identifies the benefits of the long-term use of β-blockers: analysis of data from the KAMIR-NIH registry. Circulation. 2021;14(4) doi: 10.1161/CIRCINTERVENTIONS.120.010159. [DOI] [PubMed] [Google Scholar]
- 25.Huang B.-T., Huang F.-Y., Zuo Z.-L., et al. Meta-analysis of relation between oral β-blocker therapy and outcomes in patients with acute myocardial infarction who underwent percutaneous coronary intervention. Am J Cardiol. 2015;115(11):1529–1538. doi: 10.1016/j.amjcard.2015.02.057. [DOI] [PubMed] [Google Scholar]
- 26.Wen X.S., Luo R., Liu J., et al. Short-term/long-term prognosis with or without beta-blockers in patients without heart failure and with preserved ejection fraction after acute myocardial infarction: a multicenter retrospective cohort study. BMC Cardiovasc Disord. 2022;22(1):193. doi: 10.1186/s12872-022-02631-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Puymirat E., Riant E., Aissaoui N., et al. β blockers and mortality after myocardial infarction in patients without heart failure: multicentre prospective cohort study. BMJ. 2016;354:i4801. doi: 10.1136/bmj.i4801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Konishi H., Miyauchi K., Kasai T., et al. Long-term effect of β-blocker in ST-segment elevation myocardial infarction in patients with preserved left ventricular systolic function: a propensity analysis. Heart Vessels. 2016;31(4):441–448. doi: 10.1007/s00380-014-0624-2. [DOI] [PubMed] [Google Scholar]
- 29.Misumida N., Harjai K., Kernis S., Kanei Y. Does oral beta-blocker therapy improve long-term survival in ST-segment elevation myocardial infarction with preserved systolic function? A meta-analysis. J Cardiovasc Pharmacol Ther. 2016;21(3):280–285. doi: 10.1177/1074248415608011. [DOI] [PubMed] [Google Scholar]
- 30.Montalescot G., Dallongeville J., Van Belle E., et al. STEMI and NSTEMI: are they so different? 1 year outcomes in acute myocardial infarction as defined by the ESC/ACC definition (the OPERA registry) Eur Heart J. 2007;28(12):1409–1417. doi: 10.1093/eurheartj/ehm031. [DOI] [PubMed] [Google Scholar]
- 31.Bouisset F., Ruidavets J.B., Dallongeville J., et al. Comparison of short- and long-term prognosis between ST-elevation and non-ST-elevation myocardial infarction. J Clin Med. 2021;10(2):180. doi: 10.3390/jcm10020180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kristensen A.M.D., Bovin A., Zwisler A.D., et al. Design and rationale of the Danish trial of beta-blocker treatment after myocardial infarction without reduced ejection fraction: study protocol for a randomized controlled trial. Trials. 2020;21:415. doi: 10.1186/s13063-020-4214-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Simpson S.H., Eurich D.T., Majumdar S.R., et al. A meta-analysis of the association between adherence to drug therapy and mortality. BMJ. 2006;333(7557):15. doi: 10.1136/bmj.38875.675486.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.