Abstract
Hyperpolarized (HP) magnetic resonance imaging (MRI) is a groundbreaking imaging platform advancing from research to clinical practice, offering new possibilities for real-time, non-invasive metabolic imaging. This review explores the latest advancements, challenges, and future directions of HP MRI, emphasizing its transformative impact on both translational research and clinical applications. By employing techniques such as dissolution Dynamic Nuclear Polarization (dDNP), Parahydrogen-Induced Polarization (PHIP), Signal Amplification by Reversible Exchange (SABRE), and Spin-Exchange Optical Pumping (SEOP), HP MRI achieves enhanced nuclear spin polarization, enabling in vivo visualization of metabolic pathways with exceptional sensitivity. Current challenges, such as limited imaging windows, complex pre-scan protocols, and data processing difficulties, are addressed through innovative solutions like advanced pulse sequences, bolus tracking, and kinetic modeling. We highlight the evolution of HP MRI technology, focusing on its potential to revolutionize disease diagnosis and monitoring by revealing metabolic processes beyond the reach of conventional MRI and positron emission tomography (PET). Key advancements include the development of novel tracers like [2–13C]pyruvate and [1–13C]-alpha-ketoglutarate and improved data analysis techniques, broadening the scope of clinical metabolic imaging. Future prospects emphasize integrating artificial intelligence, standardizing imaging protocols, and developing new hyperpolarized agents to enhance reproducibility and expand clinical capabilities particularly in oncology, cardiology, and neurology. Ultimately, we envisioned HP MRI as a standardized modality for dynamic metabolic imaging in clinical practice.
Keywords: Carbon-13, Dynamic nuclear polarization, Hyperpolarization, Magnetic resonance imaging, Magnetic resonance spectroscopy
Graphical abstract
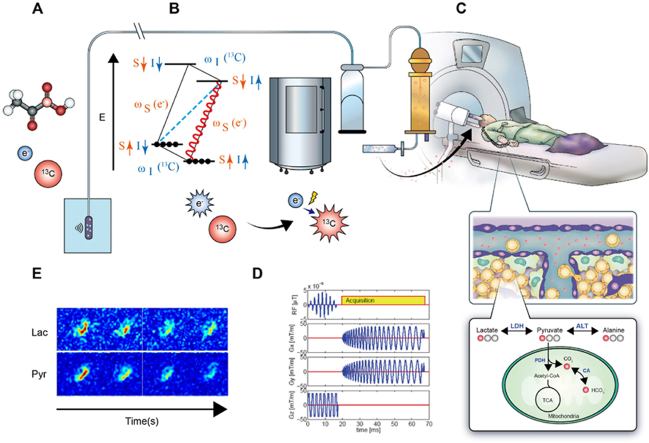
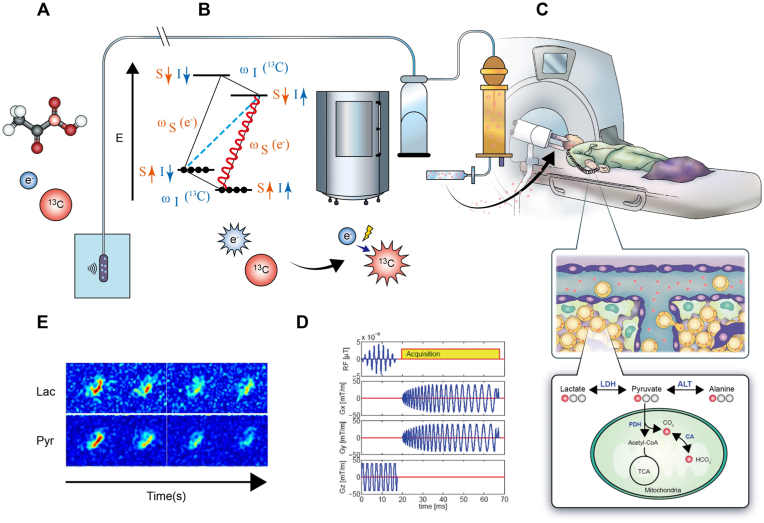
The flow of 13C substrate preparation, HP 13C polarization, MRI acquisition, and reconstruction. This diagram delineates the comprehensive process of employing HP 13C substrates, like pyruvate, to visualize downstream metabolites—lactate, alanine, and bicarbonate—in the glycolysis pathway through MRI.
1. Introduction
Hyperpolarized (HP) magnetic resonance imaging (MRI) represents a groundbreaking advancement in medical imaging, uniquely positioned between the conventional MRI and positron emission tomography (PET). Unlike conventional MRI, which visualizes anatomical structures using 1H nuclei, HP MRI uses labeled compounds with significantly enhanced nuclear spin polarization [1]. This results in a dramatic increase in signal intensity, allowing for real-time observation of metabolic processes in the body. In contrast to PET-FDG (18F-fluorodeoxyglucose), which tracks glucose metabolism using radioactive tracers, HP 13C MRI is non-radioactive and provides a different metabolic perspective. While PET-FDG is excellent for identifying areas of high glucose uptake, often correlating with tumor activity, it cannot distinguish between different metabolic pathways. HP 13C MRI, on the other hand, can provide detailed information on specific metabolic pathways, such as Krebs cycle activity in tumors, offering deeper insights into cellular metabolism and function [2,3] and the potential for early detection of treatment effects.
Even though this technique has the potential to reveal unprecedented metabolic insights, it also presents challenges, particularly in the realms of data acquisition, post-processing, data analysis, and the use of tracer [1–13C]pyruvate. These challenges stem from the short-lived nature of the hyperpolarized signal and the complexity of interpreting metabolic data. This review primarily describes the techniques and methods developed based on dissolution dynamic nuclear polarization (dDNP) and aims to address these challenges which involve developing advanced techniques for rapid and efficient data acquisition, sophisticated algorithms for post-processing to maximize signal extraction, and robust methods for data analysis to accurately interpret metabolic pathways. The use of [1–13C]pyruvate as a tracer plays a crucial role in this context, as it is pivotal in studying key metabolic processes, especially in cancer cells.
Building on this foundation, it is essential to explore the unique roles of HP 13C pyruvate in both translational research and clinical practice. In translational research, HP 13C pyruvate MRI has proven invaluable for understanding disease mechanisms, evaluating new therapies, and identifying biomarkers. For example, this technique is employed to study fundamental metabolic changes associated with diseases such as cancer [4,5], cardiovascular conditions [6], and neurological disorders such as mild Traumatic Brain Injury (TBI) [7,8], and hypoxic-ischemic encephalopathy (HIE) [9]. Researchers utilize HP 13C pyruvate MRI to visualize how diseases alter metabolic pathways, such as the conversion of pyruvate to lactate in cancer cells (Warburg effect), leading to the discovery of novel therapeutic targets and the development of new treatment strategies [10]. Additionally, in preclinical studies, HP 13C pyruvate MRI is used to assess the metabolic effects of experimental drugs or interventions, such as monitoring how a new cancer drug affects lactate production in tumor models, thus identifying potential therapeutic benefits or toxicities early in the drug development process [11,12]. The technique also facilitates the discovery of new metabolic biomarkers that indicate disease presence, progression, or response to treatment, such as changes in lactate production that correlate with tumor aggressiveness [13].
In clinical practice, HP 13C pyruvate MRI is applied directly to patients to diagnose disease, guide treatment decisions, monitor therapeutic response, and evaluate disease progression. For example, it allows for the detection of metabolic alterations in prostate cancer patients before structural changes are visible on conventional imaging, enabling earlier diagnosis and intervention [[14], [15], [16]]. Clinicians can tailor treatment plans based on individual metabolic profiles, assessing tumor activity to determine a patient's likelihood of responding to chemotherapy or radiation [17]. Moreover, HP 13C pyruvate MRI offers the advantage of detecting metabolic changes that occur much earlier than changes in tumor size, helping to quickly identify whether a treatment is effective or if a change in strategy is needed. It also enables the non-invasive tracking of disease progression over time, which is particularly valuable in conditions like cancer diseases [14,16,17], and neurological disorders [18], where early detection of progression can significantly impact clinical management.
Building on these roles, the manuscript will also present a comparative analysis of three alternative HP techniques: parahydrogen induced polarization (PHIP), spin exchange optical pumping (SEOP), and signal amplification by reversible exchange (SABRE). Each of these methods offers distinct approaches to enhancing nuclear spin polarization, thus broadening the scope of HP 13C applications.
2. HP 13C MRI: Technical advancements and unmet need
Dissolution dynamic nuclear polarization (dDNP) significantly enhances the capabilities of HP 13C imaging, a technique at the forefront of metabolic imaging. At its core, dDNP involves a process where 13C-labeled compounds are mixed with an electron-rich free radical [1]. This mixture is then cooled to extremely low temperatures, close to 1 Kelvin, within a high magnetic field. The purpose of this cooling step is to align the spins of the electrons in the free radicals to a high degree. Once this high level of electron spin polarization is achieved, microwave irradiation is applied. The microwave with specific frequencies target the interaction between the electron spins of the free radicals and the nuclear spins of the 13C nuclei. Through this interaction, polarization is transferred from the electrons to the 13C nuclei. This transfer dramatically increases the nuclear spin polarization of the 13C atoms, by a factor of 10,000 to 100,000 times greater than what is normally achieved in conventional MRI. This boosted polarization results in a significantly enhanced signal when these atoms are observed in an MRI scanner.
After the DNP process, the hyperpolarized substrate undergoes rapid dissolution in a biocompatible solvent. The liquid form of the substrate is delivered by the sterile fluid path [19]. This step is critical and time-sensitive, as the enhanced polarization of the 13C nuclei begins to decay quickly, typically within a few minutes. The dissolved, hyperpolarized agent is then ready for injection and subsequent imaging. This unique capability of dDNP to create a highly polarized state in 13C-labeled compounds is what allows for the real-time observation of metabolic processes in biological tissues, providing unprecedented insights into cellular metabolism and function. The schematic process is illustrated in [Fig. 1]. Utilizing 13C fast data acquisition and dDNP technique, these images have revealed the metabolic alterations in the human prostate [14], brain [20], breast [21], kidney [22], heart [23], as well as the immune system, both in both preclinical [24] clinical studies. Although the real-time metabolic imaging technique is promising, there are still challenges in imaging acquisition, data analysis, and the substrate of [1–13C]pyruvate. We will discuss these in the following sessions.
Fig. 1.
The flow of 13C substrate preparation, HP 13C polarization, MRI acquisition, and images.
This diagram delineates the comprehensive process of employing HP 13C substrates, like pyruvate, to visualize downstream metabolites—lactate, alanine, and bicarbonate—in the glycolysis pathway through MRI. Initially, the 13C substrate undergoes preparation in a sterile environment, setting the stage for its transformation into a highly informative metabolic tracer (A). Subsequently, the substrate is subject to polarization via the DNP technique such as GE SPINlab (B).
3. Comparison of dDNP with other hyperpolarization techniques
Parahydrogen-induced polarization (PHIP): PHIP utilizes hydrogen gas enriched in its para-spin state to hyperpolarize substrates before their incorporation into molecules of interest [25,26]. This method is cost-effective and allows for significant signal enhancement but is constrained by its requirement for a hydrogenation reaction, limiting its applicability to molecules that can undergo such a process. PHIP's application has predominantly been within preclinical studies due to its specific requirements for substrate modification and the need for further investigation into its clinical viability [27].
Signal amplification by reversible exchange (SABRE): SABRE represents a versatile hyperpolarization technique employing catalysts to transfer polarization from para-hydrogen to target nuclei, including protons [[28], [29], [30]]. This method has enabled the hyperpolarization of various compounds, such as [1–13C]- and [2–13C]pyruvate [31,32], and even [1–13C]-alpha-ketoglutarate, showcasing its capability to produce long-lived hyperpolarized states [33]. SABRE's adaptability and the ability to hyperpolarize a wide array of nuclei suggest its potential for both preclinical research and clinical applications, pending further development and validation [34].
Spin-exchange optical pumping (SEOP): HP 129Xenon (Xe) MRI, enabled by the SEOP technique, represents a significant advancement in non-invasive lung imaging. This method involves polarizing 129Xe gas in a heated cell containing rubidium vapor under a low magnetic field, where circularly polarized laser light induces spin polarization transfer from rubidium electrons to 129Xe nuclei [35]. Achieving polarization levels between 20% and 50%, SEOP greatly enhances MRI signal sensitivity compared to conventional techniques. HP 129Xe MRI has been safely applied in clinical studies [[36], [37], [38], [39]], improving the visualization of lung ventilation and gas exchange without the use of ionizing radiation. Exploiting the unique chemical shifts of 129Xe in different lung compartments allows for a detailed assessment of lung function, including ventilation defects and gas exchange efficiency [38,40,41]. This technique has proven particularly valuable in identifying and quantifying ventilation impairments, using both reader-based trials and semi-automated binning approaches for accurate ventilation defect percentage (VDP) calculation [[42], [43], [44], [45]]. Furthermore, HP 129Xe MRI facilitates the study of lung microstructures through diffusion-weighted imaging, enhancing our understanding of diseases like chronic obstructive pulmonary disease (COPD) [46]. The safe and effective use of HP 129Xe MRI underscores its potential for advancing respiratory diagnostics and research, offering a powerful tool for exploring lung physiology and pathology with unprecedented clarity and precision. [Table 1] summarizes these techniques and lists the pros and cons of each technique.
Table 1.
Comparison of hyperpolarization techniques.
| HP Technique | Principle | Primary substrate | HP Duration | Advantages | Challenges | References |
|---|---|---|---|---|---|---|
| dDNP | Polarization transfer from electron spins to 13C nuclei at low temperatures and high magnetic fields | [1–13C]pyruvate | ∼120 min | High polarization levels, real-time metabolic imaging | Requires cryogenic temperatures, and long polarization time | [1,14,[19], [20], [21], [22], [23]] |
| PHIP | Hydrogen gas enriched in para-spin state used to hyperpolarize substrates | Substrates suitable for hydrogenation | Minutes | Cost-effective, significant signal enhancement | Limited applicability due to hydrogenation requirement | [[25], [26], [27]] |
| SABRE | Use of catalysts to transfer polarization from para-hydrogen to target nuclei | Various compounds, including [1–13C]- and [2–13C]pyruvate | Minutes | Versatile, capable of hyperpolarizing a wide array of nuclei | Need for further development and validation | [[28], [29], [30], [31], [32], [33], [34]] |
| SEOP | Polarization transfer from rubidium electrons to 129Xe nuclei using circularly polarized laser light | 129Xenon | Minutes | Non-invasive lung imaging, chemical shift between blood and tissue, safe for clinical studies | Lower polarization efficiency, and limited clinical applications | [[35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46]] |
Abbreviations: dNDP: dissolution dynamic nuclear polarization; PHIP: parahydrogen induced polarization; SEOP: spin exchange optical pumping; and SABRE: signal amplification by reversible exchange.
In comparison, while PHIP offers a cost-effective approach, its utility is somewhat limited by the specificity of its substrate requirements. SABRE, on the other hand, showcases broader applicability with the ability to hyperpolarize a wide range of nuclei, positioning it as a promising technique for both preclinical and potentially clinical applications. SEOP distinguishes itself by its clinical adoption in lung imaging, facilitated by the direct use of hyperpolarized noble gases. Collectively, these techniques exemplify the ongoing evolution and diversification of hyperpolarized MRI methods, each contributing uniquely to the expansion of imaging capabilities in both research and clinical domains.
4. Challenges and remedies in imaging techniques
HP 13C MRI, a transformative imaging technique, enables the non-invasive observation of metabolic pathways by enhancing the signal of 13C-labeled substrates. This advancement illuminates the complex biochemical processes within living tissues, offering significant potential in patient diagnosis and treatment monitoring. However, its broader application is hindered by various technical and practical challenges, necessitating innovative solutions to maximize its clinical utility and unlock its full potential.
One primary challenge is the limited 13C imaging window due to the short-lived nature of the hyperpolarized signal. To address this, researchers have developed advanced pulse sequences and imaging techniques that optimize signal utilization within this constrained timeframe. For example, flyback echo-planar spectroscopic imaging (EPSI) and symmetric EPSI have improved data acquisition efficiency [47,48], while rapid 3D volume imaging techniques using spectral-spatial excitation pulse (SPSP) with echo-planar imaging (EPI) or spiral readouts have enhanced the signal-to-noise ratio (SNR) of metabolites [49,50]. Additionally, the iterative decomposition with echo asymmetry and least squares estimation (IDEAL) chemical shift imaging (CSI) offers an efficient method for decomposing multiple chemical species [51,52].
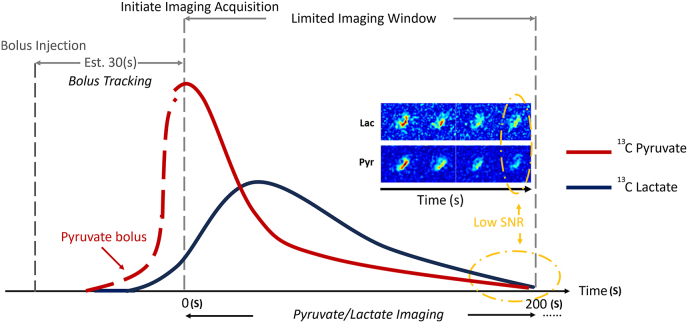
Prescan procedure complexities, arising from the small amount of natural 13C abundance, complicate automated settings like shim parameters and RF transmit gain. Remedies include utilizing Sodium 23 or Hydrogen 1 frequencies for 13C pyruvate frequency estimation and calibrating transmit gain with a 13C Urea phantom using Bloch-Siegert pulses [53,54]. Variability in HP 13C pyruvate bolus due to physiological differences is mitigated by innovative bolus tracking methods and frameworks for simultaneous automatic acquisition timing and B1 calibration to adjust for variability [55,56]. These challenges are illustrated in [Fig. 2].
Fig. 2.
Challenges in HP 13C imaging acquisition and data analysis.
This diagram vividly depicts the dynamic nature and inherent challenges of HP 13C imaging, focusing on the temporal progression of HP 13C pyruvate (red) and its metabolite lactate (blue) signals from injection up to 200 s, when signals diminish into the noise threshold. The depiction underscores the unique attribute of flux in HP 13C imaging, where the degrading metabolic signals over time highlight critical obstacles. The primary challenge illustrated is the limited imaging window, constrained to less than 200 s, demanding precise timing for optimal signal capture. The second challenge involves determining the optimal acquisition time within this narrow window to maximize data quality and relevance. Thirdly, the diagram points to the complexity of defining the pyruvate flow function, a pivotal component in the kinetic model necessary for estimating apparent exchange rate constants, crucial for accurate metabolic analysis. Lastly, the rapid degradation of the hyperpolarized state presents a significant hurdle, leading to a decreased signal-to-noise ratio (SNR) towards the end of the acquisition period. These challenges highlight the technical and temporal intricacies of HP 13C imaging, underscoring the need for precise timing and sophisticated modeling to harness its full diagnostic potential.
Challenges in achieving high spatial and temporal resolution in dynamic environments overlap with the limited imaging window issue. Using multiband excitation pulses in parallel imaging approaches is one way to address this issue. The other method involves using compartmental information from the matching high-resolution 1H images to improve the spatial resolution of HP 13C human organ images using a patch-based algorithm [13,[57], [58], [59]]. Furthermore, the application of balanced steady-state free-precession (bSSFP) sequences for the recurrent refocusing of transverse spins enhances the spatiotemporal resolution of metabolites and can also be used to reduce respiratory motion aberrations in abdominal imaging [60,61]. Lastly, addressing the production and delivery challenges of hyperpolarized agents involves investigations into cost-effective production methods and the development of preservation techniques to extend the duration of the enhanced signal, tackling issues related to cost, availability, efficient delivery, and maintaining hyperpolarization stability [62,63]. These comprehensive solutions not only enhance image quality and reliability but also pave the way for wider clinical adoption, contributing to improved patient outcomes and deeper understanding of disease mechanisms. [Table 2] summarizes the challenges and corresponding remedies.
Table 2.
Imaging Techniques of hyperpolarized 13C MRI.
| Challenges | Remedies | References |
|---|---|---|
| Limited hyperpolarization window | Advanced pulse sequences and imaging techniques: Efficiency in data acquisition, rapid 3D volume imaging, IDEAL chemical shift imaging, high-resolution imaging | EPSI [47,48]: |
| SPSP [49,50]: | ||
| IDEAL CSI [21,51,52,64,65]: | ||
| Prescan procedure complexities | Prescan procedure adaptations: Frequency estimation, transmit gain calibration | Frequency estimation [53]: |
| Transmit gain calibration [54]: | ||
| Physiological variability in HP 13C pyruvate bolus | Bolus tracking enhancements: data acquisition triggering, automatic acquisition and calibration | Data acquisition triggering [55]: |
| Automatic acquisition and calibration [56]: | ||
| Spatial and temporal resolution | Compressed sensing and multiband excitation pulses, utilization of 1H image by using patch-based algorithm | Spatial and temporal resolution [13,[57], [58], [59]]: |
| Respiratory motion in abdominal imaging | Mitigation of respiratory motion artifacts by fast acquisition | bSSFP [60,61]: |
| Production and delivery of hyperpolarized agents | Investigations into cost-effective production methods and preservation techniques | Production and preservation techniques [62,63]: |
Abbreviations: EPSI: flyback echo-planar spectroscopic imaging; SPSP: spectral-spatial excitation pulse; IDEAL: iterative decomposition with echo asymmetry and least squares estimation; CSI: chemical shift imaging; bSSEP: balanced steady-state free-precession.
5. Challenges and remedies in data processing and analysis
HP 13C MRI plays a critical role in unveiling dynamic metabolic processes, providing invaluable insights into alterations in glycolytic flux. This advanced imaging technique, through spectroscopy and imaging, offers a detailed view of metabolic fluxes, which are essential for understanding disease mechanisms and evaluating treatment responses. Accurate data analysis is pivotal in interpreting these fluxes, either through direct measurement using the area under the curve (AUC) method [66] or via kinetic modeling [[67], [68], [69]], presenting the complexities and nuances of metabolic activities within the body.
The kinetic model for analyzing HP 13C MRI data accounts for the flow of HP 13C pyruvate into an organ and its chemical exchange with lactate. This model is described by differential equations that consider the relative signal intensities of pyruvate, P(t) and lactate, L(t), the incoming pyruvate signal intensity, u(t), reaction rate constants, KP and KL, and effective relaxivity, ρ, in Equations (1), (2)). Such modeling enables the quantification of metabolic fluxes, including the conversion rate of pyruvate to lactate, which is indicative of metabolic activity and alterations in diseases.
| (1) |
| (2) |
where P and L are the relative pyruvate and lactate signal intensities, and u is the pyruvate incoming signal intensity, Kp and Kl are the forward and backward reaction rate constants, respectively, and ρ is the effective relativity given by
| (3) |
where T1 is the relaxation time of pyruvate in the medium or tissue, tR is the repetition time, and θ is the flip angle.
Addressing the challenges in this analysis requires innovative approaches. For instance, the estimation of Pyruvate Flow (u(t)) in the kinetic model is challenging due to short imaging windows and the limited signal of HP 13C pyruvate. Solutions include the use of Gd-DTPA in dynamic contrast-enhancement (DCE) MRI to estimate u(t), and co-polarization techniques of 13C urea and pyruvate for simultaneous perfusion and metabolic information, respectively [13,[70], [71], [72]]. The alternative approach is to focus solely on fitting the lactate signal, using the measured pyruvate signal as the input, u(t), for the kinetic model at every time point. This method involves fitting the estimated lactate signal at each time point based on the pyruvate signal measured at adjacent time points and the previously estimated lactate signal. This is achieved by applying the two-site model described in differential Equations (1), (2)), thereby refining the model's accuracy in capturing the dynamics between lactate and pyruvate signals [69].
The other challenge is the low signal-to-noise ratio (SNR) of 13C downstream metabolites, which can cause discrepancies in AUC results or Kp values. Remedies include denoising of HP 13C spectroscopy and images using singular value decomposition (SVD)-based low-rank methods, applying high-order SVD with low rank or wavelet methods for image denoising, and utilizing kinetic models with background noise features for accurate signal estimation of downstream metabolites [[73], [74], [75]]. The challenges of data acquisition and analysis are illustrated in [Fig. 2].
Bridging the gap between the aforementioned challenges is the overarching issue of reproducibility in 13C data analysis. Ensuring consistent outcomes across different hyperpolarized 13C MRI studies is paramount, as variability in data analysis can undermine the reliability of research findings. The introduction of the Spectroscopic Imaging, Visualization, and Computing (SIVIC) open-source software package marks a significant step towards enhancing reproducibility [76]. Furthermore, the availability of analytical tools for the kinetic model through the “Hyperpolarized MRI Toolbox” [77] via the Hyperpolarized Technology Resource Center represents an invaluable resource for researchers. By addressing the challenges of reproducibility, the scientific community can advance towards more consistent and reliable analysis in hyperpolarized 13C studies, thereby facilitating the translation of research findings into clinical applications.
These strategies highlight the concerted efforts to overcome data processing and analysis challenges in HP 13C MRI, aiming to refine techniques for a better understanding of metabolic alterations. Through such advancements, researchers and clinicians can gain deeper insights into the metabolic pathways, facilitating improved diagnosis, monitoring, and treatment of diseases. [Table 3] summarizes the challenges and corresponding remedies.
Table 3.
Data analysis of HP 13C MRI.
| Challenges | Remedies | References |
|---|---|---|
| Estimating pyruvate flow (u(t)) |
|
DCE MRI [70]: Kinetic model equations [69]: 13C urea and pyruvate co-polarization [13,71,72]: |
| Signal-to-noise ratio (SNR) discrepancy in sequential acquisition |
|
SVD-based methods [73]: High-order SVD and wavelet methods [75]: Kinetic models with noise features [74]: |
| Reproducibility |
|
SIVIC software [76]: HP 13C Kinetic model [77]: |
Abbreviations: DCE: dynamic contrast-enhancement; SVD: singular value decomposition; SNR: signal-to-noise ratio; SIVIC: Spectroscopic Imaging, Visualization, and Computing.
6. Advances in metabolic imaging: Overcoming limitations with novel 13C-labeled tracers and co-polarization techniques
HP [1–13C]pyruvate has been a cornerstone in metabolic imaging, offering critical insights into cellular metabolism and disease processes. However, its scope, particularly in probing Krebs cycle metabolites and assessing glutaminolysis—a crucial pathway in cancer metabolism—has been limited. To address these limitations, significant advancements in tracer technology have been made, expanding the utility of metabolic imaging and overcoming the constraints of [1–13C]pyruvate. The development of [2–13C]pyruvate, synthesized and polarizable via dDNP, marks a significant advancement by enabling the probing of Krebs cycle metabolites. This substrate has broadened the scope of metabolic imaging, facilitating real-time assessments of metabolism in the heart, brain, and liver across various animal models. Clinical and pre-clinical studies with [2–13C]pyruvate have showcased its efficacy in metabolic assessments, revealing its potential in diagnosing and monitoring diseases affecting these organs [[78], [79], [80], [81], [82], [83]]. Similarly, [1–13C]-Alpha-Ketoglutarate, another substrate synthesized and polarizable through dDNP, has been instrumental in advancing our understanding of glutaminolysis in cancer cell metabolism. This tracer has facilitated studies on isocitrate dehydrogenase (IDH) activity in various animal models, offering new insights into cancer metabolism and potential therapeutic targets [[84], [85], [86], [87]]. Moreover, innovations in co-polarization techniques, particularly the co-polarization of [13C,15N2]urea and [1–13C]pyruvate, have enabled simultaneous metabolic and perfusion imaging. This approach provides comprehensive insights into the interplay between metabolic processes and blood flow, further enhancing the capabilities of metabolic imaging in medical diagnostics [13,71,72]. These advancements in substrate or tracer developments not only surmount the initial limitations associated with HP [1–13C]pyruvate but also open new avenues for comprehensive metabolic imaging. The development of [2–13C]pyruvate and [1–13C]-alpha-ketoglutarate, along with the co-polarization of tracers, represents significant strides in enhancing the scope of metabolic assessments in medical imaging. This progress contributes significantly to the field of medical diagnostics and research, offering new perspectives on disease mechanisms and potential therapeutic interventions. [Table 4] summarizes the characteristics of substrates.
Table 4.
Tracers beyond HP [1–13C] pyruvate for clinical metabolic MRI.
| Tracers | Metabolic Pathway/Function and Applications | References |
|---|---|---|
| [2–13C]Pyruvate | Probing Krebs cycle metabolites - applications in heart, brain, and liver metabolism | Krebs cycle analysis [78,83]: |
| Brain metabolism [79,80]: | ||
| Diabetic muscles [81]: | ||
| Mouse liver [82]: | ||
| [1–13C]-Alpha-Ketoglutarate | Probing metabolic pathways involved in glutaminolysis - instrumental in advancing understanding of cancer cell metabolism | IDH assessment [[84], [85], [86], [87]]: |
| Co-polarization of [13C,15N2]urea and [1–13C]pyruvate | Allows simultaneous metabolic and perfusion imaging - broadening the scope of metabolic assessments | Simultaneous imaging [13,71,72]: |
7. Future perspectives
The future perspectives of technical breakthroughs and standardization in HP 13C MRI hold immense promise for advancing the field and facilitating its widespread adoption. One pivotal area of development lies in overcoming existing technical challenges, such as enhancing the duration of hyperpolarization. Technical breakthroughs in hyperpolarization methods, including innovative pulse sequences and hardware improvements, are anticipated to extend the imaging window, allowing for more comprehensive and dynamic imaging of metabolic processes.
Standardization is critical for ensuring consistency and comparability across different hyperpolarized 13C MRI studies. The establishment of standardized protocols for agent production, imaging acquisition, and data analysis is crucial. This standardization would facilitate multi-center collaborations and comparisons, fostering a more robust and reproducible research environment. Currently, several groups have reported the comprehensive survey of HP [1–13C]pyruvate in human studies [88]. This is the first step toward the standardization of HP 13C pyruvate. Moreover, future breakthroughs are expected in the development of novel hyperpolarized agents, expanding the range of metabolites that can be studied. This diversification will open new avenues for investigating various metabolic pathways and contribute to a deeper understanding of physiological and pathological processes.
The integration of artificial intelligence and machine learning into hyperpolarized 13C MRI data analysis holds great potential. These technologies can aid in automated image processing, quantitative analysis, and the extraction of meaningful metabolic information, streamlining the workflow and enhancing the clinical applicability of hyperpolarized 13C MRI. One of practical issues is the signal loss or image artifacts caused by respiratory motion. AI models inspired by 1H MRI literature may offer promise in eliminating respiratory motion artifacts [89,90].
In conclusion, the future of hyperpolarized 13C MRI envisions technical breakthroughs that address current limitations, standardization to ensure reproducibility, the development of novel hyperpolarized agents, and the integration of advanced technologies for efficient data analysis. These advancements collectively promise a transformative impact on medical imaging, fostering its evolution into a widely accepted and standardized modality for studying dynamic metabolic processes in vivo.
Upon achieving optimal polarization, the HP 13C substrate is quickly dissolved to maintain its enhanced state and then injected into the patient (B and C). This step is time-sensitive, as the hyperpolarized state degrades rapidly, necessitating swift administration to capture the peak signal. Following injection, MRI acquisition begins, tracking the distribution and metabolic conversion of the substrate into its metabolites within the body's tissues (D).
The culmination of this process is the reconstruction of detailed 13C metabolic images (E). These images provide a unique window into the body's metabolic pathways in real time, offering insights into normal physiology and the alterations that occur in diseases such as cancer. This technique represents a significant advance in medical imaging, enabling the non-invasive study of metabolism and the potential for tailored therapeutic interventions.
Declaration of competing interest
The authors declare no conflict of interest.
Data availability statement
The CC BY 4.0 license is a Creative Commons license.
Author contribution
Conceptualization: Ching-Yi Hsieh, Gigin Lin.
Data curation: Ying-Chieh Lai, Ching-Yi Hsieh.
Formal analysis: Ying-Chieh Lai, Ching-Yi Hsieh.
Funding acquisition: Ying-Chieh Lai, Ching-Yi Hsieh, Gigin Lin.
Investigation: Ching-Yi Hsieh, Gigin Lin.
Methodology: Ching-Yi Hsieh, Gigin Lin.
Project administration: Kuan-Ying Lu.
Resources: Gigin Lin.
Software: Ying-Chieh Lai, Ching-Yi Hsieh.
Supervision: Gigin Lin.
Validation: Ying-Chieh Lai, Ching-Yi Hsieh.
Visualization: Ching-Yi Hsieh.
Writing-original draft: Ching-Yi Hsieh, Gigin Lin.
Writing-review & editing: Ching-Yi Hsieh, Gigin Lin.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work the author(s) used ChatGPT in order to improve the reading and grammar of this manuscript. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
Funding
This study was funded by National Science and Technology Council, Taiwan (MOST 109- 2628-B-182A-007-, MOST 110-2628-B-182A-018-, MOST 111-2628-B-182A-012-, NSTC 112-2314-B-182A-015-, NSTC 112-2314-B-182A-127-MY3, NSTC 113-2314-B-182A-086) and Chang Gung Medical Foundation (BMRPF63, SMRPG3K0055, CLRPG3K0024 and CMRPG3M0732).
Acknowledgements
The authors would like to thank Ms. Wan Wen Chen for the assistance of illustrating figures, and organizing literature.
Footnotes
Peer review under responsibility of Chang Gung University.
References
- 1.Ardenkjær-Larsen JH, Fridlund B, Gram A, Hansson G, Hansson L, Lerche MH, et al. Increase in signal-to-noise ratio of > 10,000 times in liquid-state NMR. Proc Natl Acad Sci USA. 2003;100(18):10158–10163. doi: 10.1073/pnas.1733835100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Golman K, in ’t Zandt R, Thaning M. Real-time metabolic imaging. Proc Natl Acad Sci USA. 2006;103(30):11270–11275. doi: 10.1073/pnas.0601319103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Day SE, Kettunen MI, Gallagher FA, Hu D.-E, Lerche M, Wolber J, et al. Detecting tumor response to treatment using hyperpolarized 13C magnetic resonance imaging and spectroscopy. Nat Med. 2007;13(11):1382–1387. doi: 10.1038/nm1650. [DOI] [PubMed] [Google Scholar]
- 4.Chen AP, Albers MJ, Cunningham CH, Kohler SJ, Yen Y-F, Hurd RE, et al. Hyperpolarized C-13 spectroscopic imaging of the TRAMP mouse at 3T—initial experience. Magn Reson Med. 2007;58(6):1099–1106. doi: 10.1002/mrm.21256. [DOI] [PubMed] [Google Scholar]
- 5.Park I, Larson PE, Zierhut ML, Hu S, Bok R, Ozawa T, et al. Hyperpolarized 13C magnetic resonance metabolic imaging: application to brain tumors. Neuro Oncol. 2010;12(2):133–144. doi: 10.1093/neuonc/nop043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schroeder MA, Lau AZ, Chen AP, Gu Y, Nagendran J, Barry J, et al. Hyperpolarized (13)C magnetic resonance reveals early- and late-onset changes to in vivo pyruvate metabolism in the failing heart. Eur J Heart Fail. 2013;15(2):130–140. doi: 10.1093/eurjhf/hfs192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.DeVience SJ, Lu X, Proctor J, Rangghran P, Melhem ER, Gullapalli R, et al. Metabolic imaging of energy metabolism in traumatic brain injury using hyperpolarized [1-13C]pyruvate. Sci Rep. 2017;7(1):1907. doi: 10.1038/s41598-017-01736-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chaumeil M, Guglielmetti C, Qiao K, Tiret B, Ozen M, Krukowski K, et al. Hyperpolarized (13)C metabolic imaging detects long-lasting metabolic alterations following mild repetitive traumatic brain injury. Preprint (Version 1) available at Research Square. [DOI]
- 9.Liu X, Manninen T, Capper AM, Jiang X, Ellison J, Kim Y, et al. Brain metabolism after therapeutic hypothermia for murine hypoxia-ischemia using hyperpolarized [1-13C] pyruvate magnetic resonance spectroscopy. NMR Biomed. 2024 doi: 10.1002/nbm.5196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Park JM, Spielman D.M, Josan S, Jang T, Merchant M, Hurd RE, et al. Hyperpolarized 13C-lactate to 13C-bicarbonate ratio as a biomarker for monitoring the acute response of anti-vascular endothelial growth factor (anti-VEGF) treatment. NMR Biomed. 2016;29(5):650–659. doi: 10.1002/nbm.3509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Park I, Bok R, Ozawa T, Phillips JJ, James CD, Vigneron DB, et al. Detection of early response to temozolomide treatment in brain tumors using hyperpolarized 13C MR metabolic imaging. J Magn Reson Imag. 2011;33(6):1284–1290. doi: 10.1002/jmri.22563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Khemtong C, Carpenter NR, Lumata LL, Merritt ME, Moreno KX, Kovacs Z, et al. Hyperpolarized 13C NMR detects rapid drug-induced changes in cardiac metabolism. Magn Reson Med. 2015;74(2):312–319. doi: 10.1002/mrm.25419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen H-Y, Larson PEZ, Bok RA, von Morze C, Sriram R, Delos Santos R, et al. Assessing prostate cancer aggressiveness with hyperpolarized dual-agent 3D dynamic imaging of metabolism and perfusion. Cancer Res. 2017;77(12):3207–3216. doi: 10.1158/0008-5472.CAN-16-2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nelson SJ, Kurhanewicz J, Vigneron DB, Larson PE, Harzstark AL, Ferrone M, et al. Metabolic imaging of patients with prostate cancer using hyperpolarized [1-13C]pyruvate. Sci Transl Med. 2013;5(198):198ra08. doi: 10.1126/scitranslmed.3006070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aggarwal R, Vigneron DB, Kurhanewicz J. Hyperpolarized 1-[13C]-Pyruvate magnetic resonance imaging detects an early metabolic response to androgen ablation therapy in prostate cancer. Eur Urol. 2017;72(6):1028–1029. doi: 10.1016/j.eururo.2017.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sushentsev N, McLean MA, Warren AY, Benjamin AJV, Brodie C, Frary A, et al. Hyperpolarised (13)C-MRI identifies the emergence of a glycolytic cell population within intermediate-risk human prostate cancer. Nat Commun. 2022;13(1):466. doi: 10.1038/s41467-022-28069-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee CY, Soliman H, Bragagnolo ND, Sahgal A, Geraghty BJ, Chen AP, et al. Predicting response to radiotherapy of intracranial metastases with hyperpolarized [Formula: see text]C MRI. J Neuro Oncol. 2021;152(3):551–557. doi: 10.1007/s11060-021-03725-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hackett EP, Chen J, Ingle L, Al Nemri S, Barshikar S, da Cunha Pinho M, et al. Longitudinal assessment of mitochondrial dysfunction in acute traumatic brain injury using hyperpolarized [1-13C]pyruvate. Magn Reson Med. 2023;90(6):2432–2442. doi: 10.1002/mrm.29794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ardenkjaer-Larsen JH, Leach AM, Clarke N, Urbahn J, Anderson D, Skloss TW. Dynamic nuclear polarization polarizer for sterile use intent. NMR Biomed. 2011;24(8):927–932. doi: 10.1002/nbm.1682. [DOI] [PubMed] [Google Scholar]
- 20.Grist JT, McLean MA, Riemer F, Schulte RF, Deen SS, Zaccagna F, et al. Quantifying normal human brain metabolism using hyperpolarized [1-13C]pyruvate and magnetic resonance imaging. Neuroimage. 2019;189:171–179. doi: 10.1016/j.neuroimage.2019.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gallagher FA, Woitek R, McLean MA, Gill AB, Manzano Garcia R, Provenzano E, et al. Imaging breast cancer using hyperpolarized carbon-13 MRI. Proc Natl Acad Sci U S A. 2020;117(4):2092–2098. doi: 10.1073/pnas.1913841117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tang S, Meng MV, Slater JB, Gordon JW, Vigneron DB, Stohr BA, et al. Metabolic imaging with hyperpolarized (13) C pyruvate magnetic resonance imaging in patients with renal tumors-Initial experience. Cancer. 2021;127(15):2693–2704. doi: 10.1002/cncr.33554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cunningham CH, Lau JYC, Chen AP, Geraghty BJ, Perks WJ, Roifman I, et al. Hyperpolarized 13C metabolic MRI of the human heart: initial experience. Circ Res. 2016;119(11):1177–1182. doi: 10.1161/CIRCRESAHA.116.309769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lai YC, Hsieh CY, Lu KY, Sung CH, Ho HY, Cheng ML, et al. Monitoring early glycolytic flux alterations following radiotherapy in cancer and immune cells: hyperpolarized carbon-13 magnetic resonance imaging study. Metabolites. 2021;11(8):518. doi: 10.3390/metabo11080518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Eisenschmid TC, Kirss RU, Deutsch PP, Hommeltoft SI, Eisenberg R, Bargon J, et al. Para hydrogen induced polarization in hydrogenation reactions. J Am Chem Soc. 1987;109(26):8089–8091. [Google Scholar]
- 26.Bowers CR, Weitekamp DP. Parahydrogen and synthesis allow dramatically enhanced nuclear alignment. J Am Chem Soc. 1987;109(18):5541–5542. [Google Scholar]
- 27.Nagel L, Gierse M, Gottwald W, Ahmadova Z, Grashei M, Wolff P, et al. Parahydrogen-polarized [1-13C]pyruvate for reliable and fast preclinical metabolic magnetic resonance imaging. Adv Sci. 2023;10(30) doi: 10.1002/advs.202303441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rayner PJ, Duckett SB. Signal amplification by reversible exchange (SABRE): from discovery to diagnosis. Angew Chem Int Ed Engl. 2018;57(23):6742–6753. doi: 10.1002/anie.201710406. [DOI] [PubMed] [Google Scholar]
- 29.Adams RW, Aguilar JA, Atkinson KD, Cowley MJ, Elliott PIP, Duckett SB, et al. Reversible interactions with para-hydrogen enhance NMR sensitivity by polarization transfer. Science. 2009;323(5922):1708–1711. doi: 10.1126/science.1168877. [DOI] [PubMed] [Google Scholar]
- 30.Adams RW, Duckett SB, Green RA, Williamson DC. Green GGR A theoretical basis for spontaneous polarization transfer in non-hydrogenative parahydrogen-induced polarization. J Chem Phys. 2009;131(19):194505. doi: 10.1063/1.3254386. [DOI] [PubMed] [Google Scholar]
- 31.Iali W, Roy SS, Tickner BJ, Ahwal F, Kennerley AJ, Duckett SB. Hyperpolarising pyruvate through signal amplification by reversible exchange (SABRE) Angew Chem Int Ed Engl. 2019;58(30):10271–10275. doi: 10.1002/anie.201905483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tickner BJ, Semenova O, Iali W, Rayner PJ, Whitwood AC, Duckett SB. Optimisation of pyruvate hyperpolarisation using SABRE by tuning the active magnetisation transfer catalyst. Catal Sci Technol. 2020;10(5):1343–1355. doi: 10.1039/c9cy02498k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Adelabu I, Ettedgui J, Joshi SM, Nantogma S, Chowdhury MRH, McBride S, et al. Rapid (13)C hyperpolarization of the TCA cycle intermediate alpha-ketoglutarate via SABRE-SHEATH. Anal Chem. 2022;94(39):13422–13431. doi: 10.1021/acs.analchem.2c02160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.MacCulloch K, Browning A, Bedoya DOG, McBride SJ, Abdulmojeed MB, Dedesma C, et al. Facile hyperpolarization chemistry for molecular imaging and metabolic tracking of [1-13C]pyruvate in vivo. J Magn Reson Open. 2023:16–17. doi: 10.1016/j.jmro.2023.100129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Walker TG, Happer W. Spin-exchange optical pumping of noble-gas nuclei. Rev Mod Phys. 1997;69(2):629–642. [Google Scholar]
- 36.Driehuys B, Martinez-Jimenez S, Cleveland ZI, Metz GM, Beaver DM, Nouls JC, et al. Chronic obstructive pulmonary disease: safety and tolerability of hyperpolarized 129Xe MR imaging in healthy volunteers and patients. Radiology. 2012;262(1):279–289. doi: 10.1148/radiol.11102172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Paulin GA, Svenningsen S, Jobse BN, Mohan S, Kirby M, Lewis JF, et al. Differences in hyperpolarized 3He ventilation imaging after 4 years in adults with cystic fibrosis. J Magn Reson Imag. 2015;41(6):1701–1707. doi: 10.1002/jmri.24744. [DOI] [PubMed] [Google Scholar]
- 38.Stewart NJ, Leung G, Norquay G, Marshall H, Parra-Robles J, Murphy PS, et al. Experimental validation of the hyperpolarized 129Xe chemical shift saturation recovery technique in healthy volunteers and subjects with interstitial lung disease. Magn Reson Med. 2015;74(1):196–207. doi: 10.1002/mrm.25400. [DOI] [PubMed] [Google Scholar]
- 39.Shukla Y, Wheatley A, Kirby M, Svenningsen S, Farag A, Santyr GE, et al. Hyperpolarized 129Xe magnetic resonance imaging: tolerability in healthy volunteers and subjects with pulmonary disease. Acad Radiol. 2012;19(8):941–951. doi: 10.1016/j.acra.2012.03.018. [DOI] [PubMed] [Google Scholar]
- 40.Cleveland ZI, Cofer GP, Metz G, Beaver D, Nouls J, Kaushik SS, et al. Hyperpolarized 129Xe MR imaging of alveolar gas uptake in humans. PLoS One. 2010;5(8) doi: 10.1371/journal.pone.0012192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Collier GJ, Eaden JA, Hughes PJC, Bianchi SM, Stewart NJ, Weatherley ND, et al. Dissolved (129) Xe lung MRI with four-echo 3D radial spectroscopic imaging: quantification of regional gas transfer in idiopathic pulmonary fibrosis. Magn Reson Med. 2021;85(5):2622–2633. doi: 10.1002/mrm.28609. [DOI] [PubMed] [Google Scholar]
- 42.Virgincar RS, Cleveland ZI, Sivaram Kaushik S, Freeman MS, Nouls J, Cofer GP, et al. Quantitative analysis of hyperpolarized 129Xe ventilation imaging in healthy volunteers and subjects with chronic obstructive pulmonary disease. NMR Biomed. 2013;26(4):424–435. doi: 10.1002/nbm.2880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Svenningsen S, Kirby M, Starr D, Leary D, Wheatley A, Maksym GN, et al. Hyperpolarized 3He and 129Xe MRI: differences in asthma before bronchodilation. J Magn Reson Imag. 2013;38(6):1521–1530. doi: 10.1002/jmri.24111. [DOI] [PubMed] [Google Scholar]
- 44.Salerno M, Altes TA, Mugler JP, Nakatsu M, Hatabu H, de Lange EE. Hyperpolarized noble gas MR imaging of the lung: potential clinical applications. Eur J Radiol. 2001;40(1):33–44. doi: 10.1016/s0720-048x(01)00347-3. [DOI] [PubMed] [Google Scholar]
- 45.Marshall H, Deppe MH, Parra-Robles J, Hillis S, Billings CG, Rajaram S, et al. Direct visualisation of collateral ventilation in COPD with hyperpolarised gas MRI. Thorax. 2012;67(7):613–617. doi: 10.1136/thoraxjnl-2011-200864. [DOI] [PubMed] [Google Scholar]
- 46.Kaushik SS, Cleveland ZI, Cofer GP, Metz G, Beaver D, Nouls J, et al. Diffusion-weighted hyperpolarized 129Xe MRI in healthy volunteers and subjects with chronic obstructive pulmonary disease. Magn Reson Med. 2011;65(4):1154–1165. doi: 10.1002/mrm.22697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cunningham CH, Vigneron DB, Chen AP, Xu D, Nelson SJ, Hurd RE, et al. Design of flyback echo-planar readout gradients for magnetic resonance spectroscopic imaging. Magn Reson Med. 2005;54(5):1286–1289. doi: 10.1002/mrm.20663. [DOI] [PubMed] [Google Scholar]
- 48.Yen YF, Kohler SJ, Chen AP, Tropp J, Bok R, Wolber J, et al. Imaging considerations for in vivo 13C metabolic mapping using hyperpolarized 13C-pyruvate. Magn Reson Med. 2009;62(1):1–10. doi: 10.1002/mrm.21987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cunningham CH, Chen AP, Lustig M, Hargreaves BA, Lupo J, Xu D, et al. Pulse sequence for dynamic volumetric imaging of hyperpolarized metabolic products. J Magn Reson. 2008;193(1):139–146. doi: 10.1016/j.jmr.2008.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mayer D, Levin YS, Hurd RE, Glover GH, Spielman DM. Fast metabolic imaging of systems with sparse spectra: application for hyperpolarized 13C imaging. Magn Reson Med. 2006;56(4):932–937. doi: 10.1002/mrm.21025. [DOI] [PubMed] [Google Scholar]
- 51.Reeder SB, Brittain JH, Grist TM, Yen Y-F. Least-squares chemical shift separation for 13C metabolic imaging. J Magn Reson Imag. 2007;26(4):1145–1152. doi: 10.1002/jmri.21089. [DOI] [PubMed] [Google Scholar]
- 52.Wiesinger F, Weidl E, Menzel MI, Janich MA, Khegai O, Glaser SJ, et al. IDEAL spiral CSI for dynamic metabolic MR imaging of hyperpolarized [1-13C]pyruvate. Magn Reson Med. 2012;68(1):8–16. doi: 10.1002/mrm.23212. [DOI] [PubMed] [Google Scholar]
- 53.Grist JT, Hansen ESS, Sanchez-Heredia JD, McLean MA, Tougaard R, Riemer F, et al. Creating a clinical platform for carbon-13 studies using the sodium-23 and proton resonances. Magn Reson Med. 2020;84(4):1817–1827. doi: 10.1002/mrm.28238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Schulte RF, Sacolick L, Deppe MH, Janich MA, Schwaiger M, Wild JM, et al. Transmit gain calibration for nonproton MR using the Bloch-Siegert shift. NMR Biomed. 2011;24(9):1068–1072. doi: 10.1002/nbm.1657. [DOI] [PubMed] [Google Scholar]
- 55.Durst M, Koellisch U, Gringeri C, Janich MA, Rancan G, Frank A, et al. Bolus tracking for improved metabolic imaging of hyperpolarised compounds. J Magn Reson. 2014;243:40–46. doi: 10.1016/j.jmr.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 56.Tang S, Milshteyn E, Reed G, Gordon J, Bok R, Zhu X, et al. A regional bolus tracking and real-time B1 calibration method for hyperpolarized 13C MRI. Magn Reson Med. 2019;81(2):839–851. doi: 10.1002/mrm.27391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Larson PEZ, Hu S, Lustig M, Kerr AB, Nelson SJ, Kurhanewicz J, et al. Fast dynamic 3D MR spectroscopic imaging with compressed sensing and multiband excitation pulses for hyperpolarized 13C studies. Magn Reson Med. 2011;65(3):610–619. doi: 10.1002/mrm.22650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee H, Song J.E, Shin J, Joe E, Joo CG, Choi YS, et al. High resolution hyperpolarized 13C MRSI using SPICE at 9.4T. Magn Reson Med. 2018;80(2):703–710. doi: 10.1002/mrm.27061. [DOI] [PubMed] [Google Scholar]
- 59.Ma J, Park JM. Super-resolution hyperpolarized (13)C imaging of human brain using patch-based algorithm. Tomography. 2020;6(4):343–355. doi: 10.18383/j.tom.2020.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mansson S, Petersson JS, Scheffler K. Fast metabolite mapping in the pig heart after injection of hyperpolarized 13C-pyruvate with low-flip angle balanced steady-state free precession imaging. Magn Reson Med. 2012;68(6):1894–1899. doi: 10.1002/mrm.24183. [DOI] [PubMed] [Google Scholar]
- 61.Reed GD, von Morze C, Addy NO, Ingle RR, Johnson KO, Overall WR, et al. High spatiotemporal resolution hyperpolarized 13C angiography. J Cardiovasc Magn Reson. 2016;18(S1) [Google Scholar]
- 62.Ji X, Bornet A, Vuichoud B, Milani J, Gajan D, Rossini AJ, et al. Transportable hyperpolarized metabolites. Nat Commun. 2017;8 doi: 10.1038/ncomms13975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Capozzi A, Kilund J, Karlsson M, Patel S, Pinon AC, Vibert F, et al. Metabolic contrast agents produced from transported solid 13C-glucose hyperpolarized via dynamic nuclear polarization. Commun Chem. 2021;4(1):95. doi: 10.1038/s42004-021-00536-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Beatty PJ, Nishimura DG, Pauly JM. Rapid gridding reconstruction with a minimal oversampling ratio. IEEE Trans Med Imag. 2005;24(6):799–808. doi: 10.1109/TMI.2005.848376. [DOI] [PubMed] [Google Scholar]
- 65.Macdonald EB, Barton GP, Cox BL, Johnson KM, Strigel RM, Fain SB. Improved reconstruction stability for chemical shift encoded hyperpolarized (13) C magnetic resonance spectroscopic imaging using k-t spiral acquisitions. Magn Reson Med. 2020;84(1):25–38. doi: 10.1002/mrm.28122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Daniels CJ, McLean MA, Schulte RF, Robb FJ, Gill AB, McGlashan N, et al. A comparison of quantitative methods for clinical imaging with hyperpolarized 13C-pyruvate. NMR Biomed. 2016;29(4):387–399. doi: 10.1002/nbm.3468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Schulte RF, Sperl JI, Weidl E, Menzel MI, Janich MA, Khegai O, et al. Saturation-recovery metabolic-exchange rate imaging with hyperpolarized [1-13C] pyruvate using spectral-spatial excitation. Magn Reson Med. 2013;69(5):1209–1216. doi: 10.1002/mrm.24353. [DOI] [PubMed] [Google Scholar]
- 68.Khegai O, Schulte RF, Janich MA, Menzel MI, Farrell E, Otto AM, et al. Apparent rate constant mapping using hyperpolarized [1-13C]pyruvate. NMR Biomed. 2014; 27:1256-65 doi: 10.1002/nbm.3174. [DOI] [PubMed] [Google Scholar]
- 69.Larson PEZ, Chen HY, Gordon JW, Korn N, Maidens J, Arcak M, et al. Investigation of analysis methods for hyperpolarized 13C-pyruvate metabolic MRI in prostate cancer patients. NMR Biomed. 2018;31(11) doi: 10.1002/nbm.3997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Bankson JA, Walker CM, Ramirez MS, Stefan W, Fuentes D, Merritt ME, et al. Kinetic modeling and constrained reconstruction of hyperpolarized [1-13C]-Pyruvate offers improved metabolic imaging of tumors. Cancer Res. 2015;75(22):4708–4717. doi: 10.1158/0008-5472.CAN-15-0171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Liu X, Tang S, Mu C, Qin H, Cui D, Lai YC, et al. Development of specialized magnetic resonance acquisition techniques for human hyperpolarized [(13) C,(15) N(2) ]urea + [1-(13) C]pyruvate simultaneous perfusion and metabolic imaging. Magn Reson Med. 2022;88(3):1039–1054. doi: 10.1002/mrm.29266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Qin H, Tang S, Riselli AM, Bok RA, Delos Santos R, van Criekinge M, et al. Clinical translation of hyperpolarized (13) C pyruvate and urea MRI for simultaneous metabolic and perfusion imaging. Magn Reson Med. 2022;87(1):138–149. doi: 10.1002/mrm.28965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Brender JR, Kishimoto S, Merkle H, Reed G, Hurd RE, Chen AP, et al. Dynamic imaging of glucose and lactate metabolism by 13C-MRS without hyperpolarization. Sci Rep. 2019;9(1):3410. doi: 10.1038/s41598-019-38981-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Hsieh C-Y, Sung C-H, Shen Y-L, Lai Y-C, Lu K-Y, Lin G. Developing a method to estimate the downstream metabolite signals from hyperpolarized [1-13C]pyruvate. Sensors. 2022;22(15):5480. doi: 10.3390/s22155480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kim Y, Chen HY, Autry AW, Villanueva-Meyer J, Chang SM, Li Y, et al. Denoising of hyperpolarized (13)C MR images of the human brain using patch-based higher-order singular value decomposition. Magn Reson Med. 2021;86(5):2497–2511. doi: 10.1002/mrm.28887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Crane JC, Olson MP, Nelson SJ. SIVIC: open-source, standards-based software for DICOM MR spectroscopy workflows. Int J Biomed Imag. 2013;2013:12. doi: 10.1155/2013/169526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Larson, PE, Hyperpolarized-MRI-toolbox.
- 78.Schroeder MA, Atherton HJ, Ball DR, Cole MA, Heather LC, Griffin JL, et al. Real-time assessment of Krebs cycle metabolism using hyperpolarized 13C magnetic resonance spectroscopy. FASEB (Fed Am Soc Exp Biol) J. 2009;23(8):2529–2538. doi: 10.1096/fj.09-129171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Park JM, Josan S, Grafendorfer T, Yen YF, Hurd RE, Spielman DM, et al. Measuring mitochondrial metabolism in rat brain in vivo using MR Spectroscopy of hyperpolarized [2-13C]pyruvate. NMR Biomed. 2013;26(10):1197–1203. doi: 10.1002/nbm.2935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Chung BT, Chen HY, Gordon J, Mammoli D, Sriram R, Autry AW, et al. First hyperpolarized [2-13C]pyruvate MR studies of human brain metabolism. J Magn Reson. 2019;309 doi: 10.1016/j.jmr.2019.106617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Park JM, Josan S, Hurd RE, Graham J, Havel PJ, Bendahan D, et al. Hyperpolarized NMR study of the impact of pyruvate dehydrogenase kinase inhibition on the pyruvate dehydrogenase and TCA flux in type 2 diabetic rat muscle. Pflügers Archiv. 2021;473(11):1761–1773. doi: 10.1007/s00424-021-02613-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ragavan M, McLeod MA, Rushin A, Merritt ME. Detecting de novo Hepatic Ketogenesis Using Hyperpolarized [2-13C] Pyruvate. Front Physiol. 2022;13 doi: 10.3389/fphys.2022.832403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Chen HY, Gordon JW, Dwork N, Chung BT, Riselli A, Sivalokanathan S, et al. Probing human heart TCA cycle metabolism and response to glucose load using hyperpolarized [2-13 C] pyruvate MR spectroscopy. NMR BioMed. 2023;37(3):e5074. doi: 10.1002/nbm.5074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.AbuSalim JE, Yamamoto K, Miura N, Blackman B, Brender JR, Mushti C, et al. Simple esterification of [1-13C]-Alpha-Ketoglutarate enhances membrane permeability and allows for noninvasive tracing of glutamate and glutamine production. ACS Chem Biol. 2021;16(11):2144–2150. doi: 10.1021/acschembio.1c00561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hong D, Batsios G, Viswanath P, Gillespie AM, Vaidya M, Larson PEZ, et al. Acquisition and quantification pipeline for in vivo hyperpolarized (13) C MR spectroscopy. Magn Reson Med. 2022;87(4):1673–1687. doi: 10.1002/mrm.29081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hong D, Kim Y, Mushti C, Minami N, Wu J, Cherukuri MK, et al. Monitoring response to a clinically relevant IDH inhibitor in glioma-Hyperpolarized (13)C magnetic resonance spectroscopy approaches. Neurooncol Adv. 2023;5(1) doi: 10.1093/noajnl/vdad143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Miura N, Mushti C, Sail D, AbuSalim JE, Yamamoto K, Brender JR, et al. Synthesis of [1-13C-5-12 C]-alpha-ketoglutarate enables noninvasive detection of 2-hydroxyglutarate. NMR Biomed. 2021;34(11) doi: 10.1002/nbm.4588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Larson PE, Bernard JM, Bankson JA, Bøgh N, Bok RA, Chen AP, et al. Current methods for hyperpolarized [1-13C]pyruvate MRI human studies. Magn Reson Med. 2024;91:2204–2228. doi: 10.1002/mrm.29875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ghodrati V, Bydder M, Ali F, Gao C, Prosper A, Nguyen KL, et al. Retrospective respiratory motion correction in cardiac cine MRI reconstruction using adversarial autoencoder and unsupervised learning. NMR Biomed. 2021;34(2) doi: 10.1002/nbm.4433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Bao Q, Chen Y, Bai C, Li P, Liu K, Li Z, et al. Retrospective motion correction for preclinical/clinical magnetic resonance imaging based on a conditional generative adversarial network with entropy loss. NMR Biomed. 2022;35(12) doi: 10.1002/nbm.4809. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The CC BY 4.0 license is a Creative Commons license.