Abstract
Glioblastoma is the most common primary malignant tumor of the central nervous system, with a median survival of less than two years. While the etiology of glioblastoma is unclear, viral infection has emerged as a potential contributing factor. Cytomegalovirus (CMV) was first reported to be associated with glioblastoma in 2002. Since then, many studies have detected CMV in glioblastoma tissues suggesting it may plays a role in the glioblastoma progression. While there is no direct evidence confirmings CMV as an oncogenic virus, studies have demonstrated that CMV promotes glioblastoma development in cell and animal models, with several CMV-related genes implicated in tumorigenesis. Importantly, adjuvant CMV antiviral therapy has been proven to improve glioblastoma patient survival. This review focuses on clinical studies regarding the relationship between CMV and glioblastoma, the mechanism of CMV in tumorigenesis, advances in animal models of CMV-induced glioblastoma, and key directions for future investigations.
Keywords: Glioblastoma, Etiology, Cytomegalovirus, Mechanism, Progress
Introduction
Gliomas are the most common primary malignant tumors of the nervous system [1], with an annual incidence ranging from 4 to 11 per 100,000 individuals in developed countries [2]. The World Health Organization classifies gliomas into grades 1 to 4, with grades 3 and 4 categorized as high-grade gliomas [3]. Among gliomas, glioblastoma has the worst prognosis for patients with a median survival less than two years [4]. Current treatment options for glioblastoma include maximal surgical resection, radiotherapy, and chemotherapy [5]. Despite advances in therapeutic strategies, such as CAR T-cell therapy and immune checkpoint inhibitors, no major breakthroughs have yet been achieved [6, 7]. The onset of glioblastomas are often associated with risk factors, such as ionizing radiation, genetic mutations, chemical toxins, and pathogen infections including viral agents [2]. Yet, the contribution of pathogen infection to glioblastoma etiology remains unclear, particularly regarding whether such infections initiate or promote cellular mutations and/or contribute to tumorigenesis.
The role of pathogens in tumorigenesis, especially viruses, is well established [8, 9]. Common oncogenic viruses in humans include Epstein-Barr virus, hepatitis B virus (HBV), human papillomavirus (HPV), human T-cell lymphotropic virus, hepatitis C virus (HCV), Kaposi’s sarcoma herpesvirus, and Merkel cell polyomavirus [10]. Hepatitis B virus and HCV are linked to liver cancer [11, 12]. Human papillomavirus can cause cervical, anal, penile, and oropharyngeal cancers [13]. Epstein-Barr virus has been associated with Burkitt’s lymphoma, B-lymphoproliferative disorder, Hodgkin’s lymphoma, and nasopharyngeal carcinoma [14, 15]. Human T-cell lymphotropic virus is implicated in adult T-cell leukemia/lymphoma [16]. Merkel cell polyomavirus infection can result in Merkel cell carcinoma [17]. Kaposi’s sarcoma herpesvirus infection can lead to Kaposi’s sarcoma [18]. Over the past two decades, antigens and nucleic acids of several viruses have been detected in glioblastoma tissues, suggesting that viruses may be involved in glioblastoma development [19]. These viruses mainly include herpes virus, polyomavirus and human papillovirus [19]. Whether these viruses play a causative role in glioblastoma remains unclear. Of particular interest is human cytomegalovirus (CMV), a type 5 herpesvirus, which has garnered attention since its presence in glioblastoma tissues was reported in 2002 [20]. Since then, clinical samples and cell and animal models have been investigated to elucidate the role of CMV in glioblastoma development.
Human CMV infection is typically asymptomatic affecting up to 40–95% of the global population [10]. Numerous clinical studies have detected CMV nucleic acids or proteins in glioblastoma samples, suggesting that CMV may be involved in the initiation and/or progression of glioblastoma [1, 20]. Some studies have also reported that co-treatment with antiviral drugs such as valganciclovir showed improvement of overall patient survival [21–27]. While human CMV has been shown to promote tumor cell development and metastasis [28], the exact mechanism of CMV in glioblastoma remains to be elucidated. This review summarizes findings from epidemiological studies based on clinical samples, the role of CMV-targeted adjuvant therapy in glioblastoma in the past two decades, the tumor-promoting effect of CMV demonstrated in cell models and underlying progress in animal models of CMV-induced glioblastoma, and key areas for future research.
Clinical studies on the relationship between CMV and glioblastoma
Since the detection of CMV in glioblastoma tissues in 2002, the relationship between CMV and glioblastoma has garnered significant attention from clinicians and researchers [20]. Numerous investigations have been conducted to explore this association; however, the findings have been inconsistent [29]. While many studies have reported the presence of CMV proteins and nucleic acids in glioblastoma tissues [30–32], others have failed to detect any [33–35]. Several factors may be attributing to the inconsistency including CMV may not be present in all glioblastoma cases, the CMV genome may degrade over time in paraffin-embedded specimens, and the limitations of CMV detection techniques, which may necessitate the selection of suitable antibodies or optimization of detection methods [20, 36]. Some comprehensive reviews and meta-analyses have reported that CMV nucleic acids and proteins are detectable in over 63% of patients with cancer [1]. Although glioblastoma and CMV are closely related, most current studies have focused on detecting viral proteins or nucleic acids. However, viral proteins and nucleic acids can only suggest the presence of infection but cannot prove viral replication and activity. Measuring viral load in clinical samples and detecting CMV in serum samples may further clarify the role of CMV activity in glioblastoma [37]. Some studies have reported high viral loads of CMV in clinical samples and performed viral isolation, confirming the presence of live viruses in glioblastoma tissues [38, 39]. These isolated viruses were used to construct models that provided preliminary evidence of a relationship between CMV and glioblastoma [40]. Future clinical research should incorporate large-scale detection of viral proteins, nucleic acids, and viral load, followed by viral isolation and model validation to determine whether CMV plays a causative role in glioblastoma.
Findings from CMV-targeted adjuvant therapies also support a potential association between CMV and glioblastoma. For example, patients with low-grade CMV infection have longer overall survival than those with high-grade infection [41], suggesting that control CMV infection could be a novel therapeutic target for patients with glioblastoma. In 2013, Cecilia Söderberg-Nauclér et al. published a retrospective study involving 50 patients with glioblastoma who received valganciclovir as an adjuvant to standard therapy. The patients demonstrated significantly higher survival rates than the matched controls with similar disease stages, surgical resection grade, and baseline treatment. Patients who received valganciclovir treatment beyond six months achieved a two-year survival rate of 90% and a median overall survival of 56.4 months [23]. Giuseppe Stragliotto, et al. reported that valganciclovir treatment, when added to standard therapy, improves the outcomes of newly diagnosed glioblastoma patients [25, 42]. They further demonstrated the positive effect of valganciclovir in secondary glioblastoma. Eight patients treated with valganciclovir showed a significant increased median overall survival after progression to secondary glioblastoma compared with controls (19.1 versus 12.7 months) [26]. In another study, the addition of valganciclovir to bevacizumab slightly improved median overall survival in patients with recurrent glioblastoma compared with those receiving bevacizumab alone [24]. Patients who received valganciclovir as an add-on to their second- or third-line therapy after recurrence also exhibited significantly improved survival rates than the controls [27]. A recent meta-analysis of five randomized controlled trials with over 600 patients demonstrated that ganciclovir significantly improved glioblastoma 2-year and 4-year survival rate by 20% [43]. This warrant further case studies or large-scale clinical trials to clarify the relationship between CMV and glioblastoma, as well as the underlying mechanisms.
While antiviral therapies provide evidence for CMV’s involvement in glioblastoma and improved patient prognosis [44], it remains unclear whether CMV promotes glioblastoma development or acts as a causative agent. In patients with glioblastoma, radiotherapy and chemotherapy may reactivate latent CMV infection [45, 46]. Valganciclovir therapy may be effective in treating encephalitis symptoms and modifying the clinical course of the disease, thereby potentially influencing prognosis. Once reactivated, CMV may affect tumor aggressiveness and promote recurrence by influencing multiple cancer hallmarks. Isolating and characterizing CMV strains from patients will be critical to determining the direct role of CMV strains in brain cancer.
Mechanism of CMV in tumor development
Currently, most evidence indicates that CMV promotes tumor development; however, it has not been conclusively proven to be an oncogenic virus [47]. Cytomegalovirus can promote tumor development through multiple mechanisms (Fig. 1), including inhibiting tumor suppressor genes expression, promoting angiogenesis, enhancing tumor stem cell stemness, modifying the tumor microenvironment, promoting epithelial-mesenchymal transition (EMT), and inhibiting apoptosis (Table 1). Cytomegalovirus-infected glioma cancer stem cells can produce CMV interleukin-10 (IL-10), which can induce monocytes expressing immune suppressor B7H1 and secreting vascular endothelial growth factor, transforming growth factor-beta (TGF-β), and CMV immediate-early protein (IE), thereby facilitating glioblastoma invasion [48]. Cytomegalovirus infection rapidly activates receptor tyrosine kinase and AKT signaling pathways, promoting glioblastoma multiforme (GBM) cell invasiveness via focal adhesion kinase (FAK) activation [49]. CMV can also upregulate stemness regulators such as SOX2, phosphorylated-STAT3 (p-STAT3), and BMX in glioma stem-like cells (GSC), thereby promoting their survival, stemness, and proliferation [50]. Cytomegalovirus infected GBM tumor cells leads to NF-κB activation and the subsequent upregulation of the proto-oncogene c-MET, which promotes GBM growth [51]. Other studies indicate that CMV contributes to glioma progression by upregulating endocan, IL-6, and arginase-2 [52, 53]. Cytomegalovirus infection also increases ATF5 expression and the Bcl-2/BAX ratio, blocking apoptosis in glioblastoma cells [54, 55]. Cytomegalovirus infection also induces EMT and enhances cell migration and invasion in glioma cells by activating the RIP2/NF-κB signaling pathway, downregulating the expression of the epithelial cell marker (E-cadherin), and upregulating the expression of mesenchymal cell markers (N-cadherin and vimentin) [56, 57]. Cytomegalovirus-encoded miRNAs has also shown to promote glioma. For example, miR-UL112-3p regulates GBM pathophysiological processes by suppressing tumor suppressor candidate 3 expression [58], while miRNA CMV70-3P increases GBM cancer stem cell stemness by upregulating cellular SOX2 [59]. In a mouse model, CMV induced PDGFD expression, thereby increasing pericyte recruitment and angiogenesis [60].
Fig. 1.
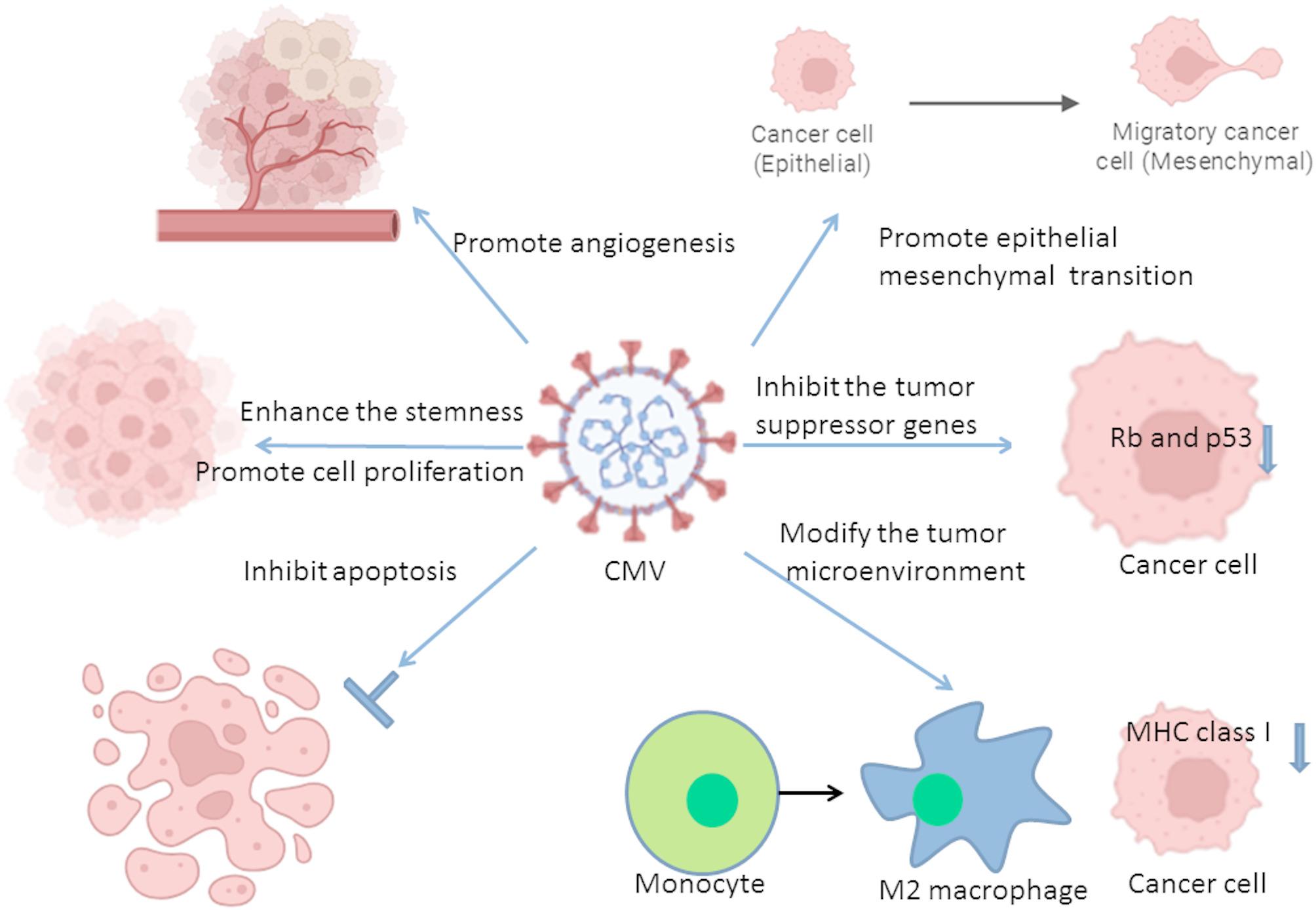
Schematic diagram of the mechanisms by which cytomegalovirus promotes glioblastoma growth. CMV: Cytomegalovirus, MHC: Major histocompatibility complex. Created in BioRender. Wu, X. (2025) https://BioRender.com/t8sq5xn
Table 1.
Mechanisms by which cytomegalovirus and its products contribute to glioblastoma development
| Virus or viral products | Target gene or signaling pathway | Effect on host | Reference |
|---|---|---|---|
| CMV IL-10 | Upregulation of VEGF and TGF-β |
Enhances migration of gCSCs |
[48] |
| CMV | Activation of PI3K/AKT, PLCγ, and FAK pathways | Increases migration of glioma cell | [49] |
| CMV | Upregulation of SOX2, p-STAT3. and BMX |
Promotes survival, stemness, and proliferation of GSC |
[50] |
| CMV | Activation of NF-κB signaling and c-MET | Promotes growth of GBM cells in vitro and in vivo | [51] |
| CMV | Upregulation of Endocan, IL-6, and arginase-2 | Contributes to glioma progression | [52, 53] |
| CMV | Upregulation of ATF5 and Bcl-2 | Inhibits apoptosis in | [54, 55] |
| GBM cell | |||
| CMV |
Activation of RIP2/ NF-κB, N-cadherin, and vimentin Downregulation of E-cadherin |
Induces EMT and enhances cell migration and invasion in glioma cells | [56, 57] |
| CMV-miR-UL112-3p | Inhibits tumor suppressor candidate 3 gene expression | Promotes cell proliferation, clone formation, migration, and invasion | [58] |
| miRNA CMV70-3P |
Increases expression of cellular SOX2 |
Increases GBM CSC stemness | [59] |
| CMV | Induces the PDGFD expression | Promotes murine GBM growth and angiogenesis | [60] |
| IE-72 | Increases hTERT promoter activity | Cellular immortalization and transformation | [64] |
| IE1 | Increases Sox2 and Nestin | Promotes stemness properties in GBM in vitro and in vivo | [65] |
| IE1 |
Reduction of Rb and p53 family proteins and induction of PI3K/AKT |
Induces cellular proliferation |
[66] |
| IE1 | Downregulation of GFAP, TSP-1, and p53 | Promotes the development of glioma | [67] |
| IE2 | Suppresses GFAP | Increases glioma cell malignancy | [68] |
| IE72 and IE86 | Degradation of connexin 43 and disrupt gap junction communication | Promotes the invasiveness of glioma cell | [69] |
| IE86 | Upregulating heterogeneous nuclear ribonucleoprotein A2B1 | Promotes migration of GBM cells and inhibits the apoptosis of GBM cells | [70, 71] |
| pp71 | Activation of NF-κB signaling and upregulation of SCF | Contributes to the aggressive phenotype of glioma | [72, 73] |
| pp71 | Downregulation of MHC class I proteins | Promotes immune suppression | [74] |
| gB | Binds to PDGFRA and activation of PI3-K/AKT | Promotes glioma cell invasion | [75, 76] |
| U28 | Activation of NF-κB and IL-6-JAK1-STAT3 | Promotes GBM cell proliferation | [77] |
| U28 | Upregulation VEGF, p-STAT3, and e-NOS | Accelerates GBM cell growth and invasion | [78]–[81] |
| U28 | Activation of S1P signaling and STAT3, AKT, and cMYC | Stimulates proliferation and survival of GBM cell | [82] |
| U28 | Activation of HIF-1α and pyruvate kinase M2 | Increased cell proliferation and metabolic reprogramming | [83] |
| US33 | Activation of STAT3 and cAMP-responsive element | Aggravates GBM tumor growth | [84, 85] |
| CMV | Increases phosphorylated STAT3 | Promotes glioma progression in a mut3 mouse model | [86] |
CMV: Cytomegalovirus, VEGF: Vascular endothelial growth factor, TGF-β: Transforming growth factor-β, gCSCs: Glioma cancer stem cells, PI3K/AKT: Phosphatidylinositol-3 kinase/Protein Kinase B, PLCγ: Phospholipase Cγ, FAK: Focal adhesion kinase, SOX2: Sex determining region Y-box 2, p-STAT3: Phosphorylated signal transduction and activators of transcription-3, BMX: Bone marrow X-linked kinase, GSC: Glioma stem-like cells, NF-κB: Nuclear factor kappa-B, GBM: glioblastoma, c-MET: Cellular-mesenchymal epithelial transition factor, ATF5:Activating transcription factor 5, Bcl-2: B-cell lymphoma-2, RIP2: Receptor-interacting protein 2, EMT: Epithelial–mesenchymal transition, PDGFD: Platelet Derived Growth Factor D, hTERT: human telomerase reverse transcriptase, Rb: retinoblastoma, GFAP: glial fibrillary acidic protein, TSP-1: thrombospondin-1, SCF: stem cell factor, MHC: Major histocompatibility complex, gB: glycoprotein B, PDGFRA: platelet derived growth factor receptor alpha, e-NOS: endothelial nitric oxide synthase, S1P: sphingosine-1-phosphate, cMYC: Cellular myelocytomatosis oncogene homolog, cAMP: cyclic adenosine monophosphate, mut3 mouse model: GFAP-cre; Nf1loxP/+; Trp53−/+ genetic mouse model
Cytomegalovirus gene products have been implicated in cell cycle dysregulation, apoptosis inhibition, enhanced cell migration and invasion, and angiogenesis [61, 62]. Specific viral gene products may influence key oncogenic signaling pathways involved in glioma progression [63]. Direct CMV infection increases glioblastoma cell migration and hTERT levels. Cytomegalovirus IE-72 may drive mitogenesis and cellular immortalization via hTERT upregulation [64]. Cytomegalovirus IE1 promotes GBM stemness, cell cycle progression, and survival by increasing SOX2 and Nestin levels [65]. Upon PI3-K/AKT pathway activation, the CMV IE1 gene product can activate Rb and p53 tumor-suppressor proteins in some GBM cells [66]. CMV IE1 gene product can also downregulate glial fibrillary acidic protein and thrombospondin-1 [67]. Cytomegalovirus IE2 has also been found to suppress glial fibrillary acidic protein expression, which declines with increasing glioma malignancy [68]. Moreover, CMV IE72 and IE86 can cause Cx43 degradation, resulting in the disruption of gap junctional intercellular communication (GJIC) in glioblastomas. GJIC degradation, which occurs at an earlier stage of tumor development, contributes to gliomagenesis [69]. Furthermore, CMV IE86 promotes GBM cell migration and inhibits apoptosis by upregulating heterogeneous nuclear ribonucleoprotein A2B1 [70, 71].
Cytomegalovirus pp71, encoded by the UL82 gene, can activate the NF-κB signaling pathway and an important angiogenic pathway. In adult neural progenitor cells and glioma cells, pp71 expression induces stem cell factor (SCF) expression in an NF-κB-dependent manner. In vivo, pp71 expression in human GBM cells may contribute to endothelial cell migration and angiogenesis, features typically associated with an aggressive tumor phenotype [72, 73]. Additionally, pp71 expression in GBM cells promotes immune suppression by reducing cell-surface expression of MHC class I proteins [74]. Cytomegalovirus glycoprotein B (gB) directly binds to PDGFRA and induces downstream activation of the oncogenic PI3-K/AKT pathway, thereby promoting glioma cell invasion [75, 76].
US28 is a constitutively active G-protein-coupled receptor that upregulates multiple oncogenic signaling pathways, including the JAK/STAT3 pathway. A study demonstrated that US28 activates the IL-6-JAK1-STAT3 signaling axis via NF-κB activation, which leads to IL-6 production [77]. US28 expression induces COX-2 expression through NF-κB activation, thereby driving the production of VEGF [78]. In GBM cells, human CMV infection or US28 overexpression is sufficient to promote the secretion of biologically active VEGF and activate multiple cellular kinases that facilitate glioma growth and invasion, such as p-STAT3 and endothelial nitric oxide synthase [79–81]. Another study demonstrated that US28 stimulates glioblastoma cell proliferation and survival by initiating S1P signaling, resulting in the concurrent activation of STAT3, AKT, and c-MYC [82]. Moreover, US28 can mediate HIF-1α and pyruvate kinase M2 expression, which drives cell proliferation, angiogenesis, and metabolic reprogramming [83]. US33, which is a viral G protein-coupled receptor, contributes to CMV-mediated STAT3 activation and can aggravate glioblastoma tumor growth in orthotopic glioblastoma xenograft models [84, 85].
Cytomegalovirus has shown to facilitate glioblastoma progression in a genetically engineered mouse model [86] (Fig. 2). Perinatal infection of these mut3 mice with mouse CMV led to a significant reduction in survival rates due to accelerated tumorigenesis. Mouse CMV infection increased p-STAT3 levels in neural stem cells prior to glioma onset [86]. However, current studies are limited to tumor cells and animal models and thus only demonstrate that CMV infection promotes tumor development [87].
Fig. 2.
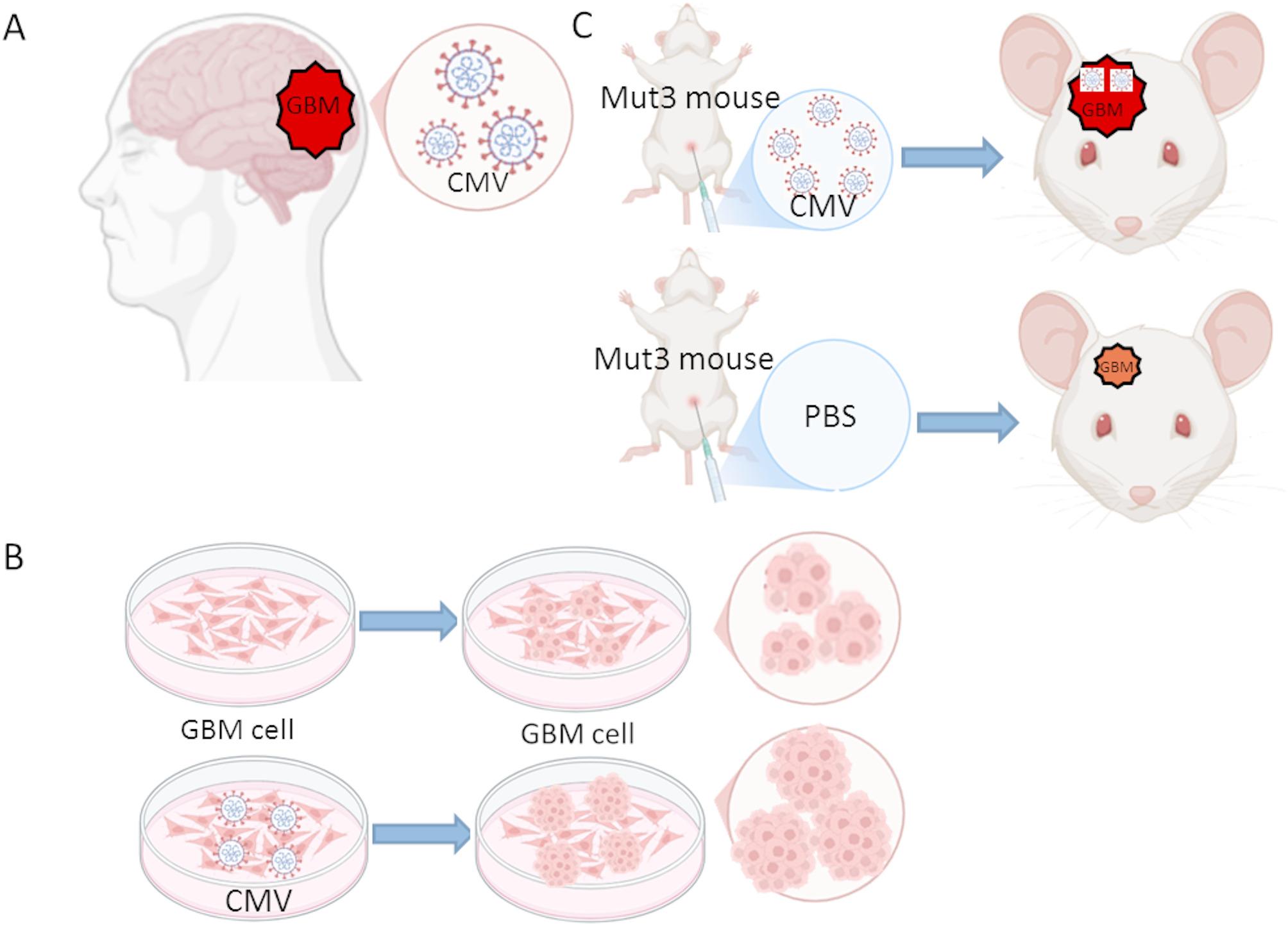
Several lines of evidence support the role of cytomegalovirus in promoting glioblastoma development. A: Cytomegalovirus is detected in the tumors of patients with glioblastoma. B: CMV promotes glioblastoma progression in cell culture models. C: CMV promotes glioblastoma progression in a genetically engineered mouse model. Mut3 mouse: GFAP-cre; Nf1loxP/+; Trp53−/+ genetic mouse. CMV: Cytomegalovirus, GBM: Glioblastoma multiforme. Created in BioRender. Wu, X. (2025) https://BioRender.com/qqzub37
Animal model for CMV-induced glioblastoma
To demonstrate the role of CMV in glioblastoma, the virus should first be isolated. Even within the same CMV species, different strains exhibit different characteristics [38]. Ideally, CMV should be isolated directly from glioblastoma cells, as this will be required for subsequent experiments. However, only one research team has succeeded in its isolation [38]. To validate the oncogenic potential of CMV, the virus should exhibit transforming potential in human cells in vitro and induce tumors in animal models, and this tumorigenesis should be preventable by viral neutralization. Georges Herbein et al. have made significant contributions promoting this research. They have isolated three CMV clinical strains from glioblastoma tissues. The strains can transform primary human astrocytes into CMV-elicited glioblastoma cells (CEGBCs). When xenografted into mice, the CEGBCs formed glioblastoma-like tumors [40]. Cytomegalovirus nucleic acids and proteins were detected in CMV-derived xenografts for up to two months post-engraftment, further indicating a potential link between CMV persistence and tumor progression. Future studies should characterize the differences between CEGBCs, primary glioblastoma cell lines, and glioblastoma tissues at the gene level and evaluate whether complete transformation from human astrocytes to glioma cells is possible.
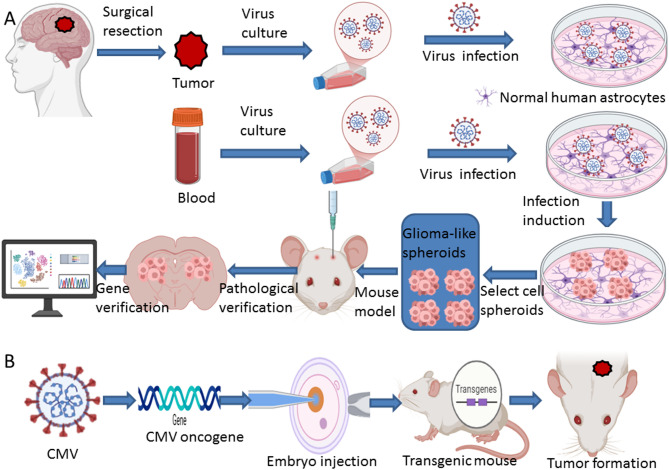
To date, no CMV oncogene has been definitively linked to glioblastoma, highlighting the need for further investigations. For example, HBV-transgenic mice expressing HBV pre-S, S, and X proteins spontaneously developed liver cancer during their lifespan [88]. Similarly, HPV E6 and E7 oncogenes are critical to tumorigenesis and have been validated in murine models [89]. To establish CMV as a causal agent in glioblastoma, it is essential to identify the key genes and develop murine tumor models that recapitulate their oncogenic activities (Fig. 3).
Fig. 3.
Schematic of an animal model for cytomegalovirus-induced glioblastoma under ideal conditions. A: Schematic of cytomegalovirus culture and in vivo and in vitro induction of glioblastoma diagram. B: Diagram of the construction of a cytomegalovirus transgenic mouse model. CMV: Cytomegalovirus. Created in BioRender. Wu, X. (2025) https://BioRender.com/bv3nsvz
Key directions for future investigations
Based on current clinical studies and in vitro and animal studies, CMV demonstrates a robust association with glioblastoma development. While CMV has been demonstrated to promote glioblastoma progression, definitive evidence linking CMV to glioblastoma etiology remains lacking, necessitating future investigations [90–92]. Establishing causality requires transitioning from correlative evidence to mechanistic validation, including the development of stable animal models and the identification of viral oncogenes [93]. Primarily, clinical research should include detailed patient data and pay more attention to tumor characteristics (primary versus recurrent), treatment history (chemotherapy and/or radiotherapy), and signs of infection or encephalitis. Such information is crucial for evaluating the role of CMV in tumor initiation and/or progression. Neuro-oncologists should continue investigate the clinical relationship between CMV and glioblastoma and the prognostic significance of antiviral therapy. Importantly, virologists and neuro-oncologists need to collaborate more to form research consortia to facilitate the collection of fresh or frozen glioblastoma and blood samples to ensure the preservation of viral nucleic acids and viral activity. Virus isolation should be prioritized for samples with high viral titers. After isolation, virus identification and research should be conducted to clarify the characteristics of the virus and study the effects of the virus on cell proliferation and tumorigenicity. The isolated virus should also be used for in vivo models. Comparative analysis with patients’ tumor cell lines and tissues should be performed at the transcriptomic level to assess whether tumorigenesis has occurred. Furthermore, CMV vaccines should be developed and tested for their preventive effects against glioblastoma.
Conclusion
Glioblastoma remains a critical oncological challenge affecting human health. Despite advances in treatment modalities, median survival for glioblastoma remains under two years. There are two primary research directions: continued development of treatment strategies and investigation into the cause of glioblastoma. Over the past two decades, increasing evidence has implicated viral involvement, particularly CMV, as a potential driver of glioblastoma development. However, existing data primarily demonstrates that CMV promotes glioblastoma progression. Antiviral therapy as an adjunct therapy for glioblastoma may extend survival and improve quality of life. Additionally, CMV-targeted immunotherapies, such as vaccines, have great prospects for future research.
To date, no direct evidence confirms that CMV can induce glioblastoma. However, CMV has been isolated from glioblastoma tissues, and preliminary in vivo models have been established. Future efforts should prioritize viral isolation and characterization from clinical samples, followed by mechanistic validation using cellular and animal models. With continued efforts, the role of CMV in glioblastoma development may be elucidated.
Acknowledgements
We thank Home for Researchers editorial team (www.home-for-researchers.com) for language editing service.
Author contributions
LZ and XW designed the research. XW and ZZ drafted the manuscript. LZ, XW and DJ Daniels contributed to the critical revision of the manuscript. All authors contributed to the manuscript and approved the submitted version.
Funding
This work was supported by the National Natural Science Foundation Youth Project (82300004) and National Key R&D Program of China (2024YFC2309904 and 2024YFC3506600). The funding sources had no role in the study design; in the writing of the report; or in the decision to submit the paper for publication.
Data availability
All data or related information supporting the conclusions of the review is included in the article.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
We have got the publication licenses for Figs. 1, 2 and 3 from BioRender.
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiaoxin Wu and Zhengyu Zhang are the co-first authors.
Contributor Information
Xiaoxin Wu, Email: xiaoxinwu@zju.edu.cn.
David J Daniels, Email: Daniels.David@mayo.edu.
References
- 1.Farias KPRA, Moreli ML, Floriano VG, da Costa VG. Evidence based on a meta-analysis of human cytomegalovirus infection in glioma. Arch Virol. 2019;164(5):1249–57. [DOI] [PubMed] [Google Scholar]
- 2.Ohgaki H, Kleihues P. Epidemiology and etiology of gliomas. Acta Neuropathol. 2005;109(1):93–108. [DOI] [PubMed] [Google Scholar]
- 3.Wang H, Tang F, Bian E, Zhang Y, Ji X, Yang Z, et al. IFITM3/STAT3 axis promotes glioma cells invasion and is modulated by TGF-β. Mol Biol Rep. 2019;47(1):433–41. [DOI] [PubMed] [Google Scholar]
- 4.Polley M-YC, Lamborn KR, Chang SM, Butowski N, Clarke JL, Prados M. Conditional probability of survival in patients with newly diagnosed glioblastoma. J Clin Oncol. 2011;29(31):4175–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. Radiotherapy plus concomitant and adjuvant Temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–96. [DOI] [PubMed] [Google Scholar]
- 6.Bagley SJ, Binder ZA, Lamrani L, Marinari E, Desai AS, Nasrallah MP, et al. Repeated peripheral infusions of anti-EGFRvIII CAR T cells in combination with pembrolizumab show no efficacy in glioblastoma: a phase 1 trial. Nat Cancer. 2024;5(3):517–31. [DOI] [PubMed] [Google Scholar]
- 7.Badani A, Ozair A, Khasraw M, Woodworth GF, Tiwari P, Ahluwalia MS, et al. Immune checkpoint inhibitors for glioblastoma: emerging science, clinical advances, and future directions. J Neurooncol. 2024;171(3):531–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Martel C, Georges D, Bray F, Ferlay J, Clifford GM. Global burden of cancer attributable to infections in 2018: a worldwide incidence analysis. Lancet Global Health. 2020;8(2):e180–90. [DOI] [PubMed] [Google Scholar]
- 9.White MK, Pagano JS, Khalili K. Viruses and human cancers: a long road of discovery of molecular paradigms. Clin Microbiol Rev. 2014;27(3):463–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Herbein G. High-Risk oncogenic human cytomegalovirus. Viruses. 2022;14(11):2462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yu DY, Moon HB, Son JK, Jeong S, Yu SL, Yoon H, et al. Incidence of hepatocellular carcinoma in Transgenic mice expressing the hepatitis B virus X-protein. J Hepatol. 1999;31(1):123–32. [DOI] [PubMed] [Google Scholar]
- 12.Lerat H, Honda M, Beard MR, Loesch K, Sun J, Yang Y, et al. Steatosis and liver cancer in Transgenic mice expressing the structural and nonstructural proteins of hepatitis C virus. Gastroenterology. 2002;122(2):352–65. [DOI] [PubMed] [Google Scholar]
- 13.Arbyn M, de Sanjosé S, Saraiya M, Sideri M, Palefsky J, Lacey C, et al. EUROGIN 2011 roadmap on prevention and treatment of HPV-related disease. Int J Cancer. 2012;131(9):1969–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ko Y-H. EBV and human cancer. Exp Mol Med. 2015;47(1):e130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Klein E, Kis LL, Klein G. Epstein–Barr virus infection in humans: from harmless to life endangering virus–lymphocyte interactions. Oncogene. 2007;26(9):1297–305. [DOI] [PubMed] [Google Scholar]
- 16.Bangham CRM. HTLV-1 persistence and the oncogenesis of adult T cell leukemia/lymphoma. Blood. 2023;141(19):2299–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu W, MacDonald M, You J. Merkel cell polyomavirus infection and Merkel cell carcinoma. Curr Opin Virol. 2016;20:20–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mesri EA, Cesarman E, Boshoff C. Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer. 2010;10(10):707–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gunasegaran B, Ashley CL, Marsh-Wakefield F, Guillemin GJ, Heng B. Viruses in glioblastoma: an update on evidence and clinical trials. BJC Rep. 2024;2(1):33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cobbs CS, Harkins L, Samanta M, Gillespie GY, Bharara S, King PH, et al. Human cytomegalovirus infection and expression in human malignant glioma. Cancer Res. 2002;62(12):3347–50. [PubMed] [Google Scholar]
- 21.Stragliotto G, Rahbar A, Solberg NW, Lilja A, Taher C, Orrego A, et al. Effects of valganciclovir as an add-on therapy in patients with cytomegalovirus‐positive glioblastoma: A randomized, double‐blind, hypothesis‐generating study. Int J Cancer. 2013;133(5):1204–13. [DOI] [PubMed] [Google Scholar]
- 22.Söderberg-Naucler C, Peredo I, Rahbar A, Hansson F, Nordlund A, Stragliotto G. Use of Cox regression with treatment status as a time-dependent covariate to re-analyze survival benefit excludes immortal time bias effect in patients with glioblastoma who received prolonged adjuvant treatment with valganciclovir. Int J Cancer. 2014;135(1):248–9. [DOI] [PubMed] [Google Scholar]
- 23.Söderberg-Nauclér C, Rahbar A, Stragliotto G. Survival in patients with glioblastoma receiving valganciclovir. N Engl J Med. 2013;369(10):985–6. [DOI] [PubMed] [Google Scholar]
- 24.Peng C, Wang J, Tanksley JP, Mobley BC, Ayers GD, Moots PL, et al. Valganciclovir and bevacizumab for recurrent glioblastoma: A single-institution experience. Mol Clin Oncol. 2016;4(2):154–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stragliotto G, Pantalone MR, Rahbar A, Bartek J, Söderberg-Naucler C. Valganciclovir as Add-on to standard therapy in glioblastoma patients. Clin Cancer Res. 2020;26(15):4031–9. [DOI] [PubMed] [Google Scholar]
- 26.Stragliotto G, Pantalone MR, Rahbar A, Söderberg-Nauclér C. Valganciclovir as Add-On to standard therapy in secondary glioblastoma. Microorganisms. 2020;8(10):1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pantalone MR, Rahbar A, Söderberg-Naucler C, Stragliotto G. Valganciclovir as Add-on to Second-Line therapy in patients with recurrent glioblastoma. Cancers. 2022;14(8):1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Barami K. Oncomodulatory mechanisms of human cytomegalovirus in gliomas. J Clin Neurosci. 2010;17(7):819–23. [DOI] [PubMed] [Google Scholar]
- 29.Lawler SE. Cytomegalovirus and glioblastoma; controversies and opportunities. J Neurooncol. 2015;123(3):465–71. [DOI] [PubMed] [Google Scholar]
- 30.Scheurer ME, Bondy ML, Aldape KD, Albrecht T, El-Zein R. Detection of human cytomegalovirus in different histological types of gliomas. Acta Neuropathol. 2008;116(1):79–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nevels M, Stangherlin LM, Castro FLF, Medeiros RSS, Guerra JM, Kimura LM, et al. Human cytomegalovirus DNA quantification and gene expression in gliomas of different grades. PLoS ONE. 2016;11(7):e0159604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shamran HA, Kadhim HS, Hussain AR, Kareem A, Taub DD, Price RL, et al. Detection of human cytomegalovirus in different histopathological types of glioma in Iraqi patients. Biomed Res Int. 2015;2015:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Holdhoff M, Guner G, Rodriguez FJ, Hicks JL, Zheng Q, Forman MS, et al. Absence of cytomegalovirus in glioblastoma and other High-grade gliomas by Real-time PCR, immunohistochemistry, and in situ hybridization. Clin Cancer Res. 2017;23(12):3150–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tang KW, Hellstrand K, Larsson E. Absence of cytomegalovirus in high-coverage DNA sequencing of human glioblastoma multiforme. Int J Cancer. 2014;136(4):977–81. [DOI] [PubMed] [Google Scholar]
- 35.Yamashita Y, Ito Y, Isomura H, Takemura N, Okamoto A, Motomura K, et al. Lack of presence of the human cytomegalovirus in human glioblastoma. Mod Pathol. 2014;27(7):922–9. [DOI] [PubMed] [Google Scholar]
- 36.Ranganathan P, Clark PA, Kuo JS, Salamat MS, Kalejta RF. Significant association of multiple human cytomegalovirus genomic loci with glioblastoma multiforme samples. J Virol. 2012;86(2):854–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mitchell DA, Xie W, Schmittling R, Learn C, Friedman A, McLendon RE, et al. Sensitive detection of human cytomegalovirus in tumors and peripheral blood of patients diagnosed with glioblastoma. Neurooncology. 2008;10(1):10–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.El Baba R, Pasquereau S, Haidar Ahmad S, Monnien F, Abad M, Bibeau F, et al. EZH2-Myc driven glioblastoma elicited by cytomegalovirus infection of human astrocytes. Oncogene. 2023;42(24):2031–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bhattacharjee B, Renzette N, Kowalik TF. Genetic analysis of cytomegalovirus in malignant gliomas. J Virol. 2012;86(12):6815–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Guyon J, Haidar Ahmad S, El Baba R, Le Quang M, Bikfalvi A, Daubon T, et al. Generation of glioblastoma in mice engrafted with human cytomegalovirus-infected astrocytes. Cancer Gene Ther. 2024;31(7):1070–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rahbar A, Stragliotto G, Orrego A, Peredo I, Taher C, Willems J, et al. Low levels of human cytomegalovirus infection in glioblastoma multiforme associates with patient survival; -a case-control study. Herpesviridae. 2012;3(1):3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu CJ, Hu YW. Immortal time bias in retrospective analysis: is there a survival benefit in patients with glioblastoma who received prolonged treatment of adjuvant valganciclovir? Int J Cancer. 2014;135(1):250–1. [DOI] [PubMed] [Google Scholar]
- 43.Chang C-T, Chen H-H, Chuang C-C, Chang S-H, Hsiao N-W. Ganciclovir as a potential treatment for glioma: a systematic review and meta-analysis. J Neurooncol. 2023;165(3):399–411. [DOI] [PubMed] [Google Scholar]
- 44.Yang T, Liu D, Fang S, Ma W, Wang Y. Cytomegalovirus and glioblastoma: A review of the biological associations and therapeutic strategies. J Clin Med. 2022;11(17):5221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Goerig NL, Frey B, Korn K, Fleckenstein B, Überla K, Schmidt MA, et al. Frequent occurrence of therapeutically reversible CMV-associated encephalopathy during radiotherapy of the brain. Neurooncology. 2016;18(12):1664–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Okita Y, Narita Y, Miyakita Y, Ohno M, Nagai S, Shibui S. Management of cytomegalovirus infection in a patient with malignant glioma treated with Temozolomide and steroids. Intern Med. 2012;51(20):2967–71. [DOI] [PubMed] [Google Scholar]
- 47.Joseph GP, McDermott R, Baryshnikova MA, Cobbs CS, Ulasov IV. Cytomegalovirus as an oncomodulatory agent in the progression of glioma. Cancer Lett. 2017;384:79–85. [DOI] [PubMed] [Google Scholar]
- 48.Dziurzynski K, Wei J, Qiao W, Hatiboglu MA, Kong L-Y, Wu A, et al. Glioma-Associated cytomegalovirus mediates subversion of the monocyte lineage to a tumor propagating phenotype. Clin Cancer Res. 2011;17(14):4642–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cobbs CS, Soroceanu L, Denham S, Zhang W, Britt WJ, Pieper R, et al. Human cytomegalovirus induces cellular tyrosine kinase signaling and promotes glioma cell invasiveness. J Neurooncol. 2007;85(3):271–80. [DOI] [PubMed] [Google Scholar]
- 50.Nevels M, Fiallos E, Judkins J, Matlaf L, Prichard M, Dittmer D, et al. Human cytomegalovirus gene expression in Long-Term infected glioma stem cells. PLoS ONE. 2014;9(12):e116178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Krenzlin H, Zdioruk M, Nowicki MO, Finkelberg T, Keric N, Lemmermann N, et al. Cytomegalovirus infection of glioblastoma cells leads to NF-κB dependent upregulation of the c-MET oncogenic tyrosine kinase. Cancer Lett. 2021;513:26–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Xing Y, Wang Y, Wang S, Wang X, Fan D, Zhou D, et al. Human cytomegalovirus infection contributes to glioma disease progression via upregulating endocan expression. Translational Res. 2016;177:113–26. [DOI] [PubMed] [Google Scholar]
- 53.Costa H, Xu X, Overbeek G, Vasaikar S, Patro CPK, Kostopoulou ON, et al. Human cytomegalovirus May promote tumour progression by upregulating arginase-2. Oncotarget. 2016;7(30):47221–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang T, Qian D, Hu M, Li L, Zhang LI, Chen HAO, et al. Human cytomegalovirus inhibits apoptosis by regulating the activating transcription factor 5 signaling pathway in human malignant glioma cells. Oncol Lett. 2014;8(3):1051–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wang X, Hu M, Xing F, Wang M, Wang B, Qian D. Human cytomegalovirus infection promotes the stemness of U251 glioma cells. J Med Virol. 2017;89(5):878–86. [DOI] [PubMed] [Google Scholar]
- 56.Cui K, Wang X, Han C, Liu S, Hu Y. Mechanism of human Cytomegalovirus-Induced Epithelial-Mesenchymal transition in glioma cells via the upregulation of RIP2 expression. Biol Pharm Bull. 2023;46(11):1506–11. [DOI] [PubMed] [Google Scholar]
- 57.Zhu X, Hu B, Hu M, Qian D, Wang B. Human cytomegalovirus infection enhances invasiveness and migration of glioblastoma cells by epithelial-to-mesenchymal transition. Int J Clin Exp Pathol. 2020;13(10):2637–47. [PMC free article] [PubMed] [Google Scholar]
- 58.Liang Q, Wang K, Wang B, Cai Q. HCMV-encoded miR-UL112-3p promotes glioblastoma progression via tumour suppressor candidate 3. Sci Rep. 2017;7(1):44705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ulasov IV, Kaverina NV, Ghosh D, Baryshnikova MA, Kadagidze ZG, Karseladze AI, et al. CMV70-3P MiRNA contributes to the CMV mediated glioma stemness and represents a target for glioma experimental therapy. Oncotarget. 2017;8(16):25989–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Krenzlin H, Behera P, Lorenz V, Passaro C, Zdioruk M, Nowicki MO, et al. Cytomegalovirus promotes murine glioblastoma growth via pericyte recruitment and angiogenesis. J Clin Invest. 2019;129(4):1671–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.McFaline-Figueroa JR, Wen PY. The viral connection to glioblastoma. Curr Infect Dis Rep. 2017;19(2):5. [DOI] [PubMed] [Google Scholar]
- 62.Dziurzynski K, Chang SM, Heimberger AB, Kalejta RF, McGregor Dallas SR, Smit M, et al. Consensus on the role of human cytomegalovirus in glioblastoma. Neurooncology. 2012;14(3):246–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cobbs CS. Cytomegalovirus and brain tumor. Curr Opin Oncol. 2013;25(6):682–8. [DOI] [PubMed] [Google Scholar]
- 64.Straat K, Liu C, Rahbar A, Zhu Q, Liu L, Wolmer-Solberg N, et al. Activation of telomerase by human cytomegalovirus. JNCI J Natl Cancer Inst. 2009;101(7):488–97. [DOI] [PubMed] [Google Scholar]
- 65.Soroceanu L, Matlaf L, Khan S, Akhavan A, Singer E, Bezrookove V, et al. Cytomegalovirus Immediate-Early proteins promote stemness properties in glioblastoma. Cancer Res. 2015;75(15):3065–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cobbs CS, Soroceanu L, Denham S, Zhang W, Kraus MH. Modulation of oncogenic phenotype in human glioma cells by cytomegalovirus IE1–Mediated mitogenicity. Cancer Res. 2008;68(3):724–30. [DOI] [PubMed] [Google Scholar]
- 67.Lee K, Jeon K, Kim JM, Kim VN, Choi DH, Kim SU, et al. Downregulation of GFAP, TSP-1, and p53 in human glioblastoma cell line, U373MG, by IE1 protein from human cytomegalovirus. Glia. 2005;51(1):1–12. [DOI] [PubMed] [Google Scholar]
- 68.Koh K, Lee K, Ahn J-H, Kim S. Human cytomegalovirus infection downregulates the expression of glial fibrillary acidic protein in human glioblastoma U373MG cells: identification of viral genes and protein domains involved. J Gen Virol. 2009;90(4):954–62. [DOI] [PubMed] [Google Scholar]
- 69.Khan Z, Yaiw K-C, Wilhelmi V, Lam H, Rahbar A, Stragliotto G, et al. Human cytomegalovirus immediate early proteins promote degradation of connexin 43 and disrupt gap junction communication: implications for a role in gliomagenesis. Carcinogenesis. 2014;35(1):145–54. [DOI] [PubMed] [Google Scholar]
- 70.Liang S, Yu B, Qian D, Zhao R, Wang B, Hu M. Human cytomegalovirus ie2 affects the migration of glioblastoma by mediating the different splicing patterns of RON through HnRNP A2B1. NeuroReport. 2019;30(12):805–11. [DOI] [PubMed] [Google Scholar]
- 71.Rou Z, Ming H, Shuzhen L, Bin W, Bo Y, Guimei Y, et al. IE86 inhibits the apoptosis and promotes the cell proliferation of glioma cells via the HnRNP A2_B1-mediated alternative splicing of Bcl-x. Int J Clin Exp Pathol. 2019;12(7):2775–85. [PMC free article] [PubMed] [Google Scholar]
- 72.Nakano I, Matlaf LA, Harkins LE, Bezrookove V, Cobbs CS, Soroceanu L. Cytomegalovirus pp71 protein is expressed in human glioblastoma and promotes Pro-Angiogenic signaling by activation of stem cell factor. PLoS ONE. 2013;8(7):e68176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sun L, Hui A-M, Su Q, Vortmeyer A, Kotliarov Y, Pastorino S, et al. Neuronal and glioma-derived stem cell factor induces angiogenesis within the brain. Cancer Cell. 2006;9(4):287–300. [DOI] [PubMed] [Google Scholar]
- 74.Trgovcich J, Cebulla C, Zimmerman P, Sedmak DD. Human cytomegalovirus protein pp71 disrupts major histocompatibility complex class I cell surface expression. J Virol. 2006;80(2):951–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cobbs C, Khan S, Matlaf L, McAllister S, Zider A, Yount G, et al. HCMV glycoprotein B is expressed in primary glioblastomas and enhances growth and invasiveness via PDGFR-alpha activation. Oncotarget. 2014;5(4):1091–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Soroceanu L, Akhavan A, Cobbs CS. Platelet-derived growth factor-α receptor activation is required for human cytomegalovirus infection. Nature. 2008;455(7211):391–5. [DOI] [PubMed] [Google Scholar]
- 77.Slinger E, Maussang D, Schreiber A, Siderius M, Rahbar A, Fraile-Ramos A, et al. HCMV-encoded chemokine receptor US28 mediates proliferative signaling through the IL-6-STAT3 axis. Sci Signal. 2010;3(133):ra58. [DOI] [PubMed] [Google Scholar]
- 78.Maussang D, Langemeijer E, Fitzsimons CP, Stigter-van Walsum M, Dijkman R, Borg MK, et al. The human Cytomegalovirus–Encoded chemokine receptor US28 promotes angiogenesis and tumor formation via Cyclooxygenase-2. Cancer Res. 2009;69(7):2861–9. [DOI] [PubMed] [Google Scholar]
- 79.Soroceanu L, Matlaf L, Bezrookove V, Harkins L, Martinez R, Greene M, et al. Human cytomegalovirus US28 found in glioblastoma promotes an invasive and angiogenic phenotype. Cancer Res. 2011;71(21):6643–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Heukers R, Fan TS, de Wit RH, van Senten JR, De Groof TWM, Bebelman MP, et al. The constitutive activity of the virally encoded chemokine receptor US28 accelerates glioblastoma growth. Oncogene. 2018;37(30):4110–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Maussang D, Verzijl D, Walsum Mv, Leurs R, Holl J, Pleskoff O, et al. Human cytomegalovirusencoded chemokine receptor US28 promotes tumorigenesis. Proc Natl Acad Sci USA. 2006;103(35):13068–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bergkamp ND, van Senten JR, Brink HJ, Bebelman MP, den Jv B, Çobanoğlu TS, et al. A virally encoded GPCR drives glioblastoma through feed-forward activation of the SK1-S1P1 signaling axis. Sci Signal. 2023;16(798):eade6737. [DOI] [PubMed] [Google Scholar]
- 83.Wit RHd, Mujić-Delić A, Senten JRv, Fraile-Ramos A, Siderius M, Smit MJ. Human cytomegalovirus encoded chemokine receptor US28 activates the HIF-1α_PKM2 axis in glioblastoma cells. Oncotarget. 2016;7(42):67966–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.van Senten JR, Bebelman MP, Fan TS, Heukers R, Bergkamp ND, van Gasselt P, et al. The human cytomegalovirus-encoded G protein–coupled receptor UL33 exhibits oncomodulatory properties. J Biol Chem. 2019;294(44):16297–308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Casarosa P, Gruijthuijsen YK, Michel D, Beisser PS, Holl J, Fitzsimons CP, et al. Constitutive signaling of the human cytomegalovirus-encoded receptor UL33 differs from that of its rat cytomegalovirus homolog R33 by promiscuous activation of G proteins of the gq, gi, and Gs classes. J Biol Chem. 2003;278(50):50010–23. [DOI] [PubMed] [Google Scholar]
- 86.Price RL, Song J, Bingmer K, Kim TH, Yi J-Y, Nowicki MO, et al. Cytomegalovirus contributes to glioblastoma in the context of tumor suppressor mutations. Cancer Res. 2013;73(11):3441–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hollon TC, Price RL, Kwon C-H, Chiocca EA. Mutations in glioblastoma oncosuppressive pathways pave the way for oncomodulatory activity of cytomegalovirus. OncoImmunology. 2014;2(9):e25620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Geninatti Crich S, Cutrin JC, Lanzardo S, Conti L, Kálmán FK, Szabó I, et al. Mn-loaded apoferritin: a highly sensitive MRI imaging probe for the detection and characterization of hepatocarcinoma lesions in a Transgenic mouse model. Contrast Media Mol Imaging. 2012;7(3):281–8. [DOI] [PubMed] [Google Scholar]
- 89.Brake T, Lambert PF. Estrogen contributes to the onset, persistence, and malignant progression of cervical cancer in a human papillomavirus-transgenic mouse model. Proc Natl Acad Sci USA. 2005;102(7):2490–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Foster H, Ulasov IV, Cobbs CS. Human cytomegalovirus-mediated immunomodulation: effects on glioblastoma progression. Biochimica et biophysica acta (BBA) -. Reviews Cancer. 2017;1868(1):273–6. [DOI] [PubMed] [Google Scholar]
- 91.Michaelis M, Baumgarten P, Mittelbronn M, Driever PH, Doerr HW, Cinatl J. Oncomodulation by human cytomegalovirus: novel clinical findings open new roads. Med Microbiol Immunol. 2010;200(1):1–5. [DOI] [PubMed] [Google Scholar]
- 92.Soroceanu L, Cobbs CS. Is HCMV a tumor promoter? Virus Res. 2011;157(2):193–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Cobbs CS. Evolving evidence implicates cytomegalovirus as a promoter of malignant glioma pathogenesis. Herpesviridae. 2011;2(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data or related information supporting the conclusions of the review is included in the article.