Abstract
Background
Early defibrillation is the foundation of treatment of shockable ventricular arrhythmias (VF, pVT) but optimal energy doses for initial and subsequent shocks in paediatric cardiac arrest remain controversial.
Objectives
To assess the use of different energy doses for initial defibrillation in infants, children and adolescents with ventricular fibrillation (VF) or pulseless ventricular tachycardia (pVT) during cardiac arrest.
Methods
A systematic review was performed by the ILCOR Paediatric Life Support Task force. This systematic review was prospectively registered as PROSPERO CRD42024548898. A search of PubMed, EMBASE, and Cochrane Controlled Register of Trials (CENTRAL) was performed for clinical trials and observational studies, published before 1 January 2025, involving cardiac defibrillation in infants and children (excluding newborn infants) in cardiac arrest. Investigators reviewed studies for relevance, extracted data, and assessed risk of bias using the ROBINS-I framework. Critical outcomes included survival to hospital discharge and return of spontaneous circulation. Results were compiled into a Summary of Findings table using the GRADEpro Guideline Development tool. Statistical calculations and Forest plot generation were performed using RevMan.
Results
We identified 7 relevant observational studies. The majority of studies involved in-hospital cardiac arrest. The overall certainty of evidence was very low. Critical (survival to hospital discharge, return of spontaneous circulation) and important (termination of VF/pVT) outcomes were not significantly better or worse when initial defibrillation doses of <1.5 J/kg or >2.5 J/kg were used for children in cardiac arrest with a shockable rhythm compared with initial doses approximating 2 J/kg.
Conclusions
The current available data suggest that outcomes are not significantly better or worse when initial defibrillation doses of <1.5 J/kg or >2.5 J/kg are used for children in cardiac arrest with a shockable rhythm (VF or pVT) compared with initial doses approximating 2 J/kg. Well-designed randomised trials are needed to address this important question.
Keywords: Paediatric, Cardiac arrest, Defibrillation, Cardioversion, Dose
Introduction and background
Shockable ventricular arrhythmias (ventricular fibrillation and pulseless ventricular tachycardia) are less frequently recorded in paediatric cardiac arrest but are associated with a higher survival rate than non-shockable rhythms (asystole, pulseless electrical activity).1 Early defibrillation is the foundation of treatment but optimal energy doses for initial and subsequent shocks remain controversial.
Differences in the first shock dose recommended by International Liaison Committee on Resuscitation (ILCOR) member councils remain, with the European Resuscitation Council (ERC) and the Australian and New Zealand Committee on Resuscitation (ANZCOR) recommending 4 J/kg for the first and all subsequent shocks2, 3 and the American Heart Association (AHA) recommending an initial dose of 2–4 J/kg (for ease of teaching, a starting dose of 2 J/kg is used in algorithms and training materials)4 For persistent VF after initial shock, the AHA guidelines recommend increasing the defibrillation dose to 4 J/kg, suggesting that subsequent energy doses should be at least 4 J/kg and noting that higher levels may be considered, not to exceed 10 J/kg.
A systematic review5 failed to show a significant benefit of one dosing regimen over another but was hampered by small sample sizes and study heterogeneity. The more recent large paediatric in-hospital registry study6 provided support for a 2 J/kg dose for initial defibrillation but did not provide guidance for subsequent doses. Current ILCOR treatment recommendations7, 8 suggest the routine use of an initial dose of 2 to 4 J/kg of monophasic or biphasic defibrillation waveforms for infants or children in ventricular fibrillation (VF) or pulseless ventricular tachycardia (pVT). They recognised that there was insufficient evidence from which to base a recommendation for second and subsequent defibrillation dosages.
This systematic review aims to review all available evidence comparing different defibrillation energy doses in infants and children with shockable rhythms to provide data that may support or change the current recommendations.
Methods
This systematic review was commissioned by the International Liaison Committee on Resuscitation (ILCOR) Paediatric Life Support Task Force. The protocol for the systematic review was developed in accordance with the ILCOR framework and registered on PROSPERO (CRD42024548898).9 The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) abstract checklist and PRISMA checklist10 are included in the Supplemental Materials (S1 and S2). Study characteristics used as criteria for eligibility are outlined in the PICOS question in Table 1.
Table 1.
PICOS framework (Population, Intervention, Comparator, Outcome, Study Design).
| PICOS | Description |
|---|---|
| Population | Infants and children (excluding newborn children) who are in ventricular fibrillation (VF) or pulseless ventricular tachycardia (pVT) during out-of-hospital or in-hospital cardiac arrest |
| Intervention | Initial defibrillation dose approximating 2 J/kg (1.5–2.5 J/kg) |
| Comparison | Compared with initial defibrillation dose of > 2.5 J/kg, <1.5 J/kg or any other specified dose |
| Outcomes | Survival to hospital discharge and return of spontaneous circulation (ROSC) were ranked as critical outcomes. Termination of VF/pVT was ranked as an important outcome. |
| Study Design | Randomised controlled trials (RCTs) and non-randomised studies (non-randomised controlled trials, interrupted time series, controlled before-and-after studies, cohort studies) and case series with a minimum of 5 number of cases were eligible for inclusion. Unpublished studies (e.g., conference abstracts, trial protocols) were excluded. All relevant publications in any language were included as long as there was an English abstract. |
Information sources & study selection
A professional librarian searched the PubMed, EMBASE and Cochrane Controlled Register of Trials (CENTRAL) databases using a peer-reviewed search strategy. All studies published up until the initial search (February 15, 2024) were included for analysis. The search was further updated on January 1, 2025. In addition, the authors reviewed international clinical trials registries, bibliographies of published systematic reviews and of included studies, and review group members provided a list of known relevant studies. The complete search strategy is provided in the Supplemental Materials (S3).
Three authors (JA, JdC, LT) independently reviewed the titles and abstracts of all articles identified in the search (2 authors per title). Study selection and screening were performed using Covidence.11 Copies of relevant articles identified by any author were obtained. Two authors (JA, JdC) then reviewed the full texts and identified articles meeting inclusion criteria. Disagreements were resolved by discussion.
Data collection
Two authors (JA, JdC) separately extracted data from each included article to a Microsoft Excel spreadsheet. Discrepancies were resolved by discussion. When necessary, authors of included studies were contacted to clarify results. Characteristics of included studies (including study location and years conducted, arrest setting, participants, shock type and outcomes assessed) were extracted and compiled in a Summary of Findings table (Table 2). From each study, the number of children receiving an initial defibrillation dose <1.5 J/kg, a dose approximating 2 J/kg (1.5–2.5 J/kg) or a dose >2.5 J/kg and the number of patients achieving successful resuscitation outcomes from each intervention were extracted. In cases where case-level data could not be obtained from the publication or author, the study was not included in the meta-analysis and instead author-calculated adjusted odds ratios (aORs) for each intervention/outcome were provided.
Table 2.
Characteristics of included studies.
| Study | Years conducted | Setting | Location | Study Participants | Interventions Compared | Shock type | Outcomes Assessed |
|---|---|---|---|---|---|---|---|
| Gutgesell 1976 | pre1976 | IHCA | USA | n = 71 defibrillation attempts on 27 children (aged 3d-16 years) with VF at single specialist children's hospital | Defibrillation dose of 2 J/kg ± 10 J v <2 J/kg ± 10 J v >2 J/kg ± 10 J | Monophasic | Termination of VF |
| Tibballs 2006 | 1999–2002 | IHCA | Australia | n = 9 children (aged 0-21y) receiving DC shock for VF or pVT at single specialist paediatric hospital (PICU or ward) | No comparison | Monophasic | ROSC |
| Rodríguez-Núñez 2006 | 1998–1999 | OHCA & IHCA | Spain | n = 44 children (aged 1 m-16y) receiving DC shock for VF/pVT in cardiac arrest (22 OHCA, 22 IHCA) | Initial shock defibrillation dose of 2 J/kg (1–3 J/kg) v >2 J/kg | Monophasic | ROSC, Initial survival (ROSC >20 min), Final Survival (to 1 year) |
| Meaney 2011 | 2000–2008 | IHCA | USA | n = 285 events in 266 children (aged 0-18y) receiving DC shock for VF/pVT within multi-site collaborative | Initial shock defibrillation dose of 2 J/kg (1–3 J/kg) v 4 J/kg (>3 to 5 J/kg) | Mixed | Termination of VF/pVT, ROSC, Sustained ROSC > 20 min |
| Tibballs 2011 | 2005–2009 | IHCA | Australia | n = 48 patients (aged 0–20 years) receiving defibrillation shock for VF/pVT | Energy (for all shocks): 0.5-<1 J/kg; 1-<1.5 J/kg; 1.5-<2 J/kg; 2-<2.5 J/kg; 2.5-<3 J/kg; 3–5 J/kg. | Biphasic | ROSC |
| Rodríguez-Núñez 2014 | 2007–2009 | IHCA | Latin-American countries, Spain, Italy, Portugal | n = 40 events in 37 children (aged 1 m-18y) receiving DC shock for VF/pVT within multi-site collaborative | Initial shock defibrillation dose of < 2 J/kg v 2–4 J/kg v > 4 J/kg | Mixed | SHD, ROSC |
| Hoyme 2020 | 2000–2016 | IHCA | USA | n = 301 paediatric patients < 12 years (primary sample) and 422 paediatric patients < 18 years (entire cohort for secondary analysis) with an index cardiac arrest and an initial documented rhythm of VF/pVT | Defibrillation dose of 1.7–2.5 J/kg v < 1.7 J/kg v > 2.5 J/kg | Mixed | SHD, ROSC |
DC: direct current; IHCA: in-hospital cardiac arrest; OHCA: out-of-hospital cardiac arrest; pVT: pulseless ventricular tachycardia; ROSC: return of spontaneous circulation; SHD: survival to hospital discharge; VF: ventricular fibrillation.
Selection of outcomes
The ILCOR P-COSCA (Pediatric Core Outcome Set for Cardiac Arrest) initiative developed a core outcome set specific for paediatric cardiac arrest studies. The design and methods of the initiative included use of a Delphi process to develop consensus on a core domain set.12 The patient centred outcomes survival to hospital discharge (SHD) and return of spontaneous circulation (ROSC) were defined by the PLS TF as critical outcomes for this review. Termination of the shockable rhythm (return to an organised rhythm) was considered an important measurable outcome. Results of analyses for all three outcomes are presented.
Synthesis of results
Risk of bias for cohort studies was assessed at an individual study level using the ROBINS-I assessment tool for human studies.13
All results are presented as Risk Ratios (RR) and absolute Risk Difference (aRD) and our assessment of statistical significance is based on the absolute risk reduction 95% confidence interval (CI 95%). A random effects model was chosen for meta-analysis to better account for study heterogeneity. Summary effects were not reported if heterogeneity between studies was very high (I2 statistic greater than 75%). The Mantel-Haenszel method was used for dichotomous outcomes.
Results were compiled into a Summary of Findings table (Table 4) using the GRADEpro Guideline Development tool.14 Statistical calculations and Forest plot generation were performed using RevMan.15
Table 4.
Summary of Findings for Critical and Important Study Outcomes.
| Outcomes (Importance) | Participants (Studies), n | Certainty of Evidence (GRADE) | RR (95% CI) | ARD with Intervention |
|---|---|---|---|---|
| Defibrillation dose < 1.5 J/kg (I) compared to defibrillation dose approximating 2 J/kg (C) for defibrillation in children in cardiac arrest | ||||
| Termination of VF/pVT (important) |
265 (2 non-randomised studies) |
Very low | RR 0.63 (0.14 to 2.84) |
179 fewer per 1,000(from 415 fewer to 888 more) |
| ROSC (critical) |
266 (4 non-randomised studies) |
Very low | RR 1.06 (0.95 to 1.18) |
51 more per 1,000(from 42 fewer to 152 more) |
| Survival to Hospital Discharge (critical) | 225 (2 non-randomised studies) |
Very low | RR 1.06 (0.80 to 1.40) |
29 more per 1,000(from 96 fewer to 192 more) |
| Defibrillation dose > 2.5 J/kg (I) compared to defibrillation dose approximating 2 J/kg (C) for defibrillation in children in cardiac arrest | ||||
| Termination of VF/pVT (important) |
265 (2 non-randomised studies) |
Very low | RR 0.96 (0.82 to 1.13) |
22 fewer per 1,000(from 99 fewer to 77 more) |
| ROSC (critical) |
596 (6 non-randomised studies) |
Very low | RR 0.95 (0.77 to 1.17) |
29 fewer per 1,000(from 133 fewer to 98 more) |
| Survival to Hospital Discharge (critical) | 225 (2 non-randomised studies) |
Very low | RR 1.20 (0.38 to 3.77) |
82 more per 1,000(from 253 fewer to 1,000 more) |
ARD: absolute risk difference; C: comparator; CI: confidence interval; GRADE: Grading of Recommendations, Assessment, Development, and Evaluation
I: intervention; RCT: randomised controlled trial; RR: risk ratio; ROSC: return of spontaneous circulation; VF: ventricular fibrillation; pVT: pulseless ventricular tachycardia.
Certainty of results was evaluated using the GRADE framework. Inconsistency was considered serious if the I2 statistic was greater than 50%, and very serious if greater than 75%. Indirectness was considered serious if more than half the resuscitations in a study category were conducted prior to 2000 because of subsequent changes to standard resuscitation. Imprecision was considered serious if the CI for risk difference crossed two of the following points: +0.05 (appreciable benefit to the intervention), 0.00 (equipoise), and −0.05 (appreciable harm), and very serious if all three points were crossed.
Additional analyses
The following subgroup analyses were prespecified at the time of protocol development: out-of-hospital cardiac arrest (OHCA), in-hospital cardiac arrest (IHCA), and patients with primary (first recorded cardiac arrest rhythm) versus secondary (later appearing) shockable rhythm.
Results
Study selection
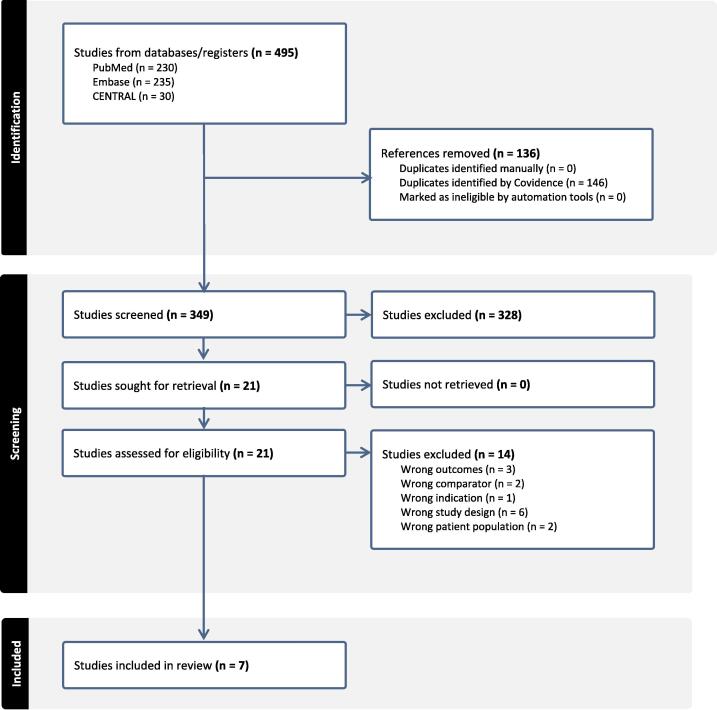
Results of each stage of the search are provided in Fig. 1. Taken together, the searches identified 349 non-duplicate articles, of which 7 met inclusion criteria.
Fig. 1.
PRISMA Diagram for systematic review.
Seven studies were included in the systematic review.6, 16, 17, 18, 19, 20, 21 None of these provided clinical trial data (no controlled trials). The identified studies were all observational studies and provided very low certainty evidence (downgraded for imprecision and risk of bias) for all outcomes. Two of the studies6, 17 reported adjusted analyses but in both cases adjusted odds ratios (aORs) were provided rather than raw data and for pooled interventions different to our intended groups (<1.5 J/kg, 1.5–2.5 J/kg, >2.5 J/kg). We chose to utilise unadjusted data from all studies rather than mix adjusted and unadjusted data and risk potential heterogeneity due to adjustment. Characteristics of these studies are described in Table 2.
Risk of bias within studies
The risk of bias in each study is presented in Table 3. Five studies were judged to have moderate overall risk of bias.6, 17, 18, 19, 21, 22 The other two studies were judged to have a serious risk of bias, because of concerns about confounding and selection.16, 20 Despite differences in study design and setting, there was little heterogeneity in study results (I2 statistic <50%). Small numbers of subjects in many studies and the low proportion of patients with good outcome contribute to imprecision in the estimate of treatment effect for most comparisons.
Table 3.
Risk of bias assessment using the ROBINS-I assessment tool for human studies.
| Study | Confounding | Selection of participants | Classification of interventions | Departure from intended interventions | Missing data | Measurement of outcomes | Selection of reported outcomes | Overall bias |
|---|---|---|---|---|---|---|---|---|
| Gutgesell 1976 | Serious | Serious | Moderate | Moderate | Moderate | Moderate | Serious | Serious |
| Hoyme 2020 | Low | Moderate | Low | Moderate | Moderate | Low | Moderate | Moderate |
| Meaney 2011 | Moderate | Moderate | Low | Moderate | Moderate | Low | Moderate | Moderate |
| Rodríguez-Núñez 2006 | Moderate | Moderate | Low | Moderate | Moderate | Low | Moderate | Moderate |
| Rodríguez-Núñez 2014 | Low | Moderate | Low | Moderate | Moderate | Low | Moderate | Moderate |
| Tibballs 2006 | Moderate | Low | Low | Moderate | Moderate | Low | Moderate | Moderate |
| Tibballs 2011 | Serious | Serious | Low | Moderate | No Information | Low | Serious | Serious |
Synthesis of results
GRADE Summary of Findings for the critical and important outcome comparisons are presented in Table 4. Forest plots for the critical and important outcome comparisons are presented in Fig. 2, Fig. 3.
Fig. 2.
Forest plots for defibrillation dose <1.5 J/kg compared to defibrillation dose approximating 2 J/kg. M−H: Cochran-Mantel-Haenszel. 95% CI: 95 percent confidence interval.
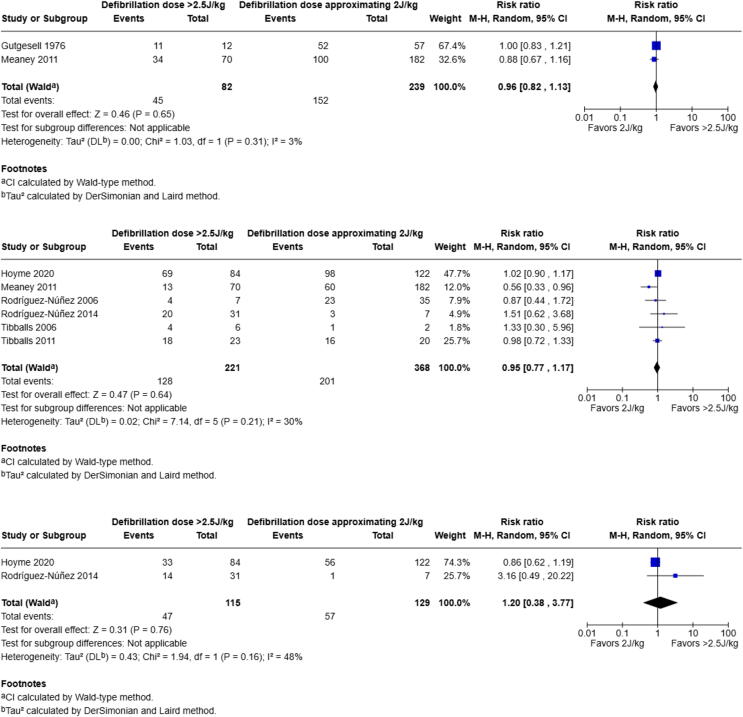
Fig. 3.
Forest plots for defibrillation dose >2.5 J/kg compared to defibrillation dose approximating 2 J/kg. M−H: Cochran-Mantel-Haenszel. 95% CI: 95 percent confidence interval.
Studies comparing defibrillation dose <1.5 J/kg to defibrillation dose approximating 2 J/kg
For the critical outcome of survival to hospital discharge, we identified very low certainty data from 2 cohort studies6, 18 involving 225 patients which showed no significant difference when comparing an initial defibrillation dose < 1.5 J/kg to that approximating 2 J/kg (29 more survivors per 1,000 resuscitations; CI 95%: 96 fewer to 192 more).
For the critical outcome of return of spontaneous circulation (ROSC), we identified very low certainty data from 4 cohort studies6, 18, 20, 21 involving 266 patients which showed no significant difference when comparing defibrillation dose <1.5 J/kg to doses approximating 2 J/kg (51 more survivors per 1,000 resuscitations; CI 95%: 42 fewer to 152 more).
Additional very low certainty evidence from two observational studies16, 17of 265 children found no significant effect on termination of VF/pVT associated with defibrillation dose <1.5 J/kg compared to that approximating 2 J/kg (179 fewer per 1,000; CI 95%: 415 fewer to 888 more).
Studies comparing defibrillation dose >2.5 J/kg to defibrillation dose approximating 2 J/kg
For the critical outcome of survival to hospital discharge, we identified very low certainty data from 2 cohort studies6, 18 involving 225 patients which showed no significant difference to SHD associated with defibrillation dose >2.5 J/kg compared to that approximating 2 J/kg (82 more survivors per 1,000 resuscitations; CI 95%: 253 fewer to 1000 more).
For the critical outcome of return of spontaneous circulation (ROSC), we identified very low certainty data from 6 cohort studies6, 17, 18, 19, 20, 21 involving 596 patients showing no significant difference to ROSC associated with defibrillation dose >2.5 J/kg compared to that approximating 2 J/kg (29 fewer survivors per 1,000 resuscitations; CI 95%: 133 fewer to 98 more).
Additional very low certainty evidence from two observational studies16, 17 including 265 children found no significant effect on termination of VF/pVT associated with defibrillation dose >2.5 J/kg compared to that approximating 2 J/kg (22 fewer per 1,000; CI 95%: 99 fewer to 77 more).
Overall summary of findings
Acknowledging the very low level of certainty, the current available data suggest that the critical (SHD, ROSC) and important (termination of VF/pVT) outcomes are not significantly better or worse when initial defibrillation doses of <1.5 J/kg or >2.5 J/kg are used for children in cardiac arrest with a shockable rhythm (VF or pVT) compared with initial doses approximating 2 J/kg.
Subgroup analyses
Of the 732 paediatric patients detailed in the included studies, only 22 received their defibrillation in an OHCA setting19 so a subgroup analysis comparing OHCA and IHCA was unable to be performed.
No data could be found to support the preplanned subgroup analyses on the effect of primary versus secondary shockable rhythms.
Discussion
Our systematic review of the current available evidence has shown that clinical outcomes are not significantly better or worse when initial defibrillation doses of <1.5 J/kg or >2.5 J/kg are used for children in cardiac arrest with a shockable rhythm (VF or pVT) compared with initial doses approximating 2 J/kg. This finding is consistent with adult data showing that initial defibrillation dose was not associated with outcome after OHCA.23
An ‘optimal’ paediatric defibrillation dose would be one that maximises the chance of reversion to a normal ‘perfusing’ rhythm whilst minimising the risk of myocardial damage (from a dose which is too high). Some animal models have demonstrated that “adult dose” biphasic defibrillators compared to “paediatric dose” shocks through attenuated biphasic defibrillators resulted in a greater frequency of myocardial damage and worse post-resuscitation myocardial function.24, 25 No specific toxic dose has been defined for paediatric defibrillation but multiple publications have reported safe and successful use of doses that are well above the currently recommended 2–4 J/kg.26
Limitations
The authors recognise that most studies in our review were conducted in sites where either 2 J/kg or 4 J/kg doses were recommended for initial defibrillation and that the observed variability of dosing was likely attributable to the limited energy dose settings on defibrillators. So, although no specific energy dose was found superior, energy selections would generally have been approximating either 2 or 4 J/kg.
Limitations to meta-analyses are well described. Mathematical combination of studies with differing designs, patient populations, and endpoint definitions is problematic, and small differences in analytic technique may change reported answers.
Most of the available outcome data was from studies that included shock types that were either all monophasic16, 19, 21 or a mix of monophasic and biphasic shocks.6, 17, 18 These findings may not be transferable to newer biphasic defibrillators. Biphasic defibrillation may be more effective and safer than monophasic defibrillation as it requires less energy to achieve the same defibrillation success rate, potentially causing less myocardial damage.27 As only one study19 included defibrillation shocks in an OHCA settings, generalisability of our findings to this setting may also be limited.
A recent study by the pediRES-Q collaborative showed that approximately 30% of defibrillation shocks delivered to children in in-hospital settings (self-reported as shockable rhythms) were actually not for shockable rhythms.28 This may represent a potential risk of bias (to NOT find a difference) if many of the shocks delivered (irrespective of dose) may not have been for shockable rhythms in the first place.
Implications and future research
Despite consistent ILCOR recommendations regarding paediatric defibrillation dosing over the last 20 years, advice from various resuscitation councils on this topic have diverged.29 In the absence of a clear benefit of either 2 J/kg or 4 J/kg as an energy dose for initial paediatric defibrillation attempts, some resuscitation councils have recommended one, some the other, and some a range (2–4 J/kg). It has been argued that these differences may provide a “natural experiment” allowing outcomes to be compared between the countries that suggest different doses. Such an approach would be limited by the potential for confounding bias.
Prehospital and in-hospital studies, ideally comparing existing different dosing strategies with planned subgroup analyses based on patient age and type of shockable rhythm (primary vs secondary) are ethical, necessary, and critically important to help guide clinicians in making these complex decisions. Examination of clinically important outcome variable, such as those defined in the P-COSCA (Pediatric Core Outcome Set for Cardiac Arrest)30 as well a exploration of specific undesirable effects (e.g. myocardial damage) of defibrillation associated with the different doses studied, would be of great utility to guideline writing groups. The rarity of shockable rhythms in paediatric cardiac arrest would necessitate a national or multinational approach to any comparative study to enable sufficient power.
Conclusions
Acknowledging the very low level of certainty, the current available data suggest that initial defibrillation doses of < 1.5 J/kg or > 2.5 J/kg are not associated with significantly better or worse outcomes compared with initial doses approximating 2 J/kg for children in cardiac arrest with a shockable rhythm (VF or pVT).
This review did not investigate the evidence for second and subsequent defibrillation dosages.
CRediT authorship contribution statement
Jason Acworth: Writing – review & editing, Writing – original draft, Visualization, Validation, Project administration, Methodology, Formal analysis, Data curation, Conceptualization. Jimena del Castillo: Writing – review & editing, Writing – original draft, Validation, Methodology, Data curation, Conceptualization. Lokesh Kumar Tiwari: Writing – review & editing, Conceptualization. Dianne Atkins: Writing – review & editing, Methodology, Conceptualization. Allan de Caen: Writing – review & editing, Methodology, Conceptualization. Arun Bansal: Writing – review & editing, Conceptualization. Thomaz Bittencourt Couto: Writing – review & editing, Conceptualization. Stephan Katzenschlager: Writing – review & editing, Conceptualization. Monica Kleinman: Writing – review & editing, Conceptualization. Jesus Lopez-Herce: Writing – review & editing, Conceptualization. Ryan W. Morgan: Writing – review & editing, Conceptualization. Michelle Myburgh: Writing – review & editing, Conceptualization. Vinay Nadkarni: Writing – review & editing, Conceptualization. Janice A. Tijssen: Writing – review & editing, Conceptualization. Barnaby R. Scholefield: Writing – review & editing, Supervision, Project administration, Conceptualization.
Funding
No funding. This Systematic Review was conducted through volunteer support from the International Liaison Committee On Resuscitation (ILCOR) which receives funding from the American Heart Association.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Monica Kleinman serves on the Editorial Board of the Resuscitation Journal. She was not involved in the editorial review or the decision to publish this article.
Acknowledgements
The authors extend their gratitude to Lars Eriksson (Library Support), and the members of the ILCOR Paediatric Life Support Task Force: Andrea Christoff, James Gray, Anne-Marie Guergerian, Hiroshi Kurosawa, Laurie J Morrison, Kee-Chong Ng, Gabrielle Nuthall, Yong-Kwang Gene Ong, Tia Raymond, Leandra Rech, Joseph Rossano, Steve Schexnayder, Ravi Thiagarajan, Alexis Topjian.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.resplu.2025.100991.
Contributor Information
Jason Acworth, Email: jason.acworth@health.qld.gov.au.
Jimena del Castillo, Email: jimenadelcastillo@yahoo.es.
Dianne Atkins, Email: Dianne-atkins@uiowa.edu.
Allan de Caen, Email: allan.decaen@albertahealthservices.ca.
Thomaz Bittencourt Couto, Email: thomaz.couto@hc.fm.usp.br.
Stephan Katzenschlager, Email: Stephan.Katzenschlager@med.uni-heidelberg.de.
Monica Kleinman, Email: monica.kleinman@childrens.harvard.edu.
Ryan W. Morgan, Email: morganr1@chop.edu.
Michelle Myburgh, Email: Myburghmc@ufs.ac.za.
Vinay Nadkarni, Email: nadkarni@email.chop.edu.
Janice A. Tijssen, Email: janice.tijssen@lhsc.on.ca.
Barnaby R. Scholefield, Email: barney.scholefield@sickkids.ca.
Appendix 1.
Non-Author Contributors:
The authors would like to formally acknowledge the following persons as contributors:
University of Queensland Library (Information Specialist):
-
1.
Lars Eriksson: l.eriksson@library.uq.edu.au
ILCOR Paediatric Life Support Task Force:
-
1.
Andrea Christoff: andrea.christoff@health.nsw.gov.au
-
2.
James M Gray: James.Gray@cchmc.org
-
3.
Anne-Marie Guergerian: anne-marie.guerguerian@sickkids.ca
-
4.
Hiroshi Kurosawa: kurohiro2@gmail.com
-
5.
Laurie J Morrison: Laurie.Morrison@sunnybrook.ca
-
6.
Kee-Chong Ng: Ng.kee.chong@kkh.com.sg
-
7.
Gabrielle Nuthall: gabriellen@adhb.govt.nz
-
8.
Yong-Kwang Gene Ong: geneong@yahoo.com
-
9.
Tia T Raymond: tiaraymond@me.com
-
10.
Leandra Rech: le.rech@gmail.com
-
11.
Joseph Rossano: rossanoj@chop.edu
-
12.
Steve Schexnayder: schexnaydersm@uams.edu
-
13.
Ravi Thiagarajan: Ravi.Thiagarajan@CARDIO.CHBOSTON.ORG
-
14.
Alexis Topjian: topjian@chop.edu
Appendix A. Supplementary material
The following are the Supplementary data to this article:
References
- 1.Atkins D.L., Everson-Stewart S., Sears G.K., et al. Epidemiology and outcomes from out-of-hospital cardiac arrest in children: the Resuscitation Outcomes Consortium Epistry-Cardiac Arrest. Circulation. 2009;119:1484–1491. doi: 10.1161/CIRCULATIONAHA.108.802678. [Resuscitation Outcomes Consortium I] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Van de Voorde P., Turner N.M., Djakow J., et al. European Resuscitation Council Guidelines 2021: paediatric life support. Resuscitation. 2021;161:327–387. doi: 10.1016/j.resuscitation.2021.02.015. [DOI] [PubMed] [Google Scholar]
- 3.ANZCOR. Guideline 12.2 – Paediatric Advanced Life Support (PALS); 2021. Available at: https://www.anzcor.org/home/paediatric-advanced-life-support/guideline-12-2-paediatric-advanced-life-support-pals/.
- 4.Topjian A.A., Raymond T.T., Atkins D., et al. Part 4: pediatric basic and advanced life support: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2020;142:S469–S523. doi: 10.1161/CIR.0000000000000901. [DOI] [PubMed] [Google Scholar]
- 5.Mercier E., Laroche E., Beck B., et al. Defibrillation energy dose during pediatric cardiac arrest: systematic review of human and animal model studies. Resuscitation. 2019;139:241–252. doi: 10.1016/j.resuscitation.2019.04.028. [DOI] [PubMed] [Google Scholar]
- 6.Hoyme D.B., Zhou Y., Girotra S., et al. Improved survival to hospital discharge in pediatric in-hospital cardiac arrest using 2 Joules/kilogram as first defibrillation dose for initial pulseless ventricular arrhythmia. Resuscitation. 2020;153:88–96. doi: 10.1016/j.resuscitation.2020.05.048. [DOI] [PubMed] [Google Scholar]
- 7.Maconochie I.K., Aickin R., Hazinski M.F., et al. International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations. Resuscitation. 2020;2020(156):A120–A155. doi: 10.1016/j.resuscitation.2020.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Maconochie I.K., Aickin R., Hazinski M.F., et al. International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations. Circulation. 2020;2020(142):S140–S184. doi: 10.1161/CIR.0000000000000894. [DOI] [PubMed] [Google Scholar]
- 9.Acworth J., del Castillo J., Tiwari L., DeCaen A. Energy doses for pediatric defibrillation during resuscitation. PROSPERO 2024. Available from https://www.crd.york.ac.uk/PROSPERO/view/CRD42024548898.
- 10.Page M.J., McKenzie J.E., Bossuyt P.M., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372 doi: 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Covidence systematic review software, Veritas Health Innovation, Melbourne, Australia. Available at http://www.covidence.org.
- 12.Topjian A.A., Scholefield B.R., Pinto N.P., et al. P-COSCA (Pediatric Core Outcome Set for Cardiac Arrest) in Children: an advisory statement from the International Liaison Committee on Resuscitation. Circulation. 2020;142:e246–e261. doi: 10.1161/CIR.0000000000000911. [DOI] [PubMed] [Google Scholar]
- 13.Sterne J.A., Hernán M.A., Reeves B.C., et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355 doi: 10.1136/bmj.i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.GRADEpro GDT: GRADEpro Guideline Development Tool [Software]. [computer program]. McMaster University (developed by Evidence Prime). Available at https://www.gradepro.org/. Accessed 20 November 2023.
- 15.Review Manager (RevMan) [Computer program], version 8.14.0. [computer program]. Version 8.14.0. Copenhagen: The Cochrane Collaboration; 2024.
- 16.Gutgesell H.P., Tacker W.A., Geddes L.A. Energy dose for ventricular defibrillation of children. Pediatrics. 1976;58:898–901. [PubMed] [Google Scholar]
- 17.Meaney P.A., Nadkarni V.M., Atkins D.L., et al. Effect of defibrillation energy dose during in-hospital pediatric cardiac arrest. Pediatrics. 2011;127:e16–e23. doi: 10.1542/peds.2010-1617. [DOI] [PubMed] [Google Scholar]
- 18.Rodríguez-Núñez A., López-Herce J., del Castillo J., Bellón J.M. Shockable rhythms and defibrillation during in-hospital pediatric cardiac arrest. Resuscitation. 2014;85:387–391. doi: 10.1016/j.resuscitation.2013.11.015. [DOI] [PubMed] [Google Scholar]
- 19.Rodríguez-Núñez A., López-Herce J., García C., Domínguez P., Carrillo A., Bellón J.M. Pediatric defibrillation after cardiac arrest: initial response and outcome. Crit Care. 2006;10:R113. doi: 10.1186/cc5005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tibballs J., Carter B., Kiraly N.J., Ragg P., Clifford M. External and internal biphasic direct current shock doses for pediatric ventricular fibrillation and pulseless ventricular tachycardia. Pediatr Crit Care Med. 2011;12:14–20. doi: 10.1097/PCC.0b013e3181dbb4fc. [DOI] [PubMed] [Google Scholar]
- 21.Tibballs J., Kinney S. A prospective study of outcome of in-patient paediatric cardiopulmonary arrest. Resuscitation. 2006;71:310–318. doi: 10.1016/j.resuscitation.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 22.Rossano J.W., Quan L., Kenney M.A., Rea T.D., Atkins D.L. Energy doses for treatment of out-of-hospital pediatric ventricular fibrillation. Resuscitation. 2006;70:80–89. doi: 10.1016/j.resuscitation.2005.10.031. [DOI] [PubMed] [Google Scholar]
- 23.Smida T., Cheskes S., Crowe R., et al. The association between initial defibrillation dose and outcomes following adult out-of-hospital cardiac arrest resuscitation: a retrospective, multi-agency study. Resuscitation. 2025;208 doi: 10.1016/j.resuscitation.2025.110507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Berg M.D., Banville I.L., Chapman F.W., et al. Attenuating the defibrillation dosage decreases postresuscitation myocardial dysfunction in a swine model of pediatric ventricular fibrillation. Pediatr Crit Care Med. 2008;9:429–434. doi: 10.1097/PCC.0b013e318172e9f8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Berg R.A., Chapman F.W., Berg M.D., et al. Attenuated adult biphasic shocks compared with weight-based monophasic shocks in a swine model of prolonged pediatric ventricular fibrillation. Resuscitation. 2004;61:189–197. doi: 10.1016/j.resuscitation.2003.12.021. [DOI] [PubMed] [Google Scholar]
- 26.Haskell S.E., Atkins D.L. Defibrillation in children. J Emerg Trauma Shock. 2010;3:261–266. doi: 10.4103/0974-2700.66526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Faddy S.C., Powell J., Craig J.C. Biphasic and monophasic shocks for transthoracic defibrillation: a meta analysis of randomised controlled trials. Resuscitation. 2003;58:9–16. doi: 10.1016/s0300-9572(03)00077-7. [DOI] [PubMed] [Google Scholar]
- 28.Gray J.M., Raymond T.T., Atkins D.L., et al. Inappropriate shock delivery is common during pediatric in-hospital cardiac arrest. Pediatr Crit Care Med. 2023;24:e390–e396. doi: 10.1097/PCC.0000000000003241. [pediRES-Q Investigators] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shepard L.N., Nadkarni V.M., Ng K.C., Scholefield B.R., Ong G.Y. ILCOR pediatric life support recommendations translation to constituent council guidelines: an emphasis on similarities and differences. Resuscitation. 2024;201 doi: 10.1016/j.resuscitation.2024.110247. [ILCOR Pediatric Life Support Task Force] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Topjian A.A., Scholefield B.R., Pinto N.P., et al. P-COSCA (Pediatric Core Outcome Set for Cardiac Arrest) in Children: an advisory statement from the International Liaison Committee on Resuscitation. Resuscitation. 2021;162:351–364. doi: 10.1016/j.resuscitation.2021.01.023. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.