Abstract
The CTG repeat at the 3′ untranslated region of the dystrophia myotonica protein kinase (DMPK) gene shows marked intergenerational and somatic instability in patients with myotonic dystrophy (DM1), when the repeat is expanded to more than ∼55 repeats. Intensive research has yielded some insights into the timing and mechanism of these intergenerational changes: (1) increases in expansion sizes occur during gametogenesis but probably not during meiosis, (2) the marked somatic mosaicism becomes apparent from the 2nd trimester of development onward and increases during adult life, and (3) DNA repair mechanisms are involved. We have performed preimplantation genetic diagnosis for DM1 since 1995, which has given us the unique opportunity to study the expanded CTG repeat in affected embryos and in gametes from affected patients. We were able to demonstrate significant increases in the number of repeats in embryos from female patients with DM1 and in their immature and mature oocytes, whereas, in spermatozoa and embryos from male patients with DM1, smaller increases were detected. These data are in concordance with data on other tissues from adults and fetuses and fill a gap in our knowledge of the behavior of CTG triplet expansions in DM1.
Myotonic dystrophy (DM1 [MIM 160900]), is a neuromuscular disorder with primary symptoms of myotonia and muscle weakness mainly involving facialmuscles and distal muscles of the extremities. Other symptoms can be cataract; endocrine changes, such as testicular atrophy; and disruptions of the CNS, such as increased somnolence, apathy, and even mental retardation in the congenital form of the disease. The molecular basis of the disease lies in the instability of a CTG triplet repeat in the 3′ UTR of the DM1 protein kinase gene (DMPK). In normal individuals, the repeat size ranges from 3 to 37 repeats, whereas patients with DM1 exhibit expansions of >50 repeats and, often, as many as several thousand repeats in the congenital form of the disease (Brook et al. 1992). Expanded CTG repeats are unstable and usually increase in number in successive generations. In DM1, an aggravation of symptoms (called “anticipation”) from asymptomatic to overt DM1 is found often when transmitted through the father, whereas children with the severe congenital form of the disease are born almost exclusively to affected mothers (Harper and Johnson 2001). In somatic cells, a high level of mosaicism can be detected also, depending on the patient’s age and repeat number (Monckton et al. 1995; Wong et al. 1995) and on the tissues examined—repeat expansions are found to be smallest in blood and largest in heart, muscle, and kidney (Jansen et al. 1994; Martorell et al. 1997).
Currently, the working hypothesis is that the instability may occur at different stages of DNA interactions, such as DNA replication before meiosis, double-strand break formation during meiosis, and DNA repair after meiosis (Pearson et al. 2002, 2003; Sinden et al. 2002). Little information is available, about the stability of trinucleotide repeats in human early embryos and in female gametes, that can confirm these hypotheses. At our center, preimplantation genetic diagnosis (PGD) for DM1 has been done since 1995 (Sermon et al. 2001). This has given us the unique opportunity to study female gametes and preimplantation embryos shown to be affected after PGD and donated for research by the patients, thus enabling us to close the gap left by the analysis of blood and offspring.
Blood, gametes, and embryos were analyzed from 32 different patients. The mean age of the patients with DM1 at PGD was 31.4 years. The local medical ethics committee approved this study, and patients signed an informed consent. PGD was performed as described elsewhere (Sermon et al. 2001).
Spermatozoa from patients with DM1 were collected either in groups of 10 or 20 cells or as single cells by micromanipulation. The zona pellucida of the germinal vesicle (GV), metaphase I (MI) and mature metaphase II (MII) oocytes with their polar bodies, and research embryos were removed with the use of acidic Tyrode solution. Embryos that were unsuitable for biopsy at day 3 were collected, as well as embryos shown to be affected after PGD and embryos shown to be unaffected after PGD that were unsuitable for transfer or cryopreservation as a result of bad morphology. All samples were collected in 0.2-ml tubes containing alkaline lysis buffer (200 mM of KOH or NaOH and 50 mM of dithiothreitol).
A specific PCR for long fragments, followed by Southern blot analysis, was performed on the samples tovisualize the expansions. However, because the small alleles could not be discriminated, a fluorescent second-round PCR, with the PCR product of the long PCR as a template, was performed and was followed by analysis on an automated DNA sequencer (ALFExpress, Amersham Pharmacia Biotech). Together with each set of gametes or embryos, 100 pg of the DNA from parents affected with DM1 was amplified for comparison. Both PCRs were performed using the Expand Long Template system (Roche Diagnostics), with 0.3 μM of primers DM101 and DM102 (Brook et al. 1992) and in accordance with the manufacturer’s instructions.
PCR fragments were separated either on a regular nondenaturing agarose gel or, owing to the occurrence of heteroduplexes, on a denaturing alkaline agarose gel (Sambrook and Russell 2001). After Southern blot, the nylon membrane was hybridized to the digoxigeninlabeled (CAG)5 probe (Eurogentec) and was processed further with the use of the DIG-CSPD system (Roche Diagnostics), in accordance with the manufacturers' recommendations.
As determined by Martorell et al. (2000), the “true germline change” is the difference between the smallest measured allele in blood DNA of the affected parent (i.e., the affected gene present in the sperm or oocyte that, after fertilization, gave rise to the affected patient who later came to us for PGD treatment) and the expansion measured in the gamete or embryo. The mean size of the smallest expanded band in the DNA of our patient group was 133.5 repeats for all 32 patients: 133.9 repeats for the 22 female patients (range 42–468 repeats) and 132.6 repeats for the 10 male patients (range 62–256 repeats).
Gametes and embryos from 64 PGD cycles were analyzed. These 64 cycles were performed for 32 patients: 48 were for the 22 female patients, and 16 were for the 10 male patients.
Because of the limited amount of data obtained from ejaculated sperm and the high interpatient variability (Martorell et al. 2000), great care must be taken in drawing conclusions from this group. The average germline change in sperm that showed an expansion was +55.9 repeats, and the highest repeat number detected was 329 repeats, which gave a germline change of +186 repeats. In total, 18 spermatozoa showed a reduction, with an average change of −41.1 repeats, whereas the repeat remained stable in 13 spermatozoa. The behavior of the triplet repeat in spermatozoa and its relation to the repeat of the progenitor agree with what has been described in the literature (Jansen et al. 1994; Monckton et al. 1995; Martorell et al. 2000): significant but not extensive enlargements were detected in spermatozoa of patients with small expansions. Also, a larger number of contractions were detected in spermatozoa and embryos from affected males than in oocytes and embryos from affected females. Because of the extensive data already existing in literature, only a few cells were tested.
A total of 56 oocytes were analyzed. In the GV oocytes, 3 (of 15) showed an expansion, and the average germline change was +337.0 repeats, with a maximal enlargement of +404 repeats. Of the MI oocytes, 8 (of 25) showed an expansion, and the average germline change was +401.1 repeats, with a maximal enlargement of +950 repeats. The average germline change for the 11 (of 16) MII oocytes was +332.2 repeats, with the largest enlargement being +614 repeats.
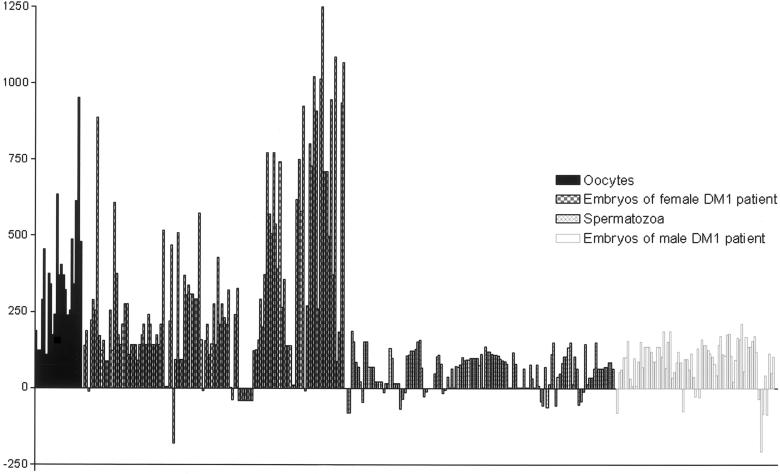
Of the 144 embryos, 77 (53%) were diagnosed as affected during PGD, and 92 (39%) of the 236 nonbiopsied embryos showed an expansion of the repeat (fig. 1). The increase in the size of the repeat in oocytes and embryos from female patients was larger; again, this was in agreement with the literature. The largest expansion we could detect was +1,372 repeats (compared with +369 repeats, the largest repeat number in samples from males), though the largest expansions were probably missed because of the limitation of the amplification procedure and the possible deterioration of DNA in the cells.
Figure 1.
CTG instability in 346 samples from patients with DM1. Each bar represents the germline change in one sample, either an oocyte, spermatozoon, or embryo. The samples on the left are from female patients (blackened boxes and checkered boxes), and the samples on the right are from male patients (grid boxes and unblackened boxes). The germline change is the difference between the smallest band detected in blood DNA of the parent and the size of the repeat in the embryos, oocytes, or spermatozoa.
The amplification rate of expanded alleles was rather low for the oocytes (39%), nonbiopsied embryos (39%), and affected embryos (53%). The low amplification efficiency in the oocytes can be explained by the fact that these are abnormal, and possibly dying, cells. In the case of the nonbiopsied embryos, this can be explained partially by the fact that approximately half of the embryos are, in fact, healthy, but this does not hold true for the affected embryos. These findings mainly illustrate the limits of our PCR assay to detect larger expansions and may be explained by the fact that some of the embryos were already degenerated at the moment of analysis.
No mosaicism was detected in any of the affected embryos. These data agree with earlier results showing that somatic mosaicism in affected fetuses starts only during the 2nd trimester of gestation (Jansen et al. 1994; Martorell et al. 1997).
In table 1, a summary is given of the germline changes measured in all spermatozoa, oocytes, and embryos after maternal or paternal transmission. Because of the limited number of oocytes, only a general trend can be deduced from this data set. The difference in range of instability of the triplet repeat after paternal and maternal transmission is remarkable. Not only is the range of instability nearly 10 times larger after maternal transmission, but more contractions of the repeat number also occur (fig. 1) after paternal transmission. Because of the small sample size, these differences are not statistically significant (P=.1046, χ2 test).
Table 1.
Germline Change for Samples with an Enlarged Repeat Number after Maternal and Paternal Transmission[Note]
|
Result in |
||||||||
| Embryos |
Oocytes |
|||||||
| Transmission Typeand Statistic | 2PNa | Cleavageb | Compactc | Blastocyst | ImmatureOocytesd | MIIOocytese | Spermatozoa | All |
| Maternal: | ||||||||
| N | 15 | 89 | 17 | 2 | 11 | 11 | … | 145 |
| Average germline change | +288.2 | +308.8 | +232.6 | +919.5 | +383.6 | +332.2 | … | +313.6 |
| Maximum germline change | +1,014 | +1,085 | +1,281 | +1,067 | +950 | +614 | … | +1,281 |
| Minimum germline change | −10 | −42 | −180 | +772 | +121 | +108 | … | −180 |
| No. of contractions | 1 | 9 | 2 | 0 | 0 | 0 | … | 12 |
| Paternal: | ||||||||
| N | 5 | 51 | 11 | 8 | … | … | 126 | 201 |
| Average germline change | +118.0 | +100.3 | +35.2 | +46.2 | … | … | +55.9 | +68.1 |
| Maximum germline change | +179 | +210 | +114 | +186 | … | … | +186 | +210 |
| Minimum germline change | −86 | −81 | −85 | −208 | … | … | −81 | −208 |
| No. of contractions | 1 | 3 | 2 | 2 | … | … | 18 | 28 |
Note.— The germline change is defined as the difference between the smallest measured allele in blood DNA of the affected parent (i.e., the allele in the gamete at the origin of the parent carrying the mutation) and the expansion measured in the gamete or embryo (Martorell et al. 2000).
Pronuclear single-cell embryo (2PN = two pronuclei).
Embryos arrested at the cleavage stage (from 2-cellular to 10-cellular cleavage stage embryos).
Embryos arrested at the compacted stage (>12 cells).
The immature oocytes include oocytes at the GV stage and oocytes arrested at the MI stage.
The mature MII oocytes are MII oocytes not suitable for microinjection.
The exceptional availability of post-PGD material allowed us to investigate immature and mature oocytes. Although the oocyte sample is too small to draw definite conclusions, our results indicate that enlargement of the repeat had occurred at the GV stage, that is, either during premeiotic proliferation of oogonia or during prophase I of meiosis I. Several models exist to explain the instability of triplet repeats during DNA replication (Cleary et al. 2002; Yang et al. 2003). Other models mainly focus on the appearance of errors in the DNA repair; several publications suggest that the DNA repair mechanism (mismatch repair system or MMR) plays an important role in the instabilities of the triplet repeats (Pearson et al. 1997; van den Broek et al. 2002; Savouret et al. 2003). This agrees with the findings of Kaytor et al. (1997), who examined the behavior of a spinocerebellar ataxia type I (SCA1, another neurodegenerative disorder caused by a dynamic mutation) cDNA containing a CTG trinucleotide repeat tract introduced into transgenic mice. Those authors hypothesized that, in oocytes, instability of the triplet repeat occurs while the oocytes are arrested in meiosis I after meiotic DNA replication. However, the timing of the expansion instability contrasts with the findings of Kovtun and McMurray (2001), who concluded from their investigation of spermatozoa from mice transgenic for the exon 1 of the human Huntington disease (HD) gene that expansion arises in the haploid stage of spermatozoa development, by gap repair. Yoon et al. (2003) could detect expansions in the HD locus in premeiotic and postmeiotic human male germ cells, leading those authors to conclude that the long lifespan and lifelong cell division of postpubertal spermatogonial stem cells make them likely targets for age-dependent mutations that arise by DNA damage repair or by cell-division–dependent DNA replication errors, respectively. This hypothesis was expanded further by Pearson (2003) to the female germ line, where the emphasis would be on DNA repair, owing to the limited number of cell divisions undergone by the female germ line. Our finding that expansion has occurred already in immature oocytes before completion of meiosis would, indeed, implicate DNA repair in these cells that have undergone a long period of quiescence. The differences in timing and/or mechanism could also account for the differences observed in transmission of the expansion between males and females. Further study of immature sperm progenitors—for example, in testicular biopsies from patients with DM1 undergoing PGD—as well as in earlier stages of female gametes, should shed more light on this question.
Acknowledgments
The authors wish to thank the clinical, paramedical, and laboratory staff at the Centers for Medical Genetics and Reproductive Medicine and J. Deconinck of the Language Education Center of the Vrije Universteit Brussel (VUB). This work was supported by a concerted research action (geconcerteerde onderzoeksactie) of the VUB. K.S. and M.D.R. were supported by grants from the Funds for Scientific Research Flanders.
Electronic-Database Information
The URL for data presented herein is as follows:
- Online Mendelian Inheritance in Man (OMIM), http://www.ncbi.nlm.nih.gov/Omim/ (for DM1) [PubMed]
References
- Brook JD, McCurrach ME, Harley HG, Buckler AJ, Church D, Aburatani H, Hunter K, Stanton VP, Thirion J-P, Hudson T, Sohn R, Zemelman B, Snell RG, Rundle SA, Crow S, Davies J, Shelbourne P, Buxton J, Jones C, Juvonen V, Johnson K, Harper PS, Shaw DJ, Housman DE (1992) Molecular basis of myotonic dystrophy: expansion of a trinucleotide (CTG) repeat at the 3′ end of a transcript encoding a protein kinase family member. Cell 68:799–808 10.1016/0092-8674(92)90154-5 [DOI] [PubMed] [Google Scholar]
- Cleary J, Nichol K, Wang Y-H, Pearson CE (2002) Evidence of cis-acting factors in replication-mediated trinucleotide repeat instability in primate cells. Nat Genet 31:37–46 10.1038/ng870 [DOI] [PubMed] [Google Scholar]
- Harper P, Johnson K (2001) Myotonic dystrophy. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic and molecular basis of inherited disease. McGraw-Hill, Colombus, pp 5525–5550 [Google Scholar]
- Jansen G, Willems P, Coerwinkel M, Nillesen W, Smeets H, Vits L (1994) Gonosomal mosaicism in myotonic dystrophy patients: involvement of mitotic events in (CTG)n repeat variation and selection against extreme expansion in sperm. Am J Hum Genet 54:575–585 [PMC free article] [PubMed] [Google Scholar]
- Kaytor MD, Burright EN, Duvick LA, Zoghbi H, Orr HT (1997) Increased trinucleotide repeat instability with advanced maternal age. Hum Mol Genet 6:2135–2139 10.1093/hmg/6.12.2135 [DOI] [PubMed] [Google Scholar]
- Kovtun IV, McMurray CT (2001) Trinucleotide expansion in haploid germ cells by gap repair. Nat Genet 27:407–411 10.1038/86906 [DOI] [PubMed] [Google Scholar]
- Martorell L, Johnson K, Boucher CA, Baiget M (1997) Somatic instability of the myotonic dystrophy (CTG)n repeat during human fetal development. Hum Mol Genet 6:877–880 10.1093/hmg/6.6.877 [DOI] [PubMed] [Google Scholar]
- Martorell L, Monckton DG, Gamez J, Baiget M (2000) Complex patterns of male germline instability and somatic mosaicism in myotonic dystrophy type 1. Eur J Hum Genet 8:423–430 10.1038/sj.ejhg.5200478 [DOI] [PubMed] [Google Scholar]
- Monckton D, Wong L-J, Ashizawa T, Caskey T (1995) Somatic mosaicism, germline expansions, germline reversions and intergenerational reductions in myotonic dystrophy males: small pool PCR analyses. Hum Mol Genet 4:1–8 [DOI] [PubMed] [Google Scholar]
- Pearson CE, Ewel A, Acharya S, Fishel RA, Sinden RR (1997) Human MSH2 binds to trinucleoide repeat DNA structures associated with neurodegenerative diseases. Hum Mol Genet 6:1117–1123 10.1093/hmg/6.7.1117 [DOI] [PubMed] [Google Scholar]
- Pearson CE, Tam M, Wang Y-H, Montgomery SE, Dar AC, Cleary JD, Nichol K (2002) Slipped-strand DNAs formed by long (CAG)*(CTG) repeats: slipped-out repeats and slip-out junctions. Nucleic Acid Res 30:4534–4547 10.1093/nar/gkf572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson CE (2003) Slipping while sleeping? Trinucleotide repeat expansions in germ cells. Trends Mol Med 9:490–495 10.1016/j.molmed.2003.09.006 [DOI] [PubMed] [Google Scholar]
- Sambrook J, Russell DW (2001) Alkaline agarose gel electrophoresis. In: Sambrook J, Russell DW (eds) Molecular cloning: a laboratory manual, 3rd ed. Cold Spring Harbor, New York, pp 5.36–5.39 [Google Scholar]
- Savouret C, Brisson E, Essers J, Kanaar R, Pastink A, te Riele H, Junien C, Gourdon G (2003) CTG repeat instability and size variation timing in DNA repair-deficient mice. EMBO Journal 22:2264–2273 10.1093/emboj/cdg202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sermon K, Seneca S, De Rycke M, Goossens V, Van de Velde H, De Vos A, Platteau P, Lissens W, Van Steirteghem A, Liebaers I (2001) PGD in the lab for triplet repeat diseases: myotonic dystrophy, Huntington's disease and Fragile-X syndrome. Mol Cell Endocrinol Suppl 183:77–85 10.1016/S0303-7207(01)00572-X [DOI] [PubMed] [Google Scholar]
- Sinden RR, Potaman VN, Oussatcheva EA, Pearson CE, Lyubchenko YL, Shlyakhtenko LS (2002) Triplet repeat DNA structures and human genetic disease: dynamic mutations from dynamic DNA. J Biosci Suppl 27:53–65 [DOI] [PubMed] [Google Scholar]
- van den Broek WJ, Nelen MR, Wansink DG, Coerwinkel MM, te Riele H, Groenen PJ, Wieringa B (2002) Somatic expansion behaviour of the (CTG)n repeat in myotonic dystrophy knock-in mice is differentially affected by Msh3 and Msh6 mismatch-repair proteins. Hum Mol Genet 11:191–198 10.1093/hmg/11.2.191 [DOI] [PubMed] [Google Scholar]
- Wong LC, Ashizawa T, Monckton DG, Caskey CT, Richards CS (1995) Somatic heterogeneity of the CTG repeat inmyotonic dystrophy is age and size dependent. Am J Hum Genet 56:114–122 [PMC free article] [PubMed] [Google Scholar]
- Yang Z, Lau R, Marcadier JL, Chitayat D, Pearson CE (2003) Replication inhibitors modulate instability of an expanded trinucleotide repeat at the myotonic dystrophy type 1 disease locus in human cells. Am J Hum Genet 73:1092–1105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon SR, Dubeau L, de Young M, Wexler NS, Arnheim N (2003) Huntington disease expansion mutations in humans can occur before meiosis is completed. PNAS 100:8834–8838 10.1073/pnas.1331390100 [DOI] [PMC free article] [PubMed] [Google Scholar]