Abstract
Hepatic stellate cells (HSCs) activation represents a central pathological mechanism in liver fibrosis, with emerging evidence implicating fatty acid metabolic reprogramming as a critical regulator of this process. Our study established the vitamin D receptor (VDR) as a key transcriptional coordinator of fatty acid metabolism during HSC activation. Genetic VDR deletion in mice exacerbated liver fibrosis progression, which was associated with elevated TGF-β1 levels and increased Smad3 phosphorylation. Mechanistically, VDR deficiency disrupted lipid homeostasis through the upregulation of lipogenic enzymes (fatty acid synthase, acetyl-CoA carboxylase 1, ATP citrate lyase) and desaturases (stearoyl-CoA desaturase-1, fatty acid desaturases 1/2) and the suppression of the β-oxidation gatekeeper carnitine palmitoyltransferase 1A (CPT1A). Pathological VDR downregulation was observed in both TGF-β1-activated HSCs and fibrotic liver tissues, suggesting a disease-associated regulatory circuit. Calcitriol-mediated VDR activation reversed TGF-β1-induced Smad3 phosphorylation and normalized metabolic enzyme expression, effectively reducing lipid accumulation and collagen deposition. We further identified senkyunolide I as a novel natural VDR agonist that rebalances fatty acid metabolism by simultaneously downregulating lipogenesis/desaturation machinery and upregulating CPT1A. The complete abolition of anti-fibrotic effects of senkyunolide I following VDR knockdown confirmed its strict receptor dependency. These findings identify VDR as a master regulator of metabolic reprogramming in HSC activation and validate pharmacological VDR activation as a promising therapeutic strategy for liver fibrosis. The dual metabolic regulatory capacity of senkyunolide I through VDR signaling highlights its potential for targeted antifibrotic intervention.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13020-025-01133-x.
Keywords: Senkyunolide I, Fatty acid metabolism reprogram, Vitamin D receptor, Liver fibrosis, Hepatic stellate cells
Introduction
Liver fibrosis represents a pivotal pathological process in chronic liver disease progression [44] and is characterized by excessive deposition and aberrant remodeling of extracellular matrix (ECM) components within hepatic tissue [40]. This condition has grave clinical implications, with epidemiological studies showing a median survival of 2–3 years postdiagnosis and five-year survival rates of < 30%. Current antiviral, including therapies-including entecavir, adefovir, and telbivudine, demonstrate clinical efficacy in viral suppression [28], although their utility is limited by dose-limiting toxicity. These adverse effects include gastrointestinal reactions, hepatic damage, visual impairment, and renal dysfunction, which collectively compromise therapeutic sustainability and negatively impact patient prognosis [46].
The activation of hepatic stellate cells (HSCs) represents a pivotal mechanism in liver fibrogenesis [22], and is characterized primarily by their differentiation into myofibroblasts and excessive extracellular matrix (ECM) deposition. Emerging evidence highlights that dysregulated fatty acid metabolism in activated HSCs exacerbates hepatic lipid accumulation and fibrosis progression, creating a self-perpetuating pathogenic cycle [41, 47]. At the molecular level, transforming growth factor-β1 (TGF-β1) orchestrates HSCs activation by inducing α-smooth muscle actin (α-SMA) expression and fibrogenic phenotype [21]. Besides, TGF-β1 upregulates key lipogenic enzymes including fatty acid synthase (FASN), acetyl-CoA carboxylase 1 (ACC1), ATP-citrate lyase (ACLY), fatty acid desaturase (FADS1/2), and stearoyl-CoA desaturase 1 (SCD1) [13], while simultaneously suppressing carnitine palmitoyltransferase 1 A (CPT1 A), the rate-limiting enzyme in fatty acid oxidation [51]. This dual modulation shifts HSCs toward lipid anabolism and impairs β-oxidation, resulting in intracellular lipid overload. The Smad3 signaling pathway serves as a critical mediator of these effects, further reinforcing the profibrotic microenvironment through disrupted hepatic lipid homeostasis and amplified ECM deposition [27]. Notably, pharmacological inhibition of these dysregulated metabolic nodes (particularly FASN, ACC1, and SCD1) has demonstrated efficacy in attenuating HSCs activation and reducing collagen accumulation [34], positioning fatty acid metabolism as a promising therapeutic target for preventing fibrosis progression.
The vitamin D receptor (VDR) plays a pivotal role in regulating lipid homeostasis in HSCs. Ligand-activated VDR has been shown to effectively suppress HSCs activation and attenuate extracellular matrix deposition in hepatic fibrosis [48]. Experimental studies using obese mouse models demonstrate that VDR activation reduces fatty acid and cholesterol levels [42], whereas VDR deficiency aggravates diet-induced hepatic lipid accumulation and promotes chronic inflammation [12, 23]. Mechanistic investigations reveal that VDR agonists modulate key fatty acid metabolism enzymes such as ACC-1 and FASN [48], with effects observed in multiple tissue types including adipose tissue, adipose-derived mesenchymal stem cells [35], and colorectal tumorigenesis models [52]. However, the exact molecular mechanisms through which VDR maintains lipogenesis balance during HSCs activation remain to be fully elucidated.
In this study, we demonstrated that VDR deficiency exacerbates hepatic fibrosis by triggering metabolic reprogramming in HSCs. Mechanistically, VDR depletion activates the TGFβ1/Smad3 signaling pathway, leading to the transcriptional upregulation of lipogenic enzymes (FASN, ACC-1, ACLY) and fatty acid desaturases (SCD1, FADS1/2), while simultaneously suppressing CPT1 A. This metabolic dysregulation results in lipid accumulation and enhanced ECM deposition in activated HSCs. Importantly, we discovered senkyunolide I (SI) as a novel VDR agonist that specifically targets the ligand-binding domain. SI demonstrates potent anti-fibrotic effects in CCl4-induced fibrotic mice by normalizing lipid metabolism homeostasis and inhibiting TGF-β1/Smad3 signaling, without causing hypercalcemia. These findings establish VDR-mediated metabolic regulation as a promising therapeutic target and underscore the SI's clinical potential for treating liver fibrosis.
Materials and methods
Animal treatment
Male C57BL/6 mice (8-weeks-old) were obtained from Hangzhou Medical College (Zhejiang, China) and housed under specific pathogen-free conditions with a 12-h light/dark cycle, ad libitum access to food and water. VDR-knockout (VDR−/−) and wild-type (WT) mice on a C57BL/6 background were generated as previously described [30]. Hepatic fibrosis was induced via intraperitoneal (i.p.) injection of 10% CCl4 (C112045, Aladdin, Shanghai, China; i.p.) three times a week [3]. The vehicle control group received equivalent volumes of olive oil (O108686, Aladdin). CCl4-treated mice were randomized into five groups: model group, low-dose SI (SI-L, 50 mg/kg, i.p.; YJ0118, Yongjian Pharmaceutical, Jiangsu, China), high-dose SI (SI–H, 100 mg/kg, i.p.) [49], positive drug silybin (SLB, 20 mg/kg, i.p.,HY-N0779 A, Yongjian Pharmaceutical) [37], and positive drug calcitriol (VD, 5 µg/kg, i.p.,#C120126, Aladdin) [38].
Assessment of liver function parameters
The alanine aminotransferase (ALT), aspartate aminotransferase (AST) alkaline phosphatase (ALP) activity, and total bilirubin (TBIL) concentrations were quantified using commercial kits (C009-2-1, C010-2-1, A059-2-2, and C019-1-1; Nanjing Jiancheng Bioengineering Institute, Jiangsu, China). The absorbance was recorded using a SpectraMax 190 microplate reader (Molecular Devices, USA).
Histopathological analysis
Right hepatic lobes were fixed in 10% neutral buffered formalin and paraffin-embedded. Hematoxylin–eosin (HE) staining was performed using a standardized protocol [25] with a commercial kit (G1076, Servicebio, Hubei, China). Histological sections were examined under an APERIO VERSA 8 microscope (Leica, Germany).
Collagen deposition assessment
Sirius red staining was conducted following established protocols [11] using a specialized staining kit (HK2021, Servicebio). Collagen fiber distribution was visualized and documented microscopically.
Fibrosis Evaluation
Masson's trichrome staining was performed according to validated methods [5] with a commercial kit (G1006, Servicebio), enabling differential visualization of collagen fibers and cellular components.
Immunohistochemistry
Liver sections were immunostained using validated protocols [53, 55] with the following primary antibodies: anti-TGF-β1 (BF8012, Affinity, Jiangsu, China,1:200), anti-p-Smad3 (AF8315, Affinity; 1:200), anti-α-SMA (BF9212, Affinity; 1:200), and anti-collagen type I α 1 (COL1 A1, PB0981, Boster; Hubei; 1:200) and imaged under a confocal laser scanning microscope (LSM800, Zeiss, Oberkochen, Germany).
Cell culture and treatment
HSCs were obtained from the Chinese Academy of Sciences Type Culture Collection and maintained in DMEM supplemented with 2% FBS (11,995,065, Gibco) at 37 °C with 5% CO2. Cells were divided into nine experimental groups: (1) untreated control; (2) TGF-β1 model (10 ng/mL, Ag24881, Proteintech, Hubei, China) [8], (3–5) TGF-β1 + SI (20, 50, 100 μM) [43], (6–7) TGF-β1 + VD (8, 16 μM) [36]; (8) TGF-β1 + SLB (100 μM) [17]; (9) TGF-β1 + TVB3664 (TVB, 150 nM, 2097262-58-1, MedChemExpress, NJ, USA) [32]. After 24 h treatment, cells were harvested for downstream analysis.
Cell viability
Cell viability was determined via 3-(4,5-dimethylthiazol-2-yl)−2,5-diphenyltetrazolium bromide kit (C0009S, Beyotime) with absorbance measurements at 490 nm (Spectra MAX 190, Molecular Devices). Results were normalized to untreated controls.
Quantitative RT-PCR analysis
Total RNA was isolated using TRIzol (99,089,501, Invitrogen, CA, US) and reverse transcribed with SweScript RT SuperMix (G3337, Servicebio). Gene expression of α-Sma, Col1a1, Timp1, Cpt1a, Fasn, Fads1, Fads2, Acc-1, Acly, and Scd1 was quantified on a 7500 Real-Time PCR System (Applied Biosystems, USA) using SYBR Green Master Mix (G3326, Servicebio). GAPDH served as endogenous control, with primer sequences detailed in Table S1.
Western blot
Total protein extracts were prepared using KeyGen Kits (KGP250, KeyGen), with nuclear fractions isolated via a nuclear and cytoplasmic protein extraction kit (P0027, Beyotime). After Bradford quantification (KGA801, KeyGen), proteins were separated by SDS-PAGE and transferred to PVDF membranes. Membranes were blocked with 5% BSA and probed overnight at 4 °C with antibodies against: α-SMA (1:2000), COL1 A1 (1:1000), FASN (10624-2-AP, Proteintech, 1:1000), VDR (AF8159, Affinity, 1:1000), p-Smad3 (1:2000), Smad3 (AF6362, Affinity, 1:2000), TGF-β1 (1:3000) and β-actin (BM3873, Boster, 1:2000). HRP-conjugated secondary antibodies were used for chemiluminescent detection (ChemiDoc MP, Bio-Rad, CA, USA).
RNA interfection
HSCs were transfected with VDR-targeting siRNA (si-VDR, GenePharma, Shanghai, China) and negative control siRNA (si-NC) using Lipo6000 transfection reagent (C0526, Beyotime). The sequences of the siRNAs used are provided in Table S2. The knockdown efficiency was confirmed through western blot analysis of VDR protein expression. The experimental groups included the following: 1) si-NC; 2) si-VDR; 3) si-NC + TGF-β1 (10 ng/mL); 4) si-VDR + TGF-β1; 5) si-NC + TGF-β1 + SI (100 μM); and 6) si-VDR + TGF-β1 + SI. The cells were harvested 24 h post-treatment for subsequent analyses.
Immunofluorescence staining
Fixed cells (4% paraformaldehyde) were permeabilized with 0.1% Triton X-100, and then incubated overnight at 4 °C with the following primary antibodies: anti-α-SMA and anti-CT-8 (PB2553, Proteintech; 1:500). Alexa Fluor-conjugated secondary antibodies were applied for 1 h. Nuclei were counterstained with DAPI (C1005, Beyotime). Images were acquired using a Zeiss LSM510 META confocal microscope and analyzed with ZEN imaging software.
Neutral lipid quantification
The cells were fixed and stained with Nile Red (C2051S, Beyotime) to detect cytoplasmic neutral lipid accumulation. Fluorescence intensity was quantified using Zeiss LSM510 META with 543 nm excitation/598 nm emission filters.
Molecular docking
The 3D structure of theVDR (PDB ID: 1 KB2) was retrieved from RCSB Protein Data Bank. The 2D structure of SI (PubChem CID: 3,085,257) was converted to 3D using Chem3D [31]. Molecular docking was performed in AutoDock 4.2. The results were visualized using PyMOL [4].
Cellular thermal shift assay (CETSA)
Protein aliquots were thermally challenged at 30, 40, 50, and 60 °C for 3 min to induce temperature-dependent denaturation [10]. Following incubation, samples were mixed with loading buffer and analyzed by western blot to determine the thermal stability profile of VDR.
Statistical analysis
Statistical analyses were performed using GraphPad Prism 8 (GraphPad Software, CA, USA). Normality and homogeneity of variances were confirmed before implementing Student's t-test for two-group comparisons and one-way ANOVA for multigroup analyses. Following significant ANOVA results (P < 0.05), Tukey's honestly significant difference test was employed for post hoc pairwise comparisons. Continuous data are expressed as mean ± standard deviation (SD), with P values < 0.05 considered statistically significant.
Results
VDR ablation exacerbates liver fibrosis and disrupts fatty-acid metabolism in mice
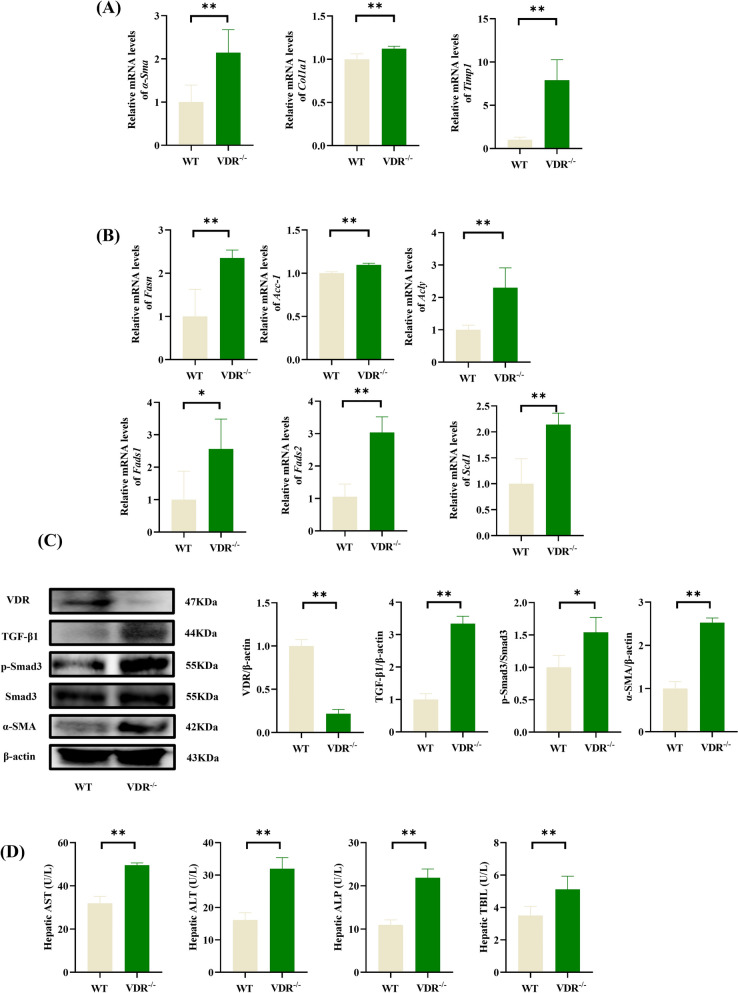
VDR, a nuclear receptor, exerts protective effects against inflammation and fibrogenesis by modulating transcriptional pathways [9]. To investigate the role of VDR in liver pathology, we analyzed the expression of profibrotic markers (α-Sma, Col1a1, and Timp1) [2, 22] and fatty acid metabolism regulators in VDR-deficient mice. Compared with those in WT controls, hepatic mRNA expression of α-Sma, Col1a1, and Timp1 was significantly elevated in VDR−/− mice (Fig. 1A), indicating aggravated fibrotic progression. Concurrently, VDR ablation markedly upregulated lipogenic enzymes (Fasn, Fads1, and Fads2) and key fatty acid synthesis regulators (Acc-1, Acly, and Scd1) (Fig. 1B), suggesting profound dysregulation of lipid homeostasis.
Fig. 1.
VDR deficiency exacerbated hepatic fibrosis and steatosis in mice. A mRNA levels of fibrogenic markers (α-Sma, Col1a1, and Timp1). B mRNA levels of lipogenic enzymes (Fasn, Acc-1, and Acly) and fatty acid desaturases (Fads1/2 and Scd1) (n = 5). C Western blot analysis of VDR and TGFβ/Smad pathway (n = 3), D Serum levels of liver function markers (AST, ALT, ALP, and TBIL) (n = 5). *P < 0.05, **P < 0.01
Western blot analysis revealed that VDR knockout mice exhibited elevated hepatic protein levels of TGF-β1 and α-SMA (Fig. 1C), which was consistent with enhanced fibrotic signaling. Phosphorylated Smad3 (p-Smad3), a downstream effector of TGF-β1, was significantly increased in the livers of VDR−/− livers. The levels of serum ALT, AST, ALP and TBIL levels, biomarkers of hepatocellular injury, were also elevated in VDR−/− mice (Fig. 1D), further supporting the role of VDR in mitigating liver damage.
VDR activation mitigates TGF-β1-induced metabolic dysregulation and fibrogenesis in HSCs
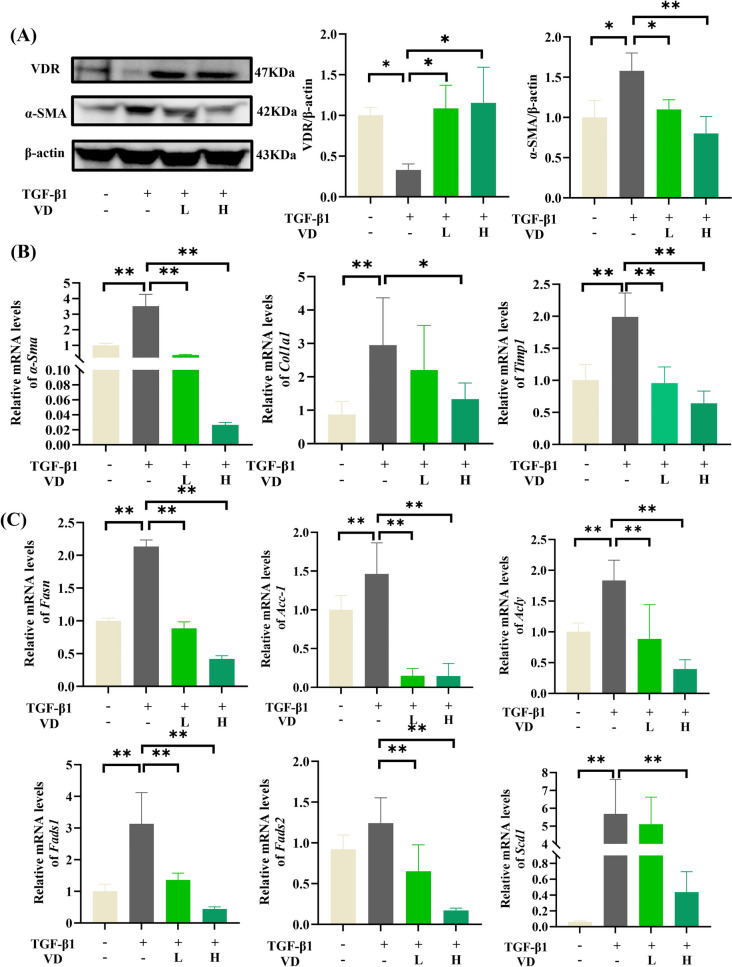
TGF-β1 stimulation significantly suppressed VDR expression (Fig. 2A) while elevating key fibrotic markers α-Sma, Col1a1, and Timp1 in HSCs (Fig. 2B). Calcitriol treatment dose-dependently reversed the TGF-β1-induced downregulation of VDR expression and upregulation of fibrotic markers.
Fig. 2.
VDR activation attenuated TGF-β1-induced fibrosteatotic changes in HSCs. A Western blot analysis of VDR and α-SMA (n = 3). B mRNA levels of fibrogenic markers (α-Sma, Col1a1, and Timp1). C mRNA levels of lipogenic enzymes (Fasn, Acc-1, and Acly) and fatty acid desaturases (Fads1/2 and Scd1) (n = 6). *P < 0.05, **P < 0.01
To investigate the metabolic regulatory mechanism, we analyzed mRNA expression of lipogenic enzymes, which showed significant elevation in TGF-β1-stimulated HSCs compared to controls (Fig. 2C). Calcitriol administration normalized these metabolic perturbations, demonstrating that VDR activation exerts dual anti-fibrotic and lipid-homeostatic effects in activated HSCs.
SI attenuates TGF-β1-mediated HSCs activation via VDR interaction
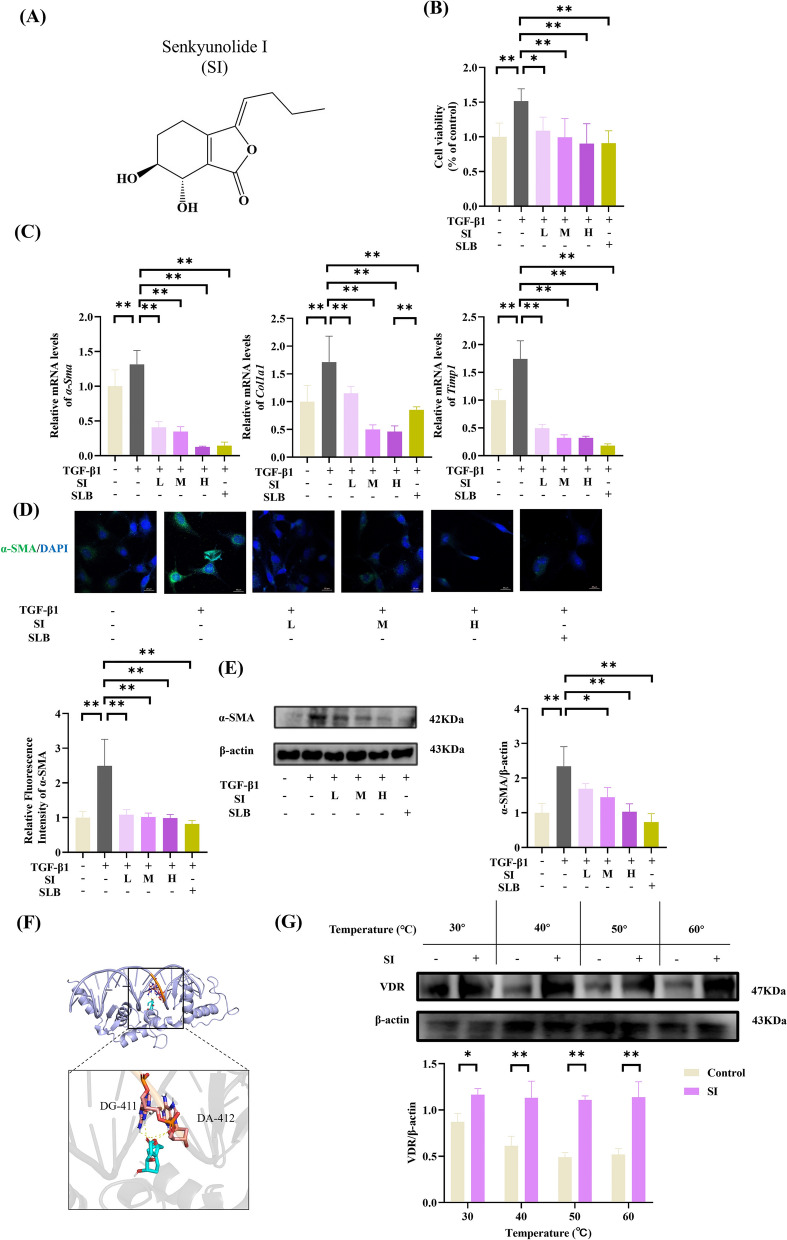
We investigated the inhibitory effects of SI on TGF-β1-induced HSCs activation. As shown in Fig. 3A, B, SI significantly reversed the TGF-β1 induced increase in HSCs. Compared with those of the controls, RT-PCR revealed dose-dependent suppression of fibrotic markers (α-Sma, Col1a1, and Timp1) by SI in the TGF-β1-stimulated cells (Fig. 3C). Immunofluorescence (Fig. 3D) and western blot (Fig. 3E) analyses confirmed SI-mediated reduction of α-SMA protein expression in TGF-β1-activated HSCs.
Fig. 3.
SI inhibited TGF-β1-induced activation of HSCs. A Chemical structure of the SI. B Cell viability of HSCs. C mRNA levels of fibrogenic markers (α-Sma, Col1a1, and Timp1). D Immunofluorescence analysis of α-SMA. scale bar = 20 μm. (n = 6). E Western blot analysis of α-SMA. F Molecular docking results of VDR and SI. G CETSA analysis. (n = 3) *P < 0.05, **P < 0.01
Molecular docking simulations predicted stable binding between SI and VDR with low binding energy (− 7.0 kcal/mol), suggesting direct interaction (Fig. 3F). CETSA demonstrated the ability of SI to stabilize VDR against thermal degradation at 30–60 °C (Fig. 3G).
SI ameliorates CCl4-induced liver fibrosis in mice
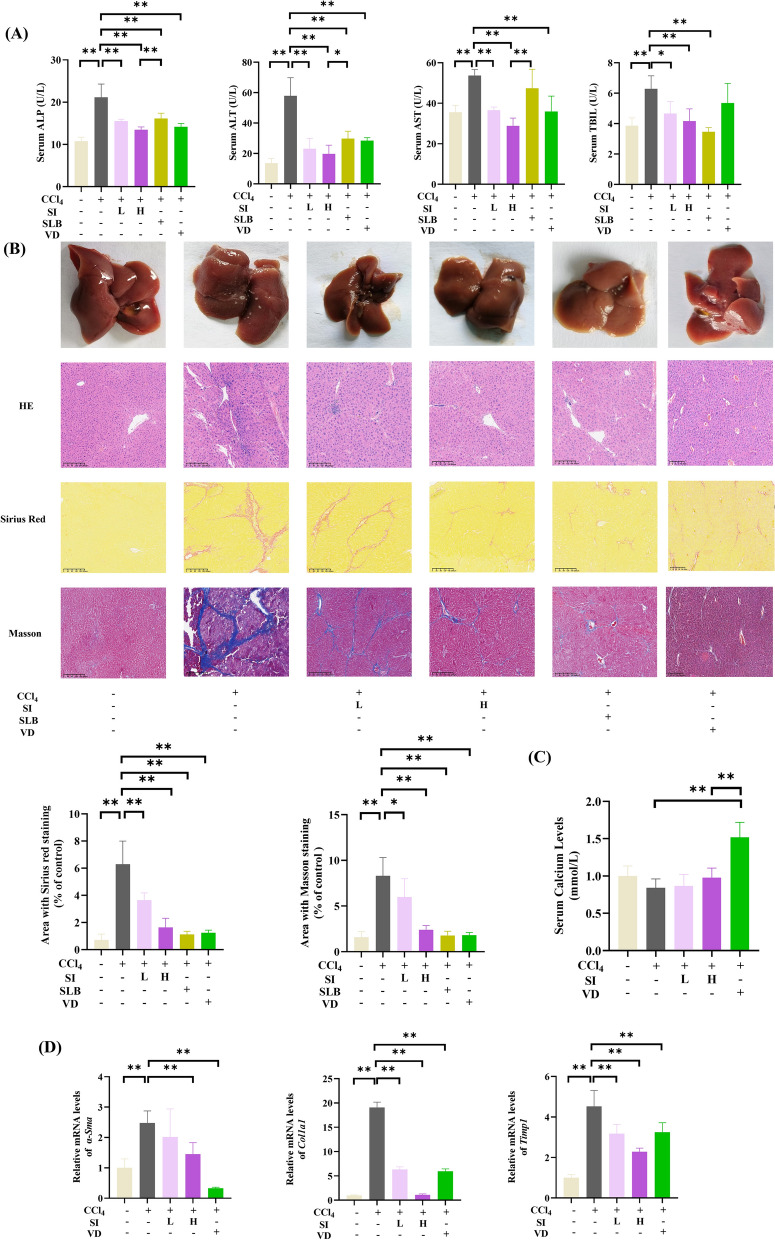
To assess the therapeutic efficacy of SI against hepatic fibrosis, we established a CCl4-induced murine liver fibrosis model. SI and VD treatment attenuated CCl4-driven elevations in the levels of serum markers of hepatic injury, including ALP, ALT, AST, and TBIL (Fig. 4A), demonstrating its hepatoprotective effects. Additionally, our study demonstrated no observable toxicity associated with SI administration in the liver fibrosis model. All animals treated with SI exhibited a 100% survival rate.
Fig. 4.
SI ameliorated CCl4-induced liver fibrosis in mice. A Serum levels of liver function markers (AST, ALT, ALP, and TBIL). B Liver tissue photograph and histopathological analysis (HE, Sirius Red, Masson), scale bar = 200 μm. C Serum calcium concentrations. D mRNA levels of fibrogenic markers (α-Sma, Col1a1, and Timp1). (n = 5) *P < 0.05, **P < 0.01
Gross morphological analysis revealed that CCl4-exposed livers presented rough surfaces with coarse granularity, blunt edges, and hardened texture. The interventions of SI, silybin, and VD improved hepatic architecture, reducing surface irregularities and fibrosis severity (Fig. 4B).
Histopathological evaluation by HE staining confirmed extensive hepatocyte disarray, inflammatory infiltration, and focal necrosis in the CCl4 group. The treatments of SI, silybin, and VD markedly reduced necrotic areas and inflammatory cell recruitment. We found that CCl4 did not cause fibrosis in kidney, and SI had no significant effect on renal α-SMA and COL1 A1 expression levels (Fig. S1).
Masson’s trichrome staining, a validated method for fibrosis quantification [54], further demonstrated that CCl4 exposure disrupted hepatic lobular architecture, inducing marked fibrous septa formation surrounding parenchymal structures (pseudolobulation). SI administration significantly decreased collagen deposition compared with that in the model group.
Notably, SI did not induce hypercalcemia, as serum calcium levels remained unchanged compared with those in the model group (Fig. 4C). In contrast, VD treatment caused significant hypercalcemia, which is consistent with the known calcium-regulatory properties of VD. RT-PCR analysis demonstrated marked upregulations of α-Sma, Timp1 and Col1a1 mRNA levels in the model group compared with those in the control group (Fig. 4D), which was attenuated by SI in a dose-dependently manner. These results collectively indicate that SI alleviates CCl4-induced hepatic fibrosis without perturbing calcium homeostasis.
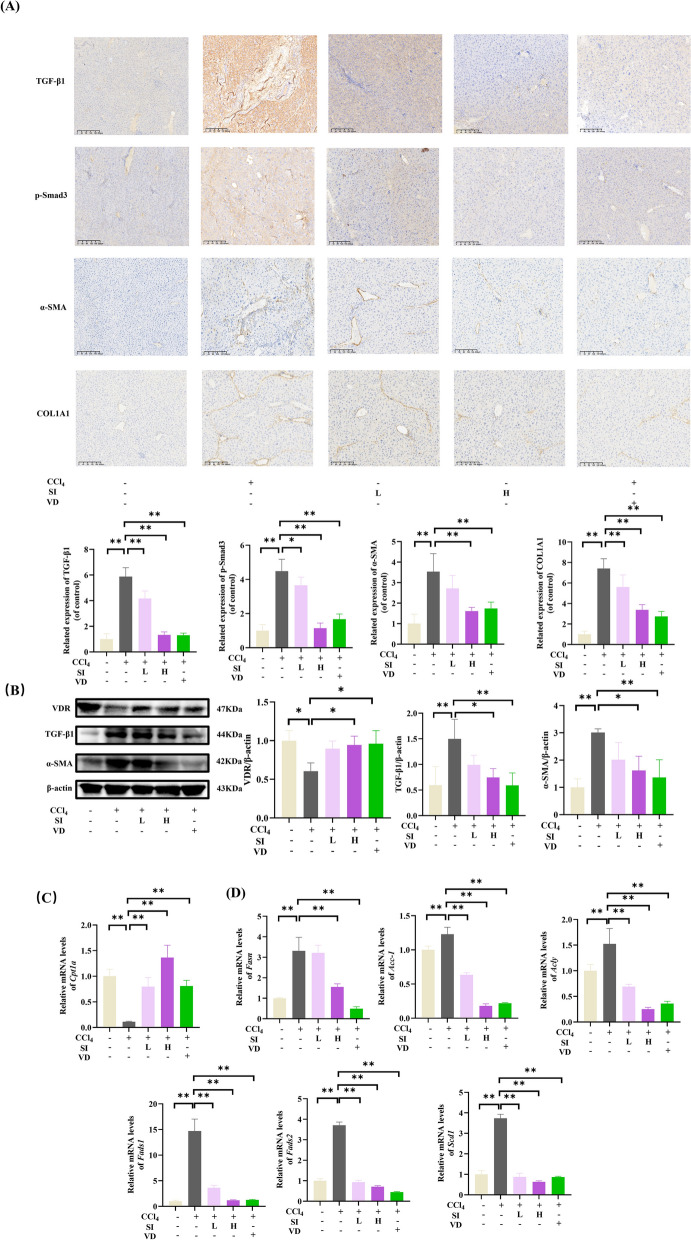
SI modulated fatty acid metabolism and ameliorated TGF-β/Smad3 pathway in liver.
Immunohistochemical analysis revealed that CCl4-induced elevations in hepatic TGF-β1, p-Smad3, α-SMA, and COL1 A1 protein levels were similarly suppressed by SI treatment in a dose-dependent manner (Fig. 5A). Western blot analysis confirmed the ability of SI to reverse CCl4-induced α-SMA overexpression (Fig. 5B). Notably, compared with control treatment, CCl4 administration significantly downregulated VDR expression, whereas SI and VD treatment restored VDR activation. This VDR activation correlated with SI-mediated metabolic reprogramming, as evidenced by reversal of CCl4-induced alterations in lipid metabolism enzymes: decreased Cpt1a (Fig. 5C) and increased Fasn, Fads1, Fads2, Acc-1, Acly and Scd1 levels (Fig. 5D) were normalized following SI intervention.
Fig. 5.
SI regulated VDR and fatty acid metabolism to suppress TGFβ/Smad pathway in CCl4-induced mice. A Immunohistochemistry analysis of TGF-β1, p-Smad3, α-SMA, COL1 A1. scale bar = 200 μm. (n = 5). B Western blot analysis of VDR, TGF-β1, and α-SMA. C mRNA levels of Cpt1a. D mRNA levels of lipogenic enzymes (Fasn, Acc-1, and Acly) and fatty acid desaturases (Fads1/2 and Scd1) (n = 6) *P < 0.05, **P < 0.01
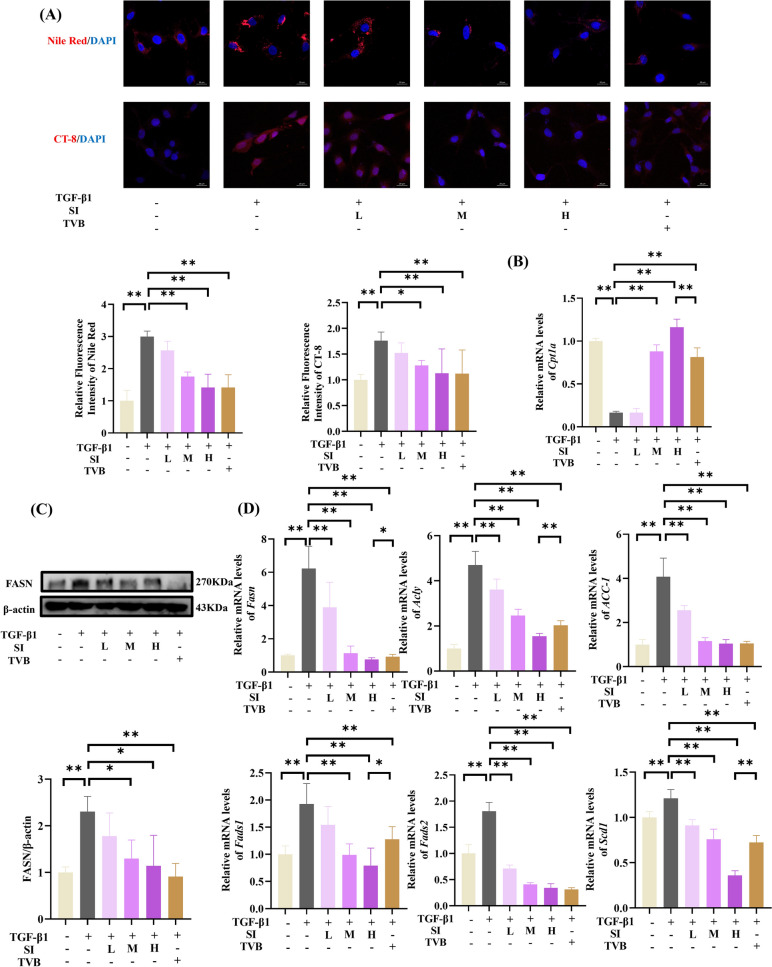
SI regulated lipid metabolism reprogramming in TGF-β1-induced HSCs
Aberrant fatty acid metabolism contributes to hepatic lipid accumulation and fibrosis progression. We investigated SI's regulatory effects on lipid metabolism in TGF-β1-activated HSCs. Nile Red staining18 revealed that TGF-β1 exposure induced intracellular lipid accumulation (Fig. 6A), which was attenuated by SI treatment. Compared with that of their TGF-β1-activated counterparts, the fluorescence intensity of SI-treated HSCs was significantly lower via complementary CT-8 quantification, confirming the capacity of SI to mitigate lipid droplet formation.
Fig. 6.
SI inhibited lipid accumulation through regulating fatty acid metabolism in TGF-β1-induced HSCs. A lipid droplet accumulation assessed by Nile Red and CT-8. scale bar = 20 μm. (n = 6). B Cpt1a expression. C Western blot analysis of FASN. (n = 3). D mRNA levels of lipogenic enzymes (Fasn, Acc-1, and Acly) and fatty acid desaturases (Fads1/2 and Scd1) (n = 6) *P < 0.05, **P < 0.01
Mechanistically, TGF-β1 activation downregulated Cpt1a (Fig. 6B), while upregulating FASN protein levels (Fig. 6C). SI treatment restored CPT1 A expression and normalized FASN overexpression. Transcriptomic analysis further showed TGF-β1-induced upregulation of lipogenic genes including Fads1, Fads2, Acc-1, Acly, and Scd1 (Fig. 6D), which were significantly suppressed by both SI and the reference inhibitor TVB.
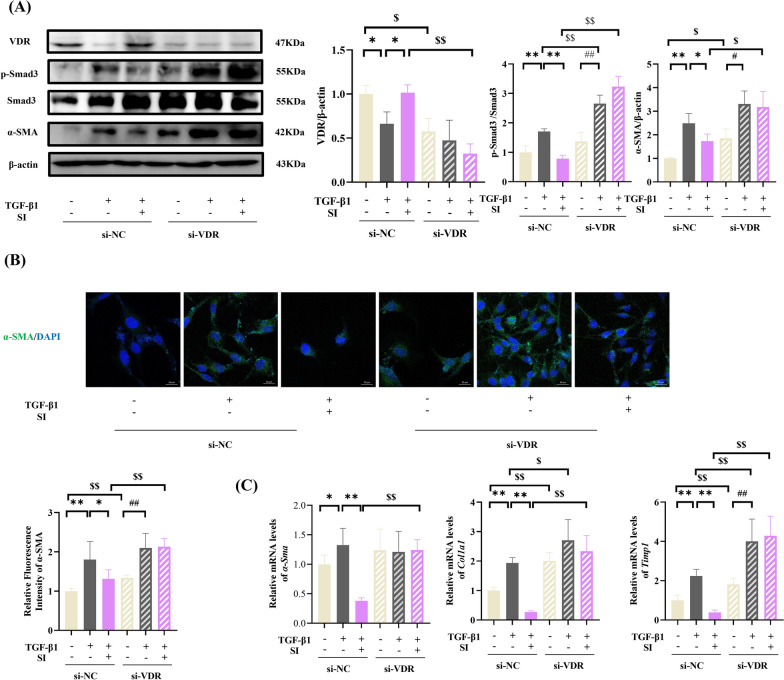
SI attenuates TGF-β1-induced HSCs activation through VDR pathway activation
Given the established role of the VDR in fatty acid metabolism regulation [48], we investigated the involvement of the VDR in the SI-mediated attenuation of hepatic fibrosis. The efficiency of siRNA-mediated VDR knockdown was confirmed in HSCs (Fig. 7A). SI treatment significantly upregulated VDR expression but suppressed Smad3 phosphorylation and α-SMA in TGF-β1-stimulated HSCs compared to TGF-β1-treated si-NC cells. Notably, VDR knockdown abolished SI's inhibitory effects on both Smad3 phosphorylation and α-SMA expression in TGF-β1-activated HSCs (Fig. 7B).
Fig. 7.
VDR knockout abolished the SI-induced inhibition of HSCs activation. A Western blot analysis (n = 3). B Immunofluorescence analysis of α-SMA. scale bar = 20 μm. (n = 6). C mRNA levels of fibrogenic markers (α-Sma, Col1a1, and Timp1). (n = 6) *P < 0.05, **P < 0.01 compared with si-NC + TGF-β1; $P < 0.05, $$P < 0.01 compared with si-NC; #P < 0.05, ##P < 0.01 compared with si-VDR + TGF-β1
Compared with those in the si-NC control cells, the mRNA levels of α-SMA, Col1 A1, and Timp1 in the si-VDR cells were increased (Fig. 7C). Importantly, SI failed to suppress TGF-β1-induced upregulation of these fibrogenic markers in VDR-deficient cells. These findings demonstrate that VDR activation is essential for the antifibrotic effects of SI, potentially through the modulation of Smad3 signaling and extracellular matrix regulation.
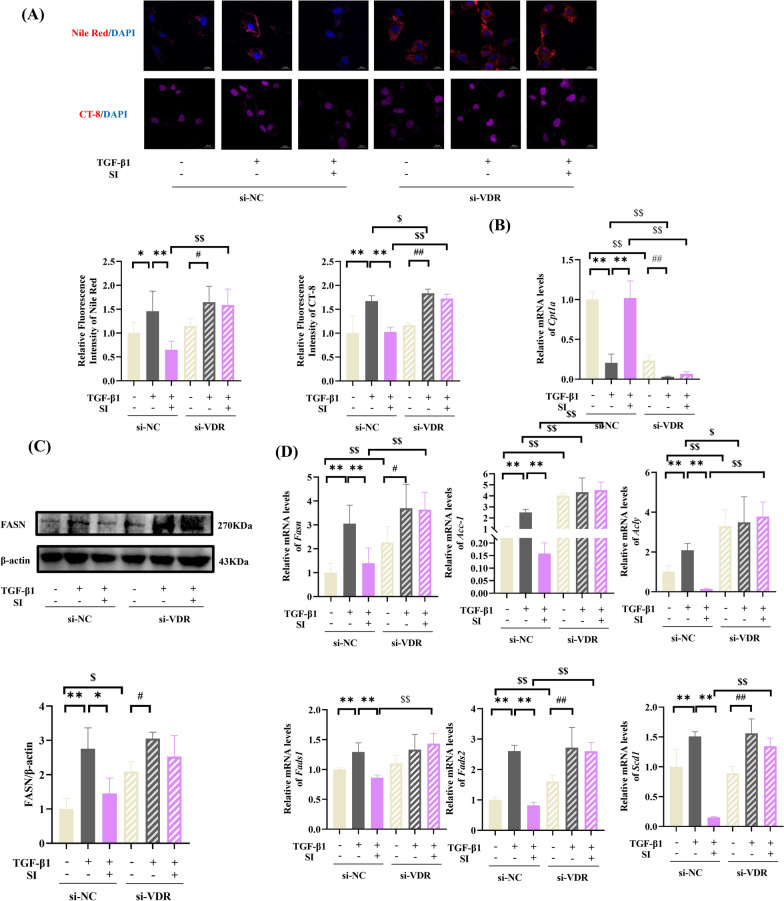
VDR silencing abrogates SI-mediated lipid metabolic reprogramming in HSCs
To investigate the role of VDR in SI-induced lipid metabolic reprogramming, we quantified Nile Red and CT-8 fluorescence quantification and detected increased lipid deposition in si-VDR-treated HSCs compared with si-NC controls (Fig. 8A). TGF-β1 cotreatment exacerbated lipid accumulation in si-VDR-treated cells, but SI failed to mitigate this effect. Molecular analysis revealed significant downregulation of the β-oxidation marker Cpt1a (Fig. 8B) and upregulation of lipogenic regulators (Fasn, Fads1 Fads2, Acc-1, Acly, and Scd1) in VDR-deficient cells (Fig. 8C, D). Notably, TGF-β1 stimulation elevated the expression of lipogenic regulators in si-VDR HSCs independent of SI intervention.
Fig. 8.
SI reprogramed fatty acid metabolism through activating VDR in TGFβ1-induced HSCs. A Lipid droplet accumulation. scale bar = 20 μm. B Cpt1a expression. C Western blot analysis of FASN. (n = 3). D mRNA levels of lipogenic enzymes (Fasn, Acc-1, and Acly) and fatty acid desaturases (Fads1/2 and Scd1). (n = 6) *P < 0.05, **P < 0.01. *P < 0.05, **P < 0.01 compared with si-NC + TGF-β1; $P < 0.05, $$P < 0.01 compared with si-NC; #P < 0.05, ##P < 0.01 compared with si-VDR + TGF-β1
Discussion
HSC activation drives liver fibrosis via fatty acid metabolic reprogramming, with VDR identified as a key transcriptional regulator. This study demonstrated that VDR deletion exacerbated fibrosis, increased TGF-β1/Smad3 signaling and disrupted lipid homeostasis by upregulating lipogenic/desaturase enzymes while suppressing the β-oxidation gatekeeper. Pathological VDR downregulation in TGF-β1-activated HSCs and fibrotic tissues highlights disease-linked regulation. Calcitriol-activated VDR normalized metabolic enzyme expression, reduced lipid/collagen accumulation, and inhibited Smad3 phosphorylation. Senkyunolide I, a novel VDR agonist, rebalances metabolism by suppressing lipogenesis/desaturation and enhancing CPT1 A, and its anti-fibrotic effects strictly VDR-dependent. These findings validate VDR as a therapeutic target and SI as a dual-action anti-fibrotic agent via VDR signaling.
The activation of HSCs serves as the principal cellular mechanism driving liver fibrogenesis [24]. Notably, CCl4 induction primarily exerts fibrogenic effects on the liver without causing fibrosis in other organs such as the kidney. Utilizing a CCl4-induced murine fibrosis model, we demonstrate that CCl4 administration triggers significant lipidomic remodeling through upregulation of lipogenic enzymes, establishing a pro-fibrogenic lipid microenvironment that amplifies HSC activation [6, 19]. This process concurrently activates the TGF-β1/Smad3 signaling axis, creating a self-reinforcing feed-forward circuit between fibrogenic signaling and metabolic dysregulation. Our findings corroborate previous reports of CCl4-induced Smad3 phosphorylation in hepatic tissue and TGF-β1-mediated HSCs activation [33]. Pharmacological restoration of fatty acid metabolism enzymes effectively attenuated collagen deposition, suggesting potential therapeutic mechanisms through metabolic modulation [27]. The mechanistic interplay between lipid overload and TGF-β1/Smad3 activation26, 50 may involve cholesterol-facilitated TGF-β receptor complex assembly [20] and saturated fatty acid-induced SMAD3 phosphorylation [16]. Further investigation is required to elucidate causal relationships between lipid metabolic dysregulation and fibrogenesis. Our findings position metabolic regulation as a novel therapeutic axis capable of disrupting the pathological cycle of HSCs activation and fibrotic matrix production.
Downregulation of VDR represents a key pathological feature in chronic liver diseases [14]. Studies demonstrate that VDR deficiency promotes spontaneous chronic liver inflammation and hepatic steatosis, whereas VDR activation mitigates fatty acid and cholesterol accumulation in obesity-associated models [1]. Mechanistically, ligand-activated VDR suppresses HSCs activation by modulating ECM production [15, 29]. Our findings further reveal that VDR depletion enhances the transcriptional activity of lipid metabolic enzymes, driving lipid accumulation that subsequently triggers TGF-β1 secretion and Smad3 phosphorylation. This aligns with prior studies demonstrating that vitamin D derivatives ameliorate hepatic fibrosis by inhibiting TGF-β/Smad signaling and HSC activation [7, 39]. Notably, VDR deficiency disrupts lipid homeostasis, elevating TC levels and promoting lipid deposition. Collectively, these data position VDR as a central regulator of lipid metabolism disorders that drive liver fibrosis progression.
The current clinical management of liver fibrosis primarily relies on antiviral nucleoside analogs combined with interferons, supplemented by anti-inflammatory/antioxidant agents like silymarin, ursodeoxycholic acid, and bicyclol [56]. Although vitamin D acts as a natural VDR agonist, its therapeutic application is constrained by hypercalcemia risks [45]. Rhizoma Chuanxiong (Ligusticum chuanxiong Hort.), a classical traditional Chinese medicine, has been documented for liver fibrosis treatment [41, 47]. Previous studies report that tetramethylpyrazine from Rhizoma Chuanxiong inhibits post-myocardial infarction cardiac fibrosis [53, 55]. Herein, SI, a principal bioactive component of Rhizoma Chuanxiong, exhibits potent anti-fibrotic efficacy in preclinical models. Through in vivo and in vitro fibrosis models, we demonstrate that SI inhibits HSCs activation and reduces ECM deposition. Mechanistically, SI selectively activates VDR to initiate fatty acid metabolic reprogramming, effectively mitigating lipid accumulation in HSCs and reversing fibrogenic activation via suppression of TGF-β/Smad3 signaling. Crucially, SI achieves VDR agonism without elevating serum calcium levels. This work elucidates the role of SI in modulating metabolic reprogramming and identifies this bioactive compound as a calcium-tolerant therapeutic candidate for liver fibrosis treatment.
Conclusion
This study elucidates the critical role of VDR in hepatic fibrogenesis, particularly its novel mechanism of attenuating liver fibrosis through modulation of fatty acid metabolism in HSCs. Our findings demonstrate that VDR deficiency induces dysregulation of fatty acid metabolism, thereby activating the TGF-β/Smad signaling pathway to promote HSCs activation and excessive collagen deposition. Notably, SI intervention effectively reverses fibrotic progression by targeting VDR-mediated suppression of TGF-β signaling. This research not only reveals a previously unrecognized metabolic regulatory mechanism of VDR in liver fibrosis but also offers novel therapeutic avenues for combating hepatic fibrosis through metabolic reprogramming strategies.
Supplementary Information
Acknowledgements
This research was funded by the National Natural Science Foundation of China (82374012), the Zhejiang Provincial Natural Science Foundation of China (LTGC24H280004), the Zhejiang Provincial Medicine Foundation (GZY-ZJ-KJ-23006, 2024ZR001, 2024ZR007, 2024 KY867), and Zhejiang Provincial Department of Science and Technology Research Institute Support Program (KJTYSZX2025, YSZX2401).
Abbreviations
- HSCs
RHuman hepatic stellate cells
- α-SMA
α-smooth muscle actin
- COL1A1
Collagen type I alpha 1
- TIMP1
Tissue inhibitor of metalloproteinase 1
- ALP
Alkaline phosphatase
- AST
Aspartate aminotransferase
- ALT
Alanine aminotransferase
- TBIL
Total bilirubin
- ACC1
Acetyl-CoA carboxylase 1
- ACLY
ATP-citrate lyase
- FADS1/2
Fatty acid desaturase
- FASN
Fatty acid synthase
- SCD1
Stearoyl-CoA desaturase 1
- CPT1A
Carnitine palmitoyltransferase 1A
- TGF-β1
Transforming growth factor-β1
- p-Smad3
Phosphorylated Smad3
- ECM
Excessive extracellular matrix
- SI
Senkyunolide I
- SLB
Silybin
- TVB
TVB3664
- VD
Vitamin D
- VDR
Vitamin D receptor
- CCl4
Carbon tetrachloride
- CETSA
Cellular thermal shift assay
- HE
Hematoxylin – eosin
- DMEM
Dulbecco’s modified eagle medium
- FBS
Fetal bovine serum
- WT
Wild-type
- i.p.
Intraperitoneal
- VDR-/-
VDR-knockout
- qRT-PCR
Quantitative reverse transcription polymerase chain reaction
- SD
Standard deviation
Author contributions
M. Zhu: Investigation, Writing—Original Draft. L. Ren: Validation. W. Xiao: Formal Analysis. L. Wang: Methodology. Z. Hu: Resources, Project Administration. N. Wang: Conceptualization, Funding Acquisition, Writing—review and editing.
Author agreement
We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us.
Funding
National Natural Science Foundation of China, 82374012, Nani Wang, Zhejiang Provincial Natural Science Foundation of China, LTGC24H280004, Nani Wang, Zhejiang Provincial Medicine Foundation, GZY-ZJ-KJ-23006, Nani Wang, 2024ZR001, Nani Wang, 2024ZR007, Nani Wang, 2024 KY867, Nani Wang, Zhejiang Provincial Department of Science and Technology Research Institute Support Program, KJTYSZX2025, Nani Wang, YSZX2401, Nani Wang.
Availability of data and materials
Data will be made available on request.
Declarations
Ethics approval and consent to participate
All animal procedures were approved by the Animal Ethics Committee of Zhejiang Academy of Traditional Chinese Medicine, Hangzhou, China (Approval No. [2021] 001) and followed the guidelines of the Basel Declaration.
Consent for publication
The author gives our consent for the publication of identifiable details, which can include data and content to be published in the above Journal and Article.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Mengyao Zhu, Lu Ren contributed equally to this work.
Contributor Information
Zhiming Hu, Email: hzm4199@163.com.
Nani Wang, Email: wnn8511@163.com.
References
- 1.Abdelrahman BA, El-Khatib AS, Attia YM. Insights into the role of vitamin D in targeting the culprits of non-alcoholic fatty liver disease. Life Sci. 2023;332: 122124. 10.1016/j.lfs.2023.122124. [DOI] [PubMed] [Google Scholar]
- 2.Aleknavičiūtė-Valienė G, Banys V. Clinical importance of laboratory biomarkers in liver fibrosis. Biochem Med (Zagreb). 2022;32(3): 030501. 10.11613/BM.2022.030501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bangen JM, Hammerich L, Sonntag R, et al. Targeting CCl4-induced liver fibrosis by RNA interference-mediated inhibition of cyclin E1 in mice. Hepatology. 2017;66(4):1242–57. 10.1002/hep.29275. [DOI] [PubMed] [Google Scholar]
- 4.Baugh EH, Lyskov S, Weitzner BD, et al. Real-time PyMOL visualization for Rosetta and PyRosetta. PLoS ONE. 2011;6(8): e21931. 10.1371/journal.pone.0021931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bai YM, Yang F, Luo P, et al. Single-cell transcriptomic dissection of the cellular and molecular events underlying the triclosan-induced liver fibrosis in mice. Mil Med Res. 2023;10(1):7. 10.1186/s40779-023-00441-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bates J, Vijayakumar A, Ghoshal S, et al. Acetyl-CoA carboxylase inhibition disrupts metabolic reprogramming during hepatic stellate cell activation. J Hepatol. 2020;73(4):896–905. 10.1016/j.jhep.2020.04.037. [DOI] [PubMed] [Google Scholar]
- 7.Beilfuss A, Sowa JP, Sydor S, et al. Vitamin D counteracts fibrogenic TGF-β signaling in human hepatic stellate cells both receptor-dependently and independently. Gut. 2015;64(5):791–9. 10.1136/gutjnl-2014-307024. [DOI] [PubMed] [Google Scholar]
- 8.Buakaew W, Krobthong S, Yingchutrakul Y, et al. Investigating the antifibrotic effects of β-citronellol on a TGF-β1-stimulated LX-2 hepatic stellate cell model. Biomolecules. 2024;14(7):800. 10.3390/biom14070800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen Y, Lin L, Yang C, et al. Ginsenoside AD-2 ameliorating lipopolysaccharide-induced activation in HSC-T6 cells and carbon tetrachloride-induced hepatic fibrosis in mice via regulation of VD-VDR axis. J Agric Food Chem. 2023. 10.1021/acs.jafc.2c06804. [DOI] [PubMed] [Google Scholar]
- 10.Chen Y, Xing Z, Chen J, et al. SIRT1 activation by Ligustrazine ameliorates migraine via the paracrine interaction of microglia and neurons. Phytomedicine. 2024;135: 156069. 10.1016/j.phymed.2024.156069. [DOI] [PubMed] [Google Scholar]
- 11.Ding J, Xu M, Du W, et al. Myeloid-specific blockade of Notch signaling ameliorates nonalcoholic fatty liver disease in mice. Int J Biol Sci. 2023;19(6):1941–54. 10.7150/ijbs.80122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dong B, Zhou Y, Wang W, et al. Vitamin D receptor activation in liver macrophages ameliorates hepatic inflammation, steatosis, and insulin resistance in mice. Hepatology. 2020;71(5):1559–74. 10.1002/hep.30937. [DOI] [PubMed] [Google Scholar]
- 13.Fondevila MF, Fernandez U, Heras V, et al. Inhibition of carnitine palmitoyltransferase 1A in hepatic stellate cells protects against fibrosis. J Hepatol. 2022;77(1):15–28. 10.1016/j.jhep.2022.02.003. [DOI] [PubMed] [Google Scholar]
- 14.Freeburg SH, Shwartz A, Kemény LV, et al. Hepatocyte vitamin D receptor functions as a nutrient sensor that regulates energy storage and tissue growth in zebrafish. Cell Rep. 2024;43(7): 114393. 10.1016/j.celrep.2024.114393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gao F, Guan C, Cheng N, et al. Design, synthesis, and anti-liver fibrosis activity of novel non-steroidal vitamin D receptor agonists based on open-ring steroid scaffold. Eur J Med Chem. 2025;286: 117250. 10.1016/j.ejmech.2025.117250. [DOI] [PubMed] [Google Scholar]
- 16.Gao T, Li J, Cheng T, et al. Ovarian cancer-derived TGF-β1 induces cancer-associated adipocytes formation by activating SMAD3/TRIB3 pathway to establish pre-metastatic niche. Cell Death Dis. 2024;15(12):930. 10.1038/s41419-024-07311-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Han X, Wang Z, Wang M, et al. Liver-targeting self-assembled hyaluronic acid-glycyrrhetinic acid micelles enhance hepato-protective effect of silybin after oral administration. Drug Deliv. 2016;23(5):1818–29. 10.3109/10717544.2015.1108374. [DOI] [PubMed] [Google Scholar]
- 18.Ioannou MS, Jackson J, Sheu SH, et al. Neuron-astrocyte metabolic coupling protects against activity-induced fatty acid toxicity. Cell. 2019;177(6):1522-1535.e14. 10.1016/j.cell.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 19.Jia H, Liu J, Fang T, et al. The role of altered lipid composition and distribution in liver fibrosis revealed by multimodal nonlinear optical microscopy. Sci Adv. 2023;9(2):eabq2937. 10.1126/sciadv.abq2937. [DOI] [PMC free article] [PubMed]
- 20.Jiao K, Yang K, Wang J, et al. 27-Hydroxycholesterol induces liver fibrosis via down-regulation of trimethylation of histone H3 at lysine 27 by activating oxidative stress; effect of nutrient interventions. Free Radic Biol Med. 2024;210:462–77. 10.1016/j.freeradbiomed.2023.11.043. [DOI] [PubMed] [Google Scholar]
- 21.Kumar V, Xin X, Ma J, et al. Therapeutic targets, novel drugs, and delivery systems for diabetes associated NAFLD and liver fibrosis. Adv Drug Deliv Rev. 2021;176: 113888. 10.1016/j.addr.2021.113888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kisseleva T, Brenner D. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat Rev Gastroenterol Hepatol. 2021;18(3):151–66. 10.1038/s41575-020-00372-7. [DOI] [PubMed] [Google Scholar]
- 23.Kusu H, Yoshida H, Kudo M, et al. Tomatidine reduces palmitate-induced lipid accumulation by activating AMPK via vitamin D receptor-mediated signaling in human HepG2 hepatocytes. Mol Nutr Food Res. 2019;63(22): e1801377. 10.1002/mnfr.201801377. [DOI] [PubMed] [Google Scholar]
- 24.Königshofer P, Brusilovskaya K, Petrenko O, et al. Nuclear receptors in liver fibrosis. Biochim Biophys Acta Mol Basis Dis. 2021;1867(12): 166235. 10.1016/j.bbadis.2021.166235. [DOI] [PubMed] [Google Scholar]
- 25.Lan T, Chen B, Hu X, et al. Tianhuang formula ameliorates liver fibrosis by inhibiting CCL2-CCR2 axis and MAPK/NF-κB signaling pathway. J Ethnopharmacol. 2024;321: 117516. 10.1016/j.jep.2023.117516. [DOI] [PubMed] [Google Scholar]
- 26.Lee JH, Kim KM, Jung EH, et al. Parkin-mediated mitophagy by TGF-β is connected with hepatic stellate cell activation. Int J Mol Sci. 2023;24(19):14826. 10.3390/ijms241914826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li L, Lei Q, Zhen Y, et al. Lactate dehydrogenase inhibition protects against hepatic fibrosis by regulating metabolic reprogramming of hepatic stellate cells. J Agric Food Chem. 2024;72(50):27953–64. 10.1021/acs.jafc.4c08211. [DOI] [PubMed] [Google Scholar]
- 28.Li P, Wang Y, Yu J, et al. Tenofovir vs entecavir among patients with HBV-related HCC after resection. JAMA Netw Open. 2023;6(10): e2340353. 10.1001/jamanetworkopen.2023.40353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu X, Wu Y, Li Y, et al. Vitamin D receptor (VDR) mediates the quiescence of activated hepatic stellate cells (aHSCs) by regulating M2 macrophage exosomal smooth muscle cell-associated protein 5 (SMAP-5). J Zhejiang Univ Sci B. 2023;24(3):248–61. 10.1631/jzus.B2200383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Li YC, Pirro AE, Amling M, et al. Targeted ablation of the vitamin D receptor: an animal model of vitamin D-dependent rickets type II with alopecia. Proc Natl Acad Sci USA. 1997;94(18):9831–5. 10.1073/pnas.94.18.9831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu YY, Yu LH, Zhang J, et al. Network pharmacology-based and molecular docking-based analysis of suanzaoren decoction for the treatment of parkinson’s disease with sleep disorder. Biomed Res Int. 2021;2021:1752570. 10.1155/2021/1752570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.O’Farrell M, Duke G, Crowley R, et al. FASN inhibition targets multiple drivers of NASH by reducing steatosis, inflammation and fibrosis in preclinical models. Sci Rep. 2022;12(1):15661. 10.1038/s41598-022-19459-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pan Q, Gao M, Kim D, et al. Hepatocyte FoxO1 deficiency protects from liver fibrosis via reducing inflammation and TGF-β1-mediated HSC activation. Cell Mol Gastroenterol Hepatol. 2024;17(1):41–58. 10.1016/j.jcmgh.2023.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ross TT, Crowley C, Kelly KL, et al. Acetyl-CoA carboxylase inhibition improves multiple dimensions of NASH pathogenesis in model systems. Cell Mol Gastroenterol Hepatol. 2020;10(4):829–51. 10.1016/j.jcmgh.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Salehpour A, Shidfar F, Hedayati M, et al. Molecular mechanisms of vitamin D plus Bisphenol A effects on adipogenesis in human adipose-derived mesenchymal stem cells. Diabetol Metab Syndr. 2021;13(1):41. 10.1186/s13098-021-00661-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shi L, Zhou L, Han M, et al. Calcitriol attenuates liver fibrosis through hepatitis C virus nonstructural protein 3-transactivated protein 1-mediated TGF β1/Smad3 and NF-κB signaling pathways. World J Gastroenterol. 2023; 29(18): 2798-2817 10.3748/wjg.v29.i18.2798. [DOI] [PMC free article] [PubMed]
- 37.Soleimani V, Delghandi PS, Moallem SA, et al. Safety and toxicity of silymarin, the major constituent of milk thistle extract: an updated review. Phytother Res. 2019;33(6):1627–38. 10.1002/ptr.6361. [DOI] [PubMed] [Google Scholar]
- 38.Song BJ, Rockey DC. Status of research on vitamin D supplementation in treating or preventing liver fibrosis. Liver Int. 2013;33(5):653–5. 10.1111/liv.12147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Song Y, Wei J, Li R, et al. Tyrosine kinase receptor B attenuates liver fibrosis by inhibiting TGF-β/SMAD signaling. Hepatology. 2023;78(5):1433–47. 10.1097/HEP.0000000000000319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tong G, Chen X, Lee J, et al. Fibroblast growth factor 18 attenuates liver fibrosis and HSCs activation via the SMO-LATS1-YAP pathway. Pharmacol Res. 2022;178: 106139. 10.1016/j.phrs.2022.106139. [DOI] [PubMed] [Google Scholar]
- 41.Wu X, Shu L, Zhang Z, et al. Adipocyte fatty acid binding protein promotes the onset and progression of liver fibrosis via mediating the crosstalk between liver sinusoidal endothelial cells and hepatic stellate cells. Adv Sci (Weinh). 2021;8(11): e2003721. 10.1002/advs.202003721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang XX, Jiang T, Shen Y, et al. Vitamin D receptor agonist doxercalciferol modulates dietary fat-induced renal disease and renal lipid metabolism. Am J Physiol Renal Physiol. 2011;300(3):F801–10. 10.1152/ajprenal.00338.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang M, Hayashi H, Horinokita I, et al. Neuroprotective effects of Senkyunolide I against glutamate-induced cells death by attenuating JNK/caspase-3 activation and apoptosis. Biomed Pharmacother. 2021;140: 111696. 10.1016/j.biopha.2021.111696. [DOI] [PubMed] [Google Scholar]
- 44.Xiang D, Zou J, Zhu X, et al. Physalin D attenuates hepatic stellate cell activation and liver fibrosis by blocking TGF-β/Smad and YAP signaling. Phytomedicine. 2020;78: 153294. 10.1016/j.phymed.2020.153294. [DOI] [PubMed] [Google Scholar]
- 45.Xing K, Wu Y, Gao F, et al. Design, synthesis and anti-hepatic fibrosis activity of novel diphenyl vitamin D receptor agonists. Eur J Med Chem. 2023;258: 115596. 10.1016/j.ejmech.2023.115596. [DOI] [PubMed] [Google Scholar]
- 46.Xu F, Zhang H, Chen J, et al. Recent progress on the application of compound formulas of traditional Chinese medicine in clinical trials and basic research in vivo for chronic liver disease. J Ethnopharmacol. 2024;321: 117514. 10.1016/j.jep.2023.117514. [DOI] [PubMed] [Google Scholar]
- 47.Wu JZ, Li YJ, Huang GR, et al. Mechanisms exploration of Angelicae Sinensis Radix and Ligusticum Chuanxiong Rhizoma herb-pair for liver fibrosis prevention based on network pharmacology and experimental pharmacologylogy. Chin J Nat Med. 2021;19(4):241–54. 10.1016/S1875-5364(21)60026-2. [DOI] [PubMed] [Google Scholar]
- 48.Xu Y, Lou Y, Kong J. VDR regulates energy metabolism by modulating remodeling in adipose tissue. Eur J Pharmacol. 2019;865: 172761. 10.1016/j.ejphar.2019.172761. [DOI] [PubMed] [Google Scholar]
- 49.Yang Q, Zhao ZZ, Xie J, et al. Senkyunolide I attenuates hepatic ischemia/reperfusion injury in mice via anti-oxidative, anti-inflammatory and anti-apoptotic pathways. Int Immunopharmacol. 2021;97: 107717. 10.1016/j.intimp.2021.107717. [DOI] [PubMed] [Google Scholar]
- 50.Yi Y, Lan X, Li Y, et al. Fatty acid synthesis and oxidation regulate human endoderm differentiation by mediating SMAD3 nuclear localization via acetylation. Dev Cell. 2023;58(18):1670-1687.e4. 10.1016/j.devcel.2023.07.005. [DOI] [PubMed] [Google Scholar]
- 51.Yin X, Peng J, Gu L, et al. Targeting glutamine metabolism in hepatic stellate cells alleviates liver fibrosis. Cell Death Dis. 2022;13(11):955. 10.1038/s41419-022-05409-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhao J, Zhou X, Chen B, et al. P53 promotes peroxisomal fatty acid β-oxidation to repress purine biosynthesis and mediate tumor suppression. Cell Death Dis. 2023;14(2):87. 10.1038/s41419-023-05625-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang K, Lin L, Zhu Y, et al. Saikosaponin d alleviates liver fibrosis by negatively regulating the ROS/NLRP3 inflammasome through activating the ERβ pathway. Front Pharmacol. 2022;13: 894981. 10.3389/fphar.2022.894981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhang H, Xia T, Xia Z, et al. KIF18A inactivates hepatic stellate cells and alleviates liver fibrosis through the TTC3/Akt/mTOR pathway. Cell Mol Life Sci. 2024;81(1):96. 10.1007/s00018-024-05114-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhang Y, Ma C, He L, et al. Tetramethylpyrazine protects endothelial injury and antithrombosis via antioxidant and antiapoptosis in HUVECs and Zebrafish. Oxid Med Cell Longev. 2022;2022:2232365. 10.1155/2022/2232365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zhao L, Tang H, Cheng Z. Pharmacotherapy of liver fibrosis and hepatitis: recent advances. Pharmaceuticals (Basel). 2024;17(12):1724. 10.3390/ph17121724. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be made available on request.