Abstract
Objective
Systemic juvenile idiopathic arthritis (sJIA) and reactive arthritis (ReA) share overlapping clinical features, posing diagnostic challenges. Early differentiation is critical for treatment decisions but lacks reliable biomarkers. This study aims to identify simple clinical indicators and develop a clinical prediction model to distinguish sJIA from ReA.
Methods
This study retrospectively included clinical data of 397 sJIA patients and 290 ReA patients who attended the Children’s Hospital of Chongqing Medical University from 2016–2024. Key predictors were identified by ANOVA, chi-square tests, univariate logistic regression, multivariate logistic regression, and stepwise analysis. The diagnostic model was established and validated by performing ROC analysis. Furthermore, we additionally included data from 20 sJIA and 20 ReA patients from two other centers to validate the above results.
Results
A total of 19 statistically different clinical indicators were identified by ANOVA and chi-square tests. These indicators were included in univariate and multivariate logistic regression analyses, lower albumin levels and significantly higher levels of gamma-glutamyl transferase (GGT) were found in sJIA patients compared to ReA in both the training and validation sets (p values were all < 0.05). In a stepwise analysis of age, gender, inflammatory cells (lymphocytes, monocytes) and inflammatory markers, it was found that albumin and GGT were still effective in differentiating between the two diseases. Clinical prediction models were developed using albumin and GGT, with AUCs of 0.842 (training) and 0.849 (validation), showing excellent diagnostic effect. These indicators also demonstrated good diagnostic efficacy in cohorts from two other centers.
Conclusion
Albumin and GGT are important clinical indicators for differentiating sJIA from ReA. The albumin-GGT prediction model provides a simple, clinically feasible tool to reduce diagnostic uncertainty.
Keywords: systemic juvenile idiopathic arthritis, reactive arthritis, clinical prediction model, albumin, gamma-glutamyl transferase
Introduction
Systemic juvenile idiopathic arthritis (sJIA) is an inflammatory disease characterized by severe systemic inflammation that may be accompanied by arthritis. Currently, the diagnosis of sJIA is primarily based on the 2001 International League of Associations for Rheumatology (ILAR) criteria.1 In recent years, advances in elucidating the immunopathogenic mechanisms of sJIA have led to significant therapeutic breakthroughs, but a subset of patients continue to present with refractory disease phenotypes with severe drug-resistant complications, highlighting the long-term challenge of achieving complete disease remission and underscoring the unmet clinical need for precision therapy.2 Recent studies indicate that the early administration of biologics markedly enhances the prognosis of sJIA patients, mitigates the risk of long-term joint damage, and attenuates the likelihood of macrophage activation syndrome (MAS) driven by systemic inflammation through the targeted inhibition of key inflammatory pathways.3,4 However, the diagnosis of sJIA is largely dependent on clinical symptoms and laboratory tests, so patients still face challenges in early diagnosis.4–6 There is an urgent need to explore biomarkers linked to conventional testing indicators to break through the limitations of the existing categorical diagnostic framework for early identification.7,8
Reactive arthritis (ReA) is a form of aseptic arthritis diagnosed based on characteristic clinical features, evidence of preceding or concurrent infection, and exclusion of alternative causes. Traditionally, ReA has been linked to infections of the genitourinary or gastrointestinal tract, particularly Chlamydia and Salmonella.9–11 However, recent studies implicate respiratory infections as potential triggers, broadening the recognized etiological spectrum.12,13 Intriguingly, respiratory infections have also been implicated as one of the sJIA predisposing factors, which may promote systemic inflammation by stimulating multiple antigen-specific pathways.14 In pediatric clinical practice, both ReA and sJIA may exhibit overlapping features, including nonsuppurative arthritis, fever, rash, and elevated inflammatory markers, complicating early differential diagnosis.3,11 This challenge is compounded in paediatric patients, who may struggle to localize pain or articulate subjective symptoms, further obscuring clinical assessment.
Moreover, therapeutic strategies for ReA and sJIA differ markedly, while sJIA typically necessitates early and aggressive immunosuppressive intervention, ReA management predominantly focuses on infection eradication and symptomatic relief.3,15 Consequently, failure to accurately distinguish between these conditions in the early disease stages may result in delayed initiation of critical therapies in patients with sJIA, adversely impacting their long-term prognosis. Currently, clinical features, HLA-B27 testing, and synovial fluid analysis are the main tools used to differentiate between the two diseases.16 However, these diagnostic tools are characterized by limited specificity, particularly among pediatric patients, and their practical utility may be restricted by logistical and technical challenges encountered in routine clinical settings. Given the significant implications associated with delayed or inappropriate therapeutic intervention, the establishment of reliable, clinically feasible biomarkers or indicators capable of clearly distinguishing ReA from sJIA is urgently warranted.
In this study, we used univariate and multivariate logistics regression analysis, stepwise analysis and other analytical methods to search for simplified clinical indicators, and constructed a clinical prediction model to assist clinicians in making a more accurate diagnosis, so that children can be diagnosed in a timely manner and receive appropriate treatment.
Methods
Study Population and Data Collection
We conducted a retrospective analysis of clinical data from patients diagnosed with sJIA and ReA who were treated at the Children’s Hospital of Chongqing Medical University between January 2016 and December 2024. Eligible sJIA participants met the diagnostic and exclusion criteria for sJIA proposed by the ILAR in 2001.1
The diagnostic criteria for all eligible children with ReA met the diagnostic criteria presented at the Fourth International Workshop on Reactive Arthritis.16,17 The diagnostic criteria are defined as the presence of documented extra-articular infections (including intestinal, urogenital, or respiratory tract infections) occurring within a timeframe of 3 days to 6 weeks preceding the onset of arthritis, coupled with characteristic articular manifestations that predominantly involve large lower limb joints in an asymmetric pattern. In addition, the diagnosis of ReA requires the exclusion of other diseases, and the exclusion criteria include: infectious arthritis, sJIA, severe immunodeficiency, arthritis due to other connective tissue diseases, and overlap syndrome.
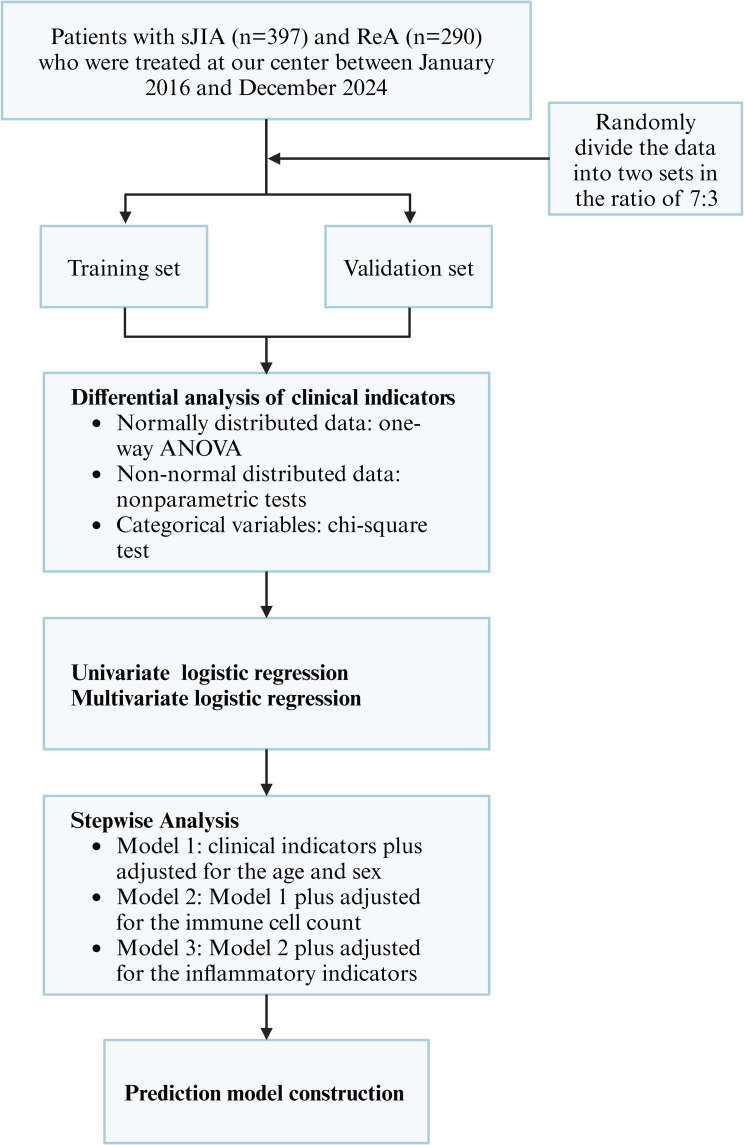
We retrospectively collected clinical data from our center’s big data platform, including patients’ demographic characteristics, diagnoses, and laboratory test indicators. The data were then randomly split into a training set and a validation set in a 7:3 ratio to ensure the robustness of the model. The flow chart of this study is detailed in Figure 1.
Figure 1.
Flow chart of the research process.
Ethics
This study was approved by the Ethics Committee of the Children’s Hospital of Chongqing Medical University (Approval No. (2022) Ethical Review (Research) No. 52). The study was conducted in accordance with the Declaration of Helsinki. As a retrospective study, it utilized medical records from previous clinical visits and posed no additional risk to the patients.
Prediction Model Construction
Prior to model development, comprehensive data preprocessing was performed. Clinical variables with more than 15% missing values were excluded from the regression analysis to ensure data integrity and robustness. Additionally, both potential confounding factors and key inflammatory markers were rigorously controlled through multivariate logistic regression, complemented by stepwise analysis to refine variable selection and minimize model overfitting. These combined strategies strengthened the validity and reliability of the findings.
A logistic regression model was fitted to the training dataset using relevant predictors and predicted probabilities were calculated for each sample. The same model was applied to the validation set and predicted probabilities were computed accordingly. Model performance was evaluated by plotting Receiver Operating Characteristic (ROC) curves and calculating the Area Under the Curve (AUC).
To evaluate the consistency of the model’s performance between the training and test datasets, a statistical comparison of the ROC curves was conducted using the DeLong test (roc.test). This test compared the AUCs of the ROC curves for both datasets, to assess the model’s performance.
To assess the generalizability of the model, external validation was performed using an independent cohort from two centers: Shenzhen Children’s Hospital and the Children’s Hospital of Nanjing Medical University. This external cohort includes clinical data from a total of 20 sJIA patients and 20 ReA patients from two centers. All patients met the same inclusion and exclusion criteria as the primary cohort. All external datasets underwent harmonized preprocessing to ensure consistency with the primary analysis. The final predictive model, developed from the primary cohort, was applied to these external datasets without modification. Model performance was evaluated using the AUCs of the ROC curves. Additionally, logistic regression was conducted to assess the association between predicted probabilities and disease status, with odds ratios (ORs) and 95% confidence intervals (CIs) calculated to quantify predictive strength. These combined steps provided complementary evidence of the model’s robustness and diagnostic accuracy.
Statistical Analysis
Statistical analyses were performed using R version 3.5. Data with a normal distribution are expressed as mean ± standard deviation and were analyzed using one-way ANOVA. Data with a non-normal distribution are presented as median (interquartile range) and were analyzed using non-parametric tests. Categorical variables are expressed as percentages, and differences between groups were assessed using the Chi-square test.
Univariate and multivariate logistic regression models were used to estimate ORs and 95%CIs for the associations between sJIA and ReA.18 Multivariate models were constructed using a stepwise approach to assess the relationship between sJIA and ReA. In the first step, we adjusted for sex and age. In the second step, we included inflammatory cells such as lymphocytes, monocytes, and neutrophils. In the final step, we adjusted for inflammatory indicators, including C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR). Statistical significance was defined as p ≤ 0.05.
Results
Clinical Characteristics
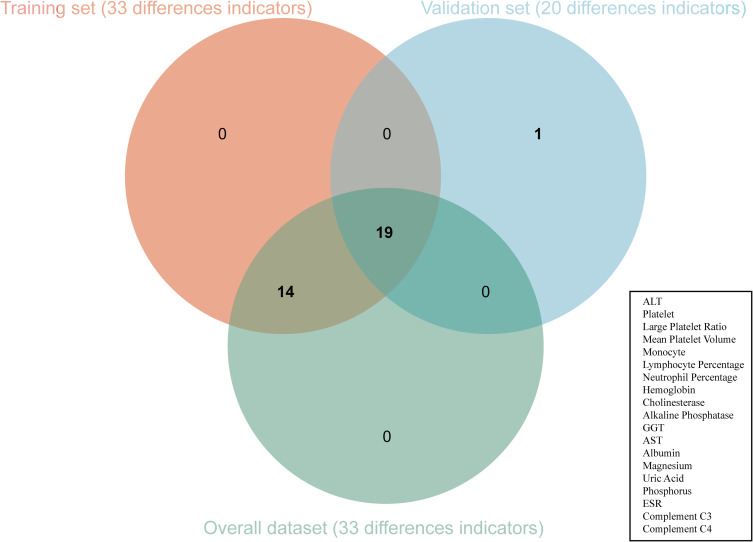
397 sJIA (female 161, male 236, mean age 13.23 years old) and 290 ReA (female 130, male 160, mean age 12.40 years old) were included in our study. Table 1 describes the baseline characteristics and differences between groups of ReA patients and sJIA patients. Meanwhile, we identified a total of 33 serologic indicators that were significantly different between the two groups, including hepatic transaminases, immune cell counts, CRP, ESR, complement, and immunoglobulins. Subsequently, we divided the dataset into training and validation sets (external test set) in a 7:3 ratio, (Table S1). No significant differences between both sets across most features, indicating good consistency in data distribution between the two. In the comparison of baseline data from the training set (Table S2), we found that clinical indicators of alanine aminotransferase, Gamma-glutamyl transferase (GGT), aspartate aminotransferase, white blood cell, neutrophil, monocyte and ESR et al were significantly elevated in the sJIA patients as compared to the ReA patients. In contrast, sJIA patients had lower red blood cell, lymphocyte percentage, and albumin et al than ReA patients. The same trend was observed in the validation sets (Table S3), suggesting that the results from the training set have good external consistency. The positive indicators in the training set, validation set and total data set were further analyzed and then the intersection between them was extracted. It was found that 19 clinical indicators such as alanine aminotransferase, monocytes, lymphocyte percentage, neutrophil percentage, GGT, aspartate aminotransferase, albumin and ESR showed significant differences (Figure 2).
Table 1.
Clinical Characteristics for Pediatric ReA and sJIA
| sJIA (n=397) | ReA (n=290) | p value | |
|---|---|---|---|
| Age (years) | 13.23±4.34 | 12.40±4.35 | 0.014 |
| Red Blood Cell (1012/L) | 4.06±0.55 | 4.52±0.43 | <0.001 |
| White Blood Cell (109/L) | 13.10±6.78 | 7.69±2.75 | <0.001 |
| Platelet (109/L) | 418.98±179.84 | 349.62±119.69 | <0.001 |
| Large Platelet Ratio (%) | 22.32±9.61 | 27.61±10.11 | <0.001 |
| Mean Platelet Volume (fL) | 9.72±1.16 | 10.34±1.22 | <0.001 |
| Monocyte (109/L) | 0.33±0.20 | 0.22±0.11 | <0.001 |
| Lymphocyte Percentage (%) | 0.25±0.15 | 0.38±0.14 | <0.001 |
| Eosinophil Percentage (%) | 0.02±0.01 | 0.03±0.02 | <0.001 |
| Neutrophil (109/L) | 9.56±6.13 | 4.50±2.34 | <0.001 |
| Neutrophil Percentage (%) | 0.70±0.16 | 0.57±0.15 | <0.001 |
| Hemoglobin (g/L) | 104.31±17.09 | 121.52±14.26 | <0.001 |
| Cholinesterase (U/L) | 6544.64±1927.88 | 8415.03±1762.03 | <0.001 |
| Alkaline Phosphatase (U/L) | 157.60±65.22 | 193.31±72.27 | <0.001 |
| GGT (U/L) | 36.26±39.65 | 15.13±15.09 | <0.001 |
| AST (U/L) | 43.81±79.37 | 25.94±10.07 | <0.001 |
| ALT (U/L) | 43.89±122.20 | 18.66±20.39 | <0.001 |
| Pre-Albumin (mg/L) | 149.79±89.69 | 185.28±60.03 | <0.001 |
| Albumin (g/L) | 38.46±5.35 | 44.64±4.14 | <0.001 |
| Globulin (g/L) | 29.44±7.12 | 26.76±6.08 | <0.001 |
| Bile Acids (umol/L) | 10.11±26.34 | 5.06±12.73 | 0.004 |
| Magnesium (mmol/L) | 0.90±0.09 | 0.86±0.06 | <0.001 |
| Urea (mmol/L) | 3.68±1.92 | 4.29±1.20 | <0.001 |
| Sodium (mmol/L) | 139.07±3.01 | 139.88±1.99 | <0.001 |
| Chloride Ion (mmol/L) | 101.68±3.72 | 102.64±2.58 | <0.001 |
| Uric Acid (umol/L) | 230.65±80.81 | 278.89±73.97 | <0.001 |
| Phosphorus (mmol/L) | 1.47±0.30 | 1.59±0.22 | <0.001 |
| Creatinine (umol/L) | 32.91±14.07 | 36.83±10.82 | <0.001 |
| CRP (mg/L) | 50.84±58.83 | 13.25±24.27 | <0.001 |
| ESR (mm/1hr) | 67.85±39.29 | 30.94±31.09 | <0.001 |
| Complement C3 (g/L) | 1.35±0.32 | 1.08±0.28 | <0.001 |
| Complement C4 (g/L) | 0.27±0.10 | 0.23±0.09 | <0.001 |
| Immunoglobulin G (g/L) | 13.37±5.50 | 12.12±3.99 | 0.002 |
Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; GGT, gamma-glutamyl transferase.
Figure 2.
Intersection of clinical indicators of difference between training set, validation set, and overall dataset.
Univariate and Multivariate Logistic Regression
We incorporated the above indicators of differential expression into univariate and multivariate logistic regression analyses, the results of which are shown in Table 2. In the training set, univariate logistic regression analysis showed that all of the above 19 indicators were strongly correlated with the occurrence of sJIA. However, because the OR values of monocyte, lymphocyte percentage, eosinophil percentage, neutrophil percentage, magnesium, complement C3, and complement C4 were too large or too small, which could have biased our results, it was excluded from further analysis. In a multivariate logistic regression model, we found a close relationship between large platelet ratio, cholinesterase, GGT, and albumin in identifying ReA and sJIA. In the validation set (Table S4), univariate logistic regression analysis showed that GGT, albumin and ESR et al were strongly associated with the development of sJIA. However, when applying multivariate logistic regression analysis, we found significant independent associations between GGT (OR:1.03, 95%CI:1.00–1.06, p=0.038), albumin (OR:0.82, 95%CI:0.71–0.96, p=0.012), and sJIA, which highlighted their potential relevance as key factors in disease progression. Subsequently in the test set and validation set, we found that decreased albumin and increased GGT may play an important role in discriminating between the two diseases.
Table 2.
Univariate and Multivariate Logistic Regression Model for Pediatric ReA and sJIA in the Training Set
| Univariate Logistic Regression | p value | Multivariate Logistic Regression | p value | |
|---|---|---|---|---|
| Alanine Aminotransferase | 1.02 (95%CI:1.00–1.03) |
p=0.012 | 0.99 (95%CI:0.96–1.01) |
p=0.192 |
| Platelet | 1.00 (95%CI:1.00–1.01) |
p<0.001 | 1.00 (95%CI:1.00–1.00) |
p=0.536 |
| Large Platelet Ratio | 0.94 (95%CI:0.91–0.96) |
p<0.001 | 0.96 (95%CI:0.93–1.00) |
p=0.045 |
| Monocyte | 165.06 (95%CI:24.70–1103.20) |
p<0.001 | – | – |
| Lymphocyte Percentage | 0.00 (95%CI:0.00–0.02) |
p<0.001 | – | – |
| Eosinophil Percentage | 0.00 (95%CI:0.00–0.00) |
p<0.001 | – | – |
| Neutrophil Percentage | 200.57 (95%CI:34.81–1155.58) |
p<0.001 | – | – |
| Hemoglobin | 0.93 (95%CI:0.91–0.95) |
p<0.001 | 0.99 (95%CI:0.96–1.01) |
p=0.280 |
| Cholinesterase | 1.00 (95%CI:1.00–1.00) |
p<0.001 | 1.00 (95%CI:1.00–1.00) |
p=0.046 |
| Alkaline Phosphatase | 0.99 (95%CI:0.99–0.99) |
p<0.001 | 1.00 (95%CI:0.99–1.00) |
p=0.219 |
| GGT | 1.07 (95%CI:1.04–1.10) |
p<0.001 | 1.07 (95%CI:1.03–1.11) |
p<0.001 |
| AST | 1.03 (95%CI:1.01–1.04) |
p=0.007 | 1.03 (95%CI:0.99–1.08) |
p=0.098 |
| Albumin | 0.77 (95%CI:0.72–0.83) |
p<0.001 | 0.91 (95%CI:0.83–1.00) |
p=0.048 |
| Magnesium | 1414.72 (95%CI:41.25–48,516.77) |
p<0.001 | – | – |
| Uric Acid | 0.99 (95%CI:0.99–1.00) |
p<0.001 | 1.00 (95%CI:0.99–1.00) |
p=0.269 |
| Phosphorus | 0.17 (95%CI:0.07–0.44) |
p<0.001 | 0.59 (95%CI:0.16–2.25) |
p=0.444 |
| Complement C3 | 12.53 (95%CI:5.26–29.86) |
p<0.001 | – | – |
| Complement C4 | 144.63 (95%CI:9.19–2276.49) |
p<0.001 | – | – |
| ESR | 1.03 (95%CI:1.02–1.03) |
p<0.001 | 1.00 (95%CI:0.99–1.01) |
p=0.378 |
Abbreviations: GGT, gamma-glutamyl transferase; AST, aspartate aminotransferase; ESR, erythrocyte sedimentation rate; 95%CI, 95% confidence intervals.
Stepwise Analysis of Albumin and GGT in sJIA
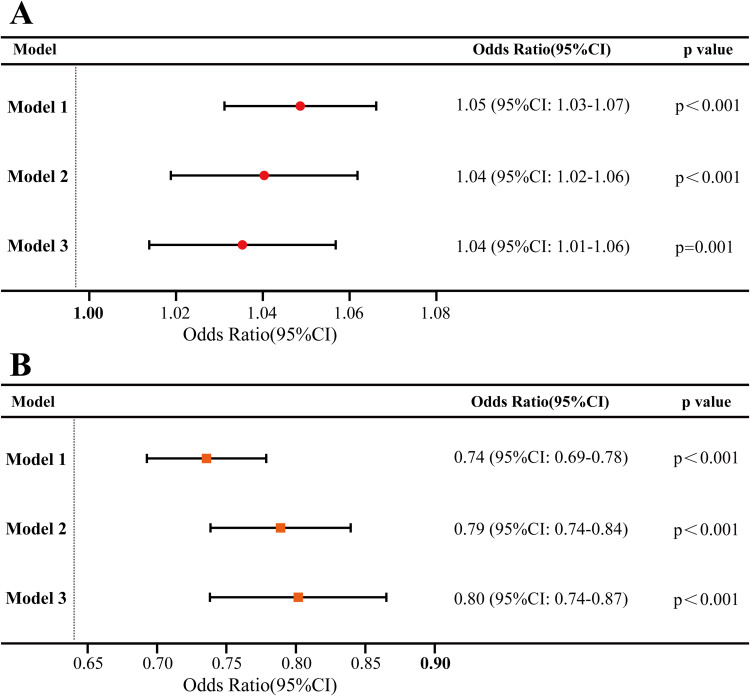
A stepwise analysis was conducted to further evaluate the relationship between albumin, GGT, and sJIA. In the training set, we first adjusted for age and gender, as shown in Model 1 of Figure 3, and found that GGT (OR:1.05, 95%CI:1.03–1.07, p<0.001) and albumin (OR:0.74, 95%CI:0.69–0.78, p<0.001) were not affected. Subsequently, we introduced inflammatory cell counts in Model 2 and found that GGT (OR:1.04, 95%CI:1.02–1.06, p<0.001) and albumin (OR:0.79, 95%CI:0.74–0.84, p<0.001) could still be used to discriminate between the two diseases. Finally, we further adjusted for CRP, and ESR in Model 3, and still found that GGT (OR:1.04, 95%CI:1.01–1.06, p=0.001) and albumin (OR:0.80, 95%CI:0.74–0.87, p<0.001) could effectively predict the occurrence of sJIA. The same pattern was observed in the validation set (Figure S1).
Figure 3.
Stepwise analysis of the relationship between clinical indicators and inflammatory indicators in training set. (A) Stepwise analysis of the relationship between GGT and inflammatory indicators in training set; (B) Stepwise analysis of the relationship between Albumin and inflammatory indicators in training set. Model 1: clinical indicators plus adjusted for the age and sex; Model 2: Model 1 plus adjusted for the immune cell count (white blood cell, monocyte, lymphocyte, neutrophil); Model 3: Model 2 plus adjusted for the classical inflammatory indicators (CRP, ESR).
Abbreviation: 95%CI: 95% confidence intervals.
Prediction Model Construction
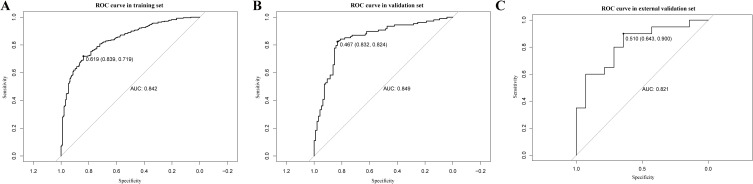
Subsequently, to assess the predictive ability of albumin and GGT for sJIA, we constructed a clinical prediction model (Figure 4). We found that both albumin and GGT effectively predicted the occurrence of sJIA in both the training (AUC=0.842) and validation sets (AUC=0.849). We further evaluated the ROC curves for both the training and test sets and found no statistically significant difference between them, further validating the reliability of the results (p=0.832). To assess the generalizability of the model, we collected an independent cohort from two clinical centers for external validation. Univariate regression analysis revealed that sJIA patients had lower albumin levels than ReA patients (OR: 0.79, 95%CI: 0.63–0.99, p=0.048), while GGT levels were higher in sJIA patients (OR: 1.47, 95%CI: 1.04–2.07, p=0.031). In the external validation cohort, the model achieved an AUC of 0.821, further confirming its generalizability. The dynamic nomogram of this predictive model can be accessed and used at https://fml-chcmu.shinyapps.io/dynnomapp.
Figure 4.
Predictive modeling of GGT in combination with albumin for the prediction of sJIA. (A) ROC curves in the training set; (B) ROC curves in the validation set; (C) ROC curves in the external validation set.
Abbreviation: AUC, Area Under the Curve.
Discussion
There is significant clinical overlap between sJIA and ReA early in the course of the disease, making accurate identification challenging.19,20 In this study, we performed multivariate logistic regression analysis to adjust for confounding factors and inflammatory markers, and for the first time identified that children with sJIA had significantly lower albumin levels and higher GGT levels compared to the ReA group. In addition, stepwise regression was conducted to further adjust for age, sex, and inflammatory markers, yielding consistent results that reinforced the robustness of this finding. To validate externally, we incorporated data from two additional centers, which similarly confirmed that children with sJIA exhibited significantly lower albumin levels and higher GGT levels than those with ReA, further strengthening the reliability of this conclusion. Such biomarker differences not only help to establish a rapid differential diagnostic model, but also may reveal differences in underlying pathologic mechanisms, providing a new direction for improving clinical decision-making.
Currently, research on clinical indicators for differentiating sJIA from ReA remains relatively limited, with most studies focusing on immune indicators or specific clinical manifestations. Liver function indicators have not been sufficiently explored in distinguishing these two diseases. Our findings suggest that albumin and GGT may have significant potential in differentiating sJIA from ReA, providing new directions for future clinical practice. Recent studies have indicated a strong relationship between albumin, GGT and sJIA. For instance, Di Donato G et al proposed that Albumin could serve as a sensitive indicator for systemic inflammation in sJIA.21 This aligns with our findings, suggesting that liver involvement may be an underappreciated aspect of this disease. Furthermore, a multicenter clinical study involving 33 sJIA patients by Zeft A et al found that albumin is closely related to the effectiveness of hormone therapy.22 These findings further support the idea that albumin could be a useful clinical indicator for monitoring disease activity in sJIA. Moreover, studies in sJIA patients with complications such as macrophage activation syndrome (MAS) have reported that a majority of patients with sJIA complicated by MAS exhibit decreased albumin, reinforcing the close association between albumin and disease activity in sJIA.23 While research on GGT in sJIA is still scarce, there is evidence from other inflammatory diseases, such as rheumatoid arthritis, where Vergneault et al demonstrated that GGT could serve as a global inflammation indicator, correlating with the severity of systemic inflammation and prognosis.24 This suggests that GGT also might hold value in evaluating the inflammatory status and disease progression in sJIA.
ReA is a sterile inflammatory disease of the joints, mainly caused by distant or systemic infections.25 However, current studies have not demonstrated significant differences in liver function indicators between ReA patients and healthy controls.26 This indicates that while ReA is associated with systemic inflammation and joint symptoms due to infection, the impact on liver function appears to be minimal. As a result, liver clinical indicators like albumin and GGT may not possess sufficient diagnostic value for distinguishing ReA from healthy controls. In contrast, our research results indicate that albumin levels in sJIA children are lower than those in children with ReA, while GGT levels are higher, highlighting the potential of these clinical indicator to differentiate between the two diseases. Given the limited liver involvement in ReA, we infer that albumin and GGT may serve as valuable clinical indicators for distinguishing sJIA from ReA. Although the exact mechanisms linking albumin and GGT to sJIA remain unclear, existing studies provide valuable insights that warrant further exploration. These indicators of liver function are believed to play significant roles in inflammatory responses and disease progression. First, sJIA is characterized by a systemic inflammatory response, primarily manifested as dysregulation of cytokines and immune responses. Research has shown that sJIA is closely associated with a severe imbalance between pro-inflammatory and anti-inflammatory cytokines, which may be related to changes in the activity of enzymes such as albumin and GGT.27 Elevations in these liver transaminases are often associated with liver inflammation and damage, which may reflect the presence of systemic inflammatory responses. Specifically, albumin and GGT may not only indicate liver damage but also serve as clinical indicators of the inflammatory state, reflecting the activation of systemic immune responses in sJIA. Secondly, the role of albumin and GGT as clinical indicators of oxidative stress and their association with metabolic risk factors have garnered widespread attention. Studies have found significant correlations between the levels of these indicators of liver function and inflammatory processes, especially in the pathogenesis of sJIA.27,28 This association suggests that albumin and GGT may not only be involved in the onset of liver injury but also play a role in exacerbating the inflammatory processes that characterize and drive the progression of sJIA. Further research also indicates that the combined effect of albumin and GGT on the incidence of diabetes highlights their potential in modulating systemic inflammation.29,30 The inflammatory environment in sJIA may be exacerbated by metabolic disturbances indicated by albumin and GGT levels, thereby accelerating disease progression. Therefore, albumin and GGT could serve not only as clinical indicators of liver dysfunction but also as sensitive indicators of systemic inflammation and metabolic abnormalities. Moreover, the involvement of albumin and GGT in liver function and their potential impact on systemic inflammation is supported by studies on their role in liver diseases.31,32 Monitoring the levels of these indicators of liver function could therefore provide valuable information for clinicians to assess disease activity and inflammation status in sJIA patients. In conclusion, albumin and GGT are not only important clinical indicators of liver function but also play a crucial role in the pathogenesis of sJIA by mediating systemic inflammation.
Our research offers a promising tool for differentiating between sJIA and ReA based on the serum levels of albumin and GGT, and the same results have been obtained from data from two other centers. This model provides clinicians with a simple and effective means for early identification and accurate diagnosis of these conditions. By reducing the risk of misdiagnosis due to overlapping clinical presentations, the model could enhance diagnostic efficiency and assist clinicians in initiating treatment earlier.
The current study has several limitations that should be acknowledged. First, although we employed multivariate logistic regression analysis and stepwise regression analysis to control for recognized confounding factors, including age, sex, and partial inflammatory markers, the retrospective design may introduce selection bias and residual confounding factors. Second, while inflammatory markers such as CRP and ESR were incorporated into the model, key cytokines including IL-6 and TNF-α were excluded due to excessive missing data, which may limit the biological interpretability of the model. Third, although we included data from two other clinical centers for external validation, sJIA is a rare disease with limited availability of clinical data at initial diagnosis. Consequently, our external validation cohort included only 20 sJIA and 20 ReA cases, which may limit the robustness and generalizability of the validation results. Future studies should focus on larger, multicenter cohorts to validate these findings and further explore the integration of this model into clinical practice.
Conclusion
Our findings suggest that albumin and GGT serve as promising clinical indicators for differentiating sJIA from ReA, providing novel evidence to advance differential diagnosis. Future work will validate these results through multicenter retrospective and prospective cohort studies and assess the integration of this model into clinical decision-making frameworks to optimize its translational utility.
Acknowledgments
We sincerely thank all participants in this study.
Funding Statement
This research was supported by the National Key R&D Program of China (2021YFC2702003).
Data Sharing Statement
All raw and processed data are available from the corresponding author upon reasonable request.
Ethics Approval and Consent to Participate
This research received ethical approval from the Ethics Committee of the Children’s Hospital of Chongqing Medical University (Approval No. (2022) Ethical Review (Research) No. 52) and adhered strictly to the principles outlined in the Declaration of Helsinki. Given the retrospective nature of this study, which involved only the analysis of previously obtained medical records, there was no additional risk posed to patients, and thus informed consent was waived.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- 1.Petty RE, Southwood TR, Manners P, et al. International league of associations for rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J Rheumatol. 2004;31(2):390–392. [PubMed] [Google Scholar]
- 2.Ambler WG, Nanda K, Onel KB, Shenoi S. Refractory systemic onset juvenile idiopathic arthritis: current challenges and future perspectives. Ann Med. 2022;54(1):1839–1850. doi: 10.1080/07853890.2022.2095431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kumar S. Systemic juvenile idiopathic arthritis: diagnosis and management. Indian J Pediatr. 2016;83(4):322–327. Epub 20160226. doi: 10.1007/s12098-016-2060-z [DOI] [PubMed] [Google Scholar]
- 4.Horneff G, Schulz AC, Klotsche J, et al. Experience with etanercept, tocilizumab and interleukin-1 inhibitors in systemic onset juvenile idiopathic arthritis patients from the biker registry. Arthritis Res Ther. 2017;19(1):256. Epub 20171122. doi: 10.1186/s13075-017-1462-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kearsley-Fleet L, Beresford MW, Davies R, et al. Short-term outcomes in patients with systemic juvenile idiopathic arthritis treated with either tocilizumab or anakinra. Rheumatology. 2019;58(1):94–102. doi: 10.1093/rheumatology/key262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Klein-Wieringa IR, Brinkman DMC, Ten Cate R, Hissink Muller PCE. Update on the treatment of nonsystemic juvenile idiopathic arthritis including treatment-to-target: is (Drug-Free) inactive disease already possible? Curr Opin Rheumatol. 2020;32(5):403–413. doi: 10.1097/BOR.0000000000000727 [DOI] [PubMed] [Google Scholar]
- 7.Ter Haar NM, van Dijkhuizen EHP, Swart JF, et al. Treatment to target using recombinant interleukin-1 receptor antagonist as first-line monotherapy in new-onset systemic juvenile idiopathic arthritis: results from a five-year follow-up study. Arthritis Rheumatol. 2019;71(7):1163–1173. Epub 20190525. doi: 10.1002/art.40865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee JJY, Schneider R. Systemic Juvenile Idiopathic Arthritis. Pediatr Clin North Am. 2018;65(4):691–709. doi: 10.1016/j.pcl.2018.04.005 [DOI] [PubMed] [Google Scholar]
- 9.Wu IB, Schwartz RA. Reiter’s syndrome: the classic triad and more. J Am Acad Dermatol. 2008;59(1):113–121. Epub 20080423. doi: 10.1016/j.jaad.2008.02.047 [DOI] [PubMed] [Google Scholar]
- 10.Stavropoulos PG, Soura E, Kanelleas A, Katsambas A, Antoniou C. Reactive Arthritis. J Eur Acad Dermatol Venereol. 2015;29(3):415–424. Epub 20140908. doi: 10.1111/jdv.12741 [DOI] [PubMed] [Google Scholar]
- 11.Garcia-Kutzbach A, Chacon-Suchite J, Garcia-Ferrer H, Iraheta I. Reactive arthritis: update 2018. Clin Rheumatol. 2018;37(4):869–874. Epub 20180217. doi: 10.1007/s10067-018-4022-5 [DOI] [PubMed] [Google Scholar]
- 12.Ahmed S, Padhan P, Misra R, Danda D. Update on post-streptococcal reactive arthritis: narrative review of a forgotten disease. Curr Rheumatol Rep. 2021;23(3):19. Epub 20210210. doi: 10.1007/s11926-021-00982-3 [DOI] [PubMed] [Google Scholar]
- 13.Kobayashi S, Ichikawa G. Reactive arthritis induced by tonsillitis: a type of ‘focal infection’. Adv Otorhinolaryngol. 2011;72:79–82. Epub 20110818. doi: 10.1159/000324612 [DOI] [PubMed] [Google Scholar]
- 14.Rigante D, Bosco A, Esposito S. The etiology of juvenile idiopathic arthritis. Clin Rev Allergy Immunol. 2015;49(2):253–261. doi: 10.1007/s12016-014-8460-9 [DOI] [PubMed] [Google Scholar]
- 15.Szamocki S, Martyn-Hemphill C, Green JSA. Reactive arthritis: can’t see, can’t pee, can’t climb a tree. Trends Urol Men’s Health. 2016;17(1):17–20. doi: 10.1002/tre.501 [DOI] [Google Scholar]
- 16.Tugelbayeva А, Ivanova R, Goremykina М, Rymbayeva Т, Toktabayeva B. Reactive arthritis in children (Review). Georgian Med News. 2021;2021:130–135. [PubMed] [Google Scholar]
- 17.Sieper J, Braun J, Kingsley GH. Report on the fourth international workshop on reactive arthritis. Arthritis Rheum. 2000;43(4):720–734. doi: [DOI] [PubMed] [Google Scholar]
- 18.Zhang Y, Luo Q, Lu K, You M, Wang H. Subclinical atherosclerosis in primary sjogren’s syndrome: comparable risk with diabetes mellitus. Clin Rheumatol. 2023;42(6):1607–1614. Epub 20230223. doi: 10.1007/s10067-023-06538-3 [DOI] [PubMed] [Google Scholar]
- 19.Liang TC, Hsu CT, Yang YH, Lin YT, Chiang BL. Analysis of childhood reactive arthritis and comparison with juvenile idiopathic arthritis. Clin Rheumatol. 2005;24(4):388–393. Epub 20041210. doi: 10.1007/s10067-004-1042-0 [DOI] [PubMed] [Google Scholar]
- 20.Nicoara DM, Munteanu AI, Scutca AC, et al. Evaluating the diagnostic performance of systemic immune-inflammation index in childhood inflammatory arthritis: a focus on differentiating juvenile idiopathic arthritis from reactive arthritis. Biomedicines. 2023;12(1):65. Epub 20231227. doi: 10.3390/biomedicines12010065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Di Donato G, Attanasi M, D’ Angelo D M, et al. Associations of C reactive protein to albumin ratio, neutrophil to lymphocyte ratio, platelet to lymphocyte ratio with disease activity in patients with juvenile idiopathic arthritis. BMC Rheumatol. 2024;8(1):26. Epub 20240617. doi: 10.1186/s41927-024-00390-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zeft A, Hollister R, LaFleur B, et al. Anakinra for systemic juvenile arthritis: the Rocky Mountain experience. J Clin Rheumatol. 2009;15(4):161–164. doi: 10.1097/RHU.0b013e3181a4f459 [DOI] [PubMed] [Google Scholar]
- 23.Zou LX, Zhu Y, Sun L, et al. Clinical and laboratory features, treatment, and outcomes of macrophage activation syndrome in 80 children: a multi-center study in China. World J Pediatr. 2020;16(1):89–98. Epub 20191014. doi: 10.1007/s12519-019-00256-0 [DOI] [PubMed] [Google Scholar]
- 24.Vergneault H, Vandebeuque E, Codullo V, Allanore Y, Avouac J. Disease activity score in 28 joints using ggt permits a dual evaluation of joint activity and cardiovascular risk. J Rheumatol. 2020;47(12):1738–1745. Epub 20200615. doi: 10.3899/jrheum.200185 [DOI] [PubMed] [Google Scholar]
- 25.Morris D, Inman RD. Reactive arthritis: developments and challenges in diagnosis and treatment. Curr Rheumatol Rep. 2012;14(5):390–394. doi: 10.1007/s11926-012-0280-4 [DOI] [PubMed] [Google Scholar]
- 26.Mileto SJ, Hutton ML, Walton SL, et al. Bezlotoxumab prevents extraintestinal organ damage induced by clostridioides difficile infection. Gut Microbes. 2022;14(1):2117504. doi: 10.1080/19490976.2022.2117504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Artigas A, Wernerman J, Arroyo V, Vincent JL, Levy M. Role of albumin in diseases associated with severe systemic inflammation: pathophysiologic and clinical evidence in sepsis and in decompensated cirrhosis. J Crit Care. 2016;33:62–70. Epub 20151229. doi: 10.1016/j.jcrc.2015.12.019 [DOI] [PubMed] [Google Scholar]
- 28.Bruck N, Schnabel A, Hedrich CM. Current understanding of the pathophysiology of systemic juvenile idiopathic arthritis (sjia) and target-directed therapeutic approaches. Clin Immunol. 2015;159(1):72–83. Epub 20150506. doi: 10.1016/j.clim.2015.04.018 [DOI] [PubMed] [Google Scholar]
- 29.Liu X, Hamnvik OP, Chamberland JP, et al. Circulating alanine transaminase (Alt) and gamma-glutamyl transferase (Ggt), but not fetuin-a, are associated with metabolic risk factors, at baseline and at two-year follow-up: the prospective Cyprus metabolism study. Metabolism. 2014;63(6):773–782. Epub 20140315. doi: 10.1016/j.metabol.2014.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Petta S, Macaluso FS, Barcellona MR, et al. Serum gamma-glutamyl transferase levels, insulin resistance and liver fibrosis in patients with chronic liver diseases. PLoS One. 2012;7(12):e51165. Epub 20121205. doi: 10.1371/journal.pone.0051165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Choi SH, Kim BT, Shin J, Kim KN. Combined effect of serum alanine aminotransferase and gamma-glutamyltransferase on incidence of diabetes mellitus: a longitudinal study. Medicine. 2020;99(11):e18963. doi: 10.1097/MD.0000000000018963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jansen E, Beekhof P, Viezeliene D, Muzakova V, Skalicky J. Long-term stability of cancer biomarkers in human serum: biomarkers of oxidative stress and redox status, homocysteine, crp and the enzymes Alt and Ggt. Biomarker Med. 2015;9(5):425–432. doi: 10.2217/bmm.15.14 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All raw and processed data are available from the corresponding author upon reasonable request.